Abstract
Most of advanced non-small cell lung cancer (NSCLC) patients will experience tumor progression with immunotherapy (IO). Preliminary data suggested an association between high plasma HGF levels and poor response to IO in advanced NSCLC. Our study aimed to evaluate further the role of the HGF/MET pathway in resistance to IO in advanced NSCLC. We included retrospectively 82 consecutive NSCLC patients from two academic hospitals. Among them, 49 patients received ICIs alone or in combination with chemotherapy (CT), while 33 patients received chemotherapy alone as the control group. We analyzed plasma HGF levels by ELISA and expression of PD-L1, MET/phospho-MET, and CD8+ T-Cell infiltration on lung tumor tissue by immunohistochemistry. We investigated the contribution of HGF/MET to IO response by culturing peripheral blood mononuclear cells (PBMC) with or without pembrolizumab, with recombinant HGF, or cocultured with NSCLC patients-derived explants. Additionally, c-MET inhibitors were used to evaluate the contribution of MET activation in NSCLC-mediated immunosuppression. High HGF levels were associated with high progression rate with IO (p = 0.0092), but not with CT. ELISA analysis of supernatants collected from cultured NSCLC cells showed that HGF was produced by tumor cells. Furthermore, when activated PBMCs were cultured in the presence of recombinant HGF or on NSCLC monolayer, the proliferation of CD3+CD8+ lymphocytes was inhibited, even in the presence of pembrolizumab. The addition of HGF/MET inhibitors restored lymphocyte activation and induced IFNγ production. In conclusion, inhibiting the HGF/MET signaling pathway could be a promising approach to enhance the efficacy of immunotherapy.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00262-024-03882-4.
Keywords: non-small cell lung cancer, immunotherapy, resistance, HGF, MET
Introduction
Lung cancer is the first cause of cancer deaths worldwide. Most of the cases are diagnosed at a metastatic stage with a survival of less than 5% at 5 years 1(1). Immune checkpoint inhibitors (ICIs) are able to restore an anti-tumor immunological response. They are the corner stone of the first-line treatment of advanced non-small cell lung cancer (NSCLC) without oncogenic addiction, either in monotherapy in case of high programmed death-ligand-1 expression (PD-L1 > 50%) by tumor cells or in combination with platinum-based chemotherapy2–4. However, nearly 50% of patients do not experience tumor response with ICIs5. The identification of biomarkers related to treatment resistance remains a major challenge.
HGF (hepatocyte growth factor) is a growth factor secreted by stromal mesenchymal cells and by neutrophils. MET (mesenchymal–epithelial transition) is a pro-oncogene encoding the epithelial MET receptor. The MET receptor is a member of the tyrosine kinase receptor family. HGF is the ligand of MET, inducing homo- or heterodimerization of MET with other receptors with tyrosine kinase activity thereby activating several downstream signaling pathways (Ras-Map kinase, PI-3K-AKT-mTOR, JAK-STAT pathways)6. Physiologically, the HGF/MET pathway plays a key role in "invasive growth"7 involving mechanisms of migration, differentiation, proliferation, and cell survival. Activation of MET by HGF promotes "epithelial-mesenchymal transition" (EMT) by disruption of intercellular contacts, detachment, and then migration of cells.
Recently, we performed a pilot study that analyzed the expression levels of 48 plasma biomarkers in 35 consecutive patients with advanced NSCLC treated with ICIs, using ELISA Multiplex assay8. We showed a significant association between circulating hepatocyte growth factor (HGF) levels at the beginning of ICI therapy and treatment efficacy. Progression-free survival (PFS) and overall survival (OS) were significantly shorter in the group with high HGF levels at baseline.
Therefore, we hypothesize that the activation of the HGF/MET pathway is associated with resistance to ICIs given in first-line treatment in advanced NSCLC. We performed translational and in vitro studies to investigate this hypothesis.
Material and methods
Patients and samples
All consecutive patients with treatment-naive stage IIIb, IIIc, or IV NSCLC (according to the 8th classification) from the Department of Pulmonology and Thoracic Oncology at two academic centers (APHP—Ambroise Paré Hospital and APHP—Bicêtre Hospital), who received first-line treatment with chemotherapy, immunotherapy, or a combination of both between September 2017 and January 2022, were included. Some patients from this cohort, specifically those included between 2017 and 2018, have been previously described8. Plasma samples were prospectively collected from NSCLC patients treated at Ambroise Paré Hospital, after signature of an informed consent (CPP IDF n°VIII). Tumor biopsies were collected at diagnosis at the Ambroise Paré Hospital and Bicêtre Hospital, and stored as formalin-fixed paraffin-embedded tissue blocks. PBMC (peripheral blood mononuclear cells) were obtained from peripheral blood samples of healthy donors. We have evaluated the overall response rate at the first tumor evaluation, PFS and OS, according to plasma concentration of the circulating biomarkers, and histologic expression of tissue biomarkers. Tumor response was evaluated every two months using RECIST 1.1 criteria.
Reagents and antibodies
ELISA assay kits for HGF (ab275901) and interferon-γ (IFN-γ, ab100537) were provided by ABCAM (Cambridge, UK). Phytohemagglutinin (00-4977-03) for PBMC activation was from Thermo Fisher Scientific (Paris, France). The antibodies used for flow cytometric analysis of lymphocytes, including mouse anti-huCD45-BV421 (HI30, 563879), mouse anti-CD3-PE-Cy7 (SK7, 557851), and anti-huCD8-APC, were all from Thermo Fisher Scientific (Paris, France). For IHC, anti-hu-phospho-Met ((Tyr1234/1235) was provided by Thermo Fisher Scientific (Paris, France). CFSE for cell proliferation assays was from Thermo Fisher Scientific (C34554, France). MTS colorimetric (ab197010) assay was acquired from ABCAM (Cambridge, UK). Pembrolizumab was a kind gift of the Pharmacy Unit of APHP-Ambroise Paré Hospital (Boulogne-Billancourt, France). c-MET/HGF inhibitors, crizotinib (PZ0191) and SGX-523 (S1112), were acquired from Sigma-Aldrich (Darmstadt, Germany) and Selleckchem (Munich, Germany), respectively.
