Abstract
Background
The Tnfrh1 gene (gene symbol Tnfrsf23) is located near one end of a megabase-scale imprinted region on mouse distal chromosome 7, about 350 kb distant from the nearest known imprinting control element. Within 20 kb of Tnfrh1 is a related gene called Tnfrh2 (Tnfrsf22) These duplicated genes encode putative decoy receptors in the tumor necrosis factor (TNF) receptor family. Although other genes in this chromosomal region show conserved synteny with genes on human Chr11p15.5, there are no obvious human orthologues of Tnfrh1 or Tnfrh2.
Results
We analyzed Tnfrh1 for evidence of parental imprinting, and characterized its tissue-specific expression. Tnfrh1 mRNA is detectable in multiple adult and fetal tissues, with highest expression in placenta, where in situ hybridization reveals a distinctive population of Tnfrh1-positive cells in maternal decidua, directly beneath the trophoblast giant cells. In offspring of interspecific mouse crosses, Tnfrh1 shows a consistent parent-of-origin-dependent allelic expression bias, with relative repression, but not silencing, of the paternal allele in several organs including fetal liver and adult spleen.
Conclusions
Genes preferentially expressed in the placenta are predicted to evolve rapidly, and Tnfrh1 appears to be an example of this phenomenon. In view of its strong expression in cells at the fetal-maternal boundary, Tnfrh1 warrants further study as a gene that might modulate immune or trophic interactions between the invasive placental trophoblast and the maternal decidua. The preferential expression of Tnfrh1 from the maternal allele indicates weak functional imprinting of this locus in some tissues.
Background
A well-studied megabase-scale region of DNA on distal mouse chromosome 7 (Chr7) contains multiple genes subject to parental imprinting. Nearly all genes in this region show conserved synteny with genes on human Chr11p15.5. The extended imprinted region appears to have a bipartite structure in that it contains at least two separate imprinted sub-domains. Each of these subdomains is regulated by a distinct imprinting control element. These correspond to short differentially methylated DNA sequences (DMRs) – one immediately upstream of the H19 gene and another within an intron of the Kcnq1 gene. These two elements control, respectively, the allele-specific expression of the H19/Igf2/Ins2 gene cluster, and the second gene cluster containing Kcnq1, the antisense transcriptKcnq1ot1, p57Kip2/Cdkn1c, Slc22a1l, Ipl/Tssc3 and possibly additional genes [1,2]. There is good evidence assigning one border of the overall imprinted region to the DNA immediately downstream of H19 [3]. However, the other border, which must lie upstream of Ipl is less well-defined [4]. Here we show evidence for weak imprinting of a gene, Tnfrh1, which is located upstream of Ipl in mice, and which encodes a putative decoy receptor in the TNF receptor family. We also map the tissue distribution of Tnfrh1 mRNA and discuss the significance of our findings in terms of possible functions of this gene in placentation.
Results and Discussion
Maps of the chromosomal region containing Tnfrh1 and Tnfrh2 and the exons of these genes are shown in Figure 1a,1b. These two genes have a similar intron/exon organization, suggesting that they arose by a local gene duplication [4]. They are also 95 percent identical in their cDNA sequences, with 84 percent overall amino acid identity (Fig.2). The proteins encoded by Tnfrh1 and Tnfrh2 share a conserved arrangement of multiple cysteine residues, characteristic of the extracellular ligand-binding domains of other TNF receptor family members (Fig.2). Tnfrh1 and Tnfrh2 lie ~350 kb distant from the KvDMR1 'imprinting center'. Two other genes, Cars and Nap1l4, lie between the strongly imprinted gene cluster regulated by this DMR and the Tnfrh1/Tnfrh2 gene pair. The Cars gene is not functionally imprinted, and the Nap1l4 gene shows a weak expression bias in some mouse tissues, but not in several human tissues examined to date [4,5]. The Obph1 gene, which shows a strong allelic bias in mRNA expression in the placenta, but not in other tissues, is located upstream of Tnfrh1. Since assessment of allelic bias in the placenta is complicated by the presence of maternal cells, it is not known with certainty whether Obph1 is imprinted. Searches of the genomic and expressed sequence databases using the Tnfrh1 nucleotide and protein sequences did not uncover any strongly similar genes in humans, and low stringency hybridization of Northern blots of human tissue RNAs with the Tnfrh1 cDNA probe did not yield specific bands. Moreover, in the interval of the human genome between CARS and the human orthologue of Obph1 (the OSBPL5 gene), there were no sequences with detectable similarity to Tnfrh1.
Figure 1.
