Abstract
The seed of Herpetospermum pedunculosum (Ser.) C.B. Clarke, known in Chinese as Bo-Leng-Gua-Zi and in Tibetan as Sejimedo, are here abbreviated as H. pedunculosum seeds. Herpetospermum pedunculosum seeds is a traditional Chinese medicine for protecting the liver, clearing heat, and detoxifying. A total of 125 chemical metabolites of H. pedunculosum seeds are found, including lignans, fatty acids, terpenes, coumarins, and others. The pharmacological activities of H. pedunculosum seeds are mainly in hepatoprotective, antioxidant, anti-cancer cells, and anticholestatic effects. In clinical application, it is mainly used in combination with other traditional Chinese medicines to play a key role in treating the liver disease. This paper gives a systematic review of above research aspects, proposes the potential limitations and put forward plausible solutions. Relevant literatures were searched in PubMed, Web of Science and Chinese National Knowledge Infrastructure with Herpetospermum as the key word. A number of studies have shown that H. pedunculosum seeds exert excellent hepatoprotective effects by acting on NF-κB, TGF-β, and Keap1-Nrf2 signaling pathways, which provide a solid base for its clinic application. However, more research is needed to explore the standard cultivation and quality evaluation of H. pedunculosum seeds and systematical structure-activity relationship of its active metabolites.
Keywords: Herpetospermum pedunculosum (Ser.) C.B. Clarke, phytochemistry, pharmacological activity, liver protection, lignan
1 Introduction
With the continuous development of various drugs, phytomedicines with fewer side effects and significant effects are gradually attracting people. Especially for some effective ethnodrugs, further development of their hidden medicinal value through in-depth research is increasingly popular and desirable. Herpetospermum pedunculosum (Ser.) C.B. Clarke (H. pedunculosum) is mainly distributed in several high-altitude areas such as Tibet, Yunnan, India and Nepal. As the main medicinal part of H. pedunculosum (Ser.) C.B. Clarke, H. pedunculosum seeds are traditional Tibetan drug, which have traditional effects of clearing heat and softening liver. At the same time, H. pedunculosum seeds are also the core ingredient of clinical traditional Chinese prescriptions, such as Jiuwei Zhangya Pill (for treating cholecystitis), Wuwei Jinse Pill (for treating jaundice hepatitis), and Songshi pill (for treating hepatitis and liver fibrosis). Modern studies have shown that H. pedunculosum seeds contain a variety of chemical metabolites, including lignans, coumarins, terpenes, etc. (Xu, 2012), and have shown a variety of pharmacological activities, including liver protection, antioxidant, anti-tumor, and anti-cholestasis effects (Gong, 2012; Shen et al., 2015).
The excellent pharmacological effects and particular characteristics of H. pedunculosum seeds undoubtedly deserve systematical induction and summary, which is hardly reported to the best of our knowledge. Therefore, we investigated relevant literatures in Web of Science, PubMed and CNKI with Herpetospermum as the key word, and focused on the literatures of the H. pedunculosum seeds with excluding that on other parts, such as stem, leaf and flesh. Based on these literatures, this paper comprehensively reviews the traditional use, botany, chemical metabolites, pharmacological effects, pharmaceutical analysis, processing and application in Chinese herbal prescriptions of H. pedunculosum seeds, which can provide scientific basis for further research and promote the potential for development.
2 Traditional uses
As a classic Tibetan medicine, H. pedunculosum seeds often used in the treatment of Tri-pa (a disease be traditionally characterized by diffusion of bile, disorders of the blood-heat, and yellow color in the muscles and eyes), which was first recorded in Yue Wang Yao Zhen (《月王药诊》) in the early 8th century. At the same time, in the middle of the 8th century, Tara Materia Medica (《度母本草》, Shivatso) recorded that H. pedunculosum seeds can treat heat disease, and bacon disease (diseases caused by the combination of food accumulation and cold). Beside these, Si Bu Yi Dian (《四部医典》, Yutog Yontan Gonpo), written and revised during the late 8th to 12th century, further proposed that H. pedunculosum seeds can remove the heat of the lower organs. In addition, it supplemented the bitter taste of H. pedunculosum seeds. Jingzhu Materia Medica (《晶珠本草》, Dema Tenpe Nyima), written in 1840, proposed that H. pedunculosum seeds could treat Tri-pa in the viscera. Diqing Tibetan medicine (《迪庆藏药》, Yang and Chu cheng) and Chinese Tibetan medicine (《中华藏本草》Luo, 1997) supplement recorded its effects of treating liver and gallbladder heat and indigestion, which was also supported by the record of Chinese Materia Medica (《中华本草》, National Administration of Traditional Chinese Medicine). In 2015, the “Interpretation of Tibetan Medicine Jinsui Materia Medica” (《藏药金穗本草诠释》, Gama Qupei) concluded that H. pedunculosum seeds could treat the liver and gallbladder diseases of the Tri-pa type. In summary, H. pedunculosum seeds have been used as its prototype medicine for over 13 centuries, and its effects on protecting the liver and treating indigestion have gained tremendous application as recorded in traditional medical books.
3 Botany
Herpetospermum pedunculosum (Ser.) C. B. Clarke, is usually harvested at around October, and adapts to grow on warm, humid subtropical roadsides, hillsides, shrubland, and forest edges at the altitude of 2,300–3,500 m (Flora Reipublicae Popularis Sinicae Commission, 1983) and its botanical organs including the flower, leaf, fruit and seed were shown in Figures 1A–D. As displayed in Figure 1D, H. pedunculosum seeds is slightly oblong with uneven carving and the surface from brown to black brown. One end of H. pedunculosum seeds has triangular protrusions, and the other end is tapered, slightly wedge-shaped and slightly concave in the center (Chinese Materia Medica Commission, 1998). The further investigation of H. pedunculosum seeds characters and sources can enhance the standardization of commercial H. pedunculosum seeds and is of great significance in cultivating it.
FIGURE 1.
Flower (A), leaf (B), fruit (C) and seed (D) of Herpetospermum pedunculosum (Ser.) C.B. Clarke.
4 Phytochemistry
The chemical metabolites of H. pedunculosum seeds are reported to include lignans, fatty acids, terpenoids, and coumarins. The other metabolites such as amino acids, alkaloids, and flavones were also discussed. Details can be found in Figures 2–6 and Table 1.
FIGURE 2.
Lignan types in Herpetospermum pedunculosum seeds.
FIGURE 6.
Structures of other metabolites from Herpetospermum pedunculosum seeds.
TABLE 1.
Metabolites extracted from Herpetospermum pedunculosum seeds.
| No. | Name | Molecular formula | Extract | Separation method | Identification method | References |
|---|---|---|---|---|---|---|
| Lignan | ||||||
| 1 | Herpetriol | C30H34O9 | Ethyl alcohol | — | UV, IR,MS, 1H-NMR, 13C-NMR | Kaouadji et al. (1979) |
| 2 | Herpetetrol | C40H44O12 | Ethyl alcohol | — | UV, MS, 1H-NMR, 13C-NMR | Kaouadji et al. (1979) |
| 3 | Herpepentol | C50H54O15 | MeOH | — | MS, 1H-NMR, 13C-NMR | Kaouadji and Pieraccini (1984a) |
| 4 | Herpetetradione | C40H42O12 | MeOH | — | MS, 1H-NMR, 13C-NMR | Kaouadji and Favre-Bonvin (1984a) |
| 5 | Herpetetrone | C40H42O13 | MeOH | Polyamide CC6 and sephadex LH-20, chromatography | UV, IR, MS, 1H-NMR | Kaouadji et al. (1987) |
| 6 | Herpetrione | C30H32O10 | Ethyl alcohol | — | MS, 1H-NMR, 13C-NMR | Kaouadji and Jean (1983) |
| 7 | Herpetone | C29H30O9 | Ethyl alcohol | Silica gel column chromatography, preparative HPLC | MS, IR, 1H-NMR, 13C-NMR | Zhang et al. (2006) |
| 8 | Herpetol | C20H20O6 | Ethyl alcohol | — | UV, MS, 1H-NMR, 13C-NMR | Kaouadji and Pieraccini (1984a) |
| 9 | Dehydrodiconiferyl alcohol | C20H22O6 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | UV, 1H-NMR, 13C-NMR | Ma (2020) |
| 10 | Herpetosin B | C20H22O7 | Ethyl acetate | Silica gel column chromatography | UV, IR, MS, 13C-NMR, 1H-NMR | Xu (2012) |
| 11 | Herpetal | C20H18O6 | Ethyl acetate | — | UV, 1H-NMR, 13C-NMR | Kaouadji et al. (1978) |
| 12 | Herpetotriol | C30H32O9 | Ethyl acetate | — | UV, 1H-NMR, 13C-NMR | Kaouadji et al. (1978) |
| 13 | Herpepropenal | C30H30O10 | Ethyl acetate | Silica gel column chromatography, RPC18, HPLC | MS, 13C-NMR, 1H-NMR, DEPT, HMBC, COSY, HSQC, NOESY | Yang et al. (2010) |
| 14 | 7,8′-didehydlroherpetotriol | C30H32O9 | Ethyl acetate | Reversed phase silica gel column chromatography, Preparation for HPLC Chromatography | UV, IR, MS, 1H-NMR,13C-NMR | Xu (2012) |
| 15 | (7S,8R,7′R,8′S)-7'-[7′-ethoxyl-7'-(4′-hydroxyl-3′-methoxylphenyl)]methyl-7-(4-hydroxyl-3-methoxylphenyl)-8-hydroxymethyl-tetrahydrofuran | C22H28O7 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi- preparative HPLC | UV, IR, 1H-NMR, 13C-NMR | Ma (2020) |
| 16 | (7S,8R)-threo-1'-[3′-hydroxy-7-(4-hydroxy-3-methoxyphenyl)-8-hydroxymethyl-7,8-dihydrobenzofuran] acrylaldehyde | C19H18O6 | Ethyl aceta | Normal phase silica gel column chromatography, MPLC, semi- preparative HPLC | UV, IR, 1H-NMR, 13C-NMR | Ma (2020) |
| 17 | (−)-Tanegool-7′-methyl etherl | C21H26O7 | Ethyl acetate | Silica gel column chromatography, sephadex LH-20 | MS, 1H-NMR, 13C-NMR | Zhou (2014) |
| 18 | Herpetin | C30H34O9 | Ethyl acetate | Silica gel column chromatography, Rp-Si -gel, semi- preparative HPLC | MS, IR, 1H-NMR, 13C-NMR | Yuan et al. (2005) |
| 19 | Lariciresino | C20H24O6 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | UV, 1H-NMR,13C-NMR | Ma (2020) |
| 20 | (+)-(7′S,7′′S,8′R,8′′R)-4,4′,4′′-Trihydroxy-3,5′,3′′-trimethoxy-7-oxo-8-ene[8-3′,7′-O-9′′,8′-8′′,9′-O-7′′] lignoid | C30H30O9 | Petroleum ether | Silica gel column chromatography, preparative HPLC | MS, IR, 1H-NMR, 13C-NMR, 1H-1H COSY, HMQC, HMBC | Yu et al. (2014) |
| 21 | Ent-isolariciresinol | C20H24O6 | Ethyl acetate | Silica gel column chromatography, MPLC, semi-preparative HPLC | UV, MS, 1H-NMR, 13C-NMR | Ma (2020) |
| 22 | Herpetenol | C20H22O6 | Ethyl acetate | Silica gel column chromatography | UV, IR, MS, 1H-NMR, 13C-NMR | Wang (2005) |
| 23 | Herpetfluorenone | C16H14O6 | Ethyl acetate | Silica gel column chromatography, sephadex LH-20 | MS, 1H-NMR, 13C-NMR | Gong et al. (2016) |
| 24 | (1S)-4hydroxy-3-[2-(4-hydroxy-3-methoxy-phenyl)-1-hydroxymethyl2-oxo-ethyl]-5-methoxy-benzaldehyde | C18H18O7 | Petroleum ether | Silica gel column chromatography, RPC18, sephadex LH-20 | MS, IR, 1H-NMR, 13C-NMR, 1H-1H COSY, HMQC, HMBC | Yu et al. (2014) |
| 25 | Hedyotol A | C30H32O9 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 26 | Picrasmalignan | C30H30O9 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 27 | Balanophonin | C20H20O6 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 28 | 1-Propanone, 3-hydroxy-1-(4-hydrpxy-3-methoxyphenyl)-2-[4-(3-hydroxy-1-propen-1-yl)-2-methoxyphenoxy] | C21H24O6 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 29 | Erythro-guaiacylglycerol-b-coniferyl ether | C20H24O7 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 30 | Threo-guaiacylglycerol- b-coiferyl ether | C20H24O7 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 31 | (7R,8S)-Dehydrodiconiferyl alcohol γ′- methyl ether | C21H24O6 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 32 | 3-Benzofuranmethanol, 2, 3-dihydro-2-(4-dydroxy-3-methoxypenyl)-7-methoxy-5-(3-methoxyl-1-propenyl)-,[2S-[2a,3b, 5(E)]]-(9CI) | C21H24O6 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 33 | Evofolin-B | C17H18O6 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 34 | 1-Propanon, 3-hydroxy-1-(2-hydrpxy-5-methoxyphenyl)-2-(4-hydroxy-3-methoxyphenyl)- | C20H24O7 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 35 | Herpetatol A | C19H18O5 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 36 | Herpetatol B | C19H16O5 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 37 | Herpetatol C | C20H22O7 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 38 | Herpetatol D | C31H32O9 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 39 | Herpetatol E | C30H30O9 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 40 | Herpetatol F | C29H28O8 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 41 | Herpetatol G | C29H28O8 | Ethyl acetate | Silica gel/gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 42 | (−)-pinoresinol monomethyl ether | C21H24O6 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | MS, IR,1H-NMR, 13C-NMR | Ma (2020) |
| 43 | epipinoresinol | C20H22O6 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | UV, MS, 1H-NMR, 13C-NMR | Ma (2020) |
| 44 | (+)-pinoresinol | C20H22O6 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | MS, IR, 1H-NMR, 13C-NMR | Ma (2020) |
