Abstract
Purpose
To evaluate the impact of Endometrial Microbiome Metagenomic Analysis and Analysis of Infectious Chronic Endometritis (EMMA & ALICE) on pregnancy outcomes following recommended treatments in women with recurrent implantation failure (RIF) or recurrent pregnancy loss (RPL).
Methods
This prospective, multicenter cohort study included 527 women under 42 years old with RIF or RPL across 14 IVF centers in Japan. Endometrial samples were analyzed using EMMA & ALICE, and patients received antibiotics, probiotics, or no treatment based on test results. Pregnancy outcomes were assessed using Kaplan–Meier survival analysis and multivariate generalized linear models.
Results
Amongst participants, 43.4% had a normal Lactobacillus‐dominated microbiota, 20.9% had dysbiosis, and 35.7% had mild dysbiosis or ultralow biomass. Kaplan–Meier analysis revealed significantly higher ongoing pregnancy rates in the dysbiosis group treated with antibiotics and probiotics compared to other groups (p = 0.031). Post‐treatment, ongoing pregnancy rates in the dysbiosis and mild dysbiosis groups were comparable to the normal group.
Conclusions
EMMA & ALICE‐guided antimicrobial and probiotic treatments improved pregnancy outcomes, enabling the dysbiosis group to achieve pregnancy earlier than the normal group. Addressing uterine dysbiosis may reduce the time to pregnancy in patients with RIF and RPL.
Trial registration
University Hospital Medical Information Network (UMIN), UMIN000036917.
Keywords: dysbiosis, endometrial microbiome, next‐generation sequencing (NGS), recurrent implantation failure (RIF), recurrent pregnancy loss (RPL)
This study evaluated the effect of endometrial microbiome analysis (EMMA & ALICE) on pregnancy outcomes in women with RIF or RPL. The dysbiosis group treated with antibiotics and probiotics achieved significantly faster pregnancies compared to the normal group, suggesting that addressing uterine dysbiosis may shorten the time to pregnancy.

1. INTRODUCTION
The Human Microbiome Project has unveiled that less than 9% of the total human microbiome resides in the female reproductive tract. 1 , 2 Historically, the uterine cavity has been considered sterile. However, research has shown that the endometrial microbiota, although comprising fewer bacteria (102–104) compared to the vaginal microbiota, 3 can significantly impact implantation and pregnancy continuation even in low numbers of bacteria. 4 , 5 The endometrial microbiota potentially modulates essential endometrial functions, with reports indicating that a healthy uterine microbiota may positively affect the immune cell subsets needed for implantation and protects against uterine infections by defending its niche and competing with pathogens. 6
Advances in next‐generation sequencing (NGS) technologies have facilitated the analysis of the endometrial microbiome through 16S ribosomal RNA (rRNA) sequencing, allowing for the detection of both culturable and non‐culturable bacterial genera in the endometrial environment. 7 , 8 Lactobacillus species, renowned for their beneficial effects in the vaginal microbiota, 9 also play a crucial role in the endometrial environment by promoting conditions conducive to embryo implantation and pregnancy. 5 , 10 , 11 A Lactobacillus‐dominated microbiota (LDM) can inhibit pathogenic bacterial colonization in the uterine cavity, ensuring an optimal environment for implantation and pregnancy. 11 , 12
Despite this understanding, the impact of therapeutic interventions, such as antimicrobials to treat dysbiosis and shift the endometrial environment towards an LDM, on pregnancy outcomes remains largely unknown. Our previous research suggested that the Endometrial Microbiome Metagenomic Analysis & Analysis of Infectious Chronic Endometritis (EMMA & ALICE) should be considered for patients with recurrent implantation failure (RIF), showing improved pregnancy rates following post‐test embryo transfer (ET) when the appropriate therapeutic interventions based on EMMA & ALICE were administered. 13 However, due to the small sample size, our previous analysis did not differentiate clinical outcomes between the normal group, which required no post‐test therapeutic intervention, and the abnormal group, which received antimicrobial treatment followed by probiotics administration. 13
Therefore, this study aims to increase the sample size of patients undergoing EMMA & ALICE in a large multicenter setting and to conduct a subgroup analysis. Additionally, the observation period to account for the effect of EMMA & ALICE has been extended to include a long‐term evaluation of cumulative pregnancy rates.
2. METHODS
2.1. Study design, participants, and criteria
This prospective multicenter cohort study was conducted between July 2019 and August 2021 at 14 facilities affiliated with the Japanese Society for Assisted Reproductive Technology (JISART), targeting patients scheduled for ET as part of assisted reproductive technology (ART) protocols.
The inclusion criteria for the study were as follows: patients who were ART‐treated infertile patients with more than three ETs with no implantation or more than two miscarriages, and who had tested negative for HIV, HBV, HCV, RPR, Chlamydia trachomatis, and gonorrhea. The following criteria were used to exclude patients from the study: patients with untreated intrauterine lesions, including endometrial polyps, submucosal uterine myoma, Asherman's syndrome, or cesarean section scarring syndrome. Additionally, patients with untreated hydrosalpinx, uncontrolled metabolic or medical complications, chromosomal abnormalities, or allergies to multiple antibiotics were excluded from the study. Furthermore, participants who had used antibiotics within the 3 months prior to sampling or who took non‐recommended antibiotics between sampling and ET were also excluded, along with those having conditions that could affect study compliance.
