Abstract
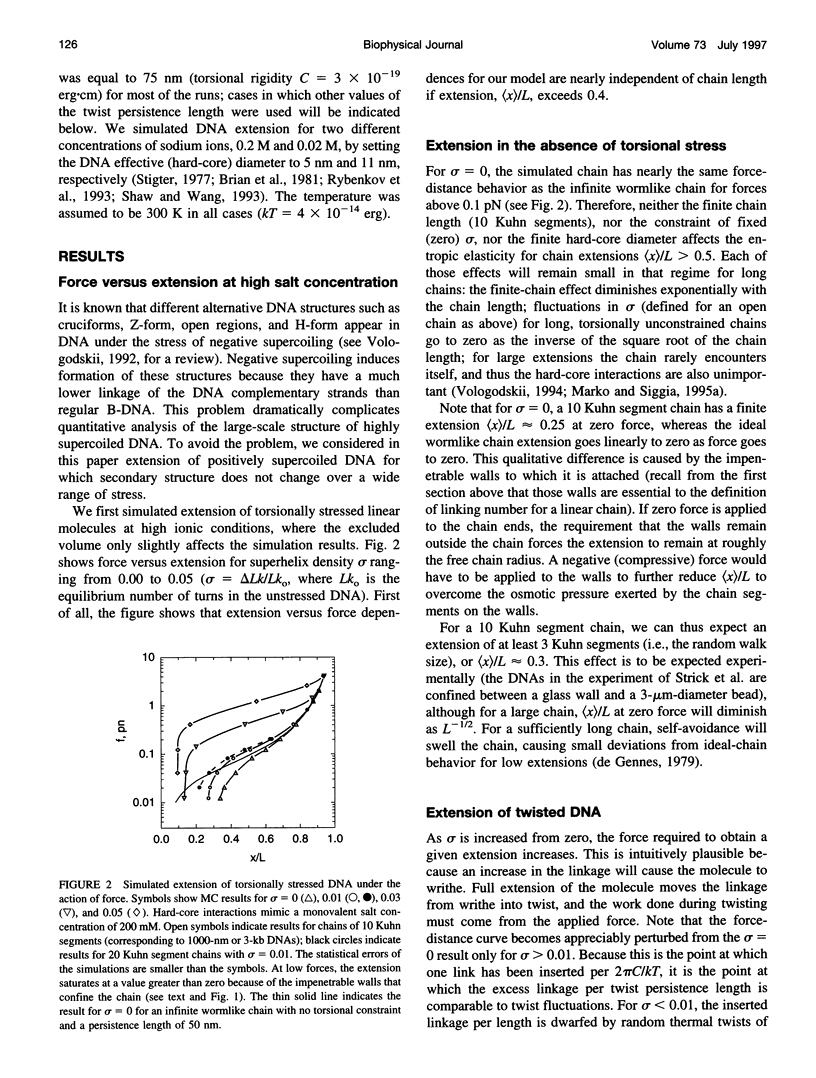
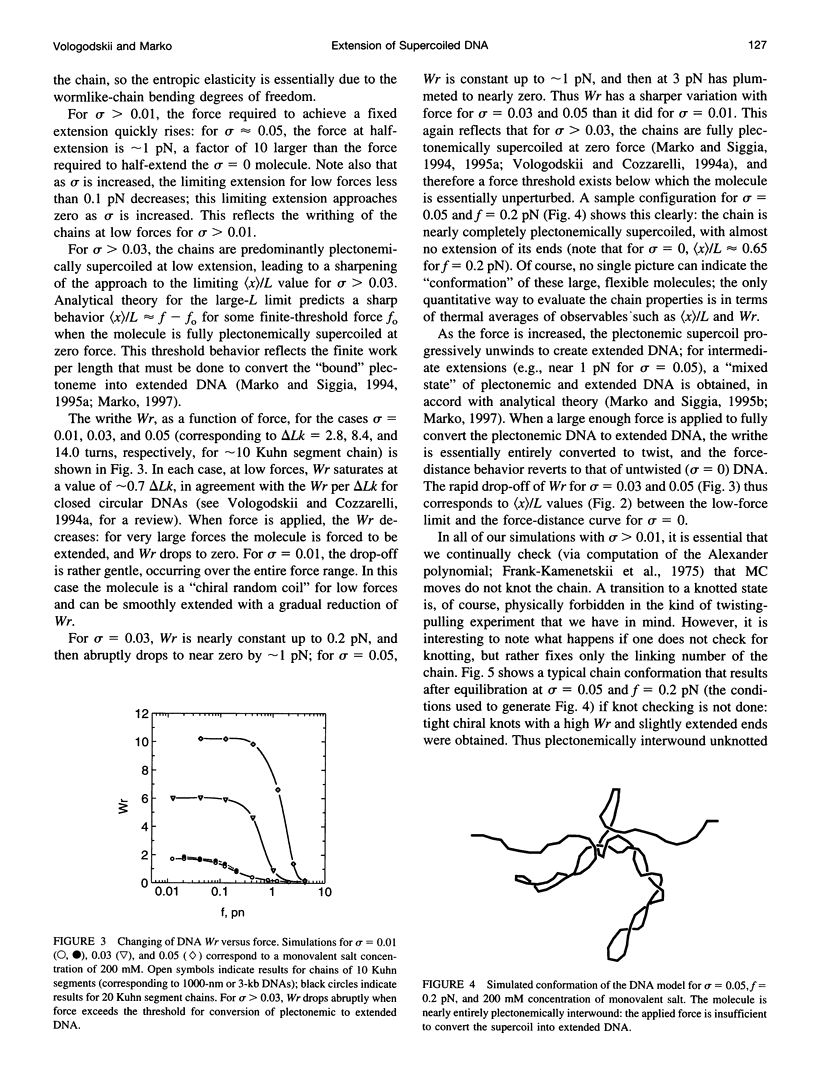
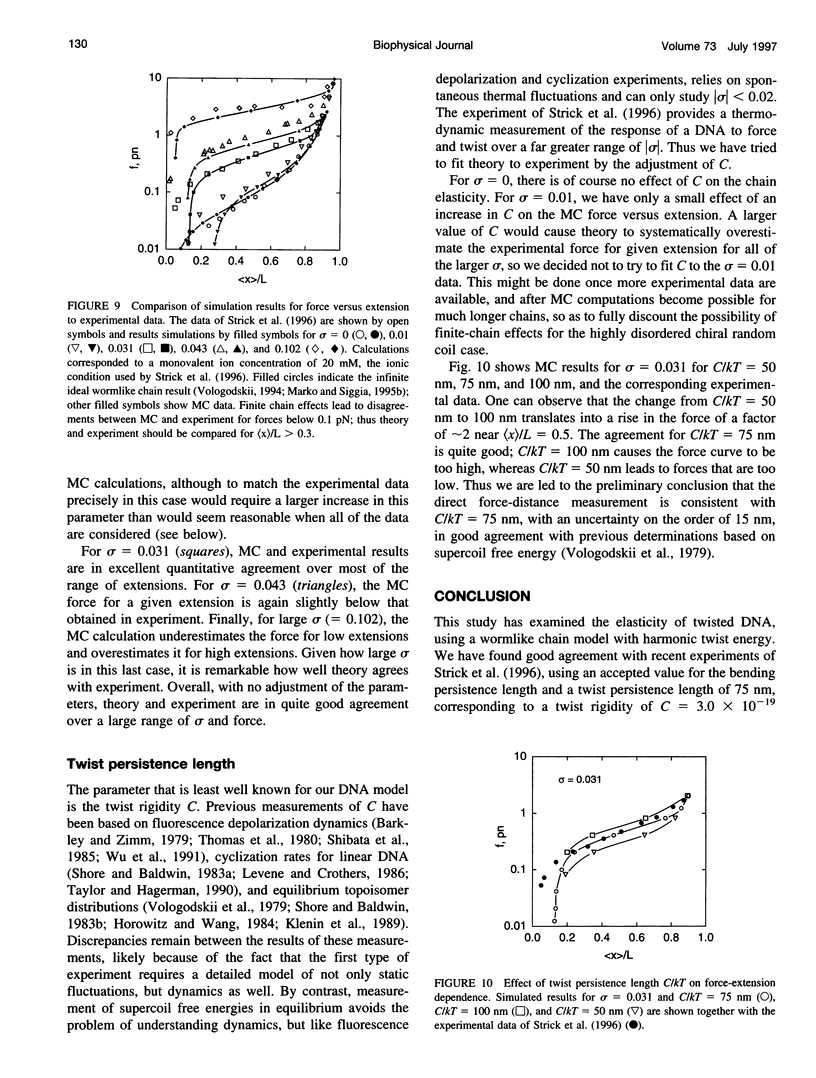
Metropolis Monte Carlo simulation was used to study the elasticity of torsionally stressed double-helical DNA. Equilibrium distributions of DNA conformations for different values of linking deficit, external force, and ionic conditions were simulated using the discrete wormlike chain model. Ionic conditions were specified in terms of DNA effective diameter, i.e., hard-core radius of the model chain. The simulations show that entropic elasticity of the double helix depends on how much it is twisted. For low amounts of twisting (less than about one turn per twist persistence length) the force versus extension is nearly the same as in the completely torsionally relaxed case. For more twisting than this, the molecule starts to supercoil, and there is an increase in the force needed to realize a given extension. For sufficiently large amounts of twist, the entire chain is plectonemically supercoiled at low extensions; a finite force must be applied to obtain any extension at all in this regime. The simulation results agree well with the results of recent micromanipulation experiments.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brian A. A., Frisch H. L., Lerman L. S. Thermodynamics and equilibrium sedimentation analysis of the close approach of DNA molecules and a molecular ordering transition. Biopolymers. 1981 Jun;20(6):1305–1328. doi: 10.1002/bip.1981.360200615. [DOI] [PubMed] [Google Scholar]
- Bustamante C., Marko J. F., Siggia E. D., Smith S. Entropic elasticity of lambda-phage DNA. Science. 1994 Sep 9;265(5178):1599–1600. doi: 10.1126/science.8079175. [DOI] [PubMed] [Google Scholar]
- Cluzel P., Lebrun A., Heller C., Lavery R., Viovy J. L., Chatenay D., Caron F. DNA: an extensible molecule. Science. 1996 Feb 9;271(5250):792–794. doi: 10.1126/science.271.5250.792. [DOI] [PubMed] [Google Scholar]
