Abstract
Genome-wide DNA methylation signatures correlate with and distinguish central nervous system (CNS) tumor types. Since the publication of the initial CNS tumor DNA methylation classifier in 2018, this platform has been increasingly used as a diagnostic tool for CNS tumors, with multiple studies showing the value and utility of DNA methylation-based classification of CNS tumors. A Consortium to Inform Molecular and Practical Approaches to CNS Tumor Taxonomy (cIMPACT-NOW) Working Group was therefore convened to describe the current state of the field and to provide advice based on lessons learned to date. Here, we provide recommendations for the use of DNA methylation-based classification in CNS tumor diagnostics, emphasizing the attributes and limitations of the modality. We emphasize that the methylation classifier is one diagnostic tool to be used alongside previously established diagnostic tools in a fully integrated fashion. In addition, we provide examples of the inclusion of DNA methylation data within the layered diagnostic reporting format endorsed by the World Health Organization (WHO) and the International Collaboration on Cancer Reporting. We emphasize the need for backward compatibility of future platforms to enable accumulated data to be compatible with new versions of the array. Finally, we outline the specific connections between methylation classes and CNS WHO tumor types to aid in the interpretation of classifier results. It is hoped that this update will assist the neuro-oncology community in the interpretation of DNA methylation classifier results to facilitate the accurate diagnosis of CNS tumors and thereby help guide patient management.
Keywords: CNS tumors, DNA methylation profiling, molecular classification, neuropathology
Key Points.
DNA methylation profiling is a valuable tool for the diagnosis of central nervous system tumors.
Results should be interpreted within the entire context of the case.
Inclusion of methylation results into a layered diagnostic format is recommended.
DNA methylation profiling has emerged as a practical platform for the classification of central nervous system (CNS) tumors. Since the initial description of its potential across CNS tumor types and the development of a classifier by the Heidelberg group in 2018,1 use of this diagnostic strategy has expanded to additional centers. Notably, The World Health Organization (WHO) 2021 Classification of Tumours of the Central Nervous System (WHO CNS “Blue Book”)2 highlights the utility of DNA methylation profiling by indicating this modality as either an “essential” or “desirable” diagnostic criterion in a large proportion of tumor types. Key attributes of DNA methylation profiling include the generation of objective, reproducible, and transferable data metrics that contribute to more precise and accurate tumor classification, prognostic information (in some tumor types), and the potential to discover new tumor types/subtypes.
With the increasing use of DNA methylation-based classification in clinical diagnostics, and in line with previous updates,3–10 the Consortium to Inform Molecular and Practical Approaches to CNS Tumor Taxonomy (cIMPACT-NOW) assembled a group of experienced neuropathologists and clinical neuro-oncologists to provide recommendations regarding the use of genome-wide DNA methylation profiling in CNS tumor diagnostics. These recommendations, based on the literature and collective experience, relate to methylation classifier results in daily practice, observed attributes and limitations of the classifier, and selection of cases for methylation profiling.
General Principles and Practice of DNA Methylation Profiling for CNS Tumor Diagnostics
DNA methylation profiling is based on the empirical observation that patterns of DNA methylation across the genome are correlated with tumor type in a reproducible manner. Epigenetic changes occurring because of specific genomic alterations can also influence the DNA methylation profile of a neoplasm. By interrogating that pattern on a given sample, and by then comparing the findings to a reference set of defined methylation classes, the closest “match” can be determined, along with a confidence score. Therefore, the reference set is a defining feature of a classifier, since it determines what methylation-defined groups can or cannot be classified. The selection of cases and groups to be included in the reference set is of primary importance for the characteristics of a classifier. A major step forward was the publication of the initial CNS tumor classifier in 2018, spearheaded by a team from Heidelberg, Germany, where the first reference set was defined for 82 tumor methylation classes and 9 nonneoplastic control tissue classes.1 The reference set data for this classifier was made publicly available. Since that time subsequent classifiers have been developed by the Heidelberg team, the most recent of which (Version 12.8) has 184 classes. The publication of this new version in a scientific journal is currently in preparation. A complementary classifier (NCI/Bethesda classifier) is in clinical use in some institutions. A manuscript describing the NCI/Bethesda classifier and its classes is in preparation (Aldape et al., in preparation). It is noted that while some classes represent well-established CNS WHO tumor types, other classes are less well-defined, often delineated based on the identification of a new group emerging from an ever-growing dataset. In that sense, it is understood that each classifier is a “data freeze” at a specific time point, of an essentially dynamic process in the development and annotation of methylation-defined groups as they emerge.
In terms of a DNA methylation profiling platform, the most used platform, and with which the initial Heidelberg classifier was developed, is based on high-throughput methylation arrays. The initial classifier was based on the Infinium HumanMethylation450 BeadChip or “450k array,” so named because it had approximately 450 000 interrogated CpG sites on the array. Subsequent iterations of this array (version 1 and version 2 of the “EPIC” array) have increased the total number of CpG sites but decreased the number of common interrogatable probes (present on all 3 versions of the array: 485 512 in 450K, 452 822 in 450K/EPICv1, and 394 380 in 450K/EPICv1/EPICv2). Consequently, this has necessitated to retrain and revalidate the classifiers for every microarray release in a backward-compatible manner so that previously established reference case cohorts could still be used. The rationale for this approach is that it allows the large number of samples profiled with older array versions to contribute to the reference set and classifier. A relevant and practical consideration is that a comprehensive reference set requires the inclusion of multiple samples per class, which can be a challenge with respect to the accrual of rare tumor types and methylation classes. Of note, additional analytical issues for the classifier arise when a new array version drops off chemically modified probes from a previous version of the array or changes the probe chemistry for a particular genetic location.
Following the determination of the reference set, classifiers are then developed using machine learning. The original Heidelberg classifier used a Random Forest algorithm.1 Alternative machine learning algorithms and classifier approaches have also been proposed.11,12 Classifier performance may differ depending on the specific machine learning algorithm, but these differences are likely modest compared to the quality and diversity of the reference set in determining the overall classifier performance. With respect to the application of the classifier to tumor diagnostic practice, the experience from multiple centers indicates improved diagnostic accuracy following the use of DNA methylation-based classification,13–16 an opportunity that may be further enhanced using multi-omic profiling.17 Beyond its application to routine diagnostics, methylation profiling also has the potential to discover new tumor types that were not (yet) discernable using more conventional pathological approaches. As an example, high-grade astrocytoma with piloid features (HGAP, recognized in the 2021 WHO classification) was discovered in this way.18 The use of methylation profiling has further led to an appreciation of clinically relevant subtypes of recognized histologic entities (eg, medulloblastoma and ependymoma) where methylation classes can define them.19,20 Further attributes of DNA methylation profiling include the ability to determine the promoter methylation status of specific genes (eg, methylguanine methyltransferase [MGMT]21), as well as in the determination of genome-wide DNA copy number profiles, particularly helpful for the diagnosis of tumor types characterized by stereotypical DNA copy number aberrations (eg, Oligodendroglioma, IDH-mutant, and 1p/19q-codeleted). Finally, the results of methylation profiling can be an important component of teaching/case review for trainees, in a manner similar to the inclusion and integration of other types of molecular data for diagnostics. By establishing additional evidence to support a specific diagnosis, cases confirmed by methylation profiling used for training purposes can be chosen that are relatively impervious to differences of opinion; this can be especially important for uncommon and rare tumor types. It may also serve as a diagnostic aid in regions of the world with a scarcity of well-trained neuropathologists. To this end, a consortium is being launched to help systematically build capacity in low- and middle-income countries. That said, it is expected that the availability of methylation profiling will likely be a challenge, for a variety of reasons, at many centers in the foreseeable future.
Methylation Classes and CNS WHO-Defined Tumor Types
Relevant to reporting of methylation profiling results is knowledge of the relationships of methylation classes, as defined in the Heidelberg classifier, to defined CNS WHO tumor types. While methylation classes generally track with WHO-defined CNS tumor types, they do not always do so in a one-to-one correlation. As an example (mentioned above), 2 methylation classes in the Heidelberg classifier correspond to Astrocytoma, IDH-mutant, and 5 methylation subtypes correspond to Supratentorial ependymoma, ZFTA fusion-positive. In other examples, the classifier name may not always fully represent the corresponding matching tumor types. For example, the methylation class Subependymoma, posterior fossa includes unequivocal posterior fossa subependymomas, but also tumors with unequivocal ependymoma histopathology, a situation that is currently being addressed by an independent cIMPACT-NOW Working Group. The “Melanocytoma” methylation class includes Melanocytoma, but also primary melanocytic tumors of higher grade. A practical consequence to this may occur when a recipient of a pathology report sees a match to the “melanocytoma” family when the tumor is actually a melanoma, which can be initially confusing to those less experienced with CNS methylation profiling results. Some CNS WHO-defined tumor types do not have identified methylation classes, including Diffuse low-grade glioma, MAPK pathway-altered, and Embryonal tumors NOS/NEC, both of which are a “basket” of tumors with diverse biological features. Additional examples include Multinodular and vacuolating neuronal tumor and Dysplastic cerebellar gangliocytoma (Lhermitte–Duclos disease), which may lack distinctive methylation signatures relative to control brain tissues. Such tumors may not yet have sufficient methylation profiling data available to be included in a classifier, but it is not clear yet whether such tumors correspond to defined methylation classes. Chondrosarcoma and Mesenchymal chondrosarcoma are WHO CNS tumor types that are not represented in the Heidelberg CNS classifier but are represented in a separate sarcoma classifier.22 Finally, many of the tumors described in the “Genetic tumor syndromes involving the CNS” of the 2021 CNS WHO and other tumors resulting from cancer predisposition syndromes are not represented well in the classifier and specific examples of such tumors may have distinct methylation signatures when compared to their sporadic counterparts.23
Conversely, a substantial number of methylation classes exist that do not correspond to a current CNS WHO tumor type, many of which have been recently described in the literature by virtue of distinctive methylation profiling signatures as well as additional defining features. Examples include “Adult-type diffuse high-grade glioma, IDH-wildtype, subtype F,” where tumors matching to this class were given a proposed name of “Gliomatosis Cerebri-like Glioma, IDH-wildtype” based on clinical presentation (gliomatosis based on imaging studies), frequent TERT and PIK3R1 mutations and an intermediate clinical course.24 Another example is “Glioneuronal tumor with ATRX alteration, kinase fusion and anaplastic features,” defined by both frequent ATRX loss as well as recurrent fusions, most often involving members of the NTRK family.25 A third instance is “High-grade glioma with pleomorphic and pseudopapillary features” (HPAP), a glioma in adults with frequent TP53 mutations and highly recurrent monosomy 13,26 a class which is present in the NCI/Bethesda classifier (https://methylscape.ccr.cancer.gov/) but not in the Heidelberg classifier. Additional examples of recently described methylation classes of CNS tumors that are not represented by a CNS WHO tumor type exist27–40 and the development of criteria for evaluation of such novel methylation classes for inclusion in future CNS WHO classifications is the subject of a separate cIMPACT-NOW Working Group. Finally, several methylation classes exist in the Heidelberg CNS tumor classifier that are in alternative WHO classifications outside the CNS, including olfactory neuroblastoma, retinoblastomas, intraocular medulloepithelioma, and several classes representing neuroblastoma. Overall, such complexities in the relationships of methylation classes to WHO-defined tumor types can be gained with experience and interdisciplinary discussion. That said, efforts to clarify these relationships more precisely to ensure that class names are, when possible, aligned with corresponding WHO tumor type designations, are recommended. A notable issue with the Heidelberg classifier is that the CNS WHO correlate is sometimes at the level of the methylation Superfamily (eg, Meningioma), sometimes at the level of the methylation Family (eg, Glioblastoma, IDH-wildtype), and sometimes at the level of the methylation Class (eg, Posterior fossa ependymoma group A). A future goal may be to harmonize the terminology used for the different methylation levels with that of the CNS WHO tumor taxonomy and nomenclature to maximize clarity and facilitate the integration of methylation results with CNS WHO designations. As a resource designed to clarify relationships of CNS WHO tumor types with currently defined methylation classes (and vice versa), we display this information in tabular form. Table 1 connects methylation classes in the Heidelberg CNS classifier that correspond to current CNS WHO tumor types. Table 2 shows CNS tumor methylation classes that do not correspond to a current CNS WHO tumor type. CNS WHO tumor types that are not represented in methylation classifiers are shown in Table 3.
