Abstract
Purpose
Esophageal cancer is the most common gastrointestinal tumor and is difficult to be eradicated with conventional treatment. Porphyrin-based photosensitizers (PSs) mediated photodynamic therapy (PDT) could kill tumor cells with less damage to normal cells. As the most widely used porphyrin-based photosensitizer in clinics, Photofrin II has excellent anti-tumor effect. However, it has some disadvantages such as weak absorption at near infrared region, the complexity of components and prolonged skin photosensitivity. Here series novel 5,15-diaryl-10,20-dihalogeno porphyrin derivatives were afforded and evaluated to develop more effective and safer photosensitizers for tumor therapy.
Methods
The photophysical properties and singlet oxygen generation rates of 5,15-diaryl-10,20-dihalogeno porphyrins (I1-6, II1-4) were tested. The cytotoxicity of I1-6 and II1-4 were measured by MTT assay. The pathway of cell death was studied by flow cytometry. In vivo photodynamic efficacy of I3 and II2-4 in Eca-109 tumor-bearing BABL/c nude mice were measured and histopathological analysis were examined.
Results
5,15-Diaryl-10,20-dihalogeno porphyrins I1-6 and II1-4 were synthesized. The longest absorption wavelength of these halogenated porphyrins (λmax = 660 nm) displayed a red shift around 30 nm compared to the unhalogenated porphyrins PS1 (λmax = 630 nm). The singlet oxygen generation rates of I1-6 and II1-4 were significantly higher than PS1 and HMME. All PSs mediated PDT showed obvious cytotoxic effect against Eca-109 cells compared to HMME in vitro and in vivo. Among these PSs, II4 exhibited appropriate absorption in the phototherapeutic window, higher 1O2 generation rate (k = 0.0061 s−1), the strongest phototoxicity (IC50 = 0.4 μM), lower dark toxicity, high generation of intracellular ROS in Eca-109 cells and excellent photodynamic anti-tumor efficacy in vivo. Besides, cell necrosis was induced by compound II4 mediated PDT.
Conclusion
All new compounds have obvious photodynamic anti-esophageal cancer effects. Among them, the photosensitizer II4 showed excellent efficacy in vitro and in vivo, which has the potential to become a photodynamic anti-tumor drug.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00432-022-04037-7.
Keywords: Photodynamic therapy, Photosensitizer, Halogenated porphyrin, Anti-tumor
Introduction
Esophageal cancer (Eca) is the common type of cancer and the leading cause of cancer deaths worldwide according to GLOBOCAN 2018. More than 570,000 people are newly diagnosed with esophageal carcinoma, and over 500,000 deaths are recorded annually (Bray et al. 2018; Uhlenhopp et al. 2020). PDT is a clinically approved novel method for the treatment of this cancer with many advantages including prominent selectivity, flexibility, minimum injury, and negligible toxicity to normal tissues (Wu et al. 2020). Exogenous light could activate the PSs from ground state to excited state whose energy could be transferred to surrounding ground oxygen to generate reactive oxygen species (ROS), such as singlet oxygen (1O2), which in turn could cause oxidative damage and kill cancer cells by reacting with biomolecules (Wu et al. 2020; Li et al. 2021a, b; Du et al. 2021; Li et al. 2021a, b; Ormond et al. 2013).
Porphyrin, also called the “color of life”, has drawn considerable attention in photodynamic therapy, cancer diagnosis, optoelectronic materials and other aspects because of its unique physical and chemical properties (Yu et al. 2020). Currently, as the representatives of porphyrin-based photosensitizers, Photofrin II and Hematoporphyrin monomethyl ether (HMME) are commonly used in photodynamic therapy (Massiot et al. 2018; Li et al. 2020; Zhu et al. 2018, Liao et al. 2017). Photofrin II is the first commercially available and most widely used photosensitizer due to its excellent anti-tumor effect. However, its clinical application is limited by the complexity of components and the prolonged skin photosensitivity (Li et al. 2021a, b). HMME, developed by our group, possesses a pair of isomers which has been clinically used for the treatment of nevus flammeus and tumors. The photophysical and photochemical properties of Photofrin II and HMME, such as the absorption ability at red light region, the 1O2 generation ability and the photodynamic effects, is still needed to be improved to develop more effective and safer photosensitizers for tumor therapy.
Among the porphyrin derivatives, numerous researches on meso-tetraarylporphyrins (Chatterjee et al., 2017) have been carried out. However, only very few studies were reported about meso-diarylporphyrins, namely 5,15-diarylporphyrins. The preliminary study was proceeded by Banfi et al. (2006) and Wiehe et al. (2001) showed that 5,15-diarylporphyrins exhibited significantly higher photocytotoxicity than Photofrin II. The dicationic 5,15-bis-[4-(3-trimethylammoniopropyloxy)phenyl] porphyrin (XF-73) was developed for the treatment of methicillin-resistant Staphylococcus aureus (MRSA) infections with satisfactory efficacy, safety and tolerability in clinical trials (Maisch et al. 2005). Constant and abundant amino acids supply is required for tumor survival to support numerous biochemical reactions, and for tumor proliferation to synthesize structural and functional proteins (Martinez-Outschoorn et al. 2017; Hosios et al. 2016). The approval of talaporfin (Laserphyrin®) which is the novel photosensitizer obtained by coupling chlorin e6 with aspartic acid proves that the introduction of amino acids into porphyrin-based derivatives is an effective strategy to improve the hydrophilicity and the biocompatibility (Kwitniewski et al. 2009; Serra et al. 2010; Tamiaki et al. 2014; Wang et al. 2008; Allison et al. 2004). A series of chlorin p6‑based water-soluble amino acid derivatives were synthesized by Meng et al (2016), and it was found that the aqueous solubility of aspartylchlorin p6 dimethylester was significantly better than its lead compound chlorin p6 dimethylester.
