Abstract
Alzheimer’s disease and related dementias are a leading cause of morbidity in our aging populations. Although influenced by genetic factors, fewer than 5% of Alzheimer’s disease and related dementia cases are due solely to genetic causes. There is growing scientific consensus that these dementias arise from complex gene by environment interactions. The 2020 Lancet Commission on dementia prevention, intervention, and care identified 12 modifiable risk factors of dementia, including lifestyle, educational background, comorbidities, and environmental exposures to environmental contaminants. In this review, we summarize the current understanding and data gaps regarding the role(s) of environmental pollutants in the etiology of Alzheimer’s disease and related dementias with a focus on air pollution. In addition to summarizing findings from epidemiological and experimental animal studies that link airborne exposures to environmental contaminants to increased risk and/or severity of Alzheimer’s disease and related dementias, we discuss currently hypothesized mechanism(s) underlying these associations, including peripheral inflammation, neuroinflammation and epigenetic changes. Key data gaps in this rapidly expanding investigative field and approaches for addressing these gaps are also addressed.
Introduction
Alzheimer’s disease (AD) and AD-related dementia (ADRD) are global public health concerns. The worldwide prevalence of ADRD is projected to rise from 57.4 million affected individuals in 2019 to 152.8 million by 20501. Alzheimer’s disease and related dementias are the most prevalent type of progressive neurodegenerative disease that is pathologically characterized by amyloid beta plaques (Aβ), phosphorylated neurofibrillary tau tangles (NFT), and persistent neuroinflammation2–5. Clinical studies have identified that AD pathology develops years and often decades prior to the onset of AD symptoms6,7. Mild cognitive impairment is a transitional phase of cognitive decline that progresses to dementia in approximately 30% of cases8,9. Currently, there are no definitive preventative or therapeutic interventions that effectively mitigate AD and ADRD risks or that prevent or reverse cognitive impairment. Given the increasing awareness that much of AD/ADRD, particularly late-onset forms, arises from complex gene × environmental (G × E) interactions, significant research effort is being devoted to identifying environmental risk factors for AD/ADRD and understanding how they modify disease risk and/or severity10–12. This is because currently it is much easier to modify our environment than our genes.
HETEROGENEITY OF ALZHEIMER’S DISEASE AND RELATED DEMENTIAS
Alzheimer’s disease was initially described as a presenile dementia occurring in individuals between 45 and 65 years old, but in 1977 it was determined that the neuropathological findings of amyloid beta (Aβ) plaques and neurofibrillary tangles (NFTs), which are the hallmark pathologies of AD were very similar in AD of early- and late-onset. However, it became increasingly recognized that with aging, late-onset AD brains frequently exhibited additional neuropathological findings, and these became categorized as ADRD. Before discussing environmental and genetic risk factors that contribute to AD and ADRD, it is important to explain terminologies used in this field.
Dementia describes a range of neurological conditions causing progressive deterioration of cognition and can be accompanied by additional neurological dysfunction. Alzheimer’s disease and related dementias are distinguished from other dementing illness by clinical assessment, neuroradiology, neuropathology, and biomarkers. Clinical assessment based on neurological and cognitive functional testing is least sensitive and least accurate at early stages of AD, and clinical confirmation utilizes other diagnostic tools including brain imaging modalities and blood and/or cerebrospinal fluid biomarker testing for amyloid and phosphorylated tau isoforms13. The current evolution of increasingly selective and sensitive biomarkers for AD is necessary to accurately distinguish AD from other dementias and to evaluate treatments for AD/ADRD whose pathology develops decades before clinical symptomatology14. Biomarker selectivity that distinguish between the different types of dementias will greatly assist with the identification of genetic and other specific risk factors, but currently consensus has not been reached on future AD/ADRD classifications. For this review, we are summarizing prior literature based on current AD/ADRD classifications and guidelines.
In the current U.S. National plan to address AD, ADRD includes frontotemporal dementia, Lewy body dementia, vascular contributions to cognitive impairment and dementia, and mixed dementias (including cerebrovascular disease or Lewy bodies)15. Each of these dementias share progressive neuronal loss, cognitive and behavioral decline, impaired daily function, and aberrant protein accumulation (e.g., Aβ, tau, or α-synuclein)16,17. The heterogeneity of AD/ADRD complicates the task of identifying the interplay between specific genetic and environmental risk factors. While it has been easier to include ADRDs with AD when identifying environmental risk factors, it is acknowledged that this inclusive categorization increases the likelihood of “masking” specific G × E interactions that drive individual risk for specific forms of AD/ADRD.
GENETICS OF EARLY AND LATE ONSET ALZHEIMER’S DISEASE
Broadly speaking, there are two age-based classifications of AD: early-onset Alzheimer’s disease (EOAD) and late-onset Alzheimer’s disease (LOAD). Early-onset Alzheimer’s disease is a less common form of AD diagnosed in individuals under the age of 65, often in their 40s and 50s. Late-onset Alzheimer’s disease, which is the more common form of AD is diagnosed after age 65 and frequently includes additional neuropathology. While both forms of AD are highly heritable (~92–100%, and ~70–80%, respectively), they differ in their inheritance pattern18,19 Roughly 90% of EOAD is inherited in an autosomal recessive fashion, and 10% follows an autosomal dominant inheritance pattern. Additional subclassifications for EOAD have emerged, including Mendelian, non-Mendelian or sporadic EOAD; however, in many studies of EOAD, the inheritance patterns are not delineated19. Common autosomal dominant forms of EOAD are due to mutations in APP, PSEN1, and PSEN2, and autosomal recessive AD is linked to mutations in approximately 27 genes, including APOE4, BIN1, TREM2, MAPT, APP, UNC5C, and CLU (Table 1). While genome-wide association studies (GWAS) have identified multiple genes associated with increased risk of AD, the relationship of many of these risk loci to pathogenic mechanisms that drive disease progression have yet to be elucidated. Importantly, the differing clinical profiles of individuals carrying these high-risk alleles is consistent with a role for environmental factors interacting with genetic risks to determine individual outcomes.
Table 1:
Alzheimer’s disease risk genes
| Early-Onset Alzheimer’s disease (EOAD) | Late-Onset Alzheimer’s disease (LOAD) | ||||
|---|---|---|---|---|---|
| Autosomal Dominant EOAD | Autosomal Recessive/Sporadic EOAD | ABCA7135,307,311–314 | TMEM106B311,313,314 | PRKD3313,314 | NTN5311 |
| BIN1135,307,311–314 | TNIP1311,313,314 | RASGEF1C313,314 | NYAP1307 | ||
| CASS4135,307,311–314 | ABCA1313,314 | RBCK1313,314 | LINC02705307 | ||
| CD2AP135,307,311–314 | ADAM17313,314 | RHOH313,314 | LINC02695307 | ||
| APP300 | APOE4302,303 | CR1135,307,311–314 | ADAMTS1313,314 | SCIMP135,314 | RNU6–560P307 |
| PSEN1300,301 | TREM2304,305 | SORL1135,307,311–314 | ANK3313,314 | SCIMP/RABEP311,313 | USP6NL-AS1307 |
| PSEN2300,301 | SORL1302 | TREM2135,307,311–314 | ANKH313,314 | SEC61G313,314 | ADAMTS4135 |
| PRNP301 | CLU307,311–314 | BLNK313,314 | SIGLEC11313,314 | HESX1135 | |
| MAPT306 | EPHA1135,307,312–314 | COX7C313,314 | SLC2A4RG313,314 | HS3ST1135 | |
| GRN307 | FERMT2307,311–314 | CTSB313,314 | SNX1313,314 | CNTNAP2135 | |
| C9orf72308 | ABI3135,311,313,314 | CTSH313,314 | SORT1313,314 | CLU/PTK2B135 | |
| NOTCH3309 | ADAM10311–313 | DOC2A313,314 | SPI1307,314 | BZRAP1-AS1135 | |
| PARK2306 | APH1B135,311,313,314 | FOXF1313,314 | SPPL2A313,314 | ||
| PLCG2300 | INPP5D307,312–314 | HS3ST5313,314 | TPCN1313,314 | SUZ12P1135 | |
| ABI3300 | PICALM135,311–313 | ICA1313,314 | TSPAN14313,314 | ALPK2135 | |
| APP300 | PTK2B307,312–314 | IDUA313,314 | TSPOAP1313,314 | AC074212.3135 | |
| PSEN1300,301 | ACE311,313,314 | IL34313,314 | UMAD1313,314 | CLNK/HS3ST1313 | |
| PSEN2300,301 | APOE135,307,311 | INPPD5311 | WDR12313,314 | ||
| SORL1300,302 | APP311,313,314 | JAZF1313,314 | WDR81313,314 | HLA313 | |
| ABCA7300 | CD33135,311,312 | KAT8313 | ZCWPW1135,312 | CELF1/SPI1313 | |
| TREM2300 | CLNK135,311,314 | KLF16313,314 | ZCWPW1/NYAP1311,313 | HLA-DQA1314 | |
| BIN1300 | ECHDC3135,313 | MAF313,314 | UNC5CL314 | ||
| UNC5C300 | GRN311,313,314 | MME313,314 | AGRN311 | TREML2314 | |
| AKAP9300 | HLA-DRB1135,311 | MS4A4A313,314 | HAVCR2311 | EPDR1314 | |
| NOTCH3300 | LILRB2311,313,314 | MYO15A313,314 | EPHA1-AS311 | SPDYE3314 | |
| 12s rRNA300 | MS4A6A135,312 | NME8313,314 | USP6NL/ECHDC3311 | USP6NL314 | |
| CLU300 | NCK2311,313,314 | PLCG313,314 | CCDC6311 | EED314 | |
| PLCG2300 | SHARPIN311,313,314 | PLEKHA1313,314 | MADD/SPI1311 | MINDY2314 | |
| ABI3300 | SLC24A4135,311,314 | PRDM7313,314 | RIN3311 | BCKDK314 | |
| APOE300 | TSPOAP1-AS1311 | WNT3314 | |||
| TET1310 | AKAP9312 | ||||
| TET2276 | UNC5C312 | ||||
The genetic contributions to LOAD are significantly more complex and include roughly 117 associated genes (Table 1). Overall, the heritability of LOAD is less than that of EOAD and findings from twin studies indicate a greater contribution of environmental factors in the development and progression of disease. A significant number of individuals with LOAD have co-morbid medical conditions, such as cardiovascular disease, type 2 diabetes20, autoimmune disorder21, or head trauma22, each of which is separately associated with cognitive decline and each of which is heavily influenced by environmental factors. In addition to age-dependent increases in Aβ plaques and NFT, as many as 50% of LOAD brains contain pathologies of cerebral amyloid angiopathy, TDP-43 inclusions (LATE-NC) and/or Lewy body pathology (α-synuclein), which increase with age and AD progression23. The presence of cerebrovascular cerebral amyloid angiopathy correlates with both Aβ plaques and NFT and is associated with expression of the APOE ε4 allele independent of dementia status. Cerebral amyloid angiopathy, like AD, is a consequence of Aβ accumulation, but the intracerebral vascular accumulation produces a separate disorder with increased risk of stroke, cerebral hemorrhages, and inflammatory encephalopathies24,25 with its own distinct genetic risk factors26 and diagnostic and potential clinical therapeutics. Given the modest risks conferred by each of these genetic mutations, it has been argued that LOAD, as well as most of ADRD, are driven by multifactorial influences, including multiple genes interacting with diverse environmental factors. Interestingly, many of the pathogenic mechanisms associated with these genetic risk alleles have been shown to be independently modified by environmental factors.
GENE BY ENVIRONMENT INTERACTION IN ALZHEIMER’s DISEASE AND RELATED DEMENTIAS ETIOLOGY
Twin studies have provided further evidence in support of the contribution of environmental factors to AD/ADRD etiology27,28. Specifically, genetically identical monozygotic twins have been compared to explore the effects of non-shared environmental risk factors. In one twin study, positron emission tomography (PET) imaging to quantify tau deposition in the entorhinal cortex and neocortical brain regions, revealed similar intensity and deposition of tau pathology27, suggesting that genetics was the predominant driver. However, when non-shared environmental risk factors were compared in twin pairs, differences in tau deposition strongly correlated with differences in depressive behaviors, social isolation, and physical inactivity27. Similarly, another study of monozygotic twins found that individuals exposed to higher levels of air pollution had lower structural integrity of the locus coeruleus, which is a brain region involved in the early stages of AD28. These results suggest that while similarities in the development of AD-relevant pathologies between monozygotic twin pairs may be explained by identical genetic composition, different trajectories of ADRD pathologies may arise from exposure to non-shared environmental factors. In a different study, analysis of the AD polygenic risk score in monozygotic and dizygotic twins suggested that total genetic contribution to AD risk accounted for 71%, while the remaining 29% was attributed to environmental factors29. This finding can help explain cases of discordant development of ADRD pathologies in monozygotic twins despite their genetic uniformity30 and highlights the importance of identifying and studying the impact of environmental risk factors that can modify ADRD outcomes (Figure 1).
Figure 1. Gene by environment interactions.
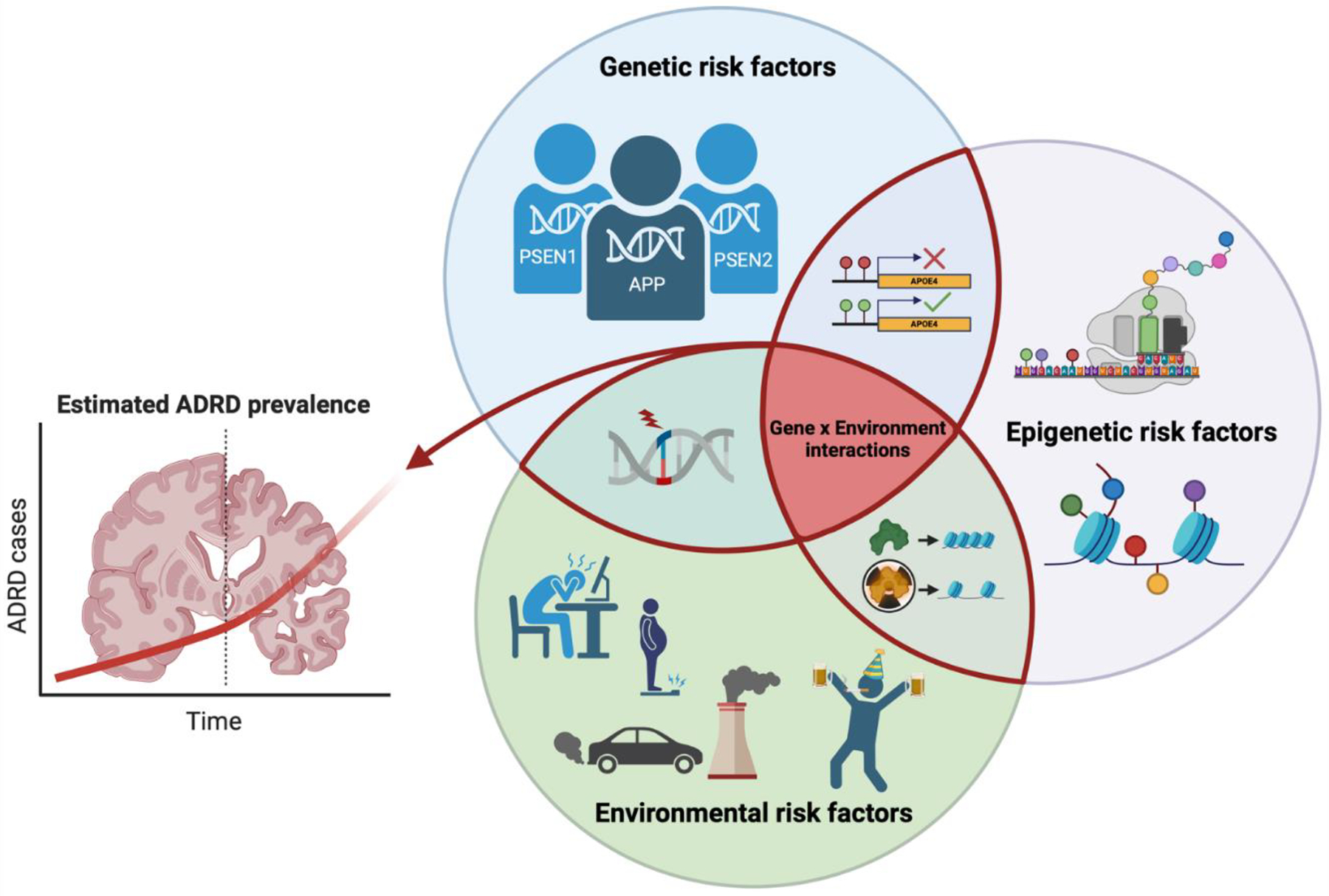
Genetic, epigenetic, and environmental risk factors interact to promote ADRD etiology and disease progression. NOTE: ADRD, Alzheimer’s disease-related dementias. Created with BioRender.com.
Environmental risk factors for Alzheimer’s disease and related dementias
The 2020 Lancet Commission on dementia prevention, intervention, and care identified 12 modifiable environmental AD/ADRD risk factors based on meta-analyses and systemic reviews of the available literature12. The report highlighted that these 12 modifiable risk factors contributed to 40% of global prevalence of dementia, suggesting that 40% of dementia cases worldwide might potentially be prevented or slowed by modifying individual exposures12. The risk factors identified by the Lancet Commission included various comorbid medical conditions (hypertension, diabetes, hearing impairment, obesity, depression, and traumatic brain injury), lifestyles (physical inactivity, excessive alcohol consumption, social isolation, smoking), less educational background, and exposure to air pollution. Medical and public health strategies targeting comorbidity or lifestyles heavily depend on individual motivation, and these applications have been limited in success at the population level31. Many of these factors are strongly influenced by environmental contaminants, and environmental exposures represent a class of risk factors that can be changed by public policy, and thus have population-level impacts31.
The contribution of environmental contaminants to the development of neurodegenerative diseases has been primarily studied in the context of occupational exposure to pesticides among agricultural workers.32 Individuals occupationally exposed to pesticides, such as organophosphate insecticides33–35 and fungicides that contain manganese36–38, were reported to have lower cognitive function and increased risk of developing neurodegenerative diseases, including AD33,39. Other synthetic persistent organic pollutants like per-and polyfluoroalkyl substances (PFAS) have also been associated with increased AD-relevant pathology and cognitive impairment in ADRD patients40. More recently, air pollution (Box 1) has garnered significant interest due to increasing evidence from epidemiological and experimental animal studies of a strong association between air pollution and AD/ADRD. Additionally, while air pollution is directly linked to increased risk for AD/ADRD, it is also linked to a number of other ADRD risk factors, such as cardiovascular disease41–46, metabolic dysfunction47, and physical inactivity48. The links between air pollution, AD/ADRD, and other risk factors suggest that air pollution may be an upstream environmental modifier that influences AD/ADRD onset and progression. It has been argued that targeting causative upstream environmental modifiers will have a broader and more significant impact on reducing the AD burden than modifying proximal individual-level risk factors49. This review focuses on current understanding and findings regarding the association between air pollution and increased risk and/or severity of AD/ADRD and discusses prevailing hypotheses regarding the mechanisms underlying these links.
Box 1. Air pollution composition, toxicology and regulations.
Six criteria air pollutants are monitored and regulated by the U.S. Environmental Protection Agency National Ambient Air Quality Standards and the World Health Organization Air Quality Guidelines. These include: (1) coarse and fine particulate matter (PM10, PMzs); (2) nitrogen dioxide (NOz); (3) ground-level ozone (03): (4) carbon monoxide (CO); (5) sulfur dioxide (SO2); and (6) lead (Pb) (Figure 2). The composition and toxicity of these pollutants are determined in large part by their sources.222 Sources of these pollutants include industrial pollution, near roadway traffic-related air pollution (TRAP), wildfire, and agriculture51,60,317,318.
Amongst the six criteria air pollutants, PM has been most intensively monitored and studied as a health hazard because it deposits in the respiratory system, and the finer particles can enter the systemic circulation and be distributed to all organs in the body, including the brain219,222,224,319–321. Particulate matter ranges in size from coarse (PM10 with an aerodynamic diameter <10 μm) to fine (PM2.5, diameter <2.5 μm) to ultrafine (UFPM, diameter <0.1 μm) (Figure 2). Fine PM0.1–2.5 are considered more toxic than coarse PM10 because of a higher penetrance into the lung parenchyma with smaller sizes. Fine PM2.5 from agriculture, TRAP, coal combustion and wildfire were found to be significant risk modifiers for dementia, including AD49,51,221,318,322,323. A wide range of organic and inorganic compounds from different sources of agriculture, wildfire, and TRAP can adsorb to PM2.5 and cause negative health outcomes in the lung and the brain.124,324,325 Likewise, the composition of gases in polluted air also depends on the source of emission. Traffic-related air pollution is one of the main sources of NOx, CO, benzene, and other volatile organic compounds (Figure 2), yet industrial boilers, gas stoves, and other fuel combustion processes also contribute to emissions of these gases. Traffic-related air pollution as a source of SO2 emissions has declined with the global transition to using low sulfur fuels for automobiles326. Yet, other sources like wildfire smoke, industrial sources, and ship emissions still contribute to the release of SO2327. Ground-level O3 is a gaseous secondary air pollutant that forms from photochemical reactions with radicals derived from NOx and volatile organic compounds in the warm ambient atmosphere328.
