Abstract
Background:
Over the last decade, the anticancer effects of Rhus verniciflua Stokes (RVS) have been reported in various preclinical or clinical studies. However, the effects of RVS on immuno-oncology, especially on the functional properties of T cells and their phenotypes, remain unclear. Here, we planned to investigate the impact of RVS on immuno-oncology, specifically focusing on its effects on T cells.
Methods
Peripheral blood mononuclear cells (PBMCs) from breast cancer patients were isolated to obtain cytokine-induced killer cell populations with >85% CD3+ T cells. The anticancer activity of these T cells was evaluated by introducing red fluorescent protein (RFP) into HLA-A02:01 type-matched breast cancer cell lines (MCF7 and MDA-MB-231) and analyzing the results using flow cytometry. The effect of RVS extracts on T cell phenotype was assessed using markers such as CTLA-4 and PD-1, as well as mRNA expression levels of key genes (IFN-γ, TNF-α, and IL-2).
Results
RVS treatment significantly enhanced the anticancer activity of T cells against breast cancer cells. Specifically, T cells treated with 100 µg/mL of RVS showed a 20.6% increase in cytotoxicity against MCF-7 cells and a 36.2% increase against MDA-MB231 cells compared to the control. Additionally, RVS treatment led to a significant reduction in PD-1 expression on T cells.
Conclusion:
Our findings demonstrate that RVS treatment enhances T cell function against breast cancer cells by reducing PD-1 expression. These results suggest that components of RVS may serve as potential candidates for restoring exhausted T cells in cancer therapy.
Keywords: Rhus verniciflua Stokes, T cell co-culture, PD-1, breast cancer, anticancer, immuno-oncology
Introduction
Rhus verniciflua Stokes (RVS), commonly known as the lacquer tree, has therapeutic properties used in Traditional East Asian medicine for centuries. 1 It has been used to treat various conditions, including abdominal tumors. In recent years, RVS has gained significant attention for its anticancer effects in preclinical and clinical studies. Previous clinical studies have suggested that RVS extract administration led to favorable outcomes and prolonged overall survival in patients with solid cancers, including non-small cell lung cancer 2 and colorectal cancer. 3 Moreover, RVS has shown complete response in treating patients with advanced renal cell carcinoma with multiple metastases. 4 These findings highlight the potential of RVS as an effective therapeutic agent against various cancers.4 -8 Research into the flavonoid-rich compounds found in RVS—specifically fustin, fisetin, sulfuretin, and butein—demonstrates their potential in inhibit cancer cell proliferation and induce programmed cell death mediated via Nuclear Factor Kappa-light-chain-enhancer of activated B cells (NF-κB) and mitogen-activated protein kinase (MAPK).9 -14 These compounds target pathways associated with cell survival, proliferation, and immune responses, making them particularly effective in preclinical studies focused on breast, prostate, lung, and liver cancers.9 -14 However, the mechanism of action in RVS primarily was focused on direct toxic effects on cancer cells without considering the tumor microenvironment which did not provide comprehensive results regarding the actual clinical response.
In recent years, programmed cell death protein 1 (PD-1), programmed cell death ligand 1 (PD-L1), and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) have emerged as a key checkpoint blockade in T cell function and has become a major target for various anticancer treatments. 15 Nowadays, immune checkpoint inhibitors such as PD-1 inhibitors, PD-L1 inhibitors, and CTLA-4 inhibitor were approved and the indications has been expanded to earlier stages across various cancer types. 16 Interestingly, RVS was reported as an immuno-oncological therapeutics by blocking the PD-1/PD-L1 and CTLA-4/CD80 interactions. 17 Also, clinical response of RVS administration in practice suggested a possibility for enhancing immune responses against cancer due to little cytotoxicity of RVS. 5 However, the action mechanism underlying its anticancer effects by immune cells remains unclear.
Here, we aimed to demonstrate the PD-1 regulatory capacity of RVS using human peripheral blood mononuclear cells (PBMCs) with breast cancer cells. Specifically, we aimed to elucidate the role of CD3+ immune cells, particularly PD-1+ cells, in mediating immune responses to RVS with 2 distinct cell models such as MCF7 and MDA-MB-231: MCF7 represents an estrogen receptor (ER)-positive subtype of breast cancer cells, whereas MDA-MB-231 cells belong to the triple-negative breast cancer (TNBC).
Materials and Methods
Cell Lines
The breast cancer cell lines MCF7 (RRID:CVCL_0031; luminal A) and MDA-MB-231(RRID:CVCL_0062; TNBC) were obtained from the Korean Cell Line Bank. The cells were cultured in RPMI-1640 (Gibco, #11875-119) medium supplemented with 10% fetal bovine serum (FBS; Gibco, #16000044) and 1% penicillin/streptomycin (Gibco, #15140122) in a humidified atmosphere with 5% CO2 at 37°C. The cells were subcultured every 2 to 3 days when they reached 80% confluence, and spent medium was replaced with fresh one. Prior to the experiments, all cells were washed twice with phosphate-buffered saline (PBS; WELGENE, #LB 001-02) to remove any residual serum or antibiotics.
