Abstract
Background/Objective
Evidence on the efficacy and safety of minimally invasive treatment for insulinoma has increased over the past decade to the point of becoming a recommendation in clinical practice guidelines for the management of this type of neuroendocrine tumor.
Case Report
We describe the case of an elderly male patient with multiple comorbidities and recurrent isolated insulinoma of 3.7 × 3.5 cm involving the uncinate process of the pancreas and contacting the splenomesenteric confluent many years after first resection, in whom, after refusing surgical management, was performed as successful arterial embolization of the pancreatic tumor.
Discussion
When addressing this pathology, it is common to encounter patients who are not candidates for surgical management, either due to the presence of comorbidities, the location of the tumor in relation to vascular structures, or refusal of the intervention. Therefore, it is important to be aware of the different therapeutic options in localized and metastatic disease.
Conclusion
Minimally invasive procedures are positioned as an effective alternative for the treatment of the hormonal overproduction in patients with insulinoma.
Key words: insulinoma, angioembolization, neuroendocrine tumor
Highlights
-
•
Insulinoma is the most frequent functioning pancreatic neuroendocrine tumor
-
•
Use of nonsurgical ablative therapies may be indicated in patients who are not candidates for surgery
-
•
Minimally invasive procedures are fast, safe, and effective alternative for the treatment of insulinoma
Clinical Relevance
The first-line therapy for localized insulinoma will always be surgery, but sometimes it is not feasible and/or represents a high risk of perioperative morbidity. In these scenarios, minimally invasive procedures are positioned as a fast, safe, and effective alternative for the treatment of the hormonal overproduction.
Introduction
Insulinoma is the most common functioning pancreatic neuroendocrine tumor, although, in general, it is considered a rare entity (1 to 4 cases per million people/year).1 In 90% of cases, it is a benign tumor, and morbidity is related to the overproduction of insulin, leading to a slowly progressive condition with devastating consequences and affecting the quality of life if not treated in time.2,3 Therapy for locoregional disease ranges from surgery to endoscopic and interventional radiology management. Tumor enucleation surgery is the treatment of choice for this disease, with cure rates between 75% and 98% of cases.3
Despite the above, some patients are not candidates for surgical management of localized disease due to a high perioperative risk associated with comorbidities, refusal of the intervention by the patient, or tumors that are in close contact with abdominal vascular structures,2,4 making surgical approach difficult. Additionally, postoperative complications (hemorrhage, fistulas, delayed gastric emptying) can occur in 14% to 58% of patients, and for all these reasons, alternative treatments for insulinoma have been developed.2
The management of symptoms related to hormonal overproduction in neuroendocrine neoplasia is based on somatostatin analogs. Diazoxide is considered as the first line of medical management in patients with insulinoma at a dose of 3-8 mg/kg/d, divided into 2-3 doses, and in cases with refractory symptoms, everolimus, sunitinib, radionuclide therapy, and/or cytotoxic chemotherapy can be used, in monotherapy or in combination, without forgetting the high frequency of adverse effects that these drugs entail.3 In patients with insulinoma, only 50% of cases achieve symptomatic control with somatostatin analogs since not all tumors express somatostatin receptor subtype 2 and they also inhibit the release of counter-regulatory hormones aggravating hypoglycemia.5
The European Neuroendocrine Tumor Society suggests the use of nonsurgical ablative therapies in patients with localized insulinoma who are not candidates for surgery.3 The approach can be performed endoscopically or percutaneously, and the limited literature on these minimally invasive procedures provides some data on efficacy and safety in patients with localized and metastatic liver disease.2 The procedures described to date are as follows: percutaneous radiofrequency ablation, high-intensity ultrasound ablation, percutaneous microwave ablation, ethanol ablation, and transcatheter arterial embolization.6
The following is the case of an elderly, obese patient with multiple comorbidities and a recurrent insulinoma many years after the resection of the primary tumor by pancreatoduodenectomy, who was proposed for arterial embolization of the pancreatic tumor after refusing a surgical reintervention.
