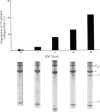
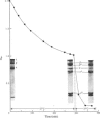
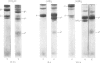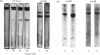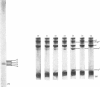Abstract
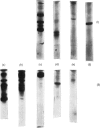
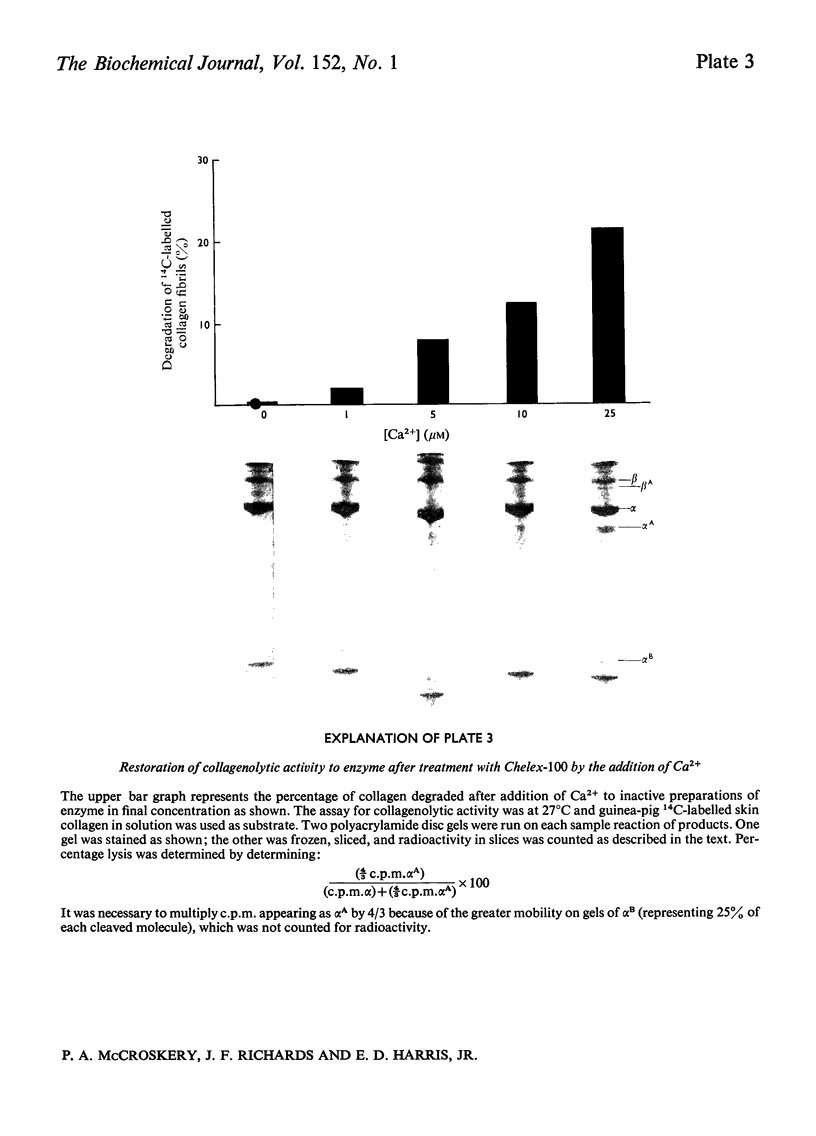
A collagenase was purified from homogenates of V2 ascites-cell carcinoma growing in rabbit muscle. (NH4)2SO4 precipitation, ion-exchange and gel-filtration chromatography, and affinity chromatography (by using the CB7 CNBr) cleavage fragment of alpha 1(I) collagen linked to agarose) gave a 268000-fold purification and a sevenfold increase in total enzyme units recovered. The specific activity, defined as mumol of collagen in solution cleaved/h per mg of enzyme at 35 degrees C, WAS 1.74.2. The collagenase had a broad pH optimum from pH7.0 to 9.5, and a mol.wt. of between 33000 and 35000. It was inhibited by dithiothreitol, L-cysteine, D-penicillamine, EDTA and 1,10-phenanthroline, and by both rabbit and human serum. 3. Removal of cations by a chelating resin (Chelex 100) produced as inactive enzyme that could be reactiviated by the addition of Ca2+ ions at concentrations as low as 1muM. Other bivalent cations were not effective. 4. The purified collagenase cleaved peptides alpha2 and alpha1-CB7 (denatured polypeptides of collagen) at 37 degrees C at one site only. [alpha1 (I)]2alpha2 and [alpha1(III)]3 collagens in solution were cleaved at the same site approximately five times more rapidly than [alpha1 (II)]3. 5. An inhibitor of the enzyme in the tumour extracts, which was dissociable from the enzyme at the (NH4) 2SO4 precipitation step of purification, had a mol. wt. of between 40000 and 50000 but was distinct from the alpha1 trypsin inhibitor. 6. Studies with zonal density-gradient centrifugation suggested that the enzyme was bound to fibrillar substrate (collagen) extracellularly, but that it was not associated with enzymes originating in cell mitochondria, microsomal preparations or lysosomes.
