Abstract
BACKGROUND
Alzheimer's disease‐related biomarkers detect pathology years before symptoms emerge, when disease‐modifying therapies might be most beneficial. Remote cognitive testing provides a means of assessing early cognitive changes. We explored the relationship between neurodegenerative biomarkers and cognition in cognitively normal individuals.
METHODS
We remotely deployed 13 computerized Cognitron tasks in 255 Insight 46 participants. We generated amyloid load and positivity, white matter hyperintensity volume (WMHV), whole brain and hippocampal volumes at age 73, plus rates of change over 2 years. We examined the relationship between Cognitron, biomarkers, and standard neuropsychological tests.
RESULTS
Slower response time on a delayed recognition task predicted amyloid positivity (odds ratio [OR] = 1.79, confidence interval [CI]: 1.15, 2.95), and WMHV (1.23, CI: 1.00, 1.56). Brain and hippocampal atrophy rates correlated with poorer visuospatial performance (b = ‐0.42, CI: ‐0.80, ‐0.05) and accuracy on immediate recognition (b = ‐0.01, CI: ‐0.012, ‐0.001), respectively. Standard tests correlated with Cognitron composites (rho = 0.50, p < 0.001).
DISCUSSION
Remote computerized testing correlates with standard supervised assessments and holds potential for studying early cognitive changes associated with neurodegeneration.
Highlights
70% of the Online 46 cohort performed a set of remote online cognitive tasks.
Response time and accuracy on a memory task predicted amyloid status and load (SUVR).
Accuracy on memory and spatial span tasks correlated with longitudinal atrophy rate.
The Cognitron tasks correlated with standard supervised cognitive tests.
Online cognitive testing can help identify early AD‐related memory deficits.
Keywords: Alzheimer's disease, amyloid, biomarkers, computerized cognitive testing, dementia, Insight 46, memory, neurodegeneration
1. BACKGROUND
The pathological changes of Alzheimer's disease (AD) begin years before symptoms emerge. 1 , 2 , 3 Recent trials of anti‐amyloid therapies showed beneficial effects in individuals with earlier symptomatic disease, 4 , 5 and these medications are now being trialed in asymptomatic individuals with AD pathology. 6 However, these therapies come with significant risks, 4 , 5 and not all individuals with AD pathology will develop cognitive decline. 7 Therefore, there is a pressing need to identify early cognitive signs associated with AD pathology, which could aid in targeting interventions to those most likely to benefit. Additionally, tools sensitive to subtle changes in memory decline are needed as outcome measures for presymptomatic clinical trials.
Among the available biomarkers included in the National Institute on Aging and Alzheimer's Association criteria, 8 cerebral β‐amyloid (Aβ) deposition, detectable in vivo through Aβ‐positron emission tomography (PET) imaging, is considered core to identify AD at both its asymptomatic and symptomatic phase. 9 Magnetic resonance imaging (MRI) provides additional information about the stage and magnitude of the disease. At the early stages of the disease, where the degree of atrophy measured cross‐sectionally may not be sufficient to indicate abnormality, techniques that measure the atrophy rate on serial MRI (e.g., the boundary shift integral) can provide a more sensitive measure of neurodegeneration. 10 , 11 Vascular burden quantified on MRI as white matter hyperintensity volume (WMHV) can be used as a marker of co‐pathology. Increased WMHV is predictive of the downstream neurodegeneration in the early symptomatic stage of AD and shorter time to AD development. 12 , 13
It is challenging to distinguish the earliest cognitive impairments emerging from AD pathology from those associated with normal aging. These impairments may be too subtle, domain‐specific, or influenced by demographic factors to be easily detected using standard on‐paper assessments. 14 , 15 Computerized cognitive assessment offers advantages, providing simultaneous measurement of multiple behavioral measures and reaction time, and greater precision. Computerized testing can be scaled for difficulty and complexity to target cognitive impairments in the clinical population of interest. This enhances sensitivity and has the potential to reduce the sample size required for appropriate statistical power in clinical trials involving individuals at the early stage of AD. 16 , 17 Computerized testing can also be deployed longitudinally with higher stimuli variability and reduced learning effects. To maximize feasibility and sensitivity, tasks should be brief, easy to perform, targeted to cognitive domains thought to be affected in AD, and validated against gold‐standard assessments.
We remotely deployed 13 computerized cognitive tasks in the Online 46 study, the remote cognitive sub‐study of the Medical Research Council (MRC) National Survey of Health and Development (NSHD) study, a population‐based cohort of individuals born in England, Scotland, and Wales in 1946. 18 , 19 The aims of this study were to (a) identify tasks that were sensitive to markers of AD pathology and neurodegeneration in individuals without dementia; and (b) to determine their degree of correlation with standard supervised neuropsychological assessments. The NSHD cohort is particularly suited for the scope of this study. At their current age, the study members are generally at risk of accumulation of AD pathology, prior to the development of dementia, enabling cognitive measures to be benchmarked in relation to these pre‐clinical biological changes. 20 , 21
Our primary hypothesis was that memory performance would demonstrate significant sensitivity to AD pathology as memory impairment is widely recognized as one of the cognitive hallmarks of AD, emerges early in the disease process, and has previously been shown to be related to amyloid deposition in this cohort. 14 , 22 , 23 However, memory impairments may occur in non‐AD dementias, 24 and amyloid deposition has been reported to impact other domains, such as language, attention, visuospatial abilities, and working memory. 24 , 25 We therefore took an additional exploratory approach, administering a broad set of cognitive tasks to investigate the relationship of AD and vascular pathology with other cognitive sub‐domains.
2. METHODS
2.1. Participants and recruitment
The NSHD cohort includes extensive clinical, physical, and cognitive characterization of a group of 2360 active study members born in in England, Scotland, and Wales within the same week of March 1946 (https://nshd.mrc.ac.uk/). Online 46 is a remote cognitive sub‐study of the NSHD designed to assess the feasibility and utility of remote computerized ‘Cognitron’ cognitive testing in 1776 eligible individuals of this cohort. Online 46 took place between June and September 2023 and resulted in a final sample of 813 participants who undertook a battery of 13 cognitive tasks within 4 weeks of invitation. Participants were invited via email and asked to complete the tasks under unsupervised conditions using any electronic device (e.g., tablets, phones, computers) and web browser. Written instructions were provided at the beginning of each task, followed by a set of practice trials to confirm that the participants properly understood how to complete the tasks.
RESEARCH IN CONTEXT
Systematic review: A PubMed search revealed growth in research on remote computerized cognitive testing for dementia detection. While many tools have been developed, most lack validation in unsupervised conditions or testing across multiple devices.
Interpretation: Remote computerized cognitive testing for dementia risk is feasible and correlates with standard assessments. Response time (RT) and accuracy on a delayed memory recognition task was positively associated with amyloid positivity and load, indicating accelerated forgetting. Cognitive impairments in β‐amyloid (Aβ)‐positive individuals were more associated with RT than accuracy. Slower RT significantly predicted poorer accuracy in amyloid‐positive individuals, potentially reflecting longer recognition time which limits performance and masks memory deficits through compensatory mechanisms. Memory and attentional tasks were linked to MRI measures of disease progression (i.e., longitudinal atrophy change and vascular load).
Future directions: Future studies should focus on the longitudinal administration of remote cognitive testing to map impairment trajectories and their association with AD pathology accumulation.
The present study focuses on 274 participants who undertook the Online 46 remote cognitive battery and were also part of the Insight 46 neuroimaging sub‐study. Insight 46 is a prospective longitudinal observational neuroimaging sub‐study of the NSHD. It involves detailed clinical, neuropsychological testing, and neuroimaging data, which have been used for the present study. The recruitment procedure is summarized in Figure 1, and the data collection methods for Insight 46 have been described previously. 18 , 19 , 26 As our focus was on presymptomatic cognitively normal individuals, 19 of the 274 participants were excluded as they had evidence of major brain disorders, which resulted in the final study sample of 255 individuals. The presence of a brain disorder was determined using phase 1 and phase 2 data of the Insight 46 neuroimaging sub‐study. Decisions were made by a panel of neurologists and study clinicians based on their clinical judgment, participants’ radiological findings, and self‐reported diagnoses. A brain disorder was defined as any clinically diagnosed neurodegenerative disorder, any psychiatric disorder requiring an antipsychotic treatment, depression necessitating electroconvulsive shock therapy, evidence of traumatic brain injury or significant neurosurgery, multiple sclerosis, evidence of an ischemic or hemorrhagic cortical stroke, radiological evidence of a brain malignancy, and mild cognitive impairment (MCI).
