Abstract
Epithelial cells are one of the prominent cell types infected by human cytomegalovirus (HCMV) within its host. However, many cultured epithelial cells, such as ARPE-19 retinal pigmented epithelial cells, are poorly infected by laboratory-adapted strains in cell culture, and little is known about the viral factors that determine HCMV epithelial cell tropism. In this report, we demonstrate that the UL131 open reading frame (ORF), and likely the entire UL131-128 locus, is required for efficient infection of epithelial cells. Repair of the mutated UL131 gene in the AD169 laboratory strain of HCMV restored its ability to infect both epithelial and endothelial cells while compromising its ability to replicate in fibroblasts. ARPE-19 epithelial cells support replication of the repaired AD169 virus as well as clinical isolates of HCMV. Productive infection of cultured epithelial cells, endothelial cells, and fibroblasts with the repaired AD169 virus leads to extensive membrane fusion and syncytium formation, suggesting that the virus may spread through cell-cell fusion.
Human cytomegalovirus (HCMV) is a widespread pathogen that can cause life-threatening disease in immunologically immature or compromised individuals, including neonates, AIDS patients, allogeneic transplant recipients, and cancer patients undergoing chemotherapy (7). In vivo, a broad range of cell types are infected by HCMV, including epithelial cells, endothelial cells, neuronal cells, smooth muscle cells, fibroblasts, monocytes, and macrophages (34). Epithelial cells play a central role in HCMV spread and persistence in immunocompetent hosts (7). Extensive involvement of other cell types is generally observed in more severe disseminated HCMV infection in immunocompromised patients (7, 20).
Although HCMV can be detected in a variety of cell types in an infected individual, the molecular aspects of its replication have been studied primarily in fibroblasts. The focus on fibroblasts has come from the widespread use of so-called laboratory strains of HCMV. The AD169 (12) and Towne (35) laboratory strains were developed as vaccine candidates by extensive serial passage in cultured fibroblasts. During propagation in fibroblasts, their genomes suffered major rearrangements as well as numerous point mutations (10, 11, 31, 32). As a consequence, laboratory strains replicate efficiently in fibroblasts but poorly in other cell types such as endothelial cells (13, 19, 27, 37, 43). This is in contrast to clinical isolates, which replicate less efficiently in cultured cells but nearly as well in endothelial cells as in fibroblasts (15, 16, 25).
What are the genetic differences among clinical as opposed to laboratory strains of HCMV that govern cell tropism? Viral receptor ligands and fusion machinery glycoproteins, such as gB, gH/gL/gO, and gM/gN, have been proposed as potential tissue and cell tropism determinants (33). However, recent work with endothelial cells has mapped a primary determinant of HCMV cell tropism to the UL131-128 locus (18). Two spliced mRNAs are encoded by this locus, and they contain three open reading frames (ORFs): UL128, UL130, and UL131. A mutation in any one of these three ORFs can abolish endothelial cell tropism (18). Laboratory-adapted strains, which do not efficiently infect endothelial cells, consistently contain mutations in this region. AD169 contains a single nucleotide insertion in UL131 exon 1 (Fig. 1A), and Towne has a frameshift mutation in the carboxy-terminal region of UL130 (1, 32). Neither the Towne nor the AD169 strain can efficiently infect human umbilical vein endothelial cells (HUVECs). Remarkably, three passages of a clinical isolate in fibroblasts can be sufficient to select for mutations in the UL131-128 locus that compromise the ability of the virus to infect endothelial cells (1). The molecular mechanism by which UL131-UL128 controls endothelial cell tropism is unknown.
FIG. 1.
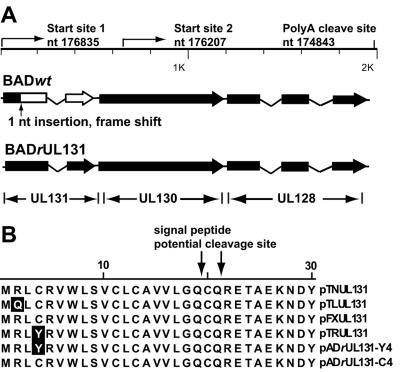
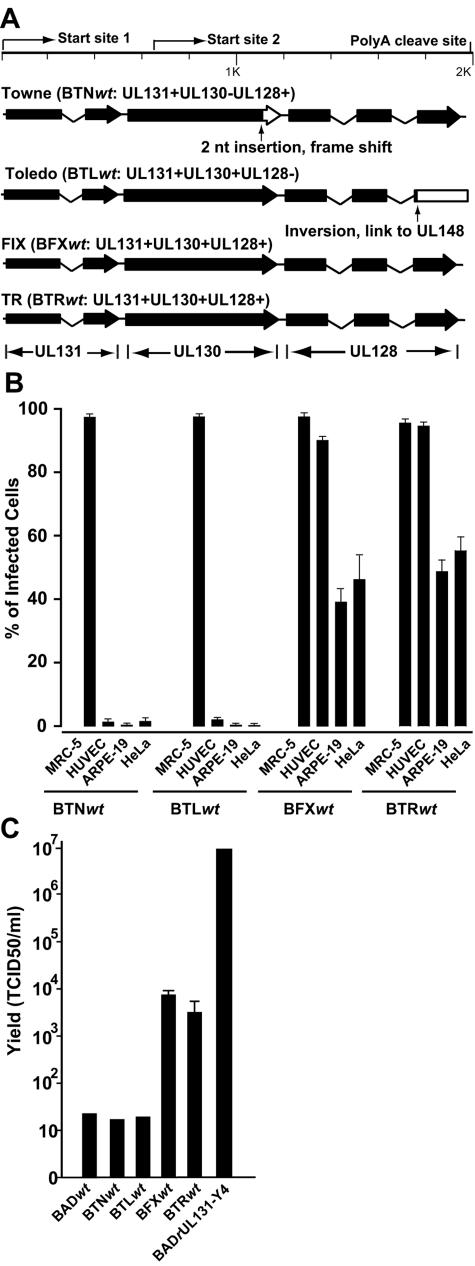
HCMV UL131-128 locus. (A) Diagram of the locus. The positions of transcriptional start sites and poly(A) cleavage sites are indicated. Solid boxes represent the sequence of wild-type ORFs. The location of the point mutation in the AD169 UL131 gene is indicated, and the portion of UL131 that is not expressed is designated by an open box. (B) Amino acid sequence of the N-terminal domain of UL131 in two repaired AD169 derivatives, BADrUL131-Y4 and BADrUL131-C4, and HCMV variant strains. The likely signal peptide cleavage sites are indicated.
