Abstract
Liver cancer represents a major global health concern, with projections indicating that the number of new cases could surpass 1 million annually by 2025. Hepatocellular carcinoma (HCC) constitutes around 90% of liver cancer cases and is primarily linked to factors incluidng aflatoxin, hepatitis B (HBV) and C (HCV), and metabolic disorders. There are no obvious symptoms in the early stage of HCC, which often leads to delays in diagnosis. Therefore, HCC patients usually present with tumors in advanced and incurable stages. Several signaling pathways are dis-regulated in HCC and cause uncontrolled cell propagation, metastasis, and recurrence of HCC. Beyond the frequently altered and therapeutically targeted receptor tyrosine kinase (RTK) pathways in HCC, pathways involved in cell differentiation, telomere regulation, epigenetic modification and stress response also provide therapeutic potential. Investigating the key signaling pathways and their inhibitors is pivotal for achieving therapeutic advancements in the management of HCC. At present, the primary therapeutic approaches for advanced HCC are tyrosine kinase inhibitors (TKI), immune checkpoint inhibitors (ICI), and combination regimens. New trials are investigating combination therapies involving ICIs and TKIs or anti-VEGF (endothelial growth factor) therapies, as well as combinations of two immunotherapy regimens. The outcomes of these trials are expected to revolutionize HCC management across all stages. Here, we provide here a comprehensive review of cellular signaling pathways, their therapeutic potential, evidence derived from late-stage clinical trials in HCC and discuss the concepts underlying earlier clinical trials, biomarker identification, and the development of more effective therapeutics for HCC.
Subject terms: Drug development, Translational research, Target identification
Introduction
Liver cancer, primarily HCC, is among the most challenging tumors to treat, ranking sixth globally in incidence and third in cancer-related deaths.1,2 HBV infection is currently the leading cause of HCC, accounting for approximately 50% of all cases. However, viral infections of HBV and HCV are declining annually due to the widespread use of antiviral drugs and vaccines, while non-alcoholic steatohepatitis (NASH) linked to metabolic disorders and diabetes as a cause of HCC is on the rise.3
The molecular etiology of HCC differs depending on specific etiologies and genotoxic damage. The distinct molecular subtypes and immune responses elicited by HBV-associated HCC differ from those in NASH-induced HCC. A deep insight into the molecular mechanisms of HCC caused by various etiologies is required to formulate more rational therapeutic strategies. This process is complicated by the notion that only approximately 25% of HCC have actionable mutations. Nearly half of HCC patients carry at least one recurring oncogenic mutation such as TP53, CTNNB1, or TERT, However, most of these mutations lack effective targeting options with conventional pharmaceutical agents.4 While inhibitors designed to target mutations in the TERT promoter and components of the WNT/β-catenin signaling pathway have been developed, achieving satisfactory therapeutic effects remain elusive.5,6 Most other mutations remain non-targetable. Studying the effect that these mutations have on downstream signaling pathways can provide clues to the future development of drugs that target these currently undruggable mutations.
The major therapeutic approaches for early stage HCC currently encompass liver resection and transplantation. In conventional approaches, local ablation utilizing radiofrequency serves as the predominant non-surgical intervention for early-stage HCC.7 Transarterial chemoembolization (TACE) has also been the predominant treatment method and the benchmark treatment over the past two decades for intermediate-stage HCC.8 Traditional methods for HCC treatment are being challenged by targeted therapy and immunotherapy (Fig. 1a). The first “targeted” therapy for advanced HCC patients is sorafenib, which gained FDA approval as a first-line therapy following successful phase III clinical trials SHARP and Asia-Pacific. The word “targeted” is between apostrophes because it is not a targeted drug in the narrow sense of the word, as it targets a number of related kinases, including RAF, MEK, VEGFR and PDGFR. The first-line treatment with sorafenib has significantly increased the median overall survival (OS), rising from 7.9 to 10.7 months. Following sorafenib’s approval, lenvatinib become another first-line agent for HCC,9 demonstrated prolonged anti-tumor activity, particularly in patients with portal vein invasion. The success of IMbrave150 trial in 2020 heralded a groundbreaking epoch in HCC treatment using anti PD-L1 immunotherapy (atezolizumab) synergistically intertwines with anti-angiogenic therapy (bevacizumab). This strategy showed superiority over sorafenib in terms of OS, progression-free survival (PFS), and objective response rate (ORR). Recently, the strategy also showed significantly improved recurrence-free survival (RFS) in early stage HCC.10 Similarly, in November 2022, FDA approved the combination of durvalumab, an anti-PD-L1 antibody, and tremelimumab, an anti-CTLA-4 (the cytotoxic T-lymphocyte-associated protein 4) antibody, which demonstrated remarkable therapeutic outcome in the HIMALAYA trial, achieving a 4-year OS rate of 25.2% for advanced HCC patients. Despite remarkable advancement, a minority of patients with advanced disease experience lasting clinical benefits, highlighting substantial therapeutic complexities.11
Fig. 1.
The development of targeted therapy and treatment regimens over time in HCC. a Describing the history of first-line and second-line therapy in HCC during 2007–2024. FDA-approved agents or agents in Phase III clinical trials framed by red and yellow colors, separately. The red represents FDA-approved drugs and black represents targeted therapy drugs. b Summarizing the active clinical trials over the last five years and emphasizing treatment strategies in Phase III trials. Information from the ClinicalTrials.gov registry, and those with the status of “not yet recruiting”, recruiting”, enrolling by invitation”, “active, not recruiting”, or “completed” were considered active and counted. The green color represents early Phase I, blue represents Phase I, orange represents Phase II, and red represents Phase III. Figure was created with biorender.com
In the last five years, phase II trials constitute over half of all clinical trials in HCC (Fig. 1b). The focus of phase III clinical trials has predominantly shifted towards second-line investigations and combination therapies (Table 1), The current focus is on the development of more potent and safer agents that target immune checkpoint pathways and angiogenesis-related pathways, such as the vascular endothelial growth factor receptor (VEGFR), transforming growth factor β (TGF-β) signaling, among others. Exploring epigenetic modifications and dysregulated pathways, including MAPK, PI3K-AKT, and Janus Kinases (JAKs)/signal transducers and activators of transcription (STATs) pathway, can be beneficial in identifying drug combinations that are both safer and more effective.
Table 1.
Phase III clinical trials of first- or second-line therapeutic strategies for hepatocellular carcinoma initiated in the last five years
| Strategy | Test Drugs/Regimen | Comparator | Line | Targets | Status and results | Estimated primary completion date | Trial identifier |
|---|---|---|---|---|---|---|---|
| Targeted therapy | Namodenoson | None | Second | ADORA3 | Recruiting | 25-Feb | NCT05201404 |
| Immunotherapy | AlloStim | FOLFOX regimen/ Sorafenib / Lenvatinib | First | Th1-like cells with anti-CD3/CD28 microbeads | Recruiting | 1-Aug-25 | NCT05033522 |
| Durvalumab + Tremelimumab | None | First | PD-L1; CTLA-4 | Recruiting | 30-Jun-25 | NCT05883644 | |
| Durvalumab + Tremelimumab | NR | First | PD-L1; CTLA-4 | Recruiting | 31-Mar-25 | NCT05557838 | |
| IBI310 + Sintilimab | Sorafenib | First | CTLA-4; PD-1 | Recruiting | 1-Dec-23 | NCT04720716 | |
| Nivolumab + Ipilimumab | Sorafenib / Lenvatinib | First | PD-1; CTLA-4 | Active, not recruiting | 6-Jul-26 | NCT04039607 | |
| PD-1/PD-L1 inhibitor hepatic artery infusion | PD-1/PD-L1 inhibitor vein infusion | First | PD-1/PD-L1 | Recruiting | 1-Jan-25 | NCT03949231 | |
| Targeted therapy combined with immunotherapy | Livmoniplimab + Budigalimab | Atezolizumab + Bevacizumab / Durvalumab + Tremelimumab | First | LRRC32, TGFβ1; PD-1 | Recruiting | 6-Sep-30 | NCT06109272 |
| Atezolizumab + Bevacizumab + Tiragolumab | Atezolizumab+ Bevacizumab | First | PD-L1; VEGF-A; TIGIT | Recruiting | 1-Sep-26 | NCT05904886 | |
| Atezolizumab + Bevacizumab + Ipilimumab | Atezolizumab+Bevaciz umab | First | PD-L1; VEGF-A; CTLA-4 | Not yet recruiting | 1-Sep-24 | NCT05665348 | |
| Lenvatinib + Rulonilimab | Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Recruiting | 1-Aug-25 | NCT05408221 | |
| Atezolizumab + Bevacizumab | None | First | PD-L1; VEGF-A | Active, not recruiting | 27-Apr-24 | NCT04732286 | |
| Atezolizumab + Bevacizumab | TACE | Not reported | PD-L1; VEGF-A | Recruiting | 1-Apr-25 | NCT04803994 | |
| Atezolizumab + Lenvatinib / Sorafenib | Lenvatinib / Sorafenib | Second | PD-1; VEGFR, FGFR, PDGFR, RET, KIT; VEGFR, PDGFR, RAF, FLT3, KIT, FGFR, RET | Recruiting | 23-Sep-24 | NCT04770896 | |
| SCT-I10A + SCT510 | Sorafenib | First | VEGF; PD-1 | Active, not recruiting | 24-Apr | NCT04560894 | |
| Bevacizumab + Toripalima | Sorafenib | First | VEGF-A; PD-1 | Active, not recruiting | 31-Oct-23 | NCT04723004 | |
| Atezolizumab + Bevacizumab | None | First | PD-L1; VEGF-A | Active, not recruiting | 13-Aug-24 | NCT04487067 | |
| Lenvatinib + Toripalimab | Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Active, not recruiting | 1-May-24 | NCT04523493 | |
| Anlotinib + AK105 | Sorafenib | Not reported | VEGFR, PDGFR, FGFR, KIT; PD-1 | Not yet recruiting | 30-Jun-24 | NCT04344158 | |
| Lenvatinib + Nofazinlimab | Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Active, not recruiting | 30-Jun-25 | NCT04194775 | |
| Apatinib + Camrelizumab | Sorafenib | First | VEGFR; PD-1 | Completed | 8-Feb-22 | NCT03764293 | |
| Lenvatinib + Pembrolizumab | Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Active, not recruiting | 21-Jun-22 | NCT03713593 | |
| Cabozantinib + Atezolizumab | Cabozantinib / Sorafenib | First | MET, VEGFR, ROS1, RET, AXL, NTRK, KIT; PD-L1 | Active, not recruiting | 8-Mar-21 | NCT03755791 | |
| Atezolizumab + Bevacizumab | Sorafenib | First | PD-L1; VEGF-A | Completed | 31-Aug-20 | NCT03434379 | |
| Localized TACE/HAIC therapy combined with systemic therapy | HAIC-FOLFOX + Lenvatinib + Toripalimab | HAIC-FOLFOX + Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Recruiting | 26-Jul-25 | NCT06201065 |
| TACE + Bevacizumab + Sintilimab | TACE + Lenvatinib | First | VEGF-A; PD-1 | Recruiting | 31-Jul-27 | NCT05985798 | |
| HAIC-FOLFOX + Apatinib + Camrelizumab | Intravenous FOLFOX + Camrelizumab + Apatinib | First | VEGFR; PD-1 | Recruiting | 31-Jul-26 | NCT06172205 | |
| HAIC-FOLFOX + Zoledronic acid | HAIC-FOLFOX | First | Farnesyl pyrophosphate synthase, hydroxylapatite, geranylgeranyl pyrophosphate synthase | Recruiting | 1-Dec-24 | NCT05866172 | |
| TACE + SBRT + Lenvatinib | Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT | Not yet recruiting | 27-Feb | NCT05718232 | |
| TACE + Lenvatinib + Sintilimab | TACE + Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Recruiting | 31-Oct-26 | NCT05608200 | |
| TACE + Anlotinib + Penpulimab | Penpulimab + Anlotinib | Not reported | VEGFR, PDGFR, FGFR, c-Kit; PD-1 | Recruiting | 31-Mar-23 | NCT05344924 | |
| TACE + Apatinibmesylate + Camrelizumab | TACE | Not reported | VEGFR; PD-1 | Recruiting | 30-Jul-24 | NCT05320692 | |
| HAIC-FOLFOX + Apatinib + Camrelizumab | Apatinib + Camrelizumab | First | VEGFR2; PD-1 | Recruiting | 1-Nov-24 | NCT05313282 | |
| TACE + synchronous Lenvatinib | TACE + sequential Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT | Recruiting | 28-Feb-24 | NCT05220020 | |
| TACE + Tremelimumab + Durvalumab + (Lenvatinib) | TACE | Not reported | CTLA-4; PD-L1; VEGFR, FGFR, PDGFR, RET, KIT | Recruiting | 31-Dec-25 | NCT05301842 | |
| HAIC-FOLFOX + Apatinib + Camrelizumab | Apatinib + Camrelizumab | First | VEGFR2; PD-1 | Recruiting | 1-Jan-26 | NCT05198609 | |
| TACE + Atezolizumab + Becavizumab | TACE | First | PD-L1; VEGF-A | Active, not recruiting | 28-Feb-29 | NCT04712643 | |
| TACE + Lenvatinib + Pembrolizumab | TACE | Not reported | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Active, not recruiting | 30-Jun-28 | NCT04246177 | |
| TACE + SBRT + Sorafenib | Sorafenib | First | VEGFR, PDGFR, RAF, FLT3, KIT, FGFR, RET | Recruiting | 1-Aug-23 | NCT04387695 | |
| TACE/TAE + Nivolumab | TACE/TAE | First | PD-1 | Recruiting | 25-Jun | NCT04268888 | |
| TACE + Bevacizumab + Durvalumab / TACE + Durvalumab | TACE | Not reported | VEGF-A; PD-L1 | Active, not recruiting | 11-Sep-23 | NCT03778957 | |
| HAIC-FOLFOX-5 FU + Concurrent Lenvatinib+ Concurrent PD-1 antibody | HAIC-FOLFOX-5 FU + Sequential Lenvatinib + Sequential PD-1 antibody | First | VEGFR, FGFR, PDGFR, RET, KIT; PD-1 | Recruiting | 27-Jul | NCT06041477 | |
| Other strategies | QL1706 + Bevacizumab + chemotherapy (Oxaliplatin + Capecitabine) / QL1706 + Bevacizumab / QL1706 + chemotherapy | Sintilimab + Bevacizumab | First | PD-1; CTLA-4; VEGF-A | Not yet recruiting | 1-Sep-27 | NCT05976568 |
| Icaritin | Huachansu | First | NR (Traditional Chinese Medicine) | Recruiting | 30-Aug-25 | NCT05594927 | |
| Lenvatinib + I-125 Seed Brachytherapy | Lenvatinib | First | VEGFR, FGFR, PDGFR, RET, KIT | Recruiting | 31-Oct-26 | NCT05608213 | |
| ADI-PEG20 | Placebo | Second | Pegargiminase | Recruiting | 25-Sep | NCT05317819 | |
| Hepatectomy + Camrelizumab + Apatinib | None | Not reported | PD-1; VEGFR | Recruiting | 30-Sep-24 | NCT05062837 | |
| Radiotherapy + Toripalimab | Sorafenib | Not reported | PD-1 | Recruiting | 31-Mar-24 | NCT04709380 |
Information retrieved from “https://classic.clinicaltrials.gov/”, as of April 15, 2024
This review summarizes essential signaling pathways including cellular signaling pathways and immune-related signaling pathways, discusses current potential therapeutic targets in HCC, and presents preclinical animal models alongside ongoing or completed clinical studies on systemic therapy, aiming to provide a prospective outlook on precision treatment for HCC in the future.
Cellular signaling pathways in HCC cells
Cellular signaling pathways in HCC cells typically bifurcate into two main segments: the RTK signaling pathways and other pathways. Examining the application of these two kinds of pathways in preclinical and clinical contexts reveals their pivotal roles in processes such as cell proliferation, survival and migration. Understanding these pathways holds promise for devising targeted therapies and advancing cancer treatment strategies.
RTKs signaling pathways
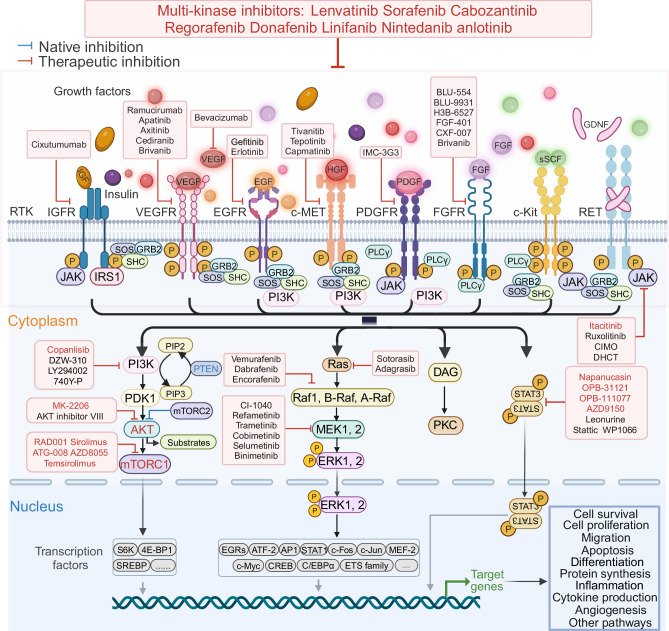
The RTKs comprise a cluster of transmembrane receptors that are activated upon binding to specific ligands, such as growth factors or hormones.12 The RTKs consist of an extracellular domain for ligand binding, transmembrane regions, and a cytoplasmic domain with tyrosine kinase activity and an ATP-binding site.13 These structural components allow RTKs to transduce extracellular signals into intracellular responses by activating downstream signaling pathways. Upon ligand binding, RTKs undergo dimerization, leading to autophosphorylation of tyrosine residues within their cytoplasmic domains. This autophosphorylation activates the intrinsic kinase activity of the receptor, allowing it to phosphorylate tyrosine residues on downstream signaling molecules. These phosphorylation events initiate intracellular signaling cascades, such as MAPK (mitogen-activated protein kinase), that are essential for cell survival, proliferation, and differentiation. Dysregulation of RTK signaling pathways is associated with HCC, making them important therapeutic targets. Based on the accumulated knowledge from existing studies, we focused on reviewing and discussing the roles of eight RTKs classes including VEGF receptor (VEGFR), Epidermal growth factor receptor (EGFR), c-Met, platelet-derived growth factor (PDGFR), fibroblast growth factor receptor (FGFR), insulin-like growth factor receptor (IGFR), KIT, RET (Rearranged during transfection) and their downstream pathways including MAPK pathway, Phosphatidylinositol 3-kinase (PI3K) pathway and JAK-STAT pathway in biological behaviors of HCC as well as their therapeutic implications.
VEGFR
“Sustained angiogenesis” is identified as one of the hallmarks of cancer proposed by Drs. Weinberg and Hanahan as early as 2000.14 Malignant tumor cells require sufficient nutrients and oxygen for rapid survival and proliferation; thus, tumor neovascularization is crucial in supporting tumor growth, infiltration, and spread.15,16 HCC is a vascular-rich solid tumor with hypervascular nature. Neovascularization plays an important role in the development of HCC, and therefore most of the existing targeted therapies are aimed at targeting its angiogenic pathways.17,18 VEGF/VEGFR pathway is one of the most typical and prominent tumor-induced pro-angiogenic factors affecting the development of HCC.19 VEGFA has a 7% ~ 14% frequency of focal amplification in HCC.20,21 Its receptors, VEGFR-1 and VEGFR-2, are usually highly expressed in HCC and correlated with the differentiation and stage of the tumors.22–25 In HCC patients, circulating VEGF serves as a marker for HCC metastasis, whilst VEGF signaling maintains an immunosuppressive tumor microenvironment.26
Almost all approved TKI for advanced HCC involve the targets of VEGFRs. Cancer neoangiogenesis is mainly mediated through the VEGFA/VEGFR2 axis, making VEGFA and VEGFR2 key targets for the development of novel drugs. Inhibition of VEGFR signaling has shown promising antitumor effects in HCC cell lines and mouse models.27,28 Furthermore, a number of selective VEGFR-related inhibitors are already in clinical practice. Bevacizumab, a recombinant humanized monoclonal antibody against VEGFA, was the first FDA-approved agent to inhibit angiogenesis and has proven to be effective against a wide range of advanced malignancies.29 The combination of bevacizumab and PD-L1 monoclonal antibody atezolizumab was authorized as a first-line therapy for advanced HCC in 2020.30 Compared with sorafenib alone, atezolizumab plus bevacizumab significantly prolonged overall and disease-free survival and maintained clinical efficacy during long-term follow-up.30,31 This combination has plausible biological mechanisms that restore or enhance the efficacy of immune checkpoint inhibitors in antitumor therapy by inhibiting the VEGFR-associated immunosuppressive microenvironment.32 Several other regimens targeting VEGFR signaling in combination with PD-1/-L1 inhibitors have now entered phase III clinical trials (NCT06172205, NCT05320692, NCT03764293). The CARES-310 study (NCT03764293), which recently disclosed data, demonstrated that apatinib, a selective VEGFR2 small molecule inhibitor, in combination with the PD-1 monoclonal antibody camrelizumab had a superior benefit over sorafenib, and therefore this combination is currently approved by the National Medical Products Administration (NMPA) as first-line therapy for unresectable or metastatic HCC.33 Moreover, apatinib monotherapy, which prolonged survival and response rates in advanced HCC compared to placebo as a second-line therapy, has now been approved by the NMPA. Following this, another VEGFR2 monoclonal antibody, ramucirumab, has been approved by the FDA for advanced HCC patients who previously recieved sorafenib and had an AFP ≥ 400 ng/mL.34 The success of these agents demonstrates the substantial role of VEGFR in HCC treatment. But there have been some failures, for example, the dual VEGFR and FGFR inhibitor brivanib failed in phase III trials as first-line, second-line, and adjuvant therapy in HCC.35–37 Two pan-VEGFR inhibitors, axitinib and cediranib, failed to advance to phase III trials after poor performance in phase II trials.38,39 These failures may be attributed in part to flaws in study design, such as imbalance in patient baseline across treatment arms and lack of specific participant eligibility criteria, but may also be due to the structure of the agents themselves being less active in humans and presenting off-target toxicity.
EGFR
EGFR belongs to the ErbB receptor family, which is expressed in many organs of the human body at low levels. Compared with other tissues, EGFR is relatively highly expressed in hepatocytes of the adult liver, indicating its significance in maintaining liver function.40 As a potential biomarker of drug resistance in tumors, EGFR exhibits intricate responses to various cellular stresses, including drug stress, UV radiation, and inflammatory cytokines. Mutations in EGFR can occur in various solid malignant tumors, especially in Asian NSCLC patients.41,42 Previous studies reported that activating mutations in exons 18-21 of EGFR are rare in HCC patients.43,44 Nevertheless, one study found 13 novel missense mutations in EGFR exon 19-23 from HCC tissues,43 and seven of these HCC-derived mutants (K757E, N808S, R831C, V897A, P937L, T940A, and M947T) showed resistant to first-generation EGFR inhibitors.45 For these patients with rare EGFR mutations, gefitinib is not suitable for combination with lenvatinib but later-generation EGFR inhibitors, such as osimertinib, could be considered.
EGFR is expressed in over 60% of HCC patients, and is positively correlated with poor differentiation, high proliferative activity, advanced stage, intrahepatic metastasis, and poorer PFS.46–49 EGFR exhibits high expression levels in HCC cell lines and tissues, and furthermore, up-regulated EGFR expression in circulating tumor microemboli maintains stability and distal metastasis of suspended HCC cells by activating Ras/MAPK pathway.50 The high EGFR-related pathway signaling in HCC may be related to alterations in its regulatory factors and overexpressed ligands.20,51,52
EGFR signaling has been found to mediate resistance to the first-line targeted drugs lenvatinib and sorafenib. Our group previously identified EGFR as a synthetic lethal target of lenvatinib in HCC by using CRISPR-Cas9 genetic screen.53 One of the mechanisms of intrinsic resistance to lenvatinib treatment in HCC is the inhibition of fibroblast growth factor receptor (FGFR) leading to a feedback activation of the EGFR-PAK2-ERK1/2 and EGFR-PAK2-ERK5 signaling pathways, which allows malignant cells to maintain survival and sustain proliferation. Subsequent phase I clinical trials demonstrated a strong synergistic effect of lenvatinib in combination with the EGFR inhibitor gefitinib in advanced HCC with high expression of EGFR and refractory to lenvatinib mono-therapy, with 9 out of 30 cases achieving partial response (NCT04642547). Recent studies have also revealed multiple other mechanisms of EGFR involved in lenvatinib resistance, including reactive oxygen species accumulation,54 activation of ABCB1 to potentiate the cytotoxic effects of lenvatinib,55 and as a downstream target for RNA modification.56,57 An analysis of circulating tumor DNA profiling of peripheral blood suggests that pre-treatment low copy number EGFR/ERBB2 amplification may serve as a genetic marker for lenvatinib resistance.58 Moreover, many studies have shown that EGFR also contributes to primary and acquired resistance to sorafenib in HCC cells and can serve as a potential predictor of resistance to sorafenib.59–62 However, our preclinical data demonstrated that sorafenib in combination with EGFR inhibitors did not show a synergistic effect in the majority of liver cancer cell lines,53 and the phase III RCT trial also found that the addition of erlotinib, a selective inhibitor targeting EGFR tyrosine kinase, to sorafenib exhibited no enhancement in survival outcomes for advanced HCC.63
C-Met
The c-Met is expressed primarily on epithelial cells and is a transmembrane receptor containing the catalytic structural domain of tyrosine kinase. The only known endogenous ligand is HGF, which is secreted mainly by mesenchymal cells and is a hepatocyte mitogen.64 HGF can activate downstream MAPK, PI3K/AKT, and STAT3 signaling pathways upon binding to c-Met via low (the SPH domain) or high (the N and kringle 1 domains) affinity binding sites. It is involved in various physiological processes such as embryonic development, tissue repair and regeneration, and inflammatory responses.65 Furthermore, it also transmits signals in a noncanonical manner independent of HGF, which is an important mechanism for malignant cells to acquire resistance to inhibitors in pathological conditions.66–68 MET is considered a proto-oncogene, and together with its ligand HGF have been found to be dysregulated in the process of HCC development.69 Downstream signaling pathways activated by c-Met contribute to malignant actions including survival, growth, invasion, metastasis, metabolic reprogramming, epithelial-mesenchymal transition (EMT), drug resistance, and enhancement of tumor stemness.65 Although c-Met mutation or amplification is detected in only approximately 1% of HCC patients,70 its overexpression occurs in about a quarter of patients, even up to 80% in advanced HCC.71,72 Activation of HGF/c-Met is one of the key mechanisms of resistance to sorafenib and lenvatinib, which is associated with the bypassing of growth factor targets of sorafenib and lenvatinib by HGF/c-Met to promote the downstream pro-carcinogenic MAPK pathway and induction of EMT.73 Therefore, c-Met levels could serve as a potential biomarker for predicting response to sorafenib and lenvatinib.24,74–77 Sorafenib or lenvatinib in combination with c-Met inhibitors exhibited therapeutic sensitization and synergistic antitumor effects.74,76 However, the prognostic association between HGF/c-Met and HCC patients is still uncertain; although most studies have shown that high c-Met or HGF expression in HCC predicts shorter survival and higher potential for recurrence and metastasis,78–81 some studies have also suggested no prognostic significance of c-Met and HGF.71,78
Several inhibitors targeting HGF/c-Met have been developed. Cabozantinib is a c-Met/RTK inhibitor, which is currently being used for the treatment of HCC patients as a second-line drug. The recent results of the phase III trial COSMIC-312 demonstrated that cabozantinib in combination with the PD-L1 inhibitor atezolizumab prolonged PFS compared to sorafenib, but did not improve OS.82 Some selective c-Met inhibitors have been tested for antitumor activity in HCC, but blocking c-Met alone does not appear to provide an adequate response.83 Tivanitib, an oral selective c-Met inhibitor, showed encouraging responses in preclinical studies and a phase II clinical trial, but a subsequent phase III trial in advanced HCC with high c-Met expression did not demonstrate similar effects.84,85 Survival was not improved with tivanitib monotherapy compared with the placebo group. The failure may be attributable to primary resistance to c-Met inhibitors and an exaggerated role for highly expressed c-Met in patients previously treated with sorafenib.86
Given the contribution of c-Met to sorafenib and lenvatinib resistance as demonstrated by preclinical data, a strategy of tivanitib in combination with sorafenib and lenvatinib has been proposed,76,77,87 but remains to be validated in clinical settings. Interestingly, two highly selective c-Met inhibitors that have been approved by the FDA for the indication of NSCLC harboring the METex14 skipping mutation, tepotinib and capmatinib, have shown promising efficacy in early clinical trials in HCC.88–90
FGFR
The FGFR gene family, including FGFR1-4, encodes seven FGFR proteins, since FGFR1-3 have two alternative splice variants.91 Eighteen ligands and four homologs have been identified in the fibroblast growth factor (FGF) family.92 Originally named for their ability to promote mitogenesis in fibroblasts, FGFs are now recognized as one of the most diverse families of peptide growth factors in mammals. A substantial amount of evidence has shown dysregulation of FGFR signaling as a pathogenic mechanism and consequence of cancer,93 and it is frequently detected to be highly expressed in HCC, contributing to tumorigenesis, progression, and drug resistance.94,95 Despite the relatively low frequency of genetic alterations, dysregulated FGF/FGFR signaling affects about half of HCC patients.96–98 Among the FGF and FGFR families, currently the most studied and promising target in HCC is the FGF19-FGFR4 signaling pathway. FGF19 is localized in the 11q13.3 amplicon, and is frequently detected co-amplified with cyclin D1 (CCND1), a known proto-oncogene, resulting in increased expression.98 They have been found to be key oncogenic driver signals in HCC and are widely expressed in patients promoting HCC progression.98,99 Transgenic mice overexpressing FGF19 in skeletal muscle led to the development of HCC,100 which was effectively inhibited by the use of either anti-FGF19 or FGFR4 monoclonal antibodies.101,102 Interestingly, clinical evidence showed that the expression of FGF19 and FGFR4 increased sequentially with the different histologic stages of carcinogenesis (steatosis-steatohepatitis–cirrhosis-HCC), suggesting a strong association with HCC development.103,104 In addition, FGF19 can exert pro-oncogenic effects by crosstalk activation of other growth factor pathways, such as increasing the expression of the EGFR ligands, connective tissue growth factor (CTGF) and amphiregulin, thereby affecting the development of HCC through indirectly or directly shared pathways.105,106 Furthermore, FGF19/FGFR4 confers acquired resistance to multikinase inhibitor therapy in HCC, and targeting the FGF19/FGFR4 axis can sensitize the antitumor effects of sorafenib and regorafenib.94,107,108
FGFR4 is specifically expressed in the liver, making it a promising target for drug development. Moreover, the hinge region of the FGFR4 complex has a unique non-conserved Cys552 structure, whereas the corresponding positions of FGFR1-3 are tyrosine, providing feasibility for the development of selective covalent FGFR4 inhibitors.109 Pan-FGFR inhibitors that have been approved by the FDA are erdafitinib, pemigatinib, and infigratinib for indications including uroepithelial cancers, bile duct cancers, or myeloid/lymphoid neoplasms.110,111 Compared to these pan-FGFR inhibitors, selective FGFR4 inhibitors have better manageable toxicity. FGFR4 inhibitors that have shown promising anti-cancer activity in HCC include BLU-9931,112,113 H3B-6527,114 FGF-401,115 CXF-007,116 and BLU-554 117. Currently, phase I or phase II clinical trials have been completed for H3B-6527, BLU-554, and FGF-401, all of which have shown good tolerability.118 The first-line RTK inhibitor lenvatinib also can suppress FGFRs, but, the degree of its FGFR4 inhibition remains uncertain.
IGFR
The IGFR consist of two isoforms, IGF-1R and IGF-2R, which together with the ligands IGF-1 and IGF-2, and the high affinity IGF-binding protein 1-6 (IGFBP 1-6), comprise the IGF family. IGF-1 exerts its biological effects by binding to IGF-1R, whereas IGF-2 binds to IGF-1R, IGF-2R, and insulin receptor.119 Both have prominent roles in the growth and development of the organism, and their deficiencies lead to developmental disorders.120 Due to shared receptors and ligands, the IGF-1R signaling cascade is also crosstalked by the insulin/insulin receptor signaling pathway.121 In contrast, IGF-2R is a type I transmembrane protein with no kinase activity and does not transduce signaling. It competitively binds to IGF-2 and enters the lysosome via endocytosis for degradation to impair IGF-IR signaling.122 The association between the IGF family and HCC development has been widely reported. Overexpressed IGF-2 and IGF-1R are frequently detected in HCC tissues and correlate with advanced stage and prognosis of HCC patients.123–126 Interestingly, although IGF-IR has no nuclear localization sequence and is normally localized to the cytoplasmic membrane, it can undergo nuclear ectoposition by SUMOylation, binding to promoter DNA of its own gene and some oncogene-related genes, such as JUN and FAM21, to promote gene expression.123,127,128 IGF-2R is considered to harbor tumor suppressor gene properties due to inhibition of IGF-1R signaling caused by competitive binding of IGF2. Besides antagonizing IGF-1R, IGF-2R promotes the transport of lysosomal enzymes and the tumor suppressor TGF-β1 into cells to exert inhibitory effects on tumor development.122,129 Thus, in contrast to the overexpression of IGF-1R in patients, the locus where the IGF-2R gene resides has been found to have frequent loss of heterozygosity (LOH) and reduced expression in different cancers, especially HCC.130–132
The IGF axis, due to its high relevance with tumorigenesis and development in HCC, has emerged as a viable target for therapeutic intervention. Considerable efforts were made to block the IGF-1R signaling pathway at multiple levels. To date, three primary classes of therapeutic agents have been assessed in both animal models and clinical trials: small molecular tyrosine kinase inhibitors, antibodies to IGF-1R, and antibodies to IGF ligands. Additionally, alternative therapeutic strategies, including small interfering RNAs, antisense oligonucleotides, recombinant IGFBPs, and pregnancy-associated plasma protein-A (PAPP-A) inhibitors, have also been explored.119 The only currently approved selective IGF-1R inhibitor is teprotumumab, a fully humanized IGF-1R monoclonal antibody, which is used to treat thyroid eye disease.133 Decades of effort have gone into the development of IGF axis-targeted drugs for indications in oncology. Unfortunately, although some therapies targeting the IGF axis have demonstrated potent antitumor activity and tolerability in preclinical and early clinical trials, no drug has successfully passed phase III trials to date.134,135 The response to targeting IGF1R as monotherapy is limited in most cases,136 and therefore it is often used in combination with other targeted therapies or chemotherapy, but the results remain disappointing. For patients with HCC, the IGF-1R inhibitor cixutumumab tested in the clinical setting showed limited clinical benefit.137,138 Failure is often attributed to the activation of compensatory signaling pathways, such as growth hormone- or insulin-related pathways, in response to IGF axis inhibition, resulting in the loss of inhibition of downstream MAPK and PI3K-AKT-mTOR cascades.138–140 In addition, crosstalk between IGF-1R and other receptors such as insulin receptor, EGFR, and integrins increases the complexity of IGF axis targeting.138,141,142 Therefore, further insight into the complex signaling network between the IGF axis and other pro-cancer pathways and the identification of biomarkers that predict therapeutic response are important and urgent.
PDGF
The PDGF family encoded by four genes, PDGFA, PDGFB, PDGFC, and PDGFD.143,144 The PDGF receptor (PDGFR) contains two isoforms, PDGFR-α and PDGFR-β, and is a type III tyrosine kinase protein.145–147 PDGFs are typically produced by discrete cell populations and act as paracrine factors to regulate cellular responses.148,149 PDGFRs are considered as an oncogenic driver, expressed or subject to activating genetic alterations in many types of cancer tissues, including HCC, which promote carcinogenesis, progression, and drug resistance through the classical MAPK and PI3K cascades.150–152 More than half of HCC patients have high PDGFR-α expression in their tumor tissues.153 Overexpressed PDGFRs promote HCC progression and are significantly associated with a worse prognosis of HCC patients.24,154 Similar to the functions of VEGF and FGF, PDGFs also can act as a pro-angiogenic factor in HCC tissues and has a role in inducing tumor neovascularization 17. Upregulated PDGFR-α may be involved in HCC development and is significantly associated with microvessel density and vascular infiltration of tumors,154 which could serve as a biomarker for predicting HCC metastasis and a potential therapeutic target.155
Several currently FDA-approved HCC-targeted agents, including lenvatinib, sorafenib, and regorafenib, have PDGFR as one of their targets. Donafenib monotherapy, which simultaneously targets PDGFR, VEGFR, and RAF kinases, was approved by the NMPA in 2021 as a first-line therapy for advanced HCC based on its ability to achieve a superior OS compared with sorafenib.156 Linifanib, a small molecule inhibitor targeting PDFGR and VEGFR, was found to have promising antitumor activity in HCC in preclinical and early clinical studies.157 However, it did not show superior efficacy over sorafenib in phase III trials and failed to meet the primary endpoint.158 Another triple angiokinase inhibitor, nintedanib, targeting PDFGR/VEGFR/FGFR, also exhibited similar results in clinical trials.159
Approaches to selectively inhibiting the PDGF/PDGFR axis primarily include receptor antibodies or small molecule inhibitors that block ligand-receptor interactions, neutralizing antibodies or ligand traps that isolate the ligand, and blocking PDGFR kinase domain activity.152 However, there are several challenges to specifically targeting PDGFR signaling. PDGFR-α and PDGFR-β, as class III RTK proteins, have greater structural similarity to some other cell surface receptors such as FLT3, kit, and CSF1R, as well as functional redundancy when PDGFR is activated.152,160 Several selective PDGFR inhibitors have been developed, including olaratumab, CP-673451, and CHMFL-PDGFRα-159, all of which have shown significant anti-cancer and antiangiogenic effects.161–165 One of these, olaratumab, was tested clinically and was received accelerated approval by the FDA for the treatment of advanced soft tissue sarcoma based on a successful phase II result, but unfortunately did not show similar benefit in a subsequent phase III trial.166 In addition, anti-PDGFR-α monoclonal antibodies have been found to have anti-proliferative and pro-apoptotic effects in multiple human and mouse HCC cell lines.153 Inhibition of PDGF signaling to suppress HCC progression was also found in mouse models.167 However, there are currently no selective PDGF/PDGFR inhibitors in clinical testing for HCC.