Epidemiological evidence linking air pollution with progressive cognitive impairment and Alzheimer’s disease and related dementias
Over 80 epidemiological studies investigating an association between air pollution and AD/ADRD were published in the peer-reviewed literature during the last six years (Table 2). Recent studies have focused on correlations between independent and combined effects of specific air pollutants and AD/ADRD risk. Zip code-based residential exposure levels of fine and course particulate matter (PM2.5, PM10), nitrogen oxides (NOx), and ground-level ozone (O3), have been leveraged to calculate odds ratios or hazard ratios to assess the risk for AD/ADRD conferred by exposure to air pollutants, with increasing risk associated with increasing levels of each air pollutant. Longitudinal monitoring studies of average air pollution exposure levels have also been leveraged to measure the time-lagged effects of long-term exposure to air pollution on cognitive decline and AD/ADRD50–54. The cohort sizes ranged from small population-based studies (n=150–8,000) to nationwide studies (n=4–12 million) of midlife to aged participants (age > 45). The main ADRD outcomes that were measured against air pollution exposure levels were risks of incident ADRD determined by clinical records of diagnosis, hospitalization, pathology of AD-relevant biomarkers, cognitive assessment, and mortality.
Table 2:
Summary of epidemiological studies testing the association between air pollutants and Alzheimer’s disease-related dementias
| Air pollutant | AD-relevant clinical pathology | Cognitive impairment | ADRD risk |
|---|---|---|---|
| PM2.5 |
↑ 5 studies reported positive association53,54,104,106,111 ∅ 1 study reported no association105 |
↑13 studies reported positive association51,81,106,115–121,124,314,315 ∅ 4 studies reported no association98,105,114,125 |
↑ 36 studies reported positive association50–52,58–90 ∅ 5 studies reported no association95–99 |
| PM10 | ↑ 4 studies reported positive association54,104–106 |
↑ 4 studies reported positive association106,121,123,314 ∅ 3 studies reported no association98,105,125 |
↑ 9 studies reported positive association67,70–72,80,82,85,87,100 ∅ 1 study reported no association98 |
| NOx | ↑ 4 studies reported positive association54,104–106 |
↑ 6 studies reported positive association106,118,121,123,314,315 ∅ 4 studies reported no association98,105,114,125 |
↑ 28 studies reported positive association50,58,59,61–63,66,67,69–73,76–80,82–87,90–93 ∅ 8 studies reported no association52,75,94–99 |
| O3 | ∅ 1 study reported no association53 |
↑ 1 study reported positive association120 ∅ 1 study reported no association123 |
↑ 3 studies reported positive association58,82,93 ∅ 6 studies reported no association58,61,76,87,94,95 |
| Black carbon |
↑ 4 studies reported positive association76,79,87,90 ∅ 4 studies reported no association52,96,97,99 |
NOTE: AD, Alzheimer’s Disease; ADRD, Alzheimer’s disease-related dementias; AD-relevant clinical pathology includes Aβ 42/40 and neurofilament light levels in the blood and cerebrospinal fluid, and brain PET/MRI scans for amyloid, tau, and cortical thickness detection.
↑: positive association reported; ∅: no association reported.
SUMMARY OF FINDINGS
In line with earlier epidemiology studies55–57, the main consensus of current epidemiological findings is that there is a strong association between exposure to air pollution and AD/ADRD; however, there were mixed results regarding associations between air pollutant type and increased risk of AD/ADRD. The majority of studies found a positive correlation between risk of all-cause dementia and exposure to PM2.550–52,58–90 and NOx50,58,59,61–63,66,67,69–73,76–80,82–87,90–93 but not to O358,61,76,87,94,95 (table 2). A few studies reported no significant association between risk of dementia and levels of PM2.595–99 and NOx52,75,94–99; and a small number of studies found a positive association between O3 level and risk of dementia58,82,93.
When records of dementia diagnoses are more specifically stratified, differential correlations between various types of air pollutants and subtypes of dementia emerge. In an analysis of data from the UK Biobank, authors reported that exposure to combined levels of NOx, PM2.5, and PM10 were significantly associated with all-cause dementia, AD, vascular dementia (VaD), and mild cognitive impairment. Yet, when each air pollutant was compared separately, VaD was no longer associated with PM2.5,10 but only influenced by NOx, while all-cause dementia, AD, and mild cognitive impairment remained significantly associated with both PM2.5,10, and NOx83. Another study leveraging the UK Biobank data found similar associations of PM2.5,10, and NOx with all-cause dementia and AD but not with VaD66. In line with these findings, other studies reported the association of PM2.5 to be more pronounced with AD than with non-AD or VaD61,64,65,74, but contrary results showed more significant association with non-AD or VaD than with AD82,84,100. These mixed findings may reflect differences across studies in modeling methods, cohort characteristics, exposure duration, confounding co-exposures, source of air pollution data, and dementia ascertainments. Due to the heterogeneity of dementia etiology and disease progression, inconsistent clinical diagnoses and records may have been used across different studies with participants from a wide range of cohorts. Additionally, the outcome measurements mainly derived from medical records may not be an accurate representation of the entire population because of underlying socioeconomic disparities in quality and accessibility of medical care101,102. This could be a critical confounding factor because studies have shown that social disadvantage can increase vulnerability to air pollution and air pollution-mediated risk of AD/ADRD101,103.
Despite these mixed results, long-term exposure to higher levels of PM2.5 and NOx was still consistently found to be associated with increased risk of AD and ADRD in most studies (Table 3). Long-term exposure to higher levels of PM2.5 and NOx was associated with greater extent of AD-relevant structural changes in the brain, which were assessed using PET and magnetic resonance imaging (MRI) to quantify amyloid deposition and cortical atrophy28,53,104–107. Individuals who were exposed to higher levels of PM2.5 and NO2 exhibited higher amyloid PET positivity and cortical atrophy53,104–107. In smaller cohorts, AD-relevant biomarkers, such as Aβ 42/40 and neurofilament light levels in the plasma and cerebrospinal fluid, were positively associated with PM2.5 and NOx exposure levels54,104,108. There were a few post-mortem autopsy studies that reported increased expression of histological biomarkers of AD/ADRD, including hyperphosphorylated tau, Aβ deposition, nanoparticle inclusions, and glial activation in the hippocampus, cortex, and olfactory bulbs of young and old individuals who lived in areas with high levels of air pollution109–113. Another outcome measurement was cognitive performance, which was assessed by a battery of cognitive tests to quantify memory, verbal fluency, and executive functions114–118. While some studies reported an inverse correlation between cognitive test scores and exposure to PM2.5 and NOx in cognitively unimpaired and impaired populations106,115–124, other studies found the relationship to be not significant98,105,114,125.
Table 3:
Epidemiological studies studying air pollution as a risk factor for Alzheimer’s disease-related dementias
| Air pollutant levels | Participant follow-up duration | Study results | Cohort type/size | Citation/location |
|---|---|---|---|---|
| Annual mean: PM2.5 = 9.3 μg/m3 NO2 = 17.1 ppb O3 = 42.6 ppb |
2000–2018 | PM2.5, NO2 ∝↑ incident AD/ADRD O3 ∅ incident AD/ADRD | Medicare Chronic Conditions Warehouse Age ≥ 65yrs n=12,233,371 |
Shi, et al.58 United States |
| Annual median: PM2.5 = 10.6 μg/m3 NO2 = 18.26 ppb O3 = 46.68 ppb |
2000–2016 | PM2.5, NO2 exposure 8–10yrs prior to diagnosis ∝↑first ADRD hospitalization | Medicare FFS beneficiaries Age > 65yrs n=8,507,437 |
Mork, et al.50 United States |
| 2010 median: PM2.5 = 10 μg/m3 PM10 = 16.1 μg/m3 NOx = 43.4 μg/m3 |
Median 12.01yrs | PM2.5, NOx∝↑ all-cause dementia risk NOx ∝↑ AD risk in individuals with higher genetic risk scores |
UK Biobank Study Age ≥ 50yrs n=437,932 |
Yuan, et al.59 United Kingdom |
| 10-year median: PM2.5 = 11.2 μg/m3 |
1998–2016 | PM2.5 ∝↑ incident dementia | Health and Retirement Study Age > 50yrs n=27,857 |
Zhang, et al.51 |
| 10-year median: PM2.5 = 21.3 μg/m3 NO2 = 32.8 μg/m3 BC = 2.3 10−5/m |
1999–2011 | PM2.5
∝↑ all-cause dementia, AD, and VaD risks NO2, BC ∅ dementia risk |
Three-City Study Age ≥ 65yrs n=7,066 |
Mortamias, et al.52 France |
| Annual mean: TRAP PM2.5 = 0.18 μg/m3 Wood-burning PM2.5 = 0.77 μg/m3 |
1993–2010 | All sources of PM2.5 ∝↑dementia incidence | Betula Study Age > 55yrs n=1,806 |
Oudin, et al.60 Sweden |
| Biennial median PM2.5 range: 2002–2003: 10.09–12.65 μg/m3 2015–2016: 7.4–8.34 μg/m3 |
2016–2018 | PM2.5 ∝↑ amyloid PET scan positivity O3 ∅ amyloid PET scan positivity | IDEAS Study (Imaging DementiaEvidence For Amyloid Scanning) Age ≥ 65yrs n=18,178 |
laccarino, et al.53 United States |
| 1- to 20-year average range: PM2.5 = 5–25 μg/m3 PM10 = 10–40 μg/m3 NO2 = 5–40 ppb |
2000–2008 | PM2.5, PM10, NO2 ∝↑ plasma Aβ1–40, Aβ1–42, Aβ 1–42/1–40 ratios | Ginkgo Evaluation of Memory Study Age ≥ 75yrs n=3,044 |
Hajat, et al.54 United States |
| 5-year mean: PM2.5 = 25.9 μg/m3 PM10 = 49.7 μg/m3 NO2 = 27.0 ppb |
2014–2017 | PM10, NO2
∝↑ MRI AD-like cortical atrophy, PM2.5 ∅ MRI AD-like cortical atrophy, PM2.5, PM10, NO2 ∝↑MRI non-AD-like cortical atrophy, PM10, NO2 ∝↓ MoCA cognitive score |
EPINEF Study (Environmental Pollution-Induced Neurological EFfects) Age ≥ 50yrs n=640 |
Cho, et al.106 Korea |
| Annual mean: PM2.5 = 17.2 μg/m3 PM10 = 37.7μg/m3 NO2 = 57.3 μg/m3 NOx = 99.1 μg/m3 |
2013–2014 | PM10, NO2
∝↓ MRI cortical thickness, PM2.5 ∅ MRI cortical thickness, PM2.5, PM10, NO2 ∅ cognitive function |
ALFA+ Study (Alzheimer and Families) Cognitively unimpaired adults Age 45–74yrs n=958 (cognition) n=228 (MRI) |
Crous-Bou, et al.105 Spain |
| Annual mean: PM2.5 = 17.3 μg/m3 PM10 = 37.9 μg/m3 NO2 = 57.6 μg/m3 |
2013–2014 | PM2.5, NO2
∝↑ PET amyloid, PM2.5, PM10 ∝↑ CSF NFL stronger in APOE-ε4 carriers |
ALFA+ Study Cognitively unimpaired adults Age 57yrs n=156 |
Alemany, et al.104 Spain |
NOTE: AD, Alzheimer’s Disease; ADRD, Alzheimer’s disease-related dementias; VaD, vascular dementia; BC, black carbon; PM, particulate matter; NO2, nitrogen dioxide; NOx, nitrogen oxides; CSF, cerebrospinal fluid; NFL, neurofilament light; MoCA, Montreal Cognitive Assessment; PET, positron emission tomography; MRI, magnetic resonance imaging.
∝↑: positively correlated to, ∝↓: inversely correlated to, ∅: not correlated to.
Orange cells indicate studies that assessed ADRD risks as outcomes; green cells indicate studies that assessed AD-relevant biomarkers as outcomes.
LIMITATIONS
All epidemiological studies are limited by several factors. In the studies described, synergistic effects between air pollution and other genetic59,126, lifestyle87,101,122,126–129, and environmental65,78,101,103,105,128,130–133 risk factors may not have been completely adjusted for in the analyses. Although carrying the APOE ε4 gene is a significant AD risk factor134–136, results from studies assessing whether it has a modifying effects on the association between air pollution and AD are inconclusive: some showed the AD/ADRD risk associated with air pollution exposure to be more pronounced in APOE ε4 carriers59,72,104,116,119,121 while others did not62,66,73,77,104,105. Genetic susceptibilities other than APOE ε4 status that can change the trajectory of AD pathogenesis were not taken into consideration for most studies. Moreover, other lifestyle factors, such as physical activity, exposure to traffic noise, sleeping patterns, and socioeconomic status, all of which are known to influence AD etiology87,129,137–140 may not have been well reflected or adjusted for in most studies. Since the G × E interactions that lead to AD/ADRD progression are complicated and not well understood, it is challenging for epidemiological studies to fully capture the true impact of air pollution on AD/ADRD. Nevertheless, consistent epidemiological findings across different cohorts and countries that show a robust association between air pollution and ADRD provide strong justification for further experimental investigation of the underlying mechanisms by which air pollution increases AD/ADRD risk.
Experimental animal studies: The challenge of replicating human exposures to air pollution
To investigate the impact of air pollution on AD/ADRD pathogenesis, experimental animal studies have primarily used wildtype or transgenic rodent models that express human AD risk genes to determine whether exposure to air pollution decreases the time to onset or the severity of AD-relevant phenotypes.
A major challenge of studying air pollution toxicology in an experimental animal model is replicating human-relevant exposures to air pollution141,142. Most earlier studies employed acute exposures to high concentrations of diesel exhaust particles (DEP) or PM2.5 administered to rodents via intratracheal143 or intranasal144 instillation, or, alternatively, oropharyngeal aspiration145 of ambient PM2.5 samples extracted from filtered media and resuspended in a delivery vehicle. The advantages of these instillation or aspiration exposure routes are that the composition of particles, dosage, and site of delivery are easier to control in testing the toxicity of a known amount and composition of air pollutants in a cost-effective way146. However, there are several critical shortcomings of these exposure paradigms in terms of replicating human exposure, including: (1) inaccurate reproduction of human-relevant exposure route, composition, pattern, and dosage of air pollutants142; and (2) omission of gaseous co-pollutants141. These factors can influence the toxicity of air pollutants and induce inconsistent biological responses even to the same air pollutants142,147,148. Hence, many recent studies have utilized in vivo whole-body inhalation exposures to aerosols of air pollutants to test toxicity in a manner that more closely replicates human exposure149–161. Some studies exposed animals to laboratory-generated aerosols of concentrated PM2.5, UFPM, or O3149–154, while other studies implemented variations of concentrated ambient particle (CAP) systems to collect real-world ambient air and concentrate PM2.5 or UFPM for direct delivery to animals155–158. The CAP systems allow more accurate representation of real-world compositions of PM, yet because CAPs methodology only concentrates particles and not gases, understanding combined effects of PM and gases on biological outcomes is not possible141,159. Lastly, both laboratory-generated and CAP-generated aerosols do not effectively emulate chronic real-life exposure dynamics, which vary hourly, weekly, monthly and seasonally141.
A few in vivo studies have developed methods for collecting ambient polluted air and delivering it to animals unchanged and in real-time at ambient concentrations via whole-body inhalation141,160–162. For example, in one study, ambient traffic-related air pollution (TRAP) was collected from a heavily trafficked freeway tunnel in northern California, USA, for subsequent delivery in real-time to animals housed in an adjacent vivarium141. This study demonstrated that chronic life-time exposure to ambient TRAP exacerbated AD-relevant phenotypes in wildtype Fischer rats and transgenic TgF344-AD rats that expressed human AD risk genes160. Another study in Taipei, Taiwan used the Taipei Air Pollutant Exposure System (TAPES) to sample and deliver outdoor ambient air mixture directly to 3xTg-AD mice housed in an exposure chamber. Transgenic mice exposed to ambient air pollution exhibited increased AD neuropathologies relative to filtered air controls161. These novel exposure methods in rodent models increase the translational value of experimental animal studies and provide corroborative evidence to support the epidemiological findings.
Experimental animal studies: Chronic exposure to air pollution exacerbates Alzheimer’s disease pathology
A number of experimental animal studies have quantified Aβ plaques, NFT, tau phosphorylation, neuronal cell loss, and cognitive behavior deficits in both wildtype and transgenic rodent models after exposure to air pollution (Table 4). In most of these studies, the exposure paradigms were short-term or subchronic exposures to PM2.5 and UFPM that ranged from 2–13 weeks149–152,155–158,161. Only a few studies examined the neurological impacts of chronic exposures, ranging from 5–14 months109,153,154,160,162. Results from these studies are mixed depending on the exposure paradigm and animal model. Studies with more chronic exposure durations (Table 4) more consistently reported air pollution-induced increases in Aβ/NFT loads, tau hyperphosphorylation, microgliosis, astrogliosis, and neuronal atrophy. Exposures longer than 3 months at either concentrated or ambient levels of UFPM, PM2.5, or O3 were shown to increase Aβ plaque load, tau phosphorylation, gliosis, and worsen learning and memory in transgenic AD rodent models109,150,153,157,158,160. Some short-term exposure studies (2–3 weeks) in rodents reported increased Aβ plaques, tau phosphorylation, neuronal atrophy, and memory deficits153,155,163, while others did not observe those changes149,161,164 or observed contrary results of reduced Aβ plaques with increased microgliosis156. The mixed results may be partially attributed to varying degrees of neuroinflammation caused by air pollution, as discussed below.
Table 4:
Summaries of in vivo studies of the effects of air pollution on Alzheimer’s disease neuropathology
| Exposure system | Exposure paradigm | Animal model | Study results | Citation |
|---|---|---|---|---|
|
System: Caldecott Tunnel Exposure Facility (CTFE) Source: TRAP drawn from tunnel bores in Oakland, CA, USA |
Whole-body exposure for 2, 5, 9, and 14 months (7days/week, 24h/day) PM2.5 = 15.6 ± 3.7 μg/m3 |
Male & female TgF344-AD & WT rats (Fisher344) 1-month-old |
↑ Refractive particle deposition in HIP after 5 months ↑ p-Tau in HIP after 14 months ↑ Aβ deposition in CO after 10 months ↑ Neuronal cell death in EC after 14 months ↑ Neuroinflammation in HIP, EC (microglia activation, IL-1 β, TNF-α,) ↓ Learning, memory after 14 months ∅ Serum cytokine |
Patten, et al.160,162 |
|
System: Taipei Air Pollutant Exposure System (TAPES) Source: Outdoor ambient air drawn from Taipei, Taiwan |
Whole-body exposure for 3 months (7days/week, 24h/day) PM2.5 = 11.38 μg/m3 |
Female 3xTg-AD mice 6-month-old |
↑ p-Tau, oxidative stress (MDA) in HIP, OB ↑ Neuronal cell death in EC ∅ Aβ−42 deposition, learning |
Lee, et al.161 |
|
System: Harvard ultrafine concentrated ambient particle system (HUCAPS) Source: TRAP UFPM drawn from Rochester, NY, USA |
Whole-body exposure for 2 weeks (4days/week, 4h/day) UFPM = 57 μg/m3 Mean UFPM size = 79 nm |
Male 3xTgAD & WT mice 12.5-month-old |
↓ Learning, memory ↓ olfactory discrimination |
Jew, et al.155 |
|
System: Harvard ultrafine concentrated ambient particle system (HUCAPS) Source: Ambient UFPM drawn from street |
Whole-body exposure for 2 weeks (4days/week, 4h/day) UFPM = 42 ± 15.7 μg/m3 |
Male 3xTgAD & WT mice 12.5–14-month-old |
↑ p-Tau (pT205) in HIP ∅ Aβ deposition in HIP ↓ Aβ deposition in subiculum ↑ Microglial ramification in HIP |
Herr, et al.156 |
|
System: Versatile Aerosol Concentration and Enrichment System (VACES) Source: TRAP UFPM drawn from Orange County, CA, USA |
Whole-body exposure for 12 weeks (4days/week, 5h/day) UFPM = 65.4 μg/m3 |
Male and female APPNL-G-f/+-Kl & WT mice (C57/BL6) 3-, 9-month-old |
↓ Learning, memory in both young and aged mice ↑ Aβ deposition in CO, HIP ↑ Neuroinflammation in CO only in old mice (microglia & astrocyte activation) ∅ Neuronal atrophy |
Kilian, et al.157 |
|
System: Versatile Aerosol Concentration and Enrichment System (VACES) Source: PM2.5 from Columbus, OH, USA |
Whole-body exposure for 3 months (5days/week, 6h/day) PM2.5 = 25.8 μg/m3 |
Male APP/PS1 & WT mice (C57BU6;C3H) 12-month-old |
↑ Neuroinflammation in HIP (microglia & astrocyte activation; TNF-α, IL-1β, IL-6, MIP-3α, IFN-γ) ↑ Aβ deposition, β-secretase, γ-secretase |
Sahu, et al.158 |
|
System: Chambers housed outdoor Source: Ambient outdoor air in Santiago, Chile |
Whole-body exposure for 7 months (7days/week, 24h/day) PM2.5 > 55 μg/m3 |
Female C57/BL6 mice 2-month-old |
↑ DNA double-strand break marker (γ-H2AX) ↑ p-Tau in CO |
Calderon-Garciduenas, et al.109 |
|
System/source: Soot UFPM generated by a Combustion Aerosol Standard burner |
Whole-body exposure for 2 weeks (5days/week, 4h/day) UFPM = 7.0×105 particles/cm3 Mean UFPM size = 39 nm |
Female 5xFAD & WT mice (C57BL/6J) 6-month-old |
↑ Inflammation (IFN-γ in plasma, OB, HIP) ↑ Oxidative stress in HIP (SOD2 protein) ∅ Aβ deposition or microglial and astrocytic activation in HIP and CO |
Saveleva, et al.149 |
|
System/source: UFPM generated by a PM generator |
Whole-body exposure for 3 weeks (5days/week, 8h/day) UFPM = 1000 μg/m3 Mean UFPM size = 178 ± 65 nm |
Male WT mice (C58BL6) 12-month-old |
↑ Oxidative stress (4-HNE) in HIP ↑ Aβ deposition, neuroinflammation (TNF-a) in HIP ∅ Neuronal cell death in HIP |
Park, et al.152 |
|
System/source: O3 generated by an HFL-10 O3 generator |
Whole-body exposure for 13 weeks (3days/week, 4h/day) O3 = 1.0 ppm |
Male 5xFAD & WT mice (C57BL/6J) 10–11-week-old |
↑ Aβ deposition in HIP and CO ↓ Plaque-associated microglia, ↓ TREM2 on plaque-associated microglia in CO ↑ Neuroinflammation in CO (NLRP3, IL-1 β) ↑ Neurite dystrophy (↑LAMP1, ↓VaChT, ChAT, AChE) in CO |
Greve, et al.150 |
|
System/source: O3 generated by an HFL-10 O3 generator |
Whole-body exposure for 13 weeks (3days/week, 4h/day) O3 = 1.0 ppm |
Male 5xFAD mice (C57BL/6J) 10–11-week-old |
↑ Aβ deposition in CO ↑ Astrocyte, astrocyte-microglia interaction in peri-plaque regions ↑ Neuroinflammation in CO (DAM astrocytic profile) |
Ahmed, et al.151 |
|
System/source: O3 generated by a flux ozone generator |
Whole-body exposure for 7, 15, 30, 60, and 90 days (7days/week, 4h/day) O3 = 0.25ppm |
Male WT rats (Wistar) 250–300g |
↑ Aβ structural destabilization overtime (↓a-helix, ↑β-sheet) ↑ Intracellular Aβ 1–42 after 60–90 days ↑ Lipid peroxidation, microglial and astrocytic activation in HIP after 15–90 days ↑ Neuronal cell death in HIP after 90 days |
Rivas-Arancibia, et al.153,154 |
NOTE: HIP, hippocampus; CO, cortex; EC, entorhinal cortex; p-Tau, phosphorylated tau; WT, wildtype.