Genes and PCR strategies.A, Map showing the positions of genes in the telomeric half of the distal mouse Chr7 imprinted domain. Direction of transcription is shown by the arrows, and imprinting is color-coded, with red indicating preferential expression of the maternal allele and blue indicating expression of the paternal allele. The Obph1 gene is coded as orange, since it shows an allelic expression bias only in the placenta, which may indicate either imprinting or expression from maternal cells. Genes that do not show an allelic expression bias, or for which complete information is not available, are in black. The KvDMR1 differentially methylated sequence, that acts as an imprinting control element and gives rise to the Kcnq1ot1 non-translated RNA, is indicated by the green bar. B, Structure of the Tnfrh1 and Tnfrh2 genes. PCR primers (arrows) are shown above the exons (rectangles). The yellow and orange shading highlights the non-conserved first exons. The last exons are highly conserved, and the remaining exons show intermediate conservation. Genetic polymorphisms in Tnfrh1 are indicated in violet (polymorphisms that distinguish CAST from BL/6) or maroon (polymorphisms that distinguish MOLD from BL/6). For analysis of allele-specific mRNA expression, we used primer pairs (1,2) or, alternatively (1,3). Estimation of relative expression from Tnfrh1 vs. Tnfrh2 by RT-PCR was done with primers 1, 4 and 5. Primers 1 and 6 were used to generate a Tnfrh1-specific cDNA probe.
Figure 2.
Amino acid sequence similarity between mouse Tnfrh1 and Tnfrh2 proteins and the extracellular domains of the prototypical TNF receptor family members TNFR and Fas/CD95. Amino acids with complete conservation are shaded, and the conserved cysteine residues are in bold. The sequence of TNFR is from F. catus and the sequence of Fas is from S. scrofa. Proteins from these species were chosen for this illustration because they yielded the most significant BLAST scores in alignments with Tnfrh1 and Tnfrh2.
Previously, Engemann et al. described Tnfrh1 as having ubiquitous expression [4]. Consistent with this, northern blotting using Tnfrh1-specific probes matching the relatively divergent 5' end of this gene showed easily detectable Tnfrh1 mRNA transcripts in nearly all adult tissues, as well as in late fetal organs and structures (Fig.3). However, by far the strongest signals were seen in the pregnant uterus and whole placenta (Fig.3). Measurements by PhosphorImaging showed that Tnfrh1 mRNA in whole placenta was 5-fold more abundant than in the non-pregnant uterus, and 10-fold more abundant than in the whole fetus. Two classes of transcripts were present: the smaller transcript, which migrated at 1.7 kb based on other blots with size standards, matches the size predicted from the cDNA sequence. The larger transcript, which migrates at 3.7 kb, must contain additional sequences, but these have not yet been mapped. To evaluate the relative expression of Tnfrh1 and Tnfrh2 in various organs and tissues, we performed reverse transcription polymerase chain reaction (RT-PCR) in multiplex reactions using a shared downstream primer and Tnfrh1- and Tnfrh2-specific upstream primers. This showed that both genes are expressed in most fetal and adult organs and structures, but that the expression in several structures including placenta and muscle is highly skewed in favor of Tnfrh1 (Fig.4).
Figure 3.
Expression of Tnfrh1 in adult and fetal mouse tissues.Tnfrh1 mRNA is detected as two alternative transcripts, both of which are present in most tissues. Both transcripts are most abundant in the placenta. The Tnfrh1 partial cDNA used to probe these blots has no detectable sequence identity with Tnfrh2 over the 5'-most 65 nt, but has 91% identity with the Tnfrh2 over the remaining 450 nt. Stripping and rehybridization at high stringency, with a smaller 5' probe with 72% overall sequence identity over 297 nt gave an identical pattern of bands. Ethidium bromide (EtBr) staining of ribosomal RNA is a loading control. The lanes contain 20 micrograms of total RNA; blots were exposed overnight.
Figure 4.
Tissue-specific ratios of Tnfrh1 to Tnfrh2 expression. cDNAs were amplified using upstream primers specific for the divergent first exons of either Tnfrh1 or Tnfrh2 (Fig.1, primers 1 and 5), with a shared downstream primer (Fig.1, primer 4). The predicted sizes for the PCR products derived from each gene are indicated. Several organs and tissues, including whole placentas at 12.5 and 14.5 dpc (asterisks), show preferential expression of Tnfrh1.
