Abstract
Background
We evaluated the outcomes, and risk factors for recurrence in patients with early stage node-negative breast cancer in this study.
Method
Retrospective data analysis was done on patient treatment records from 1988 to 2018. The patient’s demographic, clinical, pathological, and therapeutic characteristics were noted. To evaluate survival analysis and predictors of recurrence, we employed Kaplan–Meier analysis with the log-rank test.
Results
A total of 357 patients in all were enrolled in the research. At the time of diagnosis, the median age was 50 (with a range of 18–81). A total of 85.5% of patients had undergone a lumpectomy, while 14.5% had a mastectomy. 78.7% of patients had sentinel lymph node biopsy, and 21.3% had axillary lymph node dissection. In addition, the patients received adjuvant radiotherapy (88.7%), adjuvant endocrine therapy (82.1%), and adjuvant chemotherapy (48.5%). Recurrence of the tumor occurred in 31 (8.7%) patients (local recurrence 45.2% and metastatic disease 54.8%). Ten- and twenty-year recurrence-free survival rates were 92% and 77%. 19 (5.3%) patients had also developed contralateral breast cancer. Ten-year survival rates were 91.6%, and 20-year survival rates were 76.6%, respectively. Aged over 65 years (p = 0.004), necrosis (p = 0.002), mitosis (p = 0.003), and nuclear pleomorphism (p = 0.049) were found as statistically significant factors for recurrence in univariate analysis. In the ROC analysis, the largest size of the tumor (over 1.45 cm, p = 0.07) remained outside the statistical significance limit in terms of recurrence.
Conclusions
Thirty-year outcomes in individuals with early stage, node-negative breast cancer were shown in this study. We found that the recurrence ratios between 10 and 20 years were more frequent than the first 10 years during the follow-up. Despite the small number of patients who experienced a recurrence, we demonstrated that, in univariate analysis, being older than 65 and having some pathological characteristics (nuclear pleomorphism, mitosis, and necrosis) were statistically significant factors for disease recurrence.
Keywords: Breast cancer, Early stage, Lymph node, Recurrence
Introduction
Breast cancer is the most common type of cancer in women worldwide, with more than two million women diagnosed each year (Sung et al. 2021). Non-metastatic breast cancer is divided into two categories at diagnosis as early stage or locally advanced. Primary treatment in early stage breast cancer (ESBC) is breast surgery (lumpectomy or mastectomy) and adjuvant radiotherapy. An adjuvant systemic plan is made according to the tumor size, tumor grade, estrogen receptor (ER), progesterone receptor (PR), and HER2 receptor status of the patients. Today, the adjuvant systemic decision in HER2-negative and hormone-positive ESBC is taken according to the risk assessment made on the genetic characteristics of the tumor determined by MammaPrint or Oncotype (Syed 2020; Soliman et al. 2020). In patients with breast cancer, the recurrence risk ranges from 3 to 15% in 10-year follow-up after lumpectomy plus radiotherapy (Arvold et al. 2011; Braunstein et al. 2017). In patients with ESBC, molecular relapse can be detected by the ctDNA method after surgery and adjuvant treatments, and future local or metastatic recurrences can be predicted (Garcia-Murillas et al. 2019).
Numerous factors have been identified that can be an indicator of recurrence and prognosis in patients with breast cancer after treatment. Published studies have shown that patients with breast cancer under the age of 35 and over the age of 65 may have a worse prognosis (Billena et al. 2021; van de Water et al. 2012). Tumor size has been shown to be an independent variable affecting recurrence, although it is correlated with lymph node involvement in patients with breast cancer (Pan et al. 2017). However, many pathological features such as tumor grade, lymphovascular invasion, Ki-67, and hormone receptors have been shown to affect recurrence and prognosis (Lukasiewicz et al. 2021). One of the strongest independent parameters affecting recurrence and prognosis in patients with breast cancer is lymph node involvement (Siegel et al. 2023). In the literature, data on the factors influencing recurrence in patients with stage 1 or 2 breast cancer without lymph node involvement are limited. In this study, 30-year outcomes and clinical and pathological features affecting recurrence were evaluated in patients with node-negative ESBC.