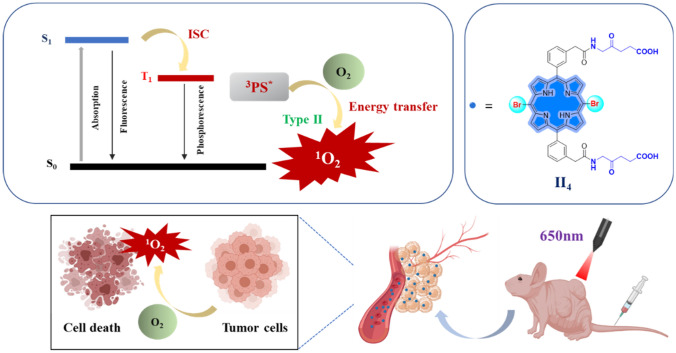
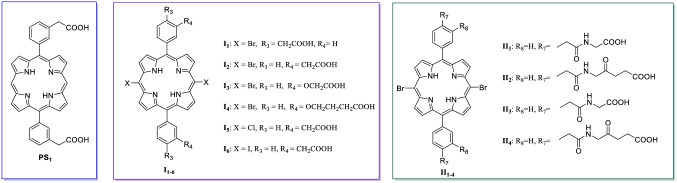
In the present study, 5,15-diaryl-10,20-dihalogeno porphyrins I1-6 and II1-4 were synthesized by introducing halogen atoms and amino acids groups into 5,15-diarylporphyrins, and their photodynamic activities in vitro and in vivo against esophageal cancer were investigated (Fig. 1).
Fig. 1.
The schematic diagram of II4 mediated PDT
Materials and methods
Materials
5,15-Diaryl-10,20-dihalogeno porphyrins (I1-6, II1-4) were synthesized in our laboratory (Fig. 2). All solvents and reagents were purchased from commercial suppliers and dichloromethane was used after further redistillation. HMME was donated by Shanghai Xianhui Pharmaceutical Co. Ltd. Thin-layer chromatography (TLC) analysis was carried out on silica gel plates GF254 and column chromatography was performed on silica gel (300–400 mesh). 1H NMR and 13C NMR spectra were recorded on a Bruker 400 or 600 MHz spectrometer. MALDI-TOF mass spectra were recorded using a JEOL JMS-S3000 Spiral-TOFMS (JEOL, Tokyo, Japan). UV–Vis absorption spectra were recorded on an UV–Vis spectrophotometer (Jasco Model V-530, Japan). Fluorescence spectra were measured on a fluorescence spectrophotometer (FluoroMax-4, France). The geometry optimization of PS1, I2, I5 and I6 in the DMSO was carried out by DFT calculations using the B3LYP functional with a 6-31G basis set for carbon, hydrogen, nitrogen, oxygen, chlorine, bromine atoms and LANL2DZ for iodine atoms. Graphics were prepared by OriginPro 9.0 and GraphPad Prism 8. All results were presented as mean ± SD. Comparison among the groups was determined by one-way ANOVA followed by Bonferroni correction for multiple testing. P< 0.05 was considered statistically significant.
Fig. 2.
The structures of 5,15-diaryl-10,20-dihalogeno porphyrins (I1-6, II1-4)
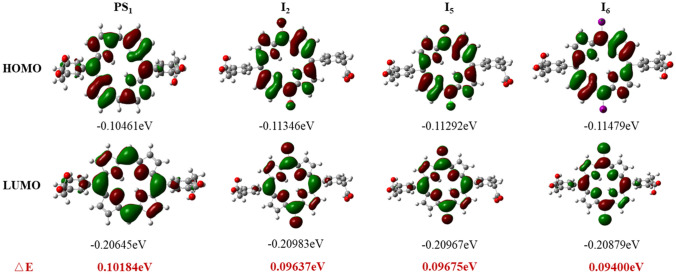
Density functional theory (DFT) studies
The geometry optimization of the compounds (PS1, I2, I5, I6) in DMSO was carried out by DFT calculations using the B3LYP functional with a 6-31G basis set for carbon, hydrogen, nitrogen, oxygen, chlorine, bromine atoms and LANL2DZ for iodine atoms. And then the FMOs of HOMO and LUMO energy (eV) level for the optimized structure of the products were calculated to evaluate the influence of the halogen atoms (Zhao et al. 2021).
Photophysical properties
UV–Vis absorption spectra of all PSs in DMSO were recorded on an ultraviolet visible spectrophotometer (Jasco Model V-530, Japan) at wavelengths from 300 to 800 nm. Fluorescence spectra were carried out using a fluorescence spectrometer (FluoroMax-4, France) in the range 500–800 nm using 425 nm excitation wavelength. All the measurements were carried out at room temperature.
Singlet oxygen generation
1,3-Diphenylisobenzofuran (DPBF) supplied by J&K Scientific was used for the detection of the singlet oxygen generation of PSs. Generally, 3 mL portions of DMF solution containing 30 μM DPBF and 1 μM PSs were placed in a sealed quartz cuvette and irradiated with 650 nm light (CNI MRL-III-650, China) at the 5 mW/cm2 laser intensity. The absorbance of the solution at 416 nm was measured every 10 s for an 80 s period with an ultraviolet visible spectrophotometer (Le et al. 2021; Durmuş et al. 2009). The natural logarithm values of absorption of DPBF at 416 nm were plotted against the irradiation time and fitted by a first-order linear least squares model to get the singlet oxygen generation rate of the photosensitized process (Zhang et al. 2014; Wolfgang et al. 1998). The rate of singlet oxygen generation was calculated by the following equation (Tang et al. 2005).
where [DPBF]t and [DPBF]0 respectively is the absorbance of DPBF after and prior irradiation. Values of k are singlet oxygen generation rates and t represent light duration.
