Abstract
Objective
Physiological blood pressure changes in pregnancy are insufficiently defined. This paper describes the blood pressure changes across healthy pregnancies in a Southern Chinese population to present gestational - age - specific blood pressure ranges with smoothed centiles (3rd, 10th, 50th, 90th, and 97th).
Methods
Antenatal blood pressure measurements [median (interquartile range) 9 (8 - 10) per woman] were repeated in 17, 776 women from a Southern China population. Multilevel cubic splines models were used to derive longitudinal reference ranges for systolic blood pressure (SBP) and diastolic blood pressure (DBP) from 6 to 42 weeks of gestation for the normal pregnancies (excluding chronic hypertension, preeclampsia, diabetic ones, and preterm birth et al.).
Results
Systolic and diastolic BP increased from 6 weeks of gestation: 50th centile (3rd - 97th centile) 106 (87 - 125); 61 (47 - 77) mm Hg to 12 weeks of gestation: 50th centile (3rd - 97th centile) 108 (88 - 129); 64 (49 - 81) mm Hg. Then, the lowest value of 107 (87 - 129); 62 (47 - 78) mm Hg was reached at 16 weeks and 20 weeks of gestation, respectively. Systolic and diastolic BP then rose to a maximum median (3rd - 97th centile) of 115 (96 - 135); 68 (53 - 85) mm Hg at 42 weeks of gestation. Additionally, the ascending tendency of SBP after 16 weeks of gestation was interrupted by two fluctuations that occurred at 24 weeks and 30 weeks of gestation.
Conclusions
In summary, our study provides blood pressure reference values for Southern Chinese women with normal pregnancies. To identify gestational hypertension and hypotension, centiles for gestational - age - specific BP should be defined in healthy pregnancies. Understanding these changes in low risk pregnancies is essential to optimize maternal blood pressure management.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12884-024-07117-3.
Keywords: Blood pressure trajectory, Normal pregnancy, Reference intervals
Introduction
The hypertensive disorders of pregnancy (HDP) collectively account for 14% of all maternal deaths worldwide [1]. Due to the ascending trend in the prevalence of HDP [2, 3], it is currently considered one of the major pregnancy health concerns. The American College of Obstetricians and Gynecologists (ACOG) has retained a definition of blood pressure greater than or equal to 140 / 90 mm Hg for hypertension in pregnancy. However, this definition is derived from hypertensive guidelines for nonpregnant individuals and is currently used as a screening tool for preeclampsia (PE). Several studies have reported a dose - response relationship between increased blood pressure (lower than 140 / 90 mm Hg) and adverse pregnancy outcomes across gestational age [2, 3]. These findings provide potential support for the redefinition of hypertension in pregnancy. Close antenatal care and proper intervention may reduce hypertension and preeclampsia - related morbidity and mortality.
Additionally, the literature on maternal hypotension is scarce, and inconsistent definitions for hypotension are used. Previous studies were based on arbitrary definitions of hypotension focusing on either the systolic blood pressure (SBP), the diastolic blood pressure (DBP), readings for both SBP and DBP, or a specific BP reading (e.g.,110 / 70 mm Hg) [4]. Pregnant women with borderline DBP (60 to 70 mm Hg) were exposed to a greater risk of stillbirth relative to normotensive pregnancies [4]. Furthermore, low risk nulliparous women with persistent maternal hypotension were found to be at an increased risk of delivering small-for-gestational age neonates [5]. However, the BP reference ranges for normal pregnancies in Southern China remain unclear.
The aim of this study was to investigate the BP trajectories of uncomplicated pregnant women across gestation in a large retrospective population using a multilevel cubic spline model. Improving the understanding of the normal BP trajectory in pregnancy may promote appropriate prevention and intervention strategies for HDP and hypotension, thus improving maternal and fetal outcomes.
Materials and methods
Study participants
The data, including maternal covariates, BP values and delivery information, were obtained by abstracted from electronic medical records. In brief, pregnant women with registration data between January 1, 2022, and May 30, 2023 were eligible for recruitment. This study was approved by the Longgang District Maternity & Child Healthcare Hospital Research Ethics Committee (No. LGFYYXLLL-2022-016). The participants provided informed consent at the initial antenatal visit. Of the 30,438 women enrolled, those with missing information on maternal characteristics at baseline (n = 4,113) and those with less than 7 BP measurements (n = 418) were excluded. In addition, women aged <17 years or >45 years (n = 108) were excluded from the study. Finally, the study included 25,799 women.
Obstetric measurements
According to the antenatal care policy of Guangdong Province, routine obstetrical visit was initiated at ~ 12 weeks of gestation, followed by repeated measurements every four weeks before 28 weeks of gestation, every two weeks at 28 - 34 weeks of gestation, and every week after 34 weeks of gestation until delivery. Blood pressure measurement is required at the antenatal visit. An automated digital sphygmomanometer was used by trained nurses to measure blood pressure twice (the average was used) at three - minute intervals, and the subject was requested to sit quietly for at least ten minutes before BP measurement. We used an automated oscillometric BP monitoring system (HEM907, Omron, Kyoto, Japan) validated for pregnancy according to the protocol of the European Society of Hypertension [6].
