Abstract
Fusion is a crucial event in the infection of animal cells by enveloped viruses (e.g., HIV or influenza). Viral fusion is mediated by glycoproteins, spanning the viral envelope, which attach to a membrane surface and induce fusion of the viral envelope to the cellular membrane. Influenza fusion protein (hemagglutinin) contains an amino-terminal segment critical to fusion, referred to as the fusion peptide. We show here that the native fusion peptide (wt-20) of hemagglutinin destabilizes membranes formed of 99% 1 -stearoyl-2-oleoylphosphatidylcholine (SOPC). The first step in destabilization is rapid insertion of the peptide into the membrane, in which membrane area increases by as much as 11% in just seconds. We visualized and quantified the area expansion by using optical video microscopy combined with micropipette aspiration. This rapid membrane area expansion is followed by the formation of membrane defects in the size range of 0.5 nm, and results in membrane rupture. Both the rate of area increase and maximum area increase are significantly higher at a pH near 5.0 compared to pH 7.0. These results suggest that enhanced membrane insertion of wt-20 and accompanying area expansion at pH 5.0 are responsible for the relatively greater lytic activity at this pH. We show that a deletion of the N-terminal glycine of wt-20 results in a lack of area expansion or membrane perturbation at pH 5.0.
Full text
PDF


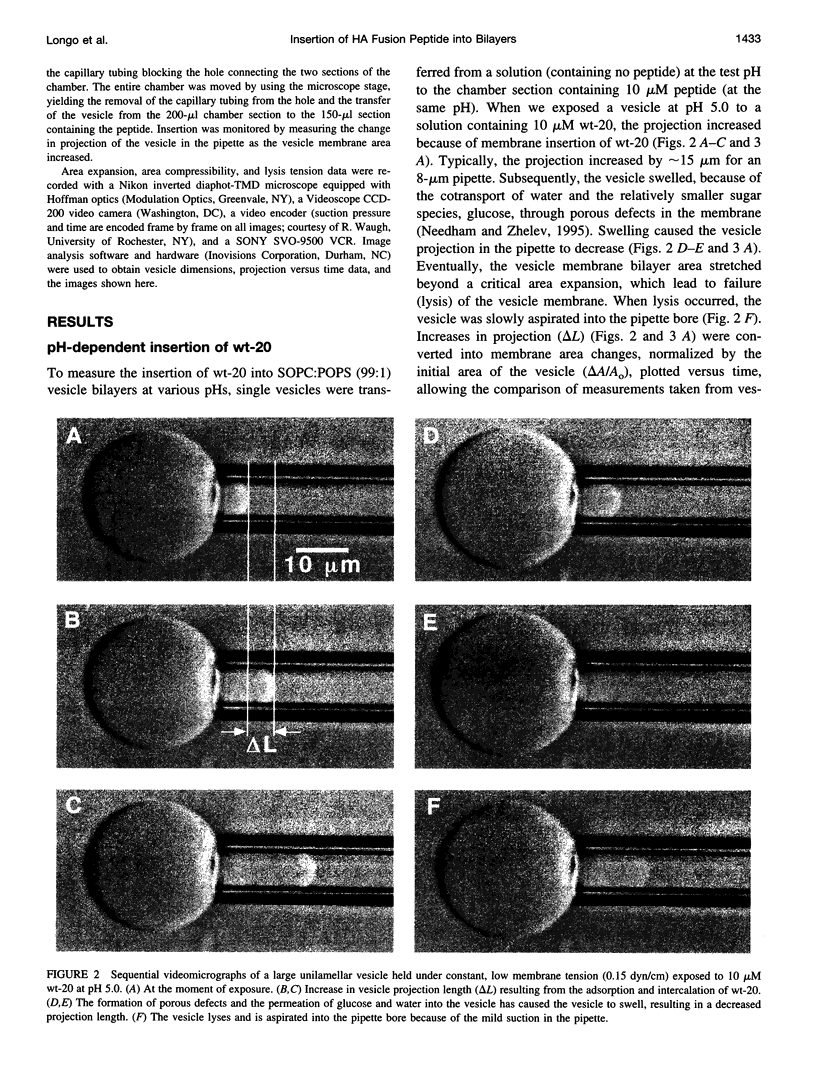
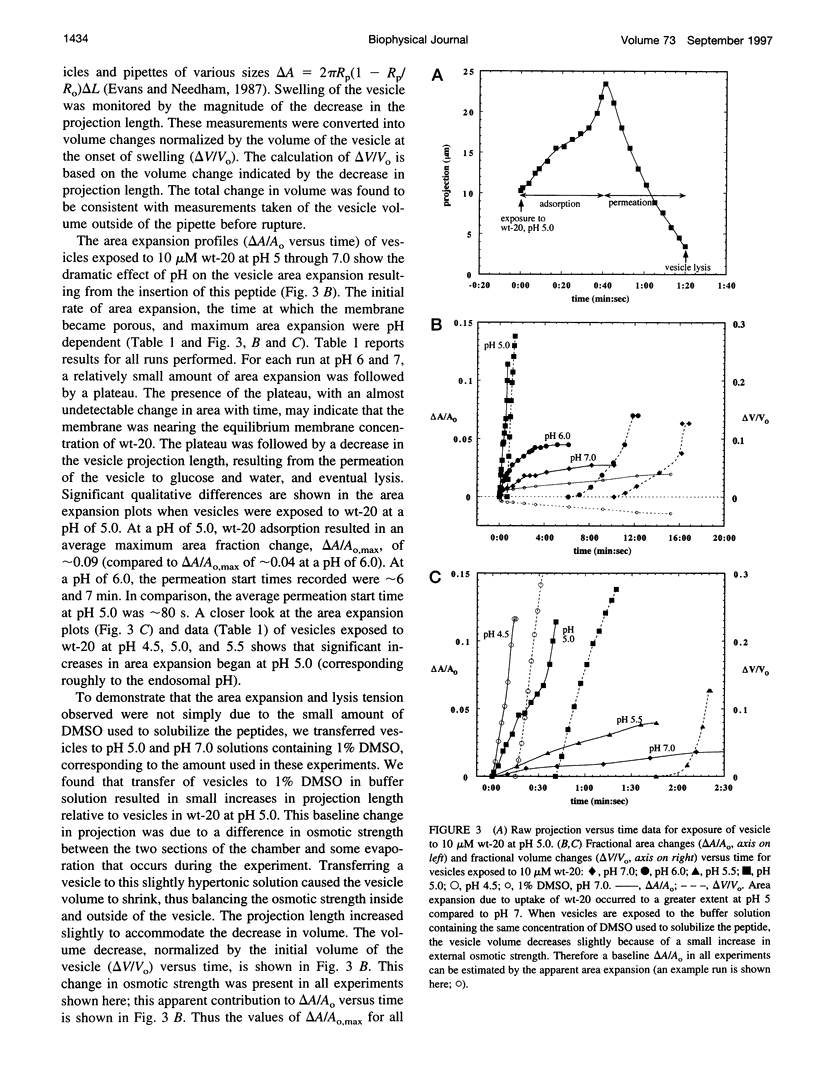

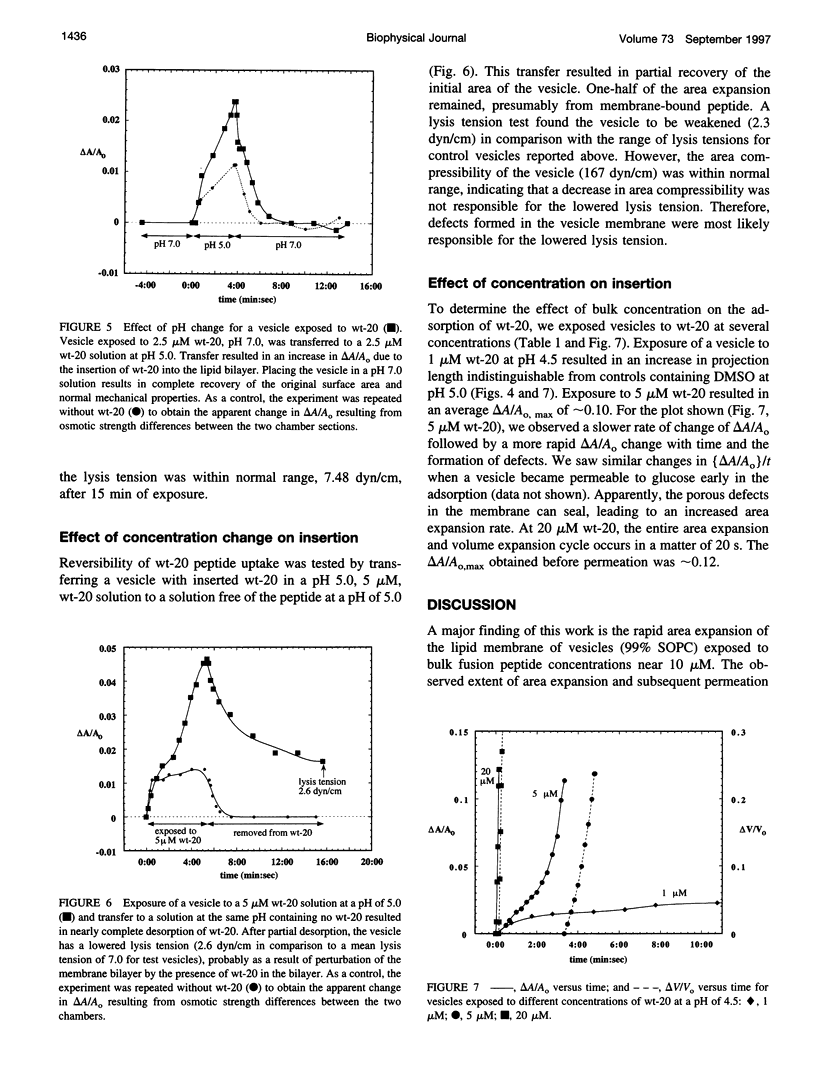



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson W. F. Gene therapy. Sci Am. 1995 Sep;273(3):124–128. [PubMed] [Google Scholar]
- Bagai S., Puri A., Blumenthal R., Sarkar D. P. Hemagglutinin-neuraminidase enhances F protein-mediated membrane fusion of reconstituted Sendai virus envelopes with cells. J Virol. 1993 Jun;67(6):3312–3318. doi: 10.1128/jvi.67.6.3312-3318.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burger K. N., Wharton S. A., Demel R. A., Verkleij A. J. The interaction of synthetic analogs of the N-terminal fusion sequence of influenza virus with a lipid monolayer. Comparison of fusion-active and fusion-defective analogs. Biochim Biophys Acta. 1991 Jun 18;1065(2):121–129. doi: 10.1016/0005-2736(91)90221-s. [DOI] [PubMed] [Google Scholar]