C-Kit
c-Kit, encoded by the KIT gene, is primarily activated by stem cell factor (SCF).168 In normal conditions, c-Kit is abundantly expressed in hematopoietic stem cells and is crucial for cell survival, proliferation, and differentiation.169 The current view is that c-Kit may play a double-edged role in the liver: on the one hand, c-Kit-positive cells participate in tissue repair by promoting target cell regeneration in the event of liver injury, but on the other hand, the overexpression or mutation of c-Kit as a proto-oncogene contributes to the development of HCC.170 An important contributor to heterogeneity in HCC is liver cancer stem/progenitor cells (LCSCs), implicated in drug resistance and tumor recurrence.171 c-Kit receptor serves as a marker for LCSCs, and its overexpression promotes the transformation of hepatic stem/progenitor cells into LCSCs.172–174 In HCC driven by HBV or HCV, c-Kit plays a mediating role. PreS1 protein of HBV and core proteins of HCV induce LCSC production and self-renewal of tumor cells by stimulating c-Kit expression, accelerating HCC onset and progression.175–177 Aberrant crosstalk exists between SCF/c-Kit and other pro-cancer pathways like TGF-β/SMAD2, forming positive feedback loops promoting malignant cell proliferation and invasion.178
The first targeted drug approved for the treatment of cancer, imatinib, has c-kit as one of its targets. Unfortunately, clinical trials of imatinib in HCC have all failed.179,180 Many inhibitors targeting c-Kit mutations have been approved, but the vast majority are multi-targeted kinase inhibitors.181 One of the targets of the current first-line targeted agents sorafenib and lenvatinib and the second-line therapies regorafenib and cabozantinib in the treatment of HCC includes c-Kit. Another tyrosine kinase inhibitor, anlotinib, which takes c-Kit as one of its targets, has shown promising efficacy and a tolerable safety profile in clinical studies. In a multicenter, phase II study enrolling patients with advanced HCC, anlotinib in combination with the PD-1 inhibitor toripalimab as first-line treatment achieved an ORR of 32% and a median survival of 18.2 months.182 Another phase II studies found that anlotinib monotherapy resulted in promising benefits as first- or second-line treatment in advanced HCC,183 while the combination of chemotherapy did not provide additional efficacy.184 Several real-world studies have also confirmed the efficacy of anlotinib in patients with HCC.185,186 Multiple Phase III clinical trials are currently testing the efficacy of anlotinib in HCC patients in different clinical settings (NCT05862337, NCT04344158, NCT05344924, NCT04665609, NCT03950518). However, anlotinib also targets some other RTKs, and the effect of selective c-Kit inhibitors on HCC has been less well-studied.
RET
RET is identified in 1985 during the transfection of mouse NIH-3T3 cells with human T-cell lymphoma.187 In the normal body, RET has a crucial role in embryonic kidney and neural development.188,189 RET is considered a proto-oncogenic driver gene, and RET fusion is the alteration of great interest, whereby the RET gene breaks and recombines with another gene to form a new gene, thus possessing the ability to self-phosphorylate for sustained activation, frequently occurring in thyroid cancer and NSCLC.190 Treatment of EGFR or KRAS inhibitors may induce acquired RET fusions, contributing to resistance to targeted therapy.191,192 RET alterations are rare in HCC patients with a frequency of less than 1%.193,194 Data from TCGA suggest that RET expression is significantly downregulated in HCC tissues compared to adjacent tissues.195 Currently, the mechanism of RET signaling in the occurrence and development of HCC has been less reported.
The first-line drug lenvatinib and the second-line drugs cabozantinib and regorafenib, used for advanced HCC treatment, target RET among other pathways. FDA-approved selective RET inhibitors, pralsetinib and selpercatinib, are indicated for metastatic RET fusion-positive NSCLC and medullary thyroid cancer, with selpercatinib additionally approved for other advanced RET fusion-positive solid tumors.196,197 However, the multicancer indication for selpercatinib was based on a phase I/II trial excluding HCC patients.190 Although lacking preclinical and clinical data in HCC, the success in thyroid and lung cancers prompts exploration of RET inhibitor efficacy in HCC, necessitating patient selection criteria.
MAPK pathway
The MAPK pathway is one of the most important signaling pathways underlying life-sustaining activities in eukaryotes, which is frequently altered in a wide range of diseases.198 Four different MAPK cascades have been identified, namely the extracellular signal regulated kinase 1/2 (ERK1/2), Jun amino terminal kinases (JNK), p38 MAPK, and BMK1 cascades. ERK1/2 is the most classical and well-studied key MAPK signaling pathway, which is activated mainly by signals transmitted through cell surface transmembrane receptors such as RTKs or G protein-coupled receptors.198,199 Almost all growth factor signals depend on the activation of ERK to complete the signaling process, thus ERK is a ubiquitous signaling pathway in the human body. Ras, Raf, MEK and ERK proteins are key components of this pathway, and abnormal function of any one of them may lead to serious consequences. Since ERK plays an important role in key cellular functions, its aberrant activation is closely linked to the development of malignant lesions. The upstream activating protein of the ERK pathway, RAS is a frequent driver mutation in human cancers, with approximately 19% of cancer patients harboring mutations in the RAS gene.200,201 Aberrant activation of the MAPK pathway is present in about half of patients with early-stage HCC, and high expression of ERK signaling is detected in almost all patients at advanced stages.202–204 Activated MAPK signaling is significantly associated with poorer prognosis and metastasis in HCC patients.205,206
Inhibitors targeting the MAPK pathway are a hot topic in antitumor drug development and have great potential for clinical application. Almost all of the currently approved RTK-targeted agents in the HCC could affect downstream activation of the MAPK pathway. One of the targets of both sorafenib and regorafenib includes the RAF protein. A number of selective inhibitors have been developed that target components of the MAPK pathway, focusing on two key kinases, MEK and ERK. The first MEK inhibitor to enter clinical testing was CI-1040 (also known as PD184352), which targets MEK1/2. Although CI-1040 in combination with sorafenib was found to have a superior antitumor effect compared to sorafenib alone in a xenograft HCC model,207 it displayed insufficient antitumor activity in a phase 2 clinical study in solid tumors,208 which may be due to its low exposure resulting from its fast clearance rate and poor solubility. Several second-generation selective MEK inhibitors have subsequently been developed, which has led to the approval of the selective MEK1/2 inhibitors trametinib, cobimetinib, selumetinib, and binimetinib. In addition, three RAF inhibitors, vemurafenib, dabrafenib, encorafenib, and two KRASG12C inhibitors, sotorasib and adagrasib, have also become available in recent years.209 Although none of their current indications include HCC, promising antitumor activity in HCC has been demonstrated in several preclinical and early clinical trials, either as a monotherapy or in conjunction with other targeted agents.210–214 For instance, a phase II clinical trial found that the MEK1/2 inhibitor refametinib in combination with sorafenib demonstrated potential synergistic effects in HCC patients with Ras mutations.215 Although therapies targeting the MAPK pathway have made encouraging progress, they are highly susceptible to drug resistance. The therapeutic effect is often short-lived and difficult to achieve complete tumor regression.216 Activating mutations of key components under drug stress, mobilization of independent alternative pathways, and feedback upregulation of the number of targeted proteins can all lead to resistance to MAPK pathway-related inhibitors.217 Several coping strategies have been proposed, including rational combinations (such as MEK plus BRAF inhibitors) and optimization of the drug’s structure (such as the second-generation RAF inhibitors PLX-8394, TAK-580, BGB-283).217 However, it remains difficult to overcome the primary or acquired resistance that almost inevitably occurs. In addition, attention should be drawn to the issue that in cells with wild-type BRAF, RAF inhibitors may transactivate ERK signaling resulting in toxicity and drug resistance.218 The low occurrence of BRAF V600 mutations in HCC targeted by vemurafenib, dabrafenib, and encorafenib also limits their application value.
PI3K-AKT pathway
PI3K-AKT has been broadly characterized to be a critical and ubiquitous pathway in regulating the cell cycle. PI3K was discovered to phosphorylate phosphatidylinositol lipids and to act as downstream of RTKs and insulin signaling.219,220 PI3K-AKT-mTOR has been found constitutively activated in cancer and acts as oncogenic pathway, regulating cell cycle, survival, metabolism, motility and angiogenesis in cancers.221,222 In HCC, activation of PI3K-AKT-mTOR signaling is found in about 50% cases, which is involved in upregulation of EGFR, PI3K, AKT and mTORC1,223–228 while the key suppressor of the PI3K/AKT cascade such as TSC1/2 and PTEN were found with loss-of function mutation or reduced expression in HCC.229 Activation of PI3K-AKT-mTOR signaling by overexpression of AKT or knockout of Pten/Tsc1/2 were validated to induce HCC hepatocarcinogenesis in mouse models,230 powerfully validating the oncogenic roles of this signaling. PIK3C3 is required to the cancer stem cells (CSCs) growth and activity.231 Hepatic mTORC2 facilitates hepatosteatosis via de novo fatty acid and lipid synthesis in HCC.232 While long-term inhibition of mTORC1 promotes HCC development through promoting IL-6/STAT3 pathway in a murine model of obesity,233 which indicates the complex function of mTORC1 in HCC might relates to the etiology. In addition to the initiation of HCC, PI3K-AKT pathways have been broadly investigated to facilitate HCC progression via metabolic reprogramming, such as glucose metabolism, lipid metabolism, amino acid metabolism, pyrimidine metabolism, and oxidative metabolism.234 HCC patients with altered PI3K-AKT-mTOR signaling showed the activation of asparagine synthetase (ASNS), glycolysis, and the pentose phosphate pathway.235 The significant tumor-promoting roles of PI3K-AKT signaling were substantially demonstrated as above, so the anti-tumor effect by suppression of this pathway has been frequently clarified in HCC, and several agents were under the investigation of clinical trials.230
Several selective inhibitors targeting PI3K-AKT-mTOR cascade have progressed in phase II studies for the treatment of liver cancer, of which the outcome is inconsistent and with significant adverse effects. Copanlisib, a specific PI3K inhibitor, was evaluated in cancer patients with activating mutation of PI3K in a phase II clinical study (NCT02465060).236 Another phase II study evaluated the efficacy of an allosteric AKT inhibitor, MK-2206, in advanced biliary cancer (NCT01239355).237 While the two trials showing discouraging results with limited efficacy and severe adverse effects.236,237 Though a phase I/II study failed to exhibit effectiveness of single RAD001 (everolimus) targeting mTOR for treating patients with advanced HCC (NCT00516165), another mTOR inhibitor sirolimus (rapamycin) was encouragingly demonstrated to improve the survival of HCC patients in 2 phase II studies.238 An orally administrated mTOR inhibitor, ATG-008 (CC-223) was tested in HBV-positive HCC in a phase II trial (NCT03591965), while terminated based on strategy development. An ATP-competitive mTOR kinase inhibitor AZD8055, has undergone an evaluation of its safety, tolerability, pharmacokinetics, and preliminary efficacy in a phase I/II study, (NCT00999882).239 Several clinical trials are ongoing to explore drug combinations of inhibitors targeting PI3K-AKT-mTOR signaling cascade.230,240 The combination treatment of rapamycin with bevacizumab demonstrated complete response (CR) in 1 case, PR in 2 cases, and stable states of the disease in 14 cases out of 20 evaluable HCC patients in a phase I study (NCT00467194).241 A single-arm phase II trial exploited the inhibition of mTOR with temsirolimus along with sorafenib’s effects on HCC,242 showing on improvement in overall survival compared with the treatment of single sorafenib (NCT01687673). Overall, therapies targeting this pathway alone or connectionally showing promising anti-HCC potential in clinical assessment, but it still requires further investigation to improve the clinical response and avoid toxicities and adverse effects.
Except for the limited agents under clinical assessment, the preclinical research of other agents targeting this signal axis is continuously attracting attention. These agents include potent PI3K inhibitors (LY294002, DZW-310,243 740Y-P,244 copanlisib245) and AKT inhibitors (MK2206, AKT inhibitor VIII).246,247 These agents elicited consistent anti-tumor effectiveness such as proliferation inhibition and apoptosis induction in HCC cells.230,248,249 Other unselective agents have also found to inhibit HCC.230,250 For example, celecoxib, a non-steroidal anti-inflammatory drug, targets the cyclooxygenase 2 (COX-2)/AKT pathway and was sufficient to inhibit the progress of HCC by inhibiting lipogenesis.250
Activation of PI3K/AKT/mTOR signaling also associates with sorafenib resistance in HCC,251 providing the rationale for the combination of targeting PI3K/AKT/mTOR cascade with sorafenib. For instance, PI3K inhibitor copanlisib exhibits synergistically anti-tumor effectiveness to sorafenib in HCC treatment,245 the synergistic effectiveness was also observed in the treatment with lenvatinib.248
JAK-STAT pathway
JAK-STAT pathway is an evolutionarily conserved signaling transduction pathway and functions in cell proliferation and survival, modulation of the immune response, as well as angiogenesis and metabolism.252–254 Nearly 60 cytokines including various interleukins, growth factors and colony-stimulating factors (CSFs) act as ligand to activate this pathway.252 Upon binding to the receptor, JAKs activate STATs by phosphorylation, which provokes STATs dimerization and subsequent translocation to the nucleus where they stimulate the transcription of specific target genes responsible for immune system development, immune regulation and hematopoiesis.253,254 The vital role of JAK-STAT pathway is well-known pivotal to neoplastic disorders including hematopoietic and solid cancers.252 Several studies affirm aberrant activation of this pathway and its promotion of malignant phenotypes in HCC via different mechanisms. Actually, STAT3 was indicated to be constitutively active in nearly 60% of the HCC cases.255 Activation of STAT3 causes the transcriptional expression of genes associated with the diverse hallmarks of cancer, like Cyclin D in cell cycle, VEGF in angiogenesis, and IL-10 in immunosuppression, indicating the crucial roles of STAT3 in HCC.256 In 9% of HBV-related HCC cases, missense mutations in JAK1 were found and led to activation of JAK1 and STAT3, allowing cytokine-independent growth.257 In contrast, the negative regulators of the JAK/STAT pathway, CIS, SOCS1 and SOCS3 were found frequently downregulated or lost in HCC, thus resulting in continuous activation of the pathway and HCC progression.258–262 The diversity of ligands and receptors, as well as JAKs and STATs can activate the JAK-STAT pathway, which contributes to its complexity and various cellular responses in cancers. Namely, STAT5A/B act as protumor factors like STAT3, while STAT1 and STAT2 present antiproliferative effects in HCC both in vitro and in vivo.263–265
As an essential immune-regulated pathway, interferon-alfa (IFN-α) mediated JAK-STAT signaling induces various target genes with antiviral and immunomodulatory functions, based on which IFN-α was used to drive host antiviral responses as the current first-line therapy for chronic hepatitis B and has been confirmed to slow the progression of liver fibrosis and even the emergence of HCC,266 indicating the promising characteristics of this pathway in HCC therapy. Actually, kinds of JAK/STAT inhibitors have been examined for their clinical significance in diverse cancers, including HCC, mainly focused on JAK and STAT3 inhibitors.
As the upstream of this signaling axis, JAKs function as a feasible target to curb the downstream effects. WP1066,267,268 pacritinib,269,270 common JAK inhibitors like cryptotanishinone and ruxolitinib are being studied in human diseases. However, these compounds are still in preclinical stages for HCC treatment.271–273 For example, WP1066 has been demonstrated to inhibit MMPs and counteract the activity of UCK2, which suppressed the migration and invasion abilities of HCC cell lines.274
STAT3 can be directly suppressed by small molecule compounds like static,275 OPB-111077,276 OPB-31121,277 napabucasin 278, and siRNA (AZD9150).279 OPB-111077 was well tolerated overall while showed limited efficacy in sorafenib-refractory HCC patients.276 OPB-31121 in a phase I research of advanced solid tumors (NCT00657176) and HCC (NCT01406574)277,280 showed limited antitumor efficacy with toxic side effects. A phase Ib/II clinical trial is evaluating napabucasin in combination with sorafenib for HCC (NCT02279719). Another Phase III trial is also underway for the combination of napabucasin and paclitaxel in gastric and gastroesophageal junction cancers (NCT02178956).278 Unlike small molecule inhibitors, AZD9150 is a siRNA targeting STAT3, showing clinically valuable antitumor activity and can be regarded as a safe treatment for diffuse large B-cell lymphoma.281 A phase I study showed that AZD9150 was well tolerated with mild and a few serious adverse events (NCT01839604) in HCC.279 Nevertheless, further studies are required to illuminate its clinical efficacy. Some agents showed therapeutic effect by indirectly targeting STAT1. An example is acyclic retinoid acts synergistically with IFNs in suppressing the proliferation of HCC cells in vitro by increased expression and DNA-binding activity of STAT1.282
Altogether, these studies reveal the potential of targeting the JAK/STAT pathway in HCC. While clinical research on these inhibitors are still in the initial phases for HCC and adverse effect need attentions. The beneficial effects observed in other tumor types offer indications of potential clinical efficacy for HCC likewise.
Although the frequency of mutations in RTKs is not high, they were detected to be overexpressed in HCC tissues of most patients and are closely associated with malignant biological behaviors such as tumor angiogenesis, proliferation, invasion, metastasis, and drug resistance. Therefore, most RTKs genes are considered as oncogenic driver genes. A series of tyrosine kinase inhibitors and antibody-based agents have been developed and tested as shown in Fig. 2.
Fig. 2.
Summary of the RTK signaling pathways and their related inhibitors in HCC. The RTK and downstream signaling pathways are described, along with a list of inhibitors targeting single or multiple targets. Sorafenib and donofenib inhibit VEGFR1–3, PDGFR, RAF, KIT; lenvatinib inhibits VEGFR1-3, PDGFR, FGFR1-4, RET; cabozatinib inhibits VEGFR1–3, MET, RET; rogerafenib inhibits VEGFR1–3, PDGFR, RAF, FGFR1-2; linifanib inhibits KDR, Flt-1, PDGFRβ, and FLT3; nintedanib inhibits VEGFR1/2/3, FGFR1/2/3, and PDGFRα/β; anlotinib inhibits VEGF R2/3, FGF R 1-4, PDGF R α / β, c-Kit, and Ret. Figure was created with biorender.com
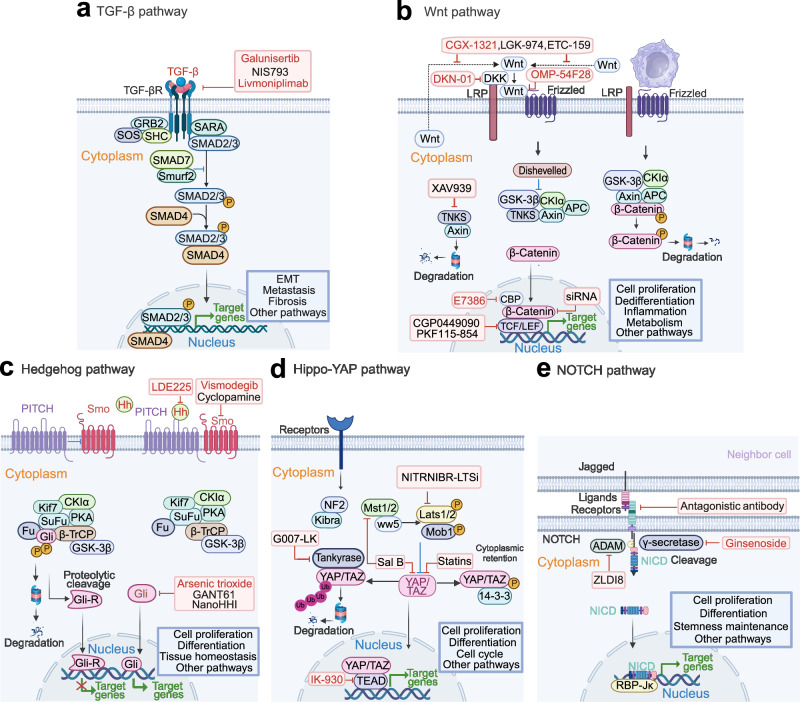
Other pathways in HCC
Beyond the frequently altered and therapeutically targeted RTK pathways in HCC. Pathways in cell differentiation such as Wnt/β-catenin, Hippo, Hedgehog and Notch are especially important for liver tissue homeostasis, and closely correlates to liver disease including HCC. Here, we review the main dysregulated signaling pathways with therapeutic advances in HCC (Table 2).
Table 2.
Summary for therapies targeting signaling pathways in clinical trials for HCC
| Signaling path way | Target | Agent | Clinical stage | Setting | Patients | Outcome | Safety | Trial identifier | Refs |
|---|---|---|---|---|---|---|---|---|---|
| PI3K/AKT/mT OR pathway | Pan-class1PI3K | Copanlisib | Phase II | Arm Z1F | Tumor with PIK3CA mutations | ORR of 16% (P5.0341) | Hyperglycemia, fatigue, diarrhea, hypertension, nausea 53% grade 3 toxicities; 3% grade 4 toxicities | NCT02465060 | 236 |
| AKT | MK-2206 | Phase II | Single agent | Advanced liver cancer (biliary cancer) did not respond to previous therapy | No clinical activity | Grade1/2 toxicities (lymphopenia, rash, fatigue, fever, vomiting, diarrhea) | NCT01239355 | 237 | |
| mTOR | RAD001 (everolimus) | Phase I/II | Single agent | Advanced HCC, iCCA | No significant difference | Anemia, Diarrhea, Lymphopenia, Stomatitis, Vomiting, Nausea, Constipation, Pain, Fatigue, et al. | NCT00516165 NCT01035229 NCT00973713 NCT00390195 | 646–648 | |
| mTOR | Sirolimus (rapamycin) | Phase II | Single agent | Liver Transplantation for HCC(immunosuppressive) | Does not decrease HCC recurrence but prolongs OS after L DLT for HCC | Wound complication and dyslipidaemia, acute cellular rejection | NCT01374750 | 238 | |
| mTOR | AZD8055 | Phase I/II | Single agent | Patients With Advanced Stage HCC and With Mild or Moderate Hepatic Impairment | Completed | NA | NCT00999882 | 239 | |
| mTOR | Sirolimus | Phase I | Combined with bevacizumab (target VEGF) | Unresected Liver cancer | No survival benefit over bevacizumab-only treatment | Grade 3 thrombocytopenia and grade 3 mucositis. hyperglycaemia (83%), thrombocytopenia (75%), fatigue (46%), mucositis (46%), anorexia (42%), diarrhea (33%) and proteinuria (12.5%) | NCT00467194 | 241 | |
| mTOR | Temsirolimus | Phase II | Combinated with Sorafenib | Advanced Hepatocellular Carcinoma | Acceptable safety but did not achieve the target threshold for efficacy | Grade 3 (hypophosphatemia, thrombocytopenia, and rash) | NCT01687673 | 242 | |
| mTOR | Temsirolimus | Phase I/II | Single agent | HCC | Disease stabilization (defined as CR + PR + SD > 12 weeks) in tumors having high and low pMTOR H-scores to be 70% and 29% respectively (OR 5.667, 95% CI1.129–28.454, p = 0.035) | Grade ≥ 3 that occurred in > 10% of patients included thrombocytopenia (4) and hyponatraemia (4) | NCT00321594 NCT01251458 NCT01567930 | 649 | |
| TORC1/2 | Onatasertib (CC-223) | Phase II | Single agent | HBV-positive advanced HCC;Advanced solid tumor including HCC | Preliminary antitumor activity | Diarrhea (60.38%), hyperglycaemia (60.38%), thrombocytopenia (30.19%), hyperbilirubinaemia (11.32%) | NCT03591965 NCT01177397 | ||
| TGF-β pathway | TGF-βRI | Galunisertib | Phase II | Combined with sorafenib | Advanced HCC and Child-Pugh A liver function without prior systemic therapy | Median OS: 18.8 months;ORR: 4.5% | The most frequent treatment emergent adverse events of any grade: palmar-plantar erythrodysesthesia (56.8%), diarrhea (43.2%), and pruritis (25.0%) | NCT01246986 | 292 |
| TGF-βRI | Galunisertib | Phase I | Combined with stereotactic body radiotherapy | Advanced HCC who progressed on, were intolerant of, or refused sorafenib | Median OS: 9.0 months; median PFS: 3.7-months; ORR: 14.2% | The most common treatment-related adverse events were fatigue (53%), abdominal pain (46.6%), nausea (40%), and increased alkaline phosphatase (40%) | NCT02906397 | 187 | |
| TGFβ1/GARP | Livmoniplimab | Phase I/II | Combined with Budigalimab | Locally advanced or metastatic and/ or unresectable HCC | Not reported | Not reported | NCT06109272 | Not available | |
| Wnt/β-catenin pathway | PORCN | CGX1321 | Phase Ib | With Pembrolizumab or Encorafenib + Cetuximab | Advanced GI Tumors | Unkwown | NA | NCT02675946 | |
| PORCN | CGX1321 | Phase I | Dose Expansion | Advanced GI Tumors | Unkwown | NA | NCT03507998 | ||
| Wnt ligands | OMP-54F28 | Phase Ib | Combined with sorafenib | HCC patients | Completed | NA | NCT02069145 | ||
| DKK1 | DKN-01, (humanized monoclonal antibody) | Phase I | Combined with gemcitabine and cisplatin | Carcinoma to Primary to the Intra-or Extra-Hepatic Biliary System or Gallbladder | No additional activity beyond historically reported efficacy with gemcitabine/cisplatin alone. | Neutropenia (60%), thrombocytopenia (34%), and anemia (23%) | NCT02375880 | 328 | |
| β-catenin-CBP | E7386 | Phase Ib/II | Combined with pembrolizumab or pembrolizumab+lenvatinib | Previously Treated Subjects With Selected Solid Tumors, including HCC | Recruiting | NA | NCT05091346 | ||
| β-catenin-CBP | E7386 | Phase Ib | Combined with lenvatinib | Solid tumor including HCC | Recruiting | NA | NCT04008797 | ||
| β-catenin-CBP | E7386 | Phase I | Single agent | Solid tumor | Recruiting | NA | NCT03264664 | ||
| JAK/STAT pathway | JAK1 | Itacitinib | Phase Ib | Single agent | Advanced HCC | Ongoing | NA | NCT04358185 | |
| STAT3 | OPB-111077 | Phase I | Single agent | Advanced HCC | Limited preliminary efficacy outcomes | Thrombocytopenia (6%), fatigue (3%), and dizziness (3%) | NCT01942083 | 276 | |
| STAT3 | OPB-31121 | PhaseI/II | Single agent | Advanced HCC | Insufficient antitumor activity for HCC | Peripheral nervous system-related toxicities | NCT01406574 | 650 | |
| STAT3 | Napanucasin(BBI608) | PhaseI/II | Combined with sorafenib | Advanced HCC | No additional effect | No dose-limiting toxicities, diarrhea (83.3%) and palmar-plantar erythrodysesthesia syndrome (66.7%) | NCT02279719 NCT02358395 | 651 | |
| STAT3 | AZD9150 | Phase I/Ib | Single agent | Advanced/Metastatic HCC | Completed | Abdominal pain; Hepatorenal failure | NCT01839604 | ||
| Hedgehog pathway | Gli | Arsenic trioxide (ATO) | Phase II | Single agent | Unresectable metastatic liver cancer | Completed | NA | NCT00128596 | |
| Gli | Arsenic trioxide (ATO) | Phase II | Single agent | Advanced liver cancer | Terminated | NA | NCT00582400 | ||
| Smo | Vismodegib | Phase I | Single agent | Advanced solid malignancies (include HCC) and hepatic impairment | Completed | Four patients experienced dose-limiting toxicity of hyperbilirubinemia on study: one in the moderate cohort and three in the severe cohort. | NCT01546519 | 363 | |
| Hedgehog | LDE225 | Phase Ib | Single agent | Advanced or metastatic HCC and Child-Pugh A/B7 Cirrhosis | Completed | NA | NCT02151864 | ||
| Hippo pathway | TEAD | IK-930 | Phase I | Single agent | Solid tumors with or without YAP1/ TAZ Fusion Genes or NF2 deficient | Recruiting | NA | NCT05228015 | |
| Notch pathway | γ-Secretase | Ginsenoside (RG3) | Phase I | Combined with TACE | HCC with high expression of Notch1 (n = 320) | Completed | NA | NCT02724358 | |
| Telomere regulation | TERT | Telomelysin (OBP-301) | Phase I | Evaluate the Safety and Efficacy, non-comparative | HCC | Improved local control in patients with advanced HCC (SD78%) | Influenza-like illness, pyrexia, fatigue, decreased platelet count, 46 abdominal distension, and anemia | NCT02293850 | 446 |
| Epigenetics | Class I, class IIa and IV HDAC | Resminostat | Phase I/II | Alone or combined with sorafenib | Advanced HCC | Resminostat at the recommended dose plus sorafenib showed no significant efficacy advantage over sorafenib monotherapy | Gastrointestinal disorders, thrombocytopenia and fatigue | NCT02400788 NCT00943449 | 466,467 |
| Pan-HDAC | Belinostat | Phase I/II | Single agent | Unresected Liver cancer | Tumor stabilization, well tolerated | Abdominal pain, hyperbilirubinemia, increased ALT, anemia, and vomiting, grade 4 anemia | NCT00321594 | 652 | |
| Pan-HDAC | Panobinostat | Phase I | Combined with sorafenib | Advanced HCC,Metastatic and/or unresectable Liver cancer | Terminated | NA | NCT00823290 NCT00873002 | ||
| Pan-HDAC | Vorinostat | Phase I | Combined with sorafenib; combined with chemotherapy | Advanced liver cancer | Ten patients (77%) had stable disease (SD), The median treatment duration was 4.7 months for response-evaluable patients. | Anorexia, dehydration, dysgeusia, fatigue, lymphocytopenia, nausea, and thrombocytopenia | NCT01075113 NCT00537121 | 465 | |
| HDAC | Tefinostat | Phase I/II | Dose escalation | HCC | Completed | NA | NCT02759601 | ||
| DNMT | Decitabine | Phase I/II | Combined with Chemo- or immunotherapy | Patients with Refractory and/or Chemotherapy Resistant Solid Tumorsor B Cell Lymphomas | The lower-dose decitabine treatment resulted in beneficial clinical response and favorable toxicity profiles(The disease control rate (CR + SD rate) was up to 46.67%. The treatment prolonged PFS and OS to 4 and 11 months) | Generally well tolerated. The most commonly reported Aes were hematologic toxicity and gastrointestinal symptoms. | NCT01799083 | 463 | |
| DNMT | Guadecitabine (SGI-110) | Phase II | After failure of prior sorafenib | Advanced HCC | Median OS: 245(148–303); Median PFS:82.5(57–113) | Pain in extremity, Abdominal pain, Febrile neutropenia | NCT01752933 | 653 | |
| DNMT | Guadecitabine(SGI-110) | Phase Ib | Combined with durvalumab | Digestive tumors including HCC | Active, not recruiting | NA | NCT03257761 | 654 | |
| P53 signaling | MDM2 | ASTX295 | Phase I | Dose escalation | Solid tumors with wild-type p53 | Active, not recruiting | NA | NCT03975387 | |
| MDM2 | HDM201 | Phase II | Different matched targeted therapy | Primary tumor or metastatic lesio solid tumors with specific oncoge advanced/metastatic solid tumors | Recruiting | NA | NCT04116541 | ||
| MDM2 | Idasanutlin MT | Phase II | Tumor-agnostic precision immuno-oncology and somatic targeting rational for you (TAPISTRY) platform study | Solid tumors with specific oncogenic genomic alterations or TMB | Recruiting | NA | NCT04589845 | ||
| MDM2 | Milademetan | Phase II | Single agent | Advanced/ metastatic solid Tumors | Terminated | NA | NCT05012397 | ||
| TP53 Y220C | PC14586 | Phase I/II | Alone and combined with pembrolizumab | Solid tumors with p53 Y220C mutation | Recruiting | NA | NCT04585750 | ||
| Mutant p53 | Arsenic trioxide | Phase II | Single agent | Refractory solid tumors with rescuable p53 mutation | Unknown status | NA | NCT04869475 | ||
| WT p53 | Ad-p53 | Phase II | Combined with immune checkpoint inhibitors | Solid tumor approved for anti-PD-1 or anti-PD-L1 Therapy | Unknown status | NA | NCT03544723 | ||
| Bcl-2 | Navitoclax | Phase I | Combined with Sorafenib | Solid tumor with HCC expansion cohort | This combination was tolerable but had limited efficacy in the HCC expansion cohort, with stable disease as best response in 6 (40%) HCC patients. | Thrombo7cytopenia, increased AST, fatigue, increased ALT, diarrhea, increased alkaline phosphatase, and rash. | NCT01364051 | 503 | |
| CDK 4/6 | Palbociclib(PD-0332991) | Phase II | Single agent | Advanced HCC | Ongoing | NA | NCT01356628 | ||
| CDK 4/6 | Ribociclib(LEE011) | Phase II | Combined with chemoembolization | Locally advanced HCC | Completed | NA | NCT02524119 | ||
| CDK 4/6 | Abemaciclib | Phase II | Combined with Nivolumab | HCC | Completed | NA | NCT03781960 |
TGF-β signaling
The transforming growth factor (TGF-β) family is the most diverse and far-reaching family of cytokines in the human body and is expressed in almost all types of tissues and cell types.283 This superfamily consists of 33 multifunctional factors, and TGF-β isoforms (I-3) are the typical members, with TGF-β1 being the most abundant and well-studied.284 Three isoforms of the TGF-β receptor exist, types I, II, and III, the first two being single transmembrane glycoproteins with serine/threonine kinase activity, and the latter being proteoglycan with no kinase activity.285 TGF-β is a major player in tumorigenesis. It has a well-known dual effect in cancer and is no exception for HCC.286 For normal tissues, TGF-β signaling inhibits cell proliferation and induces apoptosis or differentiation by blocking cells at the G1 phase or prolonging the G1 phase, thus acting as a tumor suppressor in precancerous cells.285 In the early stages of carcinogenesis, TGF-β inhibits tumorigenic inflammation or triggers apoptosis in precancerous progenitor cells carrying RAS mutations. As tumors progress to advanced stages, TGF-β signaling gradually exhibits pro-tumorigenic effects such as induction of immunosuppressive microenvironment, EMT, and promotion of angiogenesis.283 In HCC, downregulation of the TGF-β signaling pathway may imply loss of cancer suppressive activity, while upregulation leads to fibrosis and inflammation.287 Nearly 40% of HCC patients have somatic mutations in at least one member of the TGF-β pathway.287
Increasing preclinical and clinical evidence suggests that TGF-β signaling activation in established anti-cancer therapies such as targeted therapies, chemotherapy, and radiation is a driver of drug resistance,288,289 involving multiple mechanisms such as induction of EMT, metabolic reprogramming, activation of alternative pathways, promotion of an immune-suppressive microenvironment, induction of a stem cell-like phenotype, and promotion of drug uptake and efflux.290,291 Therefore, the prevailing view is that therapies combining TGF-β signaling inhibitors with other anti-tumor therapies is a promising strategy that not only inhibits advanced tumor progression but also reverses treatment resistance. However, TGF-β receptors and their ligands are widely distributed in vivo, so potential systemic cytotoxicity is a major obstacle. Currently, the only FDA-approved TGF-β inhibitor, liuspatercept, is indicated for anemia in adults with Myelodysplastic Syndromes (MDS). For HCC, a small molecule inhibitor targeting TGFβR1, galunisertib, in combination with sorafenib as first-line therapy for advanced patients has demonstrated a manageable safety profile and improved OS.292 The combination of galunisertib with stereotactic body radiotherapy demonstrated tolerable toxicity in a pilot study.187 However, galunisertib did not show sensitization to VEGFR inhibitors in HCC patients as shown in preclinical studies.293 In addition, a neutralizing antibody, NIS793, was recently tested for safety in advanced malignancies, including HCC, supporting the further exploration.294 A phase III trial was recently initiated to test the efficacy and safety of livmoniplimab, a humanized monoclonal antibody against the LRRC32/TGF β 1 complex, in combination with a PD-1 inhibitor in advanced HCC (NCT06109272). Considering the dual role of TGF-β in tumors, targeting the TGF-β pathway may only be effective in specific HCC patients, thus identification of biomarkers is warranted. For example, previous studies have found that upregulation of SMAD7, CLTC, and CXCR4 is associated with the pro-carcinogenic profile of TGF-β, and it may be worthwhile to further test whether clusters with high expression of these genes are more likely to respond to TGF-β inhibitors.295
Wnt/β-catenin signaling
Without the presence of extracellular Wnt ligands, the canonical Wnt signaling is inactive. β-catenin is located in adherent junctions and cytoplasm of the cell, where it becomes phosphorylated by the destruction complex (containing adenomatous polyposis coli protein (APC), Axin, casein kinase I isoform-α (CKIα) and glycogen synthase kinase 3 β (GSK3β) and targeted for ubiquitylation and proteasomal degradation. Wnt signaling is tightly regulated intracellularly by the canonical molecules in this pathway mentioned above. It is also modulated by extracellular antagonists and inhibitors, such as Wnt inhibitory factor 1, Dickkopf-related proteins (DKKs), and other novel potential regulators.296 Here, we focused on the role of Wnt/ β-catenin in HCC. Wnt-β-catenin signaling is commonly overactive in HCC. 20–35% of HCCs have genetic mutations and/or aberrant activations of key genes involved in this cascade.297 Human HCCs with aberrant Wnt/ β-catenin activation present distinct clinical, pathological, and molecular characteristics. The gain-of-function mutations of CTNNB1 occurred in HCCs with HCV (28%),298,299 alcohol (42%) 70 and non-cirrhotic liver in the absence of common risk factors of HCC.300 While AXIN1 is more frequently mutated in HBV-related HCCs (18%) than in HCV-related or non-viral HCCs (14% and 8%, respectively).299 Mutations of Wnt/β-catenin were more often identified to be associated with nonproliferation subgroups of HCC but also found in proliferation group.300
Wnt-β-catenin has been connected to HCC stemness, progression, metastasis and drug resistance. As an illustration, elevated activity of β-catenin prominently leads to proliferation, self-renewal and in vivo hepatocarcinogenesis of CSCs in HCC.301–303 In transgenic mice models, activation of Wnt/ β-catenin was found to induce HCC formation when cooperated with other-oncogenic signaling such as activated c-Met,304–306 K-Ras,307 Akt,308 LKB1,309 and Nrf2310. Co-expression of these oncogenes and activation of Wnt/ β-catenin were also found in human HCC tissues.83,304,310 The Wnt/β-catenin axis exerts crucial effects in HCC through regulating the downstream target genes. β-catenin induced c-MYC expression311,312 is implicated in gankyrin-driven glycolysis, glutaminolysis313 and sorafenib responsiveness in human HCC.314 Another direct target of this signaling, cyclin D1, was frequently found to be upregulated by activation of Wnt-β-catenin in mouse and human HCC.315,316 The interlinked and feedback mechanisms between cyclin D1 and Wnt-β-catenin were involved in hepatocarcinogenesis and HCC metastasis.300,317 Other target genes of Wnt/β-catenin such as glutamine synthetase (GS)318,319 and KIF2C320 were found to link with mTOR cascade and promote HCC growth.321,322 Activation of Wnt/β-catenin has also been linked to resistance against Lenvatinib, sorafenib and regorafenib in HCC patients.302,323,324 Above all, the activities of the Wnt/ β-catenin cascade during hepatocarcinogenesis have not been clearly characterized and need further investigation.