Case Report
An older male adult patient of 80 years presents to the clinic with a history of insulinoma diagnosed in 2009 and apparently surgically managed by partial pancreatoduodenectomy. Pathology result suggested a mixed neuroendocrine tumor grade 2 of World Health Organization classification, with a tumor size of 5 × 4.5 × 2 cm and Ki-67 5%. Microscopic description showed moderately differentiated malignant epithelial tumor and immunohistochemistry was negative for chromogranin, positive for keratin, and strongly and diffusely positive for synaptophysin. The differential diagnosis was between solid pseudonodular neoplasia of the pancreas vs neuroendocrine tumor, favoring the latter.
He also has other comorbidities like advanced chronic kidney disease, deep vein thrombosis of the right lower limb, prostate adenocarcinoma T4N1M1B in management with apalutamide 240 mg monthly and leuprolide acetate 45 mg biannual instated by oncology. Unfortunately, after the resection of primary neuroendocrine tumor, the patient did not follow up on this condition.
In 2023 symptomatic hypoglycemia reappeared, predominantly in the early morning hours associated with blood glucose levels below 50 mg/dl on several occasions, fulfilling clinical characteristics for Whipple's triad. In a complementary study, a contrast-enhanced abdominal computed tomography scan identified a neoplastic-looking lesion in the uncinate process of the pancreas measuring 4.7 × 3 × 4 cm, a finding consistent with Octreoscan performed later, strongly suggesting the presence of a neuroendocrine tumor.
The clinical picture worsened progressively 1 month before admission to the emergency department, linked to severe hypoglycemia. A fasting test was performed with results that confirmed endogenous hyperinsulinism (Table 1). The abdominal magnetic resonance imaging with contrast showed a 3.7 × 3.5 cm mass of hypervascularized neoplastic aspect involving the uncinate process of the pancreas, contacting the splenomesenteric confluent at 180° and infiltrating the left gastric artery, without infiltration of other arterial or venous structures (Fig. 1). The tumor size found in images is atypical in cases of benign insulinoma.
Table 1.
Fasting Test Results
| Laboratory parameter | Results | Reference |
|---|---|---|
| Blood glucose | 33 mg/dl | NA |
| Insulin | 21,3 uUI/mL | 2.6-24.9 uUI/mL |
| C peptide | 2.12 nmol/L | 0.2-2.35 nmol/L |
Figure 1.
Abdominal magnetic resonance imaging (MRI). Solid lesion with a neoplastic appearance measuring 3.7 × 3.5 cm in the axial plane, with heterogeneous signal intensity, predominantly hyperintense on T2, slightly hyperintense on T1, located in the uncinate process of the pancreas. It causes diffusion restriction and presents early arterial enhancement after the administration of contrast medium.
Surgical management with high risk of complications was proposed initially, but the patient rejected this therapeutic option. During the multidisciplinary assessment, treatment was started with diazoxide at a dose of 5 mg/kg/d distributed in 3 doses, but the patient developed anorexia, dizziness, edema, and arterial hypotension as adverse effects, leading to premature discontinuation and requiring dextrose infusion.
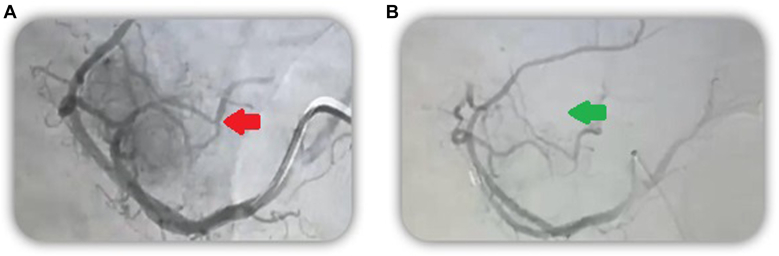
Arterial embolization of the pancreatic tumor by catheterization and angiography of the celiac trunk was proposed. On identifying the gastroduodenal artery, the inferior pancreaticoduodenal artery and the common hepatic artery, multiple abnormal arteries were observed directed towards the "tumor blush" in the topography of the pancreatic head (Fig. 2). Embolization of these branches was performed with 350–500-micron polyvinyl alcohol microparticles and 2 coils. At the end of the procedure, control angiography showed a satisfactory angiographic result (Fig. 2). During the immediate postoperative period there were no complications derived from the procedure, without dextrose infusion. Hypoglycemia episodes decreased in frequency and severity until they completely resolved 8 weeks after the procedure. So far, no complications derived from this intervention have been reported by the patient.