In this report we have investigated the molecular basis for epithelial cell tropism. Glandular and ductal epithelial cells in a number of tissues, including salivary gland, kidney, breast milk duct, and genital organs, are thought to be major sites of virus production (7, 20), enabling infectious virus to be shed into breast milk, saliva, urine, cervical secretions, and semen. Salivary gland ductal epithelial cells and renal tubular epithelial cells can be persistently infected in immunocompetent populations (20), allowing virus to be shed into urine and saliva for years even in the absence of overt HCMV disease. In immunocompromised patients, epithelial cells of many other tissues with cytopathology typical of CMV infection have been described (20). These include retinal pigmented epithelium, lung alveolar and bronchial epithelium, bile duct epithelium, lower intestinal mucous epithelium, pancreatic ductal epithelium, and inner ear columnar epithelium. Disruption of tissue epithelia, either directly by HCMV-induced cytopathology or by inflammatory cell-mediated immune responses, contributes greatly to the pathology of HCMV infection.
Despite the frequent and extensive infection of epithelial cell populations by HCMV in vivo, only a few cultured epithelial cells have been reported to be susceptible to HCMV infection, including primary lung epithelial cells (30), thyroid epithelial cells (26), HCMC and Caco-2 epithelial cells derived from colon (24, 40), cervical epithelial cells (41), and BAMB cells derived from amniotic fluid cells (42). However, most analyses of HCMV epithelial cell tropism have been performed using laboratory-adapted strains, and potential variations among virus strains were not taken into consideration. As a case in point, clinical isolates of HCMV are competent to infect retinal pigmented epithelial cells (29), while laboratory strains infect related cells at extremely low efficiencies (21). As has been shown for endothelial cell tropism (18), the inability to infect retinal pigmented epithelial cells must result from one or more mutations in the laboratory strain.
We have systematically investigated HCMV infection of epithelial cells by using a panel of cultured cells that originated from different organs and tissues. We found that cultured epithelial cells, like endothelial cells, can be efficiently infected by HCMV strains with a wild-type UL131-UL128 locus. The AD169 laboratory strain can efficiently infect both epithelial and endothelial cells when the mutation in its UL131 ORF is repaired.
MATERIALS AND METHODS
Cells and viruses.
Primary human foreskin fibroblasts (HFFs) at passage 10 to 15 were maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% newborn calf serum. Immortalized HUVECs were a generous gift of D. Gabuzda (Dana-Farber Cancer Institute, Boston, MA) and A. Moses (Oregon Health and Sciences University, Portland, OR). Lung macrovascular endothelial cells (LMVECs) were a generous gift of H. Zhu (University of Medicine and Dentistry of New Jersey-New Jersey Medical School, Newark, NJ). Both types of endothelial cells were cultured in EGM-2 medium (Cambrex, East Rutherford, NJ). HeLa cells, primary human MRC-5 embryonic lung fibroblasts, and ARPE-19 retinal pigmented epithelial cells were purchased from the American Type Culture Collection. Both MRC-5 cells and ARPE-19 cells were used at passage 24 to 30. The MRC-5 cells were cultured in DMEM supplemented with 10% fetal bovine medium, and ARPE-19 cells were propagated in DMEM/Ham's F12 (1:1) medium containing 10% of fetal bovine serum. SW480, HCT116, H1299, MCF-7, SK-N-SH, SK-N-AS, and IMR-32 cells were generous gifts of M. Cole (Dartmouth University, Hanover, NH) and were propagated in DMEM supplemented with 10% fetal bovine serum.
A derivative of the AD169 strain of HCMV (45) with a green fluorescent protein (GFP) marker (BADwt) was used as the parental AD169 virus in this study. In this variant, the UL21.5 region of the virus was replaced with a marker cassette containing the GFP coding region under control of a simian virus 40 promoter followed by an internal ribosome entry site and a puromycin resistance gene (44). To repair the AD169 UL131 coding sequence, linear recombination (6) was employed to substitute the HCMV sequence from base pairs 176685 to 176794 (sequence numbers according to reference 9) with a marker cassette containing kanamycin resistance and lacZ genes in an infectious bacterial artificial chromosome clone carrying the BADwt genome. This clone was then transformed into Escherichia coli GS500, and allelic exchange was performed with a pGS284 (38) derivative carrying the UL131 ORF and flanking sequences from the HCMV clinical strain TR (39). To determine the recombination sites between the bacterial artificial chromosome clone and the shuttle plasmid, the UL131-UL128 locus were sequenced. Two repaired UL131 bacterial artificial chromosome clones with different N-terminal protein sequences were generated by allelic exchange (Fig. 1). The viruses recovered from the repaired clones were designated BADrUL131-Y4 and BADrUL131-C4.
Wild-type virus stocks were prepared from bacterial artificial chromosome clones of HCMV laboratory strains and clinical isolates Towne (BTNwt) (28), Toledo (BLTwt) (32), VR1814-FIX (BFXwt) (18), and TR (BTRwt) (32). BTNwt lacks US1 to US12, BLTwt lacks US2 to US11, BFXwt lacks IRS1 to US6, and BTRwt lacks US2 to US5 (32) as a consequence of bacterial artificial chromosome cloning. Viruses were reconstituted by electroporation of bacterial artificial chromosome DNA into HFFs, together with a plasmid expressing the viral UL82-coded pp71 protein, which enhances the infectivity of HCMV DNA (2). Virus stocks were prepared in HFF cells, and the first passage of each virus preparation was used for cell tropism studies.
Assays for virus infection and replication.
To monitor the efficiency of infection, epithelial cells, endothelial cells, or fibroblasts were transferred into 12-well plates and incubated overnight to produce subconfluent cultures. Monolayers were washed once with serum-free RPMI 1640 and infected with virus diluted in the same medium at a multiplicity of 1 PFU/cell. During the adsorption period, the plates were first subjected to centrifugation at 25°C for 30 min at 1,000 × g and then incubated at 37°C for 1 h. The inoculum was removed and fresh medium containing the serum appropriate to the cell type was added. At 48 h after infection, cultures were fixed in 2% paraformaldehyde and permeabilized with 0.1% Triton X-100, and infected cells were identified by GFP expression and IE1 was identified by immunofluorescence using monoclonal antibody 1B10 and Alexa 546-conjugated secondary antibody. The nuclei of cells were stained with 4′,6-diamidino-2-phenylindole (DAPI). Total cell numbers were determined by the number of DAPI-stained nuclei, and efficiencies of infection were calculated as the percentage of GFP- and IE1-expressing cells in the total cell populations.