The primary outcome of this study was gestation - specific reference ranges for BP, comprising centile distributions. Preeclampsia is defined as SBP ≥ 140 mm Hg and / or DBP ≥ 90 mm Hg, along with proteinuria greater than one positive on urine dipstick testing or more than 300 mg per 24 hours after 20 weeks of pregnancy [7]. Delivery before 37 complete weeks of pregnancy was considered preterm birth.
Covariates
The covariates included in the analysis were maternal age (years), gravidity (1, 2, or ≥ 3 times), parity (nulliparous or multiparous), and prepregnancy body mass index (BMI). The BMI status of participants was classified as underweight (< 18.5 kg/m2), normal weight (18.5 - 23.9 kg/m2), overweight (24.0 - 27.9 kg/m2) and obese (≥ 28.0 kg/m2) in accordance with Chinese BMI guidelines [8]. Pregnancy complications and the information of delivery mode were also assessed.
Definition of normal pregnancy
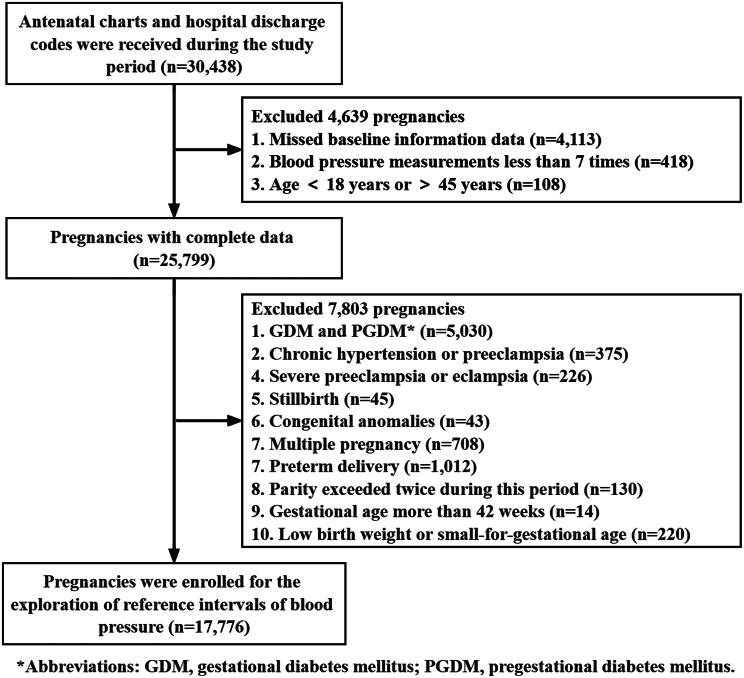
As recommended in a previous study [9], based on the current pregnancy electronic medical records, only “normal” pregnancies were retained by excluding pregnant women with pregestational diabetes mellitus (PGDM) or gestational diabetes mellitus (GDM) (n = 5,030), chronic hypertension or PE (n = 375), severe PE or eclampsia (n = 226), stillbirth (n = 45), congenital anomalies (n = 43), multiple pregnancies (n = 708), preterm delivery (n = 1,012), parity exceeded twice during this period (n = 130) and over 42 weeks of gestation (n = 14). Cases who were classified as low birth weight (birth weight less than 2500 g) or small-for-gestational age (below the 10th percentile of norms for gestational age) were also excluded (n = 220). In addition, women diagnosed with gestational hypertension were not excluded because this would have eliminated the top end of the BP distribution and biased the reference ranges, as reported by Macdonald-Wallis et al. in 2015 [9]. Thus, 17,776 women were included in the final analysis (Fig. 1).
Fig. 1.
Flowchart illustrating women enrolled in the study. Abbreviations: GDM, gestational diabetes mellitus; PGDM, pregestational diabetes mellitus
Statistical analysis
First, the statistical analysis of maternal information was conducted with SPSS V.22.0 software (IBM, Armonk, NY, United States). Continuous variables were summarized as mean (standard deviation) or median (interquartile range), while categorical variables were displayed as frequencies. An unpaired Student t-test was employed to test for differences in quantitative variables, while a χ2 test was used to compare categorical variables. A two-tailed P value of 0.05 was defined as the threshold for statistical significance.
Then, most classical statistical methods for the analysis of single measure data assume that the data is independent. However, longitudinal repeated measure data require different analysis approaches. The cubic spline smoothing technique was used to generate the smoothed centile growth curves and gestational age - specific reference intervals for BP distribution (3rd, 10th, 50th, 90th and 97th as used by the World Health Organization Multicenter Growth Reference Study [10], with corresponding 95% confidence interval) were proposed for the pregnancy women. Centiles with a 95% confidence interval (CI) for BP from 6 to 42 weeks of gestation were presented graphically and outlined at two - weekly intervals. The statistical methods were described in previous reports [11, 12], multilevel linear modeling was used to estimate BP measurements linear change over the pregnancy period (time effect), and for multiple measurements taken at each visit at the same gestational age (group effect), as well as measurements from the women at different gestational age (group-by-time interaction), accounting for the dependency among measurements. These models incorporate both fixed and random components, where the fixed components are consistent across all subjects, while the random components vary between subjects. By analyzing each subject’s change pattern over time, these models enable quantification of the between-subject variability in the given population. Overall, multilevel linear models provide a proper approach for analyzing dependent observations and characterizing individual variation in longitudinal data. Statistical analysis was performed using SPSS version 22 (IBM Corp.), and the reference range figures were produced in R version 3.40 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics
The baseline characteristics of the participants according to pregnancy complications are presented in Table 1. In total, 17,776 healthy pregnant women were enrolled in this study. The mean maternal age was 28.94 ± 4.40 (standard deviation [SD] 4.40) years. Women undergoing normal pregnancies had 16,3039 BP measurements, with a median (interquartile range) of 9 (8 - 10) measurements per woman. The participants were mostly lean, under 30 years old and had a spontaneous vaginal delivery.