| 45 | (+)-menbrine | C21H24O5 | Ethyl acetate | Normal phase silica gel column chromatography, semi-preparative HPLC | MS, IR, 1H-NMR, 13C-NMR | Ma (2020) |
| 46 | cinncassins D | C28H28O9 | Ethyl acetate | Normal phase silica gel column chromatography, semi-preparative HPLC | MS, UV, IR, 1H-NMR, 13C-NMR | Ma (2020) |
| 47 | (7R,7′R,7″R,8S,8′S,8″S)-4′,4″-dihydroxy-3,3′,3″,5-tetramethoxy-7,9':7′,9-diepoxy-4,8″-oxy-8,8′-sesqoineolignan-7″,9″-diol | C31H36O11 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Ma (2020) |
| 48 | 3-Benzofuranmethanol-2,3-dihydro-2-(4-hydroxy-3-methoxyphenyl)-4-:methoxy-6-[tetra-hydro-2-(3-hydroxy-4-methoxyphenyl)-3-methanol]-2-furanmethyl | C31H36O8 | Ethyl acetate | Normal phase silica gel column chromatograpy | MS, 1H-NMR, 13C-NMR | Yuan et al. (2006b) |
| 49 | Ehletianol C | C30H36O10 | Ethyl acetate | Normal phase silica gel column chromatography, semi-preparative HPLC | MS, UV, 1H-NMR, 13C-NMR | Ma (2020) |
| 50 | Herpetosiol G | C20H22O5 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HMBC | Ma (2020) |
| 51 | Herpetosiol H | C23H30O8 | Ethyl acetate | Normal phase silica gel column chromatography, sephadex LH-20, semi-preparative HPLC | UV, IR, MS, 1H-NMR, 13C-NMR, HSQC, HMBC, COSY, NOESY | Ma (2020) |
| 52 | Herpetosiol I | C30H34O10 | Ethyl acetate | Normal phase silica gel column chromatography, sephadex LH-20, semi-preparative HPLC | UV, IR, MS, 1H-NMR, 13C-NMR, HSQC, HMBC, COSY, NOESY | Ma (2020) |
| 53 | Herpetosiol J | C23H24O9 | Ethyl acetate | Normal phase silica gel column chromatography, semi-preparative HPLC | UV, MS, 1H-NMR, 13C-NMR, HSQC, HMBC, COSY, NOESY | Ma (2020) |
| 54 | Herpetosiol K | C30H30O9 | Ethyl acetate | Normal phase silica gel column chromatography, semi-preparative HPLC | UV, IR, MS, 1H-NMR, 13C-NMR, HSQC, HMBC, COSY, NOESY | Ma (2020) |
| 55 | Herpetosiol L | C19H18O6 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | UV, MS, 1H-NMR, 13C-NMR, HSQC, HMBC, COSY, NOESY | Ma (2020) |
| 56 | Herpetosiol M | C20H20O7 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HMBC | Ma (2020) |
| 57 | Herpetosiol N | C32H38O11 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HSQC, HMBC, COSY, NOESY | Ma (2020) |
| 58 | Phyllanglaucin B | C30H34O9 | Ethyl acetate | Normal phase silica gel column chromatography, recrystallization | MS, 1H-NMR, 13C-NMR | Huang et al. (2021) |
| 59 | Buddlenol E | C32H38O10 | Ethyl acetate | Normal phase silica gel column chromatography, sephedax LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Huang et al. (2021) |
| 60 | Spathulated | C30H34O9 | Ethyl acetate | — | HPLC | Wei et al. (2020) |
| 61 | Threo-buddlenol E | C31H36O11 | Ethyl acetate | — | HPLC | Wei et al. (2020) |
| 62 | Picrasmalignan A | C29H28O9 | Ethyl acetate | — | HPLC | Wei et al. (2020) |
| 63 | 9,3′-Dimethoxyhierochin A | C21H24O6 | Ethyl acetate | — | HPLC | Wei et al. (2020) |
| 64 | Sesquilignan | C30H34O9 | Ethyl acetate | — | HPLC | Yuan et al. (2019) |
| 65 | Herpedulin A | C50H52O16 | Ethyl acetate | Silica gel column chromatography, preparative TLC | MS, 1H-NMR, 13C-NMR, HMBC | Meng et al. (2022) |
| 66 | Herpedulin B | C30H34O10 | Ethyl acetate | Silica gel column chromatography, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HMBC | Meng et al. (2022) |
| 67 | Herpedulin C | C31H36O11 | Ethyl acetate | Silica gel column chromatography, MPLC, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, 1H-1H COSY, HSQC, HMBC, CD spectrum | Meng et al. (2022) |
| 68 | Herpedulin D | C31H36O11 | Ethyl acetate | Silica gel column chromatography, MPLC, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, 1H-1H COSY, HSQC, HMBC, CD spectrum | Meng et al. (2022) |
| 69 | Herpedulin E | C30H30O9 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HMBC, CD spectrum | Meng et al. (2022) |
| 70 | Herpedulin F | C32H38O11 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HSQC, COSY | Meng et al. (2022) |
| 71 | Herpedulin G | C30H32O11 | Ethyl acetate | Silica gel column chromatography, MPLC, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HSQC, CD spectrum | Meng et al. (2022) |
| 72 | Herpedulin H | C30H32O11 | Ethyl acetate | Silica gel column chromatography, MPLC, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, CD spectrum | Meng et al. (2022) |
| 73 | Herpedulin I | C30H32O11 | Ethyl acetate | Silica gel column chromatography, MPLC, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, CD spectrum | Meng et al. (2022) |
| 74 | Herpedulin J | C23H24O9 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HSQC, NOSY, HMBC | Meng et al. (2022) |
| 75 | Herpedulin K | C30H26O9 | Ethyl acetate | Silica gel column chromatography, recrystalization | MS, 1H-NMR, 13C-NMR, HSQC, HMBC | Meng et al. (2022) |
| 76 | Herpedulin L | C23H30O8 | Ethyl acetate | Silica gel column chromatography, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HMBC, NOESY, CD spectrum | Meng et al. (2022) |
| 77 | Herpedulin M | C19H18O6 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HSQC, 1H-1HCOSY, HMBC | Meng et al. (2022) |
| 78 | Herpedulin N | C20H20O7 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Meng et al. (2022) |
| 79 | Herpedulin O | C20H22O5 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, HMBC, CD spectrum | Meng et al. (2022) |
| 80 | Herpedulin P | C19H20O5 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR CD spectrum | Meng et al. (2022) |
| Fatty acids | ||||||
| 81 | Palmitic acid | C16H32O2 | Petroleum ether | — | GC-MS | Liu et al. (2005a) |
| 82 | Oleic acid | C18H34O2 | Petroleum ether | — | GC-MS | Liu et al. (2005a) |
| 83 | Stearic acid | C18H34O2 | Petroleum ether | — | GC-MS | Liu et al. (2005a) |
| 84 | Linoleic acid | C18H32O2 | Petroleum ether | — | GC-MS | Zhang et al. (2004) |
| 85 | Linolenic acid | C18H30O2 | Petroleum ether | Normal phase silica gel column chromatography, semi- preparative HPLC | MS, 1H-NMR, 13C-NMR | Dong et al. (2019) |
| 86 | Trilinolein | C57H98O6 | Petroleum ether | Normal phase silica gel column chromatography, preparative HPLC | MS, 1H-NMR, 13C-NMR | Dong et al. (2019) |
| 87 | 9-Octadecenoic acid | C18H34O2 | Petroleum ether | — | GC-MS | Liu et al. (2005b) |
| 88 | Octadecanoic acid | C18H36O2 | Petroleum ether | — | GC-MS | Liu et al. (2005a) |
| 89 | cis-5-Dodecaenoic acid | C12H22O2 | Petroleum ethe | Silica gel column chromatography | MS, 1H-NMR, 13C-NMR | Chen (2020) |
| 90 | Dodecanoic acid | C12H24O2 | Ethyl acetate | Silica gel column chromatography | MS, 1H-NMR, 13C-NMR | Xu, (2012) |
| 91 | 10-Eicossenoic acid | C20H38O2 | Ethyl acetate | Silica gel column chromatography, sephadex LH-20 | MS, 1H-NMR, 13C-NMR | Xu, (2012) |
| Terpenoids | ||||||
| 92 | Neocucurbitacin D | C31H44O8 | 90% EtOH | Silica gel column chromatography, sephadex LH-20, RP-HLPC | MS, IR, 1H-NMR, 13C-NMR, HMBC, NOESY | Jiang et al. (2020) |
| 93 | Cucurbitacin E | C32H44O8 | 90% EtOH | Silica gel column chromatography, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Jiang et al. (2020) |
| 94 | Cucurbitacin D | C30H44O7 | 90% EtOH | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Jiang et al. (2020) |
| 95 | Cucurbitacin B | C31H44O8 | 90% EtOH | Silica gel column chromatography, sephadex LH-20, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Jiang et al. (2020) |
| 96 | Cucurbitacin I | C30H46O7 | 90% EtOH | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR | Jiang et al. (2020) |
| 97 | 23, 24-Dihydroisocucurbitacin B | C32H48O8 | 90% EtOH | Silica gel column chromatography, sephadex LH-20, RP-HLPC | MS, 1H-NMR, 13C-NMR | Jiang et al. (2020) |
| 98 | Cucurbitacin M | C30H44O6 | Ethyl acetate | Normal phase silica gel column chromatography, MPLC, semi-preparative HPLC | MS, 1H-NMR, 13C-NMR, DEPT, HMBC, 1H-H COSY | Ma (2020) |
| 99 | Herpetosin A | C22H30O6 | Ethyl acetate | Silica gel column chromatography | UV, IR, MS, 13C-NMR, 1H-NMR | Xu (2012) |
| 100 | Cucurbitacin L | C30H44O7 | Ethyl acetate | Silica gel column chromatography, Sephadex LH-20 | MS, 1H-NMR | Dai et al. (2017) |
| 101 | Oleanic acid | C30H48O3 | Ethyl acetate | Silica gel column chromatography, Sephadex LH-20 | MS, 1H-NMR | Dai et al. (2017) |
| Coumarins | ||||||
| 102 | Herpetolide A | C16H14O6 | Ethyl acetate | Silica gel column chromatography, recrystallization | MS, IR, 1H-NMR, 13C-NMR, HMBC, NOESY, DEPT, HMQC | Zhang et al. (2008) |
| 103 | Herpetolide B | C16H12O6 | Ethyl acetate | Silica gel column chromatography, recrystallization | MS, IR, 1H-NMR, 13C-NMR, HMBC | Zhang et al. (2008) |
| 104 | Herpetosperin A | C22H24O11 | Ethyl acetate | Silica gel column chromatography, ODS silica gel CC, semi-preparative HPLC | MS, IR, 1H-NMR, 13C-NMR, HMBC | Xu et al. (2015) |
| 105 | Herpetosperin B | C22H24O11 | Ethyl acetate | Silica gel column chromatography, ODS silica gel CC, semi-preparative HPLC | MS, IR, 1H-NMR, 13C-NMR, HMBC | Xu et al. (2015) |
| 106 | Herpetospin C | C23H26O10 | Ethyl acetate | Reverse phase silica gel column chromatography | UV, IR, MS, 13C-NMR, 1H-NMR | Xu (2012) |
| 107 | Herpetolide H | C19H16O6 | Ethyl acetate | Normal phase silica gel column chromatography, recrystallization | MS, 1H-NMR, 13C-NMR | Huang et al. (2021) |
| 108 | Herpetospin D | C22H22O11 | Ethyl acetate | Normal phase silica gel column chromatography, recrystallization | MS, 1H-NMR, 13C-NMR | Xu (2012) |
| Others | ||||||
| 109 | Arginine | C6H14N4O2 | HCl | — | HITACHI 835-50 High-speed amino acid analyzer, XDY-I atomic fluorescence spectrometer | Li et al. (2005) |
| 110 | Histidine | C6H9N3O2 | HCl | — | HITACHI 835-50 High-speed amino acid analyzer, XDY-I atomic fluorescence spectrometer | Li et al. (2005) |
| 111 | Lysine | C6H14N2O2 | HCl | — | HITACHI 835-50 High-speed amino acid analyzer, XDY-I atomic fluorescence spectrometer | Li et al. (2005) |
| 112 | Leucine | C6H13NO2 | HCl | — | HITACHI 835-50 High-speed amino acid analyzer, XDY-I atomic fluorescence spectrometer | Li et al. (2005) |
| 113 | Isoleucine | C6H13NO2 | HCl | — | HITACHI 835-50 High-speed amino acid analyzer, XDY-I atomic fluorescence spectrometer | Li et al. (2005) |
| 114 | Tryptophan | C11H12N2O2 | HCl | — | HITACHI 835-50 High-speed amino acid analyzer, XDY-I atomic fluorescence spectrometer | Li et al. (2005) |
| 115 | Kaempferitrin | C27H30O14 | Ethyl acetate | Reversed phase silica gel column chromatography, semi-preparative HPLC | UV, IR, 1H-NMR, 13C-NMR | Fan et al. (2016) |
| 116 | 3′-Hydroxydaidzein | C15H10O5 | Ethyl acetate | Silica gel column chromatography, semi-preparative HPLC | MS, 1H-NMR | Dai et al. (2017) |
| 117 | Stigmasterol | C29H48O | Ethyl acetate | Silica gel column chromatography, recrystalization | MS, 1H-NMR, 13C-NMR | Liu et al. (2010) |
| 118 | β-Stigmasterol | C29H48O | Ethyl acetate | Silica gel column chromatography, Sephadex LH-20 | MS, 1H-NMR, 13C-NMR | Gong (2013) |
| 119 | Spinasterol glucoside | C35H56O6 | Ethyl acetate | Silica gel column chromatography, recrystalization | UV, IR, MS, 1H-NMR, 13C-NMR | Liu et al. (2010) |
| 120 | Arbutin, 1-acetate | C14H18O8 | Ethyl acetate | Reversed phase silica gel column chromatography | Uv, IR, MS, 1H-NMR, 13C-NMR | Hu (2016) |
| 121 | Herpetolide C | C16H14O6 | Petroleum ether | Silica gel column chromatography, sephadex LH-20, smi-preparative HPLC | UV, IR, MMS, 1H-NMR, 13C-NMR, HMQC, HMBC | Fan et al. (2016) |
| 122 | Eicosanoic acid, 2-propenyl ester | C23H44O2 | Ethyl acetate | Silica gel column chromatography, sephadex LH-20 | MS, 1H-NMR | Dai et al. (2017) |
| 123 | 3-Dodecen-1-yne | C12H20 | petroleum ether | — | GC-MS | Liu et al. (2005) |
| 124 | 2,6,10,14,18,22-Tetracosahexaen | C24H38 | petroleum ether | — | GC-MS | Zhang et al. (2004) |
| 125 | Herpecaudin | C17H22O4 | Ethyl acetate | RPHPLC, silica gel column chromatography, RP-18, sephadex LH-20 | MS,1H-NMR,13C-NMR, HMBC, NOESY, CD spectrum, X-ray | Jiang et al. (2016) |
4.1 Lignans
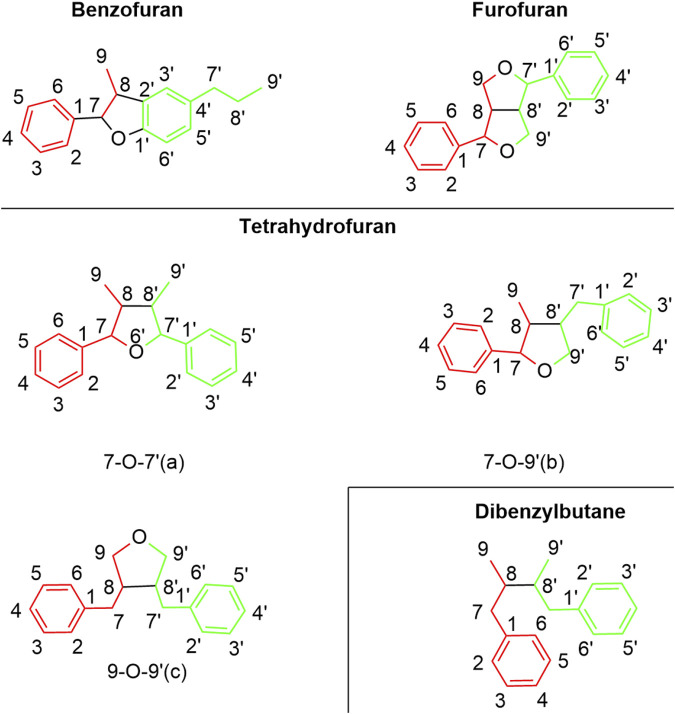
As collected in Figure 2 and summarized in Figure 3, lignans in H. pedunculosum seeds can be mainly divided into benzofurans, tetrahydrofurans and furofuran. In benzofuran lignan such as dehydrodiconiferyl alcohol (9) and herpetotriol (12), the benzene ring is linked to the side chain to form the furan oxygen ring (Figure 2; Table 1). In furfuran lignan, bimolecular phenylpropanin side chains are connected to form a bis-tetrahydrofuran ring, such as herpetetradione (4), herpetetrone (5) and herpetrione (6). Tetrahydrofurans lignans can be further divided into three types with 7-O-7' (a), 7-O-9' (b), and 9-O-9' (c) structures (Figure 2). The tetrahydrofuran of 7-O-9′ is predominant in H. pedunculosum seeds, represented by herpetriol (1) and herpetetrol (2). Beside above three main lignan types, H. pedunculosum seeds also contains dibenzylbutane (chemicals of 29 and 30) as shown in Figure 2.