- Crothers D. M., Drak J., Kahn J. D., Levene S. D. DNA bending, flexibility, and helical repeat by cyclization kinetics. Methods Enzymol. 1992;212:3–29. doi: 10.1016/0076-6879(92)12003-9. [DOI] [PubMed] [Google Scholar]
- Frank-Kamenetskii M. D., Lukashin A. V., Anshelevich V. V., Vologodskii A. V. Torsional and bending rigidity of the double helix from data on small DNA rings. J Biomol Struct Dyn. 1985 Feb;2(5):1005–1012. doi: 10.1080/07391102.1985.10507616. [DOI] [PubMed] [Google Scholar]
- Frank-Kamenetskii M. D., Lukashin A. V., Vologodskii A. V. Statistical mechanics and topology of polymer chains. Nature. 1975 Dec 4;258(5534):398–402. doi: 10.1038/258398a0. [DOI] [PubMed] [Google Scholar]
- Fuller F. B. The writhing number of a space curve. Proc Natl Acad Sci U S A. 1971 Apr;68(4):815–819. doi: 10.1073/pnas.68.4.815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebe J. A., Allison S. A., Clendenning J. B., Schurr J. M. Monte Carlo simulations of supercoiling free energies for unknotted and trefoil knotted DNAs. Biophys J. 1995 Feb;68(2):619–633. doi: 10.1016/S0006-3495(95)80223-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebe J. A., Delrow J. J., Heath P. J., Fujimoto B. S., Stewart D. W., Schurr J. M. Effects of Na+ and Mg2+ on the structures of supercoiled DNAs: comparison of simulations with experiments. J Mol Biol. 1996 Sep 20;262(2):105–128. doi: 10.1006/jmbi.1996.0502. [DOI] [PubMed] [Google Scholar]
- Hagerman P. J. Flexibility of DNA. Annu Rev Biophys Biophys Chem. 1988;17:265–286. doi: 10.1146/annurev.bb.17.060188.001405. [DOI] [PubMed] [Google Scholar]
- Horowitz D. S., Wang J. C. Torsional rigidity of DNA and length dependence of the free energy of DNA supercoiling. J Mol Biol. 1984 Feb 15;173(1):75–91. doi: 10.1016/0022-2836(84)90404-2. [DOI] [PubMed] [Google Scholar]
