Abstract
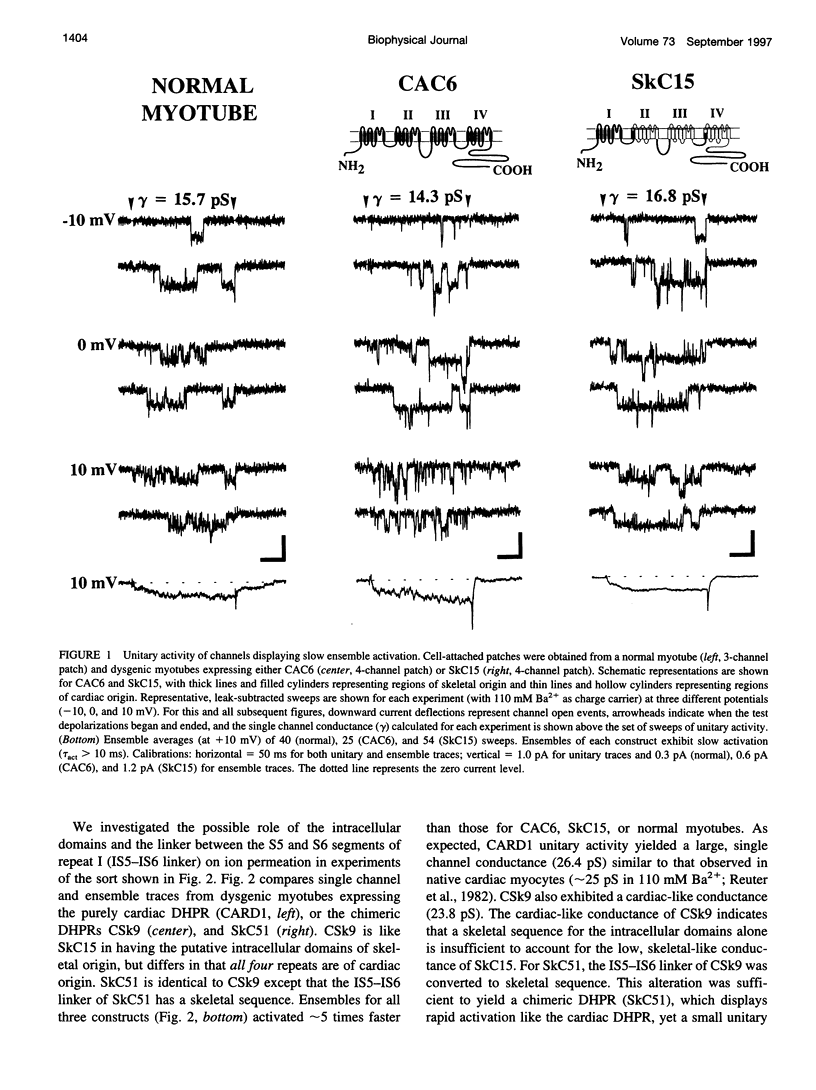
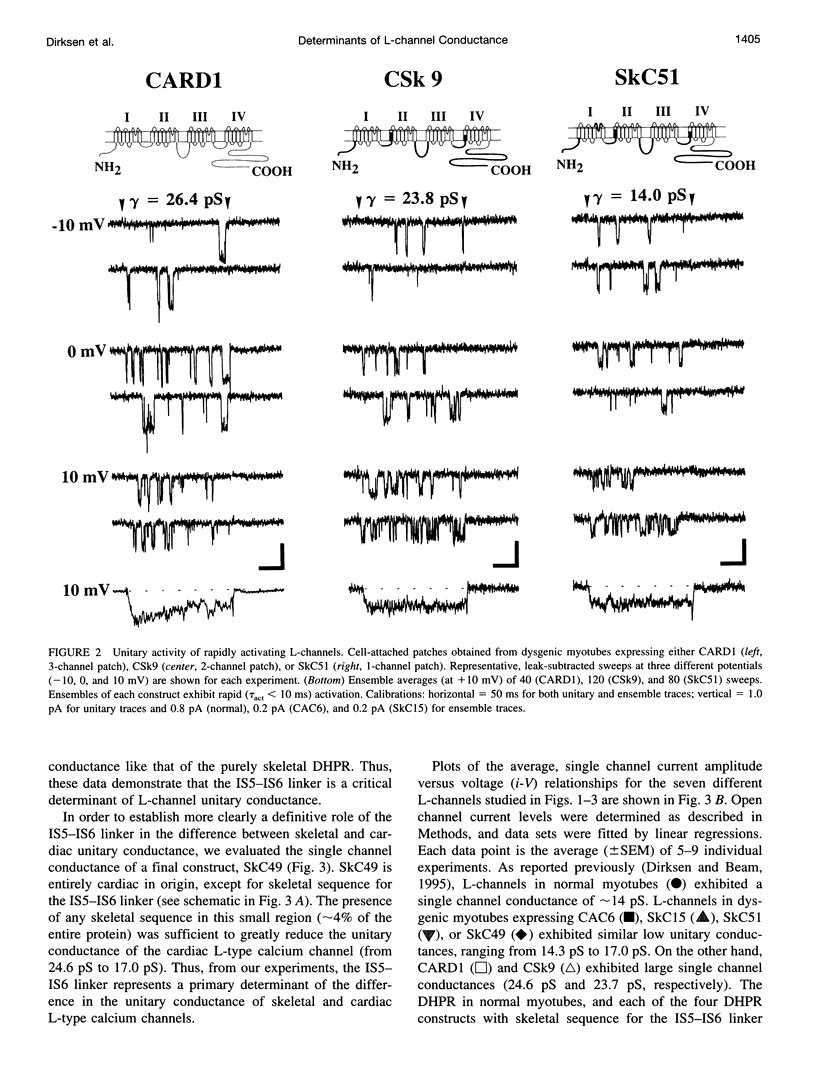
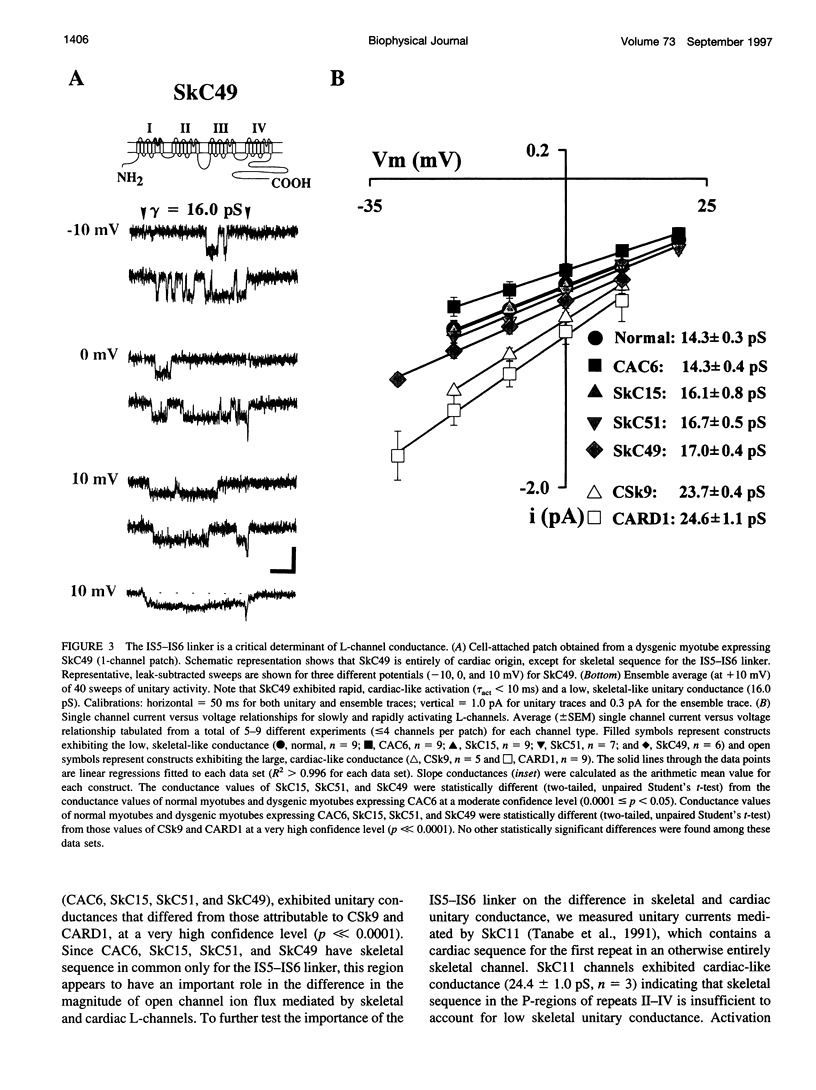
The alpha1-subunits of the skeletal and cardiac L-type calcium channels (L-channels) contain nearly identical pore regions (P-regions) in each of the four internal homology repeats. In spite of this high conservation of the P-regions, native skeletal L-channels exhibit a unitary conductance that is only about half that of native cardiac L-channels. To identify structural determinants of this difference in L-channel conductance, we have characterized unitary activity in cell-attached patches of dysgenic myotubes expressing skeletal, cardiac, and chimeric L-channel alpha1-subunits. Our results demonstrate that the S5-S6 linker of repeat I (IS5-IS6 linker) is a critical determinant of the difference in skeletal and cardiac unitary conductance. The unitary conductances attributable to the wild-type skeletal (CAC6; approximately 14 pS) and cardiac (CARD1; approximately 25 pS) alpha1-subunits expressed in dysgenic myotubes are identical to those observed in native tissues. Chimeric alpha1-subunits containing skeletal sequence for the first internal repeat and all of the putative intracellular loops (SkC15), the IS5-IS6 linker and the intracellular loops (SkC51), or only the IS5-IS6 linker (SkC49) each exhibit a low, skeletal-like unitary conductance (< or = 17 pS). Constructs in which the IS5-IS6 linker is of cardiac origin (CARD1 and CSk9) display cardiac-like conductance (approximately 25 pS). Unitary conductance and the rate of channel activation are apparently independent processes, since both SkC51 and SkC49 exhibit low, skeletal-like conductance and rapid, cardiac-like rates of ensemble activation. These results demonstrate that the IS5-IS6 linker strongly influences the single channel conductance of L-channels in a manner that is independent from the rate of channel activation.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Armstrong C. M., Bezanilla F. M., Horowicz P. Twitches in the presence of ethylene glycol bis( -aminoethyl ether)-N,N'-tetracetic acid. Biochim Biophys Acta. 1972 Jun 23;267(3):605–608. doi: 10.1016/0005-2728(72)90194-6. [DOI] [PubMed] [Google Scholar]