FIGURE 3.
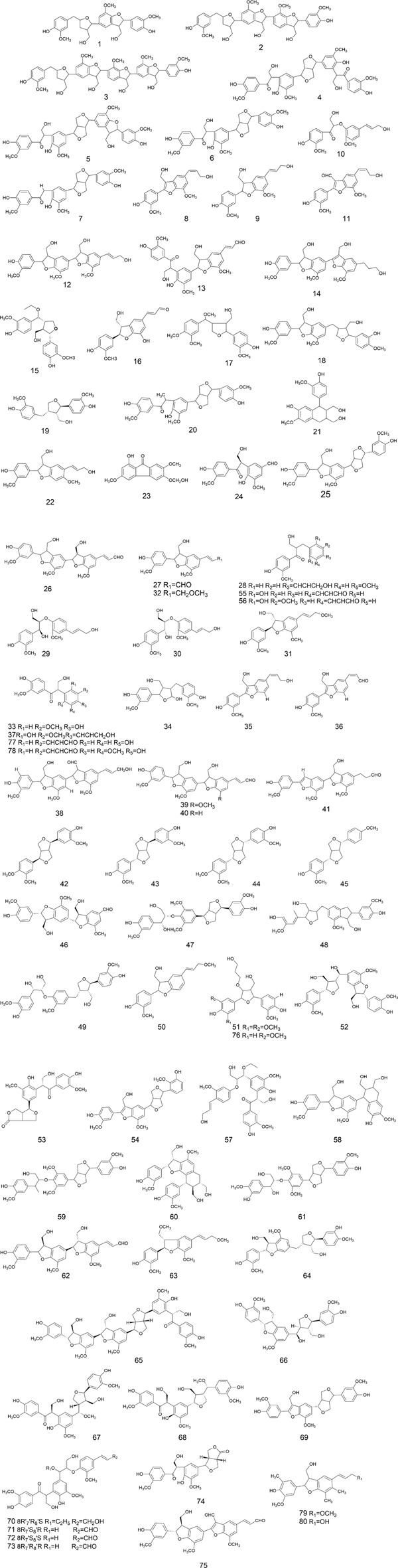
Structures of lignan metabolites in Herpetospermum pedunculosum seeds.
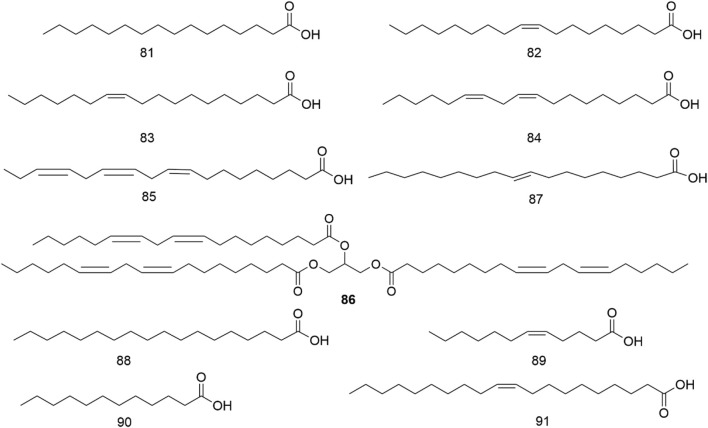
4.2 Fatty acids
It has been found that H. pedunculosum seeds contain various fatty acids (81-91, Figure 4; Table 1), with comparatively greater concentrations of linoleic (84) and linolenic acid (85) (Zhao et al., 2009). Oleic (82), palmitic (81), and linoleic acids (84) are reported to be physiologically active in decreasing blood cholesterol levels and alleviating the formation of cholesterol in the vascular wall (Dobrzyńska and Przysławski, 2020). Therefore, it is essential to study the fatty acids in H. pedunculosum seeds.
FIGURE 4.
Structures of fatty acids from Herpetospermum pedunculosum seeds.
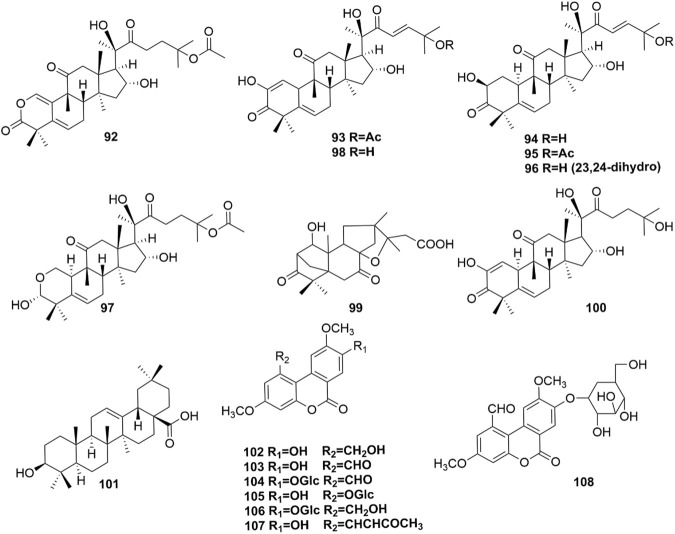
4.3 Terpenoids
Ten terpenoids (92-101, Figure 5) were identified in H. pedunculosum seeds, and triterpenoid was the dominant type among them. Triterpenoids have the activities of anti-inflammatory, antibacterial, and antiviral properties (Xiao et al., 2018). For example, cucurbitacin B (95) was reported to show anti-inflammatory, antioxidant, and neuroprotective effects (Dai et al., 2023). These bioactive triterpenoids in H. pedunculosum seeds doubtlessly contribute to its favorable pharmacologic actions.
FIGURE 5.
Structures of terpenoids (92–101) and coumarins (102–108).
4.4 Coumarins
Coumarin is widely acknowledged to have extensive biological activities including anti-tumor, anti-oxidation, anti-inflammation, and anti-coagulation (Kirsch et al., 2016; Wu et al., 2020). And there are 7 coumarins (102-108 in Figure 5) found in H. pedunculosum seeds up to now. For example, Huang et al. (2021) found that herpetolide H (107) from H. pedunculosum seeds had the effects of anti-inflammatory in vitro.
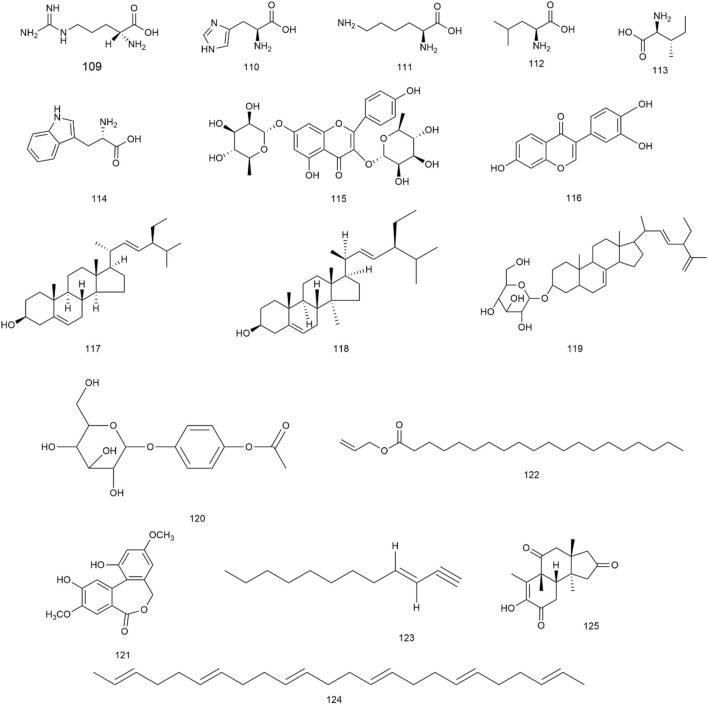
4.5 Others
In addition to the aforementioned metabolites, H. pedunculosum seeds also contain amino acids (109–114), flavonoids (115, 116), sterols (117–119), glucosides (120), esters (121, 122), olefin (123–124), and ketones (125) as illustrated in Figure 6. It is reported that leucine (112) and isoleucine (113) can prevent the fat accumulation from in hepatocyte (Zhang et al., 2022). Kaempferitrin (115) has anti-inflammatory and anti-oxidation effects (Patel D. K., 2021). The biological activity of stigmasterol (117) is found to include anti-inflammatory, antioxidant, and anti-cancer properties (Bakrim et al., 2022). Therefore, the role of these metabolites in the application of H. pedunculosum seeds deserves further research.
5 Pharmacology
Diverse studies have demonstrated the hepatoprotective, antioxidant, and anti-cholestasis effects of H. pedunculosum seeds and aforementioned metabolites. Especially, the action mechanism on liver protection effect of H. pedunculosum seeds was systematically generalized. The specific hepatoprotective action and other pharmacological effects were summarized in Table 2 and Table 3, respectively.
TABLE 2.
The hepatoprotective pharmacology of H. pedunculosum seeds.
| Liver disease | Extract/Compound | Animal/cell and intervention | Indicators and results (control, model, treatment, positive control groups) | Refs. |
|---|---|---|---|---|
| Hepatic fibrosis | Ethyl acetate (EAEHPS) | Animal: Sprague-Dawley rats (male) Model: Induction of CCl4 (50%, 3 mL/kg) Treatment: EAEHPS (1 and 3 g/kg) for 6 weeks Positive control: Silymarin (0.1 g/kg) for 6 weeks |
ALT↓, AST↓, TNF-a↓, IL-1β↓, IL-6↓, TGF-β1↓, NF-κB 65↓, IκBα↑, Smad3↑.(Compared with the model group) HA/μg . L-1: 49.35 ± 5.26, 75.37 ± 22.65, 61.27 ± 8.46 (L) 54.97 ± 8.63 (H), 39.94 ± 12.61; LN/μg . L-1: 73.55 ± 13.06, 131.74 ± 20.94, 110.38 ± 27.89 (L), 108.78 ± 6.61 (H), 112.87 ± 16.94; PCⅢ/μg . L-1: 75.57 ± 5.11, 117.65 ± 29.45, 98.38 ± 10.28 (L), 93.11 ± 10.19 (H), 88.60 ± 6.92; ColⅣ/μg L-1: 58.75 ± 23.14, 78.15 ± 14.70, 54.86 ± 16.03 (L), 46.31 ± 10.88 (H), 56.75 ± 15.14 | Feng et al. (2018a) |
| Chloroform | Animal: Sprague-Dawley rats (male) Model: Induction of CCl4 (50%, 3 mL/kg) Treatment: 1 and 3 g/kg) for 10 weeks |
GPT↓, GOT↓, TBIL↓, CP↓, HA↓, LN↓, PCIII↓, ColIV↓, TBA↓, MDA↓, CAT↑, SOD↑, ALB↑.(Compared with the model group) | Li et al. (2019) | |
| Hepatic fibrosis | Chloroform | Animal: KM mice Model: Induction of CCl4 (1%, 5 mL/kg) Treatment: 10 (L), 30 (M), 60 (H) g/kg for 1 week |
ALT/U . L-1: 26.07 ± 3.23, 121.04 ± 9.8, 53.99 ± 3.21 (L), 37.25 ± 9.80 (M), 30.40 ± 2.44 (H),/); AST/U . L-1: 66.09 ± 8.99, 231.84 ± 18.32, 139.67 ± 13.98 (L), 126.63 ± 8.53 (M), 99.63 ± 36.89 (H),/; MDA/nmol . mg-1: 6.35 ± 1.49, 11.74 ± 1.07, 8.80 ± 1.87 (L) 7.77 ± 0.32 (M) 7.01 ± 0.48 (H),/; SOD/U . mg-1: 45.20 ± 6.00, 22.80 ± 4.3, 30.99 ± 2.80 (L), 41.06 ± 1.73 (M), 36.91 ± 7.89 (H),/; Caspase-3:/, 0.1674 ± 0.0061, 0.1555 ± 0.0010 (L), 0.1356 ± 0.0099 (M), 0.1096 ± 0.0083 (H),/ | Jiang (2011) |
| Liver protection | Water | Animal: KM mice Model: Induction of CCl4 (1%, 5 mL/kg) Treatment: 10 (L), 30 (M), 60 (H) g/kg for 1 week |
ALT/U . L-1: 26.07 ± 3.23, 121.04 ± 9.8, 72.16 ± 4.9 (L), 59.59 ± 9.81 (M), 54.92 ± 7.03 (H),/; AST/U.L-1: 66.09 ± 8.99, 231.84 ± 18.32, 185.29 ± 19.63 (L) 172.25 ± 12.61 (M), 160.15 ± 12.91 (H),/; MDA/nmol . mg-1: 6.35 ± 1.49, 11.74 ± 1.07, 8.80 ± 1.87 (L), 7.77 ± 0.32 (M), 7.01 ± 0.48 (H),/; SOD/U . mg-1: 45.20 ± 6.00, 22.80 ± 4.3, 30.99 ± 2.80 (L), 41.06 ± 1.73 (M), 36.91 ± 7.89 (H),/; Caspase-3:/, 0.1674 ± 0.0061, 0.1505 ± 0.0062 (L), 0.1366 ± 0.0012 (M), 0.1026 ± 0.0096 (H),/ | |
| Chemical liver injury | Water | Animal: C57BL/6 male mice at 8–10 weeks of age Cell: BRL-3A and AML12 Model: Induction of APAP (300 mg/kg, 40 mM) Treatment: Water extract (0.3 mg/kg, 3 g/kg) in mice for 2 weeks; Water extract (100–400 μg/mL) in BRL-3A for 24 h. Water extract (100–400 μg/mL) in AML12 for 8 h |
ALT↓, AST↓, ROS↓, TNF-α↓, 1L-1β↓, HO-1↓, NQO1↓, Cell viability↑, GSH↑ | Li J. et al. (2023) |
| Drug-induced liver injury | Ethanol | Animal: C57BL/6 (male); Cell: BRL-3A Model: Induction of APAP (Cell: 40 mM, 8 h; Mice: 200 mg/kg); Treatment: ethanol extract (6.25, 12.5, 25 μg/mL) in BRL-3A; ethanol extract (0.3, 1, 3 g/kg) in mice for 15 days |
ALT↓, AST↓, ROS↓, MDA↓, Bax↓, Caspase3↓, Cleaved Caspase3↓, HO-1↑, NQO1↑, Cell viability↑, GSH↑ | Liao (2023) |
| Liver protection | Petroleum ether | Animal: Sprague-Dawley rats (male) Model: Induction of ANIT (60 mg/kg) Treatment: Petroleum ether extract of 350 (L), 700 (M), 1400 mg/kg (H) for 5 days Positive control: Ursodeoxycholic acid (UDCA) (100 mg/kg) for 5 days |
ALT↓, AST↓, ALP↓, γ-GTP↓, TBIL↓, DBIL↓, TBA↓, degree of tissue damage↓.(Compared with the model group) MDA/nmol . mg-1: 1.24 ± 0.04, 4.02 ± 0.06, 3.91 ± 0.49 (L), 2.61 ± 0.32 (M), 1.84 ± 0.09 (H), 2.65 ± 0.28); MPO/U . mg-1: 3.70 ± 0.42, 24.10 ± 4.26, 23.44 ± 3.01 (L), 19.79 ± 1.74 (M), 12.13 ± 0.64 (H), 15.62 ± 0.75; NO/μmol . L-1: 5.007 ± 2.678, 4.006 ± 0.732 (L), 3.523 ± 0.223 (M), 3.351 ± 0.194 (H), 2.678 ± 0.375; SOD/U . mg-1: 166.81 ± 10.80, (56.07 ± 4.62, 57.11 ± 4.19 (L), 62.56 ± 4.44 (M), 84.52 ± 7.02 (H), 109.02 ± 12.21; GST/nmol . min-1. mg-1: 56.15 ± 6.39, 37.40 ± 2.85, 38.66 ± 3.92 (L), 43.75 ± 2.59 (M), 47.60 ± 1.66 (H), 47.93 ± 3.27; NO/: 1.884 ± 0.122, 5.007 ± 2.678, 4.006 ± 0.732 (L) 3.523 ± 0.223 (M), 3.351 ± 0.194(H), 2.678 ± 0.375 |
Cao et al. (2017) |
| Chemical liver injury | Total lignans | Animal: ICR mice (male) Model: Induction of CCl4 (0.1%, 20 mL/kg) Treatment: Total lignans (0.375, 0.75, 1.5, 3 g/kg) for 7 days Positive control: Compound glycyrrhizin tablets of 113 mg/kg (P1) and biphenyl diester of 200 mg/kg (P2) for 7 days |
ALT/U . L-1: 50.68 ± 3.66, 259.70 ± 3.58, 231.81 ± 16.73 (0.375 g/kg) 210.71 ± 9.08 (0.75 g/kg) 218.25 ± 6.17 (1.5 g/kg) 202.86 ± 11.80 (3 g/kg), 194.85 ± 17.46 (P1) 220.29 ± 7.77 (P2); AST/U.L-1: 97.83 ± 8.04, 274.50 ± 7.35, 240.38 ± 12.23 (0.375 g/kg) 233.17 ± 17.42 (0.75 g/kg) 226.55 ± 16.93 (1.5 g/kg) 213.31 ± 27.07 (3 g/kg), 209.38 ± 11.61 (P1) 232.90 ± 11.61 (P2); ALP/U.L-1: 117.88 ± 12.99, 195.67 ± 16.08, 143.28 ± 12.46 (0.375 g/kg) 138.61 ± 10.53 (0.75 g/kg) 134.61 ± 12.73 (1.5 g/kg) 124.14 ± 14.72 (3 g/kg), 158.29 ± 9.55 (P1) 131.74 ± 21.67 (P2); MDA/nmol . mgprot-1: 12.54 ± 1.59, 35.32 ± 2.54, 23.64 ± 2.82 (0.375 g/kg) 20.72 ± 1.49 (0.75 g/kg) 19.73 ± 1.28 (1.5 g/kg) 16.03 ± 2.76 (3 g/kg), 17.43 ± 2.44 (P1) 20.67 ± 1.98 (P2); SOD/U . mgprot-1: 76.84 ± 3.59, 43.39 ± 1.72, 52.75 ± 2.58 (0.375 g/kg) 54.58 ± 3.24 (0.75 g/kg) 55.02 ± 1.20 (1.5 g/kg) 59.99 ± 2.35 (3 g/kg), 50.79 ± 1.93 (P1) 49.75 ± 1.93 (P2); GSH-Px/U . mgprot-1: 996.76 ± 81.60, 534.00 ± 50.58, 873.88 ± 96.38 (0.375 g/kg) 896.26 ± 151.70 (0.75 g/kg) 924.47 ± 125.97 (1.5 g/kg) 975.95 ± 152.21 (3 g/kg), 751.57 ± 46.27 (P1) 796.84 ± 83.47 (P2) | Zhao et al. (2015) |
| Hepatic fibrosis | Total lignans | Animal: Sprague-Dawley rats (male) Model: Induction of CCl4 (40%, 25 mg/kg) Treatment: Total lignans of 100 (L), 200 (M), 400 mg/kg (H) for 8 weeks |