Table 1.
WHO CNS5 Tumor Types That Correspond to Methylation Classes*, With Their Relationships Identified
| Site | CNS WHO category | CNS WHO family | CNS WHO tumor type | Methylation class(es) |
|---|---|---|---|---|
| Gliomas, glioneuronal tumors, and neuronal tumors | Gliomas, glioneuronal tumors, and neuronal tumors | Adult-type diffuse gliomas | Astrocytoma, IDH-mutant | Astrocytoma, IDH-mutant; high grade and lower grade |
| Oligodendroglioma, IDH-mutant, and 1p/19q-codeleted | Oligodendroglioma, IDH-mutant, and 1p/19q-codeleted | |||
| Glioblastoma, IDH-wildtype | Glioblastoma, IDH-wildtype, and subtype posterior fossa | |||
| Glioblastoma, IDH-wildtype, mesenchymal (incl. subtype B), RTK1, RTK2, and posterior fossa subtypes | ||||
| Glioblastoma, IDH-wildtype with primitive neuronal component | ||||
| Pediatric-type diffuse low-grade gliomas | Diffuse astrocytoma, MYB or MYBL1-altered | Diffuse astrocytoma, MYB or MYBL1-altered, subtypes B, C, and D | ||
| Angiocentric glioma | Angiocentric glioma, MYB/MYBL1-altered | |||
| Polymorphous low-grade neuroepithelial tumor of the young | Polymorphous low-grade neuroepithelial tumor of the young | |||
| Pediatric-type diffuse high-grade gliomas | Diffuse midline glioma, H3 K27-altered | Diffuse midline glioma, H3 K27-altered, subtype EGFR-altered | ||
| Diffuse midline glioma, H3 K27-altered, subtype H3 K27-mutant, or EZHIP expressing | ||||
| Diffuse hemispheric glioma, H3 G34-mutant | Diffuse hemispheric glioma, H3 G34-mutant | |||
| Diffuse pediatric-type high-grade glioma, H3 wildtype, and IDH-wildtype | Diffuse pediatric-type high-grade glioma, H3 wildtype and IDH-wild type, and Subtypes A and B | |||
| Diffuse pediatric-type high-grade glioma, MYCN subtype | ||||
| Diffuse pediatric-type high-grade glioma, RTK1 and RTK2 subtypes | ||||
| Infant-type hemispheric glioma | Infant-type hemispheric glioma | |||
| Circumscribed astrocytic gliomas | Pilocytic astrocytoma | Pilocytic astrocytoma (all classes) | ||
| High-grade astrocytoma with piloid features | High-grade astrocytoma with piloid features | |||
| Pleomorphic xanthoastrocytoma | Pleomorphic xanthoastrocytoma | |||
| Subependymal giant cell astrocytoma | Subependymal giant cell astrocytoma, TSC1/TSC2-altered | |||
| Chordoid glioma | Chordoid glioma, PRKCA-mutant | |||
| Astroblastoma, MN1-altered | Astroblastoma, MN1-altered, and MN1:BEND2-fused | |||
| Neuroepithelial tumor, MN1:CXXC5-fused | ||||
| Glioneuronal and neuronal tumors | Ganglioglioma/gangliocytoma | Ganglioglioma | ||
| Desmoplastic infantile astrocytoma and ganglioglioma | Desmoplastic infantile ganglioglioma/desmoplastic infantile astrocytoma | |||
| Dysembryoplastic neuroepithelial tumor | Dysembryoplastic neuroepithelial tumor | |||
| Diffuse glioneuronal tumor with oligodendroglioma-like features and nuclear clusters | Diffuse glioneuronal tumor with oligodendroglioma-like features and nuclear clusters (DGONC) | |||
| Papillary glioneuronal tumor | Papillary glioneuronal tumor; PRKCA-fused | |||
| Rosette forming glioneuronal tumor | Rosette forming glioneuronal tumor | |||
| Myxoid glioneuronal tumor | Myxoid glioneuronal tumor, PDGFRA-mutant | |||
| Diffuse leptomeningeal glioneuronal tumor | Diffuse leptomeningeal glioneuronal tumor, subtypes 1 and 2 | |||
| Central neurocytoma | Central neurocytoma | |||
| Extraventricular neurocytoma | Extraventricular neurocytoma | |||
| Cerebellar liponeurocytoma | Cerebellar liponeurocytoma | |||
| Ependymal tumors | Supratentorial ependymoma, ZFTA fusion-positive | Supratentorial ependymoma, ZFTA fusion-positive (all subclasses) | ||
| Supratentorial ependymoma, YAP1 fusion-positive | Supratentorial ependymoma, YAP1 fusion-positive | |||
| Posterior fossa group A (PFA) ependymoma | Posterior fossa group A (PFA) ependymoma (all subclasses) | |||
| Posterior fossa group B (PFB) ependymoma | Posterior fossa group B (PFB) ependymoma (all subclasses) | |||
| Spinal ependymoma | Spinal ependymoma | |||
| Spinal ependymoma, MYCN-amplified | Spinal ependymoma, MYCN-amplified | |||
| Myxopapillary ependymoma | Myxopapillary ependymoma | |||
| Subependymoma | Subependymoma (all classes)** | |||
| Choroid plexus tumors | Choroid plexus tumors | Choroid plexus tumors | Choroid plexus papilloma, atypical choroid plexus papilloma, choroid plexus carcinoma | Choroid plexus papilloma and carcinoma (all classes) |
| Embryonal tumors | Medulloblastoma | Medulloblastomas, molecularly defined | Medulloblastoma, WNT-activated | Medulloblastoma, WNT-activated |
| Medulloblastoma, SHH-activated and TP53-wildtype, Medulloblastoma, SHH-activated and TP53-mutant | Medulloblastoma, SHH-activated (subclasses 1–4 & subclass IDH-mutant) | |||
| Medulloblastoma, non-WNT/non-SHH | Medulloblastoma, non-WNT/non-SHH, groups 3 and 4 (subclasses I—VIII) | |||
| Medulloblastoma, histologically defined | Medulloblastoma, histologically defined | Medulloblastoma (all classes) | ||
| Other CNS embryonal tumors | Other CNS embryonal tumors | Atypical teratoid/rhabdoid tumor | Atypical teratoid/rhabdoid tumor (MYC, SHH, and TYR subtypes) | |
| CNS neuroblastoma, FOXR2-activated | CNS neuroblastoma, FOXR2-activated | |||
| CNS tumor with BCOR internal tandem duplication | CNS tumor with BCOR internal tandem duplication | |||
| Cribriform neuroepithelial tumor | Cribriform neuroepithelial tumor, SMARCB1-altered | |||
| Embryonal tumor with multilayered rosettes | Embryonal tumor with multilayered rosettes, C19MC-altered | |||
| Embryonal tumor with multilayered rosettes, non-C19MC-altered | ||||
| Pineal tumors | Pineal tumors | Pineal tumors | Pineocytoma | Pineocytoma |
| Pineal parenchymal tumor of intermediate differentiation | Pineal parenchymal tumor of intermediate differentiation, KBTBD4-altered, and subtypes A and B | |||
| Pineoblastoma | Pineoblastoma, (all classes) | |||
| Papillary tumor of the pineal region | Papillary tumor of the pineal region, subtypes A and B | |||
| Desmoplastic myxoid tumor of the pineal region, SMARCB1-mutant | Desmoplastic myxoid tumor of the pineal region, SMARCB1-mutant*** | |||
| Cranial and paraspinal nerve tumors | Cranial and paraspinal nerve tumors | Cranial and paraspinal nerve tumors | Schwannoma | Schwannoma |
| Neurofibroma | Neurofibroma/plexiform neurofibroma | |||
| Malignant melanotic nerve sheath tumor | Malignant melanotic nerve sheath tumor | |||
| Malignant peripheral nerve sheath tumor | Atypical malignant peripheral nerve sheath tumor | |||
| Malignant peripheral nerve sheath tumor | ||||
| Cauda equina neuroendocrine tumor [previously paraganglioma] | Cauda equina neuroendocrine tumor [paraganglioma], subtype non-CIMP | |||
| Meningioma | Meningioma | Meningioma | Meningioma | Meningioma (benign, intermediate, and malignant classes) |
| Clear cell meningioma | Meningioma, clear cell subtype, and SMARCE1-altered | |||
| Mesenchymal, non-meningothelial tumors involving the CNS | Soft tissue tumors | Fibroblastic and myofibroblastic tumors | Solitary fibrous tumor | Solitary fibrous tumor |
| Vascular tumors | Haemangioblastoma | Hemangioblastoma | ||
| Skeletal muscle tumors | Rhabdomyosarcoma | Rhabdomyosarcoma (all classes) | ||
| Tumors of uncertain differentiation | Intracranial mesenchymal tumor, FET::CREB fusion-positive | Intracranial mesenchymal tumor, FET::CREB fusion-positive*** | ||
| CIC-rearranged sarcoma | CIC-rearranged sarcoma | |||
| Primary intracranial sarcoma, DICER1-mutant | Primary intracranial sarcoma, DICER1-mutant | |||
| Ewing sarcoma | Ewing sarcoma | |||
| Chondro-osseous tumors | Chondrogenic tumors | Mesenchymal chondrosarcoma | Mesenchymal chondrosarcoma*** | |
| Chondrosarcoma | Chondrosarcoma*** | |||
| Notochordal tumors | Notochordal tumors | Chordoma | Chordoma | |
| Melanocytic tumors | Melanocytic tumors | Circumscribed meningeal melanocytic neoplasms | Primary CNS circumscribed melanocytic tumor | Melanocytoma**** |
| Haematolymphoid tumors involving the CNS | Lymphomas | CNS lymphomas | Diffuse large B-cell lymphoma of the CNS | Diffuse large B-cell lymphoma of the CNS |
| Histiocytic tumors | Histiocytic tumors | Langerhans cell histiocytosis | Langerhans cell histiocytosis | |
| Germ cell tumors | Germ cell tumors | Germ cell tumors | Germinoma | Germinoma, subtypes KIT mutant and KIT wildtype |
| Mature teratoma | Teratoma | |||
| Yolk sac tumor | Yolk sac tumor | |||
| Tumors of the sellar region | Tumors of the sellar region | Tumors of the sellar region | Adamantinomatous craniopharyngioma | Adamantinomatous craniopharyngioma |
| Papillary craniopharyngioma | Papillary craniopharyngioma | |||
| Pituicytoma, granular cell tumor of the sellar region, and spindle-cell oncocytoma | Pituicytoma, granular cell tumor of the sellar region, and spindle-cell oncocytoma | |||
| Pituitary adenoma/pituitary neuroendocrine tumor | Pituitary adenoma (all classes) | |||
| Pituitary blastoma | Pituitary blastoma*** | |||
| Metastases to the CNS | Metastases to the CNS | Metastases to the brain and spinal cord parenchyma/metastases to the meninges | Metastatic melanoma | Metastatic melanoma |
*For this comparison, the methylation classes listed in the Heidelberg version 12.8 (https://www.molecularneuropathology.org/mnp/classifiers/14) are used.