Table 1.
Descriptive frequencies of demographic and early pregnancy covariates, and the mode of delivery for the study participants according to complications
| Variable | Normal pregnancies | High risk pregnancies | |
|---|---|---|---|
| (n = 17,776) | (n = 7,803) | P value | |
| Age, years, | |||
| Mean (SD): | 28.94 (4.40) | 30.95 (4.57) | |
| <35, % (n) | 92.5 (16,448) | 77.8 (6,074) | <0.0001 |
| ≥ 35, % (n) | 7.5 (1,328) | 22.2 (1,729) | |
| Pre-pregnancy BMI kg/m2, % (n): | |||
| <18.5 | 21.8 (3,869) | 12.4 (968) | <0.0001 |
| 18.5-23.9 | 66.8 (11,877) | 65.1 (5,080) | |
| 24.0-27.9 | 9.7 (1,720) | 17.8 (1,387) | |
| ≥ 28 | 1.7 (310) | 4.7 (368) | |
| Gravidity, time, % (n): | |||
| 1 | 29.8 (5,294) | 28.6 (2,234) | <0.0001 |
| 2 | 35.2 (6,249) | 32.3 (2,524) | |
| ≥ 3 | 35.0 (6,233) | 39.1 (3,045) | |
| Parity, time, % (n) | |||
| Nulliparous | 41.6 (7,403) | 43.8 (3,421) | <0.001 |
| Multiparous | 58.4 (10,373) | 56.2 (4,382) | |
| Delivery mode, % (n) | |||
| Spontaneous vaginal delivery | 69.4 (12,343) | 52.5 (4,099) | <0.001 |
| Operative vaginal delivery | 4.2 (748) | 2.6 (200) | |
| Cesarean delivery | 26.4 (4,685) | 44.9 (3,504) | |
| GA at delivery, weeks | 39.5 (1.2) | 38.6 (1.9) | 0.02 |
Abbreviations: BMI, body mass index. BMI was calculated as: BMI = weight (kg) / height2 (m2). The subgroups were divided by BMI according to the guidelines of BMI in China. GA, gestational age. Data are presented as means (standard deviation, SD) for continuous variables and frequencies (percentages) for categorical variables
The primary outcome
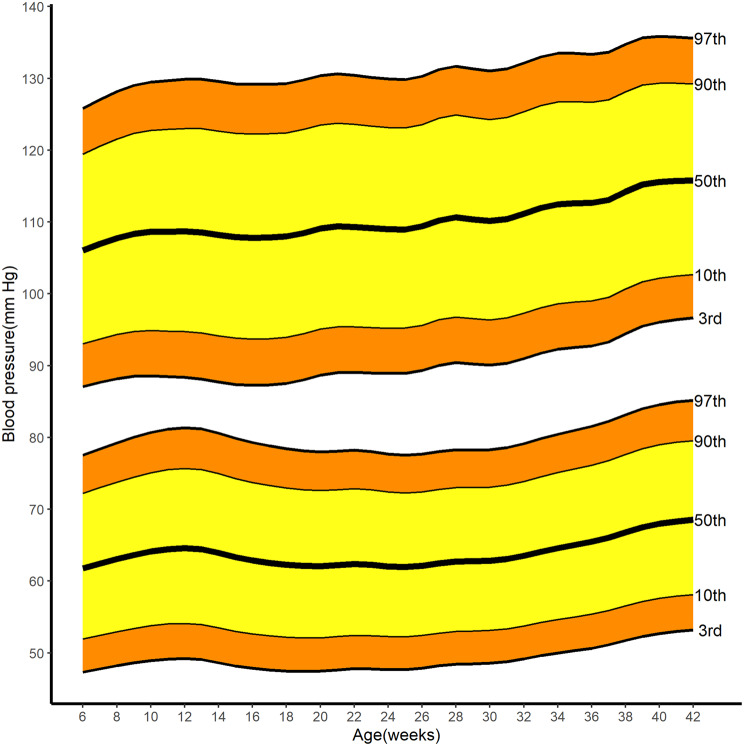
Figure 2 represents smoothed centiles for SBP and DBP for the 3rd, 10th, 50th, 90th, and 97th centiles according to gestational age. The reference values for BP from 6 to 42 weeks of gestation (at two - week intervals) are summarized in Table 2. The full references are available in Supplementary Table 1. Generally, the normal reference intervals for SBP and DBP both demonstrated a slight decrease in the first trimester, then increased from the second trimester. Specifically, SBP rose from 6 weeks of gestation to 12 weeks of gestation, then decreased to reach its lowest value of 107.8 (95% CI 87.3 - 129.2) mm Hg at 16 weeks of gestation. Subsequently, a steady rise (albeit with fluctuations) was observed from 17 weeks of gestation to a maximum median of 115.7 mm Hg (3rd to 97th centile 96.6 - 135.6) at 42 weeks, representing a difference (95% CI) of 8.0 (6.0 to 9.0) mm Hg. The two fluctuations occurred at 24 weeks (median of 109.0 [95% CI 108.9 to 109.2] mmHg) and 30 weeks (median of 110.1 [95% CI 110.0 to 110.3] mm Hg) of gestation, respectively. In addition, SBP was measured at 37 weeks: the 3rd centile was 93.3 (95% CI 93.1 to 93.4) mm Hg and 97th was 133.6 (95% CI 133.5 to 133.8) mm Hg.
