Abstract
Carriers of premutation alleles (55–200 CGG repeats) of the fragile-X mental retardation 1 (FMR1) gene are often regarded as being clinically uninvolved. However, it is now apparent that such individuals can present with one (or more) of three distinct clinical disorders: mild cognitive and/or behavioral deficits on the fragile-X spectrum; premature ovarian failure; and a newly described, neurodegenerative disorder of older adult carriers, fragile-X–associated tremor/ataxia syndrome (FXTAS). Awareness of these clinical presentations is important for proper diagnosis and therapeutic intervention, not only among families with known cases of fragile-X syndrome but also more broadly for adults with tremor, gait ataxia, and parkinsonism who are seen in movement-disorders clinics.
Introduction
In their analysis of pedigrees with fragile-X syndrome (MIM 309550) that exhibited transmission through ostensibly normal males, Pembrey et al. suggested that “the final genetic event that causes the mental retardation syndrome is preceded by a mutation that causes no harm other than predisposing to the final event, in other words, a ‘premutation’” (Pembrey et al. 1985, p. 713 [our emphasis]). The sequencing of the fragile-X mental retardation 1 (FMR1) gene in 1991 (Verkerk et al. 1991) greatly clarified the nature of the “final (genetic) event,” namely, an expansion of a trinucleotide (CGG)–repeat element in the 5′ UTR of the FMR1 gene to greater than ∼200 CGG repeats (full mutation) that is generally accompanied by methylation-coupled silencing of the gene. However, the concept of the premutation has remained imprecise, in part because there is no clear distinction between premutation and normal alleles and because premutation alleles do, indeed, cause “harm.” This latter issue, the forms of clinical involvement associated with premutation alleles, is the central focus of this review.
The Premutation: A Square Peg in a Round Hole
The concept of the premutation has remained vague on two counts since Pembrey’s original description. First, the various forms of premutation-associated clinical involvement were not generally recognized until well after the discovery of the gene itself and thus were not used to define the premutation. In fact, carrier status was often used to define, categorically, the carrier phenotype as normal (e.g., “normal transmitting male” [NTM]) despite the absence of detailed behavioral or cognitive testing. Second, following the discovery of the FMR1 gene, the premutation (allele) was defined by repeat-length instability leading to expansion to a full mutation (Fu et al. 1991; Rousseau et al. 1991). However, this definition has remained imprecise, resulting in definitions of the lower bound of the premutation with a range of 50–60 CGG repeats. If a premutation allele were defined as having a degree of repeat instability that can give rise to a full mutation in a single generation, then the lower end of the premutation range (i.e, the smallest CGG element known to satisfy this criterion) could not be >59 repeats (Nolin et al. 2003). However, significant repeat-length instability does occur for even smaller repeats in a manner that depends on both the repeat size per se and the number of AGG interruptions within the CGG tract (Zhong et al. 1995; Crawford et al. 2000; Nolin et al. 2003). Thus, as discussed by Nolin et al. (2003), for alleles in the range of ∼50–60 CGG repeats, the risk for expansion is indeterminate and can be assessed “only by observing transmissions in future generations.”
To address the ambiguity currently associated with the definition of the premutation, we recommend following the guidelines of the American College of Medical Genetics (ACMG) (Maddalena et al. 2001), with the lower bound of the premutation range operationally defined as a specific size (55 CGG repeats), irrespective of sequence composition (i.e., presence or absence of AGG interruptions). This definition requires neither sequencing nor observation of allele transmissions and takes into consideration all of the inherent uncertainty in the propensity for expansion discussed by Nolin et al. (2003). Further, such a definition provides a more consistent link to the molecular mechanisms that underlie phenotypic involvement among premutation carriers and that may have no direct association with instability/AGG interruptions per se. Defining the lower bound as 55 CGG repeats also seems prudent in view of the intrinsic inaccuracy of gel methods for repeat sizing and the continued need for genetic counseling, as will be seen below. It is also important to note that several of the large prevalence studies have used 55 repeats as the lower bound for the premutation, and the oft-quoted carrier frequencies, 1:259 females and 1:810 males (Rousseau et al. 1995; Dombrowski et al. 2002), are based on this size criterion.
Smaller CGG tracts (∼45–54 CGG repeats) are associated with some degree of size instability upon transmission (Zhong et al. 1996; Nolin et al. 2003), with the possibility of expanding to a full mutation within two generations. To account for both residual repeat instability and the possibility of repeat-length–associated clinical involvement, a gray-zone range of ∼45–54 CGG repeats that does not overlap with the premutation range should be retained, as recommended by the ACMG (Maddalena et al. 2001).
From the molecular-genetic perspective, the concept of the premutation/gray zones should probably be scrapped altogether in favor of one or more continuous scales of involvement (clinical, genetic, and biochemical); however, there is a strong legacy for the premutation that is unlikely to disappear in the short term, at least until the underlying mechanisms are better understood.
Clinical Involvement among Carriers of Premutation Alleles
The possibility of clinical involvement in premutation carriers was initially discounted, since carrier mothers of children with fragile-X syndrome are typically intellectually normal (Reiss et al. 1993; Bennetto et al. 2001). However, over the past decade, a number of studies have documented the fact that a subgroup of female carriers do have mild physical features of fragile-X syndrome, such as prominent ears or flexible finger joints (Riddle et al. 1998; Hagerman et al. 2002b). In addition, some carriers have emotional problems, including anxiety, obsessional thinking, schizotypy, and/or depression, all of which can be exacerbated by the stress of raising a child with fragile-X syndrome (Loesch et al. 1994; Sobesky 1996; Franke et al. 1998; Hagerman et al. 2002b). Johnston et al. (2001) recently observed that emotional problems, including depression and interpersonal sensitivity, were more likely to occur in carrier females with >100 CGG repeats than in those with <100 repeats. These emotional features appear to be a mild form of the anxiety and perseverative thinking that occur in those affected by fragile-X syndrome and may be due to the mild deficit of FMR1 protein (FMRP) found in the upper half of the premutation range (Tassone et al. 2000a, 2000c; Kenneson et al. 2001). Moreover, Loesch et al. (2003a, 2003b) have reported an association between the presence and extent of the physical and cognitive phenotypes and the level of FMRP, in both males and females, that begins in the premutation range and extends into the full mutation range.
Two additional studies have documented more severe clinical involvement, including mental retardation and autism-spectrum disorders, among children with premutation alleles. Tassone et al. (2000d) first reported lowered FMRP levels in some children with the premutation who presented with mental retardation and/or autism. The association between clinical involvement and protein level suggested that these forms of involvement are on the fragile-X syndrome–spectrum (i.e., severity of clinical involvement is related to relative protein deficit). These forms of involvement may be more common in male carriers; Aziz et al. (2003) noted the frequent occurrence of either learning disabilities or autism-spectrum disorder in 15 boys with the premutation.
Unlike the forms of clinical involvement described above, a disorder that is unique to the premutation range is premature ovarian failure (POF [MIM 311360]), the cessation of menses before age 40, which is seen in ∼20% of females who carry premutation alleles (Allingham-Hawkins et al. 1999; Marozzi et al. 2000). For those who have not yet gone through menopause, subclinical ovarian dysfunction leading to elevated FSH is seen in 25% of adult female carriers aged <40 years (Hundscheid et al. 2001). The finding of a clinical feature (POF) that is particular to female carriers of premutation alleles brought focus to the possibility of a specific form of molecular dysfunction in the premutation range.
