Abstract
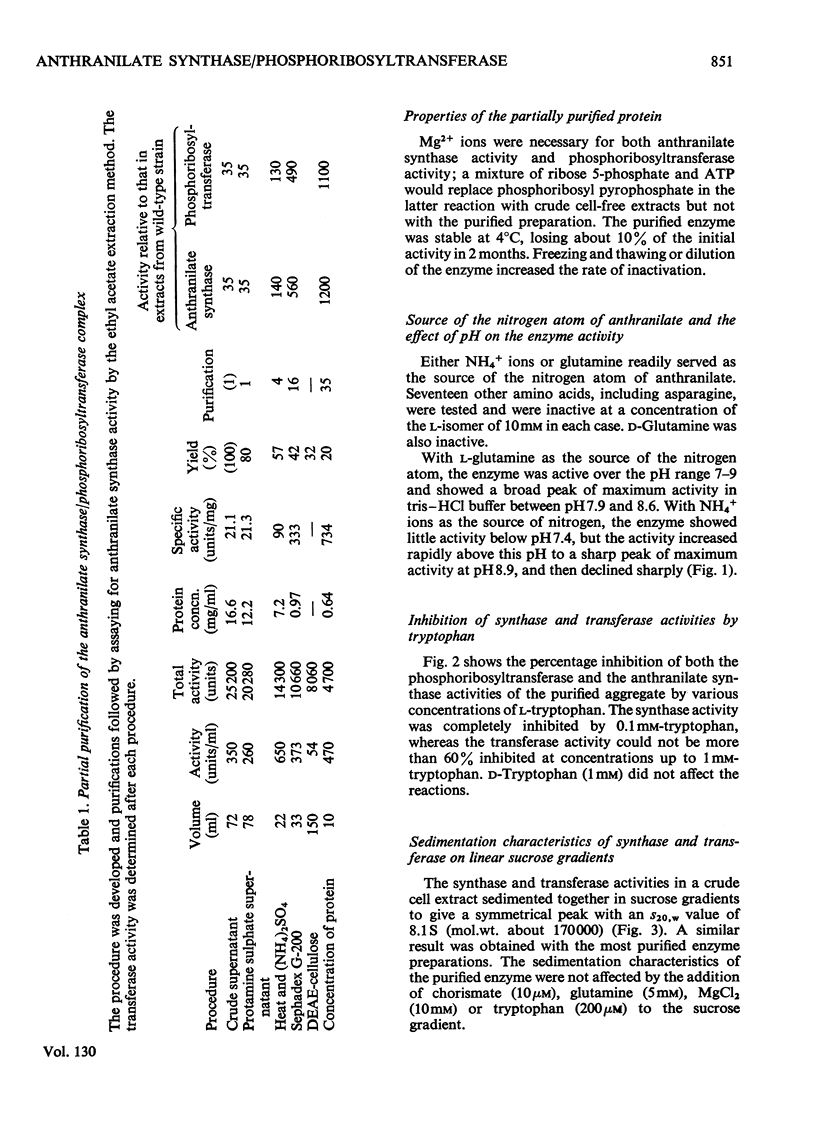
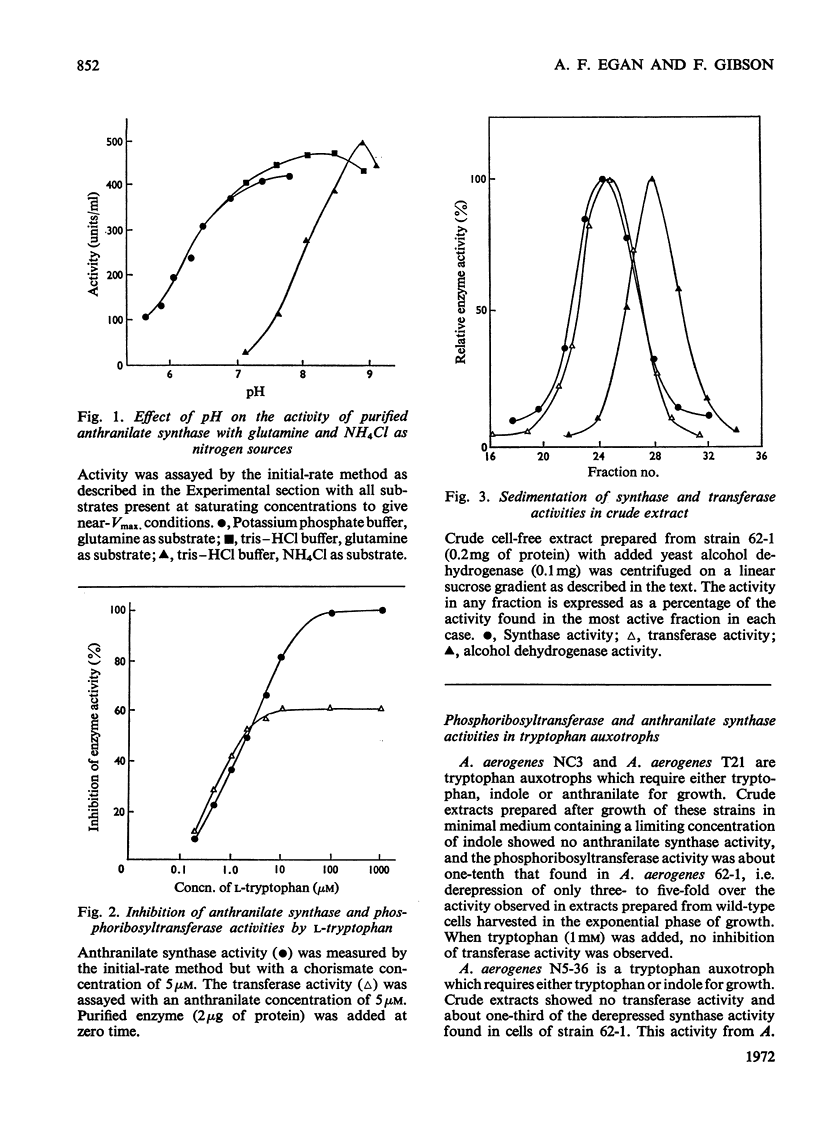
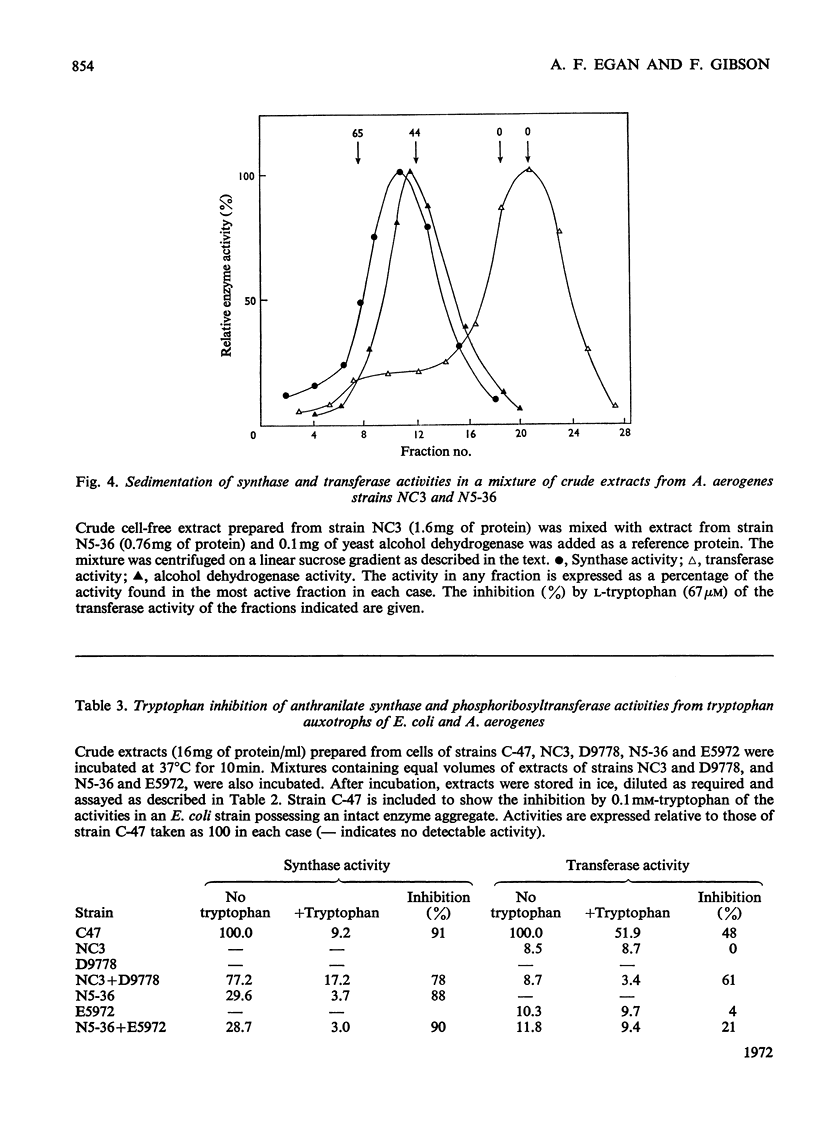
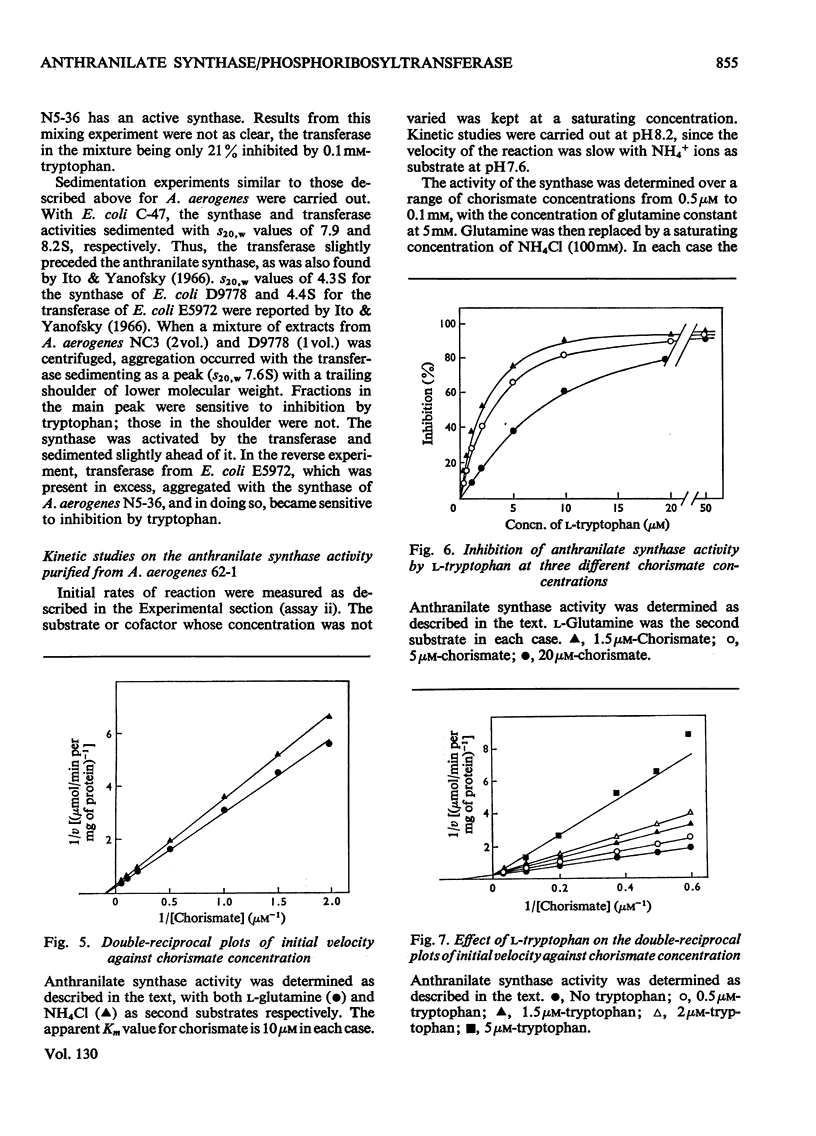
1. Anthranilate synthase and phosphoribosyltransferase from Aerobacter aerogenes purify simultaneously and sediment together on sucrose gradients, showing that they occur as an enzyme aggregate. Both activities of the intact aggregate are subject to inhibition by tryptophan. 2. By using appropriate auxotrophic mutants it was shown that an intact active enzyme aggregate is formed when the components come from separate mutant strains. An intact active aggregate can also be formed when one component is from Escherichia coli and the other from A. aerogenes. 3. Phosphoribosyltransferase of A. aerogenes is active when not in an aggregate with anthranilate synthase, but is not subject to tryptophan inhibition, indicating that the inhibitor site is on the anthranilate synthase component. 4. Anthranilate synthase can be active and sensitive to tryptophan inhibition when complexed with an inactive phosphoribosyltransferase. 5. Kinetic studies on the anthranilate synthase activity show that tryptophan is a competitive inhibitor with respect to chorismate and a non-competitive inhibitor with respect to either glutamine or NH4+ ions. This is consistent with a sequential mechanism of the ordered type in which chorismate is the first reactant.