Abstract
Introduction
The management of de novo non-specific spinal infections (spondylodiscitis - SD) remains inconsistent due to varying clinical practices and a lack of high-level evidence, particularly regarding the indications for surgery.
Research question
This study aims to develop consensus recommendations for the diagnosis and management of SD, addressing diagnostic modalities, surgical indications, and treatment strategies.
Material and methods
A Delphi process was conducted with 26 experts from the European Association of Neurosurgical Societies (EANS). Sixtytwo statements were developed on diagnostic workup, management decisions, surgical techniques, non-surgical treatment, and follow-up and submitted to the panel of experts.
Results
Consensus was reached on 38 of 62 statements. MRI was confirmed as the gold standard for diagnosis. Regarding surgical indications, the panel agreed that any new neurological deficit, even subtle, warrants surgical consideration. Motor deficits with a motor score (MRC) below 4 and bladder or bowel dysfunction were unanimously considered clear indications for surgery. For spinal deformity and instability, thresholds such as kyphosis >20°, scoliosis >10°, and vertebral body collapse >50% were established to guide surgical decision-making. Minimally invasive surgery (MIS) was endorsed whenever feasible, and a 12 week antibiotic treatment regimen was favored in cases of complicated infections.
Discussion and conclusion
This EANS consensus provides updated recommendations for SD management, incorporating recent evidence on improved outcomes with surgical therapy. While these guidelines offer a more structured approach to clinical decision-making, further research is required to optimize surgical timing and validate the long-term impact of these treatment strategies.
Keywords: De novo spinal infection, Spondylodiscitis, Pyogenic, Non-specific, Discitis, Spinal osteomyelitis, Consensus
Highlights
-
•
Motor deficits, including radicular paresis with a motor score (MRC) below 4, are strong indications for surgical intervention.
-
•
MRI remains the gold standard for diagnosing spinal infections.
-
•
Minimally invasive surgery (MIS) is the favored technique, although more extensive surgical techniques may be required.
-
•
A 12-week antibiotic course is recommended for complicated infections (resistant pathogens, older patients, immunosuppression).
-
•
Any dynamic movement, malalignment, alignment changes upon positioning, or axial pain are signs of instability, guiding decision-making.
1. Introduction
Non-specific de novo spondylodiscitis (SD) commonly results from hematogenous spread of infection, with pathogens reaching the spine through arterial circulation or venous routes, such as Batson's plexus (Skaf et al., 2010; Ratcliffe, 1985) (Fig. 1). To a smaller extent, SD can also result from direct inoculation, typically in the context of perioperative or periinterventional infections, though these forms of SD are not discussed in the present consensus statements (Fang et al., 2005). The incidence of de novo non-specific SD is significantly increasing, even in age-adjusted analyses, and the burden of this condition is becoming more apparent through recent studies (Thavarajasingam et al., 2023a; Kramer et al., 2023).
Fig. 1.
Arterial dissemination of spinal infections (A) and venous dissemination via Batson plexus (B).
Clinical consequences of SD typically include spinal pain, with or without neurological deficits from epidural spread of the infection (Lemaignen et al., 2017). Systemic signs of infection, including fever, elevated CRP levels, and leukocytosis, are commonly observed. In more severe or untreated cases, these symptoms may progress to systemic inflammatory response syndrome (SIRS) or sepsis. Furthermore, a potential destruction of disc spaces and vertebral bodies from the infection can lead to instability and consecutive deformity of the affected spinal segments. Upon suspicion of SD, imaging diagnostics are typically performed using MRI, with pathogen identification sought prior to initiating antibiotic treatment (Berbari et al., 2015). While antibiotics remain the cornerstone of SD management, surgical intervention is necessary in certain cases to debride the infection, decompress neural structures, and stabilize the affected spinal segments. Besides the presence of significant neurological deficits, there are no commonly agreed-upon indications for surgery, and a recent survey on the treatment of non-specific spinal infections among European spine surgeons identified a high level of treatment variation, particularly regarding the indications for surgery in SD (Kramer et al., 2024a). A meta-analysis assessing mortality from SD identified a clear survival benefit in patients undergoing early surgical treatment (Thavarajasingam et al., 2023b). This finding is supported by a propensity-matched cohort analysis of conservatively and surgically treated patients with SD, which also revealed a survival benefit from surgical treatment (Neuhoff et al., 2024).
Despite these publications, high level evidence for the indication for surgery in SD remains lacking as does robust evidence for many other aspects regarding diagnostics, treatment and follow-up of SD.
Several clinical scores aim to support decision-making in SD, though validation is still pending, and their relevance remains unclear (Schomig et al., 2022; Pluemer et al., 2023; Heuer et al., 2022; Appalanaidu et al., 2019).
A single prospective randomized study on SD treatment evaluated the duration of antibiotic therapy, comparing regimens of six weeks versus twelve weeks. The study found no significant benefit of the extended twelve-week regimen for infection control (Bernard et al., 2015). However, while a six-week course appears to be sufficient for uncomplicated cases of SD, elderly patients with multiple comorbidities may still require a twelve-week course to achieve adequate infection control.
With many unanswered questions regarding the diagnostics, management decisions, surgical techniques, non-surgical treatments, and follow-up of SD still unanswered—and with the IDSA guidelines now nearly a decade old—the present EANS consensus statement aims to provide updated recommendations for managing patients with SD. These recommendations are based on a Delphi consensus process conducted by an expert panel from the European Association of Neurosurgical Societies (EANS).