ALT/U . L-1: 82.25 ± 5.47, 200.00 ± 22.60, 139.86 ± 21.05 (L) 106.63 ± 16.60 (M) 92.75 ± 18.42 (H), 87.75 ± 114.47; AST/U . L-1: 169.25 ± 13.96, 217.57 ± 33.76, 225.86 ± 9.86 (L) 202.38 ± 38.03 (M) 178.00 ± 31.96 (H), 185.50 ± 30.87; ALP/U . L-1: 158.00 ± 4.04, 201.29 ± 25.45, 151.14 ± 226.17 (L) 171.25 ± 31.32 (M) 145.50 ± 18.53 (H), 136.50 ± 28.4; TGF-β1/ng . L-1: 173.37 ± 2.94, 225.15 ± 17.99, 210.64 ± 11.67 (L) 196.79 ± 15.77 (M) 188.32 ± 16.64 (H), 193.11 ± 13.22; HA/ng . L-1: 248.21 ± 9.99, 313.55 ± 16.29, 291.63 ± 11.37 (L) 273.21 ± 19.14 (M) 272.20 ± 21.30 (H), 271.04 ± 10.42; HYP/μg . L-1: 672.15 ± 10.85, 810.04 ± 25.60, 791.46 ± 21.34 (L) 742.96 ± 27.21 (M) 728.60 ± 40.68 (H), 725.27 ± 19.86; SOD/μg . L-1: 10.88 ± 0.28, 9.04 ± 0.46, 9.40 ± 0.46 (L) 10.02 ± 0.44 (M) 10.23 ± 0.67 (H), 10.23 ± 0.39 | Liu et al. (2017a) |
| Acute alcoholic liver injury | Total lignans | Animal: KM mice (male) Model: 56° Beijing Red Star Erguotou wine Treatment: Total lignans of 15 (L), 25 (M), 35 mg/kg (H) for 30 days Positive control: Polyene phosphatidylcholine (135 mg/kg) for 30 days |
AST/U . L-1: 143.7 ± 12.0, 258.7 ± 28.3, 230.0 ± 23.3 (L) 200.2 ± 25.5 (M) 222.2 ± 28.2 (H), 185.8 ± 39.6; ALT/U . L-1: 56.5 ± 6.5, 155.0 ± 27.8, 1123.3 ± 26.1 (L) 92.8 ± 14.7 (M) 98.5 ± 15.3 (H), 99.8 ± 9.6; MDA/[nmol.(mg . pro)−1]: 1.07 ± 0.14, 1.99 ± 0.87, 1.69 ± 1.26 (L) 1.14 ± 0.27 (M) 1.21 ± 0.28 (H), 1.22 ± 0.15; XOD/[U.(mg pro)−1]: 13.7 ± 1.3, 5.3 ± 3.1, 6.5 ± 1.2 (L) 8.6 ± 1.7 (M) 7.4 ± 1.0 (H), 9.9 ± 22.9; Na+-K+-ATP/[μmolPi . (mg . pro . h)−1]: 0.98 ± 0.14, 0.38 ± 0.06, 0.63 ± 0.18 (L) 0.76 ± 0.08 (M) 0.82 ± 0.10 (H), 0.75 ± 0.07; SOD/[U.(mg . pro)−1]: 65.8 ± 5.1, 61.2 ± 2.8 (L) 62.7 ± 5.7 (M) 60.0 ± 4.3 (H), 59.6 ± 2.8; GSH-Px/[U.(mg . pro)−1]: 21.1 ± 7.9, 9.5 ± 2.5, 14.3 ± 1.2 (L) 12.2 ± 1.7 (M) 13.4 ± 0.7 (H), 13.7 ± 1.0 | Huang et al. (2017) |
| Chronic alcoholic liver injury | Animal: Wistar rats (male) Model: 56° liquor (8 mL/kg-15 mL/kg) for 8 weeks Treatment: Total lignans of 100 (L), 200 (M), 400 mg/kg (H) for 8 weeks Positive control: Yishanfu (95 mg/kg) for 8 weeks |
AST/U . L-1: 24.42 ± 2.79, 58.21 ± 14.83, 41.21 ± 7.69 (L) 29.92 ± 2.99 (M) 25.69 ± 10.74 (H), 36.05 ± 15.47; ALT/U . L-1: 11.34 ± 0.69, 51.53 ± 2.18, 34.83 ± 4.77 (L) 27.45 ± 1.82 (M) 331.99 ± 2.30 (H), 331.68 ± 5.09; MDA/[nmol.(mg . prot)−1]: 1.15 ± 0.33, 3.35 ± 1.15, 1.57 ± 0.19 (L) 1.09 ± 0.29 (M) 1.17 ± 0.29 (H), 1.32 ± 0.31; ADH/[nmol/(min . mg pro)]: 3.83 ± 0.82, 12.38 ± 3.60, 7.75 ± 2.89 (L) 5.99 ± 1.77 (M) 6.91 ± 1.42 (H), 8.39 ± 44.43; TG/mmol . L-1: 7.63 ± 0.73, 10.62 ± 0.74, 9.55 ± 0.99 (L) 8.51 ± 0.67 (M) 8.75 ± 0.63 (H), 8.01 ± 1.67; SOD/[U.(mg . prot)−1]: 423.81 ± 75.64, 193.52 ± 40.85, 317.09 ± 52.41 (L) 233.66 ± 64.95 (M) 296.12 ± 34.64 (H), 196.23 ± 80.47; GSH/[mg . (g . prot)−1]: 4.47 ± 1.81, 1.47 ± 0.47, 2.77 ± 0.39 (L) 3.60 ± 0.33 (M) 2.93 ± 0.53 (H), 2.11 ± 1.04; GSH-Px/[U . (mg . prot)−1]: 40.2 ± 4.45, 34.1 ± 3.85, 39.1 ± 4.85 (L) 39.5 ± 3.25 (M) 35 ± 2.71 (H), 41.5 ± 4.23; CAT/U . mL-1: 10.03 ± 1.13, 7.09 ± 1.26, 9.08 ± 0.51 (L) 9.17 ± 1.18 (M) 8.31 ± 0.95 (H), 9.36 ± 0.93; ALDH2/[nmol/(min . mg pro)]: 9.62 ± 1.96, 3.40 ± 1.33, 4.96 ± 1.59 (L) 5.78 ± 3.53 (M) 3.07 ± 1.37 (H), 5.83 ± 3.78 | Huang et al. (2018) | |
| Cholestatic liver injury | Animal: KM mice (male) Model: Induction of ANIT (0.4%, 80 mg/kg) Treatment: Total lignans (0.05, 0.1, 0.2, 0.4 g/kg) for 7 days Positive control: Bifendate Pills group (0.15 g/kg) for 7 days |
AST/U . L-1: 36.81 ± 11.13, 197.99 ± 11.67, 173 ± 21.48 (0.05 g/kg) 127.02 ± 11.07 (0.1 g/kg) 120.56 ± 16.87 (0.2 g/kg) 107.67 ± 44.34 (0.4 g/kg), 156.83 ± 16.49; ALT/U . L-1: 26.87 ± 14.69, 470.15 ± 18.68, 275.82 ± 17.69 (0.05 g/kg) 223.29 ± 42.17 (0.1 g/kg) 206.47 ± 25.35 (0.2 g/kg) 384.08 ± 26.11 (0.4 g/kg), 220.50 ± 46.87; ALP/U . L-1: 3.4 ± 0.6, 18.27 ± 2.53, 13.40 ± 1.87 (0.05 g/kg) 11.89 ± 3.12 (0.1 g/kg) 9.98 ± 2.04 (0.2 g/kg) 11.91 ± 1.36 (0.4 g/kg), 8.26 ± 2.23; TBA/μmol . L-1: 3.63 ± 0.35, 78.10 ± 8.38, 48.13 ± 8.98 (0.05 g/kg) 44.13 ± 13.28 (0.1 g/kg) 31.83 ± 5.84 (0.2 g/kg) 50.57 ± 17.10 (0.4 g/kg), 26.94 ± 110.15; TBIL/μmol . L-1: 1.62 ± 0.66, 191.57 ± 34.47, 106.56 ± 22.48 (0.05 g/kg) 41.65 ± 17.54 (0.1 g/kg) 229.89 ± 17.11 (0.2 g/kg) 96.07 ± 18.03 (0.4 g/kg), 41.96 ± 24.65; DBIL/μmol . L-1: 0.87 ± 0.19, 124.94 ± 18.72, 27.23 ± 9.13 (0.05 g/kg) 16.76 ± 10.48 (0.1 g/kg) 10.91 ± 6.21 (0.2 g/kg) 48.08 ± 21.09 (0.4 g/kg), 8.98 ± 3.92; SOD/U . mg-1: 527.97 ± 18.82, 243.02 ± 31.43, 297.27 ± 24.09 (0.05 g/kg) 2,295.93 ± 20.08 (0.1 g/kg) 322.70 ± 20.08 (0.2 g/kg) 312.37 ± 15.70 (0.4 g/kg), 368.28 ± 15.36); MDA/nmol . mg-1: 2.41 ± 0.92, 12.66 ± 1.61, 6.91 ± 0.95 (0.05 g/kg) 8.02 ± 2.18 (0.1 g/kg) 5.69 ± 1.27 (0.2 g/kg) 8.21 ± 2.56 (0.4 g/kg), 5.93 ± 2.28; CAT/U . mg-1: 22.96 ± 1.17, 8.87 ± 1.26, 12.56 ± 1.39 (0.05 g/kg) 17.97 ± 5.30 (0.1 g/kg) 13.53 ± 4.83 (0.2 g/kg) 18.77 ± 3.78 (0.4 g/kg), 19.41 ± 3.14; GSH-Px/mg.g-1: 132.54 ± 24.50, 21.51 ± 8.74, 54.45 ± 14.00 (0.05 g/kg) 70.80 ± 9.17 (0.1 g/kg) 83.14 ± 24.01 (0.2 g/kg) 77.28 ± 10.77 (0.4 g/kg), 89.81 ± 30.43; TNF-α/ng . L-1: 43.63 ± 2.07, 65.14 ± 7.40, 52.38 ± 3.34 (0.05 g/kg) 48.20 ± 1.91 (0.1 g/kg) 45.81 ± 2.09 (0.2 g/kg) 46.75 ± 3.10 (0.4 g/kg), 44.22 ± 2.5; MCP-1/ng . L-1: 31.11 ± 2.34, 226.06 ± 43.42, 155.01 ± 30.14 (0.05 g/kg) 117.14 ± 24.86 (0.1 g/kg) 110.79 ± 19.70 (0.2 g/kg) 154.40 ± 36.39 (0.4 g/kg), 129.28 ± 32.20 | Li et al. N. J. (2023) | |
| acute alcoholic liver injury | Total sterols | Animal: ICR mice (male) Model: Induction of CCl4 (0.3%, 10 mL/kg) Treatment: Total sterols extract(10, 20, 50 mg/kg) for 7 days Positive control: Silymarin (50 mg/kg) for 7 days |
AST↓, ALT↓, IL-1β↓, IL-6↓, COX-2↓, IL-10↑ | Liu et al. (2022) |
| Immunological liver injury | Fatty acid | Animal: Swiss mice Model: 2.5 mg BCG was given by tail injection Treatment: Fatty acid extract of 7 (L), 10 (M), 14.5 mL/kg (H) for 12 days Positive control: Bifendate (200 mg/kg) for 12 days |
MDA/nmol . mg-1: 16.26 ± 4.29, 20.56 ± 3.61, 16.51 ± 2.89 (L) 19.36 ± 3.01 (M) 19.29 ± 1.99 (H), 17.73 ± 1.01; ALT/U . L-1: 7.67 ± 1.27, 90.71 ± 16.62, 23.32 ± 8.30 (L) 45.83 ± 16.92 (M) 29.16 ± 16.90 (H), 18.84 ± 8.73; AST/U . L-1: 23.48 ± 4.39, 92.39 ± 10.81, 47.61 ± 5.37 (L) 51.54 ± 13.11 (M) 44.72 ± 15.61 (H), 54.44 ± 17.37; NO/μmol . L-1: 3.33 ± 1.69, 21.26 ± 8.20, 8.45 ± 2.13 (L) 14.21 ± 9.43 (M) 10.77 ± 3.70 (H), 7.38 ± 4.66; SOD/U.mg-1: 184.40 ± 17.25, 105.00 ± 22.71, 219.95 ± 16.13 (L) 196.19 ± 23.09 (M) 228.28 ± 27.69 (H), 127.89 ± 12.69 | Chen et al. (2014) |
| Liver protection | Animal: Sprague-Dawley rats (male) Model: Induction of CCl4 (3 mL/kg) Treatment: Fatty acid extract of 1(L), 2 (M), 4 g/kg (H) for 5 days Positive control: Bifendate (200 mg/kg) for 5 days |
TG/nmol . L-1: 0.67 ± 0.21, 2.18 ± 0.53, 1.10 ± 0.38 (L) 1.03 ± 0.40 (M) 0.82 ± 0.1 (H), 1.44 ± 0.34; HDL/nmol . L-1: 0.99 ± 0.19, 1.30 ± 0.11, 0.43 ± 0.32 (L) 0.50 ± 0.13 M) 0.59 ± 0.12 (H), 0.57 ± 0.11; LDL/nmol . L-1:1.03 ± 0.24, 1.65 ± 0.10, 1.11 ± 0.37 (L) 1.08 ± 0.33 (M) 1.14 ± 0.20 (H), 1.06 ± 0.19; MDA/[nmol.(mg prot-1)]: 1.30 ± 0.11, 0.43 ± 0.32 (L) 0.50 ± 0.13 (M) 0.59 ± 0.12 (H), 0.57 ± 0.11; SOD/U . L-1: 57.69 ± 15.08, 42.86 ± 10.76, 74.28 ± 17.91 (L) 97.30 ± 12.51 (M) 102.69 ± 29.39 (H), 100.57 ± 21.66; TBIL/Umol . L-1: 1.15 ± 0.98, 11.89 ± 3.87, 6.54 ± 1.58 (L) 5.67 ± 2.07 (M) 4.75 ± 1.09 (H), 9.7 ± 2.6; AST/U . L-1: 229.00 ± 35.03, 1084.86 ± 289.13, 1181.38 ± 178.33 (L) 1039.43 ± 244.18 (M) 310.10 ± 33.99 (H), 1394.20 ± 278.11; ALT/U . L-1: 39.60 ± 5.41, 1263.43 ± 361.30, 1285.38 ± 322.05 (L) 1109.14 ± 365.50 (M) 297.40 ± 76.87 (H), 1394.20 ± 278.11; ALP/U . L-1: 136.7 ± 23.3, 281.6 ± 36.30, 220.8 ± 34.3 (L) 185.0 ± 21.3 (M) 141.3 ± 27.8 (H), 191.4 ± 29.4 | Li et al. (2014) | |
| Immunological liver injury | Polysaccharide | Animal: KM mice (male) Model: ConA (30 mg/kg) injection into the tail vein Treatment: Polysaccharide of 0.71 (L), 0.99 (M), 1.44 g/kg (H) for 8 days Positive control: Bifendate (0.2 g/kg) for 8 days |
ALT/U . L-1: 9.0 ± 0.8, 143.6 ± 7.0, 130.2 ± 6.2(L) 115.9 ± 9.4 (M) 45.8 ± 4.7 (H), 42.6 ± 6.0; AST/U . L-1: 24.6 ± 2.4, 172.3 ± 9.4, 146.2 ± 15.4 (L) 124.2 ± 8.0 (M) 55.9 ± 4.4 (H), 172.3 ± 9.4; LDH/U . L-1: 1952.7 ± 133.7, 4606.6 ± 191.6, 3,948.3 ± 232.1 (L) 3,814.3 ± 227.8 (M) 3,187.9 ± 192.9 (H), 2,742.9 ± 179.3; NO/μmol . L-1: 2.3 ± 0.2, 6.7 ± 0.5, 4.5 ± 0.3 (L) 4.1 ± 0.5 (M) 3.6 ± 0.4 (H) 3.4 ± 0.2; IL-6/pg . mL-1: 30.4 ± 1.1, 74.5 ± 2.1, 56.5 ± 3.7 (L) 50.1 ± 2.8 (M) 45.7 ± 2.9 (H), 51.4 ± 3.2; MDA/[(nmol/mg . prot)]: 6.5 ± 0.3, 9.3 ± 0.5, 11.2 ± 0.7 (L) 9.0 ± 0.4 (M) 6.4 ± 0.7 (H), 7.5 ± 0.3; SOD/[(U/mg . prot)]: 187.6 ± 4.4 59.7 ± 4.3, 74.6 ± 3.1 (L) 99.9 ± 7.0 (M) 126 ± 10.7 (H), 90.9 ± 10.0; Degree of tissue damage↓ | Li et al. (2015a) |
