Abstract
Introduction
Melanoma is the most serious form of skin cancer causing most of the skin cancer-related deaths. The incidence of melanoma has risen so dramatically over past few years that no other solid or blood malignancy comes close to it in terms of increased incidence. The main problem associated with the treatment of melanoma is low response rate to the existing treatment modalities, which in turn is due to the incomplete response by chemotherapeutic agents and inherent resistance of melanoma cells.
Materials and Methods
Conventional therapeutic strategies, as well as, recent literature on melanoma have been thoroughly studied. This review summarizes the base of anti-melanoma treatment with conventional chemotherapeutic drugs, followed by an account of recent studies which explored the potential of nanotechnology and newer strategies and agents in melanoma treatment.
Conclusion
Although melanoma is curable if detected in its early localized form, metastatic melanoma continues to be a therapeutic challenge. Metastatic melanoma has a very poor prognosis and conventional therapies have not improved the outcomes of the treatment so far. For this reason, newer combinations of anti-melanoma drugs and newer strategies utilizing nanotechnology have been constantly explored.
Keywords: Melanoma treatment, Dacarbazine, Targeted therapy, Immuno-therapy, Anti-melanoma, Nano-agents, Nanotechnology
Introduction
The cancerous growth of melanocytes (melanin producing cells) is termed Melanoma (Mandalà and Voit 2013). Skin cancer, both melanoma and non-melanoma, is the most common cancer in Caucasian population (Apalla et al. 2017). Melanoma is the most aggressive and deadly type of skin cancer. Though it accounts for only 4% of all skin cancers, it causes the highest number of skin cancer-related deaths worldwide. Incidence of Primary Cutaneous Melanoma in Caucasian populations has been constantly rising and is reported to double up every 10–14 years (Mandalà and Voit 2013). The estimated number of new cases of melanoma in 2017 is 87,110, which accounts for 5.2% of all new cancer cases. Estimated deaths in 2017 are 9730 (National Cancer Institute. SEER Stats Fact Sheets: Melanoma of the skin. http://seer.cancer.gov/statfacts/html/melan.html. Accessed on 25th January, 2018). Moreover, after lung cancer and breast cancer, melanoma is the third most common reason of brain metastases. Melanoma spreads to brain in up to 75% of melanoma patients. Brain metastases cause death in 95% of total cases (Nicholas et al. 2013).
In this review, different anti-melanoma strategies are discussed; but to understand the mechanism and utility of different treatment approaches in melanoma, a brief understanding of pathophysiology of melanoma is necessary.
c-Kit
c-Kit is a type III transmembrane receptor tyrosine kinase. Its ligand binding causes autophosphorylation and activation of several signaling pathways, thus mediates growth, proliferation, and metastasis of cancer cells, and inhibits apoptosis (Carvajal et al. 2011). The pathways activated include mitogen-activated protein kinase (MAPK), phosphatidylinositol 3-kinase (PI3K)-AKT1, and JAK-STAT-signaling pathways. c-Kit mutations are reported in 39% mucosal, 36% acral, and 28% chronic sun-damaged melanomas (Curtin et al. 2006).
RAS
RAS-signaling cascade is known to promote proliferation, survival, and invasion through two pathways, MAPK pathway and PI3K pathway. NRAS was the first component found to be activated in this pathway, and is mutated in 15–20% of all melanomas (Nikolaou et al. 2012).
RAF
BRAF is the most commonly mutated oncogene in melanoma (60% of all melanomas). Most prevalent mutation in BRAF is BRAFV600E which leads to activation of downstream protein kinases (MEK and ERK) and increased proliferation of melanoma cells (Nikolaou et al. 2012).
Challenges
Advanced malignant melanoma has a poor prognosis and chemotherapy is most of the times not effective because of the resistance of melanoma cells. According to several studies, the mechanisms of resistance in melanoma are different from hematological tumors. Though the causes of the chemoresistant phenotype are unknown, it appears that differences in the apoptotic pathways leading to apoptotic deficiency are accountable (Helmbach et al. 2001). Thus, high prominence of resistance and its multiple and complicated reasons keep pushing scientists to look for newer and efficient strategies for the treatment of melanoma.
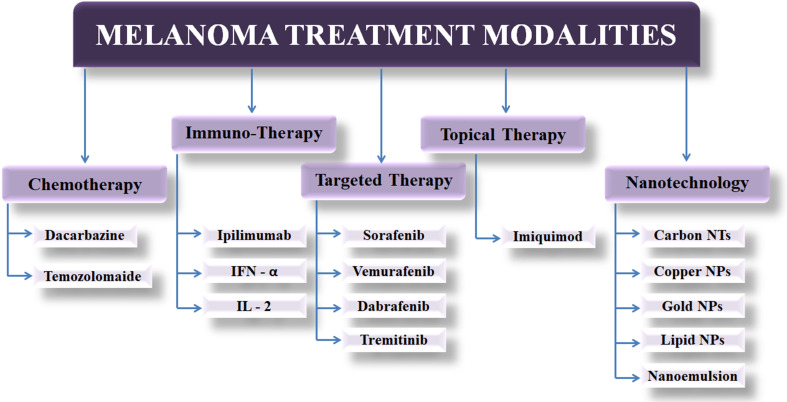
Figure 1 classifies different anti-melanoma modalities into relevant categories.
Fig. 1.
Classification of melanoma treatment modalities
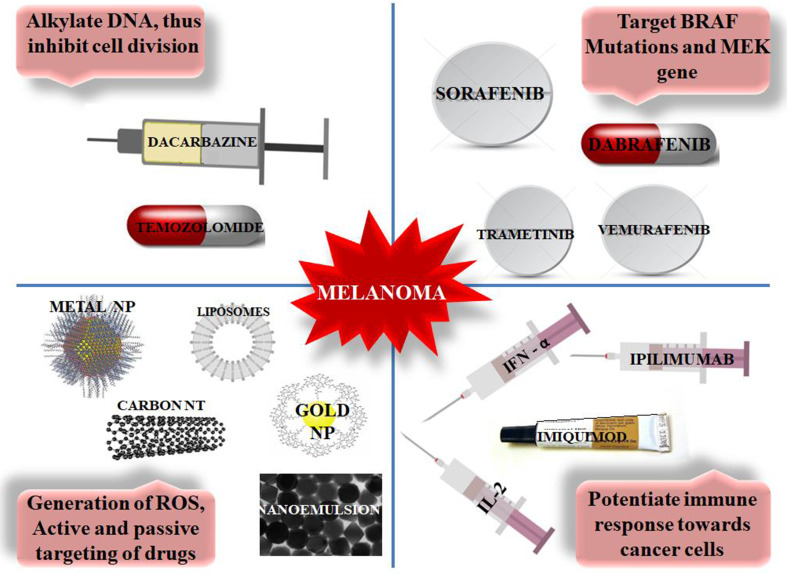
Figure 2 briefly summarizes the mechanism of action of different drugs and agents used in melanoma treatment.
Fig. 2.
Anti-melanoma drugs and agents: oldest treatment of melanoma is with chemotherapy (dacarbazine and temozolomide) which inhibit cancer cells growth by alkylating DNA. Targeted agents are those that target specific gene or mutation such as BRAF and MEK. Immunotherapeutic agents potentiate the immune response of the patient towards cancer cells damaged by chemotherapy. Nano-agents are the current interest and have shown promising results. They act by generating ROS and targeting therapeutic agents to the target site. NP nanoparticle, NT nanotube
Chemotherapy
Out of the conventional therapies for melanoma, including surgery and radiation therapy, chemotherapy always had a major role to play. Following is an account of the chemotherapeutic agents that are specifically used against melanoma, though other chemotherapeutic drugs are also explored, in combinations or alone.
Dacarbazine
Dacarbazine (DTIC) is an alkylating chemotherapeutic agent. Dacarbazine was approved by FDA in 1975 for treatment of melanoma and is the only FDA approved anti-melanoma chemotherapeutic drug until date. Several studies have been performed on efficacy of dacarbazine as anti-melanoma agent, and most of them have revealed that dacarbazine, as a single agent, offers poor overall survival benefit (Bhatia et al. 2009). Serrone et al. (2000) reported median response duration of 5–6 months; objective response rate of 20%; and complete response rate of only 5%. Middleton et al. (2000) reported median response duration of 6.4 months and Lui et al. (2007) reported objective response rate of 15.3%. Positive responses, if any, are not durable and less than 2% patients stay alive until 6 years after treatment (Hill et al. 1984).
However, despite its modest efficacy, dacarbazine still continues to be the standard treatment for metastatic melanoma and forms the basis of majority of anti-melanoma combinations. Out of several therapies that have been tried against melanoma until date, no one has shown significant benefit over dacarbazine (Bhatia et al. 2009). In addition, in a study performed on a small group of patients with lung metastases, good performance status, normal blood lactate dehydrogenase enzyme levels, and long-term disease control with a good quality of life were observed after treatment with dacarbazine (Coates and Segelov 1994).
Temozolomide
Temozolamide is an analog of dacarbazine which can be administered orally. It has been studied as anti-melanoma drug and as a substitute of dacarbazine for its convenience of oral intake. Like dacarbazine, it also gets converted to active alkylating metabolite MTIC (3-methyl-[triazen-1-yl]-imidazole-4-carboxamide); though unlike dacarbazine, this conversion takes place spontaneously at physiological pH in all tissues which is why this drug has good oral bioavailability. In addition, temozolamide can penetrate into CNS and thus has potential to prevent and treat melanoma brain metastasis (Bhatia et al. 2009; Velho 2012).
Middleton et al. (2000) studied the response of temozolomide vs dacarbazine, and found effect of temozolamide to be statistically similar to dacarbazine. In another multicenter phase III trial study, dacarbazine treatment was compared with extended dosing schedule of temozolomide (Patel et al. 2011). This study also concluded that there is no significant difference between efficiency of dacarbazine and temozolamide as anti-melanoma drugs. Resultant of this fact, dacarbazine has been replaced by temozolamide in several combinations of chemotherapy/biochemotherapy regimens. Quirt et al. (2007) have reported that using temozolamide as substitute of dacarbazine may play a vital role in preventing CNS relapse; but Chiarion-Sileni et al. (2008) compared cisplatin/IL-2/temozolomide vs cisplatin/IL-2/dacarbazine and found that temozolamide did not show any advantage in prevention of brain metastasis. Although, it is still believed that increased survival rates can be achieved in melanoma patients having brain metastasis when temozolomide is used as monotherapy or with brain radiation therapy (Hofmann et al. 2006).
Since temozolamide failed to show any therapeutic advantage over dacarbazine, choice between two drugs is made on the basis of preferred route of administration, cost factor, availability, and presence of brain metastasis (Bhatia et al. 2009).
Immuno-therapy
Melanoma cells exhibit intrinsic resistance against chemotherapy through several mechanisms (Helmbach et al. 2001), to circumvent which, researchers keep on looking for different therapeutic strategies. One such approach is to potentiate the immune response against this highly immunogenic form of skin cancer (Di Franco et al. 2017). Immuno-therapy improves immune response of melanoma patients and increases the clearance of cancer cells damaged by cytotoxic drugs. Most commonly used immuno-stimulators for treatment of melanoma are interleukin (IL)-2, interferon (IFN)-alpha, ipilimumab, and thymosin alpha 1 (Chen et al. 2013).
Ipilimumab
Ipilimumab is a monoclonal antibody which blocks CTLA-4 receptors. CTLA-4 transmembrane receptors belong to immunoglobulin superfamily and are overexpressed by T-lymphocytes (Franklin et al. 2017). Ipilimumab was approved by FDA as second-line treatment for melanoma in 2011 after Hodi et al. (2010) reported improved survival rates in ipilumumab treated melanoma patients, who also claimed that ipilimumab was the first agent to show significantly improved survival rate in metastatic malignant melanoma patients. A number of phase I/II/III studies have been conducted with different doses of ipilimumab which have yielded higher median overall survival (OS) and response rate, establishing a positive correlation with increasing dose of ipilimumab (Hodi et al. 2010; Robert et al. 2011).
Another CTLA-4 blocker is tremelimumab (ticilimumab), but it did not show any survival benefit over chemotherapy (Franklin et al. 2017).