ELISA assay
ELISA assay to quantify soluble HGF and IFN-γ in plasma samples and media collected from NSCLC culture was performed according to the manufacturer’s instructions (ab275901 and ab100537, respectively). Briefly, standards protein and plasma samples diluted at least at 1/2 ratio in sample buffers were added in ELISA well plates, then probed both with streptavidin conjugated primary antibodies and HRP conjugate secondary antibody for 90 min. The assays were revealed through the addition of HRP substrate. The plates were analyzed using a spectrophotometric plate reader (Bio rad PR 3100 EIA PhD software), set at a wavelength of 450 nm. Protein concentrations were calculated from their optical density using the standard curve. All samples standards and negative controls were tested in duplicate.
Immunohistochemistry (IHC)
Treatment-naive tumor sections (3 µm) were probed with anti-Pho-c-Met/c-MET antibodies through an automated technique (LEICA, BOND-III). Slides were read with optical microscope in blinded manner by a thoracic pathologist (CJ). Percentage of positive cells was obtained by cell counting. Tumor membrane labeling for pMET was expressed according to the number of positive tumor cells, classifying the samples in two groups: low expression (no labeling to low intensity labeling < 10% of tumor cells) and high expression (moderate 10–50% of tumor cells to high intensity labeling > 50% of tumor cells)9. Epithelial and alveolar macrophages cells on the same slide were considered as negative and positive controls, respectively.
Cell cultures and cocultures
NSCLC cell lines
Human NSCLC lines (A549 and H596) were obtained from ATCC and were maintained in complete DMEM/F12 medium (DMEM/F12 supplemented with 10% fetal bovine serum (FBS), 1% L-Glutamine (l-Glu), and 1% penicillin/streptomycin (P/S)). Cells were grown in 96 well plates as confluent monolayer for coculture experiments.
Primary NSCLC cultures
Primary NSCLC cells were isolated from immunohistochemically confirmed fresh lung cancer samples obtained at the time of NSCLC surgery. All resected samples exhibited approximately 80–100% cancer cells, as confirmed by the pathologist. The resected fresh lung cancer tissues were collected and rinsed in RPMI medium, then cut into small size, and rinsed with sterile phosphate-buffered saline (PBS); then, sample was digested in Trypsin–EDTA for 1 h at 37 °C. Digested samples were then washed and cultured in DMEM/F12 containing 10% FBS, 1% GlutaMAX, 10 mM HEPES, and Primocyn. At passage 2, cells were used for culture and coculture experiments. Cancer cells were characterized using a fluorescent-conjugated pancytokeratin antibody (MA110325, Life Technologies France).
PBMC isolation and culture/coculture
PBMCs were isolated from whole blood samples collected from a healthy donors using density gradient centrifugation with Lymphoprep (STEMCELL Technologies). Isolated PBMC, stained or not with carboxyfluorescein succinimidyl ester (CFSE, for proliferation assays), were maintained in complete RPMI or IMDM media for monoculture or coculture assays, respectively. PBMC were activated with 10 μg/mL phytohemagglutinin, and cultured in the presence of HGF or on an established primary NSCLC cells monolayer. To evaluate the contribution of HGF to lymphocyte activation or ICIs-mediated activation of lymphocytes, culture media were supplemented with pembrolizumab (20 nM corresponding to 3 µg/mL) and/or c-MET/HGF inhibitors (crizotinib (1 µM) and SGX-523 (0.5 µM)). Lymphocyte activation/proliferation was assessed either through MTS colorimetric assay or CFSE staining assay.
MTS proliferation assay
Lymphocyte proliferation was assessed using MTS proliferation assays as previously described10. Briefly, at the end of cell culture, 10 µL of MTS reagent (ABCAM, ab197010) was added into each well and keeping in incubator for 2 h. Metabolically active, viable cells converted MTS into a colored formazan. The product was then measured at 490 nm in a spectrophotometric microplate reader (Bio rad PR 3100 EIA PhD software). The viability was expressed as the percentage of optical density of treated cells compared to optical density of cells treated with the specific vehicle.
CFSE staining and flow cytometry.
To assess immunomodulatory properties of CSF-1, pembrolizumab, or NSCLC cells, PBMC were stained before each experiment with 1 μM carboxyfluorescein succinimidyl ester (CFSE; Life Technologies). The proliferation was assessed by flow cytometry on viable CD45pos cells CD3pos cells and/or CD3posCD8pos cells by using FlowJo software (TreeStar) as relative CSFE dilution of treated cells compared to cell treated with specific vehicle as previously described10,11.
Statistical analysis
All statistical analyses were conducted using Excel (Microsoft Office), XLSTAT (Addinsoft, 2023), and GraphPad Prism version 5 software (La Jolla, CA, USA). Statistical comparisons were conducted using Student's t-test and one-way ANOVA, respectively, or the Mann–Whitney and the Kruskal–Wallis methods for nonparametric distribution. Associations between variables were tested using Pearson's Chi-square or Fisher's exact test. Survival curves were generated based on HGF levels using the Kaplan–Meier method, followed by the log-rank test. Multivariate analyses were performed using logistic regression and Cox proportional hazards for drug response and patient survival, respectively. Differences were considered significant for p values < 0.05. Subsequently, the expression values were categorized as high (above the first quartile, > Q1) or low (below the first quartile, < Q1) based on the distribution of HGF expression values.
Results
Clinical characteristics of NSCLC patients
We enrolled 82 consecutive NSCLC patients. Among them, 49 patients received ICIs alone or in combination with chemotherapy, while 33 patients received chemotherapy alone as the control group. All patients had at least one pretreatment sample available, either plasma or tissue, obtained from a pretreatment biopsy. Table S1 provides a summary of the patients' characteristics. Most patients were male (70%) and had a history of smoking or were former smokers (95%). Adenocarcinoma was the most prevalent histologic type (68%). The median age at diagnosis was 66 years, ranging from 37 to 91 years. Finally, most patients (65%) had a performance status (PS) of 0–1 at the start of treatment. After 10.9 months of median follow-up, 25 (51%) patients treated with ICIs had died, including 37 (76%) patients who had experienced tumor progression.
