Abstract
Brentuximab vedotin (BV) is an antibody-drug conjugate that combines the CD30 monoclonal antibody with the microtubule-disrupting agent, monomethyl auristatin E, which induces apoptosis in the tumor cell upon its release from the conjugate. The safety and efficacy of BV have been assessed in several studies in patients with T- and B-cell lymphomas. This article reviews the currently available data on the distribution of CD30 expression in T- and B-cell lymphomas, as well as the various levels of CD30 positivity cutoff used in the literature. It also analyzes the relationship between CD30 expression levels and the clinical response to BV in clinical trials for both T- and B-cell lymphomas and investigates BV efficacy in patients with low or undetectable levels of CD30 and examines potential mechanisms by which BV exerts its effect on these patients. This review contributes to the growing evidence suggesting that CD30 expression levels do not predict the clinical benefit of BV as the drug demonstrated substantial efficacy in patients across a wide range of CD30 expression levels while suggesting that the antitumor activity was not associated with CD30 expression levels. Furthermore, the potential of BV as a targeted approach along with its mechanism of action is also summarized to explain its key role in the future treatments of lymphomas, especially for CD30-expressing lymphomas.
Keywords: Brentuximab vedotin, CD30 expression, non-Hodgkin lymphomas, T-cell lymphoma, B-cell lymphoma
Introduction
CD30, a transmembrane glycoprotein receptor and the member of tumor necrosis factor receptor superfamily, is rarely expressed on normal cells and is rapidly internalized, making it an ideal therapeutic target for monoclonal antibody therapy in patients with lymphoma with positive CD30 expression. However, naked anti-CD30 antibodies and combinations with bacterial toxins and radioimmunoconjugates were found to have limited efficacy. Nevertheless, continuous innovations on developing antibody-drug conjugates (ADCs) have reignited interest in CD30 as a tumor target in non-Hodgkin’s lymphoma (NHLs).1,2 Brentuximab vedotin (BV) is a potent ADC that targets the CD30 antigen. BV consists of a chimeric immunoglobulin G (IgG1) monoclonal antibody, which specifically targets CD30 antigen and is conjugated to the anti-microtubule agent, monomethyl auristatin E (MMAE) payload moiety.3 After binding to CD30 protein, BV is rapidly internalized and transported to lysosomes where MMAE is released and binds to tubulin, leading to cell cycle arrest and apoptosis.4 In a phase I clinical trial including 45 patients with relapsed or refractory (R/R) CD30-positive lymphomas, treatment with BV as a single agent resulted in an objective response rate (ORR) of 67%.5 In a phase II clinical trial for R/R systemic anaplastic large-cell lymphoma (sALCL), treatment with single-agent BV resulted in an ORR of 86% and a complete response (CR) rate of 57%.6 Based on these results, BV was approved by the United States Food and Drug Administration in 2011 for the treatment of R/R Hodgkin’s lymphoma and anaplastic large cell lymphoma (ALCL).7
CD30 is expressed universally in classical Hodgkin’s lymphoma and sALCL, as well as primary cutaneous anaplastic large-cell lymphoma (pcALCL) and lymphomatoid papulosis.8,9 Variable levels of CD30 expression have been found in other T- and B-cell malignancies, such as diffuse large B-cell lymphoma (DLBCL), primary mediastinal (thymic) large B-cell lymphoma (PMBCL), follicular lymphoma (FL), peripheral T-cell lymphoma not otherwise specified (PTCL-NOS), angioimmunoblastic T-cell lymphoma (AITL), adult T-cell leukemia/lymphoma, extranodal natural killer/T-cell lymphoma, mycosis fungoides (MF), Sézary syndrome (SS), and primary cutaneous gamma delta T-cell lymphoma.10
Based on the available clinical data on treatment with BV in NHL, in this article we summarize the association between the CD30 expression level and the clinical response to BV in NHL reported until now.
CD30 Expression in T- and B-Cell Lymphomas
Various cutoff values of CD30 expression levels, ranging from >0%–≥80% of tumor cells, have been reported in different studies reporting of T-cell lymphomas that reflect the lack of consensus on using a standard cutoff (Table 1). A previous study conducted in 319 patients with T-/NK-cell lymphomas showed that CD30 expression ranges from 0–64% in various types of T-cell lymphomas, including AITL and PTCL-NOS.11 The positivity of CD30 expression in ALCL is generally defined as expression of tumor cell ≥75%1 (Table 1).
Table 1.
CD30 Expression Rates in T-Cell Lymphomas Reported in Different Studies
| T-Cells | |||||||||||
| Study | Number of Patients | Treatment | CD30+ % Cell Cutoff Used | Percentage (%) of Patients With CD30 Positive Expression | |||||||
| PTCL-NOS | AITL | ATLL | ENKTL | ALK–ALCL | ALK+ALCL | EATL | CTCL | ||||
| Kawamoto et al 201839 | 97 | BV monotherapy | ≥1% | – | – | – | 57 | – | – | – | – |
| ≥10% | – | – | – | 55 | – | – | – | – | |||
| ≥20% | – | – | – | 44 | – | – | – | – | |||
| Lamarque et al 201640 | 46* | BV monotherapy | <5% | 10 | 0 | 100 | – | 0 | 0 | 0 | 6 |
| 5%–24% | 10 | 100 | 0 | – | 0 | 0 | 0 | 4 | |||
| 25%–49% | 30 | 0 | 0 | – | 0 | 20 | 0 | 6 | |||
| 50%–75% | 30 | 0 | 0 | – | 0 | 20 | 1 | 6 | |||
| >75% | 20 | 0 | 0 | – | 100 | 60 | 0 | 24 | |||
| Weisenburger et al 201141 | 217 | A | >20% | 32 | – | – | – | – | – | – | – |
| Li et al 201442 | 96 | EPOCH or GELOX | >10% | – | – | – | 31 | – | – | – | – |
| Karube et al 200811 | 319 | – | >70% | 5 | 0 | 15 | 0 | 58 | – | 9 | |
| 20%–70% | 11 | 32 | 24 | 64 | 35 | – | 9 | ||||
