Abstract
Purpose
Relapsed and/or refractory acute myeloid leukemia and high-risk myelodysplastic syndrome continue to have a poor prognosis with limited treatment options despite advancements in rational combination and targeted therapies. Belinostat (an HDAC inhibitor) and Pevonedistat (a NEDD8 inhibitor) have each been independently studied in hematologic malignancies and have tolerable safety profiles with limited single-agent activity. Preclinical studies in AML cell lines and primary AML cells show the combination to be highly synergistic, particularly in high-risk phenotypes such as p53 mutant and FLT-3-ITD positive cells. Here, we present the safety, pharmacokinetics and pharmacodynamics of belinostat and pevonedistat in a dose escalation Phase I study in AML and High-Risk MDS.
Methods
Eighteen patients (16 with AML, 2 with MDS) were treated at 5 dose levels (belinostat 800–1000 mg/m2, pevonedistat 20–50 mg/m2). Safety and tolerability were assessed according to protocol defined dose limiting toxicities (DLTs). Correlative pharmacokinetic and pharmacodynamic analyses were performed.
Results
No dose limiting toxicities were noted. Most Grade 3 or 4 toxicities were hematologic in nature. The best response was stable disease in four patients, and complete remission in one patient who qualified as an exceptional responder. Pharmakokinetic studies revealed no association between drug exposure and best response. Pharmacodynamic RT-PCR studies demonstrated post-treatment increases in several proteins, including quantitative increases in the oxidative stress protein NQO1, ferroptosis protein SLC7A11, and GSR, linked to glutathione metabolism and oxidative stress, as did the anti-oxidants SRXN1 and TXNRD1.
Conclusions
Patterns of post-treatment changes in correlative pharmacodynamic parameters may suggest possible mechanistic changes in the DNA damage response, oxidative damage, and ferroptosis pathways. The combination of pevonedistat plus belinosat is safe in an adult relapsed and/or refractory AML/High-Risk MDS population with modest but notable activity in this heavily treated, high risk population. Our findings also raise the possibility that certain extremely poor prognosis AML patients may respond to a regimen combining two targeted agents that have little or no activity when administered individually.
Trial registration
ClinicalTrials.gov ID NCT03772925, first posted 12/12/2018; CTEP Identifier 10246.
Keywords: Acute myeloid leukemia, Myelodysplastic syndrome, Pevonedistat, Belinostat
Introduction
Despite considerable progress in elucidating the underlying biological mechanisms of myeloid leukemogenesis, Acute Myeloid Leukemia (AML) continues to have a poor prognosis in adults [1, 2], particularly for those with relapsed or refractory disease, with 5 year overall survival of approximately 10% [3–5]. Extensive efforts are underway to improve these outcomes with pathophysiologically rational combination strategies targeting relevant cellular pathways.
Belinostat is a Histone Deacetylase Inhibitor (HDACi) which exerts multiple biologic effects in cancer cells, including induction of apoptosis, cell cycle arrest, and autophagic cell death [6–9]. HDACis act by modifying chromatin structure, leading to an open architecture that promotes the expression of genes associated with cell differentiation and apoptosis [10]. As malignant cells express higher levels of HDAC than their normal counterparts, the latter are relatively protected from HDACi induced DNA damage and cell death [11, 12]. Belinostat has established anti-cancer activity in a variety of tumors, including having an FDA-indication for relapsed/refractory peripheral T-cell lymphoma [13]. It also has been shown to exert single-agent activity in relapsed/refractory AML and myelodysplastic syndrome with some patients displaying stable disease over several months [14, 15]. Combination therapy with belinostat and either azacitidine or bortezomib in advanced myeloid malignancies including AML and MDS has yielded clinically meaningful results in a limited number of patients based upon reports of early phase trials involving heavily pre-treated patients [16, 17].
Pevonedistat is a first-in-class small molecule inhibitor of the NEDD8-activating enzyme (NAE): a component of the NEDD8 conjugation pathway regulating apoptosis via inhibition of the ubiquitin–proteasome system [18]. Preclinical studies of pevonedistat in mouse xenograft models of solid tumors, lymphoma, and AML have demonstrated inhibition of tumor growth, raising the possibility that this new class of agents may be of benefit in a variety of malignancies [19–22]. In the clinical setting, pevonedistat has shown modest single-agent activity in AML with, 17% complete remission (CR) + partial remission (PR) with a Maximum Tolerated Dose of 59 mg/m2 on a D1,3,5 dosing schedule in a 21-day cycle [23], and a non-statistically significant improvement in survival (23.8 vs 20.6mos) in combination with azacitidine in a subset of patients with high-risk MDS in a post-hoc analysis using a substantially lower dose of 20 mg/m2 D1,3,5 [24]. The optimal combinatorial dosing of pevonedistat with other agents is unknown.
Multiple biological considerations support the concept that combined NAE inhibition and HDAC inhibition could be effective in relapsed/refractory AML. For example, by blocking the degradation of IκBα, pevonedistat triggers NF-κB inhibition, which antagonizes myeloid leukemia-initiating cell survival and disables an HDACi compensatory cytoprotective response in AML cells [19, 25]. Both of these agents induce oxidative injury and DNA damage in leukemic cells [8, 19, 26], the latter by promoting accumulation of the DNA licensing factor CDT1 and inducing DNA re-replication [27]. Moreover, HDACis may reciprocally increase pevonedostat anti-leukemic activity by blocking DNA damage responses e.g., homologous recombination and non-homologous end-joining, as well as by disrupting DNA damage checkpoints [28, 29]. Preclinical studies in AML cell lines and primary AML cells show the combination of pevonedistat and belinostat to be highly synergistic, particularly in high-risk phenotypes such as p53 mutant and FLT-3-ITD positive cells [30]. Moreover, a regimen combining pevonedistat and belinostat was well tolerated in NSG AML xenograft models, while significantly improving survival compared to single-agent treatment [30]. Together, these concepts provide a preclinical rationale for this therapeutic combination.