Fig. 2.
Reference ranges for systolic blood pressure (upper line and centiles, mm Hg) and diastolic blood pressure (lower line and centiles, mm Hg) between 6 weeks and 42 weeks of gestation in normal pregnancies. Centiles are labelled. A normal pregnancy was defined as a woman did not have chronic hypertension, preeclampsia, nondiabetic pregnancies and delivered at term
Table 2.
Smoothed centiles for systolic blood pressure and diastolic blood pressure (mm Hg) for normal pregnancy according to gestational age for 3rd, 50th, and 97th centiles (95% CI)
| Gestational age, weeks |
Centiles for Systolic Blood Pressure (95% CI) | ||
|---|---|---|---|
| 3rd | 50th | 97th | |
| 6 | 87.0 (84.1–87.7) | 106.0 (102.4–107.4) | 125.8 (121.1–128.1) |
| 8 | 88.1 (86.4–88.2) | 107.7 (105.6–108.3) | 128.1 (125.2–129.5) |
| 10 | 88.6 (88.0–89.0) | 108.6 (107.8–108.9) | 129.5 (128.3–130.2) |
| 12 | 88.3 (87.9–88.6) | 108.6 (108.5–108.8) | 129.8 (129.6–130.1) |
| 14 | 87.7 (87.4–88.0) | 108.2 (108.0–108.4) | 129.5 (129.3–129.7) |
| 16 | 87.2 (87.1–87.5) | 107.7 (107.6–107.9) | 129.1 (128.9–129.3) |
| 18 | 87.5 (87.3–87.6) | 107.9 (107.6–108.1) | 129.3 (128.9–129.5) |
| 20 | 88.6 (88.6–88.8) | 109.1 (108.9–109.3) | 130.3 (130.1–130.6) |
| 22 | 89.0 (88.8–89.3) | 109.3 (109.2–109.5) | 130.4 (130.3–130.6) |
| 24 | 88.9 (88.6–89.2) | 109.0 (108.9–109.2) | 129.9 (129.8–130.1) |
| 26 | 89.3 (89.1–89.5) | 109.3 (109.2–109.6) | 130.2 (130.1–130.6) |
| 28 | 90.4 (90.2–90.5) | 110.6 (110.4–110.6) | 131.7 (131.4–131.8) |
| 30 | 90.0 (89.9–90.3) | 110.1 (110.0–110.3) | 131.0 (130.9–131.2) |
| 32 | 90.9 (90.8–91.1) | 111.1 (110.9–111.2) | 132.1 (131.9–132.3) |
| 34 | 92.2 (92.1–92.4) | 112.4 (112.4–112.5) | 133.5 (133.3–133.7) |
| 36 | 92.7 (92.6–92.9) | 112.6 (112.6–112.6) | 133.3 (133.2–133.4) |
| 38 | 94.4 (94.2–94.5) | 114.2 (114.1–114.3) | 134.7 (134.6–134.8) |
| 40 | 96.0 (95.7–96.2) | 115.6 (115.4–115.7) | 135.8 (135.6–136.1) |
| 41 | 96.3 (95.7–96.6) | 115.7 (115.1–116.0) | 135.7 (135.2–136.2) |
| 6 | 47.3 (44.4–50.0) | 61.8 (57.8–63.9) | 77.5 (72.0–79.2) |
| 8 | 48.2 (46.6–49.9) | 63.0 (60.9–64.3) | 79.2 (76.1–80.0) |
| 10 | 48.9 (48.3–49.8) | 64.1 (63.3–64.6) | 80.6 (79.5–81.0) |
| 12 | 49.2 (49.0–49.5) | 64.5 (64.5–64.8) | 81.3 (81.1–81.6) |
| 14 | 48.6 (48.4–48.8) | 63.9 (63.8–64.1) | 80.6 (80.4–81.0) |
| 16 | 47.8 (47.5–48.0) | 62.8 (62.7–63.0) | 79.2 (79.1–79.5) |
| 18 | 47.4 (47.3–47.5) | 62.2 (62.0–62.3) | 78.4 (78.0–78.6) |
| 20 | 47.4 (47.3–47.5) | 62.0 (61.9–62.1) | 78.0 (77.6–78.0) |
| 22 | 47.8 (47.7–47.9) | 62.3 (62.2–62.5) | 78.2 (78.0–78.4) |
| 24 | 47.6 (47.4–47.7) | 62.0 (61.9–62.1) | 77.6 (77.4–77.8) |
| 26 | 47.8 (47.7–47.9) | 62.1 (62.0–62.2) | 77.6 (77.5–77.9) |
| 28 | 48.4 (48.2–48.5) | 62.7 (62.6–62.8) | 78.2 (78.1–78.4) |
| 30 | 48.5 (48.4–48.6) | 62.8 (62.6–62.8) | 78.2 (78.0–78.4) |
| 32 | 49.1 (49.1–49.2) | 63.5 (63.4–63.6) | 79.1 (79.0–79.2) |
| 34 | 49.9 (49.8–50.1) | 64.5 (64.5–64.6) | 80.4 (80.3–80.6) |
| 36 | 50.6 (50.5–50.7) | 65.4 (65.3–65.5) | 81.5 (81.4–81.7) |
| 38 | 51.7 (51.5–51.8) | 66.8 (66.6–66.8) | 83.1 (83.0–83.2) |
| 40 | 52.7 (52.6–53.0) | 67.9 (67.8–68.1) | 84.5 (84.2–84.8) |
| 41 | 53.0 (52.8–53.4) | 68.3 (68.1–68.6) | 84.9 (84.3–85.4) |
Abbreviations: CI, confidence interval
Moreover, as shown in Table 2, the third centile for SBP was never less than 90 mm Hg and exceeded 96 mm Hg after 30 weeks of gestation.
