Abstract
Exposure to early life stress (ELS) increases the risk for developing psychopathology; however, the mechanisms underlying this association are not clear. In this study we examined systemic inflammation as a pathway that may link exposure to stress to altered neural correlates of implicit emotion regulation in adolescents with varying levels of exposure to ELS (n = 83; 52 females, 31 males; 15.63 ± 1.10 years). We measured ventrolateral prefrontal cortex (vlPFC) activation and functional connectivity (FC) between the bilateral amygdala and the vlPFC as adolescents completed an affect labeling task in the scanner and assessed concentrations of C-reactive protein (CRP) using a dried blood spot protocol. We found that CRP levels were negatively associated with vlPFC activation during implicit regulation of negatively-valenced stimuli, and that cumulative severity of ELS exposure moderated this neuroimmune association. Severity of ELS also significantly moderated the association between CRP levels and FC between the bilateral amygdala and l-vlPFC during implicit emotion regulation: in adolescents who had been exposed to more severe ELS, higher CRP was associated with more negative frontoamygdala FC during implicit regulation of negatively-valenced stimuli. Thus, ELS may disrupt the normative association between the immune system and the neural processes that underlie socioemotional functioning potentially increasing adolescents’ risk for maladaptive outcomes.
Keywords: Early life stress, Adolescence, Emotion regulation, fMRI, Inflammation, C-reactive protein
1. Introduction
Individuals exposed to early life stress (ELS) are at risk for developing mental health difficulties, such as depression (McLaughlin et al., 2012), which often have their onset in adolescence, a developmental period that is also marked by heightened emotional reactivity (Heller & Casey, 2016). ELS has been found to alter the development and functioning of neural regions and circuitry involved in emotion processing and emotion regulation, including the amygdala and prefrontal cortex (PFC) (Heim & Binder, 2012). The biological pathways linking stress exposure in childhood and adolescence to these altered neural characteristics, however, are not clear.
It is well established from animal and human research that psychosocial stressors can trigger immune responses similar to those elicited by physical pathogens (Segerstrom & Miller, 2004; Steptoe et al., 2007). In the context of ELS, researchers have posited that inflammation is an important mechanism through which early psychosocial stressors can increase biological risk for disorder (for reviews, see Danese & Lewis, 2017; Fagundes et al., 2013). For example, numerous studies have documented that ELS is associated with heightened systemic inflammation in childhood (Slopen et al., 2013) and adolescence (Danese et al., 2011); further, in adults, the relation between ELS and inflammation has been found to be independent of current stress levels, health, and health behaviors (Danese et al., 2007). Thus, the biological embedding of stressors may lead to the early development of a proinflammatory phenotype that persists into adulthood. Theorists have posited that high levels of inflammation contribute to stress-related conditions associated with ELS exposure, such as depression and anxiety (Felger, 2018; Miller et al., 2009; Slavich and Irwin, 2014).
The neuroimmune network hypothesis offers an integrative framework to examine this formulation (Nusslock & Miller, 2016). This hypothesis proposes that exposure to ELS amplifies the normative bidirectional communication between peripheral immune cells that mediate stress-related inflammatory responses and the brain circuits involved in affective processing (Salvador et al., 2021). From this framework, individuals exposed to ELS exhibit a phenotype of low-grade systemic inflammation and altered neural activation when they process emotional material. Indeed, previous studies have found that inflammation is related to activation in brain regions associated with threat processing, such as the amygdala (Inagaki et al., 2012; Mehta et al., 2018; Muscatell et al., 2016; Swartz et al., 2017). Specifically, immune challenge studies, in which inflammation is elevated experimentally using bacterial endotoxin or vaccines, have found that heightened inflammation leads to greater amygdala activation to negative stimuli (Inagaki et al., 2012; Muscatell et al., 2016). Further, in an observational study, Swartz et al. (2017) reported in college-age males that higher levels of C-reactive protein (CRP) are associated with increased amygdala activation during threat processing. Importantly, the results of a growing number of studies are providing support for the neuroimmune network hypothesis in samples of children (Miller et al., 2021), adolescents (Chat et al., 2022), and adults (Kraynak et al., 2019). Perhaps most relevant to the current study, Miller et al. (2021) reported that ELS, specifically socioeconomic disadvantage, interacts with amygdala reactivity during threat-processing to predict a proinflammatory phenotype in early adolescence.
While many of the studies described above examining neuroimmune associations focused on neural activation in response to the processing of emotional stimuli (i.e., emotion reactivity), it is likely that emotion regulation also plays a critical role in the relation between inflammation and emotion-linked neural activation (Renna, 2021). For example, Appleton et al. (2013) found that practicing adaptive emotion regulation strategies, such as cognitive reappraisal (Gross & John, 2003), is associated with lower levels of peripheral CRP. Less research, however, has examined the association between inflammation and neural aspects of emotion regulation, such as activation in the prefrontal cortex (PFC) and functional connectivity (FC) between the PFC and the amygdala (Pozzi et al., 2021). Past studies investigating FC during affective processing tasks have generally focused on emotion reactivity (Alvarez et al., 2020; Harrison et al., 2009; Muscatell et al., 2015). Three studies have described associations between inflammation levels and altered resting-state FC in brain networks underlying emotional regulation processes. Nusslock et al. (2019) reported that higher concentrations of inflammation (indexed by a composite measure of CRP, interleukin-6 [IL-6], IL-10, and tumor necrosis factor α [TNF-α]) were associated with lower resting-state FC in a network of regions involved in emotion regulation (bilateral somatomotor area, inferior frontal gyrus, precentral gyrus, middle temporal gyrus, and angular gyrus) in two samples of African-American youth. Mehta et al., (2018) reported a negative association between CRP levels and resting-state FC between the right amygdala and left ventromedial prefrontal cortex (l-vmPFC) in a sample of adults diagnosed with Major Depressive Disorder. Kraynak et al. (2019) similarly found that IL-6 was negatively related to FC between the amygdala and vmPFC in healthy adults. This study also found that the effect of childhood ELS on frontoamygdala FC was statistically mediated by IL-6, consistent with the neuroimmune network hypothesis. Such findings linking peripheral inflammation with resting-state FC patterns are important given that exposure to ELS has also been found to be associated with altered frontoamygdala FC in adolescents (Gee et al., 2013a; Miller et al., 2020; Peverill et al., 2019; Colich et al., 2017). Work from our group (based on the same cohort examined in the present study but during early pubertal development, assessed 4 years prior) indicated that ELS exposure was associated with more negative FC between bilateral amygdala and the right ventrolateral PFC (R-vlPFC) during an implicit emotion regulation task (Colich et al., 2017). That study, however, did not examine the role of inflammation in influencing neural activation. Taken together, there appears to be a significant overlap among ELS exposure, neural correlates of emotion regulation, and systemic inflammation, although this formulation has not been assessed during emotion regulation.
The purpose of the present study was to examine associations among severity of ELS, levels of peripheral inflammation, and frontoamygdala FC during implicit emotion regulation in adolescents with varying exposure to ELS as they completed an affect labeling task in the scanner. Prior studies of affect labeling indicate that labeling versus matching emotional faces recruits implicit emotion regulation processes through linguistic processing (Torre & Lieberman, 2018), eliciting robust activation in the vlPFC and reduced activation in the amygdala (Colich et al., 2017; Gee et al., 2012; Lieberman et al., 2007). In this study, we examined whether the strength of the association between inflammation and frontoamygdala FC during implicit emotion regulation varies with exposure to ELS. Based on prior literature consistent with the neuroimmune framework (Chat et al., 2022; Kraynak et al., 2019; Miller et al., 2021; Nusslock and Miller, 2016), we generated three hypotheses: 1) levels of CRP will be related to neural activation in the vlPFC during an implicit emotion regulation task; 2) ELS will moderate the association of CRP with neural activation in the vlPFC; 3a) levels of CRP will be related to FC between the bilateral amygdala and the vlPFC; and 3b) ELS will moderate the association of CRP with FC between the bilateral amygdala and the vlPFC.
2. Methods
2.1. Participants
214 adolescents participated in a longitudinal study examining the psychobiological effects of early life stress on brain structure and function through puberty. Participants were recruited using media and online advertisements posted in local communities in the surrounding Bay Area. Inclusion criteria for the study were that participants be 9–13 years of age at study entry and be proficient in English. Because of the focus of the study, at study entry males and females were matched on pubertal stage, assessed by self-report Tanner Staging (Morris & Udry, 1980). Exclusion criteria included standard MRI contraindications (e.g., metal implants or braces), as well as a history of major neurological or medical illness, severe learning disabilities and, for females, the onset of menses. Of the 214 adolescents enrolled in the study, 144 completed the third timepoint of the study (during 2018–2021), and blood samples were collected from a subset of these participants to measure inflammation. For the current analysis, we included participants who had both usable CRP and neuroimaging data at this third timepoint (n = 89). We excluded participants with poor quality fMRI data (e.g., excessive motion artifacts, poor image quality; n = 4) and poor performance on the task (n = 2) resulting in a final sample of 83 adolescents, ages 13.15–17.93 years (M = 15.63; SD = 1.10 years).
Participants completed two laboratory sessions at the third timepoint: 1) a behavioral session in which adolescents completed interviews and behavioral self-assessments; and 2) a neuroimaging session in which adolescents completed a two-hour MRI scan consisting of structural and functional sequences, followed by the collection of a blood sample using a dried blood spot protocol to assess CRP (see below). This study was approved by the Stanford University Institutional Review Board (IRB), and adolescents and their parents were compensated for their participation.