FIGURE 1.
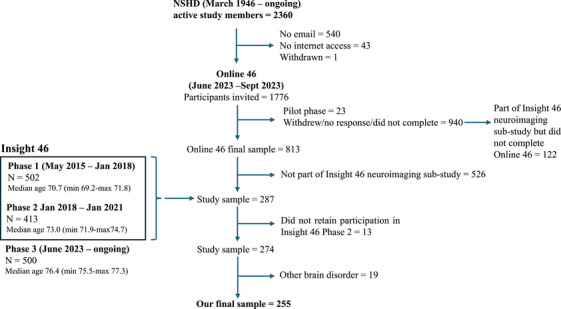
Recruitment flowchart.
As a separate group, the demographics of 122 participants from the Insight 46 neuroimaging sub‐study who declined the Online 46 cognitive testing are reported in this paper for means of comparison.
2.2. Cognitive testing
A library of computerized tasks developed in HTML5 with JavaScript is available on the Cognitron platform (https://www.cognitron.co.uk). 27 , 28 , 29 , 30 , 31 , 32 A battery of 13 of these tasks was deployed remotely to 813 participants of the Online 46 study group at age 77. Compliance, adherence, and usability of the tasks have been studied and reported elsewhere. 33 The tasks were selected to cover multiple cognitive domains and target cognitive abilities thought to be impaired early in AD, while being brief and understandable by the study participants. Task descriptions are in Figure 2 and Table 1. Participants received written instructions and completed a brief sequence of practice trials before starting each task to ensure they understood how to complete them. Average completion time for the whole battery was 39 min (mean time per task = 4.55 min, min = 1.73, max = 5.01). All tasks yielded a summary score that was accuracy‐based (total correct answers) and a secondary score that was the median response time (RT), except for motor control which was measured primarily with an RT score (Table 1). As the testing sessions were conducted under unsupervised conditions, we defined signs of non‐compliance for each Cognitron task to exclude participants who did not properly engage. Specifically, one participant was excluded from 2D Manipulations for repetitively clicking in the same location of the screen. Two participants were excluded from Digit Span—one for clicking on another browser page while performing the task and the other for achieving an accuracy score below the established threshold for engagement (located on the left tail of the distribution of accuracy scores across the entire cohort). Three participants were also excluded from Spotter as they showed an accuracy score below the expected threshold for engagement. In a separate analysis examining data from the whole Online 46 group (N = 813) and reported elsewhere, 33 9% of individuals showed indicators of lack of compliance with the choice reaction time (CRT) task. The task was therefore considered unreliable and excluded from further analysis.
FIGURE 2.

Top: Illustrations of the Cognitron tasks. Bottom: Network plot showing clustering of the primary task scores, and the cognitive domains measured. The colors of the connecting lines indicate the 5 components derived from the PCA. A smaller distance and higher opacity of the connecting line indicate a stronger correlation. PCA, principal component analysis.
TABLE 1.
Details of the Cognitron tasks.
| Task | Strategy | Cognitive domain | Mean duration (s) | Std duration | Summary score | RT score | End criteria/trials |
|---|---|---|---|---|---|---|---|
| Objects immediate and delayed recognition | Participants are shown a sequence of target objects. Afterward, they are asked to identify these targets in different arrays of objects. This task is repeated at the end of the battery to measure delayed memory recognition. | Memory |
Immediate:183.9 Delayed:104.0 |
75.5 30.9 |
Total correct | Duration to complete task | 20 objects |
| Motor control | Participants are shown a red target appearing at different locations of the screen and asked to tap on it as quickly as possible. | Processing speed | 178.3 | 41.5 | Median RT | Median RT | 30 trials |
| CRT* | Participants are shown an arrow pointing either left of right and have to respond accordingly to it tapping on the left or right‐hand side of the screen. | Processing speed | 121.2 | 8.8 | Median RT | Median RT | 60 trials |
| Blocks | Participants are asked to remove blocks of different colors and shapes from one array to match the target array. | Visuospatial abilities | 187.6 | 102.6 | Total correct | Median RT | 15 trials |
| Digit span | Participants are asked to memorise a list of digits and then repeat it by tapping the numbers on a keypad. The list of digits increases in length every correct trial. The task is interrupted after three consecutive incorrect trials. | Executive functions | 259.0 | 181.4 | Total correct | Median RT | 3 consecutive failures |
| Spatial span | Participants are asked to memorise a sequence of gray square appearing at different location of a 4X4 grid. The number of squares increases in length every correct trial. The task is interrupted after three consecutive incorrect trials. | Visuospatial abilities/attention | 145.4 | 54.7 | Total correct | Median RT | 3 consecutive failures |
| Stroop | Participants indicate the color of a tile by tapping on one of two words: "blue" and "red," which are colored either blue or red. A box will indicate which modality they will have to provide the answer in (i.e., the color, or text of the word). | Executive functions | 281.3 | 75.229 | Total correct | Median RT | 60 trials |
| 2D Manipulations | Participants are shown a target array of colored squares and asked to identify this among four. The target is rotated through either 90, 180 or 270 degrees. | Visuospatial abilities | 120.7 | 1.1 | Total correct | Median RT | 3‐min timer |
| Word definitions | Participants are shown a word and 4 possible definitions and asked to tap on the correct definition within a designated amount of time. | Language | 206.5 | 50.6 | Total correct | Median RT | 28 trials, 20 seconds per trial |
| Verbal reasoning | Participants are shown different combinations of geometric shapes and asked to indicate whether the statement describing the shapes is true of false. | Language | 172.1 | 39.0 | Total correct | Median RT | 3‐min timer |
| Spotter | Participants see numbers displayed inside a pixelated square. Their task is to click on the square immediately upon spotting the number "0." The stimuli are calibrated to be hard to detect, appearing on the screen for only 100 ms, in rapid succession and are degraded with a mask. | Processing speed | 300.6 | 0.6 | Total correct | Median RT | 4‐min timer |
| Forager | This is a reversal learning task that presents a continuous stream of shapes. Participants need to click on the shapes until they find the correct rule (e.g. tap on circles). They will do so based on the feedback they receive (correct/incorrect). After they follow the rule correctly for 6 consecutive trials, they receive negative feedback and a new rule will be generated (e.g., tap on squares). | Processing speed | 180.3 | 0.9 | Total correct | Median RT | 3‐min timer |
Abbreviations: CRT, Choice reaction time; RT, response time; s, seconds.
aExcluded from further analysis.
Participants had already completed a comprehensive Insight 46 battery of standard supervised neuropsychological assessments at age 73. 18 A subset of these standard measures mapping the cognitive domains measured by the Cognitron tasks was selected to study their association with the tasks (Table S1). These were the Mini‐Mental State Examination (MMSE), 34 the Digit Symbol Substitution Test (DSST) of the Wechsler Adult Intelligence Scale‐Revised (WAIS‐R), 35 the Logical Memory test of the Wechsler Memory Scale‐Revised (WMS‐R), 36 the 12‐item Face‐Name Associative Memory Exam (FNAME‐12), 37 the Preclinical Alzheimer cognitive composite (PACC) generated from the measures listed above, 38 and a Trail Making B test, 37 the Matrix Reasoning task of the Wechsler Abbreviated Scale of Intelligence (WASI), 39 the Adult Memory and Information Processing Battery's Complex Figure Drawing task, 40 a 15‐item word learning task, 41 the Graded Naming task, 42 and a CRT task. 43
Additional clinical and demographic measures were used in the analyses. Participants’ childhood cognitive abilities were measured at age 8, 11, and 15 using four tests of verbal and nonverbal ability devised by the National Foundation for Education Research. 44 We utilized the scores obtained at age 8. In cases where data were not available at this age, we utilized the scores derived at ages 11 or 15. Adult socioeconomic position was defined based on the occupation held at age 53 according to the UK Registrar General's classification of occupation. 26 Apolipoprotein E (ApoE) status was determined from a blood sample collected at age 53 and dichotomized into ε4 carrier or non‐carrier.
