Abstract
This study aims to investigate the association between serum copper (Cu), selenium (Se), zinc (Zn), Se/Cu and Zn/Cu ratios and the risk of sarcopenia. In this study, which involved 2766 adults aged ≥ 20 years enrolled in the National Health and Nutrition Examination Survey (NHANES) from 2011 to 2016, multivariable logistic regression, restricted cubic spline (RCS) models and mediation analyses were used. After full adjustment, multivariable logistic regression revealed that higher serum copper levels were correlated with an increased risk of sarcopenia. Conversely, higher serum Se/Cu (OR 0.45, 95% CI 0.23–0.89, P = 0.023) and Zn/Cu (OR 0.49, 95% CI 0.27–0.90, P = 0.024) were associated with a decreased risk of sarcopenia. The RCS curve indicated a non-linear, roughly inverted L-shaped relationship between serum Cu and sarcopenia risk (P non-linear < 0.001). Additionally, Se/Cu (P non-linear = 0.179) and Zn/Cu (P non-linear = 0.786) showed negative linear associations with sarcopenia risk. Furthermore, white blood cell (WBC) count, neutrophil count, and systemic inflammation index (SII) were identified as significant mediators in the relationship between serum Cu and the risk of sarcopenia, with mediation proportions of 6.34%, 6.20%, and 4.37%, respectively (all P < 0.05). Therefore, balancing essential trace metals is crucial for maintaining muscle health.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-84011-0.
Keywords: Sarcopenia, Copper, Selenium, Zinc, NHANES
Subject terms: Biomarkers, Diseases, Medical research, Pathogenesis, Risk factors, Health care, Diagnosis, Geriatrics, Health services, Nutrition, Public health
Introduction
Sarcopenia, the age-related loss of muscle mass, strength, and function, is a significant threat to the health of the elderly population1. Globally, its prevalence ranges from 5 to 10%, increasing to 11–50% among individuals over the age of 802. Associated with a higher risk of falls, disability and mortality in older adults, sarcopenia is influenced by ageing, sedentary lifestyles, prolonged bed rest and inadequate energy and protein intake3–5. Despite ongoing research, the exact pathogenesis of sarcopenia remains incompletely understood, highlighting the need for further investigation into its risk factors and mechanisms.
Essential trace metals such as copper (Cu), selenium (Se), and zinc (Zn) play a crucial role in regulating cell metabolism, antioxidant, and anti-inflammatory in the body6. Cu is an important redox cofactor for mitochondria energy production reactions7. Muscle tissue has a high energy requirement, resulting in a high demand for Cu to participate in energy metabolism8. Both the influx of Cu and the intracellular Cu are required for the differentiation of skeletal muscle cells9. Skeletal muscle stores about 20–46% of Se in the human body10. Se is involved in the formation of selenoproteins, which are involved in the main antioxidant defence system of cells and are present in various tissues as biocatalysts for the removal of harmful metabolic peroxide products to maintain redox homeostasis11. Selenoprotein W (SelW) and Selenoprotein N (SelN) play an important role in muscle formation as they are highly expressed in proliferating myoblasts and defend myoblasts against oxidative stress12,13. Zinc is mainly found in skeletal muscle (63%) and bone (20%) tissues10. Zn reduces chronic inflammatory responses by decreasing serum inflammatory cytokines and also attenuates oxidative stress by acting as a cofactor for antioxidant enzymes and as an enzyme catalyst14. Zn ions can act as biochemical signals to trigger myogenic differentiation15.
Altered levels of essential trace metals have been found in some pathological conditions. Higher levels of serum Zn, Zn/Cu ratio, and Zn/Se ratio were associated with a reduced risk of sleep disorders16. People with higher intakes of Cu, Se and Zn tended to have higher odds of osteoarthritis (OA)17. In the elderly population, reduced plasma selenium levels are associated with increased inflammation and cardiovascular disease18. Recent years have also seen research into the relationship between changes in levels of essential trace metals and muscle health. For instance, Zn deficiency can negatively impact muscle proteostasis and mitochondrial biology, leading to increased apoptosis and reduced muscle fibre cross-sectional area19. Additionally, a study has shown that higher serum Zn concentrations are linked to greater grip strength, while serum Cu levels are inversely correlated with grip strength20. In another study, low blood Se levels are associated with reduced grip strength21. Although preliminary research has delved into the relationship between essential trace metals and muscle health, the combined effects and underlying mechanisms on sarcopenia are still not fully understood.
Multiple studies have indicated a link between inflammation and the occurrence of sarcopenia22,23. Zhao et al. found that the risk of sarcopenia increased in correlation with the systemic inflammation index (SII)24. Levels of essential trace metals are closely linked to inflammation. Excessive Cu can induce the production of reactive oxygen species (ROS), leading to the activation of transcription factors associated with inflammatory responses25,26. Circulating blood Cu is significantly elevated in rheumatoid arthritis (RA) patients and positively correlates with inflammatory markers27. Elevated Cu/Zn ratio was in association with oxidative stress, inflammation28,29. Additionally, excessive exposure to Cu has been shown to decrease the grip strength of rats by triggering inflammation30. Therefore, inflammation could potentially act as a mediator in the relationship between essential trace metals and sarcopenia.
In this study, we employed multivariable logistic regression and restricted cubic spline (RCS) models to investigate the connection between serum Cu, Se, Zn, as well as Se/Cu and Zn/Cu ratios, and the risk of sarcopenia. Furthermore, we conducted mediation analyses to examine the involvement of inflammatory markers (white blood cell count, neutrophil count, and SII) in mediating the relationship between serum Cu levels and the risk of sarcopenia.
