Abstract
Objectives
Tools are needed to evaluate the risk of developing Parkinson disease (PD) in at-risk populations. In this study, we examine differences in alpha-synuclein seed amplification assay (αSyn-SAA) qualitative results and amplification parameters between nonmanifesting carriers (NMCs) of PD-related pathogenic variants, prodromal PD, and PD and the risk of developing a synucleinopathy in participants with prodromal PD.
Methods
Cross-sectional and longitudinal CSF αSyn-SAA results from participants in the Parkinson's Progression Markers Initiative were analyzed. αSyn-SAA positivity and amplification parameters (maximum fluorescence [Fmax], time-to-threshold [TTT], time-to-50% Fmax [T50], and area under the curve [AUC]) were compared between NMCs, participants with prodromal PD, and participants with PD, and their relationship with the likelihood of phenoconversion in participants with prodromal PD was investigated.
Results
Samples from 1,027 participants were analyzed (159 healthy controls [HCs], 247 NMCs, 96 participants with prodromal PD, and 525 participants with PD). TTT and T50 were faster, and AUC was higher in αSyn-SAA+ participants with prodromal PD and PD than αSyn-SAA+ NMCs and HC participants (Kruskal-Wallis χ2 = 4.15–13.96, p < 0.0002–0.04). Participants with prodromal PD with positive αSyn-SAA tests and faster TTT had higher rates of phenoconversion (log-rank p = 0.001 and log-rank test-for-trend p < 0.0001). There were no changes in 48 participants with prodromal PD with longitudinal assays.
Discussion
αSyn-SAA positivity and faster seed amplification are associated with a greater risk of developing PD in at-risk individuals and may aid in predicting phenoconversion.
Introduction
Alpha-synuclein seed amplification assay (αSyn-SAA) enables sensitive and specific detection of αSyn-seeds in CSF, which are a biomarker of α-synuclein pathology in Parkinson disease (PD) and dementia with Lewy bodies (DLB) and in patients with REM sleep behavior disorder (RBD) and hyposmia who are at risk of developing PD or DLB.1-8 Early studies showed that faster amplification, reflected in shorter time-to-threshold (TTT), shorter time-to-50% maximum fluorescence (T50), and greater area under the curve (AUC), correlated with higher quantities of synthetic αSyn-seeds.1,9 However, only modest associations have been reported between these amplification parameters and clinical changes in patients with PD.5,6,8,10,11 It is not clear whether early detection of brain α-synuclein pathology by αSyn-SAA or amplification parameters can predict the onset of motor PD in at-risk populations.7,10 In this study, we examine the cross-sectional differences in αSyn-SAA positivity and amplification parameters in participants of the Parkinson's Progression Markers Initiative (PPMI) study, including nonmanifesting carriers (NMCs) of pathogenic variants increasing the risk of developing PD, prodromal PD, and manifest PD. In addition, we examine longitudinal changes in these groups and the relationship of αSyn-SAA positivity and amplification parameters with the risk of phenoconversion in participants with prodromal PD.
Methods
Study Design and Participants
Detailed information about inclusion criteria, informed consent, demographics, and study design can be found on the PPMI website.12
The study cohort included healthy control (HC) participants, participants with PD, participants with prodromal PD (hyposmia and/or RBD with mild deficits on dopamine-transporter [DAT] SPECT scans, with or without pathogenic genetic variants that increase the risk of developing PD [LRRK2: leucine-rich repeat kinase 2, GBA: glucocerebrocidase, SNCA: α-synuclein]), and NMCs without RBD or hyposmia. The study was approved by institutional review boards at each site, and participants provided informed consent to participate. Cohort assignment was made using the most recent consensus diagnosis available at the time of data download on May 8, 2023.13 Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) part III was recorded in the off-state for treated participants.
α-Synuclein Seed Amplification Assay
CSF samples were submitted between February and August of 2022 for analysis using the Amprion αSyn-SAA as previously described.14 Amplification parameters (TTT [h], Fmax [RFU], T50 [h], and AUC [%*s]) (eFigure 1) were calculated as previously described.14 Because samples were run in triplicate, the median value of the aforementioned parameters was used for statistical analyses.
Statistical Analysis
Categorical demographic variables were compared using the Fisher exact test. Continuous demographic variables and differences in αSyn-SAA amplification parameters were compared using Kruskal-Wallis tests, with Wilcoxon tests for pairwise comparisons. Survival analyses of time-to-event were performed to compare αSyn-SAA results and the risk of phenoconversion of participants with prodromal PD to manifest PD or DLB. Log-rank tests were used for comparing 2 survival groups, and the log-rank test-for-trend was used when more than 2 groups were analyzed with Holm correction for multiple comparisons. Correlations between amplification parameters were calculated using Spearman rho.