2.3. Neuroimaging biomarkers
Participants underwent a 60‐min scan at age ∼69–71 and ∼71–73 on a single Biograph mMR 3T PET/MRI scanner (Siemens Healthcare, Erlangen), with intravenous injection of 370 MBq of 18F‐Florbetapir (Amyvid). A𝛽 burden was assessed during a 10‐min period ∼50 min correction. 45 A global standard uptake value ratio (SUVR) was calculated from a cortical gray matter composite with an eroded subcortical white matter reference region. Positive or negative A𝛽 status was determined by applying a Gaussian mixture model applied to SUVR values, with the 99th percentile of the A𝛽‐negative Gaussian as the cut‐point (0.6104). 14 In cases where data were not available at age 73 (n = 28), amyloid status and the extent of amyloid deposition were assessed based on the scans obtained at age 71. To evaluate the reliability of our imputation methodology, we performed a linear regression with SUVR at age 71 as the predictor and SUVR at age 73 as the dependent variable. To further assess the robustness of our findings, we conducted a sensitivity analysis by excluding the 28 participants with missing SUVR data at age 73. In this subset, we examined the relationship between cognitive performance on the Cognitron tasks and SUVR.
Whole brain, hippocampal, and ventricular volume at age 73 were calculated as previously reported. 18 The rates of change in volumes between age 71 and 73 were calculated using the boundary shift integral (BSI). 10 Hippocampal volumes and BSI were derived as the sum of left and right structures. We prioritized rates of atrophy accumulation over longitudinal measures of SUVR, as they are well‐established indicators of disease progression and show a strong correlation with cognitive impairments. 11 , 57 Additionally, white matter hyperintensity volumes were obtained from the MRI scans performed at age 73 using an unsupervised automated algorithm, Bayesian Model Selection to T1 and FLAIR images, generating a global WMHV, which included subcortical gray matter but not infratentorial regions. Finally, total intracranial volume (TiV) was calculated from T1 images using the tissues utility in Statistical Parametric Mapping (SPM) version 12. 46
2.4. Statistical analysis
To account for variations in participant numbers across tasks, power calculations were performed to assess the statistical power achieved in analyzing the effect of the Cognitron task with the smallest dataset (i.e., Spotter) on amyloid status and neurodegeneration. Neurodegeneration was measured using whole‐brain BSI, which has been shown to offer greater sensitivity than cross‐sectional atrophy measures for discriminating individuals with preclinical AD. 47
Demographic differences between Aβ‐positive and Aβ‐negative individuals were assessed using chi‐squared tests for gender, handedness, and education level, and two sample t‐test for childhood cognitive abilities. Differences in cognitive performance on the standard neuropsychological assessments and neuroimaging features were investigated using two‐sample t‐tests for the PACC and whole brain volume, and Wilcoxon rank‐sum tests for the remaining non‐normally distributed variables.
We used linear regression to adjust the standard supervised assessments and the Cognitron accuracy and RT scores for demographic characteristics which may influence cognitive performance. Specifically, we included sex, handedness, education level, and the device used to perform the tasks as predictors, and the cognitive scores as dependent variables. Residuals were calculated as the difference between the observed score and the predicted score. These would represent the portion of cognitive performance not explained by the demographic factors. This effectively removes the influence of these variables. The residuals were then scaled by subtracting the mean and dividing by the standard deviation.
We examined the inter‐correlations between the Cognitron tasks to confirm that they encompassed distinct cognitive domains. A principal component analysis (PCA) with orthogonal varimax rotation was conducted using the Kaiser convention (eigenvalue > 1). Components were interpreted based on the tasks that had the highest and discrete loadings on them.
We then investigated the relationship between the scaled Cognitron scores and biomarkers of neurodegeneration. Multivariable logistic regression was utilized with accuracy and RT measures of Cognitron as predictors and amyloid positivity as dependent variable. A secondary linear regression analysis tested the relationship between the Cognitron task that significantly predicted amyloid status and the extent of amyloid deposition (SUVR). We also ran a linear regression analysis to explore whether the relationship between RT and accuracy on this task differed by amyloid status. We included RT as the independent variable and accuracy as the dependent variable, with amyloid status as an interaction term. We then ran separate linear regression models for each amyloid status group, using RT as the independent variable and accuracy as the dependent variable in each model.
To examine the relationships between the online cognitive scores and MRI variables, linear regression models were conducted inputting the Cognitron accuracy and RT scores as predictors and the volume and BSI for the whole brain, hippocampi, and ventricles, as well as the WMHV as dependent variables. To handle the skewed distribution of WMHV, generalized linear models with a gamma log link function were employed. All models were conducted both with and without adjustment for childhood cognitive abilities. The models including SUVR, whole brain, hippocampal, ventricular, and WMHV as dependent variables were corrected for TiV.
Additionally, we looked at the correlation between the Cognitron battery and the battery of standard supervised cognitive assessments. We extracted a composite score from the Cognitron tasks which had shown significant associations with biomarkers of neurodegeneration, and a total composite score from all the Insight 46 standard supervised assessments. This was done using PCA, where the first principal component was taken as a global composite score. Spearman correlation was used to compare the derived Cognitron and standard assessments total composite scores. The Cognitron composite score was also correlated with the PACC to assess its utility as measure of early AD‐related cognitive changes. Finally, we examined the correlation between a memory composite score derived from the Cognitron memory tasks (i.e., Objects Memory Immediate and Delayed Recognition accuracy and RT) and a composite score generated from a subset of Insight 46 standard assessments targeting memory abilities (i.e., Logical Memory, FNAME‐12, and AMIPB—Complex Figure Drawing). These composite scores were calculated using PCA, with the first unrotated component serving as the composite memory score. The aim of these analyses was to confirm the diagnostic value of this subset of tasks, comparing their effectiveness to gold‐standard assessments, and to inform the development of a more concise Cognitron battery for future studies.
Finally, to determine the generalizability of our results, we analyzed the demographics of participants of the Insight 46 neuroimaging sub‐study who attempted the battery of Cognitron tasks compared to those who did not (N = 122). Chi‐squared tests were used to examine differences in gender, education, handedness, adult socioeconomic status, amyloid levels, and ApoE status. Two sample t‐tests were conducted to assess differences in childhood cognitive abilities and PACC scores. To evaluate differences in MMSE scores, we used the Wilcoxon rank‐sum test, as the data was not normally distributed. We also compared cognitive performance of our study group to that of participants of the Online 46 study who were not part of the Insight 46 neuroimaging sub‐study (N = 526). We used Wilcoxon rank‐sum test to look at performance differences on the Cognitron tasks and the Addenbrooke's Cognitive Examination III (ACE‐III), which was the standard cognitive assessment available in both cohorts.
3. RESULTS
A total of 255 individuals were included in the final analysis (51.76% males). Table S2 illustrates the number of participants included in the analysis for each task. A minimal statistical power of 80% was achieved when analyzing the effect of the Cognitron task with the smallest number of data points (i.e., Spotter) on amyloid status (alfa power = 81%) and whole brain BSI (alfa power = 94%) (Figure S1). The factor analysis indicated that the Cognitron tasks clustered in five interpretable cognitive domains, explaining 61.34% of variance (Figure 2 and 3).
FIGURE 3.

PCA applied to the Cognitron summary scores. Top: Scree plot showing eigenvalues (components extracted based on eigenvalues > 1). Bottom: Loadings of the Cognitron summary scores onto the derived cognitive components. PCA, principal component analysis.
3.1. Demographic and cognitive characteristics
Participants had a mean MMSE score of 29.22 (std = 0.91), a mean ACE‐III score of 94.46 (std = 3.63), and a mean PACC score of 0.14 (std = 0.62), which do not reach thresholds associated with the presence of MCI or dementia. 48 , 49 , 50 Amyloid status was missing for 14 individuals at an SUVR cutoff of 0.61, 14 and 66 (27.39%) were Aβ‐positive. No significant demographic or neuroimaging differences were observed between Aβ‐positive and Aβ‐negative participants. Full details of participants’ demographics, neuroimaging characteristics, and cognitive performance on the standard assessments are shown in Table 2.
TABLE 2.