Full text
PDF






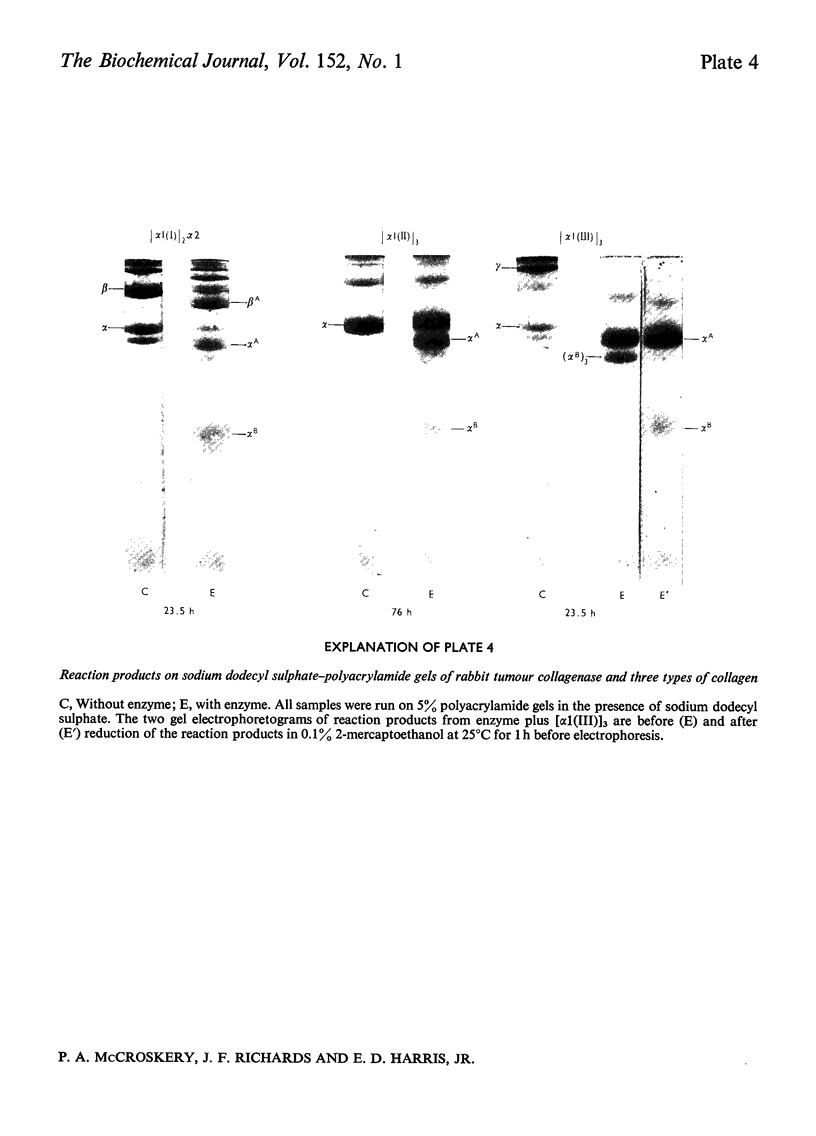
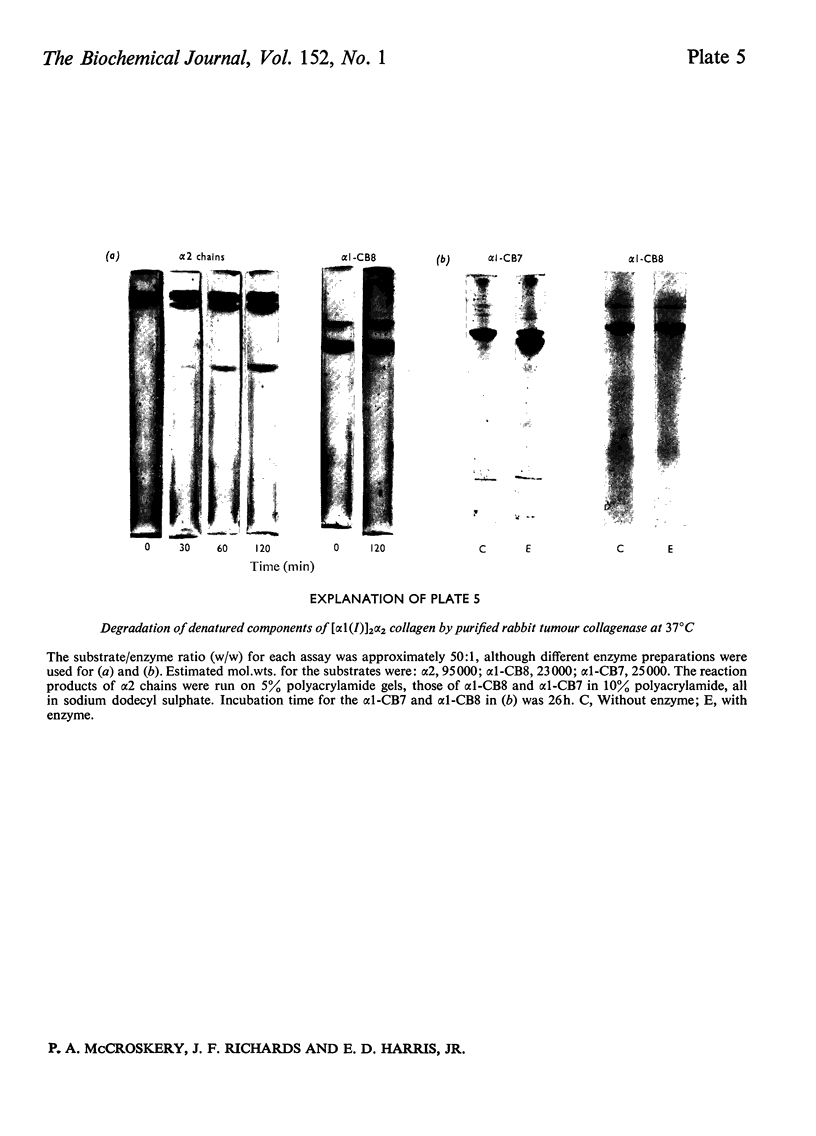
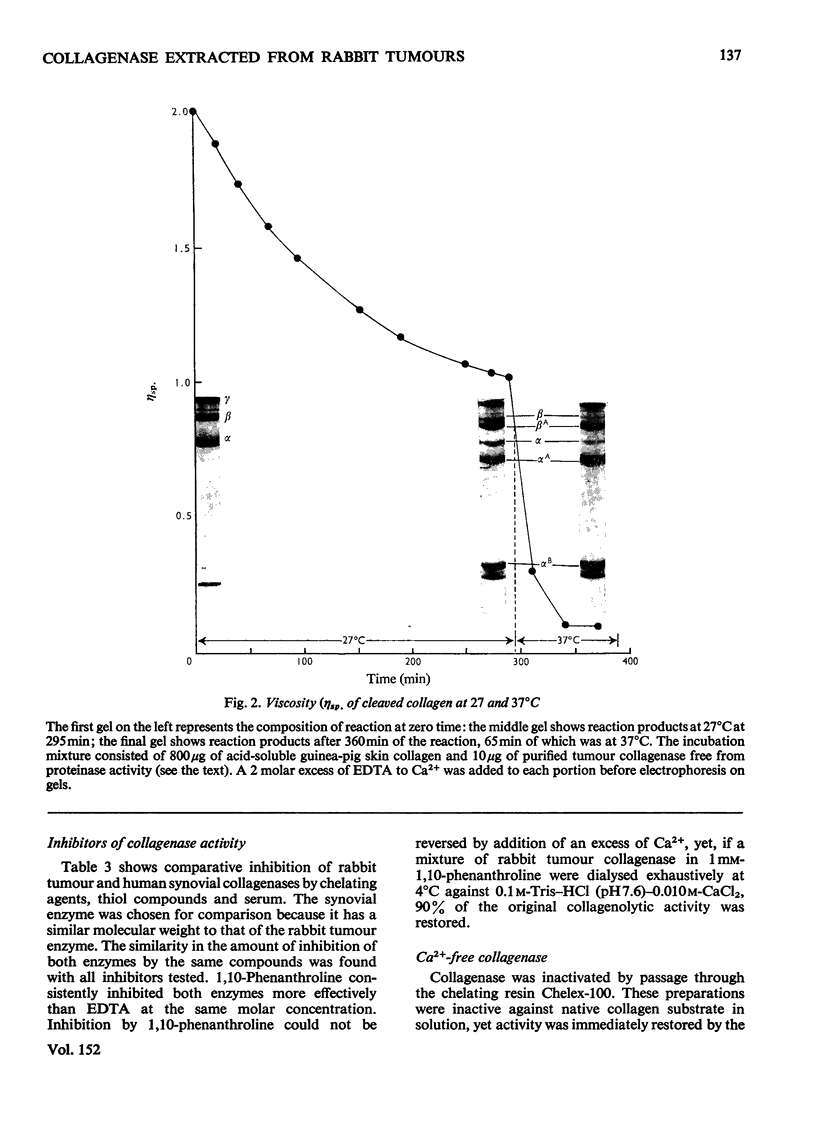
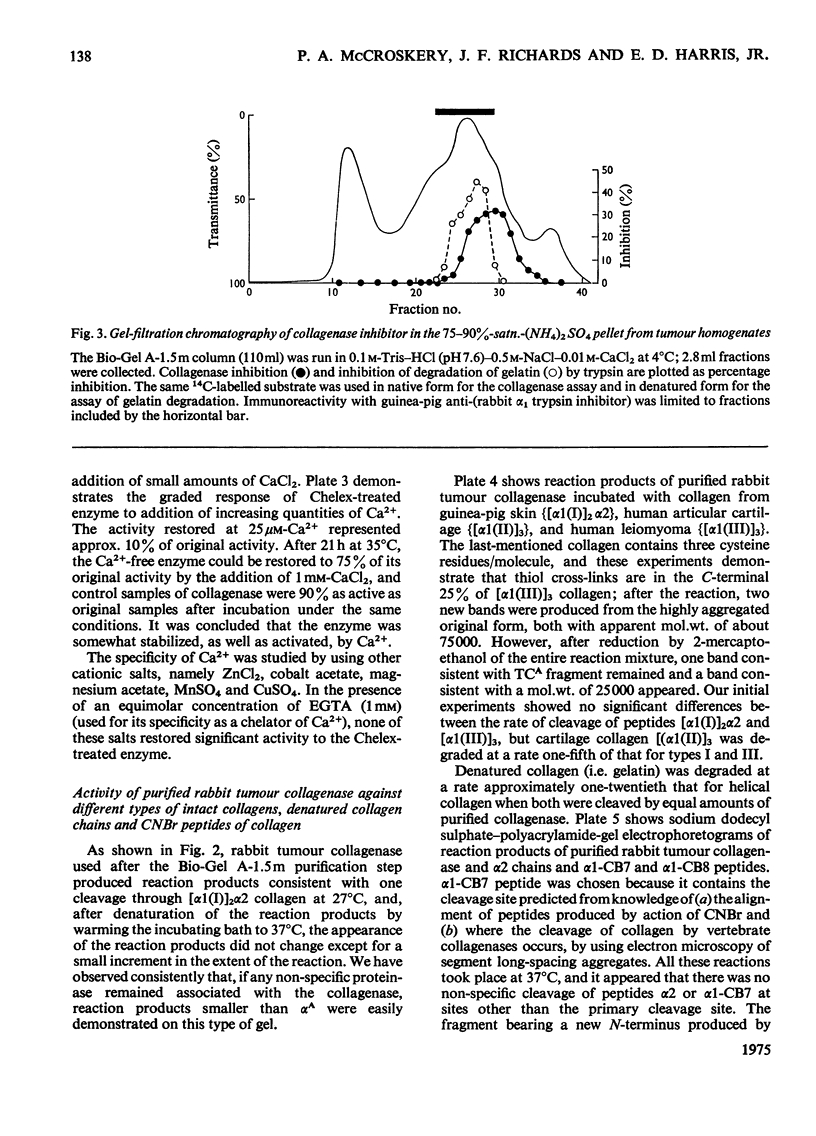




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abe S., Nagai Y. Evidence for the presence of a complex of collagenase with alpha2-macroglobulin in human rheumatoid synovial fluid: a possible regulatory mechanism of collagenase activity in vivo. J Biochem. 1973 Apr;73(4):897–900. doi: 10.1093/oxfordjournals.jbchem.a130153. [DOI] [PubMed] [Google Scholar]
- BONTING S. L., CARAVAGGIO L. L., HAWKINS N. M. Studies on sodium-potassium-activated adenosinetriphosphatase. VI. Its role in cation transport in the lens of cat, calf and rabbit. Arch Biochem Biophys. 1963 Apr;101:47–55. doi: 10.1016/0003-9861(63)90532-0. [DOI] [PubMed] [Google Scholar]
- Barrett A. J., Starkey P. M. The interaction of alpha 2-macroglobulin with proteinases. Characteristics and specificity of the reaction, and a hypothesis concerning its molecular mechanism. Biochem J. 1973 Aug;133(4):709–724. doi: 10.1042/bj1330709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer E. A., Eisen A. Z., Jeffrey J. J. Studies on purified rheumatoid synovial collagenase in vitro and in vivo. J Clin Invest. 1971 Oct;50(10):2056–2064. doi: 10.1172/JCI106699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berman M. B., Manabe R. Corneal collagenases: evidence for zinc metalloenzymes. Ann Ophthalmol. 1973 Nov;5(11):1193–passim. [PubMed] [Google Scholar]