Material and method
Patients and data collection
This study was designed as a retrospective observational cohort study. Prior to conducting the study, approval from the institutional review board was obtained, and good clinical practice principles were followed. Patients who were diagnosed and treated in our institution between 1988 and 2018 were included in the study. Patients who did not have sufficient data for the study and whose patient follow-up file could not be reached were excluded from the study. Demographic, clinical, and pathological characteristics of the patients were recorded. The treatment characteristics of the patients (surgery, radiotherapy, chemotherapy, and endocrine therapy) and the recurrence status during the follow-up period were also noted. Tumor characteristics of all patients were evaluated in a standardized pathology laboratory. Tumor histological grade evaluation was performed according to The Bloom–Richardson Grading System. Assessment of ER and PR was done by immunohistochemistry (IHC) method. HER2/neu receptor positivity was considered significant if it was found to be Score 3 by IHC or positive by the in situ hybridization method. Tumor stages were made based on the American Joint Committee on Cancer 8th edition.
The death status of the patients included in the study was checked through the death notification system of the Ministry of Health. The time from diagnosis to death from any cause was considered as overall survival (OS). Recurrence-free survival (RFS) was defined as the time from the surgery to the development of disease recurrence (local or metastatic). In addition, clinical and pathological features affecting RFS were evaluated. The significance of tumor size for the development of recurrence was also assessed.
Statistical analysis
The statistics of the study were performed via SPSS 29 (IBM, Armonk, NY, USA). Continuous variables in the study were represented by median (as well as minimum and maximum values) value numbers and percentages, while categorical variables were described by numbers and percentages. Univariate analysis was performed on clinical and pathological factors affecting RFS. When the p-value was less than 0.05, results were deemed statistically significant, and the probability ratio was calculated. Due to the low recurrence rate in the patient group included in the study, a statistically significant model could not be established in the multivariate analysis. The significance of tumor size for the development of recurrence was evaluated by ROC analysis. For survival analysis, the Kaplan–Meier method was employed.
Results
Patient characteristics and treatment modality
The statistics of the study were made with the data of 357 patients. The median age of the patients was 50 (18–81). At the time of diagnosis, 48.7% of the patients were premenopausal. The risk factors of the patients for breast cancer were evaluated, and early menarche was present in − 5.9% and nulliparity in 14.3% of the patients. The median parity number of the patients was 2 (0–10), and the median age of menarche was 13 (9–18). Active smoking was present in 24.4% of the patients at the time of diagnosis. The general characteristics of the patients are shown in Table 1. In the evaluation made in terms of pathological features, the most common histopathological subtype was found to be invasive ductal carcinoma (IDC) (70.6%). ER positivity was 75.4%, and HER2 receptor positivity was 13.7%. Postoperative surgical margin positivity was detected in 7.6% of the patients. The pathological features of the patients such as tumor histological grade, in situ component, necrosis, lymphovascular invasion, and Ki-67 positivity percentage are shown in Table 2. Patients with early stage disease frequently underwent lumpectomy (84%), and the majority of patients received adjuvant radiotherapy (83.3%). The rate of patients receiving adjuvant chemotherapy was 45.9%. Only 31 of 49 patients with HER2-positive tumor could receive adjuvant trastuzumab because some of the patients had been diagnosed in the pre-trastuzumab period. The treatment-related characteristics of the patients are presented in Table 3.
Table 1.