In the present study we assessed the safety and tolerability of pevonedistat combined with belinostat in patients with relapsed/refractory AML or myelodysplastic syndrome, and report toxicities, activity, and pharmacodynamic interactions. The primary objective was determination of the maximum tolerated dose (MTD)/Recommended Phase 2 Dose (PR2D) for pevonedistat with belinostat. Secondary objectives included description of toxicities, analysis of anti-leukemic activity/responses, pharmacokinetics, and assessment of correlative studies characterizing pharmacodynamic interactions between these agents.
Materials and methods
Drug supply
Pevonedistat HCL (MLN4924) was supplied by Takeda. Belinostat (PXD101) was supplied by Spectrum Pharmaceuticals. Both products were distributed by Pharmaceutical Management Branch of National Cancer Institute’s (NCI) Cancer Therapy Evaluation Program (CTEP) andprepared, stored, and administered according to manufacturer’s guidelines.
Study design and participants
This was a phase I multi-center single arm study dose escalation study in a 3 + 3 design of pevonedistat and belinostat in R/R AML or MDS. This NCI Experimental Therapeutics Clinical Trials Network study (NCT03772925) was approved by the Massey Cancer Center Protocol Review committee and Central Human Subjects Review Board. All subjects signed IRB- approved informed consent forms. Eighteen patients were enrolled at two sites in the United States. Patients were > 18 years old with ECOG performance status < or = 2 with adequate organ function based on liver and renal function tests, without an uncontrolled coagulopathy/bleeding disorder, uncontrolled hypertension, baseline QTc prolongation, significant cardiopulmonary disease or arrhythmia, and not having received antineoplastic therapy within 14 days of study drug (except cytoreductive hydroxyurea).
Belinostat was administered by a 30 min intravenous (IV) infusion on days 1 through 5 of each cycle; pevonedistat was administered by a 60 min IV infusion on days 1, 3 and 5 immediately after belinostat. The treatments were repeated on 21-day cycles until disease progression or unacceptable toxicity. The dose escalation schedule is described in Table 1. The regimen was administered on an outpatient basis.
Table 1.
Dose escalation schedule
| Dose level | Belinostat | MLN4G24 (pevonedistat) | ||
|---|---|---|---|---|
| Dose (mg/m2/day) | Administration and schedule | Dose (mg/m2/day) | Administration and schedule | |
| − 1 | 800 | Intravenously over 30 min once daily on days 1–5 of each 21-day cycle | 15 | Intravenously over 60 min once daily on days 1, 3, and 5 of each 21-day cycleb |
| 1a | 800 | 20 | ||
| 2 | 800 | 25 | ||
| 3 | 800 | 37 | ||
| 4 | 1000 | 37 | ||
| 5 | 1000 | 50 | ||
aStarting dose level
bWhen both drugs are given on the same day (Days 1, 3, 5), start the MLN4624 (pevonedistat) infusion within 60 min after the completion of belinostat infusion
Outcomes
Safety and tolerability were assessed according to protocol defined dose limiting toxicities (DLTs). All adverse events (AEs) were characterized and reported according to the NCI Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. Patients who were not evaluable for DLT were replaced.
Bone marrow (BM) aspiration was performed every 2 cycles for treatment response, which was classified using the International Working Group for AML and European Leukemia Net (ELN) to include complete remission (CR), complete remission with incomplete blood count recovery CRi, partial response (PR), stable disease (SD), and progressive disease (PD) [31].
Correlative pharmacokinetic studies
Blood samples for measurement of plasma concentrations were collected on day 1 of cycle 1 for both belinostat and pevonedistat. Belinostat, PK samples were collected pretreatment, 5 min prior to the end of the infusion, and at 0.25, 0.5, 1, 2, 4, 6, 8, and 24 h after the end of the infusion. Pevonedistat, PK samples were collected pretreatment and 0.5, 1, 2, 2.5, 5, and 24 h after the start of the infusion. Plasma was stored at − 80 °C until analysis. Concentrations of pevonedistat were quantified over the range of 1 to 500 ng/mL using a validated liquid chromatography-tandem mass spectrometry (LC/MS/MS) method at Tandem Labs (West Trenton, NJ) [32]. Concentrations of belinostat were determined over the range of 30–5000 ng/mL with dilutions of up to 1:100 v/v using a validated LC/MS/MS method at the Analytical Pharmacology Core Laboratory at the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins [33].
Pharmacokinetic parameters were calculated from individual concentration–time data using standard noncompartmental methods as implemented in Phoenix WinNonlin version 8.2 (Pharsight A Certara Company, Cary, North Carolina). Pharmacokinetic parameters were summarized using descriptive statistics. Differences between the pharmacokinetic parameters between dose levels or BSA-based dose were evaluated statistically via the nonparametric Kruskal–Wallis test. All statistical tests were performed using JMP Statistical Discovery software (version 7; SAS Institute, Cary, NC).