Porcupine (PORCN) is an O-acyltransferase indispensable for Wnt ligand secretion,325 whose inhibitor CGX1321 is tested in patients with advanced solid tumors, like HCC and CCA (NCT02675946, NCT03507998). OMP-54F28, which binds Wnt ligands competing with native Frizzled 8, was assessed in a phase I study in combination with sorafenib in HCC patients (NCT02069145). Dickkopf-1 (DKK1) is known as an extracellular antagonist of Wnt,326,327 thus inhibition of DKK1 can suppress β-catenin signaling. DKN-01, a DKK1-neutralizing monoclonal antibody is being evaluated in a phase I trial in combination with gemcitabine and cisplatin in patients with CCA or gallbladder cancer (NCT02375880), but did not show additional activity beyond gemicitabine/cisplatin alone, which may be due to heterogeneity of differential activity for DKN-01 to different DKK1 expression or the need for increased dose/intensity.328 Whereas the effect of DKN-01 in HCC keeps unknown.
Niclosamide is a FDA-approved drug and was used to treat taeniasis, showing inhibiting effect on tumors proliferation, stemness and metastasis with limited toxicity in other cancers,329,330 providing hopeful optimism for human HCC therapy. Besides, kinds of common drugs in clinic, such as vitamin D and retinoic acid, have been reported to block the interaction of β-catenin with TCF, NSAIDs, such as aspirin and sulindac, have been proved to strengthen the degradation of β-catenin, showing promising potential in preclinical and clinical settings for liver cancer including HCC therapy.297
The majority of agents targeting Wnt-β-catenin axis have been evaluating in preclinical studies, providing the rationale for clinical assessment. Kinds of inhibitors showed growth suppressive effect in HCC, including PORCN inhibitors (LGK-974, ETC-159),331 fungal derivatives (PKF115-854 and CGP0449090),332,333 TNKS inhibitor XAV939.334 Interfering RNA– or antisense RNA–based therapy constitutes another method to suppress the Wnt/β-catenin pathway, showing suppressive effect to HCC in vitro335,336 and in mouse models.307,337
In summary, preclinical studies have assessed the therapeutic prospect of targeting this pathway against cancers, including HCCs. The clinical development of these agents has been rather restricted to date. Therefore, the therapeutic effects for targeting Wnt-β-catenin pathway remain elusive.
Hedgehog signaling
Hedgehog (Hh) signaling regulates cell proliferation, differentiation, tissue homeostasis, and carcinogenesis.338,339 It is composed of the Hedgehog ligand, nuclear transcription factors and two transmembrane protein receptors, Patched-1 (Ptch1) and Smoothened (Smo).340 In humans, there are three types of Hedgehog ligands: Sonic Hedgehog (Shh), Indian Hedgehog (Ihh) and Desert Hedgehog (Dhh), which are disparate in the timing of expression, spatial distribution and action characteristics.341,342 The role and mechanism for Shh is the most studied.342 When Hedgehog ligands are absent, a Glioma-associated oncogene (Gli) is phosphorylated343–347 and then undergoes proteolytic cleavage, generating Gli repressor (GliR).345 GliR binds to the promoters of Hedgehog target genes and represses their transcription. When Hedgehog ligands bind with the receptors, the canonical Hedgehog signaling pathway is activated.338 The binding of Hedgehog ligands to Ptch alleviates the restraint of Smo by the receptor protein, resulting in the movement of Smo to primary cilium (PC).338,348,349 The existence of Smo in PC hinders phosphorylation and proteolytic cleavage of Gli, avoiding the generation of GliR. Thus, the promoters of Hedgehog target genes are available to transcription. Hedgehog signaling pathways are widely investigated to crosstalk with the Notch during cell development in various systems and in cancer biology.350
The activation and oncogenic effect of Hh signaling were investigated in multiple levels of HCC cell lines, mice models and clinical samples. The increased levels of Shh and its target genes like Ptch, smo and Gli were detected in human HCC tissues and HCC cell lines, suggesting activation of Hedgehog Signaling in human HCC.351–355 In detail, Shh is overly expressed in approximately 60% of human HCC, Ptch1 and Gli1 are expressed in over 50% of the tumor.355–358 Hepatic activated Hh signaling led to hepatic fibrosis and hepatocarcinogenesis.357,359 Hedgehog pathway inhibition reduce HCC growth in cell lines and mouse models.353,360 Shh signaling pathway provokes cell migration and invasion of HCC cell through Shh and Gli1.361,362 Above all, activation of Hh signaling is oncogenic in HCC. Given its participation in the initiation and development of HCC, Hh targeted therapies could be a hopeful strategy to fight against HCC.
Targeting Gli with arsenic trioxide (ATO) in has been assessed in phase II clinical trials in treating patients with metastatic liver cancer (NCT00128596), and advanced primary carcinoma of the liver (NCT00582400). A Smo inhibitor vismodegib, was assessed in phase I clinical trial includes patients with HCC (NCT01546519). However, the causal relationship between vismodegib exposure and serious adverse events are ascertain due to the high number of patients with advanced HCC with cirrhosis.363 Sonidegib is a Hedgehog pathway inhibitor approved for treatment of laBCC (US, EU, Switzerland, and Australia) and metastatic BCC (mBCC; Switzerland and Australia).364 While in HCC, it remained in phase I clinical stage (NCT02151864) with unpublished results. Thus, the clinical effectiveness of Hedgehog signaling remains unclear.
Preclinical studies targeting the Hedgehog signaling pathway mainly concentrate on suppressing the activity of Smo. Except vismodegib in clinical assessment, cyclopamine is also a smo inhibitor, which can increase apoptosis and inhibted cells growth and proliferation in diverse HCC cell lines like Hep3B and Huh7.352,355,365–367 The anti-tumor effects and impact on drug resistance of various agents targeting Gli have also been assessed. GANT61 is a selective small molecular inhibitor of Gli1 and Gli2-mediated transactivation, displaying anti-tumor effect on cell viability, proliferation and migration in HCC cell lines in vitro and growth in xenograft model, as well as the drug resistance of CD44+ HCC patient derived organoids.368–372 Another study showed that Bufalin (Bu), one of the topoisomerase II inhibitors, affecting the expression of Gli1 and Gli3 and exhibits consistent inhibitory effect in Huh7, Hep3B and HepG2 cells.373 Inhibition of Gli by RNAi can also effectively reverse sorafenib, 5-FU, doxorubicin, and cisplatin resistance in HCC cell lines.369 A previous study generated a polymeric nanoparticle-encapsulated Gli inhibitor HPI-1 (NanoHHI) and demonstrated its tumor growth inhibition and antimetastatic effects in an orthotopic model of human HCC.374 Some drugs originally intended for other pathways and diseases have also been found to inhibit the Hh pathway. They can act as unselective inhibitors of the hedgehog pathway and exerts anti-tumor effects in HCC, including ITCZ,375 Taccalonolide A,376 human sulfatase 2 inhibitor 2,4-disulfonylphenyl-tert-butylnitrone (OKN-007).377
HIPPO/YAP signaling
Hippo signaling was firstly recognized as a crucial determinant for organ size,378 emerging as a vital player in liver biology, such as liver development, cell fate determination, homeostasis and regeneration from injury.379 The cascade is initiated by neurofibromatosis 2 (NF2) and kidney and brain expressed protein (KIBRA) when transmembrane receptors transduce signals to them. The canonical Hippo signaling pathway acts to suppress the activity of YAP/TAZ. When the Hippo pathway is inactivate, the abolition of YAP phosphorylation by MST1/2 enables YAP to translocate into the nucleus, subsequently interacting with the transcription factor TEAD to mediate the target genes expression such as RUNXs, SMAD, PPARG.379 Hippo/YAP signaling exerts a deep impact on the physiopathology of liver diseases including hepatic malignancies. In HCC, Hippo signaling is involved in various oncogenic effects such as cell proliferation and apoptosis, cell cycle and differentiation. YAP activation is an early occurence in HCC development. Genomic amplification of the genomic locus which contains the YAP gene (11q22) is found in 5–10% of HCC,380 and increased YAP activity is found in 65–85% of HCC.381,382 Activation of YAP is associated with more aggressive subtypes of HCC and is an independent prognostic marker in HCC.383–385 Activation of Hippo acts as tumor suppressor, while YAP/TAZ activity has shown to promote carcinogenesis in HCC. For instance, Nf2-deletion, hepatic inactivation of HPO1/2 lead to generation of HCCs, iCCAs (intrahepatic cholangiocarcinomas), and mixed hepatocellular cholangiocarcinomas.386–389 Both YAP and TAZ are required for c-Met/sgAxin1-dependent hepatocarcinogenesis using conditional knockout (KO) mice.390 Ablation of TAZ completely prevented c-MYC-induced hepatocarcinogenesis in knockout mice.391 Multiple regulators participate in the oncogenic activity of YAP in HCC, such as NUAK2,392 Notch signaling,393 Ccl2 related immune regulation.394,395 Other signalings can interact with Hippo pathway to promote HCC, such as Notch pathway, EGFR signals, AKT pathway and DDR1 signaling396–399
Considering the oncogenic roles of YAP in liver cancer and other disease, it is attractive to develop inhibitors targeting YAP. However, the currently developed selective YAP activity inhibitors were clinically evaluated in other cancers such as non-small cell lung cancer and gastric cancer rather than HCC.391 Several agents target other pathways were found indirectly regulate Hippo/YAP signaling and against HCC, such as Statins (fluvastatin and simvastatin),400 Salvianolic acid B (Sal B)401 and Tankyrase inhibitor G007-LK.390 However, above unselective agents may exist potential off-target toxicity. More selective strategies have been developed to decrease potential off-target effects. For example, NUAK2 has been identified as a critical downstream target of YAP during liver tumorigenesis, and pharmacological inhibition of NUAK2 suppressed YAP-driven tumor growth in vivo.402 NIBR-LTSi, a selective small-molecule LATS kinase inhibitor was characterized to activate YAP signaling and blocks differentiation in vitro and in vivo, also accelerate liver regeneration following extended hepatectomy in mice,403 suggesting a clinical potential after resection therapy for HCC. However, the adverse effect due to the off-target effect and the risk for promoting proliferation of HCC cells need further investigations. A phase I clinical trial of assessment for IK-930, an oral TEAD inhibitor targeting the Hippo pathway in advanced solid tumors was ongoing (NCT05228015). Above all, the clinical assessment of agents targeting Hippo signaling in HCC is lacked and the off-target effects requires great attention.
Notch signaling
The mechanisms of the canonical and non-canonical Notch signaling pathways have been reviewed previously.404,405 Briefly, Notch signaling is initiated through the binding between a transmembrane receptor and a membrane-spanning ligand on adjacent cells. The major components and distinct steps in the Notch pathway have been widely investigated and are summarized briefly (Fig. 3e). Non-canonical Notch signaling can be started by a non-canonical ligand, or in the absence of a ligand, or may not need cleavage of the Notch receptor, or interactions with other cytoplasmic or cytosolic effectors.406,407 In addition, Notch signaling crosstalks with others like NF-κB, mTORC, TGF-β, AKT, Wnt, or Hippo to modulate target gene transcriptions.408–412 Notch pathway can function as both carcinogens and tumor suppressors depending on the context of cancers and cell populations. Abnormal activation of wild-type Notch signaling and its role in carcinogenesis and progression can be observed in HCC413 and other aggressive tumors.414–416 The oncogenic function of activated Notch signaling were more frequently found in HCC.417–419 Notch signaling is overactivated in human HCC and mouse HCC models, downregulation of Notch1/Jagged1 signaling attenuated HCC progression in mice.420,421 Notch signaling also promotes stemness, poor differentiation, epithelial-mesenchymal transition and tumor metastasis in HCC.422,423 Notably, inhibiting Notch activity using DAPT causes the differentiation of HCC CSCs into functional hepatocytes via mesenchymal–epithelial transition, indicating that targeting Notch in CSCs may be applied as differentiation therapy for HCC.424 Similarly, blocking canonical Notch signaling or treatment with the anti-Notch2 antibody in AKT/Ras mice led to the development of well-differentiated HCC and loss of intrahepatic cholangiocarcinomas-like lesions.425,426
Fig. 3.
Targeting the other critical signaling pathways in HCC cells. Canonical pathways and pharmacological inhibitors under investigation in HCC for TGF-β pathway (a), Wnt pathway (b), Hedgehog pathway (c), Hippo pathway (d), Notch pathway (e). The red fonts indicate agents under clinical assessment in HCC patients, the black fonts indicate agents under preclinical investigations. Figure was created with biorender.com
Nevertheless, pan-Notch blockade in the triple-knockout mice by using of DAPT resulted in accelerated HCC development and higher expressions of Notch-related genes, thereby supporting a tumor-suppressive role for Notch signaling in HCC.424 Recent work indicates that a portion of the tumor-suppressive role of Notch in HCC might not be cell autonomous, and could be associated with crosstalk with Wnt in liver specific tumor-associated macrophages (TAMs).427
In general, considering the crucial role of Notch in HCC and blockade of the Notch signaling exhibits alleviated effect in HCC progression, it keeps potential to explore clinical therapeutic effectiveness of targeting Notch signaling in HCC. Diverse classes of Notch-targeting therapeutics have been developed during the past decade, and several agents have been evaluated in clinical trials. Here we sum up the most recent efforts in drug discovery and development targeting Notch signaling in HCC. Present targeted strategies in the clinic mainly converge on modulating γ-secretase and blocking ligand-receptor interaction via related monoclonal antibodies.428 γ-secretase inhibitors (GSIs) were the first and most extensively studied small molecule Notch inhibitors, which were initially developed for Alzheimer disease (AD),429 then were used as anti-tumor agents due to the target of Notch signaling.430 Nirogacestat was approved in the USA for use in adult patients with progressing desmoid tumors who require systemic treatment. However, no clinical advance in HCC.431
γ-Secretase modulators (GSMs) are small molecules that keep part of Notch signaling function by modifying the catalytic activity of γ-secretase.432 Ginsenoside (Rg3), a natural product from Panax ginseng with GSM properties,433 exerts anti-tumor effect by inhibiting NOTCH-Hes1 signaling.434,435 It is the only agent targeting Notch signaling has been assessed in a phase I trial (NCT02724358). The result showed that the combination of Rg3 and TACE prolonged OS than TACE monotherapy in HCC patients with high NOTCH1 expression.436 Later, Rg3 presented a synergistic anticancer effect for sorafenib in HCC cell lines and mouse model,437 which needs further preclinical and clinical exploration. ADAM proteases act as ‘sheddases’ for a great many of membrane proteins including Notch receptors and Jagged 1. Blocking Notch pathway by ADAM17 inhibitor ZLDI-8 may sensitize HCC cells to sorafenib in vivo and in vitro by affect crosstalk between the Notch1 and Integrinβ/ILK signaling pathways in HCC in vivo and in vitro.438
Despite the limited therapeutic advance targeting Notch signaling, it keeps attractive to evaluate the clinical therapeutic effectiveness of this signaling combined with other approved treatments in HCC. Taking into account the multiple roles in the development and homeostasis of the immune system, the pharmacological manipulation of Notch has promising applications in cancer immunotherapy.
Telomere regulation pathway
Telomeres exist in all mammalian cells and their shortening with cell proliferation promotes cell arrest, senescence and apoptosis. Telomeres terminate with a 50–200 nucleotide of TTAGGG repeats single-stranded 3′ overhang that can invade preceding telomeric dsDNA to form a stable telomere loop (T-loop) structure with shelterin at each end of chromosome, which prevent against the loss of telomeres during cell division.439 Telomeres are produced by telomerase, which consists of telomerase reverse transcriptase (TERT), the telomerase RNA component (TERC), and dyskerin.440 TERT uses an TERC as RNA template to synthesize single-stranded TTAGGG repeats and has a crucial role in telomere maintenance. TERT levels typically act as the limiting factor for telomerase activity in somatic human cells.
Telomere attrition acts as a barrier to replicative immortality, dysfunction of which enables cancer cells to overcome the replicative death, thereby providing a rationale for its therapeutic application in cancer.439,441 As the most prevalent somatic mutations, TERTp was observed mutated in up to 60% of human HCC and has been proved to closely relate to sequential hepatocarcinogenesis. The reactivation of TERT mainly through various mechanisms such as mutations in the chromosomal rearrangements (5–10%), promoter (30–60%) and gene amplification, it results in a re-expression and enhanced activity of TERT, which boosts cell survival and proliferation, and against senescence.440,442
Targeting telomerase have been developed and assessed as new therapies in HCC. When the expression of TERT was silenced with antisense oligonucleotides in human liver cancer cell lines and in xenograft mouse models, it led to proliferation arrest and death of tumor cells. This provided preliminary evidence for the therapeutic potential of TERT inhibition in HCC.443,444 BIBR1532, a small molecule competitively blocking the active site of telomerase, has been linked to telomerase inhibition and HCC cell death in preclinical studies.445 However, there is no data in late phase clinical trials.
There are no agents directly targeting TERT in HCC clinical trial. The only clinical application is Telomelysin (OBP-301), a TERT-driven oncolytic adenovirus that specifically introduces the TERT promoter into tumor cells, which is being investigated in a phase I trial in HCC.446 In this study, the therapeutic efficacy of OBP-301 was less than that of other second-line systemic targeted therapies. However, OBP-301 injection increased infiltration of CD8+ T cells and <1% PD-L1 expression in tumor, suggesting its potential in combination with other immuno-therapeutics.
Conclusively, inhibitors targeting the TERT pathway have been studied in cellular and murine models of HCC (Fig. 4a); however, they have not yet undergone clinical trials for therapeutic evaluation, leading to elusive therapeutic effects. Currently, the only related product in clinical trials is the oncolytic adenovirus OBP-301, which exploits TERT promoter activity to achieve anti-tumor effects through virus replication. Nevertheless, its therapeutic effects appear inferior to those of second-line targeted agents.
Fig. 4.
Nuclear signaling pathways. Canonical pathways and pharmacological inhibitors under investigation in HCC for pathways intersecting with nuclear signaling. a Telomere regulation pathway. b Epigenetic modification. c P53 regulation and signaling. The red fonts indicate agents under clinical assessment in HCC patients, the black fonts indicate agents under preclinical investigations. Figure was created with biorender.com
Epigenetic pathways
Epigenetic modifications are inheritable variations in gene activity that do not change the DNA sequence.447 The two primary types of epigenetic modifications are DNA methylation and histone post-translational modifications (PTMs). DNA methylation occurs mainly at CpG sites, primarily found in CpG islands within regulatory regions of genes. DNA methyltransferases (DNMTs) introduce methyl groups, while ten-eleven translocation enzymes (TET1/2/3) actively remove them. Global hypomethylation of DNA is common in cancer, promoting chromosomal instability and reactivation of endogenous retroviral sequences.448 The analysis based on DNA methylation profiles of 304 HCC patients discovered a CpG methylation signature that is correlated with patient survival, among which, IGF, PI3K, TGF-β and WNT pathways were mostly dysregulated in HCC by DNA methylation.449 DNA is wrapped around histone proteins to form nucleosomes, where histone proteins undergo various PTMs such as acetylation, methylation, phosphorylation, and more. These modifications, along with DNA methylation, regulate chromatin conformation and accessibility to regulatory elements. Histone methyltransferases and demethylases, such as EZH2, EHMT2, SETDB1 and SETD2 were found to be correlated with the clinical characteristics of human HCC tissues.450 EZH2-mediated H3K27me3 signifies a major oncogenic chromatin modification and is involved in sorafenib resistance and HCC growth.451–453 Other specific histone modifications are also dysregulated in HCC. For instance, the total levels of H3K9me2 and H3K9me3 are commonly found higher in HCC tissues and positively correlates with tumor differentiation and poor prognosis.454 Further, specific DNA methylation and histone methylation can act as diagnostic biomarkers in HCC screening.455 Histone acetylation, regulated by histone acetyltransferases (HATs) and histone deacetylases (HDACs), is associated with gene activation, while histone deacetylation is correlated to gene repression and silencing.456 Histones acetylation and deacetylation were dysregulated and play important roles in HCC progression via modification enzymes. An instance is MOF, a HAT, can promote HCC growth and vascular invasion via acetylating histone H4K16.457,458 The expression of the acetylated H3 and H4 reader BRD4 is also elevated in HCC.459,460
Unlike genetic changes, the epigenetic modifications are reversible and have a more extensive impact on gene expression than genetic changes, providing potential new targets with more efficacy and safety.461,462
Epigenetic drugs in clinical trials for HCC include DNMTi and HDACi, most of which are in combined with other therapies. DNMT inhibitor Guadecitabine (SGI-110) (NCT01752933) and decitabine (NCT01799083) were assessed in phase I/II trial for pretreated patients with advanced HCC, in which decitabine showing beneficial clinical response and favorable toxicity profiles.463 A phase Ib evaluation for combination of guadecitabine with PD-L1 antibody durvalumab (NCT03257761) is underway. Several HDACs inhibitors are approved by FDA for the treatment of hematological malignancies, while remained in early phase clinical trials for HCC. For instance, Belinostat (PDX-101) exhibited tumor stabilization and generally well-tolerated in phase I/II study (NCT00321594).463 Belinostat also achieved complete tumor rejection in a murine HCC model when combined with CTLA-4 and PD-1 blockades.464 Another HDACi tefinostat was assessed in phase I/II clinical test (NCT02759601). Other phase I/II trials also tested the effect of HDACi when combined with sorafenib, including panobinostat, resminostat and vorinostat (NCT00823290, NCT00873002, NCT00943449, NCT02400788, NCT01075113). Vorinostat in combination with chemotherapy (FOLFIRI) was evaluated in patients with digestive cancers including HCC (NCT00537121). However, the results of the completed trials among above did not show significant and clear improvement over sorafenib while led to some toxicity in patients.465–467 Other agents and combined treatments showed therapeutic potential in HCC cell lines or murine models, such as pharmacological inhibition of histone methyltransferase EZH2 by GSK343,468 CM272, a dual inhibitor of DNMT1 and G9a,469–471 combined inhibition of DNMT and EZH2 by 5-Aza-CdR (DAC) and GSK126.472 Notably, a selective HDAC8 inhibitor PCI-34051 was found to stimulate antitumor immunity, and mice treated with combination of PCI-34051 and anti-PD-L1 antibody were safeguarded against subsequent tumor rechallenge and stayed tumor-free for over 15 months.473 Overall, above studies underline the potential of combining epigenetic inhibitors and ICIs in HCC treatment. Similar synergistic effect for ICIs were found in other epigenetic modulation agents, including BRD4 inhibitor (JQ-1,474–476 i-BET762477) and other BET domain inhibitors,478–480 as well as lysine demethylase 1A (KDM1A)481 and NAD-dependent deacetylase sirtuin 2,482 while the mechanisms need further study to provide more clues for therapeutic applications (Fig. 4b).
Cell death pathway
The p53 protein encoded by TP53 gene is widely recognized as a tumor suppressor protein that acts as a stress responder and transcription factor.483,484 Under physiological and non-stressed conditions, the expression of p53 is kept at a low level by negative regulators, especially MDM2 and E3 ubiquitin ligases.485 When stimulated by stress signals, mostly like DNA damage, p53 can transcriptionally regulate the expressions of genes to exert multicomplex functions. The classical and earliest recognized functions include cell-cycle arrest, apoptosis, and senescence, and other functions such as metabolism and ferroptosis.485 Mutations of TP53 typically lead to suppressed activity and occur in the majority of human cancers,486 which is observed in 31–36.1% patients with HCC.20,487 TP53 mutations are related with decreased protein levels488 and reduced OS in HCC patients.20,489 Inactivation of p53 and overactivation of MDM2 are discovered to contribute to the carcinogenesis of HCC in the context of viral infection and metabolic disease, and disrupting MDM2-p53 interaction can liberate p53.490
Targeting p53 is highly appealing for the development of anti-cancer drugs. The main strategies encompass small molecular inhibitors restoring its pro-apoptotic activity by inducing the refolding of the mutant p53 or blocking the abnormal degradation of p53 through interruption of MDM2-p53 interactions, as well as reestablishing the expression level and function of p53 with gene therapy.486,491 To date, several small molecules have been evaluated in clinical trials for the treatment of solid tumors rather than specifically HCC, including inhibitors blocking the activity of MDM2, such as HDM201(NCT04116541), ASTX295 (NCT03975387), milademetan (NCT05012397), and idasanutlin MT (NCT04589845); small molecules targeting mutant p53, PC14586 (NCT04585750) and arsenic trioxide (NCT04869475); and recombinant human adenovirus gene therapy, Ad-p53 (NCT03544723), which was approved by the China Food and Drug Administration (CFDA) in 2003 as the first gene therapy for treatment of head and neck cancer.492 For liver cancer, the combination of Ad-p53 with TACE is reviewed to enhance OS and quality of life compared to TACE monotherapy.493 Similar improvements in the clinical outcomes were observed in the combined therapy of Ad-p53 with fractionated stereotactic radiotherapy.494
Recently, p53 mRNA delivery by nanoparticles shows potential anticancer effect alone or in combination with immune checkpoint blockade in HCC mouse models.495,496 Targeting p53 is typically regarded as suppressing tumors through apoptosis and cell cycle arrest. An example is adiponectin, a wildtype p53 activator that can target p53/TRAIL/caspase-8 axis to attenuate HCC progression.497 A new molecule, SLMP53-2, can provoke cell-cycle arrest and apoptosis by activating p53 DNA-binding ability in vitro.498 Some new extracts from plants can suppress HCC by promoting apoptosis through activating p53/Bax pathway, such as oleanolic acid and acetylshikonin.499,500 Although increasing studies have investigated the therapeutic potential of restoring the pro-apoptotic effect of p53,486,491,501 no p53-specific drug has yet been approved by FDA or EMA.486,502 A few anticancer agents have been investigated in HCC for their direct targeting of apoptotic pathways through the inhibition of anti-apoptotic BCL-2 family members. For instance, navitoclax plus sorafenib demonstrated limited efficacy a phase I clinical study in patients with solid tumor with HCC expansion cohort (NCT01364051).503 Other Bcl-2 inhibitors, ABT-199 and obatoclax were evaluated in preclinical studies in combination with Mcl-inhibitor MIK665 and immune checkpoint inhibitors.504,505 Targeting apoptosis is not yet under clinical evaluation. Some agents were found to induce apoptosis in HCC cells by targeting other members of the apoptosis pathway, such as scutellarein targeting Fas/FasL,506 Garciniaxanthone I,507 Compound 19b and lycorine targeting Cyt-C.508,509 P53 also transcriptionally regulates the expressions of cyclins and cyclin-dependent kinases (CDKs), which lead to cell cycle arrest. The dysregulation of CDKs and cyclins is a hallmark of cancer, especially CDK4 and CDK6.
Three CDK inhibitors (palbociclib,501 ribociclib510 and abemaciclib511 are approved by FDA and EMA for the treatment of breast cancer, and exhibited antiproliferation activity in HCC cells and animal models,512–514 but have only been tested in clinical trials alone or in combination with Regorafenib and Lenvatinib (NCT01356628, NCT02524119 and NCT03781960) with unavailable results284 (Fig. 4c).
There are multiple dysregulated signaling pathways are involved in the development and progress in HCC, except for the well-known RTK signaling pathway (Figs. 3 and 4). These common signaling pathways applied for clinical trial keeps limited (Table 2 and Table 3). Among which, a frequently explored pathway is PI3K-AKT cascade, several inhibitors (Copanlisib, MK-2206, RAD001, sirolimus, Onatasertib, Temsirolimus) have been assessed in Phase II study, though showing limited efficacy. The majority of agents targeting these signaling axis remain in preclinical research and some in phase I study for dosage and safety evaluation. Thus, the main challenges for therapeutically targeting signaling pathway are preliminary effectiveness and toxicity. The lack of effectiveness might be related to the intrinsic and acquired drug resistance, as well as the heterogeneity of HCC.284
Table 3.
Summary for signaling pathways in preclinical stage for HCC therapy
| Signaling pathway | Target | Agent | Models | Level | Effect | Refs |
|---|---|---|---|---|---|---|
| PI3K/AKT/mTOR pathway | PI3K | DZW-310 | Hep3B, MHCC97H, Hep3B xenograft model | In vitro and in vivo | Attenuate angiogenesis and tumor growth | 243 |
| PI3K | Copanlisib | Huh7, HepG2 | In vitro | Induce apoptosis, inhibit cell growth, cell cycle arrest | 245,248 | |
| PI3K | LY294002 | MHCC97H, Huh7, Mahlavu | In vitro | Induce apoptosis, suppress cell growth | 244,246,655 | |
| PI3K | Wortmannin | Huh7, Mahlavu, HepG2 | In vitro | Induce apoptosis, suppress cell growth | 246 | |
| PI3K | Fenofbrate | Hep3B, Huh7, Hep3B xenograft model | In vitro and in vivo | Inhibit cell proliferation and migration, induce apoptosis, inhibit tumor growth | 656 | |
| AKT | AKT inhibitor VIII | Huh7, Mahlavu | In Vitro | Suppress cell growth, induce apoptosis | 246 | |
| AKT | Orlistat(FASN) or specific penetrating peptides | Akt knock-in mouse | In vivo | Suppress tumor growth | 657 | |
| mTOR | RAD001 (everolimus) | Patient-derived HCCs | In vivo | Growth inhibition | 658 | |
| mTOR | Metformin+sorafenib | HepG2, Bel-7402 xenograft model | In vitro and in vivo | Growth inhibition | 659 | |
| mTOR | miRNA-199a-3p | Huh7 xenograft model | In vivo | Growth inhibition and survival prolong | 660 | |
| mTOR | miRNA-99a | SMMC-LTNM | In vivo | Decreased tumor size | 661 | |
| PI3K/mTOR | BEZ235 | Hep3B, Huh7, PLC/RLF5 | In vitro | Growth-inhibitory, induce apoptosis | 247 | |
| AKT/mTOR | Lenvatinib | Hep3B, HepG2 | In vitro | Inhibit cell proliferation and migration | 662 | |
| AKT/mTOR | ZJQ-24 | HepG2 xenograft model | In vivo | Inhibits angiogenesis, induce apoptosis and growth inhibition | 663 | |
| PI3K/AKT | Artemisia rupestris L.(ARL) | HepG2 | In vitro | Restrict HepG2 cell proliferation, apoptosis, migration and invasion | 664 | |
| PI3K/AKT | YangzhengXiaoji capsue (YXC) | MHCC97H, HCCLM3, HepG2, Huh7, liver xenograft tumor model of MHCC 97H and HCCLM3 | In vitro and in vivo | Inhibit HCC tumor growth | 665 | |
| mTOR+VEGF | Rapamycin+bevacizumab | HepG2 xenograft model | In vivo | Growth inhibition and survival prolong | 666 | |
| PI3K/AKT/mTOR | Suaeda vermiculata Forssk (SVE) | DEN-treated rats | In vivo | Suppress the development of HCC | 667 | |
| PI3K/AKT/mTOR | Isoliquiritigenin (ISL) | SMMC-7721, MHCC97H, SMMC-7721 xenograft model | In vitro and in vivo | Induce apoptosis and autophagy and tumor growth | 655 | |
| Wnt/β-catenin pathway | PORCN | LGK-974(WNT-974), ETC-159 | HepG2 cell lines | In vitro | Enhances radiosensitivity | 668 |
| TNKS | XAV939 or WXL-8 | HepG2 and HepG2 xenograft model | In vitro and in vivo | Growth inhibition | 334 | |
| β-catenin/TCF | Fungal derivatives (PKF 115-854, CGP0449090) | HCC cell lines(HepG2, Hep40, Huh7), HepG2 xenograft model Hep3B, MHCC97, and SK-Hep-1 | In vitro and in vivo | Growth inhibition | 332,333 | |
| ß-catenin | RNAi (siRNA) | HuH-6 and HepG2 cell lines | In vitro | Proliferation inhibition | 336 | |
| ß-catenin | RNAi (siRNA) | HepG2 cell lines | In vitro | Apoptosis and angiogenesis gene expression | 335 | |
| ß-catenin | LNA-si-β-catenin | GOF Ctnnb1-mutant mouse HCC model induced by diethylnitrosa mine (DEN) and phenobarbita (PB) | In vivo | Decreased cell proliferation and increased cell death | 337 | |
| ß-catenin | siCTNNB1-LNP | Met/mutant-β-catenin HTV mice model | In vivo | Reduced tumor cell proliferation and viability | 307 | |
| BCL9 and β-catenin, PD-1/PD-L1 | sBBI&PDP nanoparticles | Orthotopic homograft mice model of HCC and a PDX HCC model | In vivo | Antitumor efficacy with a favorable biosafety profile | 669 | |
| JAK-STAT pathway | JAK2 | Dehydrocrenatidine (DHCT) | HepG2 xenograft | In vivo | Induced invasion and CSC phenotypes | 670 |
| JAK1/2 | Ruxolitinib(INC424) | Huh7, SNU182, SNU423 | In vitro | Inhibit cell proliferation, promote apoptosis | 272 | |
| JAK1,2,3 | CIMO | Huh7 orthotopic xenograft | In vivo | Reduced tumor growth | 671 | |
| STAT3 | Leonurine | HepG2, Hep3B, and HCCLM3 cells | In vitro | Induced apoptosis and also significantly potentiated the cytotoxic effect of paclitaxe l, doxorubicin, and sorafenib. | 672 | |
| STAT3 | WP1066 | Bel7402 cell lines | In vitro | Inhibit cell migration and invasion through EMT | 274,673 | |
| STAT3 | Stattic | HepG2, Bel-7402, SMMC-7721 cell lines | In vitro | Enhance radiosensitivity, reduce radio induced migration and invasion | 674 | |
| STAT3 | Cryptotanshinone (CTS) | MHCC97H,L02 and L02 xenograft model | In vitro and in vivo | Inhibit cell migration, invasion, carcinogenesis; | 675 | |
| Hepa1-6 model | Inhibit tumor growth and activation of macrophages and polarized mouse bone marrow-derived macrophages (BMM) toward anM1 phenotype; synergy with anti-PDL1 | 676 | ||||
| STAT3 | Napabucasin | Hepa1-6 xenograft | In vivo | Reduced tumor growth | 677 | |
| JAK/STAT | AZ960, TG101209 | Akt/β-catenin hepatic tumor mouse model and tumorspheres | In vivo and in vitro | Abrogate cell proliferation, n regulating the self-renewing | 678 | |
| JAK1, STAT3 | CoL-EtOH | HepG2 cell lines | In vitro | Impeded the viability | 679 | |
| JAK1, STAT3 | Nitidine chloride | HepG2 xenograft | In vivo | Reduced tumor volume | 680 | |
| JAK2/STAT3 | Etomidate | HepG2 xenograft | In vivo | Inhibition of tumor growth Increase in animal survival | 681 | |
| EphB4 receptor(JAK2, STAT3) | Cantharidin | SMMC-7721 | In vivo | Reduced tumor volume | 682 | |
| IL-6 (STAT3) | miR-515-5p | HepG2 xenograft | In vivo | Inhibit migration and invasion | 683 | |
| miR-221-3p(STAT3) | C1QTNF1-AS1 | HepG2 and Huh7 xenograft | In vivo | Reduced tumor volume | 261 | |
| Hedgehog pathway | Smo | Vismodegib (GDC-0449) | HBxTg mice, Orthotropic Transplantation mice model of hepatoma Mistheton Lectin-1 cell | In vivo | Suppress hepatic tumor development | 684,685 |
| Smo | Cyclopamine | HCC cell lines (Hep3B, Huh7, PLC/PRF5), Orthotropic Transplantation mice model of hepatoma Mistheton Lectin-1 cells | In vitro and in vivo | Decreased tumor size | 367 | |
| Gli | GANT61 | Human HCC cell lines and xenograft model of Huh 7, cd44+Patient-derived organoids | In vitro and in vivo | Inhibit proliferation, migration, in vivo growth, drug resistance(sorafenib, 5-FU, doxorubicin, and cisplatin resistance) | 368–372 | |
| Gli | RNAi | Huh7 cell lines | In vitro | Reverse sorafenib, 5-FU, doxorubicin, and cisplatin resistance | 369 | |
| Gli | NanoHHI | HCC cell lines(Huh7,MHCC97L), subcutaneous and orthotopic xenograft model of Huh7 | In vivo | Growth inhibition and antimetastatic effects in vivo | 374 | |
| SULF2 | OKN-007 | HCC cell lines (Huh7, Hep3B) and Huh7 xenografts | In vivo and in vitro | Promoted tumor cell apoptosis and inhibited cell proliferation, viability, and migration, inhibit in vivo tumor growth | 377 | |
| Shh, Smo and Gli1 | Taccalonolide A | HCC cell lines | In vitro | Reduce viability | 376 | |
| Unselective (Shh, Gli, Smo, etc) | Itraconazole | DEN-induced HCC rats model | In vivo | Anti-inflammatory, antiangiogenic, antiproliferative, and apoptotic effects | 686 | |
| Hippo pathway | TAZ((WWTR1) | Statins(fluvastatin, simvastatin) | HLF and HuH1 cell lines | In vitro | Inhibit cell proliferation | 400 |
| MST1, YAP | Salvianolic acid B (SalB) | HepG2, DEN/CCl4/C2H5OH-induced HCC model | In vitro and in vivo | Inhibit cell proliferation and migration, tumorigenesis in vivo | 401 | |
| Tankyrase | G007-LK | c-Met/sgAxin1 HCC mouse model | In vivo | Synergizes with cabozantinib leading to tumor regression | 390 | |
| LATS | NIBR-LTSi | Mice with extended hepatectomy | In vivo | Accelerates liver regeneration | 403 | |
| Notch pathway | γ-Secretase | Ginsenoside (Rg3) | HepG2, Huh7 and Huh7 xenograft mouse | In vitro and in vivo | Synergistic antitumor with sorafenib via AKT | 437 |
| ADAM17 | ZLDI-8 | HepG2, Hep3B, Hep3B xenograft mouse | In vitro and in vivo | Synergistic antitumor with sorafenib | 438 | |
| Notch2 | Antagonistic antibody | AKT/Ras HTV mice model | In vivo | Decreased tumor burden | 426 | |
| Epigenetics | Pan-HDAC | Panobinostat | HepG2, Hep3B, HepG2 xenografts | In vitro and in vivo | Delayed growth and prolonged survival | 687 |
| Pan-HDAC | Panobinostat | HCC xenografts | In vivo | Combination with sorafenib decreased tumor volume and increased surviva | 688 | |
| Pan-HDAC | Suberanilohydroxamic A cid (SAHA) | LCL-PI 11 | In vitro | Growth inhibition and apoptotic induction | 689 | |
| Pan-HDAC | HL23 | PLC/PRF/5, cMHCC97L orthotopic HCC mouse model | In vitro and in vivo | Suppressed HCC progression and metastasis | 690 | |
| Pan-HDAC | MPT0G009 | Hep3B cell lines and xenograft models | In vitro and in vivo | Induced apoptosis and growth arrest | 691 | |
| Pan-HDAC | AR-42 | Huh7 cell lines and xenograft models | In vitro and in vivo | Radiosensitize in inhibition of HCC cell growth | 692 | |
| Pan-HDAC | Belinostat | Subcutaneous Hepa129 murine HCC model | In vivo | Enhanced anti-tumor efficacy of checkpoint inhibitors | 464 | |
| Pan-HDAC | Quisinostat | HCCLM3 xenograft model | In vitro and in vivo | Synergetic with sorafenib, facilitated G0 G1 cycle arrest and apoptosis | 693 | |
| HDAC1 | Trichostatin A | Huh7, HLE, HLF, HepG2, HepG2 xenografts | In vitro and in vivo | Inhibit cell proliferation, induce apoptosis Sensitize HCC cells to enhanced NK-cell mediated killing | 694,695 | |
| HDAC-3,6,8 | Droxinostat | SMMC-7721 and HepG2 | In vitro | Induce apoptosis | 696 | |
| HDAC8 | PCI-34051 | Hepa1-6 orthotopic HCC mouse model | In vivo | Elevated intratumoral CD8+ T cell infiltration, reduced tumorigenicity, d elevated intratumoral CD8+ T cell infiltration, reduced tumorigenicity alone or combined wth anti-PDL1 | ii473 | |
| Class I and class II HDAC | Valproic Acid | HepG2, Huh7 | In vitro | TRAIL-induced apoptosis and chemotherapy sentization | 697,698 | |
| Class I, class IIa and IV HDAC | Resminostat | Huh7, HCC-LM3, SK-Hep-1, Huh7 xenografts, HepG2, Hep3B, SMMC-7721, patient-derived primary HCC cells, DEN-induced HCC mouse mode | In vitro and in vivo | Activate apoptosis | 699–701 | |
| BRD | JQ-1 | HepG2, N-Ras/c-Myc HTVi HCC mouse model; Huh7, Hep3B and Hepa129;DEN/CDAHFD diet-induced mouse model of NASH-driven HCC | In vitro and in vivo | Synergetic with anti-PD1 antibody; Reduce tumor aggressiveness | 474–476 | |
| BRD4 | i-BET762 | Hepa1-6 orthotopic HCC mouse model | In vivo | Combined treatment with anti-PD-L1 synergistically enhanced TILs, resulting in tumor eradication and prolonged survival in the fibrotic-HCC mouse model. | 477 | |
| DNMT | Guadecitabine (SGI-110) | SNU398, HepG2 and SNU475 | In vitro | Anti-proliferation effects | 702 | |
| DNMT | 5-Aza-CdR (DAC) | SNU5, HepG126, and SNU2 | In vitro | Synergistic anti-tumor effect with EZH2 inhibitor (GSK1 26) | 472 | |
| G9a and DNMT1 | CM-272 | HepG2, PLC/PRF-LX2 mixed tumors | In vitro and in vivo | Inhibit cell proliferation and tumor growth | 469 | |
| G9a (EHMT2) | UNC0638, BIX01294 | BEL7402, SMMC-7721 | In vitro | Inhibit cell growth | 470 | |
| EZH2 | GSK343 | Hepa1-6 subcutaneous xenograft mode | In vivo | Tumor suppression | 468 | |
| P53 signaling | wildtype p53 | p53 mRNA delivery by nanoparticles | Hep3B cell lines and xenograft models, p53-null orthotopic and ectopic models of murine HCC | In vitro and in vivo | Tumor suppression, synergetic with anti-PD1 antibody | 495,496 |
| wildtype p53 | Adiponectin | HepG2 | In vitro | Suppress tumor through apoptosis | 497 | |
| mutp53-Y220C | SLMP53-2 | Huh7 cell lines and xenograft models | In vitro and in vivo | Growth inhibition, synergistic effect with sorafenib | 498 | |
| p53-bax | Oleanolic acid | HepG2 cell lines and wildtype mice | In vitro and in vivo | Enhanced apoptosis and reduced hepatotoxicity, synergistic effect with cisplatin | 499 | |
| p53-bax | Acetylshikonin | SMMC-7721 cell lines and H22 mouse model | In vitro and in vivo | Growth inhibition, enhanced apoptosis | 500 | |
| Bcl-2 | ABT-199 | Huh7, Hep3B, HepG2 cell lines | In vitro | Induce cell death in combination with MIK665 | 505 | |
| Bcl-2 | Obatoclax | HepG2 and Hepa1-6 cell lines, Hepa1-6 and Hepa1c1c7 xenograft models | In vitro and in vivo | Tumor suppression, synergetic with anti-PD1 antibody | 504 | |
| Fas/FasL | Scutellarein | Hep3B cell lines | In vitro | Growth inhibition, induce apoptosis and cell cycle arrest | 506 | |
| Caspase 8 | Garciniaxanthone I | HepG2 cell lines | In vitro | Induce apoptosis and inhibit migration | 507 | |
| Cyt-C | Compound 19b | Huh7, Hep3B, HepG2 cell lines | In vitro | Induce apoptosis | 508 | |
| Cyt-C | Lycorine | HepG2 cell lines and xenograft models | In vitro and in vivo | Induced apoptosis | 509 | |
| CDK4/6 | Palbociclib(PD-0332991) | PLC/PRF/5, HepG2, Hep3B | In vitro | Growth inhibition | 513 | |
| CDK4/6 | Abemaciclib, ribociclib | HUH7, SNU398, HepG2 | In vitro | Simultaneous combination of abemaciclib with lenvatinib reduced 3D cell growth, and impaired colony formation and cell migration | 514 |
Immune-related signaling pathways
Immune-related signaling pathways refer to the intricate regulatory networks that govern the dynamic interplay between the immune system and malignant cells. These pathways can influence the immune response against HCC, thereby affecting tumor growth, progression, and the effectiveness of treatment.