↑: increase in measurement; ↓: decrease in measurement; ∅: no change in measurement.
Blue cells indicate studies that used air pollutants collected from real-world sources; yellow cells indicate studies that used air pollutants generated in the laboratory.
Prevailing hypothesis: Air pollution promotes Alzheimer’s disease-related dementias phenotypes via inflammatory mechanisms
CHRONIC NEUROINFLAMMATION IN ALZHEIMER’S DISEASE PATHOGENESIS
Persistent neuroinflammation has been posited as a key mechanism responsible for progressive neurodegenerative diseases, including ADRD165, based on accumulating evidence from human post-mortem166–168 and experimental animal studies169,170. Aggregates of misfolded proteins like Aβ and NFT, which are neurotoxic, have been shown to trigger neuroinflammation171,172. Neuroinflammatory responses are mainly orchestrated by resident brain immune cells – microglia and astrocytes – that protect the brain microenvironment not only by phagocytosing cellular damage, but also regulating neurogenesis and synapse number173,174. While microglia-mediated amyloid clearance169,175 and pruning of axosomatic inhibitory synapses following injuries176,177 likely provide neuroprotection, dysregulation of these microglial functions is observed in AD/ADRD, and an increasing number of studies report that pro-inflammatory or disease-associated microglial phenotypes are associated with worse neurotoxic outcomes and increased severity of AD/ADRD neuropathology169,178–182.
EXPOSURE TO AIR POLLUTION PROMOTES NEUROINFLAMMATION
Glial activation in response to PM exposure has been quantified by immunohistochemical analyses in many experimental animal studies. Most studies used IBA-1 as a biomarker of microglia183,184 and GFAP as a biomarker of astrocytes185,186, and some studies colocalized IBA-1 with CD68, a marker of phagocytic cells. Numerous studies have reported that PM induced recruitment of microglia and astrocytes to the olfactory bulb and hippocampus of rodents158,160,187. However, PM-induced microglial activation varied across different studies depending on the exposure paradigm, sex, age, genotype, and brain region. In one study, wildtype mice exhibited increased IBA-1+ expression with more amoeboid microglial morphology in the olfactory bulb after 4–8 weeks of exposure to concentrated PM2.5187, whereas in another study, APP/PS1 mice, but not wildtype mice, exhibited increased microglial activation in the hippocampus following a 3-month exposure158. A more chronic exposure to ambient TRAP induced microglial phagocytosis (increased ratio of CD68+/IBA-1+) in the hippocampus of both wildtype and TgF344-AD female rats after 2 months of exposure, while male wildtype and TgF344-AD rats did not exhibit TRAP-induced microglial activation until after 14 months of exposure160. Interestingly, in contrast to male rats, female rats exhibited a dampened microglial phagocytic response to TRAP after 14 months of exposure160. A different study reported that 2 weeks of UFPM exposure reduced microglial activation with more ramified microglial morphology in 3xTgAD mice compared to filtered air-exposed controls156. Like microglia, observations regarding the impacts of PM on astrocytic activation (GFAP+ immunoreactivity) varied across studies. While some studies reported PM-induced astrocytic recruitment and activation in the hippocampus152,158,188, hypothalamus189, and striatum190, other studies reported no significant effects of PM on astrocytic activation in any of the brain regions examined156,160.
These variable observations suggest that the glial responses to air pollution are complex. The functional impact of these changes also is not clear since it is now appreciated that glial activation may either exacerbate169,178–182 or protect169,191–193 against synapse loss and neurodegeneration. To better understand how air pollution disrupts neuroinflammatory homeostasis, more recent studies have distinguished glial phenotypes using molecular biomarkers of pro- vs. anti-inflammatory phenotypes or biomarkers of disease-associated microglia (DAM). Following 12 weeks of exposure to UFPM, TMEM119+ microglia, which are considered homeostatic, that were not associated with amyloid plaques were reduced, while ferritin+ DAM microglia, which are associated with senescence and reactive oxygen species (ROS), were increased in both wildtype and AppNL-G-F/+-KI mice157. The same study also showed that UFPM increased the number of highly inflammatory, neurotoxic C3+ DAM astrocytes in the cortex of both genotypes157. In vitro studies also have characterized glial phenotypes after PM treatment. Primary coculture of microglia and astrocytes exhibited upregulated expression of DAM astrocytic markers (C3 protein and DAM-specific transcripts) in response to PM2.5 treatment194. In addition, PM2.5 altered interactions between neurons and glial cells in a triculture of neuron, astrocyte, and microglia human cell lines by promoting polarization of DAM microglia characterized by CD11b, CD86, and iNOS expression195. Based on these results, the type of glial activation or polarization following PM exposure may be important in either resolving inflammation or driving it towards persistent neuroinflammation and glial dysfunction, which can further exacerbate AD pathologies and neuronal atrophy.
These observations raise important questions regarding the mechanism(s) by which air pollutants alter glial phenotypes. Microglia and astrocytes are activated by autocrine and paracrine signals in the forms of: (1) cytokines and chemokines, which are small immune signaling molecules196, and (2) reactive oxygen or nitrogen species (ROS/RNS), which are byproducts of cellular oxidative metabolism that carry unstable oxygen radicals197. Reactive oxygen/nitrogen species, such as H2O2, NOx, and superoxides, serve as short-lived paracrine signals that play important physiological roles in immune defense and maintenance of cellular homeostasis197. Due to the highly reactive property of ROS, the cellular and extracellular levels of ROS are tightly regulated by cellular and enzymatic antioxidants that reduce and stabilize ROS to maintain homeostatic levels of ROS197. However, an imbalanced and persistent increase of ROS beyond the physiological range depletes the normal antioxidant capacity, resulting in oxidative stress when ROS react with cellular components to cause lipid peroxidation, mitochondrial damage, protein oxidative damage, DNA damage, and cellular death197. Additionally, subtler changes in cellular ROS levels can alter physiological redox signaling and consequently, result in cellular dysfunctions in microglia198.
In vitro, short-term (2–3 weeks) exposure to concentrated PM or subchronic (3 months) exposure to ambient level of PM2.5 was sufficient to cause oxidative stress in the olfactory bulb and hippocampus of rodents as evidenced by increased lipid peroxidation152,161,199–201, dysregulated antioxidant metabolism149,152,202, DNA damage and epigenetic modifications consistent with oxidative stress109. These findings demonstrate that air pollution can cause oxidative stress that may trigger neuroinflammation via microglia activation. In tissue culture models, PM2.5 or DEP has been shown to increase ROS followed by microglial activation and release of proinflammatory cytokines in microglial monoculture201,203,204, neuron-glia coculture195,205 and microglia-endothelial cell coculture206. Interestingly, it has been reported that complement 3 (C3)-mediated microglial activation resulted in aggravated Aβ deposition, synaptic loss, and neuronal degeneration182,207–210 via NADPH oxidase activation208,209, which increases ROS production. In an independent study, NADPH oxidase activation was found to induce DAM microglial polarization211.
Exposure to air pollution has also been shown to cause proinflammatory cytokine release in the rodent hippocampus149,150,152,158,162,164,200,212–214. Across different studies, the main proinflammatory cytokines that were consistently upregulated by PM exposure were TNF-α, IL-1β, IL-6, and IFN-γ. However, whether and how PM-induced proinflammatory cytokines mediate DAM glial polarization and neuronal cell death are still poorly understood. Several recent in vitro studies suggest several potential mechanisms. For example, in a neuron-astrocyte coculture of human cell lines, PM2.5 treatment caused the release of chemokines (CCL1, CCL2) that can recruit microglia, as well as cytokines (IL-1β, IFN-γ, IL-5, IL-8) that can polarize microglia into DAM phenotypes195. Monocultured microglia were polarized to the DAM phenotype (CD86+, iNOS+ with increased NO release) when exposed to conditioned media from astrocyteneuron coculture exposed to PM2.5; and antibody neutralization of IL-1β, IFN-γ, CCL1, and CCL2 in the conditioned media significantly reduced DAM polarization. Interestingly, when monocultured microglia were directly exposed to PM2.5 they did not polarize to a DAM phenotype, but instead became phagocytic and anti-inflammatory (TREM2+, LC3b+, CD86−), which resulted in effective clearance of PM2.5. These authors further demonstrated that microglia and astrocytes both contributed to PM-induced synaptic impairment while DAM microglia primarily drove PM-induced neuronal cell death and tau-phosphorylation195. These findings suggest that PM-induced glial polarization occurs through bidirectional crosstalk between microglia and astrocytes in which cytokines act as mediators to not only shift glial responses, but also to cause neuronal atrophy. An independent study further confirmed these results by showing that PM0.2-induced neuronal synaptic atrophy was mainly driven via C3 signals released by DAM astrocytes that were polarized by activated microglia. Disease-associated astrocyte polarization was reversed when microglia were inhibited by minocycline even with the PM0.2 treatment, and this further reduced synaptic damages194.
The role of IL-1β has also been investigated in the context of inflammasome activation. The inflammasome is a multiprotein intracellular immune signaling complex that detects pathogen- and damage-associated molecular patterns (PAMPs and DAMPs) to initiate proinflammatory responses and has been increasingly reported and discussed as a pathogenic mechanism of AD215. In a neuron-microglia coculture, co-exposure to oligomeric Aβ and PM2.5 augmented the microglial response to lipopolysaccharide (LPS), evidenced as increased ROS and IL-1β production, resulting in activation of the NLRP3 inflammasome and subsequent neuronal apoptosis. Pretreating the coculture with the antioxidant N-acetyl-cysteine and/or caspase-1 inhibitor before oligomeric Aβ and PM2.5 exposure significantly ameliorated neuronal apoptosis, suggesting that oxidative stress and NLRP3 inflammasome activation mediate the neurotoxic effects of oligomeric Aβ and PM2.5205.
Results from animal studies and in vitro studies corroborate epidemiological findings and provide a better understanding of the molecular and cellular responses to air pollution that include oxidative stress, neuroinflammation, neurodegeneration, and aggravation of AD/ADRD pathologies. While there is ongoing progression towards identifying how air pollutants perturb brain health, mechanistic links that integrate the neuropathological processes triggered by air pollution need further investigation. It is experimentally challenging to recreate realistic air pollution exposure paradigms, but recent development of exposure systems that allow better recapitulation of human-relevant exposures will enable significant advancements in the field of air pollution and ADRD research possible. Nevertheless, discrepancies in exposure paradigms and animal models between studies has led to large variabilities in neurologic outcomes. Therefore, follow-up studies with more standardized exposures using genetically modified animal models with inclusion of both sexes are necessary to validate observations reported across different studies.
Outstanding data gap: Direct vs. indirect effects of air pollution on the brain
While in vitro studies demonstrate that PM from polluted air can directly trigger responses in neurons and glial cells, whether and how gases associated with air pollution directly affect these cell types is largely unknown. Similarly, there remains considerable uncertainty as to whether in vivo brain responses to air pollution arise from direct interactions with air pollutants or rather are the indirect consequence of air pollution effects on peripheral organs. There is experimental evidence, albeit limited, to support both possibilities, which likely are not mutually exclusive.
DIRECT PATHWAY
The direct pathway infers translocation of PM to the brain, where it can directly activate microglia. Translocation of PM to brain parenchyma may occur through retrograde transport of PM from olfactory epithelium to the olfactory cortex via olfactory nerves or via the trigeminal nerve to the brain216–218 (Figure 3A). Particulate matter inclusions in the olfactory bulbs and prefrontal cortex have been observed and associated with impaired olfactory function in both mice and humans109,110,112,113,155,160,161,187,219,220. Alternatively, PM may enter the brain by crossing the blood-brain barrier (BBB) from the systemic circulation. Inhaled fine particles that are deposited in the distal airways and alveoli can readily cross the air-blood barrier to enter the systemic circulation and cross other biological barriers like the BBB221–225. Studies using a 3-D human BBB organotypic chip have shown that PM can cross the BBB225. Moreover, an in vivo study in which transgenic AD animals were exposed to ambient levels of PM, extensive PM deposition was observed in the hippocampus, which is vulnerable to BBB leakage in AD, with minimal PM detected in the frontal cortex160, supporting this as the primary mechanism by which inhaled PM may be reaching the brain under conditions of exposure to ambient TRAP levels. Blood-brain barrier impairment is a known AD pathology226–228 that may be both a causal factor and a result of air pollution-induced AD exacerbation because PM and circulating mediators can cross an impaired BBB229 and air pollution can break down components of the neurovascular unit that forms the BBB199,230,231. Phagocytosis of PM by activated microglia has been well characterized in many in vitro studies195,205,206,232–234, and neurotoxic materials, such as metals, endotoxins, and polycyclic aromatic hydrocarbons, adhered to PM have been shown to cause neuroinflammation in the brain parenchyma115,235–237. Although there are less studies on PM-induced astrocytic activation189,194,195, results indicate that the microglia-astrocyte crosstalk can lead to bidirectional responses in both glial cells to promote neuroinflammation194,195,210,238,239.
Figure 3. Potential mechanisms by which air pollutants promote neuroinflammation.
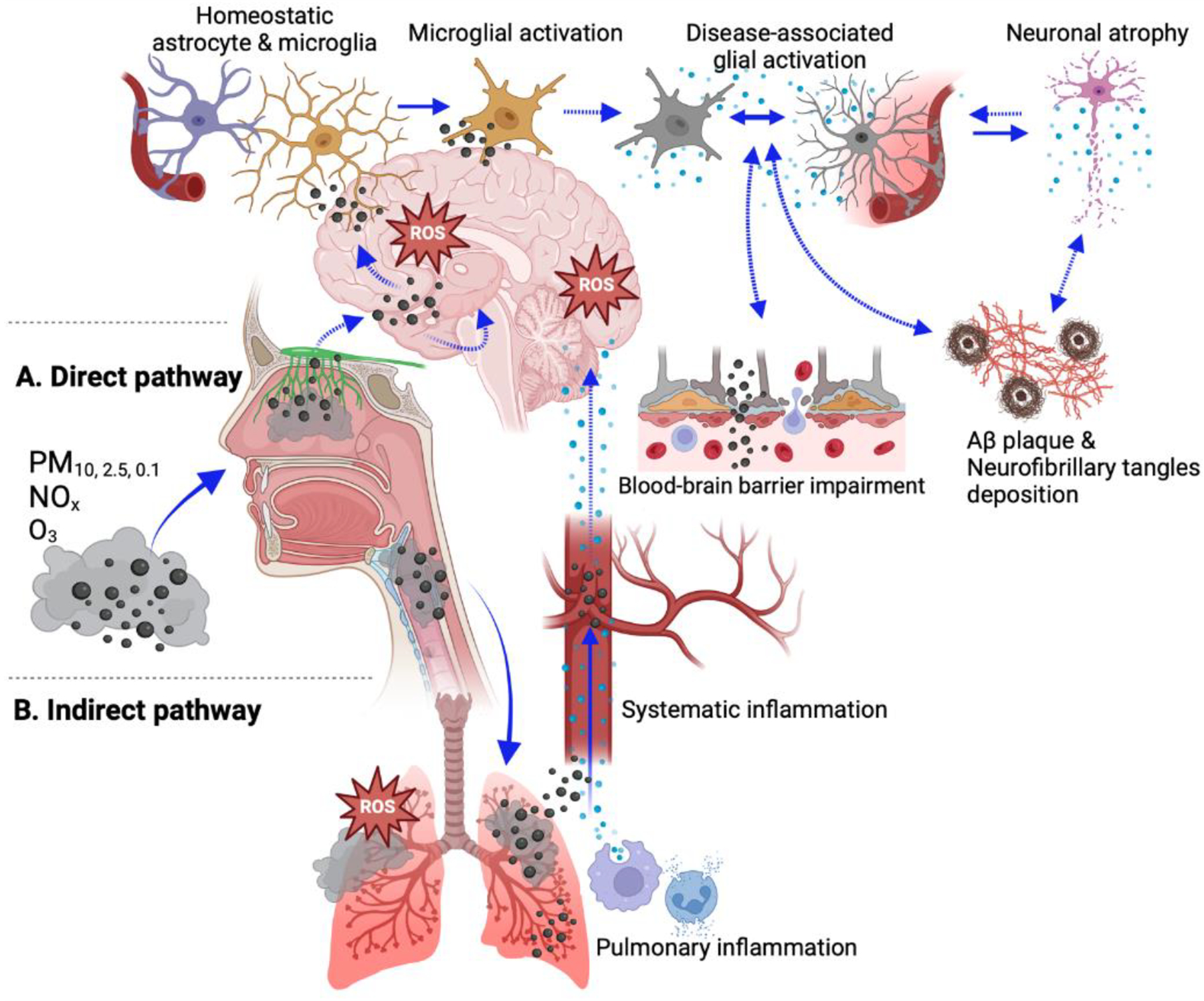
Schematic summary of potential pathways by which air pollutants mediate neuroinflammation. A. Direct pathway: Inhaled air pollutants infiltrate the brain parenchyma via retrograde transportation from olfactory nerve terminals into the olfactory cortex or by crossing the blood-brain barrier from the systemic circulation to directly activate microglia. B. Indirect pathway: Inhaled air pollutants cause neuroinflammation via the lung-brain axis. It is hypothesized that air pollutant-induced pulmonary inflammation leads to neuroinflammation via increased systemic inflammation and release of inflammatory mediators into the systemic circulation, which are then delivered to the brain and cross the blood brain barrier to activate innate immune cells in the brain. NOTE: ROS, reactive oxygen species; dotted arrows indicate hypothesized modes of actions; solid arrows indicate modes of actions supported by experimental evidence. Created with BioRender.com.
INDIRECT PATHWAY
The indirect pathway infers that air pollution-associated neuroinflammation is a consequence of inflammation induced by air pollution in peripheral targets. The respiratory toxicology of air pollution has been extensively studied, and it is well-established that inhaled air pollutants reduce lung function and increase respiratory disease susceptibility and mortality by inducing and disrupting pulmonary inflammatory responses240–243. Air pollution also causes consequent systemic inflammation, and increased serum levels of proinflammatory cytokines, dysfunctional fibrinolysis, oxidative stress, and activation of circulating immune cells41,244–248. With growing evidence suggesting that systemic inflammation can promote neuroinflammation249–254 and that higher risks of dementia and cognitive deficits are associated with impaired lung function255–258, the lung-brain axis has been proposed as a potential indirect pathway by which chronic exposure to air pollution induces persistent neuroinflammation that promotes AD/ADRD pathogenesis (Figure 3B). While the direct pathway proposes a valid explanation for PM-induced neuroinflammation, it does not adequately explain neuroinflammatory effects of gaseous components like NOx and O3150. A research group from Indiana University has conducted a series of studies demonstrating how O3-induced systemic circulating factors regulate microglial and astrocytic activation150,151,259. An acute exposure to O3 caused persistent microglial activation in rats 24 hours post-exposure, and the same study showed that the addition of serum from O3-exposed animals can prime microglia in vitro to augment their response to LPS treatment.259 The group’s more recent studies show that O3 reduced the number of peri-plaque TREM2+ microglia but increased peri-plaque colocalization of microglia and astrocytes151 with altered transcriptomic profiles shifting glial cells to more disease-associated states (lower Trem2 and higher Serpinea3n gene expressions) in 5xFAD mice150. They also reported that O3 increased the level of circulating high mobility group box 1 (HMGB1), which is a DAMP signal that enhances both innate and adaptive immune responses150. Deleting HMGB1 only in peripheral myeloid cells, but not in microglia by using Hmgb1fl/flLysM-Cre+ mouse strain, reversed O3-induced changes in glial expressions and returned Trem2 and Serpinea3n transcript levels back to the filtered air control baseline150,151. An in vitro study from a different group showed that in comparison to directly adding DEP alone to microglia, adding conditioned medium from DEP-stimulated alveolar macrophages caused a higher activation of microglial CD14, which is a pattern recognition receptor that induces DAMP signaling234. These findings and those from other studies260–262 with similar results suggest that the lung-brain axis may indirectly mediate neuroinflammatory effects of air pollution independent of the direct pathway. However, more experimental studies are needed to further investigate the lung-brain axis in different exposure paradigms that not only test acute effects of exclusive air pollutants, but also test long-term effects of exposure to combined air pollutants.