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baker T. I., Crawford I. P. Anthranilate synthetase. Partial purification and some kinetic studies on the enzyme from Escherichia coli. J Biol Chem. 1966 Dec 10;241(23):5577–5584. [PubMed] [Google Scholar]
- Bauerle R. H., Margolin P. A multifunctional enzyme complex in the tryptophan pathway of Salmonella typhimurium: comparison of polarity and pseudopolarity mutations. Cold Spring Harb Symp Quant Biol. 1966;31:203–214. doi: 10.1101/sqb.1966.031.01.028. [DOI] [PubMed] [Google Scholar]
- COTTON R. G., GIBSON F. THE BIOSYNTHESIS OF PHENYLALANINE AND TYROSINE; ENZYMES CONVERTING CHORISMIC ACID INTO PREPHENIC ACID AND THEIR RELATIONSHIPS TO PREPHENATE DEHYDRATASE AND PREPHENATE DEHYDROGENASE. Biochim Biophys Acta. 1965 Apr 12;100:76–88. doi: 10.1016/0304-4165(65)90429-0. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- EDWARDS J. M., GIBSON F., JACKMAN L. M., SHANNON J. S. THE SOURCE OF THE NITROGEN ATOM FOR THE BIOSYNTHESIS OF ANTHRANILIC ACID. Biochim Biophys Acta. 1964 Oct 9;93:78–84. doi: 10.1016/0304-4165(64)90262-4. [DOI] [PubMed] [Google Scholar]