2.2. Procedures
2.2.1. Sample collection
Endometrial biopsies (EBs) were performed during the luteal phase. For patients with a regular menstrual cycle, EBs were collected between days 15 and 25. For those on hormone replacement therapy cycles, EBs were collected between days 5 and 7 post‐progestin administration. To mitigate bacterial contamination, the vagina was thoroughly washed with saline solution, and cervical discharge was absorbed with a dry cotton pad. EBs were collected using a sterile double‐lumen catheter (Fuji Medical, Tokyo, Japan) and involved circumferential vacuum aspiration of the endometrial tissue. Samples were then decanted into sterile tubes (Cryotube, Biosigma S.p.A., Italy) containing RNA later solution (Sigma–Aldrich Co. LLC, MI), vigorously shaken for approximately 4 S, stored at 4°C for 4–72 h, and subsequently shipped to Igenomix headquarters (Valencia, Spain) at room temperature.
2.2.2. Analysis of the endometrial microbiota
The endometrial microbiota was analyzed using microbial 16S rRNA gene sequencing, as previously described by Moreno et al. 8 The protocol followed for the EMMA & ALICE tests was the one devised by Igenomix® (https://www.igenomix.com/genetic‐solutions/emma‐clinics/). EMMA & ALICE detect and quantify bacterial DNA, determining if the uterine microbial environment is optimal for pregnancy. DNA was extracted using a QIAamp cador Pathogen Mini Kit (Qiagen Inc., Venlo, The Netherlands) and quantified with Nanodrop. High‐throughput sequencing was performed using the Ion Chef Instrument: Model 4247 and Ion Torrent S5 XL Sequencer: Model 7728 (Thermo Fisher Scientific, Valencia, Spain). The test reports recommended personalized treatments based on the identified microbial composition in each sample.
The classification of EMMA & ALICE results is as follows (1): “normal”, indicating a LDM where Lactobacillus accounts for over 90% of all bacteria present in the sample, with no pathogenic bacteria were detected, (2) “abnormal”, indicating an endometrium dominated by non–Lactobacillus genera whose abundances account for more than 10% of the total bacterial composition. Abnormal in EMMA also includes the ALICE result, which is positive in ALICE if more than 10% of the CE‐causing bacteria are detected, otherwise the result is negative in ALICE. (3) “mild dysbiosis”, indicating that the endometrium is not Lactobacillus–dominated and pathogenic bacteria are not present in significant amounts, and (4) “ultralow biomass”, indicating insignificant amounts of bacteria, an almost sterile endometrium.
2.2.3. Intervention protocol
In cases where a normal result was obtained, patients continued to their ET cycles without any additional treatments (Figure 1). In cases identified as “abnormal,” a state of “dysbiosis” in the endometrium was diagnosed and antibiotic treatment followed by treatment with probiotics was recommended (Figure 1). The selection of antibiotics was tailored according to the pathogens detected and the individual clinical profile of each patient (e.g., possible existing antibiotic allergies). Probiotic treatment involved vaginal suppositories (Invag®, Biomed Krakow, Poland or Lactoflora®, Sante laboratory, Osaka, Japan) containing Lactobacillus strains available in Asia. A second sample was analyzed post‐treatment before proceeding with the ET (Figure 1). For patients with results indicating “mild dysbiosis”, or “ultralow biomass,” vaginal probiotic therapy was administered for 7–10 days starting on the pre‐ET cycle when menstrual bleeding was minimal (Figure 1).
FIGURE 1.
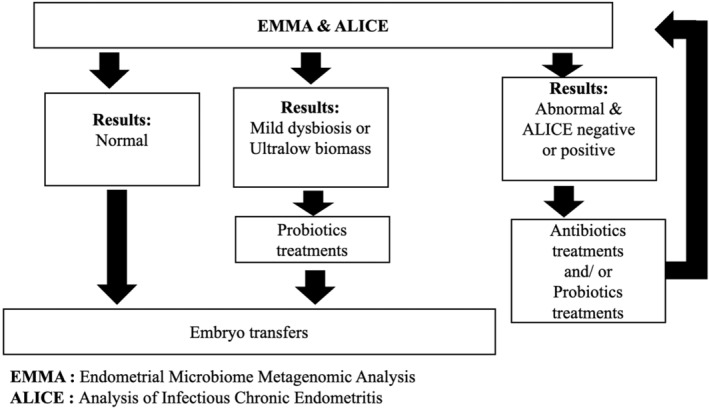
The results of EMMA & ALICE and the flow chart of post‐testing. Participant centers should treat the patients following the descriptions specified in EMMA &ALICE reports. Patients with “Normal” result, which indicate the patients with endometrial microbiome dominated by genus Lactobacillus, will continue their ART treatments according to the standard protocol of the clinic. The patients have EMMA & ALICE reports with “Mild dysbiosis” and “Ultralow biomass” results had received probiotic treatment to their vagina. The patients with Abnormal results, antibiotic treatments, probiotic treatments, re‐analysis, and embryo transfer after re‐analysis.
2.2.4. Definition of outcomes
Clinical pregnancy was defined by the presence of gestational sacs via ultrasound by the 6th week of gestation. Ongoing pregnancy was confirmed by the detection of fetal heartbeat by the 12th week of gestation. Early miscarriage was defined as pregnancy loss occurring before the 12th week of gestation.