Correlative pharmacodynamic studies
Pharmacodynamic studies were performed on samples from consenting patients with ≥ 40% blasts in the peripheral blood for RT-PCR studies. Alternatively, a flow cytometric assay involving cell permeabilization and intracellular staining of proteins [34] was used to analyze samples consisting of ≥ 10% blasts. In the first case, RNA was isolated at baseline (pre-) and 6 h post treatment. RT-qPCR relative expression levels of various genes was then analyzed. Values for each gene were normalized to the average expression of the endogenous reference genes. Results of analysis were displayed as the mean relative level of assayed target genes ± standard deviations.
Samples were also collected from consenting patients for analysis of changes in protein expression. Specimens were obtained prior to and 24 h (± 6 h) following treatment. Peripheral blood mononuclear cells were isolated from whole blood samples using Ficoll-Hypaque according to the manufacturer’s protocol, and subsequently cryopreserved at − 80 °C. For flow cytometric analysis, a previously described method applicable for AML specimens was employed as previously described with minor modifications [34]. Isolated cells were fixed and permeabilized with True/Phos Perm Buffer (Biolegend), and stained with PE Cy7 conjugated CD45, APC-CD3, and APC Cy7-CD20 antibodies (Biolegend) in conjunction with PE-Bim, PE-p-Chk1, PE-p-cdc2 Y15, FITC-p-cdc2 T14, FITC-ˠH2AX (all Cell Signaling) and FITC-p-HH3 (Biolegend) antibodies as well as the appropriate isocontrols. Cells were analyzed using a BD FACSCanto flow cytometer. Analysis of biomarkers was conducted on the gated CD45dim/low CD3-CD20- leukemic cell population. The mean fluorescence intensity ratio of signal to isocontrol for pretreatment samples for each specimen was set at 100%.
Results
Safety and efficacy measures
Eighteen patients were treated at five dose levels (belinostat 800–1000 mg/m2, pevonedistat 20–50 mg/m2). Thirty-three percent were male, with a median age of 67.5 (range 41–74), 16 of 18 patients had R/R AML (89%), 2 had Myelodysplastic Syndrome (11%) (Table 2). Median number of prior lines of therapy was 3 (range 1–11). Three patients were enrolled and evaluable for DLT in the first of three dose levels. Four subjects were enrolled at dose level four, with one non-evaluable, and five subjects were enrolled at dose level 5 with two non-evaluable. Notably, no dose limiting toxicities were encountered at any dose level.
Table 2.
Baseline characteristics
| Category | Treated patientsa n (%) |
|---|---|
| Age [Year, Mean (SD)] | 63.7 (12.1) |
| Gender | |
| Female | 12 (66.7%) |
| Male | 6 (33.3%) |
| Performance status (ECOG) | |
| 0 | 4 (22%) |
| 1 | 14 (78%) |
| Race | |
| Black of African American | 6 (33%) |
| White | 10 (55.6%) |
| Unknown | 2 (11%) |
| Ethnicity | |
| Hispanic or Latino | 2 (11.1%) |
| Not Hispanic or Latino | 15 (83.3%) |
| Unknown | 1 (5.6%) |
| Disease histology | |
| Acute myeloid leukemia | 16 (89%) |
| Myelodysplastic syndrome | 2 (11%) |
aThe denominator for the percentage is 18 (number of treated patients)
Six patients (33%) experienced one or more grade 3 or greater AEs likely attributable to the study drugs. Excluding electrolyte abnormalities, these toxicities were hematologic in nature (Table 3). Twenty-two percent of patients experienced a decrease in WBC count; 17% of patients experienced a decrease in neutrophil count; 28% of patients had a decrease in platelet counts; 17% anemia; 6% neutropenic fever.
Table 3.
Number of patients with high grade (> = 3) related adverse events
| Toxicitya | Grade 3 n (%) |
Grade 4 n (%) |
Total n (%) |
|---|---|---|---|
| Anemia | 3 (16.7) | 0 (0.0) | 3 (16.7) |
| Febrile neutropenia | 1 (5.6) | 0 (0.0) | 1 (5.6) |
| Fatigue | 1 (5.6) | 0 (0.0) | 1 (5.6) |
| Neutrophil count decreased | 1 (5.6) | 2 (11.1) | 3 (16.7) |
| Platelet count decreased | 1 (5.6) | 4 (22.2) | 5 (27.8) |
| White blood cell decreased | 3 (16.7) | 1 (5.6) | 4 (22.2) |
aWhen a particular patient had the same toxicity on more than one occasion, only the highest grade is listed
The maximum number of cycles reached was 8. A CR was achievedin one patient (6%) (on Dose Level 4), who was considered an exceptional responder.. Four patients had stable disease (22%).
Progressive disease was noted in the remaining patients. The most common reason for treatment discontinuation was disease progression 12 (67%), with 2 (11%) discontinuing due to alternative therapy pursued, and 1 (6%) due to side effects, lung infection, attribution unrelated to study drugs.
Pharmacokinetics, pharmacodynamics, and correlative studies
Belinostat and pevonedistat pharmacokinetic data were available for 16 and 11 patients, respectively (Table 4). Belinostat, belinostat glucuronide, and pevonedistat pharmacokinetic parameters were not different by dose level. When assessing by BSA-based dose, belinostat clearance (p = 0.05) was significantly higher at 1000 mg/m2 compared with 800 mg/m2. Pevonedistat AUCINF (p = 0.02) was significantly higher at 37 mg/m2 compared with 20 or 25 mg/m2. There were no associations between exposure and best response or reason for discontinuation of study.
Table 4.