For analysis of virus growth kinetics, cells were infected at a multiplicity of 0.01 or 1 PFU/cell with BADwt (laboratory strain parent) or BADrUL131 virus (repaired UL131 ORF), with the exception of HeLa cells, which were only infected at a multiplicity of infection of 1 PFU/cell because HeLa cell cultures could not be maintained for the extended period of time needed for analysis of a low-multiplicity infection. At various times after infection, cell-free virus was collected by harvesting medium from infected cultures and cell-associated virus was collected by three freeze-thaw cycles of infected cells in medium three times. Virus titers were determined by the 50% tissue culture infectious dose (TCID50) assay on MRC-5 cells. In contrast to parental AD169, the UL131-repaired viruses induced syncytia and exhibited reduced plaque-forming efficiencies. Therefore, to compare the growth of the mutants and their wild-type parent, we relied on GFP gene expression rather than cytopathic effect to identify the infected wells in our TCID50 assays. The use of the GFP marker carried on the viruses significantly increased the sensitivity and accuracy of our assays.
RESULTS
Construction and characterization of an AD169 variant with a repaired UL131 ORF.
Two spliced transcripts with a common poly(A) addition site were shown to be generated by the UL131 to UL128 locus (1, 18), but their 5′ ends were not localized. To further characterize the transcripts produced by this locus, we mapped the two start sites (Fig. 1A) by using 5′ rapid amplification of cDNA ends (RACE) analysis, and we confirmed the result using RNase protection assays (data not shown). The 5′ ends are located at sequence positions 176835, just upstream of UL131, and 176207, within UL130 (numbering according to reference 9). The mRNA with the 5′ end upstream of the UL131 coding region has the potential to encode polypeptides encoded by all three ORFs, whereas the RNA whose 5′ end maps within UL130 has the potential to encode a portion of UL130 and UL128. This 5′ mapping confirms that the 1-base-pair insertion present in the UL131 ORF of AD169 (Fig. 1A) is, indeed, present within an mRNA that has been mapped to this locus.
The frame-shifted UL131 ORF in AD169 was repaired by constructing derivatives of the laboratory strain (BADrUL131) in which the mutated UL131 ORF was replaced with a wild-type UL131 ORF derived from the HCMV TR clinical strain (Fig. 1A). Two different viruses with a repaired UL131 ORF were generated, due to recombination events at two different sites between the bacterial artificial chromosome clone and the shuttle plasmid. The sequences of the UL131 ORFs from the newly generated viruses were compared with UL131 ORFs from the Towne, Toledo, FIX, and TR strains (Fig. 1B). Except for amino acid positions 2 and 4, the sequences of the UL131 ORFs from different clinical HCMV strains are identical. The UL131 sequence of BADrUL131-Y4 derives entirely from the TR strain, while the BADrUL131-C4 sequence is identical to that of the FIX and Toledo strains (Fig. 1B).
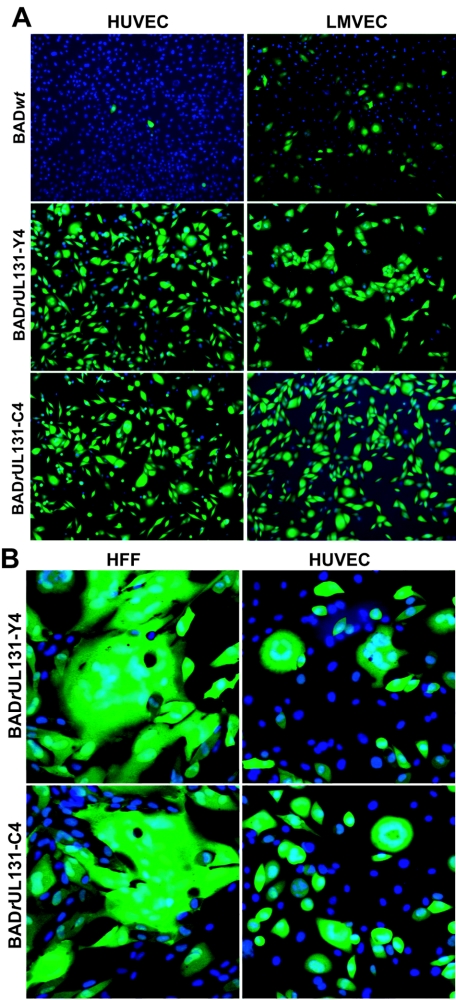
Hahn et al. (18) have shown that a UL131 deletion mutant of the clinical isolate FIX, a bacterial artificial chromosome-cloned derivative of VR1814 (17), is no longer able to replicate in endothelial cells. To confirm that endothelial cell tropism was restored by repairing the UL131 gene in AD169, two endothelial cell lines, HUVECs and LMVECs, were infected at multiplicity of 1 PFU/cell, and the cultures were assayed 48 h later for expression of the GFP marker carried by the viruses. Very few (<1%) GFP-expressing HUVECs were seen in BADwt-infected cultures; in contrast, GFP could be detected in almost all the HUVECs infected by both BADrUL131 viruses (Fig. 2A), demonstrating that the repaired AD169 variants had reacquired the ability to infect endothelial cells. LMVECs were more efficiently infected by BADwt (12%), but the efficiency of infection was considerably greater for BADrUL131-Y4 (96%) and BADrUL131-C4 (98%) (Fig. 2A). In contrast to the UL131-deficient parent, the repaired BADrUL131 viruses induced syncytia in infected cultures of both fibroblasts and endothelial cells (Fig. 2B).
FIG. 2.
Characterization of endothelial cell and fibroblast infection of UL131-repaired AD169 derivatives. (A) BADwt, BADrUL131-Y4, and BADrUL131-C4 infection of endothelial cells. HUVECs or human LMVECs were infected at a multiplicity of 1 PFU/cell, and cells were fixed 48 h after infection. Nuclei were stained with DAPI (blue), and infected cells were identified by GFP expression (green) from a marker cassette on the viruses. (B) Syncytium formation induced by BADrUL131 viruses. MRC-5 cells or HUVEC cells were infected at a multiplicity of 0.1 PFU/cell; 5 days after infection, the cells were fixed with 2% paraformaldehyde, the nuclei of the cells were stained with DAPI (blue), and GFP expression (green) demonstrated that fused cells were infected.