Patients characteristics
| Number of patients | (%) | Valid (%) | |
|---|---|---|---|
| Age at diagnosis, years | |||
| < 65 | 309 | 86.6 | |
| ≥ 65 | 48 | 13.4 | |
| Smoking status | |||
| Current | 87 | 24.4 | 27.9 |
| Never | 225 | 63 | 72.1 |
| Unknown | 45 | 12.6 | |
| Menopausal status | |||
| Premenopausal | 174 | 48.7 | 50.3 |
| Postmenopausal | 172 | 48.2 | 49.7 |
| Unknown | 11 | 3.1 | |
| Early menarche | |||
| Yes | 21 | 5.9 | 7.6 |
| No | 256 | 71.7 | 92.4 |
| Unknown | 80 | 22.4 | |
| Nulliparity | |||
| Yes | 51 | 14.3 | 17.8 |
| No | 256 | 66.1 | 82.2 |
| Unknown | 70 | 19.6 | |
| Primary tumor locations | |||
| Left side | 182 | 51 | 51.7 |
| Right side | 166 | 46.5 | 47.2 |
| Bilateral | 4 | 1.1 | 1.1 |
| Unknown | 5 | 1.4 | 1.1 |
| Stage at diagnosis | |||
| Stage 1 pT1a | 19 | 5.3 | 5.4 |
| Stage 1 pT1b | 74 | 20.7 | 21 |
| Stage 1 pT1c | 232 | 65 | 65.9 |
| Stage 2 pT2 | 27 | 7.6 | 7.7 |
| Unknown | 5 | 1.4 | |
| Recurrence | |||
| No | 326 | 91.3 | |
| Local | 14 | 3.9 | |
| Metastatic | 17 | 4.8 |
Table 2.
Pathological features of the patients
| Number of patients | (%) | Valid (%) | |
|---|---|---|---|
| Histological type | |||
| IDC | 252 | 70.6 | 71.6 |
| Invasive lobular carcinoma | 23 | 6.4 | 6.5 |
| Other types (mixed type, micropapillary, etc.) | 77 | 21.6 | 21.9 |
| Unknown | 5 | 1.4 | |
| ER status | |||
| Positive | 269 | 75.4 | 80.3 |
| Negative | 66 | 18.5 | 19.7 |
| Unknown | 22 | 6.1 | |
| PR status | |||
| Positive | 205 | 57.4 | 62.1 |
| Negative | 125 | 35 | 37.9 |
| Unknown | 27 | 7.6 | |
| HER2 overexpression | |||
| Positive | 49 | 13.7 | 84.1 |
| Negative | 259 | 72.6 | 15.9 |
| Unknown | 49 | 13.7 | |
| Tumor focality | |||
| Multifocal | 56 | 15.7 | 16 |
| Unifocal | 295 | 82.6 | 84 |
| Unknown | 6 | 1.7 | |
| Tumor centricity | |||
| Multicentric | 9 | 2.5 | 2.6 |
| Unicentric | 341 | 95.5 | 97.4 |
| Unknown | 7 | 2 | |
| Tumor histologic grade | |||
| Grade 1 | 61 | 17.1 | 18.9 |
| Grade 2 | 156 | 43.7 | 48.3 |
| Grade 3 | 106 | 29.7 | 32.8 |
| Unknown | 34 | 9.5 | |
| In situ component | |||
| Yes | 218 | 61.1 | 75.4 |
| No | 71 | 19.9 | 24.6 |
| Unknown | 68 | 19 | |
| Necrosis | |||
| Yes | 50 | 14 | 16.8 |
| No | 248 | 69.5 | 83.2 |
| Unknown | 59 | 16.5 | |
| Lenfvascular invasion | |||
| Yes | 68 | 19 | 22.4 |
| No | 235 | 65.8 | 77.6 |
| Unknown | 54 | 15.2 | |
| Surgical margin status | |||
| Positive | 27 | 7.5 | 7.8 |
| Negative | 319 | 89.4 | 92.2 |
| Unknown | 11 | 3.1 | |
| Ki67 levels % | |||
| Ki67 ≤ 20 | 119 | 33.3 | 55.1 |
| Ki67 > 20 | 97 | 27.2 | 44.9 |
| Unknown | 141 | 39.5 |
Table 3.
