Summary
BCR::ABL1-like B-lymphoblastic leukaemia (B-ALL) neoplasms lack the BCR::ABL1 translocation but have a gene expression profile like BCR::ABL1 positive B-ALL. This includes alterations in cytokine receptors and signalling genes, such as CRLF2, ABL1, ABL2, JAK2, PDGFRB and EPOR. Cases with CRLF2 rearrangements account for approximately 50% of cases of Philadelphia-like acute lymphoblastic leukaemia (Ph-like ALL), and the frequency of specific genomic lesions varies with ethnicity such that IGH::CRLF2 translocations are more common in Hispanics and Native Americans.
We report two cases of BCR::ABL1-like ALL, with significant eosinophilia. A Hispanic man in his early 20s and a Hispanic woman in her 50s presented with leukocytosis and eosinophilia. Bone marrow flow cytometry revealed lymphoblasts expressing CD19, CD10, partial CD20, CD22, CD79a, CD38, CD34, TdT and HLA-DR. Examination of the bone marrow biopsy and aspirate exhibited a hypercellular bone marrow with increased blasts and elevated eosinophils. Fluorescence in situ hybridisation (FISH) demonstrated a cryptic chromosomal rearrangement between the X chromosome and chromosome 14 at breakpoints involving IGH at 14q32 and CRLF2 at Xp22.33, t(X;14)(p22.33; q32).
These findings confirmed the diagnosis of BCR::ABL1-like B-ALL with IGH::CRLF2 rearrangement. One patient (man) attained complete remission with induction therapy using the paediatric CALGB 10403 protocol, while the other patient (woman) had a poor outcome after receiving a hyper-fractionated cyclophosphamide, vincristine, doxorubicin and dexamethasone regimen. These two cases demonstrate an unusual presentation of BCR::ABL1-like B-ALL and emphasise the importance of appropriate cytogenetic studies for correct diagnosis. When treated with conventional chemotherapy, these cases carry a poor prognosis and might require allogeneic transplantation.
Keywords: Malignant and Benign haematology, Cancer - see Oncology, Pathology, Haematology (incl blood transfusion)
Background
Philadelphia-like acute lymphoblastic leukaemia (Ph-like ALL), also referred to as BCR::ABL1-like ALL, is a high-risk cytogenetic subtype of precursor B-cell acute lymphoblastic leukaemia (B-cell ALL). Ph-like ALL patients have a gene expression similar to BCR::ABL1-positive patients; however, they lack the BCR::ABL1 fusion gene resulting from the translocation t(9;22)(q34;q11.2).1 Deletions or mutations of the lymphoid transcription factor gene IKZF1 are a hallmark of both BCR::ABL1-positive ALL and Ph-like ALL; however, other mutations including alterations in the JAK-STAT or MAPK signalling also occur.2 Approximately half of the patients with Ph-like ALL have abnormalities of the cytokine receptor gene 2 (CRLF2) either as a translocation to the immunoglobulin heavy chain enhancer region (IGH::CRLF2) or as a focal microdeletion.2
In this case series, we examine the outcomes of two patients with Ph-like ALL who were found to have CRLF2 translocation to the immunoglobulin heavy chain enhancer region (IGH::CRLF2) and discuss the challenges associated with their care and the importance of appropriate cytogenetic studies for correct diagnosis.