Figure 2.
Abdominal angiography. A. Initial arteriography and selective arterial catheterization, where tumor blush is observed. (Pre-embolization, red arrow) B. Postembolization arteriography, visualization of arteries with absence of tumor blush (green arrow).
Discussion
The recurrence rate of insulinoma after open surgical resection can be between 3% and 7% but this depends on several factors such as the experience of the surgical team, complete enucleation of the lesion, tumor grade and whether it is a case associated with a hereditary cancer syndrome.6, 7, 8, 9 Additionally, insulinomas larger than 2.5 cm suggest a malignant behavior of the tumor,4,7 so our patient’s case is very atypical because there was no evidence of metastatic or locoregional lymph node involvement, even during the initial evaluation in 2009.
The first arterial embolization of an insulinoma was published by Moore et al in 1982 and in this case report, the authors state that the success of the procedure depends on the degree of tumor necrosis achieved and, in the long term, on the capacity for recanalization and growth of possible tumor remnants.10 Given the high vascularization of insulinomas, lesion ablation has been possible through techniques other than arterial embolization, such as radiofrequency, cryoablation, and ethanol.11,12
Embolic particles coated with different chemotherapeutic and radiotherapeutic agents allow obstruction of blood flow to the tumor with consequent ischemia and tissue necrosis. The choice of the size of these particles is empirical since there is no concrete information about the diameter of intrapancreatic arteries.13 Some examples of these agents are polyvinyl alcohol particles, gelatin particles, calibrated microspheres, coils, liquid agents or gels, among others.14
When performing the review of the literature, about 44 cases reported in the literature of patients with insulinoma treated with different ablative techniques were recognized, but only 9 cases of transcatheter arterial embolization reported up to 2019.2 The present case report would count as the 10th in the world that used arterial embolization for the treatment of insulinoma and the first case to be performed in Colombia in a tertiary care institution (Table 2). The success rate of this procedure is close to 60%, defined as complete resolution of symptoms related to insulin hypersecretion with evidence of restored euglycemia (≥70 mg/dL),16, 17, 18 but more studies are needed to evaluate long-term outcomes.2,11
Table 2.
Summary of Arterial Embolization Procedures for the Management of Insulinoma (Until December 2023)
| Reference | No. of cases | Age (y) | Gender | Tumor size (mm) | Tumor location | Reason for the choice of treatment | No. of treatments | Postprocedure complications | Follow-up (mo) | Recurrence | Time of recurrence (mo) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Moore 8 | 1 | 38 | F | 20 | H | SF | 1 | Low back pain, nausea, Increase in liver enzymes | 11 | No | - |
| Uflacker 13 | 2 | 56/49 | M/M | 18/20 | H/B | SR/SE | 2/1 | Abdominal pain, hyperamylasaemia/none | 18/40 | No | - |
| Rott 14 | 1 | 84 | F | 14 | H | SR | 1 | Mild pancreatitis and pain, transitory mild diabetes | 12 | No | - |
| Peppa 11 | 1 | 30 | M | 30 | PP | DR | 1 | Abdominal pain | 12 | Yes | 12 |
| Balawejder 15 | 1 | 48 | F | 22 | UP | SR | 1 | None | N/A | Yes | 5 |
| Mele 2 | 1 | 72 | F | 17 | H | C | 2 | Mild pancreatitis and pain | 108 | Yes | 48 |
| Donohoe 9 | 2 | 83/53 | M/M | 32/NR | UP/UP | C/SR | 1/1 | None/none | 1/1 | No/No | - |
| Our case | 1 | 80 | M | 37 | H, UP | SR, C | 1 | None | 5 | No | - |
| Total | 10 | 59,3 ± 19,3 | 6 M/4 F | 23,3 ± 7,7 | H = 5; B = 1; UP = 4; PP = 1 | SR = 5; C = 3; SF = 1; SE = 1; DR = 1 | 1 in 8 cases | None in 5 cases | 23,1 ± 33,9 | 3 (30%) | 22 ± 23 |
Abbreviations: B = body; C = comorbidities; DR = disease recurrence; H = head; NR = no report; PP = peripancreatic; SE = surgical emergency; SF = surgical failure; SR = surgery refusal; UP = uncinate process.