Preparation of RVS Solution
RVS stalks, which included bark and were 10 years old, were obtained from Wonju, South Korea. The stalks were processed into sawdust, dried, and then extracted with 10 volumes of water at 90°C to 95°C for 6 hours. After extraction, the resulting solution was filtered and concentrated to yield a dark brown powder. Before the cell treatments, the obtained powder was dissolved in Dimethyl sulfoxide (DMSO; CryoStor, #210102) and diluted with PBS to a final concentration of 10%. This solution was then added to the cells at the appropriate concentrations.
LC/MS (Liquid Chromatography-Mass Spectrometry) Analysis to Determine the Components of RVS
LC was performed using a Nexera x2 instrument and MS was performed using an LCMS-8050 instrument. The column used was an ACQUITY BEH C18 (1.7 µm, 100 mm × 2.1 mm). The solvents used were DW (0.1% formic acid) and ACN (0.1% formic acid), with a flow rate of 0.25 mL/min and an injection volume of 1 µL. For MS, ionization was performed using electrospray ionization in negative mode, and the interface voltage was set to 4.0 kV. The interface temperature and DL (drying gas and nebulizing gas) temperature were set to 300°C and 250°C, respectively. Data were analyzed using LabSolution software (Shimadzu’s). Standard solutions were prepared by dissolving the standard substance in methanol to make a 1 mg/mL stock solution, which was then diluted to make the standard solution. Sample analysis was performed by dissolving the RVS powder in water to make a 3 to 4 mg/mL stock solutions, and then calculating the amount of the corresponding analyte.
Cell Viability Assay
Cell viability was determined using a 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium inner salt (MTS) assay (Promega, #G3582). 18 Briefly, confluent cells were harvested and plated in 96-well plates at a density of 5 × 103 cells/well. After overnight culture, the cells were treated with the RVS extract and incubated for 48 hours. The RVS extracts were prepared by diluting RVS powder in DMSO to a concentration of 10 mg/mL, followed by further dilution in PBS to obtain the final concentration. Complete cell culture medium was then added to the wells, and the cells were incubated for 48 hours. Cell viability was determined using the MTS assay (Promega, Madison, WI, USA) according to the manufacturer’s protocol, as previously described. The absorbance of each well was then measured at 490 nm.
Generation of RFP-Expressing Cell Lines
To generate cells that express red fluorescent protein (RFP), 293FT (RRID:CVCL_6911) cells were seeded at a density of 4 × 106 cells/10 mL in 100 mm culture dishes 1 day before transfection. 19 On the day of transfection, pALD-RFP, pALD-gag-pol, pALD-REV, and pALD-VSVg plasmids were combined with serum-free Dulbecco’s Modified Eagle Medium (DMEM) in a tube. Complexes formed by incubating the PEIpro reagent with DNA for 15 minutes were used to transfect 293FT cells. The virus was harvested from the medium at 72 hours in post-transfection and subsequently used to transduce MCF-7 and MDA-MB-231 cells in the presence of Polybrene (Sigma-Aldrich, St. Louis). Following transduction, the cells were cultured in RPMI-1640 medium (Hyclone, Logan) supplemented with 10% FBS, puromycin (Thermofisher, Waltham), and penicillin-streptomycin (Gibco, New York) to select stable transductants.
Cancer Cell Co-Culture with T Cells
MCF7 and MDA-MB-231 cells were suspended in AIM-V T cell media (ThermoFisher, Waltham) and seeded into 96-well plates (flat-bottom plates) with 5 × 103 cells/well after incubating the cells at 37ºC with 5% CO2 for at least 4 hours, immune cells were added to the wells at E:T ratios of 1:1 and 5:1, with a density of 1.5 × 105 cells/mL in AIM-V T cell media (Gibco, #0870112DK). The plate was then incubated at 37ºC with 5% CO2 for 48 hours. We confirmed the cells expressing RFP using flow cytometry at a wavelength of 570 nm, with RFP expression levels of 96.1% in MCF-7 cells and 96.5% in MDA-MB-231 cells. RFP antibodies were used to target live cells. The relative killing (%) was calculated as 100 × (remaining target cell [% of co-culture/target only]). 20 Data are presented as the mean ± standard deviation of triplicate samples from at least 3 independent experiments.
Cell Staining With Trypan Blue
After co-culturing with T cells in poly L-lysin coated 96 well plate the cells were washed with PBS. The remaining viable cells were then permeabilized with 70% ethanol (prepared in PBS) for 20 minutes at room temperature, followed by 2 additional PBS washes. 21 The cells were stained with 0.5% Trypan blue (Gibci, #15250061) diluted in PBS for 60 minutes at room temperature. After staining, the cells were washed twice with PBS to remove any remaining Trypan blue residue and to avoid artifact signals. Images were acquired using an eVOS microscope (Thermo Fisher Scientific, Massachusetts, USA).