- Beam K. G., Knudson C. M. Calcium currents in embryonic and neonatal mammalian skeletal muscle. J Gen Physiol. 1988 Jun;91(6):781–798. doi: 10.1085/jgp.91.6.781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnbaumer L., Campbell K. P., Catterall W. A., Harpold M. M., Hofmann F., Horne W. A., Mori Y., Schwartz A., Snutch T. P., Tanabe T. The naming of voltage-gated calcium channels. Neuron. 1994 Sep;13(3):505–506. doi: 10.1016/0896-6273(94)90021-3. [DOI] [PubMed] [Google Scholar]
- Dirksen R. T., Beam K. G. Single calcium channel behavior in native skeletal muscle. J Gen Physiol. 1995 Feb;105(2):227–247. doi: 10.1085/jgp.105.2.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dirksen R. T., Beam K. G. Unitary behavior of skeletal, cardiac, and chimeric L-type Ca2+ channels expressed in dysgenic myotubes. J Gen Physiol. 1996 Jun;107(6):731–742. doi: 10.1085/jgp.107.6.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujita Y., Mynlieff M., Dirksen R. T., Kim M. S., Niidome T., Nakai J., Friedrich T., Iwabe N., Miyata T., Furuichi T. Primary structure and functional expression of the omega-conotoxin-sensitive N-type calcium channel from rabbit brain. Neuron. 1993 Apr;10(4):585–598. doi: 10.1016/0896-6273(93)90162-k. [DOI] [PubMed] [Google Scholar]
- Gollasch M., Ried C., Liebold M., Haller H., Hofmann F., Luft F. C. High permeation of L-type Ca2+ channels at physiological [Ca2+]: homogeneity and dependence on the alpha 1-subunit. Am J Physiol. 1996 Sep;271(3 Pt 1):C842–C850. doi: 10.1152/ajpcell.1996.271.3.C842. [DOI] [PubMed] [Google Scholar]