Fragile-X–Associated Tremor/Ataxia Syndrome
A second form of clinical involvement, recently identified among older male carriers of premutation alleles (Hagerman et al. 2001), consists of progressive intention tremor, gait ataxia, parkinsonism, and autonomic dysfunction and has been designated “fragile-X–associated tremor/ataxia syndrome” (FXTAS) (Berry-Kravis et al. 2003; Jacquemont et al. 2003; Leehey et al. 2003). Associated features include peripheral neuropathy with decreased sensation and vibration sense in the distal lower extremities, as well as cognitive deficits involving loss of memory and executive function. The most consistent neuroradiological feature associated with FXTAS involves symmetric hyperintensities on T2-weighted MR images of the middle cerebellar peduncles (Brunberg et al. 2002). More recently, Greco et al. (2002) identified eosinophilic, intranuclear inclusions in neurons and astrocytes throughout the brain in postmortem samples from four adult male premutation carriers with FXTAS (see the “FXTAS Represents a New Class of Inclusion Disorder” section). Diagnostic criteria for FXTAS have been presented by Jacquemont et al. (2003) and are described in the appendix; the presence of the pathognomonic, eosinophilic, intranuclear inclusions has been added as a (postmortem) criterion for definite FXTAS.
Although most female premutation carriers do not appear to develop FXTAS (Berry-Kravis et al. 2003; Jacquemont et al. 2004b), a small number do suffer from this disorder, with intranuclear inclusions present in the single (female) postmortem case examined to date (Hagerman et al. 2004). One possible explanation for this difference is the partial protection afforded by random X inactivation of the premutation allele; however, there may be additional sex-specific effects that reduce penetrance among females. Larger-scale screens are required to more precisely determine both the frequency and characteristics of penetrance among females.
A critical issue with regard to FXTAS is the fact that it took so long to identify the disorder, even though fragile-X syndrome has been recognized for >25 years, and the FMR1 gene itself for >12 years. One explanation for this is that fragile-X syndrome is principally identified as a childhood (neurodevelopmental) disorder without significant progression in adulthood. Moreover, male carriers (i.e., NTMs) of premutation alleles were thought not to experience any clinical abnormality. Thus, the movement disorders experienced by the older male carriers were not associated (by adult neurologists) with a childhood disorder (fragile-X syndrome) that lacks a neurodegenerative component and affected a different group of individuals. The mothers of children with fragile-X syndrome provided the key to this association.
While their children were being seen in clinics for problems related to fragile-X syndrome, mothers often expressed concerns about their own (premutation carrier) fathers, who were experiencing problems with hand tremor (loss of daily living skills involving their hands) and unsteady gait (frequently associated with serious falls). When we began to evaluate these male carriers (grandfathers of the probands with fragile-X syndrome), it became evident that they all had a common neurological picture, consisting principally of intention tremor and gait ataxia. We have now seen 64 male carriers (aged ⩾50 years) with FXTAS, and, although at least one-third of all male carriers will develop the symptoms of FXTAS (Jacquemont et al. 2004b), there is significant variability in the progression of neurological dysfunction; some cases remain stable for years or decades, whereas others progress rapidly and develop dementia within 5–8 years of receiving their diagnosis (Jacquemont et al. 2004a).
Impact of FXTAS on Genetic Counseling
When evaluating families with fragile-X syndrome, questions should address the presence of neurological problems in all relatives who are at risk for the FMR1 premutation. Because of the frequency of learning or emotional problems among those with the premutation, we recommend FMR1 DNA testing in siblings of a child with fragile-X syndrome and in other family members at risk of carrying the mutation (Gane and Cronister 2002). In those identified with a premutation, more detailed developmental testing is recommended (Hagerman 2002b; Jacquemont et al. 2004a). Many of the symptoms—including anxiety, attention deficit hyperactivity disorder, or obsessive compulsive disorder—experienced by premutation carriers, particularly males, respond well to therapy and psychopharmacological interventions (Hagerman 2002a). Families must be counseled about the risk of developing neurological symptoms in later adulthood (⩾30% of males >50 age years) (Jacquemont et al. 2004b). For many, neurological symptoms will not arise until their 70s or 80s. The onset of symptoms may be related to allelic variations in other genes that interact with FMR1 mRNA. The prospect of developing FXTAS in later life should not preclude the testing of siblings for the premutation, since many children with premutation alleles experience emotional or behavioral problems or learning difficulties in childhood and require treatment for those forms of clinical involvement. For older carriers with neurological symptoms, magnetic resonance imaging and detailed neurological testing are warranted to detect tremor, ataxia, neuropathy, and cognitive and/or emotional problems. Treatment can often be helpful for many of these symptoms, as discussed by Jacquemont et al. (2004a).
Molecular Correlates of Clinical Involvement in the Premutation Range
The discovery that FMR1 mRNA levels are elevated by as much as fivefold in the peripheral blood leukocytes of premutation carriers (Tassone et al. 2000a, 2000c; Kenneson et al. 2001; Tassone and Hagerman 2003) provided direct biochemical evidence of altered FMR1 gene expression in the premutation range, thus establishing a molecular correlate for the observations of clinical involvement among premutation carriers. Despite the increased mRNA levels, FMRP levels gradually decrease with increasing CGG-repeat length within the premutation range (Tassone et al. 2000a, 2000c; Kenneson et al. 2001). Reduced FMRP production in the premutation range is now known to be a consequence of reduced translational efficiency of the FMR1 mRNA in cells that harbor premutation alleles (Primerano et al. 2002), a finding that extends the previous observation of reduced translation of FMR1 mRNA from full mutation alleles (Feng et al. 1995).
A more recently described effect of the premutation CGG expansion is its influence on the position of the start of transcription, both in neural and nonneural cells (Beilina et al. 2004), with increasing CGG-repeat number associated with an upstream shift of the start site. Thus, the CGG repeat appears to be modulating not only the level of FMR1 gene activity (i.e., FMR1 mRNA level) but also start-site selection. This last observation demonstrates that mRNA derived from premutation alleles differs in at least two respects from the mRNA produced by normal alleles (abnormal CGG repeat and longer non-CGG 5′ UTR extension).
Quantification of FMRP levels in the premutation range has taken on added significance, in view of the neurodevelopmental, reproductive, and neurological forms of clinical involvement that are associated with premutation alleles of <100 CGG repeats. One significant difference between the study of Kenneson et al. (2001) and those of Tassone et al. (2000a, 2000c) was in the relationship between FMRP level and CGG-repeat number. Kenneson et al. (2001), utilizing western blot analysis to quantify FMRP levels in cultured lymphoblastoid cells, observed a progressive reduction in FMRP level that commences near the lower bound of the premutation range. By contrast, the immunocytochemical approach utilized by Tassone et al. (2000a), which determines the percentage of cells that are positive-staining for FMRP by use of an anti-FMRP antibody (Willemsen et al. 1995, 1997), observed significant reductions in the percent of FMRP-positive lymphocytes only for larger premutation alleles (>100 CGG repeats). Although the western blot approach lacks the sensitivity of the immunocytochemical method to detect reduced FMRP levels in small samples of peripheral blood, it is the more quantitative of the two measures, given sufficient numbers of cells. Thus, the observations of Kenneson et al. (2001) suggest that dysregulation of protein synthesis may occur for FMR1 alleles that are within the gray-zone range.
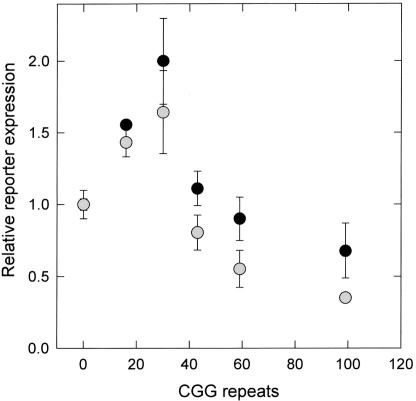
Additional evidence for a deficit in translation for only slightly expanded CGG repeats has been provided by the recent study by Chen et al. (2003). Using an expression system in which the FMR1 5′ UTR and a luciferase reporter are placed downstream of a strong (cytomegalovirus [CMV]) promoter, we examined the efficiency of translation of the reporter gene as a function of the number of CGG repeats (0–99) in the 5′ UTR. For both neural (SK) and nonneural (293) cells, translational efficiency was reduced, relative to 30 CGG repeats, even for mRNAs having repeat elements near the gray-zone (42 repeats) and low-premutation (60 repeats) ranges (fig. 1). Although this study used a strong (CMV) promoter and thus only indirectly addressed the issue of the translational efficiency of the native gene, the observed reduction in translational efficiency for even small repeat expansions underscores the need for better quantitative methods for determining FMRP levels in peripheral blood leukocytes within the premutation range.
