Abstract
In this issue of Cancer Cell, Anadon et al. perform a multi-omics analysis of ovarian tumor infiltrating tissue-resident memory T (TRM) cells at the single-cell level that supports a differentiation model from CD103−TCF1+ re-circulating T-cell precursors driven by tumor antigen recognition. CD103+ TRM cells play a dominant anti-tumor role in ovarian cancer.
One of the most significant achievements in the cancer research field in the past decade is the clinical development of immunotherapies, particularly checkpoint blockade therapies and adoptive cell therapies (ACT). These new therapies have revolutionized the treatment of certain types of cancers, such as melanoma, lung cancers, B-cell malignancies, and others. Epithelial ovarian cancers (EOCs) are considered highly immunogenic because of their expression of immunogenic tumor antigens that induce spontaneous anti-tumor immune response in patients and the strong correlation between the number of tumor-infiltrating lymphocytes (TILs) and prognosis (Sato et al., 2005; Zhang et al., 2003). However, the efficacy of checkpoint blockade therapies in EOC patients has been limited. In addition, ACT using autologous chimeric antigen receptor or T-cell receptor (TCR) gene-engineered T cells are generally not as effective as those in blood cancers. Therefore, a comprehensive understanding of the immune response against EOCs, especially in the ovarian tumor microenvironment (TME), would help in the development of novel immunotherapeutic strategies for EOC patients.
A major challenge in immune cell-mediated destruction of solid tumors is the metabolic restriction imposed by limited availability of nutrients and oxygen in the solid tumor tissues. Recently, a unique subset of T cells, called tissue-resident memory T (TRM) cells, has been identified and characterized for different peripheral tissues such as lung, liver, and others (Schenkel and Masopust, 2014). TRM cells are thought to differentiate from effector T cells that are recruited to inflammatory tissues but remain resident within the tissues after the inflammation is resolved. Through unique genetic and epigenetic reprograming, TRM cells acquire metabolic adaptation and self-renewing ability in the local tissue, while retaining their effector functions, allowing them to reside within the peripheral tissues for months to years and to play critical roles in long-term immune surveillance (Pan et al., 2017). These unique characteristics of TRM cells appear to be ideal for effective anti-tumor T-cell response and tumor immune surveillance (Mami-Chouaib et al., 2018). However, TRM cells in the solid tumor tissues have not been sufficiently defined and characterized, especially in human tumor tissues.
In this issue of Cancer Cell, Anadon et al. reported important results from a comprehensive characterization of ovarian tumor-associated TRM cells (Anadon et al., 2022). First, they found that ovarian CD8+ TILs which are in direct contact with EOC cells preferentially express canonical markers (CD103 and CD69) for TRM cells. Ovarian tumor-associated CD103+CD69+ TRM cells express a set of genes that have been defined for TRM cells in other non-tumor tissues, compared to CD103− re-circulating T cells in the same tumors. Through integrated analyses of transcriptome, chromatin accessibility, and T cell receptor (TCR) repertoire at the single-cell level, they defined the trajectory of intra-tumoral differentiation of TILs after entering the TME (Figure 1). Their data supported a linear differentiation model of TRM cells from CD103−TCF1+ re-circulating T cell precursors in the following order: TCF7lowIL7R+CCR7+CD38− TRMstem, GZMB+PRF1+GNLY+ZNF683+ effector TRMeff, TIGITlowHAVCR2+/lowHLA−DRhighENTPD1+/lowTOX+ TRMprol with proliferative signature, and finally PD-1highHAVCR2highTIGIThighENTPD1highHLA−DR+TOX+ exhausted TRMexh. By analyzing shared TCR clonotypes among TIL subsets, they also concluded that only a small repertoire of re-circulating TILs differentiate into TRM cells. They also found that TCRs that were commonly expressed by these progressive differentiation subsets recognize autologous EOC cells, supporting that the intra-tumoral TRM differentiation is driven by tumor antigen recognition. Strikingly, their data suggest that CD103+ TRM cells, but not CD103− re-circulating TILs, play dominant anti-tumor role in EOCs. This is supported by multiple observations that (1) higher TRM-like cell/re-circulating TIL ratio is associated with longer overall survival, (2) more tumor infiltration of TRMstem cells, but not other TIL subsets, is associated with longer survival, and (3) TRM cells are highly enriched in a tumor epithelial region, whereas re-circulating TILs are found in both epithelial and stromal regions. Finally, using ovalbumin (OVA)-expressing murine ovarian tumor models and OVA-specific TCR transgenic T cells, the study concluded that pre-activation of T cells before entering the TME is required for intra-tumoral differentiation of TRM cells (Figure 1). Interestingly, antigen-specific TCR stimulation in the TME appears not required for differentiation of TRM cells, although it significantly enriches TRMstem cells within the total TRM-like cells.