Demographic characteristics of the study group and differences between Aβ‐positive and Aβ‐negative individuals.
| Parameter | Total | Aβ‐positive participants | Aβ‐negative participants | Group differences |
|---|---|---|---|---|
| N | 255 a | 66 | 175 | |
| Sex, N (%) Male | 132 (51.76%) | 33 (50.00%) | 92 (52.57%) | Χ2 (1) = 0.04, p = 0.83 |
| Education (N) | Χ2 (2) = 5.17, p = 0.08 | |||
| <16 years | 43 (16.86%) | 10 (15.15%) | 30 (17.14%) | |
| High School | 155 (60.78%) | 47 (71.21%) | 99 (56.57%) | |
| Higher‐level degree | 57 (22.35%) | 9 (13.63%) | 46 (26.29%) | |
| Handedness N (%) right‐handed | 234 (91.76%) | 57 (86.36%) | 164 (93.71%) | X2 (1) = 1.52, p = 0.22 |
| Childhood cognitive ability mean (sd) | 0.46 (0.72) | 0.44 (0.70) | 0.48 (0.72) | t (119.21) = 0.78, p = 0.74, CI :[‐0.24, 0.16] |
|
MMSE mean (sd) |
29.22 (0.91) | 29.16 (0.96) | 29.22 (0.92) | W = 5559, p = 0.58 |
|
PACC mean (sd) |
0.14 (0.62) | 0.09 (0.68) | 0.15 (0.61) | t (105.68) = 0.78, p = 0.44, CI :[‐0.74, 0.05] |
|
Logical memory (WMS‐R) Mean (sd) |
12.49 (3.38) | 12.62 (3.55) | 12.40 (3.36) |
W = 5578, p = 0.68 |
|
DSST (WAIS‐R) Mean (sd) |
48.20 (10.28) | 47.53 (10.41) | 48.51 (10.47) | W = 6153.5, p = 0.43 |
|
FNAME‐12 Mean (sd) |
69.66 (17.51) | 67.55 (17.45) | 70.50 (17.60) | W = 6323.5, p = 0.26 |
|
ACE‐III Mean (sd) |
94.46 (3.63) | 94.98 (2.92) | 94.29 (3.90) | W = 3483, p = 0.44 |
|
Matrix reasoning (WASI) Mean (sd) |
25.07 (3.86) | 24.88 (4.23) | 25.13 (3.85) | W = 5798.5, p = 0.91 |
| CRT mean RT in milliseconds (sd) | 811.93 (103.00) | 812.27 (95.71) | 810.81 (104.87) | W = 5052, p = 0.61 |
|
Graded naming test Mean (sd) |
23.25 (3.38) | 23.44 (3.26) | 23.23 (3.33) | W = 5665, p = 0.93 |
|
AMIPB—Complex figure drawing (7‐day recall) Mean (sd) |
49.80 (14.20) | 48.26 (14.00) | 50.80 (14.31) | W = 6406, p = 0.13 |
| Whole brain volume | 1099.32 (95.06) | 1093.65 (91.30) | 1101.63 (97.76) |
t(117.45)= 0.57, p = 0.57, [CI: ‐19.96, 35.92] |
| Whole brain BSI | 5.75 (2.89) | 5.84 (2.90) | 5.72 (2.89) | W = 4288, p = 0.76 |
| Hippocampal volume | 3.07 (0.34) | 3.01 (0.27) | 3.10 (0.36) | W = 4973.5, p = 0.32 |
| Hippocampal BSI | 0.037 (0.039) | 0.037 (0.038) | 0.038 (0.040) | W = 4468, p = 0.88 |
| Ventricles volume | 32.60 (15.62) | 32.43 (14.26) | 32.66 (16.18) | W = 4449, p = 0.76 |
| Ventricles BSI | 1.21 (0.81) | 1.31 (0.89) | 1.16 (0.78) | W = 3956, p = 0.25 |
| WMHV | 5652.06 (6216.30) | 5960.57 (5397.22) | 5526.23 (6533.46) | W = 4161, p = 0.18 |
Abbreviations: ACE‐3, Addenbrooke's Cognitive Examination III; BSI, boundary shift integral; CRT, Choice reaction time; DSST, Digit Symbol Substitution Test; FNAME‐12, Face‐Name Associative Memory Exam; MMSE, Mini‐Mental State Examination; PACC, Preclinical Alzheimer cognitive composite; SUVR, standard uptake volume ratio; WAIS‐R, Wechsler Adult Intelligence Scale‐Revised; WASI, Wechsler Abbreviated Scale of Intelligence; WMHV, white matter hyperintensity volume; WMS‐R, Wechsler Memory Scale‐Revised.
Missing amyloid status for 14 participants.
Regarding the 122 Online 46 participants who were also part of the Insight 46 neuroimaging sub‐study but did not attempt the Cognitron battery, 36 (30.5%) were Aβ‐positive. Demographic differences between individuals who did and did not attempt the Cognitron battery are reported in Table S3. In brief, the latter group showed significantly lower childhood cognitive abilities (t(665.44) = ‐6.69, CI: ‐0.38 to ‐0.21), and lower performance on the MMSE (W = 13229, p < 0.001) and PACC (t(205.76) = ‐3.85, CI:‐0.44 to ‐0.14). We also examined cognitive differences between our study group and members of the Online 46 study cohort who did not participate in the Insight 46 neuroimaging sub‐study (N = 526) (Table S4). There was no significant difference in ACE‐III scores between the two groups. However, participants in the Insight 46 neuroimaging sub‐study scored lower on the Verbal Reasoning (W = 67009, p = 0.02) and Digit Span (W = 69210, p = 0.02) tasks, and had slower RT on the Stroop task (W = 55937, p = 0.03).
3.2. Associations with neurodegenerative biomarkers
We found that a one‐unit increase in RT on the Objects delayed recognition task at age 77 corresponded to a 1.79‐fold increase in the odds of being Aβ‐positive at age 73 (CI: 1.15, 2.95). Conversely, a one‐unit increase in accuracy on the same task was associated with a 0.54‐fold reduction in the odds of having positive amyloid status (CI: 0.32, 0.92). When examining these associations using amyloid as a continuous measure, we confirmed that higher SUVR at age 73 was associated with slower RT (0.05, CI: 0.01, 0.10) and reduced accuracy (‐0.04, CI:‐0.07, ‐0.003) on Objects delayed recognition performed at age 77 (Figure 4).
FIGURE 4.
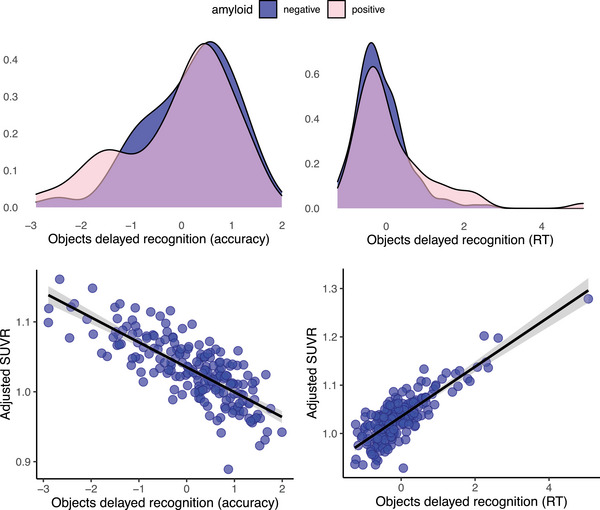
Top: Density plots showing performance differences on the Objects delayed recognition task (accuracy and RT scores) between the amyloid positive and negative groups. Bottom: Association between SUVR and the Objects delayed recognition task (accuracy and RT scores). RT, reaction time; SUVR, standard uptake value ratio.
We observed a strong correlation between SUVR at age 71 and 73 (R2 = 0.92, β = ‐1.20, CI: 1.15, 1.24), suggesting the reliability of using SUVR at age 71 when missing at age 73 for 28 participants (Figure S2). Accordingly, when removing these 28 participants from the study group, the results remained unchanged. A one‐unit increase in RT on Objects delayed recognition was associated with a 1.74‐fold increase in the odds of being amyloid positive (CI: 1.11, 2.79) and a 0.05 increase in SUVR (CI: 0.01, 0.10). In contrast, a one‐unit increase in accuracy on the Objects delayed recognition task was linked to a 0.51‐fold decrease in the odds of having a positive amyloid status (CI: 0.29, 0.87) and a 0.04 decrease in SUVR (CI: ‐0.07, ‐0.004).
Using linear regression, we also investigated whether the effect of RT on the accuracy of the Objects delayed recognition task differed in the Aβ‐positive and negative groups. We found a significant interaction between RT and amyloid status affecting task accuracy (‐0.32, CI: ‐0.63, ‐0.01). Further analysis revealed that slower performance was significantly associated with lower accuracy in the Aβ‐positive (‐0.49, CI: ‐0.73, ‐0.25) but not negative (‐0.17, CI: ‐0.38, 0.05) group.