2.2.5. Outcome measurements and statistical analysis
The primary outcome measure was a comparison of cumulative ET outcomes to achieve an ongoing pregnancy between three groups: the normal group, the abnormal group requiring antimicrobial followed by probiotic treatment, and the mild dysbiosis + ultralow biomass group that received standalone probiotic treatment. Secondary outcomes included the incidence of endometrial microbiome abnormalities in patients with RIF or recurrent pregnancy loss (RPL), cumulative clinical pregnancy rates, early miscarriage rates and adverse events related to EMMA & ALICE testing or its recommended treatments. Data were presented as means ± standard deviation (SD). A comparison of the groups was conducted using ANOVA, with Kruskal‐Wallis, and Fisher's exact tests applied as necessary. In addition, multivariate generalized linear models were used to analyze factors affecting ongoing pregnancy. For time‐to‐event analysis, the follow‐up time started on the date of the final EMMA & ALICE report. The follow‐up period ended when each woman's target event or interruption of fertility treatment was recorded. The target event was defined as the date of confirmed ongoing pregnancy and referral to obstetrics. The Kaplan–Meier (KM) survival analysis was used to compare time to pregnancy between the three groups, and the log‐rank test was used to test the differences. In addition, as a subgroup analysis for the five‐group classification of the original EMMA&ALICE results, KM survival curves were also evaluated with regard to ongoing pregnancies.
Statistical analyses were conducted using R software version 4.0.2. A p‐value <0.05 was considered as statistically significant.
3. RESULTS
This study included a total of 527 patients. The mean age of study participants was 37.7 ± 4.4 years, median duration of infertility was 33 months, and the median number of previous ETs was 3.
Out of all participants, 229 (43.4%) were classified in the normal group, exhibiting an LDM. The abnormal group included 110 patients (20.9%), characterized by a predominance of non‐Lactobacillus genera. The mild dysbiosis + ultralow biomass group included 188 patients (35.7%). Patient clinical variables for each result were compared, but there were no significant differences in the clinical variables assessed (Table 1).
TABLE 1.
Comparison of patient clinical variables according to EMMA & ALICE outcome groups.
| Normal | Abnormal | Mild + ultralow | p value | |
|---|---|---|---|---|
| Number of patients (n) | 229 | 110 | 188 | |
| Age (y) mean ± SD | 37.30 ± 4.69 | 37.65 ± 4.00 | 38.26 ± 4.12 | 0.080 |
| Duration of infertility (m) mean [IQR] |
31.50 [18.00, 57.25] |
36.00 [16.00, 62.00] |
32.50 [18.00, 53.00] |
0.527 |
|
History of delivery (n) mean [IQR] |
0.00 [0.00, 0.00] |
0.00 [0.00, 0.00] |
0.00 [0.00, 0.00] |
0.116 |
| Mean number of previous ET cycles mean [IQR] |
3.00 [2.00, 4.00] |
3.00 [2.00, 5.00] |
3.00[2.00, 4.25] | 0.788 |
|
Stage of embryo, % (n) Cleavage‐stage Blastocyst |
41.5 (131) |
38.8 (57) |
37.6 (97) |
0.633 |
|
58.5 (185) |
61.2 (90) |
62.4 (161) |
||
| PGT‐A, % (n) | 8.7 (20) | 10.9 (12) | 10.1 (19) | 0.793 |
Abbreviations: IQR, Interquartile Range; PGT, Preimplantation Genetic Testing.
The pathogenic bacteria detected in the abnormal group are shown in Figure 2. Gardnerella was the most abundant bacterium detected, found in 41.2 ± 19.3% of patients (n = 69). Following Gardnerella, Streptococcus was detected in 33.8 ± 26.4% of patients (n = 19), and Atopobium was the third most common bacterium detected, found in 28.0 ± 23.0% of patients (n = 47). The specific antimicrobial treatments administered against these bacteria are detailed in Table 2. After one course of antimicrobial treatment, 77 (70%) out of 110 patients in abnormal group were confirmed to have eliminated the pathogens upon re‐examination with EMMA & ALICE. Subsequent courses of antimicrobial treatment further improved the dysbiotic profile for all patients. No cases required doxycycline or ciprofloxacin, which have been reported as standard treatment for chronic endometritis (CE). 14 , 15
FIGURE 2.
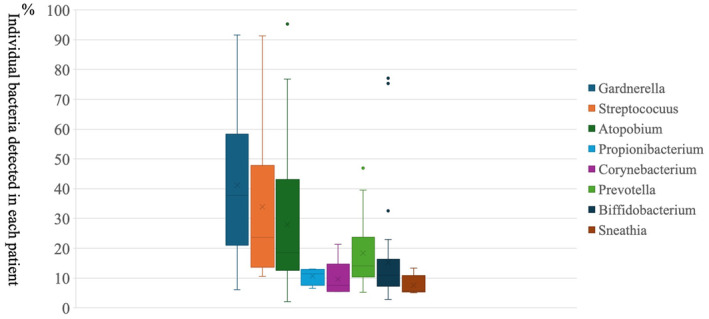
Pathogens detected in the abnormal group. The most prevalent bacteria identified in patients with abnormal results, excluding Lactobacillus, accounted for over 10% of cases. The results are presented in a box‐and‐whisker plot, which illustrates the mean and standard deviation of the percentage of bacteria detected.