- Clague M. J., Knutson J. R., Blumenthal R., Herrmann A. Interaction of influenza hemagglutinin amino-terminal peptide with phospholipid vesicles: a fluorescence study. Biochemistry. 1991 Jun 4;30(22):5491–5497. doi: 10.1021/bi00236a023. [DOI] [PubMed] [Google Scholar]
- Curiel D. T., Agarwal S., Wagner E., Cotten M. Adenovirus enhancement of transferrin-polylysine-mediated gene delivery. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8850–8854. doi: 10.1073/pnas.88.19.8850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniels R. S., Downie J. C., Hay A. J., Knossow M., Skehel J. J., Wang M. L., Wiley D. C. Fusion mutants of the influenza virus hemagglutinin glycoprotein. Cell. 1985 Feb;40(2):431–439. doi: 10.1016/0092-8674(85)90157-6. [DOI] [PubMed] [Google Scholar]
- Düzgüneş N., Gambale F. Membrane action of synthetic N-terminal peptides of influenza virus hemagglutinin and its mutants. FEBS Lett. 1988 Jan 25;227(2):110–114. doi: 10.1016/0014-5793(88)80879-2. [DOI] [PubMed] [Google Scholar]
- Düzgüneş N., Shavnin S. A. Membrane destabilization by N-terminal peptides of viral envelope proteins. J Membr Biol. 1992 May;128(1):71–80. doi: 10.1007/BF00231872. [DOI] [PubMed] [Google Scholar]
- Evans E, Rawicz W. Entropy-driven tension and bending elasticity in condensed-fluid membranes. Phys Rev Lett. 1990 Apr 23;64(17):2094–2097. doi: 10.1103/PhysRevLett.64.2094. [DOI] [PubMed] [Google Scholar]
- Fattal E., Nir S., Parente R. A., Szoka F. C., Jr Pore-forming peptides induce rapid phospholipid flip-flop in membranes. Biochemistry. 1994 May 31;33(21):6721–6731. doi: 10.1021/bi00187a044. [DOI] [PubMed] [Google Scholar]
- Fields C. G., Lloyd D. H., Macdonald R. L., Otteson K. M., Noble R. L. HBTU activation for automated Fmoc solid-phase peptide synthesis. Pept Res. 1991 Mar-Apr;4(2):95–101. [PubMed] [Google Scholar]
- Fonteijn T. A., Engberts J. B., Nir S., Hoekstra D. Asymmetric fusion between synthetic di-n-dodecylphosphate vesicles and virus membranes. Biochim Biophys Acta. 1992 Oct 5;1110(2):185–192. doi: 10.1016/0005-2736(92)90357-r. [DOI] [PubMed] [Google Scholar]