IFN-α
IFNs are cytokines released in response to pathogens or in presence of cancer cells. Consequent to improved OS in first clinical trial using high doses of IFN-α2b (J M Kirkwood et al. 1996), IFN- α was approved by US FDA in 1996 for use in adjuvant therapy for patients that possess high risk of reoccurrence after melanoma resection (Di Franco et al. 2017). Another clinical trial comparing high doses to low doses of IFN revealed improved Risk Free Survival (RFS) with no significant alteration in OS (Kirkwood et al. 2000), while another clinical trial comparing high doses of IFN with vaccines with the ganglioside GMK reported improved RFS as well as improved OS (Kirkwood et al. 2001).
Interleukin-2 (IL-2)
IL-2 was approved by FDA in 1998 for treatment of metastatic melanoma. It is a lymphokine which acts by stimulating T-cell proliferation and function (Velho 2012). Rate of complete response achieved by high dose of IL-2 was 6%, while partial response rate was 10% (Atkins et al. 1999, 2000). Better results in terms of therapeutic responses have been achieved with high doses and intravenous administration but brought along increased toxicities also, and low doses or subcutaneous administration gave poorer therapeutic results (Keilholz et al. 1998). IL-2 has been tested in combination with chemotherapy and has shown some therapeutic benefits (Table 1). Famotidine has been reported to lower the toxicity of IL-2 by enhancing lymphokine-activated killer cell activity while retaining the anti-melanoma activity (Quan et al. 2012).
Table 1.
Therapeutic outcomes of anti-melanoma combinations
| Drugs | Therapeutic outcomes | References |
|---|---|---|
|
BOLD Bleomycin Vincristine Lomustine Dacarbazine |
Complete remission of brain metastasis in three patients with stage 4 melanoma Clinical benefit in six out of eight patients Median survival: 12.5% months |
Bottoni et al. (2005) |
|
CVD Cisplatin Vinblastine Dacarbazine |
Response rate: 21% Increased to 33% after addition of IL-2 and IFN-α-2b No clear survival benefit |
Bajetta et al. (2006) |
|
DBDT Dacarbazine Carmustine Cisplatin Tamoxifen |
ORR: 26% Median survival: 9 months Improved ORR and median survival as compared to dacarbazine monotherapy However, increased toxicity with limited effect on OS |
Sileni et al. (2001) |
| DBDT + low dose of IL-2 |
Response rate increased to 32.5% Overall response increased to 11.3 months |
Su et al. (2011) |
|
Temozolomide + IFN-α-2b vs Temozolomide |
Response rate: 24 vs 13% Median OS: 9.7 months vs 8.4 months |
Kaufmann et al. (2005) |
| Temozolomide + thalidomide |
6-month PFS: 15% 1-year OS: 35% Response rate: 13% |
Clark et al. (2010) |
|
Vemurafenib + cobimetinib vs Vemurafenib |
PFS was 9.89 months as compared to 6.21 months with vemurafenib alone OS was 22.28 months as compared to 17.38 months ORR was 67.6% as compared to 44.8% |
Ascierto et al. (2015) |
|
Dabrafenib + trametinib (CombiDT) vs Dabrafenib |
Proportion of responses: 76% (combination) vs 54% (dabrafenib alone) PFS: 9.4 vs 5.8 months Median OS: 23.8 months for CombiDT group BRAF inhibitor class toxicities occurred less frequently with CombiDT than with dabrafenib monotherapy |
Flaherty et al. (2012a, b) |
|
Dabrafenib + trametinib vs Dabrafenib |
PFS only slightly improved (9.3 vs 8.8 months) Confirmed response by 140 patients vs 108 patients out of 210 subjects No significant difference between frequency of side effects |
Long et al. (2014) |
|
Dabrafenib + trametinib vs Vemurafenib |
PFS: 11.4 vs 7.3 months Overall response: 226 patients vs 180 patients Duration of response: 13.8 vs 7.5 months |
Grob et al. (2016) |
| Vemurafenib + cobimetinib |
Confirmed objective responses in 15% patients who had recently progressed on vemurafenib; median PFS: 2.8 months Confirmed objective responses in 87% patients who had never received BRAF inhibitor; Complete response: 10%; Median PFS: 13.7 months Safe and tolerable Promising antitumor activity in advanced BRAFV600-mutated melanoma, particularly in those who have never received a BRAF inhibitor |
Ribas et al. (2014) |
| Imiquimod + 5-fluorouracil |
Effective Patients did not develop new, distant lesions |
Florin et al. (2012) |
| Imiquimod + IL-2 |
Complete local response rate with long-term follow-up (24 months) Absence of malignant cells |
Shi et al. (2015a, b) |
| Imiquimod + multi-peptide cancer vaccine |
Well tolerated Circulating T-cell responses to vaccine was detected Treatment of metastases with imiquimod induced immune cell infiltration and favorable gene signatures in patients with circulating T-cell responses |
Mauldin et al. (2016) |
The above-mentioned many other immunocytokines have been known to be effective in melanoma treatment, but their use is limited due to severe side effects (Chen et al. 2013).
Topical therapy (imiquimod)
Imiquimod is a topically applied agent which acts via activation of toll-like receptor 7 and 8 (TLR7/8). Activated TLR7 induces production of different cytokines such as IFN-α, IL-12, and TNF, thus activating the innate immune system (Aspord et al. 2014).
Imiquimod has been reported to be an effective and safe treatment for metastases of melanoma. Though it does not cease progression, but it controls cutaneous metastases spreading from primary melanoma (Sisti et al. 2014). Moon and Spencer (2013) have reported that topical imiquimod therapy completely cleared an invasive melanoma of 2.75 mm depth in situ (used after surgical excision of melanoma). Sue et al. (2014) also reported no evidence of residual melanoma in situ when imiquimod was used after surgical excision. However, the efficiency of imiquimod remains unclear in the treatment of subcutaneous metastases, which may even keep progressing despite complete treatment of superficial dermis (Turza et al. 2010).
Targeted therapy
Owing to the highly resistant nature of the melanoma, the need of the targeted therapies arises which would specifically target the particular mechanisms of melanoma occurrence and progression.
BRAF inhibitors
As stated earlier, BRAF is the most frequently mutated oncogene in melanoma and induces constitutive activation of RAS–RAF–MEK–ERK-signaling cascade (Mandalà and Voit 2013).
BRAF inhibitors induce rapid regression of melanoma metastases, and response evaluation criteria in solid tumors (RECIST) has reported positive response in 50–60% patients (Menzies and Long 2014).
Sorafenib
Sorafenib is a multi-targeted tyrosine kinase inhibitor of Braf, Craf, platelet-derived growth factor receptor (PDGFR), vascular endothelial growth factor receptor (VEGFR), p38, and c-Kit. It was the first Raf inhibitor actively studied in melanoma patients (Mandalà and Voit 2013). Despite being studied in several Phase I/II/III trials, sorafenib has not yielded any promising results, neither as single agent, nor in combinations (Eisen et al. 2006; Mandalà and Voit 2013). The addition of sorafenib to carboplatin/paclitaxel was found to be of no benefit in terms of median progression free survival (PFS) or OS (Flaherty et al. 2010a, b; Hauschild et al. 2009).
Vemurafenib
Vemurafenib is an orally active BRAF inhibitor, 30 times more selective towards mutated BRAF compared to wild-type BRAF. Vemurafenib has shown anti-proliferative activity in melanoma cell lines with BRAFV600E mutation, and it was the first BRAF inhibitor which completed phase I testing with significant clinical benefit (Flaherty et al. 2010a, b; Mandalà and Voit 2013). Vemurafenib exhibited partial response and complete response too in BRAF-mutated melanoma patients, while no response was observed in patients without mutated BRAF gene (Flaherty et al. 2010a, b). In further phase II study, vemurafenib monotherapy resulted in overall response rate (ORR) of 53% and complete response of 5%; although the side effects such as arthralgia, photosensitivity, fatigue, and alopecia were observed (Sosman et al. 2012). In a phase III trial, vemurafenib resulted in improved OS as compared to dacarbazine (84 vs 64%) (Chapman et al. 2011). This revelation of clinical superiority of vemurafenib over dacarbazine led to the approval of vemurafenib by FDA in 2011 and by Health Canada in 2012. The limitation of vemurafenib is the quick development of resistance by melanoma cells (Rajakulendran and Adam 2014).
Dabrafenib
Dabrafenib is a highly potent BRAF inhibitor having 100 times greater selectivity for mutated BRAFV600E compared to wild-type BRAF gene (Mandalà and Voit 2013). It was approved by FDA in 2013 (Rajakulendran and Adam 2014). In addition to objective responses in BRAFV600E mutant patients, dabrafenib has also shown significant clinical activity against brain metastasis (Falchook et al. 2012). Another randomized, controlled phase III trial in patients with BRAF V600E mutated metastatic melanoma has claimed the efficacy of dabrafenib to be similar to vemurafenib in terms of PFS and response rates (Hauschild et al. 2012).
Major limitations in using BRAF inhibitors are the development of resistance in almost all cases and the toxicity associated with BRAF inhibition (Menzies and Long 2014).
MEK inhibitors
There are strong evidences in favor of the concept that mutation of BRAF is associated with increased selectivity and sensitivity to inhibitors of MEK as well, which is also a gene in the same signaling cascade (Mandalà and Voit 2013).
Trametinib
Trametinib is an orally active selective inhibitor of MEK. In a phase III study comparing the efficacy of trametinib with chemotherapy (dacarbazine or paclitaxel), median PFS was found to be 4.8 months with trametinib, while it was only 1.5 months in the chemotherapy group (Gilmartin et al. 2011). In another study, the 6 months OS with trametinib was 81% as compared to 67% with chemotherapy (Flaherty, Robert et al. 2011). As a result of the promising outcomes of trametinib therapy, the drug was approved by FDA in 2013 for the treatment of metastatic melanoma which involve BRAFV600E or BRAFV600K mutations (Rajakulendran and Adam 2014).
Combinatorial approach
Combinational therapy is the most common strategy towards treatment of melanoma these days due to the aggressive nature of this cancer and insufficient effect produced by dacarbazine and other chemotherapeutic drugs when used alone. Dacarbazine, though shows low and insufficient activity, still forms the basis of most of the combination chemotherapy regimens for treatment of melanoma. Dacrabazine can be combined with other cytotoxic drugs such as cisplatin, nitrosoureas, and tubular toxins (Blesa et al. 2011; Velho 2012).
The combination of BRAF and MEK inhibitors is also well established for use in patients having melanoma with BRAF mutations. This is mainly due to the development of resistance with use of BRAF inhibitors as single agent (Apalla et al. 2017). BRAF inhibitors are often used in combination with MEK inhibitors as the combination is reported to delay the onset of resistance and enhance apoptosis in comparison with BRAF inhibitor as single agent (Paraiso et al. 2010). Clinical trials performed on BRAF-mutant metastatic melanoma patients using BRAF and MEK inhibitors in combinations have revealed prolonged PFS, improved response rate, and safer toxicity profiles. First combination of BRAF and MEK inhibitors to be tried in clinical studies was dabrafenib and trametinib (CombiDT). US FDA granted accelerated approval for CombiDT in patients with BRAFV600E/K metastatic melanoma after promising results were obtained in phase II studies (Menzies and Long 2014). Another clinical trial is ongoing on the effect of dabrafenib plus trametinib in adjuvant treatment of high-risk BRAFV600 mutation-positive melanoma after surgical resection (NCT01682083). Although majority of BRAF-mutant cases respond most rapidly to the combination of BRAF and MEK inhibitors, there are non-responding or relapsing patients too (Hegedũs et al. 2017).
Table 1 summarizes therapeutic outcomes of the combinations that have been tried against melanoma.
Nanotechnology in melanoma
Despite the rigorous efforts in the field of melanoma treatment, melanoma continues to be a major health challenge due to its resistant and aggressive nature. This leaves a huge scope and need of trying newer and wiser strategies to fight against this deadly disease. Just as the nanotechnology is invading every part of the biomedical sphere, treatment of melanoma is not untouched either. Several types of nanoparticles and nanovesicles have been explored for their applications in the treatment, either as drug carriers or as anti-melanoma agents themselves.
Carbon nanotubes
Carbon nanotubes are continuous cylindrical form of one (single-wall) or more (multi-wall) layers of graphene (allotrope of carbon) with open or closed ends (Volder et al. 2013).
Sobhani et al. (2017) recently studied the efficiency of PEGylated oxidized carbon nanotubes in reducing the size of melanoma tumor after photothermal therapy. The average size of tumor in mice receiving carbon nanotubes was greatly decreased in comparison with those which received laser therapy alone.