Plasma pharmacokinetic parameters
| Belinostat | Dose (mg/m2) | preEOI Cmax (µM) | Tmax (h) | AUCINF (µM·h) | T1/2 (h) | Cl (L/hr) |
|---|---|---|---|---|---|---|
| Dose level 1 | 800 | 109.0, 144.5 (2) | 0.4, 0.5 (2) | 54.6, 75.2 (2) | 0.59, 0.67 (2) | 60.1, 87.5 (2) |
| Dose level 2 | 800 | 119.6 ± 50.3 (3) | 0.5 (0.4–0.5; 3) | 64.7 ± 27.4 (3) | 1.36 ± 0.61 (3) | 83.1 ± 30.1 (3) |
| Dose level 3 | 800 | 124.9 ± 35.0 (3) | 0.4 (0.4–0.5; 3) | 61.0, 87.4 (2) | 1.12, 1.46 (2) | 61.8, 82.4 (2) |
| 800 | 123.4 ± 34.3 (8) | 0.4 (0.4–0.5; 8) | 67.5 ± 19.1 (7) | 1.13 ± 0.50 (7) | 77.3 ± 20.7 (7) | |
| Dose level 4 | 1000 | 96.5 ± 46.0 (4) | 0.4 (0.3–0.6; 4) | 60.3 ± 39.2 (3) | 1.02 ± 0.44 (3) | 121.2 ± 60.2 (3) |
| Dose level 5 | 1000 | 113.7 ± 29.4 (3) | 0.4 (0.4–0.4, 3) | 45.1 ± 13.4 (4) | 0.79 ± 0.12 (4) | 138.0 ± 29.1 (4) |
| 1000 | 103.9 ± 37.8 (7) | 0.4 (0.3–0.6; 7) | 51.6 ± 25.8 (7) | 0.89 ± 0.30 (7) | 130.8 ± 41.4 (7) |
| Belinostat glucuronide | Dose (mg/m2) | preEOI Cmax (µM) | Tmax (h) | AUCINF (µM·h) | T1/2 (h) |
|---|---|---|---|---|---|
| Dose level 1 | 800 | 178.4, 246.7 (2) | 0.4, 0.8 (2) | 489.8, 733.6 (2) | 3.42, 3.69 (2) |
| Dose level 2 | 800 | 141.9 ± 18.8 (3) | 0.8 (0.4–0.8; 3) | 380.3 ± 127.2 (3) | 4.29 ± 0.68 (3) |
| Dose level 3 | 800 | 144.2 ± 30.0 (3) | 0.8 (0.7–0.8; 3) | 488.0, 702.3 (2) | 3.04, 4.23 (2) |
| 800 | 160.5 ± 41.5 (8) | 0.8 (0.4–0.8; 8) | 507.8 ± 168.6 (7) | 3.89 ± 0.65 (7) | |
| Dose level 4 | 1000 | 214.0 ± 86.8 (4) | 0.8 (0.3–0.9; 4) | 647.4 ± 390.3 (4) | 3.87 ± 0.91 (4) |
| Dose level 5 | 1000 | 193.0 ± 18.8 (4) | 0.7 (0.4–0.8; 4) | 624.2 ± 165.3 (4) | 4.15 ± 0.83 (4) |
| 1000 | 203.5 ± 59.2 (8) | 0.8 (0.3–0.9; 8) | 635.8 ± 277.7 (8) | 4.01 ± 0.82 (8) |
| Pevonedistat | Dose (mg/m2) | preEOI Cmax (nM) | Tmax (h) | AUCINF (nM·h) | T1/2 (h) | Cl (L/hr) |
|---|---|---|---|---|---|---|
| Dose level 1 | 20 | 278, 311 (2) | 0.4, 0.6 (2) | 2282, 2850 (2) | 5.89, 6.12 (2) | 28.5, 37.6 (2) |
| Dose level 2 | 25 | 372 ± 93.2 (3) | 0.6 (0.4–1.0; 3) | 2952 ± 666 (3) | 6.52 ± 1.38 (3) | 37.5 ± 3.4 (3) |
| Dose level 3 | 37 | 897 ± 26.0 (3) | 0.9 (0.9–1.0; 3) | 4949 ± 426 (3) | 5.97 ± 0.34 (3) | 34.9 ± 2.9 (3) |
| Dose level 4 | 37 | 841 ± 455 (3) | 1.0 (0.6–1.0; 3) | 4601 ± 1423 (3) | 5.61 ± 1.50 (3) | 35.5 ± 8.3 (3) |
| 37 | 869 ± 290 (6) | 0.9 (0.6–1.0; 6) | 4775 ± 958 (6) | 5.79 ± 0.99 (6) | 35.2 ± 5.6 (6) | |
| Dose level 5 | 50 | N.A | N.A | N.A | N.A | N.A |
Data are presented in the table as mean±SD(n). Tmax is presented as median (range; n). If n<3, the actual values are reported
AUCINF area under the plasma concentration–time curve to infinity, Cl systemic clearance, Cmax peak plasma concentration, NA not available, Tmax time to peak concentration, T1/2 half−life
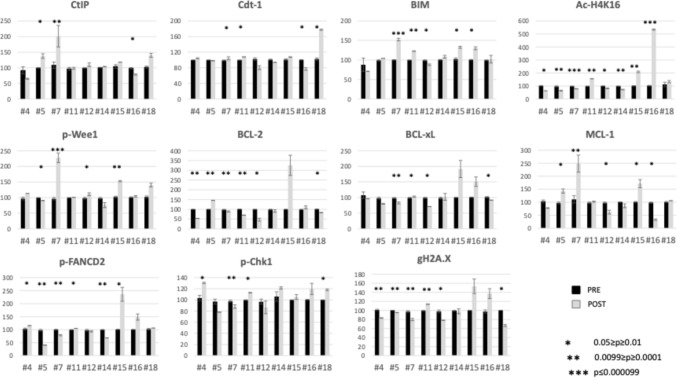
Pharmacodynamic studies employing a flow cytometric assay [34] monitoring intracellular protein levels were performed on peripheral blood leukemic specimens from 9 patients obtained prior to therapy and 24 h after administration of the first dose of pevonedistat. The goal was to determine if changes in DNA damage response proteins observed pre-clinically following pevonedistat/belinostat exposure would be recapitulated in patient cells after in vivo exposure to these agents. Figure 1 represents responses seen in intracellular protein levels. Responses appear variable, with no clear trends elucidated in the evaluated biomarkers, although a relatively consistent increase in BIM was seen in many of the patients analyzed. BIM, a member of the BCL-2 family, is important in apoptotic pathways, and treatment with chemotherapy has previously been identified as inducing BIM [35].