| Savage et al 200843 | 490 | – | >80% | 4.5 | – | – | – | 100 | 100- | – | – |
| <80% | 32 | – | – | – | – | – | |||||
| Asano et al 201144 | 47 | – | 51 | – | – | – | – | – | – | – | |
| Sabattini et al 20139 | 192 | – | 0: no staining | 36 | 51 | – | 20 | – | – | 0 | 41 (MF); 0 (t-MF) |
| 1+: >0%–<25% | 13 | 21 | – | 10 | – | – | 0 | 47 (MF) | |||
| 2+: 25%–50% | 21 | 12 | – | 30 | – | – | 22 | 6 (MF); 33 (t-MF) | |||
| 3+: >50%–75% | 13 | 10 | – | 10 | – | – | 0 | 0 (MF); 67 (t-MF); | |||
| 4+: >75% | 18 | 0 | – | 30 | – | – | 78 | 6 (MF); 0 (t-MF) | |||
| Bossard et al 201445 | 376 | – | 0: <5% | 42 | 37 | 44 | 54 | 0 | 0 | 50 | – |
| 1+: 5%–24% | 26 | 47 | 11 | 7 | 0 | 0 | 0 | – | |||
| 2+: 25%–49% | 9 | 10 | 33 | 11 | 0 | 5 | 0 | – | |||
| 3+: 50%–75% | 10 | 5 | 11 | 14 | 0 | 2 | 7 | – | |||
| 4+: >75% | 13 | 0 | 0 | 14 | 100 | 93 | 43 | – | |||
| Wang et al 201746 | 122 | – | 0: no staining | – | – | – | 30 | – | – | – | – |
| 1+: >0%–<25% | – | – | – | 38 | – | – | – | – | |||
| 2+: 25%–50% | – | – | – | 18 | – | – | – | – | |||
| 3+: >50%–75% | – | – | – | 10 | – | – | – | – | |||
| 4+: >75% | – | – | – | 5 | – | – | – | – | |||
| B-Cells | |||||||||||
| Study | Number of Patients | Treatment | CD30+ % Cell Cutoff Used | Percentage (%) of Patients With CD30 Positive Expression | |||||||
| DLBCL | Other B-cell lymphomas | ||||||||||
| Jacobsen et al 201518 | 68 | BV monotherapy | ≥1% | 25 | 48 | ||||||
| Malysz et al 201647 | 116 | BV monotherapy | ≥11% | 38 | DLBCL/FL grade 3, FL grade 3, PMLBCL, PTLD, LBCL: 46 | ||||||
| Slack et al 201448 | 308 | R-CHOP | >0% | 24 | – | ||||||
| ≥20% | 11 | – | |||||||||
| Ok et al 201449 | 732 | R-CHOP | ≥20% | 16 | – | ||||||
| Ok et al 201550 | 533 | R-CHOP | ≥20% | 14 | – | ||||||
| Hao et al 201512 | 146 | CHOP or R-CHOP | ≥20% | 16 | – | ||||||
| Song et al 201551 | 230 | EPOCH, R-CHOP, and CHOP | 36 (EBV+) | – | |||||||
| Gong et al 201552 | 232 | R-CHOP and CHOP | >0% | 12 | – | ||||||
| ≥20% | 10 | – | |||||||||
| Xu-Monette et al 201553 | 879 | R-CHOP | 17 | – | |||||||
| Wang et al 201654 | 98 | R-CHOP R+EPOCH, and doxorubicin or hyper-CVAD |
>0% | 41 | – | ||||||
| ≥20% | 22 | – | |||||||||
| ≥40% | 12 | – | |||||||||
| ≥85% | 6 | – | |||||||||
| Hu et al 201355 | 461 | R-CHOP | ≥20% | 14 | – | ||||||
| Collie et al 201356 | 94 | R-CHOP | ≥ 20% | 10 | – | ||||||
| Xu-Monette et al 201657 | 520 | R-CHOP | 15 | – | |||||||
| Xu et al 201758 | 97 | R-EPOCH | >0% | 25 | – | ||||||
| ≥20% | 12 | – | |||||||||
| Tracy et al 201859 | 362 | R-CHOP and R-CEPP | ≥20% | 9 | – | ||||||
| Gong et al 201860 | 241 | CHOP; R-CHOP |
>0%; ≥20% |
10; 8 |
– | ||||||
| Salas et al 202061 | 216 | R-CHOP | >0% | 31 | – | ||||||
| >20% | 19 | ||||||||||
| Santoso et al 202013 | 104 | – | >0% | 13 | – | ||||||
| >20% | 2 | ||||||||||
| Al-Humood et al 201362 | 44 | – | 34 | – | |||||||
| Campuzano-Zuluaga et al 201363 | 167 | – | ≥20% | 21 | – | ||||||
| 20%–49% | 16 | – | |||||||||
| 50%–79% | 32 | – | |||||||||
| ≥80% | 52 | – | |||||||||
Note: *The values are presented in numbers and not in % under different subtypes.
Abbreviations: AITL, angioimmunoblastic T-cell lymphoma; ALCL, anaplastic large-cell lymphoma; ALK–/+, anaplastic lymphoma kinase negative/positive; ATLL, adult T-cell leukemia/lymphoma; BV, brentuximab vedotin; CD30+, CD30 positive; CTCL, cutaneous T-cell lymphoma; CEPP, cyclophosphamide, etoposide, procarbazine, and prednisone; CVAD, hyper-fractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone;R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone; DLBCL, diffuse large B-cell lymphoma; EATL, enteropathy-associated T-cell lymphoma; ENKTL, extranodal natural killer/T-cell lymphoma; EPOCH, etoposide, doxorubicin, vincristine, cyclophosphamide, and prednisone;; FL, follicular lymphoma; GELOX, gemcitabine, oxaliplatin, and L-asparaginase; A, anthracycline; MF, mycosis fungoides; t-MF, transformed mycosis fungoides; PTCL-NOS, peripheral T-cell lymphoma unless otherwise specified, PMBCL, primary mediastinal large B-cell lymphoma; PTLD, post-transplant lymphoproliferative disorder; R-EPOCH, etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin; R-CEPP, etoposide and procarbazine substituting for doxorubicin and vincristine.
Studies have shown that CD30 expression in DLBCL may vary from 9.5%–40%, which could be attributed to various factors such as nonstandardized immunohistochemistry (IHC) staining methods, different methods of CD30 estimation, evaluation of results, differences in demographic factors, and variations in handling strategies of the samples.12,13 Table 1 displays the various cutoff values used in DLBCL, along with the corresponding percentages of patients who tested positive for CD30 expression. Furthermore, it has been demonstrated that evaluating the CD30 expression using a >0% cutoff in DLBCL cases is significantly influenced by factors within the tumor microenvironment.13
Response to BV Treatment with CD30 Expression ≥10% in T- and B-Cell Lymphomas
In a study involving patients with MF, BV improved ORR of 57% in patients with CD30 ≥10% compared with methotrexate or bexarotene (10.3%).14 In another study by Kim et al,15 which explored the efficacy of BV in patients with MF and SS, patients with 10–50% and >50% of CD30 expression levels reported an ORR of 79% and 100%, respectively. In the phase III ALCANZA study, patients with R/R CTCL having CD30 expression >10% and treated with BV showed a high response rate of 56%.16 Similarly, in the phase III ECHELON-2 study, patients with PTCL having CD30 expression levels ≥10% showed a prolonged progression-free survival (PFS) of 48.2 months with BV treatment combined with chemotherapy17 (Table 2).
Table 2.