- Chung E., Miller E. J. Collagen polymorphism: characterization of molecules with the chain composition (alpha 1 (3)03 in human tissues. Science. 1974 Mar;183(130):1200–1201. doi: 10.1126/science.183.4130.1200. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Eisen A. Z., Bauer E. A., Jeffrey J. J. Human skin collagenase. The role of serum alpha-globulins in the control of activity in vivo and in vitro. Proc Natl Acad Sci U S A. 1971 Jan;68(1):248–251. doi: 10.1073/pnas.68.1.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evanson J. M., Jeffrey J. J., Krane S. M. Human collagenase: identification and characterization of an enzyme from rheumatoid synovium in culture. Science. 1967 Oct 27;158(3800):499–502. doi: 10.1126/science.158.3800.499. [DOI] [PubMed] [Google Scholar]
- Fietzek P. P., Rexrodt F. W., Hopper K. E., Kühn K. The covalent structure of collagen. 2. The amino-acid sequence of alpha1-CB7 from calf-skin collagen. Eur J Biochem. 1973 Oct 5;38(2):396–400. doi: 10.1111/j.1432-1033.1973.tb03072.x. [DOI] [PubMed] [Google Scholar]
- Fullmer H. M., Taylor R. E., Guthrie R. W. Human gingival collagenase: purification, molecular weight, and inhibitor studies. J Dent Res. 1972 Mar-Apr;51(2):349–355. doi: 10.1177/00220345720510022101. [DOI] [PubMed] [Google Scholar]
- GLIMCHER M. J., FRANCOIS C. J., RICHARDS L., KRANE S. M. THE PRESENCE OF ORGANIC PHOSPHORUS IN COLLAGENS AND GELATINS. Biochim Biophys Acta. 1964 Dec 9;93:585–602. doi: 10.1016/0304-4165(64)90342-3. [DOI] [PubMed] [Google Scholar]
- GROSS J., KIRK D. The heat precipitation of collagen from neutral salt solutions: some rate-regulating factors. J Biol Chem. 1958 Aug;233(2):355–360. [PubMed] [Google Scholar]