Carbon nanotubes have also been used as delivery systems. Siu et al. (2014) used carbon nanotubes to deliver siRNA to melanoma mouse model. Significant uptake of Cy3-labeled siRNA specific to Braf and gene silencing in tumor tissue was found. Attenuation of tumor growth was achieved in 25 days.
Few researchers have utilized carbon nanotubes as an aid for immuno-therapy in different interesting ways. Fadel et al. (2014) prepared carbon nanotube–polymer composite and successfully used this composite for generating large numbers of cytotoxic T cells from mice for cancer immuno-therapy. Significant number of T cells was obtained using a 1000 times less IL-2. T cells obtained delayed tumor growth in murine melanoma model.
Copper nanoparticles
Different experiments have studied the extent and mechanism of copper nanoparticles as anti-melanoma agent. Chakraborty and Basu (2017) have recently presented evidences of killing of A375 melanoma cells by copper nanoparticles due to lowering of cell membrane rigidity, DNA degradation, chromosomal condensation, cell cycle arrest in the G2/M phase, depolarization of mitochondrial membrane, and cellular apoptosis in caspase-9-mediated intrinsic pathway. Another recent study has shown that cuprous oxide nanoparticles induced apoptosis of human melanoma stem cells (CD271+/high) in A375 and WM266-4 melanoma cell lines and significantly suppressed the expression of MITF, SOX10, and CD271 involved in maintenance and tumorigenesis of melanoma stem cells. Consequently, growth of melanoma was suppressed in tumor-bearing non-obese diabetic–severe combined immunodeficiency (NOD–SCID) mice, accompanied with tumors structural necrosis and fibrosis (Yu et al. 2017). Mukhopadhyay et al. (2018) investigated the therapeutic efficacy of copper nanoparticles in B16F10 melanoma cells and in mice-bearing B16F10 melanoma. Copper nanoparticles induced cell death by inducing oxidative stress. Abundant of apoptotic and cell cycle arrest proteins were seen in treated cell samples. A significant decrease in tumor growth was observed in tumor-bearing mice.
Wang et al. (2013) used cuprous oxide nanoparticles to treat mouse subcutaneous melanoma, where nanoparticles significantly reduced the tumor growth, inhibited metastasis of tumor cells, and increased the survival rate of mice. Nanoparticles were rapidly cleared from organs and exhibited little systemic toxicity. In another study, plasmonic copper sulfide (Cu2-xS) nanocrystals exhibited high photothermal efficacy in addition to intrinsic NIR-induced photodynamic activity by generating high levels of reactive oxygen species (ROS) (Wang et al. 2015).
The potential of copper oxide nanoparticles has also been investigated in treatment of uveal melanoma. Nanoparticles were internalized by uveal melanoma cells through lipid raft-mediated endocytosis, selectively inhibiting cancer cell growth and impairing the ability of uveal melanoma cell migration, invasion and cytoskeleton assembly, probably through damaging mitochondria, and autophagolysosomes and lysosomes, leading to elevated ROS level and over-stimulated apoptosis and autophagy (Hongyuan Song et al. 2015a, b).
Gold nanoparticles
The dynamic gold nanoparticles have been largely explored for drug carrying in addition to their photodynamic and photothermal therapeutic potential.
Zhang et al. (2015) have reported the efficacy of doxorubicin-loaded gold nanoparticles in both murine B16 and human SK-MEL-28 tumors in mice when given by intratumoral injection. Apoptosis and necrosis were revealed to be responsible for the anti-tumor action. Gold glyconanoparticles coupled to listeriolysin O peptide 91-99 (GNP-LLO91-99) have been used as novel adjuvant therapy for early stage melanoma patients. Single vaccination prevented melanoma growth and formation of lung metastases, and improved OS in mice. GNP-LLO91-99 also played the role of anti-neoplastic nanodrug as it reduced uncontrolled melanoma growth (Calderon-Gonzalez et al. 2017). Liang et al. (2017) introduced novel nanovaccine of liposome-coated gold nanocages modified with dendritic cell-specific antibody aCD11c for targeted delivery of melanoma antigen peptide TRP2 to promote activation and maturation of dendritic cells, and enhance tumor-specific T-lymphocyte responses. Nanovaccine exhibited enhanced anti-tumor immune response to inhibit tumor growth and metastasis in B16-F10 prophylactic models. Labala et al. (2017) delivered STAT3 siRNA and imatinib mesylate co-loaded in layer-by-layer assembled gold nanoparticles to melanoma-bearing mice. Significant reduction in tumor volume and suppression in STAT3 protein expression were achieved. Equally promising results are obtained in a recent similar experiment performed on curcumin liposomes (Jose et al. 2018). In another interesting study, gold nanoparticles were utilized to deliver Cas9-sgPlk-1 plasmids. Nanoformulation could enter the tumor cells and release Cas9-sgPlk-1 plasmids into cytosol by laser-triggered thermo-effects of gold nanoparticles. This way, the plasmids could reach nuclei which enabled the knock-out of target gene, thus resulting in inhibition of melanoma (Wang et al. 2018).
Camerin et al. (2016) studied photodynamic ability of gold nanoparticles by administering the conjugate of Zn(Ii)-phthalocyanine derivative photosensitizer and PEGylated gold nanoparticles in tumor melanoma-bearing mice. Tumor destruction was achieved in just 3 h. Remarkably, 40% of the treated animals showed complete survival with no regrowth of tumors. Li et al. (2016) prepared hydrogel co-loaded with doxorubicin and gold nanoparticles for intratumoral injection, which caused significant reduction in tumor sizes of mice. Role of gold nanoparticles here was of radiosensitization.
In addition to photodynamic therapy, several studies have been conducted to explore the potential of gold nanoparticles in photothermal therapy as well. Based on the results of the studies performed on B16F10 melanoma-bearing mice and comparisons done between animals groups receiving gold–ferrite nanocomposites along with laser irradiation, nanocomposites without irradiation, and only irradiations, Heidari et al. (2016) concluded that combining laser with gold–ferrite nanocomposites causes significant destruction of melanoma cells in animal model. Another recent study indicating the promising photothermal therapeutic effect of laser-stimulated PEGylated gold nano-semicubes was performed by Abo-Elfadl et al. (2016). Inflammatory mediators, nitric oxide, and cycloxygenase-2 were inhibited in mice after treatment with gold nano-semicubes, accompanied by laser exposure. Inhibition of pro-angiogenic factor VEGF was observed in addition to the induction of histone acetylation and apoptosis in tumor-bearing groups, all resulting in decreased tumor volume.
On the contrary to the above results, Jiménez Pérez et al. (2017) have recently reported gold nanoparticles to be nontoxic to murine melanoma cells. They synthesized gold and silver nanoparticles using ginseng berry extract as a reducing and capping agent. They found that gold nanoparticles (1–100 µg/ml) did not show any significant cytotoxicity in human dermal fibroblast as well as murine melanoma cell lines. However, silver nanoparticles were toxic to melanoma cells while nontoxic to fibroblasts. In addition, the cytotoxicity of biosynthesized nanoparticles was less than previously reported biogenic gold and silver nanoparticles.
Lipid nanoparticles and liposomes
Lipid nanoparticles, being a good carrier of therapeutic agents, have been used for carrying anti-melanoma agents to the target tissues.
Doxorubicin-loaded solid lipid nanoparticles (SLNs) have shown superior results as compared to doxorubicin solution both in vitro and in vivo (Tupal et al. 2016). Folic acid-conjugated poly(d,l-lactide-co-glycolide)–lipid composites have been used for targeted delivery of paclitaxel. The nanocarriers demonstrated efficient suppression in cell viability in comparison with free paclitaxel. Surface labeling with folic acid improved tumor uptake by approximately 4.41–12.8-fold. Improved tumor inhibition with no relapse and no remarkable systemic in-vivo toxicity was achieved (Wang and Sheng 2017). In another recent study, lipid nanocapsules were used to combine two anti-melanoma strategies, gene therapy to modulate anti-apoptotic proteins by use of Bcl-2 siRNA, and ferrocifens as novel anticancer agent. Specific extinction of Bcl-2 in melanoma cells demonstrated efficient gene silencing achieved by lipid nanocapsules. In addition, ferrocifens showed higher toxicity than dacarbazine. 50% reduction in tumor volume was achieved in tumor-bearing animals as compared to the control group (Resnier et al. 2017).
Drewes et al. (2016) prepared lipid-core nanocapsules using poly(ε-caprolactone), capric/caprylic triglyceride, and sorbitan monostearate and investigated their effects on human SK-Mel-28 melanoma cells and in melanoma induced C57B6 mice. Nanoparticles penetrated into SK-Mel-28 cells, and remained in cytoplasm. In-vitro treatment induced late apoptosis and necrosis, reduced cell proliferation, and delayed the cell cycle. Daily intraperitoneal or oral treatment in melanoma induced mice reduced the volume of tumor; however, intraperitoneal treatment caused toxicity, while oral treatment did not inhibit tumor development.
Liposomes have intrinsic ability of carrying lipophilic as well as hydrophilic drugs with equal ease and efficiency. Dorrani et al. (2016) demonstrated for the first time that the passive delivery of an edge-activated liposomal formulation can effectively carry siRNA through the stratum corneum and deposit it at the lower epidermis/upper dermis.
Sadhu et al. (2017) utilized liposomes to deliver glutathione disulfide to melanoma cells to increase intracellular glutathione disulfide. This increase affected cell detachment, migration, invasion, and adhesion stages of cancer metastasis. Consequently, these liposomes retarded tumor proliferation by 85–90% in melanoma-bearing mice, which was much more effective than dacarbazine. Median survival rate was also significantly improved. In another study, clodronate-containing liposomes significantly reduced the volume of primary tumors in B16/F10 melanoma-bearing mice by lowering the plasma levels of IL-10, Mo KC, TNF-α, VEGF, and PDGF-bb (Piaggio et al. 2016). Alupei et al. (2015) utilized liposomes to lower the anti-tumor dose of simvastatin. Simvastatin loaded liposomes inhibited B16.F10 melanoma growth by 85%, whereas free simvastatin at equivalent dose had no effect. This implied that liposomes resulted in better targeting of the drug to the tumor site.
Deng et al. (2017) explored the possibility of surface coating of liposomes. They administered hyaluronic acid (HA)-coated liposomes loaded with doxorubicin which were more efficiently internalized by B16F10 melanoma cells due to HA-CD44 receptor interaction. These liposomes showed more significant anti-melanoma activity than doxorubicin-loaded conventional liposomes in in-vivo studies. In addition, unwanted toxicities of doxorubicin such as myelosuppression and cardiotoxicity were also decreased due to better targeting. Shi et al. (2015a, b) designed pH-responsive liposomes containing paclitaxel and cell-penetrating peptide with the ability of active targeting to integrin αvβ3. In addition to significantly better targeting and uptake by B16F10 cells, tumor inhibition rate of 85% and higher survival rate in B16F10 tumor-bearing mice were also achieved. Saha et al. (2017) used α4β1 integrin targeted liposomes to encapsulate photosystem (protein complex present in chloroplasts which generates ROS causing apoptosis) and found them to be more potent than doxorubicin, achieving specific killing of cancer cells without affecting normal cells. Chen et al. (2016) significantly enhanced the anti-melanoma activity of siRNA through folate-coated liposomes. Liposomes substantially induced apoptosis of hypoxia-tolerant melanoma cells. The lectin from Lotus tetragonolobus (LTL) has also been used for surface functionalization to improve penetration into cells. LTL-functionalized liposomes loaded with doxorubicin greatly increased intracellular delivery of the drug to the cytoplasm of tumor cells (Della Giovampaola et al. 2017). Dicheva et al. (2015) synthesized doxorubicin-loaded thermosensitive liposomes which showed specific accumulation in melanoma vasculature. Hyperthermia caused massive release of the drug and its increased uptake in tumors.
Gowda et al. (2017) followed the combinatorial approach and developed nanoliposomes containing celecoxib and plumbagin (CelePlum-777). CelePlum-777 killed melanoma cells more effectively than normal cells and inhibited xenograft melanoma tumor growth by up to 72% without apparent toxicity. Drug combination in CelePlum-777 led to enhanced inhibition of melanoma by decreasing the levels of key cyclins important for cancer cell proliferation and survival. C8-ceramides loaded liposomes co-loaded with curcumin were found to reverse multidrug resistance. Mouse tumor growth inhibition study revealed synergistic therapeutic benefit of simultaneous delivery of curcumin and ceramide to tumor vasculature. Tumor growth was mediated via inhibition of PI3K-Akt signaling pathway (Barui et al. 2016).