Cellular signaling pathways
Mutations or activation of pathways such as CTNNB1/WNT-β-catenin, TGF-β, MYC, TP53, ARID1A, and CDK20 exert profound effects on immune responses and immune cell recruitment.4,515 For example, CTNNB1 mutations diminish CCL5 expression, impairing dendritic cell (DC) recruitment, and reducing NKG2D ligand expression, thus hindering natural killer (NK) cell-mediated responses.516,517 About a quarter of HCC harbor β-catenin mutations, correlating with reduced lymphocyte infiltration and potential resistance to PD-1 blockade, although clinical validation is needed. TP53 mutations occur in ~40% of HCCs overall but are enriched in non-inflamed HCCs, and loss of p53 function promotes the recruitment of immunosuppressive cell.11,518,519 while ARID1A mutations have a dual effect on antitumor immunity by affecting mismatch repair and IFNγ signaling.520 Genetic alterations in PTEN, RAS, and LBK1 result in lymphocyte depletion and exclusion phenotypes. MYC overexpression upregulates PD-L1, and CDK20 activation recruits myeloid-derived suppressor cells (MDSCs),521 suppressing T cell activity. Chromosomal gains at 6p21, containing VEGFA, lead to overexpression of immunosuppressive cytokines.522,523 STAT3 activation leads to the production of cytokines like TGF-β, interleukin-17 (IL-17), and VEGF.524 Additionally, STAT3 activation can inhibit the immune response orchestrated by T helper type 1 cells (Th1) and further contribute to ICI resistance.525 These effects collectively promote an immunosuppressive tumor microenvironment.
ICIs
ICIs are regulators of the immune system. These pathways are indispensable for self-tolerance, which prevents the immune system from attacking cells indiscriminately. Inhibitory checkpoint molecules are targets for cancer immunotherapy due to their potential for use in multiple types of cancers.
Currently approved checkpoint inhibitors encompasses the blockade of CTLA-4, PD-1 and PD-L1. while additional immune checkpoints in HCC, such as TIM-3 (T cell immunoglobulin domain and mucin domain-3), LAG-3 (Lymphocyte activation gene protein-3) and TIGIT (T cell immunoglobulin and ITIM domains), will also be briefly discussed hereafter.
PD-1/PD-L1
The immune checkpoint molecule, belonging to the CD28 family, dampens T cell activity during the immune response and prevents autoimmune injury by binding to its ligands PD-L1 or PD-L2. This interaction inhibits the stimulation signal of the T cell receptor (TCR).526 PD-1 is expressed on various immune cells, including activated T cells, B cells, NK cells, and dendritic cells (DCs).527–529 The expression of PD-L1 can be observed in both tumor cells and antigen-presenting cells (APCs), whereas PD-L2 is predominantly expressed on DCs and macrophages.530,531 The expression levels and impacts of PD-L1 and PD-L2 on immune responses are highly context-dependent and can vary significantly across different tissues and disease states.532 The expression of PD-L1 on TAMs can attenuate the anti-cancer immune response through its interaction with PD-1 on CD8+ and CD4+ T cells.533 The full comprehensive of the significance behind PD-L1/PD-1 expression in immune cells remains elusive.
PD-1 attenuate positive signals from TCR and CD28, affecting downstream pathways like PI3K-AKT, RAS, and ERK.534 Additionally, PD-1 inhibits T cell function by upregulating transcription factors such as BATF (Basic Leucine Zipper ATF-Like Transcription Factor), and modulating metabolic pathways to reduce glycolysis while promoting lipid degradation and β-oxidation.535 This leads to decreased cytokine Secretion, thereby aiding cancer cells in evading immune responses.536 PD-1/PD-L1 expression in Tregs exacerbates immune suppression and exhaustion in the tumor microenvironment, influencing Treg differentiation, maintenance, and function.536
It’s important to note that while PD-1/PD-L1 axis inhibition primarily affects T cells, the mechanism may differ in B cells. In B cells, PD-1 activation recruits SHP-2 to dephosphorylate BCR pathway molecules, inhibiting PI3K, ERK, and PLCγ2 pathways, disrupting calcium signaling and inhibiting B cell growth.537 PD-1 overexpression in B cells induces T cell dysfunction through an IL-10-dependent pathway, promoting tumor progression. PD-1 + B cells inhibit T cell expansion and viability, with PD-L1 blockade enhancing T cell proliferation and viability.538 HDAC6-depleted T cells stimulated PD-1/PD-L1 expression and synergistically sensitized advanced HCC to ICIs, suggesting potential for HCC immunotherapy.
The specific microenvironment created by tumor-releasing factors, LPS and hypoxic conditions can induce the expression of PD-L1 in MDSCs.539,540 The immunosuppressive effects of MDSCs on T cells activated by anti-CD3 and anti-CD28 are mediated through the binding of PD-1 on T cells and PD-L1 on MDSCs, leading to T cell inhibition. Additionally, MDSCs can activate the PI3K/AKT/NF-kB pathway in B cells via the PD-1/PD-L1 axis, inducing a subset of immunosuppressive regulatory B cells (Bregs) characterized by the absence of PD-1 and the presence of PD-L1.541
Overall, the PD-1/PD-L1 axis is indeed a key player in shaping the immunosuppressive microenvironment of HCC.542 The removal of immunosuppression by PD-1/PD-L1 inhibitors is believed to enhance the killing effect mediated by antitumor T cells, while also promoting T cell proliferation and infiltration into the tumor microenvironment (TME) for inducing an antitumor response.543 Additionally, it can enhance CD8+ T cell activation in tumor-draining lymph nodes (TDLN) and rejuvenating dysfunctional CD8+ T cells within the tumor.543 The anti-PD-L1 antibody, by activating the mTOR pathway in T cells, enhances the immune response not only through T cells but also indirectly affects macrophages. Activated T cells can promote macrophage activation, leading to their transformation into inflammatory and proliferative types that can contribute to a more effective anti-tumor response, which has potential as an alternative cancer treatment.544,545
Inhibitors of PD-1/PD-L1
In the advanced liver cancer field, there are currently FDA-approved PD-1/PD-L1 inhibitors, including nivolumab (PD-1 inhibitor), pembrolizumab (PD-1 inhibitor), camrelizumab (PD-1 inhibitor), tislelizumab (PD-1 inhibitor), durvalumab (PD-L1 inhibitor), atezolizumab (PD-L1 inhibitor) and durvalumab (PD-L1 inhibitor). Nivolumab is the first anti-PD-1 drug used for HCC, showing promising efficacy and safety in the CheckMate 040 trial, a global phase III clinical study. It demonstrates good outcomes among patients with advanced liver cancer, regardless of whether they received prior sorafenib treatment, without triggering viral outbreaks in patients with HCV or HBV hepatitis.546 Compared to sorafenib, nivolumab fail to enhance OS but demonstrated remarkable clinical efficacy, exceptional safety profile, and a notably higher rate of complete response, thereby improving patients’ quality of life.547 However, for individuals unable to use TKI or antiangiogenesis drugs, especially those at significant risk, nivolumab remains a viable treatment option. Ongoing trials, such as monotherapy in checkmate-9dx (NCT03383458) and combinations with ipilimumab (NCT01658878), continue to assess nivolumab’s potential as adjuvant therapy and in combination treatments for HCC.
In cohort 1 of the Keynote-224 clinical trial, another PD-1 inhibitor, Pembrolizumab, initially demonstrated efficacy and tolerability in patients with advanced HCC who had previously received sorafenib treatment, leading to FDA approval.548 Keynote-394 represents a milestone as the first and only phase III trial globally to achieve positive results using PD-1 inhibitor monotherapy for advanced HCC, providing a new treatment option for HCC.
Camrelizumab (SHR-1210), another human (immunoglobulin) IgG4 monoclonal antibody targeting PD-1, was evaluated in a multicenter, open, randomized phase 2 trial in China.549 Among 217 patients treated with camrelizumab, the objective response rate (ORR) was 14.7%, with a median progression-free survival (PFS) of 2.1 months and a median OS (mOS) of 13.8 months. The OS rate at 6 months was 74.4% (3%).
The monoclonal antibody Atezolizumab, targeting PD-L1, has demonstrated remarkable efficacy in the phase III imbrave150 study. When combined with bevacizumab, it exhibited a staggering 56% reduction in the risk of mortality (OS) and an impressive 40% decrease in the risk of disease progression or death (PFS), surpassing sorafenib’s performance. Furthermore, this combination therapy of Atezolizumab and bevacizumab was found to be exceptionally well-tolerated with manageable toxicity.30
Importantly, Durvalumab, a humanized IgG1 monoclonal antibody targeting PD-L1, has been recommanded as the first-line treatment for HCC in the 2022 edition of NCCN guidelines. This recommendation is based on its preliminary efficacy observed in patients with non-resectable HCC550 Specifically, the Himalaya Phase III study demonstrated that both durvalumab monotherapy and its combination with tremelimumab (D + T) significantly improved OS compared to sorafenib. This evidence supports the inclusion of durvalumab in treatment regimens for HCC, highlighting its potential to offer significant clinical benefits.551
CTLA-4
In the tumor microenvironment, CTLA-4 is predominantly expressed in Treg cells, with some effector T cells also expressing CTLA-4. Thus, CTLA-4 not only directly induces T cell function downregulation but also contributes to Treg-mediated suppression of anti-tumor immune responses.552 Treg cells have complex functions including direct suppression of DC cell function, inhibition of effector T cells, secretion of TGF-β, and competition for IL-2 and other cytokines through CD25. Tregs are central inhibitory cells in the tumor microenvironment and exhibit high CTLA-4 expression.553 Therefore, by blocking the checkpoint CTL4-4, CTLA-4 inhibitors managed to repair the collapsed immune surveillance system.
The CTLA-4 antibody, Ipilimumab, binds to CTLA-4 on effector T cells, thereby alleviating inhibition and exerting its ADCC function to eliminate Tregs. Tremelimumab was the first CTLA-4 inhibitor used for HCC treatment.554
Tremelimumab is a fully human IgG2 monoclonal antibody that blocks the interaction of CTLA-4 with its ligands CD80 and CD86. The concurrent utilization of PD-1 and CTLA-4 may potentially yield synergistic alleviation of inhibition and complementary elimination
The initial concerns regarding the potential for ICIs to trigger viral outbreaks in HCC patients with HCV or HBV were addressed in the first pivotal trials, which unequivocally demonstrated the safety of ICIs in these populations. In a groundbreaking move, researchers evaluated the antiviral activity of a CTLA-4 blocker in HCV-infected patients and observed no cases of fulminant hepatitis, establishing the favorable safety profile of ICIs.555 Similarly, in the Checkmate-040 trial’s phase 1/2 dose escalation and expansion cohort, no patients with HBV infection experienced hepatitis attacks. This marked the first inclusion of chronic HBV patients in clinical trials of immune checkpoint blockers, setting the stage for subsequent efficacy assessments of immunotherapy in HCC.556
TIM-3/LAG-3/TIGIT
TIM-3, also known as hepatitis A virus cellular receptor 2 (HAVCR2), is an immune inhibitory surface molecule expressed on various immune cells, including T cells, Treg cells, DCs, NK cells, macrophages, and cancer cells. Activation of TIM-3 leads to immune exhaustion of CD8+ T cells and can induce macrophage polarization towards the M2 type, thereby promoting tumor growth through increased secretion of interleukin-6 (IL-6).
LAG-3 is another immune checkpoint that inhibits T cell function and is expressed on tumor-infiltrating lymphocytes (CD4+ and CD8+ T cells), Treg cells, NK/T cells, B cells, NK cells, plasmacytoid DC (pDCs), and TAMs. The expression of TIM-3 and LAG-3 are associated with a poor prognosis in human cancers. TIGIT is expressed on activated NK and T cells, as well as on Treg and Th cells under resting conditions.
Several TIM-3 and LAG-3 agents are currently in clinical development, TIM3 monoclonal antibody TSR-022 (NCT03680508) and with a phase II study combined with anti-PD-1 antibody in HCC patients and mainly include INCAGN02385 (NCT03538028, monotherapy), Relatlimab (NCT04567615) and LAG-3 monoclonal antibody XmAb22841 (NCT03849469 combined with PD-1. At ESMO 2022, LAG-3 monotherapy INCAGN02385reported good tolerability for further investigation in phase Ib/II studies (NCT04370704, NCT05287113) in combination with other immunotherapies. TIGIT antibody Tiragolumab with atezolizumab and bevacizumab showed 2.83-fold higher objective response rate compared to the control therapy.
Gut microbiota
The human gastrointestinal tract harbors over 100 trillion microorganisms including bacteria, fungi, viruses and archaea that make up the gut microbiota. The gut microbiota carries out critical functions for its host.557 As the first organ to encounter enterally absorbed nutrients and microbial metabolites, the liver and the gut microbiome interacted on bile acids conversion and metabolism.558
The intricate relationship between gut microbiota and HCC is increasingly recognized, with various studies highlighting its role in both HCC development and treatment response. Microbiota profiles have been linked to clinical-pathological characteristics of HCC patients, such as AFP, ALT, and AST levels, and have shown predictive value for HBV-related HCC microvascular invasion.559,560 Additionally, the presence of specific gut microbes, such as B. longum, has been associated with improved liver function recovery in HCC patients during the perioperative period.561
Moreover, aberrations in gut microbial-derived metabolite signaling pathways, including toll-like receptor (TLR) and farnesoid X receptor (FXR) signaling, have been implicated in hepatocarcinogenesis.562–564 Gut microbiota also serve a role in regulating hepatic NKT cells and anti-tumor immunity, affecting both primary and metastatic liver tumors.565–567 Gut microbiota profiles holds promise as non-invasive biomarkers to predict treatment response and guiding therapeutic interventions in HCC patients.568
The modulation of gut microbiota is feasible through diet, probiotics, prebiotics, and antibiotics may affect the therapeutic effect of ICIs.569 Preclinically, depletion of the microbiota by gut sterilization or antibiotics administration protected against HCC development upon HCC animal models.517,562,566,570,571 Lactobacillus brevis and B. pseudolongum are promising probiotic for metabolism-related HCC prevention.572,573 Except above potential treatment, administration of atorvastatin, a cholesterol-lowering drug, and gut microbiota manipulation may also be effective strategies for NAFLD– HCC prevention.574 Furthermore, gut microbiota composition or their metabolites has been linked to treatment response and outcomes in HCC patients undergoing immunotherapy, such as with nivolumab and pembrolizumab.575,576 Antibiotic treatment can modulate the gut microbiota, affecting bile acid metabolism, hepatic inflammation, and anti-tumor immunity, ultimately impacting the development and progression of HCC.577–579 Preclinical studies suggest that antibiotic therapy may reduce secondary bile acids associated with hepatic inflammation and metabolic HCC development, which can enhance anti-tumor immunity.580
In the context of immunotherapy, conflicting results have been observed with antibiotic administration at the begining of ICI treatment, with some studies reporting decreased efficacy of ICIs while others show longer progression-free survival (NCT02021253). Probiotic BIFICO during the preoperative phase of HCC patients was capable of accelerating postoperative liver function recovery (NCT05178524). Above all, the clinical therapeutic effectiveness of probiotics in HCC patients is undefined and must considering the issue of dose- and time-finding. New strategies fecal microbiota transplantation (FMT) is becoming a novel, direct and more effective approach to restore gut homeostasis and potentially improve ICI efficacy.
The immune microenvironment and HCC etiology
HCC often arises in the setting of chronic liver inflammation, primarily triggered by innate immune activation. HCC caused by different factors may exhibit distinct immune dysfunctions. For instance, chronic viral infections can lead to pro-inflammatory innate immune responses and aberrant adaptive immune reactions, while non-viral HCCs may involve specific subsets of cells and immune cells. Studies suggest a distinctive pro-tumorigenic adaptive immune response in non-viral HCC, characterized by CXCR6+CD8+ T cells with low FOXO1 expression triggering auto-aggression in response to metabolic stimuli in NASH.581,582 Therefore, understanding the unique contributions of different HCC causes in shaping the tumor microenvironment is crucial for identifying potential mechanisms that could be targeted for effective immunotherapy strategies (Fig. 5).
Fig. 5.
The landscape of immune microenvironment and drug strategies in HCC. a The distinct viral and non-viral etiological mechanisms of hepatocellular carcinoma (HCC) are associated with an unique immune suppressive niche. b Immune effectors targeting HCC cells. c Clinical strategies for HCC. Figure was created with biorender.com
Virus-HCC
Immune responses to HBV and HCV can either promote or inhibit carcinogenesis.583–586 Platelets and HBV-specific T cells play dual roles in driving or inhibiting HCC development, respectively. Chronic HBV infection commonly induces a tolerogenic microenvironment in the liver characterized by upregulation PDL-1 and IL-10 in Breg, increasing susceptibility of HBV-specific T cells to apoptosis induced by TIGIT+NKG2D+NK cells, limiting effective anti-tumor immunity.587 Highly suppressive PD-1hi Treg cells selectively enrich in HBV-related HCC and correlate with poor prognosis.588 In contrast, HCV evades immune surveillance by inducing dysfunctional CD8+ T cells and upregulating immune checkpoint proteins. These cells exhibit reduced production of IFNγ and expression of CD127, impaired proliferation, and increased expression of PD-1316. HCV-infected cells can promote the upregulation of TIM3 expression in CD8+ T cells by releasing exosomes, stimulating monocytes to secrete galectin 9.589 Additionally, chronic HCV infection may deplete IL-2-producing CD4+ T cells and increase suppressive CD4+CD25+ Treg cells and virus-specific, IL-10-producing CD8+ T cells, which may contribute to hepatocarcinogenesis.590,591 Viral escape mutations and HCV core proteins acting as immune evasion proteins, interfering with MHC I-dependent antigen presentation, may also play roles in HCV-related HCC immune evasion.
Metabolic dysregulation also contributes to virus-related HCC pathogenesis. Disruptions in cholesterol homeostasis, driven by high expression of sterol O-acyltransferase 1 (SOAT1), promote tumor cell proliferation and migration in HBV-related HCC.592 Targeting these metabolic pathways presents a novel avenue for immunotherapeutic intervention.
In summary, chronic HBV and HCV infections drive hepatocarcinogenesis through complex interactions involving immune dysregulation and metabolic alterations. Understanding these mechanisms is crucial for developing effective preventive and therapeutic strategies for virus-related HCC.
Aflatoxin-HCC
Aflatoxin, recognized as one of the most potent naturally occurring human hepatocarcinogens, was classified as a “group 1” human carcinogen.593
Aflatoxin contamination is frequently observed in improperly stored food such as maize, peanuts, and tree nuts, significantly augmenting the incidence of HCC.594 The identification and elimination of these risk factors have substantially reduced its occurrence among young adults, underscoring the significance of aflatoxin control as a preventive measure.
The mutational landscape of Aflatoxin-HCC is characterized by frequent mutations in driver genes, including previously implicated genes such as TP53, RAS, c-fos and ADGRB1.595–598 AFB1 causes human HCC by forming a 249Ser mutation in p53. In the early stage of liver cancer, AFB1 induced mutations of the ras oncogene in liver tissues, which mainly occurred in the GG position of codons 12 and 13, most of which were the transversion of G:C to T:A. Aflatoxin can cause the overexpression of c-fos in tree shrew liver tissue, and promote the occurrence and evolution of liver cancer. Among them, ADGRB1 exhibits the highest frequency of mutation in the aflatoxin-associated cohort. Aflatoxin-associated HCCs display a distinctively high mutation rate primarily consisting of C/A mutants, which often leads to a significant increase in the generation of mutation-associated neoantigens (MANAs).595 Importantly, this elevated mutational burden is closely correlated with sensitivity to anti-PD-1/PD-L1 therapy.599 ICIs hold great potential as an effective therapeutic option for AF-HCCs originating from high-risk regions and the general population.
HBV can increase the risk of HCC in people exposed to AFB1 by 30 times. Exposure to high concentrations can cause acute hepatitis, therefore, chronic exposure can lead to the development of liver cancer. the single most effective way to reduce HCC risk in regions where AFB1 and HBV co-occur is to vaccinate against HBV in order to eliminate the synergistic effect on risk.600
NASH/NAFLD HCC
Non-alcoholic fatty liver disease (NAFLD) is globally prevalent and can progress to NASH, significantly increasing the risk of HCC.601 Unlike viral hepatitis, which features organized inflammatory foci, inflammation induced by NAFLD/NASH typically involves scattered inflammatory infiltrates.582 Preclinical evidence suggests that NAFLD/NASH-related HCC is characterized by reduced CD4+ T cell activity within tumors, loss of tumor surveillance function by CD8+PD-1+T cells, and pro-tumorigenic functions of NKT cells and Th17 cells. Responses to ICIs are weaker compared to viral HCC. Additionally, CD4+IL-17A+TH17 cells contribute to NAFLD/NASH-induced HCC by promoting neutrophil recruitment and accumulation of fatty acids.602 IgA+ plasma cells with high expression of PD-L1 and IL-10 accumulate in NASH, inhibiting CTL activation and promoting HCC. These findings suggest that PD-L1+B cells, possibly a subset of Bregs, exert potent immunosuppressive effects on T cell responses.
Moreover, obesity, commonly associated with metabolic disorders, may reduce the effectiveness of anti-VEGF therapy, although clinical evidence remains complex. Antiplatelet therapy has shown promise in reducing the development of NASH and NASH-related HCC, mediated through platelet-specific glycoprotein Ib-α (GPIbα) and its interactions with platelets and inflammatory monocytes. This is supported by a nationwide cohort study showing long-term use of aspirin is associated with reduced HCC risk.603
In summary, understanding the complex interactions between immune cells and hepatocytes in the development of NASH-related HCC is crucial for devising effective prevention and treatment strategies. Targeting platelet-mediated inflammation, immune dysregulation, and metabolic pathways holds promise in combating the occurrence of NASH-induced liver cancer.
Alcohol HCC
Alcohol intake contributes to up to 30% of the global burden of HCC. Alcohol increases gut permeability, facilitating the entry of immunomodulatory microbiota-derived molecules like LPS into the liver,604 where they have the potential to suppress hepatic immune responses, potentially affecting resident macrophages.605 Alcoholic steatohepatitis (ASH) is marked by the intrahepatic accumulation of pro-tumorigenic, immunosuppressive granulocytic MDSCs and inhibition of T cell recruitment to the liver and neutrophils in the liver parenchyma.606 Cytokines like IL-1 and IL-17 are implicated in the pathogenesis of ASH-related HCC.607,608 Thus, The presence of ASH is likely to induce profound alterations in the repertoire and states of immune cells, as well as modifications in the hepatic cytokine milieu, potentially impeding effective adaptive immune responses against HCC.
The management update of HCC
Diagnosis
HCC often shows characteristic imaging features, but approximately 10% of tumors, especially those 1-2 cm in size, may lack these hallmarks, posing diagnostic challenges. Diagnosis typically involves examining resected tissue or biopsy samples. Recently, liquid biopsy has gained traction for HCC diagnosis. Components like ct-ncRNA, cfDNA, ctDNA, CTCs, and extracellular vesicles (EVs) are released into body fluids, enabling fluid biopsy.609–611 Analytical methods that isolate cells, proteins, nucleic acids, and vesicles from fluids, could aid early diagnosis and monitor HCC progression.609 Despite its potential, challenges remain in component separation and sequencing target selection.
Molecular diagnosis is the embodiment of accurate diagnosis in precision medicine. Diagnosis of HCC is further supported by immunohistochemistry for markers glypican-3 (GPC3: NCT05003895, NCT05103631), epithelial cell adhesion molecule (NCT05028933, NCT03013712), MET (NCT01755767), mucin 1 (NCT02587689), MHC1 (NCT05195294), and TERT (NCT05595473), are being incorporated into these clinical trials. The presence of two or more of these markers increases the diagnostic specificity to 100%. The morphology of HCC has been associated with specific molecular alterations. Radiomics can serve as a clinician decision-making tool for constructing HCC diagnosis models, potentially reducing radiologist error rates.612 However, clinical application faces challenges like imaging consistency, radiomics standardization, and predominantly retrospective research. Artificial intelligence (AI), particularly deep learning, shows promise in extracting diagnostic, prognostic, and predictive insights from radiological data, achieving over 90% accuracy in classifying lesions with typical features.613 Despite potential as clinical decision support, large-scale validation is needed for differential diagnosis of hypervascular liver lesions. Advances in understanding HCC’s molecular mechanisms and technology will likely optimize diagnostic methods and introduce new detection techniques, enhancing early diagnosis and prognosis.
Therapeutic advances
Advancements in HCC therapy face challenges from complex tumor environments and resistance mechanisms. Innovations like hepatic arterial infusion chemotherapy (HAIC) combined with anti-PD-1 immunotherapy and TKI show promising transformative potential.614 The regimens included triple combination therapy (t-CT: lenvatinib, TACE, plus toripalimab) before surgery should be recommended for HCC patients with macrovascular invasion.615 Targeted protein degradation (TPD) with techniques such as intramolecular biovalent glues like CC-122 (Avadomide) in clinical trials with nivolumab or sorafenib suggests new treatment avenues.616
Antibody-drug conjugates (ADCs) targeting GPC3 and CD24 demonstrate anticancer activity, through mechanisms like antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC).617,618 CAR-T cell therapy targeting GPC3 shows efficacy, with ongoing enhancements using IL-7 and CCL19 to boost intratumoral activity.619 Additionally, CAR-T cells targeting CD147 and CD133 (NCT02541370) are also beneficial targets for the treatment HCC patients.620,621 The adoptive transfer of HBV-TCR-T cells into patients with advanced HBV-related HCC was generally well-tolerated and demonstrated a favorable safety profile. Observations of clinical efficacy provide support for the ongoing development and eventual implementation of this treatment strategy in individuals with advanced HBV-associated HCC (NCT03899415).622 DC vaccines loaded with neoantigens improve disease-free survival in post-surgery HCC.623 Innovative personalized cancer vaccines (e.g., GNOS-PV02) encoding neoantigens and immune-modulating agents demonstrate strong antitumor activity in advanced HCC, highlighting potent T cell responses (NCT04251117).624
In conclusion, these emerging therapies — HAIC combined with dual immunotherapy, TPD approaches like intramolecular biovalent glues, ADC development targeting specific markers, and advanced CAR-T cell therapies — are pivotal in expanding HCC treatment options. Continued research and clinical trials are critical to validate and optimize these approaches for improved patient outcomes.
Prognosis
During the last decade, there has been an obvious improvement in the five-year OS for HCC, from 18% to 22%.1,625 This mainly attributed to early detection of HCC, as well as the recent development of systemic drugs such as Lenvatinib and T+A (Tecentriq+Avastin). However, the prognosis of HCC with different etiology varies quite a lot. For exmaple, diagnosis for HCC caused by obesity or non-viral are not currently sensitive enough. Although new detection methods develop, The clinical evidence is limited.
Research indicates that HBV has the most significant impact on OS, followed by HCV, metabolic disorders, and alcoholic liver disease.626 A SEER study in 2020 reported median survival after HCC diagnosis as 10.3 months for HBV, 8.3 months for HCV, 7.6 months for metabolic disorders, HBV, 8.3 months for HCV, 7.6 months for metabolic disorders, and 6.1 months for alcohol-related causes. Alcohol and metabolic-related HCCs exhibit higher mortality rates potentially due to delayed diagnosis rather than inherent aggressiveness.627 Among liver-related deaths in the United States between 2008 and 2018, there was a substantial decline observed in HCV-related deaths. The prevalence of NAFLD-associated HCC increased from 2.6% in the period of 1995-1999 to 19. 5% during 2010–2014, while the proportion attributed to HCV decreased from 43.6% to 19.5%.628 This highlights the effectiveness of HBV vaccination and HCV antiviral therapy in preventing virus-associated liver cancer; however, no strategy currently exists for chemoprevention of non-viral liver diseases.
The prognosis of HCC is gender-dependent, age-dependent and regional-dependent. Across nearly every country, HCC incidence and mortality differ by sex, with rates 1.2–3.6 times higher in men than in women.629,630 The mortality rates are higher among elderly patients with HCC. HCC patients in various countries has different outcomes. Treatment choices also influence prognosis of HCC. Lenvatinib has been shown to prolong survival among non-viral HCCs and MAFLD.631 While microwave ablation may offer superior long-term recurrence rates when compared to radiofrequency ablation.632
Challenges and directions
There are still several challenges in the field of HCC. Firstly, many of the signaling pathways and targeted inhibitors that have been summarized above exhibit reduced efficacy specifically in HCC compared to other types of cancers, possibly due to the unique characteristics of HCC. Secondly, the absence of clearly defined biomarkers for HCC hinders accurate treatment strategies for patients including targeted therapy or immune therapy,633–637 especialliy in ICI. Traditional markers like PD-L1 expression have failed to predict response to nivolumab and pembrolizumab.547,548,638,639 Other potential biomarkers, such as high tumor mutational burden (TMB) or microsatellite instability (MSI), are limited in HCC. Tumor heterogeneity and clonal evolution are the underlying mechanisms.640,641 Emerging evidence suggests that activated Wnt/ β-catenin signaling is associated with primary resistance to immunotherapy, particularly in ‘cold' tumors lacking T-cell infiltration. Identifying predictive biomarkers are critical due to optimize treatment outcomes in advanced HCC.642–645 Furthermore, future clinical applications for treating HCC will likely involve combinations of two or more drugs to overcome the resistance. In additon to the agents, ICI also combined with TACE, radiotherapy and ablation. These have been tested in the clinical phase III. While determining the optimal combination and sequence of FDA-approved or investigational ICIs, as well as other treatment modalities are difficult. Specifically, the classical approach to first surgically remove resectable lesions and/or regional lymph nodes, followed by postoperative (adjuvant) therapy, is gradually giving room to treatment schedules in which neoadjuvant (chemo) immunotherapy is administered before surgery. However, there is a lack of clarity regarding the optimal combination methods, administration sequence, and timing; hence exploring new adjuvant treatment options over the next few years becomes crucial to enhance patient response rates. Lastly, With the rapid development of cellular immune technology and genetic engineering technology, application of adoptive cell therapy, CART, oncolytic virus and vaccine in HCC will benefit more HCC patients. Recent advances related to cancer genomics, proteomics, systems biology and AI suggest new perspectives in precision medicine.
Conclusion
This review provides valuable insights into the treatment landscape of HCC and its potential application across different cancer types and pathological subtypes. Novel treatment regimens have shown promise in improving survival rates and reducing severe toxicities in HCC patients. Ongoing studies hold the potential to refine therapeutic strategies and identify predictive markers for favorable outcomes. While small molecular inhibitors remain in high demand due to their moderate adverse effects, further efforts are needed to enhance treatment response. Comprehensive research efforts continue to expand our understanding of HCC mechanisms, offering potential avenues for prevention and treatment. The clinical translation of new inhibitors remains critical, and combination therapies involving novel agents and traditional regimens hold significant promise in advancing HCC treatment outcomes.
Acknowledgements
The research of the authors was funded by grants from the National Natural Science Foundation of China (82222047, 82073039), the Science and Technology Commission of Shanghai Municipality (22XD1423100, 22S11900500), and Shanghai Municipal Health Commission (2022XD057). The authors apologize to those researchers whose work was not included in this Review due to space constraints.
Author contributions
Jiaojiao Zheng, Siying Wang, Lei Xia and Zhen Sun collected information and wrote manuscript; Kui Ming Chan, René Bernards and Wenxin Qin put forward some suggestions for revision; the whole work was thoroughly supervised by Jinhong Chen, Qiang Xia and Haojie Jin. All authors have read and approved the article.
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Jiaojiao Zheng, Siying Wang, Lei Xia, Zhen Sun
Change history
4/14/2025
The original online version of this article was revised: The author name in Reference 499 has been updated for accuracy.
Contributor Information
Jinhong Chen, Email: jinhongch@hotmail.com.
Qiang Xia, Email: xiaqiang@shsmu.edu.cn.
Haojie Jin, Email: hjjin1986@shsci.org.