Cumulative data are indicated as mean ± SD, and as absolute values with percentages in parentheses.
Source: Own elaboration.
In the review of arterial embolization procedures for the management of insulinoma (up to December 2023), no case similar to that of our patient was found in terms of age and tumor size (Table 2). Tumor size seems to influence the recurrence rate of the disease; in our case, there is no evidence of recurrence until date, however, the patient is at high risk of recurrence due to tumor size and the degree of tumor differentiation.8,9
Potential adverse effects are few and consist mainly of postoperative pancreatitis, cysts, and pancreatic pseudocyst, with a frequency between 3% and 33% of cases treated by minimally invasive procedures.11,13 Mortality rates associated with these interventions are low and do not reach 5% of cases2 and a disadvantage when opting for this treatment strategy for patients with insulinoma is the limitation in obtaining a sample for histological study, with the subsequent incomplete staging of the disease.6
Conclusions
It is clear that the first-line therapy for cases of localized insulinoma will always be surgery, but sometimes it is not feasible and/or represents a high risk of perioperative morbidity. In this scenario, minimally invasive procedures position themselves as a quick, safe, and effective alternative for the treatment of hormonal overproduction causing the characteristic clinical picture in patients.
Disclosure
The authors have no conflicts of interest to disclose.
Acknowledgment
This project was funded by Hospital Universitario Fundación Valle del Lili.
Footnotes
Written informed consent was obtained from the patient's son for publication of this case report and accompanying images.
References
- 1.Effraimidis G., Knigge U., Rossing M., Oturai P., Rasmussen Å.K., Feldt-Rasmussen U. Multiple endocrine neoplasia type 1 (MEN-1) and neuroendocrine neoplasms (NENs) Semin Cancer Biol. 2022;79:141–162. doi: 10.1016/j.semcancer.2021.04.011. [DOI] [PubMed] [Google Scholar]
- 2.Mele C., Brunani A., Damascelli B., Tichà V., Castello L., Aimaretti G., et al. Non-surgical ablative therapies for inoperable benign insulinoma. J Endocrinol Invest. 2018;41:153–162. doi: 10.1007/s40618-017-0738-3. [DOI] [PubMed] [Google Scholar]
- 3.Hofland J., Falconi M., Christ E., Castaño J.P., Faggiano A., Lamarca A., et al. European neuroendocrine tumor society 2023 guidance paper for functioning pancreatic neuroendocrine tumour syndromes. J Neuroendocrinol. 2023;35(8) doi: 10.1111/jne.13318. [DOI] [PubMed] [Google Scholar]
- 4.Sultana Q., Kar J., Verma A., Sanghvi S., Kaka N., Patel N., et al. A comprehensive review on neuroendocrine neoplasms: Presentation, pathophysiology and management. J Clin Med. 2023;12 doi: 10.3390/jcm12155138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Carmona-Bayonas A., Jiménez-Fonseca P., Custodio A., Grande E., Capdevila J., López C., et al. Optimizing somatostatin analog use in well or moderately differentiated gastroenteropancreatic neuroendocrine tumors. Curr Oncol Rep [Internet] 2017;19(11):72. doi: 10.1007/s11912-017-0633-2. [DOI] [PubMed] [Google Scholar]