2. Material and Methods
As an initial step, a steering committee of members of the spine section of the European Association of Neurosurgical Societies (EANS) was established in May 2023, including FR, ES, and AD. The steering committee identified experts in the treatment of spinal pathologies associated with the EANS, based on clinical experience, relevant publications in PubMed and teaching activities in spine surgery. Thirty experts were identified and contacted, 26 agreed to contribute to the Delphi survey.
The first survey round included 54 open-ended questions related to the diagnosis and management of de novo non-specific spinal infections (supplementary material). Questions were designed by FR and edited by FR, ES, and AD. The questionnaire was sent out via the SurveyMonkey™ platform in June 2023.
From the responses to the open-ended questions, 62 statements were extracted by AK, ST, and FR, additionally edited by FR and ES. Statements were grouped into 5 categories: i) diagnostic workup (18 statements), ii) management decision (19 statements), iii) surgical technique (10 statements), iv) non-surgical treatment aspects (7 statements), and v) treatment monitoring and follow-up (8 statements). Statements were again distributed to the expert group using the SurveyMonkey™ platform and Likert scale responses (strongly agree, agree, neutral, disagree, strongly disagree) were demanded. Each statement had the additional option to add a free-text comment. Statements were sent to the expert group in October 2023.
Responses from the second round with ≥80% of experts choosing agreement (strongly agree or agree) were judged as consensus to accept the statement, while ≥80% disagreement (disagree or strongly disagree) was considered as consensus to reject the statement. An agreement or disagreement of 70–79% was categorized as borderline consensus, and any value below 70% as no consensus.
In a third round of the Delphi process experts received statements with borderline or no consensus for reevaluation. Depending on the responses and comments of the second round, statements were either unchanged, adjusted, or discarded. Experts were asked for their approval or rejection of the statements, using a two-point scale asking for agreement or disagreement.
Final responses were categorized as consensus, borderline consensus, or no consensus according to the results of round 3 and the predefined thresholds of round 2. A final document including all statements was generated and reviewed by all participants.
The Delphi process is summarized in Fig. 2.
Fig. 2.
Synopsis of the Delphi process.
3. Results
3.1. Step 1
Twenty-six experts responded to the first round. From the responses, 62 statements were generated and grouped into five categories: i) diagnostic workup (18 statements), ii) management decision (19 statements), iii) surgical technique (10 statements), iv) non-surgical treatment aspects (7 statements), and v) treatment monitoring and follow-up (8 statements).
3.2. Step 2
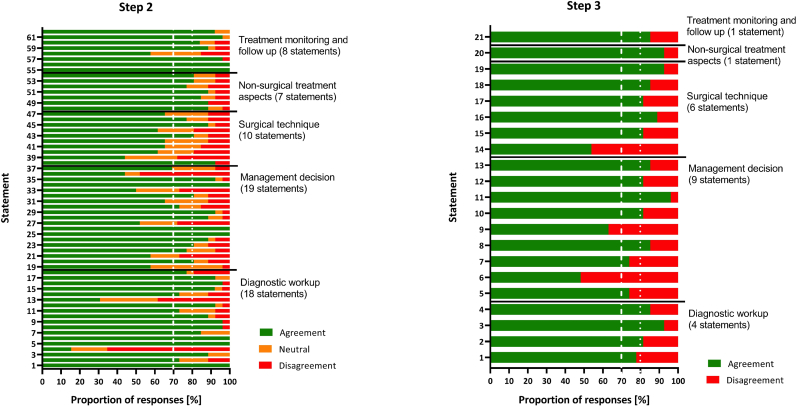
After the distribution of the statements to the expert group, 26 experts responded. According to the predefined thresholds, consensus was reached for 38 of 62 statements (Fig. 2). Borderline consensus was reached for another 8 statements, while no consensus was reached for 16 statements. Related to the five subcategories of statements, consensus, borderline consensus, and no consensus were reached for 12, 4, and 2 statements of the 18 statements related to diagnostic workup; for 10, 2, and 7 statements of the 19 statements related to management decision; for 3, 1, and 6 statements of the 10 statements related to surgical technique; for 6, 1, and 0 statements of the 7 statements related to non-surgical treatment aspects; and for 7, 0, and 1 statements of the 8 statements related to treatment monitoring and follow-up, respectively. Of the 24 statements without consensus, 7 remained unchanged for another survey round but were further discussed based on the comments, 14 statements were rephrased according to the comments, and 3 statements were discarded (2 on diagnostic workup, 1 on surgical technique) (Fig. 3).
Fig. 3.
Delphi process responses of round 2 (A) and 3 (B).
3.3. Step 3
Responses from 27 experts were available. Of the 21 statements in step 3, consensus among the expert group was reached for 15, while no final consensus could be reached for 6 statements (Fig. 3).
3.4. Consensus
The statements for which consensus, as well as no consensus, were reached are shown in the following tables: diagnostic workup in Table 1, management decisions in Table 2, surgical techniques in Table 3, non-surgical treatment aspects in Table 4, and monitoring and follow-up in Table 5.
Table 1.