- Gallaher W. R., Segrest J. P., Hunter E. Are fusion peptides really "sided" insertional helices? Cell. 1992 Aug 21;70(4):531–532. doi: 10.1016/0092-8674(92)90423-a. [DOI] [PubMed] [Google Scholar]
- Gething M. J., Doms R. W., York D., White J. Studies on the mechanism of membrane fusion: site-specific mutagenesis of the hemagglutinin of influenza virus. J Cell Biol. 1986 Jan;102(1):11–23. doi: 10.1083/jcb.102.1.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glushakova S. E., Omelyanenko V. G., Lukashevitch I. S., Bogdanov A. A., Jr, Moshnikova A. B., Kozytch A. T., Torchilin V. P. The fusion of artificial lipid membranes induced by the synthetic arenavirus 'fusion peptide'. Biochim Biophys Acta. 1992 Oct 5;1110(2):202–208. doi: 10.1016/0005-2736(92)90360-x. [DOI] [PubMed] [Google Scholar]
- Gray C., Tatulian S. A., Wharton S. A., Tamm L. K. Effect of the N-terminal glycine on the secondary structure, orientation, and interaction of the influenza hemagglutinin fusion peptide with lipid bilayers. Biophys J. 1996 May;70(5):2275–2286. doi: 10.1016/S0006-3495(96)79793-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grewe C., Beck A., Gelderblom H. R. HIV: early virus-cell interactions. J Acquir Immune Defic Syndr. 1990;3(10):965–974. [PubMed] [Google Scholar]
- Harter C., James P., Bächi T., Semenza G., Brunner J. Hydrophobic binding of the ectodomain of influenza hemagglutinin to membranes occurs through the "fusion peptide". J Biol Chem. 1989 Apr 15;264(11):6459–6464. [PubMed] [Google Scholar]
- Helenius A., Kartenbeck J., Simons K., Fries E. On the entry of Semliki forest virus into BHK-21 cells. J Cell Biol. 1980 Feb;84(2):404–420. doi: 10.1083/jcb.84.2.404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoekstra D., Klappe K., de Boer T., Wilschut J. Characterization of the fusogenic properties of Sendai virus: kinetics of fusion with erythrocyte membranes. Biochemistry. 1985 Aug 27;24(18):4739–4745. doi: 10.1021/bi00339a005. [DOI] [PubMed] [Google Scholar]
- Hoekstra D. Membrane fusion of enveloped viruses: especially a matter of proteins. J Bioenerg Biomembr. 1990 Apr;22(2):121–155. doi: 10.1007/BF00762943. [DOI] [PubMed] [Google Scholar]
- Hug P., Sleight R. G. Fusogenic virosomes prepared by partitioning of vesicular stomatitis virus G protein into preformed vesicles. J Biol Chem. 1994 Feb 11;269(6):4050–4056. [PubMed] [Google Scholar]