Immunophenotyping
Immunophenotyping of immune cells was performed by staining various surface marker combinations using the following fluorescence-conjugated antibodies: CD3 (BioLegend, #300318), CD4 (BioLegend, #63-0047-42), CD8 (BioLegend, #62-0088-42), CD56 (BioLegend, #12-0567-42), PD-1 (BioLegend, #25-2799-42), and CTLA-4 (BioLegend, #17-1529-42). All antibodies except for CD3 were purchased from eBioscience, Inc. (San Diego, CA, USA). The cells were washed twice with flow cytometry staining buffer, followed by the addition of antibodies. After incubation in the dark at 4°C for 20 minutes, the cells were washed and analyzed. Flow cytometry gating strategies for various lymphocytes were first based on low forward scatter and low side scatter gating. The T cell markers CD3, CD4, and CD8, as well as the NK cell marker CD56, were used to identify the major lymphocyte subsets. Flow cytometry experiments were performed using BD FACSLyric™ analyzer (Becton Dickinson) and the resulting data were analyzed using FlowJo software (FlowJo, LLC).
PBMC Isolation and T Cell Expansion
Blood samples were obtained from a 62-year-old female diagnosed with breast cancer. At the time of blood collection, the patient had stage IIIA luminal A A-type breast cancer. This study was approved by the Institutional Review Board of the Myoungji Hospital (IRB Nos. 2018-08-030 and 2021-12-028). Whole blood was collected into a BD Vacutainer® CPT™ Mononuclear Cell Preparation Tube (BD, #362753) containing sodium heparin. The tube was gently mixed by inverting it 8 to 10 times, and then centrifuged at 1500 rpm for 20 minutes at room temperature. After centrifugation, the liquid in the tube was separated into 3 layers, and the PBMC layer was carefully collected using a sterile transfer pipette, taking care not to disturb the underlying red blood cell layer. The collected PBMC layer was transferred to a new sterile tube, washed with 10 mL of PBS via centrifugation at 300 rpm for 10 minutes at room temperature, and resuspended in Alys media containing 5% autoplasma. For T cell expansion, on day 0, PBMCs (5 × 106) were seeded into a 25T flask coated with OKT-3 (CD3 antibody; Takara bio, #T210) and cultured in Alys media at 37°C and 5% CO2 in a humidified cell culture incubator for 24 hours. On day 1, fresh Alys media containing 200 IU/mL rhIL-2 (was added to the flasks. Thereafter, the medium was refreshed every 3 days to maintain a cell density below 1 × 106 cells/mL until the end of cell expansion on day 14.
Human Leukocyte Antigen (HLA) Testing
For HLA-A typing of the recipients, samples of peripheral blood were analyzed at the Catholic Hematopoietic Stem Cell Bank. The sequencing-based typing (SBT) method, as previously described 22 was utilized. DNA was extracted from whole blood collected in ethylenediaminetetraacetic acid (EDTA)-coated blood containers. HLA Class I and Class II gene classification were performed using the AlleleSEQR kit (Abbott, Chicago, IL, USA), following the manufacturer’s instructions. Sequencing was conducted for exons 2, 3, and 4 of HLA-A using the ABI 3130XL genetic analyzer (Applied Biosystems, Waltham, MA, USA) with POP 6 polymer. HLA types were analyzed using SBTengine (Genome Diagnostics B.V., Utrecht, Netherlands), and subsequently reanalyzed using Assign SBT v3.5 (Conexio Genomics, Applecross, Australia).
Enzyme-Linked Immunosorbent Assay (ELISA)
Both CIK (cytokine-induced killer) cells and RVS-treated CIK cells were cultured in AlyS505N-O medium (Cell Science & Technology Institute, Tokyo, Japan) supplemented with 3% CTS Immune Cell SR (Gibco) and 200 IU IL-2. The cells were seeded in a 96-well plate and treated with RVS at a concentration of 10 μg/mL. After RVS treatment, the cells were incubated at 37°C in a humidified 5% CO2 incubator for 48 hours. ELISA plates were coated with capture antibodies specific for Interferon-γ (IFN-γ; R&D, #DY285B) and Tumor Necrosis Factor-α(TNF-α; R&D, #DY210). Samples of the T cell supernatants were harvested, appropriately diluted, and standard curves were generated using known concentrations of recombinant IFN-γ and TNF-α. The amount of IFN-γ- and TNF-α-specific antibodies was measured. After incubation, an H2O2 mixed substrate solution (3,3′,5,5′-tetramethylbenzidine) was added to induce color development, and the reaction was stopped with 1N sulfuric acid (H2SO4). The absorbance of each well was then measured at 490 nm using a microplate reader. The concentrations of secreted IFN-γ and TNF-α in the T cell supernatant samples were analyzed by comparing their absorbance values to the standard curve.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) Analysis
The total RNA from cells was extracted using the AccuPrep Universal RNA Extraction kit (Bioneer, Daejeon, Korea), and cDNA was synthesized using the PrimeScript RT Master mix. Subsequently, quantitative real-time PCR was performed using TB Green and the QuantStudio 3 system (Thermo Fisher Scientific, Massachusetts, USA). The primers were obtained from Bioneer (Accutarget qPCR Screening kit, Bioneer, Daejeon, Korea), specifically from the T Cell Anergy and Immune Tolerance, T Helper Cell Differentiation, and T-Cell and B Cell Activation kits.
Statistical Analysis
Statistical analysis of the data was performed using Prism 5.01 software (GraphPad Software, USA). All experiments were repeated at least thrice, and comparable results were obtained each time. Values are expressed as the mean ± standard deviation (SD). Statistical significance (P-value < .05 for all analyses) was assessed via one-way ANOVA followed by the Dunnett comparison test for multiple comparisons or two-tailed Student’s t-test for single comparisons.