The results of the regression models examining the association between the Cognitron tasks and MRI‐based biomarkers are presented in Figure 5. A one‐unit decrease in accuracy on the Objects immediate recognition task at age 77 was associated with a 0.01 mL/year higher hippocampal atrophy rate between ages 71 and 73 (CI: 0.001, 0.012). Additionally, for each unit increase in RT on the Objects delayed recognition task performed at age 77, the WMHV quantified at age 73 showed a relative increase of 1.23 (CI: 1.00, 1.56). We also found that a one‐unit decrease in accuracy on the Spatial span task corresponded to a 0.42 mL/year increase in whole brain atrophy rate (CI: 0.05, 0.80) and a 0.84 relative increase in cross‐sectional WMHV (CI: 0.72, 0.98). Finally, we observed that every unit reduction on the Word Definition task at age 77 was associated with an increase of 2.30 mL in ventricular volume at age 73 (CI: 0.31, 4.30). When removing childhood cognitive abilities from this model, there was a reduction in effect size so that the association became non‐significant (‐1.17, CI:‐3.01, 0.66). No significant associations were observed between the cognitive tasks and cross‐sectional measures of whole brain and hippocampal volume. A full summary of the models’ results is reported in Table S5.
FIGURE 5.

Scatter plots showing the associations between the Cognitron tasks and the biomarkers of AD, white matter pathology and neurodegeneration. The values represent the predicted outcomes from the regression models, adjusted for the included covariates (i.e., childhood cognitive abilities and total intracranial volume). AD, Alzheimer's disease.
3.3. Validation against standard supervised neuropsychological assessments
The Cognitron composite score derived from the Cognitron task scores which predicted the biomarkers (Objects delayed and immediate recognition, Spatial span, and Word definitions) explained 30.04% of the variance and showed a positive correlation with the total composite score of the standard assessments (rho = 0.50, p < 0.001), which explained 26.60% of the variance, and the PACC (rho = 0.32 p < 0.001). The Cognitron memory composite score, accounting for 54.63% of the variance, significantly correlated with the standard assessments’ memory composite score, which explained 43.10% of the variance (rho = 0.43, p < 0.001) (Figure 6).
FIGURE 6.

Plots showing the association between: (A) the Cognitron composite score generated from the tasks which predicted the imaging biomarkers and the Insight 46 total composite score, (B) the Cognitron composite score generated from the tasks which predicted the imaging biomarkers and the PACC, and (C) the Cognitron memory composite score and the Insight 46 standard memory composite score. One participant had notably low Cognitron and Insight 46 memory composite scores, but excluding them from the analysis did not affect the statistical results. PACC, Preclinical Alzheimer cognitive composite.
4. DISCUSSION
In this study, we remotely deployed a battery of computerized tasks to the Online 46 cohort, a population‐based cohort of individuals born in England, Scotland, and Wales within the same week of March 1946. We identified the tasks that were associated with biomarkers of AD and neurodegeneration and verified that these correlated with standard supervised cognitive assessments.
The key finding of this study is that 4 years after receiving an amyloid PET scan, Aβ‐positive participants were significantly slower than Aβ‐negative participants on the Objects delayed recognition task. Lower accuracy on the same task was also associated with amyloid positivity, although with a weaker effect. These findings were further supported when examining the relationship between cognitive performance and amyloid deposition measured as a continuous variable (i.e., SUVR). Notably, we found that impairments in the Aβ‐positive group affected delayed rather than immediate memory recognition, likely reflecting a process of accelerated forgetting. Previous studies measured this after 30 min and 7 days, showing effects in individuals with preclinical AD only after 7 days. 23 , 51 However, we detected this deficit at a single timepoint, which is advantageous since testing over multiple days is more challenging and often leads to lower compliance.
Previous studies have found slower RT and RT inter‐individual variability in individuals with MCI and AD, as well as slower RT in attentional tasks for AD patients. 52 , 53 Additionally, lower performance on simple and choice reaction time tasks has been found to predict memory deficits in AD patients. 52 In line with these findings, we demonstrated that cognitive impairment in Aβ‐positive individuals is more evident when examining the RT required to perform a memory task rather than accuracy alone. Notably, slower RT was significantly associated with poorer accuracy only in the Aβ‐positive group, which accords with memory access being more effortful and therefore requiring more processing time. 23 This may reflect the early stages of an amnestic syndrome, where overt memory deficits are obscured by compensatory mechanisms, such as taking more time to complete a task. Therefore, RT as a measure of processing speed and as potential evidence of compensation for memory problems appears to be a good indicator of early AD‐related cognitive changes. Studies should further investigate the relationship between RT and accuracy on memory tasks in individuals with or at risk of developing dementia.
Higher rates of whole brain and hippocampal atrophy between ages 71 and 73 were associated with poorer performance on the Spatial span and Objects immediate recognition tasks at age 77. The annual atrophy rate in these regions accelerates in individuals with MCI compared to healthy controls and is even higher in progressive compared to stable MCI. 54 , 55 Atrophy rate has been shown to be a good indicator of disease progression, correlating with cognitive decline and predicting conversion from MCI to AD. 56 , 57 Our findings support that these tasks hold value in the remote assessment of disease risk and progression. Future studies should focus on the longitudinal administration of the tasks to map cognitive impairment trajectories and examine their association with the accumulation of AD pathology. Conversely, in line with previous findings, we did not find any significant association between cognitive performance and cross‐sectional brain volumes, indicating that longitudinal measurements of atrophy change may be a more sensitive measure of preclinical AD. 47
We also found that performance on the Word Definitions task at age 77, the main Cognitron measure of crystallized cognitive abilities, was associated with increased ventricular volume measured at age 73. Ventricular enlargement occurs naturally with age, but it can also predict conversion from MCI to AD and correlates with the degree of cognitive impairments in individuals with AD. 57 , 59 Late‐life cognitive ageing typically affects fluid intelligence while sparing crystallized cognitive abilities, which are usually considered a better indicator for detecting early cognitive signs of AD. 60 Therefore, the association between Word definitions and ventricular volume may reflect cognitive changes that deviate from the normal ageing trajectory. It is important to note that removing childhood cognitive abilities from the model resulted in a reduction in the estimated effect size and caused the relationship to lose statistical significance; this may reflect early life cognitive reserve having a protective effect on later‐life cognitive deterioration.
By highlighting associations between increased WMHV at age 73, slower RT on Objects delayed recognition, and reduced accuracy on the Spatial span tasks at age 77, we provide evidence that brain vascular damage plays a significant role in specific cognitive impairments. Accordingly, previous research suggests that WMHV may define a distinct subtype of AD, particularly linked to deficits in logical memory. 61 WMHV not only interacts with amyloid deposition to drive neurodegeneration, but also appears to be a more sensitive predictor of cortical thinning than amyloid deposition alone in cognitively normal individuals. 62 , 63 Furthermore, white matter lesions as measured through visual ratings of routinely performed CT scans, can predict dementia conversion in individuals with memory complaints. 64 Thus, identifying early cognitive deficits associated with WMHV may serve as a crucial marker for detecting and characterizing the preclinical stages of AD
In addition to the findings discussed above, the distinct group of 122 Insight 46 participants who declined the Online 46 study invitation and did not attempt the Cognitron tasks indicates a return rate of 70% within the Insight 46 neuroimaging sub‐study. This completion rate compares favorably with other studies utilizing remote digital assessments in adult populations, suggesting that remote computerized cognitive testing is feasible and generally well‐tolerated among older individuals at risk for dementia. 65 , 66 , 67 , 68 Notably, task completion rates were unaffected by Aβ‐positivity or ApoE status, supporting the potential of these cognitive assessments for evaluating cognitive abilities in preclinical AD.