- Enatsu T., Crawford I. P. Enzymes of the tryptophan synthetic pathway in Pseudomonas putida. J Bacteriol. 1968 Jan;95(1):107–112. doi: 10.1128/jb.95.1.107-112.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GOLDTHWAIT D. A. 5-Phosphoribosylamine, a precursor of glycinamide ribotide. J Biol Chem. 1956 Oct;222(2):1051–1068. [PubMed] [Google Scholar]
- Gibson F., Pittard J., Reich E. Ammonium ions as the source of nitrogen for tryptophan biosynthesis in whole cells of Escherichia coli. Biochim Biophys Acta. 1967 Apr 25;136(3):573–576. doi: 10.1016/0304-4165(67)90020-7. [DOI] [PubMed] [Google Scholar]
- Gibson M. I., Gibson F. Preliminary studies on the isolation and metabolism of an intermediate in aromatic biosynthesis: chorismic acid. Biochem J. 1964 Feb;90(2):248–256. doi: 10.1042/bj0900248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson E. J., Nagano H., Zalkin H., Hwang L. H. The anthranilate synthetase-anthranilate 5-phosphoribosylpyrophosphate phosphoribosyltransferase aggregate. Purification of the aggregate and regulatory properties of anthranilate synthetase. J Biol Chem. 1970 Mar 25;245(6):1416–1423. [PubMed] [Google Scholar]
- Ito J., Cox E. C., Yanofsky C. Anthranilate synthetase, an enzyme specified by the tryptophan operon of Escherichia coli: purification and characterization of component I. J Bacteriol. 1969 Feb;97(2):725–733. doi: 10.1128/jb.97.2.725-733.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito J. Hybrid anthranilate synthetase molecules from enterobacterial sources. Nature. 1969 Jul 5;223(5201):57–59. doi: 10.1038/223057a0. [DOI] [PubMed] [Google Scholar]