**The subependymoma methylation classes can include, in addition to CNS WHO grade 1 subependymoma, ependymal-like tumors that are not yet represented in the current CNS WHO classification.
***Desmoplastic myxoid tumor of the pineal region, SMARCB1-mutant, intracranial mesenchymal tumor, FET::CREB fusion-positive, mesenchymal chondrosarcoma, chondrosarcoma, and pituitary blastoma are classes in the NCI/Bethesda classifier but are not present in the current Heidelberg CNS tumor classifier.
****The melanocytoma methylation class includes Circumscribed meningeal melanocytic neoplasms of all grades, including meningeal melanoma.
Table 2.
Methylation Classes That Do Not Correspond to a WHO CNS5 Tumor Type
| Methylation class |
|---|
| Adult-type diffuse high-grade glioma, IDH-wildtype, subtypes B, E, and F |
| CNS embryonal tumor with BRD4:LEUTX fusion |
| CNS embryonal tumor with PLAG-family amplification |
| CNS schwannoma, VGLL-fused |
| CNS tumor with BCOR/BCORL1 fusion |
| Dural angioleiomyoma* |
| Glioneuronal tumor with ATRX alteration, kinase fusion, and anaplastic features |
| Glioneuronal tumor, subtype A |
| High-grade glioma with pleomorphic and pseudopapillary features (HPAP)* |
| Intraocular medulloepithelioma |
| Neuroblastoma, ALT/TERT TMM positive |
| Neuroblastoma, MYCN type |
| Neuroblastoma, TMM negative |
| Neuroepithelial tumor with PATZ1 fusion |
| Neuroepithelial tumor, PLAGL1-fused |
| Olfactory neuroblastoma, IDH-wildtype |
| Oligosarcoma, IDH-mutant** |
| Plasmacytoma of the CNS |
| Retinoblastoma |
| Retinoblastoma, subtype MYCN-altered |
| Sinonasal undifferentiated carcinoma, IDH2-mutant |
*Dural angioleiomyoma and high-grade glioma with pleomorphic and pseudopapillary features (HPAP) are classes in the NCI/Bethesda classifier but are not present in the current Heidelberg classifier.
**Oligosarcoma, IDH-mutant includes some neoplasms that do not harbor IDH mutations and/or 1p/19q-codeletion (see PMID 36520193 and PMID 34967922).
Table 3.
WHO CNS5 Tumor Types That Do Not Directly Correspond to Methylation Classes in the CNS Classifier
| CNS WHO tumor type |
|---|
| Diffuse low-grade glioma, MAPK pathway-altered |
| Multinodular and vacuolating neuronal tumor |
| Dysplastic cerebellar gangliocytoma (Lhermitte–Duclos disease) |
| CNS embryonal tumor NEC/NOS |
| Perineurioma |
| Hybrid nerve sheath tumors |
| Hemangiomas and vascular malformations |
| Rare haematolymphoid tumors involving the CNS* |
*Immunodeficiency-associated CNS lymphomas, lymphomatoid granulomatosis, intravascular large B-cell lymphoma, MALT lymphoma of the dura, other low-grade B-cell lymphomas of the CNS, anaplastic large cell lymphoma (ALK+/ALK−), T-cell and NK/T-cell lymphomas, Erdheim–Chester disease, Rosai–Dorfman disease, juvenile xanthogranuloma, histiocytic sarcoma.
Methylation Classifier Results Should Be Integrated With Appropriate Clinical, Radiological, Histological, and Genomic Information
For each tumor tested, the classifier typically assigns the neoplasm to a methylation class in the reference set that shows the most epigenetic similarity to the tumor sample being evaluated, as well as a calibrated score ranging from 0 (low-confidence) to 1 (high-confidence),1 providing the level of confidence for the tumor being placed in that class. The tumor is generally considered a “match” if the calibrated score is above a pre-determined threshold value. As with all diagnostic testing modalities, machine learning-based (supervised and unsupervised) classifier predictions from methylation profiling data in clinical diagnostics require critical review and integration with clinical, radiological, histological, and genomic data. A methylation profile does not replace a pathologist’s opinion; rather it is intended to add additional important data that helps shape their opinion. While specific confidence score cutoffs can be used as a threshold for designating the obtained result as “match” or “no-match” in clinical reporting, the scores that result from the classifier are a continuum that can be used to determine contribution to a definitive diagnosis. In this respect, subthreshold matches can nevertheless be contributory to a diagnosis, especially when ancillary data are supportive. An example is a low-confidence match to Glioblastoma IDH-wildtype in a low tumor purity sample from an older adult, in which ancillary data show a stereotypical +7/−10 chromosomal copy number signature as well as a TERT promoter mutation; in this situation, the methylation result can be seen as contributory, even in the face of a lower confidence score. Clustering and/or dimensionality reduction using t-distributed stochastic neighbor embedding (t-SNE) or uniform manifold approximation and projection for dimensionality reduction (UMAP) can also often be contributory,41 especially for tumors whose confidence scores do not reach the clinical threshold. A platform (UMAP or t-SNE) using large and well-annotated reference sets that allow embedding and visualization of the methylome profile of a tumor sample within a group can provide additional support for that class designation, even in the setting of a lower confidence score (eg, https://methylscape.ccr.cancer.gov, https://epidip.org). Class designation using such a visualization tool is not easily standardized and great caution is required when the appropriate diagnostic class is not included in the reference set, as diagnostic cases for which the appropriate class is lacking will often fall into one of the other available cases in the UMAP or t-SNE.
Methylation Class and Tumor Grading
The relationship of methylation class with WHO-defined tumor types that occur in multiple grades is complex. In some cases, a single methylation class accounts for multiple WHO grades. Examples include Pleomorphic xanthoastrocytoma and Solitary fibrous tumor (SFT), for which the current iteration of the Heidelberg classifier has a methylation class designation that correlates to the tumor type, but a relationship of methylation profile and tumor grade is not defined within each tumor type. Nonetheless, it remains possible that further work has the potential to elucidate prognostic methylation subgroups, as has been shown for SFT.42 In other instances, some relationships between methylation classes and tumor grades do exist; examples include IDH-mutant astrocytoma and meningioma. Classes corresponding to IDH-mutant astrocytoma include “Astrocytoma, IDH-mutant; high grade” and “Astrocytoma, IDH-mutant; lower grade.” While methylation class for IDH-mutant astrocytomas is not a grading tool per se, WHO grade 2 tumors tend to match the lower-grade class, while WHO grades 3 and 4 tumors more frequently match to the high-grade class, although these correlations are not absolute. A similar situation exists with meningiomas, which have Benign, Intermediate, and Malignant classes (3 classes exist for Benign and 2 for Intermediate). In such cases, the meningioma methylation class does not determine the WHO grade but may provide additional prognostic information helpful to estimate recurrence risk.43 A concurrent cIMPACT-NOW Working Group recently published recommendations on how molecular (including methylome profiling-based) risk parameters can be used for improved grading of meningiomas.10 Overall, the specific relationships of tumor grade with methylation class appear to be tumor type specific and elucidation of these relationships specifically in future classifier versions is recommended to maximize clarity.
Selection of Cases for Clinical Diagnostic DNA Methylation Profiling
A variety of approaches can be taken for case selection for diagnostic DNA methylation profiling. DNA methylation can be considered as a useful first-line molecular triage to decide what further molecular testing is needed or when the first-line of immunohistochemical stains is non-informative, particularly for samples with limited tissue.13 Experience indicates that while difficult-to-classify cases may have the highest benefit from this profiling, occasional cases can lead to unexpected findings on methylation profiling that prompt a revision of diagnostic possibilities.
One study of a series of consultation cases sent for DNA methylation profiling found that nearly 10% of cases (15/160) with a pre-methylation diagnosis of Glioblastoma, IDH-wildtype were given an alternative integrated diagnosis after DNA methylation-based classification.44
As an additional example type, it has been suggested that some methylation classes suggest a broader biology compared to a histopathology-based definition. Tumors matching to the pleomorphic xanthoastrocytoma (PXA) methylation class include both histologically defined PXAs and tumors which do not fit this histological definition. Conversely, some histologically defined PXAs matched to classes suggesting alternative tumor types, including Glioblastoma, IDH-wildtype, and Ganglioglioma.45
Tumors with histopathological characteristics of ependymoma, while largely matching to an ependymoma class, also can on occasion suggest alternative diagnoses, based on their methylation class results.46
An additional example type includes tumors that match to a methylation class that is unexpected compared to the anatomic location of the tumor. Tumors whose methylation class suggests a diagnosis of Papillary tumor of the pineal region have been described outside of the pineal region.47 Occasional cases of Supratentorial ependymoma, ZFTA fusion-positive are encountered in the posterior fossa,48 a point to be addressed separately by another cIMPACT-NOW Working Group. Tumors matching to a medulloblastoma class have been reported in the pineal region.49
DNA methylation-based classification can be specifically useful when the differential diagnosis includes a tumor type that can only be diagnosed by methylation profile, such as diffuse glioneuronal tumor with oligodendroglioma-like features and nuclear clusters and high-grade astrocytoma with piloid features.
Tumor type definition of medulloblastomas and ependymomas (eg, Posterior fossa group A [PFA] ependymoma and Posterior fossa group B [PFB] ependymoma) is efficiently performed based on methylation signatures.