BV and BV Combination Treatment Responses in T-Cell Lymphomas
| Study | Phase | Treatment | Line of Treatment | Disease Subtype | Patient Number | CD30+ % Cutoff Used | Response Rate (Overall) | Survival Outcomes (Overall) | % of Patients With Different CD30+ Levels | Response Rates | Survival Outcomes | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Younes et al 20105 | 1 | BV monotherapy | R/R | ALCL | 2 | – | ORR: 100% | CR: 100% | – | – | – | – | ||
| Pro et al 201764 | 2 | BV monotherapy | R/R | ALCL | 58 | – | ORR: 86% | CR: 66% | PR: 21% | mPFS: 20 months | mOS: NR | – | – | – |
| Song et al 202165 | 2 | BV monotherapy | R/R | sALCL | 9 | – | ORR: 67% | CR: 56% | PR: 11% | mOS: NR | – | – | – | |
| Pro et al 20126 | 2 | BV monotherapy | Recurrent disease | sALCL | 58 | ORR: 86% | CR: 57% | PR: 29% | mPFS 13.3 months |
mOS: NR | – | – | – | |
| Ogura et al 201466 | 2 | BV monotherapy | R/R | sALCL | 5 | – | ORR: 100% | CR: 80% | PR: 20% | mPFS: 10.8 months | – | – | – | |
| Kim et al 2021 (ALCANZA sub analysis)14 | 3 | BV monotherapy | R/R | MF | 100 | ≥10% | – | – | <10%*: 43% | ORR4: 41% | mPFS: 16.7 months | |||
| ≥10%*: 57% | ORR4: 57% | mPFS: 15.5 months | ||||||||||||
| Horwitz et al 201467 | 2 | BV monotherapy | R/R | PTCL-NOS | 21 | ORR: 41% | CR: 24% | PR: 18% | mPFS: 1.6 months | ≤15%: 74% | ORR: 33% CR: 14% PR: 19% |
– | ||
| AITL | 13 | mPFS: 6.7 months | ORR: 54% CR: 38% PR: 15% |
|||||||||||
| Duvic et al 201523 | 2 | BV monotherapy | Refractory | MF | 28 | – | ORR: 54% | CR: 7% | PR: 46% | PFS: 1.1 year | ≤10%: 36% | ORR: 50% (PR, n = 5) | – | |
| 10%–50%: 43% | ORR: 58% (CR, n = 1, PR, n = 6) | |||||||||||||
| >50%: 21% | ORR: 50% (CR, n = 1, PR, n = 2) | |||||||||||||
| LyP | 9 | ORR: 100% | CR: 56% | PR: 44% | – | – | – | – | ||||||
| pcALCL | 2 | ORR: 100% | CR: 100% | – | – | – | ||||||||
| Kim et al 201515 | 2 | BV monotherapy | Refractory or Advanced | MF and SS | 32 (30 for efficacy) | – | ORR: 70% | CR: 3% | PR: 67% | mPFS: NR at 12 months | <10%: 44% 10%–50%: 44% >50%:13% |
ORR: 54% ORR: 79% ORR:100% |
||
| Horwitz et al, 2022 (ECHELON-2)25 | 3 | BV + chemotherapy (A+CHP) | FL | PTCL-NOS | 89 | ≥10% | CD30 > median (25%): CR: 57% PR: 14% CD30 ≤ median: CR: 71% PR: 14% CD30 = 10%: CR: 67% PR: 33% |
mPFS: 62.3 months for A+CHP 23.8 months for CHOP mOS: not reached in either arm |
>50%: 13% | ORR: 100% | ||||
| AITL | 37 | CD30 > median (18%): CR: 57% PR: 7% CD30 ≤ median: CR: 53% PR: 20% CD30 = 10%: CR: 63% PR: 0% |
||||||||||||
| Horwitz et al 2019 (ECHELON-2)17 | 3 | BV + chemotherapy (A+CHP) |
FL | PTCLs | 226 | ≥10% | ORR: 83% | CR: 68% | PR: 15% | mPFS: 48.2 months | ||||
| chemotherapy (CHOP) |
226 | ORR: 72% | CR: 56% | PR: 16% | mPFS 20.8 months | |||||||||
| Prince et al 2017 (ALCANZA)16 | 3 | BV or physician’s choice (oral methotrexate or oral bexarotene) | R/R | MF | 97 | ≥10% | ORR4: 50% or 10% ORR: 65% or 16% CR: 10% or 0 |
mPFS: 15.9 or 3.5 months | ||||||
| pcALCL | 31 | ORR4: 75% or 20% ORR: 75% or 33% CR: 31% or 7% |
mPFS: 27.5 or 5.3 months | |||||||||||
Note: *CD30min <10% and CD30min ≥10%.
Abbreviations: A+CHP, brentuximab vedotin, cyclophosphamide, doxorubicin, and prednisone; ALCL, anaplastic large cell lymphoma; AITL, angioimmunoblastic T-cell lymphoma; CD30min, CD30 minimum; CD30+, CD30 positive; BV, brentuximab vedotin; CHOP, cyclophosphamide, doxorubicin, vincristine, and prednisone; CR, complete response; F/L, front-line; LyP, lymphomatoid papulosis; MF, mycosis fungoides; NR, not reached; ORR, overall response rate; ORR4, objective response lasting at least 4 months; OS, overall survival; pcALCL, primary cutaneous anaplastic large-cell lymphoma; PFS, progression-free survival; PTCL, peripheral T-cell lymphoma; PTCL-NOS, peripheral T-cell lymphoma not otherwise specified; PR, partial response; R, rituximab; R/R, relapsed/refractory; SS, Sézary syndrome.
Similar results were also observed in B-cell lymphomas (Table 3). In PMBCL, a B-cell lymphoma subtype that typically expresses high levels of CD30, the response rate to BV treatment was 13%–17%.18,19 In another clinical study SGN35-012, patients with B-cell NHL with CD30 expression ≥10% and treated with BV monotherapy reported an ORR of 36% (17/47; 95% confidence interval [CI], 22.7–51.5%).20
Table 3.
BV and BV Combination Treatment Responses in B-Cell Lymphomas
| Study | Phase | Treatment | Line of Treatment | Disease Subtype | Patient Number | CD30+ % Cutoff Used | Response Rate (Overall) | Survival Outcomes (Overall) | % of Patients with Different CD30+ Levels | Response Rates | Survival Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Jacobsen et al 201518 | 2 | BV monotherapy | R/R | DLBCL | 49 | ≥1% | ORR: 44% CR: 17% PR: 27% |
mPFS: 4 months | – | ||
| Gray zone lymphoma | 6 | ORR: 26% CR: 16% PR: 11% |
– | ≥1%: 100% | ORR: 50% CR: 17% PR: 33% SD: 33% |
– | |||||
| PMBL | 6 | – | ≥1%: | ORR: 17% CR: 17%SD: 50% |
– | ||||||
| FL | 3 | – | ≥1%: 100% | ORR: 0% SD: 67% |
– | ||||||
| PTLD | 3 | – | ≥1%: 100% | ORR: 33% CR: 33% |
– | ||||||
| Plasmablastic lymphoma | 1 | – | – | ORR: 0% | – | ||||||
| Bartlett et al 201768 | 2 | BV monotherapy | R/R | DLBCL | 52 | CD30 undetectable | ORR: 31% CR: 12% PR: 19% |
mPFS: 1.4 months mOS: 7.5 months |
– | – | – |
| Zinzani et al 201719 | 2 | BV monotherapy | R/R | PMBL | 15 | Not mentioned, but all patients were CD30+ | ORR: 13.3% | – | – | – | – |
| Yasenchak et al 201469 | 2 | BV monotherapy | F/L | DLBCL | 33 | ≥1%- | ORR: 92% CR: 58% PR: 33% |
– | – | – | – |
| Gandhi et al 201470 | 1/2 | BV + R | F/L | B-cell lymphoma | 7 | CR: 71% | – | – | – | – | |
| Ward et al 202271 | 1 | BV + L | R/R | DLBCL | 37 | No cutoff used | ORR: 57% CR: 35% PR: 22% |
mPFS: 10.2 months mOS: 14.3 months |
CD30+ >1%: 41% | ORR: 73% CR: 40% |
– |
| Yasenchak et al 201524 | 2 | BV + R-CHOP | F/L | DLBCL | 51 | ≥1% | CR: 69% | – | <1% (CD30-) | CR: 63% | PFS rate: 56% |
| ≥1% | CR: 76% | PFS rate: 82% | |||||||||
Abbreviations: BV, brentuximab vedotin; CD30+, CD30 positive; CR, complete response; DLBCL, diffuse large B-cell lymphoma; F/L, front-line; L, lenalidomide; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PTCL, peripheral T-cell lymphoma; PTLD, Post-transplant lymphoproliferative disorder; PR, partial response; R, rituximab; R-CHOP, rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone; PMBL, Primary mediastinal B-cell lymphoma; FL, follicular lymphoma; mOS, median overall survival; mPFS, median progression-free survival; R/R, relapsed/refractory; SD, stable disease.