There are, however, several limitations to this study. First, there is a difference in time between the data collection of the biomarkers and the standard cognitive tests and of the Cognitron tasks. Therefore, cognitive performance measured by Cognitron reflects the degree of amyloid pathology accumulated during the four years preceding its assessment. Second, recruitment and participation to the Insight 46 study have been shown to be biased toward people with better health, higher education level and socioeconomic status, which may limit the generalizability of our sample, although this is a common risk across any study cohort or epidemiological studies. 26 , 69 , 70 Additionally, there was variability in the number of participants who completed each task. Despite this, each task remained adequately powered, with a minimum of 239 subjects. Missing data might not be at random, as participants could not skip tasks, potentially excluding those with greater impairments. Nevertheless, the inability to proceed with the assessment can itself serve as an indicator of cognitive status. Moreover, it is challenging to control the conditions under which the tests were completed remotely, a common issue for all remote computerized assessments. We attempted to control for this variability by including the device used to complete the assessment in the regression models, providing participants with instructions on the testing environment to adopt, and selecting tasks whose paradigm and design have relatively lower sensitivity to device differences. Finally, the usability of the Cognitron tasks is restricted to individuals with access to Internet or a computerized device and a basic level of computer literacy. However, this limitation is becoming progressively less significant across generations. Furthermore, the tests can also be administered in‐person for such individuals, with reduced need of resources compared to on‐paper assessments and an automated scoring system. Finally, our study did not use any blood‐based biomarkers. However, amyloid PET is regarded as the gold standard for assessing AD pathology and shows a strong correlation with blood biomarkers. 71 While blood‐based measures of Aβ42/40 and p‐tau181 have previously been utilized, these have now been superseded by plasma p‐tau217, which was not available in an optimized form at the relevant time point for this study. 72 Future research within the NSHD cohort should aim to investigate the relationship between cognitive performance and these refined biomarkers.
Research investigating the use of computerized cognitive testing in the context of dementia diagnosis is growing significantly. 73 Among the tools which have been studied in relation to AD biomarkers, the C3PAD was able to discriminate Aβ‐positive from Aβ‐negative individuals who underwent Aβ‐PET scans, 74 while a digital version of the FNAME test has shown significant correlations with cerebrospinal fluid (CSF) levels of plasma pTau181 and Aβ42/40 ratio. 75 The CANTAB demonstrated to be able to identify individuals with AD and MCI, to detect cognitive changes of individuals with MCI longitudinally and to predict hippocampal volume and CSF biomarkers of T‐tau, pTau181, and tau/Aβ42 ratio in individuals with MCI. 76 , 77 , 78 Cogstate, instead, showed sensitivity to longitudinal cognitive decline in Aβ‐positive individuals, and a significant association with tau‐PET and hippocampal atrophy. 79 , 80 However, these studies were conducted in supervised settings and have yet to explore the remote applicability of the tests. Other studies have shown promise in testing older cognitively unimpaired individuals in unsupervised conditions and examined their cognitive abilities in relation to neurodegenerative biomarkers. 81 , 82 However, they have not investigated the administration of the tasks across multiple types of electronic devices or selected the most appropriate tasks from a broader set measuring different cognitive domains. In our study, we evaluated a comprehensive battery of tasks administered remotely in unsupervised conditions and run on a broad range of electronic devices. We identified a sub‐set that can specifically target cognitive impairments associated with amyloid status and biomarkers indicative of AD severity and progression, while correlating with standard face‐to‐face assessments. Being easy to deploy and score while requiring minimal amount of time to complete, the Cognitron tasks represent a cost‐efficient tool for large‐scale screening of individuals at risk of AD, and potentially for monitoring patients who require longitudinal follow‐ups, such as those with atypical presentation or who received a diagnosis at early stages of the disease. 83 This is particularly advantageous in clinical settings and for large cohorts, where the required resources for repeat assessments of patients are limited.
CONFLICTS OF INTEREST STATEMENT
A.H. is owner/director of H2 Cognitive Designs Ltd and Future Cognition Ltd, which produce online assessment technology and provide online survey data collection for third parties. P.H. is founder and director of H2 Cognitive Designs LTD, which develops and markets online cognitive tests. W.T. is an employee of H2 Cognitive Designs LTD. PM is lead for an NIHR‐funded trial with drug/placebo provided by Takeda Pharmaceuticals and sits on the Data Monitoring Committee for a trial being carried out by Johnson and Johnson. P.M. is vice chair of the Alzheimer's Society Research Strategy Council, and NIHR Specialty Lead for Dementia and Neurodegeneration, Research Delivery Network. He is also an independent member of a data monitoring committee. C.S. is a scientific BrainKey scientific advisor, part of MONAI advisory board. D.C. is chair of the Alzheimer's Association Neuroimaging professorial interest area and a member of the Scientific Programming Committee of the Alzheimer's Association. He also reports grants from the National Institute of Health. J.B. is an NFRFT 2024 Application Stage Expert Panel, director of SENBOX and Chair of Faculty Research Degrees Committee at University College London. J.M.S. is the Chief Medical Officer of Alzheimer's Research UK and clinical advisor at the UK Dementia Research Institute. He reports grants from the NIHR, LifeArc Foundation, and the British Hearth Foundation.
CONSENT STATEMENT
The study was approved by the National Research Ethics Service Committee London (REC reference 19/LO/1774) and all participants provided written informed consent.
Supporting information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
ACKNOWLEDGMENTS
Insight 46 is funded by grants from Alzheimer's Research UK (ARUK‐PG2014‐1946, ARUK‐ PG2017‐1946), Alzheimer's Association (SG‐666374‐UK BIRTH COHORT), the Medical Research Council Dementias Platform UK (CSUB19166), The Wolfson Foundation (PR/ylr/18575), The Medical Research Council (MRC) (MC_UU_10019/1, MC_UU_10019/3), and Brain Research Trust (UCC14191). Florbetapir amyloid tracer was provided in kind by AVID Radiopharmaceuticals (a wholly owned subsidiary of Eli Lilly) who had no part in the design of the study. The funders of the study had no role in study design, data collection, analysis, interpretation, report writing, or in the decision to submit the article for publication. J.M.S. acknowledges the support of University College London Hospitals Biomedical Research Centre. MR is funded by the Medical Research Council (MC_UU_00019/1 and 3). V.G. is supported by the MRC (MR/W00710X/1). A.W. and M.P. are funded by the MRC (MC_UU_00019/1). Z.C. is employed as research assistant at King's College London. JN is funded by the Alzheimer's Association (SG‐666374‐UK BIRTH COHORT. T.P. is supported by a National Institute for Health and Care Research (NIHR) lectureship. K.L. has nothing to disclose. M.D.G. is employed as research technician by Imperial College London. P.M. receives research funding from Lifearc, NIHR, MRC, Dementias Platform UK, Alzheimer's Research UK, the Football Association, FIFA and Alzheimer's Society. The study was also supported by the NIHR Imperial Biomedical Research Centre at Imperial College London. CS reports grants from the Alzheimer's Society (AS‐17‐JF‐011). JB is funded by Alzheimer's Research UK. DC is funded by the UK Dementia Research Institute, which receives its funding from DRI Ltd, Alzheimer's Research UK (ARUK‐PG2017‐1946), the Alzheimer's Association (SG‐666374‐UK BIRTH COHORT), and the University College London Hospitals (UCLH/UCL) NIHR Biomedical Research Centre.
Giovane MD, Giunchiglia V, Cai Z, et al. Remote cognitive tests predict neurodegenerative biomarkers in the Insight 46 cohort. Alzheimer's Dement. 2025;21:e14572. 10.1002/alz.14572
Adam Hampshire and Jonathan M. Schott contributed as co‐senior authors.