Methods
Study population
The data used in this study were obtained from the National Health and Nutrition Examination Survey (NHANES), a cross-sectional survey of the US civilian population. The NHANES program focuses on a range of issues related to health and nutrition. The program surveys a nationally representative sample of about 5,000 people in the US each year. This study population came from three 2-year cycles (2011/2012, 2013/2014, 2015/2016), which included a total of 29,902 participants. Eligible participants had complete data on dual-energy X-ray absorptiometry (DAX), body mass index (BMI), serum Cu, serum Se, serum Zn, white blood cell (WBC) count, neutrophil count, lymphocyte count, and platelet count. We excluded participants who were younger than 20 years (12,854) or with missing data on DAX (8,438), BMI (25), serum Cu, Se, and Zn (5,815), and inflammatory data (4). Finally, 2766 participants were included in this study (Fig. 1).
Fig. 1.
Flowchart of participant selection.
Serum trace metals
Serum samples were processed and stored at -70 °C until shipped to the Division of Laboratory Sciences, National Center for Environmental Health, Centers for Disease Control and Prevention. Serum concentrations of Cu, Se and Zn were measured by Inductively Coupled Plasma Dynamic Reaction Cell Mass Spectrometry (ICP-DRC-MS). The unit of concentration for Cu, Se and Zn is expressed in µg/dL.
Sarcopenia
Bone and soft tissue measurements of the whole body, including the head, trunk, arms and legs was measured by DAX scans in NHANES. Appendicular skeletal muscle mass (ASM) was the sum of arms and legs lean mass calculated from the DXA data. The sarcopenia index was calculated by dividing the ASM by the BMI. In this study, sarcopenia was diagnosed according to criteria defined by the Foundation for the National Institutes of Health (FNIH)31. Individuals with a sarcopenia index < 0.789 for men or < 0.512 for women were diagnosed with sarcopenia.
Inflammatory biomarkers
Peripheral blood samples in this study were obtained from the NHANES Mobile Examination Centre (MEC). The peripheral blood samples were analyzed for white blood cell, neutrophil, lymphocyte and platelet counts and the results are expressed in units of 109 cells/L. The SII was defined using platelet count * neutrophil count/lymphocyte count according to previous work32.
Covariates
Based on previous research, we selected the following variables as covariates, including age, gender, race (Mexican American, other hispanic, non-hispanic white, non-hispanic black and other race), educational level (below high school, high school and above high school), family poverty income ratio (PIR), smoking status (current smoker, former smoker, and never smoker), drinking status (1–5 drinks/month, 5–10 drinks/month, 10 + drinks/month, and no drinker), sedentary time. BMI was categorised as underweight (< 18.5 kg/m2), normal weight (18.5–25 kg/m2), overweight (25–30 kg/m2) and obesity (≥ 30 kg/m2). Diabetes was diagnosed according to the questionnaire question “Doctor told you have diabetes?”. Hypertension was diagnosed according to the questionnaire question “Has anyone ever told you that you have high blood pressure?” Estimation of 24-hour energy intake based on the average of energy intakes on day 1 and day 2 of the dietary interview (if only day 1 energy intake data was available, use this value instead of the average).
Statistical analysis
As NHANES uses a complex, multi-stage probability sampling design, weighted analyses have been carried out. Wilcoxon rank-sum test for complex samples; chi-squared test with Rao & Scott’s second-order correction was used to compare differences in baseline characteristics between sarcopenia and non-sarcopenia groups.
Serum Cu, Se, Zn, Se/Cu, and Zn/Cu ratios were categorized into three groups (Q1, Q2, Q3) based on tertiles of measured values. Multivariate logistic regression was used to examine how serum levels of Cu, Se, Zn, and the ratios of Se/Cu and Zn/Cu relate to sarcopenia. The crude model had no covariate adjustments, while Model I adjusted for age, gender, race, and education level, and Model II additionally adjusted for PIR, BMI, smoking and drinking status, diabetes, hypertension, sedentary time, and 24-hour energy intake. Moreover, the dose-response association between serum Cu, Se, Zn, Se/Cu, Zn/Cu, and sarcopenia was explored using the RCS model with three nodes set at the 10th, 50th, and 90th percentiles of the distribution.
We used linear regression to assess the connection between serum Cu, Se/Cu, Zn/Cu, and inflammatory biomarkers. Multivariate logistic regression was employed to analyze the relationship between inflammatory biomarkers and the risk of sarcopenia. Additionally, we conducted mediation analyses using the mediation package in R software to investigate whether the links between serum Cu and sarcopenia were mediated by inflammatory biomarkers. All analyses in this study were carried out using R 4.3.1.
Results
Population characteristics
Table 1 shows the basic characteristics of participants of the 2,766 individuals in this study. Among them, there were 1,401 males (50.77%) and 1,365 females (49.23%). The prevalence of sarcopenia in the study population was 7.0%. Age, race, education level, BMI, drinking status, diabetes, and 24-hour energy intake were statistically different between the sarcopenia and the non-sarcopenia groups (all P < 0.05).
Table 1.