Figure 1. Generation and differentiation of tumor-antigen specific tissue-resident CD8+ T (TRM) cells in the tumor microenvironment.
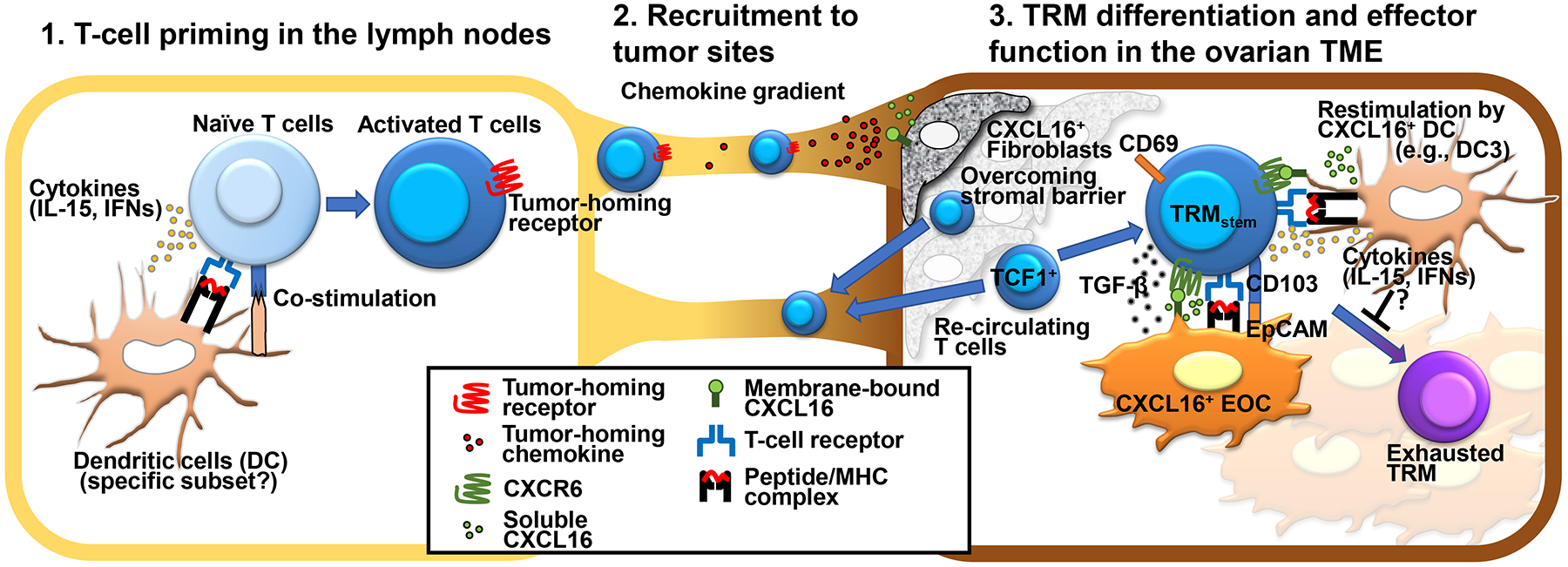
Tumor antigen-specific T cells that are primed by dendritic cells (DCs) in lymphoid tissues are recruited to ovarian tumor sites and differentiate to TCF1lowCD103+CD69+ tissue-resident memory (TRM)-like T stem (TRMstem) cells. TRMstem cells are retained in the tumor epithelial region and further differentiate to effector, proliferative and exhausted TRM subsets. CXCL16, which is expressed by multiple cell subsets in the ovarian tumor microenvironment, is considered to play a critical role in ovarian tumor tissue retention and longevity of CXCR6+ TRM cells.