References
- 1.Siegel, R. L. et al. Cancer statistics, 2024. CA Cancer J Clin.74, 12–49 (2024). [DOI] [PubMed] [Google Scholar]
- 2.Llovet, J. M. et al. Hepatocellular carcinoma. Nat. Rev. Dis. Prim. 7, 6 (2021). [DOI] [PubMed]
- 3.Anstee, Q. M., Reeves, H. L., Kotsiliti, E., Govaere, O. & Heikenwalder, M. From NASH to HCC: current concepts and future challenges. Nat. Rev. Gastroenterol. Hepatol.16, 411–428 (2019). [DOI] [PubMed] [Google Scholar]
- 4.Xu, Y. et al. Translation control of the immune checkpoint in cancer and its therapeutic targeting. Nat. Med.25, 301–311 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Goswami, V. G. & Patel, B. D. Recent updates on Wnt signaling modulators: a patent review (2014-2020). Expert Opin. Ther. Pat.31, 1009–1043 (2021). [DOI] [PubMed] [Google Scholar]
- 6.Tornesello, M. L. et al. Telomerase: a good target in hepatocellular carcinoma? An overview of relevant preclinical data. Expert Opin. Ther. Targets26, 767–780 (2022). [DOI] [PubMed] [Google Scholar]
- 7.Nevola, R. et al. Predictors of early and late hepatocellular carcinoma recurrence. World J. Gastroenterol.29, 1243–1260 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Raoul, J. L. et al. Updated use of TACE for hepatocellular carcinoma treatment: How and when to use it based on clinical evidence. Cancer Treat. Rev.72, 28–36 (2019). [DOI] [PubMed] [Google Scholar]
- 9.Sperandio, R. C., Pestana, R. C., Miyamura, B. V. & Kaseb, A. O. Hepatocellular Carcinoma Immunotherapy. Annu. Rev. Med.73, 267–278 (2022). [DOI] [PubMed] [Google Scholar]
- 10.Qin, S. et al. Atezolizumab plus bevacizumab versus active surveillance in patients with resected or ablated high-risk hepatocellular carcinoma (IMbrave050): a randomised, open-label, multicentre, phase 3 trial. Lancet402, 1835–1847 (2023). [DOI] [PubMed] [Google Scholar]
- 11.Llovet, J. M. et al. Immunotherapies for hepatocellular carcinoma. Nat. Rev. Clin. Oncol.19, 151–172 (2022). [DOI] [PubMed] [Google Scholar]
- 12.Zhang, N. & Li, Y. Receptor tyrosine kinases: biological functions and anticancer targeted therapy. MedComm.4, e446 (2023). [DOI] [PMC free article] [PubMed]
- 13.Ebrahimi, N. et al. Receptor tyrosine kinase inhibitors in cancer. Cell. Mol. Life Sci. 80, 104 (2023). [DOI] [PMC free article] [PubMed]
- 14.Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell100, 57–70 (2000). [DOI] [PubMed] [Google Scholar]
- 15.Weis, S. M. & Cheresh, D. A. Tumor angiogenesis: molecular pathways and therapeutic targets. Nat. Med. 17, 1359–1370 (2011). [DOI] [PubMed]
- 16.Liu, Z. L., Chen, H. H., Zheng, L. L., Sun, L. P. & Shi, L. Angiogenic signaling pathways and anti-angiogenic therapy for cancer. Signal Transduct. Target. Ther. 8, 198 (2023). [DOI] [PMC free article] [PubMed]
- 17.Morse, M. A. et al. The role of angiogenesis in hepatocellular carcinoma. Clin. Cancer Res.25, 912–920 (2019). [DOI] [PubMed] [Google Scholar]
- 18.Kong, F. H. et al. Current status of sorafenib nanoparticle delivery systems in the treatment of hepatocellular carcinoma. Theranostics11, 5464–5490 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Melincovici, C. S. et al. Vascular endothelial growth factor (VEGF) – key factor in normal and pathological angiogenesis. Rom. J. Morphol. Embryol.59, 455–467 (2018). [PubMed] [Google Scholar]
- 20.Ally, A. et al. Comprehensive and Integrative Genomic Characterization of Hepatocellular Carcinoma. Cell169, 1327–1341.e23 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wang, Y. & Deng, B. Hepatocellular carcinoma: molecular mechanism, targeted therapy, and biomarkers. Cancer Metastasis Rev.42, 629–652 (2023). [DOI] [PubMed] [Google Scholar]
- 22.Amaoka, N. et al. Expression of vascular endothelial growth factor receptors is closely related to the histological grade of hepatocellular carcinoma. Oncol. Rep.16, 3–10 (2006). [PubMed] [Google Scholar]
- 23.Gershtein, E. S., Dubova, E. A., Shchegolev, A. I. & Kushkinskii, N. E. Vascular endothelial growth factor and its type 2 receptor in hepatocellular carcinoma. Bull. Exp. Biol. Med.149, 749–752 (2010). [DOI] [PubMed] [Google Scholar]
- 24.Chu, J. S. et al. Expression and prognostic value of VEGFR-2, PDGFR-β, and c-Met in advanced hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 32, 16 (2013). [DOI] [PMC free article] [PubMed]
- 25.Huang, J. et al. Prognostic significance and potential therapeutic target of VEGFR2 in hepatocellular carcinoma. J. Clin. Pathol.64, 343–348 (2011). [DOI] [PubMed] [Google Scholar]
- 26.Sharma, A. et al. Onco-fetal Reprogramming of Endothelial Cells Drives Immunosuppressive Macrophages in Hepatocellular Carcinoma. Cell183, 377–394.e21 (2020). [DOI] [PubMed] [Google Scholar]
- 27.Xu, Z. H. et al. Anti-hepatocellular carcinoma activity of Sorbaria sorbifolia by regulating VEGFR and c-Met/apoptotic pathway. J. Ethnopharmacol. 324, 117758 (2024). [DOI] [PubMed]
- 28.Torimura, T. et al. Antiangiogenic and Antitumor Activities of Aflibercept, a Soluble VEGF Receptor-1 and -2, in a Mouse Model of Hepatocellular Carcinoma. Neoplasia18, 413–424 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Garcia, J. et al. Bevacizumab (Avastin®) in cancer treatment: A review of 15 years of clinical experience and future outlook. Cancer Treat. Rev. 86, 102017 (2020). [DOI] [PubMed]
- 30.Finn, R. S. et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med.382, 1894–1905 (2020). [DOI] [PubMed] [Google Scholar]
- 31.Roy, A. Updated Efficacy and Safety Data from IMbrave150: Atezolizumab Plus Bevacizumab vs. Sorafenib for Unresectable Hepatocellular Carcinoma. J. Clin. Exp. Hepatol.12, 1575–1576 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu, X., Lu, Y. & Qin, S. Atezolizumab and bevacizumab for hepatocellular carcinoma: Mechanism, pharmacokinetics and future treatment strategies. Futur. Oncol.17, 2243–2256 (2021). [DOI] [PubMed] [Google Scholar]
- 33.Qin, S. et al. Camrelizumab plus rivoceranib versus sorafenib as first-line therapy for unresectable hepatocellular carcinoma (CARES-310): a randomised, open-label, international phase 3 study. Lancet402, 1133–1146 (2023). [DOI] [PubMed] [Google Scholar]
- 34.Zhu, A. X. et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased α-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol.20, 282–296 (2019). [DOI] [PubMed] [Google Scholar]
- 35.Kudo, M. et al. Brivanib as adjuvant therapy to transarterial chemoembolization in patients with hepatocellular carcinoma: A randomized phase III trial. Hepatology60, 1697–1707 (2014). [DOI] [PubMed] [Google Scholar]
- 36.Johnson, P. J. et al. Brivanib versus sorafenib as first-line therapy in patients with unresectable, advanced hepatocellular carcinoma: Results from the randomized phase III BRISK-FL study. J. Clin. Oncol.31, 3517–3524 (2013). [DOI] [PubMed] [Google Scholar]
- 37.Llovet, J. M. et al. Brivanib in patients with advanced hepatocellular carcinoma who were intolerant to sorafenib or for whom sorafenib failed: Results from the randomized phase III BRISK-PS study. J. Clin. Oncol.31, 3509–3516 (2013). [DOI] [PubMed] [Google Scholar]
- 38.Kang, Y. K. et al. Randomized phase II study of axitinib versus placebo plus best supportive care in second-line treatment of advanced hepatocellular carcinoma. Ann. Oncol.26, 2457–2463 (2015). [DOI] [PubMed] [Google Scholar]
- 39.Zhu, A. X. et al. Efficacy, safety, pharmacokinetics, and biomarkers of cediranib monotherapy in advanced hepatocellular carcinoma: A phase II study. Clin. Cancer Res.19, 1557–1566 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Carver, R. S., Stevenson, M. C., Scheving, L. A. & Russell, W. E. Diverse expression of ErbB receptor proteins during rat liver development and regeneration. Gastroenterology123, 2017–2027 (2002). [DOI] [PubMed] [Google Scholar]
- 41.Yuankai Shi et al. A Prospective, Molecular Epidemiology Study of EGFRMutations in Asian Patients with Advanced Non–Small-CellLung Cancer of Adenocarcinoma Histology (PIONEER). J. Thorac. Oncol.9, 154–162 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Collisson, E. A. et al. Comprehensive molecular profiling of lung adenocarcinoma: The cancer genome atlas research network. Nature511, 543–550 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Panvichian, R., Tantiwetrueangdet, A., Sornmayura, P. & Leelaudomlipi, S. Missense mutations in exons 18-24 of EGFR in hepatocellular carcinoma tissues. Biomed. Res. Int. 2015, 171845 (2015). [DOI] [PMC free article] [PubMed]
- 44.Lee, S. C. et al. Lack of somatic mutations in EGFR tyrosine kinase domain in hepatocellular and nasopharyngeal carcinoma. Pharmacogenet. Genomics16, 73–74 (2006). [DOI] [PubMed] [Google Scholar]
- 45.Sueangoen, N., Tantiwetrueangdet, A. & Panvichian, R. HCC-derived EGFR mutants are functioning, EGF-dependent, and erlotinib-resistant. Cell Biosci. 10, 41 (2020). [DOI] [PMC free article] [PubMed]
- 46.Bejarano, P. A. Epidermal Growth Factor Receptor Expression and Gene Copy Number in Conventional Hepatocellular Carcinoma. Yearb. Pathol. Lab. Med.2010, 42 (2010). [DOI] [PubMed] [Google Scholar]
- 47.Su, M. C., Lien, H. C. & Jeng, Y. M. Absence of epidermal growth factor receptor exon 18-21 mutation in hepatocellular carcinoma. Cancer Lett224, 117–121 (2005). [DOI] [PubMed] [Google Scholar]
- 48.Villanueva, A., Toffanin, S. & Llovet, J. M. Linking molecular classification of hepatocellular carcinoma and personalized medicine: Preliminary steps. Curr. Opin. Oncol.20, 444–453 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ito, Y. et al. Expression and clinical significance of erb-B receptor family in hepatocellular carcinoma. Br. J. Cancer84, 1377–1383 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Song, S. et al. EGFR/MET promotes hepatocellular carcinoma metastasis by stabilizing tumor cells and resisting to RTKs inhibitors in circulating tumor microemboli. Cell Death Dis. 13, 351 (2022). [DOI] [PMC free article] [PubMed]
- 51.Dong, Z. R. et al. TMPRSS4 Drives Angiogenesis in Hepatocellular Carcinoma by Promoting HB-EGF Expression and Proteolytic Cleavage. Hepatology72, 923–939 (2020). [DOI] [PubMed] [Google Scholar]
- 52.Liu, Z., Chen, D., Ning, F., Du, J. & Wang, H. EGF is highly expressed in hepatocellular carcinoma (HCC) and promotes motility of HCC cells via fibronectin. J. Cell. Biochem.119, 4170–4183 (2018). [DOI] [PubMed] [Google Scholar]
- 53.Jin, H. et al. EGFR activation limits the response of liver cancer to lenvatinib. Nature595, 730–734 (2021). [DOI] [PubMed] [Google Scholar]
- 54.He, X. et al. EGFR inhibition reverses resistance to lenvatinib in hepatocellular carcinoma cells. Sci. Rep. 12, 8007 (2022). [DOI] [PMC free article] [PubMed]
- 55.Hu, B. et al. Inhibition of EGFR Overcomes Acquired Lenvatinib Resistance Driven by STAT3–ABCB1 Signaling in Hepatocellular Carcinoma. Cancer Res.82, 3845–3857 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wang, L. et al. METTL3-m6A-EGFR-axis drives lenvatinib resistance in hepatocellular carcinoma. Cancer Lett. 559, 216122 (2023). [DOI] [PubMed]
- 57.Huang, M. et al. METTL1-Mediated m7G tRNA Modification Promotes Lenvatinib Resistance in Hepatocellular Carcinoma. Cancer Res.83, 89–102 (2023). [DOI] [PubMed] [Google Scholar]
- 58.Lim, M. et al. EGFR/ERBB2 Amplifications and Alterations Associated With Resistance to Lenvatinib in Hepatocellular Carcinoma. Gastroenterology164, 1006–1008.e3 (2023). [DOI] [PubMed] [Google Scholar]
- 59.Gu, L., Jin, X., Liang, H., Yang, C. & Zhang, Y. Upregulation of CSNK1A1 induced by ITGB5 confers to hepatocellular carcinoma resistance to sorafenib in vivo by disrupting the EPS15/EGFR complex. Pharmacol. Res. 192, 106789 (2023). [DOI] [PubMed]
- 60.Ji, L. et al. miR-486-3p mediates hepatocellular carcinoma sorafenib resistance by targeting FGFR4 and EGFR. Cell Death Dis. 11, 2550 (2020). [DOI] [PMC free article] [PubMed]
- 61.Ezzoukhry, Z. et al. EGFR activation is a potential determinant of primary resistance of hepatocellular carcinoma cells to sorafenib. Int. J. Cancer131, 2961–2969 (2012). [DOI] [PubMed] [Google Scholar]
- 62.Niu, L. et al. New insights into sorafenib resistance in hepatocellular carcinoma: Responsible mechanisms and promising strategies. Biochim. Biophys. Acta Rev. Cancer1868, 564–570 (2017). [DOI] [PubMed] [Google Scholar]
- 63.Zhu, A. X. et al. Search: A phase III, randomized, double-blind, placebo-controlled trial of sorafenib plus erlotinib in patients with advanced hepatocellular carcinoma. J. Clin. Oncol.33, 559–566 (2015). [DOI] [PubMed] [Google Scholar]
- 64.Lin, Y. et al. HGF/R-spondin1 rescues liver dysfunction through the induction of Lgr5+ liver stem cells. Nat. Commun. 8, 1175 (2017). [DOI] [PMC free article] [PubMed]
- 65.Fu, J. et al. HGF/c-MET pathway in cancer: from molecular characterization to clinical evidence. Oncogene40, 4625–4651 (2021). [DOI] [PubMed] [Google Scholar]
- 66.Wang, H. et al. The Function of the HGF/c-Met Axis in Hepatocellular Carcinoma. Front. Cell Dev. Biol. 8, 55 (2020). [DOI] [PMC free article] [PubMed]
- 67.García-Vilas, J. A. & Medina, M. Á. Updates on the hepatocyte growth factor/c-Met axis in hepatocellular carcinoma and its therapeutic implications. World J. Gastroenterol.24, 3695–3706 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Corso, S. & Giordano, S. Cell-autonomous and non-cell-autonomous mechanisms of HGF/MET-driven resistance to targeted therapies: From basic research to a clinical perspective. Cancer Discov.3, 978–992 (2013). [DOI] [PubMed] [Google Scholar]
- 69.Nakayama, N. et al. Hepatocyte growth factor and c-met expression in Long-Evans Cinnamon rats with spontaneous hepatitis and hepatoma. Hepatology24, 596–602 (1996). [DOI] [PubMed] [Google Scholar]
- 70.Schulze, K. et al. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat. Genet.47, 505–511 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lee, S. J. et al. A survey of c-MET expression and amplification in 287 patients with hepatocellular carcinoma. Anticancer Res.33, 5179–5186 (2013). [PubMed] [Google Scholar]
- 72.Ang, C. et al. Comprehensive multiplatform biomarker analysis of 350 hepatocellular carcinomas identifies potential novel therapeutic options. J. Surg. Oncol.113, 55–61 (2016). [DOI] [PubMed] [Google Scholar]
- 73.Yu, J., Chen, G. G. & Lai, P. B. S. Targeting hepatocyte growth factor/c-mesenchymal–epithelial transition factor axis in hepatocellular carcinoma: Rationale and therapeutic strategies. Med. Res. Rev.41, 507–524 (2021). [DOI] [PubMed] [Google Scholar]
- 74.Ma, Y., Liu, X. & Tang, X. ETS-1/c-Met drives resistance to sorafenib in hepatocellular carcinoma. Am. J. Transl. Res.15, 896–913 (2023). [PMC free article] [PubMed] [Google Scholar]
- 75.Chen, S. & Xia, X. Long noncoding RNA NEAT1 suppresses sorafenib sensitivity of hepatocellular carcinoma cells via regulating miR-335–c-Met. J. Cell. Physiol.234, 14999–15009 (2019). [DOI] [PubMed] [Google Scholar]
- 76.Fu, L. et al. Synergistic antitumor activity of low-dose c-met tyrosine kinase inhibitor and sorafenib on human non-small cell lung cancer cells. Oncol. Lett.15, 5081–5086 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Fu, R., Jiang, S., Li, J., Chen, H. & Zhang, X. Activation of the HGF/c-MET axis promotes lenvatinib resistance in hepatocellular carcinoma cells with high c-MET expression. Med. Oncol. 37, 24 (2020). [DOI] [PubMed]
- 78.Ueki, T., Fujimoto, J., Suzuki, T., Yamamoto, H. & Okamoto, E. Expression of hepatocyte growth factor and its receptor c-met proto-oncogene in hepatocellular carcinoma. Hepatology25, 862–866 (1997). [DOI] [PubMed] [Google Scholar]
- 79.Ke, A. W. et al. Role of overexpression of CD151 and/or c-Met in predicting prognosis of hepatocellular carcinoma. Hepatology49, 491–503 (2009). [DOI] [PubMed] [Google Scholar]
- 80.Wu, F. S. et al. Study on the prognostic value of hepatocyte growth factor and c-met for patients with hepatocellular carcinoma. Zhonghua Wai Ke Za Zhi44, 603–608 (2006). [PubMed] [Google Scholar]
- 81.Rimassa, L. et al. Tumor and circulating biomarkers in patients with second-line hepatocellular carcinoma from the randomized phase II study with tivantinib. Oncotarget7, 72622–72633 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yau, T. et al. Cabozantinib plus atezolizumab versus sorafenib for advanced hepatocellular carcinoma (COSMIC-312): final results of a randomised phase 3 study. Lancet Gastroenterol. Hepatol.9, 310–322 (2024). [DOI] [PubMed] [Google Scholar]
- 83.Zhan, N. et al. The effect of selective c-MET inhibitor on hepatocellular carcinoma in the MET-Active, β-Catenin-Mutated mouse model. Gene Expr.18, 135–147 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Rimassa, L. et al. Tivantinib for second-line treatment of MET-high, advanced hepatocellular carcinoma (METIV-HCC): a final analysis of a phase 3, randomised, placebo-controlled study. Lancet Oncol.19, 682–693 (2018). [DOI] [PubMed] [Google Scholar]
- 85.Santoro, A. et al. Tivantinib for second-line treatment of advanced hepatocellular carcinoma: A randomised, placebo-controlled phase 2 study. Lancet Oncol.14, 55–63 (2013). [DOI] [PubMed] [Google Scholar]
- 86.Zhao, S. et al. Selective Inhibitor of the c-Met Receptor Tyrosine Kinase in Advanced Hepatocellular Carcinoma: No Beneficial Effect With the Use of Tivantinib? Front. Immunol. 12, 731527 (2021). [DOI] [PMC free article] [PubMed]
- 87.Puzanov, I. et al. Phase 1 trial of tivantinib in combination with sorafenib in adult patients with advanced solid tumors. Invest. New Drugs33, 159–168 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Qin, S. et al. A phase II study of the efficacy and safety of the MET inhibitor capmatinib (INC280) in patients with advanced hepatocellular carcinoma. Ther. Adv. Med. Oncol. 11, 1758835919889001 (2019). [DOI] [PMC free article] [PubMed]
- 89.Decaens, T. et al. Phase 1b/2 trial of tepotinib in sorafenibpretreated advanced hepatocellular carcinoma with MET overexpression. Br. J. Cancer125, 190–199 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ryoo, B. Y. et al. Randomised Phase 1b/2 trial of tepotinib vs sorafenib in Asian patients with advanced hepatocellular carcinoma with MET overexpression. Br. J. Cancer125, 200–208 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kalinina, J. et al. The alternatively spliced acid box region plays a key role in FGF receptor autoinhibition. Structure20, 77–88 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Olsen, S. K. et al. Fibroblast Growth Factor (FGF) Homologous Factors Share Structural but Not Functional Homology with FGFs. J. Biol. Chem.278, 34226–34236 (2003). [DOI] [PubMed] [Google Scholar]
- 93.Touat, M., Ileana, E., Postel-Vinay, S., André, F. & Soria, J. C. Targeting FGFR signaling in cancer. Clin. Cancer Res.21, 2684–2694 (2015). [DOI] [PubMed] [Google Scholar]
- 94.Gao, L. et al. FGF19/FGFR4 signaling contributes to the resistance of hepatocellular carcinoma to sorafenib. J. Exp. Clin. Cancer Res. 36, 8 (2017). [DOI] [PMC free article] [PubMed]
- 95.Paur, J. et al. Fibroblast growth factor receptor 3 isoforms: Novel therapeutic targets for hepatocellular carcinoma? Hepatology62, 1767–1778 (2015). [DOI] [PubMed] [Google Scholar]
- 96.Chen, Z. et al. The Role of Fibroblast Growth Factor 19 in Hepatocellular Carcinoma. Am. J. Pathol.191, 1180–1192 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wang, H. et al. Advances of Fibroblast Growth Factor/Receptor Signaling Pathway in Hepatocellular Carcinoma and its Pharmacotherapeutic Targets. Front. Pharmacol. 12, 650388 (2021). [DOI] [PMC free article] [PubMed]
- 98.Sawey, E. T. et al. Identification of a Therapeutic Strategy Targeting Amplified FGF19 in Liver Cancer by Oncogenomic Screening. Cancer Cell19, 347–358 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wu, X. et al. FGF19-induced hepatocyte proliferation is mediated through FGFR4 activation. J. Biol. Chem.285, 5165–5170 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Nicholes, K. et al. A mouse model of hepatocellular carcinoma: Ectopic expression of fibroblast growth factor 19 in skeletal muscle of transgenic mice. Am. J. Pathol.160, 2295–2307 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Desnoyers, L. R. et al. Targeting FGF19 inhibits tumor growth in colon cancer xenograft and FGF19 transgenic hepatocellular carcinoma models. Oncogene27, 85–97 (2008). [DOI] [PubMed] [Google Scholar]
- 102.French, D. M. et al. Targeting FGFR4 inhibits hepatocellular carcinoma in preclinical mouse models. PLoS One7, e36713 (2012). [DOI] [PMC free article] [PubMed]
- 103.Cui, G. et al. Up-regulation of FGF15/19 signaling promotes hepatocellular carcinoma in the background of fatty liver. J. Exp. Clin. Cancer Res. 37, 136 (2018). [DOI] [PMC free article] [PubMed]
- 104.Li, Y. et al. Up-regulation of fibroblast growth factor 19 and its receptor associates with progression from fatty liver to hepatocellular carcinoma. Oncotarget7, 52329–52339 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shen, B. et al. EGFR Inhibition Overcomes Resistance to FGFR4 Inhibition and Potentiates FGFR4 Inhibitor Therapy in Hepatocellular Carcinoma. Mol. Cancer Ther.22, 1479–1492 (2023). [DOI] [PubMed] [Google Scholar]
- 106.Latasa, M. U. et al. Regulation of Amphiregulin Gene Expression by β-Catenin Signaling in Human Hepatocellular Carcinoma Cells: A Novel Crosstalk between FGF19 and the EGFR System. PLoS One7, e52711 (2012). [DOI] [PMC free article] [PubMed]
- 107.Tovar, V. et al. Tumour initiating cells and IGF/FGF signalling contribute to sorafenib resistance in hepatocellular carcinoma. Gut66, 530–540 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Kanzaki, H. et al. The impact of FGF19/FGFR4 signaling inhibition in antitumor activity of multi-kinase inhibitors in hepatocellular carcinoma. Sci. Rep. 11, 5303 (2021). [DOI] [PMC free article] [PubMed]
- 109.Lu, X., Chen, H., Patterson, A. V., Smaill, J. B. & Ding, K. Fibroblast Growth Factor Receptor 4 (FGFR4) Selective Inhibitors as Hepatocellular Carcinoma Therapy: Advances and Prospects. J. Med. Chem. 62, 2905–2915 (2019). [DOI] [PubMed]
- 110.Abou-Alfa, G. K. et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: a multicentre, open-label, phase 2 study. Lancet Oncol.21, 671–684 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Loriot, Y. et al. Erdafitinib in Locally Advanced or Metastatic Urothelial Carcinoma. N. Engl. J. Med.381, 338–348 (2019). [DOI] [PubMed] [Google Scholar]
- 112.Hagel, M. et al. First selective small molecule inhibitor of FGFR4 for the treatment of hepatocellular carcinomas with an activated FGFR4 signaling pathway. Cancer Discov5, 424–437 (2015). [DOI] [PubMed] [Google Scholar]
- 113.Chen, J. et al. Fibroblast Growth Factor 19–Mediated Up-regulation of SYR-Related High-Mobility Group Box 18 Promotes Hepatocellular Carcinoma Metastasis by Transactivating Fibroblast Growth Factor Receptor 4 and Fms-Related Tyrosine Kinase 4. Hepatology71, 1712–1731 (2020). [DOI] [PubMed] [Google Scholar]
- 114.Joshi, J. J. et al. H3B-6527 Is a potent and selective inhibitor of FGFR4 in FGF19-Driven hepatocellular carcinoma. Cancer Res.77, 6999–7013 (2017). [DOI] [PubMed] [Google Scholar]
- 115.Huynh, H. et al. FGF401 and vinorelbine synergistically mediate antitumor activity and vascular normalization in FGF19-dependent hepatocellular carcinoma. Exp. Mol. Med.52, 1857–1868 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Chen, X. et al. Design, synthesis, and biological evaluation of selective covalent inhibitors of FGFR4. Eur. J. Med. Chem. 268, 116281 (2024). [DOI] [PubMed]
- 117.Kim, R. D. et al. First-in-human phase i study offisogatinib (BLU-554) validates aberrant FGF19 signaling as a driver event in hepatocellular carcinoma. Cancer Discov9, 1696–1707 (2019). [DOI] [PubMed] [Google Scholar]
- 118.Chen, X. et al. Insight into the design of FGFR4 selective inhibitors in cancer therapy: Prospects and challenges. Eur. J. Med. Chem. 263, 115947 (2024). [DOI] [PubMed]
- 119.Werner, H. The IGF1 Signaling Pathway: From Basic Concepts to Therapeutic Opportunities. Int. J. Mol. Sci. 24, 14882 (2023). [DOI] [PMC free article] [PubMed]
- 120.Kraemer, W. J., Ratamess, N. A., Hymer, W. C., Nindl, B. C. & Fragala, M. S. Growth Hormone(s), Testosterone, Insulin-Like Growth Factors, and Cortisol: Roles and Integration for Cellular Development and Growth With Exercise. Front. Endocrinol.11, 33 (2020). [DOI] [PMC free article] [PubMed]
- 121.Taniguchi, C. M., Emanuelli, B. & Kahn, C. R. Critical nodes in signalling pathways: insights into insulin action. Nat. Rev. Mol. Cell Biol.7, 85–96 (2006). [DOI] [PubMed] [Google Scholar]
- 122.Martin-Kleiner, I. & Gall Troselj, K. Mannose-6-phosphate/insulin-like growth factor 2 receptor (M6P/IGF2R) in carcinogenesis. Cancer Lett.289, 11–22 (2010). [DOI] [PubMed] [Google Scholar]
- 123.Aleksic, T. et al. Nuclear IGF1R interacts with regulatory regions of chromatin to promote RNA polymerase II recruitment and gene expression associated with advanced tumor stage. Cancer Res.78, 3497–3509 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Peloso Maia, M. L. et al. Immunohistochemical Expression of Insulin-Like Growth Factor-1 Receptor and Its Association with Clinicopathological Parameters in Hepatocellular Carcinoma. Oncology 1–9, 10.1159/000535332 (2023). [DOI] [PMC free article] [PubMed]
- 125.Chun, Y. S., Huang, M., Rink, L. & Von Mehren, M. Expression levels of insulin-like growth factors and receptors in hepatocellular carcinoma: A retrospective study. World J. Surg. Oncol. 12, 231 (2014). [DOI] [PMC free article] [PubMed]
- 126.Kang, G. H. et al. Prognostic significance of p53, mTOR, c-Met, IGF-1R, and HSP70 overexpression after the resection of hepatocellular carcinoma. Gut Liver8, 79–87 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Sarfstein, R. et al. Insulin-like Growth Factor-I Receptor (IGF-IR) translocates to nucleus and autoregulates IGF-IR gene expression in breast cancer cells. J. Biol. Chem.287, 2766–2776 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Guerard, M. et al. Nuclear translocation of IGF1R by intracellular amphiregulin contributes to the resistance of lung tumour cells to EGFR-TKI. Cancer Lett420, 146–155 (2018). [DOI] [PubMed] [Google Scholar]
- 129.Takeda, T. et al. Upregulation of IGF2R evades lysosomal dysfunction-induced apoptosis of cervical cancer cells via transport of cathepsins. Cell Death Dis. 10, (2019). [DOI] [PMC free article] [PubMed]
- 130.Yamada, T., De Souza, A. T., Finkelstein, S. & Jirtle, R. L. Loss of the gene encoding mannose 6-phosphate/insulin-like growth factor II receptor is an early event in liver carcinogenesis. Proc. Natl. Acad. Sci. USA94, 10351–10355 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Jang, H. S. et al. Clinical significance of loss of heterozygosity for M6P/IGF2R in patients with primary hepatocellular carcinoma. World J. Gastroenterol.14, 1394–1398 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sue, S. R. et al. Transforming growth factor-beta receptors and mannose 6-phosphate/insulin-like growth factor-II receptor expression in human hepatocellular carcinoma. Ann. Surg.222, 171–178 (1995). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Douglas, R. S. et al. Teprotumumab for the Treatment of Active Thyroid Eye Disease. N. Engl. J. Med.382, 341–352 (2020). [DOI] [PubMed] [Google Scholar]
- 134.Wang, P., Mak, V. C. & Cheung, L. W. Drugging IGF-1R in cancer: New insights and emerging opportunities. Genes Dis.10, 199–211 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Beck, H. & Yee, D. Minireview: Were the IGF signaling inhibitors all bad? Mol. Endocrinol.29, 1549–1557 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Reidy-Lagunes, D. L. et al. A phase 2 study of the insulin-like growth factor-1 receptor inhibitor MK-0646 in patients with metastatic, well-differentiated neuroendocrine tumors. Cancer118, 4795–4800 (2012). [DOI] [PubMed] [Google Scholar]
- 137.El-Khoueiry, A. B. et al. A phase I trial of escalating doses of cixutumumab (IMC-A12) and sorafenib in the treatment of advanced hepatocellular carcinoma. Cancer Chemother. Pharmacol.81, 957–963 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Abou-Alfa, G. K. et al. A phase II study of cixutumumab (IMC-A12, NSC742460) in advanced hepatocellular carcinoma. J. Hepatol.60, 319–324 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Haluska, P. et al. Phase I dose escalation study of the anti-IGF-1R monoclonal antibody CP-751,871 in patients with refractory solid tumors. J. Clin. Oncol.25, 3586–3586 (2007). [DOI] [PubMed] [Google Scholar]
- 140.Goncalves, M. D., Hopkins, B. D. & Cantley, L. C. Phosphatidylinositol 3-Kinase, Growth Disorders, and Cancer. N. Engl. J. Med.379, 2052–2062 (2018). [DOI] [PubMed] [Google Scholar]
- 141.Kiely, P. A., O’Gorman, D., Luong, K., Ron, D. & O’Connor, R. Insulin-Like Growth Factor I Controls a Mutually Exclusive Association of RACK1 with Protein Phosphatase 2 A and β 1 Integrin To Promote Cell Migration. Mol. Cell. Biol.26, 4041–4051 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Riedemann, J., Takiguchi, M., Sohail, M. & Macaulay, V. M. The EGF receptor interacts with the type 1 IGF receptor and regulates its stability. Biochem. Biophys. Res. Commun.355, 707–714 (2007). [DOI] [PubMed] [Google Scholar]
- 143.Kazlauskas, A. PDGFs and their receptors. Gene614, 1–7 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Heldin, C. H. & Lennartsson, J. Structural and functional properties of platelet-derived growth factor and stem cell factor receptors. Cold Spring Harb. Perspect. Biol. 5, a009100 (2013). [DOI] [PMC free article] [PubMed]
- 145.Karlsson, L., Lindahl, P., Heath, J. K. & Betsholtz, C. Abnormal gastrointestinal development in PDGF-A and PDGFR-α deficient mice implicates a novel mesenchymal structure with putative instructive properties in villus morphogenesis. Development127, 3457–3466 (2000). [DOI] [PubMed] [Google Scholar]
- 146.Ishii, Y., Hamashima, T., Yamamoto, S. & Sasahara, M. Pathogenetic significance and possibility as a therapeutic target of platelet derived growth factor. Pathol. Int.67, 235–246 (2017). [DOI] [PubMed] [Google Scholar]
- 147.Heldin, C. H. Targeting the PDGF signaling pathway in the treatment of non-malignant diseases. J. Neuroimmune Pharmacol.9, 69–79 (2014). [DOI] [PubMed] [Google Scholar]
- 148.Betsholtz, C. Insight into the physiological functions of PDGF through genetic studies in mice. Cytokine Growth Factor Rev.15, 215–228 (2004). [DOI] [PubMed] [Google Scholar]
- 149.Hoch, R. V. & Soriano, P. Roles of PDGF in animal development. Development130, 4769–4784 (2003). [DOI] [PubMed] [Google Scholar]
- 150.Wang, Y. et al. The platelet-derived growth factors (PDGFs) and their receptors (PDGFRs) are major players in oncogenesis, drug resistance, and attractive oncologic targets in cancer. Growth Factors34, 64–71 (2016). [DOI] [PubMed] [Google Scholar]
- 151.Heldin, C. H., Lennartsson, J. & Westermark, B. Involvement of platelet-derived growth factor ligands and receptors in tumorigenesis. J. Intern. Med.283, 16–44 (2018). [DOI] [PubMed] [Google Scholar]
- 152.Papadopoulos, N. & Lennartsson, J. The PDGF/PDGFR pathway as a drug target. Mol. Aspects Med.62, 75–88 (2018). [DOI] [PubMed] [Google Scholar]
- 153.Stock, P. et al. Platelet-derived growth factor receptor-α: A novel therapeutic target in human hepatocellular cancer. Mol. Cancer Ther.6, 1932–1941 (2007). [DOI] [PubMed] [Google Scholar]
- 154.Wei, T. et al. Overexpression of platelet-derived growth factor receptor alpha promotes tumor progression and indicates poor prognosis in hepatocellular carcinoma. Oncotarget5, 10307–10317 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Zhang, T. et al. Overexpression of platelet-derived growth factor receptor α in endothelial cells of hepatocellular carcinoma associated with high metastatic potential. Clin. Cancer Res.11, 8557–8563 (2005). [DOI] [PubMed] [Google Scholar]
- 156.Qin, S. et al. Donafenib Versus Sorafenib in First-Line Treatment of Unresectable or Metastatic Hepatocellular Carcinoma: A Randomized, Open-Label, Parallel-Controlled Phase II-III Trial. J. Clin. Oncol.39, 3002–3011 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Toh, H. C. et al. Phase 2 trial of linifanib (ABT-869) in patients with unresectable or metastatic hepatocellular carcinoma. Cancer119, 380–387 (2013). [DOI] [PubMed] [Google Scholar]
- 158.Cainap, C. et al. Linifanib versus sorafenib in patients with advanced hepatocellular carcinoma: Results of a randomized phase III trial. J. Clin. Oncol.33, 172–179 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Yen, C. J. et al. A Phase I/Randomized Phase II Study to Evaluate the Safety, Pharmacokinetics, and Efficacy of Nintedanib versus Sorafenib in Asian Patients with Advanced Hepatocellular Carcinoma. Liver Cancer7, 165–178 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Blume-Jensen, P. & Hunter, T. Oncogenic kinase signalling. Nature411, 355–365 (2001). [DOI] [PubMed] [Google Scholar]
- 161.Loizos, N. et al. Targeting the platelet-derived growth factor receptor α with a neutralizing human monoclonal antibody inhibits the growth of tumor xenografts: Implications as a potential therapeutic target. Mol. Cancer Ther.4, 369–379 (2005). [DOI] [PubMed] [Google Scholar]
- 162.Wang, Q. et al. Discovery of 4-((N-(2-(dimethylamino)ethyl)acrylamido)methyl)-N-(4-methyl-3-((4-(pyridin-3-yl)pyrimidin-2-yl)amino)phenyl)benzamide (CHMFL-PDGFR-159) as a highly selective type II PDGFRα kinase inhibitor for PDGFRα driving chronic eosinophilic leukemia. Eur. J. Med. Chem.150, 366–384 (2018). [DOI] [PubMed] [Google Scholar]
- 163.Yin, L. et al. PDGFR-β inhibitor slows tumor growth but increases metastasis in combined radiotherapy and Endostar therapy. Biomed. Pharmacother.99, 615–621 (2018). [DOI] [PubMed] [Google Scholar]
- 164.Roberts, W. G. et al. Antiangiogenic and antitumor activity of a selective PDGFR tyrosine kinase inhibitor, CP-673,451. Cancer Res.65, 957–966 (2005). [PubMed] [Google Scholar]
- 165.Yang, Y. et al. Inhibition of PDGFR by CP-673451 induces apoptosis and increases cisplatin cytotoxicity in NSCLC cells via inhibiting the Nrf2-mediated defense mechanism. Toxicol. Lett.295, 88–98 (2018). [DOI] [PubMed] [Google Scholar]
- 166.Tap, W. D. et al. Effect of Doxorubicin Plus Olaratumab vs Doxorubicin Plus Placebo on Survival in Patients with Advanced Soft Tissue Sarcomas: The ANNOUNCE Randomized Clinical Trial. JAMA J. Am. Med. Assoc.323, 1266–1276 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Gotzmann, J. et al. A crucial function of PDGF in TGF-β-mediated cancer progression of hepatocytes. Oncogene25, 3170–3185 (2006). [DOI] [PubMed] [Google Scholar]
- 168.Pathania, S., Pentikäinen, O. T. & Singh, P. K. A holistic view on c-Kit in cancer: Structure, signaling, pathophysiology and its inhibitors. Biochim. Biophys. Acta-Rev. Cancer1876, 188631 (2021). [DOI] [PubMed]
- 169.Miettinen, M. & Lasota, J. KIT (CD117): A review on expression in normal and neoplastic tissues, and mutations and their clinicopathologic correlation. Appl. Immunohistochem. Mol. Morphol.13, 205–220 (2005). [DOI] [PubMed] [Google Scholar]
- 170.Wang, W., Shui, L., Liu, Y. & Zheng, M. C-Kit, a Double-Edged Sword in Liver Regeneration and Diseases. Front. Genet. 12, 598855 (2021). [DOI] [PMC free article] [PubMed]
- 171.Fujii, T. et al. Participation of liver cancer stem/progenitor cells in tumorigenesis of scirrhous hepatocellular carcinoma-human and cell culture study. Hum. Pathol.39, 1185–1196 (2008). [DOI] [PubMed] [Google Scholar]
- 172.Xu, J. et al. Evaluation of NCAM and c-Kit as hepatic progenitor cell markers for intrahepatic cholangiocarcinomas. Pathol. Res. Pract.214, 2011–2017 (2018). [DOI] [PubMed] [Google Scholar]
- 173.Yan, W., Zhu, Z., Pan, F., Huang, A. & Dai, G. H. Overexpression of c-kit(CD117), relevant with microvessel density, is an independent survival prognostic factor for patients with HBV-related hepatocellular carcinoma. Onco. Targets. Ther.11, 1285–1292 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Chen, L. et al. Precancerous stem cells have the potential for both benign and malignant differentiation. PLoS One2, e293 (2007). [DOI] [PMC free article] [PubMed]
- 175.Nazzal, M. et al. Establishment of a Patient-Derived Xenograft Tumor From Hepatitis C–Associated Liver Cancer and Evaluation of Imatinib Treatment Efficacy. Hepatology72, 379–388 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kwon, Y.-C. et al. Promotion of Cancer Stem-Like Cell Properties in Hepatitis C Virus-Infected Hepatocytes. J. Virol.89, 11549–11556 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Liu, Z. et al. Hepatitis B virus PreS1 facilitates hepatocellular carcinoma development by promoting appearance and self-renewal of liver cancer stem cells. Cancer Lett400, 149–160 (2017). [DOI] [PubMed] [Google Scholar]
- 178.Rojas, A. et al. A Positive TGF-β/c-KIT Feedback Loop Drives Tumor Progression in Advanced Primary Liver Cancer. Neoplasia18, 371–386 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Lin, A. Y., Fisher, G. A., So, S., Tang, C. & Levitt, L. Phase II study of imatinib in unresectable hepatocellular carcinoma. Am. J. Clin. Oncol. Cancer Clin. Trials31, 84–88 (2008). [DOI] [PubMed] [Google Scholar]
- 180.Eckel, F. et al. Pharmacokinetic and clinical phase II trial of imatinib in patients with impaired liver function and advanced hepatocellular carcinoma. Oncology69, 363–371 (2005). [DOI] [PubMed] [Google Scholar]
- 181.Abdellateif, M. S., Bayoumi, A. K. & Mohammed, M. A. c-Kit Receptors as a Therapeutic Target in Cancer: Current Insights. Onco. Targets. Ther.16, 785–799 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Zhang, C. S. et al. Anlotinib Combined With Toripalimab as First-Line Therapy for Unresectable Hepatocellular Carcinoma: A Prospective, Multicenter, Phase II Study. Oncologist28, e1239–e1247 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Sun, Y. et al. Anlotinib in the treatment of advanced hepatocellular carcinoma: an open-label phase II study (ALTER-0802 study). Hepatol. Int.15, 621–629 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Kang, M., Xue, F., Xu, S., Shi, J. & Mo, Y. Effectiveness and safety of anlotinib with or without S-1 in the treatment of patients with advanced hepatocellular carcinoma in a Chinese population: A prospective, phase 2 study. Radiol. Oncol.57, 405–410 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Chen, X. Q. et al. Effectiveness and Safety of Anlotinib with or without PD-1 Blockades in the Treatment of Patients with Advanced Primary Hepatocellular Carcinoma: A Retrospective, Real-World Study in China. Drug Des. Devel. Ther.16, 1483–1493 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Li, Q. et al. A Real-World Study of Optimal Treatment with Anlotinib First-Line Therapy in Advanced Hepatocellular Carcinoma. Cancer Manag. Res.14, 3037–3046 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Reiss, K. A. et al. A pilot study of galunisertib plus stereotactic body radiotherapy in patients with advanced hepatocellular carcinoma. Mol. Cancer Ther.20, 389–397 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Costantini, F. & Shakya, R. GDNF/Ret signaling and the development of the kidney. BioEssays28, 117–127 (2006). [DOI] [PubMed] [Google Scholar]
- 189.Schuchardt, A., D’Agati, V., Larsson-Blomberg, L., Costantini, F. & Pachnis, V. Defects in the kidney and enteric nervous system of mice lacking the tyrosine kinase receptor Ret. Nature367, 380–383 (1994). [DOI] [PubMed] [Google Scholar]