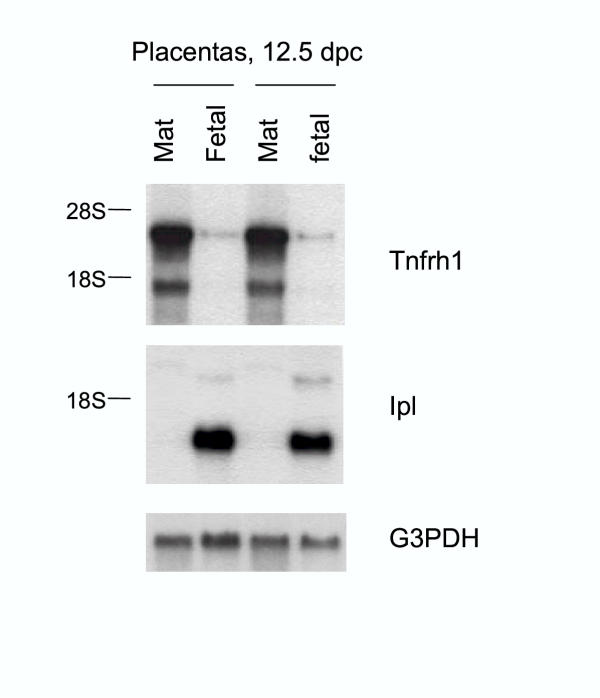
The whole placenta, after dissection from the uterus in mid-gestation, contains both a fetal component (the placenta proper, consisting of the labyrinth, spongiotrophoblast and giant cell layers) and a maternal component (the decidua basalis, as well as maternal blood vessels). We were therefore interested to determine the precise localization of the Tnfrh1-positive cells in this organ. To this end we first dissected each of two frozen placentas, obtained at 12.5 dpc, into a superficial half, enriched in the superficial fetal components, and a deeper half, enriched in deep fetal and superficial maternal components, and extracted RNA from each half. This procedure crudely separates the labyrinthine trophoblast from all of the deeper tissues. Hybridizing the northern blot with a probe for Ipl mRNA (Fig.4) validated this procedure. Ipl is specifically expressed in the placental labyrinth [6,7], and this probe gave a strong signal only in RNA from the superficial halves. The same blot re-hybridized with the Tnfrh1 cDNA probe gave the opposite pattern, clearly showing that Tnfrh1 mRNA is restricted to layers of the placenta deep to the labyrinth (Fig.5).
Figure 5.
Tnfrh1 mRNA is restricted to the deep layers of the placenta. Two placentas from 12.5 dpc conceptuses were frozen and divided into superficial (fetal) and deep (maternal; mat) halves prior to RNA extraction. The Ipl cDNA probe is a specific marker for the labyrinthine trophoblast and therefore hybridizes to the lanes from the superficial halves of the placentas.
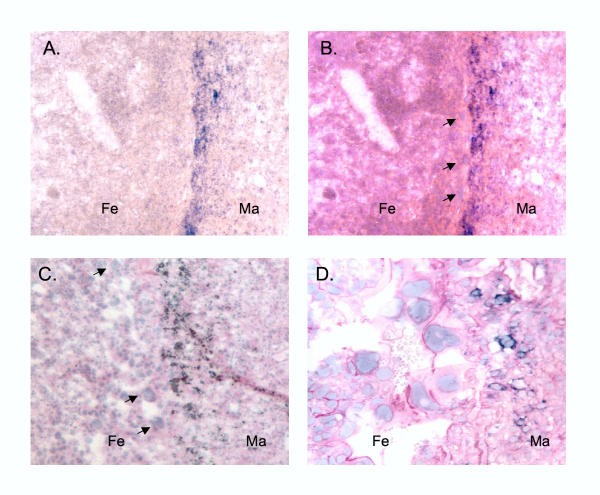
To determine whether Tnfrh1 is expressed in the deep fetal component or, alternatively, in the maternal component of the placenta, we next carried out in situ hybridization with a digoxigenin-labeled Tnfrh1 cDNA probe. As shown in Figure 6a,6b,6c, this gave a detectable signal only in a narrow band of cells, situated immediately deep to the trophoblast giant cell layer. This restricted distribution of Tnfrh1-positive cells was observed both at 10.5 dpc, when the definitive placental layers have recently formed, and at 12.5 dpc, after these layers have begun to mature (Fig.6a,6b,6c). At high magnification, the Tnfrh1-positive cells were seen closely juxtaposed to trophoblast giant cells (Fig.6d). The morphology of these cells, and their location deep to the giant cell layer, suggested decidual parenchyma. These cells did not coincide with PAS-positive granulated uterine natural killer lymphocytes, or with CD3-positive infiltrating T-lymphocytes (data not shown). However, additional characterization will be necessary for a definitive assignment of cell type.
Figure 6.