| Acute alcoholic liver injury | Herpetfluorenone | Animal: C57BL/6 mice; Cell: BMSCs Model: Induction of CCl4 Treatment: 100 μM of Herpetfluorenone |
AST↓, ALT↓, ALP↓, TBA↓, MDA↓, ALB↑, SOD↑, GSH↑ | Yang et al. (2023) |
| Acute alcoholic liver injury | Herpetin | Animal: C57BL/6 mice (male)Cell: BMSCs; Model: Induction of CCl4
Treatment: 10 μM of Herpetin |
AST↓, ALT↓, AKP↓, ALB↑ | Ding et al. (2023) |
| Immunological liver injury | Herpetin | Animal: ICR mice (male); Model: ConA (20 mg/kg) injection into the tail vein; Treatment: 10 (L), 20 mg/kg (H) of herpetin for 7 days Positive control: Qingkailing injection (20 mg/kg) for 5 days |
iNOS: 0.215 ± 0.004, 0.290 ± 0.013, 0.275 ± 0.012 (L) 0.239 ± 0.009 (H), 0.237 ± 0.008; TNF-α: 0.130 ± 0.006, 0.166 ± 0.008, 0.145 ± 0.004 (L) 0.139 ± 0.005 (H), 0.141 ± 0.005; NF-κB: 0.129 ± 0.006, 0.150 ± 0.004, 0.153 ± 0.006 (L) 0.130 ± 0.002 (H), 0.141 ± 0.003; IFN-γ: 0.131 ± 0.006, 0.149 ± 0.006, 0.134 ± 0.003 (L) 0.132 ± 0.003 (H), 0.132 ± 0.005; IL-4: 0.104 ± 0.002, 0.129 ± 0.004, 0.121 ± 0.004 (L) 0.118 ± 0.002 (H) 0.117 ± 0.003; SOCS1: 0.120 ± 0.007, 0.081 ± 0.005, 0.087 ± 0.007 (L) 0.091 ± 0.008 (H), 0.105 ± 0.011 | Wang et al. (2016) |
| Immunological liver injury | Herpetin | Animal: ICR mice (male) Model: Induction of BCG (2.5 mg) +LPS (7.5 μg) Treatment: 10 (L), 20 mg/kg (H) of herpetin for 12 days Positive control: Qingkailing injection (20 mg/kg) for 12 days |
AST/U . L-1: 96.01 ± 9.40, 197.55 ± 8.1, 184.33 ± 15.86 (L) 161.59 ± 18.20 (H), 123.55 ± 11.07; ALT/U . L-1: 42.24 ± 1.52, 101.61 ± 5.05, 92.69 ± 2.75 (L), 68.35 ± 0.94 (M), 58.32 ± 2.44; LDH/U . L-1: 239.11 ± 20.05, 546.87 ± 18.16, 481.84 ± 9.04 (L) 393.70 ± 32.96 (H), 340.78 ± 16.13; MDA/nmol . mgprot-1: 15.98 ± 1.39, 38.41 ± 1.59, 35.61 ± 1.87 (L) 29.6 ± 1.52 (H), 24.38 ± 2.03; SOD/U . (mg . prot-1:97.47 ± 9.15, 68.08 ± 12.80, 75.75 ± 9.09 (L) 85.04 ± 8.75 (H), 88.64 ± 11.92; GSH-Px/U . mg . prot-1:545.37 ± 54.86, 292.78 ± 57.38, 380.12 ± 33.94 (L) 414.53 ± 48.03 (H), 463.56 ± 32.30 | Liu (2017b) |
TABLE 3.
The other pharmacology effects of Herpetospermum pedunculosum seeds.
| Effects | Extract/Compound | Animal/Cell and intervention | Indicators and results (control, model, treatment, positive control groups) | Refs. |
|---|---|---|---|---|
| Antioxidation | Chloroform | Animal: SD rats Model: Induction of CCl4 (50%, 0.6 mg/kg) Treatment: chloroform extract of 200 (L), 400 mg/kg (H) for 7 days. Water extract of 200 (L), 400 mg/kg (H) for 7 days Positive control: VitE (400 mg/kg) for 7 days |
MDA/nmol . mg-1 protein: 8.65 ± 2.89, 16.92 ± 4.75, 7.22 ± 1.94 (L) 7.34 ± 0.97 (H), 5.16 ± 0.64; SOD/unit . mg-1 protein: 316.68 ± 19.05, 237.62 ± 17.81, 292.83 ± 41.64 (L) 289.52 ± 40.07 (H), 289.00 ± 25.29; GSH-px/unit . mg-1 protein: 146.36 ± 24.67, 101.82 ± 24.17, 118.51 ± 18.36 (L) 121.68 ± 223.16 (H), 142.56 ± 16.61 | Fang et al. (2008) |
| Water | MDA/nmol . mg-1 protein: 8.65 ± 2.89, 6.89 ± 1.26 (L) 6.11 ± 0.48 (H), 5.16 ± 0.64 SOD/unit . mg-1 protein: 316.68 ± 19.05, 272.29 ± 25.97 (L) 308.15 ± 13.34 (H), 289.00 ± 25.29; GSH-px/unit . mg-1 protein: 146.36 ± 24.67, 142.48 ± 10.83 (L) 148.25 ± 12.35 (H), 142.56 ± 16.61 | |||
| Anti-fatigue | Chloroform | Animal: KM mice (the mice that could learn to swim, male) Treatment: chloroform extract of 80 (L), 160 (M), 320 mg/kg (H); Ethyl acetate extract of 80 (L), 160 (M), 320 mg/kg (H); n-Butanol extract of 80 (L), 160 (M) 320 mg/kg (H) for 30 days; herpetrione of 15 (L), 30 (M), 60 mg/kg(H) for 30 days |
Swimming time↑, survival time↑; HG/mg . g-1:9.99 ± 1.58,/, 10.40 ± 1.47 (L) 10.53 ± 1.56 (M) 10.58 ± 1.97 (H),/; LDH/U . L-1: 874.50 ± 64.22,/, 900.56 ± 143.87 (L) 942.11 ± 127.10 (M) 961.84 ± 70.95 (H),/; SOD/U . mL-1: 69.52 ± 9.79,/, 119.84 ± 16.13 (L) 118.50 ± 9.52 (M) 121.28 ± 8.44 (H),/; GSH-Px/U . L-1: 109.56/, 119.84 ± 16.13 (L) 118.50 ± 9.52 (M) 121.28 ± 8.44 (H),/; BLA/ng.100mL-1: 24.49 ± 1.99,/, 23.91 ± 2.85 (L) 23.19 ± 1.84 (M) 23.37 ± 1.67 (H),/; MDA/nmol . L-1:14.04 ± 2.07,/, 13.92 ± 1.58 (L) 13.56 ± 1.91 (M) 13.43 ± 1.42 (H),/ | Jin et al. (2016) |
| Ethyl acetate | Swimming time↑, survival time↑; HG/mg . g-1: 9.99 ± 1.58,/, 10.55 ± 1.60 (L) 10.90 ± 1.58 (M) 11.56 ± 1.28 (H),/; LDH/U . L-1: 874.50 ± 64.22,/, 916.63 ± 137.80 (L) 996.50 ± 112.53 (M) 1073.66 ± 140.79 (H),/; SOD/U . mL-1 : 69.52 ± 9.79,/, 118.69 ± 9.38 (L) 120.54 ± 11.04 (M) 123.78 ± 8.18 (H),/; GSH-Px/U . L-1: 109.56 ± 9.58,/, 118.69 ± 9.38 (L) 120.54 ± 11.04 (M) 123.78 ± 8.18 (H) 1,/; BLA/ng.100mL-1: 24.49 ± 1.99,/, 23.09 ± 1.70 (L) 22.16 ± 2.16 (M) 21.94 ± 2.24 (H),/; MDA/nmol . L-1: 14.04 ± 2.07,/, 13.26 ± 1.47 (L) 12.261 ± 1.13 (M) 11.77 ± 1.44 (H),/ | |||
| n-Butanol | HG/mg . g-1: 9.99 ± 1.58,/, 10.04 ± 1.44 (L) 10.40 ± 1.79 (M) 11.45 ± 1.14 (H),/; LDH/U . L-1: 874.50 ± 64.22,/, 914.42 ± 153.03 (L) 926.89 ± 111.32 (M) 1028.14 ± 104.08 (H),/; SOD/U . mL-1: 69.52 ± 9.79,/, 117.02 ± 17.47 (L) 120.28 ± 17.46 (M) 119.15 ± 8.56 (H),/; GSH-Px/U . L-1:/, 117.02 ± 17.47 (L) 120.28 ± 17.46 (M) 119.15 ± 8.56 (H),/; BLA/ng.100mL-1: 24.49 ± 1.99,/, 25.41 ± 2.18 (L) 24.54 ± 2.64 (M)23.44 ± 2.56 (H),/; MDA/nmol . L-1 : 14.04 ± 2.07,/, 13.63 ± 2.11 (L) 13.27 ± 1.70 (M) 13.09 ± 1.21 (H),/ | |||
| herpetrione | Swimming time↑, survival time↑; HG/mg . g-1; 9.99 ± 1.58,/, 10.87 ± 1.38(L) 11.67 ± 1.37 (M) 11.75 ± 1.25 (H),/; LDH/U . L-1:874.50 ± 64.22,/,1003.27 ± 92.20 (L) 1046.10 ± 109.91 (M) 1092.73 ± 109.60 (H),/; SOD/U . mL-1: 69.52 ± 9.79,/, 115.18 ± 11.96 (L) 10,220.44 ± 8.07 (M) 123.04 ± 11.36 (H),/; GSH-Px/U . L-1: 109.56 ± 9.58, 109.56 ± 9.58/, 115.18 ± 11.96 (L) 120.44 ± 8.07 (M) 123.04 ± 11.36 (H),/; BLA/ng.100mL-1: 24.49 ± 1.99,/, 22.40 ± 1.85 (L) 21.75 ± 1.78 (M) 20.91 ± 1.91 (H),/; MDA/nmol . L-1: 14.04 ± 2.07/, 12.15 ± 1.14 (L) 11.65 ± 1.24 (M) 11.50 ± 1.21 (H),/ | |||
| Anti-tumor | Lignans | Cell: BEL-7402, BEL-7404, HCT | IC50: 1.45 μg/mL, 1.68 μg/mL, 2.36 μg/mL | Yuan (2006a) |
| Anti-hyperuricemia | Ethanol | Animal: KM mice (male); Model: Intraperitoneal injection of potassium oxonate emulsion (300 mg/kg). Treatment: ethanol extract (100, 200, 400 mg/kg) for 10 days. Positive control: colchicine (0.3 mg/kg) for 10 days | UA↓, XO(/) | Wang et al. (2022b) |
| Anti-gouty arthritis | Weight↑, Articular swelling↓, IL-1β↓, TNF-α↓, UA↓ | Wang et al. (2022b) | ||
| Anti-cholestasis | Ethyl acetate | Animal: SD rats (male); Model: Induction of ANIT (60 mg/kg); Treatment: Ethyl acetate extract (100, 200, 400 mg/kg) for 7 days Positive control: UDCA (100 mg/kg) for 7 days |
ALT↓, AST↓, ALP↓, γ-GTP↓, TBIL↓, DBIL↓ TBA↓, GSH↓, SOD↓, GPx↓, CAT↓ | Wei et al. (2020) |
| Anti-skin inflammation | Ethanol | Animal: BALB/c mice (female); Cell: HaCat Model: Mice induced by IMQ for 7 days; Hacat cell induced by IFN-γ (2 ng/mL) for 24 h Treatment: ethanol extract (0.125, 1.25 and 12.5 μg/g) in mice for 7 days. Ethanol extract (12.5 mg/mL) in HaCat cell |
IFN-γ↓, TNF-α↓, IL-17A↓, ICAM-1↓, CXCL9↓ | Zhong et al. (2023) |
| Anti-candida albicans | Herpetin, herpetrione | — | Minimal inhibitory concentration of 10.5 μM and 9.2 μM, respectively | Dai et al. (2019) |
5.1 Hepatoprotective effect
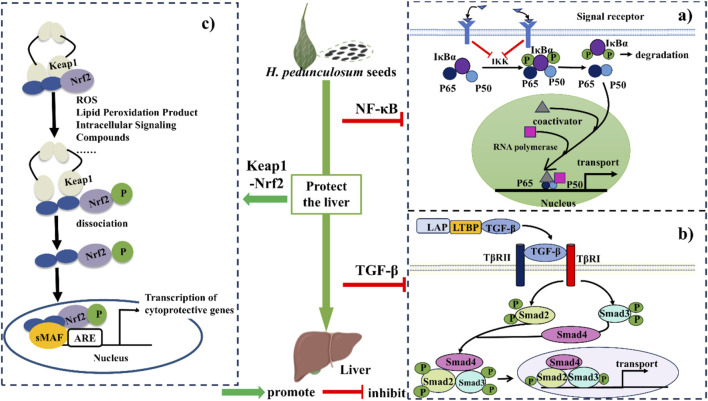
Liver is a vital metabolic organ implementing multiple functions such as toxicant detoxification, protein synthesis, and special compound production, thus the increasing prevalence of liver illnesses including fatty liver, liver damage, fibrosis, cirrhosis, and cancer aroused great attention nowadays (Asrani et al., 2019). As collected in Table 2, plentiful researches showed the remarkable hepatoprotective effect of H. pedunculosum seeds through the adjustment of some enzymes in animal models with the induction of CCl4, paracetamol (APAP), concanavalin A (ConA), α-naphthyl isothiocyanate (ANIT), liquor, bacillus calmette-guérin (BCG) and lipopolysaccharides (LPS). For instance, the ethyl acetate extract of H. pedunculosum seeds (EAEHPS) showed hepatoprotective activity against CCl4-induced hepatic fibrosis in rats via the inflammatory pathway with obviously inhibiting the expression of NF-κB (IκBα), Samd3, and TGF-β1 proteins (Feng et al., 2018a). The water extract of H. pedunculosum seeds could alleviate APAP-induced liver injury by inhibiting oxidative stress and ferroptosis through activating the Nrf2 signal pathway (Li N. Z. et al., 2023). In addition, some proteins, such as NLRP3, TLR-2, TLR-4, and JNK, will have their expression reduced by the total lignan of H. pedunculosum seeds (TLHPS), so as to protect mice against ANIT-induced liver damage (Li J. et al., 2023). Some metabolites such as herpetfluorenone (HPF, 23) and herpetin (18) from H. pedunculosum seeds were further found to have a positive pharmaceutical effect on acute liver injury by promoting the differentiation of bone marrow mesenchymal stem cells into hepatocellular-like cells and controlling autoimmune oxidation (Yang et al., 2023; Ding et al., 2023).