Expression of phospho-MET, HGF, and correlation with demographics
To investigate the impact of HGF/MET pathway expression in NSCLC patients treated with ICIs or chemotherapy, we assessed the expression of markers of this pathway: phospho-MET (pMET), the activated form of the MET receptor, and HGF. pMET was assessed through IHC of tumor biopsies collected before treatment initiation in both groups (ICIs-treated patients n = 22 patients; CT-treated patients n = 8 patients) (Figure S1A, B). In patients treated with ICIs, pMET was highly expressed in 19 out of 21 patients (90%) and weakly expressed in 2 patients (10%). Besides, all 8 patients in the CT-treated group exhibited high pMET expression (100%) (n = 8/8; 100%) (Figure S1B).
We next analyzed the expression of HGF in plasma samples collected from patients at the diagnostic, first evaluation and progression (Figure S1C, D). The median HGF expression level in the total population was 1153 pg/ml (IQR 545-2300) at diagnosis. In the ICIs-treated population (ICIs alone or in combination with chemotherapy), the median HGF level before treatment was 892 pg/ml (IQR 376.8-1580). For patients treated with ICIs alone, the median HGF level before treatment was 891 pg/ml (IQR 379.4-1488). For patients treated with combination therapy (ICIs + CT), the median HGF level was 1092 pg/ml (IQR 375-1661) before treatment. The median expression level for the CT group was 1530 pg/ml (IQR 1009-2681) before treatment. We also analyzed longitudinal HGF levels according to the time, demonstrating a significant decrease in HGF levels between diagnosis and first tumor evaluation during treatment (Figure S1D). We did not find any correlation between pMET expression and plasma HGF levels (data not shown). Next, we analyzed the association between plasma HGF levels and demographic data, including age, gender, PS, tumor stage, and number of metastatic sites (Table S2). Except for tumor histology type, there was no significant association between HGF levels and demographic or tumor aggressiveness factors including age, sex, smoking status, number of metastatic sites, and PD-L1 (Table S2). For patients treated with CT, there was no significant association between pretreatment plasma HGF levels and patient demographics.
Correlation with patient’s outcomes for ICIs therapy
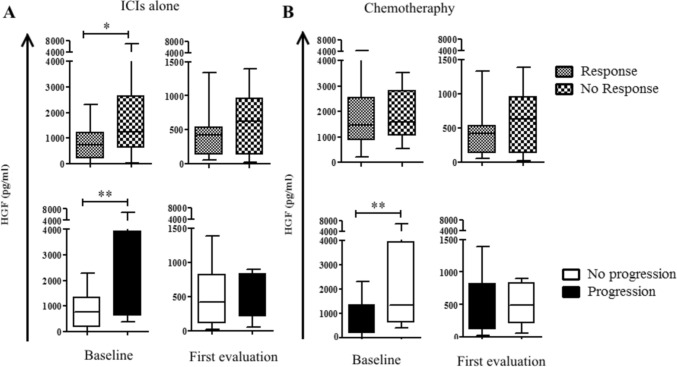
For patients treated with ICIs (alone or in combination with chemotherapy), the objective response rate at the first evaluation (ORR) was 57% (n = 28/49), and 16 out of 49 patients (33%) experienced tumor progression. Patients with stable disease and tumor progression were classified as non-responders. HGF levels at treatment initiation were higher in non-responders (1611 ± 432.7 pg/ml) and progressing patients (1888 ± 555.0 pg/ml) compared to responders (1128 ± 232.8 pg/ml) and non-progressing patients (1076 ± 202.6 pg/ml) (Table S3). These differences were more significant in patients treated with ICIs alone including non-responders vs responders (2061 vs 790.8; p = 0.0287) and progressing vs non-progressing patients (2408 ± 786.9 vs 811.1 ± 167.7; p = 0.0093) (Fig. 1A; Table S3).
Fig. 1.
Plasma HGF levels at baseline and first evaluation according to patient’s outcomes. Plasma samples were obtained from NSCLC patients at baseline and during the first evaluation. ELISA assays were utilized to quantify HGF levels. Subsequently, expression values were categorized based on drug response and disease progression, with A the group receiving ICIs alone and B the group receiving chemotherapy alone. Data are presented as mean ± SEM. *p < 0.05; **p < 0.01
Then, we analyzed the potential correlation between HGF levels and patients’ progression. In patients treated with ICIs (alone or in combination with chemotherapy), we observed that low HGF levels at baseline correlated with lower progression rate (Odds Ratio, OR = 0.14; 95% CI (0.02–1.23); p = 0.024) (Table 1A). In patients treated with ICIs alone, no progression was observed in patients displaying lower HGF levels (OR = 0.07 (0.003–1.32); p = 0.0092) (Table 1B). To ascertain if HGF levels in plasma samples were predictive of immunotherapy response, we also analyzed the correlation with tumor progression in patients treated with chemotherapy alone. In these patients, no association nor correlation between HGF rate and tumor progression were observed (Fig. 1B and Table 1C–D), confirming that HGF levels predict drug response only in patients treated with immunotherapy. Univariate analyses revealed that, in addition to HGF levels, the progression rate of patients treated with ICIs alone was also related to adenocarcinoma histology (OR = 0.10 (0.02–0.63); p = 0.014) (Table S4). Multivariate analysis based on logistic regression confirmed that adenocarcinoma histology (OR = 31.65 (1.05–949.11); p = 0.05); and low baseline HGF levels (OR = 1.17 (1.02–1.35); p = 0.03) were both predictors of low progression rate (Table 2).
Table 1.