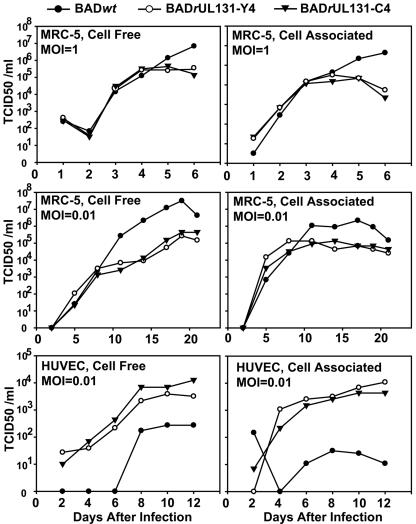
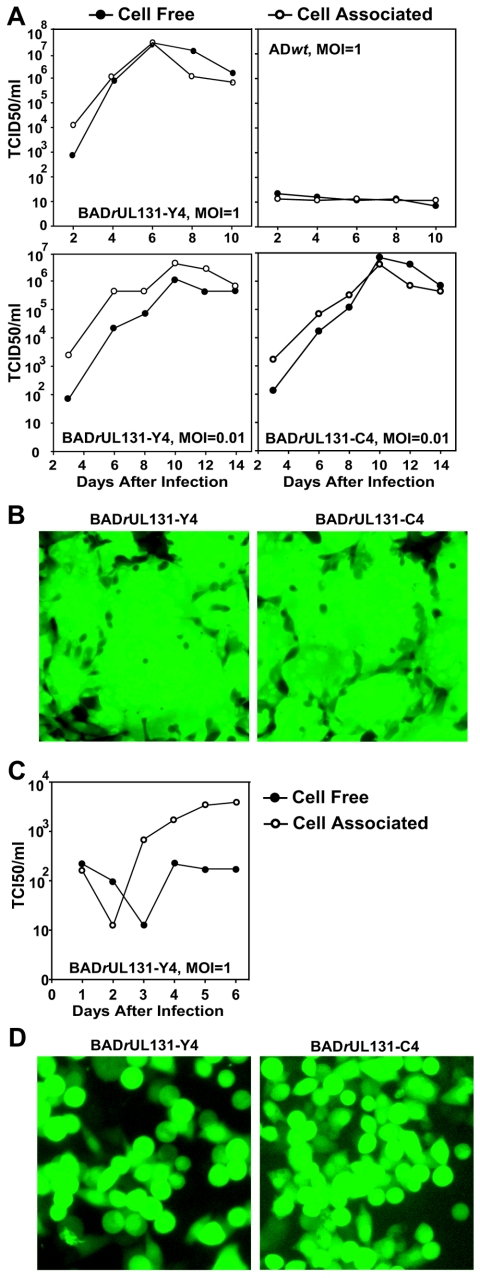
To evaluate the growth characteristics of the repaired viruses, MRC-5 fibroblasts and HUVECs were infected with BADwt or BADrUL131 variants, and the production of cell-free and cell-associated virus in single-step growth or multistep growth analysis was assayed (Fig. 3). The repaired viruses grew more poorly than their AD169 parent in fibroblasts, generating a 10- to 100-fold-reduced yield of both cell-free and cell-associated virus. The reduced yields correlated with extensive cell-cell fusion induced by the repaired viruses, although it is not clear that cell fusion compromises the efficiency of virus replication in fibroblasts. In contrast to fibroblasts, the repaired viruses grew more efficiently in endothelial cells than their laboratory strain parent, producing about 20-fold more cell-free and 300-fold more cell-associated virus.
FIG. 3.
Growth kinetics of UL131-repaired AD169 derivatives in MRC-5 cells and HUVEC cells. Cells were infected with BADwt (•), BADrUL131-Y4 (○), or BADrUL131-C4 (▾) at a multiplicity of 1 or 0.01 PFU/cell. Cell-free and cell-associated virus was collected at the indicated times after infection, and infectious virus was quantified by TCID50 assay on MRC-5 cells.
Collectively, our data demonstrate that repair of the mutated UL131 gene in AD169 compromises virus replication in fibroblasts and facilitates growth in endothelial cells.
UL128-131 locus-dependent infection of epithelial cells.
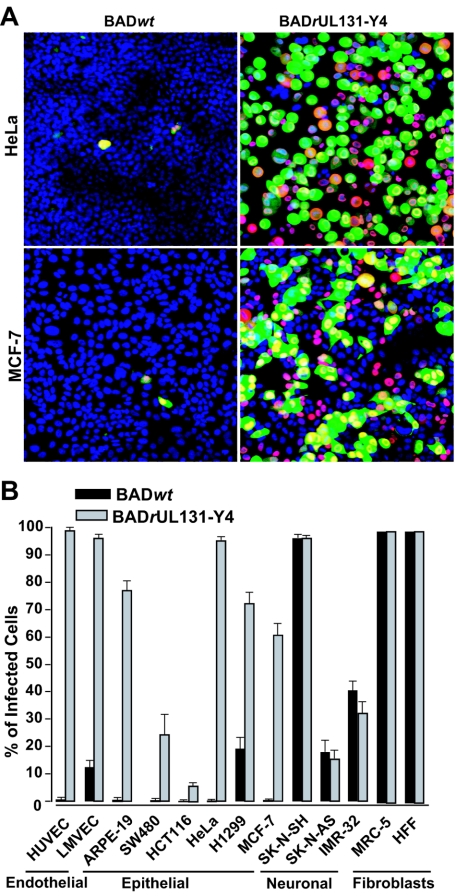
To test the possibility that UL131 is also important for HCMV replication in epithelial cells, we examined the susceptibility of a panel of epithelial cell lines to BADwt and BADrUL131-Y4 (Fig. 4). Susceptibility to infection was assayed by monitoring expression of the UL123-coded IE1 protein and expression of the GFP marker genes carried by the two viruses. The epithelial cell lines originated from a variety of tissues: retina (ARPE-19), cervix (HeLa), colon (SW480 and HCT116), lung (H1299), and breast (MCF-7). As shown in Fig. 4A, HeLa and MCF-7 cells could be efficiently infected by BADrUL131, and the infection was highly dependent on UL131 since a very low incidence of infection was seen for the AD169 parent, BADwt. Cell-cell fusion in the MCF-7 epithelial cells, but not in HeLa cell cultures, was seen after infection with BADrUL131-Y4 (Fig. 4A, lower panels). This experiment also demonstrated that GFP expression can lead to an underestimate of the percentage of cells that are infected, because the HCMV UL123-coded IE1 protein was detected in a greater proportion of cells (Fig. 4A, compare IE1-red to GFP-green signals). Presumably this reflects differences in the activity of the simian virus 40 early promoter, which controls the expression of GFP in these viruses, relative to the HCMV immediate-early promoter.
FIG. 4.
Cell tropism of a UL131-rapaired AD169 derivative. (A) Identification of infected HeLa and MCF-7 cells by fluorescence. Cells were infected with BADrUL131-Y4 at a multiplicity of 1 PFU/cell and fixed 48 h after infection. Nuclei were stained with DAPI (blue), and infected cells were identified by IE1 antigen staining (red) and GFP expression (green) from a marker cassette on the viruses. (B) Quantification of UL131-UL128 locus-dependent host cell tropism. Cultures of epithelial cells, endothelial cells, and fibroblasts were infected at a multiplicity of 1 PFU/cell with BADwt (black bars) or BADrUL131-Y4 (gray bars); 48 h later, cells were fixed with 2% paraformaldehyde, and infected cells were identified by monitoring GFP expression.