Results
Cross-Sectional Analysis
Of 1,119 possible participants, 1,027 were studied. Excluded cases included participants with inconclusive αSyn-SAA results (n = 16), CSF amount not sufficient for testing (n = 5), participants without consensus diagnoses or with discordant cohort assignments (n = 69), and participants classified as “scans without evidence of dopamine deficiency (SWEDD)” (n = 2) (eFigure 2). While some cross-sectional results have been previously reported,4 here we used the most updated consensus diagnoses, excluded SWEDD participants, included those with SNCA pathogenic variants, and analyzed participants with GBA or LRRK2 pathogenic variants with hyposmia or RBD in the prodromal PD group (eTable 1). Demographics and αSyn-SAA positivity are provided in Table 1.
Table 1.
Clinical Variables in Cohorts Assessed and αSyn-SAA Positivity
| Clinical values | Healthy controls (N = 159) | Nonmanifesting carriers (N = 247) | Prodromal PD cohort (N = 96) | PD cohort (N = 525) |
| Age, mean (SD) y | 60.6 (11.2) | 62.0 (7.4) | 64.3 (8.4) | 61.5 (9.5) |
| Sexa, no. (%) | F: 55 (35) M: 104 (65) |
F: 138 (56) M: 109 (44) |
F: 39 (41) M: 57 (59) |
F: 201 (38) M: 324 (62) |
| Racea, no. (%) | Black/African American: 8 (5) White: 148 (93) Other/multiple races: 1 (<1) Not reported: 2 (1) |
White: 242 (98) Not reported: 5 (2) |
Black/African American: 1 (1) White: 91 (95) Not reported: 4 (4) |
American Indian/Alaskan Native: 1 (<1) Asian: 9 (2) Black/African American: 7 (1) White: 496 (95) Other/multiple races: 7 (1) Not reported: 5 (<1) |
| Hispanic/Latino ethnicitya, no. (%) | 3 (2%) | 11 (5%) | 21 (22% | 30 (6%) |
| Education, mean years (SD) | 16.2 (3.1) | 16.1 (3.8) | 15.2 (4.9) | 15.6 (3.4) |
| αSyn-SAA positivity | ||||
| Totala | 3 (2%, 95% CI 0.4–5.4) | 13 (5%, 95% CI 3.1–9.3) | 54 (56%, 95% CI 45.7–66.4) | 463 (88%, 95% CI 85.1–90.8) |
| Sporadic | NA | NA | 44/51 (86%, 95% CI 73.7–94.3) |
329/353 (93%, 95% CI 90.1–95.6) |
| GBAa | NA | 5/122 (4%, 95% CI 1.3–9.3) | 5/18 (28%, 95% CI 9.7–54.3) | 47/49 (96%, 95% CI 86.0–99.5) |
| LRRK2a | NA | 8/125 (6%, 95% CI 2.8–12.2) | 5/25 (20%, 95% CI 6.8–40.7) | 76/112 (68%, 95% CI 58.4–76.4) |
| SNCAa | NA | NA | 0/2 (0%, NA) | 11/11 (100%, NA) |
Abbreviations: F = female; M = male; NA = not applicable.
Binomial exact 95% confidence intervals are reported.
p < 0.05 between cohorts.
Lower rates of αSyn-SAA positivity were observed in participants with prodromal PD with pathogenic variants than in participants with sporadic prodromal PD [prodromal PD-LRRK2: 20% (5/25), prodromal PD-GBA: 28% (5/18), prodromal PD-SNCA: 0% (0/2), and sporadic prodromal PD: 86% (44/51); χ2 = 40.5, p < 0.001]. Clinical differences between αSyn-SAA+ and αSyn-SAA- participants with prodromal PD, stratified by genetic status, are shown in the supplemental material (eTable 2). Different rates of αSyn-SAA positivity were observed between participants with sporadic prodromal PD and participants with sporadic PD but were not statistically significant (χ2 = 3.02, p = 0.08) (Table 1).
αSyn-SAA+ participants with PD and prodromal PD had faster TTT and T50 and higher AUC than the small number of αSyn-SAA+ HC and NMC participants (Figure 1A, eFigure 3). There were no major differences in amplification parameters between participants with prodromal PD with RBD and/or hyposmia (Figure 1B). Among αSyn-SAA+ participants with prodromal PD, sporadic cases had shorter TTT than those with GBA or LRRK2 pathogenic variants (Figure 1C). Participants with PD-SNCA had the shortest TTT and T50 and highest AUC, followed by participants with PD-GBA and sporadic PD and participants with PD-LRRK2 (Figure 1D, eFigure 4).