Response to BV Treatment with Undetectable or Low CD30 Expression in T- and B-Cell Lymphomas
Several studies have shown that BV has single-agent activity in ALCL as well as NHLs with very low CD30 expression or undetectable CD30.15,18,21 A phase II study by Jacobsen assessing the efficacy and safety of BV in patients with B-cell lymphomas with variable CD30 expression (≥1%) reported an ORR of 44% (including CR rate of 17%) in patients with DLBCL and an ORR of 26% in other B-cell lymphomas18 (Table 3). In another study, SGN35-012, B-cell NHL treated with BV monotherapy demonstrated an ORR of 36% in patients the CD30 <10% (24/66; 95% confidence interval [CI], 24.9%–49.1%, p = 0.983). Additionally, an ORR of 34% (17/50; 95% CI, 21.2–48.8%) was also reported in patients with B-cell NHL with an undetectable CD30 expression.20 Furthermore, a phase II study by Bartlett et al has shown a response rate of 27% in patients with DLBCL with undetectable CD30 expression levels.22
In SGN35-012 study of PTCL, the ORR was reported to be 33% (4/12; 95% CI, 9.9–65.1%) in patients with CD30 <10%. In the same study, the ORR was 33% (2/6; 95% CI, 4.3–77.7%) in patients with PTCL with an undetectable CD30 expression.20 The ALCANZA study reported an ORR of 43% (6/14; 95% CI, 17.7–71.1%) in patients with MF and pcALCL and CD30average (CD30avg)<10%. In addition, ORR lasting for four months was reported to be 56·3% (36/64 patients) in the CD30 positive patients.16 In a study involving patients with CD30 positive (CD30+) CTCL and lymphoplasmacytic lymphoma treated with BV, a response rate of 50% was observed in patients with CD30 expression levels <10%.23 Furthermore, a study of BV in SS and MF with variable CD30 levels showed an ORR of 54% in patients with CD30 levels <10%.15 In another study, 35-IST-002, 12 patients with MF were reported to have undetectable CD30, and partial responses (PRs) were achieved in 8 patients with an ORR of 67% (95% CI, 34.9–90.1%).20
Correlation Between BV Treatment Response and CD30 Expression in T- and B-Cell Lymphomas
Considering that CD30 is its molecular target, theoretically, the level of CD30 expression should influence the efficacy of BV. However, the published literature suggests otherwise, as evidenced from the results of various studies conducted in patients with T- and B-cell lymphomas (Tables 2 and 3).
In 2015, Jacobsen et al, investigating the efficacy and safety of BV in R/R mature T- and B-cell lymphomas with a variable CD30 expression in a phase II, open-label, multicenter study reported no correlation between the BV therapy response and level of CD30 expression in patients with R/R DLBCL. In that study, 50% (n = 8) of patients achieving CR had a low level of CD30 (<10%).18 However, patients with R/R DLBCL and other B-cell lymphomas with CD30 >1% and treated with BV showed a CR of 50% and CR + PR of 21% in patients with <10% CD30 levels.18 A study conducted by Yasenchak et al further supported these findings, demonstrating a CR of 63% in patients with DLBCL who had CD30 <1%.24 Furthermore, a study by Jagadeesh et al, which assessed the response to BV based on the CD30 expression across 5 different clinical studies in NHL, demonstrated no significant differences in the overall response between patients with CD30 expression ≥10% and <10%, indicating that CD30 expression does not predict the clinical response to BV.20 In SGN35-012 B-cell NHL, SGN35-012 PTCL, ALCANZA (avg), ALCANZA (min), 35-IST-001, 35-IST-002 (avg) and 35-IST-002 (min) treated with BV monotherapy, the ORR was 36% (17/47), 45% (10/22), 72% (26/36), 71% (20/28), 55% (11/20), 80% (8/10) and 80% (4/5) respectively in the CD30avg≥ 10% group. Similarly, 36% (24/66), 33% (4/12), 43% (6/14), 55% (12/22), 55% (11/20), 59% (13/22) and 63% (17/27) in the CD30avg<10%. In the CD30min group, the ORR was 34% (17/50) in SGN35-012 B-cell NHL, 40% (4/10) in ALCANZA (min), and 67% (8/12) in 35-IST-002 (min) treated with BV monotherapy.
Among one of these studies, Phase III ALCANZA study involving patients with MF, characterized patients as CD30min <10% (1 biopsy with <10% CD30 expression) or CD30min ≥10% (all biopsies with ≥10% CD30 expression). Compared with the physician’s choice of treatment, BV improved the ORR and PFS in both patient groups regardless of baseline CD30 expression levels, indicating no correlation between the CD30 expression level and BV treatment response.14,20 Similarly, in the ECHELON-2 study in patients with PTC, no correlation was observed between the median CD30 expression level (of 18%) or above or below the median level and treatment response with BV plus cyclophosphamide, doxorubicin, and prednisone as responses were observed across CD30 expression levels. The median CD30 expression was found to be 18% in AITL and 25% in PTCL-NOS. However, there appeared to be no correlation between the level of CD30 expression, when evaluated as above or below the median for the subtype, and the probability of achieving CR in patients with either AITL or PTCL-NOS.25 The ECHELON-1 Phase-III trial showed that BV in combination with chemotherapeutic is robust and durable in improving the PFS with a consistent safety profile. Treatment with BV in combination with other chemotherapeutic drugs resulted in a 5-year PFS of 82.2% (95% CI 79·0–85·0) at the follow-up period of 60.9 months with only peripheral neuropathy (19%) as major AE.26
A recent long-term study demonstrated that patients with R/R PMBCL with a varied range of CD30 expression were able to achieve a CR or PR when treated with a combination of BV and nivolumab.27 In addition, an ongoing recent trial has demonstrated that BV combined with cyclophosphamide, doxorubicin, and prednisone was effective in patients with PTCL regardless of CD30 expression, with an ORR of 83% in CD30 negative patients (CD30 expression <1%) and 74% in patients with low levels of CD30 (CD30 expression 1–10%).28 Furthermore, a study by Duvic et al23 had shown similar rates of ORR in both low and high CD30-expressing patients with MF.