- Katritch V., Vologodskii A. The effect of intrinsic curvature on conformational properties of circular DNA. Biophys J. 1997 Mar;72(3):1070–1079. doi: 10.1016/S0006-3495(97)78757-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klenin K. V., Vologodskii A. V., Anshelevich V. V., Dykhne A. M., Frank-Kamenetskii M. D. Computer simulation of DNA supercoiling. J Mol Biol. 1991 Feb 5;217(3):413–419. doi: 10.1016/0022-2836(91)90745-r. [DOI] [PubMed] [Google Scholar]
- Klenin K. V., Vologodskii A. V., Anshelevich V. V., Klishko VYu, Dykhne A. M., Frank-Kamenetskii M. D. Variance of writhe for wormlike DNA rings with excluded volume. J Biomol Struct Dyn. 1989 Feb;6(4):707–714. doi: 10.1080/07391102.1989.10507731. [DOI] [PubMed] [Google Scholar]
- Levene S. D., Crothers D. M. Ring closure probabilities for DNA fragments by Monte Carlo simulation. J Mol Biol. 1986 May 5;189(1):61–72. doi: 10.1016/0022-2836(86)90381-5. [DOI] [PubMed] [Google Scholar]
- Marko J. F., Siggia E. D. Fluctuations and supercoiling of DNA. Science. 1994 Jul 22;265(5171):506–508. doi: 10.1126/science.8036491. [DOI] [PubMed] [Google Scholar]
- Marko JF, Siggia ED. Statistical mechanics of supercoiled DNA. Phys Rev E Stat Phys Plasmas Fluids Relat Interdiscip Topics. 1995 Sep;52(3):2912–2938. doi: 10.1103/physreve.52.2912. [DOI] [PubMed] [Google Scholar]
- Perkins T. T., Quake S. R., Smith D. E., Chu S. Relaxation of a single DNA molecule observed by optical microscopy. Science. 1994 May 6;264(5160):822–826. doi: 10.1126/science.8171336. [DOI] [PubMed] [Google Scholar]
- Perkins T. T., Smith D. E., Larson R. G., Chu S. Stretching of a single tethered polymer in a uniform flow. Science. 1995 Apr 7;268(5207):83–87. doi: 10.1126/science.7701345. [DOI] [PubMed] [Google Scholar]
- Rybenkov V. V., Cozzarelli N. R., Vologodskii A. V. Probability of DNA knotting and the effective diameter of the DNA double helix. Proc Natl Acad Sci U S A. 1993 Jun 1;90(11):5307–5311. doi: 10.1073/pnas.90.11.5307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw S. Y., Wang J. C. Knotting of a DNA chain during ring closure. Science. 1993 Apr 23;260(5107):533–536. doi: 10.1126/science.8475384. [DOI] [PubMed] [Google Scholar]