Figure 1. αSyn-SAA Amplification Parameters Across Cohorts.
Baseline speed of αSyn-SAA amplification evaluated using TTT (h) for different cohorts. (A) TTT for all αSyn-SAA+ participants, including healthy controls, GBA and LRRK2 nonmanifesting carriers, prodromal PD, and PD. (B) TTT for αSyn-SAA+ participants with prodromal PD grouped by prodromal symptoms (hyposmia and RBD) compared with nonmanifesting carriers. (C) TTT for αSyn-SAA+ participants with PD by genetic status. (D) Correlations of different amplification parameters. Size of the circle indicated magnitude of the correlation along with color indicating direction of association. **p < 0.01 for Pearson correlation between amplification parameters. αSyn-SAA = alpha-synuclein seed amplification assay; AUC = area under the curve, Fmax = maximum fluorescence; PD = Parkinson disease; T50 = time-to-50% maximum fluorescence; TTT = time-to-threshold.
Longitudinal Analysis
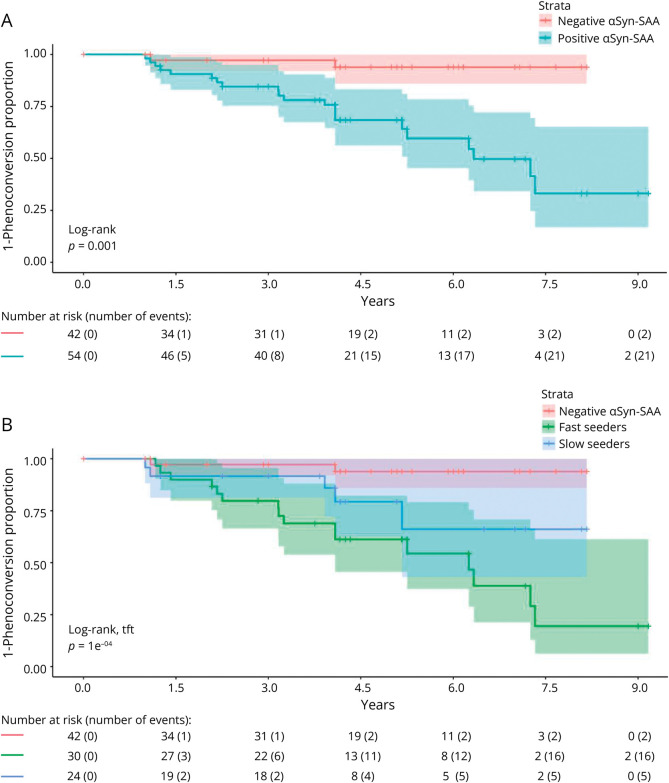
Longitudinal clinical data were available for 96 participants with prodromal PD (sporadic prodromal PD n = 51, prodromal PD-LRRK2 n = 25, prodromal PD-GBA n = 18, prodromal PD-SNCA n = 2) with baseline αSyn-SAA tests [56.2% (54/96) αSyn-SAA+]. The maximum follow-up time was 9.2 years (median 5.0 years, IQR 2.92 years). At the time of data abstraction, 23 of these participants (19 with sporadic prodromal PD and 4 with prodromal PD-LRRK2) had phenoconverted to PD (n = 21) or DLB (n = 2). Prodromal PD αSyn-SAA+ participants were more likely to phenoconvert (p = 0.001), suggesting that αSyn-SAA positivity could be used as an enrichment tool for individuals at risk of phenoconversion within 5–10 years (Figure 2A).
Figure 2. αSyn-SAA Positivity and Amplification Parameters and the Risk of Phenoconversion.
(A) Participants with prodromal PD with positive αSyn-SAA had a greater likelihood of phenoconversion to PD or DLB. (B) Of those with positive αSyn-SAA tests, participants with prodromal PD with faster TTT (by median split) had the greatest likelihood of phenoconversion, followed by those with slower TTT values and participants with negative αSyn-SAA. αSyn-SAA = alpha-synuclein seed amplification assay; DLB = dementia with Lewy bodies; PD = Parkinson disease; TTT = time-to-threshold.