To quantify the relative susceptibilities of the epithelial cell lines, we calculated the percentage of cells expressing GFP from the viral genome in different epithelial cell populations after infection at identical input multiplicities with the parental virus or repaired virus (Fig. 4B). A repaired UL131 ORF was absolutely required to infect ARPE-19, HeLa, SW480, HCT116, and MCF-7 epithelial cells. The repaired ORF was not essential but dramatically enhanced the efficiency of infection in H1299 cells. Fibroblasts and endothelial cells were included as controls. Although no detectable differences in susceptibility to infection with BADwt versus BADrUL131-Y4 were evident in MRC-5 or HFF fibroblasts, GFP expression in HUVEC and LMVEC endothelial cells was dramatically enhanced. Finally, several neuron-derived cell lines were tested. The SK-N-AS, SK-N-SH, and IMR-32 neuroblastoma cell lines showed variable susceptibilities to infection, but GFP expression was not UL131 gene dependent.
The production of infectious progeny by the BADrUL131 repaired viruses was examined in ARPE-19 cells. The epithelial cells were infected at a multiplicity of 0.01 or 1 PFU/cell and the production of cell-associated and cell-free virus was monitored by TCID50 assay on fibroblasts (Fig. 5A). In epithelial cells, BADrUL131-Y4 and BADrUL131-C4 produced a ≥10-fold-greater yield than in fibroblasts and a ≈100-fold greater yield than in endothelial cells (compare Fig. 5A and Fig. 3). Further, BADrUL131 induced extensive cell-cell fusion in epithelial cell cultures (Fig. 5B), as it did in fibroblasts and endothelial cells (Fig. 2B).
FIG.5.
Infection of ARPE-19 and HeLa epithelial cells by UL131-repaired AD169 derivatives. (A) Growth kinetics of BADwt, BADrUL131-Y4, and BADrUL131-C4 in ARPE-19 cells. Cells were infected at a multiplicity of 1 or 0.01 PFU/cell, cell-free (•) and cell-associated (○) viruses were collected at the indicated times, and infectious virus was quantified by TCID50 assay on MRC-5 cells. (B) Induction of cell-cell fusion by BADrUL131 viruses infection in ARPE-19 cells. ARPE-19 cells were infected at a multiplicity of 0.1 PFU/cell, and cultures were observed 3 or 7 days later by phase contrast and fluorescent microscopy. (C) Growth kinetics of BADrUL131-Y4 in HeLa cells. HeLa cells were infected at a multiplicity of 0.01 PFU/cell, cell-free (•) and cell-associated (○) virus was collected at the indicated times, and infectious virus was quantified by TCID50 assay on MRC-5 cells. (D) Cytopathic effect of BADrUL131-Y4 and BADrUL131-C4 in HeLa cells. HeLa cells were infected at a multiplicity of 1 PFU/cell, and cells were observed 3 or 7 days later by fluorescent microscopy.
The replication of the two BADrUL131 repaired viruses was also monitored in HeLa cells, an epithelial tumor cell line. Infection of HeLa cells generated a yield of cell-free virus that was reduced by a factor of ≈1,000 in comparison to the yield from APRE-19 cells (Fig. 5C). It is interesting that 98% of HeLa cells and 77% of ARPE-19 cells expressed the GFP marker after infection at a multiplicity, determined by TCID50 assay on fibroblasts, of 1 PFU/cell with BADrUL131-Y4 (Fig. 4B). In spite of the higher efficiency with which the marker was expressed in HeLa cells, the yield of infectious progeny was greater in ARPE-19 cells. Apparently, there is a block to efficient replication of HCMV in HeLa cells, relative to the efficiency of replication in ARPE-19 cells, which occurs after the genome reaches the nucleus and expresses the marker gene. In contrast to ARPE-19 cells, HeLa cells fail to undergo fusion after infection with BADrUL131 viruses (Fig. 4A and 5D), although significant cytopathic effects with characteristic cell rounding can be detected as early as 24 h after infection. This might reflect a failure of the virus to efficiently advance to the late stage of the replication cycle. This would inhibit expression of the UL131 ORF, which is expressed during the late phase (8).
A mutation in UL131 is responsible for the inability of AD169 to replicate efficiently in epithelial cells. To further explore the role of the UL131-128 locus in HCMV epithelial cell tropism, we assayed for infection by four additional HCMV strains: Towne, Toledo, FIX, and TR. UL130 is mutated in Towne and UL128 is disrupted in Toledo, whereas FIX and TR contain wild-type UL128, -130, and -131 ORFs (Fig. 6A). All of the virus strains infected the MRC-5 fibroblasts and expressed their IE1 protein equally well, but only TR and FIX efficiently infected HUVEC endothelial or ARPE-19 and HeLa epithelial cells (Fig. 6B). This experiment demonstrates that the Towne and Toledo strains do not efficiently enter epithelial cells and express their IE1 genes, and raises the possibility that the defect might result from mutations in the UL130 and UL128 ORFs, as has been shown for replication in endothelial cells (18).
FIG.6.
Cell tropism of HCMV strains. (A) Diagram of the UL131-UL128 locus in the Towne, Toledo, FIX, and TR strains of HCMV. The positions of transcriptional start sites and poly(A) cleavage sitesare indicated. Solid boxes represent wild-type ORFs. The point mutation in the Towne UL130 gene and the deletion in the Toledo UL128 gene are shown, and the positions of open reading frames whose expression is disrupted are represented by open boxes. (B) Entry of HCMV strains into MRC-5, HUVEC, HeLa, and ARPE-19 cells. Cells were infected at a multiplicity of 1 PFU/cell, and 48 h later, the cultures were fixed with 2% paraformaldehyde, and infected cells were identified by IE1 expression. (C) Production of infectious progeny by HCMV strains in ARPE-19 cells. Cells were infected at a multiplicity of 1 PFU/cell. Six days (BADrUL131-Y4) or 12 days (all other viruses) later, the cells and media were collected together, samples were frozen and thawed three times, and virus titers were determined by TCID50 assay on MRC-5 cells.
Finally, the yields of infectious progeny were determined after infection of ARPE-19 epithelial cells with the set of HCMV strains (Fig. 6C). The repaired AD169 strain BADrUL131-Y4 produced the highest yield in epithelial cells (107 PFU/ml). AD169, Towne, and Toledo, each of which contain a defect in the UL131-128 locus, generated little infectious progeny (≈1.5 × 101 PFU/ml), consistent with their inability to efficiently express their IE1 genes in the epithelial cells. FIX and TR, which contain wild-type UL131-128 loci, generated intermediate yields (7 × 103 and 3 × 103 PFU/ml, respectively).