These observations in EOC patient-derived specimens and a murine EOC model provide new insights into our understanding of anti-tumor T cell responses in the ovarian TME, especially regarding dominant anti-tumor roles by TRM cells and strongly warrant development of TRM cell-based immunotherapies for EOC patients. For development of TRM cell-based immunotherapies, there are a few outstanding questions to be answered. First, it is important to elucidate mechanisms by which re-circulating (CD103−) T cells are specifically recruited and infiltrate into epithelial tumor regions to differentiate into TRM cells (Figure 1). Such mechanisms can be utilized to promote T cell infiltration and TRM differentiation to improve efficacy of immunotherapies. In this regard, we recently reported that the CXCR6-CXCL16 axis plays a critical role in the generation and retention of tumor antigen-specific TRM cells in a murine ovarian cancer model (Muthuswamy et al., 2021). A recent study also showed that CXCL16+ dendritic cells (CCR7+ DC3) play a critical role in supporting survival of CXCR6+ T cells by providing IL-15 in the TME (Di Pilato et al., 2021). Importantly, the study in the current issue (Anadon et al., 2022) identified CXCR6 as one of genes that are overexpressed in TRM compared to CD103− TILs. Another study also identified that EOC cells highly express CXCL16 in EOC patients (Zsiros et al., 2015). Strategies to overexpress CXCR6 on anti-tumor effector T cells and/or CXCL16 in the TME can be incorporated in immunotherapies to promote recruitment and retention of TRM cells. Next, it is known that expression of tumor-specific neoantigens and tumor-associated shared antigens is highly variable between patients, and it is still to be determined how interpatient heterogeneity in tumor immunogenicity affects generation, anti-tumor function, and differentiation fate of TRM cells. Using OVA-expressing murine EOC models, this study demonstrated that antigen-specific priming with high affinity TCR ligand (OVA peptide) before entering the TME is preferred for efficient generation of TRM cells, especially TRMstem cells that show prognostic impact. This observation may suggest that the generation and anti-tumor effect of ovarian TRM cells are dependent on the expression of tumor antigens and affinity of TCR-pMHC interactions. Elucidating mechanisms by which a diverse repertoire of tumor antigens shape TRM cells in the TME could help with efforts to discover effective targets for immunotherapies of EOCs. Finally, previous studies on normal tissue-associated TRM cells have identified several key molecules such as TGF-β and IL-15 that are required for generation of TRM cells (Figure 1). It is important to elucidate molecular and environmental conditions that promote generation of ovarian tumor-associated TRM cells.
In conclusion, this important study from the group of Conejo-Garcia highlights the indispensable roles of TRM cells in anti-tumor T-cell response. Although this study exclusively focused on EOCs, it is highly likely that the conclusions of this study are applicable to other solid tumors because of widespread prognostic roles of TILs in many different tumors. Future studies should focus on promoting TRMstem cell generation, retention and function from re-circulating TILs, and developing TRM cell-based immunotherapies.
Declaration of interests:
K.O. is a co-founder of Tactiva Therapeutics and receives research support from AstraZeneca and Tesaro. T.T. and J.M. declare no competing interests.
References
- Anaton CM, Yu X, Hanggi K, Biswas S, Chaurio R, Martin A, Payne KK, Mandal G, Innamarato PP, et al. (2022) Ovarian cancer immunogenicity is governed by a narrow subset of progenitor tissue-resident memory T cells. Cancer Cell [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Pilato M, Kfuri-Rubens R, Pruessmann JN, Ozga AJ, Messemaker M, Cadilha BL, Sivakumar R, Cianciaruso C, Warner RD, Marangoni F, et al. (2021). CXCR6 positions cytotoxic T cells to receive critical survival signals in the tumor microenvironment. Cell 184, 4512–4530 e4522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mami-Chouaib F, Blanc C, Corgnac S, Hans S, Malenica I, Granier C, Tihy I, and Tartour E (2018). Resident memory T cells, critical components in tumor immunology. J Immunother Cancer 6, 87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muthuswamy R, McGray AR, Battaglia S, He W, Miliotto A, Eppolito C, Matsuzaki J, Takemasa T, Koya R, Chodon T, et al. (2021). CXCR6 by increasing retention of memory CD8(+) T cells in the ovarian tumor microenvironment promotes immunosurveillance and control of ovarian cancer. J Immunother Cancer 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan Y, Tian T, Park CO, Lofftus SY, Mei S, Liu X, Luo C, O’Malley JT, Gehad A, Teague JE, et al. (2017). Survival of tissue-resident memory T cells requires exogenous lipid uptake and metabolism. Nature 543, 252–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato E, Olson SH, Ahn J, Bundy B, Nishikawa H, Qian F, Jungbluth AA, Frosina D, Gnjatic S, Ambrosone C, et al. (2005). Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc Natl Acad Sci U S A 102, 18538–18543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schenkel JM, and Masopust D (2014). Tissue-resident memory T cells. Immunity 41, 886–897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Conejo-Garcia JR, Katsaros D, Gimotty PA, Massobrio M, Regnani G, Makrigiannakis A, Gray H, Schlienger K, Liebman MN, et al. (2003). Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med 348, 203–213. [DOI] [PubMed] [Google Scholar]
- Zsiros E, Duttagupta P, Dangaj D, Li H, Frank R, Garrabrant T, Hagemann IS, Levine BL, June CH, Zhang L, et al. (2015). The Ovarian Cancer Chemokine Landscape Is Conducive to Homing of Vaccine-Primed and CD3/CD28-Costimulated T Cells Prepared for Adoptive Therapy. Clin Cancer Res 21, 2840–2850. [DOI] [PMC free article] [PubMed] [Google Scholar]