Results
Quantification and Comparison of Major Marker Chemicals in RVS Extracts
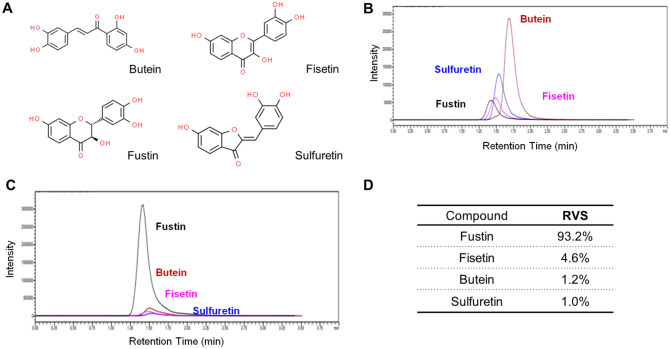
The quantification and comparison of the major marker compounds in RVS extracts were conducted to better understand the chemical profile and ensure consistency in its composition. The chromatograms and chemical structures of the marker compounds were investigated to understand the composition and structure of the RVS extract (Figure 1A). Peak areas of the marker compounds were identified at a concentration of 1 ppm using LC chromatograms (Figure 1B). The concentrations and compositions of these compounds in the RVS extract samples were then determined (Figure 1C). Fustin was identified as the predominant component, followed by fisetin, butein, and sulfuretin, in descending order of abundance (Figure 1D).
Figure 1.
Quantitative analysis of marker compounds in RVS extract. The chemical structure of four reference marker compounds such as butein, fisetin, fustin, and sulfuretin in the extract of RVS (A). The Chromatogram of the four reference marker compounds at 1 ppm concentration (B). The Chromatograms (C) and composition pie chart (D) comparing the peak areas of butein, fisetin, fustin, and sulfuretin in RVS extract.
Evaluation of RVS Concentration in Cancer Cells
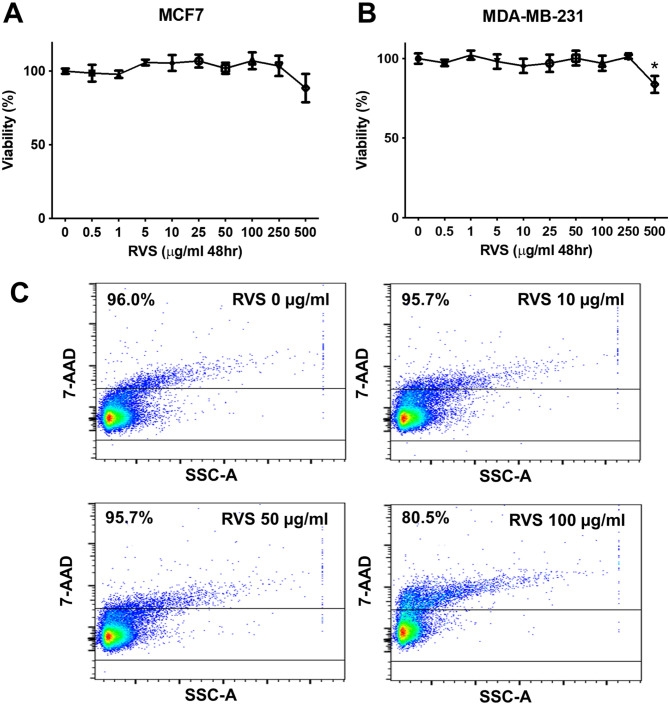
The toxicity assessment at various concentrations of RVS was performed to determine safe and effective dosing for subsequent experiments. At a concentration of 500 µg/mL RVS, the observed toxicity was 11.48% in MCF7 cells and 16.26% in MDA-MB-231 cells. However, no significant toxicity was observed at concentrations below 250 µg/mL in both cell lines (Figure 2A and B). While slight toxicity was observed at 100 µg/mL, no specific toxicity to T-cells was detected at 10 and 50 µg/mL (Figure 2C). Therefore, we used RVS concentrations below 100 µg/mL in our co-culture experiments to avoid any potential toxicity to target T-cells. This approach allowed us to accurately assess the effects of RVS treatment on T-cells and establish appropriate experimental conditions.
Figure 2.
The viability of breast cancer cells treated with RVS extract. MCF-7 and MDA-MB-231 cells were treated with varying concentrations of RVS (0.5, 1, 5, 10, 25, 50, 100, 250, and 500 µg/mL) for 48 hours ((A and B), respectively). The cells were then subjected to the MTT assay to determine cell viability. Data are presented as the mean ± standard deviation of 3 independent experiments performed in triplicate. Immune cell toxicity analysis of RVS treatment using 7AAD staining and flow cytometry (C). Representative flow cytometry plots of 7AAD staining for untreated and RVS-treated immune cells. Statistical significance is indicated as follows: P < .05*.