2.2. Early life stress assessment
At the first timepoint, a modified version of the Traumatic Events Screening Inventory for Children (TESI-C) (Ribbe, 1996) was administered to participants to assess more than 30 types of stressful life experiences (e.g. domestic violence, abuse, parental separation). For each event, participants described the stressful event and provided a subjective severity rating. An external panel of three or four coders blind to the participants’ subjective ratings then rated the objective severity of each event on a scale ranging from 0 (non-event) to 4 (high severity), in halfpoint increments, using a modified version of the UCLA Life Stress Interview coding system (Rudolph et al., 2000). A cumulative severity score was computed by summing the maximum objective severity scores from each type of endorsed stressful experience category (King et al., 2017, King et al., 2020).
2.3. Socioeconomic status (SES)
At the third timepoint, parents reported their annual household income and the number of people in their household. Income data was categorized into bins. Income-to-needs ratio (INR) was calculated by dividing the median of the household income bin by the 2019 Department of Housing and Urban Development low-income limit for Santa Clara County (https://www.huduser.gov/portal/datasets/il.html#2019) with respect to the number household members (King et al., 2019; Noble et al., 2015; Yu et al., 2018). Parental income data were obtained for 75 participants, and missing INR values (n = 8) were imputed using the mean INR of the sample.
2.4. C-reactive protein (CRP)
Peripheral CRP levels were assessed for each participant at the Stanford Human Immune Monitoring Center using a dried blood spot protocol (Ho et al., 2021, Ho et al., 2022). Five blood spots, consisting of approximately 150–250 μL per spot, were collected from the participants’ fingertips (non-dominant hand) using sterile contact-activated lancets (BD 366,594 Microtainer, BD Biosciences, San Jose, CA) onto filter paper cards (Whatman #903, GE Healthcare, Pascataway, NJ). These spots were then dried overnight at room temperature before being placed in biohazard bags with a dessicant and stored in a freezer at −20 °C. For the CRP assay, a single dried blood spot was extracted and placed in a 1.5 ml tube containing 400 μL of radioimmunoprecipitation buffer and protease inhibitor and was incubated overnight with orbital shaking (4 °C; 500–600 rpm). Following extraction the vials were centrifuged (room temperature; 17000 rpm, 10 min.) and liquid transferred to a clean polypropylene plate. The polypropylene plate was the source for CRP assay which required a 2000-fold dilution of extracted samples. The assay plate containing anti-CRP antibody beads was incubated with 25ul of diluted sample overnight at 4 °C with shaking. The plate was then washed with an ELx 405 Biotek washer. Biotinylated detection antibody was added for 60 min at room temperature with shaking. The plate was washed again and streptavidin-PE was added for 30 min at room temperature with shaking. The plate was washed again and PBS reading buffer was added to the wells. Next, the plate was read in the Luminex FlexMap3D Instrument. All data were analyzed using MasterPlex software (Hitachi Software Engineering America ltd., MiraiBio Group). CRP concentrations are reported in log-transformed median fluorescence intensity values (MFI), with higher MFI values indicating higher peripheral inflammatory levels of CRP. CRP data were assayed in four batches, which were controlled for in all statistical analyses. The intra-assay coefficient of variation for CRP was 5.7 % ± 5.4 % and the lower limits of detection was 10 pg/mL.
2.5. MRI acquisition
Neuroimaging data were acquired at the Stanford Center for Cognitive and Neurobiological Imaging (CNI). The CNI scanner was upgraded during the data collection period from a 3 T General Electric (GE) Discovery MR750 scanner to a 3 T GE Ultra High Performance (UHP) system. Thus, MRI data from 68 participants were acquired with the MR750 and data from 15 participants were acquired with the UHP, both using the same 32-channel head coil (Nova Medical). Scanner model was included as a covariate in the analyses. Structural imaging included a T1-weighted IR-prep, fast SPGR (3D BRAVO) sequence (repetition time [TR] = 6.3 ms; echo time [TE] = 3.1 ms; flip angle = 12°; matrix = 256 × 256; FOV = 23.04 cm, slice thickness = 0.9 mm, voxel resolution = 0.9 mm isotropic. The T1w acquisition time was 5:16. Functional imaging was acquired using two T2*-weighted gradient echo EPI pulse sequences (TR = 2000 ms; TE = 30 ms; flip angle = 77°; matrix = 70 × 70; FOV = 22.4 cm, slice thickness = 3 mm, voxel resolution = 3.2 mm × 3.2 mm × 3.0 mm). Each fMRI sequence acquisition time was 5:54, with a total task scan time of 12 min across both runs.
2.6. Implicit emotion regulation fMRI task
Participants completed a modified affect labeling task that we used to assess the neural correlates of implicit emotion regulation (Hariri et al., 2000; Lieberman et al., 2007; Colich et al., 2017). Participants saw a target facial expression on the top of the screen and were instructed to select the correct option located in the bottom left or bottom right of the screen. During the Label conditions, the two options were words describing the emotion; during Match conditions, the two options were images of facial expressions. Emotions within a block were either all positively-valenced (happy, calm, excited) or all negatively-valenced (fearful, angry, sad). A sensorimotor control condition was included where participants matched shapes (Shape Match), resulting in five separate conditions. Each condition had a block of 10 stimuli trials, each presented on the screen for 5 s, and task conditions were interspersed with a fixation rest block lasting 15 s. A single run consisted of one block of each task condition: Positive Match and Positive Label (i.e., positive emotions), Negative Match and Negative Label (i.e., negative emotions), and Shape Match, and the interspersed rest blocks. Conditions and images were presented randomly across all participants, who completed two runs of the task. While the Label condition involves implicit emotion regulation processes, it also involves shared processes that are engaged as participants complete the Match condition (e.g., lower-level visual processing of basic facial features, identification of emotion categories). Therefore, we examined the contrast between Positive Label > Positive Match and Negative Label > Negative Match in order to isolate the processes specific to implicit emotion regulation, as has been done in other studies (Colich et al., 2017; Lieberman et al., 2007).
2.7. Image preprocessing
All neuroimaging data were initially preprocessed using the fMRIPrep (20.2.1) pipeline (Esteban et al., 2019, Esteban et al., 2020). This workflow conducted automated preprocessing steps on both anatomical (T1w) and functional (task fMRI) data, based on empirically optimized steps. A summary of the preprocessing is presented below; see the Supplement for a full reproduction of the fMRIPrep summary output. The T1w anatomical image was bias field corrected (Tustison et al., 2010), which adjusted for intensity non-uniformity, using Advanced Normalization Tools (ANTs v.2.2.0; Avants et al., 2008). ANTs was also used to skull-strip the image, and the resultant brain tissue was segmented into cerebrospinal fluid, white matter, and gray matter with fast from the FMRIB Software Library (FSL v.5.0.9; Smith et al., 2004; Zhang et al., 2001). The brain surface was reconstructed using recon_all in Freesurfer (v.6.0.1; Dale et al., 1999), followed by a volume-based spatial normalization to the MNIPediatricAsym (Cohort 6) template, an unbiased, age-appropriate MRI template designed for post-pubertal participants ages 13–18.5 years (Fonov et al., 2011). Normalization was conducted using the ANTs nonlinear registration tool antsRegistration.
The fMRI data were preprocessed by creating an unwarped reference volume and skull-stripping the image using a custom fMRIPrep algorithm. The BOLD reference was co-registered to the T1w reference with FreeSurfer’s boundary-based registration tool bbregister with 6 degrees of freedom (Greve & Fischl, 2009). Head-motion was estimated with FSL’s MCFLIRT (Jenkinson et al., 2002), and the BOLD runs were slicetime corrected with 3dTshift from the Analysis of Functional NeuroImages (AFNI v.20160207) software package (Cox & Hyde, 1997). Frames that exceeded a threshold of 0.5 mm framewise displacement (FD) or 1.5 standardized derivative of root mean square variance over voxels (DVARS) (Power et al., 2014; Smyser et al., 2011) were annotated as motion outliers and censored (“scrubbed”) from the fMRI model estimation by adding single-TR regressors at censored volumes (as described in Siegel et al., 2014). The time-series were resampled into MNIPediatricAsym (Cohort 6) standard spaces to match the anatomical images. Confounding time-series were calculated including FD, DVARS, and three global signals from the cerebrospinal fluid, white matter, and whole-brain masks. Physiological regressors were also extracted for component-based noise correction (CompCor); the first six were used (Behzadi et al., 2007). These were based on high-pass filtering of the BOLD data to yield temporal and anatomical components. In summary, the resultant fMRIPrep outputs were preprocessed anatomical and affect task fMRI data spatially registered to age-appropriate template space and a table of confound regressors. These data were visually inspected for image quality and motion artifacts. Participants were excluded if they met any of the following quality control criteria: more than five non-steady-state volumes for both runs (n = 0); more than 20 % of the imaging data were marked for censoring by fMRIPrep (n = 0); and they had significant motion artifacts upon visual inspection (n = 4). We also excluded participants if they performed poorly during the affect labeling task, defined as having<50 % average accuracy on any of the task conditions (n = 2). These exclusion criteria resulted in an overall final sample of n = 83.