Based on above discussions and previous literatures, the hepatoprotective mechanism of H. pedunculosum seeds can be summarized into three pathways as illustrated in Figure 7. The first one is the inhibition of NF-κB signaling pathway to alleviate the inflammation during liver diseases (Figure 7A). Herpetospermum pedunculosum seeds can inhibit the phosphorylation of IκB through the inhibition of IKK, which in turn has anti-inflammatory and hepatoprotective effects (Li N. Z. et al., 2023). The second mechanism is inhibiting the TGF-β signaling pathway (Figure 7B). EAEHPS an inhibit the phosphorylation of Smad3, which in turn inhibits the expression of relevant genes after the complex enters the nucleus, thus playing a role in inhibiting liver fibrosis (Feng et al., 2018a). The third one is the promotion of Keap1-Nrf2 signaling pathway (Figure 7C). Nrf2 plays a crucial role in cellular defense against oxidative stress. When activated by H. pedunculosum seeds, the stability of Nrf2 increases, leading to reduced degradation and subsequent activation of genes driven by the antioxidant response element (ARE), thereby exerting a protective effect against liver damage (Li N. Z. et al., 2023; Liao, 2023).
FIGURE 7.
The mechanism of protective effect of Herpetospermum pedunculosum seeds on liver. (A) NF-κB signaling pathway; (B) TGF-β signaling pathway; (C) Keap1-Nrf2 signaling pathway.
5.2 Antioxidation
Fang et al. (2007), Fang et al. (2008) demonstrated the antioxidant activities of CHCl3, water and ethanol extracts of H. pedunculosum seeds to prevent lipid peroxidation brought on by CCl4 in vivo experiments. Jiang et al. (2020) tested the significant inhibitory activity of neocucurbitacin D (92) (IC50 = 15.27 ± 0.29 μM) and 23, 24-dihydrocucurbitacin B (97) (IC50 = 24.18 ± 0.26 μM) on XOD. Gong, (2013) showed that herpetone (7) has good DPPH free radical scavenging ability and antioxidant activity. Although many studies have shown that H. pedunculosum seeds has antioxidant effects, there are still some problems, such as the simplistic evaluation index and the unclear relationship between dose and activity.
5.3 Anti-cancer cells
The lignan of H. pedunculosum seeds demonstrated considerable in vitro inhibitory action against several cancer cells. The IC50 of lignans in H. pedunculosum seeds were 1.45 μg/mL, 1.68 μg/mL, and 2.36 μg/mL for human hepatocellular carcinoma cells (BEL-7402, BEL-7404), and HCT, respectively (Yuan, 2006a). Zhang et al. (2007) demonstrated the inhibitory effects of herpetolide A (102) and herpetolide B (116) on the growth of human promyelocytic leukemia cells (HL-60). The metabolites of H. pedunculosum seeds including including herpetosiol A (42), herpetosiol C (44), 7′, 8′-didehydlroherpepetotriol (14), herpetetrol (2), herpepropenal (13), herpetrione (6) showed significant cytotoxicity on human gastric adenocarcinoma cells (SGC7901), human lung cancer cells (A549), human breast cancer cells (MDA-MB-231), and human hepatocellular carcinoma cells (HepG2) (Ma, 2020; Kong et al., 2023). However, these studies only perform a simple detection of IC 50 and cytotoxicity, and lack other powerful indicators to reflect the efficacy of the drug. In addition, it is worth noting that the anti-tumor effects are mainly tested at the cellular level, lacking in animal and mechanism investigations, which are noteworthy in further research.
5.4 Anticholestasis effects
EAEHPS exerted an anti-cholestatic effect with increasing bile flow in a dose-dependent manner, which promoted bile acid transport by activating the farnesoid X receptor (FXR) signaling pathway (Wei, 2020). Meanwhile, the EAEHPS activated the Keap1-Nrf2 pathway to alleviate oxidative stress and inhibit of NF-κB/Are signaling pathway to inhibit inflammatory response, which could prevent and treat ANIT-induced cholestasis in rats (Wei et al., 2020).
5.5 Other effects
Wang S. W. et al. (2022) found that EAEHPS also had anti-hyperuricemia and anti-gouty arthritis activities, through reducing serum uric acid (UA) levels, suppressing the production and releasing pertinent inflammatory components, and lessening inflammatory damage and pathological tissue necrosis. Jin et al. (2016) demonstrated the anti-fatigue effects of ethanol extract of H. pedunculosum seeds with longer swimming time and hypoxia tolerance of experimental mice than that of the control group. The ethanol extract further showed a therapeutic effect on skin inflammation caused by imiquimod (Zhong et al., 2023). Moreover, Dai et al. (2019) showed that herpetin (18) and herpetrione (6) had favorable anti-candida albicans effects with minimal inhibitory connection of 10.5 μM and 9.2 μM, respectively.
6 Structure-activity relationship of lignan
Considering the key role of lignans in H. pedunculosum seeds, their structure-activity relationship was summarized according to previous literatures. For benzofuran lignans (Figure 2), H-5 can improve its anti-inflammatory capacity when it remains unchanged (Wang L. X. et al., 2022). The electron-withdrawing or electron-donor groups on the benzene ring of benzofuran lignans can decrease their anti-tuberculosis activity (Xu Z. et al., 2019). The anti-tumor activity of tetrahydrofuran lignans with the 7-O-9′ structure (Figure 2B.) can be increased by fixing the following sites: C-7′ is carbonyl group, H-5/5′is not substituted, and C-4/4′is methoxy (Wang L. X. et al., 2022). The antioxidant capacity of furofuran lignans is reported to decrease with the number of substituted methoxy groups on their benzene ring (Wang L. X. et al., 2022). Meanwhile, the presence of methoxy benzene in furofuran lignans enhances its toxicity to tumor cells (Xu W. H. et al., 2019), which could provide a structure-activity basis for the anti-tumor effect of herpetrione (6, Figure 3) (Yuan, 2006a; Yuan et al., 2006b; Yuan et al., 2011). For dibenzylbutane lignans (Figure 2), stronger antiviral activity can be achieved when the hydrogens at C-4 and C-5 are substituted by hydroxyl and methoxy groups respectively, and that at C-3'/4'/5′are substituted by methoxy or hydroxyl groups (Xu et al., 2022). Therefore, the separation and structural modification of lignan compounds from H. pedunculosum seeds show great potential for the development of drug leads.
7 Pharmaceutical analysis
Herpetospermum pedunculosum seeds are only stipulated qualitatively in the Chinese Pharmacopoeia, and their quantitative provisions are still lacking. The existing regulations are not enough to accurately evaluate the quality of H. pedunculosum seeds. Therefore, this section briefly introduces the latest research on modern analytical methods to provide guidance for quality evaluation for H. pedunculosum seeds.
Lignans such as herpetrione (6), herpetotriol (12), herpetin (18), and herpetfluorenone (23) are considered to be typical metabolites of the genus Herpetospermum and also the main active metabolites of H. pedunculosum seeds, which undoubtedly have a direct effect on the quality research of H. pedunculosum seeds and are indispensable to be detected. Wang, (2014) used herpetotriol (12) as the chemical reference materials in TLC to compare H. pedunculosum seeds from different areas. Cong et al. (2008) detected seven lignans from different areas by reversed-phase HPLC method. The results showed that herpetrione (6) was the most abundant among the seven metabolites, followed by herpetotriol (12). Qian et al. (2011) further accurately analyzed the average content of herpetrione (6) in 10 batches of H. pedunculosum seeds from different areas by UPLC, which was found to be 3.7223 mg g-1. Except lignan, other metabolites such as fatty acids and polysaccharides also contribute to the bioactivity of H. pedunculosum seeds, and their analysis are meaningful for the quality evaluation of H. pedunculosum seeds. Ling et al. (2018) detected four fatty acids in H. pedunculosum seeds by GC, the result showed that the content of oleic acid (82) was highest, followed by palmitic acid (81). Liu M. L., (2017a) combined UV-Vis and phenol-sulfuric acid methods to detect the polysaccharides in 10 batches of H. pedunculosum seeds, indicating higher polysaccharide content (2.16%) of H. pedunculosum seeds produced in Yunnan province. However, it is difficult to accurately evaluate the quality of H. pedunculosum seeds based on single or several metabolite analyses owing to its plentiful active metabolites, and establishing their fingerprint for similarity evaluation and principal component analysis could be a feasible choice in this aspect. Wang, (2014) found that there were 18 common peaks in the HPLC fingerprint, and the content of herpetolide A (102) was relatively high in all the samples to be analyzed. Subsequently, the HPLC fingerprint of H. pedunculosum seeds from Nyingchi region of Tibet was also studied, and 17 common peaks were identified, among which herpetrione (6) was the highest (Chen et al., 2020b).
In brief, among the active metabolites suitable for quantitative analysis, herpetrione (6) and herpetolide (102) exhibit various pharmacological activities with relatively high content, which have the potential to serve as markers for evaluating the quality of H. pedunculosum seeds. The current analysis methods for H. pedunculosum seeds are still far from perfect to establishing their quality evaluation system. It is urgent to elucidate the key indicative metabolites of H. pedunculosum seeds and develop standard determination methods capable of evaluating its quality comprehensively.
8 Processing
Processing methods are able to change the effect of H. pedunculosum seeds. For example, the stir-frying with grit can effectively alleviate the side effects of diarrhea caused by the shell of H. pedunculosum seeds. Meanwhile, the content of herpetrione (6) significantly decreased by 40.9% during this process, which can affect the clinical efficacy (Ling et al., 2018). Research further revealed that H. pedunculosum seeds processed by stir-frying with vinegar has better effects on protecting the liver and reducing enzymes, compared with sand owing to the lower herpetrione loss (12%) than that stir-frying with sand (41.4%). (Chen et al., 2016). Additionally, preparing the lignans of H. pedunculosum seeds into nanosuspension can improve their bioavailability and stability (Li et al., 2018; Shen et al., 2016). Therefore, the processing optimization could be a feasible approach to enhance the efficacy of H. pedunculosum seeds, which deserves more detailed research.
9 Application
The commercial herbal formulae including H. pedunculosum seeds and related details were collected in Table 4. For example, H. pedunculosum seeds is often combined with Swertia bimaculata, Terminalia schedule, and Carthami flos (1, 2, 3 in Table 4) to soothe the liver, promote bile flow, clear heat, and detoxify. When it is paired with Rosa multiflora, T. schedule, Phylanthus emblica (1, 2, 3, 4 in Table 4), formed compound medicines have the effects of strengthening the spleen, as well as promoting digestion. These summarizations and analyses supported the clinical practice of H. pedunculosum seeds and provided reference value for the development of other H. pedunculosum seed-derived prescriptions.
TABLE 4.
Formulations and preparations of H. pedunculosum seeds.
| No | Name | Composition | Efficacy | Refs. |
|---|---|---|---|---|
| 1 | Sanwei Qiangwei powder | Rosa multiflora, Herpetospermum caudigerum, Terminalia chebula | Clear heat and remove toxins, promote bile flow, For the treatment of Tri-pa and gallbladder diseases | Chinese Pharmacopoeia Commission (1995) |
| 2 | Jiuwei Zhangyacai pill | Swertia bimaculata, Herpetospermum caudigerum, Aconitum tanguticum, Ixeris polycephala, Berberis kansuensis, Lagotis Gaertn, Hypecoum erectum, Radix Aucklandiae, Chrysosplenium sinicum | Clear heat, anti-inflammatory, alleviates pain. For cholecystitis and incipient icteric hepatitis | |
| 3 | Wuwei Jinse pill | Terminalia chebula, Herpetospermum caudigerum, semen punicae granati, faeces soris scrofae, Radix aucklandiae | Clear heat and promote bile flow, promote digestion. For the treatment of jaundice hepatitis, and gallbladder pain | |
| 4 | Shiwei gaining pill | Carthami flos, Crocus sativus, Herpetospermum caudigerum, Meconopsis integrifolia, Dracocephalum tanguticum, Saxifraga stolonifera, Corydalis impatiens, Bear gallbladder, Calculus bovis, Brag-zhun, Calciosinti, and Turquoisis | It is used to treat fatty liver, viral hepatitis, liver fibrosis, cirrhosis, and other liver injuries | Feng et al. (2018b) |
| 5 | Qiwei hezi powder | Terminalia chebula, Herpetospermum caudigerum, Bombax ceiba, Amomum tsaoko Crevost, Syzygium aromaticum, Nardostachys chinensis, Piper longum | Clear heat and relieve pain. It is used for spleen enlargement, pain and spleen heat caused by strain injury | Yuandan and Song (1987) |
| 6 | Songshi pill | Songshi, Borneol, Syzygium aromaticum, Santalum album, Pulvis fellis ursi, Forest musk abelmosk, Herpetospermum caudigerum | Clear heat and remove toxins, soothe liver. It is used to treat liver pain, cirrhosis, hepatitis and cholecystitis | Xu et al. (2023) |
| 7 | Shiwei heibingpian powder | Borneol, Pomegranate seed, Cinnamomum cassia, Myristica fragrans, Piper longum、Terminalia chebula、Light halititum, Herpetospermum caudigerum、Holarrhena antidysenteriaca, Pulvis fellis ursi | It is used to treat nausea, cholecystitis, gallstones, and jaundice | Cang and De (2020) |
10 Conclusions and prospects
Herpetospermum pedunculosum seeds is a traditional Tibetan medicine with long history, rich chemical metabolites and high medicinal value. The research of H. pedunculosum seeds has achieved fruitful results and provided a scientific basis for the clinical medication. However, there are some shortcomings that need to be addressed in follow-up studies.