Immunomodulatory Effects of RVS on MCF7 and MDA-MB-231 Cells
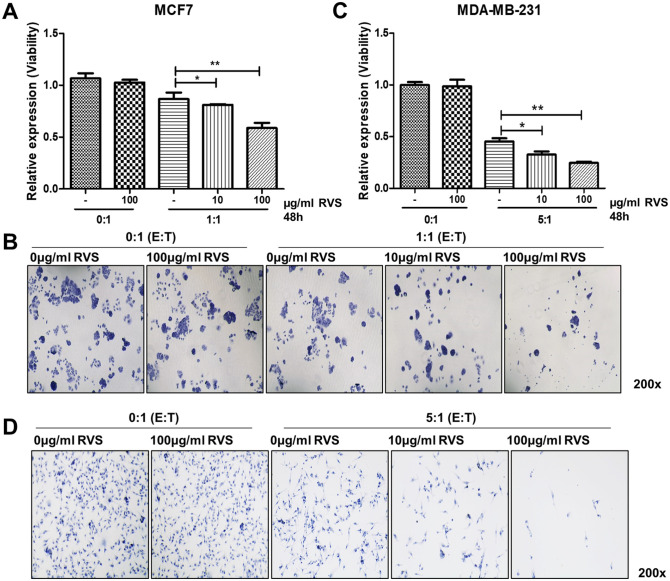
To investigate the immunomodulatory effects of RVS, we conducted an RFP killing assay and Trypan blue staining. Immune cells and cancer cells were co-cultured at effector cell: target cell (E:T) ratios of 1:1 and 5:1, treated with RVS, and incubated for 48 hours. The results showed that RVS increased the cancer cell-killing effect in MCF7 cells by 6.4% and 20.6% at concentrations of 10 and 100 µg/mL, respectively, compared to immune cells alone at an E:T ratio of 1:1 (Figure 3A). In MDA-MB-231 cells, RVS increased the cell-killing effect by 8.2% and 22% at 10 and 100 µg/mL, respectively, at an E:T ratio of 1:1, and by 23.9% and 36.2%, at the same concentrations at an E:T ratio of 5:1 (Figure 3C). However, at the 5:1 E:T ratio, immune cells completely eliminated the MCF7 cells during co-culture, making it impossible to evaluate immune cell-mediated cytotoxicity in this group. The results from the RFP killing assay were further validated through Trypan blue staining, which showed similar cell confluencies (Figure 3B and D).
Figure 3.
Effect of RVS treatment on cancer cell killing by immune cells in co-culture at different E:T ratios. RFP killing assay was performed by co-culturing immune cells with breast cancer cells and treating them with RVS at 10 and 100 µg/mL for 48 hours, followed by RFP measurement using flow cytometry in MCF-7 (A) and MDA-MB-231 (C). The experiment was repeated 3 times and presented as mean ± standard deviation. Statistical significance was determined by t-test. Trypan blue staining was performed to observe the cell morphology under co-culture conditions with Effector:Target (E:T) ratios of 1:1 and 1:5 in MCF7 (B) and MDA-MB-231 (D). Magnification was 200x and statistical significance is indicated as follows: P < .05* and P < .01**.
RVS Treatment Induces Downregulation of PD-1 Expression in Immune Cells
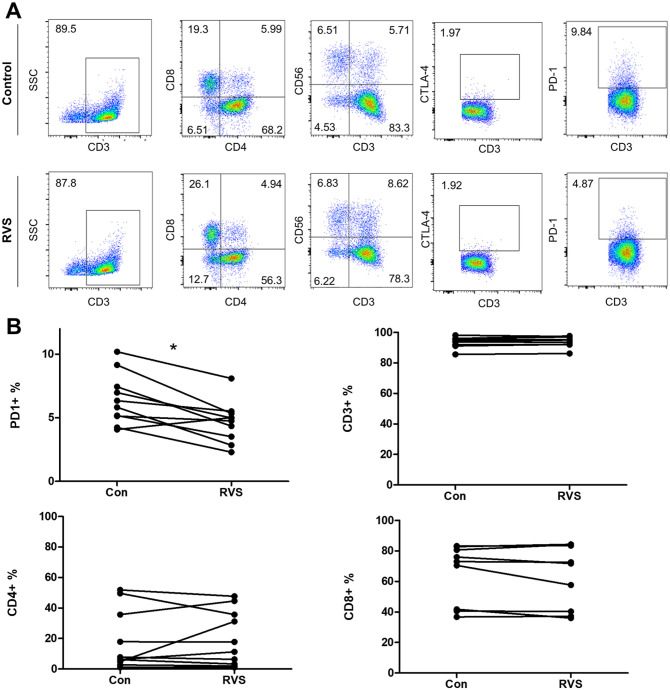
To explore the potential immunomodulatory effects of RVS, we assessed whether RVS treatment could alter immune cell phenotypes and alleviate immune exhaustion. Specifically, the focus was on identifying changes in key markers related to immune activation and exhaustion, such as PD-1 and CTLA-4, which are crucial in regulating immune responses. To examine the phenotypic changes in immune cells in response to RVS treatment, we treated the cells with 10 µg/mL of RVS for 48 hours and then stained them with various phenotype markers (CD3, CD4, CD8, CD56, PD-1, and CTLA-4). Despite variations in immune cell phenotypes among different patients, we observed a significant decrease in PD-1 expression, from 9.84% to 4.87%, which was more pronounced than changes in the other markers (Figure 4A). Additionally, CD3+CD56+ (NKT-like) cells slightly increased. This decrease in PD-1 expression was consistently observed across most samples, in contrast to the expression of another exhaustion marker, CTLA-4. These findings suggest that RVS effectively inhibits the PD-1 pathway (Figure 4B).