When dividing the αSyn-SAA+ participants with prodromal PD into those with faster (TTT < median) and slower (TTT ≥ median) amplification, there was a stepwise increase in the likelihood of phenoconversion, with participants with “fast amplification” being the most likely to phenoconvert, followed by participants with “slow amplification” and αSyn-SAA- participants (log-rank test-for-trend p < 0.001, Figure 2B). Concordantly, participants with “fast amplification” had greater increases in MDS-UPDRS part III scores than participants with “slow amplification” over time (eFigure 5, A–C). Addition of age and sex as covariates further illustrated these differences (adjusted Cox proportional hazard “slow amplification” vs αSyn-SAA-, p = 0.0001, and “slow amplification” vs “fast amplification,” p = 0.03). There were no significant changes in TTT or T50 over time in longitudinally collected samples from 48 participants with prodromal PD (eFigure 6).
Discussion
CSF αSyn-SAA is an accepted biomarker of underlying α-synuclein pathology in PD and DLB.3,5,9,14,15 Because αSyn-seeds have been detected in those with prodromal PD, we evaluated αSyn-SAA positivity and amplification parameters and their association with phenoconversion to a PD or DLB diagnosis. We did not investigate genetic differences between prodromal participants because our goal was to evaluate whether underlying synuclein neuropathology detected by αSyn-SAA could be an indicator of clinical phenoconversion, regardless of the potential etiology of the α-synuclein pathology. Future appropriately powered studies will be needed to understand biomarker-related progression in these groups with potentially different pathobiologies. In this study, we showed that αSyn-SAA positivity is a phenoconversion predictor for those with prodromal PD symptoms (RBD and/or hyposmia) with mild DAT deficits.7,16 Moreover, the speed of amplification seems to be a potential tool to further increase the chances of identifying phenoconverters in this cohort. Larger, more diverse prodromal PD cohorts will be needed to confirm these findings and assess generalizability. Recent publications have shown associations between speed of amplification and cognitive decline in PD and other Lewy body disorders.17,18 Our results suggest that those same parameters combined with qualitative αSyn-SAA results could aid in identifying those with prodromal PD, who have the highest chances of phenoconversion, for neuroprotective clinical trial enrollment. However, this version of the αSyn-SAA seems unlikely to independently predict imminent phenoconversion at the individual level. Thus, additional biomarkers and/or newer αSyn-SAAs are probably needed to translate group findings to individual participants.
Acknowledgment
Data used in the preparation of this article were obtained on May 8, 2023, from the Parkinson's Progression Markers Initiative (PPMI) database (ppmi-info.org/access-data-specimens/download-data), RRID: SCR 006431. Up-to-date information on the study can be obtained from ppmi-info.org. PPMI—a public-private partnership—is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, BioArctic, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Capsida Biotherapeutics, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Jazz Pharmaceuticals, Johnson & Johnson Innovative Medicine, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Neuron23, Neuropore, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics.
Footnotes
Editorial, page e213384
Author Contributions
D.G. Coughlin: drafting/revision of the manuscript for content, including medical writing for content; study concept or design; analysis or interpretation of data. B. Shifflett: drafting/revision of the manuscript for content, including medical writing for content; analysis or interpretation of data. C.M. Farris: major role in the acquisition of data. Y. Ma: major role in the acquisition of data. D. Galasko: drafting/revision of the manuscript for content, including medical writing for content; study concept or design. S.D. Edland: drafting/revision of the manuscript for content, including medical writing for content; analysis or interpretation of data. B. Mollenhauer: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data. M.C. Brumm: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data. K.L. Poston: drafting/revision of the manuscript for content, including medical writing for content. K. Marek: major role in the acquisition of data; study concept or design. A.D. Siderowf: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; study concept or design. C. Soto: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data. L. Concha-Marambio: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis or interpretation of data.
Study Funding
D. Galasko is funded by P30 AG062429. D.G. Coughlin is funded by K23 NS122038. C. Soto is funded by U24 AG079685 and RF1 AG055053 and the Michael J. Fox Foundation. The Michael J. Fox Foundation funds the PPMI study.
Disclosure
Y. Ma, C.M. Farris, C. Soto, and L. Concha-Marambio are Amprion employees and declare employee stock option ownership and invention of patents related to SAA assigned to Amprion. The other authors report no relevant disclosures. Go to Neurology.org/N for full disclosures.