Statements regarding ‘diagnostic workup’ and proportion of expert agreement leading to consensus or no consensus.
| Diagnostic workup | ||
|---|---|---|
| No | Statement | Level of consensus |
| 1 | For initial diagnosis of a spinal infection MRI−/+C of the suspected spinal region should be done | 100% |
| 2 | Upon imaging diagnosis of a spinal infection a whole spine MRI−/+C should be performed to exclude non-contiguous spinal lesions | 77%, no consensus |
| 3 | A CT scan of the infected segments should be done to assess bony destruction | 88% |
| 4 | A CT scan of the whole spine is not necessary, a CT scan of the affected levels to assess for bony destructions should be done | 81% |
| 5 | A set of blood cultures should be taken upon suspected diagnosis of a spinal infection and prior to beginning of any antibiotic therapy | 100% |
| 6 | If blood cultures remain negative and further therapy is non-surgical or surgical therapy is delayed a CT-guided biopsy should be performed | 100% |
| 7 | For maximal biopsy yield, the contrast enhancing part of the disc should be targeted for biopsy | 85% |
| 8 | Biopsy or surgical specimen should always be sent for microbiological and histopathological evaluation | 96% |
| 9 | CRP is the most relevant laboratory parameter for initial diagnosis and treatment monitoring | 96% |
| 10 | WBCC can be used for diagnosis and follow up as well but is less sensitive than CRP and might be not elevated | 88% |
| 11 | Despite its sensitivity, ESR is in general no longer performed at many institutions. Therefore, it is dispensable in the diagnosis and monitoring of spondylodiscitis | 93% |
| 12 | To identify a potential primary focus of infection a CT thorax/abdomen/pelvis, a cardiac ultrasound, dental and skin examination, urinary laboratory and evaluation of any implants should be performed | 92% |
| 13 | Even in cases of an obvious source of infection a full workup should be done to exclude any further foci | 85% |
| 14 | FDG-PET is reserved for selected cases and not part of a routine workup or routine follow-up | 92% |
| 15 | Standing x-rays are used as comparison to supine MRI/CT to rule out instability/deformity where suspected | 96% |
| 16 | Parameters making instability/deformity likely: Back pain upon recumbency, severe bony destruction, deformity in supine position | 92% |
Table 2.
tatements regarding ‘management decision’ and proportion of expert agreement leading to consensus or no consensus.
| Management decision | ||
|---|---|---|
| No | Statement | Level of consensus |
| 1 | Although advanced patient age, frailty and comorbidities are relevant risk factors for surgery, old/frail/multimorbid patients will benefit the most from surgery regarding control of the spinal infection | 74%, no consensus |
| 2 | Higher age/frailty/relevant comorbidities should not be factors to detain surgery but should rather lead to the consideration how surgery can be made possible | 81% |
| 3 | High CRP values, supposedly from the spinal infection, are an argument for earlier surgical treatment to achieve infection control | 48%, no consensus |
| 4 | Significant pain upon axial loading, refractory to conservative therapy should be an indication for surgical stabilization | 81% |
| 5 | A septic or preseptic status is an indication for early surgical treatment (empyema evacuation, stabilization, debridement) | 81% |
| 6 | Patients with multiresistant pathogens and pathogens hard to control by antibiotic therapy (as decided in conjunction with microbiologists) should undergo surgical treatment | 88% |
| 7 | A new neurological deficit is an indication for surgical decompression and potentially stabilization | 100% |
| 8 | A radicular motor deficit of <4/5, any neurogenic bladder or bowel dysfunction, any cord symptoms including sensory symptoms are an indication for surgery. A pure radicular paresis of 4/5 and better allows conservative treatment but remains a relative indication for surgery | 100% |
| 9 | There are no clearly defined parameters of instability or deformity in spinal infections | 74%, no consensus |
| 10 | Any dynamic movement, relevant malalignment (for orientation: kyphosis >20o, scoliosis >10°), alignment changes in supine versus upright imaging, vertebral body height loss, deformity or pain upon axial loading are considered as signs of instability | 88% |
| 11 | Spinal instability of the infected segment is an indication for surgical stabilization | 92% |
| 12 | Any infection-associated dynamic movement, relevant malalignment (for orientation: kyphosis >20o, scoliosis >10°) or longitudinal changes in alignment in spinal infections are considered an indication for surgery | 85% |
| 13 | Infection associated deformities are an indication for surgical stabilization | 85% |
| 14 | Bony destructions influence the treatment decision towards surgery | 81% |
| 15 | An epidural empyema is almost always an indication for surgery, even in the absence of a neurological deficit | 63%, no consensus |
| 16 | An epidural empyema causing a neurological deficit is a clear indication for decompressive surgery | 100% |
| 17 | A sizable psoas abscess should either undergo surgical decompression or percutaneous image-guided drainage | 92% |
| 18 | The spinal surgery decision is not necessarily influenced by the size of a psoas abscess | 81% |
| 19 | An isolated discitis without relevant bony involvement, without epidural empyema is a typical candidate for conservative treatment | 96% |
Table 3.