- 190.Subbiah, V. et al. Tumour-agnostic efficacy and safety of selpercatinib in patients with RET fusion-positive solid tumours other than lung or thyroid tumours (LIBRETTO-001): a phase 1/2, open-label, basket trial. Lancet Oncol23, 1261–1273 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Awad, M. M. et al. Acquired Resistance to KRAS G12C Inhibition in Cancer. N. Engl. J. Med.384, 2382–2393 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Wang, C. et al. RET fusions as primary oncogenic drivers and secondary acquired resistance to EGFR tyrosine kinase inhibitors in patients with non-small-cell lung cancer. J. Transl. Med. 20, 390 (2022). [DOI] [PMC free article] [PubMed]
- 193.Shi, M. et al. Identification of RET fusions in a Chinese multicancer retrospective analysis by next-generation sequencing. Cancer Sci.113, 308–318 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Kato, S. et al. RET aberrations in diverse cancers: Next-generation sequencing of 4,871 patients. Clin. Cancer Res.23, 1988–1997 (2017). [DOI] [PubMed] [Google Scholar]
- 195.Sun, Y. et al. A novel four-gene signature for predicting the prognosis of hepatocellular carcinoma. Scand. J. Gastroenterol.57, 1227–1237 (2022). [DOI] [PubMed] [Google Scholar]
- 196.Duke, E. S. et al. FDA Approval Summary: Selpercatinib for the Treatment of Advanced RET Fusion-Positive Solid Tumors. Clin. Cancer Res.29, 3573–3578 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Kim, J. et al. FDA Approval Summary: Pralsetinib for the Treatment of Lung and Thyroid cancers with RET gene mutations or fusions. Clin. Cancer Res. 27, 5452–5456 (2021). [DOI] [PubMed]
- 198.Lee, S., Rauch, J. & Kolch, W. Targeting MAPK signaling in cancer: Mechanisms of drug resistance and sensitivity. Int. J. Mol. Sci. 21, 1102 (2020). [DOI] [PMC free article] [PubMed]
- 199.Ullah, R., Yin, Q., Snell, A. H. & Wan, L. RAF-MEK-ERK pathway in cancer evolution and treatment. Semin. Cancer Biol.85, 123–154 (2022). [DOI] [PubMed] [Google Scholar]
- 200.Forbes, S. A. et al. COSMIC: Mining complete cancer genomes in the catalogue of somatic mutations in cancer. Nucleic Acids Res. 39, D945-50 (2011). [DOI] [PMC free article] [PubMed]
- 201.Fernández-Medarde, A. & Santos, E. Ras in cancer and developmental diseases. Genes Cancer2, 344–358 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Delire, B. & Stärkel, P. The Ras/MAPK pathway and hepatocarcinoma: Pathogenesis and therapeutic implications. Eur. J. Clin. Invest.45, 609–623 (2015). [DOI] [PubMed] [Google Scholar]
- 203.Huynh, H. et al. Over-expression of the mitogen-activated protein kinase (MAPK) kinase (MEK)-MAPK in hepatocellular carcinoma: Its role in tumor progression and apoptosis. BMC Gastroenterol. 3, 19 (2003). [DOI] [PMC free article] [PubMed]
- 204.Moon, H. & Ro, S. W. Mapk/erk signaling pathway in hepatocellular carcinoma. Cancers. 13, 3026 (2021). [DOI] [PMC free article] [PubMed]
- 205.Zhao, X., Yang, C., Wu, J. & Nan, Y. ADAMTS8 targets ERK to suppress cell proliferation, invasion, and metastasis of hepatocellular carcinoma. Onco. Targets. Ther.11, 7569–7578 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Li, D. et al. Prognostic analysis of RAS-related lncRNAs in liver hepatocellular carcinoma. Ann. Transl. Med.10, 1356–1356 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Ou, D. L. et al. Induction of Bim expression contributes to the antitumor synergy between sorafenib and mitogen-activated protein kinase/extracellular signal-regulated kinase kinase inhibitor CI-1040 in hepatocellular carcinoma. Clin. Cancer Res.15, 5820–5828 (2009). [DOI] [PubMed] [Google Scholar]
- 208.Rinehart, J. et al. Multicenter phase II study of the oral MEK inhibitor, CI-1040, in patients with advanced non-small-cell lung, breast, colon, and pancreatic cancer. J. Clin. Oncol.22, 4456–4462 (2004). [DOI] [PubMed] [Google Scholar]
- 209.Bahar, M. E., Kim, H. J. & Kim, D. R. Targeting the RAS/RAF/MAPK pathway for cancer therapy: from mechanism to clinical studies. Signal Transduct. Target. Ther. 8, 455 (2023). [DOI] [PMC free article] [PubMed]
- 210.Charette, N. et al. Salirasib inhibits the growth of hepatocarcinoma cell lines in vitro and tumor growth in vivo through ras and mTOR inhibition. Mol. Cancer9, 256 (2010). [DOI] [PMC free article] [PubMed]
- 211.Da Silva Morais, A., Saliez, A., Leclercq, I., Horsmans, Y. & Stärkel, P. Inhibition of the Ras oncoprotein reduces proliferation of hepatocytes in vitro and in vivo in rats. Clin. Sci.114, 73–83 (2008). [DOI] [PubMed] [Google Scholar]
- 212.Tong, X. et al. MEK inhibition by cobimetinib suppresses hepatocellular carcinoma and angiogenesis in vitro and in vivo. Biochem. Biophys. Res. Commun.523, 147–152 (2020). [DOI] [PubMed] [Google Scholar]
- 213.Hennig, M. et al. Targeting mitogen-activated protein Kinase Kinase with the inhibitor pd0325901 decreases hepatocellular carcinoma growth in vitro and in mouse model systems. Hepatology51, 1218–1225 (2010). [DOI] [PubMed] [Google Scholar]
- 214.Tai, W. M. et al. A phase Ib study of selumetinib (AZD6244, ARRY-142886) in combination with sorafenib in advanced hepatocellular carcinoma (HCC). Ann. Oncol.27, 2210–2215 (2016). [DOI] [PubMed] [Google Scholar]
- 215.Lim, H. Y. et al. Phase II studies with refametinib or refametinib plus sorafenib in patients with RAS-mutated hepatocellular Carcinoma. Clin. Cancer Res.24, 4650–4661 (2018). [DOI] [PubMed] [Google Scholar]
- 216.Lito, P., Rosen, N. & Solit, D. B. Tumor adaptation and resistance to RAF inhibitors. Nat. Med.19, 1401–1409 (2013). [DOI] [PubMed] [Google Scholar]
- 217.Subbiah, V., Baik, C. & Kirkwood, J. M. Clinical Development of BRAF plus MEK Inhibitor Combinations. Trends in Cancer6, 797–810 (2020). [DOI] [PubMed] [Google Scholar]
- 218.Poulikakos, P. I., Zhang, C., Bollag, G., Shokat, K. M. & Rosen, N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature464, 427–430 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Vasan, N. & Cantley, L. C. At a crossroads: how to translate the roles of PI3K in oncogenic and metabolic signalling into improvements in cancer therapy. Nat. Rev. Clin. Oncol.19, 471–485 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Hoxhaj, G. & Manning, B. D. The PI3K–AKT network at the interface of oncogenic signalling and cancer metabolism. Nat. Rev. Cancer20, 74–88 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Fruman, D. A. & Rommel, C. PI3K and cancer: Lessons, challenges and opportunities. Nat. Rev. Drug Discov.13, 140–156 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Golob-Schwarzl, N. et al. New liver cancer biomarkers: PI3K/AKT/mTOR pathway members and eukaryotic translation initiation factors. Eur. J. Cancer83, 56–70 (2017). [DOI] [PubMed] [Google Scholar]
- 223.Wang, Q. et al. Spontaneous Hepatocellular Carcinoma after the Combined Deletion of Akt Isoforms. Cancer Cell29, 523–535 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Xin, Z. et al. Cancer Genomic Alterations Can Be Potential Biomarkers Predicting Microvascular Invasion and Early Recurrence of Hepatocellular Carcinoma. Front. Oncol. 12, 783109 (2022). [DOI] [PMC free article] [PubMed]
- 225.Thorpe, L. M., Yuzugullu, H. & Zhao, J. J. PI3K in cancer: Divergent roles of isoforms, modes of activation and therapeutic targeting. Nat. Rev. Cancer15, 7–24 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Fujiwara, Y. et al. PTEN/MMAC1 mutation and frequent loss of heterozygosity identified in chromosome 10q in a subset of hepatocellular carcinomas. Japanese J. Cancer Res.91, 287–292 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Kawamura, N. et al. PTEN/MMAC1 mutations in hepatocellular carcinomas: Somatic inactivation of both alleles in tumors. Japanese J. Cancer Res.90, 413–418 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Sun, E. J., Wankell, M., Palamuthusingam, P., McFarlane, C. & Hebbard, L. Targeting the pi3k/akt/mtor pathway in hepatocellular carcinoma. Biomedicines9, 1639 (2021). [DOI] [PMC free article] [PubMed]
- 229.Villanueva, A. et al. Pivotal Role of mTOR Signaling in Hepatocellular Carcinoma. Gastroenterology135, 1972–1983 (2008). [DOI] [PMC free article] [PubMed]
- 230.Bang, J., Jun, M., Lee, S., Moon, H. & Ro, S. W. Targeting EGFR/PI3K/AKT/mTOR Signaling in Hepatocellular Carcinoma. Pharmaceutics15, 2130 (2023). [DOI] [PMC free article] [PubMed]
- 231.Liu, F. et al. PIK3C3 regulates the expansion of liver CSCs and PIK3C3 inhibition counteracts liver cancer stem cell activity induced by PI3 Kinhibitor. Cell Death Dis.11, 427 (2020). [DOI] [PMC free article] [PubMed]
- 232.Guri, Y. et al. mTORC2 Promotes Tumorigenesis via Lipid Synthesis. Cancer Cell32, 807–823.e12 (2017). [DOI] [PubMed] [Google Scholar]
- 233.Umemura, A. et al. Liver damage, inflammation, and enhanced tumorigenesis after persistent mTORC1 inhibition. Cell Metab.20, 133–144 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Tian, L. Y., Smit, D. J. & Jücker, M. The Role of PI3K/AKT/mTOR Signaling in Hepatocellular Carcinoma Metabolism. Int. J. Mol. Sci. 24, 2652 (2023). [DOI] [PMC free article] [PubMed]
- 235.Bidkhori, G. et al. Metabolic network-based stratification of hepatocellular carcinoma reveals three distinct tumor subtypes. Proc. Natl. Acad. Sci. USA115, E11874–E11883 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Damodaran, S. et al. Phase II Study of Copanlisib in Patients with Tumors with PIK3CA Mutations: Results from the NCI-MATCH ECOG-ACRIN Trial (EAY131) Subprotocol Z1F. J. Clin. Oncol.40, 1552–1561 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Ahn, D. H. et al. Results of an abbreviated phase-II study with the Akt Inhibitor MK-2206 in Patients with Advanced Biliary Cancer. Sci. Rep. 5, 12122 (2015). [DOI] [PMC free article] [PubMed]
- 238.Lee, K. W. et al. Sirolimus prolongs survival after living donor liver transplantation for hepatocellular carcinoma beyond milan criteria: A prospective, randomised, open-label, multicentre phase 2 trial. J. Clin. Med.9, 1–11 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Cervello, M. et al. Targeted therapy for hepatocellular carcinoma: Novel agents on the horizon. Oncotarget3, 236–260 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Lu, X., Paliogiannis, P., Calvisi, D. F. & Chen, X. Role of the Mammalian Target of Rapamycin Pathway in Liver Cancer: From Molecular Genetics to Targeted Therapies. Hepatology73, 49–61 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Choo, S. P. et al. A Phase 1 dose-finding and pharmacodynamic study of rapamycin in combination with bevacizumab in patients with unresectable hepatocellular carcinoma. Eur. J. Cancer49, 999–1008 (2013). [DOI] [PubMed] [Google Scholar]
- 242.Kelley, R. K. et al. Phase ii trial of the combination of temsirolimus and sorafenib in advanced hepatocellular carcinoma with tumor mutation profiling. Liver Cancer10, 561–571 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Wu, Y. et al. DZW-310, a novel phosphoinositide 3-kinase inhibitor, attenuates the angiogenesis and growth of hepatocellular carcinoma cells via PI3K/AKT/mTOR axis. Biochem. Pharmacol. 201, 115093 (2022). [DOI] [PubMed]
- 244.Gong, C. et al. NCAPG promotes the proliferation of hepatocellular carcinoma through PI3K/AKT signaling. Onco. Targets. Ther.12, 8537–8552 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Ye, L. et al. The PI3K inhibitor copanlisib synergizes with sorafenib to induce cell death in hepatocellular carcinoma. Cell Death Discov. 5, 86 (2019). [DOI] [PMC free article] [PubMed]
- 246.Buontempo, F. et al. Inhibition of Akt signaling in hepatoma cells induces apoptotic cell death independent of Akt activation status. Invest. New Drugs29, 1303–1313 (2011). [DOI] [PubMed] [Google Scholar]
- 247.Ou, D. L. et al. Vertical blockade of the IGFR-PI3K/Akt/mTOR pathway for the treatment of hepatocellular carcinoma: The role of survivin. Mol. Cancer13, 2 (2014). [DOI] [PMC free article] [PubMed]
- 248.Sun, D., Liu, J., Wang, Y. & Dong, J. Co-administration of MDR1 and BCRP or EGFR/PI3K inhibitors overcomes lenvatinib resistance in hepatocellular carcinoma. Front. Oncol. 12, 944537 (2022). [DOI] [PMC free article] [PubMed]
- 249.Li, K. S. et al. NT5DC2 promotes tumor cell proliferation by stabilizing EGFR in hepatocellular carcinoma. Cell Death Dis. 11, 335 (2020). [DOI] [PMC free article] [PubMed]
- 250.Qiu, Z. et al. Celecoxib alleviates AKT/c-Met-triggered rapid hepatocarcinogenesis by suppressing a novel COX-2/AKT/FASN cascade. Mol. Carcinog.58, 31–41 (2019). [DOI] [PubMed] [Google Scholar]
- 251.Samarin, J. et al. PI3K/AKT/mTOR-dependent stabilization of oncogenic far-upstream element binding proteins in hepatocellular carcinoma cells. Hepatology63, 813–826 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Philips, R. L. et al. The JAK-STAT pathway at 30: Much learned, much more to do. Cell185, 3857–3876 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Xue, C. et al. Evolving cognition of the JAK-STAT signaling pathway: autoimmune disorders and cancer. Signal Transduct. Target. Ther. 8, 204 (2023). [DOI] [PMC free article] [PubMed]
- 254.McLornan, D. P., Pope, J. E., Gotlib, J. & Harrison, C. N. Current and future status of JAK inhibitors. Lancet398, 803–816 (2021). [DOI] [PubMed] [Google Scholar]
- 255.He, G. & Karin, M. NF-κB and STAT3-key players in liver inflammation and cancer. Cell Res.21, 159–168 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Hin Tang, J. J., Hao Thng, D. K., Lim, J. J. & Toh, T. B. JAK/STAT signaling in hepatocellular carcinoma. Hepatic Oncol. 7, HEP18 (2020). [DOI] [PMC free article] [PubMed]
- 257.Kan, Z. et al. Whole-genome sequencing identifies recurrent mutations in hepatocellular carcinoma. Genome Res.23, 1422–1433 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Calvisi, D. F. et al. Ubiquitous Activation of Ras and Jak/Stat Pathways in Human HCC. Gastroenterology130, 1117–1128 (2006). [DOI] [PubMed] [Google Scholar]
- 259.Zhao, Z. et al. CircSOD2 induced epigenetic alteration drives hepatocellular carcinoma progression through activating JAK2/STAT3 signaling pathway. J. Exp. Clin. Cancer Res. 39, 2559 (2020). [DOI] [PMC free article] [PubMed]
- 260.Liu, Z. K. et al. EYA2 suppresses the progression of hepatocellular carcinoma via SOCS3-mediated blockade of JAK/STAT signaling. Mol. Cancer20, 79 (2021). [DOI] [PMC free article] [PubMed]
- 261.Li, H. et al. C1QTNF1-AS1 regulates the occurrence and development of hepatocellular carcinoma by regulating miR-221-3p/SOCS3. Hepatol. Int.13, 277–292 (2019). [DOI] [PubMed] [Google Scholar]
- 262.Khan, M. G. M. et al. Prognostic significance of SOCS1 and SOCS3 tumor suppressors and oncogenic signaling pathway genes in hepatocellular carcinoma. BMC Cancer20, 774 (2020). [DOI] [PMC free article] [PubMed]
- 263.Verhoeven, Y. et al. The potential and controversy of targeting STAT family members in cancer. Semin. Cancer Biol.60, 41–56 (2020). [DOI] [PubMed] [Google Scholar]
- 264.Loh, C. Y. et al. Signal transducer and activator of transcription (STATs) proteins in cancer and inflammation: Functions and therapeutic implication. Front. Oncol. 9, 48 (2019). [DOI] [PMC free article] [PubMed]
- 265.Thomas, S. J., Snowden, J. A., Zeidler, M. P. & Danson, S. J. The role of JAK/STAT signalling in the pathogenesis, prognosis and treatment of solid tumours. Br. J. Cancer113, 365–371 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Caraglia, M. et al. Alpha-Interferon and Its Effects on Signalling Pathways Within Cells. Curr. Protein Pept. Sci.5, 475–485 (2005). [DOI] [PubMed] [Google Scholar]
- 267.Iwamaru, A. et al. A novel inhibitor of the STAT3 pathway induces apoptosis in malignant glioma cells both in vitro and in vivo. Oncogene26, 2435–2444 (2007). [DOI] [PubMed] [Google Scholar]
- 268.Jensen, K. V., Cseh, O., Aman, A., Weiss, S. & Luchman, H. A. The JAK2/STAT3 inhibitor pacritinib effectively inhibits patient-derived GBM brain tumor initiating cells in vitro and when used in combination with temozolomide increases survival in an orthotopic xenograft model. PLoS One12, e0189670 (2017). [DOI] [PMC free article] [PubMed]
- 269.Mascarenhas, J. et al. Pacritinib vs best available therapy, including ruxolitinib, in patients with myelofibrosis: A randomized clinical trial. JAMA Oncol.4, 652–659 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Al-Fayoumi, S., Hashiguchi, T., Shirakata, Y., Mascarenhas, J. & Singer, J. W. Pilot study of the antifibrotic effects of the multikinase inhibitor pacritinib in a mouse model of liver fibrosis. J. Exp. Pharmacol.10, 9–17 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Yang, S. et al. Activating JAK1 mutation may predict the sensitivity of JAK-STAT inhibition in hepatocellular carcinoma. Oncotarget7, 5461–5469 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Wilson, G. S. et al. Tumoricidal effects of the JAK inhibitor Ruxolitinib (INC424) on hepatocellular carcinoma in vitro. Cancer Lett.341, 224–230 (2013). [DOI] [PubMed] [Google Scholar]
- 273.Eghtedar, A. et al. Phase 2 study of the JAK kinase inhibitor ruxolitinib in patients with refractory leukemias, including postmyeloproliferative neoplasm acute myeloid leukemia. Blood119, 4614–4618 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Zhou, Q. et al. Uridine-cytidine kinase 2 promotes metastasis of hepatocellular carcinoma cells via the stat3 pathway. Cancer Manag. Res.10, 6339–6355 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Schust, J., Sperl, B., Hollis, A., Mayer, T. U. & Berg, T. Stattic: A Small-Molecule Inhibitor of STAT3 Activation and Dimerization. Chem. Biol.13, 1235–1242 (2006). [DOI] [PubMed] [Google Scholar]
- 276.Yoo, C. et al. Phase I dose-finding study of OPB-111077, a novel STAT3 inhibitor, in patients with advanced hepatocellular carcinoma. Cancer Res. Treat.51, 510–518 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Bendell, J. C. et al. Phase 1, open-label, dose-escalation, and pharmacokinetic study of STAT3 inhibitor OPB-31121 in subjects with advanced solid tumors. Cancer Chemother. Pharmacol.74, 125–130 (2014). [DOI] [PubMed] [Google Scholar]
- 278.Shitara, K., Yodo, Y. & Iino, S. A phase I study of napabucasin plus paclitaxel for Japanese patients with advanced/recurrent gastric cancer. In Vivo Brooklyn33, 933–937 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Lee, C. & Cheung, S. T. STAT3: An emerging therapeutic target for hepatocellular carcinoma. Cancers. 11, 1646 (2019). [DOI] [PMC free article] [PubMed]
- 280.Oh, D. Y. et al. Phase I study of OPB-31121, an oral STAT3 inhibitor, in patients with advanced solid tumors. Cancer Res. Treat.47, 607–615 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Reilley, M. J. et al. STAT3 antisense oligonucleotide AZD9150 in a subset of patients with heavily pretreated lymphoma: Results of a phase 1b trial. J. Immunother. Cancer6, 119 (2018). [DOI] [PMC free article] [PubMed]
- 282.Obora, A. et al. Synergistic induction of apoptosis by acyclic retinoid and interferon-β in human hepatocellular carcinoma cells. Hepatology36, 1115–1124 (2002). [DOI] [PubMed] [Google Scholar]
- 283.Liu, S., Ren, J. & ten Dijke, P. Targeting TGF β signal transduction for cancer therapy. Signal Transduct. Target. Ther. 6, 8 (2021). [DOI] [PMC free article] [PubMed]
- 284.Pessino, G., Scotti, C. & Maggi, M. Hepatocellular Carcinoma: Old and Emerging Therapeutic Targets. Cancers. 16, 901 (2024). [DOI] [PMC free article] [PubMed]
- 285.Peng, D., Fu, M., Wang, M., Wei, Y. & Wei, X. Targeting TGF-β signal transduction for fibrosis and cancer therapy. Mol. Cancer21, 8 (2022). [DOI] [PMC free article] [PubMed]
- 286.David, C. J. & Massagué, J. Contextual determinants of TGF β action in development, immunity and cancer. Nat. Rev. Mol. Cell Biol.19, 419–435 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Chen, J. et al. Analysis of Genomes and Transcriptomes of Hepatocellular Carcinomas Identifies Mutations and Gene Expression Changes in the Transforming Growth Factor-β Pathway. Gastroenterology154, 195–210 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Oshimori, N., Oristian, D. & Fuchs, E. TGF-β Promotes Heterogeneity and Drug Resistance in Squamous. Cell Carcinoma. Cell160, 963–976 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Huang, S. et al. MED12 controls the response to multiple cancer drugs through regulation of TGF-β receptor signaling. Cell151, 937–950 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Wang, X., Eichhorn, P. J. A. & Thiery, J. P. TGF-β, EMT, and resistance to anti-cancer treatment. Semin. Cancer Biol.97, 1–11 (2023). [DOI] [PubMed] [Google Scholar]
- 291.Turati, M., Mousset, A., Issa, N., Turtoi, A. & Ronca, R. TGF-β mediated drug resistance in solid cancer. Cytokine Growth Factor Rev.71–72, 54–65 (2023). [DOI] [PubMed] [Google Scholar]
- 292.Kelley, R. K. et al. A phase 2 study of galunisertib (TGF-b1 Receptor Type i Inhibitor) and sorafenib in patients with advanced hepatocellular carcinoma. Clin. Transl. Gastroenterol. 10, e00056 (2019). [DOI] [PMC free article] [PubMed]
- 293.Harding, J. J. et al. Phase 1b study of galunisertib and ramucirumab in patients with advanced hepatocellular carcinoma. Cancer Med.10, 3059–3067 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Bauer, T. M. et al. Phase I/Ib, open-label, multicenter, dose-escalation study of the anti-TGF-β monoclonal antibody, NIS793, in combination with spartalizumab in adult patients with advanced tumors. J. Immunother. Cancer11, e007353 (2023). [DOI] [PMC free article] [PubMed]
- 295.Gonzalez-Sanchez, E. et al. The tgf-β pathway: A pharmacological target in hepatocellular carcinoma? Cancers. 13, 3248 (2021). [DOI] [PMC free article] [PubMed]
- 296.Liu, J. et al. Wnt/β-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct. Target. Ther. 7, 3 (2022). [DOI] [PMC free article] [PubMed]
- 297.Perugorria, M. J. et al. Wnt–β-catenin signalling in liver development, health and disease. Nat. Rev. Gastroenterol. Hepatol.16, 121–136 (2019). [DOI] [PubMed] [Google Scholar]
- 298.Tornesello, M. L. et al. Mutations in TP53, CTNNB1 and PIK3CA genes in hepatocellular carcinoma associated with hepatitis B and hepatitis C virus infections. Genomics102, 74–83 (2013). [DOI] [PubMed] [Google Scholar]
- 299.Ding, S. L. et al. Integrative analysis of aberrant Wnt signaling in hepatitis B virus-related hepatocellular carcinoma. World J. Gastroenterol.21, 6317–6328 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Xu, C. et al. β-Catenin signaling in hepatocellular carcinoma. J. Clin. Invest. 132, e154515 (2022). [DOI] [PMC free article] [PubMed]
- 301.Fan, Z. et al. PTK2 promotes cancer stem cell traits in hepatocellular carcinoma by activating Wnt/ β-catenin signaling. Cancer Lett.450, 132–143 (2019). [DOI] [PubMed] [Google Scholar]
- 302.Leung, H. W. et al. EPHB2 activates β-catenin to enhance cancer stem cell properties and drive sorafenib resistance in hepatocellular carcinoma. Cancer Res.81, 3229–3240 (2021). [DOI] [PubMed] [Google Scholar]
- 303.Lo, R. C. L. et al. Cripto-1 contributes to stemness in hepatocellular carcinoma by stabilizing Dishevelled-3 and activating Wnt/β-catenin pathway. Cell Death Differ.25, 1426–1441 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Bachmann, R. et al. Quality of operative treatment of pelvic fractures is not influenced by an additional abdominal injury: A monocentric registry study. Chirurg91, 483–490 (2020). [DOI] [PubMed] [Google Scholar]
- 305.Patil, M. A. et al. Role of cyclin D1 as a mediator of c-Met- and B-catenin-induced hepatocarcinogenesis. Cancer Res.69, 253–261 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Tao, J. et al. Modeling a human hepatocellular carcinoma subset in mice through coexpression of met and point-mutant β-catenin. Hepatology64, 1587–1605 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Tao, J. et al. Targeting β-catenin in hepatocellular cancers induced by coexpression of mutant β-catenin and K-Ras in mice. Hepatology65, 1581–1599 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Stauffer, J. K. et al. Coactivation of AKT and β-catenin in mice rapidly induces formation of lipogenic liver tumors. Cancer Res.71, 2718–2727 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Charawi, S. et al. LKB1 signaling is activated in CTNNB1-mutated HCC and positively regulates β-catenin-dependent CTNNB1-mutated HCC. J. Pathol.247, 435–443 (2019). [DOI] [PubMed] [Google Scholar]
- 310.Tao, J. et al. Nuclear factor erythroid 2–related factor 2 and β-Catenin Coactivation in Hepatocellular Cancer: Biological and Therapeutic Implications. Hepatology74, 741–759 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Tong, Z. et al. Steroid receptor coactivator 1 promotes human hepatocellular carcinoma progression by enhancing Wnt/ β-catenin signaling. J. Biol. Chem.290, 18596–18608 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Shang, X. Z. et al. Stabilized β-catenin promotes hepatocyte proliferation and inhibits TNFα-induced apoptosis. Lab. Investig.84, 332–341 (2004). [DOI] [PubMed] [Google Scholar]
- 313.Liu, R. et al. Gankyrin drives metabolic reprogramming to promote tumorigenesis, metastasis and drug resistance through activating β-catenin/c-Myc signaling in human hepatocellular carcinoma. Cancer Lett.443, 34–46 (2019). [DOI] [PubMed] [Google Scholar]
- 314.Qiao, Y. et al. Oncogenic potential of N-terminal deletion and S45Y mutant β-catenin in promoting hepatocellular carcinoma development in mice. BMC Cancer18, 1093 (2018). [DOI] [PMC free article] [PubMed]
- 315.Delgado, E. et al. β-Catenin Knockdown in Liver Tumor Cells by a Cell Permeable Gamma Guanidine-based Peptide Nucleic Acid. Curr. Cancer Drug Targets13, 867–878 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Shtutman, M. et al. The cyclin D1 gene is a target of the β-catenin/LEF-1 pathway. Proc. Natl. Acad. Sci. USA96, 5522–5527 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Tang, B. et al. Overexpression of CTNND1 in hepatocellular carcinoma promotes carcinous characters through activation of Wnt/ β-catenin signaling. J. Exp. Clin. Cancer Res. 35, 82 (2016). [DOI] [PMC free article] [PubMed]
- 318.Cadoret, A. et al. New targets of β-catenin signaling in the liver are involved in the glutamine metabolism. Oncogene21, 8293–8301 (2002). [DOI] [PubMed] [Google Scholar]
- 319.Lee, J. M. et al. β-Catenin signaling in hepatocellular cancer: Implications in inflammation, fibrosis, and proliferation. Cancer Lett.343, 90–97 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Wei, S. et al. KIF2C: a novel link between Wnt/ β-catenin and mTORC1 signaling in the pathogenesis of hepatocellular carcinoma. Protein Cell12, 788–809 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.Chiu, M. et al. Glutamine depletion by crisantaspase hinders the growth of human hepatocellular carcinoma xenografts. Br. J. Cancer111, 1159–1167 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Adebayo Michael, A. O. et al. Inhibiting Glutamine-Dependent mTORC1 Activation Ameliorates Liver Cancers Driven by β-Catenin Mutations. Cell Metab.29, 1135–1150.e6 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Karabicici, M. et al. Changes in Wnt and TGF-β Signaling Mediate the Development of Regorafenib Resistance in Hepatocellular Carcinoma Cell Line HuH7. Front. Cell Dev. Biol. 9, 639779 (2021). [DOI] [PMC free article] [PubMed]
- 324.Wang, J. et al. N6-Methyladenosine–Mediated Up-Regulation of FZD10 Regulates Liver Cancer Stem Cells’ Properties and Lenvatinib Resistance Through WNT/β-Catenin and Hippo Signaling Pathways. Gastroenterology164, 990–1005 (2023). [DOI] [PubMed] [Google Scholar]
- 325.Takada, R. et al. Monounsaturated Fatty Acid Modification of Wnt Protein: Its Role in Wnt Secretion. Dev. Cell11, 791–801 (2006). [DOI] [PubMed] [Google Scholar]
- 326.Yu, B. et al. Elevated expression of DKK1 is associated with cytoplasmic/nuclear β-catenin accumulation and poor prognosis in hepatocellular carcinomas. J. Hepatol.50, 948–957 (2009). [DOI] [PubMed] [Google Scholar]
- 327.Shi, X. D. et al. Dickkopf-1 expression is associated with tumorigenity and lymphatic metastasis in human hilar cholangiocarcinoma. Oncotarget7, 70378–70387 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Goyal, L. et al. Phase I and biomarker study of the wnt pathway modulator dkn-01 in combination with gemcitabine/ cisplatin in advanced biliary tract cancer. Clin. Cancer Res.26, 6158–6167 (2020). [DOI] [PubMed] [Google Scholar]
- 329.Wang, Y. C. et al. Drug Screening Identifies Niclosamide as an Inhibitor of Breast Cancer Stem-Like Cells. PLoS One8, e74538 (2013). [DOI] [PMC free article] [PubMed]
- 330.Ye, T. et al. The anthelmintic drug niclosamide induces apoptosis, impairs metastasis and reduces immunosuppressive cells in breast cancer model. PLoS One9, e85887 (2014). [DOI] [PMC free article] [PubMed]
- 331.Yang, X. G., Zhu, L. C., Wang, Y. J., Li, Y. Y. & Wang, D. Current Advance of Therapeutic Agents in Clinical Trials Potentially Targeting Tumor Plasticity. Front. Oncol. 9, 887 (2019). [DOI] [PMC free article] [PubMed]
- 332.Yamashita, T., Budhu, A., Forgues, M. & Xin, W. W. Activation of hepatic stem cell marker EpCAM by Wnt-β-catenin signaling in hepatocellular carcinoma. Cancer Res.67, 10831–10839 (2007). [DOI] [PubMed] [Google Scholar]
- 333.Wei, W., Chua, M. S., Grepper, S. & So, S. Small molecule antagonists of Tcf4/β-catenin complex inhibit the growth of HCC cells in vitro and in vivo. Int. J. Cancer126, 2426–2436 (2010). [DOI] [PubMed] [Google Scholar]
- 334.Ma, L. et al. Tankyrase inhibitors attenuate WNT/ß-catenin signaling and inhibit growth of hepatocellular carcinoma cells. Oncotarget6, 25390–25401 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Wang, X. H. et al. β-catenin siRNA regulation of apoptosis- and angiogenesis-related gene expression in hepatocellular carcinoma cells: Potential uses for gene therapy. Oncol. Rep.24, 1093–1099 (2010). [PubMed] [Google Scholar]
- 336.Sangkhathat, S. et al. In vitro RNA interference against β-catenin inhibits the proliferation of pediatric hepatic tumors. Int. J. Oncol.28, 715–722 (2006). [PubMed] [Google Scholar]
- 337.Delgado, E. et al. Complete response of Ctnnb1-mutated tumours to b-catenin suppression by locked nucleic acid antisense in a mouse hepatocarcinogenesis model. J. Hepatol.62, 380–387 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Jeng, K. S. et al. Sonic Hedgehog signaling pathway as a potential target to inhibit the progression of hepatocellular carcinoma (Review). Oncol. Lett.18, 4377–4384 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Jia, Y., Wang, Y. & Xie, J. The Hedgehog pathway: role in cell differentiation, polarity and proliferation. Arch. Toxicol.89, 179––1191 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Dimri, M. & Satyanarayana, A. Molecular signaling pathways and therapeutic targets in hepatocellular carcinoma. Cancers. 12, 491 (2020). [DOI] [PMC free article] [PubMed]
- 341.Zhang, Y. & Beachy, P. A. Cellular and molecular mechanisms of Hedgehog signalling. Nat. Rev. Mol. Cell Biol.24, 668–687 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Jing, J. et al. Hedgehog signaling in tissue homeostasis, cancers, and targeted therapies. Signal Transduct. Target. Ther. 8, 315 (2023). [DOI] [PMC free article] [PubMed]
- 343.Ding, J., Li, H. Y., Zhang, L., Zhou, Y. & Wu, J. Hedgehog signaling, a critical pathway governing the development and progression of hepatocellular carcinoma. Cells10, 1–18 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Skoda, A. M. et al. The role of the hedgehog signaling pathway in cancer: A comprehensive review. Bosn. J. Basic Med. Sci.18, 8–20 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Pan, Y. & Wang, B. A novel protein-processing domain in Gli2 and Gli3 differentially blocks complete protein degradation by the proteasome. J. Biol. Chem.282, 10846–10852 (2007). [DOI] [PubMed] [Google Scholar]
- 346.Wang, B. & Li, Y. Evidence for the direct involvement of βTrCP in Gli3 protein processing. Proc. Natl. Acad. Sci. USA103, 33–38 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Machado, M. V. & Diehl, A. M. Hedgehog signalling in liver pathophysiology. J. Hepatol.68, 550–562 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Goetz, S. C. & Anderson, K. V. The primary cilium: A signalling centre during vertebrate development. Nat. Rev. Genet.11, 331–344 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 349.Briscoe, J. & Thérond, P. P. The mechanisms of Hedgehog signalling and its roles in development and disease. Nat. Rev. Mol. Cell Biol.14, 418–431 (2013). [DOI] [PubMed] [Google Scholar]
- 350.Xia, R., Xu, M., Yang, J. & Ma, X. The role of Hedgehog and Notch signaling pathway in cancer. Mol. Biomed. 3, 44 (2022). [DOI] [PMC free article] [PubMed]
- 351.Tada, M. et al. Down-regulation of hedgehog-interacting protein through genetic and epigenetic alterations in human hepatocellular carcinoma. Clin. Cancer Res.14, 3768–3776 (2008). [DOI] [PubMed] [Google Scholar]
- 352.Huang, S. et al. Activation of the hedgehog pathway in human hepatocellular carcinomas. Carcinogenesis27, 1334–1340 (2006). [DOI] [PubMed] [Google Scholar]
- 353.Kim, Y. et al. Selective down-regulation of glioma-associated oncogene 2 inhibits the proliferation of hepatocellular carcinoma cells. Cancer Res.67, 3583–3593 (2007). [DOI] [PubMed] [Google Scholar]
- 354.Che, L., Yuan, Y. H., Jia, J. & Ren, J. Activation of sonic hedgehog signaling pathway is an independent potential prognosis predictor in human hepatocellular carcinoma patients. Chinese J. Cancer Res.24, 323–331 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Patil, M. A. et al. Hedgehog signaling in human hepatocellular carcinoma. Cancer Biol. Ther.5, 111–117 (2006). [DOI] [PubMed] [Google Scholar]
- 356.Di Magliano, M. P. & Hebrok, M. Hedgehog signalling in cancer formation and maintenance. Nat. Rev. Cancer3, 903–911 (2003). [DOI] [PubMed] [Google Scholar]
- 357.Philips, G. M. et al. Hedgehog signaling antagonist promotes regression of both liver fibrosis and hepatocellular carcinoma in a murine model of primary liver cancer. PLoS One6, e23943 (2011). [DOI] [PMC free article] [PubMed]
- 358.Bollati, V. et al. Dysregulation of the Hedgehog pathway in human hepatocarcinogenesis. Carcinogenesis30, 1330–1335 (2017). [DOI] [PubMed] [Google Scholar]
- 359.Chung, S. I. et al. Hepatic expression of Sonic Hedgehog induces liver fibrosis and promotes hepatocarcinogenesis in a transgenic mouse model. J. Hepatol.64, 618–627 (2016). [DOI] [PubMed] [Google Scholar]
- 360.Syn, W. K. et al. Hedgehog-Mediated Epithelial-to-Mesenchymal Transition and Fibrogenic Repair in Nonalcoholic Fatty Liver Disease. Gastroenterology137, 1478-1488.e8 (2009). [DOI] [PMC free article] [PubMed]
- 361.Cheng, W. T. et al. Role of Hedgehog signaling pathway in proliferation and invasiveness of hepatocellular carcinoma cells. Int. J. Oncol.34, 829–836 (2009). [DOI] [PubMed] [Google Scholar]
- 362.Chen, J. S. et al. Down-regulation of Gli-1 inhibits hepatocellular carcinoma cell migration and invasion. Mol. Cell. Biochem.393, 283–291 (2014). [DOI] [PubMed] [Google Scholar]
- 363.Abou-Alfa, G. K. et al. Pharmacokinetics and safety of vismodegib in patients with advanced solid malignancies and hepatic impairment. Cancer Chemother. Pharmacol.80, 29–36 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Lear, J. T. et al. Concomitant medication use does not appear to alter long-term efficacy of sonidegib for advanced basal cell carcinoma: a post hoc analysis based on the 42-month BOLT study. Eur. J. Dermatology33, 280–286 (2023). [DOI] [PubMed] [Google Scholar]
- 365.Chen, B., Hu, Z., Chen, B. & Li, B. Effects and mechanism of Lanthanum Citrate on the proliferation and apoptosis of hepatocellular carcinoma cell line SMMC-7721. Turkish J. Gastroenterol.31, 264–271 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Sicklick, J. K. et al. Dysregulation of the Hedgehog pathway in human hepatocarcinogenesis. Carcinogenesis27, 748–757 (2006). [DOI] [PubMed] [Google Scholar]
- 367.Jeng, K. S. et al. Blockade of the sonic hedgehog pathway effectively inhibits the growth of hepatoma in mice: An in vivo study. Oncol. Lett.4, 1158–1162 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Wang, S. et al. Hedgehog signaling promotes sorafenib resistance in hepatocellular carcinoma patient-derived organoids. J. Exp. Clin. Cancer Res. 39, 22 (2020). [DOI] [PMC free article] [PubMed]
- 369.Zhou, X. T. et al. Hedgehog signalling mediates drug resistance through targeting TAP1 in hepatocellular carcinoma. J. Cell. Mol. Med.24, 4298–4311 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.Liu, B. W. et al. GANT61, an inhibitor of Gli1, inhibits the proliferation and migration of hepatocellular carcinoma cells. J. Investig. Med.72, 181–192 (2024). [DOI] [PubMed] [Google Scholar]
- 371.Wang, Y., Han, C., Lu, L., Magliato, S. & Wu, T. Hedgehog signaling pathway regulates autophagy in human hepatocellular carcinoma cells. Hepatology58, 995–1010 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 372.Harada, K., Ohashi, R., Naito, K. & Kanki, K. Hedgehog signal inhibitor GANT61 inhibits the malignant behavior of undifferentiated hepatocellular carcinoma cells by targeting non-canonical GLI signaling. Int. J. Mol. Sci. 21, (2020). [DOI] [PMC free article] [PubMed]
- 373.Sheng, X. et al. Inhibitory effect of bufalin combined with Hedgehog signaling pathway inhibitors on proliferation and invasion and metastasis of liver cancer cells. Int. J. Oncol.49, 1513–1524 (2016). [DOI] [PubMed] [Google Scholar]
- 374.Xu, Y. et al. Polymeric nanoparticle-encapsulated hedgehog pathway inhibitor HPI-1 (NanoHHI) inhibits systemic metastases in an orthotopic model of human hepatocellular carcinoma. Clin. Cancer Res.18, 1291–1302 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.Li, K., Fang, D., Xiong, Z. & Luo, R. Inhibition of the hedgehog pathway for the treatment of cancer using Itraconazole. Onco. Targets. Ther.12, 6875–6886 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.Tian, H. & He, Z. Anti-hepatoma effect of taccalonolide A through suppression of sonic hedgehog pathway. Artif. Cells Nanomedicine Biotechnol.48, 939–947 (2020). [DOI] [PubMed] [Google Scholar]
- 377.Zheng, X. et al. The human sulfatase 2 inhibitor 2,4-disulfonylphenyl-tert-butylnitrone (OKN-007) has an antitumor effect in hepatocellular carcinoma mediated via suppression of TGFB1/SMAD2 and Hedgehog/GLI1 signaling. Genes Chromosom. Cancer52, 225–236 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Dong, J. et al. Elucidation of a Universal Size-Control Mechanism in Drosophila and Mammals. Cell130, 1120–1133 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Russell, J. O. & Camargo, F. D. Hippo signalling in the liver: role in development, regeneration and disease. Nat. Rev. Gastroenterol. Hepatol.19, 297–312 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Takamori, S. et al. Molecular Anatomy of a Trafficking Organelle. Cell127, 831–846 (2006). [DOI] [PubMed] [Google Scholar]
- 381.Tao, J. et al. Activation of β-catenin and Yap1 in human hepatoblastoma and induction of hepatocarcinogenesis in mice. Gastroenterology147, 690–701 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 382.Li, H. et al. Deregulation of Hippo kinase signalling in Human hepatic malignancies. Liver Int.32, 38–47 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Sohn, B. H. et al. Inactivation of hippo pathway is significantly associated with poor prognosis in hepatocellular carcinoma. Clin. Cancer Res.22, 1256–1264 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Fitamant, J. et al. YAP Inhibition Restores Hepatocyte Differentiation in Advanced HCC, Leading to Tumor Regression. Cell Rep.10, 1692–1707 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Xu, M. Z. et al. Yes-associated protein is an independent prognostic marker in hepatocellular carcinoma. Cancer115, 4576–4585 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Yi, C. et al. The p130 isoform of angiomotin is required for yap-mediated hepatic epithelial cell proliferation and tumorigenesis. Sci. Signal. 6, ra77 (2013). [DOI] [PMC free article] [PubMed]