Although H. pedunculosum seeds is present in many Chinese patent medicines for the treatment of liver diseases, the interaction between the chemical metabolites in the prescriptions remains unclear and needs further investigation. Secondly, the supply of H. pedunculosum seeds is restricted due to the particular growth environment and limited wild resources. The large-scale cultivation of H. pedunculosum seeds could be of high research and economy value. Herpetospermum pedunculosum seeds are reported to contain 125 chemical metabolites, and lignan, terpenoids and coumarin are the main metabolites. Among them, lignan has been widely studied, which is usually recognized as the main pharmacological metabolite of H. pedunculosum seeds to exert hepatoprotective effect. However, the research on many other potentially active components such as polysaccharide is still in shortage. More advanced technologies can be used to extract, enrich, separate and purify the metabolites with low content and attention for better understanding of the medicinal material base of H. pedunculosum seeds. Moreover, there is a lack of structure-activity relationship studies of other active mmetabolites except lignans in H. pedunculosum seeds. The systematic structure-activity relationship studies can accelerate the synthesis of active metabolites and the development of related drugs derived from H. pedunculosum seeds.
The pharmacological effects of H. pedunculosum seeds, especially its effects on liver diseases, have been extensively researched. However, there are few in-depth studies on other pharmacological effects, and the current pharmacological research only remains at the cell and animal levels without comprehensive clinical research. Future research should take this as the direction to accelerate the clinical translation of drugs. Moreover, the quality standard of H. pedunculosum seeds still lacks the indicative components and standard detection method, which can be disadvantageous for standard pharmacology research and clinic practice. At present, some analytical methods have been used to detect the main bioactive ingredients with relatively high content such as herpetrione (6) and herpetolide (102), which may be a promising direction for better quality evaluation.
Although the current medical use of H. pedunculosum seeds is without processing, some studies have shown that H. pedunculosum seeds stir-fried with sand and vinegar can reduce their side effect of diarrhea and also the content of active ingredients. Therefore, the effect of processing method needs to be systematically determinated and optimized in combination with pharmacology and clinical research. In summary, this paper has comprehensively reviewed and analyzed the botany, phytochemistry, pharmacology, analytical methods and quality evaluation, processing and application of H. pedunculosum seeds, which can provide more insights for further research and development of traditional Tibetan medicine.
Acknowledgments
It is also appreciated for the assistance from the Public Platform of Pharmaceutical Research Center, Academy of Chinese Medical Science, Zhejiang Chinese Medical University and Shiyanjia Lab (www.shiyanjia.com).
Funding Statement
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. Financial support from Zhejiang Provincial Natural Science Foundation of China (LY24H290005), National Natural Science Foundation of China (82004208), and Research Project of Zhejiang Chinese Medical University (2023GJYY16; GQD23SH23, BZXCG-2022-37).
Author contributions
ZJ: Writing–original draft. CZ: Writing–original draft. XY: Writing–original draft. KW: Writing–original draft. ZS: Writing–original draft. WG: Writing–original draft. QaZ: Writing–original draft. XM: Formal analysis, Funding acquisition, Writing–review and editing. LQ: Writing–review and editing. QmZ: Writing–review and editing.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
- Asrani S. K., Devarbhavi H., Eaton J., Kamath P. S. (2019). Burden of liver diseases in the world. J. Hepatol. 70 (1), 151–171. 10.1016/j.jhep.2018.09.014 [DOI] [PubMed] [Google Scholar]
- Bakrim S., Benkhaira N., Bourais I., Benali T., Lee L. H., El Omari N., et al. (2022). Health benefits and pharmacological properties of stigmasterol. Antioxidants (Basel.) 11 (10), 1912. 10.3390/antiox11101912 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cang J., De J. (2020). Clinical observation on the treatment of jaundice hepatitis with Tibetan medicine Shiwei heibing tables. J. Med. Pharm. Chin. Minor. 26, 3–5. 10.16041/j.cnki.cn15-1175.2020.05.002 [DOI] [Google Scholar]
- Cao W. R., Ge J. Q., Xie X., Fan M. L., Fan X. D., Wang H., et al. (2017). Protective effects of petroleum ether extracts of Herpetospermum caudigerum against α-naphthylisothiocyanate-induced acute cholestasis of rats. J. Ethnopharmacol. 198, 139–147. 10.1016/j.jep.2017.01.003 [DOI] [PubMed] [Google Scholar]
- Chen H. G., Shen B. D., Shen C. Y., Li Z. M., Yuan H. L. (2016). Effects of different processing technology on active ingredients diarrhea and hepatoprotection of Herpetospermum caudigerum . Pharm. J. Chin. People's Lib. Army. 32, 289–292+298. [Google Scholar]
- Chen J. F. (2020). Study on potential toxicity, antibacterial activity, and chemical constituents of the shell of Herpetospermum pedunculosum. Master. Jiangsu: Nanjing University of Science and Technolog. [Google Scholar]
- Chen L., Zhang M., Yao H. E., Li C. Q., He C. Y. (2014). Protective effect of Herpetospermum seed oil on immunologic liver injury in the mice. West China J. Pharm. Sci. 29, 143–145. [Google Scholar]
- Chen X., Lu X. G., Feng X., Zhong G. J. (2020b). HPLC fingerprint of Tibetan medicine hertospermi semen from Linzhi Tibet. Cent. South Pharm. 18, 664–667. [Google Scholar]
- Chinese Materia Medica Commission (1998). Chinese Materia Medica. Shanghai: Shanghai Science and Technology Press. [Google Scholar]
- Chinese Pharmacopoeia Commission (1995). Pharmacopoeia of the people’s Republic of China, Beijing: China. Med. Sci. Press. [Google Scholar]
- Cong L. B., Yuan H. L., Wang Q., Li X. Y., Gong Q. F., Xiao X. H. (2008). Simultaneous determination of seven bioactive lignans in Herpetospermum caudigerum by RP-HPLC method. Biomed. Chromatogr. BMC 22 (10), 1084–1090. 10.1002/bmc.1028 [DOI] [PubMed] [Google Scholar]
- Dai S., Wang C., Zhao X. T., Ma C., Fu K., Liu Y. F., et al. (2023). Cucurbitacin B: a review of its pharmacology, toxicity, and pharmacokinetics. Pharmacol. Res. 187, 106587. 10.1016/j.phrs.2022.106587 [DOI] [PubMed] [Google Scholar]
- Dai Y. X., Hu S., Jiang H. Z., Tan R. (2019). Study on phenolic constituents from Herpetospermum caudigerum . Nat. Prod. R&D. 31 (02), 280–283. 10.16333/j.1001-6880.2019.2.016 [DOI] [Google Scholar]
- Dai Y. X., Wu W. J., Tan R. (2017). Study on the chemical components of the ethyl acetate extract from Herpetospermum caudigerum . North Am. J. Med. Sci. 10 (04), 136–138. [Google Scholar]
- Ding Y., Tan R., Gu J., Gong P. Y. (2023). Herpetin promotes bone marrow mesenchymal stem cells to alleviate carbon tetrachloride-induced acute liver injury in mice. Molecules 28 (9), 3842. 10.3390/molecules28093842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobrzyńska M. A., Przysławski J. (2020). The effect of camelina oil (α-linolenic acid) and canola oil (oleic acid) on lipid profile, blood pressure, and anthropometric parameters in postmenopausal women. Arch. Med. Sci. 17 (6), 1566–1574. 10.5114/aoms.2020.94033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong Z. Y., Wang H., Ma Y. X., Lan X. Z., Chen M. (2019). Chemical constituents from Herpetospermum caudigerum . Chin. Tradit. Pat. Med. 41, 341–344. [Google Scholar]
- Fan X. D., Wang G. W., Wang H., Ma J. X., Lan X. Z., Liao Z. H., et al. (2016). Herpetolide C: one new 7H-dibenzo[c,e]oxepin-5-one from Herpetospermum caudigerum . Acta Pharm. Sin. 51 (5), 770–774. 10.16438/j.0513-4870.2015-0938 [DOI] [PubMed] [Google Scholar]
- Fang Q. M., Zhang H., Cao Y. (2008). Anti-oxidant activities of the seed extracts of Herpetospermum pedunculosum against liver injury in rats. West China J. Pharm. Sci. 02, 147–149. 10.13375/j.cnki.wcjps.2008.02.007 [DOI] [Google Scholar]
- Fang Q. M., Zhang H., Cao Y., Wang C. (2007). Anti-inflammatory and free radical scavenging activities of ethanol extracts of three seeds used as “Bolengguazi”. J. Ethnopharmacol. 114 (1), 61–65. 10.1016/j.jep.2007.07.024 [DOI] [PubMed] [Google Scholar]
- Feng X., Li M. H., Xia J., Deng Ba D. J., Ruan L. Y., Xing Y. X., et al. (2018b). Tibetan medical formula Shi-Wei-Gan-Ning-Pill protects against carbon tetrachloride-induced liver fibrosis-an NMR-based metabolic profiling. Front. Pharmacol. 9, 965. 10.3389/fphar.2018.00965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng X., Zhong G. J., Deng Ba D. J., Yang B., Chen L. Y., Du Y. (2018a). Hepatoprotective effect of Herpetospermum caudigerum Wall. on carbon tetrachloride-induced hepatic fibrosis in rats. J. Cell. Mol. Med. 22 (07), 3691–3697. 10.1111/jcmm.13568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flora Reipublicae Popularis Sinicae Commission (1983). Flora Reipublicae Popularis Sinicae. Beijing: Science Publishing House. [Google Scholar]
- Gong F. Q. (2013). Studies on chemical constituents and pharmacological activity of the seeds of Herpetospermum caudigerum Wall. Master. Shandong: Shandong University of Traditional Chinese Medicine. [Google Scholar]
- Gong P. Y., Yuan Z. X., Gu J., Tan R., Li J. C., Ren Y., et al. (2016). Anti-HBV activities of three compounds extracted and purified from Herpetospermum seeds. Molecules 22 (1), 14. 10.3390/molecules22010014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu S. (2016). Study on the chemical constitutions of the ethyl acetate extract from Herpetospermum caudigerum. Master. Shandong: Southwest Jiaotong University. [Google Scholar]
- Huang D., Ma Y. X., Wei L., Sun Y., Zeng Q. H., Lan X. Z., et al. (2021). One new coumarin from seeds of Herpetospermum pedunculosum . China. J. Chin. Mater. Med. 46 (10), 2514–2518. 10.19540/j.cnki.cjcmm.20210125.601 [DOI] [PubMed] [Google Scholar]
- Huang S. Y., Gu J., Tan R., Shi Y. C., Liu W., Shi L. L. (2018). The protective effect and mechanism of total lignans from Tibetan medicinal Herpetospermum pedunculosum seeds on chronic alcohol-induced hepatic injury in rats. J. Chin. Med. Mater. 41 (02), 432–436. 10.13863/j.issn1001-4454.2018.02.041 [DOI] [Google Scholar]
- Huang S. Y., Sun N. Y., Sun W., Fu X. H., Gu J. (2017). Protective effect and mechanism of total lignans from Tibetan medicinal Herpetospermum seeds on alcohol-inducing acute hepatic injury in mice. China J. Chin. Mater. Med. 33, 66–70. 10.13412/j.cnki.zyyl.2017.05.018 [DOI] [PubMed] [Google Scholar]
- Jiang H. Z., Hu S., Tan R. X., Tan R., Jiao R. H. (2020). Neocucurbitacin D, a novel lactone-type norcucurbitacin as xanthine oxidase inhibitor from Herpetospermum pedunculosum . Nat. Prod. Res. 34 (8), 1728–1734. 10.1080/14786419.2018.1528592 [DOI] [PubMed] [Google Scholar]
- Jiang H. Z., Tan R., Jiao R. H., Deng X. Z., Tan R. X. (2016). Herpecaudin from Herpetospermum caudigerum, a xanthine oxidase inhibitor with a novel isoprenoid scaffold. Planta. Med. 82 (11-12), 1122–1127. 10.1055/s-0042-108210 [DOI] [PubMed] [Google Scholar]
- Jiang X. (2011). Protective effect of extracting solution of the Tibetan medicine Herpetospermum caudigerum seeds on acute liver injury mice produced by carbon tetrachloride. Master. Hubei: Huazhong Agricultural University. [Google Scholar]
- Jin S. Y., Li R. S., Shen B. D., Bai J. X., Xu P. H., Dai L., et al. (2016). Lignans-rich extract from Herpetospermum caudigerum alleviate physical fatigue in mice. Chin. J. Integr. Med. 22 (11), 840–845. 10.1007/s11655-016-2254-2 [DOI] [PubMed] [Google Scholar]
- Kaouadji M., Favre-Bonvin J. (1984). Herpetol, a new dimeric lignoid. from Herpetospermum caudigerum Wall. Z. Naturforsch., C. J. Biosci. 39, 307–308. 10.1515/znc-1984-3-419 [DOI] [Google Scholar]
- Kaouadji M., Jean F. B. (1983). Herpetrione, lignoide trimere isoled’Herpetospermum caudigerum. Tetrahedron Lett. 24, 5881–5884. 10.1016/S0040-4039(00)94226-6 [DOI] [Google Scholar]
- Kaouadji M., Jean F. B., Mariotte A. M. (1978). Herpetal, benzofuranne isolede Herpetospermum caudigerum . Phytochemistry 17, 2134–2135. 10.1016/s0031-9422(00)89299-7 [DOI] [Google Scholar]
- Kaouadji M., Jean F. B., Mariotte A. M. (1979). Herpetriol and Herpetetrol, new lignoids isolated from Herpetospermum caudigerum Wall. Z. Naturforsch., C. J. Biosci. 34, 1129–1132. 10.1515/znc-1979-1208 [DOI] [Google Scholar]
- Kaouadji M., Jean F. B., Sarrazin F., Davoust D. (1987). Herpetetrone, another tetrameric lignoid from Herpetospermum caudigerum seeds. J. Nat. Prod. 50, 1089–1094. 10.1021/np50054a013 [DOI] [Google Scholar]
- Kaouadji M., Pieraccini E. (1984a). Herpetetradione, nouveau lignoide tetramere isoled’Herpetospermum caudigerum wall. Tetrahedron Lett. 25, 5135–5136. 10.1016/s0040-4039(01)81544-6 [DOI] [Google Scholar]