- GROSS J. [Studies on the formation of collagen. III. Time-dependent solubility changes of collagen in vitro]. J Exp Med. 1958 Aug 1;108(2):215–226. doi: 10.1084/jem.108.2.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross J., Harper E., Harris E. D., McCroskery P. A., Highberger J. H., Corbett C., Kang A. H. Animal collagenases: specificity of action, and structures of the substrate cleavage site. Biochem Biophys Res Commun. 1974 Nov 27;61(2):605–612. doi: 10.1016/0006-291x(74)91000-6. [DOI] [PubMed] [Google Scholar]
- HEPPEL L. A., HILMORE R. J. Purification and properties of 5-nucleotidase. J Biol Chem. 1951 Feb;188(2):665–676. [PubMed] [Google Scholar]
- Harpel P. C. Studies on human plasma alpha 2-macroglobulin-enzyme interactions. Evidence for proteolytic modification of the subunit chain structure. J Exp Med. 1973 Sep 1;138(3):508–521. doi: 10.1084/jem.138.3.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper E., Bloch K. J., Gross J. The zymogen of tadpole collagenase. Biochemistry. 1971 Aug 3;10(16):3035–3041. doi: 10.1021/bi00792a008. [DOI] [PubMed] [Google Scholar]
- Harris E. D., Jr, DiBona D. R., Krane S. M. Collagenases in human synovial fluid. J Clin Invest. 1969 Nov;48(11):2104–2113. doi: 10.1172/JCI106177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris E. D., Jr, Farrell M. E. Resistance to collagenase: a characteristic of collagen fibrils cross-linked by formaldehyde. Biochim Biophys Acta. 1972 Aug 31;278(1):133–141. doi: 10.1016/0005-2795(72)90114-6. [DOI] [PubMed] [Google Scholar]
- Harris E. D., Jr, Faulkner C. S., 2nd, Wood S., Jr Collagenase in carcinoma cells. Biochem Biophys Res Commun. 1972 Sep 5;48(5):1247–1253. doi: 10.1016/0006-291x(72)90845-5. [DOI] [PubMed] [Google Scholar]