Based on these considerations, cases that appear unusual show unexpected results on ancillary studies, or which cannot be precisely classified with “conventional” pathology diagnostics warrant the use of methylation profiling where possible. While an argument can be made for routine methylation profiling of all CNS tumors, this may not be practical in the current environment for many institutions, and decisions in routine practice need to be made in the context of test availability, resource-based considerations, and local expertise.
The classification of pediatric tumors, in general, appears to benefit from methylation profiling. For instance, one study selected tumors that were deemed diagnostically uncertain after 2 rounds of review by neuropathology experts. In this study, DNA methylation was considered contributory to a final diagnosis in 71% of these cases.50 A population-based study in the United Kingdom assessed the impact of DNA methylation profiling on 306 pediatric brain tumors, finding that the classifier contributed to the diagnosis in 35% of cases.51 An additional feature of this study was an examination of misleading results, in which 3 (1%) cases were determined to be initially misleading, but these tumors could be readily resolved with a re-review of associated clinical and ancillary data.51 Another study focused on tumors in adult patients, which included both diagnostically difficult cases, as well as tumors for which methylation-based subtyping was required (eg, medulloblastoma and ependymoma). In this series, only cases above the pre-specified 0.84 confidence score threshold were considered in the analysis. Among these cases, the diagnosis was changed in 45 (25%), refined in 86 (48%), and confirmed in 44 cases (25%).52 It is worth noting that these series were analyzed by highly experienced neuropathologists. The expectation would be that the impact might be even higher in regions with pathologists less experienced in CNS tumor diagnostics. Additional scenarios that are observed with some frequency are histologically low-grade diffuse gliomas in older adults, where methylation profiling of such cases can at times reveal features of Glioblastoma, IDH-wildtype, both by virtue of a methylation match to this family as well as the presence of characteristic DNA copy number changes (eg, +7/−10 changes and/or EGFR amplification). Conversely, occasional IDH-wildtype high-grade diffuse gliomas are encountered where Glioblastoma, IDH-wildtype is considered in the differential diagnosis, but where such accompanying genomic changes are not observed. Such cases can at times be resolved into alternative diagnoses (eg, diffuse pediatric-type high-grade glioma, H3-wildtype and IDH-wildtype or diffuse hemispheric glioma, and H3 G34-mutant).
Special cases exist in which the diagnosis may not be in question, but the methylation profiling data may provide additional information. As mentioned above, another use of methylation profiling is in the determination of prognostic subclass in meningiomas.43 Yet another additional use of the methylation array is in the determination of copy number status such as the status of 1p/19q in glial neoplasms for the diagnosis of Oligodendroglioma, IDH-mutant, and 1p/19q-codeleted.53 Additional examples exist, including the identification of MYCN amplification in subsets of pediatric-type high-grade gliomas and ependymomas.54–56 This technique can also be used as a single tool to obtain synchronously different molecular data as copy number variations (CNVs–deletions, amplifications), although it is noted that the resolution of CNVs from methylation data is not as high as it is from dedicated copy number platforms. Determination of MGMT promoter methylation status in Glioblastoma, IDH-wildtype,21 is important because the benefit from temozolomide is likely to be lower if their tumors can be shown to be unambiguously unmethylated at the MGMT promoter.57 Methylguanine methyltransferase promoter methylation status is also readily obtained from the methylation array data,21,58,59 which is particularly useful when tumoral material is limited.
While both methylation profiling and next-generation sequencing (NGS) provide valuable information, a blanket recommendation cannot be made in the choice of one over the other when tumor tissue is limiting, and decisions are best made on a case-by-case basis. DNA methylation profiling generally provides a wider range of possibilities for challenging cases, but NGS can be helpful to rule in or rule out specific diagnostically meaningful alterations. Given the challenges of DNA methylation profiling in a setting of low tumor purity, NGS may be more generally informative in such cases. In addition, precise delineation of low-grade glial/glioneuronal tumors can be a challenge for methylation-based classification, a consideration which may influence the choice of molecular platforms to provide helpful diagnostic information. While not all cases can be resolved, collectively, the evidence from multiple studies and institutions demonstrates the value and contribution of DNA methylation-based classification in many, but not all, CNS neoplasms encountered in daily practice, especially for cases that either require subtyping or cases in which the diagnosis cannot be readily resolved using other diagnostic modalities.
An additional consideration arises when the tumor with a high-confidence score matches to a methylation class of a tumor type whose definition requires the determination of a specific genomic alteration. A relevant example is a finding of a Supratentorial ependymoma, ZFTA fusion-positive class on methylation. While the WHO CNS5 criteria require the determination of the defining ZFTA fusion to make the diagnosis, there may be cases where testing for the defining fusion is not available or practical. In this situation, a final diagnosis of Supratentorial ependymoma, ZFTA fusion-positive is considered reasonable. Addition of a comment is advisable, indicating the particular circumstances of the case and notation that a determination of the defining genomic alteration is not possible, but that a diagnosis can nonetheless be rendered.
Use of Additional Data Layers for Diagnostics and Molecular Classification
Current methylation classifiers are based only on the methylation data per se, even though the methylation array data enable alternate outputs, including DNA copy number determination. Although the DNA copy number profile can be helpful diagnostically, copy number data are not formally included in currently available CNS tumor classifiers. DNA copy number profiles derived from DNA methylation profiling data can be used to orthogonally support a suspected diagnosis, which can be helpful in cases of low-confidence-score matches. High-level amplifications for specific genes/small genomic regions can also be helpful for individual cases (eg, EGFR amplifications in cases of Glioblastoma, IDH-wildtype, and C19MC amplification in Embryonal tumor with multilayered rosettes, C19MC-altered). An additional use of the copy number data is the finding of breakpoints within specific chromosomal regions that suggest the presence of diagnostically helpful fusions, such as KIAA1549::BRAF.60 Some limitations of DNA copy number data derived from array-based methylation profiling include the inability to detect copy number-neutral loss of heterozygosity, as well as its inability to determine ploidy levels. Still, the determination of copy number from DNA methylation data is of value in many cases, and improvements to the originally described method (“conumee”) are in development.61 Multi-omic approaches, such as leveraging methylation data with next-generation sequencing in formal classification algorithms, have been suggested to improve the ability to classify CNS tumors,17 but specific recommendations cannot be made at this time.
DNA Methylation Detection Platforms
Currently, the methylation array is the most widely used platform for CNS tumors, and methylation array-derived data serve as the basis for all commonly used classifier reference sets. More recently alternative platforms have emerged, including the use of Nanopore technology41,62–64 for the detection of diagnostic DNA methylation profiles, which has the potential advantage of faster turnaround time. Next-generation sequencing (eg, following bisulfite or enzymatic conversion) may also be used in the future for DNA methylation profiling41,63 emphasizing that technological advances may allow for alternative platforms to be used. Existing experience, including current classifier reference sets, can potentially be used for training these newer approaches, saving time and resources when new platforms are being developed and evaluated.
DNA Methylation Profiling Results and Diagnostic Pathology Reports
Reporting methylome results in pathology reports has recently been addressed,64 and key elements can be emphasized here. Briefly, a diagnostic pathology report that includes methylome profiling analysis should contain information on the specific classifier version(s) used, the classifier match, estimated tumor cell content, calibrated confidence score, and range or threshold for positive, indeterminate, and negative results. Where applicable, reporting on both the diagnostic category (eg, the confidence score of a match to the methylation family “Glioblastoma, IDH-wildtype”) and any additional subclass determination (eg, the confidence score, in this hypothetical case, of a match to the “Glioblastoma RTK2 subclass”) should be included.
A “layered” reporting format has been suggested for CNS tumor diagnostic reports that incorporate both histologic and molecular information.65,66 Such a format promotes the communication of relevant data and emphasizes the integration of relevant information. Because methylation profiling provides information distinct from other data sources, the inclusion of methylation classifier results (class match and confidence score) is recommended in the molecular line of the layered report format. Suggested formats are shown with examples of a case with a high-confidence match to family and class (Figure 1); a case that showed no match on methylation-based classification but sufficient alternative/orthogonal data were available to render a definitive diagnosis (Figure 2); a case that showed a high-confidence match to a methylation family, but not to a specific methylation class, in which a descriptive (not elsewhere classified [NEC]) diagnosis is warranted (Figure 3); and a case showing a high-confidence match to a class that does not correspond to a current CNS WHO tumor type (Figure 4). Finally, an example of a case that leverages the recent cIMPACT-NOW recommendation on meningioma10 is shown (Figure 5). For further clarifications within a pathology report and/or the accompanying genome-wide methylation report, additional information is suggested, including the specifics of the methylation classifier used, and the matches and confidence scores within the hierarchy of the methylation classifier (eg, Superfamily, Family, Class, and Subclass, example shown in Figure 6A). Additional technical results that are recommended for inclusion include the Sample type (eg, FFPE/frozen), tumor cell fraction/tumor purity, amount of DNA input, quality of bisulfite conversion, reference set used, classifier/algorithm/version used (example shown in Figure 6B).
Figure 1.
Layered diagnosis example: high-confidence match to family and class.
Figure 2.
Layered diagnosis example: no match on methylation-based classification, with sufficient orthogonal data for definitive diagnosis.
Figure 3.
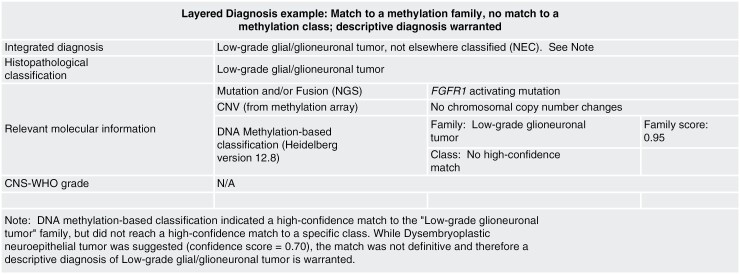
Layered diagnosis example: match to a methylation family, no match to a methylation class; descriptive diagnosis warranted.
Figure 4.
Layered diagnosis example: high-confidence match to a class that does not correspond to a current WHO tumor type.
Figure 5.
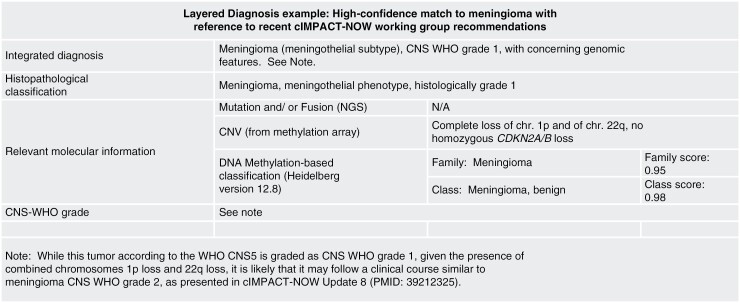
Layered diagnosis example: high-confidence match to meningioma with reference to recent cIMPACT-NOW Working Group recommendations.