Treatment approaches of the patients
| Number of patients | % | Valid (%) | |
|---|---|---|---|
| Breast surgery | |||
| Mastectomy | 51 | 14.3 | 14.5 |
| Lumpectomy | 300 | 84 | 85.5 |
| Unknown | 6 | 1.7 | |
| Lymph node surgery | |||
| Sentinel node biopsy | 269 | 75.4 | 78.7 |
| Axillary lymph node dissection | 73 | 20.4 | 21.3 |
| Unknown | 15 | 4.2 | |
| Adjuvant radiotherapy | |||
| Yes | 297 | 83.2 | 88.7 |
| No | 38 | 10.6 | 11.3 |
| Unknown | 22 | 6.2 | |
| Adjuvant chemotherapy | |||
| Yes | 164 | 45.9 | 51.5 |
| No | 174 | 48.8 | 48.5 |
| Unknown | 19 | 5.3 | |
| Adjuvant endocrine therapy | |||
| Yes | 275 | 77 | 82.1 |
| No | 60 | 16.8 | 17.9 |
| Unknown | 22 | 6.2 |
Prognosis and long-term outcomes
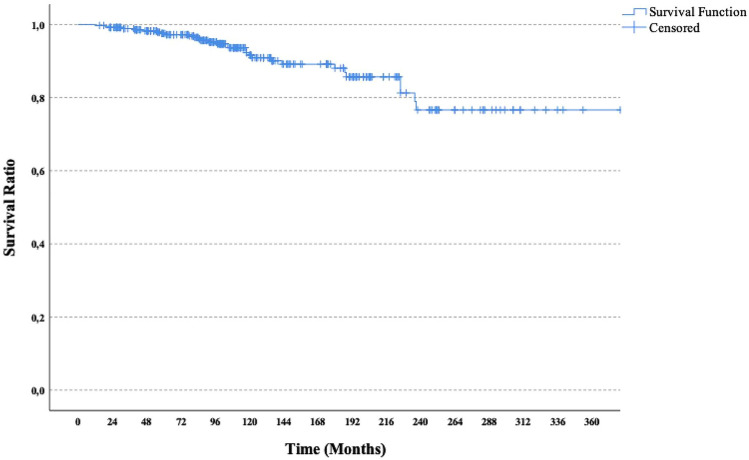
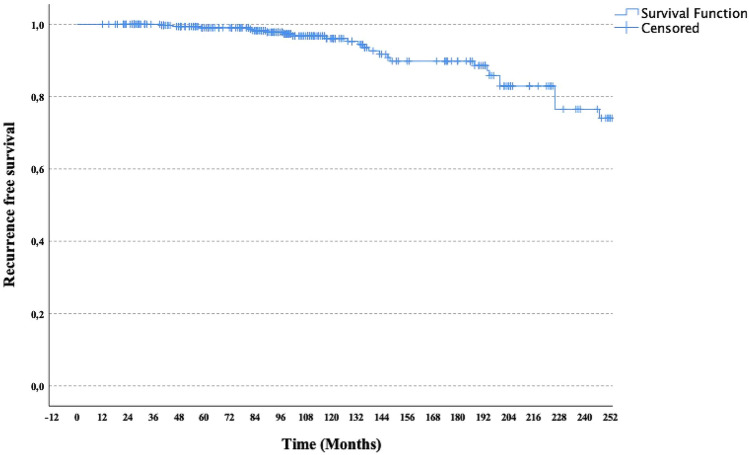
The median follow-up period was 100 months. During the follow-up period, 30 (8.4%) patients died. The 10-year and 20-year OS rates of the patients were 91.6% and 76.6%, respectively (Fig. 1). Relapse occurred in 31 (8.7%) patients during the follow-up period, and the 5-year, 10-year and 20-year RFS rates were 96.1, 92% and 77%, respectively (Fig. 2). 45.2% of the recurrences presented as local and 54.8% as metastatic disease. Clinical factors other than age (p = 0.004); smoking, menopausal status, early menarche, nulliparity onset stage, and type of surgery were not found to be statistically significant factors for recurrence (Table 4). When pathological factors are evaluated; except for pleomorphism (p = 0.049), mitosis score (p = 0.003), and necrosis (p = 0.002) other pathological features were not statistically significant in terms of recurrence (Table 5). In addition, Ki-67 level (p = 0.088), perineural invasion status (p = 0.061), breast surgery type (p = 0.064), and tumor localization (p = 0.087) remained within the statistical significance limit for recurrence. The median tumor size was 1.5 (0.1–5) cm. In ROC analysis, tumor size remained outside the statistical significance limit in terms of recurrence (p = 0.07 and AUC 0.599, for tumor size 1.45 cm; sensitivity 71%, specificity 53.6%) (Fig. 3).
Fig. 1.
Kaplan–Meier Curve for OS in the patients
Fig. 2.
Kaplan–Meier Curve for RFS in the patients
Table 4.