In vitro photosensitizing efficacy
Cell lines and culture conditions
Human esophageal cancer cell line Eca-109 was obtained from the Type Culture Collection of the Chinese Academy of Sciences. All cell culture reagents were purchased from MesGen Biotech (Shanghai, China). Cells were cultured in normal RPMI-1640 culture medium with 10% fetal bovine serum (FBS), 50 units per mL penicillin and 50 mg mL−1 streptomycin in 5% CO2 at 37 ℃.
Cytotoxicity
The dark- and photocytotoxicity of PSs were analyzed in Eca-109 cells following the procedures as described in the literature (Tang et al. 2005). Eca-109 cells were cultured in RPMI-1640 medium with 10% (v/v) FBS, collected with 0.25% (w/v) trypsin, and seeded in 96-well plates at 5 × 104 cells per well. The cells were allowed to attach to the bottom of the wells for 24 h prior to starting the experiment. RPMI-1640 medium containing I1-6 and II1-4 in different concentrations (range from 0 to 30 μM) was administered to cells, and allowed to uptake for 24 h, and irradiated with different light doses (0, 2, 4, 8, 12 J/cm2) using an Nd: YAG laser at 650 nm. Non-irradiated cells were used to investigate the dark cytotoxicity. After treatment, the cell viability was determined by adding 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-terazolium bromide (MTT) solution (5 μg/mL in PBS) for 4 h at 37 ℃. Subsequently, the MTT solution was removed and 150 μL DMSO was added. The samples were shaken for 10 min at room temperature and their absorbance was measured by the microplate reader at 570 nm.
Intracellular ROS generation
The intracellular ROS generation of compounds I3, II2, II3 and II4 was measured by fluorescent probe 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA) (Wang et al. 2014; Rubio et al. 2014; Kim et al. 2011). Eca-109 cells were successively incubated with 1 μM photosensitizer for 4 h, and then irradiated by 650 nm laser (2 J/cm2, 40 mW/cm2) for 50 s, followed by incubating with DCFH-DA for 30 min. The fluorescence imaging was monitored by inverted fluorescence microscope (Leica, DMi8, Germany).
Flow cytometry analysis
Flow cytometry analysis was conducted using Annexin V-EGFP/PI Apoptosis Detection Kit (KeyGEN BioTECH, China) following the manufacturer’s protocol. Briefly, Eca-109 cells were seeded in a six-well plate and incubated with the compounds at the concentration of 4 μM for 4 h and then irradiated with a light dose of 2 J/cm2 (λ = 650 nm), followed by incubation for 6 h at 37 °C. After the treatment, the media and cells were collected for each sample. The cells were resuspended with binding buffer in a 12 × 75 test tube, and then 5 μL of Annexin V and 5 μL of PI staining solution were added to each tube followed by incubation for 20 min at room temperature in the dark. The fluorescence of the cells was immediately determined by BD FACS Calibur Flow cytometer (Franklin Lakes, NJ).
Different PSs mediated PDT in vivo
Animals model
The BALB/c nude mice (female, 18–20 g) were obtained from Shanghai SLAC Laboratory Animal Company and housed in an air-conditioned room at 23 ± 2 °C with dedicated pathogen-free barrier facilities. For the establishment of tumor model, the 5 × 106 Eca-109 cells in 1 mL PBS were subcutaneously injected into the right region of BALB/c mice. When tumors reached a volume range from 100–150 mm3, the following experiments were carried out. All animal procedures were performed according to the Guidelines of the Laboratory Protocol of Animal Handling and with approval from the Animal Care and Use Committee of Donghua University.
In vivo photosensitizing efficacy
The tumor xenograft mice were randomly divided into the following groups of five each: (1) control, (2) 120 J/cm2 light alone, (3) 2 mg/kg HMME + 120 J/cm2 light, (4) 2 mg/kg I3 + 120 J/cm2 light, (5) 2 mg/kg II2 + 120 J/cm2 light, (6) 2 mg/kg II3 + 120 J/cm2 light, (7) 2 mg/kg II4 + 120 J/ cm2 light. The control mice were injected with the same dose of PBS. Five minutes after the caudal vein injection, the tumor sites of mice were irradiated with laser light (400 mW/cm2) for 5 min, and the total light dose was 120 J/cm2. After the treatment, the length (L) and width (W) of tumors were measured with a caliper every other day. The tumor volume was calculated using the formula: V = L × W2 × 0.5. After 14 days post-treatment, the mice were sacrificed and the tumors were removed and weighed.
Histsological analysis
After 1 day-post treatment, tumor tissues from different groups were fixed with 4% paraformaldehyde fix solution for 24 h, dehydrated through graded alcohols, and embedded in paraffin. The paraffin-embedded section (6 μm thick) were stained with hematoxylin and eosin (H&E). Histopathological changes were observed under a light microscope (Nikon E600, Japan).
Results
Chemistry
5,15-Diaryl-10,20-dihalogeno porphyrins I1-6 were obtained by halogenation and hydrolysis of porphyrins synthesized by standard Lindsey protocol in 30–50% total yields, as shown in Scheme S1 (Vinogradova et al. 2009). The synthesis of 5,15-diaryl-10,20-dibromoporphyrins (II1-4) was shown in Scheme S2. An amide condensation reaction occurred among compounds (I1, I2) and amino-acid esters. The compounds (4a-d) were hydrolyzed to give the target compounds (II1-4) in 56–70% total yields. The structures of these porphyrins (I1-6, II1-4) were characterized by 1H NMR, 13C NMR and HR-MS (Figure S1–25).