Fig. 1.
Flow cytometry analysis of pre- and post-treatment patient samples. Peripheral blood mononuclear cells (PBMNCs) were isolated at baseline (Pre) and 24 h after treatment (Post). PBMNCs for each sample were analyzed for levels of CtIP, Cdt-1, BIM, Ac-H4K16, p-Wee1, BCL-2, BCL-xL, MCL-1, p-FANCD2, p-Chk1 and gH2AX. Analysis of biomarkers were performed on the CD45dim SSlow CD3- CD20- population. The Mean fluorescence Intensity (MFI) ratios of biomarker signals to their isocontrols for pre-treatment samples for each patient was set as 100%. Analysis for each individual biomarker is represented as a bar chart showing the relative level of the assayed protein in samples after averaging, including standard deviation bars. The Student t test was performed to indicate the significance of changes in post versus pre-treatment samples
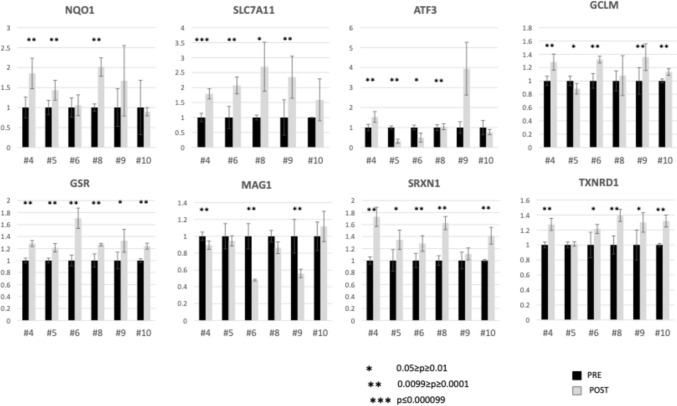
RT-PCR studies performed on samples obtained from 6 patients demonstrated consistent post-treatment increases in several proteins tested (Fig. 2). For example, quantitative increases in the oxidative stress protein NQO1 in all but one patient. Statistically significant increases in SLC7A11, a protein associated with ferroptosis, were noted in all but one sample [36, 37]. GSR, linked to glutathione metabolism and oxidative stress [38] increased in all specimens assayed. Similarly, the anti-oxidants SRXN1 and TXNRD1 increased in 5 specimens analyzed.
Fig. 2.
RT-PCR analysis of pre- and post-treatment patient samples. RNA was isolated from cells at baseline (Pre) and 6 h after treatment (Post). RT-qPCR analysis was performed to determine the relative expression levels of NQO1, SLCA11, ATF3, GCLM, GSR, MAG1, SRXN1 and TXNRD1. The expression of each gene was normalized to the average expression levels of the 4 endogenous reference genes e.g., 18S, B2M, RPLP0 and UBC. Analysis of each individual biomarker is shown as a bar chart illustrating the relative level of the assayed target gene, including standard deviation bars. The Student t test was performed to show the significance of post- versus pre-treatment changes
In summary, results of pharmacodynamic studies monitoring post-treatment protein expression were variable. Trends warranting further analysis have emerged, including those in ferroptosis, glutathione metabolism, and oxidative stress pathways that warrant further exploration. Unfortunately, the exceptional responder did not consent to these studies, thus, conclusions regarding the response patterns of responders could not be drawn.
Underlying leukemic cytogenetic and molecular mutations were not available for most patients, however, the exceptional responder’s AML signature included Trisomy 8, and BRAF V600E and TET2 mutations. A larger sample size would be necessary to determine if any correlates between disease characteristics, Pharmacodynamic, and clinical responses to this regimen exist.
Discussion
This study is the first combination of a NEDD 8 inihibitor and an HDAC inhibitor in the treatment of relapsed/refractory AML or High Risk MDS. The results of this study indicate that the combination of pevonedistat plus belinostat is safe in adult patients with relapsed/refractory AML or high-grade myelodysplastic syndrome. It confers a manageable, primarily hematologic toxicity profile, with a modest activity in this heavily pre-treated population. This indicates that a subset of patients with extremely poor prognostic features may respond to a regimen combining agents with modest (pevonedistat) [23] or minimal (belinostat) [13] single-agent activity. Notably, there was potentially increased signal for anti-leukemic activity as dose levels progressed. Two patients at dose level 3 achieved stable disease and underwent four and eight cycles of therapy, respectively. The patient achieving CR (primary refractory AML with TET 2 and Trisomy 8) was treated at dose level 4. A maximum tolerated dose was not reached and no DLTs were noted at the highest dose level, which was significantly higher than previous combination therapy studies e.g., with 5-azacytidine [24]. Maximum tolerated dose, while a common Phase I endpoint, may not be as clinically relevant as optimal biologic dose (OBD), particularly in targeted agents [39]. Correlative PD studies, such as those included herein, are critical for efforts at determining OBD rather than MTD and can provide background information for future studies with OBD endpoints using either of these agents.