Statements regarding ‘surgical technique’ and proportion of expert agreement leading to consensus or no consensus.
| Surgical technique | ||
|---|---|---|
| No | Statement | Level of consensus |
| 1 | In cases exclusively presenting with epidural empyema, decompression only is a surgical option | 92% |
| 2 | Surgical cases not presenting with an isolated epidural empyema should undergo stabilization in addition to an eventual decompression | 53%, no consensus |
| 3 | An anterior column support or reconstruction is necessary for cases of significant bony destruction or deformity, only | 81% |
| 4 | The majority of non-destructed, non-deformed infection cases will fuse without anterior interbody support | 88% |
| 5 | In surgical cases debridement of the infected disc space should be done for infection control | 81% |
| 6 | Titanium alloy is the preferred implant material | 81% |
| 7 | Although titanium implants are preferred, PEEK implants are acceptable in spinal infections, as well | 85% |
| 8 | Whenever possible minimally-invasive techniques should be preferred | 88% |
| 9 | An extensive irrigation of the whole surgical field by saline or an antiseptic solution should be performed | 93% |
Table 4.
Statements regarding ‘non-surgical treatment aspects’ and proportion of expert agreement leading to consensus or no consensus.
| Non-surgical treatment aspects | ||
|---|---|---|
| No | Statement | Level of consensus |
| 1 | Antibiotic treatment should be initiated after blood sampling for cultures and/or tissue biopsy in non-septic patients | 88% |
| 2 | Antibiotic treatment should be initiated after blood sampling for cultures in septic patients | 88% |
| 3 | Empiric antibiotic treatment depends on patient specifics, local aspects and should be defined for each institution in conjunction with microbiologists | 85% |
| 4 | For non-complicated infections (younger patient, sensitive pathogen, no immunosupression) 6 weeks of antibiotic therapy initiated by 2 weeks of iv therapy are usually sufficient | 88% |
| 5 | For complicated infections (older patients, resistant pathogen, immunosupression) 12 weeks of antibiotic therapy initiated by at least 2 weeks of iv therapy are usually necessary | 92% |
| 6 | Immobilization by bed rest is not part of the non-surgical treatment | 81% |
| 7 | Bracing is an optional aspect of the conservative approach, however, with unclear relevance | 81% |
Table 5.
Statements regarding ‘monitoring and follow-up of expert agreement leading to consensus or no consensus.
| Treatment monitoring and follow-up | ||
|---|---|---|
| No | Statement | Level of consensus |
| 1 | Most important parameters for monitoring of treatment success are CRP and pain | 100% |
| 2 | A decreasing CRP (ideally CRP reduction by 50% per week) and pain decrease are treatment response criteria | 100% |
| 3 | CRP values should be controlled twice weekly for the first two weeks and upon treatment response weekly afterwards | 96% |
| 4 | Conservatively treated patients should have regular standing x-rays to exclude secondary deformities | 85% |
| 5 | Instrumented patients should have postoperative imaging (at least x-ray) and follow-up imaging at 6 weeks, 3 months and 1 year | 88% |
| 6 | The use of follow-up MRI is optional and relevance for monitoring of treatment success is unclear as imaging changes can persist even after resolution of the infection | 84% |
| 7 | Antibiotics are discontinued after 6 or 12 weeks if CRP is normalized and pain resolved | 96% |
| 8 | After discontinuation of antibiotic therapy CRP and clinical symptoms should be monitored weekly for another 2–4 weeks | 92% |
3.5. Recommendations
3.5.1. Diagnostic workup
Imaging Modalities.
-
1.
MRI with Contrast: MRI with contrast is essential for the initial diagnosis of spinal infections and should be performed on the suspected spinal region. This was unanimously agreed upon as the gold standard for accurate diagnosis.
-
2.
Whole Spine MRI: While a whole spine MRI can be considered to exclude non-contiguous spinal lesions, especially in cases where multifocal disease is suspected, this recommendation did not achieve strong consensus (77%). Therefore, its use should be guided by resource availability and patient-specific factors.
-
3.
CT Scans: CT scans are recommended to assess bony destruction at the infected segments. Selective CT imaging of the affected levels is preferred over a whole spine CT, which is generally not necessary unless specific circumstances dictate otherwise.
-
4.
FDG-PET: FDG-PET is reserved for selected cases where MRI results are inconclusive or compromised by artifacts, particularly in patients with metal implants. It is not recommended as part of a routine workup.
Culture Sampling and Histological Analyses.
-
1.
Blood Cultures: Blood cultures should be taken upon suspected diagnosis of a spinal infection and prior to the initiation of antibiotic therapy. This step is critical for identifying the causative pathogen and guiding targeted antimicrobial treatment.
-
2.
Biopsy: If blood cultures are negative and further therapy is non-surgical or delayed, a CT-guided biopsy should be performed. For optimal results, the contrast-enhancing part of the disc should be targeted during the biopsy. Biopsy or surgical specimens must always be sent for both microbiological and histopathological evaluation to confirm the diagnosis and plan treatment.
Laboratory Parameters.
-
1.
CRP: CRP is highlighted as the most relevant laboratory parameter for both the initial diagnosis and monitoring of treatment response in spondylodiscitis.
-
2.
WBCC and ESR: While the white blood cell count (WBCC) can be used for diagnosis and follow-up, it is less sensitive than CRP and may not be elevated in all cases. The erythrocyte sedimentation rate (ESR), although sensitive, is increasingly seen as dispensable in many institutions and is not necessary for routine monitoring.
Additional Diagnostic Workup.
-
1.
Identifying the Primary Focus: A comprehensive diagnostic workup should be undertaken to identify any potential primary focus of infection. This includes a CT of the thorax/abdomen/pelvis, cardiac ultrasound, dental and skin examination, urinary laboratory tests, and evaluation of any implants.
-
2.