REFERENCES
- 1. De Strooper B, Karran E. The Cellular Phase of Alzheimer's Disease. Cell. 2016;164(4):603‐615. [DOI] [PubMed] [Google Scholar]
- 2. Jia J, Ning Y, Chen M, et al. Biomarker Changes during 20 Years Preceding Alzheimer's Disease. N Engl J Med. 2024;390(8):712‐722. [DOI] [PubMed] [Google Scholar]
- 3. Jack CR, Bennett DA, Blennow K, et al. NIA‐AA Research Framework: toward a biological definition of Alzheimer's disease. Alzheimer's & Dementia. 2018;14(4):535‐562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mintun MA, Lo AC, Duggan Evans C, et al. Donanemab in Early Alzheimer's Disease. N Engl J Med. 2021;384(18):1691‐1704. [DOI] [PubMed] [Google Scholar]
- 5. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in Early Alzheimer's Disease. N Engl J Med. 2023;388(1):9‐21. [DOI] [PubMed] [Google Scholar]
- 6. A Donanemab (LY3002813) Prevention Study in Participants With Alzheimer's Disease (TRAILBLAZER‐ALZ 3) [Internet]. [cited 2024 Apr 24]. Available from: https://trials.lilly.com/en‐US/trial/302480
- 7. Brookmeyer R, Abdalla N. Estimation of lifetime risks of Alzheimer's disease dementia using biomarkers for preclinical disease. Alzheimer's & Dementia. 2018;14(8):981‐988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Jack CR, Andrews JS, Beach TG, et al. Revised criteria for diagnosis and staging of Alzheimer's disease: alzheimer's Association Workgroup. Alzheimer's & Dementia. 2024;20:5143‐5169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Jack CR, Bennett DA, Blennow K, et al. NIA‐AA Research Framework: toward a biological definition of Alzheimer's disease. Alzheimer's & Dementia. 2018;14(4):535‐562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Freeborough PA, Fox NC. The boundary shift integral: an accurate and robust measure of cerebral volume changes from registered repeat MRI. IEEE Trans Med Imaging. 1997;16(5):623‐629. [DOI] [PubMed] [Google Scholar]
- 11. Leung KK, Bartlett JW, Barnes J, Manning EN, Ourselin S, Fox NC. Cerebral atrophy in mild cognitive impairment and Alzheimer disease. Neurology. 2013;80(7):648‐654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Brickman AM. Contemplating Alzheimer's Disease and the Contribution of White Matter Hyperintensities. Curr Neurol Neurosci Rep. 2013;13(12):415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Keuss SE, Coath W, Nicholas JM, et al. Associations of β‐Amyloid and Vascular Burden With Rates of Neurodegeneration in Cognitively Normal Members of the 1946 British Birth Cohort. Neurology. 2022;99(2):e129‐e141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Lu K, Nicholas JM, Collins JD, et al. Cognition at age 70. Neurology. 2019;93(23). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bradfield NI. Mild Cognitive Impairment: diagnosis and Subtypes. Clin EEG Neurosci. 2023;54(1):4‐11. [DOI] [PubMed] [Google Scholar]
- 16. Brooker H, Williams G, Hampshire A, et al. FLAME: a computerized neuropsychological composite for trials in early dementia. Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring. 2020;12(1):e12098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Veitch DP, Weiner MW, Aisen PS, et al. Understanding disease progression and improving Alzheimer's disease clinical trials: recent highlights from the Alzheimer's Disease Neuroimaging Initiative. Alzheimer's & Dementia. 2019;15(1):106‐152. [DOI] [PubMed] [Google Scholar]
- 18. Lane CA, Parker TD, Cash DM, et al. Study protocol: insight 46 – a neuroscience sub‐study of the MRC National Survey of Health and Development. BMC Neurol. 2017;17(1):75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Murray‐Smith H, Barker S, Barkhof F, et al. Updating the study protocol: insight 46 – a longitudinal neuroscience sub‐study of the MRC National Survey of Health and Development – phases 2 and 3. BMC Neurol. 2024;24(1):40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kern S, Zetterberg H, Kern J, et al. Prevalence of preclinical Alzheimer disease. Neurology. 2018;90(19). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Jansen WJ, Ossenkoppele R, Knol DL, et al. Prevalence of Cerebral Amyloid Pathology in Persons Without Dementia. JAMA. 2015;313(19):1924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Weintraub S, Wicklund AH, Salmon DP. The Neuropsychological Profile of Alzheimer Disease. Cold Spring Harb Perspect Med. 2012;2(4):a006171‐a006171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Lu K, Pavisic IM, James S, et al. Accelerated forgetting is sensitive to β‐amyloid pathology and cerebral atrophy in cognitively normal 72‐year‐olds. Alzheimer's & Dementia. 2020;16(S6). [Google Scholar]
- 24. Loreto F, Gunning S, Golemme M, et al. Evaluating cognitive profiles of patients undergoing clinical amyloid‐PET imaging. Brain Commun. 2021;3(2). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Lim YY, Maruff P, Pietrzak RH, et al. Effect of amyloid on memory and non‐memory decline from preclinical to clinical Alzheimer's disease. Brain. 2014;137(1):221‐231. [DOI] [PubMed] [Google Scholar]
- 26. James SN, Lane CA, Parker TD, et al. Using a birth cohort to study brain health and preclinical dementia: recruitment and participation rates in Insight 46. BMC Res Notes. 2018;11(1):885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Del Giovane M, Trender WR, Bălăeţ M, et al. Computerized cognitive assessment in patients with traumatic brain injury: an observational study of feasibility and sensitivity relative to established clinical scales. EClinicalMedicine. 2023;59:101980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Hampshire A, Chatfield DA, MPhil AM, et al. Multivariate profile and acute‐phase correlates of cognitive deficits in a COVID‐19 hospitalised cohort. EClinicalMedicine. 2022;47:101417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Hampshire A, Azor A, Atchison C, et al. Cognition and Memory after Covid‐19 in a Large Community Sample. N Engl J Med. 2024;390(9):806‐818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Bălăeţ M, Alhajraf F, Zerenner T, et al. Online cognitive monitoring technology for people with Parkinson's disease and REM sleep behavioral disorder. NPJ Digit Med. 2024;7(1):118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Shibata K, Attaallah B, Tai XY, et al. Remote digital cognitive assessment reveals cognitive deficits related to hippocampal atrophy in autoimmune limbic encephalitis: a cross‐sectional validation study. EClinicalMedicine. 2024;69:102437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Taquet M, Skorniewska Z, De Deyn T, et al. Cognitive and psychiatric symptom trajectories 2‐3 years after hospital admission for COVID‐19: a longitudinal, prospective cohort study in the UK. Lancet Psychiatry. 2024;11(9):696‐708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Cai Z, Giunchiglia V, Street R, et al. Online46: online cognitive assessments in elderly cohorts—the British 1946 birth cohort case study. medRxiv:2024091924313984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Folstein MF, Folstein SE, McHugh PR. “Mini‐mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189‐198. [DOI] [PubMed] [Google Scholar]
- 35. Wechsler D. The psychometric tradition: developing the wechsler adult intelligence scale. Contemp Educ Psychol. 1981;6(2):82‐85. [Google Scholar]
- 36. Wechsler D. Wechsler memory scale‐revised edition. Psychological Corp., Harcourt Brace Jovanovich; 1987. [Google Scholar]
- 37. Papp KV, Amariglio RE, Dekhtyar M, et al. Development of a Psychometrically Equivalent Short Form of the Face–Name Associative Memory Exam for use Along the Early Alzheimer's Disease Trajectory. Clin Neuropsychol. 2014;28(5):771‐785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Donohue MC, Sperling RA, Salmon DP, et al. The Preclinical Alzheimer Cognitive Composite. JAMA Neurol. 2014;71(8):961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Wechsler D. The Wechsler abbreviated scale of intelligence. The Psychological Corporation; 1999. [Google Scholar]
- 40. Coughlan Anthony K, HSE. The adult memory and information processing battery (AMIPB) : test manual. A.K. Coughlin, Psychology Dept, St James’ Hospital, Leeds; 1985.