DISCUSSION
The UL131-128 locus has been shown previously to be essential for efficient replication of HCMV in endothelial cells (18). Deletion of UL128, UL130, or UL131 from the FIX clinical isolate of HCMV blocked replication in HUVECs. The AD169 strain of HCMV carries a defective UL131 ORF (Fig. 1A), and we generated repaired derivatives of AD169, termed BADrUL131-Y4 and BADrUL131-C4 (Fig. 1B). The two repaired viruses behaved identically in all of our assays. As expected, they were able to enter (Fig. 2A) and produce infectious progeny (Fig. 3) in endothelial cells. They also entered (Fig. 4) and replicated (Fig. 5) to generate progeny virus in epithelial cells, in contrast to their UL131-deficient AD169 parent. We conclude that UL131 function is required for HCMV replication not only in endothelial cells but also in epithelial cells.
The Towne laboratory strain, which has been extensively passaged in fibroblasts, and the Toledo strain, which has been passaged to a more limited extent in fibroblasts, failed to efficiently infect epithelial cells (Fig. 6B) or produce progeny in epithelial cells (Fig. 6C). Since Towne carries a mutation in the UL130 ORF and Toledo contains a mutated UL128 ORF (Fig. 6A), it is likely that these two ORFs, in addition to UL131, are important for infection of epithelial cells, as has been shown previously for endothelial cells (18).
The mechanism by which the UL131-128 locus contributes to HCMV infection of cells other than fibroblasts is unknown. However, it is clear that the defect exhibited by HCMV strains with mutations in these ORFs occurs early in the replication cycle. For example, the UL131-deficient AD169 variant BADwt failed to express either the viral IE1 protein or a GFP marker protein in endothelial (Fig. 1) or epithelial (Fig. 3) cells. Perhaps the products of the UL131-128 locus are needed for HCMV to efficiently contact or enter epithelial and endothelial cells. It is possible that viruses lacking a product of this locus enter these cells via a route that causes an abortive viral infection. Bodaghi et al. (4) reported that AD169 enters ARPE-19 cells via receptor-mediated endocytosis instead of the normal mode of entry documented for fibroblasts, which involves fusion of the virion envelope with the plasma membrane. Alternatively, viruses lacking a product of the UL131-128 locus might enter endothelial and epithelial cells normally but fail to efficiently execute a postentry step. For example, the virus might not fully disassemble the capsid-tegument complex within the cytoplasm (36) and, as a consequence, fail to deliver its genome to the nucleus. We favor the view that the UL131-128 locus facilitates the initial interaction of the virion with the surface of epithelial cells, since the block to AD169 infection of ARPE-19 cells can be relieved by neuraminidase treatment of the cells in combination with centrifugal enhancement (21).
Cell-specific effects downstream of entry also influence the ability of HCMV to produce a yield in epithelial cells. The UL131-repaired AD169 derivative efficiently entered and expressed a marker gene in both ARPE-19 and HeLa cells (Fig. 4B), but it generates 1,000-fold more infectious progeny in ARPE-19 cells (Fig. 5A) than in HeLa cells (Fig. 5C). This observation is consistent with earlier work suggesting that HCMV generally replicates more efficiently in well-differentiated diploid cells than in anuploid cells (40). Downstream virus-specific effects also influence virus yields in epithelial cells. The UL131-repaired AD169 derivative and clinical isolates efficiently entered and expressed IE1 or a marker gene in ARPE-19 cells, but the AD169 derivative produced a significantly greater yield of infectious progeny than did the FIX and TR clinical isolates (Fig. 5A and 6C). Apparently, some of the changes that occurred in AD169 during its sequential passage in fibroblasts can facilitate efficient replication in other cell types if it is able to enter them.
The rescue of epithelial and endothelial cell tropism by repairing the UL131 gene in AD169 concurrently induced a syncytial phenotype. Cell-cell fusion was evident in fibroblasts, endothelial cells, and epithelial cells infected with BADrUL131 viruses (Fig. 2B, 4A, and 5B). Fusion was not observed with the parental UL131-deficient AD169 virus (Fig. 2A and data not shown). Several reports have demonstrated syncytium formation by HCMV clinical isolates in fibroblasts and epithelial cells (5, 30, 42), and the phenotype of the repaired AD169 derivative indicates that the UL131 product is needed for cell-cell fusion. The formation of syncytia would mediate cell-to-cell spread of HCMV while allowing it to evade neutralizing antibodies.
A variety of herpesviruses can form syncytia in certain cell types, including some well-characterized syn variants of herpes simplex virus type 1. However, the syncytial phenotype of HCMV is associated with viruses that contain a wild-type UL131 gene, whereas the phenotype is more commonly seen in mutants of herpes simplex virus, such as UL20 (3), UL24 (23), UL27-gB (14), and UL53-gK (22) mutants. Expression of the UL131 to UL128 ORFs alone did not result in syncytium formation (data not shown), indicating that other viral proteins are likely involved. Perhaps the UL131-128 products function in cooperation with one or more virus-coded fusogenic glycoproteins, such as gB, gH-gL-gO, or gM-gN.
The repaired viruses, BADrUL131-Y4 and -C4, grew more poorly in fibroblasts than their UL131-deficient AD169 parent, producing a 10- to 100-fold-reduced yield (Fig. 2). Hahn et al. (18) compared the replication of an HCMV clinical isolate (FIX) and a derivative lacking UL131 and found that the two viruses grew equally well. The AD169 and FIX genomes differ at numerous locations (32), and the different genetic backgrounds of the laboratory and clinical viruses apparently influence their response to the presence or absence of UL131. The relatively poor replication of BADrUL131 helps to explain why the UL131-128 locus is mutated in laboratory strains of HCMV and why clinical isolates accumulate mutations in this locus when they are serially passaged in fibroblasts.
It is not clear why an intact UL131 ORF interferes with the production of infectious AD169 progeny in fibroblasts. However, this observation serves to highlight a major problem in the production of stocks of HCMV clinical isolates in the laboratory. If a clinical isolate is passaged in fibroblasts, it can very rapidly accumulate a mutation in the UL131-128 locus, precluding its ability to infect other cell types (17). Indeed, as few as three passages in fibroblasts can be sufficient for such mutations to appear in virus stocks (1). Since clinical isolates, but not laboratory strains, efficiently infect and generate infectious progeny in epithelial cells (Fig. 6C), this cell type, like endothelial cells, holds promise as a laboratory host for the production of clinical HCMV stocks that are not selected for mutations in the UL131-UL128 locus.
Acknowledgments
We thank D. Gabuzda and A. Moses for generously providing the immortalized HUVEC cells, H. Zhu for providing the LMVEC cells, M. Cole for providing SW480, HCT116, H1299, MCF-7, SK-N-SH, SK-N-AS, and IMR-32 cells, J. Schroer for help with microscopy, and D. Brown for critical reading of the manuscript.