Full Workup Despite Obvious Source: Even when an obvious source of infection is identified, a full workup is recommended to exclude additional foci of infection.
-
3.
Standing X-rays: For cases where instability or deformity is suspected, standing x-rays should be used as a comparison to supine MRI/CT to accurately assess these conditions.
3.5.2. Management decisions
Surgical Considerations.
-
•
Advanced age, frailty, and comorbidities should not automatically exclude surgery; rather, these factors should guide how surgery can be made feasible.
-
•
Significant pain upon axial loading that is unresponsive to conservative treatment, along with uncontrolled antibiotic-resistant pathogens, warrants surgical stabilization.
-
•
In a septic patient early surgery should be considered, particularly for empyema evacuation, stabilization, and debridement.
-
•
Motor deficits with a motor score (MRC) below 4, cord symptoms/myelopathy including sensory symptoms and neurogenic bladder or bowel dysfunction, are clear indications for surgical decompression and stabilization.
-
•
High CRP values alone are not sufficient to dictate surgical timing but may contribute to the decision-making process.
-
•
Epidural empyema, especially when causing neurological deficits, typically requires surgical decompression.
-
•
A sizeable psoas abscess should be managed with either surgical decompression or percutaneous image-guided drainage, although the size of the abscess alone does not dictate the need for surgery.
-
•
Isolated discitis without significant bony involvement or epidural empyema is generally suitable for conservative treatment.
Surgical Technique.
-
•
Decompression in Epidural Empyema: In cases presenting exclusively with epidural empyema, surgical decompression alone is a viable option.
-
•
Anterior Column Support and Fusion: Anterior column support or reconstruction is recommended only in cases with significant bony destruction or deformity, while non-destructed, non-deformed infection cases are likely to achieve fusion without the need for anterior interbody support.
-
•
Debridement and Implant Materials: In surgical cases, debridement of the infected disc space is recommended for infection control. Titanium alloy is the preferred implant material, although PEEK implants are also acceptable in spinal infections.
-
•
Minimally Invasive Techniques and Irrigation: Whenever possible, minimally invasive techniques should be preferred. Additionally, extensive irrigation of the entire surgical field with saline or an antiseptic solution should be performed.
Non-Surgical Treatment Aspects.
-
•
Timing of Antibiotic Treatment: In non-septic patients antibiotic treatment should be initiated only after blood samples for cultures and/or tissue biopsy have been collected. In septic patients antibiotics should be started after obtaining blood samples for cultures.
-
•
Empiric Antibiotic Therapy: Empiric antibiotic treatment should be tailored to patient specifics and defined in conjunction with microbiologists or infectious disease specialists.
-
•
Duration of Antibiotic Therapy: For uncomplicated infections (younger patients, sensitive pathogen, no immunosuppression), a six-week antibiotic course, starting with two weeks of intravenous therapy, is usually sufficient. For complicated infections (older patients, resistant pathogen, immunosuppression), a twelve-week course, beginning with at least two weeks of intravenous therapy, is recommended.
-
•
Immobilization and Bracing: Immobilization by bed rest is not recommended as part of non-surgical treatment. Bracing may be considered as an optional aspect of the conservative approach, though its relevance remains unclear.
Treatment Monitoring and Follow-Up.
-
•
CRP Monitoring: CRP values should be monitord twice a week for the initial two weeks, and weekly thereafter once a response to after treatment is observed. A decrease in CRP (ideally by 50% per week) along with pain reduction are key indicators of favourable treatment response.
-
•
Follow-up Imaging: Conservatively treated patients should undergo regular standing X-rays to exclude secondary deformities. For instrumented patients, postoperative imaging (at least X-ray) is recommended, with follow-up imaging at 6 weeks, 3 months, and 1 year. The use of follow-up MRI is optional, as its relevance for monitoring treatment success is unclear due to potential persistence of imaging changes even after infection resolution.
-
•
Antibiotic Therapy Discontinuation: Antibiotics should be discontinued after 6 or 12 weeks if CRP levels have normalized and pain has resolved. After discontinuation, CRP levels and clinical symptoms should be monitored weekly for an additional 2–4 weeks.
4. Discussion
4.1. Interpretation of consensus statements
The management of SD has historically been marked by variability in both diagnostic and therapeutic approaches, largely due to a lack of high-level evidence. This variability has led to a divergence in clinical practices, with some advocating for early surgical intervention and others favoring conservative, long-term antibiotic therapy (Kramer et al., 2024a). Since the publication of the 2015 IDSA guidelines for the diagnosis and management of native vertebral osteomyelitis (NVO), these have served as a valuable resource for clinicians managing spinal infections (Berbari et al., 2015). However, the evolving diagnostic tools and therapeutic advancements necessitate a reassessment of certain recommendations. In particular, the IDSA guidelines’ recommendations for surgical therapy leave considerable room for interpretation, especially regarding factors such as progressive neurological deficits, spinal deformity, and instability, which remain loosely defined in both the literature and clinical practice.
Against this backdrop, the consensus statements presented here reflect the collective expertise of a panel of spine surgeons. The goal of this consensus statement is to provide a comprehensive framework for clinical decision-making based on current diagnostic modalities, microbiological sampling techniques, and standardized treatment regimens. The consensus seeks to address the gaps left by previous guidelines and to refine the criteria for both diagnostic and therapeutic measures in SD.