Figure 4.
Phenotypic analysis of immune cells following RVS treatment. Flow cytometry analysis (lower) and table (upper) showing the phenotype of immune cells following marker staining (A). Immune cells from 7 cases were treated with RVS at 10 µg/mL for 48 hours, and the phenotype was compared using marker staining (B). Data represents mean ± standard deviation of n = 3 independent experiments, analyzed using t-test. Statistical significance is indicated as follows: P < .05 *.
RVS Treatment Modulates T Cell-Related RNA Expression
To explore the impact of RVS treatment on the RNA expression levels of genes associated with immune cell function, qPCR analysis of 288 genes was performed. A total of 75 genes were found to be significantly upregulated or downregulated after RVS treatment (Supplemental Figure 1A and B). Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis revealed that these genes were involved in several biological processes, including inflammatory cytokine regulation, Th17 cell differentiation, Th1 and Th2 cell differentiation, and T cell receptor signaling pathways (Supplemental Figure 1C). Notably, we observed an increase in the RNA level of forkhead box P3 (FOXP3; fold change = 4.0 ± 1.62), a gene involved in 3 of the identified pathways, upon treatment with RVS. We also found that the expression of IFN-γ was significantly increased (fold change = 4.43 ± 0.74), while the RNA level of PD-1 was decreased (fold change = 0.24) compared to that in the control cells (Supplemental Figure 1D). These findings suggest that RVS treatment enhances T cell function by modulating the expression of PD-1 and T cell receptor signaling.
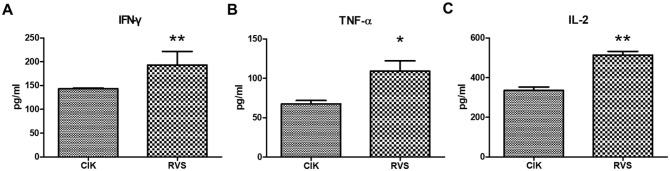
RVS Increases the Secretion of IFN-γ, TNF-α, and IL-2 in CIK Cells
We measured the secretion of major cytokines in T cell activation and the immune response, such as IFN-γ and TNF-α, to observe the activation of RVS-treated CIK cells. The results revealed increased secretions of IFN-γ, TNF-α, and IL-2 in RVS-treated CIK cells compared to untreated cells (Figure 5). Specifically, the levels of IFN-γ and TNF-α were elevated in the RVS-treated group, indicating the stimulatory effect of RVS on the production of these pro-inflammatory cytokines in CIK cells. These findings demonstrated the immunomodulatory effects of RVS on CIK cells.
Figure 5.
Increased Secretion of IFN-γ and TNF-α in RVS Treated CIK Cells. IFN-γ and TNF-α detected by ELISA assay. The serum cytokine concentrations of (A) IFN-γ, (B) TNF-α and (C) IL-2 were detected by ELISA assay. The levels of IFN-γ and TNF-α were significantly increased in the RVS treated group. Statistical significance is indicated as follows: P < .05* and P < .01**.
Discussion
In this study, we established a co-culture model of cancer and immune cells mimicking the tumor microenvironment, where RVS extracts showed the immunomodulatory effect for T cells against breast cancer cells. To evaluate the cancer cell-killing effect of T cells, it is essential to determine whether cancer cells express matching types of HLA (Human leukocyte Antigen). Only after the HLA type of cancer cells aligns with the specificity of the T cell receptor, T cells could exhibit a killing effect by recognizing and attacking cancer cells through specific antigen peptides, such as Human Epidermal Growth Factor Receptor 2 (HER2), Wilms’ tumor gene (WT1), and survivin. 23 Therefore, in our study, we selected HLA-A type 02:01 from human patient-derived PBMCs and conducted research using cells with the same HLA type, such as MCF-7 and MDA-MB-231 cells. In both cells, the significant difference of RVS was not observed even though the anti-cancer effect of RVS was slightly higher in MDA-MB-231 cells.
RVS was previously reported to have anticancer effects on breast cancers.24 -27 Specifically, butein, a component of RVS, has been shown to inhibit the growth of breast cancer cells when co-cultured with fibroblasts. 11 Moreover, RVS extracts induced apoptosis in MCF-7 cells by modulating the Adenosine Monophosphate-Activated Protein Kinase pathway (AMPK) pathway 24 and led to the inhibition of cell viability via G1 cell cycle arrest. 27 Here, we demonstrated a decrease in PD-1 expression in T cells and an increase in the cancer cell-killing effect following the specific increase in the secretion of cytokines such as IFN-γ and TNF-α, which suggests another role of RVS for restoring T cells exhausted against breast cancer. By directly assessing PD-1 expression in T cells, we uncovered a pivotal mechanism underlying RVS-mediated immune regulation. Therefore, RVS could be used as a supplement to conventional cancer treatment, particularly for augmenting T cell function within the tumor microenvironment. Delving deeper into the specific signaling pathways implicated could facilitate the development of immunotherapeutic strategies employing RVS.