Additionally, the median DBP increased from 6 weeks to 12 weeks of gestation, with a difference of 6.7 (95% CI 5.0 to 7.0) mm Hg, then decreased to its lowest value of 62.0 (95% CI 47.4 - 78.0) mm Hg at 20 weeks, representing a difference (95% CI) of -2.0 (-3.0 to 0.0) mm Hg. Thereafter, DBP increased to a maximum median of (3rd - 97th centile) 68.5 (53.2 - 85.1) mm Hg at 42 weeks, displaying a steady growth of 6.0 (95% CI, 5.0 - 8.0) mm Hg from the nadir to the peak point of DBP. Specifically, DBP was measured at 37 weeks: the 3rd centile was 51.1 (95% CI 51.0 to 51.2) mm Hg and 97th was 82.2 (95% CI 82.1 to 82.4) mm Hg.
Finally, as presented in Table 2, the 97th centile of SBP and DBP values were all above 130 mm Hg and 80 mm Hg after 34 weeks of gestation.
Discussion
Our study examined the BP gestational course trajectories in women with uncomplicated pregnancies. The results revealed that both SBP and DBP progressively increased from 6 weeks to 12 weeks of gestation, then decreased to reach the nadir in the second trimester, reflecting the expected physiological decline by the second trimester for normal pregnancy. Then, BP gradually increased until term. Generally, BP showed upward changes across gestation. Nevertheless, the rise in BP may not be linear throughout gestation. In addition, the bilateral cut off limits defining hypertension and hypotension for women at 37 weeks of gestation in Southern China may be 133 / 82 mm Hg and 93 / 51 mm Hg, respectively. According to the American College of Cardiology and American Heart Association (ACC / AHA) guidelines, stage 1 hypertension was defined as SBP ≥ 130 mm Hg or DBP ≥ 80 mm Hg, improving the management of women at risk of HDP. Our results also support this recommendation. These findings may have important implications in identifying abnormal BP during pregnancy in China.
Changes in the BP pattern during pregnancy have been observed. Studies showed that BP declined from first to second trimester [13, 14], while recent publications challenged this notion and demonstrated a progressive increase in BP as the gestation progresses [15–17]. We observed a rise in BP from 6 weeks to 12 weeks of gestation, followed by a slight decline from 12 weeks to 16 weeks of gestation, which might indicate a physiological decline in second trimester in a Southern Chinese population. That the reports used different methods of assessing BP may contribute to the variations in the results. Furthermore, largely unexplained but marked ethnic and geographic differences exist in BP levels observed during pregnancy and the risk of HDP [18]. In accordance with other reports [9, 13, 15, 16], blood pressure naturally increases from 19 weeks of gestation, although our study identified slight BP fluctuations at 24 weeks and 30 weeks of gestation. The findings allow clinicians to recognize relative hypotension from the BP measurements. The third centile for SBP did not fall below 90 mm Hg, and was greater than 96 mm Hg after 30 weeks of gestation, which concurred with obstetric early warning score charts [19]. These thresholds can be used to recognize sepsis and require urgent attention in pregnancy [20]. Categorizing elevated BP (120 - 129 / < 80 mm Hg) and stage 1 hypertension (130 - 139 or 80 - 89 mm Hg) improves identification of subjects at risk for PE [21]. Reconsideration of the threshold of BP values in specific territory may result in the implementation of appropriate measures to management PE [22]. Therefore, the BP trajectory variance suggests appropriate gestational thresholds may improve the detection and management of gestational hypertension and preeclampsia.