Epigenetics, Environment, and Alzheimer’s disease and related dementias
Epigenetic factors, which mediate gene and environment interactions, may serve as a mechanism underlying G × E interactions and explain a portion of the missing heritability of AD/ADRD.83 Epigenetic control is highly complex, with at least 28 known histone, 53 DNA, and 160 RNA modifications, as well as non-coding RNAs, that work in concert to regulate chromatin accessibility, gene expression, mRNA stability, and mRNA translation263–265. The location and context-dependent function of these modifications further adds to their complexity but also enables their targeted use as biomarkers and targeted therapeutics266–268. Here, while we do not cover the full breadth of epigenetic modifications and their functions, we do summarize recent findings related to their association with and potential functional link between air pollution and AD/ADRD pathogenicity.
Prior experimental and population-based epigenetic studies characterizing G × E interactions identified dynamic and disease-associated epigenetic modifications and transcriptional profiles that varied in response to environmental factors268. The modifiable nature of epigenetic modifications and their control over gene expression positions them as prime tools and targets for therapeutic intervention. Additionally, their dynamism makes them well suited to function as biomarkers indicative of disease predisposition, pathological outcomes and disease susceptibility, or of environmental exposures.
Generally, epigenetic modifications, whether located on histones, DNA, or RNA, are regulated by writer and erasers and interpreted by reader proteins or protein/RNA complexes. The expression of these regulatory factors enables fine control over tissue and cell-type specific spatiotemporal expressional programming269–272. Dysregulation of epigenetic control, either through the alteration of specific markers or the abundance of the readers/writers/erasers serves as an additional mechanism of control and potential contributor towards disease state.
DNA MODIFICATIONS
Recent work characterizing the relative contributions of genetic and environmental factors in the development and progression of AD highlighted epigenetic modifications as critical mediators of G × E interactions. In a cohort of older women from the Women’s Health Initiative Memory Study (WHIMS), it was found that nanoparticles exposure increased the risk of cognitive decline by 81% and all-cause dementia by 92% in a dose-dependent manner for APOE ε4 carriers273. Following up on these findings, the authors utilized the 5xFAD mouse model and found that exposure to nanoparticles was associated with greater AD pathology and the APOE status of the exposed individual functioned as a response modifier. In the 5xFAD mouse model, over the course of 15 weeks of nanoparticles exposure, the presence of ApoE ε4+/+ over ApoE ε3+/+ alleles significantly increased amyloid plaque pathology, reduced hippocampal CA1 neurites, and decreased the glutamate GluR1 subunit. In a similar study investigating the G × E interaction between cadmium exposure and ApoE ε4 dosage, 14 weeks of cadmium exposure in the ApoE knock-in mouse model identified accelerated cognitive impairment and reduced hippocampal neurogenesis in ApoE ε4 carriers relative to their ApoE ε3 counterparts, indicating G × E interactions modulate AD pathogenicity274. In effort to explore the missing heritability observed in AD, Panitch, et al. recently investigated the relationship between APOE DNA methylation (5-methylcytosine; 5mC) and ε4 carrier status and found differential methylation at 25 CpG sites in the dorsolateral prefrontal cortex and 36 CpG sites in blood, with the majority of sites being hypomethylated275. Furthermore, this group identified seven CpG sites in the APOE region (including TOMM40, APOE, and APOC1 genes) that significantly differed between APOE ε4 carriers and non-carriers in brain and blood (P<5×10−8), with three sites in the APOE gene showing hypermethylation in ε4 carriers and a nominal association with APOE expression in the brain (P<10−5)275. One of the three genes that contained differentially methylated CpG sites in APOE ε4 carriers relative to control was TET1, a 5mC eraser and DNA hydroxymethylation (5-hydroxymethylcytosine, 5hmC) writer. In the same family of Ten-Eleven Translocation proteins, TET2, a 5mC eraser and 5hmC writer, was also found to be moderately differentially methylated in APOE ε4 carriers relative to control (p = 7.6×10−6)275. Interestingly, both TET1 and TET2 function in similar but distinct capacities, and prior sequencing studies have identified an enrichment of TET1 and TET2 variants associated with EOAD/frontotemporal dementia276,277. Lastly, utilizing an APP mouse model, tethering the de novo 5mC writer, Dnmt3a, to dCas9 targeted to APP was sufficient to methylate the APP gene, reduce APP expression and amyloid pathology, and ameliorate cognitive and behavioral impairment278.
HISTONE MODIFICATIONS
In addition to DNA modifications, histone modifications have emerged as crucial players in the pathogenesis of AD and often function with DNA modifications to regulate gene expression279,280. Recent studies reveal links between air pollution and histone modifications. Zheng et al. explored how different components of air pollution (PM2.5, PM10, black carbon, and elemental components (potassium, sulfur, iron, silicon, aluminum, zinc, calcium, and titanium)) influenced various histone modifications (H3K9ac, H3K9me3, H3K27me3, and H3K36me3) in the blood leukocytes of exposed truck drivers and office workers in Beijing281. Their group identified differential associations between pollutants and various histone markers. Specifically, they noted an increase in ambient PM10 associated with lower H3K27me3 and H3K36me3 levels. They also observed that office workers had a stronger association between black carbon and H3K9ac and H3K36me3 than truck drivers, and that the association between black carbon exposure and H3K9ac and H3K9me3 status was sex-dependent281.
Similarly, exposure to air pollutants, particularly PM2.5 and PM10, induced significant epigenetic alterations in rat blood and lung tissue in a dose-dependent manner282. This study revealed that increased exposure to PM2.5 and PM10 generally led to decreased DNA methylation of LINE-1 and iNOS, while simultaneously increasing histone acetylation (H3K9ac) in both blood and lung tissue. Interestingly, PM2.5 exposure was also associated with increased methylation of p16CDKN2A and APC promoters. The effects of NOx were more variable, showing mixed impacts on methylation patterns. Notably, these epigenetic changes were often more pronounced in blood compared to lung tissue, with H3K9ac consistently increasing in response to PM2.5and PM10 exposure in both tissue types282.
RNA MODIFICATIONS
RNA N6-methyladenosine (m6A) is the most studied and prevalent RNA modification, it is highly enriched in the brain, and associated with a suite of neurodevelopmental and neurodegenerative diseases, including AD. m6A influences transcript splicing, stability, translation, nuclear export, and RNA structure283,284. There is evidence that environmental exposure influences RNA modifications and subsequently, AD pathophysiology285 Li et al., 2023 found that exposure to PM2.5 is associated with a greater prevalence of m6A modifications, a global increase in gene expression, and a significant increase in the expression of prostaglandin-endoperoxide synthase 2 (PTGS2), which is involved in synthesizing the prostaglandin inflammation signaling molecules286.
Recent research shed light on the molecular mechanisms underlying the effects of PM2.5 exposure on lung cells287. In the A549 lung cell line, PM2.5 exposure upregulated m6A RNA methylation coincident with increased expression of TGF-β, SMAD3, and the methyltransferases METTL3 and METTL14 (m6A writers). Importantly, inhibition of TGF-β reversed the PM2.5 -induced changes, suggesting a pivotal role for the TGF-β signaling pathway in regulating m6A RNA methylation following PM2.5 exposure287. These findings revealed a potential mechanism by which lung inflammation triggered by PM2.5 exposure leads to m6A modifications observed in AD.
Using postmortem AD patient data, another study identified significantly reduced expression and soluble protein levels of METTL3, and no significant changes for METTL14 in the hippocampus288. Notably, insoluble fractions of AD brain samples had accumulated METTL3 that positively correlated with insoluble tau protein levels, although no direct interaction between METTL3 and tau was observed288. This research suggests that while tau pathology is a better predictor of disrupted m6A signaling than Aβ load, it likely does not directly cause the altered METTL3 expression seen in AD hippocampal neurons.
These findings were corroborated in a study of individuals diagnosed with AD vs. controls that reported a marked decrease in m6A modifications and METTL3, METTL14, WTAP, FTO, and YTHDF protein levels in cortex and hippocampus tissue with AD. The functional significance of this observation is suggested by experimental animal studies: knockdown of METTL3 in mouse hippocampus significantly increased memory loss, neurodegeneration, gliosis, oxidative stress, and apoptotic processes289. Knocking down METTL3, METTL14, and YTHDF in flies expressing TauR406W was found to worsen eye phenotype and reduce motor function, highlighting that reduced expression of m6A or the ability to read m6A is associated with worsened AD-associated phenotypes290.
Collectively, these studies suggest that the regulation of RNA writers, readers and erasers is a critical component for mediating G × E interactions, and aberrant regulation of these proteins has a large knock-on effect, influencing pathological outcomes.
NON-CODING RNA
A separate class of RNAs, non-coding RNAs, exert epigenetic control via control of transcription, transcript stability, and translation efficiency. These molecules are highly dynamic, and their expression and stability can be influenced by exposure to environmental factors291–294.
To determine whether DEP induces pulmonary inflammatory response, Wang and colleagues explored the involvement of RNA binding protein LIN28B and non-coding microRNA Iet7 axis in the inflammatory response to DEP exposure in mice295. Let7-d and Iet7-g were significantly downregulated, while LIN28B protein levels were elevated from 6 to 24 hours after DEP exposure. Furthermore, they found that LIN28B is responsible for the downregulation of Iet7, subsequently increasing expression of the pro-inflammatory cytokine IL-6295. A study involving a protein of the same class, LIN28A, in a VaD rat model, where VaD was introduced by bilateral common carotid artery occlusion, found that treatment with Lin28a siRNA ameliorated cognitive impairment, as well as upregulated expression of GFAP, and IBA-1 glial markers296. Moreover, treatment with siRNA alleviated BBB damage as measured by expression of PECAM-1, PDGFRβ, occludin, claudin-9, and ZO-1, and Ccr6296.
Furthering the link between environmental exposure, non-coding RNAs and modulation of inflammation, microarray analyses of blood samples from foundry workers exposed to metal-rich PM identified significantly increased expression of miR-421, miR-146a, miR-29a, and let-7g after 3 days of work exposure relative to baseline (first day of the work week after two days off).297 In addition to sampling miRNA expression, PCR was performed on 18 inflammatory genes, including TGFß1, TGFß2, TNF, ITGA2, ITGAX, NFKB1, NOS2, CCL2, CCL5. Significant differences were found in individual miRNAs and inflammatory genes that have differing targets and regulatory activity. This included miR-421 associated with NOS2 expression; miR-146a with TGFß1, CCL2 and CCL5 expression; and let-7g with TGFß1, ITGAX, and NFKß1 expression297.
Additional research exploring the relationship between PM2.5 exposure and expression of non-coding RNAs has identified increased expression of the long non-coding RNA, HCG18, in the lung. HCG18 suppresses miR-195 expression and leads to upregulated ATG14 expression and increased autophagy and progression of lung adenocarcinoma298. In addition to playing a significant role in the development of lung pathologies, reduced mir-195 expression is observed with human aging and in AD progression, while increased mir-195 prevalence is associated with improved cognitive performance and negatively correlated with cerebrospinal fluid tau levels. Human expression patterns of mir-195 are mirrored in ApoE ε4+/+ mice when compared with ApoE ε3+/+ mice. Furthermore, elevating miR-195 levels was found to reduce amyloid burden and tau hyper-phosphorylation and improve cognitive impairment. Lastly, when cortical neurons and astrocytes generated from inducible pluripotent stem cells derived from ApoE ε4+/+ mice were co-cultured, miR-195 inhibition exacerbated AD-associated lysosomal phenotypes299.
In this section, we highlighted epigenetic mechanisms by which air pollutants may contribute to cognitive impairment and AD-associated pathologies. These studies underscore the crucial role of epigenetics as a mediator of G × E interactions in AD. Importantly, they indicate that the development of epigenetic markers of air pollution exposure may advance the identification of specific air pollutants that promote AD/ADRD-associated risk and progression. Epigenetic analyses also hold the promise of providing targets for therapeutic interventions, offering modifiable pathways to potentially reduce AD pathophysiology.
Conclusions
Consistent with previous epidemiological findings regarding the association between air pollution and ADRD, recent epidemiological studies have largely reported positive correlations between air pollution and AD/ADRD. This association is mostly substantiated by experimental animal studies. Although there is general consensus that air pollution is an environmental risk factor for AD/ADRD, associations between specific air pollutants and ADRD subtypes are variable across different studies. This variability in part stems from the complexity of mixed ADRD pathologies and equivocal etiological links between genetic status, pathological manifestation, cognitive impairment, and clinical diagnoses. Currently, the field is reassessing diagnostic criteria utilizing biomarkers that more accurately delineate between AD and related dementias to better characterize pathology-specific disease progression. These refinements in diagnostic precision will improve efforts to identify environmental factors that modify risk and severity of AD/ADRD. Elucidating ADRD stratification and G × E contributions implicated in the development of ADRD will inform effective public health regulations and guidelines for air pollution and other environmental contaminants, with the aim of preventing and minimizing adverse neurological health outcomes.
Figure 2. Composition of air pollutants.

Air pollutants consist of gaseous and particulate matter (PM) fractions with heterogenous sources, compositions, and sizes, all of which influence toxicity to the respiratory system and the brain. Created with BioRender.com.
Acknowledgements:
The authors thank Dr. Suzette Smiley-Jewell (UC Davis) for her assistance in proofreading and editing this manuscript.
Funding Statement:
This work was supported by the National Institute on Aging (grant # RF1 AG074709 to PJL), the National Institute of Neurological Disorders and Stroke (grant # RF1 NS130659 to PJL) and the National Heart, Lung, and Blood Institute predoctoral training grant program (grant # T32 HL007013 to HHP).
List of abbreviations
- 5hmC
5-hydroxymethylcytosine
- 5mC
5-methylcytosine
- AD
Alzheimer’s disease
- ADRD
Alzheimer’s disease-related dementias
- Aβ
Amyloid beta plaques
- BBB
Blood-brain barrier
- BC
Black carbon
- C3
Complement 3
- CAP
Concentrated ambient particle
- CO
Carbon monoxide
- CSF
Cerebrospinal fluid
- DAM
Disease-associated molecular biomarkers
- DAMP
Damage-associated molecular pattern
- DEP
Diesel exhaust particles
- EOAD
Early-onset Alzheimer’s disease
- G × E
Gene by environment interaction
- HMGB1
High mobility group box 1
- LOAD
Late-onset Alzheimer’s disease
- LPS
Lipopolysaccharide
- m6A
N6-methyladenosine
- MRI
Magnetic resonance imaging
- NFL
Neurofilament light
- NFT
Neurofibrillary tau tangles
- NOx
Nitrogen oxides
- O3
Ground-level ozone
- PAMP
Pathogen-associated molecular pattern
- PET
Positron emission tomography
- PFAS
Per-and polyfluoroalkyl substances
- PM
Particulate matter
- PTGS2
prostaglandin-endoperoxide synthase 2
- ROS/RNS
Reactive oxygen or nitrogen species
- SO2
Sulfur dioxide
- TRAP
Traffic-related air pollution
- UFPM
Ultrafine particulate matter
- VaD
Vascular dementia
Footnotes
Conflict of Interest:
The authors declare that they have no conflicts of interest.
References:
- 1.Collaborators GBDDF. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. Feb 2022;7(2):e105–e125. doi: 10.1016/S2468-2667(21)00249-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Selkoe DJ, Hardy J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol Med. Jun 2016;8(6):595–608. doi: 10.15252/emmm.201606210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jack CR Jr., Bennett DA, Blennow K, et al. NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement Apr 2018;14(4):535–562. doi: 10.1016/j.jalz.2018.02.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Khan SS, Bloom GS. Tau: The Center of a Signaling Nexus in Alzheimer’s Disease. Front Neurosci. 2016;10:31. doi: 10.3389/fnins.2016.00031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.2024 Alzheimer’s disease facts and figures. Alzheimers Dement. May 2024;20(5):3708–3821. doi: 10.1002/alz.13809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dubois B, Hampel H, Feldman HH, et al. Preclinical Alzheimer’s disease: Definition, natural history, and diagnostic criteria. Alzheimers Dement. Mar 2016;12(3):292–323. doi: 10.1016/j.jalz.2016.02.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sperling RA, Aisen PS, Beckett LA, et al. Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. May 2011;7(3):280–92. doi: 10.1016/j.jalz.2011.03.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Petersen RC, Caracciolo B, Brayne C, Gauthier S, Jelic V, Fratiglioni L. Mild cognitive impairment: a concept in evolution. J Intern Med. Mar 2014;275(3):214–28. doi: 10.1111/joim.12190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Petersen RC, Lopez O, Armstrong MJ, et al. Practice guideline update summary: Mild cognitive impairment: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology. Jan 16 2018;90(3):126–135. doi: 10.1212/WNL.0000000000004826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dunn AR, O’Connell KMS, Kaczorowski CC. Gene-by-environment interactions in Alzheimer’s disease and Parkinson’s disease. Neurosci Biobehav Rev. Aug 2019;103:73–80. doi: 10.1016/j.neubiorev.2019.06.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Eid A, Mhatre I, Richardson JR. Gene-environment interactions in Alzheimer’s disease: A potential path to precision medicine. Pharmacol Ther. Jul 2019;199:173–187. doi: 10.1016/j.pharmthera.2019.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Livingston G, Huntley J, Sommerlad A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. Aug 8 2020;396(10248):413–446. doi: 10.1016/S0140-6736(20)30367-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schindler SE, Galasko D, Pereira AC, et al. Acceptable performance of blood biomarker tests of amyloid pathology - recommendations from the Global CEO Initiative on Alzheimer’s Disease. Nat Rev Neurol. Jul 2024;20(7):426–439. doi: 10.1038/s41582-024-00977-5 [DOI] [PubMed] [Google Scholar]
- 14.Ritchie C, Smailagic N, Noel-Storr AH, Ukoumunne O, Ladds EC, Martin S. CSF tau and the CSF tau/ABeta ratio for the diagnosis of Alzheimer’s disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. Mar 22 2017;3(3):CD010803. doi: 10.1002/14651858.CD010803.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.National Plan to Address Alzheimer’s Disease: 2023 Update. Updated 2024, April 22. https://aspe.hhs.aov/reports/national-plan-2023-update
- 16.Schneider JA. Neuropathology of Dementia Disorders. Continuum (Minneap Minn). Jun 1 2022;28(3):834–851. doi: 10.1212/CON.0000000000001137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Arvanitakis Z, Shah RC, Bennett DA. Diagnosis and Management of Dementia: Review. JAMA. Oct 22 2019;322(16):1589–1599. doi: 10.1001/jama.2019.4782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Caldwell AB, Anantharaman BG, Ramachandran S, et al. Transcriptomic profiling of sporadic Alzheimer’s disease patients. Mol Brain. Oct 12 2022;15(1):83. doi: 10.1186/s13041-022-00963-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Reitz C, Rogaeva E, Beecham GW. Late-onset vs nonmendelian early-onset Alzheimer disease: A distinction without a difference? Neurol Genet. Oct 2020;6(5):e512. doi: 10.1212/NXG.0000000000000512 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dao L, Choi S, Freeby M. Type 2 diabetes mellitus and cognitive function: understanding the connections. CurrOpin Endocrinol Diabetes Obes. Feb 1 2023;30(1):7–13. doi: 10.1097/MED.0000000000000783 [DOI] [PubMed] [Google Scholar]
- 21.Benedict RHB, Amato MP, DeLuca J, Geurts JJG. Cognitive impairment in multiple sclerosis: clinical management, MRI, and therapeutic avenues. Lancet Neurol. Oct 2020;19(10):860–871. doi: 10.1016/S1474-4422(20)30277-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mclnnes K, Friesen CL, MacKenzie DE, Westwood DA, Boe SG. Mild Traumatic Brain Injury (mTBI) and chronic cognitive impairment: A scoping review. PLoS One. 2017;12(4):e0174847. doi: 10.1371/journal.pone.0174847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Robinson JL, Richardson H, Xie SX, et al. The development and convergence of co-pathologies in Alzheimer’s disease. Brain. Apr 12 2021;144(3):953–962. doi: 10.1093/brain/awaa438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Greenberg SM, Bacskai BJ, Hernandez-Guillamon M, Pruzin J, Sperling R, van Veluw SJ. Cerebral amyloid angiopathy and Alzheimer disease - one peptide, two pathways. Nat Rev Neurol. Jan 2020;16(1):30–42. doi: 10.1038/s41582-019-0281-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Grasso D, Castorani G, Borreggine C, Simeone A, De Blasi R. Cerebral amyloid angiopathy related inflammation: A little known but not to be underestimated disease. Radiol Case Rep. Sep 2021;16(9):2514–2521. doi: 10.1016/j.radcr.2021.05.080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hereditary cerebral amyloid angiopathy (2022).