Figure 1.
Relative in vivo translational efficiencies of FMR1 5′ UTRs (expressed per mRNA molecule), plotted as a function of the number of CGG-trinucleotide repeats. The FMR1 5′ UTRs were placed upstream of a reporter (firefly luciferase) coding sequence, and the resulting CMV-promoter constructs were transfected into either SK-N-MC cells (neuroepithelial origin) (black circles) or 293 cells (fetal kidney epithelium) (gray circles). The graph is normalized to 0 CGG repeats. Figure adapted from Chen et al. (2003).
An RNA Gain-of-Function Model for FXTAS
One of the striking features of FXTAS is that it appears to be confined to the premutation range; we have not observed the characteristic features of intention tremor and gait ataxia among the older adult patients with a full mutation whom we have seen clinically, nor have there been published reports of an increased incidence of combined tremor and ataxia among adult males with full-mutation alleles. The apparent lack of cases among males with full-mutation alleles who produce little or no FMRP argues against a protein-deficiency model for FXTAS; that is, the mild reductions in FMRP observed for some patients with FXTAS who harbor premutation alleles are unlikely to be causative of the tremor/ataxia syndrome. Thus, FXTAS is mechanistically distinct from fragile-X syndrome and affects a different group of individuals.
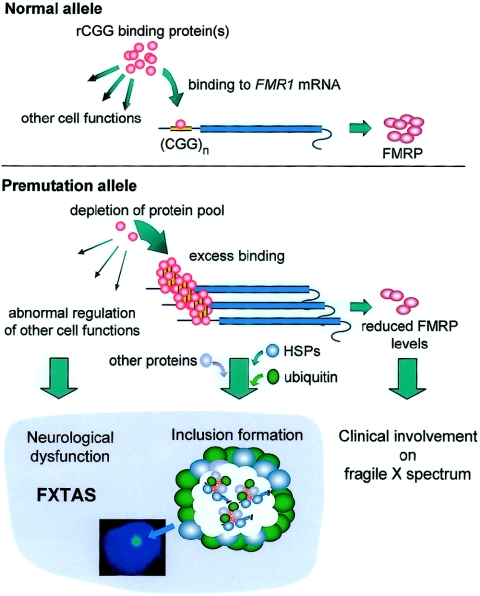
The presence of elevated levels of abnormal (expanded CGG repeat) FMR1 mRNA, coupled with the absence of FXTAS cases in the full-mutation range, led us to propose an RNA “toxic gain-of-function” model for FXTAS (Hagerman et al. 2001; Greco et al. 2002; Jacquemont et al. 2003) in which the FMR1 mRNA itself is causative of the neurological disorder (fig. 2).
Figure 2.
Schematic representation of our working hypothesis for an RNA toxic gain-of-function model for FXTAS. In this model, (hypothesized) specific protein interactions with the 5′ UTR of the FMR1 mRNA are altered as a consequence of expansion of the CGG repeat; this model is analogous to the RNA gain-of-function model proposed for myotonic dystrophy (see the “An RNA Gain-of-Function Model for FXTAS” section). In the premutation range, the expanded CGG repeat would lead to excess binding of one or more proteins (pink spheres), owing to (i) increased mRNA copy number, (ii) increased number of CGG repeats (increased CGG molarity), and/or (iii) altered secondary/tertiary RNA structure. This excess binding depletes the proteins from the cellular pool, resulting in the loss of their normal functions in other regulatory processes. The sequestration process would also trigger the accumulation or abnormal processing of proteins by the proteasomal degradation pathway, leading to inclusion formation with associated ubiquitinated proteins, proteasomal subunits, and stress-response (HSP) proteins. In the full-mutation range, altered protein-RNA interactions do not occur in the absence of FMR1 mRNA but could occur in the case of a full mutation allele that remains transcriptionally active. Image inset, Inclusion within an isolated neural cell nucleus (frontal cortex) from a patient with FXTAS; the inclusion is stained with fluorescent antiubiquitin antibody, and the nucleus is counterstained with DAPI.
Our hypothesis was based on the well-characterized RNA gain-of-function model proposed for myotonic dystrophy (MIM 160900 and MIM 602688), in which either expansion of a CUG repeat in the 3′ UTR of the DMPK (MIM 605377) mRNA (DM1) or expansion of the CCUG repeat in the first intron of the ZNF9 (MIM 116955) gene (DM2) leads to sequestration of one or more C(C)UG-binding proteins (see reviews by Finsterer [2002], Mankodi and Thornton [2002], and Ranum and Day [2002]). This gain-of-function effect has already been demonstrated for DM1 by placing an expanded CTG repeat within the noncoding region of a heterologous gene in a transgenic mouse (Mankodi and Thornton 2002) or within a transgene containing a human DMPK gene with an expanded CTG repeat (Seznec et al. 2001); both forms of transgenic mice developed myotonia. There is a further parallel between FXTAS and myotonic dystrophy, namely, the presence of intranuclear inclusions in FXTAS (Greco et al. 2002) and nuclear foci in both DM1 and DM2 (Taneja et al. 1995; Davis et al. 1997; Fardaei et al. 2002; Mankodi and Thornton 2002). Toxic RNA gain-of-function mechanisms have also been proposed for several of the spinocerebellar ataxias (SCAs)—types 8, 10, and 12—and Huntington disease–like type 2 (HDL2) (reviewed by Ranum and Day [2002]). The possibility of an RNA-based pathogenesis for POF among female carriers of premutation FMR1 alleles has also been considered (Conway et al. 1998).
Taken together, the myotonic dystrophies, DM1 and DM2, along with SCAs 8, 10, and 12 and HDL2, provide ample precedent for an RNA gain-of-function mechanism for FXTAS. Furthermore, the absence of cases of FXTAS among older adult males with full mutation has largely ruled out FMRP deficiency as the basis for FXTAS. However, the RNA-based model does make the prediction that a small subgroup of adults with full-mutation alleles, those whose FMR1 gene continues to produce elevated FMR1 mRNA levels (Tassone et al. 2000b, 2001), remains at risk for developing tremor and/or ataxia.
To test the RNA gain-of-function hypothesis for FXTAS, Willemsen et al. (2003) utilized a “knock-in” mouse in which the endogenous CGG element had been replaced with an ∼100 CGG-repeat element within the Fmr1 gene. Although the mice did not demonstrate neurological abnormalities (up to 72 wk), they did demonstrate the presence of ubiquitin-positive intranuclear inclusions in broad distribution throughout all of the brains examined. The presence of inclusions in the expanded-CGG–repeat mouse that has normal levels of FMRP provides further evidence against a protein-deficiency model for FXTAS. More recently, Jin et al. (2003) made similar findings in a Drosophila model in which an expanded CGG element (90 CGG repeats) was placed in a heterologous reporter gene; selective overexpression of the gene containing the expanded CGG repeat (but not a smaller repeat) in the Drosophila eye led to both inclusion formation and neurodegenerative changes in the eye. Thus, the CGG repeat itself, as RNA, may well be sufficient to induce the neuropathic changes that have been proposed to be causative of FXTAS. Although the dfxr gene does not itself possess a CGG-repeat element (Schenck et al. 2002), the putative RNA-based disease mechanism is likely to have little, if anything, to do with the function of the FMR1 (or dfxr) gene product.
It should be noted that the inclusions in Drosophila are quite different in form (electron dense) and distribution (substantial cytoplasmic distribution) than those found in either patients with FXTAS or the knock-in mouse (exclusively nuclear), perhaps reflecting differences in the sets of proteins that interact with (and localize) RNA species possessing the CGG repeat. This caveat notwithstanding, the Drosophila model holds promise for identifying potential homologues for proteins that may be interacting with the CGG-repeat element within the 5′ UTR region of the human FMR1 mRNA.