- Harris E. D., Jr, Krane S. M. An endopeptidase from rheumatoid synovial tissue culture. Biochim Biophys Acta. 1972 Feb 28;258(2):566–576. doi: 10.1016/0005-2744(72)90249-5. [DOI] [PubMed] [Google Scholar]
- Harris E. D., Jr, Krane S. M. Collagenases (first of three parts). N Engl J Med. 1974 Sep 12;291(11):557–563. doi: 10.1056/NEJM197409122911105. [DOI] [PubMed] [Google Scholar]
- Harris E. D., Jr, Krane S. M. Collagenases (second of three parts). N Engl J Med. 1974 Sep 19;291(12):605–609. doi: 10.1056/NEJM197409192911205. [DOI] [PubMed] [Google Scholar]
- Harris E. D., Jr, Krane S. M. Collagenases (third of three parts). N Engl J Med. 1974 Sep 26;291(13):652–661. doi: 10.1056/NEJM197409262911305. [DOI] [PubMed] [Google Scholar]
- Kruze D., Wojtecka E. Activation of leucocyte collagenase proenzyme by rheumatoid synovial fluid. Biochim Biophys Acta. 1972 Dec 28;285(2):436–446. doi: 10.1016/0005-2795(72)90330-3. [DOI] [PubMed] [Google Scholar]
- LEWIS M. S., PIEZ K. A. SEDIMENTATION-EQUILIBRIUM STUDIES OF THE MOLECULAR WEIGHT OF SINGLE AND DOUBLE CHAINS FROM RAT-SKIN COLLAGEN. Biochemistry. 1964 Aug;3:1126–1131. doi: 10.1021/bi00896a020. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROBERTS N. R., WU M. L., HIXON W. S., CRAWFORD E. J. The quantitative histochemistry of brain. II. Enzyme measurements. J Biol Chem. 1954 Mar;207(1):19–37. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lazarus G. S., Daniels J. R., Lian J., Burleigh M. C. Role of granulocyte collagenase in collagen degradation. Am J Pathol. 1972 Sep;68(3):565–578. [PMC free article] [PubMed] [Google Scholar]