- 387.Zhang, N. et al. The Merlin/NF2 Tumor Suppressor Functions through the YAP Oncoprotein to Regulate Tissue Homeostasis in Mammals. Dev. Cell19, 27–38 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Hoffer, K. M. Resolving payment disputes in small claims court. Patient Acc.21, 2–3 (1998). [PubMed] [Google Scholar]
- 389.Qi, S. et al. Two Hippo signaling modules orchestrate liver size and tumorigenesis. EMBO J. 42, e112126 (2023). [DOI] [PMC free article] [PubMed]
- 390.Liang, B. et al. Differential requirement of Hippo cascade during CTNNB1 or AXIN1 mutation-driven hepatocarcinogenesis. Hepatology77, 1929–1942 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Wang, H. et al. TAZ is indispensable for c-MYC-induced hepatocarcinogenesis. J. Hepatol.76, 123–1134 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Yuan, W. C. et al. NUAK2 is a critical YAP target in liver cancer. Nat. Commun. 9, 4834 (2018). [DOI] [PMC free article] [PubMed]
- 393.Tschaharganeh, D. F. et al. Yes-associated protein up-regulates jagged-1 and activates the NOTCH pathway in human hepatocellular carcinoma. Gastroenterology144, 1530–1542.e12 (2013). [DOI] [PMC free article] [PubMed]
- 394.Kim, W. et al. Hepatic Hippo signaling inhibits protumoural microenvironment to suppress hepatocellular carcinoma. Gut67, 1692–1703 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Guo, X. et al. Single tumor-initiating cells evade immune clearance by recruiting type II macrophages. Genes Dev31, 247–259 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Xia, H. et al. EGFR-PI3K-PDK1 pathway regulates YAP signaling in hepatocellular carcinoma: The mechanism and its implications in targeted therapy article. Cell Death Dis. 9, 269 (2018). [DOI] [PMC free article] [PubMed]
- 397.Jeong, S. H. et al. Hippo-mediated suppression of IRS2/AKT signaling prevents hepatic steatosis and liver cancer. J. Clin. Invest.128, 1010–1025 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 398.Xiong, Y. X. et al. Collagen I-DDR1 signaling promotes hepatocellular carcinoma cell stemness via Hippo signaling repression. Cell Death Differ30, 1648–1665 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Kim, W. et al. Hippo signaling interactions with Wnt/ β-catenin and Notch signaling repress liver tumorigenesis. J. Clin. Invest.127, 137–152 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Higashi, T. et al. Statin attenuates cell proliferative ability via TAZ (WWTR1) in hepatocellular carcinoma. Med. Oncol. 33, 123 (2016). [DOI] [PubMed]
- 401.Xu, W. et al. Salvianolic acid B exerts an anti-hepatocellular carcinoma effect by regulating the Hippo/YAP pathway and promoting pSmad3L to pSmad3C simultaneously. Eur. J. Pharmacol. 939, 175423 (2023). [DOI] [PubMed]
- 402.Lee, D. H. et al. LATS-YAP/TAZ controls lineage specification by regulating TGF β signaling and Hnf4α expression during liver development. Nat. Commun. 7, 11961 (2016). [DOI] [PMC free article] [PubMed]
- 403.Namoto, K. et al. NIBR-LTSi is a selective LATS kinase inhibitor activating YAP signaling and expanding tissue stem cells in vitro and in vivo. Cell Stem Cell31, 554–569.e17 (2024). [DOI] [PubMed] [Google Scholar]
- 404.Kovall, R. A., Gebelein, B., Sprinzak, D. & Kopan, R. The Canonical Notch Signaling Pathway: Structural and Biochemical Insights into Shape, Sugar, and Force. Dev. Cell41, 228–241 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 405.Andersson, E. R. & Lendahl, U. Therapeutic modulation of Notch signalling-are we there yet? Nat. Rev. Drug Discov.13, 357–378 (2014). [DOI] [PubMed] [Google Scholar]
- 406.Ayaz, F. & Osborne, B. A. Non-canonical Notch signaling in cancer and immunity. Front. Oncol. 4, 345 (2014). [DOI] [PMC free article] [PubMed]
- 407.Espinoza, I. & Miele, L. Notch inhibitors for cancer treatment. Pharmacol. Ther.139, 95–110 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Katoh, M. & Katoh, M. Transcriptional mechanisms of WNT5A based on NF-κB, Hedgehog, TGF β, and Notch signaling cascades. Int. J. Mol. Med.23, 763–769 (2009). [DOI] [PubMed] [Google Scholar]
- 409.Kim, W., Khan, S. K. & Yang, Y. Interacting network of Hippo, Wnt/β-catenin and Notch signaling represses liver tumor formation. BMB Rep.50, 1–2 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.Mangolini, M. et al. Notch2 controls non-autonomous Wnt-signalling in chronic lymphocytic leukaemia. Nat. Commun. 9, 3839 (2018). [DOI] [PMC free article] [PubMed]
- 411.Chen, L. J. et al. Gm364 coordinates MIB2/DLL3/Notch2 to regulate female fertility through AKT activation. Cell Death Differ. 29, 366–380 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Song, L. L. et al. Notch-1 associates with IKKα and regulates IKK activity in cervical cancer cells. Oncogene27, 5833–5844 (2008). [DOI] [PubMed] [Google Scholar]
- 413.Razumilava, N. & Gores, G. J. Notch-driven carcinogenesis: The merging of hepatocellular cancer and cholangiocarcinoma into a common molecular liver cancer subtype. J. Hepatol.58, 1244–1245 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Yuan, X. et al. Meta-analysis reveals the correlation of Notch signaling with non-small cell lung cancer progression and prognosis. Sci. Rep. 5, 10338 (2015). [DOI] [PMC free article] [PubMed]
- 415.Fre, S. et al. Notch and Wnt signals cooperatively control cell proliferation and tumorigenesis in the intestine. Proc. Natl. Acad. Sci. USA106, 6309–6314 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Reedijk, M. et al. High-level coexpression of JAG1 and NOTCH1 is observed in human breast cancer and is associated with poor overall survival. Cancer Res.65, 8530–8537 (2005). [DOI] [PubMed] [Google Scholar]
- 417.Giovannini, C. et al. Notch3 inhibition enhances sorafenib cytotoxic efficacy by promoting GSK3ß phosphorylation and p21 down-regulation in hepatocellular carcinoma. Oncotarget4, 1618–1631 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Dill, M. T. et al. Constitutive Notch2 signaling induces hepatic tumors in mice. Hepatology57, 1607–1619 (2013). [DOI] [PubMed] [Google Scholar]
- 419.Giovannini, C. et al. Molecular and proteomic insight into Notch1 characterization in hepatocellular carcinoma. Oncotarget7, 39609–39626 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 420.Villanueva, A. et al. Notch signaling is activated in human hepatocellular carcinoma and induces tumor formation in mice. Gastroenterology143, 1660–1669.e7 (2012). [DOI] [PMC free article] [PubMed]
- 421.Viatour, P. et al. Notch signaling inhibits hepatocellular carcinoma following inactivation of the RB pathway. J. Exp. Med.208, 1963–1976 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Xie, Q. et al. Tspan5 promotes epithelial–mesenchymal transition and tumour metastasis of hepatocellular carcinoma by activating Notch signalling. Mol. Oncol.15, 3184–3202 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Fang, S. et al. Lymphoid enhancer-binding factor-1 promotes stemness and poor differentiation of hepatocellular carcinoma by directly activating the NOTCH pathway. Oncogene38, 4061–4074 (2019). [DOI] [PubMed] [Google Scholar]
- 424.Luo, H. et al. Differentiation-inducing therapeutic effect of Notch inhibition in reversing malignant transformation of liver normal stem cells via MET. Oncotarget9, 18885–18895 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Zhang, S. et al. Hippo Cascade Controls Lineage Commitment of Liver Tumors in Mice and Humans. Am. J. Pathol.188, 995–1006 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 426.Huntzicker, E. G. et al. Differential effects of targeting Notch receptors in a mouse model of liver cancer. Hepatology61, 942–952 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Ye, Y. C. et al. Notch signaling via Wnt regulates the proliferation of alternative, CCR2-independent tumor-associated macrophages in hepatocellular carcinoma. Cancer Res.79, 4160–4172 (2019). [DOI] [PubMed] [Google Scholar]
- 428.Moore, G., Annett, S., McClements, L. & Robson, T. Top Notch Targeting Strategies in Cancer: A Detailed Overview of Recent Insights and Current Perspectives. Cells9, 1503 (2020). [DOI] [PMC free article] [PubMed]
- 429.Hur, J. Y. γ-Secretase in Alzheimer’s disease. Exp. Mol. Med.54, 433–446 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Feng, M., Santhanam, R. K., Xing, H., Zhou, M. & Jia, H. Inhibition of γ-secretase/Notch pathway as a potential therapy for reversing cancer drug resistance. Biochem. Pharmacol. 220, 115991 (2024). [DOI] [PubMed]
- 431.Keam, S. J. Nirogacestat: First Approval. Drugs84, 355–361 (2024). [DOI] [PubMed] [Google Scholar]
- 432.Golde, T. E., Koo, E. H., Felsenstein, K. M., Osborne, B. A. & Miele, L. γ-Secretase inhibitors and modulators. Biochim. Biophys. Acta Biomembr.1828, 2898–2907 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 433.Kang, M. S. et al. Modulation of lipid kinase PI4KIIα activity and lipid raft association of presenilin 1 underlies γ-secretase inhibition by ginsenoside (20S)-Rg3. J. Biol. Chem.288, 20868–20882 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 434.Song, J. H. et al. Inhibitory effect of ginsenoside Rg3 on cancer stemness and mesenchymal transition in breast cancer via regulation of myeloidderived suppressor cells. PLoS One15, e0240533 (2020). [DOI] [PMC free article] [PubMed]
- 435.Li, X. et al. Ginsenoside Rg3 Suppresses Epithelial-Mesenchymal Transition via Downregulating Notch-Hes1 Signaling in Colon Cancer Cells. Am. J. Chin. Med.49, 217–235 (2021). [DOI] [PubMed] [Google Scholar]
- 436.Zhou, B. et al. Prospective study of Transcatheter arterial chemoembolization (Tace) with ginsenoside rg3 versus Tace alone for the Treatment of Patients with advanced hepatocellular carcinoma. Radiology280, 630–639 (2016). [DOI] [PubMed] [Google Scholar]
- 437.Lu, M., Fei, Z. & Zhang, G. Synergistic anticancer activity of 20(S)-Ginsenoside Rg3 and Sorafenib in hepatocellular carcinoma by modulating PTEN/Akt signaling pathway. Biomed. Pharmacother.97, 1282–1288 (2018). [DOI] [PubMed] [Google Scholar]
- 438.Xu, C. et al. The ADAM17 inhibitor ZLDI-8 sensitized hepatocellular carcinoma cells to sorafenib through Notch1-integrin β-talk. Pharmacol. Res. 203, 107142 (2024). [DOI] [PubMed]
- 439.Guterres, A. N. & Villanueva, J. Targeting telomerase for cancer therapy. Oncogene39, 5811–5824 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 440.Nault, J. C., Ningarhari, M., Rebouissou, S. & Zucman-Rossi, J. The role of telomeres and telomerase in cirrhosis and liver cancer. Nat. Rev. Gastroenterol. Hepatol.16, 544–558 (2019). [DOI] [PubMed] [Google Scholar]
- 441.Kotiyal, S. & Evason, K. J. Exploring the interplay of telomerase reverse transcriptase and β-catenin in hepatocellular carcinoma. Cancers. 13, 4202 (2021). [DOI] [PMC free article] [PubMed]
- 442.Garcia-Lezana, T., Lopez-Canovas, J. L. & Villanueva, A. Signaling pathways in hepatocellular carcinoma. Adv. Cancer Res.149, 63–101 (2021). [DOI] [PubMed] [Google Scholar]
- 443.Djojosubroto, M. W. et al. Telomerase antagonists GRN163 and GRN163L inhibit tumor growth and increase chemosensitivity of human hepatoma. Hepatology42, 1127–1136 (2005). [DOI] [PubMed] [Google Scholar]
- 444.Ningarhari, M. et al. Telomere length is key to hepatocellular carcinoma diversity and telomerase addiction is an actionable therapeutic target. J. Hepatol.74, 1155–1166 (2021). [DOI] [PubMed] [Google Scholar]
- 445.Tahtouh, R. et al. Telomerase inhibition decreases alpha-fetoprotein expression and secretion by hepatocellular carcinoma cell lines: In vitro and in vivo study. PLoS One10, e0119512 (2015). [DOI] [PMC free article] [PubMed]
- 446.Heo, J. et al. Safety and dose escalation of the targeted oncolytic adenovirus OBP-301 for refractory advanced liver cancer: Phase I clinical trial. Mol. Ther.31, 2077–2088 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 447.Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov12, 31–46 (2022). [DOI] [PubMed] [Google Scholar]
- 448.Tan Y. et al. Alternative polyadenylation reprogramming of MORC2 induced by NUDT21 loss promotes KIRC carcinogenesis. JCI Insight. 8, e162893 (2023). [DOI] [PMC free article] [PubMed]
- 449.Villanueva, A. et al. DNA methylation-based prognosis and epidrivers in hepatocellular carcinoma. Hepatology61, 1945–1956 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Zheng, Y., Tang, L., Chen, G. & Liu, Z. Comprehensive Bioinformatics Analysis of Key Methyltransferases and Demethylases for Histone Lysines in Hepatocellular Carcinoma. Technol. Cancer Res. Treat. 19, 1533033820983284 (2020). [DOI] [PMC free article] [PubMed]
- 451.Panera, N., Crudele, A., Romito, I., Gnani, D. & Alisi, A. Focal adhesion kinase: Insight into molecular roles and functions in hepatocellular carcinoma. Int. J. Mol. Sci. 18, 93–103 (2017). [DOI] [PMC free article] [PubMed]
- 452.Gnani, D. et al. Focal adhesion kinase depletion reduces human hepatocellular carcinoma growth by repressing enhancer of zeste homolog 2. Cell Death Differ.24, 889–902 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 453.Kusakabe, Y. et al. EZH1/2 inhibition augments the anti-tumor effects of sorafenib in hepatocellular carcinoma. Sci. Rep. 11, 21396 (2021). [DOI] [PMC free article] [PubMed]
- 454.Qian, Y. et al. High methylation levels of histone H3 lysine 9 associated with activation of hypoxia-inducible factor 1α (HIF-1α) predict patients’ worse prognosis in human hepatocellular carcinomas. Cancer Genet245, 17–26 (2020). [DOI] [PubMed] [Google Scholar]
- 455.Davalos, V. & Esteller, M. Cancer epigenetics in clinical practice. CA. Cancer J. Clin73, 376–424 (2023). [DOI] [PubMed] [Google Scholar]
- 456.Zhang, S. et al. Targeting histone modifiers in bladder cancer therapy — preclinical and clinical evidence. Nat. Rev. Urol. 21, 495–511 (2024). [DOI] [PubMed]
- 457.Poté, N. et al. The histone acetyltransferase hMOF promotes vascular invasion in hepatocellular carcinoma. Liver Int.40, 956–967 (2020). [DOI] [PubMed] [Google Scholar]
- 458.Zhang, J. et al. The histone acetyltransferase hMOF suppresses hepatocellular carcinoma growth. Biochem. Biophys. Res. Commun.452, 575–580 (2014). [DOI] [PubMed] [Google Scholar]
- 459.Stathis, A. & Bertoni, F. BET proteins as targets for anticancer treatment. Cancer Discov.8, 24–36 (2018). [DOI] [PubMed] [Google Scholar]
- 460.Tsang, F. H. C. et al. Aberrant Super-Enhancer Landscape in Human Hepatocellular Carcinoma. Hepatology69, 2502–2517 (2019). [DOI] [PubMed] [Google Scholar]
- 461.Du, G., Yang, R., Qiu, J. & Xia, J. Multifaceted Influence of Histone Deacetylases on DNA Damage Repair: Implications for Hepatocellular Carcinoma. J. Clin. Transl. Hepatol.11, 231––2243 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 462.Zheng, J. et al. NAT10 regulates mitotic cell fate by acetylating Eg5 to control bipolar spindle assembly and chromosome segregation. Cell Death Differ.29, 846–860 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 463.Mei, Q. et al. An open-label, single-arm, phase I/II study of lowerdose decitabine based therapy in patients with advanced hepatocellular carcinoma. Oncotarget6, 16698–16711 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 464.Llopiz, D. et al. Enhanced anti-tumor efficacy of checkpoint inhibitors in combination with the histone deacetylase inhibitor Belinostat in a murine hepatocellular carcinoma model. Cancer Immunol. Immunother.68, 379–393 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 465.Gordon, S. W. et al. Phase i study of sorafenib and vorinostat in advanced hepatocellular carcinoma. Am. J. Clin. Oncol. Cancer Clin. Trials42, 649–654 (2019). [DOI] [PubMed] [Google Scholar]
- 466.Bitzer, M. et al. Resminostat plus sorafenib as second-line therapy of advanced hepatocellular carcinoma – The SHELTER study. J. Hepatol.65, 280–288 (2016). [DOI] [PubMed] [Google Scholar]
- 467.Tak, W. Y. et al. Phase I/II study of first-line combination therapy with sorafenib plus resminostat, an oral HDAC inhibitor, versus sorafenib monotherapy for advanced hepatocellular carcinoma in east Asian patients. Invest. New Drugs36, 1072–1084 (2018). [DOI] [PubMed] [Google Scholar]
- 468.Bugide, S., Gupta, R., Green, M. R. & Wajapeyee, N. EZH2 inhibits NK cell–mediated antitumor immunity by suppressing CXCL10 expression in an HDAC10-dependent manner. Proc. Natl. Acad. Sci. USA118, e2102718118 (2021). [DOI] [PMC free article] [PubMed]
- 469.Bárcena-Varela, M. et al. Dual Targeting of Histone Methyltransferase G9a and DNA-Methyltransferase 1 for the Treatment of Experimental Hepatocellular Carcinoma. Hepatology69, 587–603 (2019). [DOI] [PubMed] [Google Scholar]
- 470.Wei, L. et al. Histone methyltransferase G9a promotes liver cancer development by epigenetic silencing of tumor suppressor gene RARRES3. J. Hepatol.67, 758–769 (2017). [DOI] [PubMed] [Google Scholar]
- 471.Barcena-Varela, M. et al. Epigenetic mechanisms and metabolic reprogramming in fibrogenesis: Dual targeting of G9a and DNMT1 for the inhibition of liver fibrosis. Gut70, 388–400 (2021). [DOI] [PubMed] [Google Scholar]
- 472.Zhang, L. et al. DNMT and EZH2 inhibitors synergize to activate therapeutic targets in hepatocellular carcinoma: Combination treatment of DNMT and EZH2 inhibitors for HCC. Cancer Lett. 548, 215899 (2022). [DOI] [PMC free article] [PubMed]
- 473.Yang, W. et al. A selective HDAC8 inhibitor potentiates antitumor immunity and efficacy of immune checkpoint blockade in hepatocellular carcinoma. Sci. Transl. Med. 13, eaaz6804 (2021). [DOI] [PubMed]
- 474.Jühling, F. et al. Targeting clinical epigenetic reprogramming for chemoprevention of metabolic and viral hepatocellular carcinoma. Gut70, 157–169 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 475.Bayo, J. et al. A comprehensive study of epigenetic alterations in hepatocellular carcinoma identifies potential therapeutic targets. J. Hepatol.71, 78–90 (2019). [DOI] [PubMed] [Google Scholar]
- 476.Liu, C. et al. Bromo- and extraterminal domain protein inhibition improves immunotherapy efficacy in hepatocellular carcinoma. Cancer Sci.111, 3503–3515 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 477.Liu, M. et al. Targeting monocyte-intrinsic enhancer reprogramming improves immunotherapy efficacy in hepatocellular carcinoma. Gut69, 365–379 (2020). [DOI] [PubMed] [Google Scholar]
- 478.Lai, X. et al. Modeling combination therapy for breast cancer with BET and immune checkpoint inhibitors. Proc. Natl. Acad. Sci. USA115, 5534–5539 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Hogg, S. J. et al. BET-Bromodomain Inhibitors Engage the Host Immune System and Regulate Expression of the Immune Checkpoint Ligand PD-L1. Cell Rep.18, 2162–2174 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Wang, N., Wu, R., Tang, D. & Kang, R. The BET family in immunity and disease. Signal Transduct. Target. Ther. 6, 23 (2021). [DOI] [PMC free article] [PubMed]
- 481.Wang, Y. & Cao, K. KDM1A Promotes Immunosuppression in Hepatocellular Carcinoma by Regulating PD-L1 through Demethylating MEF2D. J. Immunol. Res. 2021, 9965099 (2021). [DOI] [PMC free article] [PubMed]
- 482.Hisada, R. et al. The deacetylase SIRT2 contributes to autoimmune disease pathogenesis by modulating IL-17A and IL-2 transcription. Cell. Mol. Immunol.19, 738–750 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 483.Levine, A. J. P53: 800 Million Years of Evolution and 40 Years of Discovery. Nat. Rev. Cancer20, 471–480 (2020). [DOI] [PubMed] [Google Scholar]
- 484.Voskarides, K. & Giannopoulou, N. The Role of TP53 in Adaptation and Evolution. Cells12, 512 (2023). [DOI] [PMC free article] [PubMed]
- 485.Liu, Y., Su, Z., Tavana, O. & Gu, W. Understanding the complexity of p53 in a new era of tumor suppression. Cancer Cell42, 946–967 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 486.Peuget, S., Zhou, X. & Selivanova, G. Translating p53-based therapies for cancer into the clinic. Nat. Rev. Cancer24, 192–215 (2024). [DOI] [PubMed] [Google Scholar]
- 487.Midorikawa, Y. et al. Accumulation of molecular aberrations distinctive to hepatocellular carcinoma progression. Cancer Res.80, 3810–3819 (2020). [DOI] [PubMed] [Google Scholar]
- 488.Fujita, M. et al. Proteo-genomic characterization of virus-associated liver cancers reveals potential subtypes and therapeutic targets. Nat. Commun. 13, 6481 (2022). [DOI] [PMC free article] [PubMed]
- 489.Liu, J. et al. Alterations of TP53 are associated with a poor outcome for patients with hepatocellular carcinoma: Evidence from a systematic review and meta-analysis. Eur. J. Cancer48, 2328–2338 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 490.Cao, H. et al. The role of MDM2–p53 axis dysfunction in the hepatocellular carcinoma transformation. Cell Death Discov. 6, 53 (2020). [DOI] [PMC free article] [PubMed]
- 491.Hassin, O. & Oren, M. Drugging p53 in cancer: one protein, many targets. Nat. Rev. Drug Discov.22, 127–144 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 492.Zhang, W. W. et al. The First Approved Gene Therapy Product for Cancer Ad-p53 (Gendicine): 12 Years in the Clinic. Hum. Gene Ther.29, 160–179 (2018). [DOI] [PubMed] [Google Scholar]
- 493.Guo, Y. et al. Recombinant human adenovirus p53 combined with transcatheter arterial chemoembolization for liver cancer: A meta-analysis. PLoS One18, e0295323 (2023). [DOI] [PMC free article] [PubMed]
- 494.Yang, Z. X. et al. Clinical study of recombinant adenovirus-p53 combined with fractionated stereotactic radiotherapy for hepatocellular carcinoma. J. Cancer Res. Clin. Oncol.136, 625–630 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 495.Xiao, Y. et al. Combining p53 mRNA nanotherapy with immune checkpoint blockade reprograms the immune microenvironment for effective cancer therapy. Nat. Commun. 13, 758 (2022). [DOI] [PMC free article] [PubMed]
- 496.Kong, N. et al. Synthetic mRNA nanoparticle-mediated restoration of p53 tumor suppressor sensitizes p53-deficient cancers to mTOR inhibition. Sci. Transl. Med. 11, eaaw1565 (2019). [DOI] [PMC free article] [PubMed]
- 497.Liu, Y. et al. A Novel Indolizine Derivative Induces Apoptosis Through the Mitochondria p53 Pathway in HepG2 Cells. Front. Pharmacol. 10, 762 (2019). [DOI] [PMC free article] [PubMed]
- 498.Gomes, S. et al. SLMP53-2 restoreswild-type-like function to mutant p53 through hsp70: Promising activity in hepatocellular carcinoma. Cancers11, 1151 (2019). [DOI] [PMC free article] [PubMed]
- 499.Khan, M. W. et al. Synergism of cisplatin-oleanolic acid co-loaded calcium carbonate nanoparticles on hepatocellular carcinoma cells for enhanced apoptosis and reduced hepatotoxicity. Int. J. Nanomedicine14, 3753–3771 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 500.Wu, Y. Y. et al. The anti-proliferation effect of Aikete injection on hepatocellular carcinoma in vitro and in vivo. Pharm. Biol.49, 531–538 (2011). [DOI] [PubMed] [Google Scholar]
- 501.Wang, H., Guo, M., Wei, H. & Chen, Y. Targeting p53 pathways: mechanisms, structures, and advances in therapy. Signal Transduct. Target. Ther. 8, 92 (2023). [DOI] [PMC free article] [PubMed]
- 502.Nishikawa, S. & Iwakuma, T. Drugs Targeting p53 Mutations with FDA Approval and in Clinical Trials. Cancers. 15, 429 (2023). [DOI] [PMC free article] [PubMed]
- 503.Emiloju, O. E. et al. Phase 1 trial of navitoclax and sorafenib in patients with relapsed or refractory solid tumors with hepatocellular carcinoma expansion cohort. Invest. New Drugs42, 127–135 (2024). [DOI] [PubMed] [Google Scholar]
- 504.Li, J., Xu, J. & Li, Z. Obatoclax, the pan-Bcl-2 inhibitor sensitizes hepatocellular carcinoma cells to promote the anti-tumor efficacy in combination with immune checkpoint blockade. Transl. Oncol. 14, 101116 (2021). [DOI] [PMC free article] [PubMed]
- 505.Michalski, M. et al. Simultaneous Inhibition of Mcl-1 and Bcl-2 Induces Synergistic Cell Death in Hepatocellular Carcinoma. Biomedicines11, 1666 (2023). [DOI] [PMC free article] [PubMed]
- 506.Eun, H. S. et al. Scutellarein induces fas-mediated extrinsic apoptosis and G2/M cell cycle arrest in Hep3B hepatocellular carcinoma cells. Nutrients11, 263 (2019). [DOI] [PMC free article] [PubMed]
- 507.Jin, S., Shi, K., Liu, L., Chen, Y. & Yang, G. Xanthones from the bark of Garcinia xanthochymus and the mechanism of induced apoptosis in human hepatocellular carcinoma HepG2 cells via the mitochondrial pathway. Int. J. Mol. Sci. 20, 4803 (2019). [DOI] [PMC free article] [PubMed]
- 508.Güzelcan, E. A., Baxendale, I. R., Cetin-Atalay, R. & Baumann, M. Synthesis of new derivatives of boehmeriasin A and their biological evaluation in liver cancer. Eur. J. Med. Chem.166, 243–255 (2019). [DOI] [PubMed] [Google Scholar]
- 509.Liu, W. Y. et al. Lycorine induces mitochondria-dependent apoptosis in hepatoblastoma HepG2 cells through ROCK1 activation. Front. Pharmacol. 10, 651 (2019). [DOI] [PMC free article] [PubMed]
- 510.Shah, A. et al. FDA approval: Ribociclib for the treatment of postmenopausal women with hormone receptor-positive, HER2-negative advanced or metastatic breast cancer. Clin. Cancer Res.24, 2999–3004 (2018). [DOI] [PubMed] [Google Scholar]
- 511.Royce, M. et al. FDA Approval Summary: Abemaciclib With Endocrine Therapy for High-Risk Early Breast Cancer. J. Clin. Oncol.40, 1155–1162 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 512.Limousin, W. et al. Molecular-based targeted therapies in patients with hepatocellular carcinoma and hepato-cholangiocarcinoma refractory to atezolizumab/bevacizumab. J. Hepatol.79, 1450–1458 (2023). [DOI] [PubMed] [Google Scholar]
- 513.Wang, H., Liao, P., Zeng, S. X. & Lu, H. Co-targeting p53-R249S and CDK4 synergistically suppresses survival of hepatocellular carcinoma cells. Cancer Biol. Ther.21, 269–277 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 514.Digiacomo, G. et al. CDK4/6 inhibitors improve the anti-tumor efficacy of lenvatinib in hepatocarcinoma cells. Front. Oncol. 12, 942341 (2022). [DOI] [PMC free article] [PubMed]
- 515.Moeini, A. et al. An Immune Gene Expression Signature Associated With Development of Human Hepatocellular Carcinoma Identifies Mice That Respond to Chemopreventive Agents. Gastroenterology157, 1383–1397.e11 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 516.Cadoux, M. et al. Expression of NKG2D ligands is downregulated by β-catenin signalling and associates with HCC aggressiveness. J. Hepatol.74, 1386–1397 (2021). [DOI] [PubMed] [Google Scholar]
- 517.de Galarreta, M. R. et al. β-catenin activation promotes immune escape and resistance to anti–PD-1 therapy in hepatocellular carcinoma. Cancer Discov9, 1124–1141 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 518.Montironi, C. et al. Inflamed and non-inflamed classes of HCC: A revised immunogenomic classification. Gut72, 129–140 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 519.Long, J. et al. Development and validation of a TP53-associated immune prognostic model for hepatocellular carcinoma. EBioMedicine42, 363–374 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 520.Shen, J. et al. ARID1A deficiency promotes mutability and potentiates therapeutic antitumor immunity unleashed by immune checkpoint blockade. Nat. Med.24, 556–562 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 521.Zhou, J. et al. Hepatoma-intrinsic CCRK inhibition diminishes myeloid-derived suppressor cell immunosuppression and enhances immune-checkpoint blockade efficacy. Gut67, 931–944 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 522.Chiang, D. Y. et al. Focal gains of VEGFA and molecular classification of hepatocellular carcinoma. Cancer Res.68, 6779–6788 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 523.Huinen, Z. R., Huijbers, E. J. M., van Beijnum, J. R., Nowak-Sliwinska, P. & Griffioen, A. W. Anti-angiogenic agents — overcoming tumour endothelial cell anergy and improving immunotherapy outcomes. Nat. Rev. Clin. Oncol.18, 527–540 (2021). [DOI] [PubMed] [Google Scholar]
- 524.Sun, X. et al. MiR-146a is directly regulated by STAT3 in human hepatocellular carcinoma cells and involved in anti-tumor immune suppression. Cell Cycle14, 243–252 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 525.Wang, Y., Shen, Y., Wang, S., Shen, Q. & Zhou, X. The role of STAT3 in leading the crosstalk between human cancers and the immune system. Cancer Lett.415, 117–128 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 526.Butte, M. J., Keir, M. E., Phamduy, T. B., Sharpe, A. H. & Freeman, G. J. Programmed Death-1 Ligand 1 Interacts Specifically with the B7-1 Costimulatory Molecule to Inhibit T Cell Responses. Immunity27, 111–122 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 527.Kinter, A. L. et al. The Common γ-Chain Cytokines IL-2, IL-7, IL-15, and IL-21 Induce the Expression of Programmed Death-1 and Its Ligands. J. Immunol.181, 6738–6746 (2008). [DOI] [PubMed] [Google Scholar]
- 528.Kulpa, D. A. et al. PD-1 coinhibitory signals: The link between pathogenesis and protection. Semin. Immunol.25, 219–227 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 529.Keir, M. E., Butte, M. J., Freeman, G. J. & Sharpe, A. H. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol.26, 677–704 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 530.Topalian, S. L., Taube, J. M., Anders, R. A. & Pardoll, D. M. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat. Rev. Cancer16, 275–287 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 531.Tang, H. et al. PD-L1 on host cells is essential for PD-L1 blockade– mediated tumor regression. J. Clin. Invest.128, 580–588 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 532.Zhou, D., Luan, J., Huang, C. & Li, J. Tumor-associated macrophages in hepatocellular carcinoma: Friend or foe? Gut Liver15, 500–516 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 533.Park, D. J. et al. Preferential expression of programmed death ligand 1 protein in tumor-associated macrophages and its potential role in immunotherapy for hepatocellular carcinoma. Int. J. Mol. Sci. 22, 4710 (2021). [DOI] [PMC free article] [PubMed]
- 534.Sharpe, A. H. & Pauken, K. E. The diverse functions of the PD1 inhibitory pathway. Nat. Rev. Immunol.18, 153–167 (2018). [DOI] [PubMed] [Google Scholar]
- 535.Li, B. et al. Anti–PD-1/PD-L1 Blockade Immunotherapy Employed in Treating Hepatitis B Virus Infection–Related Advanced Hepatocellular Carcinoma: A Literature Review. Front. Immunol. 11, 1037 (2020). [DOI] [PMC free article] [PubMed]
- 536.Daassi, D., Mahoney, K. M. & Freeman, G. J. The importance of exosomal PDL1 in tumour immune evasion. Nat. Rev. Immunol.20, 209–215 (2020). [DOI] [PubMed] [Google Scholar]
- 537.Cai, J., Wang, D., Zhang, G. & Guo, X. The role of PD-1/PD-L1 axis in treg development and function: Implications for cancer immunotherapy. Onco. Targets. Ther.12, 8437–8445 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 538.Xiao, X. et al. PD-1hi identifi es a novel regulatory b-cell population in human hepatoma that promotes disease progression. Cancer Discov.6, 546–559 (2016). [DOI] [PubMed] [Google Scholar]
- 539.Nam, S., Lee, A., Lim, J. & Lim, J. S. Analysis of the expression and regulation of PD-1 protein on the surface of myeloid-derived suppressor cells (MDSCs). Biomol. Ther.27, 63–70 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 540.Lu, C., Redd, P. S., Lee, J. R., Savage, N. & Liu, K. The expression profiles and regulation of PD-L1 in tumor-induced myeloid-derived suppressor cells. Oncoimmunology5, e1247135 (2016). [DOI] [PMC free article] [PubMed]
- 541.Liu, M. et al. Myeloid-derived suppressor cells regulate the immunosuppressive functions of PD-1-PD-L1+ Bregs through PD-L1/PI3K/AKT/NF-κB axis in breast cancer. Cell Death Dis. 12, 362 (2021). [DOI] [PMC free article] [PubMed]
- 542.Li, Q., Han, J., Yang, Y. & Chen, Y. PD-1/PD-L1 checkpoint inhibitors in advanced hepatocellular carcinoma immunotherapy. Front. Immunol. 13, 1070961 (2022). [DOI] [PMC free article] [PubMed]
- 543.Kuzume, A., Chi, S., Yamauchi, N. & Minami, Y. Immune-checkpoint blockade therapy in lymphoma. Int. J. Mol. Sci.21, 1–15 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 544.Kudo, M. Targeted and immune therapies for hepatocellular carcinoma: Predictions for 2019 and beyond. World J. Gastroenterol.25, 789–807 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 545.Hartley, G. P., Chow, L., Ammons, D. T., Wheat, W. H. & Dow, S. W. Programmedcell death ligand 1 (PD-L1) signaling regulates macrophage proliferation and activation. Cancer Immunol. Res.6, 1260–1273 (2018). [DOI] [PubMed] [Google Scholar]
- 546.El-Khoueiry, A. B. et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet389, 2492–2502 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 547.Yau, T. et al. Nivolumab versus sorafenib in advanced hepatocellular carcinoma (CheckMate 459): a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol.23, 77–90 (2022). [DOI] [PubMed] [Google Scholar]
- 548.Zhu, A. X. et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol.19, 940–952 (2018). [DOI] [PubMed] [Google Scholar]
- 549.Qin, S. et al. Camrelizumab in patients with previously treated advanced hepatocellular carcinoma: a multicentre, open-label, parallel-group, randomised, phase 2 trial. Lancet Oncol.21, 571–580 (2020). [DOI] [PubMed] [Google Scholar]
- 550.Wainberg, Z. A. et al. Safety and clinical activity of durvalumab monotherapy in patients with hepatocellular carcinoma (HCC). J. Clin. Oncol.35, 4071–4071 (2017). [Google Scholar]
- 551.Abou-Alfa, G. K. et al. Phase 3 randomized, open-label, multicenter study of tremelimumab (T) and durvalumab (D) as first-line therapy in patients (pts) with unresectable hepatocellular carcinoma (uHCC): HIMALAYA. J. Clin. Oncol.40, 379–379 (2022). [Google Scholar]
- 552.Vignali, D. A. A., Collison, L. W. & Workman, C. J. How regulatory T cells work. Nat. Rev. Immunol.8, 523–532 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 553.Wing, K. et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science322, 271–275 (2008). [DOI] [PubMed] [Google Scholar]
- 554.Grosso, J. F. & Jure-Kunkel, M. N. CTLA-4 blockade in tumor models: An overview of preclinical and translational research. Cancer Immun. 13, 5 (2013). [PMC free article] [PubMed]
- 555.Sangro, B. et al. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J. Hepatol.59, 81–88 (2013). [DOI] [PubMed] [Google Scholar]
- 556.Finkelmeier, F., Waidmann, O. & Trojan, J. Nivolumab for the treatment of hepatocellular carcinoma. Expert Rev. Anticancer Ther18, 1169–1175 (2018). [DOI] [PubMed] [Google Scholar]
- 557.Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol.19, 55–71 (2021). [DOI] [PubMed] [Google Scholar]
- 558.Hsu, C. L. & Schnabl, B. The gut–liver axis and gut microbiota in health and liver disease. Nat. Rev. Microbiol.21, 719–733 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 559.Huo, R. et al. Altered Gut Microbiota Composition and Its Potential Association in Patients with Advanced Hepatocellular Carcinoma. Curr. Oncol.30, 1818–1830 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 560.Peng, Y. C. et al. Specific gut microbiome signature predicts hepatitis B virus-related hepatocellular carcinoma patients with microvascular invasion. Ann. Med. 55, 2283160 (2023). [DOI] [PMC free article] [PubMed]
- 561.Yu, J. et al. Bifidobacterium longum promotes postoperative liver function recovery in patients with hepatocellular carcinoma. Cell Host Microbe32, 131–144.e6 (2024). [DOI] [PubMed] [Google Scholar]
- 562.Dapito, D. H. et al. Promotion of Hepatocellular Carcinoma by the Intestinal Microbiota and TLR4. Cancer Cell21, 504–516 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 563.Golonka, R. M. et al. Loss of toll-like receptor 5 potentiates spontaneous hepatocarcinogenesis in farnesoid X receptor-deficient mice. Hepatol. Commun. 7, e0166 (2023). [DOI] [PMC free article] [PubMed]
- 564.Rajapakse, J. et al. Unveiling the complex relationship between gut microbiota and liver cancer: opportunities for novel therapeutic interventions. Gut Microbes15, 2240031 (2023). [DOI] [PMC free article] [PubMed]
- 565.Schöler, D. & Schnabl, B. The role of the microbiome in liver disease. Curr. Opin. Gastroenterol.40, 134–142 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 566.Ma, C. et al. Gut microbiome–mediated bile acid metabolism regulates liver cancer via NKT cells. Science. 360, eaan5931 (2018). [DOI] [PMC free article] [PubMed]
- 567.Liu, T., Guo, Y., Liao, Y. & Liu, J. Mechanism-guided fine-tuned microbiome potentiates anti-tumor immunity in HCC. Front. Immunol. 14, 1333864 (2023). [DOI] [PMC free article] [PubMed]
- 568.Pallozzi, M. et al. Non-Invasive Biomarkers for Immunotherapy in Patients with Hepatocellular Carcinoma: Current Knowledge and Future Perspectives. Cancers14, 4631 (2022). [DOI] [PMC free article] [PubMed]
- 569.Das, B. K. Altered gut microbiota in hepatocellular carcinoma: Insights into the pathogenic mechanism and preclinical to clinical findings. Apmis130, 719–740 (2022). [DOI] [PubMed] [Google Scholar]
- 570.Yamada, S. et al. Bile acid metabolism regulated by the gut microbiota promotes non-alcoholic steatohepatitis-associated hepatocellular carcinoma in mice. Oncotarget9, 9925–9939 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 571.Yu, L. X. et al. Endotoxin Accumulation Prevents Carcinogen-Induced Apoptosis and Promotes Liver Tumorigenesis in Rodents. Hepatology52, 1322–1333 (2010). [DOI] [PubMed] [Google Scholar]
- 572.Song, Q. et al. Bifidobacterium pseudolongum-generated acetate suppresses non-alcoholic fatty liver disease-associated hepatocellular carcinoma. J. Hepatol.79, 1352–1365 (2023). [DOI] [PubMed] [Google Scholar]
- 573.Chen, S. et al. Lactobacillus brevis alleviates the progress of hepatocellular carcinoma and type 2 diabetes in mice model via interplay of gut microflora, bile acid and NOTCH 1 signaling. Front. Immunol. 14, 1179014 (2023). [DOI] [PMC free article] [PubMed]
- 574.Zhang, X. et al. Dietary cholesterol drives fatty liver-associated liver cancer by modulating gut microbiota and metabolites. Gut70, 761–774 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 575.Ponziani, F. R. et al. Gut Dysbiosis and Fecal Calprotectin Predict Response to Immune Checkpoint Inhibitors in Patients With Hepatocellular Carcinoma. Hepatol. Commun.6, 1492–1501 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 576.Li, L., Ye, J. & Liu, M. Characterization of gut microbiota in patients with primary hepatocellular carcinoma received immune checkpoint inhibitors: A Chinese population-based study. Medicine99, E21788 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 577.Alshammari, K. et al. Antibiotic Exposure Concurrently with Anti-PD1 Blockade Therapy Reduces Overall Survival in Patients with Child–Pugh Class A Advanced Hepatocellular Carcinoma. Cancers. 16, 133 (2024). [DOI] [PMC free article] [PubMed]
- 578.Pinato, D. J. et al. Association between antibiotics and adverse oncological outcomes in patients receiving targeted or immune-based therapy for hepatocellular carcinoma. JHEP Rep.5, 100747(2023). [DOI] [PMC free article] [PubMed]
- 579.Spahn, S. et al. Clinical and genetic tumor characteristics of responding and non-responding patients to pd-1 inhibition in hepatocellular carcinoma. Cancers12, 1–17 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 580.Che, Y. et al. Gut microbial metabolite butyrate improves anticancer therapy by regulating intracellular calcium homeostasis. Hepatology78, 88–102 (2023). [DOI] [PubMed] [Google Scholar]
- 581.Dudek, M. et al. Auto-aggressive CXCR6 + CD8 T cells cause liver immune pathology in NASH. Nature592, 444–449 (2021). [DOI] [PubMed] [Google Scholar]
- 582.Pfister, D. et al. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature592, 450–456 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 583.Roberts, L. R. Hepatocellular carcinoma. Pract. Gastroenterol. Hepatol. Liver Biliary Dis. 144–151, 10.7863/jum.2003.22.9.887 (2010).