This work was supported by Public Health Service grants CA82396, CA085786, AI54430, and GM071508.
REFERENCES
- 1.Akter, P., C. Cunningham, B. P. McSharry, A. Dolan, C. Addison, D. J. Dargan, A. F. Hassan-Walker, V. C. Emery, P. D. Griffiths, G. W. Wilkinson, and A. J. Davison. 2003. Two novel spliced genes in human cytomegalovirus. J. Gen. Virol. 84:1117-1122. [DOI] [PubMed] [Google Scholar]
- 2.Bacsi, A., J. Aranyosi, Z. Beck, P. Ebbesen, I. Andirko, J. Szabo, L. Lampe, J. Kiss, L. Gergely, and F. D. Toth. 1999. Placental macrophage contact potentiates the complete replicative cycle of human cytomegalovirus in syncytiotrophoblast cells: role of interleukin-8 and transforming growth factor-beta1. J. Interferon Cytokine Res. 19:1153-1160. [DOI] [PubMed] [Google Scholar]
- 3.Baines, J. D., P. L. Ward, G. Campadelli-Fiume, and B. Roizman. 1991. The UL20 gene of herpes simplex virus 1 encodes a function necessary for viral egress. J. Virol. 65:6414-6424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bodaghi, B., M. E. Slobbe-van Drunen, A. Topilko, E. Perret, R. C. Vossen, M. C. van Dam-Mieras, D. Zipeto, J. L. Virelizier, P. LeHoang, C. A. Bruggeman, and S. Michelson. 1999. Entry of human cytomegalovirus into retinal pigment epithelial and endothelial cells by endocytosis. Investig. Ophthalmol. Vis. Sci. 40:2598-2607. [PubMed] [Google Scholar]
- 5.Booth, J. C., J. E. Beesley, and H. Stern. 1978. Syncytium formation caused by human cytomegalovirus in human embryonic lung fibroblasts. Arch. Virol. 57:143-152. [DOI] [PubMed] [Google Scholar]
- 6.Borst, E. M., S. Mathys, M. Wagner, W. Muranyi, and M. Messerle. 2001. Genetic evidence of an essential role for cytomegalovirus small capsid protein in viral growth. J. Virol. 75:1450-1458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Britt, W. J., and C. A. Alford. 1996. Cytomegalovirus, p. 2493-2523. In B. N. Fields, D. M. Knipe, and P. M. Howley (ed.), Fields virology, 3rd ed. Lippincott-Raven, Philadelphia, Pa.
- 8.Chambers, J., A. Angulo, D. Amaratunga, H. Guo, Y. Jiang, J. S. Wan, A. Bittner, K. Frueh, M. R. Jackson, P. A. Peterson, M. G. Erlander, and P. Ghazal. 1999. DNA microarrays of the complex human cytomegalovirus genome: profiling kinetic class with drug sensitivity of viral gene expression. J. Virol. 73:5757-5766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chee, M. S., A. T. Bankier, S. Beck, R. Bohni, C. M. Brown, R. Cerny, T. Horsnell, C. A. Hutchison 3rd, T. Kouzarides, J. A. Martignetti, et al. 1990. Analysis of the protein-coding content of the sequence of human cytomegalovirus strain AD169. Curr. Top. Microbiol. Immunol. 154:125-169. [DOI] [PubMed] [Google Scholar]
- 10.Dargan, D. J., F. E. Jamieson, J. MacLean, A. Dolan, C. Addison, and D. J. McGeoch. 1997. The published DNA sequence of human cytomegalovirus strain AD169 lacks 929 base pairs affecting genes UL42 and UL43. J. Virol. 71:9833-9836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dolan, A., C. Cunningham, R. D. Hector, A. F. Hassan-Walker, L. Lee, C. Addison, D. J. Dargan, D. J. McGeoch, D. Gatherer, V. C. Emery, P. D. Griffiths, C. Sinzger, B. P. McSharry, G. W. Wilkinson, and A. J. Davison. 2004. Genetic content of wild-type human cytomegalovirus. J. Gen. Virol. 85:1301-1312. [DOI] [PubMed] [Google Scholar]
- 12.Elek, S. D., and H. Stern. 1974. Development of a vaccine against mental retardation caused by cytomegalovirus infection in utero. Lancet i:1-5. [DOI] [PubMed] [Google Scholar]
- 13.Friedman, H. M., E. J. Macarak, R. R. MacGregor, J. Wolfe, and N. A. Kefalides. 1981. Virus infection of endothelial cells. J. Infect. Dis. 143:266-273. [DOI] [PubMed] [Google Scholar]
- 14.Gage, P. J., M. Levine, and J. C. Glorioso. 1993. Syncytium-inducing mutations localize to two discrete regions within the cytoplasmic domain of herpes simplex virus type 1 glycoprotein B. J. Virol. 67:2191-2201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gerna, G., E. Percivalle, F. Baldanti, and M. G. Revello. 2002. Lack of transmission to polymorphonuclear leukocytes and human umbilical vein endothelial cells as a marker of attenuation of human cytomegalovirus. J. Med. Virol. 66:335-339. [DOI] [PubMed] [Google Scholar]
- 16.Gerna, G., E. Percivalle, A. Sarasini, and M. G. Revello. 2002. Human cytomegalovirus and human umbilical vein endothelial cells: restriction of primary isolation to blood samples and susceptibilities of clinical isolates from other sources to adaptation. J. Clin. Microbiol. 40:233-238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Grazia Revello, M., F. Baldanti, E. Percivalle, A. Sarasini, L. De-Giuli, E. Genini, D. Lilleri, N. Labo, and G. Gerna. 2001. In vitro selection of human cytomegalovirus variants unable to transfer virus and virus products from infected cells to polymorphonuclear leukocytes and to grow in endothelial cells. J. Gen. Virol. 82:1429-1438. [DOI] [PubMed] [Google Scholar]
- 18.Hahn, G., M. G. Revello, M. Patrone, E. Percivalle, G. Campanini, A. Sarasini, M. Wagner, A. Gallina, G. Milanesi, U. Koszinowski, F. Baldanti, and G. Gerna. 2004. Human cytomegalovirus UL131-128 genes are indispensable for virus growth in endothelial cells and virus transfer to leukocytes. J. Virol. 78:10023-10033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ho, D. D., T. R. Rota, C. A. Andrews, and M. S. Hirsch. 1984. Replication of human cytomegalovirus in endothelial cells. J. Infect. Dis. 150:956-957. [DOI] [PubMed] [Google Scholar]
- 20.Ho, M. 1991. Cytomegalovirus biology and infection. Plenum Press, New York, N.Y.