Figure 6.
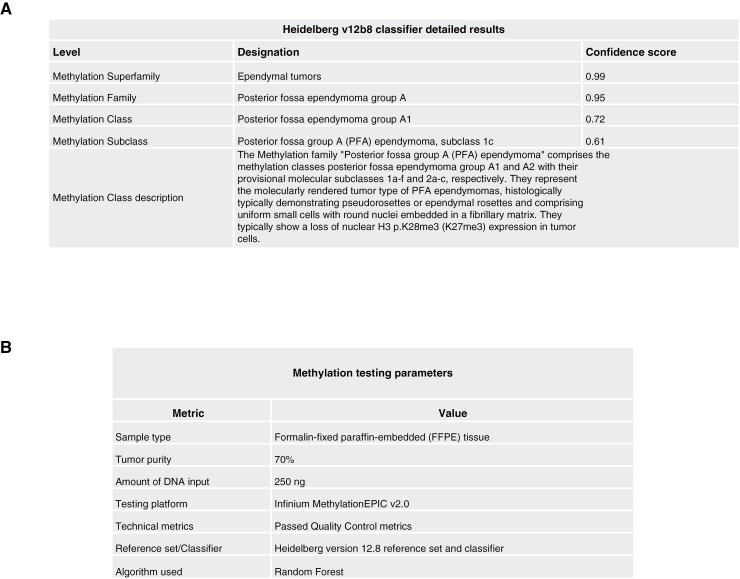
Additional reporting recommendations: (A) classifier detailed results; (B) methylation testing parameters.
Given the importance of integration of the methylation classifier result within the context of the entire clinical and histomolecular aspects of the case, interpretation of methylation profiling data is optimal when a neuropathologist experienced in molecular diagnostics is directly involved. One salient reason for this is that misleading methylation classifier results with a high calibrated score can occasionally be obtained, highlighting the need for an interdisciplinary tumor board interpretation by content experts who are aware of specific attributes and limitations of the classifier under use. As discussed above, knowledge of such examples from a specific classifier would clearly be of benefit in coming to an accurate diagnosis in specific cases.
Biologic and Clinical Significance of Methylation Subclasses
For several CNS tumor types, multiple methylation subclasses exist. For example, 5 classes exist for Medulloblastoma, Sonic Hedgehog (SHH)-activated, encompassing both TP53-wildtype and TP53-mutant CNS WHO types. Medulloblastoma groups 3 and 4 comprise a total of 8 subclasses, and enrichment for specific genomic aberrations is described in these subclasses. As examples, of the groups 3 and 4 medulloblastoma, methylation subclass 1 generally shows a balanced CNV profile but is enriched for OTX2 amplifications, while subclass 2 is enriched for MYC amplification and subclass 5 is enriched for both MYC and MYCN amplifications.67 In SHH medulloblastomas, subclass 1 is highly enriched for TP53 mutations.68 As such, methylation subclasses can inform as to the likelihood of corresponding genomic aberrations in medulloblastoma. Most Glioblastoma, IDH-wildtype fall into one of 3 subclasses, mesenchymal, RTK1, and RTK2. Mesenchymal glioblastomas have been shown to have increased myeloid cell compartment, including macrophages,69 and there have been suggestions of survival differences among these methylation-defined subclasses.70 The RTK2 subclass of Glioblastoma IDH-wildtype is described as showing a high frequency of EGFR amplification in the Heidelberg classifier (https://www.molecularneuropathology.org/mnp/classifiers/14). While direct clinical translation of these glioblastoma methylation subclasses has yet to be demonstrated, they provide insights into biology that may suggest directions for future therapeutic approaches. As a practical matter, methylation profiling of Glioblastoma, IDH-wildtype can on occasion reveal a high-confidence match to the Glioblastoma IDH-wildtype methylation family, but the scores can split among the glioblastoma methylation subclasses. In this context, a low score for a specific glioblastoma class would not affect the integrated diagnosis. A hypothetical example of such a case, in the layered diagnostic format, is shown in Figure 7. Most cases of the diffuse pediatric-type high-grade glioma, H3 wildtype, and IDH-wildtype fall into 3 broad methylation-defined categories: diffuse pediatric-type high-grade glioma, MYCN subtype; diffuse pediatric-type high-grade glioma, RTK1 subtype; or diffuse pediatric-type high-grade glioma, RTK2 subtype.71 The RTK1 subtype is then divided into subgroups A, B, and C, and the RTK2 subtype is divided into A and B subtypes. As with the glioblastoma classes, enrichments of specific biological events correlate with these classes. For example, the diffuse pediatric-type high-grade glioma, MYCN subtype is associated with MYCN amplification,71 and the diffuse pediatric-type high-grade glioma, RTK1 subtype is associated with both prior cranial radiation as well as patients with constitutional mismatch repair deficiency syndrome or Lynch syndrome (https://www.molecularneuropathology.org/mnp/classifiers/14). It has been noted that patients whose tumors that fall into these methylation classes are not restricted to children and adolescents,72 and can (not infrequently) occur in adults, when it is important to distinguish them from glioblastoma IDH-wildtype based on genomic aberrations and biological characteristics. Currently, the significance of the clinical distinction between diffuse pediatric-type high-grade glioma, H3 wildtype, and IDH-wildtype versus Glioblastoma IDH-wildtype in adult patients is not clear, although biological differences are identified with respect to epigenomic and genomic characteristics. Overall, methylation profiling is useful to elucidate the diagnosis of Diffuse pediatric-type high-grade glioma, H3-wildtype, and IDH-wildtype in specific cases, but future work is required to establish the clinical relevance, if any, of the subclasses in this methylation family.
Figure 7.
Layered diagnosis example: high-confidence match to the glioblastoma IDH-wildtype methylation family, but low-confidence matches to glioblastoma classes.
DNA Methylation-Based Classifiers for Tumors Outside the CNS
The utility of CNS DNA methylation classification has prompted the exploration of this platform for additional tumor types and organ systems. A comprehensive review of these is beyond the scope of the current work, but awareness of their existence is important. Classifiers for sarcomas22,73 and sinonasal tumors74 have been reported. Additional classifiers have been proposed41,75 and are in various stages of development. Their existence may be useful in the diagnostic process of CNS neoplasms as such lesions are occasionally encountered in neuropathology practice. For example, the class “Malignant peripheral nerve sheath tumor” is included in both the CNS and sarcoma classifiers, and similarly “Olfactory neuroblastoma” is included in both the CNS and sinonasal classifiers. In these instances, dual classifiers can potentially be leveraged in a complementary fashion to evaluate such diagnoses from methylation data. Since some classes overlap among different classifiers, the same data (methylation IDAT files) can be used for multiple applications, adding efficiencies and lowering costs. Additionally, secondary involvement of a tumor to the CNS (direct extension, metastasis) can potentially be interrogated productively using extra-CNS classifiers that may be developed in the future.
Methylation Classifier Limitations
Optimal interpretation of methylation classifier results for tumor diagnostics is ideally reviewed by individuals with specific expertise in CNS tumor diagnostics, as it requires content expertise as well as awareness of specific classifier characteristics. Central nervous system tumors are highly diverse, and a precise diagnosis can require knowledge of the relevant literature and clinical, histopathologic, genomic, and epigenomics features of the tumor type at hand. This point is even more relevant with uncommon CNS tumors. Here we highlight some examples of limitations of methylation-based classification that have been commonly encountered in current practice:
- A low content of neoplastic cells in a sample can mask the neoplastic signal in the methylation profile. The methylation profile of a tumor sample is a composite of the cell types included, which inevitably includes nonneoplastic cells. Samples with relatively high content of nonneoplastic cells (eg, preexistent glial cells, neurons, inflammatory/reactive cells) can lead to either low-confidence matches to tumor types or matches to control/reactive CNS tissue. In this context, one must review samples for appropriate neoplastic content in selecting tissues for methylation profiling, and interpretation of methylation profiling results must occur with awareness of overall neoplastic content in the specimen. Concurrent evaluation of the amplitudes of copy number-alterations, if applicable, and/or sequence variant allele frequency may aid in the interpretation of tumor cell content. The Heidelberg classifier has some inbuilt quality measures in this regard, but some cases can still remain ambiguous. It is possible that the addition of deconvolution algorithms will help to resolve such cases. On a practical level, we note that some cases exist that match to the “Control tissue, reactive tumor microenvironment” or to the “Inflammatory microenvironment” classes which can be demonstrated by other means to represent neoplasms of low tumor purity. As such, cases that match to these classes are often not contributory to diagnosis. Of note, class descriptions are available for each class in the Heidelberg classifier (https://www.molecularneuropathology.org/mnp/classifiers) which can clarify the characteristics and common features of each class. For the “Control tissue, reactive tumor microenvironment” and the “Inflammatory microenvironment” classes, the descriptions of each of these 2 classes clarify this point, which highlights the need to provide such descriptions in the pathology report (as noted below) for clarity of interpretation.
- A substantial proportion of cases that undergo DNA methylation profiling do not result in a high-confidence match. Most studies report that a subset of cases do not match with a confidence score at a pre-specified threshold range.1,44,50,76 Reasons for “no-match” cases vary, ranging from issues with pre-analytical variables, assay quality metrics, and low tumor purity. An additional important consideration in no match cases is whether the tumor type in question is represented in the classifier reference set since the classifier cannot determine the tumor type otherwise. Experience indicates that CNS tumors arising in the setting of predisposition syndromes (eg, Li-Fraumeni syndrome, neurofibromatosis type 1) more often do not match to specific classes with high-confidence scores.23,51 Still the fact that the classifier does not force each sample into a class is beneficial, since a low classifier score immediately raises a concern regarding the precise diagnosis.
- Glioblastoma, matching to Ganglioglioma. Experience of some of the authors of this work indicates that occasional cases of Glioblastoma IDH-wildtype, especially in the setting of low tumor purity, can match (often, but not always, with low-confidence score) to Ganglioglioma in the version 12 iterations of the Heidelberg classifier. Such cases can usually be resolved using, for example, orthogonal data as outlined above and require awareness to ensure diagnostic accuracy.
- SMARCB1- and SMARCA4-deficient tumors (of various types) matching to Atypical teratoid/rhabdoid tumor (ATRT). The methylation profile of a tumor cell is a composite of cell-of-origin as well as epigenetic changes occurring as a result to specific genomic alterations.77 Loss of SMARCB1 (or SMARCA4) gene function appears to result in a profound and stereotypic epigenetic change such that non-ATRT tumor types usually match to an ATRT class (often MYC subtype) in the CNS tumor classifier. Anecdotal cases of SMARCB1-altered tumor types originating outside the CNS (eg, metastatic malignant rhabdoid tumors of the kidney) that match to AT/RT are identified. Caution is therefore warranted in the setting of matches or high scores for ATRT depending on the histopathological setting.