- MORTON R. K. Alkaline phosphatase of milk. 2. Purification of the enzyme. Biochem J. 1953 Dec;55(5):795–800. doi: 10.1042/bj0550795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCroskery P. A., Wood S., Jr, Harris E. D., Jr Gelatin: a poor substrate for a mammalian collegenase. Science. 1973 Oct 5;182(4107):70–71. doi: 10.1126/science.182.4107.70. [DOI] [PubMed] [Google Scholar]
- NAGAI Y., GROSS J., PIEZ K. A. DISC ELECTROPHORESIS OF COLLAGEN COMPONENTS. Ann N Y Acad Sci. 1964 Dec 28;121:494–500. doi: 10.1111/j.1749-6632.1964.tb14221.x. [DOI] [PubMed] [Google Scholar]
- NORDLIE R. C., ARION W. J. EVIDENCE FOR THE COMMON IDENTITY OF GLUCOSE 6-PHOSPHATASE, INORGANIC PYROPHOSPHATASE, AND PYROPHOSPHATE-GLUCOSE PHOSPHOTRANSFERASE. J Biol Chem. 1964 Jun;239:1680–1685. [PubMed] [Google Scholar]
- OUCHTERLONY O. Diffusion-in-gel methods for immunological analysis. II. Prog Allergy. 1962;6:30–154. doi: 10.1159/000313795. [DOI] [PubMed] [Google Scholar]
- Oronsky A. L., Perper R. J., Schroder H. C. Phagocytic release and activation of human leukocyte procollagenase. Nature. 1973 Dec 14;246(5433):417–419. doi: 10.1038/246417a0. [DOI] [PubMed] [Google Scholar]
- PENNINGTON R. J. Biochemistry of dystrophic muscle. Mitochondrial succinate-tetrazolium reductase and adenosine triphosphatase. Biochem J. 1961 Sep;80:649–654. doi: 10.1042/bj0800649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PROCKOP D. J., UDENFRIEND S. A specific method for the analysis of hydroxyproline in tissues and urine. Anal Biochem. 1960 Nov;1:228–239. doi: 10.1016/0003-2697(60)90050-6. [DOI] [PubMed] [Google Scholar]
- Sakai T., Gross J. Some properties of the products of reaction of tadpole collagenase with collagen. Biochemistry. 1967 Feb;6(2):518–528. doi: 10.1021/bi00854a021. [DOI] [PubMed] [Google Scholar]