Yang et al. (2016) introduced a new concept of immuno-chemotherapy combination by formulating paclitaxel and alpha-galactosylceramide (αGC) co-loaded TH peptide-modified liposomes. Paclitaxel initiated cytotoxicity attack in tumor cells, and meanwhile, αGC triggered anti-tumor immune response at spleen tissue. This formulation also adjusted the in-vivo immune status and induced more remarkable systemic anti-tumor immunity that could further suppress the growth of tumor. Neumann et al. (2016), to reduce immuno-suppressive cell populations, administered anti-melanoma long-peptide vaccine, and licofelone (non-steroidal anti-inflammatory drug) loaded in cationic liposomes to mice which showed delayed tumor growth compared to mice given the vaccine alone. Song et al. (2015a, b) prepared Vincristine containing immunoliposomes conjugated to CD20 antibodies (VCR-Lip-CD20). CD20 is a phenotype of melanoma cancer initiating cells (CIC) and is responsible for melanoma drug resistance. These liposomes could selectively kill CD20+ melanoma CIC in populations of WM266-4 cells both in vitro and in vivo.
GuhaSarkar et al. (2016) developed injectable liposome-in-gel system for regional delivery of radiosensitizer paclitaxel and evaluated with concurrent fractionated radiation. The formulation efficiently localized the drug at tumor site and synergistically enhanced the effect of concurrent radiotherapy.
Jang et al. (2017) prepared 2-hydroxyoleic acid (2OHOA) inserted liposomes to modulate the membrane properties of liposomes and also to make use of the anti-tumor potential of 2-hydroxyoleic acid. These liposomes caused greater and more selective inhibition of cancer cells compared with free 2-hydroxyoleic acid. Insertion of 2-hydroxyoleic acid greatly increased the loading of hydrophobic model drugs such as retinoic acid. These liposomes possessed significantly higher anticancer activity than retinoic acid-loaded conventional liposomes. Delayed tumor growth was achieved in B16-F10 melanoma syngeneic mouse model due to increased induction of apoptosis.
Out of several interesting studies performed on liposomes, one is where Fu et al. (2015) developed paclitaxel (PTX)-loaded liposomes functionalized with TAT, the most frequently used cell-penetrating peptide, and cleavable PEG via a redox-responsive disulfide linker (PTX-C-TAT-LP). Under physiological conditions, TAT was shielded by PEG layer and liposomes exhibited a long blood circulation. At tumor site, PEG could be detached in presence of exogenous reducing agent (glutathione) and TAT was exposed to facilitate cell internalization. Tumor inhibition of around 70% was achieved in B16F1-bearing mice.
Nanoemulsion
Nanoemulsions, made up of components having anti-melanoma activity, have been successfully used for melanoma treatment.
Monge-Fuentes et al. (2017) formulated nanoemulsion using acai oil as a novel photosensitizer for photodynamic therapy of melanoma. 85% cell death was achieved in B16F10 melanoma cell lines, while high viability was maintained in NIH/3T3 normal cells after treatment. Tumor-bearing C57BL/6 mice showed 82% tumor volume reduction as compared to control. Paclitaxel-loaded lipid nanoemulsion was tested in B16F10 melanoma-bearing mice. 95% tumor growth inhibition was achieved in the group given paclitaxel lipid nanoemulsion along with simvastatin (given as adjuvant) which was way more than tumor growth inhibition (44%) with paclitaxel and simvastatin. Survival rates were also higher in this group. After more research, the researchers concluded that simvastatin could increase the anti-tumor activity of paclitaxel when carried in lipid nanoemulsion but not of paclitaxel as it is (Kretzer et al. 2016). Kretzer et al. (2012) had already proved the superiority of lipid nanoemulsions earlier by loading them with paclitaxel and etoposide, and testing and proving their better efficacy in B16F10 melanoma-bearing mice. Piplartine, a poorly soluble alkaloid having anticancer activity, exhibited 1.5 times improved oral bioavailability and showed marked anti-tumor activity in melanoma-bearing mice when loaded in nanoemulsions (Fofaria et al. 2016).
In addition to these studies performed on in-vivo models, Table 2 enlists few more anti-melanoma studies performed on melanoma cell lines using above-mentioned nano-agents.
Table 2.
Cell line studies using nano-agents for treatment of melanoma
| Formulation | Cell lines used | Results | References |
|---|---|---|---|
| Liposomes containing pH-responsive phytosterol derivatives | B16-F10 |
Contents delivered into endosomes and cytosol of B16-F10 cells Liposomes penetrated 3D skin models and reached basement membrane |
Yamazaki et al. (2017) |
| Liposomes containing ferrous chlorophyllin | B16-F10 |
Increased cellular uptake of liposomes via endocytosis Preferential accumulation in mitochondria and nucleus Decreased LC50 with increased incubation time after PDT Mechanism of cell death: apoptosis and necrosis |
Gomaa et al. (2017) |
| Curcumin-loaded liposome gold nanoparticles | B16-F10 |
Significant temperature rise upon laser irradiation causing irreversible cellular damage Significantly enhanced cellular uptake Enhanced cancer cell cytotoxicity upon laser irradiation |
Singh et al. (2017) |
| Liposomes containing C6 (ceramide) | SK-Mel2, WM-266.4, A-375, WM-115 |
Caspase-dependent apoptotic death Activated protein phosphatase 1 (PP1) to inactivate Akt-mammalian target of rapamycin (mTOR) signaling, inhibiting melanoma cells |
Jiang et al. (2016) |
| Liposomes containing Cuphen |
MNT-1 HaCaT B16-F10 |
Anti-proliferative effects of Cuphen in different cancer cell lines, in free form or after incorporation in liposomes Main function of liposomes was to enhance stability of Cuphen and its accumulation in cancerous tissues via EPR effect |
Nave et al. (2016) |
| pH-sensitive liposomes containing doxorubicin | A375 |
Fused with endosomal membrane under acidic conditions of endosome to release doxorubicin into cytoplasm, gathered into nucleus, thus achieving “endosomal escape” Lower cell viability under low pH conditions |
Xu et al. (2015) |
| Layer-by-layer polymer-coated gold nanoparticles containing imatinib mesylate | B16-F10 |
Iontophoresis application enhanced skin penetration of nanoparticles by 6.2-fold as compared to passive application Greater retention in stratum corneum and viable skin Rapid uptake Significant decrease in cell viability |
Labala et al. (2015) |
| Gold Nanoparticles | HTB-72 |
After irradiation, progression of treated cells towards G2/M phase was more rapid than that of non-treated cells, release of former from G2/M phase was slower than that of latter Irradiation with gold nanoparticles increased vulnerability of cells to radiation damage |
Kim and Kim, (2018) |
| Gold nanoparticles and mitoxantrone with microwave hyperthermia | DFW | Mitoxantrone and gold nanoparticles under irradiation caused maximum cell death compared to other groups | Shanei et al. (2017) |
| Gold nanoparticles combined with antibodies targeting phosphorylated FAK (p-FAK-GNP) |
G361 HaCaT |
Used non-thermal atmospheric pressure plasma to stimulate gold nanoparticles within p-FAK-GNP Much higher lethality of combined treatment against G361 cells than HaCaT keratinocyte cells Immediate killing of G361 cells by plasma and p-FAK-GNP |
Choi et al. (2017) |
| Phthalocyanines attached on surface of gold nanorods |
B16-F10 B16-G4F |
Photodynamic properties of phthalocyanines were enhanced Combination of PDT and hyperthermia eliminated over 90% of melanoma cells |
Freitas et al. (2017) |
| PEGylated gold nanoparticles | B16F10 |
Proliferation efficiency and survival fraction decreased with increasing concentration of nanoparticles Significant sensitization of nanoparticles and radiosensitization occurred in presence of 6 MeV electrons |
Mousavi et al. (2017) |
| Chitosan-coated gold nanoparticles carrying STAT3 siRNA | B16-F10 |
Inhibited cell growth by 49.0 ± 0.6 and 66.0 ± 0.2% at 0.25 nM and 0.5 nM STAT3 siRNA concentration, respectively Time dependent cell uptake up to 120 min Clathrin mediated endocytosis as predominant cell uptake mechanism Apoptosis assay showed 29 and 44% of early and late apoptotic events Application of anodal iontophoresis enhanced skin penetration to reach viable epidermis |
Labala et al. (2016) |
| Curcumin-loaded gold nanoparticles | B16-F10s | Efficient uptake and decreased cell viability compared to free curcumin | Muddineti et al. (2016) |
| Chitosan-coated gold nanoparticles as hidden cargo of endothelial colony forming cells (ECFCs) | A375 | Heavily Gold-doped ECFCs efficiently warmed up tumor environment and killed cancer cells via hyperthermic heating both in vitro and in vivo | Margheri et al. (2016) |
| Lipid-coated gold nanohybrids containing docetaxel | B16-F10 |
Significantly greater cytotoxicity compared to free docetaxel Improved cellular uptake Effective tumor cell suppression |
Kang and Ko (2015) |
| Anti-NEU antibody-labeled gold nanoparticles |
G361 HaCaT |
Preferentially targeted melanoma cells than normal keratinocytes Melanoma cells had higher death rate than normal keratinocyte cells Cancer cell death was due to selective destruction of NEU protein and downstream effector of NEU |
Choi et al. (2015) |
| Gold nanoparticles surrounded by amphiphilic-mixed organic ligand shell | B16-F10 |
Amphiphilic nanoparticles initially delivered into endosomes by gold core transferred over a period of hours to intracellular membranes through tumor cells Greater intracellular spread in melanoma cells than breast carcinoma cells Enhanced radiotherapeutic killing of melanoma cells |
Yang et al. (2014) |
|
Carbon nanotube, multi-walled carbon nanotube, iron oxide nanoparticles |
F10 |
All nanoparticles induced selective toxicity and caspase 3 activation through mitochondria pathway Caused generation of ROS, mitochondrial membrane potential decline, mitochondria swelling and cytochrome c release |
Naserzadeh et al. (2017) |
| Zinc monoamino phthalocyanine–folic acid-conjugated single-walled carbon nanotubes | A375 | 60–63% cell death after irradiation of treated melanoma A375 cells | Ogbodu et al. (2015) |
| Anti-GD2 antibody-attached gold nanoparticle conjugated, single-wall carbon nanotube (SWCNT) | UACC903 |
Huge enhancement of two-photon luminescence intensity due to strong resonance enhancement coupled with stronger electric field enhancement Serves as local nanoantennae to enhance photothermal capability via strong optical energy absorption Selective two-photon imaging using 1100 nm light 100% melanoma cells killed after 8 min of exposure |
Tchounwou et al. (2015) |
| Coffee oil–algae oil-based nanoemulsions | B16 F10 |
Effective inhibition of melanoma cell growth Cell cycle arrested at G2/M phase Dose-dependent upregulation of p53, p21, cyclin B, and cyclin A, bax, and cytochrome c expressions and downregulation of CDK1, CDK2 and bcl-2 expression Rise in caspase-3, caspase-8, and caspase-9 activities for apoptosis execution |
Yang et al. (2017) |
| Oil-in-water nanoemulsions of tectona grandis leaf extract | B16 F10 |
Possessed ability to sensitize cells to red light of LED in vitro Photodynamic effect observed as toxicity increased under illumination with red light Reasonable photocytotoxicity and much less toxic towards normal cells in dark |
de Menezes Furtado et al. (2017) |
| Nanoemulsion of 5-FU | SK-MEL-5 | Much more efficacious than free 5-FU when used for topical delivery | Shakeel et al. (2015) |
|
Multi-peptide and toll-like receptor 4 agonist codelivery system based on lipid coated Zinc phosphate hybrid nanoparticles |
B16-F10 |
Exhibited anti-tumor immunity evident by secretion of cytokines in vitro and increased CD8+ T-cell response from IFN-γ ELISPOT analysis ex vivo Improved anti-tumor effects evidenced from prophylactic, therapeutic and metastatic melanoma tumor models compared with free antigens and single peptide-loaded nano-vaccines |
Zhuang et al. (2016) |
Other nano-agents
In addition to the above-mentioned nano-agents, several others are also exploited as anti-melanoma agents; recent ones of them are enlisted in Table 3.