FXTAS Represents a New Class of Inclusion Disorder
The neuropathology associated with FXTAS has been presented and reviewed elsewhere (Greco et al. 2002; Hagerman et al. 2003b); its principal features include general cerebral and cerebellar atrophy, spongiosis of white matter and the middle cerebellar peduncles, Purkinje-cell drop out, and neuronal and astrocytic intranuclear inclusions. This last feature, the presence of intranuclear inclusions in broad distribution throughout the cerebrum and brain stem, is the most consistent finding associated with FXTAS, found in all nine brains of patients with FXTAS examined to date (Greco et al. 2002; Hagerman et al. 2003a, 2003b).
The inclusions are positive for ubiquitin by immunocytochemical staining; however, the presence of ubiquitin is quite widespread among various inclusion disorders and probably reflects a general degradative response to the inclusions themselves or the proteins involved in the underlying pathology. What is more important is that the intranuclear inclusions associated with FXTAS are immunocytochemically negative for staining with either anti-tau– or anti-α–synuclein antibodies (Greco et al. 2002; Hagerman et al. 2003b), further distinguishing the current inclusion disorder from the cytoplasmic inclusions found in the tauopathies (e.g., Pick disease) or synucleinopathies (e.g., Lewy body dementias and Parkinson disease [PD]). The inclusions associated with FXTAS are also polyglutamine negative, which distinguishes them from the morphologically similar intranuclear inclusions found in the CAG-repeat disorders (Cummings and Zoghbi 2000) or the (rare) neuronal intranuclear inclusion disease (Michaud and Gilbert 1981; Kimber et al. 1998; Zannolli et al. 2002). Last, the inclusions are distinct from those found in the phenotypically similar condition, multiple-system atrophy (MSA) (Gilman et al. 1999), both in their location (exclusively nuclear, neuronal, and astrocytic vs. cytoplasmic and oligodendrocytic) and in the absence of α-synuclein in the FXTAS-associated inclusions (present in MSA). The characteristic features of the intranuclear inclusions thus appear to establish FXTAS as a new class of inclusion disorder, and these inclusions appear to be pathognomonic for FXTAS (see the appendix).
The distinct features of the intranuclear inclusions in FXTAS undoubtedly reflect a novel mechanism for their genesis, as part of the underlying molecular pathology leading to FXTAS. If, as now seems likely, the molecular basis of FXTAS is a toxic gain of function of the abnormal (premutation) FMR1 mRNA, then it is possible that the mRNA is playing a direct role as a nucleation center for as-yet-unidentified proteins, again by analogy with DM1/2. Identification of FMR1 mRNA within the inclusions would be helpful in this regard, and such studies are currently under way. But there is another intriguing aspect to the inclusions that also may address pathogenic mechanism; namely, why the inclusions in humans are present in neurons and astrocytes but (apparently) not in oligodendroglia and why the inclusions are present in the nucleus rather than the cytoplasm. In this regard, differences in the location of the inclusions between human and mouse (exclusively nuclear) and the fly (substantial cytoplasmic component) may reflect a species difference in the proteins involved with handling the mRNA itself. Moreover, differences between the human (both clinical and neurodegenerative phenotypes) and the mouse (no evident neurological or neurodegenerative features discovered thus far) may be related to the absence of astrocytic inclusions in the latter, although neurological dysfunction may appear in the mice upon further aging or may depend on strain-dependent genetic modifiers. Finally, as a model system capable of generating both inclusions and neurodegeneration, the fly model may prove to be an important tool for understanding the mechanistic details underlying inclusion formation.
Contribution of FXTAS to Neurological Dysfunction among Older Adults
In the absence of large-scale epidemiological studies of the prevalence of FXTAS in the general population, it is difficult to gauge the contribution of the premutation allele to the movement disorders of aging. Recent estimates of the penetrance of neurological dysfunction among male carriers of premutation alleles suggest that at least one-third of the males aged >50 years, ascertained through analysis of families of children with fragile-X syndrome, will develop the neurological features of FXTAS (Smits et al. 2002; Rogers et al. 2003; Jacquemont et al. 2004b), and that the percentage of affected males appears to increase with age (Jacquemont et al. 2004b). On the basis of a carrier frequency of ∼1/813 (95% CI 1/527 to 1/1,781) for males in the general population (Dombrowski et al. 2002), the expectation for the prevalence of probable FXTAS (combined intention tremor and ataxia), absent biasing influences, would be ∼1/3,000 for males aged >50 years (∼1/10,000 males of all ages) in the general population, subject to a nearly threefold uncertainty, on the basis of the CIs of the prevalence figure.
The projected prevalence estimate for FXTAS is substantially less than the reported prevalence of 1%–5% for essential tremor (ET) in older adults (Louis et al. 1995; Brin and Koller 1998; Dogu et al. 2003), although it does approach the combined prevalence of ∼1/2,000 quoted for all forms of late-onset ataxia (National Ataxia Foundation). This latter figure is largely reflective of sporadic ataxias, with hereditary (dominant and recessive) ataxias accounting for only ∼10% of the total prevalence (Polo et al. 1991; Filla et al. 1992; Leone et al. 1995; Pujana et al. 1999; Mori et al. 2001; van de Warrenburg et al. 2002).
In separate assessments of the prevalence of premutation alleles within populations with movement disorders, four clinical cohorts were screened for carriers of premutation alleles among cases of sporadic ataxia or ET; none of these studies was tied to ascertainment through families known to have fragile-X syndrome. In a screen of 59 males presenting with neurological disorders involving ataxia and originally referred for SCA testing, Macpherson et al. (2003) found 3 (5%) carriers of premutation or gray-zone alleles (87, 66, and 51 CGG repeats). This finding is highly significant (P<.0001) for association between carrier status and ataxia. It is interesting that one of the three patients (with 66 repeats) had a childhood onset of ataxia, which has not been observed in any of the studies of FXTAS among families with fragile-X syndrome. This individual may have a different origin of his neurological dysfunction, with the premutation allele only an incidental finding; alternatively, the earlier onset may reflect a synthetic/epistatic effect of the premutation allele and one or more additional genes. In a second screen of males with sporadic ataxia who were originally referred for SCA testing, Di Maria et al. (2003) found 2 (7%) carriers with premutation alleles (84 and 86 CGG repeats) among 28 males aged >50 years.
In two screens of older adult males presenting with ET, no premutation carriers were found among 40 males (Garcia Arocena et al. 2003) or among 114 males (Di Maria et al. 2003). This result is not unexpected, in view of the high prevalence of ET in the general population, wherein only 1%–2% of ET cases among older males would be expected to be associated with premutation alleles. It is interesting that, in a separate clinical observation, two subjects who initially received the diagnosis of ET were later found to be carriers of premutation alleles (Leehey et al. 2003). Although no conclusions regarding prevalence can be drawn from this last observation, it does underscore the fact that cases of premutation-associated tremor can be present within clinical populations who have received other neurological diagnoses.
Taken together, the pilot screening studies for carriers of premutation (FMR1) alleles among cases of ataxia, a major feature of FXTAS, suggest that ⩾5% of cases of sporadic ataxia among older adult males may be attributable to the effects of premutation alleles. On the basis of the projected prevalence of males with FXTAS in the general population, one might have expected that a larger fraction of those with sporadic ataxia would have had premutation alleles. There are at least three possible explanations for this difference, all requiring further examination through systematic, large-scale epidemiological studies of both fragile-X–carrier populations and of populations with movement disorders.