- 27.Coomans EM, Tomassen J, Ossenkoppele R, et al. Genetically identical twins show comparable tau PET load and spatial distribution. Brain. Oct 21 2022;145(10):3571–3581. doi: 10.1093/brain/awac004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Puckett OK, Fennema-Notestine C, Hagler DJ Jr., et al. The Association between Exposure to Fine Particulate Matter and MRI-Assessed Locus Coeruleus Integrity in the Vietnam Era Twin Study of Aging (VETSA). Environ Health Perspect. Jul 2024;132(7):77006. doi: 10.1289/EHP14344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Karlsson IK, Escott-Price V, Gatz M, et al. Measuring heritable contributions to Alzheimer’s disease: polygenic risk score analysis with twins. Brain Commun. 2022;4(1):fcab308. doi: 10.1093/braincomms/fcab308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Varjonen A, Schwarz C, Vuoksimaa E. Co-twin design in brain imaging-review on biomarkers of Alzheimer’s disease. Cereb Cortex. Jul 5 2023;33(14):9054–9066. doi: 10.1093/cercor/bhad181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Livingston G, Sommerlad A, Orgeta V, et al. Dementia prevention, intervention, and care. Lancet. Dec 16 2017;390(10113):2673–2734. doi: 10.1016/S0140-6736(17)31363-6 [DOI] [PubMed] [Google Scholar]
- 32.Gunnarsson LG, Bodin L. Occupational Exposures and Neurodegenerative Diseases-A Systematic Literature Review and Meta-Analyses. Int J Environ Res Public Health. Jan 26 2019;16(3) doi: 10.3390/ijerph16030337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sarailoo M, Afshari S, Asghariazar V, Safarzadeh E, Dadkhah M. Cognitive Impairment and Neurodegenerative Diseases Development Associated with Organophosphate Pesticides Exposure: a Review Study. Neurotox Res. Oct 2022;40(5):1624–1643. doi: 10.1007/s12640-022-00552-0 [DOI] [PubMed] [Google Scholar]
- 34.Kim JY, Park SJ, Kim SK, et al. Pesticide exposure and cognitive decline in a rural South Korean population. PLoS One. 2019;14(3):e0213738. doi: 10.1371/journal.pone.0213738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hayden KM, Norton MC, Darcey D, et al. Occupational exposure to pesticides increases the risk of incident AD: the Cache County study. Neurology. May 11 2010;74(19):1524–30. doi: 10.1212/WNL.0b013e3181dd4423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sturm ET, Castro C, Mendez-Colmenares A, et al. Risk Factors for Brain Health in Agricultural Work: A Systematic Review. Int J Environ Res Public Health. Mar 13 2022;19(6) doi: 10.3390/ijerph19063373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Palzes VA, Sagiv SK, Baker JM, et al. Manganese exposure and working memory-related brain activity in smallholder farmworkers in Costa Rica: Results from a pilot study. Environ Res. Jun 2019;173:539–548. doi: 10.1016/j.envres.2019.04.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liu CH, Huang CY, Huang CC. Occupational neurotoxic diseases in taiwan. Saf Health Work. Dec 2012;3(4):257–67. doi: 10.5491/SHAW.2012.3.4.257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Baldi I, Lebailly P, Mohammed-Brahim B, Letenneur L, Dartigues JF, Brochard P. Neurodegenerative diseases and exposure to pesticides in the elderly. Am J Epidemiol. Mar 1 2003;157(5):409–14. doi: 10.1093/aje/kwf216 [DOI] [PubMed] [Google Scholar]
- 40.Delcourt N, Pouget AM, Grivaud A, et al. First Observations of a Potential Association Between Accumulation of Per- and Polyfluoroalkyl Substances in the Central Nervous System and Markers of Alzheimer’s Disease. J Gerontol A Biol Sci Med Sci. Mar 1 2024;79(3) doi: 10.1093/gerona/glad208 [DOI] [PubMed] [Google Scholar]
- 41.Lederer AM, Fredriksen PM, Nkeh-Chungag BN, et al. Cardiovascular effects of air pollution: current evidence from animal and human studies. Am J Physiol Heart Circ Physiol. Apr 1 2021;320(4):H1417–H1439. doi: 10.1152/ajpheart.00706.2020 [DOI] [PubMed] [Google Scholar]
- 42.Anderson JO, Thundiyil JG, Stolbach A. Clearing the air: a review of the effects of particulate matter air pollution on human health. J Med Toxicol. Jun 2012;8(2):166–75. doi: 10.1007/s13181-011-0203-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bennett M, Nault I, Koehle M, Wilton S. Air Pollution and Arrhythmias. Can J Cardiol. Sep 2023;39(9):1253–1262. doi: 10.1016/j.cjca.2023.03.023 [DOI] [PubMed] [Google Scholar]
- 44.Brook RD, Rajagopalan S, Pope CA 3rd, et al. Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation. Jun 1 2010;121 (21):2331–78. doi: 10.1161/CIR.0b013e3181dbece1 [DOI] [PubMed] [Google Scholar]
- 45.Fiordelisi A, Piscitelli P, Trimarco B, Coscioni E, laccarino G, Sorriento D. The mechanisms of air pollution and particulate matter in cardiovascular diseases. Heart Fail Rev. May 2017;22(3):337–347. doi: 10.1007/s10741-017-9606-7 [DOI] [PubMed] [Google Scholar]
- 46.Miller MR, Newby DE. Air pollution and cardiovascular disease: car sick. Cardiovasc Res. Feb 1 2020;116(2):279–294. doi: 10.1093/cvr/cvz228 [DOI] [PubMed] [Google Scholar]
- 47.Rajagopalan S, Brook RD, Salerno P, et al. Air pollution exposure and cardiometabolic risk. Lancet Diabetes Endocrinol. Mar 2024;12(3):196–208. doi: 10.1016/S2213-8587(23)00361-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tainio M, Jovanovic Andersen Z, Nieuwenhuijsen MJ, et al. Air pollution, physical activity and health: A mapping review of the evidence. Environ Int. Feb 2021;147:105954. doi: 10.1016/j.envint.2020.105954 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Paul KC, Haan M, Mayeda ER, Ritz BR. Ambient Air Pollution, Noise, and Late-Life Cognitive Decline and Dementia Risk. Annu Rev Public Health. Apr 1 2019;40:203–220. doi: 10.1146/annurev-publhealth-040218-044058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mork D, Braun D, Zanobetti A. Time-lagged relationships between a decade of air pollution exposure and first hospitalization with Alzheimer’s disease and related dementias. Environ Int. Jan 2023;171:107694. doi: 10.1016/j.envint.2022.107694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhang B, Weuve J, Langa KM, et al. Comparison of Particulate Air Pollution From Different Emission Sources and Incident Dementia in the US. JAMA Intern Med. Aug 14 2023; doi: 10.1001/jamainternmed.2023.3300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mortamais M, Gutierrez LA, de Hoogh K, et al. Long-term exposure to ambient air pollution and risk of dementia: Results of the prospective Three-City Study. Environ Int. Mar 2021;148:106376. doi: 10.1016/j.envint.2020.106376 [DOI] [PubMed] [Google Scholar]
- 53.laccarino L, La Joie R, Lesman-Segev OH, et al. Association Between Ambient Air Pollution and Amyloid Positron Emission Tomography Positivity in Older Adults With Cognitive Impairment. JAMA Neurol. Feb 1 2021;78(2):197–207. doi: 10.1001/jamaneurol.2020.3962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hajat A, Park C, Adam C, et al. Air pollution and plasma amyloid beta in a cohort of older adults: Evidence from the Ginkgo Evaluation of Memory study. Environ Int. Feb 2023;172:107800. doi: 10.1016/j.envint.2023.107800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Weuve J, Puett RC, Schwartz J, Yanosky JD, Laden F, Grodstein F. Exposure to particulate air pollution and cognitive decline in older women. Arch Intern Med. Feb 13 2012;172(3):219–27. doi: 10.1001/archinternmed.2011.683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ranft U, Schikowski T, Sugiri D, Krutmann J, Kramer U. Long-term exposure to traffic-related particulate matter impairs cognitive function in the elderly. Environ Res. Nov 2009;109(8):1004–11. doi: 10.1016/j.envres.2009.08.003 [DOI] [PubMed] [Google Scholar]
- 57.Jung CR, Lin YT, Hwang BF. Ozone, particulate matter, and newly diagnosed Alzheimer’s disease: a population-based cohort study in Taiwan. J Alzheimers Dis. 2015;44(2):573–84. doi: 10.3233/JAD-140855 [DOI] [PubMed] [Google Scholar]
- 58.Shi L, Steenland K, Li H, et al. A national cohort study (2000–2018) of long-term air pollution exposure and incident dementia in older adults in the United States. Nat Commun. Nov 19 2021;12(1):6754. doi: 10.1038/s41467-021-27049-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yuan S, Huang X, Zhang L, et al. Associations of air pollution with all-cause dementia, Alzheimer’s disease, and vascular dementia: a prospective cohort study based on 437,932 participants from the UK biobank. Front Neurosci. 2023;17:1216686. doi: 10.3389/fnins.2023.1216686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Oudin A, Segersson D, Adolfsson R, Forsberg B. Association between air pollution from residential wood burning and dementia incidence in a longitudinal study in Northern Sweden. PLoS One. 2018;13(6):e0198283. doi: 10.1371/journal.pone.0198283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Carey IM, Anderson HR, Atkinson RW, et al. Are noise and air pollution related to the incidence of dementia? A cohort study in London, England. BMJ Open. Sep 11 2018;8(9):e022404. doi: 10.1136/bmjopen-2018-022404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Parra KL, Alexander GE, Raichlen DA, Klimentidis YC, Furlong MA. Exposure to air pollution and risk of incident dementia in the UK Biobank. Environ Res. Jun 2022;209:112895. doi: 10.1016/j.envres.2022.112895 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhang H, Shi L, Ebelt ST, et al. Short-term associations between ambient air pollution and emergency department visits for Alzheimer’s disease and related dementias. Environ Epidemiol. Feb 2023;7(1):e237. doi: 10.1097/EE9.0000000000000237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rhew SH, Kravchenko J, Lyerly HK. Exposure to low-dose ambient fine particulate matter PM2.5 and Alzheimer’s disease, non-Alzheimer’s dementia, and Parkinson’s disease in North Carolina. PLoS One. 2021;16(7):e0253253. doi: 10.1371/journal,pone.0253253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wu J, Jackson L. Greenspace Inversely Associated with the Risk of Alzheimer’s Disease in the Mid-Atlantic United States. Earth (Basel). Feb 28 2021;2(1):140–150. doi: 10.3390/earth2010009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhang Z, Chen L, Wang X, et al. Associations of Air Pollution and Genetic Risk With Incident Dementia: A Prospective Cohort Study. Am J Epidemiol. Feb 1 2023;192(2):182–194. doi: 10.1093/aje/kwac188 [DOI] [PubMed] [Google Scholar]
- 67.Grande G, Ljungman PLS, Eneroth K, Bellander T, Rizzuto D. Association Between Cardiovascular Disease and Long-term Exposure to Air Pollution With the Risk of Dementia. JAMA Neurol. Jul 1 2020;77(7):801–809. doi: 10.1001/jamaneurol.2019.4914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Grande G, Hooshmand B, Vetrano DL, et al. Association of Long-term Exposure to Air Pollution and Dementia Risk: The Role of Homocysteine, Methionine, and Cardiovascular Burden. Neurology. Jul 13 2023; doi: 10.1212/WNL.0000000000207656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ilango SD, Chen H, Hystad P, et al. The role of cardiovascular disease in the relationship between air pollution and incident dementia: a population-based cohort study. Int J Epidemiol. Feb 1 2020;49(1):36–44. doi: 10.1093/ije/dyz154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ilango SD, Leary CS, Ritchie E, et al. An Examination of the Joint Effect of the Social Environment and Air Pollution on Dementia Among US Older Adults. Environ Epidemiol. Jun 2023;7(3):e250. doi: 10.1097/EE9.0000000000000250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Liu T, Zhou Y, Wei J, et al. Association between short-term exposure to ambient air pollution and dementia mortality in Chinese adults. Sci Total Environ. Nov 25 2022;849:157860. doi: 10.1016/j.scitotenv.2022.157860 [DOI] [PubMed] [Google Scholar]
- 72.Ma H, Li X, Zhou T, Wang M, Heianza Y, Qi L. Long-term exposure to low-level air pollution, genetic susceptibility and risk of dementia. Int J Epidemiol. Jun 6 2023;52(3):738–748. doi: 10.1093/ije/dyac146 [DOI] [PubMed] [Google Scholar]
- 73.Oudin A, Andersson J, Sundstrom A, et al. Traffic-Related Air Pollution as a Risk Factor for Dementia: No Clear Modifying Effects of APOEvarepsilon4 in the Betula Cohort. J Alzheimers Dis. 2019;71(3):733–740. doi: 10.3233/JAD-181037 [DOI] [PubMed] [Google Scholar]
- 74.Shi L, Zhu Q, Wang Y, et al. Incident dementia and long-term exposure to constituents of fine particle air pollution: A national cohort study in the United States. Proc Natl Acad Sci USA. Jan 3 2023;120(1):e2211282119. doi: 10.1073/pnas.2211282119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Smargiassi A, Sidi EAL, Robert LE, et al. Exposure to ambient air pollutants and the onset of dementia in Quebec, Canada. Environ Res. Nov 2020;190:109870. doi: 10.1016/j.envres.2020.109870 [DOI] [PubMed] [Google Scholar]
- 76.So R, Andersen ZJ, Chen J, et al. Long-term exposure to air pollution and mortality in a Danish nationwide administrative cohort study: Beyond mortality from cardiopulmonary disease and lung cancer. Environ Int. Jun 2022;164:107241. doi: 10.1016/j.envint.2022.107241 [DOI] [PubMed] [Google Scholar]
- 77.Wang X, Younan D, Millstein J, et al. Association of improved air quality with lower dementia risk in older women. Proc Natl Acad Sci USA. Jan 11 2022;119(2)doi: 10.1073/pnas.2107833119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yu Y, Su J, Jerrett M, et al. Air pollution and traffic noise interact to affect cognitive health in older Mexican Americans. Environ Int. Mar 2023;173:107810. doi: 10.1016/j.envint.2023.107810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yuchi W, Sbihi H, Davies H, Tamburic L, Brauer M. Road proximity, air pollution, noise, green space and neurologic disease incidence: a population-based cohort study. Environ Health. Jan 21 2020;19(1):8. doi: 10.1186/s12940-020-0565-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhu Z, Yang Z, Yu L, et al. Residential greenness, air pollution and incident neurodegenerative disease: A cohort study in China. Sci Total Environ. Jun 20 2023;878:163173. doi: 10.1016/j.scitotenv.2023.163173 [DOI] [PubMed] [Google Scholar]
- 81.Tzivian L, Dlugaj M, Winkler A, et al. Long-Term Air Pollution and Traffic Noise Exposures and Mild Cognitive Impairment in Older Adults: A Cross-Sectional Analysis of the Heinz Nixdorf Recall Study. Environ Health Perspect. Sep 2016;24(9):1361–8. doi: 10.1289/ehp.1509824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cerza F, Renzi M, Gariazzo C, et al. Long-term exposure to air pollution and hospitalization for dementia in the Rome longitudinal study. Environ Health. Aug 9 2019;18(1):72. doi: 10.1186/s12940-019-0511-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ge R, Wang Y, Zhang Z, Sun H, Chang J. Association of long-term exposure to various ambient air pollutants, lifestyle, and genetic predisposition with incident cognitive impairment and dementia. BMC Public Health. Jan 15 2024;24(1):179. doi: 10.1186/s12889-024-17702-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Peters S, Bouma F, Hoek G, Janssen N, Vermeulen R. Air pollution exposure and mortality from neurodegenerative diseases in the Netherlands: A population-based cohort study. Environ Res. Jul 2 2024;259:119552. doi: 10.1016/j.envres.2024.119552 [DOI] [PubMed] [Google Scholar]
- 85.Tian F, Qian Z, Zhang Z, et al. Air pollution, APOE genotype and risk of dementia among individuals with cardiovascular diseases: A population-based longitudinal study. Environ Pollut. Apr 15 2024;347:123758. doi: 10.1016/j.envpol.2024.123758 [DOI] [PubMed] [Google Scholar]
- 86.Wang J, Hu X, Yang T, et al. Ambient air pollution and the dynamic transitions of stroke and dementia: a population-based cohort study. EClinicalMedicine. Jan 2024;67:102368. doi: 10.1016/j.eclinm.2023.102368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Tuffier S, Zhang J, Bergmann M, et al. Long-term exposure to air pollution and road traffic noise and incidence of dementia in the Danish Nurse Cohort. Alzheimers Dement. Jun 2024;20(6):4080–4091. doi: 10.1002/aIz.13814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhang B, Langa KM, Weuve J, et al. Hypertension and Stroke as Mediators of Air Pollution Exposure and Incident Dementia. JAMA NetwOpen. Sep 5 2023;6(9):e2333470. doi: 10.1001/jamanetworkopen.2023.33470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Shaffer RM, Blanco MN, Li G, et al. Fine Particulate Matter and Dementia Incidence in the Adult Changes in Thought Study. Environ Health Perspect. Aug 2021;129(8):87001. doi: 10.1289/EHP9018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wu QZ, Zeng HX, Andersson J, et al. Long-term exposure to major constituents of fine particulate matter and neurodegenerative diseases: A population-based survey in the Pearl River Delta Region, China. J Hazard Mater. May 15 2024;470:134161. doi: 10.1016/j.jhazmat.2024.134161 [DOI] [PubMed] [Google Scholar]
- 91.Paul KC, Haan M, Yu Y, et al. Traffic-Related Air Pollution and Incident Dementia: Direct and Indirect Pathways Through Metabolic Dysfunction. J Alzheimers Dis. 2020;76(4):1477–1491. doi: 10.3233/JAD-200320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Andersson J, Oudin A, Sundstrom A, Forsberg B, Adolfsson R, Nordin M. Road traffic noise, air pollution, and risk of dementia - results from the Betula project. Environ Res. Oct 2018;166:334–339. doi: 10.1016/j.envres.2018.06.008 [DOI] [PubMed] [Google Scholar]
- 93.Fania A, Monaco A, Amoroso N, et al. Machine learning and XAI approaches highlight the strong connection between O3 and NO2 pollutants and Alzheimer’s disease. Sci Rep. Mar 5 2024;14(1):5385. doi: 10.1038/s41598-024-55439-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.He F, Tang J, Zhang T, et al. Impact of air pollution exposure on the risk of Alzheimer’s disease in China: A community-based cohort study. Environ Res. Apr 1 2022;205:112318. doi: 10.1016/j.envres.2021.112318 [DOI] [PubMed] [Google Scholar]
- 95.Wood D, Evangelopoulos D, Beevers S, Kitwiroon N, Katsouyanni K. Exposure to Ambient Air Pollution and the Incidence of Dementia in the Elderly of England: The ELSA Cohort. Int J Environ Res Public Health. Nov 29 2022;19(23) doi: 10.3390/ijerph192315889 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Trevenen ML, Heyworth J, Almeida OP, et al. Ambient air pollution and risk of incident dementia in older men living in a region with relatively low concentrations of pollutants: The Health in Men Study. Environ Res. Dec 2022;215(Pt 2):114349. doi: 10.1016/j.envres.2022.114349 [DOI] [PubMed] [Google Scholar]
- 97.Andersen ZJ, Zhang J, Jorgensen JT, et al. Long-term exposure to air pollution and mortality from dementia, psychiatric disorders, and suicide in a large pooled European cohort: ELAPSE study. Environ Int. Dec 2022;170:107581. doi: 10.1016/j.envint.2022.107581 [DOI] [PubMed] [Google Scholar]
- 98.de Crom TOE, Ginos BNR, Oudin A, Ikram MK, Voortman T, Ikram MA. Air Pollution and the Risk of Dementia: The Rotterdam Study. J Alzheimers Dis. 2023;91(2):603–613. doi: 10.3233/JAD-220804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Blanco MN, Shaffer RM, Li G, et al. Traffic-related air pollution and dementia incidence in the Adult Changes in Thought Study. Environ Int. Jan 2024;183:108418. doi: 10.1016/j.envint.2024.108418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Shim JI, Byun G, Lee JT. Long-term exposure to particulate matter and risk of Alzheimer’s disease and vascular dementia in Korea: a national population-based Cohort Study. Environ Health. Apr 14 2023;22(1):35. doi: 10.1186/s12940-023-00986-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Li Z, Christensen GM, Lah JJ, et al. Neighborhood characteristics as confounders and effect modifiers for the association between air pollution exposure and subjective cognitive functioning. Environ Res. Sep 2022;212(Pt A):113221. doi: 10.1016/j.envres.2022.113221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.McMaughan DJ, Oloruntoba O, Smith ML. Socioeconomic Status and Access to Healthcare: Interrelated Drivers for Healthy Aging. Front Public Health. 2020;8:231. doi: 10.3389/fpubh.2020.00231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Frndak S, Deng Z, Ward-Caviness CK, Gorski-Steiner I, Thorpe RJ, Dickerson AS. Risk of dementia due to Co-exposure to air pollution and neighborhood disadvantage. Environ Res. Jun 15 2024;251(Pt 2):118709. doi: 10.1016/j.envres.2024.118709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Alemany S, Crous-Bou M, Vilor-Tejedor N, et al. Associations between air pollution and biomarkers of Alzheimer’s disease in cognitively unimpaired individuals. Environ Int. Dec 2021;157:106864. doi: 10.1016/j.envint.2021.106864 [DOI] [PubMed] [Google Scholar]