Overall, the expression levels of CD30 did not appear to exert an impact on the response to the BV treatment, indicating a lack of direct correlation between the CD30 expression levels and treatment response. To better guide the clinical treatment, no threshold should be used, and pathologists should report the CD30 expression based on what is observed at any staining intensity, estimate the percent positive expression in tumor cells or total cells, and report in number or range expression with any expression level.10
Potential Mechanisms of Action of BV in Patients with All Levels of CD30 Expression or Not Visually Detectable CD30 Expression
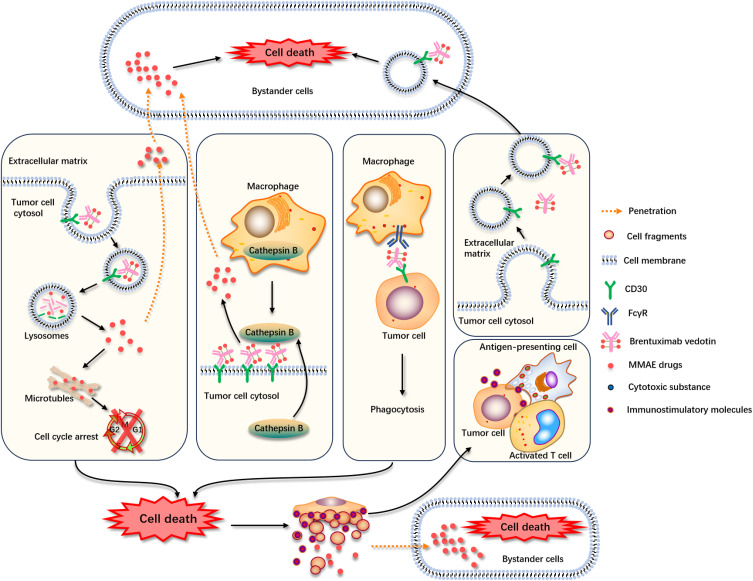
Various studies have shown that the clinical responses/outcomes to the treatment with BV were not associated with CD30 expression levels in various CD30-expressing lymphomas. These findings suggest the presence of some off-target immune modulatory mechanism of BV action, other than the standard ADC internalization, and MMAE release followed by apoptosis of the tumor cells.4 Several hypotheses have been proposed to explain the observable clinical response to BV, independent of the CD30 expression level. The foremost hypothesis is the bystander effect of BV where MMAE crosses the cell membrane and is released into the surrounding extracellular matrix (ECM), exerting its cytotoxic activity on adjacent tumor cells (Figure 1).29 The bystander effect occurs when the payload is released from the cell that has been internalized and degraded the ADC.30,31 Another proposed mechanism for the bystander effect of BV is the cleavage of MMAE from CD30+ bound to BV on tumor cells by extracellular enzymes such as cathepsin B released by surrounding tumor cells and tumor-associated macrophages, thereby releasing MMAE in ECM and killing the tumor cells in the vicinity. Furthermore, targeted tumor cells can undergo antibody-dependent cellular phagocytosis releasing free MMAE in ECM that can induce apoptosis in the surrounding tumor cells.29,31–33 Another proposed mechanism is the depletion of regulatory T cells by BV and induction of immunogenic cell death.27,34 All these alternative mechanisms of BV action do not require CD30 antigen on all targeted cells. Moreover, it has been demonstrated in vitro that CD30-negative cells cultured in isolation are not sensitive to BV. However, when they were cocultured with CD30-expressing cell lines and treated with BV, cell death was observed in the CD30-negative cells.15,20,30,31 One study also suggested that CD30-based BV not only targets CD30+ tumor cells but also CD30– DLBCL cells in the presence of CD30+ extracellular vesicles (EVs). This study provides a feasible explanation for the clinical impact of BV on CD30– DLBCL and warrants confirmatory studies in animal models.35
Figure 1.
Proposed alternative mechanisms of action for BV with all levels of CD30 expression. Binding of BV to the CD30 antigen leading to its internalization via endocytosis. BV is transferred to the lysosomes where the dissociation of the linker occurs resulting in the release of free diffusible MMAE drug. This free drug is released into the cytosol where it binds to microtubules and at the cellular level induces cell cycle arrest and ultimately leads to cell death. MMAE can penetrate surrounding bystander cells after diffusing out of the target cell and cause cell death. There is an alternative route where BV binds to CD30 but does not get internalized. Before internalization process an extracellular enzyme such as cathepsin B, released by tumor cells and tumor-associated macrophages (TAMs) leads to generate diffusible MMAE. Therefore, MMAE can penetrate surrounding bystander cells after diffusing out of the target cell and cause cell death. Also, there is another potential drug delivery path where the BV-bound target tumor cell, internalized through Fc-mediated phagocytosis which involves recognition and engulfment and upon degradation of the target tumor cell, it would lead to the release of free and diffusible drug. BV may also stimulate immune effector cell recruitment via MMAE-mediated immunogenic cell death. CD30 is also released in large EVs (I-EVs) by budding from the surface and in small EVs (s-EVs) by plasma membrane fusion of multivesicular endosomes. CD30+ EVs bind BV and target difference cells within the tumor microenvironment, such as CD30+ or CD30- DLBCL cells.
Abbreviations: BV, Brentuximab vedotin; MMAE, monomethyl auristatin E; TAMS, tumor associated macrophages; EV, extracellular vesicles.
Several factors may contribute to the observation that a response to BV can occur in patients with not visually detectable CD30 expression. The first factor involved is the standard immunohistochemistry (IHC), which is a semiquantitative assay, may exhibit a limited sensitivity, particularly for lower CD30 levels crucial for therapeutic decisions. Tests designed for diagnostic calibration may not identify clinically significant lower expression levels. Technical challenges also add complexities to the accuracy of IHC. Nevertheless, standard IHC remains to be appropriate for characterizing CD30-expressing malignancies, holding promise for optimization as a predictive biomarker to guide the treatment.36 Another potentially significant factor involved is the intra- and inter-lesional heterogeneity of CD30 expression in tumor samples. CD30 expression within a single tumor can vary, and a random biopsy sample could be misrepresentative of the overall CD30 expression status of the patient’s lymphoma. In the ALCANZA study, inter-lesional variability was prominent among the screened patients with MF, 44% (55 of 125) of patients had demonstrated at least 1 biopsy with low (<10%) or undetectable CD30 in lymphoid cells. This increases the possibility that these patients may not have been eligible for recruitment if only a single biopsy had been required.14,20 Furthermore, another assessment revealed inter- and intra-patient variability in CD30 levels, with several patients demonstrating large differences in CD30 expression between biopsies, ranging from 0%–70%. Therefore, if the inclusion criteria for the patients are based on the results of a single biopsy, then most of the patients would be excluded from the studies and remain devoid of treatment with BV or any other CD30-targeted treatment.20
The heterogeneity observed in biopsy tissue can be influenced by the dynamic nature of CD30 expression, involving protein cleavage leading to soluble CD30 expressions in the blood.20,37,38 Increased soluble CD30 levels observed in inflammatory and oncologic parameters may be associated with tumor burden. The dynamic partitioning between surface-bound and secreted CD30 forms in tumors implies that the surface levels of CD30 are not constant, complicating the assessment of CD30 expression at a single time point.20
Future Prospects
Surface expression of CD30 receptors has been recognized as a clinically potential target to treat cancers, especially lymphomas. CD30-targeted therapies, such as BV, have demonstrated significant efficacy, highlighting the potential for personalized treatment strategies. Previously, it was considered that BV could provide clinical benefits only in NHLs with substantial CD30 expression. However, this review adds to the growing evidence that treatment with BV benefits patients with NHL, with all levels of CD30 expression or undetectable CD30 expression. For therapeutic decision-making, the protocol of CD30 tests used in clinical IHC laboratory should be optimized to guarantee that it should be capable of detecting clinically important low levels of CD30. In addition, numerous studies have used more sensitive methods than IHC to assess the CD30 expression. In several studies, computer-assisted digital image analysis revealed CD30 expression of tumors, which is classified as undetectable by IHC. Furthermore, multispectral image analysis identified CD30 expression in biopsies with negligible IHC-detected CD30 expression. With more standardization of these new methods, they can also become more widely used in the future. On the other hand, artificial intelligence-assisted interpretation software is also encouraged to be developed for better quantification of CD30 expression levels, especially at low expression levels.