We speculate that some prominent mechanisms may explain our findings. First, early in pregnancy, the high resistance circulation within the uterus is transformed into a low resistance circulation by trophoblastic invasion and spiral artery remodeling. The remodeling is believed to begin around the end of the first trimester and is completed by 18 to 20 weeks [23]. In our study, the lowest point of SBP occurred at 16 weeks of gestation, and that of the DBP occurred at 20 weeks of gestation. Pregnancy triggers significant physiologic adaptations in the cardiovascular system. Maternal cardiac output (CO) can increase by around 30-50% throughout gestation. These changes primarily occur during early trimesters of pregnancy, then plateau between 28 weeks’ and 32 weeks’ gestation and are maintained until delivery [24]. Moreover, the myocardial contractility and cardiac compliance increase as gestation progresses [25]. Second, dilatation of the microvasculature causes a decrease in peripheral resistance, which is disproportional to the raised CO, thereby lowering maternal BP early in the second trimester [26]. Previous studies have reported variations in aortic stiffness during pregnancy, indicated by the pulse wave velocity (PWV) and the augmentation index (AIx). These studies have consistently concluded that AIx decreases in the first trimester, reaching its lowest point in the second trimester before rising again. Robb et al. [27] reported a similar pattern with PWV, the trough occurring at 24 weeks of gestation, with a progressive increase until delivery. Both AIx and PWV showed a modest U - shaped relationship with gestational age in previous research [28]. The nadir of the curve for AIx occurred at 25-27 weeks of gestation. For PWV, the trough was observed at 18- 25 weeks of gestation. Third, the placental growth factor (PlGF) serves as an endocrine signal to promote normal systemic endothelial function. In normal pregnancy, maternal PlGF levels rise and then plateau at 28 to 30 weeks of gestation, subsequently falling beyond 36 weeks when fetal growth velocity decelerates [29, 30]. Interestingly, uterine artery Doppler studies reported a significant decrease in the resistance index from the nonpregnant state to early pregnancy, the decline continuing onward 30 weeks of gestation [31]. Collectively, the changes in hemodynamic parameters and endothelial activation molecules may account for the BP changes, resulting in BP decline in the second trimester, followed by a gradual increase until delivery with slight fluctuations at 24 weeks and 30 weeks of gestation.
The strengths of our study include the large sample size. Moreover, a longitudinal analysis of BP was conducted using statistical methods accommodating the unstructured nature of BP measurements. Cross sectional analysis of longitudinal data could enhance the likelihood of a type I error owing to multiple comparisons in these non-independent data sets. While other approaches are useful for modeling longitudinal data, they typically assume a biologically piecewise linear growth pattern [32]. In this study, trajectories were outlined without relying on these restrictive assumptions, allowing a more natural fit.
Nevertheless, the potential limitations should be mentioned. First, single center, retrospective observational studies are prone to various sources of bias, including confounding and selection bias. The participants in our study were ethnically homogeneous, the generalizability of our findings to another population of normal pregnant women needs further validation. A larger and more diverse participant population may help to answer this question. In addition, residual confounding cannot be ruled out, as we did not have data on covariates, including a history of pregnancy complications, although our findings are relevant to the clinical setting. Moreover, the 97th percentile blood pressure (135 / 85 mm Hg) in the third trimester that we used was not the exact cut-off values, and future studies are warranted to validate the best blood pressure cut-off for predicting hypertensive disorders of pregnancy. Second, although the physiological BP trajectory in our study might be of interest, it should be considered the clinical implications. Previous research found that women with high normal and abnormal BP trajectories were at increased risk of preterm birth [33], using the group-based trajectory modeling could confirm the clinical significance of our findings. Third, in an analysis of successive pregnancies, Bernstein et al. showed that mean arterial pressure was reduced in the subsequent pregnancies compared with the index pregnancies [34]. Furthermore, the reduction in mean arterial pressure between the two pregnancies was negatively correlated with the length of the interpregnancy intervals [35]. Blood pressure values stratified by parity and pregnancy intervals should be determined.
In summary, our study presents blood pressure reference intervals for Southern Chinese women with uncomplicated pregnancies. Our results support the notion that a physiological decline in BP exists in the second trimester, and women diagnosed with ACC / AHA recommended stage 1 hypertension should receive close follow up. Defining centiles for SBP and DBP in healthy pregnancies would facilitate the identification of individuals with normal and abnormal blood pressure, based on the gestational age. Understanding these changes in uncomplicated pregnancy is essential to optimize maternal blood pressure management.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1: Smoothed centiles for systolic blood pressure and diastolic blood pressure (mm Hg) from 6 weeks to 42 weeks of gestation for normal pregnancy according to gestational age for 3rd, 10th, 50th, 90th and 97th centiles (95% CI).
Acknowledgements
We are extremely grateful to all the participants who took part in this study.