- Hamill O. P., Marty A., Neher E., Sakmann B., Sigworth F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981 Aug;391(2):85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Mikami A., Imoto K., Tanabe T., Niidome T., Mori Y., Takeshima H., Narumiya S., Numa S. Primary structure and functional expression of the cardiac dihydropyridine-sensitive calcium channel. Nature. 1989 Jul 20;340(6230):230–233. doi: 10.1038/340230a0. [DOI] [PubMed] [Google Scholar]
- Nakai J., Adams B. A., Imoto K., Beam K. G. Critical roles of the S3 segment and S3-S4 linker of repeat I in activation of L-type calcium channels. Proc Natl Acad Sci U S A. 1994 Feb 1;91(3):1014–1018. doi: 10.1073/pnas.91.3.1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noda M., Suzuki H., Numa S., Stühmer W. A single point mutation confers tetrodotoxin and saxitoxin insensitivity on the sodium channel II. FEBS Lett. 1989 Dec 18;259(1):213–216. doi: 10.1016/0014-5793(89)81531-5. [DOI] [PubMed] [Google Scholar]
- Parent L., Gopalakrishnan M., Lacerda A. E., Wei X., Perez-Reyes E. Voltage-dependent inactivation in a cardiac-skeletal chimeric calcium channel. FEBS Lett. 1995 Feb 27;360(2):144–150. doi: 10.1016/0014-5793(95)00090-v. [DOI] [PubMed] [Google Scholar]
- Reuter H., Stevens C. F., Tsien R. W., Yellen G. Properties of single calcium channels in cardiac cell culture. Nature. 1982 Jun 10;297(5866):501–504. doi: 10.1038/297501a0. [DOI] [PubMed] [Google Scholar]
- Tanabe T., Adams B. A., Numa S., Beam K. G. Repeat I of the dihydropyridine receptor is critical in determining calcium channel activation kinetics. Nature. 1991 Aug 29;352(6338):800–803. doi: 10.1038/352800a0. [DOI] [PubMed] [Google Scholar]
- Tanabe T., Beam K. G., Adams B. A., Niidome T., Numa S. Regions of the skeletal muscle dihydropyridine receptor critical for excitation-contraction coupling. Nature. 1990 Aug 9;346(6284):567–569. doi: 10.1038/346567a0. [DOI] [PubMed] [Google Scholar]
- Tanabe T., Beam K. G., Powell J. A., Numa S. Restoration of excitation-contraction coupling and slow calcium current in dysgenic muscle by dihydropyridine receptor complementary DNA. Nature. 1988 Nov 10;336(6195):134–139. doi: 10.1038/336134a0. [DOI] [PubMed] [Google Scholar]