Basic characteristics of participants.
| Characteristic | Overall, N = 2766 (100%)1 | Non-sarcopenia, N = 2535 (93%)1 | Sarcopenia, N = 231 (7.0%)1 | P Value2 |
|---|---|---|---|---|
| Age (years) | 39.4 (11.6) | 39.1 (11.5) | 43.3 (12.1) | < 0.001 |
| Gender, n (%) | > 0.9 | |||
| Female | 1,365 (49.23%) | 1,243 (49.19%) | 122 (49.77%) | |
| Male | 1,401 (50.77%) | 1,292 (50.81%) | 109 (50.23%) | |
| Race, n (%) | < 0.001 | |||
| Mexican American | 419 (10.53%) | 338 (9.37%) | 81(25.85%) | |
| Other Hispanic | 299 (7.30%) | 262 (6.94%) | 37 (12.12%) | |
| Non-Hispanic White | 994 (62.13%) | 937 (63.18%) | 57 (48.13%) | |
| Non-Hispanic Black | 569 (11.32%) | 554 (11.85%) | 15 (4.33%) | |
| Other race | 485 (8.73%) | 444 (8.66%) | 41 (9.57%) | |
| PIR | 2.98 (1.68) | 2.99 (1.68) | 2.80 (1.64) | 0.3 |
| Education level, n (%) | < 0.001 | |||
| Below high school | 506 (13.23%) | 434 (12.55%) | 72 (22.27%) | |
| High school | 590 (20.73%) | 532 (20.44%) | 58 (24.55%) | |
| Above high school | 1,670 (66.04%) | 1,569 (67.01%) | 101 (53.18%) | |
| BMI, n (%) | < 0.001 | |||
| Underweight | 55 (1.61%) | 55 (1.73%) | 0.00 (0.00%) | |
| Normal weight | 850 (30.40%) | 825 (32.13%) | 25 (7.48%) | |
| Overweight | 868 (33.42%) | 812 (34.29%) | 56 (21.92%) | |
| Obesity | 993 (34.58%) | 843 (31.85%) | 150 (70.61%) | |
| Smoking status, n (%) | 0.067 | |||
| Current smoker | 614 (21.89%) | 581 (22.48%) | 33 (14.10%) | |
| Former smoker | 466 (18.30%) | 415 (17.90%) | 51 (23.49%) | |
| Never smoker | 1,686 (59.81%) | 1,539 (59.61%) | 147 (62.41%) | |
| Drinking status, n (%) | < 0.001 | |||
| 1–5 drinks/month | 1,330 (48.49%) | 1,215 (48.40%) | 115 (49.76%) | |
| 5–10 drinks/month | 261 (11.35%) | 254 (12.02%) | 7 (2.43%) | |
| 10 + drinks/month | 353 (16.88%) | 335 (17.04%) | 18 (14.71%) | |
| No drinker | 623 (17.30%) | 546 (16.50%) | 77 (27.89%) | |
| Not recorded | 199 (5.98%) | 185 (6.03%) | 14 (5.22%) | |
| Sedentary time (min) | 398 (208) | 397 (207) | 419 (224) | 0.4 |
| Hypertension, n (%) | 0.3 | |||
| Yes | 629 (21.82%) | 562 (21.53%) | 67 (25.67%) | |
| No | 2,136 (78.17%) | 1,972 (78.46%) | 164 (74.33%) | |
| Not recorded | 1 (0.01%) | 1 (0.01%) | 0 (0.00%) | |
| Diabetes, n (%) | < 0.001 | |||
| Yes | 207 (6.43%) | 171 (5.72%) | 36 (15.80%) | |
| No | 2,556 (93.52%) | 2,361 (94.23%) | 195 (84.20%) | |
| Not recorded | 3 (0.05%) | 3 (0.05%) | 0 (0.00%) | |
| Energy intake, kcal/day | 2,169.8 (859.0) | 2,182.1 (866.3) | 2,003.5 (735.8) | 0.039 |
1Mean (SD); n (unweighted) (%).
2Wilcoxon rank-sum test for complex survey samples; chi-squared test with Rao and Scott’s second-order correction.
Significant values are in bold.
Multivariate logistic regression models
Table 2 demonstrates a significant correlation between serum Cu levels and sarcopenia risk. Sarcopenia risk increased in higher tertiles of serum Cu. In the crude model, compared to the lowest tertile (Q1), the risk of sarcopenia rose by 2.73-fold (95% CI 1.81–4.13, P < 0.001) and 3.25-fold (95% CI 2.13–4.98, P < 0.001) in the middle (Q2) and highest (Q3) tertiles, respectively. In model I and model II, the sarcopenia risk remained significantly elevated in Q2 and Q3 compared to Q1. Serum Se and Zn levels showed no association with sarcopenia risk in all models. However, in model II, participants in the highest tertiles of Se/Cu (OR 0.45, 95% CI 0.23–0.89, P = 0.023) and Zn/Cu (OR 0.49, 95% CI 0.27–0.90, P = 0.024) exhibited a lower risk of sarcopenia compared to those in the lowest tertiles.
Table 2.
Associations of serum Cu, Se, Zn, Se/Cu and Zn/Cu ratios with Sarcopenia.