Density functional theory (DFT) studies
In general, a smaller gap of HOMO and LUMO energy level (ΔE) corresponds to a stronger driving force toward the electron-transfer state. Frontier molecular orbitals (FMOs) of the HOMO and LUMO energy (eV) levels for the optimized structures of PS1, I2, I5 and I6 (Fig. 3) were calculated. According to our DFT calculations, ΔE of compounds I2 (0.09637 eV), I5 (0.09675 eV) and I6 (0.09400 eV) were smaller than compound PS1 (0.10184 eV) due to the existence of halogen atoms (Fig. 3). In addition, it was observed that their corresponding ΔE values decreased with the increase of relative atomic mass of halogen atoms of meso-substituents.
Fig. 3.
FMOs and ΔE of PS1, I2, I5, I6
Photophysical properties
The UV–Vis absorption spectrum of the 5,15-diaryl-10,20-dihalogeno porphyrins (I1-6 and II1-4) in DMSO were presented in Fig. 4A. All compounds exhibited an intense B band at ~ 425 nm, and four Q bands at around 523, 558, 602 and 660 nm. Among these compounds, I6 showed the longest absorption wavelength at 664 nm. The unhalogenated porphyrins PS1 had fluorescence while 5,15-diaryl-10,20-dihalogeno porphyrins (I1-6 and II1-4) have no fluorescence (Fig. 4B).
Fig. 4.
The spectrum properties of compounds. A UV-Vis absorption spectra of compounds PS1, I1-6 and II1-4 at 10 μM in DMSO. B Fluorescence excitation and emission spectra of compounds PS1 in DMSO at the concentration of 10 μM
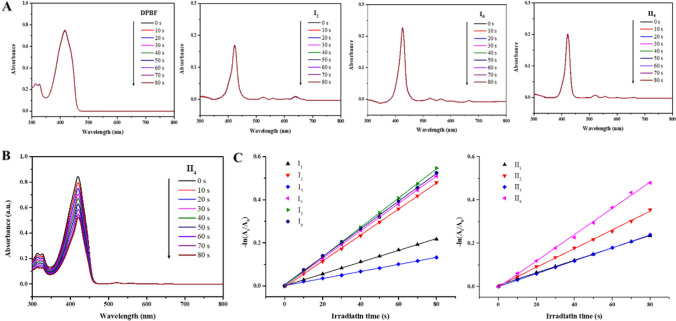
Singlet oxygen generation
To evaluate the ability to produce singlet oxygen, 1,3-diphenylisobenzofuran (DPBF) was used as the quencher. The generation rate of singlet oxygen was measured by monitoring the decrease in absorbance at 415 nm with regular intervals of 10 s upon irradiation with 650 nm laser light. As shown in Fig. 5A, decomposition of DPBF did not occur in the absence of singlet oxygen. The representative halogenated porphyrins (I2, I6, II4) were irradiated with 650 nm laser and their absorbance did not decrease with the increase of irradiation time, which indicated that halogenated porphyrins had good photostability. Time-dependent changes of the absorption spectra of various PSs containing DPBF under light irradiation were shown in Fig. 5B, C and Figure S26. The singlet oxygen generation rates of all halogeno compounds were summarized in Table 1. The results showed that the ability of singlet oxygen generation of halogenated porphyrins was significantly higher than that of unhalogenated porphyrins PS1 and HMME.
Fig. 5.
The singlet oxygen generation of compounds. A Photodegradation of DPBF and compounds I2, I6, II4 under 650 nm laser irradiation (5 mW/cm2) every 10 s. B Photodecomposition of DPBF in the presence of II4 under 650 nm laser irradiation (5 mW/cm2) every 10 s. C First-order plot for the photodecomposition of DPBF after PDT with the compounds
Table 1.
Singlet oxygen generation rate of compounds I1-6, II1-4
| Compound | k× 10–2( s−1) | Compound | k × 10–2(s−1) |
|---|---|---|---|
| I1 | 0.27 | II1 | 0.29 |
| I2 | 0.60 | II2 | 0.43 |
| I3 | 0.16 | II3 | 0.30 |
| I4 | 0.64 | II4 | 0.61 |
| I5 | 0.68 | PS1 | 0.014 |
| I6 | 0.64 | HMME | 0.026 |
In vitro photosensitizing efficacy
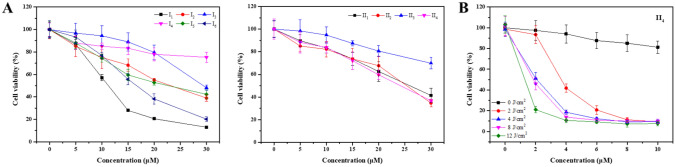
MTT assays were used to test the in vitro cytotoxicity of the target compounds against Eca-109 cells. As shown in Fig. 6A and Table 2, the cell viabilities after the treatment with compounds I3-4, and II1-4 at 10 μM were higher than 80% under dark condition while other compounds were lower than 80% under the same conditions, which suggested that compounds I3-4, and II1-4 had negligible dark cytotoxicity. There was little difference between the IC50 values of halogenated porphyrins (I2, I5 and I6) with Cl, Br and I under dark or light irradiation, as shown in Figure S27. The IC50 values of all target compounds were evidently smaller than HMME under light irradiation of 12 J/cm2 (Table 2). It was noteworthy that significant decrease in cell viability of Eca-109 cells after the treatment with compounds I1-3, I5-6, and II1-4 at concentration of 4 μM and a light dose of 12 J/cm2 was observed (Figure S27 and Fig. 6B), which indicated that these compounds had obvious photodynamic activities against Eca-109 cells.