The preprocessed and inspected fMRIPrep outputs were then analyzed using FSL (v. 6.0.1) with FEAT (fMRI Expert Analysis Tool; (Smith et al., 2004; Woolrich et al., 2001). The first five volumes of each dataset were removed to allow for the stabilization of the longitudinal magnetization. The data were spatially smoothed with a Gaussian kernel (FWHM = 5 mm) and underwent high-pass temporal filtering (Gaussian-weighted least squares straight line fitting, sigma = 100.0 s) prior to first-level statistical analysis. First-level individual analysis was conducted using FSL’s FILM (FMRIB’s Improved Linear Model) with prewhitening to improve estimation of each voxel’s time series. The voxel-wise general linear model included regressors for each task condition and selected confounds generated from fMRIPrep: FD, anatomical CompCor components 1–5, and motion parameters (six rigid-body parameters and their first temporal derivatives and quadratics, censored motion outliers). A double-gamma convolution was used to model the hemodynamic response function. Each participant’s within-subject runs were combined using a fixed-effects model using FLAME I (FMIRB’s Local Analysis of Mixed Effects; Beckmann et al., 2003); this procedure averaged the contrast estimates over the two runs within subjects by setting the random effects variance to zero.
2.8. Statistical analyses
2.8.1. Participant characteristics
We computed bivariate Pearson correlations to assess the association between CRP and demographic and physical participant characteristics to assess for potential confounding effects. We conducted an independent samples t-test to examine sex differences in levels of CRP, and a one-way analysis of variance (ANOVA) to examine differences in levels of CRP as a function of race. All analyses were conducted using R (v. 4.1.2; R Core Team, 2021).
2.8.2. ELS associations with CRP
We constructed linear regression models to assess whether severity of ELS predicted levels of CRP. The linear models, and all subsequent analyses, included the following covariates of noninterest known to affect the BOLD signal or inflammation: age, sex (coded categorically), BMI, recent steroid, antibiotic, or allergy medication use (coded categorically; n = 13), recent illness (coded categorically; n = 8), race (coded categorically; see Table 1 for race composition of sample), INR, assay batch (coded categorically), and, when analyses included fMRI data, scanner model (coded categorically).
Table 1.
Participant characteristics. Demographic, physical, and psychological characteristics of the adolescent sample are reported, in full and separated by sex. Psychological assessments included the TESI interview for ELS. Bivariate Pearson correlations between relevant variables and CRP are also reported. Differences in inflammation in race were assessed with a one-way ANOVA. Units: age-years; CRP-logMFI; ELS-raw scores. *indicates significance at the α = 0.05 level.
| Full | Male | Female | Corr. With CRP | |
|---|---|---|---|---|
| n | 83 | 31 | 52 | |
| Age, M (SD) | 15.63 (1.10) | 16.10 (0.82) | 15.35 (1.15) | r = −0.15 |
| Range | 13.15–17.93 | 14.61–17.54 | 13.15–17.93 | |
| Race | F(5,77) = 0.76 | |||
| African American | 4 (4.8 %) | 1 (3.2 %) | 3 (5.8 %) | p = 0.58 |
| Asian | 10 (12 %) | 4 (12.9 %) | 6 (11.5 %) | |
| Biracial | 12 (14.5 %) | 4 (12.9 %) | 8 (15.4 %) | |
| Caucasian | 44 (53 %) | 18 (58.1 %) | 26 (50 %) | |
| Hispanic | 8 (9.6 %) | 2 (6.5 %) | 6 (11.5 %) | |
| Other | 5 (6 %) | 2 (6.5 %) | 3 (5.8 %) | |
| Physical Status | ||||
| BMI, M (SD) | 21.6 (4.2) | 22.4 (4.3) | 21.2 (4.1) | r = 0.47* |
| Range | 15.4–37.1 | 16.4–35.5 | 15.4–37.1 | |
| Recent Illness | 8 | 4 | 4 | |
| Corticosteroid, Antibiotic, or Allergy Med. | 13 | 5 | 8 | |
| Psychiatric Medication | 8 | 2 | 6 | |
| CRP, M (SD) | 3.99 (1.05) | 4.19 (1.14) | 3.87 (0.98) | |
| Range | 2.71–7.09 | 2.71–7.09 | 2.71–6.78 | |
| Socioeconomic Status | ||||
| Income-to-Needs ratio, M (SD) | 1.15 (0.37) | 1.20 (0.24) | 1.12 (0.43) | r = −0.03 |
| Range | 0.07–1.80 | 0.60–1.60 | 0.07–1.80 | |
| Low Income, % (n) | 21.7 % (18) | 16.1 % (5) | 25.0 % (13) | |
| Psychological Assessments | ||||
| Early Life Stress Severity, M (SD) | 6.86 (5.36) | 6.85 (4.78) | 6.87 (5.73) | r = 0.04 |
| Range | 0–22 | 1–22 | 0–20 | |
| Imaging Quality | ||||
| Percent Censored M (SD) | 3.1 (3.7) | 3.7 (4.0) | 2.8 (3.5) | r = 0.13 |
| Range | 0–18.0 | 0–14.5 | 0–18.0 |
2.8.3. Task performance
Participants’ mean reaction time (RT) and accuracy were computed across all conditions. We conducted paired t-tests to compare these performance metrics during the Positive Label vs Positive Match and Negative Label vs Negative Match conditions. We also conducted linear regression analyses to determine whether CRP concentration levels predicted task accuracy in each of the five conditions, controlling for age, sex, BMI, recent steroid, antibiotic, or allergy medication use, recent illness, race, INR, and assay batch. Regression significance levels were adjusted with a multiple-comparison-corrected α = 0.01. Two participants were excluded from the task performance analysis due to corrupted performance data, resulting in n = 81 for all analyses involving fMRI task data. We also conducted linear regression analyses examining whether stress severity predicted task accuracy using a similar multiple-comparison-corrected α = 0.01. We also conducted whole-brain cluster analyses, described below, to examine the main effects of the Label and Match task conditions on neural activation, controlling for age and sex. These results are reported in the Supplement.
2.8.4. Emotion regulation-related brain regions exhibiting significant associations with CRP (H1)
For Hypothesis 1, we tested whether CRP was related to neural correlates of implicit emotion regulation by conducting a data-driven whole-brain cluster analysis. We conducted a higher-level group mixed effects model analysis of the fixed effects contrast parameter estimates (PEs) (Smith et al., 2004) using FSL’s FLAME I to investigate the main effect of CRP on the BOLD signal during the two implicit emotion regulation conditions (Negative Label > Negative Match and Positive Label > Positive Match). The cluster defining threshold was set to Z = 3.1, with a multiple-comparisons-corrected cluster threshold of α = 0.025 for the two emotion conditions. Resultant clusters were defined based on maximal overlap of regions from the Brainnetome atlas, which was defined using a connectivity-based parcellation framework (Fan et al., 2016). In a post-hoc exploratory analysis, we tested whether the CRP-related activation from the resultant clusters were associated with the severity of ELS. PEs of cluster activation were extracted using FSL’s fslmeants tool and regressed onto CRP levels. The linear model controlled for age, sex, and scanner model.
2.8.5. ELS moderating associations of CRP with neural activation during implicit emotion regulation (H2)
For Hypothesis 2, we conducted two multiple regression analyses to examine the moderating effect of ELS severity on the linear association between CRP concentration and neural activation during implicit emotion regulation in the two vlPFC clusters identified from the data-driven cluster analysis (l-vlPFC and R-vlPFC). The models assessed participant ELS severity scores, CRP levels, and extracted PEs from the l-vlPFC and R-vlPFC clusters, and they included an interaction term between ELS severity and CRP levels. Analyses were conducted using a multiple-comparisons-corrected level of α = 0.025 for the two frontal clusters. Significant interactions were probed using simple slopes analysis (Aiken et al., 1991) and the Johnson-Neyman technique (Johnson & Neyman, 1936).
2.8.6. ELS moderating associations of CRP with frontoamygdala functional connectivity during implicit emotion regulation (H3)
For Hypothesis 3, we examined whether CRP levels moderated the linear association between ELS severity and frontoamygdala FC during implicit emotion regulation. To assess this, we conducted two psychophysiological interaction (PPI) analyses to examine the FC between the bilateral amygdala and the two vlPFC regions exhibiting a significant effect of CRP during implicit emotion regulation (results from analyses described in the previous section, 2.8.4.). The first PPI analysis examined FC between the bilateral amygdala and the l-vlPFC; the second PPI analysis examined FC between the bilateral amygdala and the R-vlPFC. The PPI method measures the task-specific interaction between a brain region (e.g. bilateral amygdala in the current analysis) and a seed region (e.g. one of the two frontal clusters with significant CRP-related activation) (Friston et al., 1997; O’Reilly et al., 2012). For each participant, we conducted an individual PPI GLM analysis in FSL that included the original individual-level task regressors including a regressor for the implicit emotion regulation contrast (Negative Label > Negative Match), the extracted timeseries from the frontal cluster, and an interaction between the task contrast and the cluster time series. Individual-level PPI results from the two task runs were combined in a fixed-effects model using FLAME I to average the contrast estimates, generating whole-brain maps of FC with the frontal seed region. To measure frontoamygdala FC, we used a bilateral amygdala mask to extract PEs of FC from the whole-brain maps. The mask was defined using the Harvard-Oxford Subcortical Structural atlas (75 % threshold) (Desikan et al., 2006). The amygdala was assessed bilaterally, as in our previous work (see Colich et al., 2017), given that we did not have hemisphere-specific hypotheses (hemisphere-specific findings are reported in the Supplement).
For Hypothesis 3a, we examined whether CRP levels predicted frontoamygdala FC between 1) the bilateral amygdala and the l-vlPFC; and 2) the bilateral amygdala and the R-vlPFC. Two linear models were constructed and analyzed with an α = 0.025, corrected for the two regression analyses.