- Classifier matches to low-grade glial/glioneuronal tumors can be ambiguous. The methylation profiles of several WHO-defined low-grade glial/glioneuronal tumors appear to be quite similar to each other, making precise distinction difficult. An example is the difficulty in distinguishing Ganglioglioma from Polymorphous low-grade neuroepithelial tumor of the young in the methylation classifier. An additional ambiguity is the finding of frequent matches to dysembryoplastic neuroepithelial tumor (DNT) in cases that lack typical histologic features of this diagnosis. Whether and how this finding contributes to our understanding of DNT awaits further study. Detailed studies on these tumors indicated that diagnostic criteria of low-grade glial/glioneuronal tumors does not adequately reflect their tumor biology, and in particular the histopathologic characteristics of this group of tumors is only partially reflective of their underlying molecular profiles.78,79 As a solution, it has been suggested that integration of methylation and radiological data can improve diagnostic precision for these tumors.79
- Reproducible misclassification of Intracranial mesenchymal tumor, FET::CREB fusion-positive as meningioma. The commonly used Heidelberg version 12 classifier does not include Intracranial mesenchymal tumor, FET::CREB Fusion-Positive (ICMT-FET-CREB) as a defined class, and experience indicates that most of these tumors match on the version 12 classifier to meningioma, sometimes with high-confidence scores. Future classifier iterations that include ICMT-FET-CREB as a distinct class with well-curated reference cases may serve to resolve this issue, but in the interim genomic testing for a fusion involving genes in the FET and CREB families will provide a definitive diagnosis.
Overall, therefore, while DNA methylation-based classification has many attributes and can be instrumental in specific cases to reach a definitive diagnosis, awareness of its limitations is important in assessing its performance in individual cases. It is also prudent to comment on logistical challenges, which include the time and costs required to perform the assay, as well as regulatory issues that may be operative in relation to the launch of a clinically approved test. On that point, progress is evident, and as one example, in the United States, a Current Procedural Terminology (CPT) code has been made available for the Epignostix CNS Tumor Methylation Classifier from Heidelberg Epignostix GmbH, which is an important step towards broader adoption of such a test in the United States. A CPT code designation for a clinical test in the United States is a step that can lead to health insurance reimbursement and potential economic viability for institutions conducting the test.
Classifier Updates and Use
While technical aspects of test validation are outside the scope of this manuscript, a brief comment is in order. Minimum criteria have been proposed80 and include determination of assay sensitivity and specificity, establishment of minimum sample criteria, as well as implementation of quality control metrics. Challenges to clinical testing include tumor purity estimation, availability of appropriate reference set data, availability of and access to specialized bioinformatics resources, and changes that occur with new versions of the array.
The initial Heidelberg classifier described 91 classes (82 of these concerning neoplasms and nine concerning nonneoplastic/reactive CNS tissue) in a “version 11” classifier.1 Subsequently, several iterations of a “version 12” classifier were developed, with an increased number of classes. Such classifiers are freely available for use (https://www.molecularneuropathology.org/mnp/) and their existence and availability have enabled the adoption of methylation profiling at a number of centers. Concurrently, several centers have developed in-house classifiers, which are undergoing evaluation for diagnostics, and such classifiers may also be subject to broader use (eg, https://methylscape.ccr.cancer.gov, https://epidip.org41). Differences in existing classifiers include alternative machine learning algorithms, differences in specific samples included in reference sets, as well as differences in specific class types and designations and the number of cases that make up the reference sets. However, the original Heidelberg classifiers are currently the most widely known and used in the community. It is expected that the choice of classifiers will depend on the specific needs of individual institutions as well as specific applications. As the use of methylation profiling broadens and new classes are proposed, it is expected that classifiers will continue to evolve, with the refinement of classes and designations. The field will thus benefit from consensus and harmonization of agreed-upon methylation classes and class designations. Alongside the need for harmonization is the knowledge that no single classifier is likely to be perfect, and each will have its strengths and limitations. In this context, a case can be made for the use of multiple, complementary classifiers based, for example, on nonoverlapping reference sets and complementary machine learning algorithms. These points are raised for discussion and specific recommendations on the use of specific classifiers, either singularly or as an ensemble, cannot be made at this time. Nonetheless, it is important to use different pieces of information (eg, histology, methylation class, NGS, etc.) to support a diagnosis. In this context, a case where multiple classifiers give the same answer could be considered “higher” confidence. Relevant to this point is the potential for retroactive identification of classifier matches as new classes are defined. In other words, a specific case, while not yielding a match to a prior classifier version, might represent a newly identified class in a future classifier version. In the same spirit, DNA methylation profiling would be suitable for retrospective or prospective clinical trials to ensure reproducibility of diagnosis, but also to find prognostic or predictive markers.
The utility and accuracy of DNA methylation-based classification as a diagnostic tool are highly dependent on a comprehensive and well-annotated reference set that serves as the basis for the classifier. There is a need for the inclusion of all recognized classes in a classifier, based on the principle that classes can be detected only if they are included in the reference set and are included in the classifier. This highlights the benefit of comprehensive “data warehouses” that collate very large numbers of samples, including collections of rare classes that cannot practically be obtained in a single institution. Given these considerations, evaluation of classifiers, and criteria for their use should be prioritized based on the quality of the reference set. The Heidelberg classifiers, in particular, have leveraged this principle to positive effect, by virtue of the inclusion of data obtained from multiple institutions worldwide. In this context, data sharing initiatives that promote the collection and annotation of large datasets are likely to result in further improvements in methylation-based classification. Regulatory policies of specific laboratories or countries may require the availability of both a specific algorithm and the primary reference set data, a limitation that may exist in some situations,81 which further illustrates the advantages of data sharing to promote the availability and expansion of this important diagnostic modality.
Conclusions
DNA methylation profiling represents an important and useful tool in the armamentarium of CNS tumor diagnostics. Appropriate application of this platform requires knowledge of its strengths and limitations, as well as the need to integrate the profiling results within the overall context of a specific case. While recommendations of the use of specific classifiers or inclusion of specific classes are not within the scope of this cIMPACT Working Group, it is evident that harmonization and sharing of classifier reference sets, along with a collaborative process that allows for consensus recommendations and periodic updates is warranted to maximize the utility of this tool to promote best practices for CNS tumor diagnostics.
Summary of Recommendations
DNA methylation profiling of CNS tumors should be considered whenever it may help resolve a differential diagnosis. Prioritization is recommended for cases where the diagnosis is otherwise challenging, where there is a discrepancy among the findings of a case, where further molecular subtyping is needed to come to a diagnosis, or where DNA methylation-based classification is the only method to define a tumor type.
DNA methylation profiling is included in essential or desirable criteria for the diagnosis of many CNS WHO tumor types, and therefore awareness of both attributes and limitations of this platform as a diagnostic tool is important for pathologists and clinicians involved in the care of patients with CNS tumors.
As with any diagnostic modality (eg, histologic interpretation, IHC, and genomic findings) DNA Methylation classifier results should be interpreted in the context of all appropriate information available for the case. A DNA methylation classifier result that is unexpected or that does not fit within the context of these additional data requires expert, sometimes multidisciplinary review to determine an appropriate diagnosis.
DNA methylation classifiers may exhibit limitations in the resolution of specific methylation classes, as well as reveal findings that are unexpected compared to prior knowledge. Experience with the classifier and awareness of these limitations and attributes are therefore critical for the appropriate use of DNA methylation data in clinical diagnostics. Review of DNA methylation classifier results by a (neuro)pathologist who can interpret and integrate within the context of the case as a whole is therefore recommended as good clinical practice.
Current classifiers utilize all 3 BeadChip versions of the array, and the recent versions developed were not fully compatible with the original (“450k”) version. As new versions of the methylation array are formulated, ensuring that these new versions are fully backward-compatible is highly recommended, to provide maximal probe overlap across versions as well as to eliminate the need to re-develop a classifier with each array version.
Inclusion of DNA methylation results into a layered diagnostic format is recommended, with the inclusion of specifics as to the classifier used as well as the family and class match, including the confidence scores(s). The inclusion of additional data on quality control and the full output of the classifier results in the pathology report is also recommended.
As more cases are profiled by DNA methylation, classifiers will be improved by the inclusion of additional samples of rare tumor types into the reference set. This will also provide the opportunity for the discovery of new methylation classes. Data sharing to promote the development of comprehensive datasets will thus be of benefit to the field.
Acknowledgments
This paper has been reviewed by the Steering Committee and Clinical Advisory Panel of cIMPACT-NOW and by the International Society of Neuropathology Executive. The full cIMPACT Steering Committee comprises Drs. Kenneth Aldape, Dan Brat, David Capper, Andreas von Deimling, Dominique Figarella-Branger, Cynthia Hawkins, Thomas Jacques, Takashi Komori, Arie Perry, Guido Reifenberger, Brent Orr, Felix Sahm, Chitra Sarkar, Pascale Varlet, and Pieter Wesseling, with David Louis as cIMPACT-NOW advisor. The cIMPACT clinical advisory panel comprises Drs. Martin van den Bent, Mark Gilbert, Sabine Mueller, Stefan Pfister, Uri Tabori, and Michael Weller. We thank P.J. Cimino for helpful suggestions on the manuscript.
Contributor Information
Kenneth Aldape, Laboratory of Pathology, Center for Cancer Research, National Cancer Institute, Bethesda, Maryland, USA.
David Capper, German Cancer Consortium (DKTK), Partner Site Berlin, German Cancer Research Center (DKFZ), Heidelberg, Germany; Department of Neuropathology, Charité – Universitätsmedizin Berlin, corporate member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Germany.
Andreas von Deimling, Clinical Cooperation Unit Neuropathology, German Consortium for Translational Cancer Research (DKTK), Deutsches Krebsforschungszentrum (DKFZ), Heidelberg, Germany; Department of Neuropathology, University Hospital Heidelberg, Heidelberg, Germany.
Caterina Giannini, Department of Biomedical and Neuromotor Sciences (DIBINEM), Alma Mater Studiorum, Bologna, Italy; Department of Pathology, Mayo Clinic, Rochester, Minnesota, USA.
Mark R Gilbert, Neuro-Oncology Branch, Center for Cancer Research, National Cancer Institute, Bethesda, Maryland, USA.
Cynthia Hawkins, Department of Paediatric Laboratory Medicine, The Hospital for Sick Children, University of Toronto, Toronto, Ontario, Canada.
Jürgen Hench, Institut für Medizinische Genetik und Pathologie, Universitätsspital Basel, Basel, Switzerland.
Thomas S Jacques, Department of Histopathology, Great Ormond Street Hospital for Children, London, UK; Paediatric Neuropathology, University College London, UCL GOS Institute of Child Health, London, UK.
David Jones, Division of Pediatric Glioma Research, German Cancer Research Center (DKFZ) and German Cancer Consortium (DKTK), Heidelberg, Germany.