Fig. 6.
The efficacy of compounds against Eca-109 cells in vitro. A Dark cytotoxicity of compound I1-6, II1-4 at concentrations ranging from 0 to 30 μM in Eca-109 cells. B The cell viabilities treated by II4 at concentrations ranging from 2 to 10 μM under different light doses. Data represents mean ± SD
Table 2.
IC50 values of I1-6, II1-4 in Eca-109 cells upon different light dose irradiation
| Compound | IC50 (μM) | ||||
|---|---|---|---|---|---|
| 0 J/cm2 | 2 J/cm2 | 4 J/cm2 | 8 J/cm2 | 12 J/cm2 | |
| I1 | 11.1 | 8.1 | 6.2 | 4.6 | 3.5 |
| I2 | 23.4 | 7.6 | 4.8 | 3.9 | 3.0 |
| I3 | 28.6 | 4.6 | 3.9 | 3.2 | 2.4 |
| I4 | > 30 | > 30 | 23.3 | 14.0 | 7.7 |
| I5 | 22.2 | 20.0 | 5.8 | 3.7 | 2.9 |
| I6 | 16.5 | 10.0 | 6.4 | 3.8 | 2.5 |
| II1 | 26.6 | 13.8 | 7.2 | 4.1 | 2.2 |
| II2 | 26.3 | 4.9 | 2.7 | 1.9 | 0.9 |
| II3 | > 30 | 5.4 | 4.2 | 3.4 | 1.6 |
| II4 | 24.4 | 3.9 | 1.9 | 1.6 | 0.4 |
| HMME | 27.9 | 22.4 | 18.6 | 15.0 | 9.2 |
Intracellular ROS generation
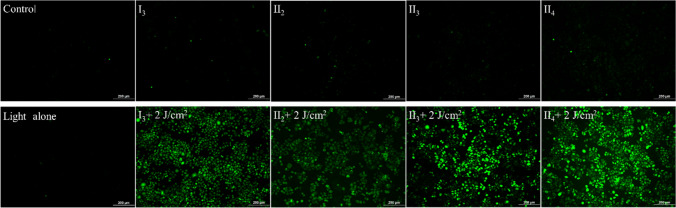
To confirm ROS generation ability of compounds in Eca-109 cells, fluorescence probe DCFH-DA was chosen as the indicator of 1O2 after PDT treatment. As illustrated in Fig. 7, the cells in the control group, light alone group and PSs alone group exhibited dim green fluorescence, while the cells in PDT groups showed intense green fluorescence under same irradiation condition. It indicated that compounds I3, II2, II3 and II4 could generate ROS under irradiation in Eca-109 cells. The fluorescence intensity of II4-PDT group was visually higher than I3-PDT group, II2-PDT group and II3-PDT group.
Fig. 7.
The fluorescence images of Eca-109 cells incubated with different PSs and probe DCFH-DA under light irradiation. Scale bar: 200 μm
Flow cytometry analysis
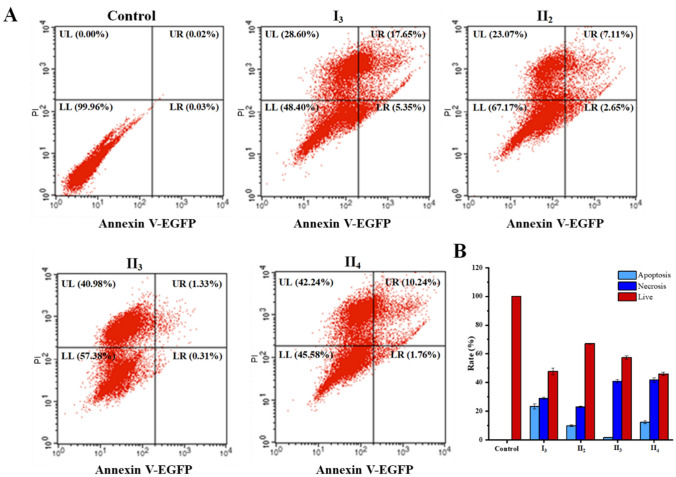
Tumor cells are killed by PDT through apoptotic and/or non-apoptotic (necrosis, autophagy) pathways, through damaging tumor vasculature and immune response. The effects relied on the type, dosage and localization of PSs, incubation time, oxygen level, genotype of cells subjected to PDT etc. Apoptosis is likely caused by the reactive oxygen radicals produced by the photodynamic effect of the PSs localized in mitochondria, while necrosis is the major form of tumor cells death induced by photosensitizers that localize in plasma membrane and lysosomes (Chilakamarthi and Giribabu 2017). To investigate the cell death induced by different PSs, Eca-109 cells after PDT were incubated for 5 h and stained with the Annexin V-FITC/PI apoptosis assay kit for flow cytometry analysis. As shown in Fig. 8, no necrotic cells were detected in the control group. After 5 h post-PDT, the proportion of living cells were decreased in all PDT-treatment groups, especially dropped to 48.40 and 45.58% in I3 and II4 groups respectively. II4-PDT mainly induced cell necrosis while I3-PDT could induce cell necrosis and apoptotic.
Fig. 8.