Ascertainment Bias
The vast majority of cases of FXTAS identified thus far have been ascertained through families with a known proband (with fragile-X syndrome), with the affected carriers typically (although not exclusively) being the grandfathers of the proband. The distribution of premutation alleles among transmitting males is likely to be biased toward larger CGG-repeat lengths, since larger alleles, when transmitted through daughters, are more likely to give rise to full-mutation alleles and an affected proband. This form of allele-size bias could potentially lead to an overestimation of the population prevalence of FXTAS, if the penetrance of neurological dysfunction increases with increasing CGG-repeat size, since our use of the general population prevalence of 1/813 (Dombrowski et al. 2002) assumes that there is neither allele-size bias nor allele-size dependence of the penetrance of the neurological dysfunction. However, we have not observed a strong CGG-repeat–size dependence of the penetrance, severity, or age at onset of FXTAS, with the preponderance of alleles falling within the range of 55–100 CGG repeats (Jacquemont et al. 2004a). A second (as-yet-hypothetical) form of ascertainment bias (in the opposite direction) would arise as a consequence of reduced fitness of males with large premutation alleles, perhaps owing to a reduced propensity to marry and have children if they are significantly learning disabled. The degree to which this effect is operating is difficult to assess, although we suspect that it has only a minor influence on prevalence estimates. We do see alleles with as many as 160 CGG repeats within our population of transmitting males with FXTAS (Jacquemont et al. 2004a); a relatively small fraction of premutation alleles in males is comparable to or exceeds this size (only ∼8% of male premutation alleles with >140 CGG repeats [Zhong et al. 1995]).
Incorrect Initial Diagnosis and Classification
For those individuals whose level of neurological dysfunction was severe enough to warrant clinical evaluation, the majority received neurological diagnoses that would not have been counted among cases of sporadic ataxia. In the survey by Rogers et al. (2003), four male carriers had received a diagnosis of PD (a fifth carrier possibly had PD), with additional diagnoses (one each) of Alzheimer disease, transient ischemic attack, and motor neuron disease; only one individual received a diagnosis of cerebellar ataxia. In the study by Berry-Kravis et al. (2003), four of seven male carriers had previously received neurological diagnoses, two with atypical PD and two with ET. Although the range of previous diagnoses has not been addressed in a systematic fashion, the common feature of parkinsonism among patients with FXTAS (Hagerman et al. 2001; Berry-Kravis et al. 2003; Jacquemont et al. 2003) has led to a frequent diagnosis of PD or an atypical variant to account for cerebellar findings. In light of these findings, we suspect that most cases of FXTAS have been classified as neurological disorders that would not be counted among cases of ataxia.
Absence of a Clinical Presentation
Many of the cases of neurological dysfunction (probable FXTAS) identified through probands with fragile-X syndrome (Berry-Kravis et al. 2003; Jacquemont et al. 2003, 2004b; Rogers et al. 2003) were not of sufficient clinical severity to warrant a detailed neurological assessment and therefore would not have been counted among cases of sporadic ataxia (or ET). In fact, the gait ataxia and/or action tremor experienced by some carriers had been considered “normal aging” or had been believed to be the sequelae of strokes or microvascular disease. In this regard, a powerful advantage of genetic association (e.g., the FMR1-FXTAS association) is the ability to identify those features of aging that are not normal but that are part of a specific process of neurological dysfunction. However, in the absence of such a genetic association, it is very difficult to properly identify cases with only mild neurological involvement; many additional cases of probable FXTAS have fallen into this category.
A Maturing Perspective
The identification of at least three forms of clinical involvement among carriers of premutation alleles (FXTAS, POF, and learning disabilities/autism-spectrum disorder), coupled with the identification of specific biochemical abnormalities within the premutation range, has fundamentally changed the way we view this group of individuals. This maturing perspective has moved us from a categorical to a more nuanced view of the way the FMR1 gene contributes to clinical involvement, much as our perspective on the range of clinical involvement with other disorders, such as cystic fibrosis or Rett syndrome, has broadened following the discoveries of the CFTR and MECP2 genes. With the FMR1 gene, our perspective has matured in another—more literal—way. It is now clear that the same gene can give rise to both the most common heritable form of cognitive impairment (a disorder of childhood) and an important neurological disorder (a disease of aging), and it does so by what appear to be completely distinct disease mechanisms.
With respect to FXTAS, what is needed now is a concerted effort to better define the disorder through large-scale epidemiological studies that address the contributions of premutation alleles to various categories of neurological disorders that involve parkinsonism with cerebellar and autonomic dysfunction (e.g., atypical PD and multiple-system disorders). At the same time, a better understanding of the molecular underpinnings of FXTAS should shed light on common mechanisms in other neurodegenerative diseases, which will lead to treatments, for those disorders, that are both more effective and more specific to the molecular dysfunction.
Acknowledgments
The authors thank Drs. S. Nolin and W. T. Brown for helpful discussions regarding the premutation range. This study was supported by National Institute of Neurological Diseases and Stroke grant NS43532 (P.J.H.), National Institute for Child Health and Development grants HD40661 (P.J.H.) and HD36071 (R.J.H.), and the M.I.N.D. Institute (general support).
Appendix: Diagnostic Criteria for FXTAS (Mandatory Criterion: FMR1 Allele Size of 55–200 CGG Repeats)
-
1.
Definite:
-
A.
one clinical major criterion (clinical major criteria: intention tremor and gait ataxia; clinical minor criterion: parkinsonism) and
-
B.
one radiological major criterion (radiological major criterion: symmetric white-matter lesions involving the middle cerebellar peduncles; radiological minor criteria: white-matter lesions in cerebral white matter, moderate-to-severe generalized atrophy) or
-
C.
presence of inclusions (the presence of intranuclear—neuronal and astrocytic—inclusions has been added as an additional criterion for FXTAS, on the basis of examination of postmortem brain tissue).
-
A.
-
2.
Probable:
-
A.
two clinical major criteria or
-
B.
one radiological major criterion and
-
C.
one clinical minor criterion.
-
A.
-
3.
Possible:
-
A.
one clinical major criterion and
-
B.
one radiological minor criterion.
-
A.
Electronic-Database Information