- Ito J., Yanofsky C. Anthranilate synthetase, an enzyme specified by the tryptophan operon of Escherichia coli: Comparative studies on the complex and the subunits. J Bacteriol. 1969 Feb;97(2):734–742. doi: 10.1128/jb.97.2.734-742.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito J., Yanofsky C. The nature of the anthranilic acid synthetase complex of Escherichia coli. J Biol Chem. 1966 Sep 10;241(17):4112–4114. [PubMed] [Google Scholar]
- Kane J. F., Jensen R. A. The molecular aggregation of anthranilate synthase in Bacillus subtilis. Biochem Biophys Res Commun. 1970 Oct 23;41(2):328–333. doi: 10.1016/0006-291x(70)90507-3. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- MOYED H. S., FRIEDMAN M. Interference with feedback control; a mechanism of antimetabolite action. Science. 1959 Apr 10;129(3354):968–969. doi: 10.1126/science.129.3354.968. [DOI] [PubMed] [Google Scholar]
- Nagano H., Zalkin H., Henderson E. J. The anthranilate synthetase-anthranilate-5-phosphorribosylpyrophosphate phosphoribosyltransferase aggregate. On the reaction mechanism of anthranilate synthetase from Salmonella typhimurium. J Biol Chem. 1970 Aug 10;245(15):3810–3820. [PubMed] [Google Scholar]