Response rate and patients’ progression in univariate analysis
| Parameters | Response rate (%) | Odds ratio (OR); 95% CI; p value | Progression rate (%) | Odds ratio (OR); 95% CI; p value | |
|---|---|---|---|---|---|
| (A) Immunotherapy (ICIs alone and ICIs + chemotherapy) | |||||
| HGF Baseline (pg/mL) | Low (12) | 75 | 2.7 (0.62–11.58); p = 0.09 | 8 | 0.14 (0.02–1.23); p = 0.02 |
| High (36) | 53 | 39 | |||
| HGF First Evaluation (pg/mL) | Low (9) | 78 | 0.62 (0.18–2.16); p = 0.23 | 11 | 0.30 (0.03–2.78); p = 0.133 |
| High (27) | 56 | 30 | |||
| (B) ICIs alone | |||||
| HGF Baseline (pg/mL) | Low (6) | 85 | 9.00 (0.9–89.66); p = 0,02 | 0 | 0.07 (0.003–1.32); p = 0,0092 |
| High (21) | 33 | 50 | |||
| HGF First Evaluation (pg/mL) | Low (6) | 83 | 55.00 (2.83–1069); p < 0.001 | 0 | 0.5 (0.04–6.02); p = 0.29 |
| High (12) | 43 | 48 | |||
| (C) COMBO (ICIs + chemotherapy) | |||||
| HGF Baseline (pg/mL) | Low (5) | 60 | 2.55 (0.23–27.72); p = 0.22 | 20 | 0.75 (0.06–8.84); p = 0.41 |
| High (16) | 69 | 25 | |||
| HGF First Evaluation (pg/mL) | Low (5) | 80 | 0.23 (0.21–26.84); p = 0.23 | 20 | 0.75 (0.06–5.61); p = 0.41 |
| High (16) | 63 | 0 | |||
| (D) Chemotherapy (CT) | |||||
| HGF Baseline (pg/mL) | Low (10) | 40 | 1.17 (0.25–5.42); p = 0.42 | 30 | 1.46 (0.27–7.82); p = 0.33 |
| High (22) | 36 | 23 | |||
| HGF First Evaluation (pg/mL) | Low (7) | 43 | 0.84 (0.14–4.98); p = 0.43 | 14 | 0.16 (0.04–5.00); p = 0.26 |
| High (17) | 47 | 24 | |||
Table 2.
Logistic regression analysis of response and progression rate in the ICIs alone group
| Response | Progression | |
|---|---|---|
| Parameters | Odds ratio (OR); 95% CI; p value | |
| PD-L1 | 1.04 (0.97–1.10); p = 0.292 | 0.97 (0.89–1.05); p = 0.40 |
| HGF baseline (pg/ml) | 0.87 (0.77–0.99); p = 0.04 | 1.17 (1.02–1.35); p = 0.03 |
| HGF first evaluation | 0.80 (0.514–1.24); p = 0.312 | 0.97 (0.59–1.58); p = 0.90 |
| PS < 1 | 4.73 (0.144–155.47); p = 0.38 | 0.37 (0.002–64.12); p = 0.71 |
| Histology (adenocarcinoma, ADK) | 0.02 (0.001–0.62); p = 0.03 | 31.65 (1.05–949.11); p = 0.05 |
| Metastasis < 2 | 6.84 (0.27–170.93); p = 0.24 | 8.98 (0.17–473.93); p = 0.28 |
| Presence of a mutation | 0.02 (0.0003–1.769); p = 0.009 | 173.47 (0.19–161,045.95); p = 0.14 |
Correlation with patient’s survival
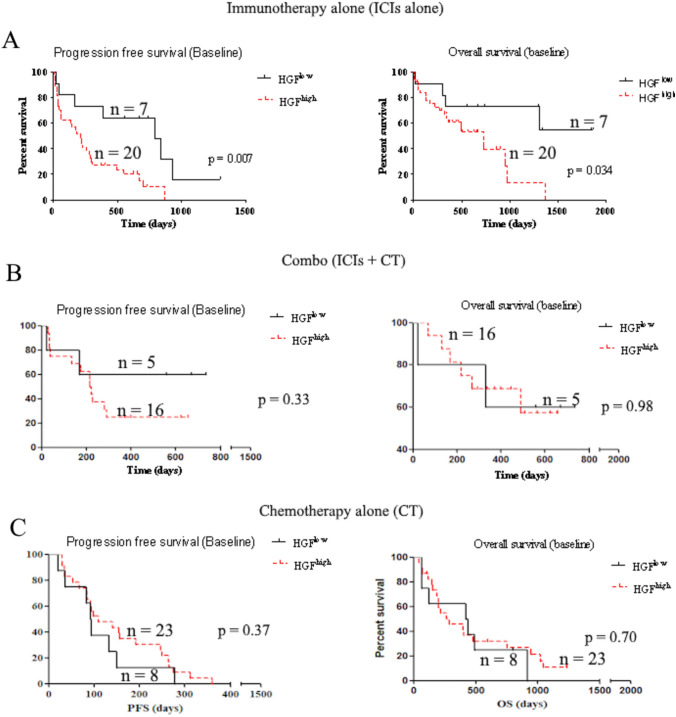
In patients treated with ICIs (alone and in combination with chemotherapy), very low HGF (below the first quartile) concentrations at baseline were associated with longer PFS (HR = 3.05; 95% CI (1.33–7.01); p = 0.01). In the group of patients treated with ICIs alone, lower levels of HGF correlated with prolonged survival (both PFS and OS) (Fig. 2A; Table S5). In this patients’ group, we found that performance status (PS) < 1 and adenocarcinoma histology correlated with longer PFS and OS (Table S5). The COX proportional hazard models revealed that only adenocarcinoma histology and low baseline HGF levels were predictors of longer PFS and OS (Table 3). There was no correlation between HGF levels and patient survival in either the chemotherapy group or the combination (COMBO) group, supporting that HGF levels were predictive of patient survival only in the ICIs alone group (Fig. 2B, C).
Fig. 2.
Patient’s survival according to HGF expression levels. Kaplan–Meier analyses of patient survival (PFS and OS) were conducted based on baseline HGF levels (low or high) within each treatment group, including immunotherapy alone (A), immunotherapy associated with chemotherapy (B), and chemotherapy alone (C). The log-rank test was utilized to compare survival within each subgroup
Table 3.