Expression of Tnfrh1 is restricted to a discrete sub-population of cells near the fetal-maternal boundary.A, low-magnification image of 10.5 dpc placenta subjected to ISH with the Tnfrh1-antisense probe, showing a band of Tnfrh1-positive cells near the fetal-maternal junction. Signal in other areas is not higher than that seen with the sense control probe (not shown). Fe=fetal side of placenta, Ma=maternal side. B, the same section stained with hematoxylin-eosin (HE) to show cellular morphology. The arrows indicate trophoblast giant cells at the fetal-maternal boundary. The in situ image has been superimposed on the HE image, revealing Tnfrh1-positive cells restricted to the zone immediately under the trophoblast giant cells. C, superimposed ISH and HE images obtained from a placenta at 12.5 dpc, showing a distribution of Tnfrh1-positive cells similar to that at the earlier stage. Trophoblast giant cells are indicated by arrows. D, high-magnification field of another section at 12.5 dpc, which was first subjected to ISH and then lightly counterstained with periodic acid Schiff (PAS). The ISH reaction product persists under these conditions, revealing Tnfrh1-positive cells in close proximity to trophoblast giant cells.
The expression of Tnfrh1 only at the fetal-maternal boundary obviously suggests that this gene may play a role in modulating either immune or trophic interactions between the invasive placental trophoblast and the uterine host tissue. Since Tnfrh1 encodes a receptor in the TNFR family lacking a cytoplasmic domain (i.e., a decoy receptor), this gene might function to block the action of TNF-related ligands. A similar scenario has been proposed by Phillips et al. [8,9] for another TNF ligand-receptor system, namely TRAIL and TRAIL receptors, which are expressed in human placentas. However, in their studies the major decoy receptor for TRAIL, DcR1, was found expressed in trophoblast, not in decidua, while another decoy receptor, DcR2, was expressed by placental macrophages. Fas ligand is also highly expressed by human and murine trophoblast, although its functional role in the placenta is unknown [10,11].
To investigate potential imprinting of Tnfrh1 and Tnfrh2, we next searched for genetic polymorphisms in these genes that would allow us to distinguish maternal from paternal alleles in interspecific mouse crosses. We failed to identify polymorphisms in Tnfrh2, but we found multiple single-nucleotide polymorphisms (SNPs) in the Tnfrh1 gene (Table 2). Using cDNAs from fetal and adult tissues derived from interspecific crosses, we amplified a region of Tnfrh1 containing several of these SNPs. The PCR strategies generated products that crossed either 4 or 5 exon-exon boundaries, thereby eliminating the possibility of genomic contamination (Fig.1b). Since one of the SNPs that distinguished the standard C57BL/6 laboratory strain from the divergent strain M. m. castaneus (CAST) created an MboII restriction site in the CAST sequence, we used RFLP analysis to assess allelic representation in cDNAs from F1 progeny of BL/6 × CAST and CAST × BL/6 reciprocal crosses. This indicated an obvious but partial maternal bias in the fetal liver, and a nearly complete bias towards the maternal allele in placenta (Fig.7). This was confirmed by direct sequencing of the cDNAs, and the parent-of-origin dependence of this effect was emphasized by the opposite patterns observed in the reciprocal crosses (Fig.7). The bias in fetal liver must reflect parental imprinting of Tnfrh1, but based on the information from Northern blotting and ISH, the pattern seen in the placental cDNAs in these experiments reflects expression from maternal cells, not imprinting.
Table 2.
Sequence variants in Tnfrh1 exons
| AJ276505 (nt) | Seq. Variant | C57/Bl6 | MOLD | CAST | Exon |
| 31323 | T→C | T | C | T | 1 |
| 37064 | C→A | C | A | C | 3 |
| 37129 | A→G | A | A | G | 3 |
| 46227 | G→C | G | C | G | 4 |
| 46260 | A→G | A | A | G | 4 |
| 47119 | T→A | T | A | nd | 5 |
| 47127 | G→A | G | A | nd | 5 |
Additional variants were found in Tnfrh1 exon 6, but were outside of the PCR products used for the allelic expression assays. Sequencing of PCR products generated from genomic DNAs using outer flanking primers verified that the binding sites for the primers used in the allelic expression assays were free of polymorphisms.
Figure 7.