| Characteristic | Crude model | Model I | Model II | ||||||
|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | P-value | OR | 95% CI | P-value | OR | 95% CI | P-value | |
| Cu | |||||||||
| Q1 (43.9–101.7) | Ref | Ref | – | Ref | Ref | – | Ref | Ref | – |
| Q2 (101.7–123.2) | 2.73 | 1.81, 4.13 | < 0.001 | 2.96 | 1.88, 4.65 | < 0.001 | 3.02 | 1.85, 4.91 | < 0.001 |
| Q3 (123.2–289.8) | 3.25 | 2.13, 4.98 | < 0.001 | 4.62 | 2.62, 8.15 | < 0.001 | 3.38 | 1.82, 6.26 | < 0.001 |
| Se | |||||||||
| Q1 (80.5–121.7) | Ref | Ref | – | Ref | Ref | – | Ref | Ref | – |
| Q2 (80.5–135.4) | 1.06 | 0.70, 1.61 | 0.778 | 1.01 | 0.66, 1.53 | 0.974 | 0.86 | 0.52, 1.42 | 0.546 |
| Q3 (135.4–299.1) | 1.18 | 0.71, 1.96 | 0.528 | 1.07 | 0.64, 1.79 | 0.795 | 1.09 | 0.63, 1.87 | 0.745 |
| Zn | |||||||||
| Q1 (44.0–75.5) | Ref | Ref | – | Ref | – | – | Ref | Ref | – |
| Q2 (75.5–87.6) | 0.82 | 0.52, 1.31 | 0.407 | 0.81 | 0.50, 1.31 | 0.387 | 0.84 | 0.47, 1.51 | 0.542 |
| Q3 (87.6–232.5) | 1.00 | 0.62, 1.61 | 0.995 | 1.02 | 0.63, 1.67 | 0.921 | 1.02 | 0.57, 1.80 | 0.952 |
| Se/Cu | |||||||||
| Q1 (0.46–1.03) | Ref | Ref | – | Ref | Ref | – | Ref | Ref | – |
| Q2 (1.03–1.29) | 0.79 | 0.54, 1.16 | 0.222 | 0.64 | 0.40, 1.05 | 0.074 | 0.88 | 0.55, 1.39 | 0.557 |
| Q3 (1.29–4.93) | 0.42 | 0.27, 0.67 | < 0.001 | 0.30 | 0.16, 0.56 | < 0.001 | 0.45 | 0.23, 0.89 | 0.023 |
| Zn/Cu | |||||||||
| Q1 (0.19–0.64) | Ref | Ref | – | Ref | Ref | – | Ref | Ref | – |
| Q2 (0.64–0.82) | 0.77 | 0.53, 1.11 | 0.157 | 0.65 | 0.42, 1.00 | 0.050 | 0.92 | 0.60, 1.42 | 0.702 |
| Q3 (0.82–3.08) | 0.42 | 0.26, 0.69 | < 0.001 | 0.35 | 0.20, 0.61 | < 0.001 | 0.49 | 0.27, 0.90 | 0.024 |
Crude Model: not adjusted for covariates. Model I: Adjusted for age, gender, race, educational level. Model II: Adjusted for age, gender, race, educational level, PIR, smoking status, drinking status, sedentary time, BMI, diabetes, hypertension, 24-hour energy intake.
OR odds ratio, CI confidence interval.
Significant values are in bold.
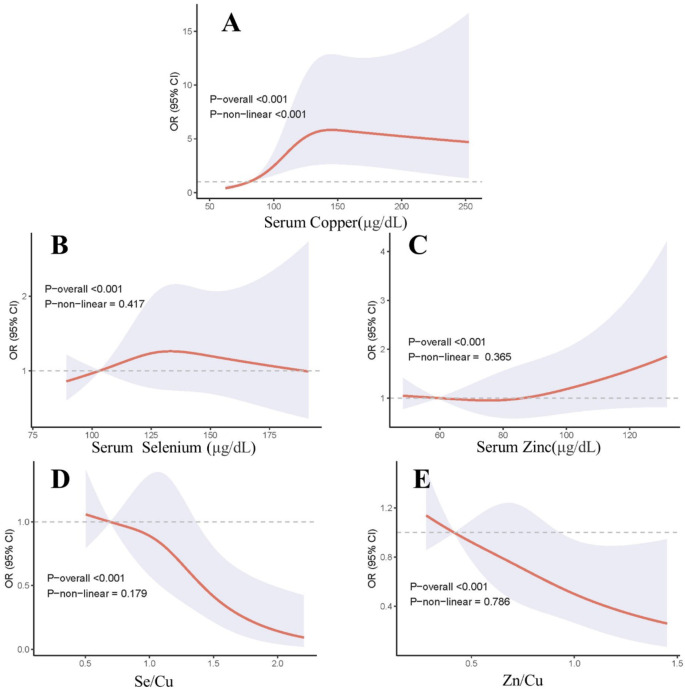
RCS analyses
The dose-response relationship between serum Cu, Se, Zn, Se/Cu and Zn/Cu and the risk of sarcopenia was investigated using the RCS model. As shown in Fig. 2, after adjustment for all covariates, a roughly inverted L-shaped relationship existed between serum Cu and the risk of sarcopenia with non-linearity (P non-linear < 0.001). The risk of sarcopenia gradually increased with increasing serum Cu, with a maximum risk of sarcopenia at 145.51 µg/dL, after which the curve became relatively flat. There was no non-linear relationship between serum Se, Zn and the risk of sarcopenia (P non-linear > 0.05). In addition, we found that Se/Cu (P non-linear = 0.179) and Zn/Cu (P non-linear = 0.786) were negatively linearly associated with the risk of sarcopenia.
Fig. 2.
Restricted cubic spline (RCS) model of the OR of sarcopenia with serum Cu, Se, Zn, Se/Cu and Zn/Cu. Models were adjusted for age, gender, race, educational level, PIR, smoking status, drinking status, sedentary time, BMI, diabetes, hypertension, 24-hour energy intake.
Mediation analyses
As shown in Table 3, serum Cu was significantly positively correlated with WBC (β = 0.01, 95% CI 0.01–0.02, P < 0.001), neutrophils (β = 0.01, 95% CI 0.00-0.01, P < 0.001) and SII (β = 1.3, 95% CI 0.75–1.90, P < 0.001) in linear regression. In addition, we observed that Se/Cu and WBC (β = − 0.72, 95% CI − 1.1 to −0.30, P = 0.002), neutrophils (β = − 0.58, 95% CI − 0.94 to −0.22, P = 0.003), SII (β = − 78, 95% CI − 136 to −20, P = 0.011) were significantly negatively correlated. Similarly, Zn/Cu was significantly negatively associated with WBC (β = − 2.1, 95% CI − 3.1 to −1.1, P < 0.001), neutrophils (β = − 1.7, 95% CI − 2.5 to −0.82, P < 0.001), SII (β = − 182, 95% CI − 271 to −93, P < 0.001). As shown in Table 4, WBC (OR 1.15, 95% CI 1.03–1.27, P = 0.013), neutrophils (OR 1.18, 95% CI 1.05–1.33, P = 0.008) and SII (OR 1.0008, 95% CI 1.0004–1.0013, P = 0.001) were associated with an increased risk of sarcopenia. We further analyzed the mediating effect of inflammatory biomarkers between serum Cu and the risk of sarcopenia. Figure 3 shows the mediating effect of inflammatory biomarkers in the association between serum Cu and the risk of sarcopenia. We found that WBC, neutrophils and SII all significantly mediated the relationship between serum Cu and the risk of sarcopenia, with a mediation proportion of 6.34%, 6.20%, and 4.37%, respectively (all P < 0.05). However, inflammatory biomarkers were not observed to mediate between Se/Cu (Supplementary Fig. S1) or Zn/Cu (Supplementary Fig. S2) and the risk of sarcopenia.