Cox proportional hazard analysis of patient’s survival
| Variable | PFS | OS | ||||
|---|---|---|---|---|---|---|
| Relative risk (RR) | 95% CI | p value | Relative risk (RR) | 95% CI | p value | |
| PD-L1 | 0.98 | 0.96–1.00 | 0.01 | 0.99 | 0.97–1.02 | 0.63 |
| HGF baseline (pg/ml) | 1.08 | 0.96–1.11 | < 0.001 | 1.08 | 1.03–1.13 | 0.002 |
| HGF first evaluation | 1.02 | 0.91–1.15 | 0.74 | 1.03 | 0.82–1.30) | 0.826 |
| PS < 1 | 1.18 | 0.34–4.03 | 0.80 | 0.293 | 0.04–2.23) | 0.236 |
| Histology (Adenocarcinoma, ADK) | 0.10 | 0.0001–0.37 | < 0.001 | 0.10 | 0.000–0.43 | 0.002 |
| Metastasis < 2 | 2.13 | 0.78–5.76 | 0.14 | 0.80 | 0.22–2.82 | 0.72 |
| Presence of a mutation | 0.88 | 0.29–2.7 | 0.81 | 1.69 | 0.28–10.25 | 0.57 |
HGF suppresses lymphocyte (LT) proliferation and interferes with immunotherapy.
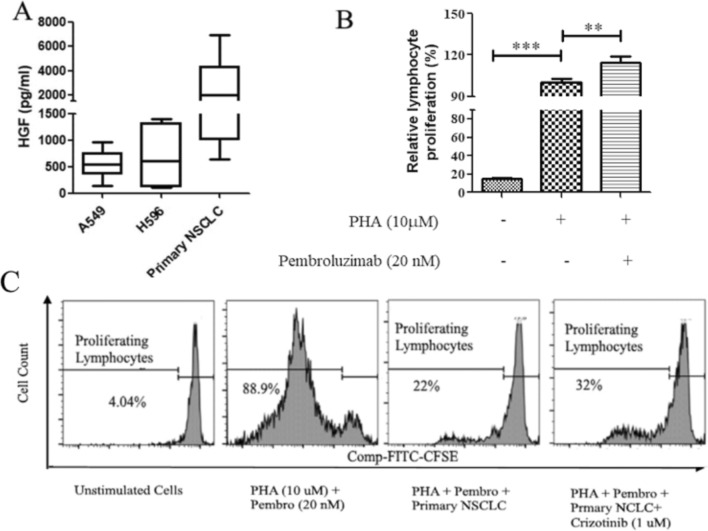
We conducted a series of in vitro and ex vivo experiments to investigate the impact of the HGF/MET pathway on the modulation of ICI-mediated lymphocyte (LT) activation. Specifically, we analyzed the influence of HGF on the immune properties of immune cells in the presence and absence of pembrolizumab. First, we investigated whether NSCLC cells were a source of HGF production. Therefore, we analyzed the supernatants from A549 and H596 cell line cultures, as well as primary NSCLC cultures. Our data confirmed the production of high levels of HGF by NSCLC cells (Fig. 3A). HGF production by primary samples was comparable to HGF levels measured in plasma samples from NSCLC patients (see Sect. 3.2), highlighting the relevance of our primary culture model in replicating HGF production in the tumor microenvironment. Then, we activated PBMC with PHA (10 µM) and cultured them on a monolayer of primary NSCLC cells. Our findings indicated that pembrolizumab (20 nM) enhanced lymphocyte activation mediated by PHA. When lymphocytes were cocultured with primary NSCLC cells, pembrolizumab-mediated lymphocyte activation was reduced. Furthermore, the addition of crizotinib (a MET inhibitor) into the culture medium partially restored T-cell proliferation in 3/5 of the coculture (Fig. 3B). These experiments suggest that HGF produced by primary NSCLC cells can hinder the ability of pembrolizumab to restore LT activation.
Fig. 3.
HGF production by NSCLC cells and inhibition of lymphocyte activation in the presence of pembrolizumab (“Pembro”) A expression levels of HGF in NSCLC cell cultures were assessed using an ELISA assay. B Peripheral blood mononuclear cells (PBMCs) were stained with CFSE and activated with phytohemagglutinin (PHA) alone or in combination with pembrolizumab. C PHA-activated cells were cultured either alone or on a primary NSCLC monolayer in the presence of pembrolizumab and/or crizotinib. After 96 h of incubation, CFSE-stained cells were collected and further stained with PE-Cy7-conjugated anti-CD3, BV421-conjugated anti-CD45, and APC-conjugated anti-CD8 antibodies. The cells were analyzed using flow cytometry (B). The presented data are representative of at least 6 patients
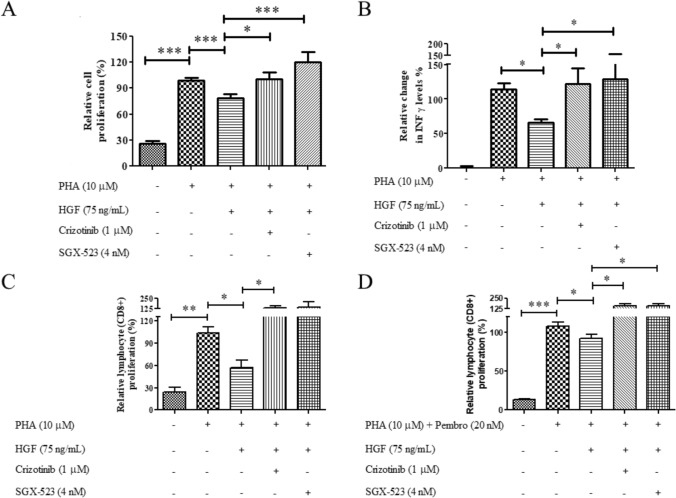
To validate the immunosuppressive properties of HGF, we treated stimulated PBMC with recombinant HGF. We compared the proliferation of LT cells in the presence of recombinant HGF and MET inhibitors (crizotinib, SGX-523). Following the administration of recombinant HGF, we observed a significant inhibition of PBMC proliferation and a decrease in IFN-γ production compared to the control group stimulated with PHA alone (Fig. 4A, B). These findings indicated that the presence of HGF suppressed PBMC proliferation and hampered the production of IFN-γ, which is associated with the anti-tumor lymphocyte response. To further investigate this hypothesis, we performed flow cytometric analysis of the activated population of PBMC. The results revealed that recombinant HGF suppresses CD8+ activation, even in the presence of pembrolizumab. The CD8+ LT activation was restored using MET inhibitors (Fig. 4C, D). These findings provide strong support for the notion that the release of HGF in the tumor microenvironment plays a significant role in immune evasion and contributes to resistance to immune checkpoint inhibitors. Consequently, the use of HGF/MET inhibitors has the potential to prevent immunosuppression and enhance the effectiveness of ICIs in the treatment of NSCLC.