Case presentation
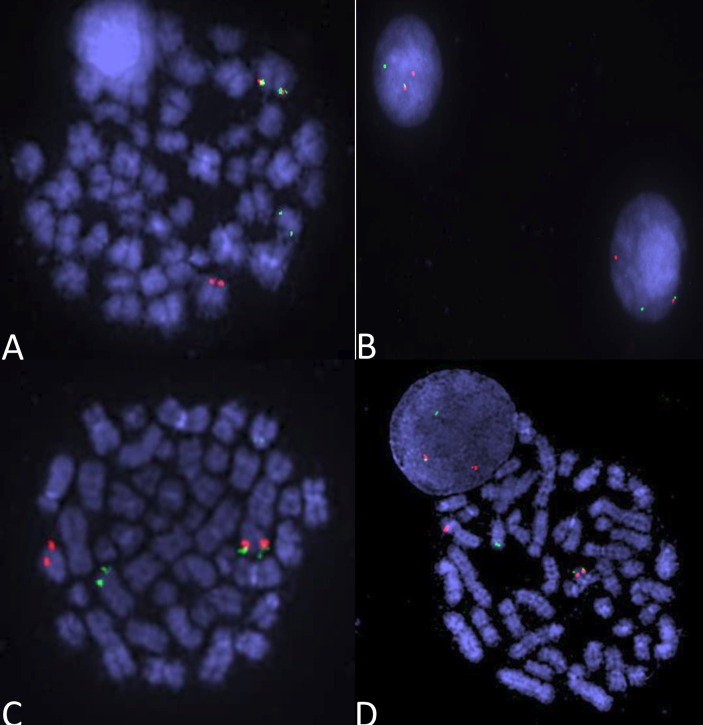
A Hispanic woman in her 50s with no significant medical history presented with leucocytosis (WBC 47/Blasts 75%/Plt 14/Hb 9.7. Diff: N:3% L:5% Mo:1% Eo:13% Baso:0%). Other labs including coagulation studies were normal. Bone marrow flow cytometry revealed 70% of lymphoblasts with CD19, bright CD10, dim CD20, variable CD22, CD38, CD34, variable HLA-DR, CD79a and TDT with dim CD45. Examination of the bone marrow biopsy and aspirate showed hypercellular marrow with increased blasts (more than 90%) consistent with B-lymphoblastic leukaemia. Chromosome analysis showed a normal female karyotype. Fluorescence in situ hybridisation (FISH) was positive for a chromosomal rearrangement between the X chromosome and chromosome 14 at breakpoints involving Xp22.33, ie, /t(X;14)(p22.33;14q32.2) by metaphase, with 88% of cells showing CRLF2 rearrangement by interphase (see figure 1A,B). These findings confirmed the diagnosis of BCR::ABL1-like B-ALL with IGH::CRLF2 rearrangement.
Figure 1. Rearrangement of CRLF2 gene at Xp22.33 on metaphase FISH study (A) and interphase FISH study (B) using CRLF2 breakapart probe.Rearrangement of IGH gene at 14q32.3 on metaphase FISH study (C) and interphase FISH study (D) using IGH breakapart probe.
Case 2
A Hispanic man in his early 20s with no significant medical history presented with leucocytosis. His labs showed (WBC 81/Blasts 8%/Plt 376/Hb 16. Diff: N:15% L:10% Mo:0% Eo:59% Baso:4%). Other laboratory findings were unremarkable. Bone marrow flow cytometry was consistent with B-lymphoblastic leukaemia, with 31% lymphoblasts expressing CD19, CD10, partial CD20, CD22, CD79a, CD38, CD34, TdT and HLA-DR. Examination of the bone marrow biopsy and aspirate exhibited a hypercellular bone marrow with increased blasts and elevated atypical eosinophils. Per discussion with pathology, the presence of eosinophilia with abnormal eosinophil segmentation and granulation raised the possibility of a lymphoid neoplasm with eosinophilia and rearrangement of PDGFRA, PDGFRB, or FGFR1 or with PCM1::JAK2. Chromosome analysis showed a normal male karyotype, whereas FISH demonstrated a cryptic chromosomal rearrangement between the X chromosome and chromosome 14 at breakpoints involving IGH at 14q32 and CRLF2 at Xp22.33, i.e., t(X;14)(p22.33; q32) (see figure 1C,D). These findings confirmed the diagnosis of BCR::ABL1-like B-ALL with IGH::CRLF2 rearrangement.
The patient attained complete remission with induction therapy using the paediatric protocol, CALGB 10403. The patient’s course was complicated by persistent transaminitis that was believed to be due to the peg-asparaginase. The liver biopsy showed medication-induced injury. Maintenance therapy was initiated per protocol; however, methotrexate was held given the liver injury. The patient continues to follow up in the clinic and is stable on maintenance therapy. Unfortunately, due to his visa status, he was not a candidate for bone marrow transplantation and he remains stable on POMP (6-Mercaptopurine + Vincristine + Methotrexate + Prednisone) therapy.