- Kirsch G., Abdelwahab A. B., Chaimbault P. (2016). Natural and synthetic coumarins with effects on inflammation. Molecules 21, 1322. 10.3390/molecules21101322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong F. M., Wang C. R., Zhao L. L., Liao D. Y., Wang X. Q., Sun B. X., et al. (2023). Traditional Chinese medicines for non-small cell lung cancer: therapies and mechanisms. Chin. Herb. Med. 15 (4), 509–515. 10.1016/j.chmed.2023.05.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li C. Q., Wang Z. Y., Tang H. J., Wang C. D., Zhang M. (2015a). Protective effects of Herpetospermum polysaccharide on immunological liver injury induced by concanavalin A in mice. Pharmacol. Clin. Chin. Mater. Med. 31 (04), 94–97. 10.13412/j.cnki.zyyl.2015.04.029 [DOI] [Google Scholar]
- Li G., Wang X. Y., Suo Y. R., Wang H. L. (2014). Protective effect of seed oil of Herpetospermum pedunculosum against carbon tetrachloride-induced liver injury in rats. Saudi Med. J. 35 (9), 981–987. [PubMed] [Google Scholar]
- Li J., Lu Q., Peng M., Liao J., Zhang B., Yang D., et al. (2023). Water extract from Herpetospermum pedunculosum attenuates oxidative stress and ferroptosis induced by acetaminophen via regulating Nrf2 and NF-κB pathways. J. Ethnopharmacol. 305, 116069. 10.1016/j.jep.2022.116069 [DOI] [PubMed] [Google Scholar]
- Li J. J., Cheng L., Shen G., Qiu L., Shen C. Y., Zheng J., et al. (2018). Improved stability and oral bioavailability of Ganneng dropping pills following transforming lignans of Herpetospermum caudigerum into nanosuspensions. Chin. J. Nat. Med. 16 (1), 70–80. 10.1016/S1875-5364(18)30031-1 [DOI] [PubMed] [Google Scholar]
- Li L. Y., Shu S., Wei Y. F., Zhong G. Y., Chen J. H. (2005). Morphological and histological studies of Herpetospermum pedunculosum seeds and other substitutes. China J. Chin. Mater. Med. 14, 1073–1076. [PubMed] [Google Scholar]
- Li M. H., Feng X., Deng Ba D. J., Chen C., Ruan L. Y., Xing Y. X., et al. (2019). Hepatoprotection of Herpetospermum caudigerum Wall. against CCl4-induced liver fibrosis on rats. J. Ethnopharmacol. 229, 1–14. 10.1016/j.jep.2018.09.033 [DOI] [PubMed] [Google Scholar]
- Li N. Z., Shi Y. W., Guo M. X., Zhu J. X., Liang J., Feng Y. L. (2023). Study on protective effect and mechanism of total lignans from Tibetan medicine Herpetospermun caudigerum Wall against α-Naphthylisothiocyanate-induced liver injury in mice. Tradit. Chin. Drug Res. Clin. Pharmacol. 34 (01), 49–56. 10.19378/j.issn.1003-9783.2023.01.007 [DOI] [Google Scholar]
- Li Y. L., Zhou F. C., Wang H. L., Ding C. X., Suo Y. R. (2005). Analysis of amino acids in Tibetan medicine. Herpetospermum penduculosum Biot. Resour. 04, 11–12. [Google Scholar]
- Liao J. Q. (2023). Molecular mechanism research of ethanol extract of Herpetospermum pedunculosum seeds alleviating acetaminophen-induced liver injury . Master. Sichuan: Chengdu University. [Google Scholar]
- Ling H. L., Liu M. L., Zhang M. (2018). Simultaneous determination of four fatty acids in the fatty oil from Bolenggua seeds by GC-MS. Asia-Pacific Tradit. Med. 14, 37–40. [Google Scholar]
- Liu F., Zhu M. Q., Liu W. Y. (2022). Hepatoprotective effects of total sterols from Herpetospermum caudigerum Wall. shell on CCl4-induced acute liver injury in mice and quantification of major ingredients by RP-HPLC. World Sci. Technology-Modernization Traditional Chin. Med. 24, 2319–2330. [Google Scholar]
- Liu J., Chen X., Zhang Y., Zhang M. (2010). Chemical constituents of the ethyl acetate extract from Semen herpetospermi. Pharmacol. Clin. Chin. Mater. Med. 1 (03), 15–18. [Google Scholar]
- Liu M. L. (2017a). Primary studies on the methods for quality control and liver injury of active fractions of Herpetospermum caudigerum. Master. Sichuan: Chengdu University of Traditional Chinese Medicine. [Google Scholar]
- Liu S. Y. (2017b). Study on the protective effect and mechanism of herpetin, an effective component of Tibetan medicine Herpetospermum caudigerum Wall, on immune liver injury. Master. Sichuan: Southwest Minzu University. [Google Scholar]
- Liu W., Fu X. H., Huang S. Y., Shi Y. C., Shi L. L., Gu J. (2017a). Protective effect and mechanism of total lignans from Herpetospermum seeds on carbon tetrachloride-induced liver fibrosis in rats. China J. Chin. Mater. Med. 42 (3), 567–571. 10.19540/j.cnki.cjcmm.20161222.063 [DOI] [PubMed] [Google Scholar]
- Liu X. L., Han Y. P., Xu X. S., Song X. W. (2005). Analysis of fat oil from Bolenggua seeds by GC-MS. J. Southwest Univ. Nat. Sci. 05, 710–711. [Google Scholar]
- Luo D. S. (1997). Chinese Tibetan medicine. Beijing: Minzu press. [Google Scholar]
- Ma Y. X. (2020). Study on the chemical constitutions of the ethyl acetate extract from Herpetospermum pedunculosum. Master. Sichuan: Southwest University. [Google Scholar]
- Meng F. C., Ma Y. X., Zhan H. H., Zong W., Linghu L., Wang Z., et al. (2022). Lignans from the seeds of Herpetospermum pedunculosum and their farnesoid X receptor-activating effect. Phytochemistry 193, 113010. 10.1016/j.phytochem.2021.113010 [DOI] [PubMed] [Google Scholar]
- Patel D. K. (2021). Pharmacological activities and therapeutic potential of kaempferitrin in medicine for the treatment of human disorders: a review of medicinal importance and health benefits. Cardiovasc Hematol. Disord. Drug Targets 21 (2), 104–114. 10.2174/1871529X21666210812111931 [DOI] [PubMed] [Google Scholar]
- Qian Y. Z., Han J., Liu L. P., Zhang Y. Y., Guo J. J., Yuan H. L. (2011). Determination of Herpetrione in Herpetospermum caudigerum by ultrahigh performance liquid chromatography. Pharm. J. Chin. People's Lib. Army. 27, 527–528+531. [Google Scholar]
- Shen B., Chen H. G., Shen C. Y., Xu P. H., Li J. J., Shen G., et al. (2015). Hepatoprotective effects of lignans extract from Herpetospermum caudigerum against CCl4-induced acute liver injury in mice. J. Ethnopharmacol. 164, 46–52. 10.1016/j.jep.2015.01.044 [DOI] [PubMed] [Google Scholar]
- Shen G., Chen L., Wang L. Q., Zhong L. H., Shen B. D., Liao W. B., et al. (2016). Formulation of dried lignans nanosuspension with high redispersibility to enhance stability, dissolution, and oral bioavailability. Chin. J. Nat. Med. 14 (10), 757–768. 10.1016/S1875-5364(16)30090-5 [DOI] [PubMed] [Google Scholar]
- Wang H. (2005). Studies on the chemical components in bioactive part and HPLC fingerprint of the Tibetan medicinal substance Herpetospermum seed. Master. Sichuan: Chengdu University of Traditional Chinese Medicine. [Google Scholar]
- Wang L., Liu S. Y., Sun N. Y., Li J. C., Gu J. (2016). Protection of herpetin from Tibetan medicine Herpetospermum seeds on ConA-induced immunologic liver injury in mice. J. Southwest Univ. Nat. Sci. 42, 492–495. [Google Scholar]
- Wang L. X., Wang H. L., Huang J., Chu T. Z., Peng C., Zhang H., et al. (2022a). Review of lignans from 2019 to 2021: newly reported compounds, diverse activities, structure-activity relationships and clinical applications. Phytochemistry 202, 113326. 10.1016/j.phytochem.2022.113326 [DOI] [PubMed] [Google Scholar]
- Wang P. L. (2014). Research on quality control of the seeds of Herpetospermum pedunculosum (Sex.). Master. Chongqing: Southwest University. [Google Scholar]
- Wang S. W., Li Y. X., Du C. Y., Fan H. B., Wu X. Q., Chen X., et al. (2022b). Chemical constituents, anti-hyperuricemic and anti-gouty arthritis activities of extract of Herpetospermum caudigerum . Pharmacol. Res. 3, 100102. 10.1016/j.prmcm.2022.100102 [DOI] [Google Scholar]
- Wei X. D. (2020). Study on FXR-mediated anti-cholestasis effect and mechanism of the seeds of Herpetospermum pedunculosum. Master. Chongqing: Southwest University. [Google Scholar]
- Wei X. D., Fan X. D., Feng Z. Y., Ma Y. X., Lan X. Z., Chen M. (2020). Ethyl acetate extract of Herpetospermum pedunculosum alleviates α-naphthylisothiocyanate-induced cholestasis by activating the farnesoid x receptor and suppressing oxidative stress and inflammation in rats. Phytomedicine 76, 153257. 10.1016/j.phymed.2020.153257 [DOI] [PubMed] [Google Scholar]
- Wu Y., Xu J., Liu Y. T., Zeng Y. Y., Wu G. J. (2020). A review on anti-tumor mechanisms of coumarins. Front. Oncol. 10, 592853. 10.3389/fonc.2020.592853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao S. L., Tian Z. Y., Wang Y. F., Si L. L., Zhang L. Z., Zhou D. M. (2018). Recent progress in the antiviral activity and mechanism study of pentacyclic triterpenoids and their derivatives. Med. Res. Rev. 38 (3), 951–976. 10.1002/med.21484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu B. (2012). Studies on the chemical constituents of Herpetospermum caudigerum Wall. Master. Chongqing: Southwest University. [Google Scholar]
- Xu B., Liu S., Fan X. D., Deng L. Q., Ma W. H., Chen M. (2015). Two new coumarin glycosides from Herpetospermum caudigerum . J. Asian Nat. Prod. Res. 17 (7), 738–743. 10.1080/10286020.2014.996137 [DOI] [PubMed] [Google Scholar]
- Xu W. H., Zhao P., Wang M., Liang Q. (2019b). Naturally occurring furofuran lignans: structural diversity and biological activities. Nat. Prod. Res. 33 (9), 1357–1373. 10.1080/14786419.2018.1474467 [DOI] [PubMed] [Google Scholar]
- Xu X., Lu H. G., Xie X., Liu Y. T., Zhao H. Q., Lei C., et al. (2023). Ershiwuwei Songshi Pills improve CCL4-induced liver fibrosis in rats by regulating macrophage phenotype. Pharmacol. Clin. Chin. Mater. Med. 39, 17–23. 10.13412/j.cnki.zyyl.20230525.002 [DOI] [Google Scholar]
- Xu X. Y., Wang D. Y., Li Y. P., Deyrup S. T., Zhang H. J. (2022). Plant-derived lignans as potential antiviral agents: a systematic review. Phytol. Clin. Chin.Mater.Med. 21 (1), 239–289. 10.1007/s11101-021-09758-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z., Zhao S., Lv Z., Feng L., Wang Y., Zhang F., et al. (2019a). Benzofuran derivatives and their anti-tubercular, anti-bacterial activities. Eur.J. Med. Chem. 162, 266–276. 10.1016/j.ejmech.2018.11.025 [DOI] [PubMed] [Google Scholar]
- Yang B., Luo Q. L., Wang N., Hu Y. T., Zheng W. X., Li H., et al. (2023). HPF modulates the differentiation of BMSCs into HLCs and promotes the recovery of acute liver injury in mice. Int. J. Mol. Sci. 24 (6), 5686. 10.3390/ijms24065686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang F., Zhang H. J., Zhang Y. Y., Chen W. S., Yuan H. L., Lin H. W. (2010). A hepatitis B virus inhibitory neolignan from Herpetospermum caudigerum . Chem. Pharm. Bull. 58 (3), 402–404. 10.1248/cpb.58.402 [DOI] [PubMed] [Google Scholar]
- Yu J. Q., Hang W., Duan W. J., Wang X., Wang D. J., Qin X. M. (2014). Two new anti-HBV lignans from Herpetospermum caudigerum. Phytochem. Lett. 10, 230–234. 10.1016/j.phytol.2014.10.001 [DOI] [Google Scholar]
- Yuan H. L. (2006a). Extracts from Herpetospermum caudigerum Wall, their Dropping Pills, preparation methods, and applications. Patent. No: CN1857367A. [Google Scholar]
- Yuan H. L., Guo J. J., Li X. Y., Han J., Lv J. L., Fu S. S. (2011). Preparation and application of herperione and its capsule. Patent. No: CN102140101A. [Google Scholar]
- Yuan H. L., Liu Y., Zhao Y. L., Xiao X. H. (2005). Herpetin, a new bioactive lignan isolated from Herpetospermum caudigerum . J. Chin. Pharm. Sci. 03, 140–143. [Google Scholar]
- Yuan H. L., Yang M., Li X. Y., You R. H., Liu Y., Zhu J., et al. (2006b). Hepatitis B virus inhibiting constituents from Herpetospermum caudigerum . Chem. Pharm. Bull. 54 (11), 1592–1594. 10.1248/cpb.54.1592 [DOI] [PubMed] [Google Scholar]
- Yuandan K., Song T. (1987). Si Bu Yi dian. Shanghai: Shanghai Science and Technology Press. [Google Scholar]
- Zhang M., Deng Y., Zhang H. B., Chen H. L. (2007). Herpetolide, preparation method and application. Patent. No: CN101311173. [Google Scholar]
- Zhang M., Deng Y., Zhang H. B., Su X. L., Chen H. L., Yu T., et al. (2008). Two new coumarins from Herpetospermum caudigerum . Chem. Pharm. Bull. 56, 192–193. 10.1248/cpb.56.192 [DOI] [PubMed] [Google Scholar]
- Zhang M., Dong X. P., Deng Y., Wang H., Li X. N., Song Q. (2006). A new sesqui-norlignan from Herpetospermum pedunculosum . Acta Pharmacol. Sin. 41 (07), 659–661. 10.16438/j.0513-4870.2006.07.015 [DOI] [PubMed] [Google Scholar]
- Zhang M., Dong X. P., Wang H., Li X. N., Song Q. (2004). GC-MS analysis on the fatty oils of the Tibetan medicinal substance Herpetospermum. J. Chengdu Univ. Tradit. Chin. Med. 04, 49–52. [Google Scholar]
- Zhang Y., Lin S., Peng J., Liang X., Yang Q., Bai X., et al. (2022). Amelioration of hepatic steatosis by dietary essential amino acid-induced ubiquitination. Mol. cell 82 (8), 1528–1542.e10. 10.1016/j.molcel.2022.01.021 [DOI] [PubMed] [Google Scholar]
- Zhao X. E., Liu R. J., Wang H. L., Su Y. R. (2009). HPLC and GC-MS analysis of fatty acids Tibetan medicine Herpetospermum seed oil. Nat. Prod. R&D. 21 (01), 76–83. 10.16333/j.1001-6880.2009.01.022 [DOI] [Google Scholar]
- Zhao X. R., Zhang L., Li J. C., Gu J. (2015). Protective effect of total lignans from Tibetan medicinal Herpetospermum seeds on experimental liver injury by CCl4 . J. Southwest Univ. Nat. Sci. 41 (02), 181–185. [Google Scholar]
- Zhong Y., Zhang B. W., Li J. T., Zeng X., Pei J. X., Zhang Y. M., et al. (2023). Ethanol extract of Herpetospermum caudigerum Wall ameliorates psoriasis-like skin inflammation and promotes degradation of keratinocyte-derived ICAM-1 and CXCL9. J. Integr. Med. 21 (6), 584–592. 10.1016/j.joim.2023.11.004 [DOI] [PubMed] [Google Scholar]
- Zhou X. S. (2014). Systematic study on the lignans compounds of Herpetospermum caudigerum. Master. Sichuan: Southwest Jiaotong University. [Google Scholar]