Table 3.
Association between Cu, Se/Cu, Zn/Cu and inflammation biomarkers.
| Inflammatory markers | Beta | 95% CI | P-value |
|---|---|---|---|
| Cu | |||
| WBC | 0.01 | 0.01, 0.02 | < 0.001 |
| Neutrophils | 0.01 | 0.00, 0.01 | < 0.001 |
| SII | 1.3 | 0.75, 1.90 | < 0.001 |
| Se/Cu | |||
| WBC | − 0.72 | − 1.1, − 0.30 | 0.002 |
| Neutrophils | − 0.58 | − 0.94, − 0.22 | 0.003 |
| SII | − 78 | − 136, − 20 | 0.011 |
| Zn/Cu | |||
| WBC | − 2.1 | − 3.1, − 1.1 | < 0.001 |
| Neutrophils | − 1.7 | − 2.5, − 0.82 | < 0.001 |
| SII | − 182 | − 271, − 93 | < 0.001 |
Models were adjusted for age, gender, race, educational level, PIR, smoking status, drinking status, sedentary time, BMI, diabetes, hypertension, 24-hour energy intake.
CI confidence interval.
Table 4.
Association between the inflammation biomarkers and the risk of Sarcopenia.
| Inflammatory markers | OR | 95% CI | P-value |
|---|---|---|---|
| WBC | 1.15 | 1.03, 1.27 | 0.013 |
| Neutrophils | 1.18 | 1.05, 1.33 | 0.008 |
| SII | 1.0008 | 1.0004, 1.0013 | 0.001 |
Models were adjusted for age, gender, race, educational level, PIR, smoking status, drinking status, sedentary time, BMI, diabetes, hypertension, 24-hour energy intake.
OR odds ratio, CI confidence interval.
Fig. 3.
Mediating effect and proportions of inflammatory biomarkers between serum Cu and the risk of sarcopenia. Models were adjusted for age, gender, race, educational level, PIR, smoking status, drinking status, sedentary time, BMI, diabetes, hypertension, 24-hour energy intake.
Discussion
As the global population ages, sarcopenia has been the focus of increasing attention. Essential trace metals are essential for muscle health because they are involved in important physiological activities and structural composition. A deficiency of essential trace metals in the body can lead to serious physiological disorders, but an excess of essential trace metals can also increase the risk of certain diseases. It has been reported that Zn deficiency increases the risk of sarcopenia in people with chronic liver disease33. A negative correlation between serum Cu and muscle mass has been found in a recent study34. Although researchers have conducted several studies on the link between single essential trace metal and muscle health, it is not yet clear what the underlying mechanisms are between essential trace metals and the development of sarcopenia. In addition, metal elements can often be additive, synergistic or antagonistic35–37. Therefore, it is necessary to take the interactions between essential trace metals into account. Our study revealed that serum Cu was associated with the risk of sarcopenia, and we clarified the mediating role of inflammation. Additionally, we observed a decrease in the risk of sarcopenia with higher Se/Cu and Zn/Cu ratios. These findings emphasize the importance of considering the interplay between essential trace metals and their potential impact on sarcopenia.
There is growing evidence of a link between inflammation and sarcopenia. Elevated levels of circulating inflammatory factors contribute to decreased muscle mass and strength38. Chronic inflammation activates the ubiquitin proteasome system to induce muscle protein degradation39. Another study showed that chronic inflammation can destroy satellite cells, which play a key role in repairing injured muscle, leading to muscle regeneration being inhibited40. Our study observed a positive correlation between sarcopenia and inflammation biomarkers. Moreover, there has been evidence that Cu can be a promoter of inflammatory responses. Guo et al. found that Cu supplementation enhanced the inflammatory response in macrophages induced by lipopolysaccharide (LPS)41. Researches have observed that excessive Cu exposure upregulates pro-inflammatory cytokines, increases secretion of inflammatory mediators, and activates inflammatory responses by downregulating anti-inflammatory cytokines in various organs through a variety of molecular and cellular signalling pathways, including the NF-κB pathway, the MAPKs pathway, the JAK-STAT pathway, and the NLRP3 inflammasome42. In this study, we also found a linear positive correlation between inflammatory biomarkers and serum Cu. Therefore, it is worth considering whether serum Cu increases the risk of sarcopenia mediated by inflammation. Furthermore, we observed that WBC, neutrophils and SII were involved in the positive correlation between serum Cu and the risk of sarcopenia, accounting for 6.34%, 6.20%, and 4.37% of the proportion using mediation analysis, respectively. In recent years, Cu has been released into the ecosystem in large quantities as a result of increased agricultural, industrial, medical industrial and metal processing activities43,44. As a result, through the consumption of contaminated food and drinking water, Cu enters the food chain and accumulates in humans and animals45,46. Therefore, the adverse effects of excessive Cu exposure on humans should be taken seriously.