Univariate analysis of clinical factors for recurrence in the patients
| Total number | Number of events | Events (%) | p value | |
|---|---|---|---|---|
| Age | 0.004 | |||
| < 65 | 309 | 24 | 7.8 | |
| ≥ 65 | 48 | 7 | 14.6 | |
| Smoking status | 0.723 | |||
| Yes | 87 | 5 | 5.7 | |
| No | 225 | 18 | 8 | |
| Menopausal status | 0.943 | |||
| Premenopausal | 174 | 16 | 9.2 | |
| Postmenopausal | 172 | 13 | 7.6 | |
| Early menarche | 0.320 | |||
| Yes | 21 | 1 | 4.8 | |
| No | 256 | 19 | 7.4 | |
| Nulliparity | 0.820 | |||
| Yes | 51 | 4 | 7.8 | |
| No | 236 | 18 | 7.6 | |
| Primary tumor locations | 0.087 | |||
| Right side | 166 | 11 | 6.6 | |
| Left side | 182 | 19 | 10.4 | |
| Bilateral | 4 | 1 | 25 | |
| Stage at diagnosis | 0.618 | |||
| Stage 1 pT1a | 19 | 0 | 0 | |
| Stage 1 pT1b | 74 | 5 | 6.8 | |
| Stage 1 pT1c | 232 | 22 | 9.5 | |
| Stage 2 pT2 | 27 | 4 | 14.8 | |
| Breast surgery | 0.064 | |||
| Lumpectomy | 200 | 21 | 7 | |
| Mastectomy | 51 | 10 | 19.6 | |
| Lymph node surgery | 0.745 | |||
| Sentinel node biopsy | 269 | 19 | 7.1 | |
| Axillary LND | 73 | 12 | 16.4 | |
| Adjuvant radiotherapy | 0.142 | |||
| Yes | 297 | 23 | 7.7 | |
| No | 38 | 8 | 21.1 | |
| Adjuvant chemotherapy | 0.450 | |||
| Yes | 164 | 18 | 11 | |
| No | 174 | 12 | 6.9 | |
| Adjuvant trastuzumab | 0.578 | |||
| Yes | 31 | 3 | 9.7 | |
| No | 18 | 2 | 11.1 | |
| Adjuvant endocrine therapy | 0.372 | |||
| Yes | 275 | 25 | 9.1 | |
| No | 60 | 5 | 8.3 |
Table 5.
Univariate analysis of pathological features for recurrence in the patients
| Total number | Number of events | Events (%) | p value | |
|---|---|---|---|---|
| Histological type | 0.463 | |||
| IDC | 252 | 20 | 7.9 | |
| Other types | 100 | 11 | 11 | |
| ER status | 0.773 | |||
| Positive | 269 | 20 | 7.4 | |
| Negative | 66 | 7 | 10.6 | |
| PR status | 0.153 | |||
| Positive | 205 | 13 | 6.4 | |
| Negative | 125 | 13 | 10.4 | |
| HER2 overexpression | 0.314 | |||
| Negative | 259 | 19 | 7.3 | |
| Positive | 49 | 5 | 10.2 | |
| Tumor focality | 0.517 | |||
| Unifocal | 295 | 25 | 8.5 | |
| Multifocal | 56 | 6 | 10.7 | |
| Tumor centricity | 0.510 | |||
| Unicentric | 341 | 30 | 8.8 | |
| Multicentric | 9 | 1 | 11.1 | |
| Tumor histologic grade | 0.100 | |||
| Grade 1 | 61 | 1 | 1.6 | |
| Grade 2 | 156 | 11 | 7.1 | |
| Grade 3 | 106 | 15 | 14.2 | |
| Pleomorphism | 0.049 | |||
| Score 1 | 14 | 0 | 0 | |
| Score 2 | 127 | 3 | 2.4 | |
| Score 3 | 84 | 8 | 9.5 | |
| Mitosis score | 0.003 | |||
| Score 1 | 98 | 2 | 2 | |
| Score 2 | 77 | 2 | 2.6 | |
| Score 3 | 49 | 7 | 14.3 | |
| Tubule score | 0.224 | |||
| Score 1 | 22 | 0 | 0 | |
| Score 2 | 97 | 3 | 3.1 | |
| Score 3 | 106 | 8 | 7.5 | |
| In situ component | 0.328 | |||
| No | 71 | 6 | 8.5 | |
| Yes | 218 | 14 | 6.4 | |
| Necrosis | 0.002 | |||
| No | 248 | 12 | 4.8 | |
| Yes | 50 | 11 | 22 | |
| Lymphovascular invasion | 0.943 | |||
| No | 235 | 17 | 7.2 | |
| Yes | 68 | 6 | 8.8 | |
| Perineuronal invasion | 0.061 | |||
| No | 36 | 1 | 2.8 | |
| Yes | 6 | 2 | 33.3 | |
| Surgical margin status | 0.929 | |||
| Negative | 319 | 27 | 8.5 | |
| Positive | 27 | 3 | 11.1 |
If p < 0.05, values are shown in bold
Fig. 3.