- Ishiguro R., Kimura N., Takahashi S. Orientation of fusion-active synthetic peptides in phospholipid bilayers: determination by Fourier transform infrared spectroscopy. Biochemistry. 1993 Sep 21;32(37):9792–9797. doi: 10.1021/bi00088a034. [DOI] [PubMed] [Google Scholar]
- Lear J. D., DeGrado W. F. Membrane binding and conformational properties of peptides representing the NH2 terminus of influenza HA-2. J Biol Chem. 1987 May 15;262(14):6500–6505. [PubMed] [Google Scholar]
- Matlin K. S., Reggio H., Helenius A., Simons K. Infectious entry pathway of influenza virus in a canine kidney cell line. J Cell Biol. 1981 Dec;91(3 Pt 1):601–613. doi: 10.1083/jcb.91.3.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClure M. O., Marsh M., Weiss R. A. Human immunodeficiency virus infection of CD4-bearing cells occurs by a pH-independent mechanism. EMBO J. 1988 Feb;7(2):513–518. doi: 10.1002/j.1460-2075.1988.tb02839.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melikyan G. B., Niles W. D., Peeples M. E., Cohen F. S. Influenza hemagglutinin-mediated fusion pores connecting cells to planar membranes: flickering to final expansion. J Gen Physiol. 1993 Dec;102(6):1131–1149. doi: 10.1085/jgp.102.6.1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mobley P. W., Lee H. F., Curtain C. C., Kirkpatrick A., Waring A. J., Gordon L. M. The amino-terminal peptide of HIV-1 glycoprotein 41 fuses human erythrocytes. Biochim Biophys Acta. 1995 Jun 9;1271(2-3):304–314. doi: 10.1016/0925-4439(95)00048-9. [DOI] [PubMed] [Google Scholar]
- Morris S. J., Sarkar D. P., White J. M., Blumenthal R. Kinetics of pH-dependent fusion between 3T3 fibroblasts expressing influenza hemagglutinin and red blood cells. Measurement by dequenching of fluorescence. J Biol Chem. 1989 Mar 5;264(7):3972–3978. [PubMed] [Google Scholar]
- Morsy M. A., Mitani K., Clemens P., Caskey C. T. Progress toward human gene therapy. JAMA. 1993 Nov 17;270(19):2338–2345. [PubMed] [Google Scholar]
- Needham D. Measurement of interbilayer adhesion energies. Methods Enzymol. 1993;220:111–129. doi: 10.1016/0076-6879(93)20078-h. [DOI] [PubMed] [Google Scholar]
- Needham D., Zhelev D. V. Lysolipid exchange with lipid vesicle membranes. Ann Biomed Eng. 1995 May-Jun;23(3):287–298. doi: 10.1007/BF02584429. [DOI] [PubMed] [Google Scholar]