Preferential expression of the maternal allele of Tnfrh1 in fetal liver from CAST × BL/6 reciprocal crosses.A, Analysis of the MboII RFLP, in which the BL/6 allele is undigested and the CAST allele yields two fragments. There is a strong maternal allele bias in placenta, and a weaker but still obvious parent-of-origin dependent biases in fetal liver. This imprinting effect is superimposed on a non-imprinted bias for higher expression of the BL/6 allele. The allelic bias in placenta reflects expression from maternal cells, but the bias in fetal liver reflects parental imprinting. Similar results were obtained with multiple aliquots of cDNA (not shown). B=BL/6, C=CAST. (Note that only the internal comparisons of alleles in single PCR reactions are valid; the stronger band obtained from the CAST maternal kidney should not be compared to that from the BL/6 maternal kidney for intensity of expression, since these were separate RT-PCR reactions). B, Direct sequencing of RT-PCR products. This shows an allelic expression bias that depends on the direction of the cross, consistent with the RFLP results.
Other fetal and adult tissues were also assessed in these crosses, and showed a maternal expression bias, but weaker than that observed in fetal liver (data not shown). The parent-of-origin dependent allelic bias in Tnfrh1 mRNA was superimposed on a 'baseline' bias towards hyper-expression of the BL/6 allele (Fig.7). This most likely reflects the effects of polymorphisms in regulatory promoter and/or enhancer sequences. Indeed, sequencing of the BL/6 and CAST Tnfrh1 promoter sequences revealed multiple SNPs in the region -200 to +100 relative to the transcriptional start site (data not shown). The alternative trivial explanation for the baseline bias, polymorphisms within primer binding sites, was excluded by direct sequencing of genomic DNAs.
We next repeated the imprinting analysis using cDNAs from reciprocal crosses between BL/6 and another divergent strain, M. m. mollosinus (MOLD). Since the polymorphisms present in MOLD do not affect restriction sites, this experiment was done using SSCP to distinguish the alleles. The results again showed an incomplete bias towards expression of the maternal allele in several fetal and adult tissues, reflecting parental imprinting, as well as a stronger bias in the placenta, reflecting expression from maternal cells (Fig.8). Direct sequencing of cDNAs confirmed the SSCP data (Fig. 8). As was true of the BL/6 × CAST crosses, the parent-of-origin dependence of the allele-specific expression was superimposed on a baseline bias towards hyper-expression of the BL/6 allele. As was true of CAST, the MOLD Tnfrh1 promoter region differed from BL/6 at several SNPs, thereby providing a possible explanation for this constitutive bias (data not shown).
Figure 8.
Preferential expression of the maternal allele of Tnfrh1 in fetal and adult tissues from MOLD × BL/6 reciprocal crosses.A, SSCP analysis of RT-PCR products from the BL/6 and MOLD alleles. The mixing experiment validates this method. There is a bias towards greater expression of the maternal allele in several organs, indicated by a relative increase in the MOLD allele intensity in the MxB lanes compared to the BxM lanes. This is superimposed on a non-imprinted baseline of hyper-expression of the BL/6 allele. The allelic bias in placenta reflects expression from maternal cells, but the parent-of-origin dependent bias in other tissues reflects imprinting. Brackets indicate duplicate or triplicate assays using independent aliquots of cDNA. B=BL/6, M=MOLD. B, Direct sequencing of RT-PCR products. Consistent with the SSCP analysis, this shows an expression bias favoring the maternal allele, which is superimposed on a baseline of hyper-expression of the BL/6 allele.
As a control for these assays, we assessed the allele-specific expression of the Obph1 gene, using the same cDNA preparations. Consistent with the previous report from Engemann et al. [4], this showed equal allelic representation in cDNAs from various fetal and adult tissues, and a strong maternal bias in the placenta (Fig.9). As mentioned above, additional work will be needed to determine whether the results in the placenta reflect imprinting of Obph1, or alternatively indicate expression from maternal cells.
Figure 9.
Lack of allelic expression bias for Obph1 in fetal and adult tissues from MOLD × BL/6 reciprocal crosses.A, Separation of BL/6 and MOLD alleles of Obph1 by SSCP analysis of RT-PCR products. This shows equal biallelic expression in all organs, except for the placenta. B, Confirmation of the SSCP data by direct sequencing of Obph1 cDNAs. An allelic bias is only present in the placenta.
Conclusions
The findings described here include 'leaky' but consistent imprinting of Tnfrh1 in several organs, and high-level expression of this gene in a distinctive population of cells restricted to the interface between the placental trophoblast and the uterine lining. The absence of identifiable orthologues of Tnfrh1 and Tnfrh2 in the human genome suggests that both of these genes arose subsequent to the divergence of placental mammals. This fact, together with our observation of preferential expression of Tnfrh1 mRNA in cells at the fetal-maternal boundary, highlights Tnfrh1 as a potential example of the rapid evolution of genes with functions specific to the placenta, a process postulated to be driven by conflict between fetal and maternal alleles. Anecdotal examples, including pregnancy-associated glycoproteins, trophoblast interferons, the Pem and Psx homeobox genes, and the placental lactogen genes support the notion that 'placental genes' evolve rapidly [12-17], but counter-examples can also be adduced. Two imprinted genes with placenta-specific expression and function, namely Ipl and Mash2 [6,7,18,19] are highly conserved. This contrasts with the lack of conservation of Tnfrh1, and the species-specific imprinting of at least one placental lactogen gene [20]. Of more immediate interest is the biological function of Tnfrh1, which will need to be determined by knockout experiments. Since the sequence of this gene predicts that it encodes a TNF decoy receptor, a likely possibility is that it acts to dampen immune responses to the fetal semi-allograft.