- Shibata J. H., Fujimoto B. S., Schurr J. M. Rotational dynamics of DNA from 10(-10) to 10(-5) seconds: comparison of theory with optical experiments. Biopolymers. 1985 Oct;24(10):1909–1930. doi: 10.1002/bip.360241006. [DOI] [PubMed] [Google Scholar]
- Shore D., Baldwin R. L. Energetics of DNA twisting. I. Relation between twist and cyclization probability. J Mol Biol. 1983 Nov 15;170(4):957–981. doi: 10.1016/s0022-2836(83)80198-3. [DOI] [PubMed] [Google Scholar]
- Shore D., Baldwin R. L. Energetics of DNA twisting. II. Topoisomer analysis. J Mol Biol. 1983 Nov 15;170(4):983–1007. doi: 10.1016/s0022-2836(83)80199-5. [DOI] [PubMed] [Google Scholar]
- Smith S. B., Cui Y., Bustamante C. Overstretching B-DNA: the elastic response of individual double-stranded and single-stranded DNA molecules. Science. 1996 Feb 9;271(5250):795–799. doi: 10.1126/science.271.5250.795. [DOI] [PubMed] [Google Scholar]
- Smith S. B., Finzi L., Bustamante C. Direct mechanical measurements of the elasticity of single DNA molecules by using magnetic beads. Science. 1992 Nov 13;258(5085):1122–1126. doi: 10.1126/science.1439819. [DOI] [PubMed] [Google Scholar]
- Stigter D. Interactions of highly charged colloidal cylinders with applications to double-stranded. Biopolymers. 1977 Jul;16(7):1435–1448. doi: 10.1002/bip.1977.360160705. [DOI] [PubMed] [Google Scholar]
- Strick T. R., Allemand J. F., Bensimon D., Bensimon A., Croquette V. The elasticity of a single supercoiled DNA molecule. Science. 1996 Mar 29;271(5257):1835–1837. doi: 10.1126/science.271.5257.1835. [DOI] [PubMed] [Google Scholar]
- Tan R. K., Harvey S. C. Molecular mechanics model of supercoiled DNA. J Mol Biol. 1989 Feb 5;205(3):573–591. doi: 10.1016/0022-2836(89)90227-1. [DOI] [PubMed] [Google Scholar]
- Taylor W. H., Hagerman P. J. Application of the method of phage T4 DNA ligase-catalyzed ring-closure to the study of DNA structure. II. NaCl-dependence of DNA flexibility and helical repeat. J Mol Biol. 1990 Mar 20;212(2):363–376. doi: 10.1016/0022-2836(90)90131-5. [DOI] [PubMed] [Google Scholar]