4.2. Diagnostic and therapeutic possibilities
The diagnostic landscape for SD has remained largely stable, with MRI continuing to be the gold standard due to its high sensitivity and ability to provide detailed soft tissue and bone visualization (Dunbar et al., 2010; Arbelaez et al., 2014). The consensus strongly supports MRI as the primary diagnostic tool, with unanimous agreement on its use for the initial diagnosis of spinal infections. CT scans are also recommended, particularly for assessing bony destruction in the infected segments, and are considered an important adjunct for surgical decision-making particularly in view of the need for additional instrumentation. Standing X-rays serve as a comparison to supine MRI/CT in cases where instability or deformity is suspected. While FDG-PET has emerged as an alternative imaging technology that may offer enhanced diagnostic precision, especially in cases where MRI is inconclusive or compromised by artifacts like metal implants, its routine use remains limited to selected challenging cases (Treglia et al., 2012; Heyde et al., 2023; Prodi et al., 2016). CT and X-ray, however, remain crucial for everyday assessments and should be integrated into the standard diagnostic workup.
4.3. Neurological deficits and surgical indications
One of the most challenging aspects of SD management is determining when neurological deficits warrant surgical intervention. While severe neurological symptoms, such as significant motor deficits or bladder and bowel dysfunction, are widely accepted as clear indications for decompressive surgery, the management of subtle or slowly evolving neurological deficits remains less defined (Herren et al., 2017; Guerado and Cervan, 2012). In cases of mild radicular weakness or sensory symptoms without profound motor impairment, clinicians often face uncertainty in deciding whether conservative management is appropriate. The consensus provides a structured approach to this ambiguity by endorsing surgical consideration for any new neurological deficit while accepting conservative treatment in cases of radicular paresis with a motor score (MRC) of ≥4.
4.4. Spinal deformity and instability: thresholds for surgery
The consensus also seeks to provide clarity on spinal deformity and instability as indications for surgery. We proposed specific thresholds, including kyphosis >20°, scoliosis >10°, and alignment changes in supine versus upright imaging. Vertebral body collapse >50% or translation >5 mm are also considered surgical indications (Herren et al., 2017). The value of these thresholds lies not only in providing immediate clinical guidance but also in their potential to inform future studies on long-term outcomes. Persisting back pain, spinal deformity, and instability significantly impact patients' quality of life. Early surgical intervention in these cases may prevent further deterioration, although the correlation between these thresholds and long-term outcomes, such as functional status and chronic pain, remains underexplored. Further prospective studies are required to validate these criteria and better understand their influence on patient recovery and quality of life (Stoop et al., 2021; Yagdiran et al., 2021).
4.5. Minimally invasive techniques: a growing role in SD management
Minimally invasive surgery (MIS) has gained traction in the management of SD, offering benefits such as reduced morbidity, quicker recovery, and fewer complications compared to traditional open approaches. Techniques like percutaneous drainage, endoscopic debridement, and minimally invasive stabilization have demonstrated their potential in minimizing the surgical burden on patients. (Scheyerer et al., 2021; Schwendner et al., 2023; Yu, 2020). However, the consensus highlights that traditional open surgery remains necessary in cases involving extensive bony destruction, significant deformity, or large abscess formation (Neuhoff et al., 2023). Thus, the choice of surgical approach must be individualized based on patient-specific factors and the extent of the infection.
The consensus strongly supports the use of MIS techniques whenever feasible (88% agreement) while emphasizing the importance of anterior column support or reconstruction in cases with significant bony destruction or deformity (81% agreement)(Timothy et al., 2019). As MIS becomes more widely adopted, future research must rigorously evaluate its efficacy and long-term outcomes in managing SD, particularly in complex cases.
4.6. Antibiotic treatment duration in SD
The optimal duration of antibiotic treatment in spinal infections remains a debated issue. The consensus supports a longer course of antibiotics—typically 12 weeks for complicated infections (older patients, resistant pathogens, or immunosuppressed individuals). However, many authors have advocated for shorter antibiotic regimens. Bernard et al., in a landmark prospective randomized study, demonstrated that 6 weeks of antibiotic treatment was non-inferior to 12 weeks in terms of clinical cure at one year (Bernard et al., 2015). Their findings suggest that in cases of uncomplicated pyogenic vertebral osteomyelitis, a shorter course of antibiotics may be sufficient, thus reducing treatment burden and associated risks such as antibiotic intolerance. While this evidence is compelling, it is important to note that the consensus reflects a more cautious approach for complicated infections. In these cases, the potential for incomplete resolution of the infection and risk of relapse warrants a longer duration of treatment.
4.7. Gaps in evidence and future research directions
Despite the comprehensive framework provided by the consensus, several critical areas of SD management remain underexplored. For example, the correlation between spinal deformity, instability, and long-term outcomes is poorly understood, and prospective studies are urgently needed to validate the proposed thresholds for surgical intervention. Concerning the distinct subset of patients presenting in a critically ill condition, we found in an international retrospective analysis, that delaying surgery significantly reduced mortality in this particular subgroup (Kramer et al., 2024b). However, further exploration of this and other subgroups of SD patients is required to refine treatment decision-making and to understand its impact on long term functional outcome.