Se is a component of antioxidant enzymes involved in maintaining redox homeostasis47. Glutathione peroxidase (GSH-Px), one of the first selenoproteins to be characterised, helps reduce cellular damage caused by free radicals produced during oxidation48. Studies have shown that reactive oxygen species (ROS) accumulated during strenuous exercise cause membrane lesions in skeletal muscle cells, leading to skeletal muscle damage, but selenoproteins may protect muscle cells from oxidative stress damage49. Selenoproteins act as antioxidants to control free radical overproduction at inflammatory sites. In addition, selenoprotein S reduces the release of inflammatory factors from macrophages and is involved in the regulation of inflammatory response processes50. Se deficiency or inhibiting selenoprotein expression leads to increased inflammatory cytokine levels in tissues47. Therefore, Se may help to reduce the inflammatory response caused by some metals, reduce the damage to muscle tissue and help to maintain the health of the muscles. Liang et al. reported that Se has antioxidant and anti-inflammatory properties that may counteract the damage caused to muscles by heavy metals and reduce the negative effects of Cd and Pb on grip strength51. Zn is an essential trace metal closely associated with normal cell development, proliferation and differentiation. It has important anti-inflammatory and antioxidant effects52. An in vitro experiment found that Zn improved muscle contraction53. In a double-blind crossover study, middle-aged women showed improvements in muscle strength and endurance after Zn supplementation (30 mg/day)54. In addition, a study found that Zn supplementation improved growth performance and reduced muscle fatigue in cattle55. The above studies indicate that Se and Zn have numerous protective effects on muscle health. In our study, Se/Cu and Zn/Cu are negatively associated with the risk of sarcopenia. These findings emphasise the importance of maintaining a balance of essential trace metals for muscle health.
Although serum Zn and Se levels were not associated with sarcopenia in this investigation, Se/Cu and Zn/Cu may provide some new insights. High levels of Zn/Cu and Se/Cu were associated with a reduced risk of sarcopenia. This may be because the level of serum Cu is increased in people with sarcopenia, leading to a decrease in serum Zn and Se due to redistribution by activation of inflammatory cytokines in tissues18,56. Cu/Zn ratio is considered as a sensitive biomarker of harmful inflammatory or nutritional changes in the elderly57. High serum Cu/Zn has been reported to be associated with lower lean mass, strength and power, and lower limb function58. There were now fewer studies on serum Se/Cu and this study suggests that the effects of serum Se/Cu on muscle health merit further investigation. From a clinical perspective, the study results suggest that a higher serum Zn/Cu and Se/Cu ratio may reflect a better anti-inflammatory status, which has a protective effect on muscle health. Supplementation of Se and Zn to maintain serum Cu, Se and Zn and their ratios in the appropriate range may be potentially beneficial in sarcopenia.
This research has several limitations. Firstly, as this was a cross-sectional study based on NHANES, it was not able to clarify the causal relationship between serum Cu, Se/Cu, Zn/Cu and the risk of sarcopenia. Secondly, it was not possible to examine the relationship between serum trace metals and muscle strength and function because of the lack of data in NHANES. Thirdly, only participants aged 8–59 years had DAX scans in NHANES, so those older than 59 years were not included in this study.
Conclusion
This study examined the correlation between serum Cu, Se, Zn, Se/Cu and Zn/Cu ratios, and the risk of sarcopenia. Elevated serum levels of Cu increase the inflammatory response, leading to an increased risk of sarcopenia. High levels of Se/Cu and Zn/Cu ratios are beneficial for the reducing the risk of sarcopenia. Therefore, supplementation with Se and Zn may be helpful in preventing sarcopenia in populations with excess Cu exposure.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Author contributions
D.Z. designed this study. B.C., H.W. collected data. S.X. and S.L. analyzed and interpreted the data. Z.Y. validated the data. X.P. visualized the outcome. X.T. and Y.Q. critically reviewed, edited, and approved the manuscript. All authors read and approved the final manuscript.
Funding
This study was supported by funding from the Key Project of the National Natural Science Foundation of China (U21A20390), the National Natural Science Foundation of China (823B2059), Jilin Scientific and Technological Development Program (20230204075YY, 20240305034YY), the Graduate Innovation Program of Jilin University (2024CX142).
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Xiongfeng Tang, Email: tangxf921@gmail.com.
Yanguo Qin, Email: qinyg@jlu.edu.cn.