For Hypothesis 3b, we assessed the moderating effect of ELS on the association between CRP and FC between the 1) bilateral amygdala and the l-vlPFC; and 2) the bilateral amygdala and the R-vlPFC. We conducted two multiple regression analyses that included an interaction between ELS and CRP, using an α = 0.025. We analyzed significant interactions with simple slopes analysis and the Johnson-Neyman technique.
3. Results
3.1. Participant characteristics
Participant characteristics and associations between CRP and select variables are presented in Table 1. Eight participants were taking psychiatric medication, predominantly antidepressant medication.1 Participants came from relatively high-SES households; 21.7 % of parents reported low-income status (INR < 1). There was a large range of ELS severity scores (range = 0–22; M = 6.9; SD = 5.4). Male and female participants did not differ significantly in ELS severity (t(81) = −0.01, p = 0.99) and in CRP levels (t(81) = −1.36, p = 0.18). Overall, the majority of imaging data passed the motion scrubbing thresholds (range = 0–18.0 %; M = 3.1 %; SD = 3.7 %).
3.2. Association of ELS with CRP
Regression analysis indicated that severity of ELS did not significantly predict adolescents’ CRP concentrations (β = −0.03, t(73) = −1.32, p = 0.191), controlling for age, sex, BMI, recent steroid, antibiotic, or allergy medication use, recent illness, race, INR, and assay batch.
3.3. Task performance
Participants had significantly better accuracy (t(80) = 6.39, p < 0.001) and faster RTs (t(80) = −6.14, p < 0.001) in the Negative Label condition than in the Negative Match condition. Participants had significantly better accuracy (t(80) = 3.48, p < 0.001) during the Positive Match condition than during the Positive Label condition; there was no significant difference in RT between the positive emotion conditions (t (80) = −0.94, p = 0.35). Task descriptive statistics are reported in the Supplement. Linear regression analyses indicated that CRP significantly predicted participants’ accuracy during both Label conditions (Negative Label condition (β = 0.32, t(71) = 2.29, p = 0.025) and Positive Label condition (β = 0.40, t(71) = 2.82, p = 0.006), but the Negative Label finding was not significant after correcting for multiple comparisons. ELS severity did not significantly predict task accuracy in any of the five conditions (all p > 0.08). Whole-brain cluster analyses examining the main effect of the task conditions identified clusters spanning visual, inferior frontal, and amygdala regions. Specific cluster results are reported in the Supplement.
3.4. Brain regions exhibiting significant effect of CRP (H1)
The whole-brain analysis identified five significant clusters with CRP-related activation during the Negative Label > Negative Match contrast, visualized in Fig. 1. Two of the clusters were located in the l-vlPFC and R-vlPFC. Additional areas in which there was CRP-related activation included two SFG clusters and an anterior insula cluster. Specific cluster information is presented in Table 2. The association between CRP and PEs of activation was negative: specifically, higher CRP levels were associated with reduced activation during the implicit emotion regulation contrast. When examining associations between CRP and cluster activation during the main effects of the task, CRP was significantly associated with activation in the l-vlPFC during both the Negative Label and Negative Match conditions (Label: β = −0.29, t(66) = −2.33, p = 0.023; Match: β = 0.46, t(66) = 3.65, p = 0.001). CRP was significantly associated with activation in the R-vlPFC during the Negative Label condition only (Label: β = −0.43, t(66) = −3.33, p = 0.001; Match: β = 0.25 t(66) = 1.88, p = 0.064). Neuroimmune associations for the vlPFC clusters are visualized in Fig. 2 and results for all clusters are reported in the Supplement. No significant clusters were identified in the Positive Label > Positive Match contrast; thus, only the negative emotion conditions were examined in further analyses. Post-hoc analyses showed that severity of ELS did not significantly predict activation in any of the five clusters in the Negative Label > Negative Match contrast (all p > 0.274).
Fig. 1.

CRP is negatively associated with neural activation during implicit regulation of negative emotions. Data-driven analysis identified five clusters with significant CRP-related BOLD activation during the Negative Label > Negative Match contrast: l-vlPFC (red), R-vlPFC (orange), bilateral SFG (blue), R-SFG (not shown), and l-anterior insula (green); Z-threshold = 3.1 and α = 0.025. The two vlPFC clusters were used as seeds in the functional connectivity analysis. Cluster images displayed over a 1 mm MNI standard template.
Table 2.
CRP-Related Cluster Details. Results from the whole-brain cluster analysis identifying CRP-related activation during implicit emotion regulation of negatively-valenced faces. All neuroimmune associations were negative: at higher CRP, activation in frontal clusters was more negative. Z-threshold = 3.1; α = 0.025. All coordinates reported in MNI standard space. SFG = superior frontal gyrus, vlPFC = ventrolateral prefrontal cortex, Ant. = anterior, L = left, R = left, A = Brodmann Area, COG = center of gravity.
| Z-Statistics | Z-Max | COG | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Region | Brainnetome Label | Voxels | p | Mean | Max | X | Y | Z | X | Y | Z |
| Bilateral SFG | A8m | 449 | <0.001 | 3.61 | 5.02 | 4 | 20 | 64 | 4 | 22 | 60 |
| l-vlPFC | A44op, A44v, A45c | 264 | <0.001 | 3.66 | 4.90 | −42 | 20 | 4 | −44 | 18 | 2 |
| R-SFG | A9m | 178 | 0.002 | 3.42 | 4.16 | 8 | 42 | 26 | 6 | 42 | 24 |
| R-vlPFC | A44v, A45c, A44op | 163 | 0.004 | 3.47 | 4.40 | 58 | 18 | 0 | 52 | 20 | 4 |
| l-Ant. Insula | vIa | 134 | 0.010 | 3.41 | 3.99 | −26 | 16 | −10 | −32 | 18 | −14 |
Fig. 2.

Neuroimmune associations during implicit emotion regulation, labeling, and matching of negative emotions. PEs from the two vlPFC clusters displaying significant CRP-related activation were extracted during the negative implicit emotion regulation contrast (Label > Match) (blue), Negative Label condition (green), and Negative Match condition (red). Plots show that activation was negatively associated with CRP during the Label > Match and Label conditions, and activation was positively associated with CRP during the Match condition. PEs are reported in arbitrary units; CRP reported in log-MFI; *denotes significant association. PE = parameter estimate; vlPFC = ventrolateral prefrontal cortex; L = left; R = right.
3.5. ELS moderating associations of CRP with neural activation during implicit emotion regulation (H2)
We conducted two multiple regressions to test whether severity of ELS moderated the association of CRP with neural activation in the l-vlPFC and R-vlPFC clusters during implicit emotion regulation. There was a significant interaction between ELS and CRP that predicted l-vlPFC activation (β = −0.28, t(64) = −2.32, p = 0.024); the interaction for R-vlPFC activation was not significant (β = −0.11, t(64) = −0.87, p = 0.390). The results remained significant after controlling for participants’ average accuracy in the two negative conditions (β = −0.28, p = 0.048), although they were not significant when conservatively controlling for multiple comparisons.
Follow-up simple slopes analysis indicated that in adolescents who experienced a greater severity of ELS, higher CRP was associated with lower activation in the l-vlPFC during the implicit emotion regulation contrast. The Johnson-Neyman interval specifying the lower- and upperbounds for which ELS had a significant moderating effect on the association between CRP and l-vlPFC activation was −15.50, −0.99 (z-scored values). Simple slopes and the Johnson-Neyman interval are visualized in Fig. 3.
Fig. 3.

ELS severity moderates the association of CRP with activation in the l-vlPFC during implicit emotion regulation. Top) Simple slopes of the ELS-CRP interaction. At greater ELS severity (solid red), adolescents with higher CRP show reduced activation in the l-vlPFC during the Negative Label > Negative Match contrast. Bottom) Johnson-Neyman plot displaying the levels where ELS has a significant moderating effect on the CRP-l-vlPFC activation relation (sig. range in red). The dotted red line marks the upper boundary of the interval (−0.99). The black horizontal bar represents the observed ELS severity levels (z-scored).
3.6. ELS moderating associations of CRP with frontoamygdala functional connectivity during implicit emotion regulation (H3)
For Hypothesis 3a, we conducted two linear models to examine whether CRP predicted frontoamygdala FC between the bilateral amygdala and the l-vlPFC or R-vlPFC during implicit emotion regulation. CRP did not predict amygdala-based FC with either the l-vlPFC (β = −0.21, t(66) = −1.40, p = 0.167) or the R-vlPFC (β = −0.18, t(66) = −1.25, p = 0.215). The results remained nonsignificant when controlling for negative condition accuracy.
For Hypothesis 3b, we conducted two multiple regressions to test whether severity of ELS moderated the association of CRP with FC between the bilateral amygdala and the l-vlPFC, and with FC between the bilateral amygdala and the R-vlPFC during implicit emotion regulation. There was a significant interaction between ELS severity and CRP (β = −0.37, t(64) = −2.43, p = 0.018) that predicted FC between the bilateral amygdala and the l-vlPFC; the interaction term predicting FC between the bilateral amygdala and the R-vlPFC was not statistically significant (β = −0.16, t(64) = −1.00, p = 0.323). The interaction remained significant when controlling for negative condition accuracy (β = −0.46, p = 0.009).