Investigations
Laboratory blood workup: complete blood count with differential,
-
Comprehensive metabolic panel
Coagulation studiesStool studies
Flow cytometry
Peripheral smear
Bone marrow biopsy
FISH studies
Differential diagnosis
Philadelphia-positive ALL was a consideration. It was important to perform a FISH test in order to assess if this was the case. We also needed to rule out Philadelphia-negative ALL. These two diseases are separate entities and differ in their prognosis and treatment.
Treatment
Patients received high-dose chemotherapy in order to control their acute leukaemia.
Outcome and follow-up
The woman patient in her 50s passed away in hospital due to infectious complications. The man patient in his early 20s achieved remission and is still being followed up at the haematology clinic 2 years after diagnosis. He is currently undergoing maintenance therapy and surveillance every month.
Discussion
B-lineage ALL is a disease with several subgroups some of which have an unfavourable prognosis. The outcome of B-ALL not only depends on the dose intensity of chemotherapy given but also depends strongly on the identification of specific genetic aberrations that can establish an accurate diagnosis and determine optimal therapy.3 B-ALL comprises multiple genetic subtypes which indicate underlying molecular lesions that are important for risk stratification.3 In childhood ALL, numerous good and high-risk cytogenetic subgroups have been identified. This, along with intensive chemotherapy, has led to great outcomes in paediatric patients with complete remission rates that exceed 95% and long-term event-free survival that exceeds 80%.4,6 Outcomes in adult B-cell ALL are inferior compared with paediatric patients. The inferior survival is believed to be a result of a higher frequency of poor prognostic cytogenetic subtypes such as BCR::ABL1 translocation and MLL rearrangements, and a lower frequency of favourable prognostic subtypes, such as ETV6::RUNX1 and high hyperdiploidy.7 Previous studies have shown that adults with ALL have approximately an 80% complete response rate and a 5-year overall survival of 40–50%.7 BCR::ABL1-like or Ph-like ALL is one such high-risk entity of B-ALL that has a gene expression signature like Ph-positive ALL but lacks the BCR::ABL1 fusion gene.7 Ph-like ALL is a diverse subtype of ALL because it can include rearrangements, copy number alterations, and sequence mutations that lead to activation of kinase or affect signalling pathways.8 This subtype can include rearrangements of CRLF2, JAK2 or EPOR, fusions that involve the ABL gene, or alterations in the pathways that affect JAK-STAT or RAS.8 The frequency of kinase subgroups differs with age, particularly as we discuss CRLF2 rearrangements such as in our patients. IGH::CRLF2 accounts for almost 50% of Ph-like ALL in young adults and adults (rather than the paediatric population).8 Ph-like ALL was first identified in the paediatric population and accounts for 10–15% of childhood B-ALL.9 10 The frequency of kinase subgroups differs with age, particularly as we discuss CRLF2 rearrangements such as in our patients. IGH::CRLF2 accounts for almost 50% of Ph-like ALL in young adults and adults (rather than the paediatric population). Like BCR::ABL1-positive ALL, the incidence of Ph-like ALL increases with age.9,11 In a comprehensive retrospective study by Roberts et al, 798 adult patients with B-ALL were studied and the incidence of Ph-like ALL was reported as 51.1% in young adults, 46.8% in adults and 58.4% in older adults.11 Roberts et al.11 provided a comprehensive understanding of the prevalence and outcome of Ph-like ALL. They found that patients with Ph-like ALL were associated with inferior treatment outcomes and a 5-year overall survival of 25.4% vs 64.2% in patients with non-Ph-like ALL.11 These patients also had a higher risk of relapse compared with non-Ph-like patients. Their 5-year event-free survival was 40.4% in young adults, 29.8% in adults and 18.9% in older adults.11 As evident by these studies, increasing age led to worse outcomes. Another interesting finding was that patients with Ph-like ALL also had more MRD positivity at the end of treatment than patients with BCR::ABL1 ALL.10 11