The extent and mode of cell death induced by different PSs-PDT. A Flow cytometric assay of I3, II2, II3 and II4 at 4 μM exposed to 2 J/cm2 of light. LL: annexin V (−) PI (–), lived cell; LR: annexin V (+) PI (−), early apoptotic cells; UR: annexin V (+) PI (+), late apoptotic cells; UL annexin V (−), necrotic cells. B Histogram of apoptotic cells, necrosis cells and live cells after the treatment. Data represents mean ± SD
Different PSs mediated PDT in vivo
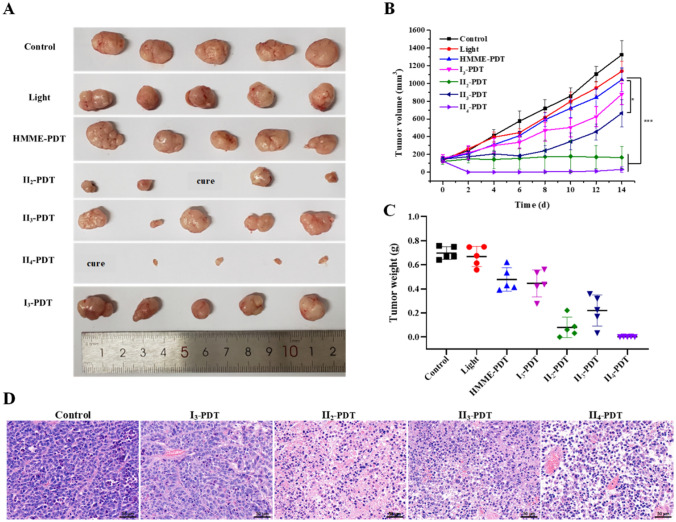
Since compounds I3, II2, II3 and II4 had more obvious photodynamic cytotoxicity than others under the same conditions, their photodynamic efficacy in Eca-109 tumor-bearing BABL/c nude mice were evaluated. The compounds at a dose of 2 mg/kg were injected intravenously into mice, followed by irradiation of 120 J/cm2 light at tumor site for single time. As shown in Fig. 9A–C, almost negligible inhibition of tumor growth was observed from the mice treated with light only. Notably, II2 and II4 showed more prominent anti-tumor PDT effect compared to the control group, light group and HMME group, especially II4. After 1-day post treatment, the tumor tissue sections were separated then stained with hematoxylin and eosin (H&E) reagent. As shown in Fig. 9D, significant tumor tissue damage in the PDT group could be observed under microscopy compared to the control group. The results of the tumor growth curve and histological examination showed that II4 had the best photodynamic antitumor activity in vivo.
Fig. 9.
Evaluation of tumor growth inhibition in the Eca-109-tumor-bearing model mediated by PDT. A Tumor images after 14 days. B Tumor growth curves after different treatments. *P < 0.05, **P < 0.001, ***P < 0.0001 vs HMME-PDT group. C Tumor weight. D Histological sections of tumor tissues stained with hematoxylin and eosin. Scale bar = 50 μm
Discussion and conclusion
In this study, series novel photosensitizers were prepared and their photodynamic activities were investigated. The longest absorption wavelength of halogenated porphyrins was superior to unhalogenated porphyrins as shown in Fig. 4, which was correlated with their small ΔE calculated by the DFT. It is interesting that all halogenated porphyrins showed no fluorescence emission,which is caused by the corresponding smaller ΔE values to transfer most of the first excited state populations of PSs into triplet excited state. In addition, as shown in Table 1, the singlet oxygen generation rates of all halogeno compounds were remarkably higher than PS1 and HMME, because the increase of the triplet excited state populations of halogenated porphyrins was beneficial to the generation of singlet oxygen. The singlet oxygen generation rates of porphyrin-amino acid conjugates have been improved to a certain extent, especially bromoporphyrins linked with 5-aminolevulinic acid (II2, II4). However, the singlet oxygen generation rates of bromoporphyrin (I2), chloroporphyrin (I5) and iodoporphyrin (I6) are slightly different. It was also showed that all compounds (I1-6 and II1-4) had more obvious photodynamic anti-Eca-109 cell activities in vitro than compounds HMME at the same treatment conditions, and the compounds I3-4 II1-4 had negligible dark cytotoxicity. Moreover, with 12 J/cm2 irradiation of 650 nm laser, compounds II4 at 4 μM had the excellent anti-tumor effect and low dark toxicity in vitro. Besides, compounds I3, II2, II3 and II4 could generate intracellular ROS under irradiation, especially II4-PDT showed high generation level of intracellular ROS. Necrosis is the major form of tumor cells death induced by PSs that localize in plasma membrane and lysosomes. Cell necrosis rate of II4-PDT was significantly higher than that of I3-PDT, which indicated their subcellular localization may be different. II4-PDT mainly induced cell necrosis, while I3-PDT could induce cell necrosis and apoptotic. In addition, compounds II3 and II4 with amino acid groups have higher cell necrosis rates compared to compound I3 when the substituents located at the same positions, while compound II4 with substituents on the meta position of the benzene ring had higher one compared to compound II2 with substituents on the para position when the substituents were the same (Fig. 8). These results implied that the kind and position of the substituents on benzene ring greatly affected the cell death pathway. Notably, compound II4 showed more prominent anti-tumor photodynamic efficacy in Eca-109 tumor-bearing BABL/c nude mice (Fig. 9), which was consistent with the phototoxicity in vitro and intracellular ROS generation level. In summary, II4, with 5-aminolevulinic acid group at periphery of tetrapyrrole ring, showed excellent photodynamic efficacy on Eca-109 cells in vitro and in vivo. So it has the potential to act as a photodynamic anti-tumor drug.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21977016), Foundation of Shanghai Science and Technology Committee (No. 19410711000, 20430730900, 20490740400, 21430730100).