Follow-up simple slopes analysis for the significant bilateral amygdala-l-vlPFC interaction indicated that in adolescents who were exposed to greater severity of ELS, higher CRP was associated with more negative FC between bilateral amygdala and l-vlPFC. Conversely, in adolescents who were exposed to lower severity of ELS, higher CRP was associated with more positive FC between bilateral amygdala and l-vlPFC FC. The Johnson-Neyman interval specifying the lower- and upperbounds for which ELS had a significant moderating effect on the association between CRP and bilateral amygdala and l-vlPFC FC was −4.04, 0.12 (z-scored values). Simple slopes and the Johnson-Neyman interval are visualized in Fig. 4.
Fig. 4.
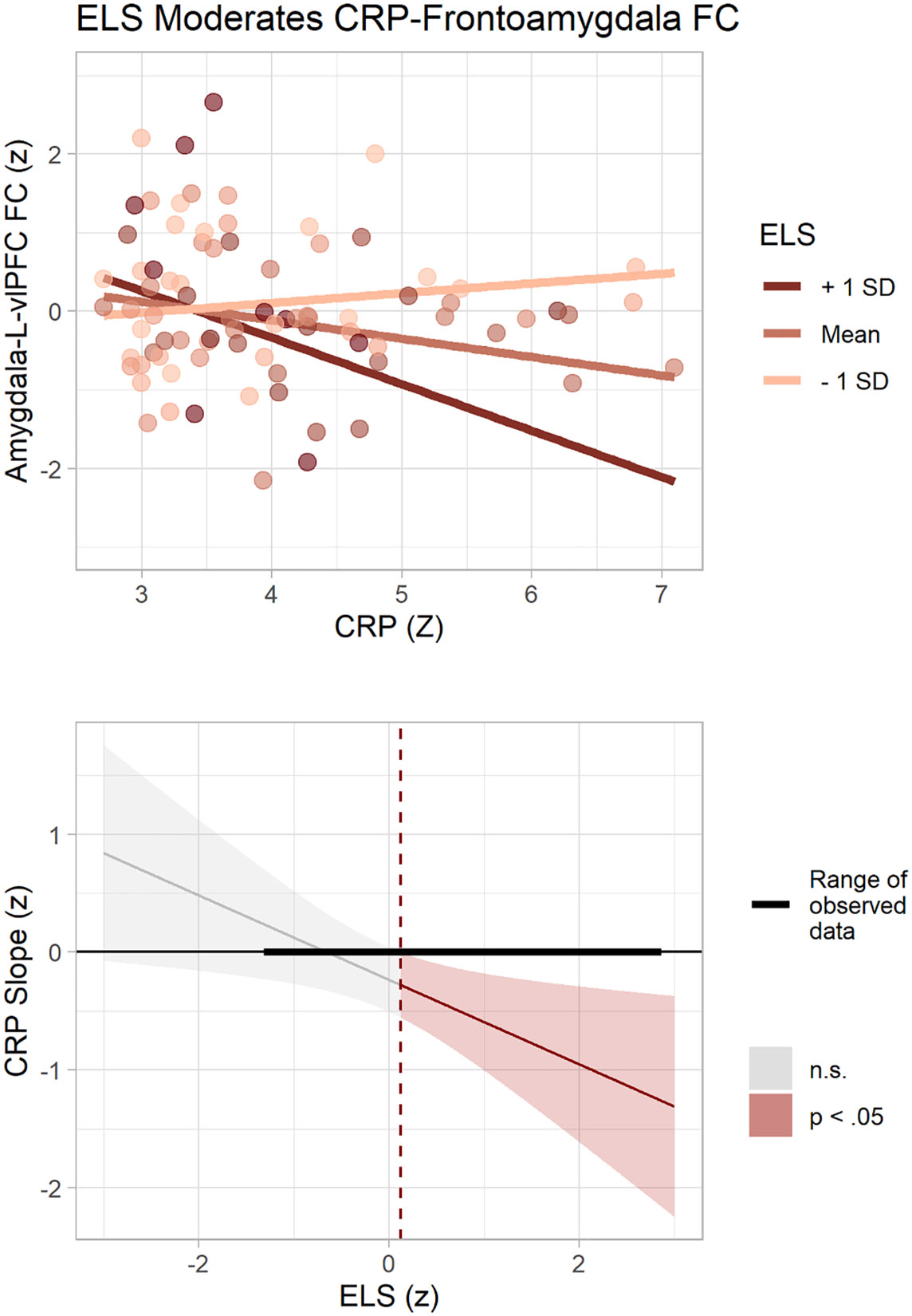
ELS severity moderates the association of CRP with frontoamygdala FC during implicit emotion regulation. Top) Simple slopes of the ELS-CRP interaction. At greater ELS severity (solid red), adolescents with higher CRP show more negative FC between the bilateral amygdala and l-vlPFC during the Negative Label > Negative Match contrast. Bottom) Johnson-Neyman plot displaying the levels where ELS has a significant moderating effect on the CRP-FC relation (sig. range in red). The dotted red line marks the upper boundary of the interval (0.12). The black horizontal bar represents the observed ELS severity levels (z-scored).
4. Discussion
Elevated systemic inflammation has been found to be associated with altered neural function in emotion processing circuitry. Exposure to ELS may potentiate this neuroimmune association and contribute to the increased risk for maladaptive outcomes associated with ELS. In this context, the present study was conducted to examine how severity of ELS affects the relation between systemic inflammation levels and neural function during implicit emotion regulation in adolescents with varying exposure to ELS during childhood. Our study had three primary findings: 1) CRP levels were negatively associated with activation in the R- and l-vlPFC during implicit emotion regulation of negative emotional faces; 2) severity of ELS moderated the association of CRP with l-vlPFC activation; and 3) severity of ELS moderated the association of CRP with functional connectivity between the bilateral amygdala and the l-vlPFC during implicit emotion regulation; however, we did not find that adolescents’ levels of CRP predicted FC between bilateral amygdala and l-vlPFC. This study is the first to demonstrate both that peripheral inflammation is associated with neural activation during implicit emotion regulation in adolescents, and that experiences of ELS moderate these associations.
Our results support previous findings that elevated systemic inflammation is associated with altered neural activation during the processing of negative emotions (H1). Previous research has documented the sensitizing effects of inflammation on limbic reactivity during the processing of negatively valenced stimuli (i.e., amygdala activation during the matching of fearful or angry faces). In our study, CRP was not associated with amygdala activation during negative emotion matching (see Supplemental analyses); rather, our findings suggest that heightened inflammation is associated with reduced activation during implicit emotion regulation in the vlPFC, SFG, and anterior insula. The two vlPFC cluster findings are particularly relevant in this context. The R-vlPFC has been previously implicated in affect labeling (Lieberman et al., 2007). The l-vlPFC is particularly associated with language processing (Nozari & Thompson-Schill, 2016), and thus, its activation makes sense given the linguistic component of the labeling task. Further, the vlPFC plays a significant role in the processing of negative emotions (Pozzi et al., 2021) and has been found to be recruited during both implicit and explicit forms of emotion regulation (Morawetz et al., 2017). Further, Bayesian dynamic causal modeling of neural activation during affect labeling supports the interpretation that the vlPFC down-regulates amygdala responsiveness (Torrisi et al., 2013). Thus, the negative direction of the neuroimmune association suggests that higher levels of inflammation are associated with a diminished ability to down-regulate, or reduce, negative affect, which may explain the heightened sensitivity to negative stimuli often reported in neuroimmune studies (Inagaki et al., 2012; Muscatell et al., 2016; Swartz et al., 2017).
Results of the two interaction analyses (for H2 and H3b) add to a growing body of evidence supporting the neuroimmune network hypothesis, which posits that exposure to ELS alters the bidirectional communication between the immune system and the nervous system (Nusslock & Miller, 2016; see Chat et al., 2022; Kraynak et al., 2019; Miller et al., 2021). Mirroring Miller et al.’s (2021) findings in children living in poverty, our neural activation results from H2 indicate that experiencing more severe ELS in childhood is associated with potentiated neuroimmune associations in adolescence during the implicit emotion regulation of negatively-valenced emotions. In fact, we obtained a similar effect size for our interaction of early life stress and inflammation predicting l-vlPFC activation (β = −0.28) as Miller et al. did for their interaction of poverty and amygdala activation predicting childhood inflammatory status (β = 0.22). Interestingly, the frontoamygdala FC effect size we obtained in testing H3 was even larger (β = −0.37), suggesting that the relation between inflammation and frontoamygdala FC is particularly affected by ELS exposure. This interpretation is speculative, of course, given the differences in racial and SES composition between our study and Miller et al.’s study and the possibility that our larger effect size is inflated because of our smaller sample (see Limitations below). Kraynak et al. assessed resting-state FC between the amygdala and the vmPFC, similar to the current study. They found that in healthy midlife adults, the effect of experiencing childhood abuse on resting-state frontoamygdala FC was mediated by levels of peripheral concentration of IL-6. Considered in concert with the current findings, this supports the prediction of the neuroimmune network hypothesis that a significant interaction of ELS, inflammation, and frontoamygdala FC persists into adulthood but is detectable at an earlier developmental stage (i.e., adolescence). Interestingly, in contrast to other findings (e.g. Slopen et al., 2013), severity of ELS alone did not predict levels of inflammation in adolescence in this study, due perhaps to the wide variability of ELS exposure reported by our sample. Oversampling for exposure to stressors would likely yield a sample that exhibits the proinflammatory phenotype that has been reported in previous research.