- Plank C., Oberhauser B., Mechtler K., Koch C., Wagner E. The influence of endosome-disruptive peptides on gene transfer using synthetic virus-like gene transfer systems. J Biol Chem. 1994 Apr 29;269(17):12918–12924. [PubMed] [Google Scholar]
- Rafalski M., Ortiz A., Rockwell A., van Ginkel L. C., Lear J. D., DeGrado W. F., Wilschut J. Membrane fusion activity of the influenza virus hemagglutinin: interaction of HA2 N-terminal peptides with phospholipid vesicles. Biochemistry. 1991 Oct 22;30(42):10211–10220. doi: 10.1021/bi00106a020. [DOI] [PubMed] [Google Scholar]
- Simon S. A., Disalvo E. A., Gawrisch K., Borovyagin V., Toone E., Schiffman S. S., Needham D., McIntosh T. J. Increased adhesion between neutral lipid bilayers: interbilayer bridges formed by tannic acid. Biophys J. 1994 Jun;66(6):1943–1958. doi: 10.1016/S0006-3495(94)80988-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinangil F., Loyter A., Volsky D. J. Quantitative measurement of fusion between human immunodeficiency virus and cultured cells using membrane fluorescence dequenching. FEBS Lett. 1988 Oct 24;239(1):88–92. doi: 10.1016/0014-5793(88)80551-9. [DOI] [PubMed] [Google Scholar]
- Slepushkin V. A., Melikyan G. B., Sidorova M. S., Chumakov V. M., Andreev S. M., Manulyan R. A., Karamov E. V. Interaction of human immunodeficiency virus (HIV-1) fusion peptides with artificial lipid membranes. Biochem Biophys Res Commun. 1990 Oct 30;172(2):952–957. doi: 10.1016/0006-291x(90)90768-i. [DOI] [PubMed] [Google Scholar]
- Soltesz S. A., Hammer D. A. Micropipette manipulation technique for the monitoring of pH-dependent membrane lysis as induced by the fusion peptide of influenza virus. Biophys J. 1995 Jan;68(1):315–325. doi: 10.1016/S0006-3495(95)80190-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stegmann T., Delfino J. M., Richards F. M., Helenius A. The HA2 subunit of influenza hemagglutinin inserts into the target membrane prior to fusion. J Biol Chem. 1991 Sep 25;266(27):18404–18410. [PubMed] [Google Scholar]
- Stegmann T., Hoekstra D., Scherphof G., Wilschut J. Kinetics of pH-dependent fusion between influenza virus and liposomes. Biochemistry. 1985 Jun 18;24(13):3107–3113. doi: 10.1021/bi00334a006. [DOI] [PubMed] [Google Scholar]