Acknowledgments
Medical writing support for the development of this narrative review, under the direction of the authors, was provided by Mehar Naseem, PhD and Kusuma Kumari G, PhD of Indegene Private Limited, Bangalore, India and funded by Takeda (China) International Trading Co., Ltd. The mechanism diagram (Figure 1) was drawn by Rumeng Bai, MD of Department of Pathology, the First Affiliated Hospital of Nanjing Medical University. Takeda reviewed the manuscript. This narrative review complies with the Good Publication Practice 2022 guidelines. Medical writing and the journal’s Open Access Fee is being funded by Takeda (China) International Trading Co., Ltd., an affiliation of Takeda Pharmaceutical company. Medical review was supported by Takeda.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.van der Weyden CA, Pileri SA, Feldman AL, Whisstock J, Prince HM. Understanding CD30 biology and therapeutic targeting: a historical perspective providing insight into future directions. Blood Cancer J. 2017;7(9):e603–e603. doi: 10.1038/bcj.2017.85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schirrmann T, Steinwand M, Wezler X, Ten Haaf A, Tur MK, Barth S. CD30 as a therapeutic target for lymphoma. BioDrugs Clin Immunother Biopharm Gene Ther. 2014;28(2):181–209. doi: 10.1007/s40259-013-0068-8 [DOI] [PubMed] [Google Scholar]
- 3.Yi JH, Kim SJ, Kim WS. Brentuximab vedotin: clinical updates and practical guidance. Blood Res. 2017;52(4):243–253. doi: 10.5045/br.2017.52.4.243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van de Donk NWCJ, Dhimolea E. Brentuximab vedotin. mAbs. 2012;4(4):458–465. doi: 10.4161/mabs.20230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Younes A, Bartlett NL, Leonard JP, et al. Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N Engl J Med. 2010;363(19):1812–1821. doi: 10.1056/NEJMoa1002965 [DOI] [PubMed] [Google Scholar]
- 6.Pro B, Advani R, Brice P, et al. Brentuximab vedotin (SGN-35) in patients with relapsed or refractory systemic anaplastic large-cell lymphoma: results of a Phase II study. J Clin Oncol off J Am Soc Clin Oncol. 2012;30(18):2190–2196. doi: 10.1200/JCO.2011.38.0402 [DOI] [PubMed] [Google Scholar]
- 7.Kim HK, Moon SM, Moon JH, Park JE, Byeon S, Kim WS. Complete remission in CD30-positive refractory extranodal NK/T-cell lymphoma with brentuximab vedotin. Blood Res. 2015;50(4):254–6. doi: 10.5045/br.2015.50.4.254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Oflazoglu E, Grewal IS, Gerber H. Targeting CD30/CD30L in oncology and autoimmune and inflammatory diseases. Adv Exp Med Biol. 2009;647:174–185. doi: 10.1007/978-0-387-89520-8_12 [DOI] [PubMed] [Google Scholar]
- 9.Sabattini E, Pizzi M, Tabanelli V, et al. CD30 expression in peripheral T-cell lymphomas. Haematologica. 2013;98(8):e81–82. doi: 10.3324/haematol.2013.084913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gru AA, Lim MS, Dogan A, et al. Best practices in CD30 immunohistochemistry testing, interpretation, and reporting: an expert panel consensus. Arch Pathol Lab Med. 2023;147(1):79–86. doi: 10.5858/arpa.2021-0270-OA [DOI] [PubMed] [Google Scholar]
- 11.Karube K, Aoki R, Nomura Y, et al. Usefulness of flow cytometry for differential diagnosis of precursor and peripheral T-cell and NK‐cell lymphomas: analysis of 490 cases. Pathol Int. 2008;58(2):89–97. doi: 10.1111/j.1440-1827.2007.02195.x [DOI] [PubMed] [Google Scholar]
- 12.Hao X, Wei X, Huang F, et al. The expression of CD30 based on immunohistochemistry predicts inferior outcome in patients with diffuse large B-cell lymphoma. PLoS One. 2015;10(5):e0126615. doi: 10.1371/journal.pone.0126615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Santoso MR, Hardianti MS, Indrawati I, Anggorowati N. CD30 expression and its correlation with clinicopathologic features in Indonesian diffuse large B-cell lymphoma. Asian Pac J Cancer Biol. 2020;5(3):107–113. doi: 10.31557/apjcb.2020.5.3.107-113 [DOI] [Google Scholar]
- 14.Kim YH, Prince HM, Whittaker S, et al. Response to brentuximab vedotin versus physician’s choice by CD30 expression and large cell transformation status in patients with mycosis fungoides: an ALCANZA sub-analysis. Eur J Cancer Oxf Engl. 2021;148:411–421. doi: 10.1016/j.ejca.2021.01.054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim YH, Tavallaee M, Sundram U, et al. Phase II investigator-initiated study of Brentuximab vedotin in mycosis fungoides and Sézary syndrome with variable CD30 expression level: a multi-institution collaborative project. J Clin Oncol off J Am Soc Clin Oncol. 2015;33(32):3750–3758. doi: 10.1200/JCO.2014.60.3969 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Prince HM, Kim YH, Horwitz SM, et al. Brentuximab vedotin or physician’s choice in CD30-positive cutaneous T-cell lymphoma (ALCANZA): an international, open-label, randomised, Phase 3, multicentre trial. Lancet. 2017;390(10094):555–566. doi: 10.1016/S0140-6736(17)31266-7 [DOI] [PubMed] [Google Scholar]
- 17.Horwitz S, O’Connor OA, Pro B, et al. Brentuximab vedotin with chemotherapy for CD30-positive peripheral T-cell lymphoma (ECHELON-2): a global, double-blind, randomised, phase 3 trial. Lancet. 2019;393(10168):229–240. doi: 10.1016/S0140-6736(18)32984-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jacobsen ED, Sharman JP, Oki Y, et al. Brentuximab vedotin demonstrates objective responses in a Phase 2 study of relapsed/refractory DLBCL with variable CD30 expression. Blood. 2015;125(9):1394–1402. doi: 10.1182/blood-2014-09-598763 [DOI] [PubMed] [Google Scholar]
- 19.Zinzani PL, Pellegrini C, Chiappella A, et al. Brentuximab vedotin in relapsed primary mediastinal large B-cell lymphoma: results from a phase 2 clinical trial. Blood. 2017;129(16):2328–2330. doi: 10.1182/blood-2017-01-764258 [DOI] [PubMed] [Google Scholar]
- 20.Jagadeesh D, Horwitz S, Bartlett NL, et al. Response to Brentuximab vedotin by CD30 expression in Non-Hodgkin lymphoma. Oncologist. 2022;27(10):864–873. doi: 10.1093/oncolo/oyac137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kim WY, Nam SJ, Kim S, et al. Prognostic implications of CD30 expression in extranodal natural killer/T-cell lymphoma according to treatment modalities. Leuk Lymphoma. 2015;56(6):1778–1786. doi: 10.3109/10428194.2014.974048 [DOI] [PubMed] [Google Scholar]
- 22.Bartlett NL, Smith MR, Siddiqi T, et al. Brentuximab vedotin activity in diffuse large B-cell lymphoma with CD30 undetectable by visual assessment of conventional immunohistochemistry. Leuk Lymphoma. 2017;58(7):1607–1616. doi: 10.1080/10428194.2016.1256481 [DOI] [PubMed] [Google Scholar]