Imprinting of genes on mouse distal Chr7 is controlled by two DMRs, which act as 'imprinting centers'. Of these two control elements, the closest to Tnfrh1 is KvDMR1, located in an intron of the Kcnq1 gene and giving rise to the Kcnq1ot1 non-translated RNA. This element, which is ~350 kb distant from Tnfrh1, is essential in cis for the imprinting of at least 4 genes, Kcnq1, Cdkn1c, Slc22a1l and Ipl/Tssc3 ([21-23] and M. Higgins, personal communication). All of these genes are relatively repressed on the paternal allele and active on the maternal allele. The simplest explanation for our finding of weak but consistent functional imprinting of Tnfrh1, with relative hyper-expression from the maternal allele, is that the KvDMR1 control element exerts distance-dependent effects. However, studies using KvDMR1-mutant mice will be necessary to confirm this.
Materials and Methods
Genomic and cDNA PCR
Trizol™ reagent (Life Technologies, Gaithersburg, MD) was used to prepare total RNA from fetal and adult mouse tissues. cDNA was prepared by reverse transcription of total RNA using the Superscript Preamplification System (Life Technologies). For PCR, 50 l volume reaction mixtures contained 400 ng of genomic DNA, 50 mM KCl, 10 mM Tris pH8.3, 1.5 mM MgCl2, 200 M of each dNTP, 5%DMSO and 0.5 units of Taq DNA polymerase (Roche, Indianapolis, IN). Cycling parameters were an initial denaturation of 94°C for 4 min, followed by 30 cycles of denaturation at 94°C for 1 min, annealing for 45 sec and extension at 72°C for 1 min 30 sec, followed by a final extension of 72°C for 7 min. The annealing temperatures for each primer pair are listed in Table 1. Sequencing was with dye-labeled terminators (ABI 377 Sequencer). Primers for RT-PCR analysis of the Obph1 cDNAs were: OBP-For (gaagctgtggtgtgtactg) and OBP-Rev (cgtctgattcagaagcggc).
Table 1.
PCR primers
| Primer Name | Sequence | Gene, Exon |
| 1 (forward) | gctcacagccatggttacct | Tnfrh1, exon 1 |
| 2 (reverse) | gccagtttctgggatttgaa | Tnfrh1, exons 5/6 |
| 3 (reverse) | agcaccatgtgtgcctccg | Tnfrh1, exon 6 |
| 4 (reverse) | aatgcttgcctcataagaaa | Tnfrh1, exon 6 |
| 5 (forward) | aattacctggatgcttgtat | Tnfrh2, exon 1 |
| 6 (reverse) | gattgtgaactttgctccacctg | Tnfrh1, exon 3 |
SSCP and RFLP analysis
PCR labeling was by incorporation of alpha32P-dCTP, using 10 ng of gel isolated PCR product as a template. Cycling employed an initial denaturation at 94°C for 4 min, followed by 6 cycles of denaturation at 94°C for 1 min, annealing at 59°C for 15 sec and extension at 72°C for 1 min with a final extension at 72°C for 7 min. The radiolabeled PCR products were digested with AluI or HpaII, denatured and electrophoresed for 10–16 h at 500 V on non-denaturing 6% acrylamide gels at room temperature [24]. For RFLP analysis, PCR products were digested with MboII and analyzed by electrophoresis on non-denaturing 6% acrylamide gels at 500 V for 3–4 hours.
Northern blotting
Fractionation of the placenta by dissection into maternal and fetal portions was done as described previously [6]. RNA was extracted using Trizol, and 6–10 micrograms of total RNA was electrophoresed and transferred to nylon membranes. Northern blots containing total RNA from panels of mouse tissues were purchased from SeeGene (Seoul, Korea). The Tnfrh1 cDNA probes (the longer probe made using primers 1 and 2, and matching the first 506 nt of Genbank Acc. AY046550 plus an additional 11 nt at the 5' end; and a smaller probe made with primers 1 and 6 matching the first 297 nt) were labeled with α-32P (Random primers DNA labeling system, Life Technologies). Blots were prehybridized and hybridized at 42°C in ExpressHyb (Clontech, Palo Alto, CA) and washed in 0.1× SSC/1% SDS for one hour at 65°C.