References
- 1.Yang, A. et al. The effect of vitamin D on Sarcopenia depends on the level of physical activity in older adults. J. Cachexia Sarcopenia Muscle11, 678–689 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Golabi, P. et al. Contribution of Sarcopenia and physical inactivity to mortality in people with non-alcoholic fatty liver disease. JHEP Rep.2, 100171 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mo, Y., Zhou, Y., Chan, H., Evans, C. & Maddocks, M. The association between sedentary behaviour and sarcopenia in older adults: a systematic review and meta-analysis. BMC Geriatr.23, 877 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sieber, C. C. Malnutrition and sarcopenia. Aging Clin. Exp. Res.31, 793–798 (2019). [DOI] [PubMed] [Google Scholar]
- 5.Güngör, Ö., Güzel, F. B., Ulu, S. & Zadeh, K. K. Sarcopenia in patients with chronic kidney disease. Turk. J. Nephrol.31, 3–6 (2022). [Google Scholar]
- 6.Yu, J. et al. Associations between mild cognitive impairment and whole blood zinc and selenium in the elderly cohort. Biol. Trace Elem. Res.201, 51–64 (2023). [DOI] [PubMed] [Google Scholar]
- 7.Garza, N. M., Swaminathan, A. B., Maremanda, K. P., Zulkifli, M. & Gohil, V. M. Mitochondrial copper in human genetic disorders. Trends Endocrinol. Metab.34, 21 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McCann, C. et al. The mitochondrial cu + transporter PiC2 (SLC25A3) is a target of MTF1 and contributes to the development of skeletal muscle in vitro. Front. Mol. Biosci.9, 1 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Whitlow, T. J. et al. Regulation of Atp7a RNA contributes to differentiation-dependent Cu redistribution in skeletal muscle cells. Met. Integr. Biometal Sci.15, mfad042 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lu, H. H., Ege, D., Salehi, S. & Boccaccini, A. R. Ionic medicine: exploiting metallic ions to stimulate skeletal muscle tissue regeneration. Acta Biomater.10.1016/j.actbio.2024.10.033 (2024). [DOI] [PubMed] [Google Scholar]
- 11.Qi, Z., Duan, A. & Ng, K. Selenoproteins in health. Molecules29, 136 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Loflin, J., Lopez, N., Whanger, P. D. & Kioussi, C. Selenoprotein W during development and oxidative stress. J. Inorg. Biochem.100, 1679–1684 (2006). [DOI] [PubMed] [Google Scholar]
- 13.Petit, N. et al. Selenoprotein N: an endoplasmic reticulum glycoprotein with an early developmental expression pattern. Hum. Mol. Genet.12, 1045–1053 (2003). [DOI] [PubMed] [Google Scholar]
- 14.Hosseini, R., Ferns, G. A., Sahebkar, A., Mirshekar, M. A. & Jalali, M. Zinc supplementation is associated with a reduction in serum markers of inflammation and oxidative stress in adults: a systematic review and meta-analysis of randomized controlled trials. Cytokine138, 155396 (2021). [DOI] [PubMed] [Google Scholar]
- 15.Mnatsakanyan, H., Serra, R. S., i, Rico, P. & Salmerón-Sánchez, M. Zinc uptake promotes myoblast differentiation via Zip7 transporter and activation of akt signalling transduction pathway. Sci. Rep.8, 13642 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Deng, M. G. et al. Associations of serum zinc, copper, and selenium with sleep disorders in the American adults: data from NHANES 2011–2016. J. Affect. Disord.323, 378–385 (2023). [DOI] [PubMed] [Google Scholar]
- 17.Yang, W. et al. The daily intake levels of copper, selenium, and zinc are associated with osteoarthritis but not with rheumatoid arthritis in a cross-sectional study. Biol. Trace Elem. Res.201, 5662–5670 (2023). [DOI] [PubMed] [Google Scholar]
- 18.Giacconi, R. et al. Reduced levels of plasma selenium are associated with increased inflammation and cardiovascular disease in an Italian elderly population. Exp. Gerontol.145, 111219 (2021). [DOI] [PubMed] [Google Scholar]
- 19.Reddy, S. S., Addi, U. R., Pullakhandam, R. & Reddy, G. B. Dietary zinc deficiency disrupts skeletal muscle proteostasis and mitochondrial biology in rats. Nutrition98, 111625 (2022). [DOI] [PubMed] [Google Scholar]
- 20.Chen, K., Zhou, J., Liu, N. & Meng, X. Association of serum concentrations of copper, selenium, and zinc with grip strength based on NHANES 2013–2014. Biol. Trace Elem. Res.202, 824–834 (2024). [DOI] [PubMed] [Google Scholar]
- 21.Gbemavo, M. C. J. & Bouchard, M. F. Concentrations of lead, mercury, selenium, and manganese in blood and hand grip strength among adults living in the United States (NHANES 2011–2014). Toxics9, 189 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Meng, S. J. & Yu, L. J. Oxidative stress, molecular inflammation and sarcopenia. Int. J. Mol. Sci.11, 1509–1526 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Picca, A. & Calvani, R. Molecular mechanism and pathogenesis of Sarcopenia: an overview. Int. J. Mol. Sci.22, 3032 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhao, J. et al. Higher systemic immune-inflammation index is associated with Sarcopenia in individuals aged 18–59 years: a population-based study. Sci. Rep.13, 22156 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pereira, T. C. B., Campos, M. M. & Bogo, M. R. Copper toxicology, oxidative stress and inflammation using zebrafish as experimental model. J. Appl. Toxicol.36, 876–885 (2016). [DOI] [PubMed] [Google Scholar]
- 26.Ciaffaglione, V. & Rizzarelli, E. Carnosine, zinc and copper: a menage a trois in bone and cartilage protection. Int. J. Mol. Sci.24, 16209 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang, H. et al. Circulating level of blood iron and copper associated with inflammation and disease activity of rheumatoid arthritis. Biol. Trace Elem. Res.201, 90 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Khadim, R. M. & Al-Fartusie, F. S. Evaluation of some trace elements and antioxidants in sera of patients with rheumatoid arthritis: a case–control study. Clin. Rheumatol.42, 55–65 (2023). [DOI] [PubMed] [Google Scholar]
- 29.Guo, C. H., Chen, P. C., Yeh, M. S., Hsiung, D. Y. & Wang, C. L. Cu/Zn ratios are associated with nutritional status, oxidative stress, inflammation, and immune abnormalities in patients on peritoneal dialysis. Clin. Biochem.44, 275–280 (2011). [DOI] [PubMed] [Google Scholar]