David N Louis, Department of Pathology, Massachusetts General Hospital, Brigham and Women’s Hospital, Harvard Medical School, Boston Massachusetts, USA.
Sabine Mueller, Department of Pediatric, University of Zurich, Zürich, Switzerland; Department of Neurology, Neurosurgery, and Pediatrics, University of California San Francisco, San Francisco, California, USA.
Brent A Orr, Department of Pathology, St. Jude Children’s Research Hospital, Memphis, Tennessee, USA.
MacLean Nasrallah, Division of Neuropathology, Department of Pathology and Laboratory Medicine, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, Pennsylvania, USA.
Stefan M Pfister, Department of Pediatric Hematology and Oncology, Heidelberg University Hospital and National Center for Tumor Diseases (NCT), Heidelberg, Germany; Hopp Children´s Cancer Center Heidelberg (KiTZ), Division of Pediatric Neurooncology, German Cancer Research Center (DKFZ) and German Cancer Consortium (DKTK), Heidelberg, Germany.
Felix Sahm, Clinical Cooperation Unit Neuropathology, German Consortium for Translational Cancer Research (DKTK), Deutsches Krebsforschungszentrum (DKFZ), Heidelberg, Germany; Department of Neuropathology, University Hospital Heidelberg, Heidelberg, Germany.
Matija Snuderl, Department of Pathology, New York University Langone Health and Grossman School of Medicine, New York, New York, USA.
David Solomon, Department of Pathology, University of California San Francisco, San Francisco, California, USA.
Pascale Varlet, Department of Neuropathology, GHU Paris - Psychiatry and Neuroscience, Sainte-Anne Hospital, Paris, France.
Pieter Wesseling, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands; Department of Pathology, Amsterdam University Medical Centers/VU University, Amsterdam, The Netherlands.
Funding
Funding for this work was provided to K.A. by the support from the intramural program of the National Cancer Institute at the National Institutes of Health. T.S.J. is grateful for funding from the Brain Tumour Charity (including the EVEREST Centre [GN-000707]). Olivia Hodson Cancer Fund, Cancer Research UK, and the National Institute of Health Research. All research at Great Ormond Street Hospital NHS Foundation Trust and UCL Great Ormond Street Institute of Child Health is made possible by the NIHR Great Ormond Street Hospital Biomedical Research Centre. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR, or the Department of Health.
Conflict of interest statement
A.D., D.C., F.S., M.S., and S.M.P. are co-founders and shareholders of Heidelberg Epignostix GmbH.
Authorship statement
All authors participated in consensus discussions, contributed to writing, and approved the manuscript.
Data availability
Not applicable.
References
- 1. Capper D, Jones DTW, Sill M, et al. DNA methylation-based classification of central nervous system tumours. Nature. 2018;555(7697):469–474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. WHO Classification of Tumours Ed. Board. World Health Organization Classification of Tumours of the Central Nervous System. 5th ed.Lyon: International Agency for Research on Cancer; 2021. [Google Scholar]
- 3. Louis DN, Wesseling P, Paulus W, et al. cIMPACT-NOW update 1: Not Otherwise Specified (NOS) and Not Elsewhere Classified (NEC). Acta Neuropathol. 2018;135(3):481–484. [DOI] [PubMed] [Google Scholar]
- 4. Louis DN, Giannini C, Capper D, et al. cIMPACT-NOW update 2: diagnostic clarifications for diffuse midline glioma, H3 K27M-mutant and diffuse astrocytoma/anaplastic astrocytoma, IDH-mutant. Acta Neuropathol. 2018;135(4):639–642. [DOI] [PubMed] [Google Scholar]
- 5. Brat DJ, Aldape K, Colman H, et al. cIMPACT-NOW update 3: recommended diagnostic criteria for “Diffuse astrocytic glioma, IDH-wildtype, with molecular features of glioblastoma, WHO grade IV”. Acta Neuropathol. 2018;136(5):805–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ellison DW, Hawkins C, Jones DTW, et al. cIMPACT-NOW update 4: diffuse gliomas characterized by MYB, MYBL1, or FGFR1 alterations or BRAF(V600E) mutation. Acta Neuropathol. 2019;137(4):683–687. [DOI] [PubMed] [Google Scholar]
- 7. Brat DJ, Aldape K, Colman H, et al. cIMPACT-NOW update 5: recommended grading criteria and terminologies for IDH-mutant astrocytomas. Acta Neuropathol. 2020;139(3):603–608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Louis DN, Wesseling P, Aldape K, et al. cIMPACT-NOW update 6: new entity and diagnostic principle recommendations of the cIMPACT-Utrecht meeting on future CNS tumor classification and grading. Brain Pathol. 2020;30(4):844–856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ellison DW, Aldape KD, Capper D, et al. cIMPACT-NOW update 7: advancing the molecular classification of ependymal tumors. Brain Pathol. 2020;30(5):863–866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Sahm F, Aldape KD, Brastianos PK, et al. cIMPACT-NOW Update 8: clarifications on molecular risk parameters and recommendations for WHO grading of meningiomas. Neuro-Oncol. 2024:noae170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Santana-Santos L, Kam KL, Dittmann D, et al. Validation of whole genome methylation profiling classifier for central nervous system tumors. J Mol Diagn. 2022;24(8):924–934. [DOI] [PubMed] [Google Scholar]
- 12. Tran QT, Alom MZ, Orr BA.. Comprehensive study of semi-supervised learning for DNA methylation-based supervised classification of central nervous system tumors. BMC Bioinf. 2022;23(1):223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Galbraith K, Vasudevaraja V, Serrano J, et al. Clinical utility of whole-genome DNA methylation profiling as a primary molecular diagnostic assay for central nervous system tumors-A prospective study and guidelines for clinical testing. Neurooncol Adv. 2023;5(1):vdad076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Halliday GC, Junckerstorff RC, Bentel JM, et al. The case for DNA methylation based molecular profiling to improve diagnostic accuracy for central nervous system embryonal tumors (not otherwise specified) in adults. J Clin Neurosci. 2018;47:163–167. [DOI] [PubMed] [Google Scholar]
- 15. Tam OCH, Ho RSL, Chan S, et al. Genome-Wide DNA methylation profiling as frontline diagnostics for central nervous system embryonal tumors in Hong Kong. Cancers (Basel). 2023;15(19):4880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. White CL, Kinross KM, Moore MK, et al. Implementation of DNA methylation array profiling in pediatric central nervous system tumors: the AIM BRAIN project: an Australian and New Zealand Children’s Haematology/Oncology Group Study. J Mol Diagn. 2023;25(10):709–728. [DOI] [PubMed] [Google Scholar]
- 17. Sturm D, Capper D, Andreiuolo F, et al. Multiomic neuropathology improves diagnostic accuracy in pediatric neuro-oncology. Nat Med. 2023;29(4):917–926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Reinhardt A, Stichel D, Schrimpf D, et al. Anaplastic astrocytoma with piloid features, a novel molecular class of IDH wildtype glioma with recurrent MAPK pathway, CDKN2A/B and ATRX alterations. Acta Neuropathol. 2018;136(2):273–291. [DOI] [PubMed] [Google Scholar]
- 19. Schwalbe EC, Williamson D, Lindsey JC, et al. DNA methylation profiling of medulloblastoma allows robust subclassification and improved outcome prediction using formalin-fixed biopsies. Acta Neuropathol. 2013;125(3):359–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Pajtler KW, Witt H, Sill M, et al. Molecular classification of ependymal tumors across all CNS compartments, histopathological grades, and age groups. Cancer Cell. 2015;27(5):728–743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Bady P, Sciuscio D, Diserens AC, et al. MGMT methylation analysis of glioblastoma on the Infinium methylation BeadChip identifies two distinct CpG regions associated with gene silencing and outcome, yielding a prediction model for comparisons across datasets, tumor grades, and CIMP-status. Acta Neuropathol. 2012;124(4):547–560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Koelsche C, Schrimpf D, Stichel D, et al. Sarcoma classification by DNA methylation profiling. Nat Commun. 2021;12(1):498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Lucas CG, Sloan EA, Gupta R, et al. Multiplatform molecular analyses refine classification of gliomas arising in patients with neurofibromatosis type 1. Acta Neuropathol. 2022;144(4):747–765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Muench A, Teichmann D, Spille D, et al. A novel type of IDH-wildtype glioma characterized by gliomatosis cerebri-like growth pattern, TERT promoter mutation, and distinct epigenetic profile. Am J Surg Pathol. 2023;47(12):1364–1375. [DOI] [PubMed] [Google Scholar]
- 25. Bogumil H, Sill M, Schrimpf D, et al. Glioneuronal tumor with ATRX alteration, kinase fusion and anaplastic features (GTAKA): a molecularly distinct brain tumor type with recurrent NTRK gene fusions. Acta Neuropathol. 2023;145(5):667–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Pratt D, Abdullaev Z, Papanicolau-Sengos A, et al. High-grade glioma with pleomorphic and pseudopapillary features (HPAP): a proposed type of circumscribed glioma in adults harboring frequent TP53 mutations and recurrent monosomy 13. Acta Neuropathol. 2022;143(3):403–414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Metais A, Tauziede-Espariat A, Garcia J, et al. ; Biopathology RENOCLIP-LOC network. Clinico-pathological and epigenetic heterogeneity of diffuse gliomas with FGFR3::TACC3 fusion. Acta Neuropathol Commun. 2023;11(1):14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Mata DA, Benhamida JK, Lin AL, et al. Genetic and epigenetic landscape of IDH-wildtype glioblastomas with FGFR3-TACC3 fusions. Acta Neuropathol Commun. 2020;8(1):186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wu Z, Lopes Abath Neto O, Bale TA, et al. DNA methylation analysis of glioblastomas harboring FGFR3-TACC3 fusions identifies a methylation subclass with better patient survival. Acta Neuropathol. 2022;144(1):155–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Sievers P, Sill M, Schrimpf D, et al. Epigenetic profiling reveals a subset of pediatric-type glioneuronal tumors characterized by oncogenic gene fusions involving several targetable kinases. Acta Neuropathol. 2022;144(5):1049–1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Suwala AK, Felix M, Friedel D, et al. Oligosarcomas, IDH-mutant are distinct and aggressive. Acta Neuropathol. 2022;143(2):263–281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Wu Z, Rajan S, Chung HJ, et al. Molecular and clinicopathologic characteristics of gliomas with EP300::BCOR fusions. Acta Neuropathol. 2022;144(6):1175–1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Torre M, Meredith DM, Dubuc A, et al. Recurrent EP300-BCOR fusions in pediatric gliomas with distinct clinicopathologic features. J Neuropathol Exp Neurol. 2019;78(4):305–314. [DOI] [PubMed] [Google Scholar]