The URLs for data presented herein are as follows:
- National Ataxia Foundation, http://www.ataxia.org/
- Online Mendelian Inheritance in Man (OMIM), http://www.ncbi.nlm.nih.gov/Omim/ (for fragile-X syndrome, POF, myotonic dystrophy, DMPK, and ZNF9)
References
- Allingham-Hawkins DJ, Babul-Hirji R, Chitayat D, Holden JJ, Yang KT, Lee C, Hudson R, et al (1999) Fragile X premutation is a significant risk factor for premature ovarian failure: the International Collaborative POF in Fragile X Study: preliminary data. Am J Med Genet 83:322–325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aziz M, Stathopulu E, Callias M, Taylor C, Turk J, Oostra B, Willemsen R, Patton M (2003) Clinical features of boys with fragile X premutations and intermediate alleles. Am J Med Genet 121B:119–127 12898586 [DOI] [PubMed] [Google Scholar]
- Beilina A, Tassone F, Schwartz PH, Sahota P, Hagerman PJ (2004) Redistribution of transcription start sites within the FMR1 promoter region with expansion of the downstream CGG-repeat element. Hum Mol Genet 13:543–549 14722156 [DOI] [PubMed] [Google Scholar]
- Bennetto L, Pennington BF, Porter D, Taylor AK, Hagerman RJ (2001) Profile of cognitive functioning in women with the fragile X mutation. Neuropsychology 15:290–299 [PubMed] [Google Scholar]
- Berry-Kravis E, Lewin F, Wuu J, Leehey M, Hagerman R, Hagerman P, Goetz CG (2003) Tremor and ataxia in fragile X premutation carriers: blinded videotape study. Ann Neurol 53:616–623 10.1002/ana.10522 [DOI] [PubMed] [Google Scholar]
- Brin MF, Koller W (1998) Epidemiology and genetics of essential tremor. Mov Disord 13:55–63 [DOI] [PubMed] [Google Scholar]
- Brunberg JA, Jacquemont S, Hagerman RJ, Berry-Kravis EM, Grigsby J, Leehey MA, Tassone F, Brown WT, Greco CM, Hagerman PJ (2002) Fragile X premutation carriers: characteristic MR imaging findings of adult male patients with progressive cerebellar and cognitive dysfunction. AJNR Am J Neuroradiol 23:1757–1766 [PMC free article] [PubMed] [Google Scholar]
- Chen LS, Tassone F, Sahota P, Hagerman PJ (2003) The (CGG)n repeat element within the 5′ untranslated region of the FMR1 message provides both positive and negative cis effects on in vivo translation of a downstream reporter. Hum Mol Genet 12:3067–3074 10.1093/hmg/ddg331 [DOI] [PubMed] [Google Scholar]
- Conway GS, Payne NN, Webb J, Murray A, Jacobs PA (1998) Fragile X premutation screening in women with premature ovarian failure. Hum Reprod 13:1184–1187 9647544 [DOI] [PubMed] [Google Scholar]
- Crawford DC, Zhang F, Wilson B, Warren ST, Sherman SL (2000) Fragile X CGG repeat structures among African-Americans: identification of a novel factor responsible for repeat instability. Hum Mol Genet 9:1759–1769 10.1093/hmg/9.12.1759 [DOI] [PubMed] [Google Scholar]
- Cummings CJ, Zoghbi HY (2000) Trinucleotide repeats: mechanisms and pathophysiology. Annu Rev Genomics Hum Genet 1:281–328 10.1146/annurev.genom.1.1.281 [DOI] [PubMed] [Google Scholar]
- Davis BM, McCurrach ME, Taneja KL, Singer RH, Housman DE (1997) Expansion of a CUG trinucleotide repeat in the 3′ untranslated region of myotonic dystrophy protein kinase transcripts results in nuclear retention of transcripts. Proc Natl Acad Sci USA 94:7388–7393 10.1073/pnas.94.14.7388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Maria E, Grasso M, Pigullo S, Faravelli F, Abbruzzese G, Barone P, Martinelli P, Ratto S, Sciolla R, Bellone E, Dagna-Bricarelli F, Ajmar F, Mandich P (2003) Further evidence that a tremor/ataxia syndrome may occur in fragile X premutation carriers. Paper presented at American Society of Human Genetics, 53rd Annual Meeting, Los Angeles, November 4–8 [Google Scholar]
- Dogu O, Sevim S, Camdeviren H, Sasmaz T, Bugdayci R, Aral M, Kaleagasi H, Un S, Louis ED (2003) Prevalence of essential tremor: door-to-door neurologic exams in Mersin province, Turkey. Neurology 61:1804–1806 [DOI] [PubMed] [Google Scholar]
- Dombrowski C, Levesque S, Morel ML, Rouillard P, Morgan K, Rousseau F (2002) Premutation and intermediate-size FMR1 alleles in 10,572 males from the general population: loss of an AGG interruption is a late event in the generation of fragile X syndrome alleles. Hum Mol Genet 11:371–378 10.1093/hmg/11.4.371 [DOI] [PubMed] [Google Scholar]
- Fardaei M, Rogers MT, Thorpe HM, Larkin K, Hamshere MG, Harper PS, Brook JD (2002) Three proteins, MBNL, MBLL and MBXL, co-localize in vivo with nuclear foci of expanded-repeat transcripts in DM1 and DM2 cells. Hum Mol Genet 11:805–814 10.1093/hmg/11.7.805 [DOI] [PubMed] [Google Scholar]
- Feng Y, Zhang F, Lokey LK, Chastain JL, Lakkis L, Eberhart D, Warren ST (1995) Translational suppression by trinucleotide repeat expansion at FMR1. Science 268:731–734 [DOI] [PubMed] [Google Scholar]
- Filla A, De Michele G, Marconi R, Bucci L, Carillo C, Castellano AE, Iorio L, Kniahynicki C, Rossi F, Campanella G (1992) Prevalence of hereditary ataxias and spastic paraplegias in Molise, a region of Italy. J Neurol 239:351–353 [DOI] [PubMed] [Google Scholar]
- Finsterer J (2002) Myotonic dystrophy type 2. Eur J Neurol 9:441–447 10.1046/j.1468-1331.2002.00453.x [DOI] [PubMed] [Google Scholar]
- Franke P, Leboyer M, Gansicke M, Weiffenbach O, Biancalana V, Cornillet-Lefebre P, Croquette MF, Froster U, Schwab SG, Poustka F, Hautzinger M, Maier W (1998) Genotype-phenotype relationship in female carriers of the premutation and full mutation of FMR-1. Psychiatry Res 80:113–127 10.1016/S0165-1781(98)00055-9 [DOI] [PubMed] [Google Scholar]
- Fu YH, Kuhl DP, Pizzuti A, Pieretti M, Sutcliffe JS, Richards S, Verkerk AJ, Holden JJ, Fenwick RG Jr, Warren ST, Oostra BA, Nelson DL, Caskey CT (1991) Variation of the CGG repeat at the fragile X site results in genetic instability: resolution of the Sherman paradox. Cell 67:1047–1058 [DOI] [PubMed] [Google Scholar]
- Gane LW, Cronister A (2002) Genetic counseling. In: Hagerman RJ, Hagerman PJ (eds) Fragile X syndrome: diagnosis, treatment, and research. Johns Hopkins University Press, Baltimore, pp 251–286 [Google Scholar]
- Garcia Arocena D, Breece KE, Hagerman PJ (2003) Distribution of CGG repeat sizes within the fragile X mental retardation 1 (FMR1) homologue in a non-human primate population. Hum Genet 113:371–376 10.1007/s00439-003-0982-9 [DOI] [PubMed] [Google Scholar]
- Gilman S, Low PA, Quinn N, Albanese A, Ben-Shlomo Y, Fowler CJ, Kaufmann H, Klockgether T, Lang AE, Lantos PL, Litvan I, Mathias CJ, Oliver E, Robertson D, Schatz I, Wenning GK (1999) Consensus statement on the diagnosis of multiple system atrophy. J Neurol Sci 163:94–98 10.1016/S0022-510X(98)00304-9 [DOI] [PubMed] [Google Scholar]
- Greco CM, Hagerman RJ, Tassone F, Chudley AE, Del Bigio MR, Jacquemont S, Leehey M, Hagerman PJ (2002) Neuronal intranuclear inclusions in a new cerebellar tremor/ataxia syndrome among fragile X carriers. Brain 125:1760–1771 10.1093/brain/awf184 [DOI] [PubMed] [Google Scholar]
- Hagerman P, Iwahashi C, Babineau B, Yasui D, Greco CM, Duncan M, Graw S, Kim F, Hagerman RJ (2003a) Fragile X-associated tremor/ataxia syndrome (FXTAS): a common heritable neuronal inclusion disorder. Neurology 60:A469 [Google Scholar]