- 30.Kalita, J., Kumar, V., Misra, U. K. & Bora, H. K. Movement disorder in copper toxicity rat model: role of inflammation and apoptosis in the corpus striatum. Neurotoxicol. Res.37, 904–912 (2020). [DOI] [PubMed] [Google Scholar]
- 31.Yang, Y. et al. Association of urinary phthalate metabolites with Sarcopenia in US adults: NHANES 1999–2006. Environ. Sci. Pollut. Res.29, 7573–7582 (2022). [DOI] [PubMed] [Google Scholar]
- 32.You, Y. et al. The association between sedentary behavior, exercise, and sleep disturbance: a mediation analysis of inflammatory biomarkers. Front. Immunol.13, 1080782 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nishikawa, H. et al. Serum zinc concentration and sarcopenia: a close linkage in chronic liver diseases. J. Clin. Med.8, 336 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen, Z. et al. Association between serum copper levels and muscle mass: results from NHANES 2011–2016. Environ. Sci. Pollut. Res.31, 6847–6856 (2024). [DOI] [PubMed] [Google Scholar]
- 35.Wang, Y. et al. Association between blood metals mixture and chronic kidney disease in adults: NHANES 2013–2016. J. Trace Elem. Med. Biol.83, 127395 (2024). [DOI] [PubMed] [Google Scholar]
- 36.Hasani, M. et al. Beneficial effects of Se/Zn co-supplementation on body weight and adipose tissue inflammation in high‐fat diet‐induced obese rats. Food Sci. Nutr.9, 3414 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cheng, B. J. et al. Selenium attenuates the association of co-exposure to arsenic, cadmium, and lead with cognitive function among Chinese community-dwelling older adults. Environ. Sci. Pollut. Res.30, 36377–36391 (2023). [DOI] [PubMed] [Google Scholar]
- 38.Pan, L. et al. Inflammation and sarcopenia: a focus on circulating inflammatory cytokines. Exp. Gerontol.154, 111544 (2021). [DOI] [PubMed] [Google Scholar]
- 39.Cao, P. R., Kim, H. J. & Lecker, S. H. Ubiquitin–protein ligases in muscle wasting. Int. J. Biochem. Cell. Biol.37, 2088–2097 (2005). [DOI] [PubMed] [Google Scholar]
- 40.Ogura, Y., Mishra, V., Hindi, S. M., Kuang, S. & Kumar, A. Proinflammatory cytokine tumor necrosis factor (TNF)-like weak inducer of apoptosis (TWEAK) suppresses satellite cell self-renewal through inversely modulating notch and NF-B signaling pathways. J. Biol. Chem.288, 35159–35169 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Guo, H. et al. Copper promotes LPS-induced inflammation via the NF-кB pathway in bovine macrophages. Biol. Trace Elem. Res.10.1007/s12011-024-04107-6 (2024). [DOI] [PubMed] [Google Scholar]
- 42.Deng, H. et al. The dysregulation of inflammatory pathways triggered by copper exposure. Biol. Trace Elem. Res.201, 539–548 (2023). [DOI] [PubMed] [Google Scholar]
- 43.Genome-wide analysis of. Transcription and photosynthesis inhibition in the harmful dinoflagellate Prorocentrum minimum in response to the biocide copper sulfate. Harmful Algae57, 27–38 (2016). [DOI] [PubMed] [Google Scholar]
- 44.Influence of CuSO. 4 and chelated copper algaecide exposures on biodegradation of microcystin-LR. Chemosphere174, 538–544 (2017). [DOI] [PubMed] [Google Scholar]
- 45.Pastorelli, A. A. et al. Human exposure to lead, cadmium and mercury through fish and seafood product consumption in Italy: a pilot evaluation. Food Addit. Contam. Part. Chem. Anal. Control Expo Risk Assess.29, 1913–1921 (2012). [DOI] [PubMed] [Google Scholar]
- 46.Tian, F. et al. Protective effects of Lactobacillus plantarum CCFM8246 against copper toxicity in mice. PLoS ONE10, e0143318 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Avery, J. C. & Hoffmann, P. R. Selenium, selenoproteins, and immunity. Nutrients10, 1203 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kieliszek, M. & Błażejak, S. Current knowledge on the importance of selenium in food for living organisms: a review. Molecules21, 609 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fernández-Lázaro, D. et al. The role of selenium mineral trace element in exercise: antioxidant defense system, muscle performance, hormone response, and athletic performance. A systematic review. Nutrients12, 1790 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hariharan, S. & Dharmaraj, S. Selenium and selenoproteins: it’s role in regulation of inflammation. Inflammopharmacology28, 667–695 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liang, Y. et al. Moderate selenium mitigates hand grip strength impairment associated with elevated blood cadmium and lead levels in middle-aged and elderly individuals: insights from NHANES 2011–2014. Front. Pharmacol.14, 1 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Himoto, T. & Masaki, T. Associations between zinc deficiency and metabolic abnormalities in patients with chronic liver disease. Nutrients10, 88 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lukaski, H. C. Vitamin and mineral status: effects on physical performance. Nutrition20, 632–644 (2004). [DOI] [PubMed] [Google Scholar]
- 54.Krotkiewski, M., Gudmundsson, M., Backström, P. & Mandroukas, K. Zinc and muscle strength and endurance. Acta Physiol. Scand.116, 309–311 (1982). [DOI] [PubMed] [Google Scholar]
- 55.Heiderscheit, K. J. & Hansen, S. L. Effect of increasing zinc supplementation on post-transit performance, behavior, blood and muscle metabolites, and gene expression in growing beef feedlot steers. J. Anim. Sci.100, 246 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yang, M., Li, Y., Yao, C., Wang, Y. & Yan, C. Association between serum copper-zinc ratio and respiratory tract infection in children and adolescents. PLoS ONE18, e0293836 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Malavolta, M. et al. Plasma copper/zinc ratio: an inflammatory/nutritional biomarker as predictor of all-cause mortality in elderly population. Biogerontology11, 309–319 (2010). [DOI] [PubMed] [Google Scholar]
- 58.Gaier, E. D. et al. High serum Cu and Cu/Zn ratios correlate with impairments in bone density, physical performance and overall health in a population of elderly men with frailty characteristics. Exp. Gerontol.47, 491–496 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.