Our findings support previous findings that exposure to ELS is associated with accelerated development, indexed by a variety of biological markers, including pubertal timing, cellular health, and neurodevelopment (for a review of ELS and biological aging, see Colich et al., 2020). For example, the stress acceleration hypothesis posits that ELS exposure hastens the development of neural networks that underlie socioemotional processing, and, in particular connectivity between the amygdala and PFC (Callaghan & Tottenham, 2016). The direction of amygdala-PFC coupling changes during adolescence: whereas children have more positive amygdala-PFC FC during processing of negative stimuli, adults exhibit more negative FC, a pattern that has been posited to reflect top-down regulation of the PFC on emotional stimuli processed in the amygdala (Gee et al., 2013b). Several studies have reported that adolescents exposed to various types of ELS have a more adult-like pattern of frontoamygdala FC (i.e. more negative) during emotion processing than do their non-exposed peers (e.g., Gee et al., 2013a; Miller et al., 2020; Peverill et al., 2019; Wolf and Herringa, 2016; Colich et al., 2017). In this context, immune function, specifically inflammatory mechanisms, appears to play an important role in the expression of this more mature neurophenotype. Interestingly, prior longitudinal analyses of the current cohort at a younger age indicate that this neurophenotype may confer protection against accelerated biological aging. Specifically, Miller et al. (2020) found that more negative frontoamygdala FC during a different emotion processing fMRI task was related to slower telomere shortening and slower pubertal tempo; further, ELS moderated this association such that these relations were stronger in adolescents who had experienced higher ELS. This stands in contrast to the formulation that exposure to ELS is associated with accelerated development across all markers of aging and suggests that more mature socioemotional functioning preferentially buffers adolescents who experienced ELS from other forms of biological aging. Given the current findings linking inflammation to frontoamygdala FC, future work should examine whether the immune system confers a similar protective effect against these maturational outcomes in adolescents exposed to high levels of ELS.
In contrast to other studies that found associations of inflammation with altered FC between brain regions involved in the processing and regulation of emotion (e.g., Alvarez et al., 2020; Harrison et al., 2009; Muscatell et al., 2015; Nusslock et al., 2019), we did not find a significant main effect of CRP on bilateral amygdala and l-vlPFC FC (H3a). It is noteworthy that researchers have found direct associations of CRP concentration and frontoamygdala FC in adults (Kraynak et al., 2019; Mehta et al., 2018) but not in adolescents (Swartz et al., 2021). Thus, the association of inflammation with frontoamygdala FC may follow a developmental trajectory that may not be observable until adulthood; however, future studies are needed to definitively test this possibility.
We should note four limitations of this study. First, as we noted above, the sample size is relatively modest, which, in the context of estimating effect sizes of associations of neural measures with psychological phenotypes, may yield inflated effect sizes (Marek et al., 2022). In particular, our frontoamygdala FC results warrant replication in larger samples. Second, the observational design of this study prevents us from establishing causal relations among ELS, inflammation, and brain function. Longitudinal designs, coupled with immune-challenge paradigms, are needed to address this issue, and would also improve measurement reliability for smaller-n studies. Third, we were not sufficiently powered to examine sex differences in the associations of levels of CRP with neural activation to emotional stimuli, nor were we able to examine whether different dimensions of stressful experiences, such as threat or deprivation, are associated with unique neuroimmune associations. There is evidence that neuroimmune relations are stronger in males than in females (Swartz et al., 2017), and that different dimensions of ELS are associated with unique patterns of neural activation (Chahal et al., 2022; McLaughlin et al., 2019). These questions should be addressed in future research with larger samples. Finally, half our participants were Caucasian, and many were from high-income backgrounds. Although we controlled for race and SES in our analyses, we cannot generalize our results to adolescents from other sociodemographic backgrounds.
5. Conclusions
The present findings support the predictions of the neuroimmune network hypothesis: severity of childhood ELS moderates the association of systemic inflammation and neural correlates of implicit emotion regulation in adolescents. Altered activation and FC may contribute to the overall sensitivity to negatively-valenced emotions associated with heightened inflammation. We speculate that inflammatory mechanisms play an important role in accelerated development following ELS exposure. From a clinical perspective, peripheral inflammatory processes could serve as targets for interventions and therapies aimed at reducing the many negative outcomes associated with ELS exposure.
Supplementary Material
Acknowledgements
We thank Anna Cichocki, Miranda Edwards, Jordan Garcia, Abigail Graber, Madelaine Graber, Melissa Hansen, Emma Jaeger, Vanessa Lopez, Amar Ojha, Holly Pham, Michelle Sanabria, Jill Segarra, Lucinda Sisk, Alexess Sosa, Megan Strickland, Giana Teresi, Johanna Walker, and Rachel Weisenburger for their assistance in data collection and management. We also thank Yael Rosenberg-Hasson and Holden Maecker for their assistance with the dried blood spot protocol, and Anthony Gifuni and Katy Werwath for their assistance in the early stages of this project. Finally, we thank all our participants and their families for participating in this research.
Funding
This research was supported by grants from the National Institute of Mental Health (R37MH101495 to IHG, F32MH120975 to RC, F32MH114317 to NLC, and K01MH117442 to TCH), from the Stanford University Precision Health and Integrated Diagnostics (PHIND) Center (to IHG and TCH), from the Klingenstein Third Generation Foundation (to RC), the Stanford Institute for Research in the Social Sciences (IRiSS) (to JPY), and the National Science Foundation Graduate Research Fellowship Program (NSF GRFP) (to JPY).
Footnotes
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Appendix A. Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bbi.2022.07.007.
We repeated the analyses for the three hypotheses controlling for participants taking psychiatric medication (n=8) and obtained the same pattern of significant findings (full results reported in the Supplement).
References
- Aiken LS, West SG, Reno RR, 1991. Multiple Regression: Testing and Interpreting Interactions. SAGE. [Google Scholar]
- Alvarez GM, Hackman DA, Miller AB, Muscatell KA, 2020. Systemic inflammation is associated with differential neural reactivity and connectivity to affective images. Social Cognit. Affective Neurosci 15 (10), 1024–1033. 10.1093/scan/nsaa065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Appleton AA, Buka SL, Loucks EB, Gilman SE, Kubzansky LD, 2013. Divergent associations of adaptive and maladaptive emotion regulation strategies with inflammation. Health Psychol. 32 (7), 748–756. 10.1037/a0030068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avants BB, Epstein CL, Grossman M, Gee JC, 2008. Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Med. Image Anal 12 (1), 26–41. 10.1016/j.media.2007.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beckmann CF, Jenkinson M, Smith SM, 2003. General multilevel linear modeling for group analysis in FMRI. NeuroImage 20 (2), 1052–1063. 10.1016/S1053-8119(03)00435-X. [DOI] [PubMed] [Google Scholar]
- Behzadi Y, Restom K, Liau J, Liu TT, 2007. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. NeuroImage 37 (1), 90–101. 10.1016/j.neuroimage.2007.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callaghan BL, Tottenham N, 2016. The stress acceleration hypothesis: effects of early-life adversity on emotion circuits and behavior. Curr. Opin. Behav. Sci 7, 76–81. 10.1016/j.cobeha.2015.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chahal R, Miller JG, Yuan JP, Buthmann JL, Gotlib IH, 2022. An exploration of dimensions of early adversity and the development of functional brain network connectivity during adolescence: implications for trajectories of internalizing symptoms. Dev. Psychopathol 34 (2), 557–571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chat I-K-Y, Gepty AA, Kautz M, Giollabhui NM, Adogli ZV, Coe CL, Abramson LY, Olino TM, Alloy LB, 2022. Residence in high-crime neighborhoods moderates the association between interleukin 6 and social and nonsocial reward brain responses. Biol. Psychiatry Global Open Sci 10.1016/j.bpsgos.2022.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colich NL, Williams ES, Ho TC, King LS, Humphreys KL, Price AN, Ordaz SJ, & Gotlib IH, 2017. The association between early life stress and prefrontal cortex activation during implicit emotion regulation is moderated by sex in early adolescence. Development and Psychopathology, 29(5), 1851–1864. doi: 10.1017/S0954579417001444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colich NL, Rosen ML, Williams ES, McLaughlin KA, 2020. Biological Aging in Childhood and Adolescence Following Experiences of Threat and Deprivation: A Systematic Review and Meta-Analysis. Psychol. Bull 146 (9), 721–764. 10.1037/bul0000270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox RW, Hyde JS, 1997. Software tools for analysis and visualization of fMRI data. NMR Biomed. 10 (4–5), 171–178. 10.1002/(SICI)1099-1492(199706/08)10:4/5<171::AID-NBM453>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- Dale AM, Fischl B, Sereno MI, 1999. Cortical Surface-Based Analysis: I. Segmentation and Surface Reconstruction. NeuroImage 9 (2), 179–194. 10.1006/nimg.1998.0395. [DOI] [PubMed] [Google Scholar]