According to a genome-wide analysis of paediatric patients with high-risk ALL,10 50% of patients with Ph-like ALL have high expression of the type I cytokine receptor, CRLF2.10 CRLF2 rearrangement with concomitant Janus Kinase (JAK) mutations were detected in 50% of those patients.10 These rearrangements were seen in a similar distribution in adults with an overall CRLF2 frequency noted of 51%.11 Deregulation of CRLF2 expression occurs via three genomic rearrangements. According to Russell et al, the three genomic mutations are: translocation with the immunoglobulin heavy chain locus (IGH), an interstitial deletion within the pseudo autosomal region of chromosomes X and Y (P2RY8::CRLF2) and recurrent CRLF2 mutations.12 This leads to overexpression of CRLF2 mRNA and protein, the cause of which is not well understood, however, is common in Down syndrome ALL.2 12 13
Both patients described in our cases were of Hispanic origin and had CRLF2 rearrangements. Previous studies of children with high-risk ALL have shown that CRLF2 gene rearrangement is more common in the Hispanic ethnicity as compared with the non-Hispanic and Latino population of patients.14 15 As for adult patients, Jain et al16 studied 148 patients with Ph-like ALL and performed genomic mapping of these patients.16 This study found that patients with Ph-like ALL had worse outcomes with a 5-year overall survival for Ph-like ALL of 23% compared with 59% for other B-cell ALLs.16 An interesting observation in this study was that adult patients with ALL who were of Hispanic ethnicity with overexpression of CRLF2, however, outcomes in these patients were similar to outcomes previously reported.16 This observation had not been previously described in the literature. There seems to be a genetic predisposition in the Hispanic community to this gene rearrangement as in the literature discussed.14,16 This group of patients, although predominantly uniformly treated with an aggressive chemotherapy regimen, the hyper-CVAD regimen, had a poor outcome with a 5-year survival of <20% if they had a CRLF2 rearrangement.16
Despite advances in genomic sequencing, Ph-like ALL remains a high-risk and poor outcome disease. Understanding the mechanisms of oncogenic signalling by CRLF2 in ALL and identifying co-mutations will provide important insights into the role of CRLF2 in this disease entity. The reason for the inferior outcomes of patients with Ph-like ALL and CRLF2 overexpression compared with non-CRLF2 Ph-like ALL is unclear. Whether owing to concomitant mutations such as IKZF1 or JAK2 mutations as was previously reported or to a population of patients who cannot tolerate dose-intense chemotherapy remains unclear.2 Furthermore, the link between ethnicity and Ph-like ALL (especially those with CRLF2 rearrangement) is worth exploring in order to identify whether treatment outcomes are different among races. Once better characterised, this may lead to new therapeutic approaches to improve outcomes in CRLF2 overexpression and Ph-like ALL. Our case series highlights the importance of genomic characterisation of Ph-like ALL, as this may have significant therapeutic implications with the emerging use of kinase inhibitors in this patient population.
Learning points.
Despite advances in genomic sequencing, Ph-like ALL remains a high-risk and poor outcome disease.
Understanding the mechanisms of oncogenic signalling by CRLF2 in ALL and identifying co-mutations will provide important insights into the role of CRLF2 in this disease entity.
The link between ethnicity and Ph-like ALL (especially those with CRLF2 rearrangement) is worth exploring in order to identify whether treatment outcomes are different among races.
We need new therapeutic approaches to improve outcome in CRLF2 overexpression and Ph-like ALL.
Footnotes
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Provenance and peer review: Not commissioned; externally peer reviewed.
Patient consent for publication: Consent obtained directly from patient(s).