- Thomas J. C., Allison S. A., Appellof C. J., Schurr J. M. Torison dynamics and depolarization of fluorescence of linear macromolecules. II. Fluorescence polarization anisotropy measurements on a clean viral phi 29 DNA. Biophys Chem. 1980 Oct;12(2):177–188. doi: 10.1016/0301-4622(80)80050-0. [DOI] [PubMed] [Google Scholar]
- Vologodskii A. V., Anshelevich V. V., Lukashin A. V., Frank-Kamenetskii M. D. Statistical mechanics of supercoils and the torsional stiffness of the DNA double helix. Nature. 1979 Jul 26;280(5720):294–298. doi: 10.1038/280294a0. [DOI] [PubMed] [Google Scholar]
- Vologodskii A. V., Cozzarelli N. R. Conformational and thermodynamic properties of supercoiled DNA. Annu Rev Biophys Biomol Struct. 1994;23:609–643. doi: 10.1146/annurev.bb.23.060194.003141. [DOI] [PubMed] [Google Scholar]
- Vologodskii A. V., Cozzarelli N. R. Monte Carlo analysis of the conformation of DNA catenanes. J Mol Biol. 1993 Aug 20;232(4):1130–1140. doi: 10.1006/jmbi.1993.1465. [DOI] [PubMed] [Google Scholar]
- Vologodskii A. V., Levene S. D., Klenin K. V., Frank-Kamenetskii M., Cozzarelli N. R. Conformational and thermodynamic properties of supercoiled DNA. J Mol Biol. 1992 Oct 20;227(4):1224–1243. doi: 10.1016/0022-2836(92)90533-p. [DOI] [PubMed] [Google Scholar]
- Vologodskii A. V., Lukashin A. V., Anshelevich V. V., Frank-Kamenetskii M. D. Fluctuations in superhelical DNA. Nucleic Acids Res. 1979 Mar;6(3):967–982. doi: 10.1093/nar/6.3.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu P. G., Fujimoto B. S., Song L., Schurr J. M. Effect of ethidium on the torsion constants of linear and supercoiled DNAs. Biophys Chem. 1991 Dec;41(3):217–236. doi: 10.1016/0301-4622(91)85038-r. [DOI] [PubMed] [Google Scholar]
- Zechiedrich E. L., Cozzarelli N. R. Roles of topoisomerase IV and DNA gyrase in DNA unlinking during replication in Escherichia coli. Genes Dev. 1995 Nov 15;9(22):2859–2869. doi: 10.1101/gad.9.22.2859. [DOI] [PubMed] [Google Scholar]