TABLE 2.
Recommended antibiotic treatments for patients with abnormal results in EMMA & ALICE.
| Recommended first‐line antibiotic treatments for patients with abnormal results (n = 110) | |
|---|---|
| Amoxicillin/clavulanic acid (500 mg–125 mg every 8 h for 8 days) | 62 (56.4%) |
| Metronidazole (500 mg twice a day for 7 days) | 45 (40.9%) |
| Clindamycin (300 mg twice a day for 7 days) | 1 (0.9%) |
| None | 2 (1.8%) |
| Recommended second‐line antibiotic treatments for patients with abnormal results (n = 33) | |
| Clindamycin (300 mg twice a day for 7 days) | 21 (63.6%) |
| Amoxicillin/Clavulanic acid (500 mg–125 mg every 8 h for 8 days) | 5 (15.2%) |
| Metronidazole (500 mg twice a day for 7 days) | 6 (18.2%) |
| Azithromycin (1000 mg, only one dose) | 1 (3.0%) |
| Recommended third‐line antibiotic treatments for patients with abnormal results (n = 12) | |
| Metronidazole (500 mg twice a day for 7 days) | 5 (41.7%) |
| Clindamycin (300 mg twice a day for 7 days) | 4 (33.3%) |
| Amoxicillin/Clavulanic acid (500 mg–125 mg every 8 h for 8 days) | 2 (16.7%) |
| Clarithromycin (200 mg twice a day for 7 days) | 1 (8.3%) |
The efficacy of the treatments based on EMMA & ALICE results were evaluated through subsequent post‐test ETs. The pregnancy outcome following ET is the result of frozen ET in all cases except three fresh ET cases. A comparison of the transferred embryos amongst the three groups revealed no significant differences in the stage of the embryos (blastocyst vs. cleavage). Furthermore, the presence or absence of preimplantation genetic testing did not differ significantly amongst the groups (Table 1). Pregnancy outcomes after the interventions based on the EMMA & ALICE reports are shown in Table 3 for the three study groups. The clinical pregnancy, ongoing pregnancy and miscarriage rates were not significantly different between three groups.
TABLE 3.
Cumulative pregnancy outcomes per embryo transfer after administration of the recommended treatments in EMMA & ALICE reports.
| Normal | Abnormal | Mild dysbiosis+ultralow biomass | p value | |
|---|---|---|---|---|
| Rate of positive hCG % (n) |
62.9 (144/229) |
68.2 (75/110) |
63.8 (120/188) |
0.641 |
| Clinical pregnancy rate % (n) |
55.0 (126/229) |
53.6 (59/110) |
57.4 (108/188) |
0.794 |
| Ongoing pregnancy rate % (n) |
47.6 (109/229) |
48.2 (53/110) |
49.5 (93/188) |
0.935 |
| Miscarriage rate % (n) |
13.5 (17/126) |
10.2 (6/59) |
13.9 (15/108) |
0.831 |
A KM survival analysis was conducted to assess the impact of post‐ EMMA & ALICE interventions on pregnancy outcomes. The results indicated that the abnormal group, who required EMMA & ALICE based antimicrobial treatment, achieved pregnancy significantly earlier than the other two groups at 6 months' time (p = 0.011). This trend persisted until the 12‐month mark (p = 0.049) (Figure 3). The results of the subgroup analysis in the five groups showed that the “abnormal in EMMA/negative in ALICE” group achieved significantly earlier pregnancies at 6 months (p = 0.02) compared to the other groups. This trend was not observed in the “abnormal in EMMA/positive in ALICE” group. No significant differences were found between the mild and ultralow groups (Figure 4). Tables S1 show the results of patient background and cumulative pregnancy outcomes for the five groups.
FIGURE 3.
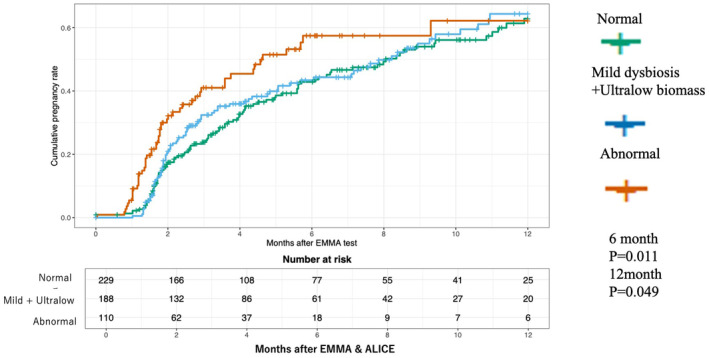
Kaplan–Meier survival analysis of time to ongoing pregnancy for each treatment intervention. The figure below demonstrates the cumulative pregnancy rates over time (in months) following EMMA & ALICE testing across three groups. Normal: Patients with normal endometrial microbiota (green). Mild dysbiosis + Ultralow biomass: Patients with mild dysbiosis or ultralow microbiota biomass (blue). Abnormal: Patients with significant dysbiosis requiring therapeutic intervention (orange).
FIGURE 4.