Abbreviations
- PDT
Photodynamic therapy
- PSs
Photosensitizers
- ROS
Reactive oxygen species
- 1O2
Singlet oxygen
- ISC
Intersystem crossing
- DFT
Density functional theory
- TLC
Thin layer chromatography
- NMR
Nuclear magnetic resonance
- MS
Mass spectra
- FMOs
Frontier molecular orbitals
- HMME
Hematoporphyrin monomethyl ether
- DMSO
Dimethyl sulfoxide
- DMF
Dimethylformamide
- DPBF
1,3-Diphenylisobenzofuran
- DDQ
2,3-Dicyano-5,6-dichlorobenzoquinone
- H&E reagent
Hematoxylin–eosin reagent
- FBS
Fetal bovine serum
- PBS
Phosphate buffered saline
- MTT
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-terazolium bromide
Author contributions
The manuscript was written through contributions of all authors. All authors have read and agreed to the final version of the manuscript.
Declarations
Conflict of interest
The authors have declared no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yi Jia Yan, Email: y_yan11@sina.com.
Zhi Long Chen, Email: zlchen1967@qq.com.
References
- Allison RR, Downie GH, Sibata CH et al (2004) Photosensitizers in clinical PDT. Photodiagn Photodyn 1(1):27–42. 10.1016/S1572-1000(04)00007-9 [DOI] [PubMed] [Google Scholar]
- Banfi S, Caruso E, Gramatica P et al (2006) Comparison between 5,10,15,20-tetraaryl- and 5,15-diarylporphyrins as photosensitizers: synthesis, photodynamic activity, and quantitative structure-activity relationship modeling. J Med Chem 49(11):3293–3304. 10.1021/jm050997m [DOI] [PubMed] [Google Scholar]
- Bray F, Ferlay J, Jemal A et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424. 10.3322/caac.21492 [DOI] [PubMed] [Google Scholar]
- Chatterjee T, Shetti VS, Ravikanthet M et al (2017) Heteroatom-containing porphyrin analogues. Chem Rev 117:3254–3328. 10.1021/acs.chemrev.6b00496 [DOI] [PubMed] [Google Scholar]
- Chilakamarthi U, Giribabu L (2017) Photodynamic therapy: past, present and future. Chem Rec 17:775–802. 10.1002/tcr.201600121 [DOI] [PubMed] [Google Scholar]
- Du J, Shi T, Peng X et al (2021) Enhanced photodynamic therapy for overcoming tumor hypoxia: from microenvironment regulation to photosensitizer innovation. Coordin Chem Rev 427(6):213604. 10.1016/j.ccr.2020.213604 [Google Scholar]
- Durmuş M, Erdoğmuş A, Nyokong T et al (2009) The synthesis and photophysicochemical behaviour of novel water-soluble cationic indium(III) phthalocyanine. Dyes Pigments 82(2):244–250. 10.1016/j.dyepig.2009.01.008 [Google Scholar]
- Hosios AM, Hecht VC, Vanderheiden M et al (2016) Amino acids rather than glucose account for the majority of cell mass in proliferating mammalian cells. Dev Cell 36(5):540–549. 10.1016/j.devcel.2016.02.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong HG, Choi MS et al (2016) Design and properties of porphyrin-based singlet oxygen generator. Isr J Chem 56:110–118. 10.1002/ijch.201500026 [Google Scholar]
- Kim CH, Chung CW, Kang DH et al (2011) Effect of 5-aminolevulinic acid-based photodynamic therapy via reactive oxygen species in human cholangiocarcinoma cells. Int J Nanomed 6:1357–1363. 10.2147/ijn.s21395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwitniewski M, Juzeniene A, Moan J et al (2009) Diamino acid derivatives of PpIX as potential photosensitizers for photodynamic therapy of squamous cell carcinoma and prostate cancer: in vitro studies. J Photochem Photobiol B Biol 94(3):214–222. 10.1016/j.jphotobiol.2008.11.005 [DOI] [PubMed] [Google Scholar]
- Le NA, Babu V, Spingler B et al (2021) Photostable platinated bacteriochlorins as potent photodynamic agents. J Med Chem 64(10):6792–6801. 10.1021/acs.jmedchem.1c00052 [DOI] [PubMed] [Google Scholar]
- Li X, Lovell JF, Chen X et al (2020) Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat Rev Clin Oncol 17(11):657–674. 10.1038/s41571-020-0410-2 [DOI] [PubMed] [Google Scholar]
- Li J, Ou H, Ding D et al (2021a) Recent progress in boosted PDT induced immunogenic cell death for tumor immunotherapy. Chem Res Chin Univ 37(1):83–89. 10.1007/s40242-021-0402-5 [Google Scholar]
- Li MY, Gao YH, Chen ZL et al (2021b) Synthesis and evaluation of novel fluorinated hematoporphyrin ether derivatives for photodynamic therapy. Bioorg Chem 107:104528. 10.1016/j.bioorg.2020.104528 [DOI] [PubMed] [Google Scholar]
- Liao PY, Wang XR, Chen ZL et al (2017) Synthesis of 2-morpholine tetraphenylporphyrins and their photodynamic activities. Bioorg Chem 71:299–304. 10.1016/j.bioorg.2017.02.015 [DOI] [PubMed] [Google Scholar]
- Maisch T, Bosl C, Abels C et al (2005) Photodynamic effects of novel XF porphyrin derivatives on prokaryotic and eukaryotic cells. Antimicrob Agents Chemother 49(4):1542–1552. 10.1128/AAC.49.4.1542-1552.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Outschoorn UE, Peiris-Pages M, Lisanti MP et al (2017) Cancer metabolism: a therapeutic perspective. Nat Rev Clin Oncol 14(1):11–31. 10.1038/nrclinonc.2017.1 [DOI] [PubMed] [Google Scholar]