RNA in situ hybridization
Placentas were fixed in 4% paraformaldehyde overnight at 4 C, transferred to 30% sucrose in 0.1 MPB, equilibrated overnight, and then embedded and snap frozen in standard glycerol-based medium (TBS, Durham, NC). After cryo-sectioning, the sections were post-fixed in paraformaldehyde and then subjected to in situ hybridization (ISH) with digoxigenin-labeled probes (Dig RNA Labeling Kit, Roche), followed by alkaline phosphatase-mediated detection. Anti-sense and sense probes for Tnfrh1 were synthesized from cDNA clone Image 1479846 (Genbank AI156311). The sense probe was used as a control and did not produce a signal.
Acknowledgments
Acknowledgements
This work was supported by grants to B.T. from the N.I.H and the Human Frontiers Science Project.
Contributor Information
Lorraine Clark, Email: lc654@columbia.edu.
Michelle Wei, Email: mw380@columbia.edu.
Giorgio Cattoretti, Email: gc87@columbia.edu.
Cathy Mendelsohn, Email: clm20@columbia.edu.
Benjamin Tycko, Email: bt12@columbia.edu.
References
- Maher ER, Reik W. Beckwith-Wiedemann syndrome: imprinting in clusters revisited. J Clin Invest. 2000;105:247–252. doi: 10.1172/JCI9340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson-Smith AC, Surani MA. Imprinting and the epigenetic asymmetry between parental genomes. Science. 2001;293:1086–1089. doi: 10.1126/science.1064020. [DOI] [PubMed] [Google Scholar]
- Yuan L, Qian N, Tycko B. An extended region of biallelic gene expression and rodent-human synteny downstream of the imprinted H19 gene on chromosome 11p15.5. Hum Mol Genet. 1996;5:1931–1937. doi: 10.1093/hmg/5.12.1931. [DOI] [PubMed] [Google Scholar]
- Engemann S, Strodicke M, Paulsen M, Franck O, Reinhardt R, Lane N, Reik W, Walter J. Sequence and functional comparison in the Beckwith-Wiedemann region: implications for a novel imprinting centre and extended imprinting. Hum Mol Genet. 2000;9:2691–2706. doi: 10.1093/hmg/9.18.2691. [DOI] [PubMed] [Google Scholar]
- Hu RJ, Lee MP, Johnson LA, Feinberg AP. A novel human homologue of yeast nucleosome assembly protein, 65 kb centromeric to the p57KIP2 gene, is biallelically expressed in fetal and adult tissues. Hum Mol Genet. 1996;5:1743–1748. doi: 10.1093/hmg/5.11.1743. [DOI] [PubMed] [Google Scholar]
- Frank D, Mendelsohn CL, Ciccone E, Svensson K, Ohlsson R, Tycko B. A novel pleckstrin homology-related gene family defined by Ipl/Tssc3, TDAG51, and Tih1: tissue-specific expression, chromosomal location, and parental imprinting. Mamm Genome. 1999;10:1150–1159. doi: 10.1007/s003359901182. [DOI] [PubMed] [Google Scholar]
- Frank D, Fortino W, Clark L, Musalo R, Wang W, Saxena A, Li C-M, Reik W, Ludwig T, Tycko B. Isolated placental overgrowth in mice lacking the imprinted gene Ipl. Proc Natl Acad Sci U S A. 2002;99:7490–7495. doi: 10.1073/pnas.122039999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phillips TA, Ni J, Hunt JS. Death-inducing tumour necrosis factor (TNF) superfamily ligands and receptors are transcribed in human placentae, cytotrophoblasts, placental macrophages and placental cell lines. Placenta. 2001;22:663–672. doi: 10.1053/plac.2001.0703. [DOI] [PubMed] [Google Scholar]
- Phillips TA, Ni J, Pan G, Ruben SM, Wei YF, Pace JL, Hunt JS. TRAIL (Apo-2L) and TRAIL receptors in human placentas: implications for immune privilege. J Immunol. 1999;162:6053–6059. [PubMed] [Google Scholar]
- Hunt JS, Vassmer D, Ferguson TA, Miller L. Fas ligand is positioned in mouse uterus and placenta to prevent trafficking of activated leukocytes between the mother and the conceptus. J Immunol. 1997;158:4122–4128. [PubMed] [Google Scholar]