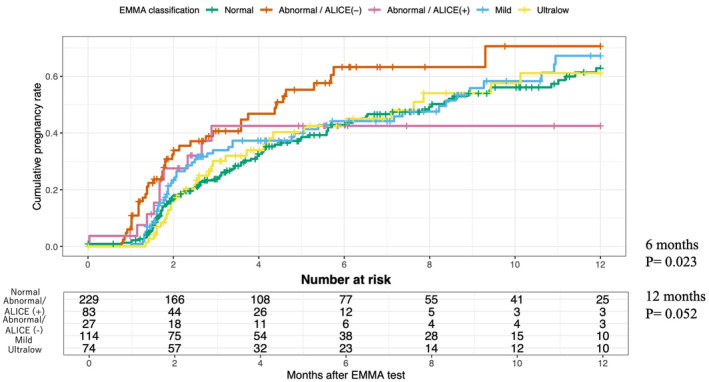
Kaplan–Meier survival analysis of time to ongoing pregnancy for each of the five EMMA & ALICE results. This figure shows the cumulative pregnancy rates over time (in months) following EMMA & ALICE testing across five patient subgroups classified by their microbiota: Normal: Patients with a normal endometrial microbiota profile (green). Abnormal/ALICE (−): Patients with dysbiosis but no pathogenic bacteria detected by ALICE (orange). Abnormal/ALICE (+): Patients with dysbiosis and pathogenic bacteria detected by ALICE (pink).Mild: Patients with mild dysbiosis (light blue). Ultralow: Patients with ultralow microbiota biomass (yellow).
A multivariate analysis using a generalized linear model was conducted with the primary outcome of the trial, ongoing pregnancy, as the endpoint. The relative risk for cumulative ongoing pregnancy compared with the normal group, in which no post‐test interventions were required, was 0.988 (95% CI 0.78–1.25, p = 0.921) for the abnormal group and 0.961(95% CI 0.79–1.17, p = 0.694) for the mild dysbiosis + ultralow biomass group. Maternal age was the only factor that significantly impacted ongoing pregnancy rates (Table 4).
TABLE 4.
The results of a multivariate analysis with ongoing pregnancy as the endpoint.
| Odds ratio | 95% CI | p value | |
|---|---|---|---|
| Age | 0.848 | 0.806–0.890 | <0.0001 |
| Duration of infertility | 0.995 | 0.989–1.001 | 0.104 |
| Mean number of previous ET cycles | 0.945 | 0.876–1.016 | 0.133 |
| History of miscarriage | 1.191 | 0.666–2.160 | 0.558 |
| History of delivery | 1.361 | 0.693–2.696 | 0.370 |
| EMMA & ALICE result: Abnormal | 1.103 | 0.671–1.816 | 0.700 |
| EMMA & ALICE result: mild dysbiosis + ultralow biomass | 1.229 | 0.803–1.886 | 0.342 |
Note: Multivariate analysis was performed using a generalized linear model with ongoing endpoints. Odds ratios for ongoing pregnancy according to the EMMA & ALICE results compared to the normal group, which did not require post‐test interventions.
Although multivariate analysis revealed no significant difference in cumulative ongoing pregnancy rates between EMMA & ALICE results, age was identified as a significant moderating factor. Consequently, the impact of age was subjected to further investigation. After stratifying the KM curve data by age, the analysis showed that interventions in individuals aged 35 years and younger resulted in pregnancy outcomes comparable to those observed in the normal group. However, in participants aged 36 years and older, those in the abnormal group achieved pregnancy significantly earlier than their counterparts in the normal and mild dysbiosis + ultra‐low biomass groups. (Figure 5).
FIGURE 5.
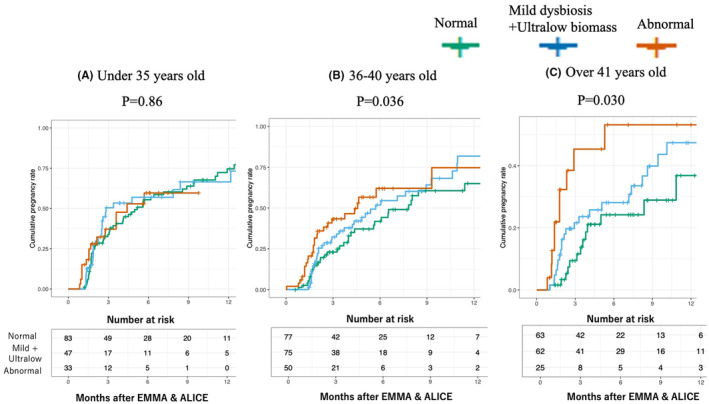
Kaplan–Meier survival analysis of time to ongoing pregnancy as comparison of ongoing pregnancies by age group. This figure presents Kaplan–Meier survival analyses of cumulative ongoing pregnancy rates following EMMA & ALICE testing, stratified by three age groups: (A) Under 35 years old (p = 0.86): No significant differences in ongoing pregnancy rates between the groups were observed. (B) 36–40 years old (p = 0.036): Significant differences were observed, with the abnormal group (orange line) achieving higher cumulative pregnancy rates. (C) Over 41 years old (p = 0.030): Significant differences were also noted, with the abnormal group showing the highest pregnancy rates, followed by the mild dysbiosis + ultralow biomass group (blue line) and the normal group (green line). These results highlight that therapeutic interventions for abnormal microbiota are particularly effective in older patients (36 years and above), suggesting a greater impact of dysbiosis correction in this demographic.