It was not possible to correlate pharmacodynamics features of the regimen with clinical activity given low CR rate. However, the observed patterns of post-treatment changes in pharmacodynamic parameters suggest that for such agents, perturbations in the DDR [40, 41], oxidative damage pathways [38], and potentially ferroptosis [36, 37] represent plausible candidate pharmacodynamic determinants.
As in the case of all such trials employing targeted agents, it is possible that the failure to achieve adequate drug plasma levels might contribute to the limited responses observed. However, it should be noted that pharmacokinetic studies of belinostat [33] and pevonedistat [42] administered at doses similar to those employed here yielded Cmax levels far in excess of those shown to exert activity in preclinical studies e.g., 100–200 nM [38, 39]. The belinostat, belinostat glucuronide, and pevonedistat exposure were similar to previously reported values [33, 43].
After the Phase III PANTHER trial assessing azacitidine vs azacitidine and pevonedistat in the first line setting for patients with high-risk MDS and AML did not achieve its primary endpoint of event-free survival, development of pevonedistat was halted by the sponsor [24]. While the Panther III trial was substantially different from this study in that it involved a different patient population (treatment-naïve vs relapsed/refractory) as well as a different drug combination (pevonedistat plus 5-azacytidine vs an HDACI), important lessons learned from PANTHER III trial may still apply. For example, a signal for improvement in overall survival in a post-hoc analysis was noted for patients receiving greater than three cycles [24], underscoring the importance of continued treatment with regimens incorporating a hypomethylating agent backbone. Our study is suggestive of similar findings, with CR having been achieved only after four cycles.
Conclusions
In summary, the results of the present study indicate that a regimen combining the NEDD8 inhibitor pevonedistat with the HDAC inhibitor belinostat is safe and unassociated with DLTs at the dose levels examined. Significantly, a bona fide exceptional responder was observed at a dose level (37 mg/m2) well below the maximally tolerated pevonedistat dose (59 mg/m2) previously shown to exhibit significant single-agent activity. The PK and PD data as presented herein could have important implications for future iterations of this drug in combination in AML. The strategy of enhancing the anti-leukemic activity of this class of agents with HDACIs might be extended to include alternative clinically relevant agents which disrupt the ubiquitin–proteasome system and induce proteotoxic stress. The latter might include agents such as inhibitors of p97 [44] and ubiquitin-activating enzymes (e.g., TAK-243) [45]. Accordingly, preclinical studies designed to test this possibility are currently underway.
Acknowledgements
The study was supported by the following grants from the National Institutes of Health (NIH): Experimental Therapeutics Clinical Trials Network (grants UM1CA186644 [SAG] and U24CA247648 [MAR]). The project described was also supported by an award from the Leukemia and Lymphoma Society of America (#R6508-18; to S.G.), and the Analytical Pharmacology Core of the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins (NIH grants P30CA006973 [MAR], UL1TR003098 [MAR], and the Shared Instrument Grant S10OD020091 [MAR]). The project was also supported by grant number UL1TR003098 from the National Center for Advancing Translational Sciences (NCATS), a component of the NIH, and the NIH Roadmap for Medical Research and by the WINN career development award. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NCATS or NIH. Services in support of this research were provided by the VCU Massey Comprehensive Cancer Center including Biostatistics Shared Resource, supported in part with funding from NIH-NCI Cancer Center Support Grant P30 CA016059.
Author contributions
KM: Develop and conduct study, collect and analyze data, write paper DS: Conduct study, collect data, write paper DS: Conduct study DB: Analyze data, write paper XD: Analyze data, write paper JW: Study design RP: Study Design MR: Write paper, PK/PD analyses RDH: Pharmacokinetic studies SG: Study design, analyze data, write paper.
Data availability
All supporting data and materials will be made available upon reasonable request.
Declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval and consent to participate
NCI Central IRB served as the IRB or Record for this study. All patients provided signed informed consent prior to participation in this study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bouligny IM, Maher KR, Grant S (2023) Mechanisms of myeloid leukemogenesis: current perspectives and therapeutic objectives. Blood Rev 57:100996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Döhner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H et al (2022) Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 140(12):1345–1377 [DOI] [PubMed] [Google Scholar]
- 3.Nath R, Reddy V, Kapur A, Gebregergish S, Gurskyte L, Kulakova M et al (2019) Survival of relapsed/refractory acute myeloid leukemia (R/R AML) patients receiving stem cell transplantation (SCT). Biol Blood Marrow Transplant 25(3):S125 [Google Scholar]
- 4.Tenold ME, Moskoff BN, Benjamin DJ, Hoeg RT, Rosenberg AS, Abedi M et al (2021) Outcomes of adults with relapsed/refractory acute myeloid leukemia treated with venetoclax plus hypomethylating agents at a comprehensive cancer center. Front Oncol 11:649209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.DeWolf S, Tallman MS (2020) How I treat relapsed or refractory AML. Blood 136(9):1023–1032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen S, Dai Y, Pei X-Y, Grant S (2009) Bim upregulation by histone deacetylase inhibitors mediates interactions with the Bcl-2 antagonist ABT-737: evidence for distinct roles for Bcl-2, Bcl-x<sub>L</sub>, and Mcl-1. Mol Cell Biol 29(23):6149–6169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Aron JL, Parthun MR, Marcucci G, Kitada S, Mone AP, Davis ME et al (2003) Depsipeptide (FR901228) induces histone acetylation and inhibition of histone deacetylase in chronic lymphocytic leukemia cells concurrent with activation of caspase 8-mediated apoptosis and down-regulation of c-FLIP protein. Blood 102(2):652–658 [DOI] [PubMed] [Google Scholar]
- 8.Rosato RR, Almenara JA, Grant S (2003) The histone deacetylase inhibitor MS-275 promotes differentiation or apoptosis in human leukemia cells through a process regulated by generation of reactive oxygen species and induction of p21CIP1/WAF1 1. Cancer Res 63(13):3637–3645 [PubMed] [Google Scholar]
- 9.Robert T, Vanoli F, Chiolo I, Shubassi G, Bernstein KA, Rothstein R et al (2011) HDACs link the DNA damage response, processing of double-strand breaks and autophagy. Nature 471(7336):74–79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Carrier F (2013) Chromatin modulation by histone deacetylase inhibitors: impact on cellular sensitivity to ionizing radiation. Mol Cell Pharmacol 5(1):51–59 [PMC free article] [PubMed] [Google Scholar]
- 11.Ungerstedt JS, Sowa Y, Xu WS, Shao Y, Dokmanovic M, Perez G et al (2005) Role of thioredoxin in the response of normal and transformed cells to histone deacetylase inhibitors. Proc Natl Acad Sci USA 102(3):673–678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee JH, Choy ML, Ngo L, Foster SS, Marks PA (2010) Histone deacetylase inhibitor induces DNA damage, which normal but not transformed cells can repair. Proc Natl Acad Sci USA 107(33):14639–14644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.O’Connor OA, Horwitz S, Masszi T, Van Hoof A, Brown P, Doorduijn J et al (2015) Belinostat in patients with relapsed or refractory peripheral T-cell lymphoma: results of the pivotal phase II BELIEF (CLN-19) study. J Clin Oncol 33(23):2492–2499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kirschbaum MH, Foon KA, Frankel P, Ruel C, Pulone B, Tuscano JM et al (2014) A phase 2 study of belinostat (PXD101) in patients with relapsed or refractory acute myeloid leukemia or patients over the age of 60 with newly diagnosed acute myeloid leukemia: a California Cancer Consortium Study. Leuk Lymphoma 55(10):2301–2304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cashen A, Juckett M, Jumonville A, Litzow M, Flynn PJ, Eckardt J et al (2012) Phase II study of the histone deacetylase inhibitor belinostat (PXD101) for the treatment of myelodysplastic syndrome (MDS). Ann Hematol 91(1):33–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Odenike O, Halpern A, Godley LA, Madzo J, Karrison T, Green M et al (2015) A phase I and pharmacodynamic study of the histone deacetylase inhibitor belinostat plus azacitidine in advanced myeloid neoplasia. Invest New Drugs 33(2):371–379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Holkova B, Tombes MB, Shrader E, Cooke SS, Wan W, Sankala H et al (2011) Phase I trial of belinostat and bortezomib in patients with relapsed or refractory acute leukemia, myelodysplastic syndrome, or chronic myelogenous leukemia in blast crisis. Blood 118(21):2598 [Google Scholar]
- 18.Soucy TA, Smith PG, Milhollen MA, Berger AJ, Gavin JM, Adhikari S et al (2009) An inhibitor of NEDD8-activating enzyme as a new approach to treat cancer. Nature 458(7239):732–736 [DOI] [PubMed] [Google Scholar]
- 19.Swords RT, Kelly KR, Smith PG, Garnsey JJ, Mahalingam D, Medina E et al (2010) Inhibition of NEDD8-activating enzyme: a novel approach for the treatment of acute myeloid leukemia. Blood 115(18):3796–3800 [DOI] [PubMed] [Google Scholar]
- 20.Milhollen MA, Traore T, Adams-Duffy J, Thomas MP, Berger AJ, Dang L et al (2010) MLN4924, a NEDD8-activating enzyme inhibitor, is active in diffuse large B-cell lymphoma models: rationale for treatment of NF-κB–dependent lymphoma. Blood 116(9):1515–1523 [DOI] [PubMed] [Google Scholar]
- 21.Nawrocki ST, Kelly KR, Smith PG, Espitia CM, Possemato A, Beausoleil SA et al (2013) Disrupting protein NEDDylation with MLN4924 Is a novel strategy to target cisplatin resistance in ovarian cancer. Clin Cancer Res 19(13):3577–3590 [DOI] [PubMed] [Google Scholar]