- 105.Crous-Bou M, Gascon M, Gispert JD, et al. Impact of urban environmental exposures on cognitive performance and brain structure of healthy individuals at risk for Alzheimer’s dementia. Environ Int May 2020;138:105546. doi: 10.1016/j.envint.2020.105546 [DOI] [PubMed] [Google Scholar]
- 106.Cho J, Jang H, Park H, et al. Alzheimer’s disease-like cortical atrophy mediates the effect of air pollution on global cognitive function. Environ Int Jan 2023;171:107703. doi: 10.1016/j.envint.2022.107703 [DOI] [PubMed] [Google Scholar]
- 107.Falcon C, Gascon M, Molinuevo JL, et al. Brain correlates of urban environmental exposures in cognitively unimpaired individuals at increased risk for Alzheimer’s disease: A study on Barcelona’s population. Alzheimers Dement (Amst). 2021;13(1):e12205. doi: 10.1002/dad2.12205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Casey E, Li Z, Liang D, et al. Association between Fine Particulate Matter Exposure and Cerebrospinal Fluid Biomarkers of Alzheimer’s Disease among a Cognitively Healthy Population-Based Cohort. Environ Health Perspect. Apr 2024;132(4):47001. doi: 10.1289/EHP13503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Calderon-Garciduenas L, Herrera-Soto A, Jury N, et al. Reduced repressive epigenetic marks, increased DNA damage and Alzheimer’s disease hallmarks in the brain of humans and mice exposed to particulate urban air pollution. Environ Res. Apr 2020;183:109226. doi: 10.1016/j.envres.2020.109226 [DOI] [PubMed] [Google Scholar]
- 110.Calderon-Garciduenas L, Gonzalez-Maciel A, Reynoso-Robles R, et al. Alzheimer’s disease and alpha-synuclein pathology in the olfactory bulbs of infants, children, teens and adults </=40 years in Metropolitan Mexico City. APOE4 carriers at higher risk of suicide accelerate their olfactory bulb pathology. Environ Res. Oct 2018;166:348–362. doi: 10.1016/j.envres.2018.06.027 [DOI] [PubMed] [Google Scholar]
- 111.Shaffer RM, Li G, Adar SD, et al. Fine Particulate Matter and Markers of Alzheimer’s Disease Neuropathology at Autopsy in a Community-Based Cohort. J Alzheimers Dis. 2021;79(4):1761–1773. doi: 10.3233/JAD-201005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Calderon-Garciduenas L, Stommel EW, Torres-Jardon R, et al. Alzheimer and Parkinson diseases, frontotemporal lobar degeneration and amyotrophic lateral sclerosis overlapping neuropathology start in the first two decades of life in pollution exposed urbanites and brain ultrafine particulate matter and industrial nanoparticles, including Fe, Ti, Al, V, Ni, Hg, Co, Cu, Zn, Ag, Pt, Ce, La, Pr and W are key players. Metropolitan Mexico City health crisis is in progress. Front Hum Neurosci. 2023;17:1297467. doi: 10.3389/fnhum.2023.1297467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Vanbrabant K, Van Dam D, Bongaerts E, et al. Accumulation of Ambient Black Carbon Particles Within Key Memory-Related Brain Regions. JAMA Netw Open. Apr 1 2024;7(4):e245678. doi: 10.1001/jamanetworkopen.2024.5678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Cullen B, Newby D, Lee D, et al. Cross-sectional and longitudinal analyses of outdoor air pollution exposure and cognitive function in UK Biobank. Sci Rep. Aug 14 2018;8(1):12089. doi: 10.1038/s41598-018-30568-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Calderon-Garciduenas L, Chavez-Franco DA, Luevano-Castro SC, et al. Metals, Nanoparticles, Particulate Matter, and Cognitive Decline. Front Neurol. 2021;12:794071. doi: 10.3389/fneur.2021.794071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lee YG, Yoon SJ, Yoon SH, et al. Air pollution is associated with faster cognitive decline in Alzheimer’s disease. Ann Clin Transl Neurol. Jun 2023;10(6):964–973. doi: 10.1002/acn3.51779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Yang L, Wan W, Yu C, Xuan C, Zheng P, Yan J. Associations between PM(2.5) exposure and Alzheimer’s Disease prevalence Among elderly in eastern China. Environ Health. Nov 29 2022;21(1):119. doi: 10.1186/s12940-022-00937-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Younan D, Wang X, Millstein J, et al. Air quality improvement and cognitive decline in community-dwelling older women in the United States: A longitudinal cohort study. PLoS Med. Feb 2022;19(2):e1003893. doi: 10.1371/journal.pmed.1003893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Franz CE, Gustavson DE, Elman JA, et al. Associations Between Ambient Air Pollution and Cognitive Abilities from Midlife to Early Old Age: Modification by APOE Genotype. J Alzheimers Dis. 2023;93(1):193–209. doi: 10.3233/JAD-221054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ilango SD, Gonzalez K, Gallo L, et al. Long-Term Exposure to Ambient Air Pollution and Cognitive Function Among Hispanic/Latino Adults in San Diego, California. J Alzheimers Dis. 2021;79(4):1489–1496. doi: 10.3233/JAD-200766 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kulick ER, Elkind MSV, Boehme AK, et al. Long-term exposure to ambient air pollution, APOE-epsilon4 status, and cognitive decline in a cohort of older adults in northern Manhattan. Environ Int Mar 2020;136:105440. doi: 10.1016/j.envint.2019.105440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Lo CC, Liu WT, Lu YH, et al. Air pollution associated with cognitive decline by the mediating effects of sleep cycle disruption and changes in brain structure in adults. Environ Sci Pollut Res Int. Jul 2022;29(35):52355–52366. doi: 10.1007/s11356-022-19482-7 [DOI] [PubMed] [Google Scholar]
- 123.Park SY, Han J, Kim SH, Suk HW, Park JE, Lee DY. Impact of Long-Term Exposure to Air Pollution on Cognitive Decline in Older Adults Without Dementia. J Alzheimers Dis. 2022;86(2):553–563. doi: 10.3233/JAD-215120 [DOI] [PubMed] [Google Scholar]
- 124.Ard K, Thomas J, Bullock C. Toxic air pollution and cognitive decline: Untangling particulate matter. Health Place. Aug 16 2024;89:103330. doi: 10.1016/j.healthplace.2024.103330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wang X, Younan D, Petkus AJ, et al. Ambient Air Pollution and Long-Term Trajectories of Episodic Memory Decline among Older Women in the WHIMS-ECHO Cohort. Environ Health Perspect. Sep 2021;129(9):97009. doi: 10.1289/EHP7668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Younan D, Wang X, Gruenewald T, et al. Racial/Ethnic Disparities in Alzheimer’s Disease Risk: Role of Exposure to Ambient Fine Particles. J Gerontol A Biol Sci Med Sci. May 5 2022;77(5):977–985. doi: 10.1093/gerona/glab231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Raichlen DA, Furlong M, Klimentidis YC, et al. Association of Physical Activity with Incidence of Dementia Is Attenuated by Air Pollution. Med Sci Sports Exerc. Jul 1 2022;54(7):1131–1138. doi: 10.1249/MSS.0000000000002888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Yu Y, Haan M, Paul KC, et al. Metabolic dysfunction modifies the influence of traffic-related air pollution and noise exposure on late-life dementia and cognitive impairment: A cohort study of older Mexican-Americans. Environ Epidemiol. Dec 2020;4(6):e122. doi: 10.1097/EE9.0000000000000122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zhang Y, Fu Y, Guan X, et al. Associations of ambient air pollution exposure and lifestyle factors with incident dementia in the elderly: A prospective study in the UK Biobank. Environ Int Jul 4 2024;190:108870. doi: 10.1016/j.envint.2024.108870 [DOI] [PubMed] [Google Scholar]
- 130.Hu HY, Ma YH, Deng YT, et al. Residential greenness and risk of incident dementia: A prospective study of 375,342 participants. Environ Res. Jan 1 2023;216(Pt 3):114703. doi: 10.1016/j.envres.2022.114703 [DOI] [PubMed] [Google Scholar]
- 131.Astell-Burt T, Navakatikyan MA, Feng X. Urban green space, tree canopy and 11-year risk of dementia in a cohort of 109,688 Australians. Environ Int Dec 2020;145:106102. doi: 10.1016/j.envint.2020.106102 [DOI] [PubMed] [Google Scholar]
- 132.Rodriguez-Loureiro L, Gadeyne S, Bauwelinck M, Lefebvre W, Vanpoucke C, Casas L. Long-term exposure to residential greenness and neurodegenerative disease mortality among older adults: a 13-year follow-up cohort study. Environ Health. May 7 2022;21(1):49. doi: 10.1186/s12940-022-00863-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Yu Y, Mayeda ER, Paul KC, et al. Traffic-related Noise Exposure and Late-life Dementia and Cognitive Impairment in Mexican-Americans. Epidemiology. Nov 1 2020;31(6):771–778. doi: 10.1097/EDE.0000000000001249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Fortea J, Pegueroles J, Alcolea D, et al. APOE4 homozygozity represents a distinct genetic form of Alzheimer’s disease. Nat Med. May 2024;30(5):1284–1291. doi: 10.1038/s41591-024-02931-w [DOI] [PubMed] [Google Scholar]
- 135.Jansen IE, Savage JE, Watanabe K, et al. Genome-wide meta-analysis identifies new loci and functional pathways influencing Alzheimer’s disease risk. Nat Genet. Mar 2019;51(3):404–413. doi: 10.1038/s41588-018-0311-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Kim J, Basak JM, Holtzman DM. The role of apolipoprotein E in Alzheimer’s disease. Neuron. Aug 13 2009;63(3):287–303. doi: 10.1016/j.neuron.2009.06.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Klimentidis YC, Raichlen DA, Bea J, et al. Genome-wide association study of habitual physical activity in over 377,000 UK Biobank participants identifies multiple variants including CADM2 and APOE. Int J Obes (Lond). Jun 2018;42(6):1161–1176. doi: 10.1038/s41366-018-0120-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Nuzum H, Stickel A, Corona M, Zeller M, Melrose RJ, Wilkins SS. Potential Benefits of Physical Activity in MCI and Dementia. Behav Neurol. 2020;2020:7807856. doi: 10.1155/2020/7807856 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Shi L, Chen SJ, Ma MY, et al. Sleep disturbances increase the risk of dementia: A systematic review and meta-analysis. Sleep Med Rev. Aug 2018;40:4–16. doi: 10.1016/j.smrv.2017.06.010 [DOI] [PubMed] [Google Scholar]
- 140.Zhang Y, Chen SD, Deng YT, et al. Identifying modifiable factors and their joint effect on dementia risk in the UK Biobank. Nat Hum Behav. Jul 2023;7(7):1185–1195. doi: 10.1038/s41562-023-01585-x [DOI] [PubMed] [Google Scholar]
- 141.Bein KJ, Wallis CD, Silverman JL, Lein PJ, Wexler AS. Emulating Near-Roadway Exposure to Traffic-Related Air Pollution via Real-Time Emissions from a Major Freeway Tunnel System. Environ Sci Technol. Mar 2 2022; doi: 10.1021/acs.est.1c07047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Shang Y, Sun Q. Particulate air pollution: major research methods and applications in animal models. Environ Dis. Jul-Sep 2018;3(3):57–62. doi: 10.4103/ed.ed_16_18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Bendtsen KM, Gren L, Malmborg VB, et al. Particle characterization and toxicity in C57BL/6 mice following instillation of five different diesel exhaust particles designed to differ in physicochemical properties. Part Fibre Toxicol. Aug 8 2020;17(1):38. doi: 10.1186/s12989-020-00369-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Moon KY, Park MK, Leikauf GD, Park CS, Jang AS. Diesel exhaust particle-induced airway responses are augmented in obese rats. Int J Toxicol. Jan-Feb 2014;33(1):21–8. doi: 10.1177/1091581813518355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Daniel S, Phillippi D, Schneider LJ, Nguyen KN, Mirpuri J, Lund AK. Exposure to diesel exhaust particles results in altered lung microbial profiles, associated with increased reactive oxygen species/reactive nitrogen species and inflammation, in C57Bl/6 wildtype mice on a high-fat diet. Part Fibre Toxicol. Jan 8 2021;18(1):3. doi: 10.1186/s12989-020-00393-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Zavala J, Freedman AN, Szilagyi JT, et al. New Approach Methods to Evaluate Health Risks of Air Pollutants: Critical Design Considerations for In Vitro Exposure Testing. Int J Environ Res Public Health. Mar 23 2020;17(6) doi: 10.3390/ijerph17062124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Lakatos HF, Burgess HA, Thatcher TH, et al. Oropharyngeal aspiration of a silica suspension produces a superior model of silicosis in the mouse when compared to intratracheal instillation. Exp Lung Res. May 2006;32(5):181–99. doi: 10.1080/01902140600817465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Silva RM, Doudrick K, Franzi LM, et al. Instillation versus inhalation of multiwalled carbon nanotubes: exposure-related health effects, clearance, and the role of particle characteristics. ACS Nano. Sep 23 2014;8(9):8911–31. doi: 10.1021/nn503887r [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Saveleva L, Vartiainen P, Gorova V, et al. Subacute inhalation of ultrafine particulate matter triggers inflammation without altering amyloid beta load in 5xFAD mice. Neurotoxicology. Mar 2022;89:55–66. doi: 10.1016/j.neuro.2022.01.001 [DOI] [PubMed] [Google Scholar]
- 150.Greve HJ, Dunbar AL, Lombo CG, et al. The bidirectional lung brain-axis of amyloid-beta pathology: ozone dysregulates the peri-plaque microenvironment. Brain. Mar 1 2023;146(3):991–1005. doi: 10.1093/brain/awac113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Ahmed C, Greve HJ, Garza-Lombo C, et al. Peripheral HMGB1 is linked to O(3) pathology of disease-associated astrocytes and amyloid. Alzheimers Dement. May 2024;20(5):3551–3566. doi: 10.1002/alz.13825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Park SJ, Lee J, Lee S, et al. Exposure of ultrafine particulate matter causes glutathione redox imbalance in the hippocampus: A neurometabolic susceptibility to Alzheimer’s pathology. Sci Total Environ. May 20 2020;718:137267. doi: 10.1016/j.scitotenv.2020.137267 [DOI] [PubMed] [Google Scholar]
- 153.Rivas-Arancibia S, Rodriguez-Martinez E, Badillo-Ramirez I, Lopez-Gonzalez U, Saniger JM. Structural Changes of Amyloid Beta in Hippocampus of Rats Exposed to Ozone: A Raman Spectroscopy Study. Front Mol Neurosci. 2017;10:137. doi: 10.3389/fnmol.2017.00137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Rivas-Arancibia S, Guevara-Guzman R, Lopez-Vidal Y, et al. Oxidative stress caused by ozone exposure induces loss of brain repair in the hippocampus of adult rats. Toxicol Sci. Jan 2010;113(1):187–97. doi: 10.1093/toxsci/kfp252 [DOI] [PubMed] [Google Scholar]
- 155.Jew K, Herr D, Wong C, et al. Selective memory and behavioral alterations after ambient ultrafine particulate matter exposure in aged 3xTgAD Alzheimer’s disease mice. Part Fibre Toxicol. Nov 26 2019;16(1):45. doi: 10.1186/s12989-019-0323-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Herr D, Jew K, Wong C, et al. Effects of concentrated ambient ultrafine particulate matter on hallmarks of Alzheimer’s disease in the 3xTgAD mouse model. Neurotoxicology. May 2021;84:172–183. doi: 10.1016/j.neuro.2021.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Kilian JG, Mejias-Ortega M, Hsu HW, et al. Exposure to quasi-ultrafine particulate matter accelerates memory impairment and Alzheimer’s disease-like neuropathology in the AppNL-G-F knock-in mouse model. Toxicol Sci. May 31 2023;193(2):175–191. doi: 10.1093/toxsci/kfad036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Sahu B, Mackos AR, Floden AM, Wold LE, Combs CK. Particulate Matter Exposure Exacerbates Amyloid-beta Plaque Deposition and Gliosis in APP/PS1 Mice. J Alzheimers Dis. 2021;80(2):761–774. doi: 10.3233/JAD-200919 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Jung HA C; Zhao Y; Paulson S,; Anastasio C; Wexler A. Impact of the Versatile Aerosol Concentration Enrichment System (VACES) on Gas Phase Species. Aerosol Science and Technology. 2010:44(12):1113–1121. doi: 10.1080/02786826.2010.512028 [DOI] [Google Scholar]
- 160.Patten KT, Valenzuela AE, Wallis C, et al. The Effects of Chronic Exposure to Ambient Traffic-Related Air Pollution on Alzheimer’s Disease Phenotypes in Wildtype and Genetically Predisposed Male and Female Rats. Environ Health Perspect. May 2021;129(5):57005. doi: 10.1289/EHP8905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Lee SH, Chen YH, Chien CC, et al. Three month inhalation exposure to low-level PM2.5 induced brain toxicity in an Alzheimer’s disease mouse model. PLoS One. 2021;16(8):e0254587. doi: 10.1371/journal.pone.0254587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Patten KT, Valenzuela AE, Wallis C, et al. Hippocampal but Not Serum Cytokine Levels Are Altered by Traffic-Related Air Pollution in TgF344-AD and Wildtype Fischer 344 Rats in a Sex- and Age-Dependent Manner. Front Cell Neurosci. 2022;16:861733. doi: 10.3389/fncel.2022.861733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Fu P, Zhao Y, Dong C, Cai Z, Li R, Yung KKL. An integrative analysis of miRNA and mRNA expression in the brains of Alzheimer’s disease transgenic mice after real-world PM(2.5) exposure. J Environ Sci (China). Dec 2022;122:25–40. doi: 10.1016/j.jes.2021.10.007 [DOI] [PubMed] [Google Scholar]
- 164.Ehsanifar M, Yavari Z, Rafati M. Exposure to urban air pollution particulate matter: neurobehavioral alteration and hippocampal inflammation. Environ Sci Pollut Res Int. Jul 2022;29(33):50856–50866. doi: 10.1007/s11356-022-19367-9 [DOI] [PubMed] [Google Scholar]
- 165.Zhang W, Xiao D, Mao Q, Xia H. Role of neuroinflammation in neurodegeneration development. Signal Transduct Target Ther. Jul 12 2023;8(1):267. doi: 10.1038/s41392-023-01486-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Hopperton KE, Mohammad D, Trepanier MO, Giuliano V, Bazinet RP. Markers of microglia in post-mortem brain samples from patients with Alzheimer’s disease: a systematic review. Mol Psychiatry. Feb 2018;23(2):177–198. doi: 10.1038/mp.2017.246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Wetering JV, Geut H, Bol JJ, et al. Neuroinflammation is associated with Alzheimer’s disease co-pathology in dementia with Lewy bodies. Acta Neuropathol Commun. May 7 2024;12(1):73. doi: 10.1186/s40478-024-01786-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Liu WS, Zhang YR, Ge YJ, Wang HF, Cheng W, Yu JT. Inflammation and Brain Structure in Alzheimer’s Disease and Other Neurodegenerative Disorders: a Mendelian Randomization Study. Mol Neurobiol. Mar 2024;61(3):1593–1604. doi: 10.1007/s12035-023-03648-6 [DOI] [PubMed] [Google Scholar]
- 169.Rangaraju S, Dammer EB, Raza SA, et al. Identification and therapeutic modulation of a proinflammatory subset of disease-associated-microglia in Alzheimer’s disease. Mol Neurodegener. May 21 2018;13(1):24. doi: 10.1186/s13024-018-0254-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Saito T, Saido TC. Neuroinflammation in mouse models of Alzheimer’s disease. Clin Exp Neuroimmunol. Nov 2018;9(4):211–218. doi: 10.1111/cen3.12475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Mucke L, Selkoe DJ. Neurotoxicity of amyloid beta-protein: synaptic and network dysfunction. Cold Spring Harb Perspect Med. Jul 2012;2(7):a006338. doi: 10.1101/cshperspect.a006338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Chen Y, Yu Y. Tau and neuroinflammation in Alzheimer’s disease: interplay mechanisms and clinical translation. J Neuroinflammation. Jul 14 2023;20(1):165. doi: 10.1186/s12974-023-02853-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Rauti R, Cellot G, D’Andrea P, et al. BDNF impact on synaptic dynamics: extra or intracellular long-term release differently regulates cultured hippocampal synapses. Mol Brain. Mar 17 2020;13(1):43. doi: 10.1186/s13041-020-00582-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Stevens B, Allen NJ, Vazquez LE, et al. The classical complement cascade mediates CNS synapse elimination. Cell. Dec 14 2007;131 (6):1164–78. doi: 10.1016/j.cell.2007.10.036 [DOI] [PubMed] [Google Scholar]