Regarding adverse events observed in the study, no patient developed serious complications such as uterine perforation. In terms of adverse reactions due to therapeutic interventions, four patients experienced rashes due to amoxicillin‐clavulanic acid, which improved after 7 days of treatment with an anti‐allergic drug (fexofenadine hydrochloride). No serious complications such as anaphylactic shock due to antibiotic administration were observed.
4. DISCUSSION
Previous studies have indicated that non‐Lactobacillus‐dominated microbiotas (non‐LDMs) are associated with lower pregnancy rates and that dysbiosis negatively affects pregnancy outcomes. 10 , 16 However, evidence on whether therapeutic interventions for dysbiosis can improve pregnancy rates has been limited. This study is the first to demonstrate that appropriate therapeutic interventions based on the EMMA & ALICE report for patients in the abnormal group can result in ongoing pregnancy rates that are comparable to those of the normal patient group, which did not require treatment. A previous report showed significantly higher implantation and ongoing pregnancy rates in RIF patients who underwent EMMA & ALICE compared to those who did not. 13 However, that study did not compare pregnancy outcomes between the treatment‐free normal group and the treatment‐requiring abnormal group, leaving it unclear whether therapeutic intervention improved pregnancy rates.
By increasing the sample size and directly comparing pregnancy outcomes between the normal and abnormal groups, this study provides robust evidence supporting the clinical utility of microbiota‐targeted interventions. Patients with dysbiosis who underwent the recommended therapeutic interventions were able to achieve ongoing pregnancies at an earlier point in time than those in the control group. These findings indicate that the early identification and treatment of dysbiosis can significantly reduce the time to pregnancy, emphasizing the importance of incorporating endometrial microbiota testing into the clinical management of infertility.
Furthermore, Mitter et al. 17 reported that antibiotic treatment following diagnostic endometrial biopsy significantly shortened the time to live birth in women with CE experiencing RIF or RPL. However, the study did not differentiate between specific microbiota profiles, such as those detected by EMMA & ALICE. A subgroup analysis revealed that the efficiency of antimicrobial therapy differs based on the microbiota profile. In particular, patients who were classified as “Abnormal in EMMA/Positive in ALICE” may have CE, which could result in persistently lower pregnancy rates even after antimicrobial treatment has eliminated pathogenic bacteria. In contrast, the group classified as “Abnormal in EMMA/Negative in ALICE” demonstrated a more favorable response to antimicrobial interventions, resulting in improved pregnancy outcomes. These findings indicate that CE, particularly when accompanied by dysbiosis, is characterized by distinct pathological mechanisms that may require the application of tailored treatment strategies.
Consistent with previous studies, disruptions in the endometrial microbiota are associated with compromised immune function, reduced implantation rates, and increased miscarriage rates. 18 While both dysbiosis and CE have been shown to impair reproductive outcomes, their coexistence may result in a more pronounced impact, emphasizing the importance of distinguishing between these pathologies. Furthermore, our findings suggest that dysbiosis without active CE may be more responsive to correction through targeted antimicrobial or probiotic therapies.
The use of NGS‐based precision diagnostics offers a notable advantage in this context. It allows for the identification of specific pathogenic bacteria while simultaneously preserving beneficial species like Lactobacillus. In this study, targeted antimicrobial therapies based on EMMA & ALICE reports achieved a 70% pathogen elimination rate within one week, which is markedly higher than the rates reported with conventional CE treatments. 19 This approach minimizes the risks associated with broad‐spectrum antibiotics, including the development of antibiotic resistance and the depletion of essential microbiota components that are necessary for endometrial health.
Multivariate analysis identified age as the only significant factor influencing cumulative ongoing pregnancy rates. KM curve analysis validated by age group revealed that pregnancy outcomes in the normal group and the treated abnormal group were comparable in women under 35 years old. However, as age increased, the treated abnormal group showed better pregnancy outcomes compared to the normal group. This finding suggests that age‐related changes in systemic microflora, including the gut, 20 , 21 may make EMMA & ALICE more critical for identifying the causes of RIF and RPL in older women.
Subgroup analysis further emphasized the clinical significance of specific pathogens associated with dysbiosis, including Gardnerella, Atopobium and Streptococcus. These bacteria, which are commonly identified in dysbiotic endometria, are well‐known causative agents of CE. 22 , 23 In this study, they were effectively targeted using antibiotic regimens tailored to NGS findings. Our findings confirm the previously established correlation between dysbiosis and CE. However, our results also demonstrate that there are instances where dysbiosis is present without CE, 13 and vice versa. This indicates that the early diagnosis and intervention of dysbiosis may address a more extensive range of infertility‐related issues than treatments that solely target CE.
Overall, this study provides robust evidence that EMMA & ALICE‐guided therapeutic interventions enhance reproductive outcomes, particularly in patients with dysbiosis. The administration of early and precise treatment, such as a brief targeted course of antibiotics, has been demonstrated to beneficially modulate the endometrial microbiota, reduce the time to pregnancy, and increase ongoing pregnancy rates. These findings underscore the potential of NGS‐based diagnostics to transform the management of infertility by enabling tailored and effective interventions for RIF and RPL.