In general, herb extracts exhibit characteristics such as multi-component, multi-target, and multi-pathway effects. 28 RVS also contains a variety of active substances including fustin, fisetin, butein, and sulfuretin, which exerts diverse activities, including anti-inflammatory, antibacterial, antiviral, antioxidant, anti-tumor, and immune system-modulating properties in various pharmacological studies. 1 Therefore, it is needed to investigate the mechanisms of RVS from the perspective of multiple components and targets against cancer. In addition, a combination strategy involving RVS and existing immunotherapies or conventional cancer treatments should be extensively studied for enhancing the overall efficacy of cancer treatment. 17 This exploration could potentially lead to improved cancer cell-killing effects compared to RVS alone and contribute to advancing breast cancer immunotherapy strategies.
Our investigations have several limitations. First, we used the crude extract of RVS, where the effect of RVS was not further analyzed in relation to its individual components. However, the concentrations and compositions of RVS tested were analyzed and could serve as a standardized reference for the subsequent investigations of RVS extracts. In future research, we plan to explore the specific contributions of individual RVS components such as butein, fisetin, fustin, and sulfuretin to better understand their distinct roles in the observed immunomodulatory effects. Second, other widespread subtypes of breast cancer, for example HER2, were not tested for comparison, even though the difference between luminal A and TNBC was not observed in our analysis. Additionally, we did not evaluate the effect of RVS on normal breast cells to assess potential toxicity at the concentrations used. However, previous report indicates that normal cells exhibit lower toxicity than cancer cells when exposed to the same concentration of RVS. 14 Finally, further elucidation is needed by confirming various surface markers contributing to T-cell function 29 (eg, T cell Immunoglobulin and Mucin Domain-3; TIM-3, Lymphocyte Activation Gene-3; LAG-3) and verifying additional limitations such as the expression of PD-L1 or apoptosis markers in target cells altered by both T-cells and RVS.
Conclusions
Our study demonstrated the immunomodulatory effects of RVS on immune cells, particularly in the context of breast cancer. We found that RVS enhanced cancer cell-killing efficacy, downregulated PD-1 expression, induced changes in gene expression, and increased the release of pro-inflammatory cytokines. These findings suggest that RVS holds promise as a potential immunotherapeutic agent for breast cancer, especially in improving T cell function and modulating PD-1 expression. Further research is needed to identify the active compounds within RVS, evaluate their clinical efficacy, and explore combination strategies for different breast cancer subtypes.
Supplemental Material
Supplemental material, sj-pdf-1-ict-10.1177_15347354241308220 for Rhus Verniciflua Stokes Inhibits PD-1 Expression and Induces Anticancer Effects by Enhancing T Cell Function by Seoyoung Kim, Young-Kwan Lee, Wang-Jun Lee, Hyoun Jong Moon and Sanghun Lee in Integrative Cancer Therapies
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Myongji Hospital (2103-07-05) and the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), Republic of Korea (grant number: RS-2023-KH139182 and RS-2023-KH139933).
ORCID iD: Sanghun Lee  https://orcid.org/0000-0002-0573-9555
https://orcid.org/0000-0002-0573-9555
Supplemental Material: Supplemental material for this article is available online.
References
- 1. Li MC, Zhang Y-Q, Meng C-W, et al. Traditional uses, phytochemistry, and pharmacology of Toxicodendron vernicifluum (Stokes) F.A. Barkley - a review. J Ethnopharmacol. 2021;267:113476. [DOI] [PubMed] [Google Scholar]
- 2. Lee SH, Kim KS, Choi WC, Yoon SW. The concurrent use of rhus verniciflua stokes as complementary therapy with second or more line regimens on advanced non-small-cell lung cancer: case series. J Korean Med. 2009;30:112-117. [Google Scholar]
- 3. Lee SH, Choi WC, Yoon SW. Impact of standardized Rhus verniciflua stokes extract as complementary therapy on metastatic colorectal cancer: a Korean single-center experience. Integr Cancer Ther. 2009;8:148-152. [DOI] [PubMed] [Google Scholar]
- 4. Lee SK, Jung HS, Eo WK, et al. Rhus verniciflua Stokes extract as a potential option for treatment of metastatic renal cell carcinoma: report of two cases. Ann Oncol. 2010;21:1383-1385. [DOI] [PubMed] [Google Scholar]
- 5. Choi W, Jung H, Kim K, et al. Rhus verniciflua stokes against advanced cancer: a perspective from the Korean Integrative Cancer Center. J Biomed Biotechnol. 2011;2012:874276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Nam TG, Lee BH, Choi H-K, et al. Rhus verniciflua stokes extract and its flavonoids protect PC-12 cells against H(2)O(2)-induced cytotoxicity. J Microbiol Biotechnol. 2017;27:1090-1097. [DOI] [PubMed] [Google Scholar]