ROC curve by tumor size for recurrence. (p = 0.07 and AUC 0.599, for tumor size 1.45 cm; sensitivity 71%, specificity 53.6%)
Discussion
In our study, we showed the clinicopathological features of node-negative ESBC patients. We found that this patient group generally had a good prognosis. The 10-year OS of the patients was 92%, and the 20-year OS was 77%. Similar to our study, in a multicenter study including patients with node-negative ESBC, 5-year survival was found to be 98%, and patients in Her-2 positive, triple negative, and pT1b subgroups had a worse prognosis (Gamucci et al. 2013). In another study conducted with patient data from the National Comprehensive Cancer Network Database, 5-year recurrence rates were found to be less than 10% in patients with small node-negative breast cancer without chemotherapy, and also, it was shown that patients with pT1a and pT1b tumors had an excellent prognosis (Vaz-Luis et al. 2014). In a study in the Danish Breast Cancer Group clinical database in which patients with breast cancer diagnosed between 1987 and 2004 and who did not develop recurrence within the first 10 years were included, the cumulative incidence of recurrence between 10 and 32 years after diagnosis was found to be 16.6% (Pedersen et al. 2022). Similar results were found in our study, and relapse occurred in 31 (8.7%) patients during the follow-up period. The 10-year and 20-year RFS rates were found to be 92% and 77%, respectively. The incidence of recurrence between 10 and 20 years after diagnosis was found to be 15%. In addition, we found that contralateral breast cancer developed in 5.3% of the patients in our study. In the SEER database analysis, in which breast cancer patients diagnosed and followed up between 1990 and 2015 were evaluated for contralateral breast cancer, contralateral breast cancer development was found in 3.2% of the patients after 25 years of follow-up, and it was determined that black women and patients with ER-negative tumors were at higher risk (Giannakeas et al. 2021).
In our study, we found that age at diagnosis and some pathological characteristics (nuclear pleomorphism, mitosis, and necrosis) increased the risk of recurrence. Contrary to the results of our study, in an analysis by Sharma et al., it was determined that patients younger than 40 years were more at risk for recurrence than patients ≥ 40 years of age in patients with T1 or T2 tumors and up to three lymph node positivity (Sharma et al. 2010). In a study conducted on a population-based database in Sweden, it was stated that the poor prognosis in elderly breast cancer patients was due to less diagnostic activity, later diagnosis, and less aggressive treatment, and the poor prognosis could not be explained only by comorbidities (Eaker et al. 2006). In our study, the high recurrence rate in patients over 65 years of age can be explained by reducing the intensity of adjuvant treatments. Tumor grade showing the aggressiveness of breast tumor is made by looking at tubule formation, mitosis count, and nuclear pleomorphism in The Bloom–Richardson Grading System (Gandhi et al. 2023). Rakha et al. showed that histological grade calculated by tubule formation, mitosis number, and nuclear pleomorphism scoring is associated with disease-free survival and breast cancer-related mortality (Rakha et al. 2008). In our study, although there was no statistical relationship between histologic grade and RFS, a trend was found between grade level and percentage of recurrence events. Similarly, it was shown that the rate of recurrence events increased with the level of tubule formation, mitosis number, and nuclear pleomorphism. The inability to reach statistical significance can be explained by the relatively limited number of patients. In a study evaluating patients with node-negative breast cancer by Zemni et al., the 5-year disease-free survival of patients with a median tumor size of 3.5 cm was 82.1%, and tumor size was found to be a prognostic factor in patients with breast cancer (Zemni et al. 2017). In our study, patients had a lower median tumor size (1.5 cm) and a better 10-year RFS (92%). This can be explained by the earlier diagnosis of our patient group. Also, the significance of tumor size for the development of recurrence was evaluated by ROC analysis and remained outside the statistical significance limit (for above 1.45 cm, p = 0.07) in our study.