References
- 1.Shahnawaz M, Tokuda T, Waragai M, et al. Development of a biochemical diagnosis of Parkinson disease by detection of α-synuclein misfolded aggregates in cerebrospinal fluid. JAMA Neurol. 2017;74(2):163-172. doi: 10.1001/jamaneurol.2016.4547 [DOI] [PubMed] [Google Scholar]
- 2.Rossi M, Candelise N, Baiardi S, et al. Ultrasensitive RT-QuIC assay with high sensitivity and specificity for Lewy body-associated synucleinopathies. Acta Neuropathol. 2020;140(1):49-62. doi: 10.1007/s00401-020-02160-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Arnold MR, Coughlin DG, Brumbach BH, et al. α-Synuclein seed amplification in CSF and brain from patients with different brain distributions of pathological α-synuclein in the context of co-pathology and non-LBD diagnoses. Ann Neurol. 2022;92(4):650-662. doi: 10.1002/ANA.26453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Siderowf A, Concha-Marambio L, Lafontant D-E, et al. Assessment of heterogeneity among participants in the Parkinson's progression markers initiative cohort using α-synuclein seed amplification: a cross-sectional study. Lancet Neurol. 2023;22(5):407-417. doi: 10.1016/S1474-4422(23)00109-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kang UJ, Boehme AK, Fairfoul G, et al. Comparative study of cerebrospinal fluid α‐synuclein seeding aggregation assays for diagnosis of Parkinson's disease. Mov Disord. 2019;34(4):536-544. doi: 10.1002/mds.27646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Russo MJ, Orru CD, Concha-Marambio L, et al. High diagnostic performance of independent alpha-synuclein seed amplification assays for detection of early Parkinson's disease. Acta Neuropathol Commun. 2021;9(1):179. doi: 10.1186/S40478-021-01282-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Iranzo A, Fairfoul G, Ayudhaya ACN, et al. Detection of α-synuclein in CSF by RT-QuIC in patients with isolated rapid-eye-movement sleep behaviour disorder: a longitudinal observational study. Lancet Neurol. 2021;20(3):203-212. doi: 10.1016/S1474-4422(20)30449-X [DOI] [PubMed] [Google Scholar]
- 8.Concha-Marambio L, Weber S, Farris CM, et al. Accurate detection of α-synuclein seeds in cerebrospinal fluid from isolated rapid eye movement sleep behavior disorder and patients with Parkinson's disease in the denovo Parkinson (DeNoPa) cohort. Mov Disord. 2023;38(4):567-578. doi: 10.1002/mds.29329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Groveman BR, Orrù CD, Hughson AG, et al. Rapid and ultra-sensitive quantitation of disease-associated α-synuclein seeds in brain and cerebrospinal fluid by αSyn RT-QuIC. Acta Neuropathol Commun. 2018;6(1):7. doi: 10.1186/s40478-018-0508-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Poggiolini I, Gupta V, Lawton M, et al. Diagnostic value of cerebrospinal fluid alpha-synuclein seed quantification in synucleinopathies. Brain. 2022;145(2):584-595. doi: 10.1093/BRAIN/AWAB431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brockmann K, Quadalti C, Lerche S, et al. Association between CSF alpha-synuclein seeding activity and genetic status in Parkinson's disease and dementia with Lewy bodies. Acta Neuropathol Commun. 2021;9(1):175. doi: 10.1186/s40478-021-01276-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Parkinson's Progression Markers Initiative. ppmi-info.org
- 13.USC Stevens Neuroimaging and Informatics Institute, Image and Data Archive. http://ida.loni.usc.edu/home/projectPage.jsp?project=PPMI
- 14.Concha-Marambio L, Pritzkow S, Shahnawaz M, Farris CM, Soto C. Seed amplification assay for the detection of pathologic alpha-synuclein aggregates in cerebrospinal fluid. Nat Protoc. 2023;18(4):1179-1196. doi: 10.1038/s41596-022-00787-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fairfoul G, McGuire LI, Pal S, et al. Alpha-synuclein RT-Qu IC in the CSF of patients with alpha-synucleinopathies. Ann Clin Transl Neurol. 2016;3(10):812-818. doi: 10.1002/acn3.338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Siderowf A, Jennings D, Stern M, et al. Clinical and imaging progression in the PARS cohort: long‐term follow‐up. Mov Disord. 2020;35(9):1550-1557. doi: 10.1002/mds.28139 [DOI] [PubMed] [Google Scholar]
- 17.Brockmann K, Quadalti C, Lerche S, et al. Association between CSF alpha-synuclein seeding activity and genetic status in Parkinson's disease and dementia with Lewy bodies. Acta Neuropathol Commun. 2021;9(1):175. doi: 10.1186/s40478-021-01276-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bräuer S, Rossi M, Sajapin J, et al. Kinetic parameters of alpha-synuclein seed amplification assay correlate with cognitive impairment in patients with Lewy body disorders. Acta Neuropathol Commun. 2023;11(1):162. doi: 10.1186/s40478-023-01653-3 [DOI] [PMC free article] [PubMed] [Google Scholar]