- 175.Jantti H, Sitnikova V, Ishchenko Y, et al. Microglial amyloid beta clearance is driven by PIEZO1 channels. J Neuroinflammation. Jun 15 2022;19(1):147. doi: 10.1186/s12974-022-02486-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Chen Z, Jalabi W, Shpargel KB, et al. Lipopolysaccharide-induced microglial activation and neuroprotection against experimental brain injury is independent of hematogenous TLR4. J Neurosci. Aug 22 2012;32(34):11706–15. doi: 10.1523/JNEUROSCI.0730-12.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Fan J, Dong X, Tang Y, et al. Preferential pruning of inhibitory synapses by microglia contributes to alteration of the balance between excitatory and inhibitory synapses in the hippocampus in temporal lobe epilepsy. CNS Neurosci Ther. Oct 2023;29(10):2884–2900. doi: 10.1111/cns.14224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Taddei RN, Perbet R, Mate de Gerando A, et al. Tau Oligomer-Containing Synapse Elimination by Microglia and Astrocytes in Alzheimer Disease. JAMA Neurol. Nov 1 2023;80(11):1209–1221. doi: 10.1001/jamaneurol.2023.3530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Chung HY, Wickel J, Hahn N, et al. Microglia mediate neurocognitive deficits by eliminating C1q-tagged synapses in sepsis-associated encephalopathy. Sci Adv. May 26 2023;9(21):eabq7806. doi: 10.1126/sciadv.abq7806 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Shu J, Fang XH, Li YJ, Deng Y, Wei WS, Zhang L. Microglia-induced autophagic death of neurons via IL-6/STAT3/miR-30d signaling following hypoxia/ischemia. Mol Biol Rep. Aug 2022;49(8):7697–7707. doi: 10.1007/s11033-022-07587-8 [DOI] [PubMed] [Google Scholar]
- 181.Liang P, Zhang X, Zhang Y, et al. Neurotoxic A1 astrocytes promote neuronal ferroptosis via CXCL10/CXCR3 axis in epilepsy. Free Radic Biol Med. Feb 1 2023;195:329–342. doi: 10.1016/j.freeradbiomed.2023.01.002 [DOI] [PubMed] [Google Scholar]
- 182.Hong S, Beja-Glasser VF, Nfonoyim BM, et al. Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science. May 6 2016;352(6286):712–716. doi: 10.1126/science.aad8373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Imai Y, Ibata I, Ito D, Ohsawa K, Kohsaka S. A novel gene iba1 in the major histocompatibility complex class III region encoding an EF hand protein expressed in a monocytic lineage. Biochem Biophys Res Commun. Jul 25 1996;224(3):855–62. doi: 10.1006/bbrc.1996.1112 [DOI] [PubMed] [Google Scholar]
- 184.Ito D, Imai Y, Ohsawa K, Nakajima K, Fukuuchi Y, Kohsaka S. Microglia-specific localisation of a novel calcium binding protein, lba1. Brain Res Mol Brain Res. Jun 1 1998;57(1):1–9. doi: 10.1016/s0169-328x(98)00040-0 [DOI] [PubMed] [Google Scholar]
- 185.Middeldorp J, Hoi EM. GFAP in health and disease. Prog Neurobiol. Mar 2011;93(3):421–43. doi: 10.1016/j.pneurobio.2011.01.005 [DOI] [PubMed] [Google Scholar]
- 186.Eng LF. Glial fibrillary acidic protein (GFAP): the major protein of glial intermediate filaments in differentiated astrocytes. J Neuroimmunol. Jun 1985;8(4–6):203–14. doi: 10.1016/s0165-5728(85)80063-1 [DOI] [PubMed] [Google Scholar]
- 187.Ji X, Liu R, Guo J, et al. Olfactory bulb microglia activation mediated neuronal death in real-ambient particulate matter exposure mice with depression-like behaviors. Sci Total Environ. May 15 2022;821:153456. doi: 10.1016/j.scitotenv.2022.153456 [DOI] [PubMed] [Google Scholar]
- 188.Jang S, Kim EW, Zhang Y, et al. Particulate matter increases beta-amyloid and activated glial cells in hippocampal tissues of transgenic Alzheimer’s mouse: Involvement of PARP-1. Biochem Biophys Res Commun. Jun 2 2018;500(2):333–338. doi: 10.1016/j.bbrc.2018.04.068 [DOI] [PubMed] [Google Scholar]
- 189.Xu MX, Zhu YF, Chang HF, Liang Y. Nanoceria restrains PM2.5-induced metabolic disorder and hypothalamus inflammation by inhibition of astrocytes activation related NF-kappaB pathway in Nrf2 deficient mice. Free Radic Biol Med. Oct 2016;99:259–272. doi: 10.1016/j.freeradbiomed.2016.08.021 [DOI] [PubMed] [Google Scholar]
- 190.Andrade-Oliva MD, Aztatzi-Aguilar OG, Garcia-Sierra F, De Vizcaya-Ruiz A, Arias-Montano JA. Effect of in vivo exposure to ambient fine particles (PM(2.5)) on the density of dopamine D(2)-like receptors and dopamine-induced [(35)S]-GTPgammaS binding in rat prefrontal cortex and striatum membranes. Environ Toxicol Pharmacol. Jun 2018;60:58–65. doi: 10.1016/j.etap.2018.04.001 [DOI] [PubMed] [Google Scholar]
- 191.Zhong L, Sheng X, Wang W, et al. TREM2 receptor protects against complement-mediated synaptic loss by binding to complement C1q during neurodegeneration. Immunity. Aug 8 2023;56(8):1794–1808 e8. doi: 10.1016/j.immuni.2023.06.016 [DOI] [PubMed] [Google Scholar]
- 192.Saha P, Sarkar S, Paidi RK, Biswas SC. TIMP-1: A key cytokine released from activated astrocytes protects neurons and ameliorates cognitive behaviours in a rodent model of Alzheimer’s disease. Brain Behav Immun. Jul 2020;87:804–819. doi: 10.1016/j.bbi.2020.03.014 [DOI] [PubMed] [Google Scholar]
- 193.Chen Z, Jalabi W, Hu W, et al. Microglial displacement of inhibitory synapses provides neuroprotection in the adult brain. Nat Commun. Jul 22 2014;5:4486. doi: 10.1038/ncomms5486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Li B, Chang X, Liang X, et al. The role of reactive astrocytes in neurotoxicity induced by ultrafine particulate matter. Sci Total Environ. Apr 1 2023;867:161416. doi: 10.1016/j.scitotenv.2023.161416 [DOI] [PubMed] [Google Scholar]
- 195.Kang YJ, Tan HY, Lee CY, Cho H. An Air Particulate Pollutant Induces Neuroinflammation and Neurodegeneration in Human Brain Models. Adv Sci (Weinh). Nov 2021;8(21):e2101251. doi: 10.1002/advs.202101251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Weaver DF. Amyloid-beta is a cytokine. Alzheimers Dement. Sep 2023;19(9):4237–4247. doi: 10.1002/alz.13165 [DOI] [PubMed] [Google Scholar]
- 197.Bai R, Guo J, Ye XY, Xie Y, Xie T. Oxidative stress: The core pathogenesis and mechanism of Alzheimer’s disease. Ageing Res Rev. May 2022;77:101619. doi: 10.1016/j.arr.2022.101619 [DOI] [PubMed] [Google Scholar]
- 198.Vilhardt F, Haslund-Vinding J, Jaquet V, McBean G. Microglia antioxidant systems and redox signalling. Br J Pharmacol. Jun 2017;174(12):1719–1732. doi: 10.1111/bph.13426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Suwannasual U, Lucero J, McDonald JD, Lund AK. Exposure to traffic-generated air pollutants mediates alterations in brain microvascular integrity in wildtype mice on a high-fat diet. Environ Res. Jan 2018;160:449–461. doi: 10.1016/j.envres.2017.10.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Ehsanifar M, Montazeri Z, Taheri MA, Rafati M, Behjati M, Karimian M. Hippocampal inflammation and oxidative stress following exposure to diesel exhaust nanoparticles in male and female mice. Neurochem Int. May 2021;145:104989. doi: 10.1016/j.neuint.2021.104989 [DOI] [PubMed] [Google Scholar]
- 201.Cacciottolo M, Morgan TE, Saffari AA, et al. Traffic-related air pollutants (TRAP-PM) promote neuronal amyloidogenesis through oxidative damage to lipid rafts. Free Radic Biol Med. Feb 1 2020;147:242–251. doi: 10.1016/j.freeradbiomed.2019.12.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Lee SH, Lin CY, Chen TF, et al. Distinct brain lipid signatures in response to low-level PM(2.5) exposure in a 3xTg-Alzheimer’s disease mouse inhalation model. Sci Total Environ. Sep 10 2022;838(Pt 4):156456. doi: 10.1016/j.scitotenv.2022.156456 [DOI] [PubMed] [Google Scholar]
- 203.Bai KJ, Chuang KJ, Wu SM, et al. Effects of diesel exhaust particles on the expression of tau and autophagy proteins in human neuroblastoma cells. Environ Toxicol Pharmacol. Sep 2018;62:54–59. doi: 10.1016/j.etap.2018.06.007 [DOI] [PubMed] [Google Scholar]
- 204.Milani C, Corsetto PA, Farina F, et al. Early evidence of stress in immortalized neurons exposed to diesel particles: the role of lipid reshaping behind oxidative stress and inflammation. Toxicology. Nov 1 2018;409:63–72. doi: 10.1016/j.tox.2018.07.017 [DOI] [PubMed] [Google Scholar]
- 205.Wang BR, Shi JQ, Ge NN, et al. PM2.5 exposure aggravates oligomeric amyloid beta-induced neuronal injury and promotes NLRP3 inflammasome activation in an in vitro model of Alzheimer’s disease. J Neuroinflammation. May 2 2018;15(1):132. doi: 10.1186/s12974-018-1178-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Aquino GV, Dabi A, Odom GJ, Zhang F, Bruce ED. Evaluating the endothelial-microglial interaction and comprehensive inflammatory marker profiles under acute exposure to ultrafine diesel exhaust particles in vitro. Toxicology. Apr 30 2021;454:152748. doi: 10.1016/j.tox.2021.152748 [DOI] [PubMed] [Google Scholar]
- 207.Shi Q, Chowdhury S, Ma R, et al. Complement C3 deficiency protects against neurodegeneration in aged plaque-rich APP/PS1 mice. Sci Transl Med. May 31 2017;9(392) doi: 10.1126/scitranslmed.aaf6295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Zhang J, Malik A, Choi HB, Ko RW, Dissing-Olesen L, MacVicar BA. Microglial CR3 activation triggers long-term synaptic depression in the hippocampus via NADPH oxidase. Neuron. Apr 2 2014;82(1):195–207. doi: 10.1016/j.neuron.2014.01.043 [DOI] [PubMed] [Google Scholar]
- 209.Hou L, Wang K, Zhang C, et al. Complement receptor 3 mediates NADPH oxidase activation and dopaminergic neurodegeneration through a Src-Erk-dependent pathway. Redox Biol. Apr 2018;14:250–260. doi: 10.1016/j.redox.2017.09.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Lian H, Litvinchuk A, Chiang AC, Aithmitti N, Jankowsky JL, Zheng H. Astrocyte-Microglia Cross Talk through Complement Activation Modulates Amyloid Pathology in Mouse Models of Alzheimer’s Disease. J Neurosci. Jan 13 2016;36(2):577–89. doi: 10.1523/JNEUROSCI.2117-15.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Kumar A, Barrett JP, Alvarez-Croda DM, Stoica BA, Faden Al, Loane DJ. NOX2 drives M1-like microglial/macrophage activation and neurodegeneration following experimental traumatic brain injury. Brain Behav Immun. Nov 2016;58:291–309. doi: 10.1016/j.bbi.2016.07.158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Zhang H, Haghani A, Mousavi AH, et al. Cell-based assays that predict in vivo neurotoxicity of urban ambient nano-sized particulate matter. Free Radic Biol Med. Dec 2019;145:33–41. doi: 10.1016/j.freeradbiomed.2019.09.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Velazquez-Perez R, Rodriguez-Martinez E, Valdes-Fuentes M, Gelista-Herrera N, Gomez-Crisostomo N, Rivas-Arancibia S. Oxidative Stress Caused by Ozone Exposure Induces Changes in P2X7 Receptors, Neuroinflammation, and Neurodegeneration in the Rat Hippocampus. Oxid Med Cell Longev. 2021;2021:3790477. doi: 10.1155/2021/3790477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Solleiro-Villavicencio H, Hernandez-Orozco E, Rivas-Arancibia S. Effect of exposure to low doses of ozone on interleukin 17A expression during progressive neurodegeneration in the rat hippocampus. Neurologia (Engl Ed). Nov-Dec 2021;36(9):673–680. doi: 10.1016/j.nrleng.2018.08.003 [DOI] [PubMed] [Google Scholar]
- 215.Singh J, Habean ML, Panicker N. Inflammasome assembly in neurodegenerative diseases. Trends Neurosci. Oct 2023;46(10):814–831. doi: 10.1016/j.tins.2023.07.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Bourganis V, Kammona O, Alexopoulos A, Kiparissides C. Recent advances in carrier mediated nose-to-brain delivery of pharmaceutics. Eur J Pharm Biopharm. Jul 2018;128:337–362. doi: 10.1016/j.ejpb.2018.05.009 [DOI] [PubMed] [Google Scholar]
- 217.Ajmani GS, Suh HH, Pinto JM. Effects of Ambient Air Pollution Exposure on Olfaction: A Review. Environ Health Perspect. Nov 2016;124(11):1683–1693. doi: 10.1289/EHP136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Gartziandia O, Egusquiaguirre SP, Bianco J, et al. Nanoparticle transport across in vitro olfactory cell monolayers. Int J Pharm. Feb 29 2016;499(1–2):81–89. doi: 10.1016/j.ijpharm.2015.12.046 [DOI] [PubMed] [Google Scholar]
- 219.Elder A, Gelein R, Silva V, et al. Translocation of inhaled ultrafine manganese oxide particles to the central nervous system. Environ Health Perspect. Aug 2006;114(8):1172–8. doi: 10.1289/ehp.9030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Liang C, Jiang Y, Zhang T, et al. Atmospheric particulate matter impairs cognition by modulating synaptic function via the nose-to-brain route. Sci Total Environ. Jan 20 2023;857(Pt 3):159600. doi: 10.1016/j.scitotenv.2022.159600 [DOI] [PubMed] [Google Scholar]
- 221.Kilian J, Kitazawa M. The emerging risk of exposure to air pollution on cognitive decline and Alzheimer’s disease - Evidence from epidemiological and animal studies. Biomed J. Jun 2018;41(3):141–162. doi: 10.1016/j.bj.2018.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Wang W, Lin Y, Yang H, et al. Internal Exposure and Distribution of Airborne Fine Particles in the Human Body: Methodology, Current Understandings, and Research Needs. Environ Sci Technol. Jun 7 2022;56(11):6857–6869. doi: 10.1021/acs.est.1c07051 [DOI] [PubMed] [Google Scholar]
- 223.You R, Ho YS, Chang RC. The pathogenic effects of particulate matter on neurodegeneration: a review. J Biomed Sci. Feb 22 2022;29(1):15. doi: 10.1186/s12929-022-00799-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Calderon-Garciduenas L, Perez-Calatayud AA, Gonzalez-Maciel A, et al. Environmental Nanoparticles Reach Human Fetal Brains. Biomedicines. Feb 9 2022;10(2)doi: 10.3390/biomedicines10020410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Li Y, Liu Y, Hu C, et al. Study of the neurotoxicity of indoor airborne nanoparticles based on a 3D human blood-brain barrier chip. Environ Int. Oct 2020;143:105598. doi: 10.1016/j.envint.2020.105598 [DOI] [PubMed] [Google Scholar]
- 226.Yamazaki Y, Shinohara M, Shinohara M, et al. Selective loss of cortical endothelial tight junction proteins during Alzheimer’s disease progression. Brain. Apr 1 2019;142(4):1077–1092. doi: 10.1093/brain/awz011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Kurz C, Walker L, Rauchmann BS, Perneczky R. Dysfunction of the blood-brain barrier in Alzheimer’s disease: Evidence from human studies. Neuropathol Appl Neurobiol. Apr 2022;48(3):e12782. doi: 10.1111/nan.12782 [DOI] [PubMed] [Google Scholar]
- 228.Cai Z, Qiao PF, Wan CQ, Cai M, Zhou NK, Li Q. Role of Blood-Brain Barrier in Alzheimer’s Disease. J Alzheimers Dis. 2018;63(4):1223–1234. doi: 10.3233/JAD-180098 [DOI] [PubMed] [Google Scholar]
- 229.Bernardino PN, Hobson BA, Huddleston SL, et al. Time- and region-dependent blood-brain barrier impairment in a rat model of organophosphate-induced status epilepticus. Neurobiol Dis. Oct 15 2023;187:106316. doi: 10.1016/j.nbd.2023.106316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Park JH, Choi JY, Lee HK, Jo C, Koh YH. Notch 1-mediated inflammation is associated with endothelial dysfunction in human brain microvascular endothelial cells upon particulate matter exposure. Arch Toxicol. Feb 2021;95(2):529–540. doi: 10.1007/s00204-020-02942-9 [DOI] [PubMed] [Google Scholar]
- 231.Adivi A, Lucero J, Simpson N, McDonald JD, Lund AK. Exposure to traffic-generated air pollution promotes alterations in the integrity of the brain microvasculature and inflammation in female ApoE(−/−) mice. Toxicol Lett. Mar 15 2021;339:39–50. doi: 10.1016/j.toxlet.2020.12.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Roque PJ, Dao K, Costa LG. Microglia mediate diesel exhaust particle-induced cerebellar neuronal toxicity through neuroinflammatory mechanisms. Neurotoxicology. Sep 2016;56:204–214. doi: 10.1016/j.neuro.2016.08.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Han B, Li X, Ai RS, et al. Atmospheric particulate matter aggravates cns demyelination through involvement of TLR-4/NF-kB signaling and microglial activation. Elife. Feb 24 2022;11 doi: 10.7554/eLife.72247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Pradhan SH, Gibb M, Kramer AT, Sayes CM. Peripheral (lung-to-brain) exposure to diesel particulate matter induces oxidative stress and increased markers for systemic inflammation. Environ Res. Aug 15 2023;231(Pt 3):116267. doi: 10.1016/j.envres.2023.116267 [DOI] [PubMed] [Google Scholar]
- 235.Ljubimova JY, Braubach O, Patil R, et al. Coarse particulate matter (PM(2.5–10)) in Los Angeles Basin air induces expression of inflammation and cancer biomarkers in rat brains. Sci Rep. Apr 9 2018;8(1):5708. doi: 10.1038/s41598-018-23885-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Wallin C, Sholts SB, Osterlund N, et al. Alzheimer’s disease and cigarette smoke components: effects of nicotine, PAHs, and Cd(ll), Cr(lll), Pb(ll), Pb(IV) ions on amyloid-beta peptide aggregation. Sci Rep. Oct 31 2017;7(1):14423. doi: 10.1038/s41598-017-13759-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Best EA, Juarez-Colunga E, James K, LeBlanc WG, Serdar B. Biomarkers of Exposure to Polycyclic Aromatic Hydrocarbons and Cognitive Function among Elderly in the United States (National Health and Nutrition Examination Survey: 2001–2002). PLoS One. 2016;11(2):e0147632. doi: 10.1371/journal,pone.0147632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Liddelow SA, Guttenplan KA, Clarke LE, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. Jan 26 2017;541(7638):481–487. doi: 10.1038/nature21029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Goshi N, Morgan RK, Lein PJ, Seker E. A primary neural cell culture model to study neuron, astrocyte, and microglia interactions in neuroinflammation. J Neuroinflammation. May 11 2020;17(1):155. doi: 10.1186/s12974-020-01819-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Monoson A, Schott E, Ard K, et al. Air pollution and respiratory infections: the past, present, and future. Toxicol Sci. Mar 20 2023;192(1):3–14. doi: 10.1093/toxsci/kfad003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Carraro S, Ferraro VA, Zanconato S. Impact of air pollution exposure on lung function and exhaled breath biomarkers in children and adolescents. J Breath Res. Aug 19 2022;16(4) doi: 10.1088/1752-7163/ac8895 [DOI] [PubMed] [Google Scholar]
- 242.Wang M, Aaron CP, Madrigano J, et al. Association Between Long-term Exposure to Ambient Air Pollution and Change in Quantitatively Assessed Emphysema and Lung Function. JAMA. Aug 13 2019;322(6):546–556. doi: 10.1001/jama.2019.10255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Melbourne CA, Mesut Erzurumluoglu A, Shrine N, et al. Genome-wide gene-air pollution interaction analysis of lung function in 300,000 individuals. Environ Int. Jan 15 2022;159:107041. doi: 10.1016/j.envint.2021.107041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Laratta CR, Kendzerska T, Carlsten C, et al. Air Pollution and Systemic Inflammation in Patients With Suspected OSA Living in an Urban Residential Area. Chest. Oct 2020; 158(4): 1713–1722. doi: 10.1016/j.chest.2020.05.596 [DOI] [PubMed] [Google Scholar]
- 245.Lucking AJ, Lundback M, Mills NL, et al. Diesel exhaust inhalation increases thrombus formation in man. Eur Heart J. Dec 2008;29(24):3043–51. doi: 10.1093/eurheartj/ehn464 [DOI] [PubMed] [Google Scholar]
- 246.Mills NL, Tornqvist H, Robinson SD, et al. Diesel exhaust inhalation causes vascular dysfunction and impaired endogenous fibrinolysis. Circulation. Dec 20 2005;112(25):3930–6. doi: 10.1161/CIRCULATIONAHA.105.588962 [DOI] [PubMed] [Google Scholar]
- 247.Peretz A, Peck EC, Bammler TK, et al. Diesel exhaust inhalation and assessment of peripheral blood mononuclear cell gene transcription effects: an exploratory study of healthy human volunteers. Inhal Toxicol. Nov 2007;19(14):1107–19. doi: 10.1080/08958370701665384 [DOI] [PubMed] [Google Scholar]
- 248.Zhang Z, Hoek G, Chang LY, et al. Particulate matter air pollution, physical activity and systemic inflammation in Taiwanese adults. Int J Hyg Environ Health. Jan 2018;221(1):41–47. doi: 10.1016/j.ijheh.2017.10.001 [DOI] [PubMed] [Google Scholar]