Additionally, while advanced imaging modalities such as FDG-PET and MRI spectroscopy offer promising diagnostic capabilities, their application in routine practice remains limited. Further research is required to determine their impact on decision-making, especially in complex cases where traditional diagnostics may fall short. Similarly, in a recent retrospective MRI study on spinal deformity progression in conservatively treated patients we highlighted the substantial risk of deformity, particularly with >50% vertebral body destruction (Kramer et al., 2024c). Yet, more evidence is needed to determine the appropriate timing for surgical intervention in cases with high risk of deformity progression.
As the incidence of SD continues to rise, there is a growing demand for robust clinical evidence, particularly from randomized controlled trials, to inform decision-making for distinct patient subsets. This includes patients with evolving neurological deficits, geriatric patients, those with severe systemic infections, or significant comorbidities, where the balance between conservative and surgical treatment remains uncertain.
5. Conclusion
The EANS consensus provides up-to-date recommendations for the diagnosis and management of SD, integrating recent advances in the literature and offering a more nuanced exploration of surgical treatment options. By taking into account evolving imaging modalities and minimally invasive techniques, this consensus refines clinical decision-making in an area still characterized by variability. Despite these advancements, the field of optimal SD treatment remains under exploration, with limited long-term outcomes in many patients. This highlights the ongoing need for further research to optimize both diagnostic approaches and therapeutic strategies, ensuring that treatment can be tailored more effectively to the individual needs of this growing patient population.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Handling Editor: Prof F Kandziora
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bas.2024.104178.
Appendix A. Supplementary data
The following is the Supplementary data to this article.
References
- Appalanaidu N., Shafafy R., Gee C., et al. Predicting the need for surgical intervention in patients with spondylodiscitis: the Brighton Spondylodiscitis Score (BSDS) Eur. Spine J. 2019;28(4):751–761. doi: 10.1007/s00586-018-5775-x. [DOI] [PubMed] [Google Scholar]
- Arbelaez A., Restrepo F., Castillo M. Spinal infections: clinical and imaging features. Top. Magn. Reson. Imag. 2014;23(5):303–314. doi: 10.1097/RMR.0000000000000032. [DOI] [PubMed] [Google Scholar]
- Berbari E.F., Kanj S.S., Kowalski T.J., et al. Infectious diseases society of America (IDSA) clinical practice guidelines for the diagnosis and treatment of native vertebral osteomyelitis in adults. Clin. Infect. Dis. 2015;61(6):e26–e46. doi: 10.1093/cid/civ482. 2015. [DOI] [PubMed] [Google Scholar]
- Bernard L., Dinh A., Ghout I., et al. Antibiotic treatment for 6 weeks versus 12 weeks in patients with pyogenic vertebral osteomyelitis: an open-label, non-inferiority, randomised, controlled trial. Lancet. 2015;385(9971):875–882. doi: 10.1016/S0140-6736(14)61233-2. [DOI] [PubMed] [Google Scholar]
- Dunbar J.A., Sandoe J.A., Rao A.S., Crimmins D.W., Baig W., Rankine J.J. The MRI appearances of early vertebral osteomyelitis and discitis. Clin. Radiol. 2010;65(12):974–981. doi: 10.1016/j.crad.2010.03.015. [DOI] [PubMed] [Google Scholar]
- Fang A., Hu S.S., Endres N., Bradford D.S. Risk factors for infection after spinal surgery. Spine. 2005;30(12):1460–1465. doi: 10.1097/01.brs.0000166532.58227.4f. [DOI] [PubMed] [Google Scholar]
- Guerado E., Cervan A.M. Surgical treatment of spondylodiscitis. An update. Int. Orthop. 2012;36(2):413–420. doi: 10.1007/s00264-011-1441-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herren C., Jung N., Pishnamaz M., Breuninger M., Siewe J., Sobottke R. Spondylodiscitis: diagnosis and treatment options. Dtsch Arztebl Int. 2017;114(51–52):875–882. doi: 10.3238/arztebl.2017.0875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heuer A., Strahl A., Viezens L., Koepke L.G., Stangenberg M., Dreimann M. The hamburg spondylodiscitis assessment score (HSAS) for immediate evaluation of mortality risk on hospital admission. J. Clin. Med. 2022;11(3) doi: 10.3390/jcm11030660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heyde C.E., Spiegl U.J.A., Voelker A., von der Hoeh N., Henkelmann J. Imaging in the diagnosis of nonspecific pyogenic spondylodiskitis. J. Neurol. Surg. Cent. Eur. Neurosurg. 2023;84(1):69–76. doi: 10.1055/s-0040-1709168. [DOI] [PubMed] [Google Scholar]
- Kramer A., Thavarajasingam S.G., Neuhoff J., et al. Epidemiological trends of pyogenic spondylodiscitis in Germany: an EANS spine section study. Sci. Rep. 2023;13(1) doi: 10.1038/s41598-023-47341-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kramer A., S G.T., Neuhoff J., et al. Variation of practice in the treatment of pyogenic spondylodiscitis: a European association of neurosurgical Societies spine section study. J. Neurosurg. Spine. 2024;41(2):263–272. doi: 10.3171/2024.2.SPINE231202. [DOI] [PubMed] [Google Scholar]