- ORNSTEIN L. DISC ELECTROPHORESIS. I. BACKGROUND AND THEORY. Ann N Y Acad Sci. 1964 Dec 28;121:321–349. doi: 10.1111/j.1749-6632.1964.tb14207.x. [DOI] [PubMed] [Google Scholar]
- Pittard J., Wallace B. J. Distribution and function of genes concerned with aromatic biosynthesis in Escherichia coli. J Bacteriol. 1966 Apr;91(4):1494–1508. doi: 10.1128/jb.91.4.1494-1508.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Queener S. F., Gunsalus I. C. Anthranilate synthase enzyme system and complementation in Pseudomonas species. Proc Natl Acad Sci U S A. 1970 Nov;67(3):1225–1232. doi: 10.1073/pnas.67.3.1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivasan P. R. The biosynthesis of anthranilate from [3,4-'+C]glucose in Escherichia coli. Biochemistry. 1965 Dec;4(12):2860–2865. doi: 10.1021/bi00888a043. [DOI] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- Wegman J., Crawford I. P. Tryptophan synthetic pathway and its regulation in Chromobacterium violaceum. J Bacteriol. 1968 Jun;95(6):2325–2335. doi: 10.1128/jb.95.6.2325-2335.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitt D. D., Carlton B. C. Characterization of mutants with single and multiple defects in the tryptophan biosynthetic pathway in Bacillus subtilis. J Bacteriol. 1968 Oct;96(4):1273–1280. doi: 10.1128/jb.96.4.1273-1280.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]