Abbreviations
- BP
Blood Pressure
- SBP
Systolic Blood Pressure
- DBP
Diastolic Blood Pressure
- HDP
Hypertensive Disorders of pregnancy
- ACOG
American College of Obstetricians and Gynecologists
- PE
preeclampsia
- BMI
Body Mass Index
- PGDM
Pregestational diabetes mellitus
- GDM
Gestational diabetes mellitus
- CI
Confidence interval
- ACC / AHA
American College of Cardiology and American Heart Association
- CO
Cardiac output
- PWV
Pulse wave velocity
- AIx
Augmentation index; PlGF: placental growth factor
Author contributions
YD conceived and designed the study, implemented data collection, perform statistical analysis, and wrote the paper. LL and YG analyzed the data, reviewed the paper. YG and GL are responsible for clinical data collection. QC and ZH analyzed the data and reviewed the paper. JY acquired the data and funding, administrated project, and revised the paper. All authors read and approved the final manuscript.
Funding
This work was supported by National Natural Science Foundation of China (81971417), Medical and Health Science and Technology Plan attached to Longgang District Science and Technology Development Special Funding (No. LGKCYLWS2021000025), Shenzhen Municipal S&T programme (No. JCYJ20220530162415035) and Research Initiation Fund of Longgang District Maternity & Child Healthcare Hospital of Shenzhen City (No. Y2024012).
Data availability
The data that support the findings of this study are availability from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study has ethical approval from the Longgang District Maternal & Child Healthcare Hospital Research Ethics Committee (No. LGFYYXLLL-2022-016). All participants have signed the written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Say L, Chou D, Gemmill A, Tuncalp O, Moller AB, Daniels J, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014;2(6):e323–33. [DOI] [PubMed] [Google Scholar]
- 2.Reddy M, Rolnik DL, Harris K, Li W, Mol BW, Da Silva Costa F, et al. Challenging the definition of hypertension in pregnancy: a retrospective cohort study. Am J Obstet Gynecol. 2020;222(6):e6061–21. [DOI] [PubMed] [Google Scholar]
- 3.Bone JN, Magee LA, Singer J, Nathan H, Qureshi RN, Sacoor C, et al. Blood pressure thresholds in pregnancy for identifying maternal and infant risk: a secondary analysis of community-level interventions for pre-eclampsia (CLIP) trial data. Lancet Global Health. 2021;9(8):e1119–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Warland J, McCutcheon H, Baghurst P. Maternal blood pressure in pregnancy and stillbirth: a case-control study of third-trimester stillbirth. Am J Perinatol. 2008;25(5):311–17. [DOI] [PubMed] [Google Scholar]
- 5.de Los Reyes S, Plunkett BA, Dude A. The association between persistent maternal hypotension and small for gestational age neonates. Am J Obstet Gynecol MFM. 2021;3(6):100449. [DOI] [PubMed] [Google Scholar]
- 6.Hurrell A, Webster L, Chappell LC, Shennan AH. The assessment of blood pressure in pregnant women: pitfalls and novel approaches. Am J Obstet Gynecol. 2022;226(2):S804–18. [DOI] [PubMed] [Google Scholar]
- 7.Espinoza JVAP. ACOG Practice Bulletin 202 Summary: gestational hypertension and Preeclampsia. Obstet Gynecol. 2019;133(1):1. [DOI] [PubMed] [Google Scholar]
- 8.Zhang C, Lai J, Liu K, Yang N, Zeng G, Mao L, et al. Optimal gestational weight gain in Chinese pregnant women by chinese-specific BMI categories: a multicenter prospective cohort study. Public Health Nutr. 2021;24(11):3210–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Macdonald-Wallis C, Silverwood RJ, Fraser A, Nelson SM, Tilling K, Lawlor DA, et al. Gestational-age-specific reference ranges for blood pressure in pregnancy. J Hypertens. 2015;33(1):96–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.De Onis M. WHO Child Growth standards based on length/height, weight and age. Acta Paediatr Suppl. 2006;450:76–85. [DOI] [PubMed] [Google Scholar]
- 11.Altman DG, Ohuma EO. Statistical considerations for the development of prescriptive fetal and newborn growth standards in the INTERGROWTH-21st Project. BJOG 2013;120 Suppl 2:71 – 6, v. [DOI] [PubMed]
- 12.Ohuma EO, Altman DG. Design and other methodological considerations for the construction of human fetal and neonatal size and growth charts. Stat Med. 2019;38(19):3527–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Costantine MM. Physiologic and pharmacokinetic changes in pregnancy. Front Pharmacol. 2014;5:65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Grindheim G, Estensen ME, Langesaeter E, Rosseland LA, Toska K. Changes in blood pressure during healthy pregnancy: a longitudinal cohort study. J Hypertens. 2012;30(2):342–50. [DOI] [PubMed] [Google Scholar]
- 15.Green LJ, Mackillop LH, Salvi D, Pullon R, Loerup L, Tarassenko L, et al. Gestation-Specific Vital sign reference ranges in pregnancy. Obstet Gynecol. 2020;135(3):653–64. [DOI] [PubMed] [Google Scholar]