- Massiot J, Rosilio V, Makky A et al (2018) Newly synthesized lipid-porphyrin conjugates: evaluation of their self-assembling properties, their miscibility with phospholipids and their photodynamic activity in vitro. Chem 24(72):19179–19194. 10.1002/chem.201804865 [DOI] [PubMed] [Google Scholar]
- Meng Z, Yu B, Yao J et al (2016) Chlorin p6-based water-soluble amino acid derivatives as potent photosensitizers for photodynamic therapy. J Med Chem 59(10):4999–5010. 10.1021/acs.jmedchem.6b00352 [DOI] [PubMed] [Google Scholar]
- Ormond AB, Freeman HS et al (2013) Dye sensitizers for photodynamic therapy. Materials (Basel) 6(3):817–840. 10.3390/ma6030817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubio N, Verrax J, Agostinis JP et al (2014) p38(MAPK)-regulated induction of p62 and NBR1 after photodynamic therapy promotes autophagic clearance of ubiquitin aggregates and reduces reactive oxygen species levels by supporting Nrf2-antioxidant signaling. Free Radic Biol Med 67:292–303. 10.1016/j.freeradbiomed.2013.11.010 [DOI] [PubMed] [Google Scholar]
- Serra VV, Zamarrón A, Sanz-Rodríguez F et al (2010) New porphyrin amino acid conjugates: Synthesis and photodynamic effect in human epithelial cells. Bioorg Med Chem 18(16):6170–6178. 10.1016/j.bmc.2010.06.030 [DOI] [PubMed] [Google Scholar]
- Tamiaki H, Isoda Y, Machida S et al (2014) Synthesis of chlorophyll-amino acid conjugates as models for modification of proteins with chromo/fluorophores. Bioorg Med Chem 22(4):1421–1428. 10.1016/j.bmc.2013.12.059 [DOI] [PubMed] [Google Scholar]
- Tang W, Xu H, Kopelman R et al (2005) Photodynamic characterization and in vitro application of methylene blue-containing nanoparticle platforms. Photochem Photobiol 81:242–249. 10.1562/2004-05-24-ra-176.1 [DOI] [PubMed] [Google Scholar]
- Uhlenhopp DJ, Then EO, Gaduputi V et al (2020) Epidemiology of esophageal cancer: update in global trends, etiology and risk factors. Clin J Gastroenterol 13(6):1010–1021. 10.1007/s12328-020-01237-x [DOI] [PubMed] [Google Scholar]
- Vinogradova EV, Enakieva YY, Tsivadze AY et al (2009) Synthesis of meso-substituted porphyrins as precursors in creating highly ordered electroluminescent polymer materials. Prot Met Phys Chem 45(5):529–534. 10.1134/S2070205109050050 [Google Scholar]
- Wang HM, Jiang JQ, Liu XY et al (2008) Porphyrin with amino acid moieties: a tumor photosensitizer. Chem Biol Interact 172(2):154–158. 10.1016/j.cbi.2007.11.016 [DOI] [PubMed] [Google Scholar]
- Wang B, Wang JH, Chu PK et al (2014) Rose-bengal-conjugated gold nanorods for in vivo photodynamic and photothermal oral cancer therapies. Biomater 35:1954–1966. 10.1016/j.biomaterials.2013.11.066 [DOI] [PubMed] [Google Scholar]
- Wiehe A, SimonenkoB RÖDer, EJ et al (2001) Hydrophilicity vs hydrophobicity—varying the amphiphilic structure of porphyrins related to the photosensitizer m-THPC. J Porphyr Phthalocya 5(10):758–761. 10.1002/jpp.389 [Google Scholar]
- Wolfgang S et al (1998) Singlet oxygen quantum yields of different photosensitizers in polar solvents and micellar solutions. J Porphyr Phthalocya 2(2):145–158. 10.1002/(SICI)1099-1409(199803/04)2:2%3c145::AID-JPP60%3e3.0.C [Google Scholar]
- Wu J, Sha J, Wang P et al (2020) Recent advances in theranostic agents based on natural products for photodynamic and sonodynamic therapy. View 1(3):20200090. 10.1002/VIW.20200090 [Google Scholar]
- Yu WM, Zhen WQ, Liu YM et al (2020) Porphyrin-based metal-organic framework compounds as promising nanomedicines in photodynamic therapy. ChemMedChem 15:1766–1775. 10.1002/cmdc.202000353 [DOI] [PubMed] [Google Scholar]
- Zhang LJ, Bian J, Chen ZL et al (2014) Photosensitizing effectiveness of a novel chlorin-based photosensitizer for photodynamic therapy in vitro and in vivo. J Cancer Res Clin Oncol 140(9):1527–1536. 10.1007/s00432-014-1717-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X, Yao Q, Long S et al (2021) An approach to developing cyanines with simultaneous intersystem crossing enhancement and excited-state lifetime elongation for photodynamic antitumor metastasis. J Am Chem Soc 143:12345–12354. 10.1021/jacs.1c06275 [DOI] [PubMed] [Google Scholar]
- Zhu W, Gao YH, Chen ZL et al (2018) Comparison between porphin, chlorin and bacteriochlorin derivatives for photodynamic therapy: synthesis, photophysical properties, and biological activity. Eur J Med Chem 160:146–156. 10.1016/j.ejmech.2018.10.005 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.