Fig. 4.
MET inhibitors suppress the immunosuppressive capabilities of recombinant HGF. A, B Peripheral blood mononuclear cells (PBMCs) activated with phytohemagglutinin (PHA, 10 µg/ml) were cultured either alone (A) or in the presence of hepatocyte growth factor (HGF, 75 ng/mL) and/or MET inhibitors (crizotinib, SGX-523). The MTS proliferation assay was employed to quantify lymphocyte/PBMC proliferation at 72 h, while cell supernatants were collected at 24 h for interferon-gamma (IFNγ) quantification. C, D Activated PBMCs were stained with CFSE and treated for 96 h with HGF (75 ng/mL) alone or in combination with pembrolizumab (20 nm) and/or MET inhibitors (crizotinib, SGX-523). The stained cells were collected, probed with PE-Cy7-conjugated anti-CD3, BV421-conjugated anti-CD45, and APC-conjugated anti-CD8 antibodies, and analyzed using flow cytometry. Data are presented as mean ± SEM of at least four independent experiments. *p < 0.05 and ***p < 0.001
Discussion
Identification of predictive biomarkers of IO resistance remains a significant challenge in lung cancer. In this study, we investigated the role of the HGF/MET pathway in immunotherapy resistance among NSCLC patients undergoing first-line treatment with ICIs, either alone or in combination with chemotherapy. Our data support the association of higher levels of plasma HGF with poor ORR and reduced survival among patients treated with ICIs.
The MET activation pathway plays a pivotal role in supporting the growth, survival, and drug resistance of various cancer types. Consequently, the predictive potential of HGF/MET expression has been explored across multiple malignancies12. Within the context of lung cancer, elevated MET expression and/or increased circulating HGF levels have been identified as signatures for both NSCLC and SCLC samples13,14. MET overexpression is associated with the activation of its receptor, the phosphorylated form of MET (pMET). Consistent with this pattern, we observed robust and consistent expression of pMET across most patients in the analyzed cohorts, including those treated with ICIs and chemotherapy (CT). However, as discussed by Moosavi et al.15, the widespread and consistent expression of pMET observed across samples, while distinguishing tumor samples from normal tissues, cannot be used to stratify patients based on pMET levels, complicating the assessment of its influence on drug response and patient survival. Therefore, our investigation focused on plasma HGF levels, revealing that low levels of plasma HGF were associated with a favorable response to ICI treatment and a reduced progression rate. Similarly, patients with low HGF levels experienced significantly extended survival. These correlations were not observed in the CT group. Consequently, our results suggest that high baseline levels of HGF may not only serve as a prognostic factor, as previously suggested16,17, but also as a predictive factor for resistance to ICIs when used as first-line therapy in NSCLC.
HGF is primarily secreted by stromal cells, often through paracrine secretion, including mesenchymal cells. However, it can also be secreted directly by tumor cells, indicating autocrine secretion. This is highlighted by the presence of high levels of HGF in the supernatant of NSCLC primary cells and cell lines. The adverse prognostic significance of elevated serum HGF levels in cancer is substantiated by the fact that the activation of MET receptors located on cancer cells promotes tumor cell growth, survival, and metastatic spread16–18. In the context of the immune response and ICI therapy, the HGF/MET pathway has been reported as a mechanism of immune suppression. Through the modulation of dendritic cells, activation of regulatory T-cells, and the control of cytotoxic CD8 + T-cells19,20 MET activation in tumor cells promotes various tumor properties, including the EMT, which may consequently enhance the immunosuppressive characteristics of tumor cells21,22. Notably, MET activation induces an increase in PD-L1 expression23. Interestingly, PD-L1 expression is also elevated after HGF administration, and there is a correlation between high MET activation and high PD-L1 expression24. Therefore, inhibiting MET-induced signaling could potentially block the HGF-induced PD-L1 expression25, thus enhancing the efficacy of ICIs. Consistently, we present evidence in our work that the inhibition of MET activity using crizotinib interferes with NSCLC-mediating inhibition of CD8+ activation in the context of ICI inhibition, confirming the contribution of MET signaling in immune suppression as a mechanism of tumor-mediating resistance to ICIs. However, despite variations in HGF levels among patients, the levels of active MET in cancer cells did not differ significantly among patients, implying that MET activation in cancer cells may not be the main mechanism of immunosuppression mediated by HGF in NSCLC. In fact, it has been observed that CD8+ cytotoxic T lymphocytes (CTLs), when primed with specific antigens or exposed to the immune environment, express MET. Consequently, HGF could directly interfere with CTLs. We validated this hypothesis by demonstrating that recombinant HGF suppresses CTL activation and the production of IFN-γ, even in the presence of pembrolizumab.
Our study supports a scenario in which HGF/MET pathway could participate in a concerted immunosuppressive mechanism within the tumor microenvironment, by enhancing the immunosuppressive properties of the microenvironment components and interfering with CTL activation. We provided evidence that the use of two MET pathway inhibitors, crizotinib and SGX-523, interferes with HGF-mediated suppression of CTL activation. Thus, the use of MET inhibitors appears to be an interesting therapeutic approach to enhance the efficacy of ICIs in patients with advanced NSCLC. Currently, various HGF/MET inhibitors are available or under development in clinical trials, including monoclonal antibodies (e.g., rilotumumab), drug-conjugated antibodies (e.g., telisotuzumab vedotin), bi-specific antibodies (e.g., amivantamab)26, and MET-selective tyrosine kinase inhibitors (e.g., tivantinib capmatinib, tepotinib, or savolitinib).