4.1. Limitations
This study provides valuable insights into the role of the endometrial microbiota in fertility; however, several limitations warrant consideration. Despite stringent protocols to minimize contamination, the risk of introducing external bacteria during endometrial sample collection cannot be entirely eliminated, potentially confounding the microbial analysis. 8
Moreover, while NGS enables detailed profiling of the microbiota, it cannot distinguish between live and dead bacteria, which may complicate the interpretation of microbial activity and clinical significance. Additionally, NGS is limited in its ability to detect non‐bacterial microorganisms, such as fungi and viruses, leaving their potential impact on reproductive health poorly understood. 24
The positive outcomes observed in the antimicrobial and probiotic treatment groups for the dysbiosis cohort suggest that bacterial genera other than Lactobacillus species may contribute to RIF or RPL. This is supported by a report demonstrating an association between the dominance of specific bacterial genera, such as Pseudomonas, Acinetobacter, and Dysgonomonas, with infertility‐related diseases like adenomyosis and endometriosis. 25 Conversely, previous studies have reported that bacteria commonly detected in non‐LDM patients are also frequently found in the endometria of healthy reproductive‐aged women. 26 , 27 , 28 Accordingly, the pathological nature of the endometrium in non‐LDM patients remains a matter of debate.
Addressing these limitations in future research will be crucial for enhancing our understanding and improving interventions focusing on the endometrial microbiota.
5. CONCLUSION
This multicenter prospective study underscores the potential of analyzing and intervening in the endometrial microbiota to reduce the time to pregnancy in patients with dysbiosis. Despite valuable insights, the study highlights the need for improved sample collection techniques to minimize contamination and enhance reliability. EMMA & ALICE‐based personalized treatment approaches are crucial for improving pregnancy outcomes by addressing dysbiosis effectively.
Future research should focus on optimizing endometrial microbiota sampling, elucidating the interplay between dysbiosis and CE, and developing tailored therapeutic strategies for different microbiota profiles. A multifaceted evaluation of the effectiveness of EMMA & ALICE is essential to refine their clinical application. Strategically optimizing the endometrial environment through microbiota‐targeted diagnostics and interventions has the potential to significantly enhance pregnancy rates and redefine infertility treatment.
CONFLICT OF INTEREST STATEMENT
All authors have no conflicts of interest to declare, relevant to this study. Hiroaki Yoshida are Editorial Board members of Reproductive Medicine and Biology and co‐authors of this article. To minimize bias, they were excluded from all editorial decision‐making related to the acceptance of this article for publication.
ETHICS STATEMENT
This study adhered to the Declaration of Helsinki regarding medical research involving human subjects and followed Good Clinical Practice guidelines. It was approved by the ethics committee of the Institutional Review Board of JISART. This trial is registered in the University Hospital Medical Information Network (UMIN000036917) clinical trial registry in Japan. Written informed consent was obtained from all enrolled patients.
Supporting information
Appendix S1.
Appendix S2.
ACKNOWLEDGMENTS
We express our gratitude to all participating JISART facilities for their invaluable cooperation in providing data for this study. Special thanks to Ms. Viviane Casaroli, MSc, for her assistance with English proofreading. This research was supported by funding from Igenomix Japan K.K.
Iwami N, Komiya S, Asada Y, Tatsumi K, Habara T, Kuramoto T, et al. “Shortening time to pregnancy in infertile women by personalizing treatment of microbial imbalance through Emma & Alice: A multicenter prospective study”. Reprod Med Biol. 2025;24:e12634. 10.1002/rmb2.12634
REFERENCES
- 1. Turnbaugh PJ, Ley RE, Hamady M, Fraser‐Liggett CM, Knight R, Gordon JI. The human microbiome project. Nature. 2007;449(7164):804–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Peterson J, Garges S, Giovanni M, McInnes P, Wang L, Schloss JA, et al. The NIH human microbiome project. Genome Res. 2009;19(12):2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Mitchell CM, Haick A, Nkwopara E, Garcia R, Rendi M, Agnew K, et al. Colonization of the upper genital tract by vaginal bacterial species in nonpregnant women. Am J Obstet Gynecol. 2015;212(5):61:e1–611.e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mlodzik N, Lukaszuk K, Sieg W, Jakiel G, Smolarczyk R. Endometrial microbiota—do they mean more than we have expected? Ginekol Pol. 2020;91(1):45–48. [DOI] [PubMed] [Google Scholar]
- 5. Toson B, Simon C, Moreno I. The endometrial microbiome and its impact on human conception. Int J Mol Sci. 2022;23(1):1–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Benner M, Ferwerda G, Joosten I, Van Der Molen RG. How uterine microbiota might be responsible for a receptive, fertile endometrium. Hum Reprod Update. 2018;24(4):393–415. [DOI] [PubMed] [Google Scholar]