- 584.Qian, Z. et al. HBV integrations reshaping genomic structures promote hepatocellular carcinoma. Gut, 10.1136/gutjnl-2023-330414 (2024). [DOI] [PMC free article] [PubMed]
- 585.Chen, L. et al. Deep whole-genome analysis of 494 hepatocellular carcinomas. Nature627, 586–593 (2024). [DOI] [PubMed] [Google Scholar]
- 586.Zoulim, F., Chen, P.-J., Dandri, M., Kennedy, P. & Seeger, C. Hepatitis B Virus DNA integration: Implications for diagnostics, therapy, and outcome. J. Hepatol. 10.1016/j.jhep.2024.06.037 (2024). [DOI] [PubMed]
- 587.Maini, M. K. & Pallett, L. J. Defective T-cell immunity in hepatitis B virus infection: why therapeutic vaccination needs a helping hand. Lancet Gastroenterol. Hepatol.3, 192–202 (2018). [DOI] [PubMed] [Google Scholar]
- 588.Lim, C. J. et al. Multidimensional analyses reveal distinct immune microenvironment in hepatitis B virus-related hepatocellular carcinoma. Gut68, 916–927 (2019). [DOI] [PubMed] [Google Scholar]
- 589.Hou, J., Zhang, H., Sun, B. & Karin, M. The immunobiology of hepatocellular carcinoma in humans and mice: Basic concepts and therapeutic implications. J. Hepatol.72, 167–182 (2020). [DOI] [PubMed] [Google Scholar]
- 590.Cabrera, R. et al. An immunomodulatory role for CD4 + CD25+regulatory T lymphocytes in hepatitis C virus infection. Hepatology40, 1062–1071 (2004). [DOI] [PubMed] [Google Scholar]
- 591.Abel, M. et al. Intrahepatic virus-specific IL-10-producing CD8 T cells prevent liver damage during chronic hepatitis C virus infection. Hepatology44, 1607–1616 (2006). [DOI] [PubMed] [Google Scholar]
- 592.Schmidt, N. M. et al. Targeting human Acyl-CoA:cholesterol acyltransferase as a dual viral and T cell metabolic checkpoint. Nat. Commun. 12, 2814 (2021). [DOI] [PMC free article] [PubMed]
- 593.Datta, K. & Kulkarni, A. P. Oxidative metabolism of aflatoxin B1 by lipoxygenase purified from human term placenta and intrauterine conceptal tissues. Teratology50, 311–317 (1994). [DOI] [PubMed] [Google Scholar]
- 594.Williams, J. H. et al. Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences, and interventions. Am. J. Clin. Nutr.80, 1106–1122 (2004). [DOI] [PubMed] [Google Scholar]
- 595.Zhang, W. et al. Genetic Features of Aflatoxin-Associated Hepatocellular Carcinoma. Gastroenterology153, 249–262.e2 (2017). [DOI] [PubMed] [Google Scholar]
- 596.Hong, S. W. & Park, C. The Effect of Aflatoxin B1 on the Expression of Early Response Genes and Transforming Growth Factor-α in CCl4 Induced Rat Liver Injury. Yonsei Med. J.38, 167–177 (1997). [DOI] [PubMed] [Google Scholar]
- 597.Shen, H. M. & Ong, C. N. Mutations of the p53 tumor suppressor gene and ras oncogenes in aflatoxin hepatocarcinogenesis. Mutat. Res. Rev. Genet. Toxicol.366, 23–44 (1996). [DOI] [PubMed] [Google Scholar]
- 598.Hussain, S. P., Schwank, J., Staib, F., Wang, X. W. & Harris, C. C. TP53 mutations and hepatocellular carcinoma: Insights into the etiology and pathogenesis of liver cancer. Oncogene26, 2166–2176 (2007). [DOI] [PubMed] [Google Scholar]
- 599.Zhu, Q. et al. AHR mediates the aflatoxin B1 toxicity associated with hepatocellular carcinoma. Signal Transduct. Target. Ther. 6, 299 (2021). [DOI] [PMC free article] [PubMed]
- 600.Jin, J., Kouznetsova, V. L., Kesari, S. & Tsigelny, I. F. Synergism in actions of HBV with aflatoxin in cancer development. Toxicology499, 153652 (2023). [DOI] [PubMed]
- 601.Li, M. et al. Spatial proteomics of immune microenvironment in nonalcoholic steatohepatitis-Associated hepatocellular carcinoma. Hepatology79, 560–574 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 602.Gomes, A. L. et al. Metabolic Inflammation-Associated IL-17A Causes Non-alcoholic Steatohepatitis and Hepatocellular Carcinoma. Cancer Cell30, 161–175 (2016). [DOI] [PubMed] [Google Scholar]
- 603.Malehmir, M. et al. Platelet GPIbα is a mediator and potential interventional target for NASH and subsequent liver cancer. Nat. Med.25, 641–655 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 604.Ganne-Carrié, N. & Nahon, P. Hepatocellular carcinoma in the setting of alcohol-related liver disease. J. Hepatol.70, 284–293 (2019). [DOI] [PubMed] [Google Scholar]
- 605.Parlesak, A., Schäfer, C., Schütz, T., Bode, J. C. & Bode, C. Increased intestinal permeability to macromolecules and endotoxemia in patients with chronic alcohol abuse in different stages of alcohol-induced liver disease. J. Hepatol.32, 742–747 (2000). [DOI] [PubMed] [Google Scholar]
- 606.Gao, M. et al. Granulocytic myeloid-derived suppressor cell population increases with the severity of alcoholic liver disease. J. Cell. Mol. Med.23, 2032–2041 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 607.Ma, H. Y. et al. IL-17 signaling in steatotic hepatocytes and macrophages promotes hepatocellular carcinoma in alcohol-related liver disease. J. Hepatol.72, 946–959 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 608.Tilg, H., Moschen, A. R. & Szabo, G. Interleukin-1 and inflammasomes in alcoholic liver disease/acute alcoholic hepatitis and nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology64, 955–965 (2016). [DOI] [PubMed] [Google Scholar]
- 609.Ahn, J. C. et al. Detection of Circulating Tumor Cells and Their Implications as a Biomarker for Diagnosis, Prognostication, and Therapeutic Monitoring in Hepatocellular Carcinoma. Hepatology73, 422–436 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 610.Xu, R. H. et al. Circulating tumour DNA methylation markers for diagnosis and prognosis of hepatocellular carcinoma. Nat. Mater.16, 1155–1162 (2017). [DOI] [PubMed] [Google Scholar]
- 611.Foda, Z. H. et al. Detecting Liver Cancer Using Cell-Free DNA Fragmentomes. Cancer Discov13, 616–631 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 612.Feng, Z. et al. CT radiomics to predict macrotrabecular-massive subtype and immune status in hepatocellular carcinoma. Radiology307, e221291 (2023). [DOI] [PubMed]
- 613.Chatzipanagiotou, O. P. et al. Artificial intelligence in hepatocellular carcinoma diagnosis: a comprehensive review of current literature. J. Gastroenterol. Hepatol. 10.1111/jgh.16663 (2024). [DOI] [PubMed]
- 614.Gao, Q. et al. Dual transformation therapy for giant hepatocellular carcinoma: Two case reports and review of literature. World J. Gastrointest. Surg.15, 2089–2097 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 615.Qu, W. F. et al. Conversion therapy for initially unresectable hepatocellular carcinoma using a combination of toripalimab, lenvatinib plus TACE: Real-world study. BJS Open6, zrac114 (2022). [DOI] [PMC free article] [PubMed]
- 616.Tomimaru, Y., Aihara, A., Wands, J. R., Aloman, C. & Kim, M. A Novel Drug, CC-122, Inhibits Tumor Growth in Hepatocellular Carcinoma through Downregulation of an Oncogenic TCF-4 Isoform. Transl. Oncol.12, 1345–1356 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 617.Murali, M. et al. Antibody–drug conjugate as targeted therapeutics against hepatocellular carcinoma: preclinical studies and clinical relevance. Clin. Transl. Oncol.24, 407–431 (2022). [DOI] [PubMed] [Google Scholar]
- 618.Fu, Y. et al. Glypican-3-Specific Antibody Drug Conjugates Targeting Hepatocellular Carcinoma. Hepatology70, 563–576 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 619.Pang, N. et al. IL-7 and CCL19-secreting CAR-T cell therapy for tumors with positive glypican-3 or mesothelin. J. Hematol. Oncol. 14, 118 (2021). [DOI] [PMC free article] [PubMed]
- 620.Zhang, R. Y. et al. Doxycycline Inducible Chimeric Antigen Receptor T Cells Targeting CD147 for Hepatocellular Carcinoma Therapy. Front. Cell Dev. Biol. 7, 233 (2019). [DOI] [PMC free article] [PubMed]
- 621.Dai, H. et al. Efficacy and biomarker analysis of CD133-directed CAR T cells in advanced hepatocellular carcinoma: a single-arm, open-label, phase II trial. Oncoimmunology9, 1846926 (2020). [DOI] [PMC free article] [PubMed]
- 622.Meng, F. et al. Immunotherapy of HBV-related advanced hepatocellular carcinoma with short-term HBV-specific TCR expressed T cells: results of dose escalation, phase I trial. Hepatol. Int.15, 1402–1412 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 623.Tagliamonte, M. et al. Potentiating cancer vaccine efficacy in liver cancer. Oncoimmunology7, e1488564 (2018). [DOI] [PMC free article] [PubMed]
- 624.Yarchoan, M. et al. Personalized neoantigen vaccine and pembrolizumab in advanced hepatocellular carcinoma: a phase 1/2 trial. Nat. Med.30, 1044–1053 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 625.Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 74, 229–263 (2024). [DOI] [PubMed]
- 626.Chien, S. C. et al. Higher Risk of Tumor Recurrence in NASH-Related Hepatocellular Carcinoma Following Curative Resection. Viruses14, 2427 (2022). [DOI] [PMC free article] [PubMed]
- 627.Brar, G. et al. Hepatocellular Carcinoma Survival by Etiology: A SEER-Medicare Database Analysis. Hepatol. Commun.4, 1541–1551 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 628.Huang, D. Q. et al. Changing global epidemiology of liver cancer from 2010 to 2019: NASH is the fastest growing cause of liver cancer. Cell Metab.34, 969–977.e2 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 629.Rumgay, H. et al. Global burden of primary liver cancer in 2020 and predictions to 2040. J. Hepatol.77, 1598–1606 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 630.Tan, D. J. H. et al. Global burden of liver cancer in males and females: Changing etiological basis and the growing contribution of NASH. Hepatology77, 1150–1163 (2023). [DOI] [PubMed] [Google Scholar]
- 631.Shimose, S. et al. The beneficial impact of metabolic dysfunction-associated fatty liver disease on lenvatinib treatment in patients with non-viral hepatocellular carcinoma. Hepatol. Res.53, 104–115 (2023). [DOI] [PubMed] [Google Scholar]
- 632.Xiong, Y., Zhang, Y. & Hu, C. Radiofrequency ablation versus microwave ablation for hepatocellular carcinoma with cirrhosis: a propensity score analysis. Transl. Cancer Res.13, 1807–1820 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 633.Abou-Alfa, G. K. et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med.379, 54–63 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 634.Bruix, J. et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet389, 56–66 (2017). [DOI] [PubMed] [Google Scholar]
- 635.Llovet, J. M. et al. Sorafenib in Advanced Hepatocellular Carcinoma. N. Engl. J. Med.359, 378–390 (2008). [DOI] [PubMed] [Google Scholar]
- 636.Kudo, M. et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet391, 1163–1173 (2018). [DOI] [PubMed] [Google Scholar]
- 637.Cheng, A. L. et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol.10, 25–34 (2009). [DOI] [PubMed] [Google Scholar]
- 638.Yau, T. et al. Efficacy and Safety of Nivolumab plus Ipilimumab in Patients with Advanced Hepatocellular Carcinoma Previously Treated with Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 6, e204564 (2020). [DOI] [PMC free article] [PubMed]
- 639.Doroshow, D. B. et al. PD-L1 as a biomarker of response to immune-checkpoint inhibitors. Nat. Rev. Clin. Oncol.18, 345–362 (2021). [DOI] [PubMed] [Google Scholar]
- 640.McGranahan, N. & Swanton, C. Clonal Heterogeneity and Tumor Evolution: Past, Present, and the Future. Cell168, 613–628 (2017). [DOI] [PubMed] [Google Scholar]
- 641.Fisher, R., Pusztai, L. & Swanton, C. Cancer heterogeneity: Implications for targeted therapeutics. Br. J. Cancer108, 479–485 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 642.Zhou, C. et al. Lenvatinib Induces Immunogenic Cell Death and Triggers Toll-Like Receptor-3/4 Ligands in Hepatocellular Carcinoma. J. Hepatocell. Carcinoma10, 697–712 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 643.Zhu, A. X. et al. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): A randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol.16, 859–870 (2015). [DOI] [PubMed] [Google Scholar]
- 644.Zhu, A. X. et al. Biomarker analyses of clinical outcomes in patients with advanced hepatocellular carcinoma treated with sorafenib with or without erlotinib in the SEARCH trial. Clin. Cancer Res.22, 4870–4879 (2016). [DOI] [PubMed] [Google Scholar]
- 645.Qin, S. et al. Apatinib as second-line or later therapy in patients with advanced hepatocellular carcinoma (AHELP): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Gastroenterol. Hepatol.6, 559–568 (2021). [DOI] [PubMed] [Google Scholar]
- 646.Zhu, A. X. et al. Effect of everolimus on survival in advanced hepatocellular carcinoma after failure of sorafenib: The EVOLVE-1 randomized clinical trial. Jama312, 57–67 (2014). [DOI] [PubMed] [Google Scholar]
- 647.Lau, D. K. et al. Phase II study of everolimus (RAD001) monotherapy as first-line treatment in advanced biliary tract cancer with biomarker exploration: The RADiChol Study. Br. J. Cancer118, 966–971 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 648.Shiah, H. S. et al. Randomised clinical trial: Comparison of two everolimus dosing schedules in patients with advanced hepatocellular carcinoma. Aliment. Pharmacol. Ther.37, 62–73 (2013). [DOI] [PubMed] [Google Scholar]
- 649.Yeo, W. et al. Phase I/II study of temsirolimus for patients with unresectable Hepatocellular Carcinoma (HCC)- a correlative study to explore potential biomarkers for response. BMC Cancer15, 395 (2015). [DOI] [PMC free article] [PubMed]
- 650.Okusaka, T. et al. Phase 1 and pharmacological trial of OPB-31121, a signal transducer and activator of transcription-3 inhibitor, in patients with advanced hepatocellular carcinoma. Hepatol. Res.45, 1283–1291 (2015). [DOI] [PubMed] [Google Scholar]
- 651.Okusaka, T. et al. A Phase I Study to Investigate the Safety, Tolerability and Pharmacokinetics of Napabucasin Combined with Sorafenib in Japanese Patients with Unresectable Hepatocellular Carcinoma. Drugs R D23, 99–107 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 652.Yeo, W. et al. Epigenetic therapy using belinostat for patients with unresectable hepatocellular carcinoma: A multicenter phase I/II study with biomarker and pharmacokinetic analysis of tumors from patients in the mayo phase II consortium and the cancer therapeutics res. J. Clin. Oncol.30, 3361–3367 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 653.Villanueva, L., Álvarez-Errico, D. & Esteller, M. The Contribution of Epigenetics to Cancer Immunotherapy. Trends Immunol.41, 676–691 (2020). [DOI] [PubMed] [Google Scholar]
- 654.Sanceau, J. & Gougelet, A. Epigenetic mechanisms of liver tumor resistance to immunotherapy. World J. Hepatol.13, 979–1002 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 655.Song, L. et al. Isl induces apoptosis and autophagy in hepatocellular carcinoma via downregulation of pi3k/akt/mtor pathway in vivo and in vitro. Drug Des. Devel. Ther.14, 4363–4376 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 656.Chen, W. et al. Fenofibrate suppresses the progression of hepatoma by downregulating osteopontin through inhibiting the PI3K/AKT/Twist pathway. Naunyn. Schmiedebergs. Arch. Pharmacol.397, 1025–1035 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 657.Bu, L. et al. High-fat diet promotes liver tumorigenesis via palmitoylation and activation of AKT. Gut.73, 1156–1168 (2024). [DOI] [PubMed] [Google Scholar]
- 658.Huynh, H. et al. RAD001 (everolimus) inhibits tumour growth in xenograft models of human hepatocellular carcinoma. J. Cell. Mol. Med.13, 1371–1380 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 659.Li, Y. et al. Combination of metformin and sorafenib suppresses proliferation and induces autophagy of hepatocellular carcinoma via targeting the mTOR pathway. Int. J. Oncol.50, 297–309 (2017). [DOI] [PubMed] [Google Scholar]
- 660.Varshney, A. et al. Targeted delivery of microRNA-199a-3p using self-assembled dipeptide nanoparticles efficiently reduces hepatocellular carcinoma in mice. Hepatology67, 1392–1407 (2018). [DOI] [PubMed] [Google Scholar]
- 661.Li, D. et al. MicroRNA-99a inhibits hepatocellular carcinoma growth and correlates with prognosis of patients with hepatocellular carcinoma. J. Biol. Chem.286, 36677–36685 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 662.Zhao, Z. et al. Oxysophocarpine suppresses FGFR1-overexpressed hepatocellular carcinoma growth and sensitizes the therapeutic effect of lenvatinib. Life Sci.264, 118642 (2021). [DOI] [PubMed] [Google Scholar]
- 663.Liu, J. et al. Indole hydrazide compound ZJQ-24 inhibits angiogenesis and induces apoptosis cell death through abrogation of AKT/mTOR pathway in hepatocellular carcinoma. Cell Death Dis.11, 926 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 664.Zhang, S. et al. Exploring the molecular mechanism of Artemisia rupestris L. for the treatment of hepatocellular carcinoma via PI3K/AKT pathway. J. Ethnopharmacol.322, 117572 (2024). [DOI] [PubMed] [Google Scholar]
- 665.Zhou, T. T. et al. A network pharmacology integrated serum pharmacochemistry strategy for uncovering efficacy of YXC on hepatocellular carcinoma. J. Ethnopharmacol.319, 117125 (2024). [DOI] [PubMed] [Google Scholar]
- 666.Ong, L. C. et al. Effective inhibition of xenografts of hepatocellular carcinoma (hepg2) by rapamycin and bevacizumab in an intrahepatic model. Mol. Imaging Biol.11, 334–342 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 667.Abdel-Bakky, M. S. et al. Targeting the PI3K/pAKT/mTOR/NF-κB/FOXO3a signaling pathway for suppressing the development of hepatocellular carcinoma in rats: Role of the natural remedic Suaeda vermiculata forssk. Environ. Toxicol.39, 3666–3678 (2024). [DOI] [PubMed] [Google Scholar]
- 668.Tian, D., Shi, Y., Chen, D., Liu, Q. & Fan, F. The Wnt inhibitor LGK-974 enhances radiosensitivity of HepG2 cells by modulating Nrf2 signaling. Int. J. Oncol.51, 545–554 (2017). [DOI] [PubMed] [Google Scholar]
- 669.Zhou, Z. et al. Targeting β-catenin and PD-L1 simultaneously by a racemic supramolecular peptide for the potent immunotherapy of hepatocellular carcinoma. Theranostics13, 3371–3386 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 670.Li, C. L. et al. The Natural Compound Dehydrocrenatidine Attenuates Nicotine-Induced Stemness and Epithelial-Mesenchymal Transition in Hepatocellular Carcinoma by Regulating a7nAChR-Jak2 Signaling Pathways. Dis. Markers2022, 8316335 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 671.Mohan, C. D. et al. Development of a novel azaspirane that targets the Janus Kinase-signal transducer and activator of transcription (STAT) pathway in hepatocellular carcinoma in vitro and in vivo. J. Biol. Chem.289, 34296–34307 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 672.Jung, Y. Y. et al. Leonurine ameliorates the STAT3 pathway through the upregulation of SHP-1 to retard the growth of hepatocellular carcinoma cells. Cell. Signal.114, 111003 (2024). [DOI] [PubMed] [Google Scholar]
- 673.Song, L., Guo, L. & Li, Z. Molecular mechanisms of 3,3′4,4′,5-pentachlorobiphenyl-induced epithelial-mesenchymal transition in human hepatocellular carcinoma cells. Toxicol. Appl. Pharmacol.322, 75–88 (2017). [DOI] [PubMed] [Google Scholar]
- 674.Xu, G. et al. Stattic Enhances Radiosensitivity and Reduces Radio-Induced Migration and Invasion in HCC Cell Lines through an Apoptosis Pathway. Biomed. Res. Int.2017, 1832494 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 675.Zhao, Z. et al. Peri-tumor fibroblasts promote tumorigenesis and metastasis of hepatocellular carcinoma via Interleukin6/STAT3 signaling pathway. Cancer Manag. Res.11, 2889–2901 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 676.Han, Z. et al. Inhibition of murine hepatoma tumor growth by cryptotanshinone involves TLR7-dependent activation of macrophages and induction of adaptive antitumor immune defenses. Cancer Immunol. Immunother.68, 1073–1085 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 677.Li, Y., Han, Q., Zhao, H., Guo, Q. & Zhang, J. Napabucasin Reduces Cancer Stem Cell Characteristics in Hepatocellular Carcinoma. Front. Pharmacol.11, 597520 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 678.Toh, T. B., Lim, J. J., Hooi, L., Rashid, M. B. M. A. & Chow, E. K. H. Targeting Jak/Stat pathway as a therapeutic strategy against SP/CD44+ tumorigenic cells in Akt/β-catenin-driven hepatocellular carcinoma. J. Hepatol.72, 104–118 (2020). [DOI] [PubMed] [Google Scholar]
- 679.Shekh, R. et al. Ethanolic extract of Coleus aromaticus leaves impedes the proliferation and instigates apoptotic cell death in liver cancer HepG2 cells through repressing JAK/STAT cascade. J. Food Biochem.46, e14368 (2022). [DOI] [PubMed] [Google Scholar]
- 680.Liao, J. et al. Nitidine chloride inhibits hepatocellular carcinoma cell growth in vivo through the suppression of the JAK1/STAT3 signaling pathway. Int. J. Mol. Med.32, 79–84 (2013). [DOI] [PubMed] [Google Scholar]
- 681.Xu, J. et al. Etomidate elicits anti-tumor capacity by disrupting the JAK2/STAT3 signaling pathway in hepatocellular carcinoma. Cancer Lett.552, 215970 (2023). [DOI] [PubMed] [Google Scholar]
- 682.Zhu, M. et al. Cantharidin treatment inhibits hepatocellular carcinoma development by regulating the JAK2/STAT3 and PI3K/Akt pathways in an EphB4-dependent manner. Pharmacol. Res.158, 104868 (2020). [DOI] [PubMed] [Google Scholar]
- 683.Ni, J. S. et al. miR-515–5p suppresses HCC migration and invasion via targeting IL6/JAK/STAT3 pathway. Surg. Oncol.34, 113–120 (2020). [DOI] [PubMed] [Google Scholar]
- 684.Arzumanyan, A. et al. Hedgehog signaling blockade delays hepatocarcinogenesis induced by hepatitis B virus X protein. Cancer Res.72, 5912–5920 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 685.Jeng, K. S. et al. Sonic hedgehog pathway inhibitor mitigates mouse hepatocellular carcinoma. Am. J. Surg.210, 554–560 (2015). [DOI] [PubMed] [Google Scholar]
- 686.Mohammed, O. A. et al. Itraconazole halts hepatocellular carcinoma progression by modulating sonic hedgehog signaling in rats: A novel therapeutic approach. Pathol. Res. Pract.253, 155086 (2024). [DOI] [PubMed] [Google Scholar]
- 687.Di Fazio, P. et al. The pan-deacetylase inhibitor panobinostat inhibits growth of hepatocellular carcinoma models by alternative pathways of apoptosis. Cell. Oncol.32, 285–300 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 688.Lachenmayer, A. et al. Combination therapy for hepatocellular carcinoma: Additive preclinical efficacy of the HDAC inhibitor panobinostat with sorafenib. J. Hepatol.56, 1343–1350 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 689.Sanaei, M., Kavoosi, F. & Esmi, Z. The effect of 5-aza-2′-deoxycytidine in combination to and in comparison with vorinostat on dna methyltransferases, histone deacetylase 1, glutathione s-transferase 1 and suppressor of cytokine signaling 1 genes expression, cell growth inhibition and apop. Int. J. Hematol. Stem Cell Res.14, 45–55 (2020). [PMC free article] [PubMed] [Google Scholar]
- 690.Lu, Y. et al. Thioredoxin-interacting protein-activated intracellular potassium deprivation mediates the anti-tumour effect of a novel histone acetylation inhibitor HL23, a fangchinoline derivative, in human hepatocellular carcinoma. J. Adv. Res.51, 181–196 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 691.Chen, M. C. et al. Novel histone deacetylase inhibitor MPT0G009 induces cell apoptosis and synergistic anticancer activity with tumor necrosis factor-related apoptosis-inducing ligand against human hepatocellular carcinoma. Oncotarget7, 402–417 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 692.Lu, Y. S. et al. Radiosensitizing effect of a phenylbutyrate-derived histone deacetylase inhibitor in hepatocellular carcinoma. Int. J. Radiat. Oncol. Biol. Phys.83, e181–e189 (2012). [DOI] [PubMed] [Google Scholar]
- 693.He, B. et al. The HDAC inhibitor quisinostat (JNJ-26481585) supresses hepatocellular carcinoma alone and synergistically in combination with sorafenib by G0/G1 phase arrest and apoptosis induction. Int. J. Biol. Sci.14, 1845–1858 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 694.Buurman, R. et al. Histone deacetylases activate hepatocyte growth factor signaling by repressing microRNA-449 in hepatocellular carcinoma cells. Gastroenterology143, 811–820.e15 (2012). [DOI] [PubMed] [Google Scholar]
- 695.Shin, S., Kim, M., Lee, S. J., Park, K. S. & Lee, C. H. Trichostatin a sensitizes hepatocellular carcinoma cells to enhanced NK cell-mediated killing by regulating immune-related genes. Cancer Genomics Proteomics14, 349–362 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 696.Liu, J. et al. Droxinostat, a histone deacetylase inhibitor, induces apoptosis in hepatocellular carcinoma cell lines via activation of the mitochondrial pathway and downregulation of FLIP. Transl. Oncol.9, 70–78 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 697.Pathil, A. et al. HDAC inhibitor treatment of hepatoma cells induces both TRAIL-independent apoptosis and restoration of sensitivity to TRAIL. Hepatology43, 425–434 (2006). [DOI] [PubMed] [Google Scholar]
- 698.Schuchmann, M. et al. Histone deacetylase inhibition by valproic acid down-regulates c-FLIP/CASH and sensitizes hepatoma cells towards CD95- and TRAIL receptor-mediated apoptosis and chemotherapy. Oncol. Rep.15, 227–230 (2006). [DOI] [PubMed] [Google Scholar]
- 699.Sun, W. J. et al. Romidepsin induces G2/M phase arrest via Erk/cdc25C/cdc2/cyclinB pathway and apoptosis induction through JNK/c-Jun/caspase3 pathway in hepatocellular carcinoma cells. Biochem. Pharmacol.127, 90–100 (2017). [DOI] [PubMed] [Google Scholar]
- 700.Fu, M., Shi, W., Li, Z. & Liu, H. Activation of mPTP-dependent mitochondrial apoptosis pathway by a novel pan HDAC inhibitor resminostat in hepatocellular carcinoma cells. Biochem. Biophys. Res. Commun.477, 527–533 (2016). [DOI] [PubMed] [Google Scholar]
- 701.Afaloniati, H. et al. HDAC1/2 inhibitor romidepsin suppresses den-induced hepatocellular carcinogenesis in mice. Onco. Targets. Ther.13, 5575–5588 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 702.Liu, M. et al. Integrative Epigenetic Analysis Reveals Therapeutic Targets to the DNA Methyltransferase Inhibitor Guadecitabine (SGI-110) in Hepatocellular Carcinoma. Hepatology68, 1412–1428 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]