Table 3.
Miscellaneous nano-agents for treatment of melanoma
| Formulation | Tested on | Results | References |
|---|---|---|---|
| (Dacarbazine encapsulated hollow mesoporous silica nanoparticles)-enveloped in folic acid-grafted liposomes |
Human melanoma cell line A875 Mouse melanoma cell line B16/F10 |
Significant improvement against lung metastasis of melanoma via targeting melanoma cells and tumor-associated macrophages | Liu et al. (2017) |
| [Porous silicon-based micro/nano composite loaded with BRAF proto-oncogene serine/threonine kinase (BRAF) siRNA-containing liposomes]- conjugated with docetaxel-encapsulated polymeric nanoparticles |
Melanoma cell line A375 Mice-bearing A375SM melanoma lung metastases |
Superior therapeutic efficacy and increased accumulation in metastatic melanoma lesions in the lungs | Mi et al. (2016) |
| Curcumin-loaded silica mesoporous nanoparticles | B16F10 mouse melanoma cells |
Biocompatible in non-cancerous CHO cell line Significant cytotoxicity against cancer cells by generating ROS and downregulating anti-apoptotic proteins |
Bollu et al. (2016) |
| Titanium-dioxide-nanoparticle–gold-nanocluster–graphene heterogeneous nanocomposites | B16F1-tumor-xenograft-bearing mice |
Under irradiation, triggered intracellular ROS production, glutathione depletion, heme oxygenase-1 expression, and mitochondrial dysfunctions, resulting in severe cell death Significantly inhibited tumor growth and caused severe pathological tumor tissue changes |
Cheng et al. (2017) |
| Cholrambucil-loaded liposomes/albumin hybrid nanoparticles | B16F10 melanoma-bearing mice |
Better tumor-targeting capacity and significantly increased drug accumulation in tumor Longest median survival of treated group: 30 days |
Zhang et al. (2017) |
| N-lauryl glucosamine coated Doxorubicin-loaded niosomes | Melanoma-bearing BDF mice model | Commendable targeting potential with significant reduction in tumor volume and higher survival rate | Pawar et al. (2016) |
| Acridine orange charged exosomes | Monolayer and spheroid models of melanoma cells lines Me 30,966 |
Taken much more rapidly by melanoma cells and retained for longer time Enhanced anti-cancer effect of acridine orange by increasing exposure time of target cells Significant tumoricidal action sparing normal cells |
Iessi et al. (2017) |
Conclusion
Challenge in the treatment of melanoma is low response rate with existing treatment modalities. Chemotherapy fails to treat melanoma due to its resistance, and instead kills normal body cells. This results in poor outcome of chemotherapy with toxic side effects. Though the standard treatment drugs such as dacarbazine and temozolomide still constitute the mainstay of the melanoma treatment, the strategies need to be improvised by combining these agents with newer targeted and more effective drugs. Nanotechnology has given a big platform to try out new strategies against this deadly cancer with a hope of improving response rate and overall survival of melanoma patients. During the last few years, targeted nanoformulations have been explored for their potential in melanoma treatment and promising results have been obtained. All these experiments open up new avenues for future studies in melanoma treatment expected to lead to better therapeutic outcomes of melanoma treatment.
Compliance with ethical standards
Conflict of interest
The authors declare no competing financial interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
References
- Abo-Elfadl MT, Gamal-Eldeen AM, Elshafey MM, Abdalla GM, Ali SS, Ali MRK, Zawrah MFM (2016) Photothermal therapeutic effect of PEGylated gold nano-semicubes in chemically-induced skin cancer in mice. J Photochem Photobiol B 164:21–29. 10.1016/j.jphotobiol.2016.09.012 [DOI] [PubMed] [Google Scholar]
- Alupei MC, Licarete E, Patras L, Banciu M (2015) Liposomal simvastatin inhibits tumor growth via targeting tumor-associated macrophages-mediated oxidative stress. Cancer Lett 356(2, part B):946–952. 10.1016/j.canlet.2014.11.010 [DOI] [PubMed] [Google Scholar]
- Apalla Z, Nashan D, Weller RB, Castellsagué X (2017) Skin cancer: epidemiology, disease burden, pathophysiology, diagnosis, and therapeutic approaches. Dermatol Therapy 7(1):5–19. 10.1007/s13555-016-0165-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ascierto PA, McArthur GA, Dréno B, Larkin J, Liszkay G, Maio M, Ribas A (2015) coBRIM: a phase 3, double-blind, placebo-controlled study of vemurafenib versus vemurafenib + cobimetinib in previously untreated BRAFV600 mutation—positive patients with unresectable locally advanced or metastatic melanoma (NCT01689519). J Transl Med 13(1):O4. 10.1186/1479-5876-13-S1-O4 [Google Scholar]
- Aspord C, Tramcourt L, Leloup C, Molens J-P, Leccia M-T, Charles J, Plumas J (2014) Imiquimod inhibits melanoma development by promoting pDC cytotoxic functions and impeding tumor vascularization. J Invest Dermatol 134(10):2551–2561. 10.1038/jid.2014.194 [DOI] [PubMed] [Google Scholar]
- Atkins MB, Lotze MT, Dutcher JP, Fisher RI, Weiss G, Margolin K, Rosenberg SA (1999) High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J Clin Oncol 17(7):2105–2105. 10.1200/JCO.1999.17.7.2105 [DOI] [PubMed] [Google Scholar]
- Atkins MB, Kunkel L, Sznol M, Rosenberg SA (2000) High-dose recombinant interleukin-2 therapy in patients with metastatic melanoma: long-term survival update. Cancer J Sci Am 6(Suppl 1):S11–S14 [PubMed] [Google Scholar]
- Bajetta E, Del Vecchio M, Nova P, Fusi A, Daponte A, Sertoli MR, Cascinelli N (2006) Multicenter phase III randomized trial of polychemotherapy (CVD regimen) versus the same chemotherapy (CT) plus subcutaneous interleukin-2 and interferon-α2b in metastatic melanoma. Ann Oncol 17(4):571–577. 10.1093/annonc/mdl007 [DOI] [PubMed] [Google Scholar]
- Barui S, Saha S, Yakati V, Chaudhuri A (2016) Systemic codelivery of a homoserine derived ceramide analogue and curcumin to tumor vasculature inhibits mouse tumor growth. Mol Pharm 13(2):404–419. 10.1021/acs.molpharmaceut.5b00644 [DOI] [PubMed] [Google Scholar]
- Bhatia S, Tykodi SS, Thompson JA (2009) Treatment of metastatic melanoma: an overview. Oncology (Williston Park NY) 23(6):488–496 [PMC free article] [PubMed] [Google Scholar]
- Blesa JMG, Pulido EG, Candel VA, Pulla MP (2011) Melanoma: from darkness to promise. Am J Clin Oncol 34(2):179. 10.1097/COC.0b013e3181d6b427 [DOI] [PubMed] [Google Scholar]
- Bollu VS, Barui AK, Mondal SK, Prashar S, Fajardo M, Briones D, Gómez-Ruiz S (2016) Curcumin-loaded silica-based mesoporous materials: synthesis, characterization and cytotoxic properties against cancer cells. Mater Sci Eng C 63:393–410. 10.1016/j.msec.2016.03.011 [DOI] [PubMed] [Google Scholar]
- Bottoni U, Bonaccorsi P, Devirgiliis V, Panasiti V, Borroni RG, Trasimeni G, Calvieri S (2005) Complete remission of brain metastases in three patients with stage IV melanoma treated with BOLD and G-CSF. Jpn J Clin Oncol 35(9):507–513. 10.1093/jjco/hyi141 [DOI] [PubMed] [Google Scholar]
- Calderon-Gonzalez R, Terán-Navarro H, García I, Marradi M, Salcines-Cuevas D, Yañez-Diaz S, Álvarez-Domínguez C (2017) Gold glyconanoparticles coupled to listeriolysin O 91–99 peptide serve as adjuvant therapy against melanoma. Nanoscale 9(30):10721–10732. 10.1039/C7NR02494K [DOI] [PubMed] [Google Scholar]
- Camerin M, Moreno M, Marín MJ, Schofield CL, Chambrier I, Cook MJ, Russell DA (2016) Delivery of a hydrophobic phthalocyanine photosensitizer using PEGylated gold nanoparticle conjugates for the in vivo photodynamic therapy of amelanotic melanoma. Photochem Photobiol Sci 15(5):618–625. 10.1039/C5PP00463B [DOI] [PubMed] [Google Scholar]
- Carvajal RD, Antonescu CR, Wolchok JD, Chapman PB, Roman R-A, Teitcher J, Schwartz GK (2011) KIT as a therapeutic target in metastatic melanoma. JAMA 305(22):2327–2334. 10.1001/jama.2011.746 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakraborty R, Basu T (2017) Metallic copper nanoparticles induce apoptosis in a human skin melanoma A-375 cell line. Nanotechnology 28(10):105101. 10.1088/1361-6528/aa57b0 [DOI] [PubMed] [Google Scholar]
- Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J, McArthur GA (2011) Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med 364(26):2507–2516. 10.1056/NEJMoa1103782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J, Shao R, Zhang XD, Chen C (2013) Applications of nanotechnology for melanoma treatment, diagnosis, and theranostics. Int J Nanomed 8:2677–2688. 10.2147/IJN.S45429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z, Zhang T, Wu B, Zhang X (2016) Insights into the therapeutic potential of hypoxia-inducible factor-1α small interfering RNA in malignant melanoma delivered via folate-decorated cationic liposomes. Int J Nanomed 11:991–1002. 10.2147/IJN.S101872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y, Chang Y, Feng Y, Liu N, Sun X, Feng Y, Zhang H (2017) Simulated sunlight-mediated photodynamic therapy for melanoma skin cancer by titanium-dioxide-nanoparticle–gold-nanocluster–graphene heterogeneous nanocomposites. Small. 10.1002/smll.201603935 [DOI] [PubMed] [Google Scholar]
- Chiarion-Sileni V, Guida M, Ridolfi R, Romanini A, Brugnara S, Bianco D, De Salvo PG (2008) Temozolomide (TMZ) as prophylaxis for melanoma brain metastases (BrM): results from a phase III, multicenter study. J Clin Oncol 26(15_suppl):20014–20014. 10.1200/jco.2008.26.15_suppl.20014 [Google Scholar]
- Choi BB, Kim MS, Song KW, Kim UK, Hong JW, Lee HJ, Kim GC (2015) Targeting NEU protein in melanoma cells with non-thermal atmospheric pressure plasma and gold nanoparticles. J Biomed Nanotechnol 11(5):900–905. 10.1166/jbn.2015.1999 [DOI] [PubMed] [Google Scholar]
- Choi BBR, Choi JH, Hong JW, Song KW, Lee HJ, Kim UK, Kim GC (2017) Selective killing of melanoma cells with non-thermal atmospheric pressure plasma and p-FAK antibody conjugated gold nanoparticles. Int J Med Sci 14(11):1101–1109. 10.7150/ijms.20104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark JI, Moon J, Hutchins LF, Sosman JA, Kast WM, Da Silva DM, Sondak VK (2010) Phase 2 trial of combination thalidomide plus temozolomide in patients with metastatic malignant melanoma: Southwest Oncology Group S0508. Cancer 116(2):424–431. 10.1002/cncr.24739 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coates AS, Segelov E (1994) Long term response to chemotherapy in patients with visceral metastatic melanoma. Ann Oncol 5(3):249–251. 10.1093/oxfordjournals.annonc.a058802 [DOI] [PubMed] [Google Scholar]
- Curtin JA, Busam K, Pinkel D, Bastian BC (2006) Somatic activation of KIT in distinct subtypes of melanoma. J Clin Oncol 24(26):4340–4346. 10.1200/JCO.2006.06.2984 [DOI] [PubMed] [Google Scholar]