- Danese A, Lewis S, 2017. Psychoneuroimmunology of early-life stress: the hidden wounds of childhood trauma? Neuropsychopharmacology 42 (1), 99–114. 10.1038/npp.2016.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danese A, Pariante CM, Caspi A, Taylor A, Poulton R, 2007. Childhood maltreatment predicts adult inflammation in a life-course study. Proc. Natl. Acad. Sci 104 (4), 1319–1324. 10.1073/pnas.0610362104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danese A, Caspi A, Williams B, Ambler A, Sugden K, Mika J, Werts H, Freeman J, Pariante CM, Moffitt TE, Arseneault L, 2011. Biological embedding of stress through inflammation processes in childhood. Mol. Psychiatry 16 (3), 244–246. 10.1038/mp.2010.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, Buckner RL, Dale AM, Maguire RP, Hyman BT, Albert MS, Killiany RJ, 2006. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage 31 (3), 968–980. 10.1016/j.neuroimage.2006.01.021. [DOI] [PubMed] [Google Scholar]
- Esteban O, Markiewicz CJ, Blair RW, Moodie CA, Isik AI, Erramuzpe A, Kent JD, Goncalves M, DuPre E, Snyder M, Oya H, Ghosh SS, Wright J, Durnez J, Poldrack RA, Gorgolewski KJ, 2019. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat. Methods 16 (1), 111–116. 10.1038/s41592-018-0235-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esteban O, Ciric R, Finc K, Blair RW, Markiewicz CJ, Moodie CA, Kent JD, Goncalves M, DuPre E, Gomez DEP, Ye Z, Salo T, Valabregue R, Amlien IK, Liem F, Jacoby N, Stojić H, Cieslak M, Urchs S, Halchenko YO, Ghosh SS, De La Vega A, Yarkoni T, Wright J, Thompson WH, Poldrack RA, Gorgolewski KJ, 2020. Analysis of task-based functional MRI data preprocessed with fMRIPrep. BioRxiv 15 (7), 2186–2202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fagundes CP, Glaser R, Kiecolt-Glaser JK, 2013. Stressful early life experiences and immune dysregulation across the lifespan. Brain Behav. Immun 27, 8–12. 10.1016/j.bbi.2012.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan L, Li H, Zhuo J, Zhang Y, Wang J, Chen L, Yang Z, Chu C, Xie S, Laird AR, Fox PT, Eickhoff SB, Yu C, & Jiang T, 2016. The Human Brainnetome Atlas: A New Brain Atlas Based on Connectional Architecture. Cerebral Cortex (New York, N.Y.: 1991), 26(8), 3508–3526. doi: 10.1093/cercor/bhw157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Felger JC, 2018. Imaging the role of inflammation in mood and anxiety-related disorders. Curr. Neuropharmacol 16 (5), 533–558. 10.2174/1570159X15666171123201142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fonov V, Evans AC, Botteron K, Almli CR, McKinstry RC, Collins DL, 2011. Unbiased average age-appropriate atlases for pediatric studies. NeuroImage 54 (1), 313–327. 10.1016/j.neuroimage.2010.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friston KJ, Buechel C, Fink GR, Morris J, Rolls E, Dolan RJ, 1997. Psychophysiological and modulatory interactions in neuroimaging. NeuroImage 6 (3), 218–229. 10.1006/nimg.1997.0291. [DOI] [PubMed] [Google Scholar]
- Gee DG, Karlsgodt KH, van Erp TGM, Bearden CE, Lieberman MD, Belger A, Perkins DO, Olvet DM, Cornblatt BA, Constable T, Woods SW, Addington J, Cadenhead KS, McGlashan TH, Seidman LJ, Tsuang MT, Walker EF, Cannon TD, 2012. Altered age-related trajectories of amygdala-prefrontal circuitry in adolescents at clinical high risk for psychosis: A preliminary study. Schizophr. Res 134 (1), 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gee DG, Gabard-Durnam LJ, Flannery J, Goff B, Humphreys KL, Telzer EH, Hare TA, Bookheimer SY, Tottenham N, 2013a. Early developmental emergence of human amygdala–prefrontal connectivity after maternal deprivation. Proc. Natl. Acad. Sci 110 (39), 15638–15643. 10.1073/pnas.1307893110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gee DG, Humphreys KL, Flannery J, Goff B, Telzer EH, Shapiro M, Hare TA, Bookheimer SY, Tottenham N, 2013b. A developmental shift from positive to negative connectivity in human amygdala-prefrontal circuitry. J. Neurosci 33 (10), 4584–4593. 10.1523/JNEUROSCI.3446-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greve DN, Fischl B, 2009. Accurate and robust brain image alignment using boundary-based registration. NeuroImage 48 (1), 63–72. 10.1016/j.neuroimage.2009.06.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross JJ, John OP, 2003. Individual differences in two emotion regulation processes: Implications for affect, relationships, and well-being. J. Pers. Soc. Psychol 85 (2), 348–362. 10.1037/0022-3514.85.2.348. [DOI] [PubMed] [Google Scholar]
- Hariri AR, Bookheimer SY, Mazziotta JC, 2000. Modulating emotional responses: Effects of a neocortical network on the limbic system. NeuroReport 11 (1), 43–48. 10.1097/00001756-200001170-00009. [DOI] [PubMed] [Google Scholar]
- Harrison NA, Brydon L, Walker C, Gray MA, Steptoe A, Critchley HD, 2009. Inflammation causes mood changes through alterations in subgenual cingulate activity and mesolimbic connectivity. Biol. Psychiatry 66 (5), 407–414. 10.1016/j.biopsych.2009.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heim C, Binder EB, 2012. Current research trends in early life stress and depression: review of human studies on sensitive periods, gene–environment interactions, and epigenetics. Exp. Neurol 233 (1), 102–111. 10.1016/j.expneurol.2011.10.032. [DOI] [PubMed] [Google Scholar]
- Heller AS, Casey BJ, 2016. The neurodynamics of emotion: Delineating typical and atypical emotional processes during adolescence. Dev. Sci 19 (1), 3–18. 10.1111/desc.12373. [DOI] [PubMed] [Google Scholar]
- Ho TC, Teresi GI, Segarra JR, Ojha A, Walker JC, Gu M, Spielman DM, Sacchet MD, Jiang F, Rosenberg-Hasson Y, Maecker H, Gotlib IH, 2021. Higher levels of pro-inflammatory cytokines are associated with higher levels of glutamate in the anterior cingulate cortex in depressed adolescents. Front. Psychiatry 12, 642976. 10.3389/fpsyt.2021.642976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho TC, Kulla A, Teresi GI, Sisk LM, Rosenberg-Hasson Y, Maecker HT, Gotlib IH, 2022. Inflammatory cytokines and callosal white matter microstructure in adolescents. Brain Behav. Immun 100, 321–331. 10.1016/j.bbi.2021.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inagaki TK, Muscatell KA, Irwin MR, Cole SW, Eisenberger NI, 2012. Inflammation selectively enhances amygdala activity to socially threatening images. NeuroImage 59 (4), 3222–3226. 10.1016/j.neuroimage.2011.10.090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson M, Bannister P, Brady M, Smith S, 2002. Improved optimization for the robust and accurate linear registration and motion correction of brain images. NeuroImage 17 (2), 825–841. 10.1006/nimg.2002.1132. [DOI] [PubMed] [Google Scholar]
- Johnson PO, Neyman J, 1936. Tests of certain linear hypotheses and their application to some educational problems. Statistical Res. Memoirs 1, 57–93. [Google Scholar]
- King LS, Colich NL, LeMoult J, Humphreys KL, Ordaz SJ, Price AN, Gotlib IH, 2017. The impact of the severity of early life stress on diurnal cortisol: the role of puberty. Psychoneuroendocrinology 77, 68–74. 10.1016/j.psyneuen.2016.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King LS, Humphreys KL, Camacho MC, Gotlib IH, 2019. A person-centered approach to the assessment of early life stress: associations with the volume of stresssensitive brain regions in early adolescence. Dev. Psychopathol 31 (02), 643–655. 10.1017/S0954579418000184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King LS, Graber MG, Colich NL, Gotlib IH, 2020. Associations of waking cortisol with DHEA and testosterone across the pubertal transition: effects of threat-related early life stress. Psychoneuroendocrinology 115, 104651. 10.1016/j.psyneuen.2020.104651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraynak TE, Marsland AL, Hanson JL, Gianaros PJ, 2019. Retrospectively reported childhood physical abuse, systemic inflammation, and resting corticolimbic connectivity in midlife adults. Brain Behav. Immun 82, 203–213. 10.1016/j.bbi.2019.08.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lieberman MD, Eisenberger NI, Crockett MJ, Tom SM, Pfeifer JH, Way BM, 2007. Putting feelings into words. Psychol. Sci 18 (5), 421–428. 10.1111/j.1467-9280.2007.01916.x. [DOI] [PubMed] [Google Scholar]