- Hammer A, Blaschitz A, Daxbock C, Walcher W, Dohr G. Fas and Fas-ligand are expressed in the uteroplacental unit of first-trimester pregnancy. Am J Reprod Immunol. 1999;41:41–51. doi: 10.1111/j.1600-0897.1999.tb00074.x. [DOI] [PubMed] [Google Scholar]
- Roberts RM, Ealy AD, Alexenko AP, Han CS, Ezashi T. Trophoblast interferons. Placenta. 1999;20:259–264. doi: 10.1053/plac.1998.0381. [DOI] [PubMed] [Google Scholar]
- Hughes AL, Green JA, Garbayo JM, Roberts RM. Adaptive diversification within a large family of recently duplicated, placentally expressed genes. Proc Natl Acad Sci U S A. 2000;97:3319–3323. doi: 10.1073/pnas.050002797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chun JY, Han YJ, Ahn KY. Psx homeobox gene is X-linked and specifically expressed in trophoblast cells of mouse placenta. Dev Dyn. 1999;216:257–266. doi: 10.1002/(SICI)1097-0177(199911)216:3<257::AID-DVDY4>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- Forsyth IA. Comparative aspects of placental lactogens: structure and function. Exp Clin Endocrinol. 1994;102:244–251. doi: 10.1055/s-0029-1211288. [DOI] [PubMed] [Google Scholar]
- Wallis M. Remarkably high rate of molecular evolution of ruminant placental lactogens. J Mol Evol. 1993;37:86–88. doi: 10.1007/BF00170466. [DOI] [PubMed] [Google Scholar]
- Maiti S, Doskow J, Sutton K, Nhim RP, Lawlor DA, Levan K, Lindsey JS, Wilkinson MF. The Pem homeobox gene: rapid evolution of the homeodomain, X chromosomal localization, and expression in reproductive tissue. Genomics. 1996;34:304–316. doi: 10.1006/geno.1996.0291. [DOI] [PubMed] [Google Scholar]
- Guillemot F, Caspary T, Tilghman SM, Copeland NG, Gilbert DJ, Jenkins NA, Anderson DJ, Joyner AL, Rossant J, Nagy A. Genomic imprinting of Mash2, a mouse gene required for trophoblast development. Nat Genet. 1995;9:235–242. doi: 10.1038/ng0395-235. [DOI] [PubMed] [Google Scholar]
- Guillemot F, Nagy A, Auerbach A, Rossant J, Joyner AL. Essential role of Mash-2 in extraembryonic development. Nature. 1994;371:333–336. doi: 10.1038/371333a0. [DOI] [PubMed] [Google Scholar]
- Vrana PB, Matteson PG, Schmidt JV, Ingram RS, Joyce A, Prince KL, Dewey MJ, Tilghman SM. Genomic imprinting of a placental lactogen gene in Peromyscus. Dev Genes Evol. 2001;211:523–532. doi: 10.1007/s00427-001-0188-x. [DOI] [PubMed] [Google Scholar]
- Cleary MA, van Raamsdonk CD, Levorse J, Zheng B, Bradley A, Tilghman SM. Disruption of an imprinted gene cluster by a targeted chromosomal translocation in mice. Nat Genet. 2001;29:78–82. doi: 10.1038/ng715. [DOI] [PubMed] [Google Scholar]
- Smilinich NJ, Day CD, Fitzpatrick GV, Caldwell GM, Lossie AC, Cooper PR, Smallwood AC, Joyce JA, Schofield PN, Reik W, Nicholls RD, Weksberg R, Driscoll DJ, Maher ER, Shows TB, Higgins MJ. A maternally methylated CpG island in KvLQT1 is associated with an antisense paternal transcript and loss of imprinting in Beckwith-Wiedemann syndrome. Proc Natl Acad Sci U S A, 1999;96:8064–8069. doi: 10.1073/pnas.96.14.8064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horike S, Mitsuya K, Meguro M, Kotobuki N, Kashiwagi A, Notsu T, Schulz TC, Shirayoshi Y, Oshimura M. Targeted disruption of the human LIT1 locus defines a putative imprinting control element playing an essential role in Beckwith-Wiedemann syndrome. Hum Mol Genet. 2000;9:2075–2083. doi: 10.1093/hmg/9.14.2075. [DOI] [PubMed] [Google Scholar]
- Orita M, Iwahana H, Kanazawa H, Hayashi K, Sekiya T. Detection of polymorphisms of human DNA by gel electrophoresis as single-strand conformation polymorphisms. Proc Natl Acad Sci U S A. 1989;86:2766–2770. doi: 10.1073/pnas.86.8.2766. [DOI] [PMC free article] [PubMed] [Google Scholar]