- de Menezes Furtado C, de Faria FSEDV, Azevedo RB, Py-Daniel K, dos Santos Camara AL, da Silva JR, Degterev IA (2017) Tectona grandis leaf extract, free and associated with nanoemulsions, as a possible photosensitizer of mouse melanoma B16 cell. J Photochem Photobiol B 167:242–248. 10.1016/j.jphotobiol.2017.01.004 [DOI] [PubMed] [Google Scholar]
- Della Giovampaola C, Capone A, Ermini L, Lupetti P, Vannuccini E, Finetti F, Bonechi C (2017) Formulation of liposomes functionalized with Lotus lectin and effective in targeting highly proliferative cells. Biochim Biophys Acta Gen Subj 1861(4):860–870. 10.1016/j.bbagen.2017.01.015 [DOI] [PubMed] [Google Scholar]
- Deng C, Zhang Q, Fu Y, Sun X, Gong T, Zhang Z (2017) Coadministration of oligomeric hyaluronic acid-modified liposomes with tumor-penetrating peptide-iRGD enhances the antitumor efficacy of doxorubicin against melanoma. ACS Appl Mater Interfaces 9(2):1280–1292. 10.1021/acsami.6b13738 [DOI] [PubMed] [Google Scholar]
- Di Franco S, Turdo A, Todaro M, Stassi G (2017) Role of type I and II interferons in colorectal cancer and melanoma. Front Immunol. 10.3389/fimmu.2017.00878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dicheva BM, Hagen TLM, ten Seynhaeve ALB, Amin M, Eggermont AMM, Koning GA (2015) Enhanced specificity and drug delivery in tumors by cRGD—anchoring thermosensitive liposomes. Pharm Res 32(12):3862–3876. 10.1007/s11095-015-1746-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorrani M, Garbuzenko OB, Minko T, Michniak-Kohn B (2016) Development of edge-activated liposomes for siRNA delivery to human basal epidermis for melanoma therapy. J Controll Release 228:150–158. 10.1016/j.jconrel.2016.03.010 [DOI] [PubMed] [Google Scholar]
- Drewes CC, Fiel LA, Bexiga CG, Asbahr ACC, Uchiyama MK, Cogliati B, Farsky SP (2016) Novel therapeutic mechanisms determine the effectiveness of lipid-core nanocapsules on melanoma models. Int J Nanomed 11:1261–1279. 10.2147/IJN.S101543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisen T, Ahmad T, Flaherty KT, Gore M, Kaye S, Marais R, Ratain MJ (2006) Sorafenib in advanced melanoma: a Phase II randomised discontinuation trial analysis. Br J Cancer 95(5):581. 10.1038/sj.bjc.6603291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fadel TR, Sharp FA, Vudattu N, Ragheb R, Garyu J, Kim D, Fahmy TM (2014) A carbon nanotube–polymer composite for T-cell therapy. Nat Nanotechnol 9(8):639. 10.1038/nnano.2014.154 [DOI] [PubMed] [Google Scholar]
- Falchook GS, Long GV, Kurzrock R, Kim KB, Arkenau TH, Brown MP, Kefford RF (2012) Dabrafenib in patients with melanoma, untreated brain metastases, and other solid tumours: a phase 1 dose-escalation trial. Lancet 379(9829):1893–1901. 10.1016/S0140-6736(12)60398-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flaherty KT, Lee SJ, Schuchter LM, Flaherty LE, Wright JJ, Leming PD, Kirkwood JM (2010a) Final results of E2603: a double-blind, randomized phase III trial comparing carboplatin (C)/paclitaxel (P) with or without sorafenib (S) in metastatic melanoma. J Clin Oncol 28(15_suppl):8511–8511. 10.1200/jco.2010.28.15_suppl.8511 [Google Scholar]
- Flaherty KT, Puzanov I, Kim KB, Ribas A, McArthur GA, Sosman JA, Chapman PB (2010b) Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med 363(9):809–819. 10.1056/NEJMoa1002011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flaherty KT, Infante JR, Daud A, Gonzalez R, Kefford RF, Sosman J, Weber J (2012a) Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med 367(18):1694–1703. 10.1056/NEJMoa1210093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flaherty KT, Robert C, Hersey P, Nathan P, Garbe C, Milhem M, Schadendorf D (2012b) Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med 367(2):107–114. 10.1056/NEJMoa1203421 [DOI] [PubMed] [Google Scholar]
- Florin V, Desmedt E, Vercambre-Darras S, Mortier L (2012) Topical treatment of cutaneous metastases of malignant melanoma using combined imiquimod and 5-fluorouracil. Invest New Drugs 30(4):1641–1645. 10.1007/s10637-011-9717-2 [DOI] [PubMed] [Google Scholar]
- Fofaria NM, Qhattal HSS, Liu X, Srivastava SK (2016) Nanoemulsion formulations for anti-cancer agent piplartine—characterization, toxicological, pharmacokinetics and efficacy studies. Int J Pharm 498(1):12–22. 10.1016/j.ijpharm.2015.11.045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franklin C, Livingstone E, Roesch A, Schilling B, Schadendorf D (2017) Immunotherapy in melanoma: recent advances and future directions. Eur J Surg Oncol 43(3):604–611. 10.1016/j.ejso.2016.07.145 [DOI] [PubMed] [Google Scholar]
- Freitas LF, Hamblin MR, Anzengruber F, Perussi JR, Ribeiro AO, Martins VCA, Plepis AMG (2017) Zinc phthalocyanines attached to gold nanorods for simultaneous hyperthermic and photodynamic therapies against melanoma in vitro. J Photochem Photobiol B 173:181–186. 10.1016/j.jphotobiol.2017.05.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu H, Shi K, Hu G, Yang Y, Kuang Q, Lu L, He Q (2015) Tumor-targeted paclitaxel delivery and enhanced penetration using TAT-decorated liposomes comprising redox-responsive poly(ethylene glycol). J Pharm Sci 104(3):1160–1173. 10.1002/jps.24291 [DOI] [PubMed] [Google Scholar]
- Gilmartin AG, Bleam MR, Groy A, Moss KG, Minthorn EA, Kulkarni SG, Laquerre SG (2011) GSK1120212 (JTP-74057) is an inhibitor of MEK activity and activation with favorable pharmacokinetic properties for sustained in vivo pathway inhibition. Clin Cancer Res. 10.1158/1078-0432.CCR-10-2200 [DOI] [PubMed] [Google Scholar]
- Gomaa I, Sebak A, Afifi N, Abdel-Kader M (2017) Liposomal delivery of ferrous chlorophyllin: a novel third generation photosensitizer for in vitro PDT of melanoma. Photodiagn Photodyn Ther 18:162–170. 10.1016/j.pdpdt.2017.01.186 [DOI] [PubMed] [Google Scholar]
- Gowda R, Kardos G, Sharma A, Singh S, Robertson GP (2017) Nanoparticle-based celecoxib and plumbagin for the synergistic treatment of melanoma. Mol Cancer Ther 16(3):440–452. 10.1158/1535-7163.MCT-16-0285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grob JJ, Robert C, Long GV, Stroyakovskiy D, Levchenko E, Chiarion-Sileni V, Schadendorf D (2016) Health-related quality-of-life (HRQOL) impact of dabrafenib (D) and trametinib (T) vs BRAF inhibitor (BRAFi) monotherapy by lactate dehydrogenase (LDH) in patients (pts) with BRAF V600—mutant melanoma. Ann Oncol. 10.1093/annonc/mdw379.3227502712 [Google Scholar]
- GuhaSarkar S, Pathak K, Sudhalkar N, More P, Goda JS, Gota V, Banerjee R (2016) Synergistic locoregional chemoradiotherapy using a composite liposome-in-gel system as an injectable drug depot. Int J Nanomed 11:6435–6448. 10.2147/IJN.S110525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauschild A, Agarwala SS, Trefzer U, Hogg D, Robert C, Hersey P, Keilholz U (2009) Results of a Phase III, randomized, placebo-controlled study of sorafenib in combination with carboplatin and paclitaxel as second-line treatment in patients with unresectable stage III or stage IV melanoma. J Clin Oncol 27(17):2823–2830. 10.1200/JCO.2007.15.7636 [DOI] [PubMed] [Google Scholar]
- Hauschild A, Grob J-J, Demidov LV, Jouary T, Gutzmer R, Millward M, Chapman PB (2012) Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet 380(9839):358–365. 10.1016/S0140-6736(12)60868-X [DOI] [PubMed] [Google Scholar]
- Hegedũs L, Garay T, Molnár E, Varga K, Bilecz Á, Török S, Enyedi A (2017) The plasma membrane Ca2+ pump PMCA4b inhibits the migratory and metastatic activity of BRAF mutant melanoma cells. Int J Cancer 140(12):2758–2770. 10.1002/ijc.30503 [DOI] [PubMed] [Google Scholar]
- Heidari M, Sattarahmady N, Azarpira N, Heli H, Mehdizadeh AR, Zare T (2016) Photothermal cancer therapy by gold-ferrite nanocomposite and near-infrared laser in animal model. Lasers Med Sci 31(2):221–227. 10.1007/s10103-015-1847-x [DOI] [PubMed] [Google Scholar]
- Helmbach H, Rossmann E, Kern MA, Schadendorf D (2001) Drug-resistance in human melanoma. Int J Cancer 93(5):617–622. 10.1002/ijc.1378 [DOI] [PubMed] [Google Scholar]
- Hill GJ, Krementz ET, Hill HZ (1984) Dimethyl triazeno imidazole carboxamide and combination therapy for melanoma IV. Late results after complete response to chemotherapy (central oncology group protocols 7130, 7131, and 7131A). Cancer 53(6):1299–1305. 10.1002/1097-0142(19840315)53:6%3C1299::AID-CNCR2820530613%3E3.0.CO;2-4 [DOI] [PubMed] [Google Scholar]
- Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Urba WJ (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363(8):711–723. 10.1056/NEJMoa1003466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofmann M, Kiecker F, Wurm R, Schlenger L, Budach V, Sterry W, Trefzer U (2006) Temozolomide with or without radiotherapy in melanoma with unresectable brain metastases. J Neuro Oncol 76(1):59–64. 10.1007/s11060-005-2914-0 [DOI] [PubMed] [Google Scholar]
- Iessi E, Logozzi M, Lugini L, Azzarito T, Federici C, Spugnini EP, Fais S (2017) Acridine orange/exosomes increase the delivery and the effectiveness of acridine orange in human melanoma cells: a new prototype for theranostics of tumors. J Enzyme Inhib Med Chem 32(1):648–657. 10.1080/14756366.2017.1292263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang E-J, Choi WR, Kim S-Y, Hong S-S, Rhee I, Lee S-J, Lim S-J (2017) 2-Hydroxyoleic acid-inserted liposomes as a multifunctional carrier of anticancer drugs. Drug Deliv 24(1):1587–1597. 10.1080/10717544.2017.1388452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang F, Jin K, Huang S, Bao Q, Shao Z, Hu X, Ye J (2016) Liposomal C6 ceramide activates protein phosphatase 1 to inhibit melanoma cells. PLoS One 11(9):e0159849. 10.1371/journal.pone.0159849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiménez Pérez ZE, Mathiyalagan R, Markus J, Kim Y-J, Kang HM, Abbai R, Yang DC (2017) Ginseng-berry-mediated gold and silver nanoparticle synthesis and evaluation of their in vitro antioxidant, antimicrobial, and cytotoxicity effects on human dermal fibroblast and murine melanoma skin cell lines. Int J Nanomed 12:709–723. 10.2147/IJN.S118373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jose A, Labala S, Ninave KM, Gade SK, Venuganti VVK (2018) Effective skin cancer treatment by topical co-delivery of curcumin and STAT3 siRNA using cationic liposomes. AAPS Pharm Sci Tech 19(1):166–175. 10.1208/s12249-017-0833-y [DOI] [PubMed] [Google Scholar]