Study limitations
Due to the retrospective nature of our study, some patient data were missing. The patient group was heterogeneous, and patients with sufficient 30-year follow-up data were included in the study. Despite long-term follow-up with ESBC, the number of patients who developed recurrence was limited. For this reason, a statistically significant multivariate analysis model could not be established for the factors affecting recurrence. Some of the HER2 positive patients could not use trastuzumab because they were diagnosed in the pre-trastuzumab period.
Conclusions
The clinical and pathological features of patients with node-negative ESBC were shown in the study, and the features of recurrence and contralateral breast cancer development in the long-term follow-up of these patients were demonstrated. In this study, clinical and pathological features affecting recurrence were evaluated. It has been shown that age at diagnosis and some pathological features (nuclear pleomorphism, mitosis, and necrosis) may affect recurrence. This study makes an important contribution to the literature in terms of including the results of 30 years of long-term follow-up. In the future, with a better understanding of the genetic and biological characteristics of breast cancer, the selection of adjuvant therapy and the factors affecting the development of recurrence will be better understood.
Author contributions
Conception and design: Dr. ID and Professor Dr. AA; Development of methodology, analysis, and interpretation of data: Dr. ID; Data acquisition: All authors; Manuscript writing: All authors; Final approval of manuscript: All authors.
Funding
Neither financial nor of other nature.
Data availability statement
This published paper contains all of the data produced or analyzed during this investigation. Further enquiries can be directed to the corresponding author.
Declarations
Conflict of interest
The authors declared that there are no potential conflicts of interest.
Ethical approval
The academic board approved this study at the Istanbul University Institute of Oncology (Number: 1723150).
Informed consent
For this type of research, informed consent is not required.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Arvold ND, Taghian AG, Niemierko A, Abi Raad RF, Sreedhara M, Nguyen PL, Bellon JR, Wong JS, Smith BL, Harris JR (2011) Age, breast cancer subtype approximation, and local recurrence after breast-conserving therapy. J Clin Oncol 29(29):3885–3891 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billena C, Wilgucki M, Flynn J, Modlin L, Tadros A, Razavi P, Braunstein LZ, Gillespie E, Cahlon O, McCormick B, Zhang Z, Morrow M, Powell S, Khan AJ (2021) 10-year breast cancer outcomes in women </=35 years of age. Int J Radiat Oncol Biol Phys 109(4):1007–1018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braunstein LZ, Taghian AG, Niemierko A, Salama L, Capuco A, Bellon JR, Wong JS, Punglia RS, MacDonald SM, Harris JR (2017) Breast-cancer subtype, age, and lymph node status as predictors of local recurrence following breast-conserving therapy. Breast Cancer Res Treat 161(1):173–179 [DOI] [PubMed] [Google Scholar]
- Eaker S, Dickman PW, Bergkvist L, Holmberg L, Uppsala G, Cancer OB (2006) Differences in management of older women influence breast cancer survival: results from a population-based database in Sweden. PLoS Med 3(3):e25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gamucci T, Vaccaro A, Ciancola F, Pizzuti L, Sperduti I, Moscetti L, Longo F, Fabbri MA, Giampaolo MA, Mentuccia L, Di Lauro L, Vici P (2013) Recurrence risk in small, node-negative, early breast cancer: a multicenter retrospective analysis. J Cancer Res Clin Oncol 139(5):853–860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandhi H, Maru A, Shah N, Mansuriya RK, Rathod G, Parmar P (2023) Correlation of Robinson’s cytological grading with Elston and Ellis’ Nottingham modification of bloom Richardson score