- 34. Tauziede-Espariat A, Uro-Coste E, Sievers P, et al. ; RENOCLIP-LOC. CNS tumor with EP300::BCOR fusion: discussing its prevalence in adult population. Acta Neuropathol Commun. 2023;11(1):26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Sievers P, Henneken SC, Blume C, et al. Recurrent fusions in PLAGL1 define a distinct subset of pediatric-type supratentorial neuroepithelial tumors. Acta Neuropathol. 2021;142(5):827–839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Sievers P, Bielle F, Gobel K, et al. Identification of a putative molecular subtype of adult-type diffuse astrocytoma with recurrent MAPK pathway alterations. Acta Neuropathol. 2024;148(1):7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Keck MK, Sill M, Wittmann A, et al. Amplification of the PLAG-family genes-PLAGL1 and PLAGL2-is a key feature of the novel tumor type CNS embryonal tumor with PLAGL amplification. Acta Neuropathol. 2023;145(1):49–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Barresi V, Cardoni A, Miele E, et al. CNS tumor with CREBBP::BCORL1 fusion and pathogenic mutations in BCOR and CREBBP: expanding the spectrum of BCOR-altered tumors. Acta Neuropathol Commun. 2024;12(1):8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Andreiuolo F, Ferrone CK, Rajan S, et al. Molecular and clinicopathologic characteristics of CNS embryonal tumors with BRD4::LEUTX fusion. Acta Neuropathol Commun. 2024;12(1):42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Alhalabi KT, Stichel D, Sievers P, et al. PATZ1 fusions define a novel molecularly distinct neuroepithelial tumor entity with a broad histological spectrum. Acta Neuropathol. 2021;142(5):841–857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Hench J, Hultschig C, Brugger J, et al. EpiDiP/NanoDiP: a versatile unsupervised machine learning edge computing platform for epigenomic tumour diagnostics. Acta Neuropathol Commun. 2024;12(1):51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Eschbacher KL, Tran QT, Moskalev EA, et al. NAB2::STAT6 fusions and genome-wide DNA methylation profiling: predictors of patient outcomes in meningeal solitary fibrous tumors. Brain Pathol. 2024;34:e13256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Maas SLN, Stichel D, Hielscher T, et al. ; German Consortium on Aggressive Meningiomas (KAM). Integrated molecular-morphologic meningioma classification: a multicenter retrospective analysis, retrospectively and prospectively validated. J Clin Oncol. 2021;39(34):3839–3852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Wu Z, Abdullaev Z, Pratt D, et al. Impact of the methylation classifier and ancillary methods on CNS tumor diagnostics. Neuro-Oncology. 2022;24(4):571–581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Ebrahimi A, Korshunov A, Reifenberger G, et al. Pleomorphic xanthoastrocytoma is a heterogeneous entity with pTERT mutations prognosticating shorter survival. Acta Neuropathol Commun. 2022;10(1):5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Trager M, Schweizer L, Perez E, et al. Adult intracranial ependymoma-relevance of DNA methylation profiling for diagnosis, prognosis, and treatment. Neuro-Oncol. 2023;25(7):1286–1298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Capper D, Stichel D, Sahm F, et al. Practical implementation of DNA methylation and copy-number-based CNS tumor diagnostics: the Heidelberg experience. Acta Neuropathol. 2018;136(2):181–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Paulson VA, Liu YJ, Fang H, et al. Infantile ZFTA fusion-positive tumor of the posterior fossa: molecular tumor board. JCO Precis Oncol. 2023;7:e2200226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Liu APY, Priesterbach-Ackley LP, Orr BA, et al. WNT-activated embryonal tumors of the pineal region: ectopic medulloblastomas or a novel pineoblastoma subgroup? Acta Neuropathol. 2020;140(4):595–597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Pages M, Uro-Coste E, Colin C, et al. ; On Behalf Of The Renoclip-Loc Network. The implementation of DNA methylation profiling into a multistep diagnostic process in pediatric neuropathology: a 2-year real-world experience by the French neuropathology network. Cancers (Basel). 2021;13(6):1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Pickles JC, Fairchild AR, Stone TJ, et al. DNA methylation-based profiling for paediatric CNS tumour diagnosis and treatment: a population-based study. Lancet Child Adolesc Health. 2020;4(2):121–130. [DOI] [PubMed] [Google Scholar]
- 52. Jaunmuktane Z, Capper D, Jones DTW, et al. Methylation array profiling of adult brain tumours: diagnostic outcomes in a large, single centre. Acta Neuropathol Commun. 2019;7(1):24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Wiestler B, Capper D, Hovestadt V, et al. Assessing CpG island methylator phenotype, 1p/19q codeletion, and MGMT promoter methylation from epigenome-wide data in the biomarker cohort of the NOA-04 trial. Neuro-Oncol. 2014;16(12):1630–1638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Raffeld M, Abdullaev Z, Pack SD, et al. High level MYCN amplification and distinct methylation signature define an aggressive subtype of spinal cord ependymoma. Acta Neuropathol Commun. 2020;8(1):101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Shatara M, Schieffer KM, Klawinski D, et al. Clinically aggressive pediatric spinal ependymoma with novel MYC amplification demonstrates molecular and histopathologic similarity to newly described MYCN-amplified spinal ependymomas. Acta Neuropathol Commun. 2021;9(1):192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Tauziede-Espariat A, Uro-Coste E, Nicaise Y, et al. Refinement of diagnostic criteria for pediatric-type diffuse high-grade glioma, IDH- and H3-wildtype, MYCN-subtype including histopathology, TP53, MYCN and ID2 status. Acta Neuropathol Commun. 2023;11(1):170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Hegi ME, Oppong FB, Perry JR, et al. No benefit from TMZ treatment in GB with truly unmethylated MGMT promoter: reanalysis of the CE.6 and the pooled Nordic/NOA-08 trials in elderly GB patients. Neuro-Oncol. 2024;26:1867–1875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Bady P, Delorenzi M, Hegi ME.. Sensitivity analysis of the MGMT-STP27 model and impact of genetic and epigenetic context to predict the MGMT methylation status in gliomas and other tumors. J Mol Diagn. 2016;18:350–361. [DOI] [PubMed] [Google Scholar]
- 59. van den Bent MJ, Erdem-Eraslan L, Idbaih A, et al. MGMT-STP27 methylation status as predictive marker for response to PCV in anaplastic Oligodendrogliomas and Oligoastrocytomas. A report from EORTC study 26951. Clin Cancer Res. 2013;19(19):5513–5522. [DOI] [PubMed] [Google Scholar]
- 60. Stichel D, Schrimpf D, Sievers P, et al. Accurate calling of KIAA1549-BRAF fusions from DNA of human brain tumours using methylation array-based copy number and gene panel sequencing data. Neuropathol Appl Neurobiol. 2021;47(3):406–414. [DOI] [PubMed] [Google Scholar]
- 61. Daenekas B, Perez E, Boniolo F, et al. Conumee 2.0: enhanced copy-number variation analysis from DNA methylation arrays for humans and mice. Bioinformatics. 2024;40(2):btae029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Djirackor L, Halldorsson S, Niehusmann P, et al. Intraoperative DNA methylation classification of brain tumors impacts neurosurgical strategy. Neurooncol Adv. 2021;3(1):vdab149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Patel A, Dogan H, Payne A, et al. Rapid-CNS(2): rapid comprehensive adaptive nanopore-sequencing of CNS tumors, a proof-of-concept study. Acta Neuropathol. 2022;143(5):609–612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Sahm F, Brandner S, Bertero L, et al. Molecular diagnostic tools for the World Health Organization (WHO) 2021 classification of gliomas, glioneuronal and neuronal tumors; an EANO guideline. Neuro-Oncol. 2023;25(10):1731–1749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, eds. WHO Classification of Tumours of the Central Nervous System. 4th ed. Lyon, France: WHO Publications; 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Louis DN, Perry A, Wesseling P, et al. The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro-Oncol. 2021;23(8):1231–1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Sharma T, Schwalbe EC, Williamson D, et al. Second-generation molecular subgrouping of medulloblastoma: an international meta-analysis of Group 3 and Group 4 subtypes. Acta Neuropathol. 2019;138(2):309–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Cavalli FMG, Remke M, Rampasek L, et al. Intertumoral heterogeneity within medulloblastoma subgroups. Cancer Cell. 2017;31(6):737–754.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Kaffes I, Szulzewsky F, Chen Z, et al. Human Mesenchymal glioblastomas are characterized by an increased immune cell presence compared to Proneural and Classical tumors. Oncoimmunol. 2019;8(11):e1655360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Dejaegher J, Solie L, Hunin Z, et al. DNA methylation based glioblastoma subclassification is related to tumoral T-cell infiltration and patient survival. Neuro-Oncol. 2021;23(2):240–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Korshunov A, Schrimpf D, Ryzhova M, et al. H3-/IDH-wild type pediatric glioblastoma is comprised of molecularly and prognostically distinct subtypes with associated oncogenic drivers. Acta Neuropathol. 2017;134(3):507–516. [DOI] [PubMed] [Google Scholar]
- 72. Bender K, Kahn J, Perez E, et al. Diffuse paediatric-type high-grade glioma, H3-wildtype and IDH-wildtype: case series of a new entity. Brain Tumor Pathol. 2023;40(4):204–214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Roohani S, Ehret F, Perez E, et al. Sarcoma classification by DNA methylation profiling in clinical everyday life: the Charite experience. Clin Epigenetics. 2022;14(1):149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Jurmeister P, Gloss S, Roller R, et al. DNA methylation-based classification of sinonasal tumors. Nat Commun. 2022;13(1):7148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Kommoss FKF, Stichel D, Schrimpf D, et al. DNA methylation-based profiling of uterine neoplasms: a novel tool to improve gynecologic cancer diagnostics. J Cancer Res Clin Oncol. 2020;146(1):97–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Priesterbach-Ackley LP, Boldt HB, Petersen JK, et al. Brain tumour diagnostics using a DNA methylation-based classifier as a diagnostic support tool. Neuropathol Appl Neurobiol. 2020;46(5):478–492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Plass C, Pfister SM, Lindroth AM, et al. Mutations in regulators of the epigenome and their connections to global chromatin patterns in cancer. Nat Rev Genet. 2013;14(11):765–780. [DOI] [PubMed] [Google Scholar]
- 78. Stone TJ, Keeley A, Virasami A, et al. Comprehensive molecular characterisation of epilepsy-associated glioneuronal tumours. Acta Neuropathol. 2018;135(1):115–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Stone TJ, Mankad K, Tan AP, et al. DNA methylation-based classification of glioneuronal tumours synergises with histology and radiology to refine accurate molecular stratification. Neuropathol Appl Neurobiol. 2023;49(2):e12894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Leung ML, Abdullaev Z, Santana-Santos L, et al. Microarray-Based DNA methylation profiling: validation considerations for clinical testing. J Mol Diagn. 2024;26(6):447–455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Park JW, Lee K, Kim EE, Kim SI, Park SH.. Brain tumor classification by methylation profile. J Korean Med Sci. 2023;38(43):e356. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.