- Kang JH, Ko YT (2015) Lipid-coated gold nanocomposites for enhanced cancer therapy. Int J Nanomed 10(Spec Iss):33–45. 10.2147/IJN.S88307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufmann R, Spieth K, Leiter U, Mauch C, von den Driesch P, Vogt T, Dummer R (2005) Temozolomide in combination with interferon-alfa versus temozolomide alone in patients with advanced metastatic melanoma: a randomized, phase III, multicenter study from the Dermatologic Cooperative Oncology Group. J Clin Oncol 23(35):9001–9007. 10.1200/JCO.2005.01.1551 [DOI] [PubMed] [Google Scholar]
- Keilholz U, Conradt C, Legha SS, Khayat D, Scheibenbogen C, Thatcher N, Pritsch M (1998) Results of interleukin-2-based treatment in advanced melanoma: a case record-based analysis of 631 patients. J Clin Oncol 16(9):2921–2929. 10.1200/JCO.1998.16.9.2921 [DOI] [PubMed] [Google Scholar]
- Kim S-R, Kim E-H (2018) Feasibility study on the use of gold nanoparticles in fractionated kilovoltage X-ray treatment of melanoma. Int J Radiat Biol 94(1):8–16. 10.1080/09553002.2018.1393579 [DOI] [PubMed] [Google Scholar]
- Kirkwood JM, Strawderman MH, Ernstoff MS, Smith TJ, Borden EC, Blum RH (1996) Interferon alfa-2b adjuvant therapy of high-risk resected cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J Clin Oncol 14(1):7–17. 10.1200/JCO.1996.14.1.7 [DOI] [PubMed] [Google Scholar]
- Kirkwood JM, Ibrahim JG, Sondak VK, Richards J, Flaherty LE, Ernstoff MS, Blum RH (2000) High- and low-dose interferon Alfa-2b in high-risk melanoma: first analysis of intergroup trial E1690/S9111/C9190. J Clin Oncol 18(12):2444–2458. 10.1200/JCO.2000.18.12.2444 [DOI] [PubMed] [Google Scholar]
- Kirkwood JM, Ibrahim JG, Sosman JA, Sondak VK, Agarwala SS, Ernstoff MS, Rao U (2001) High-dose interferon Alfa-2b significantly prolongs relapse-free and overall survival compared with the GM2-KLH/QS-21 vaccine in patients with resected stage IIB-III melanoma: results of intergroup trial E1694/S9512/C509801. J Clin Oncol 19(9):2370–2380. 10.1200/JCO.2001.19.9.2370 [DOI] [PubMed] [Google Scholar]
- Kretzer IF, Maria DA, Maranhão RC (2012) Drug-targeting in combined cancer chemotherapy: tumor growth inhibition in mice by association of paclitaxel and etoposide with a cholesterol-rich nanoemulsion. Cell Oncol 35(6):451–460. 10.1007/s13402-012-0104-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kretzer IF, Maria DA, Guido MC, Contente TC, Maranhão RC (2016) Simvastatin increases the antineoplastic actions of paclitaxel carried in lipid nanoemulsions in melanoma-bearing mice. Int J Nanomed 11:885–904. 10.2147/IJN.S88546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labala S, Mandapalli PK, Kurumaddali A, Venuganti VVK (2015) Layer-by-layer polymer coated gold nanoparticles for topical delivery of imatinib mesylate to treat melanoma. Mol Pharm 12(3):878–888. 10.1021/mp5007163 [DOI] [PubMed] [Google Scholar]
- Labala S, Jose A, Venuganti VVK (2016) Transcutaneous iontophoretic delivery of STAT3 siRNA using layer-by-layer chitosan coated gold nanoparticles to treat melanoma. Colloids Surf B 146:188–197. 10.1016/j.colsurfb.2016.05.076 [DOI] [PubMed] [Google Scholar]
- Labala S, Jose A, Chawla SR, Khan MS, Bhatnagar S, Kulkarni OP, Venuganti VVK (2017) Effective melanoma cancer suppression by iontophoretic co-delivery of STAT3 siRNA and imatinib using gold nanoparticles. Int J Pharm 525(2):407–417. 10.1016/j.ijpharm.2017.03.087 [DOI] [PubMed] [Google Scholar]
- Li T, Zhang M, Wang J, Wang T, Yao Y, Zhang X, Zhang N (2016) Thermosensitive hydrogel co-loaded with gold nanoparticles and doxorubicin for effective chemoradiotherapy. AAPS J 18(1):146–155. 10.1208/s12248-015-9828-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang R, Xie J, Li J, Wang K, Liu L, Gao Y, Tao J (2017) Liposomes-coated gold nanocages with antigens and adjuvants targeted delivery to dendritic cells for enhancing antitumor immune response. Biomaterials 149:41–50. 10.1016/j.biomaterials.2017.09.029 [DOI] [PubMed] [Google Scholar]
- Liu Q, Xu N, Liu L, Li J, Zhang Y, Shen C, Tao J (2017) Dacarbazine-loaded hollow mesoporous silica nanoparticles grafted with folic acid for enhancing antimetastatic melanoma response. ACS Appl Mater Interfaces 9(26):21673–21687. 10.1021/acsami.7b05278 [DOI] [PubMed] [Google Scholar]
- Long GV, Stroyakovsky DL, Gogas H, Levchenko E, de Braud F, Larkin JMG, Flaherty K (2014) COMBI-d: a randomized, double-blinded, Phase III study comparing the combination of dabrafenib and trametinib to dabrafenib and trametinib placebo as first-line therapy in patients (pts) with unresectable or metastatic BRAFV600E/K mutation-positive cutaneous melanoma. J Clin Oncol 32(15_suppl):9011–9011. 10.1200/jco.2014.32.15_suppl.9011 [Google Scholar]
- Lui P, Cashin R, Machado M, Hemels M, Corey-Lisle PK, Einarson TR (2007) Treatments for metastatic melanoma: synthesis of evidence from randomized trials. Cancer Treat Rev 33(8):665–680. 10.1016/j.ctrv.2007.06.004 [DOI] [PubMed] [Google Scholar]
- Mandalà M, Voit C (2013) Targeting BRAF in melanoma: biological and clinical challenges. Crit Rev Oncol Hematol 87(3):239–255. 10.1016/j.critrevonc.2013.01.003 [DOI] [PubMed] [Google Scholar]
- Margheri G, Zoppi A, Olmi R, Trigari S, Traversi R, Severi M, Rosso MD (2016) Tumor-tropic endothelial colony forming cells (ECFCs) loaded with near-infrared sensitive Au nanoparticles: a “cellular stove” approach to the photoablation of melanoma. Oncotarget 7(26):39846–39860. 10.18632/oncotarget.9511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauldin IS, Wages NA, Stowman AM, Wang E, Olson WC, Deacon DH, Slingluff CL (2016) Topical treatment of melanoma metastases with imiquimod, plus administration of a cancer vaccine, promotes immune signatures in the metastases. Cancer Immunol Immunother 65(10):1201–1212. 10.1007/s00262-016-1880-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menzies AM, Long GV (2014) Systemic treatment for BRAF-mutant melanoma: where do we go next? Lancet Oncol 15(9):e371–e381. 10.1016/S1470-2045(14)70072-5 [DOI] [PubMed] [Google Scholar]
- Mi Y, Mu C, Wolfram J, Deng Z, Hu TY, Liu X, Ferrari M (2016) A micro/nano composite for combination treatment of melanoma lung metastasis. Adv Healthc Mater 5(8):936–946. 10.1002/adhm.201500910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Middleton Mr, Grob Jj, Aaronson N, Fierlbeck G, Tilgen W, Seiter S, Thatcher N (2000) Randomized phase III study of temozolomide versus dacarbazine in the treatment of patients with advanced metastatic malignant melanoma. J Clin Oncol 18(1):158–158. 10.1200/JCO.2000.18.1.158 [DOI] [PubMed] [Google Scholar]
- Monge-Fuentes V, Muehlmann LA, Longo JPF, Silva JR, Fascineli ML, de Souza P, Azevedo RB (2017) Photodynamic therapy mediated by acai oil (Euterpe oleracea Martius) in nanoemulsion: a potential treatment for melanoma. J Photochem Photobiol B 166:301–310. 10.1016/j.jphotobiol.2016.12.002 [DOI] [PubMed] [Google Scholar]
- Moon SD, Spencer JM (2013) Clearance of invasive melanoma with topical imiquimod. J Drugs Dermatol 12(1):107–108 [PubMed] [Google Scholar]
- Mousavi M, Nedaei HA, Khoei S, Eynali S, Khoshgard K, Robatjazi M, Rad RI (2017) Enhancement of radiosensitivity of melanoma cells by pegylated gold nanoparticles under irradiation of megavoltage electrons. Int J Radiat Biol 93(2):214–221. 10.1080/09553002.2017.1231944 [DOI] [PubMed] [Google Scholar]
- Muddineti OS, Kumari P, Ajjarapu S, Lakhani PM, Bahl R, Ghosh B, Biswas S (2016) Xanthan gum stabilized PEGylated gold nanoparticles for improved delivery of curcumin in cancer. Nanotechnology 27(32):325101. 10.1088/0957-4484/27/32/325101 [DOI] [PubMed] [Google Scholar]
- Mukhopadhyay R, Kazi J, Debnath MC (2018) Synthesis and characterization of copper nanoparticles stabilized with Quisqualis indica extract: evaluation of its cytotoxicity and apoptosis in B16F10 melanoma cells. Biomed Pharmacother 97:1373–1385. 10.1016/j.biopha.2017.10.167 [DOI] [PubMed] [Google Scholar]
- Naserzadeh P, Esfeh FA, Kaviani M, Ashtari K, Kheirbakhsh R, Salimi A, Pourahmad J (2017) Single-walled carbon nanotube, multi-walled carbon nanotube and Fe2O3 nanoparticles induced mitochondria mediated apoptosis in melanoma cells. Cutan Ocular Toxicol 0(0):1–10. 10.1080/15569527.2017.1363227 [DOI] [PubMed] [Google Scholar]
- Nave M, Castro RE, Rodrigues CM, Casini A, Soveral G, Gaspar MM (2016) Nanoformulations of a potent copper-based aquaporin inhibitor with cytotoxic effect against cancer cells. Nanomedicine 11(14):1817–1830. 10.2217/nnm-2016-0086 [DOI] [PubMed] [Google Scholar]
- Neumann S, Shirley SA, Kemp RA, Hook SM (2016) Improved antitumor activity of a therapeutic melanoma vaccine through the use of the dual COX-2/5-LO inhibitor licofelone. Front Immunol. 10.3389/fimmu.2016.00537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicholas S, Mathios D, Jackson C, Lim M (2013) Metastatic melanoma to the brain: surgery and radiation is still the standard of care. Curr Treat Options Oncol 14(2):264–279. 10.1007/s11864-013-0228-6 [DOI] [PubMed] [Google Scholar]
- Nikolaou VA, Stratigos AJ, Flaherty KT, Tsao H (2012) Melanoma: new insights and new therapies. J Investig Dermatol 132(3, part 2):854–863. 10.1038/jid.2011.421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogbodu RO, Ndhundhuma I, Karsten A, Nyokong T (2015) Photodynamic therapy effect of zinc monoamino phthalocyanine–folic acid conjugate adsorbed on single walled carbon nanotubes on melanoma cells. Spectrochim Acta Part A Mol Biomol Spectrosc 137:1120–1125. 10.1016/j.saa.2014.09.033 [DOI] [PubMed] [Google Scholar]
- Paraiso KHT, Fedorenko IV, Cantini LP, Munko AC, Hall M, Sondak VK, Smalley KSM (2010) Recovery of phospho-ERK activity allows melanoma cells to escape from BRAF inhibitor therapy. Br J Cancer 102(12):1724. 10.1038/sj.bjc.6605714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel PM, Suciu S, Mortier L, Kruit WH, Robert C, Schadendorf D, Spatz A (2011) Extended schedule, escalated dose temozolomide versus dacarbazine in stage IV melanoma: final results of a randomised phase III study (EORTC 18032). Eur J Cancer 47(10):1476–1483. 10.1016/j.ejca.2011.04.030 [DOI] [PubMed] [Google Scholar]
- Pawar S, Shevalkar G, Vavia P (2016) Glucosamine-anchored doxorubicin-loaded targeted nano-niosomes: pharmacokinetic, toxicity and pharmacodynamic evaluation. J Drug Target 24(8):730–743. 10.3109/1061186X.2016.1154560 [DOI] [PubMed] [Google Scholar]
- Piaggio F, Kondylis V, Pastorino F, Di Paolo D, Perri P, Cossu I, Brignole C (2016) A novel liposomal Clodronate depletes tumor-associated macrophages in primary and metastatic melanoma: anti-angiogenic and anti-tumor effects. J Controll Release 223:165–177. 10.1016/j.jconrel.2015.12.037 [DOI] [PubMed] [Google Scholar]
- Quan WDY, Quan FM, Perez M, Johnson E (2012) Outpatient intravenous interleukin-2 with famotidine has activity in metastatic melanoma. Cancer Biother Radiopharm 27(7):442–445. 10.1089/cbr.2012.1239 [DOI] [PubMed] [Google Scholar]