- 16.Green LJ, Kennedy SH, Mackillop L, Gerry S, Purwar M, Staines Urias E, et al. International gestational age-specific centiles for blood pressure in pregnancy from the INTERGROWTH-21st Project in 8 countries: a longitudinal cohort study. PLoS Med. 2021;18(4):e1003611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nama V, Antonios TF, Onwude J, Manyonda IT. Mid-trimester blood pressure drop in normal pregnancy: myth or reality? J Hypertens. 2011;29(4):763–8. [DOI] [PubMed] [Google Scholar]
- 18.Bouthoorn SH, Gaillard R, Steegers EA, Hofman A, Jaddoe VW, van Lenthe FJ, et al. Ethnic differences in blood pressure and hypertensive complications during pregnancy: the Generation R study. Hypertension. 2012;60(1):198–205. [DOI] [PubMed] [Google Scholar]
- 19.Mhyre JM, D’Oria R, Hameed AB, Lappen JR, Holley SL, Hunter SK, et al. The maternal early warning criteria: a proposal from the national partnership for maternal safety. Obstet Gynecol. 2014;124(4):782–86. [DOI] [PubMed] [Google Scholar]
- 20.Acosta CD, Kurinczuk JJ, Lucas DN, Tuffnell DJ, Sellers S, Knight M. Severe maternal sepsis in the UK, 2011–2012: a national case-control study. PLoS Med. 2014;11(7):e1001672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hauspurg A, Parry S, Mercer BM, Grobman W, Hatfield T, Silver RM, et al. Blood pressure trajectory and category and risk of hypertensive disorders of pregnancy in nulliparous women. Am J Obstet Gynecol. 2019;221(3):e2771–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hauspurg A, Sutton EF, Catov JM, Caritis SN. Aspirin effect on Adverse Pregnancy Outcomes Associated with Stage 1 hypertension in a high-risk cohort. Hypertension. 2018;72(1):202–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lam C, Lim KH, Karumanchi SA. Circulating angiogenic factors in the pathogenesis and prediction of preeclampsia. Hypertension. 2005;46(5):1077–85. [DOI] [PubMed] [Google Scholar]
- 24.Robson SC, Hunter S, Boys RJ, Dunlop W. Serial study of factors influencing changes in cardiac output during human pregnancy. Am J Physiol. 1989;256(4 Pt 2):H1060–5. [DOI] [PubMed] [Google Scholar]
- 25.Rubler S, Damani PM, Pinto ER. Cardiac size and performance during pregnancy estimated with echocardiography. Am J Cardiol. 1977;40(4):534–40. [DOI] [PubMed] [Google Scholar]
- 26.Clapp JR, Seaward BL, Sleamaker RH, Hiser J. Maternal physiologic adaptations to early human pregnancy. Am J Obstet Gynecol. 1988;159(6):1456–60. [DOI] [PubMed] [Google Scholar]
- 27.Robb AO, Mills NL, Din JN, Smith IB, Paterson F, Newby DE, et al. Influence of the menstrual cycle, pregnancy, and preeclampsia on arterial stiffness. Hypertension. 2009;53(6):952–8. [DOI] [PubMed] [Google Scholar]
- 28.Anness AR, Nath M, Melhuish K, Osman MW, Webb D, Robinson T, et al. Arterial stiffness throughout pregnancy: Arteriograph device-specific reference ranges based on a low-risk population. J Hypertens. 2022;40(5):870–7. [DOI] [PubMed] [Google Scholar]
- 29.Levine RJ, Maynard SE, Qian C, Lim KH, England LJ, Yu KF, et al. Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med. 2004;350(7):672–83. [DOI] [PubMed] [Google Scholar]
- 30.McLaughlin K, Hobson SR, Chandran AR, Agrawal S, Windrim RC, Parks WT, et al. Circulating maternal placental growth factor responses to low-molecular-weight heparin in pregnant patients at risk of placental dysfunction. Am J Obstet Gynecol. 2022;226(2):S1145–56. [DOI] [PubMed] [Google Scholar]
- 31.Ling HZ, Guy GP, Bisquera A, Poon LC, Nicolaides KH, Kametas NA. The effect of parity on longitudinal maternal hemodynamics. Am J Obstet Gynecol. 2019;221(3):e2491–14. [DOI] [PubMed] [Google Scholar]
- 32.Howe LD, Tilling K, Matijasevich A, Petherick ES, Santos AC, Fairley L, et al. Linear spline multilevel models for summarising childhood growth trajectories: a guide to their application using examples from five birth cohorts. Stat Methods Med Res. 2016;25(5):1854–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Malaba TR, Cois A, Madlala HP, Matjila M, Myer L, Newell M. Blood pressure trajectories during pregnancy and associations with adverse birth outcomes among HIV-infected and HIV-uninfected women in South Africa: a group-based trajectory modelling approach. BMC Pregnancy Childbirth. 2020;20(1):742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bernstein IM, Thibault A, Mongeon JA, Badger GJ. The influence of pregnancy on arterial compliance. Obstet Gynecol. 2005;105(3):621–5. [DOI] [PubMed] [Google Scholar]
- 35.Howe L, Hammer E, Badger G, Bernstein IM. Effect of pregnancy interval on second pregnancy blood pressure following prior Preeclampsia. Reproductive Sci. 2018;25(5):727–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material 1: Smoothed centiles for systolic blood pressure and diastolic blood pressure (mm Hg) from 6 weeks to 42 weeks of gestation for normal pregnancy according to gestational age for 3rd, 10th, 50th, 90th and 97th centiles (95% CI).
Data Availability Statement
The data that support the findings of this study are availability from the corresponding author on reasonable request.