- 21.Huber, M. T., R. Tomazin, T. Wisner, J. Boname, and D. C. Johnson. 2002. Human cytomegalovirus US7, US8, US9, and US10 are cytoplasmic glycoproteins, not found at cell surfaces, and US9 does not mediate cell-to-cell spread. J. Virol. 76:5748-5758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hutchinson, L., K. Goldsmith, D. Snoddy, H. Ghosh, F. L. Graham, and D. C. Johnson. 1992. Identification and characterization of a novel herpes simplex virus glycoprotein, gK, involved in cell fusion. J. Virol. 66:5603-5609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jacobson, J. G., S. L. Martin, and D. M. Coen. 1989. A conserved open reading frame that overlaps the herpes simplex virus thymidine kinase gene is important for viral growth in cell culture. J. Virol. 63:1839-1843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jarvis, M. A., C. E. Wang, H. L. Meyers, P. P. Smith, C. L. Corless, G. J. Henderson, J. Vieira, W. J. Britt, and J. A. Nelson. 1999. Human cytomegalovirus infection of caco-2 cells occurs at the basolateral membrane and is differentiation state dependent. J. Virol. 73:4552-4560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kahl, M., D. Siegel-Axel, S. Stenglein, G. Jahn, and C. Sinzger. 2000. Efficient lytic infection of human arterial endothelial cells by human cytomegalovirus strains. J. Virol. 74:7628-7635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Knowles, W. A. 1976. In-vitro cultivation of human cytomegalovirus in thyroid epithelial cells. Arch. Virol. 50:119-124. [DOI] [PubMed] [Google Scholar]
- 27.MacCormac, L. P., and J. E. Grundy. 1999. Two clinical isolates and the Toledo strain of cytomegalovirus contain endothelial cell tropic variants that are not present in the AD169, Towne, or Davis strains. J. Med. Virol. 57:298-307. [DOI] [PubMed] [Google Scholar]
- 28.Marchini, A., H. Liu, and H. Zhu. 2001. Human cytomegalovirus with IE-2 (UL122) deleted fails to express early lytic genes. J. Virol. 75:1870-1878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Miceli, M. V., D. A. Newsome, L. C. Novak, and R. W. Beuerman. 1989. Cytomegalovirus replication in cultured human retinal pigment epithelial cells. Curr. Eye Res. 8:835-839. [DOI] [PubMed] [Google Scholar]
- 30.Michelson-Fiske, S., J. Arnoult, and H. Febvre. 1975. Cytomegalovirus infection of human lung epithelial cells in vitro. Intervirology 5:354-363. [DOI] [PubMed] [Google Scholar]
- 31.Mocarski, E. S., M. N. Prichard, C. S. Tan, and J. M. Brown. 1997. Reassessing the organization of the UL42-UL43 region of the human cytomegalovirus strain AD169 genome. Virology 239:169-175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Murphy, E., D. Yu, J. Grimwood, J. Schmutz, M. Dickson, M. A. Jarvis, G. Hahn, J. A. Nelson, R. M. Myers, and T. E. Shenk. 2003. Coding potential of laboratory and clinical strains of human cytomegalovirus. Proc. Natl. Acad. Sci. USA 100:14976-14981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pignatelli, S., P. D. Monte, G. Rossini, and M. P. Landini. 2004. Genetic polymorphisms among human cytomegalovirus (HCMV) wild-type strains. Rev. Med. Virol. 14:383-410. [DOI] [PubMed] [Google Scholar]
- 34.Plachter, B., C. Sinzger, and G. Jahn. 1996. Cell types involved in replication and distribution of human cytomegalovirus. Adv. Virus Res. 46:195-261. [DOI] [PubMed] [Google Scholar]
- 35.Plotkin, S. A., T. Furukawa, N. Zygraich, and C. Huygelen. 1975. Candidate cytomegalovirus strain for human vaccination. Infect. Immun. 12:521-527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sinzger, C., M. Kahl, K. Laib, K. Klingel, P. Rieger, B. Plachter, and G. Jahn. 2000. Tropism of human cytomegalovirus for endothelial cells is determined by a post-entry step dependent on efficient translocation to the nucleus. J. Gen. Virol. 81:3021-3035. [DOI] [PubMed] [Google Scholar]
- 37.Sinzger, C., K. Schmidt, J. Knapp, M. Kahl, R. Beck, J. Waldman, H. Hebart, H. Einsele, and G. Jahn. 1999. Modification of human cytomegalovirus tropism through propagation in vitro is associated with changes in the viral genome. J. Gen. Virol. 80:2867-2877. [DOI] [PubMed] [Google Scholar]
- 38.Smith, G. A., and L. W. Enquist. 1999. Construction and transposon mutagenesis in Escherichia coli of a full-length infectious clone of pseudorabies virus, an alphaherpesvirus. J. Virol. 73:6405-6414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Smith, I. L., I. Taskintuna, F. M. Rahhal, H. C. Powell, E. Ai, A. J. Mueller, S. A. Spector, and W. R. Freeman. 1998. Clinical failure of CMV retinitis with intravitreal cidofovir is associated with antiviral resistance. Arch. Ophthalmol. 116:178-185. [DOI] [PubMed] [Google Scholar]
- 40.Smith, J. D. 1986. Human cytomegalovirus: demonstration of permissive epithelial cells and nonpermissive fibroblastic cells in a survey of human cell lines. J. Virol. 60:583-588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vesterinen, E., P. Leinikki, and E. Saksela. 1975. Cytopathogenicity of cytomegalovirus to human ecto- and endocervical epithelial cells in vitro. Acta Cytol. 19:473-481. [PubMed] [Google Scholar]
- 42.Vonka, V., E. Anisimova, and M. Macek. 1976. Replication of cytomegalovirus in human epithelioid diploid cell line. Arch. Virol. 52:283-296. [DOI] [PubMed] [Google Scholar]
- 43.Waldman, W. J., J. M. Sneddon, R. E. Stephens, and W. H. Roberts. 1989. Enhanced endothelial cytopathogenicity induced by a cytomegalovirus strain propagated in endothelial cells. J. Med. Virol. 28:223-230. [DOI] [PubMed] [Google Scholar]
- 44.Wang, D., W. Bresnahan, and T. Shenk. 2004. Human cytomegalovirus encodes a highly specific RANTES decoy receptor. Proc. Natl. Acad. Sci. USA 101:16642-16647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yu, D., G. A. Smith, L. W. Enquist, and T. Shenk. 2002. Construction of a self-excisable bacterial artificial chromosome containing the human cytomegalovirus genome and mutagenesis of the diploid TRL/IRL13 gene. J. Virol. 76:2316-2328. [DOI] [PMC free article] [PubMed] [Google Scholar]