- Quirt I, Verma S, Petrella T, Bak K, Charette M (2007) Temozolomide for the treatment of metastatic melanoma: a systematic review. Oncologist 12(9):1114–1123. 10.1634/theoncologist.12-9-1114 [DOI] [PubMed] [Google Scholar]
- Rajakulendran T, Adam DN (2014) Bench to bedside: mechanistic principles of targeting the RAF kinase in melanoma. Int J Dermatol 53(12):1428–1433. 10.1111/ijd.12724 [DOI] [PubMed] [Google Scholar]
- Resnier P, Galopin N, Sibiril Y, Clavreul A, Cayon J, Briganti A, Passirani C (2017) Efficient ferrocifen anticancer drug and Bcl-2 gene therapy using lipid nanocapsules on human melanoma xenograft in mouse. Pharmacol Res 126:54–65. 10.1016/j.phrs.2017.01.031 [DOI] [PubMed] [Google Scholar]
- Ribas A, Gonzalez R, Pavlick A, Hamid O, Gajewski TF, Daud A, McArthur GA (2014) Combination of vemurafenib and cobimetinib in patients with advanced BRAFV600-mutated melanoma: a phase 1b study. Lancet Oncol 15(9):954–965. 10.1016/S1470-2045(14)70301-8 [DOI] [PubMed] [Google Scholar]
- Robert C, Thomas L, Bondarenko I, O’Day S, Weber J, Garbe C, Wolchok JD (2011) Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med 364(26):2517–2526. 10.1056/NEJMoa1104621 [DOI] [PubMed] [Google Scholar]
- Sadhu SS, Wang S, Dachineni R, Averineni RK, Yang Y, Yin H, Guan X (2017) In vitro and in vivo tumor growth inhibition by glutathione disulfide liposomes. Cancer Growth Metast. 10.1177/1179064417696070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saha A, Mohapatra S, Das G, Jana B, Ghosh S, Bhunia D, Ghosh S (2017) Cancer cell specific delivery of photosystem I through integrin targeted liposome shows significant anticancer activity. ACS Appl Mater Interfaces 9(1):176–188. 10.1021/acsami.6b13352 [DOI] [PubMed] [Google Scholar]
- Serrone L, Zeuli M, Sega FM, Cognetti F (2000) Dacarbazine-based chemotherapy for metastatic melanoma: thirty-year experience overview. J Exp Clin Cancer Re 19(1):21–34 [PubMed] [Google Scholar]
- Shakeel F, Haq N, Al-Dhfyan A, Alanazi FK, Alsarra IA (2015) Chemoprevention of skin cancer using low HLB surfactant nanoemulsion of 5-fluorouracil: a preliminary study. Drug Deliv 22(4):573–580. 10.3109/10717544.2013.868557 [DOI] [PubMed] [Google Scholar]
- Shanei A, Sazgarnia A, Dolat E, Hojaji-Najafabadi L, Sehhati M, Baradaran-Ghahfarokhi M (2017) Dual function of gold nanoparticles in synergism with mitoxantrone and microwave hyperthermia against melanoma cells. Asian Pac J Cancer Prev 18(11):2911–2917. 10.22034/APJCP.2017.18.11.2911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi K, Li J, Cao Z, Yang P, Qiu Y, Yang B, He Q (2015a) A pH-responsive cell-penetrating peptide-modified liposomes with active recognizing of integrin αvβ3 for the treatment of melanoma. J Controll Release 217:138–150. 10.1016/j.jconrel.2015.09.009 [DOI] [PubMed] [Google Scholar]
- Shi VY, Tran K, Patel F, Leventhal J, Konia T, Fung MA, Maverakis E (2015b) 100% Complete response rate in patients with cutaneous metastatic melanoma treated with intralesional interleukin (IL)-2, imiquimod, and topical retinoid combination therapy: results of a case series. J Am Acad Dermatol 73(4):645–654. 10.1016/j.jaad.2015.06.060 [DOI] [PubMed] [Google Scholar]
- Sileni VC, Nortilli R, Aversa SML, Paccagnella A, Medici M, Corti L, Monfardini S (2001) Phase II randomized study of dacarbazine, carmustine, cisplatin and tamoxifen versus dacarbazine alone in advanced melanoma patients. Melanoma Res 11(2):189 [DOI] [PubMed] [Google Scholar]
- Singh SP, Alvi SB, Pemmaraju DB, Singh AD, Manda SV, Srivastava R, Rengan AK (2017) NIR triggered liposome gold nanoparticles entrapping curcumin as in situ adjuvant for photothermal treatment of skin cancer. Int J Biol Macromol. 10.1016/j.ijbiomac.2017.11.163 [DOI] [PubMed] [Google Scholar]
- Sisti A, Sisti G, Oranges CM (2014) Topical treatment of melanoma skin metastases with imiquimod: a review. Dermatol Online J 21:2 [PubMed] [Google Scholar]
- Siu KS, Chen D, Zheng X, Zhang X, Johnston N, Liu Y, Min W-P (2014) Non-covalently functionalized single-walled carbon nanotube for topical siRNA delivery into melanoma. Biomaterials 35(10):3435–3442. 10.1016/j.biomaterials.2013.12.079 [DOI] [PubMed] [Google Scholar]
- Sobhani Z, Behnam MA, Emami F, Dehghanian A, Jamhiri I (2017) Photothermal therapy of melanoma tumor using multiwalled carbon nanotubes. Int J Nanomed 12:4509–4517. 10.2147/IJN.S134661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song H, Su X, Yang K, Niu F, Li J, Song J, Gao J (2015a) CD20 antibody-conjugated immunoliposomes for targeted chemotherapy of melanoma cancer initiating cells. J Biomed Nanotechnol 11(11):1927–1946. 10.1166/jbn.2015.2129 [DOI] [PubMed] [Google Scholar]
- Song H, Xu Q, Zhu Y, Zhu S, Tang H, Wang Y, Zhao S (2015b) Serum adsorption, cellular internalization and consequent impact of cuprous oxide nanoparticles on uveal melanoma cells: implications for cancer therapy. Nanomedicine 10(24):3547–3562. 10.2217/nnm.15.178 [DOI] [PubMed] [Google Scholar]
- Sosman JA, Kim KB, Schuchter L, Gonzalez R, Pavlick AC, Weber JS, Ribas A (2012) Survival in BRAF V600—mutant advanced melanoma treated with vemurafenib. N Engl J Med 366(8):707–714. 10.1056/NEJMoa1112302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su P-J, Chen J-S, Liaw C-C, Chang H-K, Wang H-M, Yang T-S, Chang JW-C (2011) Biochemotherapy with carmustine, cisplatin, dacarbazine, tamoxifen and low-dose interleukin-2 for patients with metastatic malignant melanoma. Chang Gung Med J 34(5):478–486 [PubMed] [Google Scholar]
- Sue GR, Hanlon A, Lazova R, Narayan D (2014) Use of imiquimod for residual acral melanoma. BMJ Case Rep. 10.1136/bcr-2014-203826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tchounwou C, Sinha SS, Nellore VBP, Pramanik A, Kanchanapally R, Jones S, Ray PC (2015) Hybrid theranostic platform for second near-IR window light triggered selective two-photon imaging and photothermal killing of targeted melanoma cells. ACS Appl Mater Interfaces 7(37):20649–20656. 10.1021/acsami.5b05225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tupal A, Sabzichi M, Ramezani F, Kouhsoltani M, Hamishehkar H (2016) Dermal delivery of doxorubicin-loaded solid lipid nanoparticles for the treatment of skin cancer. J Microencapsul 33(4):372–380. 10.1080/02652048.2016.1200150 [DOI] [PubMed] [Google Scholar]
- Turza K, Dengel LT, Harris RC, Patterson JW, White K, Grosh WW, Slingluff CL (2010) Effectiveness of imiquimod limited to dermal melanoma metastases, with simultaneous resistance of subcutaneous metastasis. J Cutan Pathol 37(1):94–98. 10.1111/j.1600-0560.2009.01290.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velho TR (2012) Metastatic melanoma—a review of current and future drugs. Drugs Context. 10.7573/dic.212242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volder MFLD, Tawfick SH, Baughman RH, Hart AJ (2013) Carbon nanotubes: present and future commercial applications. Science 339(6119):535–539. 10.1126/science.1222453 [DOI] [PubMed] [Google Scholar]
- Wang H, Sheng W (2017) 131I-traced PLGA-lipid nanoparticles as drug delivery carriers for the targeted chemotherapeutic treatment of melanoma. Nanoscale Res Lett 12:365. 10.1186/s11671-017-2140-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Yang F, Zhang H-X, Zi X-Y, Pan X-H, Chen F, Hu Y-P (2013) Cuprous oxide nanoparticles inhibit the growth and metastasis of melanoma by targeting mitochondria. Cell Death Dis 4(8):e783. 10.1038/cddis.2013.314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S, Riedinger A, Li H, Fu C, Liu H, Li L, Pellegrino T (2015) Plasmonic copper sulfide nanocrystals exhibiting near-infrared photothermal and photodynamic therapeutic effects. ACS Nano 9(2):1788–1800. 10.1021/nn506687t [DOI] [PubMed] [Google Scholar]
- Wang P, Zhang L, Zheng W, Cong L, Guo Z, Xie Y, Jiang X (2018) Thermo-triggered release of CRISPR-Cas9 system by lipid-encapsulated gold nanoparticles for tumor therapy. Angew Chem Int Ed. 10.1002/anie.201708689 [DOI] [PubMed] [Google Scholar]
- Xu H, Hu M, Yu X, Li Y, Fu Y, Zhou X, Li J (2015) Design and evaluation of pH-sensitive liposomes constructed by poly(2-ethyl-2-oxazoline)-cholesterol hemisuccinate for doxorubicin delivery. Eur J Pharm Biopharm 91:66–74. 10.1016/j.ejpb.2015.01.030 [DOI] [PubMed] [Google Scholar]
- Yamazaki N, Yamakawa S, Sugimoto T, Yoshizaki Y, Teranishi R, Hayashi T, Kono K (2017) Carboxylated phytosterol derivative-introduced liposomes for skin environment-responsive transdermal drug delivery system. J Liposome Res 0(0):1–10. 10.1080/08982104.2017.1369995 [DOI] [PubMed] [Google Scholar]
- Yang Y-S, Carney RP, Stellacci F, Irvine DJ (2014) Enhancing radiotherapy by lipid nanocapsule-mediated delivery of amphiphilic gold nanoparticles to intracellular membranes. ACS Nano 8(9):8992–9002. 10.1021/nn502146r [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Tai X, Shi K, Ruan S, Qiu Y, Zhang Z, He Q (2016) A new concept of enhancing immuno-chemotherapeutic effects against B16F10 tumor via systemic administration by taking advantages of the limitation of EPR effect. Theranostics 6(12):2141–2160. 10.7150/thno.16184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang C-C, Hung C-F, Chen B-H (2017) Preparation of coffee oil-algae oil-based nanoemulsions and the study of their inhibition effect on UVA-induced skin damage in mice and melanoma cell growth. Int J Nanomed 12:6559–6580. 10.2147/IJN.S144705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu B, Wang Y, Yu X, Zhang H, Zhu J, Wang C, Zhu H (2017) Cuprous oxide nanoparticle-inhibited melanoma progress by targeting melanoma stem cells. Int J Nanomed 12:2553–2567. 10.2147/IJN.S130753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Teodoro JG, Nadeau JL (2015) Intratumoral gold-doxorubicin is effective in treating melanoma in mice. Nanomed Nanotechnol Biol Med 11(6):1365–1375. 10.1016/j.nano.2015.04.001 [DOI] [PubMed] [Google Scholar]
- Zhang Q, Zhang L, Li Z, Xie X, Gao X, Xu X (2017) Inducing controlled release and increased tumor-targeted delivery of chlorambucil via albumin/liposome hybrid nanoparticles. AAPS Pharm Sci Tech 18(8):2977–2986. 10.1208/s12249-017-0782-5 [DOI] [PubMed] [Google Scholar]
- Zhuang X, Wu T, Zhao Y, Hu X, Bao Y, Guo Y, Zhang Z (2016) Lipid-enveloped zinc phosphate hybrid nanoparticles for codelivery of H-2Kb and H-2Db-restricted antigenic peptides and monophosphoryl lipid A to induce antitumor immunity against melanoma. J Controll Release 228:26–37. 10.1016/j.jconrel.2016.02.035 [DOI] [PubMed] [Google Scholar]