- 249.Thome AD, Faridar A, Beers DR, et al. Functional alterations of myeloid cells during the course of Alzheimer’s disease. Mol Neurodegener. Nov 13 2018;13(1):61. doi: 10.1186/s13024-018-0293-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Butterfield DA, Halliwell B. Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat Rev Neurosci. Mar 2019;20(3):148–160. doi: 10.1038/s41583-019-0132-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Heneka MT, Carson MJ, El Khoury J, et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. Apr 2015;14(4):388–405. doi: 10.1016/S1474-4422(15)70016-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Ransohoff RM. How neuroinflammation contributes to neurodegeneration. Science. Aug 19 2016;353(6301):777–83. doi: 10.1126/science.aag2590 [DOI] [PubMed] [Google Scholar]
- 253.Xie J, Van Hoecke L, Vandenbroucke RE. The Impact of Systemic Inflammation on Alzheimer’s Disease Pathology. Front Immunol. 2021;12:796867. doi: 10.3389/fimmu.2021.796867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Calsolaro V, Edison P. Neuroinflammation in Alzheimer’s disease: Current evidence and future directions. Alzheimers Dement. Jun 2016; 12(6):719–32. doi: 10.1016/j.jalz.2016.02.010 [DOI] [PubMed] [Google Scholar]
- 255.Lutsey PL, Chen N, Mirabelli MC, et al. Impaired Lung Function, Lung Disease, and Risk of Incident Dementia. Am J Respir Crit Care Med. Jun 1 2019;199(11):1385–1396. doi: 10.1164/rccm.201807-1220OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Grande G, Li Y, Trevisan C, et al. Lung function in relation to brain aging and cognitive transitions in older adults: A population-based cohort study. Alzheimers Dement. Jul 5 2024;doi: 10.1002/aIz.14079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Russ TC, Kivimaki M, Batty GD. Respiratory Disease and Lower Pulmonary Function as Risk Factors for Dementia: A Systematic Review With Meta-analysis. Chest. Jun 2020;157(6):1538–1558. doi: 10.1016/j.chest.2019.12.012 [DOI] [PubMed] [Google Scholar]
- 258.Gilsanz P, Mayeda ER, Flatt J, Glymour MM, Quesenberry CP Jr., Whitmer RA. Early Midlife Pulmonary Function and Dementia Risk. Alzheimer Dis Assoc Disord. Oct-Dec 2018;32(4):270–275. doi: 10.1097/WAD.0000000000000253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Mumaw CL, Levesque S, McGraw C, et al. Microglial priming through the lung-brain axis: the role of air pollution-induced circulating factors. FASEB J. May 2016;30(5):1880–91. doi: 10.1096/fj.201500047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Erickson MA, Jude J, Zhao H, et al. Serum amyloid A: an ozone-induced circulating factor with potentially important functions in the lung-brain axis. FASEB J. Sep 2017;31(9):3950–3965. doi: 10.1096/fj.201600857RRR [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Mao Y, Bajinka O, Tang Z, Qiu X, Tan Y. Lung-brain axis: Metabolomics and pathological changes in lungs and brain of respiratory syncytial virus-infected mice. J Med Virol. Dec 2022;94(12):5885–5893. doi: 10.1002/jmv.28061 [DOI] [PubMed] [Google Scholar]
- 262.Hosang L, Canals RC, van der Flier FJ, et al. The lung microbiome regulates brain autoimmunity. Nature. Mar 2022;603(7899):138–144. doi: 10.1038/s41586-022-04427-4 [DOI] [PubMed] [Google Scholar]
- 263.Millan-Zambrano G, Burton A, Bannister AJ, Schneider R. Histone post-translational modifications - cause and consequence of genome function. Nat Rev Genet. Sep 2022;23(9):563–580. doi: 10.1038/s41576-022-00468-7 [DOI] [PubMed] [Google Scholar]
- 264.Sood AJ, Viner C, Hoffman MM. DNAmod: the DNA modification database. J Cheminform. Apr 23 2019;11(1):30. doi: 10.1186/s13321-019-0349-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Zhao LY, Song J, Liu Y, Song CX, Yi C. Mapping the epigenetic modifications of DNA and RNA. Protein Cell. Nov 2020;11(11):792–808. doi: 10.1007/s13238-020-00733-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Creyghton MP, Cheng AW, Welstead GG, et al. Histone H3K27ac separates active from poised enhancers and predicts developmental state. Proc Natl Acad Sci USA. Dec 14 2010;107(50):219316. doi: 10.1073/pnas.1016071107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Martinez-Feduchi P, Jin P, Yao B. Epigenetic modifications of DNA and RNA in Alzheimer’s disease. Front Mol Neurosci. 2024;17:1398026. doi: 10.3389/fnmol.2024.1398026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Cavalli G, Heard E. Advances in epigenetics link genetics to the environment and disease. Nature. Jul 2019;571(7766):489–499. doi: 10.1038/s41586-019-1411-0 [DOI] [PubMed] [Google Scholar]
- 269.Greenberg MVC, Bourc’his D. The diverse roles of DNA methylation in mammalian development and disease. Nat Rev Mol Cell Biol. Oct 2019;20(10):590–607. doi: 10.1038/s41580-019-0159-6 [DOI] [PubMed] [Google Scholar]
- 270.Yang Y, Hsu PJ, Chen YS, Yang YG. Dynamic transcriptomic m(6)A decoration: writers, erasers, readers and functions in RNA metabolism. Cell Res. Jun 2018;28(6):616–624. doi: 10.1038/s41422-018-0040-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Delaunay S, Helm M, Frye M. RNA modifications in physiology and disease: towards clinical applications. Nat Rev Genet. Feb 2024;25(2):104–122. doi: 10.1038/s41576-023-00645-2 [DOI] [PubMed] [Google Scholar]
- 272.Dan J, Chen T. Chapter 3 - Writers, erasers, and readers of DNA and histone methylation marks. In: Gray SG, ed. In Translational Epigenetics, Epigenetic Cancer Therapy. Second Edition ed. 2023:39–63:chap 3. [Google Scholar]
- 273.Cacciottolo M, Wang X, Driscoll I, et al. Particulate air pollutants, APOE alleles and their contributions to cognitive impairment in older women and to amyloidogenesis in experimental models. Transl Psychiatry. Jan 31 2017;7(1):e1022. doi: 10.1038/tp.2016.280 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Zhang L, Wang H, Abel GM, Storm DR, Xia Z. The Effects of Gene-Environment Interactions Between Cadmium Exposure and Apolipoprotein E4 on Memory in a Mouse Model of Alzheimer’s Disease. Toxicological Sciences. 2020 2020;173(1):189–201. doi: 10.1093/toxsci/kfz218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Panitch R, Sahelijo N, Hu J, et al. APOE genotype-specific methylation patterns are linked to Alzheimer disease pathology and estrogen response. Transl Psychiatry. Feb 29 2024;14(1):129. doi: 10.1038/s41398-024-02834-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Cochran JN, Geier EG, Bonham LW, et al. Non-coding and Loss-of-Function Coding Variants in TET2 are Associated with Multiple Neurodegenerative Diseases. Am J Hum Genet. May 7 2020;106(5):632–645. doi: 10.1016/j.ajhg.2020.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Armstrong MJ, Jin Y, Vattathil SM, et al. Role of TET1-mediated epigenetic modulation in Alzheimer’s disease. Neurobiol Dis. Sep 2023;185:106257. doi: 10.1016/j.nbd.2023.106257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Park H, Shin J, Kim Y, Saito T, Saido TC, Kim J. CRISPR/dCas9-Dnmt3a-mediated targeted DNA methylation of APP rescues brain pathology in a mouse model of Alzheimer’s disease. Transl Neurodegener. Sep 15 2022;11(1):41.doi: 10.1186/s40035-022-00314-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Santana DA, Smith MAC, Chen ES. Histone Modifications in Alzheimer’s Disease. Genes (Basel). Jan 29 2023;14(2)doi: 10.3390/genes14020347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.De Jager PL, Srivastava G, Lunnon K, et al. Alzheimer’s disease: early alterations in brain DNA methylation at ANK1, BIN1, RHBDF2 and other loci. Nat Neurosci. Sep 2014;17(9):1156–63. doi: 10.1038/nn.3786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Zheng Y, Sanchez-Guerra M, Zhang Z, et al. Traffic-derived particulate matter exposure and histone H3 modification: A repeated measures study. Environ Res. Feb 2017;153:112–119. doi: 10.1016/j.envres.2016.11.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Ding R, Jin Y, Liu X, et al. Dose- and time-effect responses of DNA methylation and histone H3K9 acetylation changes induced by traffic-related air pollution. Sci Rep. Mar 3 2017;7:43737. doi: 10.1038/srep43737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Jiang X, Liu B, Nie Z, et al. The role of m6A modification in the biological functions and diseases. Signal Transduct Target Ther. Feb 21 2021;6(1):74. doi: 10.1038/s41392-020-00450-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Wang S, Lv W, Li T, et al. Dynamic regulation and functions of mRNA m6A modification. Cancer Cell Int. Jan 29 2022;22(1):48. doi: 10.1186/s12935-022-02452-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Zhu X, Fu H, Sun J, Xu Q. Interaction between N6-methyladenosine (m6A) modification and environmental chemical-induced diseases in various organ systems. Chem Biol Interact. Mar 1 2023;373:110376. doi: 10.1016/j.cbi.2023.110376 [DOI] [PubMed] [Google Scholar]
- 286.Li Z, Su Q, Xu R, et al. Effect of acute PM(2.5) exposure on PTGS2 and RNA m6A modification. Environ Pollut. Oct 15 2023;335:122264. doi: 10.1016/j.envpol.2023.122264 [DOI] [PubMed] [Google Scholar]
- 287.Wu T, Liu B, Wei Y, Li Z. TGF-beta Regulates m(6)A RNA Methylation after PM(2.5) Exposure. Toxics. Dec 16 2023;11(12)doi: 10.3390/toxics11121026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Huang H, Camats-Perna J, Medeiros R, Anggono V, Widagdo J. Altered Expression of the m6A Methyltransferase METTL3 in Alzheimer’s Disease. eNeuro. Sep/Oct 2020;7(5)doi: 10.1523/ENEURO.0125-20.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Zhao F, Xu Y, Gao S, et al. METTL3-dependent RNA m(6)A dysregulation contributes to neurodegeneration in Alzheimer’s disease through aberrant cell cycle events. Mol Neurodegener. Sep 30 2021;16(1):70. doi: 10.1186/s13024-021-00484-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Shafik AM, Zhang F, Guo Z, et al. N6-methyladenosine dynamics in neurodevelopment and aging, and its potential role in Alzheimer’s disease. Genome Biol. Jan 5 2021;22(1):17. doi: 10.1186/s13059-020-02249-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Miguel V, Lamas S, Espinosa-Diez C. Role of non-coding-RNAs in response to environmental stressors and consequences on human health. Redox biology. 2020/10// 2020;37:101580. doi: 10.1016/j.redox.2020.101580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Diao Q, Qin X, Hu N, et al. Long non-coding RNAs mediate the association between short-term PM2.5 exposure and circulating biomarkers of systemic inflammation. Environmental Pollution. 2023/10/15/ 2023;335:122299. doi: 10.1016/j.envpol.2023.122299 [DOI] [PubMed] [Google Scholar]
- 293.Aghaei-Zarch SM, Alipourfard I, Rasoulzadeh H, et al. Non-coding RNAs: An emerging player in particulate matter 2.5-mediated toxicity. Int J Biol Macromol. 2023/04/30/ 2023;235:123790. doi: 10.1016/j.ijbiomac.2023.123790 [DOI] [PubMed] [Google Scholar]
- 294.Zheng R, Gao F, Xiao Y, et al. PM2.5-derived exosomal long noncoding RNA PAET participates in childhood asthma by enhancing DNA damage via m6A-dependent OXPHOS regulation. Environment International. 2024 2024;183:108386. doi: 10.1016/j.envint.2023.108386 [DOI] [PubMed] [Google Scholar]
- 295.Wang L, Rowe RG, Jaimes A, et al. Small-Molecule Inhibitors Disrupt let-7 Oligouridylation and Release the Selective Blockade of let-7 Processing by LIN28. Cell Rep. Jun 5 2018;23(10):3091–3101. doi: 10.1016/j.celrep.2018.04.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Lee JM, Lee JH, Kim YJ. Blockade on Lin28a Prevents Cognitive Impairment and Disruption of the Blood-Brain Barrier Induced by Chronic Cerebral Hypoperfusion. Biomedicines. Apr 5 2022;10(4)doi: 10.3390/biomedicines10040852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Motta V, Angelici L, Nordio F, et al. Integrative Analysis of miRNA and inflammatory gene expression after acute particulate matter exposure. Toxicol Sci. Apr 2013; 132(2):307–16. doi: 10.1093/toxsci/kft013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Luo F, Wu Y, Li Y, et al. PM2.5 regulates the progression of lung adenocarcinoma through the axis of HCG18, miR-195 and ATG14. Clin Exp Pharmacol Physiol. 2024/06// 2024;51(6):e13861. doi: 10.1111/1440-1681.13861 [DOI] [PubMed] [Google Scholar]
- 299.Cao J, Huang M, Guo L, et al. MicroRNA-195 rescues ApoE4-induced cognitive deficits and lysosomal defects in Alzheimer’s disease pathogenesis. Molecular Psychiatry. 2021/09//2021;26(9):4687–4701. doi: 10.1038/s41380-020-0824-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Hoogmartens J, Cacace R, Van Broeckhoven C. Insight into the genetic etiology of Alzheimer’s disease: A comprehensive review of the role of rare variants. Alzheimers Dement (Amst). 2021;13(1):e12155. doi: 10.1002/dad2.12155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Giau W, Bagyinszky E, Yang YS, Youn YC, An SSA, Kim SY. Genetic analyses of early-onset Alzheimer’s disease using next generation sequencing. Sci Rep. Jun 10 2019;9(1):8368. doi: 10.1038/s41598-019-44848-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Cacace R, Sleegers K, Van Broeckhoven C. Molecular genetics of early-onset Alzheimer’s disease revisited. Alzheimers Dement. Jun 2016;12(6):733–48. doi: 10.1016/j.jalz.2016.01.012 [DOI] [PubMed] [Google Scholar]
- 303.Genin E, Hannequin D, Wallon D, et al. APOE and Alzheimer disease: a major gene with semi-dominant inheritance. Mol Psychiatry. Sep 2011;16(9):903–7. doi: 10.1038/mp.2011.52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Yaghmoor F, Noorsaeed A, Alsaggaf S, et al. The Role of TREM2 in Alzheimer’s Disease and Other Neurological Disorders. J Alzheimers Dis Parkinsonism. Nov 2014;4(5) doi: 10.4172/2161-0460.1000160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Kiianitsa K, Lukes ME, Hayes BJ, et al. TREM2 variants that cause early dementia and increase Alzheimer’s disease risk affect gene splicing. Brain. Jul 5 2024;147(7):2368–2383. doi: 10.1093/brain/awae014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Barber IS, Braae A, Clement N, et al. Mutation analysis of sporadic early-onset Alzheimer’s disease using the NeuroX array. Neurobiol Aging. Jan 2017;49:215 e1–215 e8. doi: 10.1016/j.neurobiolaging.2016.09.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Kunkle BW, Grenier-Boley B, Sims R, et al. Genetic meta-analysis of diagnosed Alzheimer’s disease identifies new risk loci and implicates Abeta, tau, immunity and lipid processing. Nat Genet. Mar 2019;51(3):414–430. doi: 10.1038/s41588-019-0358-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Konig T, Wurm R, Parvizi T, et al. C9orf72 repeat length might influence clinical sub-phenotypes in dementia patients. Neurobiol Dis. Dec 2022;175:105927. doi: 10.1016/j.nbd.2022.105927 [DOI] [PubMed] [Google Scholar]
- 309.Guerreiro RJ, Lohmann E, Kinsella E, et al. Exome sequencing reveals an unexpected genetic cause of disease: NOTCH3 mutation in a Turkish family with Alzheimer’s disease. Neurobiol Aging. May 2012;33(5):1008 e17–23. doi: 10.1016/j.neurobiolaging.2011.10.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Armstrong MJ, Jin Y, Allen EG, Jin P. Diverse and dynamic DNA modifications in brain and diseases. Hum Mol Genet. Nov 21 2019;28(R2):R241–R253. doi: 10.1093/hmg/ddz179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Wightman DP, Jansen IE, Savage JE, et al. A genome-wide association study with 1,126,563 individuals identifies new risk loci for Alzheimer’s disease. Nat Genet. Sep 2021;53(9):1276–1282. doi: 10.1038/s41588-021-00921-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Giri M, Zhang M, Lu Y. Genes associated with Alzheimer’s disease: an overview and current status. Clin Interv Aging. 2016;11:665–81. doi: 10.2147/CIA.S105769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Bellenguez C, Kucukali F, Jansen IE, et al. New insights into the genetic etiology of Alzheimer’s disease and related dementias. Nat Genet. Apr 2022;54(4):412–436. doi: 10.1038/s41588-022-01024-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.(NIAGADS) NIoAGoAsDDSS. Top Variants List from the Alzheimer’s Disease Sequencing Project Genome Variant Call. https://adsp.niagads.org/gvc-top-hits-list/
- 315.Wu J, Grande G, Stafoggia M, et al. Air pollution as a risk factor for Cognitive Impairment no Dementia (CIND) and its progression to dementia: A longitudinal study. Environ Int. Feb 2022;160:107067. doi: 10.1016/j.envint.2021.107067 [DOI] [PubMed] [Google Scholar]
- 316.Ronaldson A, Stewart R, Mueller C, et al. Associations between air pollution and mental health service use in dementia: a retrospective cohort study. BMJ Ment Health. Jul 2023;26(1) doi: 10.1136/bmjment-2023-300762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Hu J, Zhang H, Chen S, et al. Identifying PM2.5 and PM0.1 sources for epidemiological studies in California. Environ Sci Technol. May 6 2014;48(9):4980–90. doi: 10.1021/es404810z [DOI] [PubMed] [Google Scholar]
- 318.Calderon-Garciduenas L, Reynoso-Robles R, Gonzalez-Maciel A. Combustion and friction-derived nanoparticles and industrial-sourced nanoparticles: The culprit of Alzheimer and Parkinson’s diseases. Environ Res. Sep 2019;176:108574. doi: 10.1016/j.envres.2019.108574 [DOI] [PubMed] [Google Scholar]
- 319.Lu D, Luo Q, Chen R, et al. Chemical multi-fingerprinting of exogenous ultrafine particles in human serum and pleural effusion. Nat Commun. May 22 2020;11(1):2567. doi: 10.1038/s41467-020-16427-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Kreyling WG, Moller W, Holzwarth U, et al. Age-Dependent Rat Lung Deposition Patterns of Inhaled 20 Nanometer Gold Nanoparticles and their Quantitative Biokinetics in Adult Rats. ACS Nano. Aug 28 2018;12(8):7771–7790. doi: 10.1021/acsnano.8b01826 [DOI] [PubMed] [Google Scholar]
- 321.Lopez-Granero C, Polyanskaya L, Ruiz-Sobremazas D, Barrasa A, Aschner M, Alique M. Particulate Matter in Human Elderly: Higher Susceptibility to Cognitive Decline and Age-Related Diseases. Biomolecules. Dec 26 2023;14(1)doi: 10.3390/biom14010035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Costa LG, Cole TB, Dao K, Chang YC, Coburn J, Garrick JM. Effects of air pollution on the nervous system and its possible role in neurodevelopmenta I and neurodegenerative disorders. Pharmacol Ther. Jun 2020;210:107523. doi: 10.1016/j.pharmthera.2020.107523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Russ TC, Reis S, van Tongeren M. Air pollution and brain health: defining the research agenda. Curr Opin Psychiatry. Mar 2019;32(2):97–104. doi: 10.1097/YCO.0000000000000480 [DOI] [PubMed] [Google Scholar]
- 324.Cho J, Sohn J, Noh J, et al. Association between exposure to polycyclic aromatic hydrocarbons and brain cortical thinning: The Environmental Pollution-Induced Neurological EFfects (EPINEF) study. Sci Total Environ. Oct 1 2020;737:140097. doi: 10.1016/j.scitotenv.2020.140097 [DOI] [PubMed] [Google Scholar]
- 325.Kaumbekova S, Torkmahalleh MA, Shah D. Ambient Benzo[a]pyrene’s Effect on Kinetic Modulation of Amyloid Beta Peptide Aggregation: A Tentative Association between Ultrafine Particulate Matter and Alzheimer’s Disease. Toxics. Dec 14 2022;10(12) doi: 10.3390/toxics10120786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Environment. EPARot. Sulfur Dioxide Emissions. https://cfpub.epa.qov/roe/indicator.cfm?i=22
- 327.Tapia V, Carbajal L, Vasquez V, et al. [Traffic regulation and environmental pollution by particulate material (2.5 and 10), sulfur dioxide, and nitrogen dioxide in Metropolitan Lima, Peru]. Rev Peru Med Exp Salud Publica. Apr-Jun 2018;35(2):190–197. Reordenamiento vehiculary contaminacion ambiental por material particulado (2,5 y 10), dioxido de azufre y dioxido de nitrogeno en Lima Metropolitana, Peru. doi: 10.17843/rpmesp.2018.352.3250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Gaffney JS, Marley NA. Atmospheric chemistry and air pollution. ScientificWorldJournal. Apr 7 2003;3:199–234. doi: 10.1100/tsw.2003.18 [DOI] [PMC free article] [PubMed] [Google Scholar]