Overall, despite the limitation in our study due to the insufficient number of available samples, notably for IHC, we managed to overcome this challenge by conducting a prospective collection of plasma samples throughout the disease follow-up. This approach allowed us to perform a comparative analysis and longitudinally monitor the patients. Concurrently, we implemented functional in vitro and ex vivo studies using a model of primary cultures derived from surgical explants, providing a valuable representation of the tumor microenvironment. The results of these efforts collectively support the proposition that the HGF/MET pathway may serve as a potential predictive factor for the response to ICIs in advanced NSCLC.
Conclusion
Our study's overall experiments emphasize the significant role of the HGF/MET pathway in inhibiting lymphocyte activation mediated by ICI in advanced NSCLC. In the ICI-treated patient group, we observed a correlation between high plasma HGF levels and criteria indicating tumor aggressiveness, a less favorable therapeutic response, and shorter survival. However, no such correlation was found in the group of patients treated with CT, suggesting a unique predictive role of this pathway in ICI resistance. As a result, assessing the expression of the HGF/MET pathway at the time of diagnosis, particularly through elevated plasma HGF levels, may serve as a predictive factor for a less favorable response to ICIs. Considering this, exploring the combination of HGF/MET inhibitors with ICIs in this context appears promising and warrants dedicated clinical trials for evaluation.
Supplementary Information
Below is the link to the electronic supplementary material.
Author contributions
AA and PTK built the methodology, collected the data, performed the experiments, performed the statistical analyses, and wrote the manuscript. CJ, CG, and EL interpreted the pathological results. CC performed the experiments. AC, CD, JD, VG, and MG collected the samples. JFE supervised the project. EGL built the methodology, supervised the project. All authors reviewed the manuscript.
Funding
This work was supported by Fondation du Souffle (2021).
Data availability
No datasets were generated or analyzed during the current study.
Declarations
Conflict of interest
Etienne Giroux Leprieur declares to have received honoraria/personal fees (advisory boards) from AstraZeneca, Bristol-Myers-Squibb MSD, and Roche. Other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Primary samples were collected at Ambroise Paré Hospital after obtaining signed informed consent (CPP IDF n°8) and stored within the Centre de Ressources Biologiques (CRB) of Ambroise Paré Hospital (ID CRB 2014-A00187-40).
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Assya Akli and Paul Takam Kamga have contributed equally to this work.
References
- 1.Pérez-Ruiz E, Melero I, Kopecka J, Sarmento-Ribeiro AB, García-Aranda M, De Las Rivas J (2020) Cancer immunotherapy resistance based on immune checkpoints inhibitors: targets, biomarkers, and remedies. Drug Resist. Update Rev. Comment. Antimicrob. Anticancer Chemother. 53:100718. 10.1016/j.drup.2020.100718 [DOI] [PubMed] [Google Scholar]
- 2.Gandhi L, Rodríguez-Abreu D, Gadgeel S, Esteban E, Felip E, De Angelis F, Domine M, Clingan P, Hochmair MJ, Powell SF et al (2018) Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 378(22):2078–2092. 10.1056/NEJMoa1801005 [DOI] [PubMed] [Google Scholar]
- 3.Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe S et al (2016) Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 375(19):1823–1833. 10.1056/NEJMoa1606774 [DOI] [PubMed] [Google Scholar]
- 4.Paz-Ares L, Luft A, Vicente D, Tafreshi A, Gümüş M, Mazières J, Hermes B, Çay Şenler F, Csőszi T, Fülöp A et al (2018) Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med 379(21):2040–2051. 10.1056/NEJMoa1810865 [DOI] [PubMed] [Google Scholar]
- 5.Darvin P, Toor SM, Sasidharan Nair V, Elkord E (2018) Immune checkpoint inhibitors: recent progress and potential biomarkers. Exp Mol Med 50(12):1–11. 10.1038/s12276-018-0191-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Trusolino L, Bertotti A, Comoglio PM (2010) MET signalling: principles and functions in development, organ regeneration and cancer. Nat Rev Mol Cell Biol 11(12):834–848. 10.1038/nrm3012 [DOI] [PubMed] [Google Scholar]
- 7.Mazzone M, Comoglio PM (2006) The Met pathway: master switch and drug target in cancer progression. FASEB 20(10):1611–1621. 10.1096/fj.06-5947rev [DOI] [PubMed] [Google Scholar]
- 8.Costantini A, Takam Kamga P, Julie C, Corjon A, Dumenil C, Dumoulin J, Ouaknine J, Giraud V, Chinet T, Rottman M et al (2020) Plasma biomarkers screening by multiplex ELISA assay in patients with advanced non-small cell lung cancer treated with immune checkpoint inhibitors. Cancers 13(1):97. 10.3390/cancers13010097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cheng Y, Wang C, Wang Y, Dai L (2022) Soluble PD-L1 as a predictive biomarker in lung cancer: a systematic review and meta-analysis. Future Oncol 18(2):261–273. 10.2217/fon-2021-0641 [DOI] [PubMed] [Google Scholar]