References
- 1.Den Boer ML, van Slegtenhorst M, De Menezes RX, et al. A subtype of childhood acute lymphoblastic leukaemia with poor treatment outcome: a genome-wide classification study. Lancet Oncol . 2009;10:125–34. doi: 10.1016/S1470-2045(08)70339-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Roberts KG, Li Y, Payne-Turner D, et al. Targetable kinase-activating lesions in Ph-like acute lymphoblastic leukemia. N Engl J Med. 2014;371:1005–15. doi: 10.1056/NEJMoa1403088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Byun JM, Koh Y, Shin D-Y, et al. BCR-ABL translocation as a favorable prognostic factor in elderly patients with acute lymphoblastic leukemia in the era of potent tyrosine kinase inhibitors. Haematologica. 2017;102:e187–90. doi: 10.3324/haematol.2016.159988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.DeAngelo DJ, Stevenson KE, Dahlberg SE, et al. Long-term outcome of a pediatric-inspired regimen used for adults aged 18-50 years with newly diagnosed acute lymphoblastic leukemia. Leukemia. 2015;29:526–34. doi: 10.1038/leu.2014.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Silverman LB, Stevenson KE, O’Brien JE, et al. Long-term results of Dana-Farber Cancer Institute ALL Consortium protocols for children with newly diagnosed acute lymphoblastic leukemia (1985-2000) Leukemia. 2010;24:320–34. doi: 10.1038/leu.2009.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gaynon PS, Angiolillo AL, Carroll WL, et al. Long-term results of the children’s cancer group studies for childhood acute lymphoblastic leukemia 1983-2002: a Children’s Oncology Group Report. Leukemia. 2010;24:285–97. doi: 10.1038/leu.2009.262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Boer JM, Koenders JE, van der Holt B, et al. Expression profiling of adult acute lymphoblastic leukemia identifies a BCR-ABL1-like subgroup characterized by high non-response and relapse rates. Haematologica. 2015;100:e261–4. doi: 10.3324/haematol.2014.117424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Roberts KG. Genetics and prognosis of ALL in children vs adults. Hematology Am Soc Hematol Educ Program. 2018;2018:137–45. doi: 10.1182/asheducation-2018.1.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Roberts KG, Pei D, Campana D, et al. Outcomes of children with BCR-ABL1–like acute lymphoblastic leukemia treated with risk-directed therapy based on the levels of minimal residual disease. J Clin Oncol. 2014;32:3012–20. doi: 10.1200/JCO.2014.55.4105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roberts KG, Morin RD, Zhang J, et al. Genetic alterations activating kinase and cytokine receptor signaling in high-risk acute lymphoblastic leukemia. Cancer Cell. 2012;22:153–66. doi: 10.1016/j.ccr.2012.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Roberts KG, Gu Z, Payne-Turner D, et al. High Frequency and Poor Outcome of Philadelphia Chromosome-Like Acute Lymphoblastic Leukemia in Adults. J Clin Oncol. 2017;35:394–401. doi: 10.1200/JCO.2016.69.0073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Russell LJ, Jones L, Enshaei A, et al. Characterisation of the genomic landscape of CRLF2-rearranged acute lymphoblastic leukemia. Genes Chromosomes Cancer. 2017;56:363–72. doi: 10.1002/gcc.22439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mullighan CG, Collins-Underwood JR, Phillips LAA, et al. Rearrangement of CRLF2 in B-progenitor- and Down syndrome-associated acute lymphoblastic leukemia. Nat Genet. 2009;41:1243–6. doi: 10.1038/ng.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Harvey RC, Mullighan CG, Chen I-M, et al. Rearrangement of CRLF2 is associated with mutation of JAK kinases, alteration of IKZF1, Hispanic/Latino ethnicity, and a poor outcome in pediatric B-progenitor acute lymphoblastic leukemia. Blood. 2010;115:5312–21. doi: 10.1182/blood-2009-09-245944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Raca G, Abdel-Azim H, Yue F, et al. Increased Incidence of IKZF1 deletions and IGH-CRLF2 translocations in B-ALL of Hispanic/Latino children-a novel health disparity. Leukemia. 2021;35:2399–402. doi: 10.1038/s41375-021-01133-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jain N, Roberts KG, Jabbour E, et al. Ph-like acute lymphoblastic leukemia: a high-risk subtype in adults. Blood. 2017;129:572–81. doi: 10.1182/blood-2016-07-726588. [DOI] [PMC free article] [PubMed] [Google Scholar]