- 23.Duvic M, Tetzlaff MT, Gangar P, Clos AL, Sui D, Talpur R. Results of a phase II trial of Brentuximab vedotin for CD30+ cutaneous T-cell lymphoma and lymphomatoid papulosis. J Clin Oncol off J Am Soc Clin Oncol. 2015;33(32):3759–3765. doi: 10.1200/JCO.2014.60.3787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yasenchak CA, Farber CM, Budde LE, et al. Brentuximab vedotin in combination with RCHOP as front-line therapy in patients with DLBCL: interim results from a phase 2 study. Blood. 2014;124(21):1745. doi: 10.1182/blood.V124.21.1745.1745 [DOI] [Google Scholar]
- 25.Horwitz S, O’Connor OA, Pro B, et al. The ECHELON-2 trial: 5-year results of a randomized, phase III study of brentuximab vedotin with chemotherapy for CD30-positive peripheral T-cell lymphoma. Ann Oncol. 2022;33(3):288–298. doi: 10.1016/j.annonc.2021.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Straus DJ, Długosz-Danecka M, Connors JM, et al. Brentuximab vedotin with chemotherapy for stage III or IV classical Hodgkin lymphoma (ECHELON-1): 5-year update of an international, open-label, randomised, phase 3 trial. Lancet Haematol. 2021;8(6):e410–e421. doi: 10.1016/S2352-3026(21)00102-2 [DOI] [PubMed] [Google Scholar]
- 27.Zinzani PL, Santoro A, Gritti G, et al. Nivolumab combined with Brentuximab vedotin for relapsed/refractory primary mediastinal large B-Cell lymphoma: efficacy and safety from the phase II CHECKMATE 436 study. J Clin Oncol off J Am Soc Clin Oncol. 2019;37(33):3081–3089. doi: 10.1200/JCO.19.01492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Iyer S, Jagadeesh D, Domingo Domènech E, et al. P1130: frontline brentuximab vedotin and CHP (A+ CHP) in patients with peripheral T-cell lymphoma with less than 10% CD30 expression: initial safety and efficacy results from the phase 2 study SGN35-032. HemaSphere. 2023;7(Suppl):e4008899. doi: 10.1097/01.HS9.0000971416.40088.99 [DOI] [Google Scholar]
- 29.Staudacher AH, Brown MP. Antibody drug conjugates and bystander killing: is antigen-dependent internalisation required? Br J Cancer. 2017;117(12):1736–1742. doi: 10.1038/bjc.2017.367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Okeley NM, Miyamoto JB, Zhang X, et al. Intracellular activation of SGN-35, a potent anti-CD30 antibody-drug conjugate. Clin Cancer Res off J Am Assoc Cancer Res. 2010;16(3):888–897. doi: 10.1158/1078-0432.CCR-09-2069 [DOI] [PubMed] [Google Scholar]
- 31.Li F, Emmerton KK, Jonas M, et al. Intracellular released payload influences potency and bystander-killing effects of antibody-drug conjugates in preclinical models. Cancer Res. 2016;76(9):2710–2719. doi: 10.1158/0008-5472.CAN-15-1795 [DOI] [PubMed] [Google Scholar]
- 32.Karube K, Kakimoto Y, Tonozuka Y, Ohshima K. The expression of CD30 and its clinico-pathologic significance in peripheral T-cell lymphomas. Expert Rev Hematol. 2021;14(8):777–787. doi: 10.1080/17474086.2021.1955344 [DOI] [PubMed] [Google Scholar]
- 33.Oflazoglu E, Stone IJ, Gordon KA, et al. Macrophages contribute to the antitumor activity of the anti-CD30 antibody SGN-30. Blood. 2007;110(13):4370–4372. doi: 10.1182/blood-2007-06-097014 [DOI] [PubMed] [Google Scholar]
- 34.Gardai SJ, Epp A, Law CL. Abstract 2469: brentuximab vedotin-mediated immunogenic cell death. Cancer Res. 2015;75(15_Supplement):2469. doi: 10.1158/1538-7445.AM2015-2469 [DOI] [Google Scholar]
- 35.Lobastova L, Lettau M, Babatz F, et al. CD30-positive extracellular vesicles enable the targeting of CD30-negative DLBCL cells by the CD30 antibody-drug conjugate Brentuximab vedotin. Front Cell Dev Biol. 2021;9:698503. doi: 10.3389/fcell.2021.698503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Onaindia A, Martínez N, Montes-Moreno S, et al. CD30 expression by B and T cells: a frequent finding in angioimmunoblastic T-Cell lymphoma and peripheral T-Cell lymphoma-not otherwise specified. Am J Surg Pathol. 2016;40(3):378–385. doi: 10.1097/PAS.0000000000000571 [DOI] [PubMed] [Google Scholar]
- 37.Horn-Lohrens O, Tiemann M, Lange H, et al. Shedding of the soluble form of CD30 from the Hodgkin-analogous cell line L540 is strongly inhibited by a new CD30-specific antibody (Ki-4). Int J Cancer. 1995;60(4):539–544. doi: 10.1002/ijc.2910600419 [DOI] [PubMed] [Google Scholar]
- 38.Hansen HP, Recke A, Reineke U, et al. The ectodomain shedding of CD30 is specifically regulated by peptide motifs in its cysteine-rich domains 2 and 5. FASEB J off Publ Fed Am Soc Exp Biol. 2004;18(7):893–895. doi: 10.1096/fj.03-0901fje [DOI] [PubMed] [Google Scholar]
- 39.Kawamoto K, Miyoshi H, Suzuki T, et al. Frequent expression of CD30 in extranodal NK/T-cell lymphoma: potential therapeutic target for anti-CD30 antibody-based therapy. Hematol Oncol. 2018;36(1):166–173. doi: 10.1002/hon.2482 [DOI] [PubMed] [Google Scholar]
- 40.Lamarque M, Bossard C, Contejean A, et al. Brentuximab vedotin in refractory or relapsed peripheral T-cell lymphomas: the French named patient program experience in 56 patients. Haematologica. 2016;101(3):e103–106. doi: 10.3324/haematol.2015.135400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Weisenburger DD, Savage KJ, Harris NL, et al. Peripheral T-cell lymphoma, not otherwise specified: a report of 340 cases from the international peripheral T-cell lymphoma project. Blood. 2011;117(12):3402–3408. doi: 10.1182/blood-2010-09-310342 [DOI] [PubMed] [Google Scholar]
- 42.Li P, Jiang L, Zhang X, Liu J, Wang H. CD30 expression is a novel prognostic indicator in extranodal natural killer/T-cell lymphoma, nasal type. BMC Cancer. 2014;14(1):890. doi: 10.1186/1471-2407-14-890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Savage KJ, Harris NL, Vose JM, et al. ALK- anaplastic large-cell lymphoma is clinically and immunophenotypically different from both ALK+ ALCL and peripheral T-cell lymphoma, not otherwise specified: report from the international peripheral T-cell lymphoma project. Blood. 2008;111(12):5496–5504. doi: 10.1182/blood-2008-01-134270 [DOI] [PubMed] [Google Scholar]
- 44.Asano N, Kinoshita T, Tamaru JI, et al. Cytotoxic molecule-positive classical Hodgkin’s lymphoma: a clinicopathological comparison with cytotoxic molecule-positive peripheral T-cell lymphoma of not otherwise specified type. Haematologica. 2011;96(11):1636–1643. doi: 10.3324/haematol.2011.041079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bossard C, Dobay MP, Parrens M, et al. Immunohistochemistry as a valuable tool to assess CD30 expression in peripheral T-cell lymphomas: high correlation with mRNA levels. Blood. 2014;124(19):2983–2986. doi: 10.1182/blood-2014-07-584953 [DOI] [PubMed] [Google Scholar]
- 46.Wang GN, Zhao WG, Li L, et al. Prognostic significance of CD30 expression in nasal natural killer/T-cell lymphoma. Oncol Lett. 2017;13(3):1211–1215. doi: 10.3892/ol.2017.5592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Malysz J, Erdman P, Klapper J, Zhu J, Creer M, Bayerl MG. Clinical implications of CD30 expression in aggressive B-cell lymphomas. Clin Lymphoma Myeloma Leuk. 2016;16(8):429–433. doi: 10.1016/j.clml.2016.04.011 [DOI] [PubMed] [Google Scholar]