- 41. Richards M, Sacker A. Lifetime Antecedents of Cognitive Reserve. J Clin Exp Neuropsychol. 2003;25(5):614‐624. [DOI] [PubMed] [Google Scholar]
- 42. McKenna P, Warrington EK. Testing for nominal dysphasia. J Neurol Neurosurg Psychiatry. 1980;43(9):781‐788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Lu K, Nicholas JM, James S, et al. Increased variability in reaction time is associated with amyloid beta pathology at age 70. Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring. 2020;12(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Nishida A, Cadar D, Xu MK, et al. Adolescent Self‐Organization and Adult Smoking and Drinking over Fifty Years of Follow‐Up: the British 1946 Birth Cohort. PLoS One. 2016;11(1):e0146731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Burgos N, Cardoso MJ, Thielemans K, et al. Multi‐contrast attenuation map synthesis for PET/MR scanners: assessment on FDG and Florbetapir PET tracers. Eur J Nucl Med Mol Imaging. 2015;42(9):1447‐1458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Malone IB, Leung KK, Clegg S, et al. Accurate automatic estimation of total intracranial volume: a nuisance variable with less nuisance. NeuroImage, Neuroimage. 2015;104:366‐372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Xie L, Wisse LEM, Das SR, et al. Longitudinal atrophy in early Braak regions in preclinical Alzheimer's disease. Hum Brain Mapp. 2020;41(16):4704‐4717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. McCarthy L, Rubinsztein J, Lowry E, et al. Cut‐off scores for mild and moderate dementia on the Addenbrooke's Cognitive Examination‐III and the Mini‐Addenbrooke's Cognitive Examination compared with the Mini‐Mental State Examination. BJPsych Bull. 2024;48(1):12‐18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Donohue MC, Sperling RA, Salmon DP, et al. The Preclinical Alzheimer Cognitive Composite. JAMA Neurol. 2014;71(8):961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Salis F, Costaggiu D, Mandas A. Mini‐Mental State Examination: optimal Cut‐Off Levels for Mild and Severe Cognitive Impairment. Geriatrics. 2023;8(1):12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Weston PSJ, Nicholas JM, Henley SMD, et al. Accelerated long‐term forgetting in presymptomatic autosomal dominant Alzheimer's disease: a cross‐sectional study. Lancet Neurol. 2018;17(2):123‐132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Christ BU, Combrinck MI, Thomas KGF. Both Reaction Time and Accuracy Measures of Intraindividual Variability Predict Cognitive Performance in Alzheimer's Disease. Front Hum Neurosci. 2018;12:124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Bailon O, Roussel M, Boucart M, Krystkowiak P, Godefroy O. Psychomotor Slowing in Mild Cognitive Impairment, Alzheimer's Disease and Lewy Body Dementia: mechanisms and Diagnostic Value. Dement Geriatr Cogn Disord. 2010;29(5):388‐396. [DOI] [PubMed] [Google Scholar]
- 54. Frankó E, Joly O. Evaluating Alzheimer's Disease Progression Using Rate of Regional Hippocampal Atrophy. PLoS One. 2013;8(8):e71354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Whitwell JL. Progression of Atrophy in Alzheimer's Disease and Related Disorders. Neurotox Res. 2010;18(3‐4):339‐346. [DOI] [PubMed] [Google Scholar]
- 56. Leung KK, Bartlett JW, Barnes J, Manning EN, Ourselin S, Fox NC. Cerebral atrophy in mild cognitive impairment and Alzheimer disease. Neurology. 2013;80(7):648‐654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Evans MC, Barnes J, Nielsen C, et al. Volume changes in Alzheimer's disease and mild cognitive impairment: cognitive associations. Eur Radiol. 2010;20(3):674‐682. [DOI] [PubMed] [Google Scholar]
- 58. Jack CR, Shiung MM, Gunter JL, et al. Comparison of different MRI brain atrophy rate measures with clinical disease progression in AD. Neurology. 2004;62(4):591‐600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Thompson PM, Hayashi KM, de Zubicaray GI, et al. Mapping hippocampal and ventricular change in Alzheimer disease. Neuroimage. 2004;22(4):1754‐1766. [DOI] [PubMed] [Google Scholar]
- 60. Harrington KD, Dang C, Lim YY, et al. The effect of preclinical Alzheimer's disease on age‐related changes in intelligence in cognitively normal older adults. Intelligence. 2018;70:22‐29. [Google Scholar]
- 61. Prosser L, Sudre CH, Oxtoby NP, Young AL, Malone IB, Manning EN. Biomarker pathway heterogeneity of amyloid‐positive individuals. Alzheimer's Dement. 2024:1‐13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Parker TD, Cash DM, Lane CA, et al. Amyloid β influences the relationship between cortical thickness and vascular load. Alzheimers Dement (Amst). 2020;12(1):e12022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Keuss SE, Coath W, Cash DM, et al. Rates of cortical thinning in Alzheimer's disease signature regions associate with vascular burden but not with β‐amyloid status in cognitively normal adults at age 70. J Neurol Neurosurg Psychiatry. 2024;95(8):748‐752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Sacuiu S, Eckerström M, Johansson L, et al. Increased Risk of Dementia in Subjective Cognitive Decline if CT Brain Changes are Present. J Alzheimers Dis. 2018;66(2):483‐495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Cheetham NJ, Penfold R, Giunchiglia V, et al. The effects of COVID‐19 on cognitive performance in a community‐based cohort: a COVID symptom study biobank prospective cohort study. EClinicalMedicine. 2023;62:102086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Taquet M, Skorniewska Z, De Deyn T, et al. Cognitive and psychiatric symptom trajectories 2‐3 years after hospital admission for COVID‐19: a longitudinal, prospective cohort study in the UK. Lancet Psychiatry. 2024;11(9):696‐708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Bălăeţ M, Alhajraf F, Zerenner T, et al. Online cognitive monitoring technology for people with Parkinson's disease and REM sleep behavioral disorder. NPJ Digit Med. 2024;7(1):118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Bălăeţ M, Alhajraf F, Bourke NJ, et al. Metacognitive accuracy differences in Parkinson's disease and REM sleep behavioral disorder relative to healthy controls. Front Neurol. 2024;15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Baquet CR, Commiskey P, Daniel Mullins C, Mishra SI. Recruitment and participation in clinical trials: socio‐demographic, rural/urban, and health care access predictors. Cancer Detect Prev. 2006;30(1):24‐33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Strandhagen E, Berg C, Lissner L, et al. Selection bias in a population survey with registry linkage: potential effect on socioeconomic gradient in cardiovascular risk. Eur J Epidemiol. 2010;25(3):163‐172. [DOI] [PubMed] [Google Scholar]
- 71. Warmenhoven N, Salvadó G, Janelidze S, et al. A comprehensive head‐to‐head comparison of key plasma phosphorylated tau 217 biomarker tests. Brain. 2024:awae346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Arranz J, Zhu N, Rubio‐Guerra S, et al. Diagnostic performance of plasma pTau217, pTau181, Aβ1‐42 and Aβ1‐40 in the LUMIPULSE automated platform for the detection of Alzheimer disease. Alzheimers Res Ther. 2024;16(1):139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Staffaroni AM, Tsoy E, Taylor J, Boxer AL, Possin KL. Digital Cognitive Assessments for Dementia: digital assessments may enhance the efficiency of evaluations in neurology and other clinics. Pract Neurol (Fort Wash Pa). 2020;2020:24‐45. [PMC free article] [PubMed] [Google Scholar]
- 74. Papp KV, Rentz DM, Maruff P, et al. The Computerized Cognitive Composite (C3) in A4, an Alzheimer's Disease Secondary Prevention Trial. J Prev Alzheimers Dis. 2020:1‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Alegret M, Sotolongo‐Grau O, de Antonio EE, et al. Automatized FACEmemory scoring is related to Alzheimer's disease phenotype and biomarkers in early‐onset mild cognitive impairment: the BIOFACE cohort. Alzheimers Res Ther. 2022;14(1):43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Chipi E, Salvadori N, Montanucci C, et al. Subtle cognitive deficits assessed by means of CANTAB in a cohort of subjects with CSF AD profile. Alzheimer's & Dementia. 2022;18(S7). [Google Scholar]
- 77. O'Connell H, Coen R, Kidd N, Warsi M, Chin A, Lawlor BA. Early detection of Alzheimer's disease (AD) using the CANTAB paired Associates Learning Test. Int J Geriatr Psychiatry. 2004;19(12):1207‐1208. [DOI] [PubMed] [Google Scholar]
- 78. Campos‐Magdaleno M, Leiva D, Pereiro AX, et al. Changes in visual memory in mild cognitive impairment: a longitudinal study with CANTAB. Psychol Med. 2021;51(14):2465‐2475. [DOI] [PubMed] [Google Scholar]
- 79. Lim YY, Pietrzak RH, Bourgeat P, et al. Relationships Between Performance on the Cogstate Brief Battery, Neurodegeneration, and A Accumulation in Cognitively Normal Older Adults and Adults with MCI. Arch Clin Neuropsychol. 2015;30(1):49‐58. [DOI] [PubMed] [Google Scholar]
- 80. Lim YY, Villemagne VL, Laws SM, et al. Performance on the Cogstate Brief Battery Is Related to Amyloid Levels and Hippocampal Volume in Very Mild Dementia. J Mol Neurosci. 2016;60(3):362‐370. [DOI] [PubMed] [Google Scholar]
- 81. Berron D, Olsson E, Andersson F, et al. Remote and unsupervised digital memory assessments can reliably detect cognitive impairment in Alzheimer's disease. Alzheimer's & Dementia. 2024;20:4775‐4479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Thompson LI, Kunicki ZJ, Emrani S, et al. Remote and in‐clinic digital cognitive screening tools outperform the MoCA to distinguish cerebral amyloid status among cognitively healthy older adults. Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring. 2023;15(4):e12500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Howard R, Schott JM. When dementia is misdiagnosed. Int J Geriatr Psychiatry. 2021;36(6):799‐801. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information
Supporting Information