of histopathology for breast carcinoma. Maedica (bucur) 18(1):55–60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Murillas I, Chopra N, Comino-Mendez I, Beaney M, Tovey H, Cutts RJ, Swift C, Kriplani D, Afentakis M, Hrebien S, Walsh-Crestani G, Barry P, Johnston SRD, Ring A, Bliss J, Russell S, Evans A, Skene A, Wheatley D, Dowsett M, Smith IE, Turner NC (2019) Assessment of molecular relapse detection in early-stage breast cancer. JAMA Oncol 5(10):1473–1478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giannakeas V, Lim DW, Narod SA (2021) The risk of contralateral breast cancer: a SEER-based analysis. Br J Cancer 125(4):601–610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lukasiewicz S, Czeczelewski M, Forma A, Baj J, Sitarz R, Stanislawek A (2021) Breast cancer-epidemiology, risk factors, classification, prognostic markers, and current treatment strategies-an updated review. Cancers (basel) 13(17):4287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan H, Gray R, Braybrooke J, Davies C, Taylor C, McGale P, Peto R, Pritchard KI, Bergh J, Dowsett M, Hayes DF, Ebctcg. (2017) 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med 377(19):1836–1846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen RN, Esen BO, Mellemkjaer L, Christiansen P, Ejlertsen B, Lash TL, Norgaard M, Cronin-Fenton D (2022) The incidence of breast cancer recurrence 10–32 years after primary diagnosis. J Natl Cancer Inst 114(3):391–399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rakha EA, El-Sayed ME, Lee AH, Elston CW, Grainge MJ, Hodi Z, Blamey RW, Ellis IO (2008) Prognostic significance of Nottingham histologic grade in invasive breast carcinoma. J Clin Oncol 26(19):3153–3158 [DOI] [PubMed] [Google Scholar]
- Sharma R, Bedrosian I, Lucci A, Hwang RF, Rourke LL, Qiao W, Buchholz TA, Kronowitz SJ, Krishnamurthy S, Babiera GV, Gonzalez-Angulo AM, Meric-Bernstam F, Mittendorf EA, Hunt KK, Kuerer HM (2010) Present-day locoregional control in patients with t1 or t2 breast cancer with 0 and 1 to 3 positive lymph nodes after mastectomy without radiotherapy. Ann Surg Oncol 17(11):2899–2908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73(1):17–48 [DOI] [PubMed] [Google Scholar]
- Soliman H, Shah V, Srkalovic G, Mahtani R, Levine E, Mavromatis B, Srinivasiah J, Kassar M, Gabordi R, Qamar R, Untch S, Kling HM, Treece T, Audeh W (2020) MammaPrint guides treatment decisions in breast cancer: results of the IMPACt trial. BMC Cancer 20(1):81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249 [DOI] [PubMed] [Google Scholar]
- Syed YY (2020) Oncotype DX breast recurrence score((R)): a review of its use in early-stage breast cancer. Mol Diagn Ther 24(5):621–632 [DOI] [PubMed] [Google Scholar]
- van de Water W, Markopoulos C, van de Velde CJ, Seynaeve C, Hasenburg A, Rea D, Putter H, Nortier JW, de Craen AJ, Hille ET, Bastiaannet E, Hadji P, Westendorp RG, Liefers GJ, Jones SE (2012) Association between age at diagnosis and disease-specific mortality among postmenopausal women with hormone receptor-positive breast cancer. JAMA 307(6):590–597 [DOI] [PubMed] [Google Scholar]
- Vaz-Luis I, Ottesen RA, Hughes ME, Mamet R, Burstein HJ, Edge SB, Gonzalez-Angulo AM, Moy B, Rugo HS, Theriault RL, Weeks JC, Winer EP, Lin NU (2014) Outcomes by tumor subtype and treatment pattern in women with small, node-negative breast cancer: a multi-institutional study. J Clin Oncol 32(20):2142–2150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zemni I, Ghalleb M, Jbir I, Slimane M, Ben Hassouna J, Ben Dhieb T, Bouzaiene H, Rahal K (2017) Identifying accessible prognostic factors for breast cancer relapse: a case-study on 405 histologically confirmed node-negative patients. World J Surg Oncol 15(1):206 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This published paper contains all of the data produced or analyzed during this investigation. Further enquiries can be directed to the corresponding author.