- Steinhauer D. A., Wharton S. A., Skehel J. J., Wiley D. C. Studies of the membrane fusion activities of fusion peptide mutants of influenza virus hemagglutinin. J Virol. 1995 Nov;69(11):6643–6651. doi: 10.1128/jvi.69.11.6643-6651.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Struck D. K., Hoekstra D., Pagano R. E. Use of resonance energy transfer to monitor membrane fusion. Biochemistry. 1981 Jul 7;20(14):4093–4099. doi: 10.1021/bi00517a023. [DOI] [PubMed] [Google Scholar]
- Wagner E., Plank C., Zatloukal K., Cotten M., Birnstiel M. L. Influenza virus hemagglutinin HA-2 N-terminal fusogenic peptides augment gene transfer by transferrin-polylysine-DNA complexes: toward a synthetic virus-like gene-transfer vehicle. Proc Natl Acad Sci U S A. 1992 Sep 1;89(17):7934–7938. doi: 10.1073/pnas.89.17.7934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber T., Paesold G., Galli C., Mischler R., Semenza G., Brunner J. Evidence for H(+)-induced insertion of influenza hemagglutinin HA2 N-terminal segment into viral membrane. J Biol Chem. 1994 Jul 15;269(28):18353–18358. [PubMed] [Google Scholar]
- Wharton S. A., Martin S. R., Ruigrok R. W., Skehel J. J., Wiley D. C. Membrane fusion by peptide analogues of influenza virus haemagglutinin. J Gen Virol. 1988 Aug;69(Pt 8):1847–1857. doi: 10.1099/0022-1317-69-8-1847. [DOI] [PubMed] [Google Scholar]
- White J. M. Membrane fusion. Science. 1992 Nov 6;258(5084):917–924. doi: 10.1126/science.1439803. [DOI] [PubMed] [Google Scholar]
- White J. M. Viral and cellular membrane fusion proteins. Annu Rev Physiol. 1990;52:675–697. doi: 10.1146/annurev.ph.52.030190.003331. [DOI] [PubMed] [Google Scholar]
- Zhelev D. V., Needham D. Tension-stabilized pores in giant vesicles: determination of pore size and pore line tension. Biochim Biophys Acta. 1993 Apr 8;1147(1):89–104. doi: 10.1016/0005-2736(93)90319-u. [DOI] [PubMed] [Google Scholar]
- Zimmerberg J., Blumenthal R., Sarkar D. P., Curran M., Morris S. J. Restricted movement of lipid and aqueous dyes through pores formed by influenza hemagglutinin during cell fusion. J Cell Biol. 1994 Dec;127(6 Pt 2):1885–1894. doi: 10.1083/jcb.127.6.1885. [DOI] [PMC free article] [PubMed] [Google Scholar]