- 10.Takam Kamga P, Mayenga M, Sebane L, Costantini A, Julie C, Capron C, Parent F, Seferian A, Guettier C, Emile J-F et al (2024) Colony stimulating factor-1 (CSF-1) signalling is predictive of response to immune checkpoint inhibitors in advanced non-small cell lung cancer. Lung Cancer Amst Neth 188:107447. 10.1016/j.lungcan.2023.107447 [DOI] [PubMed] [Google Scholar]
- 11.Martkamchan S, Onlamoon N, Wang S, Pattanapanyasat K, Ammaranond P (2016) The Effects of Anti-CD3/CD28 Coated Beads and IL-2 on Expanded T Cell for Immunotherapy. Adv Clin Exp 25(5):821–828. 10.17219/acem/35771 [DOI] [PubMed] [Google Scholar]
- 12.Gherardi E, Birchmeier W, Birchmeier C, Vande WG (2012) Targeting MET in cancer: rationale and progress. Nat Rev Cancer 12(2):89–103. 10.1038/nrc3205 [DOI] [PubMed] [Google Scholar]
- 13.Tretiakova M, Salama AKS, Karrison T, Ferguson MK, Husain AN, Vokes EE, Salgia R (2011) MET and phosphorylated MET as potential biomarkers in lung cancer. J Environ Pathol Toxicol Oncol 30(4):341–354. 10.1615/jenvironpatholtoxicoloncol.v30.i4.70 [DOI] [PubMed] [Google Scholar]
- 14.Arriola E, Cañadas I, Arumí-Uría M, Dómine M, Lopez-Vilariño JA, Arpí O, Salido M, Menéndez S, Grande E, Hirsch FR et al (2011) MET phosphorylation predicts poor outcome in small cell lung carcinoma and its inhibition blocks HGF-induced effects in MET mutant cell lines. Br J Cancer 105(6):814–823. 10.1038/bjc.2011.298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moosavi F, Giovannetti E, Saso L, Firuzi O (2019) HGF/MET pathway aberrations as diagnostic, prognostic, and predictive biomarkers in human cancers. Crit Rev Clin Lab Sci 56(8):533–566. 10.1080/10408363.2019.1653821 [DOI] [PubMed] [Google Scholar]
- 16.Masuya D, Huang C, Liu D, Nakashima T, Kameyama K, Haba R, Ueno M, Yokomise H (2004) The tumour-stromal interaction between intratumoral c-Met and stromal hepatocyte growth factor associated with tumour growth and prognosis in non-small-cell lung cancer patients. Br J Cancer 90(8):1555–1562. 10.1038/sj.bjc.6601718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Navab R, Liu J, Seiden-Long I, Shih W, Li M, Bandarchi B, Chen Y, Lau D, Zu Y-F, Cescon D et al (2009) Co-overexpression of met and hepatocyte growth factor promotes systemic metastasis in NCI-H460 non-small cell lung carcinoma cells. Neoplasia N Y N 11(12):1292–1300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tsuta K, Kozu Y, Mimae T, Yoshida A, Kohno T, Sekine I, Tamura T, Asamura H, Furuta K, Tsuda H (2012) c-MET/phospho-MET protein expression and MET gene copy number in non-small cell lung carcinomas. J Thorac Oncol 7(2):331–339. 10.1097/JTO.0b013e318241655f [DOI] [PubMed] [Google Scholar]
- 19.Benkhoucha M, Santiago-Raber M-L, Schneiter G, Chofflon M, Funakoshi H, Nakamura T, Lalive PH (2010) Hepatocyte growth factor inhibits CNS autoimmunity by inducing tolerogenic dendritic cells and CD25+Foxp3+ regulatory T cells. Proc Natl Acad Sci USA 107(14):6424–6429. 10.1073/pnas.0912437107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Okunishi K, Dohi M, Nakagome K, Tanaka R, Mizuno S, Matsumoto K, Miyazaki J-I, Nakamura T, Yamamoto K (2005) A novel role of hepatocyte growth factor as an immune regulator through suppressing dendritic cell function. J Immunol Baltim Md 1950 175(7):4745–4753. 10.4049/jimmunol.175.7.4745 [DOI] [PubMed] [Google Scholar]
- 21.Rudisch A, Dewhurst MR, Horga LG, Kramer N, Harrer N, Dong M, van der Kuip H, Wernitznig A, Bernthaler A, Dolznig H et al (2015) High EMT signature score of invasive non-small cell lung cancer (NSCLC) cells correlates with NFκB driven colony-stimulating factor 2 (CSF2/GM-CSF) secretion by neighboring stromal fibroblasts. PLoS ONE 10(4):e0124283. 10.1371/journal.pone.0124283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Taki M, Abiko K, Ukita M, Murakami R, Yamanoi K, Yamaguchi K, Hamanishi J, Baba T, Matsumura N, Mandai M (2021) Tumor immune microenvironment during epithelial-mesenchymal transition. Clin Cancer Res 27(17):4669–4679. 10.1158/1078-0432.CCR-20-4459 [DOI] [PubMed] [Google Scholar]
- 23.Garcia-Diaz A, Shin DS, Moreno BH, Saco J, Escuin-Ordinas H, Rodriguez GA, Zaretsky JM, Sun L, Hugo W, Wang X et al (2017) Interferon receptor signaling pathways regulating PD-L1 and PD-L2 expression. Cell Rep 19(6):1189–1201. 10.1016/j.celrep.2017.04.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Saigi M, Alburquerque-Bejar JJ, Mc Leer-Florin A, Pereira C, Pros E, Romero OA, Baixeras N, Esteve-Codina A, Nadal E, Brambilla E et al (2018) MET-oncogenic and JAK2-inactivating alterations are independent factors that affect regulation of PD-L1 expression in lung cancer. Clin Cancer Res 24(18):4579–4587. 10.1158/1078-0432.CCR-18-0267 [DOI] [PubMed] [Google Scholar]
- 25.Benkhoucha M, Tran NL, Breville G, Senoner I, Jandus C, Lalive P (2021) c-Met enforces proinflammatory and migratory features of human activated CD4+ T cells. Cell Mol Immunol 18(8):2051–2053. 10.1038/s41423-021-00721-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Glisson B, Besse B, Dols MC, Dubey S, Schupp M, Jain R, Jiang Y, Menon H, Nackaerts K, Orlov S et al (2017) A randomized, placebo-controlled, phase 1b/2 study of Rilotumumab or Ganitumab in combination with platinum-based chemotherapy as first-line treatment for extensive-stage small-cell lung cancer. Clin Lung Cancer 18(6):615-625.e8. 10.1016/j.cllc.2017.05.007 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analyzed during the current study.