- Hagerman PJ, Greco CM, Hagerman RJ (2003b) A cerebellar tremor/ataxia syndrome among fragile X premutation carriers. Cytogenet Genome Res 100:206–212 10.1159/000072856 [DOI] [PubMed] [Google Scholar]
- Hagerman RJ (2002a) Medical follow-up and pharmacotherapy. In: Hagerman RJ, Hagerman PJ (eds) Fragile X syndrome: diagnosis, treatment and research. The Johns Hopkins University Press, Baltimore, pp 287–338 [Google Scholar]
- Hagerman RJ (2002b) Physical and behavioral phenotype. In: Hagerman RJ, Hagerman PJ (eds) Fragile X syndrome: diagnosis, treatment and research. The Johns Hopkins University Press, Baltimore, pp 3–109 [Google Scholar]
- Hagerman RJ, Leavitt BR, Farzin F, Jacquemont S, Greco CM, Brunberg JA, Tassone F, Hessl D, Harris SW, Zhang L, Jardini T, Gane LW, Ferranti J, Ruiz L, Leehey MA, Grigsby J, Hagerman PJ (2004) Fragile X–associated tremor/ataxia syndrome (FXTAS) in females with the FMR1 premutation. Am J Hum Genet 74:1051–1056 (in this issue) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagerman RJ, Leehey M, Heinrichs W, Tassone F, Wilson R, Hills J, Grigsby J, Gage B, Hagerman PJ (2001) Intention tremor, parkinsonism, and generalized brain atrophy in male carriers of fragile X. Neurology 57:127–130 [DOI] [PubMed] [Google Scholar]
- Hundscheid RD, Braat DD, Kiemeney LA, Smits A, Thomas CM (2001) Increased serum FSH in female fragile X premutation carriers with either regular menstrual cycles or on oral contraceptives. Hum Reprod 16:457–462 10.1093/humrep/16.3.457 [DOI] [PubMed] [Google Scholar]
- Jacquemont S, Farzin F, Hall DA, Leehey MA, Tassone F, Gane LW, Zhang L, Grigsby J, Jardini T, Lewin F, Berry-Kravis E, Hagerman PJ, Hagerman RJ (2004a) Aging in individuals with the FMR1 mutation. Am J Ment Retard 109:154–164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacquemont S, Hagerman RJ, Leehey M, Grigsby J, Zhang L, Brunberg JA, Greco C, Des Portes V, Jardini T, Levine R, Berry-Kravis E, Brown WT, Schaeffer S, Kissel J, Tassone F, Hagerman PJ (2003) Fragile X premutation tremor/ataxia syndrome: molecular, clinical, and neuroimaging correlates. Am J Hum Genet 72:869–878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacquemont S, Hagerman RJ, Leehey MA, Hall DA, Levine RA, Brunberg JA, Zhang L, Jardini T, Gane LW, Harris SW, Herman K, Grigsby J, Greco C, Berry-Kravis E, Tassone F, Hagerman PJ (2004b) Penetrance of the fragile X-associated tremor/ataxia syndrome (FXTAS) in a premutation carrier population: initial results from a California family-based study. JAMA 291:460–469 [DOI] [PubMed] [Google Scholar]
- Jin P, Zarnescu DC, Zhang F, Pearson CE, Lucchesi JC, Moses K, Warren ST (2003) RNA-mediated neurodegeneration caused by the fragile X premutation rCGG repeats in Drosophila. Neuron 39:739–747 [DOI] [PubMed] [Google Scholar]
- Johnston C, Eliez S, Dyer-Friedman J, Hessl D, Glaser B, Blasey C, Taylor A, Reiss A (2001) Neurobehavioral phenotype in carriers of the fragile X premutation. Am J Med Genet 103:314–319 10.1002/ajmg.1561.abs [DOI] [PubMed] [Google Scholar]
- Kenneson A, Zhang F, Hagedorn CH, Warren ST (2001) Reduced FMRP and increased FMR1 transcription is proportionally associated with CGG repeat number in intermediate-length and premutation carriers. Hum Mol Genet 10:1449–1454 10.1093/hmg/10.14.1449 [DOI] [PubMed] [Google Scholar]
- Kimber TE, Blumbergs PC, Rice JP, Hallpike JF, Edis R, Thompson PD, Suthers G (1998) Familial neuronal intranuclear inclusion disease with ubiquitin positive inclusions. J Neurol Sci 160:33–40 10.1016/S0022-510X(98)00169-5 [DOI] [PubMed] [Google Scholar]
- Leehey MA, Munhoz RP, Lang AE, Brunberg JA, Grigsby J, Greco C, Jacquemont S, Tassone F, Lozano AM, Hagerman PJ, Hagerman RJ (2003) The fragile X premutation presenting as essential tremor. Arch Neurol 60:117–121 [DOI] [PubMed] [Google Scholar]
- Leone M, Bottacchi E, D’Alessandro G, Kustermann S (1995) Hereditary ataxias and paraplegias in Valle d’Aosta, Italy: a study of prevalence and disability. Acta Neurol Scand 91:183–187 [DOI] [PubMed] [Google Scholar]
- Loesch DZ, Hay DA, Mulley J (1994) Transmitting males and carrier females in fragile X: revisited. Am J Med Genet 51:392–399 [DOI] [PubMed] [Google Scholar]
- Loesch DZ, Huggins RM, Bui QM, Taylor AK, Hagerman RJ (2003a) Relationship of deficits of FMR1 gene specific protein with physical phenotype of fragile X males and females in pedigrees: a new perspective. Am J Med Genet 118A:127–134 12655493 [DOI] [PubMed] [Google Scholar]
- Loesch DZ, Huggins RM, Bui QM, Taylor AK, Pratt C, Epstein J, Hagerman RJ (2003b) Effect of fragile X status categories and FMRP deficits on cognitive profiles estimated by robust pedigree analysis. Am J Med Genet 122A:13–23 12949966 [DOI] [PubMed] [Google Scholar]
- Louis ED, Marder K, Cote L, Pullman S, Ford B, Wilder D, Tang MX, Lantigua R, Gurland B, Mayeux R (1995) Differences in the prevalence of essential tremor among elderly African Americans, whites, and Hispanics in northern Manhattan, NY. Arch Neurol 52:1201–1205 [DOI] [PubMed] [Google Scholar]
- Macpherson J, Waghorn A, Hammans S, Jacobs P (2003) Observation of an excess of fragile-X premutations in a population of males referred with spinocerebellar ataxia. Hum Genet 112:619–620 [DOI] [PubMed] [Google Scholar]
- Maddalena A, Richards CS, McGinniss MJ, Brothman A, Desnick RJ, Grier RE, Hirsch B, Jacky P, McDowell GA, Popovich B, Watson M, Wolff DJ (Quality Assurance Subcommittee of the Laboratory Practice Committee) (2001) Technical standards and guidelines for fragile X: the first of a series of disease-specific supplements to the Standards and Guidelines for Clinical Genetics Laboratories of the American College of Medical Genetics. Genet Med 3:200–205 10.1097/00125817-200105000-00010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mankodi A, Thornton CA (2002) Myotonic syndromes. Curr Opin Neurol 15:545–552 10.1097/00019052-200210000-00005 [DOI] [PubMed] [Google Scholar]
- Marozzi A, Vegetti W, Manfredini E, Tibiletti MG, Testa G, Crosignani PG, Ginelli E, Meneveri R, Dalpra L (2000) Association between idiopathic premature ovarian failure and fragile X premutation. Hum Reprod 15:197–202 10.1093/humrep/15.1.197 [DOI] [PubMed] [Google Scholar]
- Michaud J, Gilbert JJ (1981) Multiple system atrophy with neuronal intranuclear hyaline inclusions: report of a new case with light and electron microscopic studies. Acta Neuropathol 54:113–119 [DOI] [PubMed] [Google Scholar]
- Mori M, Adachi Y, Kusumi M, Nakashima K (2001) A genetic epidemiological study of spinocerebellar ataxias in Tottori prefecture, Japan. Neuroepidemiology 20:144–149 10.1159/000054775 [DOI] [PubMed] [Google Scholar]
- Nolin SL, Brown WT, Glicksman A, Houck GE Jr, Gargano AD, Sullivan A, Biancalana V, Bröndum-Nielsen K, Hjalgrim H, Holinski-Feder E, Kooy F, Longshore J, Macpherson J, Mandel J-L, Matthijs G, Rousseau F, Steinbach P, Väisänen M-L, von Koskull H, Sherman SL (2003) Expansion of the fragile X CGG repeat in females with premutation or intermediate alleles. Am J Hum Genet 72:454–464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pembrey ME, Winter RM, Davies KE (1985) A premutation that generates a defect at crossing over explains the inheritance of fragile X mental retardation. Am J Med Genet 21:709–717 [DOI] [PubMed] [Google Scholar]