- Sakamoto S., Sakamoto M., Goldhaber P., Glimcher M. J. Studies on the interaction between heparin and mouse bone collagenase. Biochim Biophys Acta. 1975 Mar 14;385(1):41–50. doi: 10.1016/0304-4165(75)90072-0. [DOI] [PubMed] [Google Scholar]
- Sakamoto S., Sakamoto M., Goldhaber P., Glimcher M. J. The inhibition of mouse bone collagenase by lysozyme. Calcif Tissue Res. 1974 May 28;14(4):291–299. doi: 10.1007/BF02060303. [DOI] [PubMed] [Google Scholar]
- Steers E., Jr, Cuatrecasas P., Pollard H. B. The purification of beta-galactosidase from Escherichia coli by affinity chromatography. J Biol Chem. 1971 Jan 10;246(1):196–200. [PubMed] [Google Scholar]
- Traub W., Piez K. A. The chemistry and structure of collagen. Adv Protein Chem. 1971;25:243–352. doi: 10.1016/s0065-3233(08)60281-8. [DOI] [PubMed] [Google Scholar]
- Vaes G. The release of collagenase as an inactive proenzyme by bone explants in culture. Biochem J. 1972 Jan;126(2):275–289. doi: 10.1042/bj1260275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- Werb Z., Burleigh M. C. A specific collagenase from rabbit fibroblasts in monolayer culture. Biochem J. 1974 Feb;137(2):373–385. doi: 10.1042/bj1370373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Burleigh M. C., Barrett A. J., Starkey P. M. The interaction of alpha2-macroglobulin with proteinases. Binding and inhibition of mammalian collagenases and other metal proteinases. Biochem J. 1974 May;139(2):359–368. doi: 10.1042/bj1390359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woessner J. F., Jr, Ryan J. N. Collagenase activity in homogenates of the involuting rat uterus. Biochim Biophys Acta. 1973 Jun 6;309(2):397–405. doi: 10.1016/0005-2744(73)90038-7. [DOI] [PubMed] [Google Scholar]