- 22.Luo Z, Yu G, Lee HW, Li L, Wang L, Yang D et al (2012) The Nedd8-activating enzyme inhibitor MLN4924 induces autophagy and apoptosis to suppress liver cancer cell growth. Can Res 72(13):3360–3371 [DOI] [PubMed] [Google Scholar]
- 23.Swords RT, Erba HP, DeAngelo DJ, Bixby DL, Altman JK, Maris M et al (2015) Pevonedistat (MLN4924), a first-in-class NEDD8-activating enzyme inhibitor, in patients with acute myeloid leukaemia and myelodysplastic syndromes: a phase 1 study. Br J Haematol 169(4):534–543 [DOI] [PubMed] [Google Scholar]
- 24.Adès L, Girshova L, Doronin VA, Díez-Campelo M, Valcárcel D, Kambhampati S et al (2022) Pevonedistat plus azacitidine vs azacitidine alone in higher-risk MDS/chronic myelomonocytic leukemia or low-blast-percentage AML. Blood Adv 6(17):5132–5145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guzman ML, Swiderski CF, Howard DS, Grimes BA, Rossi RM, Szilvassy SJ et al (2002) Preferential induction of apoptosis for primary human leukemic stem cells. Proc Natl Acad Sci USA 99(25):16220–16225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Swords RT, Kelly KR, Smith PG, Garnsey JJ, Mahalingam D, Medina E et al (2010) Inhibition of NEDD8-activating enzyme: a novel approach for the treatment of acute myeloid leukemia. Blood 115(18):3796–3800 [DOI] [PubMed] [Google Scholar]
- 27.Glozak MA, Seto E (2009) Acetylation/deacetylation modulates the stability of DNA replication licensing factor Cdt1 *. J Biol Chem 284(17):11446–11453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Blank JL, Liu XJ, Cosmopoulos K, Bouck DC, Garcia K, Bernard H et al (2013) Novel DNA damage checkpoints mediating cell death induced by the NEDD8-activating enzyme inhibitor MLN4924. Cancer Res 73(1):225–234 [DOI] [PubMed] [Google Scholar]
- 29.Brazelle W, Kreahling JM, Gemmer J, Ma Y, Cress WD, Haura E et al (2010) Histone deacetylase inhibitors downregulate checkpoint kinase 1 expression to induce cell death in non-small cell lung cancer cells. PLoS One 5(12):e14335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhou L, Chen S, Zhang Y, Kmieciak M, Leng Y, Li L et al (2016) The NAE inhibitor pevonedistat interacts with the HDAC inhibitor belinostat to target AML cells by disrupting the DDR. Blood 127(18):2219–2230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T et al (2017) Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 129(4):424–447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shah JJ, Jakubowiak AJ, O’Connor OA, Orlowski RZ, Harvey RD, Smith MR et al (2016) Phase I Study of the novel investigational NEDD8-activating enzyme inhibitor pevonedistat (MLN4924) in patients with relapsed/refractory multiple myeloma or lymphoma. Clin Cancer Res 22(1):34–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shafer D, Kagan AB, Rudek MA, Kmieciak M, Tombes MB, Shrader E et al (2023) Phase 1 study of belinostat and adavosertib in patients with relapsed or refractory myeloid malignancies. Cancer Chemother Pharmacol 91(3):281–290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ito S, Ishida Y, Murai K, Kuriya S (2001) Flow cytometric analysis of aberrant antigen expression of blasts using CD45 blast gating for minimal residual disease in acute leukemia and high-risk myelodysplastic syndrome. Leuk Res 25(3):205–211 [DOI] [PubMed] [Google Scholar]
- 35.Harada H, Grant S (2012) Targeting the regulatory machinery of BIM for cancer therapy. Crit Rev Eukaryot Gene Expr 22(2):117–129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Koppula P, Zhuang L, Gan B (2021) Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell 12(8):599–620 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen X, Li J, Kang R, Klionsky DJ, Tang D (2021) Ferroptosis: machinery and regulation. Autophagy 17(9):2054–2081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.McLoughlin MR, Orlicky DJ, Prigge JR, Krishna P, Talago EA, Cavigli IR et al (2019) TrxR1, Gsr, and oxidative stress determine hepatocellular carcinoma malignancy. Proc Natl Acad Sci 116(23):11408–11417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fraisse J, Dinart D, Tosi D, Bellera C, Mollevi C (2021) Optimal biological dose: a systematic review in cancer phase I clinical trials. BMC Cancer 21(1):60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Miller KM, Tjeertes JV, Coates J, Legube G, Polo SE, Britton S et al (2010) Human HDAC1 and HDAC2 function in the DNA-damage response to promote DNA nonhomologous end-joining. Nat Struct Mol Biol 17(9):1144–1151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kinner A, Wu W, Staudt C, Iliakis G (2008) Gamma-H2AX in recognition and signaling of DNA double-strand breaks in the context of chromatin. Nucleic Acids Res 36(17):5678–5694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhou X, Vaishampayan U, Mahalingam D, Harvey RD, Chung KY, Sedarati F et al (2022) Phase 1 study to evaluate the effects of rifampin on pharmacokinetics of pevonedistat, a NEDD8-activating enzyme inhibitor in patients with advanced solid tumors. Invest New Drugs 40(5):1042–1050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Faessel HM, Mould DR, Zhou X, Faller DV, Sedarati F, Venkatakrishnan K (2019) Population pharmacokinetics of pevonedistat alone or in combination with standard of care in patients with solid tumours or haematological malignancies. Br J Clin Pharmacol 85(11):2568–2579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Magnaghi P, D’Alessio R, Valsasina B, Avanzi N, Rizzi S, Asa D et al (2013) Covalent and allosteric inhibitors of the ATPase VCP/p97 induce cancer cell death. Nat Chem Biol 9(9):548–556 [DOI] [PubMed] [Google Scholar]
- 45.Hyer ML, Milhollen MA, Ciavarri J, Fleming P, Traore T, Sappal D et al (2018) A small-molecule inhibitor of the ubiquitin activating enzyme for cancer treatment. Nat Med 24(2):186–193 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All supporting data and materials will be made available upon reasonable request.