- 6.Jayawickreme K.P., Muthukuda D.T., Kariyawasam C., Piyarisi L., Abeywickrama B.A. A rare case of insulinoma presenting with deep vein thrombosis, successfully treated with minimally invasive procedures including microwave ablation. Endocrinol Diabetes Metab Case Rep. 2022;2022(1) doi: 10.1530/EDM-21-0127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Castillón J.C., Gordoa T.A., Bayonas A.C., Carretero A.C., García-Carbonero R., Pulido E.G., et al. SEOM-GETNE clinical guidelines for the diagnosis and treatment of gastroenteropancreatic and bronchial neuroendocrine neoplasms (NENs) (2022) Clin Transl Oncol. 2023;25(9):2692–2706. doi: 10.1007/s12094-023-03205-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kalra P., Anakal M.G., Dharmalingam M., Indushekhar S., Rao V., Kumar K.P. Insulinoma case series: experience of a tertiary care center. Indian J. Endocrinol. Metab. 2014;18:858–862. doi: 10.4103/2230-8210.141385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Crippa S., Zerbi A., Boninsegna L., Capitanio V., Partelli S., Balzano G., et al. Surgical management of insulinomas: short- and long-term outcomes after enucleations and pancreatic resections. Arch Surg. 2012;147(3):261–266. doi: 10.1001/archsurg.2011.1843. [DOI] [PubMed] [Google Scholar]
- 10.Moore T.J., Peterson L.M., Harrington D.P., Smith R.J. Successful arterial embolization of an insulinoma [Internet] 1982. http://jama.jamanetwork.com/ [PubMed]
- 11.O’Donohoe R.L., Patel S., Bryant T.J.C., Stedman B., Hamady Z., Takhar A.S., et al. Transarterial embolization for mitigation of severe hypoglycemia in 2 patients with primary pancreatic insulinoma. J Vasc Intervent Radiol. 2019;30:476–479. doi: 10.1016/j.jvir.2018.10.032. [DOI] [PubMed] [Google Scholar]
- 12.Ghorashi N.S., Ghodasara N., Liddell R.P. Percutaneous cryoablation of an insulinoma in a patient with symptomatic hypoglycemia refractory to bland microsphere embolization. Cardiovasc Intervent Radiol. 2022;45:530–532. doi: 10.1007/s00270-021-03046-2. [DOI] [PubMed] [Google Scholar]
- 13.Peppa M., Brountzos E., Economopoulos N., Boutati E., Pikounis V., Patapis P., et al. Embolization as an alternative treatment of insulinoma in a patient with multiple endocrine neoplasia type 1 syndrome. Cardiovasc Intervent Radiol. 2009;32(4):807–811. doi: 10.1007/s00270-008-9499-x. [DOI] [PubMed] [Google Scholar]
- 14.Wang N., Jiang X., Wang J. Progress in Biomedical Engineering Emerging embolic agents in endovascular embolization: an overview. Prog Biomed Eng (Bristol) 2020;2(1) doi: 10.1088/2516-1091/ab6c7d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ufacker R. Arterial embolization as defnitive treatment for benign insulinoma of the pancreas. J Vasc Interv Radiol. 1992;3:639–644. doi: 10.1016/S1051-0443(92)72912-1. [DOI] [PubMed] [Google Scholar]
- 16.Rott G., Biggemann M., Pfohl M. Embolization of an insulinoma of the pancreas with trisacryl gelatin microspheres as defnitive treatment. Cardiovasc Interv Radiol. 2008;31:659–662. doi: 10.1007/s00270-007-9185-4. [DOI] [PubMed] [Google Scholar]
- 17.Balawejder M., Skorkowska-Telichowska K., Kuniej T., Tuchendler R. Insulinoma located in the head of the pancreas: is there an alternative to surgery? Int J Case Rep Images. 2014;5:285–294. doi: 10.5348/ijcri-201459-CR-10370. [DOI] [Google Scholar]
- 18.Crino S.F., Napoleón B., Facciorusso A., Lakhtakia S., Borbath I., Caillol F., et al. Endoscopic ultrasound-guided radiofrequency ablation versus surgical resection for treatment of pancreatic insulinoma. Clin Gastroenterol Hepatol. 2023;21(11):2834–2843.e2. doi: 10.1016/j.cgh.2023.02.022. [DOI] [PubMed] [Google Scholar]