- Marek S, Tervo-Clemmens B, Calabro FJ, Montez DF, Kay BP, Hatoum AS, Donohue MR, Foran W, Miller RL, Hendrickson TJ, Malone SM, Kandala S, Feczko E, Miranda-Dominguez O, Graham AM, Earl EA, Perrone AJ, Cordova M, Doyle O, Dosenbach NUF, 2022. Reproducible brain-wide association studies require thousands of individuals. Nature 603 (7902), 654–660. 10.1038/s41586-022-04492-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin KA, Greif Green J, Gruber MJ, Sampson NA, Zaslavsky AM, Kessler RC, 2012. Childhood adversities and first onset of psychiatric disorders in a national sample of US adolescents. Arch. Gen. Psychiatry 69 (11), 1151–1160. 10.1001/archgenpsychiatry.2011.2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin KA, Weissman D, Bitrán D, 2019. Childhood adversity and neural development: a systematic review. Annu. Rev. Dev. Psychol 1 (1), 277–312. 10.1146/annurev-devpsych-121318-084950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta ND, Haroon E, Xu X, Woolwine BJ, Li Z, Felger JC, 2018. Inflammation negatively correlates with amygdala-ventromedial prefrontal functional connectivity in association with anxiety in patients with depression: preliminary results. Brain Behav. Immun 73, 725–730. 10.1016/j.bbi.2018.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller JG, Ho TC, Humphreys KL, King LS, Foland-Ross LC, Colich NL, Ordaz SJ, Lin J, Gotlib IH, 2020. Early life stress, frontoamygdala connectivity, and biological aging in adolescence: a longitudinal investigation. Cereb. Cortex 30 (7), 4269–4280. 10.1093/cercor/bhaa057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller AH, Maletic V, Raison CL, 2009. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 65 (9), 732–741. 10.1016/j.biopsych.2008.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller GE, White SF, Chen E, Nusslock R, 2021. Association of inflammatory activity with larger neural responses to threat and reward among children living in poverty. Am. J. Psychiatry 178 (4), 313–320. 10.1176/appi.ajp.2020.20050635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morawetz C, Bode S, Derntl B, Heekeren HR, 2017. The effect of strategies, goals and stimulus material on the neural mechanisms of emotion regulation: a meta-analysis of fMRI studies. Neurosci. Biobehav. Rev 72, 111–128. 10.1016/j.neubiorev.2016.11.014. [DOI] [PubMed] [Google Scholar]
- Morris NM, Udry JR, 1980. Validation of a self-administered instrument to assess stage of adolescent development. J. Youth Adolesc 9 (3), 271–280. 10.1007/BF02088471. [DOI] [PubMed] [Google Scholar]
- Muscatell KA, Dedovic K, Slavich GM, Jarcho MR, Breen EC, Bower JE, Irwin MR, Eisenberger NI, 2015. Greater amygdala activity and dorsomedial prefrontal–amygdala coupling are associated with enhanced inflammatory responses to stress. Brain Behav. Immun 43, 46–53. 10.1016/j.bbi.2014.06.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muscatell KA, Moieni M, Inagaki TK, Dutcher JM, Jevtic I, Breen EC, Irwin MR, Eisenberger NI, 2016. Exposure to an inflammatory challenge enhances neural sensitivity to negative and positive social feedback. Brain Behav. Immun 57, 21–29. 10.1016/j.bbi.2016.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noble KG, Houston SM, Brito NH, Bartsch H, Kan E, Kuperman JM, Akshoomoff N, Amaral DG, Bloss CS, Libiger O, Schork NJ, Murray SS, Casey BJ, Chang L, Ernst TM, Frazier JA, Gruen JR, Kennedy DN, Van Zijl P, Mostofsky S, Kaufmann WE, Kenet T, Dale AM, Jernigan TL, Sowell ER, 2015. Family income, parental education and brain structure in children and adolescents. Nat. Neurosci 18 (5), 773–778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nozari N, Thompson-Schill SL, 2016. Chapter 46—Left Ventrolateral Prefrontal Cortex in Processing of Words and Sentences. In: Hickok G, Small SL (Eds.), Neurobiology of Language. Academic Press, pp. 569–584. 10.1016/B978-0-12-407794-2.00046-8. [DOI] [Google Scholar]
- Nusslock R, Brody GH, Armstrong CC, Carroll AL, Sweet LH, Yu T, Barton AW, Hallowell ES, Chen E, Higgins JP, Parrish TB, Wang L, Miller GE, 2019. Higher peripheral inflammatory signaling associated with lower resting-state functional brain connectivity in emotion regulation and central executive networks. Biol. Psychiatry 86 (2), 153–162. 10.1016/j.biopsych.2019.03.968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nusslock R, Miller GE, 2016. Early-life adversity and physical and emotional health across the lifespan: a neuroimmune network hypothesis. Biol. Psychiatry 80 (1), 23–32. 10.1016/j.biopsych.2015.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Reilly JX, Woolrich MW, Behrens TEJ, Smith SM, Johansen-Berg H, 2012. Tools of the trade: Psychophysiological interactions and functional connectivity. Social Cognit. Affect. Neurosci 7 (5), 604–609. 10.1093/scan/nss055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peverill M, Sheridan MA, Busso DS, McLaughlin KA, 2019. Atypical prefrontal-amygdala circuitry following childhood exposure to abuse: links with adolescent psychopathology. Child Maltreatment 24 (4), 411–423. 10.1177/1077559519852676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Power JD, Mitra A, Laumann TO, Snyder AZ, Schlaggar BL, Petersen SE, 2014. Methods to detect, characterize, and remove motion artifact in resting state fMRI. NeuroImage 84, 320–341. 10.1016/j.neuroimage.2013.08.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pozzi E, Vijayakumar N, Rakesh D, Whittle S, 2021. Neural correlates of emotion regulation in adolescents and emerging adults: a meta-analytic study. Biol. Psychiatry 89 (2), 194–204. 10.1016/j.biopsych.2020.08.006. [DOI] [PubMed] [Google Scholar]
- R Core Team, 2021. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. [Google Scholar]
- Renna ME, 2021. A review and novel theoretical model of how negative emotions influence inflammation: the critical role of emotion regulation. Brain, Behav., Immun. - Health 18, 100397. 10.1016/j.bbih.2021.100397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribbe D., 1996. Psychometric review of traumatic event screening instrument for children (TESI-C). [Google Scholar]
- Rudolph KD, Hammen C, Burge D, Lindberg N, Herzberg D, Daley SE, 2000. Toward an interpersonal life-stress model of depression: the developmental context of stress generation. Dev. Psychopathol 12 (2), 215–234. 10.1017/S0954579400002066. [DOI] [PubMed] [Google Scholar]
- Salvador AF, de Lima KA, Kipnis J, 2021. Neuromodulation by the immune system: a focus on cytokines. Nat. Rev. Immunol 21 (8), 526–541. 10.1038/s41577-021-00508-z. [DOI] [PubMed] [Google Scholar]
- Segerstrom SC, Miller GE, 2004. Psychological stress and the human immune system: a meta-analytic study of 30 years of inquiry. Psychol. Bull 130 (4), 601–630. 10.1037/0033-2909.130.4.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel JS, Power JD, Dubis JW, Vogel AC, Church JA, Schlaggar BL, Petersen SE, 2014. Statistical improvements in functional magnetic resonance imaging analyses produced by censoring high-motion data points. Hum. Brain Mapp 35 (5), 1981–1996. 10.1002/hbm.22307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavich GM, Irwin MR, 2014. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychol. Bull 140 (3), 774–815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slopen N, Kubzansky LD, McLaughlin KA, Koenen KC, 2013. Childhood adversity and inflammatory processes in youth: a prospective study. Psychoneuroendocrinology 38 (2), 188–200. 10.1016/j.psyneuen.2012.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TEJ, Johansen-Berg H, Bannister PR, De Luca M, Drobnjak I, Flitney DE, Niazy RK, Saunders J, Vickers J, Zhang Y, De Stefano N, Brady JM, & Matthews PM, 2004. Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage, 23, Supplement 1, S208–S219. doi: 10.1016/j.neuroimage.2004.07.051. [DOI] [PubMed] [Google Scholar]
- Smyser CD, Snyder AZ, Neil JJ, 2011. Functional connectivity MRI in infants: exploration of the functional organization of the developing brain. NeuroImage 56 (3), 1437–1452. 10.1016/j.neuroimage.2011.02.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steptoe A, Hamer M, Chida Y, 2007. The effects of acute psychological stress on circulating inflammatory factors in humans: a review and meta-analysis. Brain Behav. Immun 21 (7), 901–912. 10.1016/j.bbi.2007.03.011. [DOI] [PubMed] [Google Scholar]
- Swartz JR, Prather AA, Hariri AR, 2017. Threat-related amygdala activity is associated with peripheral CRP concentrations in men but not women. Psychoneuroendocrinology 78, 93–96. 10.1016/j.psyneuen.2017.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swartz JR, Carranza AF, Tully LM, Knodt AR, Jiang J, Irwin MR, Hostinar CE, 2021. Associations between peripheral inflammation and resting state functional connectivity in adolescents. Brain Behav. Immun 95, 96–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torre JB, Lieberman MD, 2018. Putting feelings into words: affect labeling as implicit emotion regulation. Emotion Rev. 10 (2), 116–124. 10.1177/1754073917742706. [DOI] [Google Scholar]
- Torrisi SJ, Lieberman MD, Bookheimer SY, Altshuler LL, 2013. Advancing understanding of affect labeling with dynamic causal modeling. NeuroImage 82, 481–488. 10.1016/j.neuroimage.2013.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tustison NJ, Avants BB, Cook PA, Zheng Y, Egan A, Yushkevich PA, Gee JC, 2010. N4ITK: improved N3 bias correction. IEEE Trans. Med. Imaging 29 (6), 1310–1320. 10.1109/TMI.2010.2046908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf RC, Herringa RJ, 2016. Prefrontal-amygdala dysregulation to threat in pediatric posttraumatic stress disorder. Neuropsychopharmacology 41 (3), 822–831. 10.1038/npp.2015.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woolrich MW, Ripley BD, Brady M, Smith SM, 2001. Temporal autocorrelation in univariate linear modeling of FMRI data. NeuroImage 14 (6), 1370–1386. 10.1006/nimg.2001.0931. [DOI] [PubMed] [Google Scholar]
- Yu Q, Daugherty AM, Anderson DM, Nishimura M, Brush D, Hardwick A, Lacey W, Raz S, Ofen N, 2018. Socioeconomic status and hippocampal volume in children and young adults. Dev. Sci 21 (3), e12561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Brady M, Smith S, 2001. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imaging 20 (1), 45–57. 10.1109/42.906424. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