- 7. Franasiak JM, Werner MD, Juneau CR, Tao X, Landis J, Zhan Y, et al. Endometrial microbiome at the time of embryo transfer: next‐generation sequencing of the 16S ribosomal subunit. J Assist Reprod Genet. 2016;33(1):129–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Moreno I, Cicinelli E, Garcia‐Grau I, Gonzalez‐Monfort M, Bau D, Vilella F, et al. The diagnosis of chronic endometritis in infertile asymptomatic women: a comparative study of histology, microbial cultures, hysteroscopy, and molecular microbiology. Am J Obstet Gynecol. 2018;218(6):602.e1–602.e16. [DOI] [PubMed] [Google Scholar]
- 9. Ravel J, Gajer P, Abdo Z, Schneider GM, Koenig SSK, McCulle SL, et al. Vaginal microbiome of reproductive‐age women. Proc Natl Acad Sci USA. 2011;15:4680–4687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Moreno I, Codoñer FM, Vilella F, Valbuena D, Martinez‐Blanch JF, Jimenez‐Almazán J, et al. Evidence that the endometrial microbiota has an effect on implantation success or failure. Am J Obstet Gynecol. 2016;215(6):684–703. [DOI] [PubMed] [Google Scholar]
- 11. Moreno I, Garcia‐Grau I, Perez‐Villaroya D, Gonzalez‐Monfort M, Bahçeci M, Barrionuevo MJ, et al. Endometrial microbiota composition is associated with reproductive outcome in infertile patients. Microbiome. 2022;10(1):1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Carosso A, Revelli A, Gennarelli G, Canosa S, Cosma S, Borella F, et al. Controlled ovarian stimulation and progesterone supplementation affect vaginal and endometrial microbiota in IVF cycles: a pilot study. J Assist Reprod Genet. 2020;37(9):2315–2326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Iwami N, Kawamata M, Ozawa N, Yamamoto T, Watanabe E, Mizuuchi M, et al. Therapeutic intervention based on gene sequencing analysis of microbial 16S ribosomal RNA of the intrauterine microbiome improves pregnancy outcomes in IVF patients: a prospective cohort study. J Assist Reprod Genet. 2023;40(1):125–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Vitagliano A, Saccardi C, Noventa M, Di Spiezio SA, Saccone G, Cicinelli E, et al. Effects of chronic endometritis therapy on in vitro fertilization outcome in women with repeated implantation failure: a systematic review and meta‐analysis. Fertil Steril. 2018;110(1):103–112.e1. [DOI] [PubMed] [Google Scholar]
- 15. Kitaya K, Takeuchi T, Mizuta S, Matsubayashi H, Ishikawa T. Endometritis: new time, new concepts. Fertil Steril. 2018;110(2):344–350. [DOI] [PubMed] [Google Scholar]
- 16. Cela V, Daniele S, Obino MER, Ruggiero M, Zappelli E, Ceccarelli L, et al. Endometrial dysbiosis is related to inflammatory factors in women with repeated implantation failure: a pilot study. J Clin Med. 2022;11(9):1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Mitter VR, Meier S, Rau TT, Gillon T, Mueller MD, Zwahlen M, et al. Treatment following hysteroscopy and endometrial diagnostic biopsy increases the chance for live birth in women with chronic endometritis. Am J Reprod Immunol. 2021;86(5):1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Al‐Nasiry S, Ambrosino E, Schlaepfer M, Morré SA, Wieten L, Voncken JW, et al. The interplay between reproductive tract microbiota and immunological system in human reproduction. Front Immunol. 2020;11(March):1–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cicinelli E, Resta L, Loizzi V, Pinto V, Santarsiero C, Cicinelli R, et al. Antibiotic therapy versus no treatment for chronic endometritis: a case‐control study. Fertil Steril. 2021;115(6):1541–1548. [DOI] [PubMed] [Google Scholar]
- 20. Takagi T, Naito Y, Inoue R, Kashiwagi S, Uchiyama K, Mizushima K, et al. Differences in gut microbiota associated with age, sex, and stool consistency in healthy Japanese subjects. J Gastroenterol. 2019;54(1):53–63. [DOI] [PubMed] [Google Scholar]
- 21. Hopkins MJ, Sharp R, Macfarlane GT. Age and disease related changes in intestinal bacterial populations assessed by cell culture, 16S rRNA abundance, and community cellular fatty acid profiles. Gut. 2001;48(2):198–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Cicinelli E, Matteo M, Tinelli R, Pinto V, Marinaccio M, Indraccolo U, et al. Chronic endometritis due to common bacteria is prevalent in women with recurrent miscarriage as confirmed by improved pregnancy outcome after antibiotic treatment. Reprod Sci. 2014;21(5):640–647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Liu Y, Ko EYL, Wong KKW, Chen X, Cheung WC, Law TSM, et al. Endometrial microbiota in infertile women with and without chronic endometritis as diagnosed using a quantitative and reference range‐based method. Fertil Steril. 2019;112(4):707–717.e1. [DOI] [PubMed] [Google Scholar]
- 24. Molina NM, Sola‐Leyva A, Haahr T, Aghajanova L, Laudanski P, Castilla JA, et al. Analysing endometrial microbiome: methodological considerations and recommendations for good practice. Hum Reprod. 2021;36(4):859–879. [DOI] [PubMed] [Google Scholar]
- 25. Chen C, Song X, Wei W, Zhong H, Dai J, Lan Z, et al. The microbiota continuum along the female reproductive tract and its relation to uterine‐related diseases. Nat Commun. 2017;8(1):1–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Li F, Chen C, Wei W, Wang Z, Dai J, Hao L, et al. The metagenome of the female upper reproductive tract. Gigascience. 2018;7(10):1–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Winters AD, Romero R, Gervasi MT, Gomez‐Lopez N, Tran MR, Garcia‐Flores V, et al. Does the endometrial cavity have a molecular microbial signature? Sci Rep. 2019;9(1):9905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Sola‐Leyva A, Andrés‐Leóe E, Molina NM, Terron‐Camero LC, Plaza‐Diaz J, Sáez‐Lara MJ, et al. Mapping the entire functionally active endometrial microbiota. Hum Reprod. 2021;36(4):1021–1031. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1.
Appendix S2.