- 7. Lee S-H, Choi W-C, Kim K-S, et al. Shrinkage of gastric cancer in an elderly patient who received Rhus verniciflua Stokes extract. J Altern Complement Med. 2010;16:497-500. [DOI] [PubMed] [Google Scholar]
- 8. Cheon SH, Kim KS, Kim S, et al. Efficacy and safety of Rhus verniciflua stokes extracts in patients with previously treated advanced non-small cell lung cancer. Complement Med Res. 2011;18:2-83. [DOI] [PubMed] [Google Scholar]
- 9. Choi HS, Kim MK, Choi YK, et al. Rhus verniciflua Stokes (RVS) and butein induce apoptosis of paclitaxel-resistant SKOV-3/PAX ovarian cancer cells through inhibition of AKT phosphorylation. BMC Complement Altern Med. 2016;16:122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Li Y, Ma C, Qian M, et al. Butein induces cell apoptosis and inhibition of cyclooxygenase-2 expression in A549 lung cancer cells. Mol Med Rep. 2014;9:763-767. [DOI] [PubMed] [Google Scholar]
- 11. Samoszuk M, Tan J, Chorn G. The chalcone butein from Rhus verniciflua Stokes inhibits clonogenic growth of human breast cancer cells co-cultured with fibroblasts. BMC Complement Altern Med. 2005;5:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Afroze N, Pramodh S, Shafarin J, et al. Fisetin deters cell proliferation, induces apoptosis, alleviates oxidative stress and inflammation in human cancer cells, HeLa. Int J Mol Sci. 2022;23:1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Kumar RM, Kumar H, Bhatt T, et al. Fisetin in cancer: attributes, developmental aspects, and nanotherapeutics. Pharmaceuticals. 2023;16:196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Jang I-S, Park JW, Jo EB, et al. Growth inhibitory and apoptosis-inducing effects of allergen-free Rhu s verniciflua Stokes extract on A549 human lung cancer cells. Oncol Rep. 2016;36:3037-3043. [DOI] [PubMed] [Google Scholar]
- 15. Christodoulou M-I, Zaravinos A. New clinical approaches and emerging evidence on immune-checkpoint inhibitors as anti-cancer therapeutics: CTLA-4 and PD-1 pathways and beyond. Crit Rev Immunol. 2019;39:379-408. [DOI] [PubMed] [Google Scholar]
- 16. Postow MA, Callahan MK, Wolchok JD. Immune checkpoint blockade in cancer therapy. World J Clin Oncol. 2015;33:1974-1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Li W, Kim TI, Kim JH, Chung H-S. Immune checkpoint PD-1/PD-L1 CTLA-4/CD80 are blocked by Rhus verniciflua Stokes and its active compounds. Molecules. 2019;24:4062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Barba D, Saris SC, Holder C, Rosenberg SA, Oldfield EH. Intratumoral LAK cell and interleukin-2 therapy of human gliomas. J Neurosurg. 1989;70:175-182. [DOI] [PubMed] [Google Scholar]
- 19. Tario JD, Muirhead KA, Pan D, Munson ME, Wallace PK. Tracking immune cell proliferation | cytotoxic potential using flow cytometry. Methods Mol Biol. 2011;699:119-164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Pimentel VO, Yaromina A, Marcus D, Dubois LJ, Lambin P. A novel co-culture assay to assess anti-tumor CD8+ T cell cytotoxicity via luminescence and multicolor flow cytometry. J Immunol Methods. 2020;487:112899. [DOI] [PubMed] [Google Scholar]
- 21. Crowley LC, Marfell BJ, Christensen ME, Waterhouse NJ. Measuring cell death by trypan blue uptake and light microscopy. Cold Spring Harb Protoc. 2016;2016:b. prot087155. [DOI] [PubMed] [Google Scholar]
- 22. Min GJ, Kim HJ, Kim TG, et al. Specific donor HLA allotypes as predictors of cytomegalovirus disease risk in acute myeloid leukemia. HLA. 2020;96:445-455. [DOI] [PubMed] [Google Scholar]
- 23. Andersen M. Tumor microenvironment antigens. Paper presented at: Seminars in Immunopathology, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Lee JO, Moon JW, Lee SK, et al. Rhus verniciflua extract modulates survival of MCF-7 breast cancer cells through the modulation of AMPK-pathway. Biol Pharm Bull. 2014;37:794-801. [DOI] [PubMed] [Google Scholar]
- 25. Yang L-H, Ho YJ, Lin J-F, et al. Butein inhibits the proliferation of breast cancer cells through generation of reactive oxygen species and modulation of ERK and p38 activities. Mol Med Rep. 2012;6:1126-1132. [DOI] [PubMed] [Google Scholar]
- 26. Li Y, Li S, Meng X, et al. Dietary natural products for prevention and treatment of breast cancer. Nutrients. 2017;9:728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kim MS, Lee CW, Kim J-H, Lee J-C, An WG. Extract of Rhus verniciflua Stokes induces p53-mediated apoptosis in MCF-7 breast cancer cells. Evid Based Complement Alternat Med. 2019;2019:9407340. 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Zheng H, Wang G, Liu M, Cheng H. Traditional Chinese medicine inhibits PD-1/PD-L1 axis to sensitize cancer immunotherapy: a literature review. Front Oncol. 2023;13:1168226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lu C, Tan Y. Promising immunotherapy targets: TIM3, LAG3, and TIGIT joined the party. Mol Ther Oncol. 2024;32:200773. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-pdf-1-ict-10.1177_15347354241308220 for Rhus Verniciflua Stokes Inhibits PD-1 Expression and Induces Anticancer Effects by Enhancing T Cell Function by Seoyoung Kim, Young-Kwan Lee, Wang-Jun Lee, Hyoun Jong Moon and Sanghun Lee in Integrative Cancer Therapies