- Tanabe T., Mikami A., Numa S., Beam K. G. Cardiac-type excitation-contraction coupling in dysgenic skeletal muscle injected with cardiac dihydropyridine receptor cDNA. Nature. 1990 Mar 29;344(6265):451–453. doi: 10.1038/344451a0. [DOI] [PubMed] [Google Scholar]
- Tanabe T., Takeshima H., Mikami A., Flockerzi V., Takahashi H., Kangawa K., Kojima M., Matsuo H., Hirose T., Numa S. Primary structure of the receptor for calcium channel blockers from skeletal muscle. Nature. 1987 Jul 23;328(6128):313–318. doi: 10.1038/328313a0. [DOI] [PubMed] [Google Scholar]
- Tareilus E., Roux M., Qin N., Olcese R., Zhou J., Stefani E., Birnbaumer L. A Xenopus oocyte beta subunit: evidence for a role in the assembly/expression of voltage-gated calcium channels that is separate from its role as a regulatory subunit. Proc Natl Acad Sci U S A. 1997 Mar 4;94(5):1703–1708. doi: 10.1073/pnas.94.5.1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terlau H., Heinemann S. H., Stühmer W., Pusch M., Conti F., Imoto K., Numa S. Mapping the site of block by tetrodotoxin and saxitoxin of sodium channel II. FEBS Lett. 1991 Nov 18;293(1-2):93–96. doi: 10.1016/0014-5793(91)81159-6. [DOI] [PubMed] [Google Scholar]
- Williams M. E., Feldman D. H., McCue A. F., Brenner R., Velicelebi G., Ellis S. B., Harpold M. M. Structure and functional expression of alpha 1, alpha 2, and beta subunits of a novel human neuronal calcium channel subtype. Neuron. 1992 Jan;8(1):71–84. doi: 10.1016/0896-6273(92)90109-q. [DOI] [PubMed] [Google Scholar]
- Yang J., Ellinor P. T., Sather W. A., Zhang J. F., Tsien R. W. Molecular determinants of Ca2+ selectivity and ion permeation in L-type Ca2+ channels. Nature. 1993 Nov 11;366(6451):158–161. doi: 10.1038/366158a0. [DOI] [PubMed] [Google Scholar]
- Yellen G., Jurman M. E., Abramson T., MacKinnon R. Mutations affecting internal TEA blockade identify the probable pore-forming region of a K+ channel. Science. 1991 Feb 22;251(4996):939–942. doi: 10.1126/science.2000494. [DOI] [PubMed] [Google Scholar]