- 48.Slack GW, Steidl C, Sehn LH, Gascoyne RD. CD30 expression in de novo diffuse large B-cell lymphoma: a population-based study from British Columbia. Br J Haematol. 2014;167(5):608–17. doi: 10.1111/bjh.13085 [DOI] [PubMed] [Google Scholar]
- 49.Ok CY, Li L, Xu-Monette ZY, et al. Prevalence and clinical implications of epstein-barr virus infection in de novo diffuse large B-cell lymphoma in Western countries. Clin Cancer Res. 2014;20(9):2338–49. doi: 10.1158/1078-0432.CCR-13-3157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ok CY, Xu-Monette ZY, Li L, et al. Evaluation of NF-κB subunit expression and signaling pathway activation demonstrates that p52 expression confers better outcome in germinal center B-cell-like diffuse large B-cell lymphoma in association with CD30 and BCL2 functions. Mod Pathol. 2015;28(9):1202–1213. doi: 10.1038/modpathol.2015.76 [DOI] [PubMed] [Google Scholar]
- 51.Song CG, Huang JJ, Li YJ, et al. Epstein-Barr virus-positive diffuse large B-cell lymphoma in the elderly: a matched case-control analysis. PLoS One. 2015;10(7):e0133973. doi: 10.1371/journal.pone.0133973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gong QX, Lu TX, Liu C, et al. Prevalence and clinicopathologic features of CD30-positive de novo diffuse large B-cell lymphoma in Chinese patients: a retrospective study of 232 cases. Int J Clin Exp Pathol. 2015;8(12):15825–15835. [PMC free article] [PubMed] [Google Scholar]
- 53.Xu-Monette ZY, Tu M, Jabbar KJ, et al. Clinical and biological significance of de novo CD5+ diffuse large B-cell lymphoma in Western countries. Oncotarget. 2015;6(8):5615–5633. doi: 10.18632/oncotarget.3479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang XJ, Seegmiller AC, Reddy NM, Li S. CD30 expression and its correlation with MYC rearrangement in de novo diffuse large B-cell lymphoma. Eur J Haematol. 2016;97(1):39–47. doi: 10.1111/ejh.12680 [DOI] [PubMed] [Google Scholar]
- 55.Hu S, Xu-Monette ZY, Balasubramanyam A, et al. CD30 expression defines a novel subgroup of diffuse large B-cell lymphoma with favorable prognosis and distinct gene expression signature: a report from the International DLBCL Rituximab-CHOP consortium program study. Blood. 2013;121(14):2715–2724. doi: 10.1182/blood-2012-10-461848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Collie AMB, Hill BT, Manilich EA, Smith MR, Hsi ED. CD30 immunohistochemical expression in diffuse large B-Cell lymphoma is associated with decreased overall survival and the non-germinal center molecular subtype. Blood. 2013;122(21):4318. doi: 10.1182/blood.V122.21.4318.4318 [DOI] [Google Scholar]
- 57.Xu-Monette ZY, Zhang S, Li X, et al. p63 expression confers significantly better survival outcomes in high-risk diffuse large B-cell lymphoma and demonstrates p53-like and p53-independent tumor suppressor function. Aging. 2016;8(2):345–365. doi: 10.18632/aging.100898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Xu J, Oki Y, Saksena A, et al. CD30 expression and prognostic significance in R-EPOCH-treated patients with diffuse large B-cell lymphoma. Hum Pathol. 2017;60:160–166. doi: 10.1016/j.humpath.2016.10.009 [DOI] [PubMed] [Google Scholar]
- 59.Tracy SI, Habermann TM, Feldman AL, et al. Outcomes among North American patients with diffuse large B-cell lymphoma are independent of tumor Epstein-Barr virus positivity or immunosuppression. Haematologica. 2018;103(2):297–303. doi: 10.3324/haematol.2017.176511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gong QX, Wang Z, Liu C, et al. CD30 expression and its correlation with MYC and BCL2 in de novo diffuse large B-cell lymphoma. J Clin Pathol. 2018;71(9):795–801. doi: 10.1136/jclinpath-2018-205039 [DOI] [PubMed] [Google Scholar]
- 61.Salas MQ, Climent F, Tapia G, et al. Clinicopathologic features and prognostic significance of CD30 expression in de novo diffuse large B-cell lymphoma (DLBCL): results in a homogeneous series from a single institution. Biomarkers. 2020;25(1). doi: 10.1080/1354750X.2019.1691656 [DOI] [PubMed] [Google Scholar]
- 62.Al-Humood S, Alqallaf A, Al-Shemmari S, Al-Faris L, Al-Ayadhy B. Genetic and immunohistochemical characterization of Epstein-Barr virus-associated diffuse large B-cell lymphoma. Acta Haematol. 2014;131(1):1–10. doi: 10.1159/000350493 [DOI] [PubMed] [Google Scholar]
- 63.Campuzano-Zuluaga G, Cioffi-Lavina M, Lossos IS, Chapman-Fredricks JR. Frequency and extent of CD30 expression in diffuse large B-cell lymphoma and its relation to clinical and biologic factors: a retrospective study of 167 cases. Leuk Lymphoma. 2013;54(11):2405–2411. doi: 10.3109/10428194.2013.778407 [DOI] [PubMed] [Google Scholar]
- 64.Pro B, Advani R, Brice P, et al. Five-year results of brentuximab vedotin in patients with relapsed or refractory systemic anaplastic large cell lymphoma. Blood. 2017;130(25):2709–2717. doi: 10.1182/blood-2017-05-780049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Song Y, Guo Y, Huang H, et al. Phase II single-arm study of brentuximab vedotin in Chinese patients with relapsed/refractory classical Hodgkin lymphoma or systemic anaplastic large cell lymphoma. Expert Rev Hematol. 2021;14(9):867–875. doi: 10.1080/17474086.2021.1942831 [DOI] [PubMed] [Google Scholar]
- 66.Ogura M, Tobinai K, Hatake K, et al. Phase I / II study of brentuximab vedotin in Japanese patients with relapsed or refractory CD30-positive Hodgkin’s lymphoma or systemic anaplastic large-cell lymphoma. Cancer Sci. 2014;105(7):840–846. doi: 10.1111/cas.12435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Horwitz SM, Advani RH, Bartlett NL, et al. Objective responses in relapsed T-cell lymphomas with single-agent brentuximab vedotin. Blood. 2014;123(20):3095–3100. doi: 10.1182/blood-2013-12-542142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bartlett NL, Smith MR, Advani R, et al. Brentuximab vedotin monotherapy in DLBCL patients with undetectable CD30: preliminary results from a phase 2 study. Blood. 2014;124(21):629. doi: 10.1182/blood.V124.21.629.629 [DOI] [Google Scholar]
- 69.Yasenchak CA, Halwani A, Advani R, et al. Brentuximab vedotin with RCHOP as frontline therapy in patients with high-intermediate/high-risk Diffuse Large B Cell Lymphoma (DLBCL): results from an ongoing phase 2 study. Blood. 2015;126(23):814. doi: 10.1182/blood.V126.23.814.814 [DOI] [Google Scholar]
- 70.Gandhi M, Ma S, Smith SM, et al. Brentuximab Vedotin (BV) Plus Rituximab (R) as frontline therapy for Patients (Pts) with Epstein Barr Virus (EBV)+ and/or CD30+ lymphoma: phase I results of an ongoing phase I-II study. Blood. 2014;124(21):3096. doi: 10.1182/blood.V124.21.3096.3096 [DOI] [Google Scholar]
- 71.Ward JP, Berrien-Elliott MM, Gomez F, et al. Phase 1/dose expansion trial of brentuximab vedotin and lenalidomide in relapsed or refractory diffuse large B-cell lymphoma. Blood. 2022;139(13):1999–2010. doi: 10.1182/blood.2021011894 [DOI] [PMC free article] [PubMed] [Google Scholar]