- Kramer A., Thavarajasingam S.G., Neuhoff J., et al. An international multicentric evaluation of septic patients with primary spondylodiscitis – the 2SICK study of the EANS spine section. Brain and Spine. 2024;4 [Google Scholar]
- Kramer A., Neuhoff J., Thavarajasingam S.G., et al. Radiographic progression and spinal deformity in conservatively treated pyogenic spondylodiscitis: a monocentric, retrospective analysis of MRI data from 59 patients. Brain and Spine. 2024;4 [Google Scholar]
- Lemaignen A., Ghout I., Dinh A., et al. Characteristics of and risk factors for severe neurological deficit in patients with pyogenic vertebral osteomyelitis: a case-control study. Medicine (Baltim.) 2017;96(21) doi: 10.1097/MD.0000000000006387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuhoff J., Berkulian O., Kramer A., et al. Single- and Multilevel Corpectomy and Vertebral body replacement for treatment of spinal infections. A retrospective single-center study of 100 cases. Brain and Spine. 2023 doi: 10.1016/j.bas.2023.102721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuhoff J., Kramer A., Thavarajasingam S.G., et al. Comparing conservative and early surgical treatments for pyogenic spondylodiskitis: an international propensity score-matched retrospective outcome analysis. Neurosurgery. 2024 doi: 10.1227/neu.0000000000003223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pluemer J., Freyvert Y., Pratt N., et al. A novel scoring system concept for de novo spinal infection treatment, the Spinal Infection Treatment Evaluation Score (SITE Score): a proof-of-concept study. J. Neurosurg. Spine. 2023;38(3):396–404. doi: 10.3171/2022.11.SPINE22719. [DOI] [PubMed] [Google Scholar]
- Prodi E., Grassi R., Iacobellis F., Cianfoni A. Imaging in spondylodiskitis. Magn. Reson. Imag. Clin. N. Am. 2016;24(3):581–600. doi: 10.1016/j.mric.2016.04.005. [DOI] [PubMed] [Google Scholar]
- Ratcliffe J.F. Anatomic basis for the pathogenesis and radiologic features of vertebral osteomyelitis and its differentiation from childhood discitis. A microarteriographic investigation. Acta Radiol. Diagn. 1985;26(2):137–143. doi: 10.1177/028418518502600204. [DOI] [PubMed] [Google Scholar]
- Scheyerer M.J., Herren C., Kühne C., Neufang J., Pieroh P., von der Höh N.H. Surgical treatment strategies for pyogenic spondylodiscitis of the thoracolumbar spine. Z. für Orthop. Unfallchirurgie. 2021;160(6):621–628. doi: 10.1055/a-1527-7939. [DOI] [PubMed] [Google Scholar]
- Schomig F., Li Z., Perka L., et al. Georg schmorl prize of the German spine society (DWG) 2021: spinal Instability Spondylodiscitis Score (SISS)-a novel classification system for spinal instability in spontaneous spondylodiscitis. Eur. Spine J. 2022;31(5):1099–1106. doi: 10.1007/s00586-022-07157-3. [DOI] [PubMed] [Google Scholar]
- Schwendner M., Liang R., Butenschon V.M., Meyer B., Ille S., Krieg S.M. The one-stop-shop approach: navigating lumbar 360-degree instrumentation in a single position. Front Surg. 2023;10 doi: 10.3389/fsurg.2023.1152316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skaf G.S., Domloj N.T., Fehlings M.G., et al. Pyogenic spondylodiscitis: an overview. J Infect Public Health. 2010;3(1):5–16. doi: 10.1016/j.jiph.2010.01.001. [DOI] [PubMed] [Google Scholar]
- Stoop N., Zijlstra H., Ponds N.H.M., Wolterbeek N., Delawi D., Kempen D.H.R. Long-term quality of life outcome after spondylodiscitis treatment. Spine J. 2021;21(12):1985–1992. doi: 10.1016/j.spinee.2021.06.019. [DOI] [PubMed] [Google Scholar]
- Thavarajasingam S.G., Ponniah H.S., Philipps R., et al. Increasing incidence of spondylodiscitis in England: an analysis of the national health service (NHS) hospital episode statistics from 2012 to 2021. Brain and Spine. 2023 doi: 10.1016/j.bas.2023.101733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thavarajasingam S.G., Vemulapalli K.V., Vishnu K.S., et al. Conservative versus early surgical treatment in the management of pyogenic spondylodiscitis: a systematic review and meta-analysis. Sci. Rep. 2023;13(1) doi: 10.1038/s41598-023-41381-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timothy J., Pal D., Akhunbay-Fudge C., et al. Extreme lateral interbody fusion (XLIF) as a treatment for acute spondylodiscitis: leeds spinal unit experience. J. Clin. Neurosci. 2019;59:213–217. doi: 10.1016/j.jocn.2018.10.063. [DOI] [PubMed] [Google Scholar]
- Treglia G., Focacci C., Caldarella C., et al. The role of nuclear medicine in the diagnosis of spondylodiscitis. Eur. Rev. Med. Pharmacol. Sci. 2012;16(Suppl. 2):20–25. [PubMed] [Google Scholar]
- Yagdiran A., Otto-Lambertz C., Lingscheid K.M., et al. Quality of life and mortality after surgical treatment for vertebral osteomyelitis (VO): a prospective study. Eur. Spine J. 2021;30(6):1721–1731. doi: 10.1007/s00586-020-06519-z. [DOI] [PubMed] [Google Scholar]
- Yu C.H. Full-endoscopic debridement and drainage treating spine infection and psoas muscle abscess. J Spine Surg. 2020;6(2):415–423. doi: 10.21037/jss.2020.01.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.