- Polo JM, Calleja J, Combarros O, Berciano J (1991) Hereditary ataxias and paraplegias in Cantabria, Spain: an epidemiological and clinical study. Brain 114:855–866 [DOI] [PubMed] [Google Scholar]
- Primerano B, Tassone F, Hagerman RJ, Hagerman P, Amaldi F, Bagni C (2002) Reduced FMR1 mRNA translation efficiency in fragile X patients with premutations. RNA 8:1482–1488 [PMC free article] [PubMed] [Google Scholar]
- Pujana MA, Corral J, Gratacos M, Combarros O, Berciano J, Genis D, Banchs I, Estivill X, Volpini V, the Ataxia Study Group (1999) Spinocerebellar ataxias in Spanish patients: genetic analysis of familial and sporadic cases. Hum Genet 104:516–522 10.1007/s004390050997 [DOI] [PubMed] [Google Scholar]
- Ranum LP, Day JW (2002) Dominantly inherited, non-coding microsatellite expansion disorders. Curr Opin Genet Dev 12:266–271 10.1016/S0959-437X(02)00297-6 [DOI] [PubMed] [Google Scholar]
- Reiss AL, Freund L, Abrams MT, Boehm C, Kazazian H (1993) Neurobehavioral effects of the fragile X premutation in adult women: a controlled study. Am J Hum Genet 52:884–894 [PMC free article] [PubMed] [Google Scholar]
- Riddle JE, Cheema A, Sobesky WE, Gardner SC, Taylor AK, Pennington BF, Hagerman RJ (1998) Phenotypic involvement in females with the FMR1 gene mutation. Am J Ment Retard 102:590–601 9606468 [DOI] [PubMed] [Google Scholar]
- Rogers C, Partington M, Turner G (2003) Tremor, ataxia and dementia in older men may indicate a carrier of the fragile X syndrome. Clin Genet 64:54–56 10.1034/j.1399-0004.2003.00089.x [DOI] [PubMed] [Google Scholar]
- Rousseau F, Heitz D, Biancalana V, Blumenfeld S, Kretz C, Boue J, Tommerup N, Van Der Hagen C, DeLozier Blanchet C, Croquette MF, Gilgenkrantz S, Jalbert P, Voelckel MA, Oberlé I, Mandel JL (1991) Direct diagnosis by DNA analysis of the fragile X syndrome of mental retardation. N Engl J Med 325:1673–1681 [DOI] [PubMed] [Google Scholar]
- Rousseau F, Rouillard P, Morel ML, Khandjian EW, Morgan K (1995) Prevalence of carriers of premutation-size alleles of the FMRI gene—and implications for the population genetics of the fragile X syndrome. Am J Hum Genet 57:1006–1018 [PMC free article] [PubMed] [Google Scholar]
- Schenck A, Van de Bor V, Bardoni B, Giangrande A (2002) Novel features of dFMR1, the Drosophila orthologue of the fragile X mental retardation protein. Neurobiol Dis 11:53–63 10.1006/nbdi.2002.0510 [DOI] [PubMed] [Google Scholar]
- Seznec H, Agbulut O, Sergeant N, Savouret C, Ghestem A, Tabti N, Willer JC, Ourth L, Duros C, Brisson E, Fouquet C, Butler-Browne G, Delacourte A, Junien C, Gourdon G (2001) Mice transgenic for the human myotonic dystrophy region with expanded CTG repeats display muscular and brain abnormalities. Hum Mol Genet 10:2717–2726 10.1093/hmg/10.23.2717 [DOI] [PubMed] [Google Scholar]
- Smits A, van der Bruggen W, Sistermans E, Kiemeney B, Renier W, Kremer B (2002) Striking neurological symptoms in normal transmitting males of the fragile X syndrome. Paper presented at The 8th International Fragile X Conference. Chicago, July 16–21 [Google Scholar]
- Sobesky WE (1996) The treatment of emotional and behavioral problems. In: Hagerman RJ, Cronister A (eds) Fragile X syndrome: diagnosis, treatment, and research, 2nd ed. The Johns Hopkins University Press, Baltimore, pp 332–348 [Google Scholar]
- Taneja KL, McCurrach M, Schalling M, Housman D, Singer RH (1995) Foci of trinucleotide repeat transcripts in nuclei of myotonic dystrophy cells and tissues. J Cell Biol 128:995–1002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tassone F, Hagerman PJ (2003) Expression of the FMR1 gene. Cytogenet Genome Res 100:124–128 10.1159/000072846 [DOI] [PubMed] [Google Scholar]
- Tassone F, Hagerman RJ, Chamberlain WD, Hagerman PJ (2000a) Transcription of the FMR1 gene in individuals with fragile X syndrome. Am J Med Genet 97:195–203 11449488 [DOI] [PubMed] [Google Scholar]
- Tassone F, Hagerman RJ, Loesch DZ, Lachiewicz A, Taylor AK, Hagerman PJ (2000b) Fragile X males with unmethylated, full mutation trinucleotide repeat expansions have elevated levels of FMR1 messenger RNA. Am J Med Genet 94:232–236 [DOI] [PubMed] [Google Scholar]
- Tassone F, Hagerman RJ, Taylor AK, Gane LW, Godfrey TE, Hagerman PJ (2000c) Elevated levels of FMR1 mRNA in carrier males: a new mechanism of involvement in fragile-X syndrome. Am J Hum Genet 66:6–15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tassone F, Hagerman RJ, Taylor AK, Hagerman PJ (2001) A majority of fragile X males with methylated, full mutation alleles have significant levels of FMR1 messenger RNA. J Med Genet 38:453–456 10.1136/jmg.38.7.453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tassone F, Hagerman RJ, Taylor AK, Mills JB, Harris SW, Gane LW, Hagerman PJ (2000d) Clinical involvement and protein expression in individuals with the FMR1 premutation. Am J Med Genet 91:144–152 [DOI] [PubMed] [Google Scholar]
- van de Warrenburg BP, Sinke RJ, Verschuuren-Bemelmans CC, Scheffer H, Brunt ER, Ippel PF, Maat-Kievit JA, Dooijes D, Notermans NC, Lindhout D, Knoers NV, Kremer HP (2002) Spinocerebellar ataxias in the Netherlands: prevalence and age at onset variance analysis. Neurology 58:702–708 [DOI] [PubMed] [Google Scholar]
- Verkerk AJ, Pieretti M, Sutcliffe JS, Fu YH, Kuhl DP, Pizzuti A, Reiner O, Richards S, Victoria MF, Zhang FP, Eussen BE, van Ommen GJB, Blonden LAJ, Riggins GJ, Chastain JL, Kunst CB, Galjaard H, Caskey CT, Nelson DL, Oostra BA, Warren ST (1991) Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell 65:905–914 [DOI] [PubMed] [Google Scholar]
- Willemsen R, Hoogeveen-Westerveld M, Reis S, Holstege J, Severijnen LA, Nieuwenhuizen IM, Schrier M, Van Unen L, Tassone F, Hoogeveen AT, Hagerman PJ, Mientjes EJ, Oostra BA (2003) The FMR1 CGG repeat mouse displays ubiquitin-positive intranuclear neuronal inclusions: implications for the cerebellar tremor/ataxia syndrome. Hum Mol Genet 12:949–959 10.1093/hmg/ddg114 [DOI] [PubMed] [Google Scholar]
- Willemsen R, Los F, Mohkamsing S, van den Ouweland A, Deelen W, Galjaard H, Oostra B (1997) Rapid antibody test for prenatal diagnosis of fragile X syndrome on amniotic fluid cells: a new appraisal. J Med Genet 34:250–251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willemsen R, Mohkamsing S, de Vries B, Devys D, van den Ouweland A, Mandel JL, Galjaard H, Oostra B (1995) Rapid antibody test for fragile X syndrome. Lancet 345:1147–1148 10.1016/S0140-6736(95)90979-6 [DOI] [PubMed] [Google Scholar]
- Zannolli R, Gilman S, Rossi S, Volpi N, Bernini A, Galluzzi P, Galimberti D, Pucci L, D’Ambrosio A, Morgese G, Giannini F (2002) Hereditary neuronal intranuclear inclusion disease with autonomic failure and cerebellar degeneration. Arch Neurol 59:1319–1326 10.1001/archneur.59.8.1319 [DOI] [PubMed] [Google Scholar]
- Zhong N, Ju W, Pietrofesa J, Wang D, Dobkin C, Brown WT (1996) Fragile X “gray zone” alleles: AGG patterns, expansion risks, and associated haplotypes. Am J Med Genet 64:261–265 [DOI] [PubMed] [Google Scholar]
- Zhong N, Yang W, Dobkin C, Brown WT (1995) Fragile X gene instability: anchoring AGGs and linked microsatellites. Am J Hum Genet 57:351–361 [PMC free article] [PubMed] [Google Scholar]