Abstract
Effective T-cell responses are critical to eradicate acute viral infections and prevent viral persistence. Emerging evidence indicates that robust, early CD4 T-cell responses are important in effectively sustaining CD8 T-cell activity. Herein, we illustrate that virus-specific CD4 T cells are functionally inactivated early during the transition into viral persistence and fail to produce effector cytokines (i.e., interleukin-2 and tumor necrosis factor alpha), thereby compromising an efficient and effective antiviral immune response. Mechanistically, the inactivation occurs at the cellular level and is not an active process maintained by regulatory T cells or antigen-presenting cells. Importantly, a small subpopulation of cells is able to resist inactivation and persist into the chronic phase of infection. However, the virus-specific CD4 T-cell population ultimately undergoes a second round of inactivation, and the cells that had retained functional capacity fail to respond to rechallenge in an acute time frame. Based on these results we propose a biological mechanism whereby early CD4 T-cell inactivation leads to a subsequent inability to sustain cytotoxic T-lymphocyte function, which in turn facilitates viral persistence. Moreover, these studies are likely relevant to chronic/persistent infections of humans (e.g., human immunodeficiency virus, hepatitis C virus, and hepatitis B virus) by providing evidence that a reservoir of virus-specific CD4 T cells can remain functional during chronic infection and represent a potential therapeutic target to stimulate the immune response and establish control of infection.
Viral infection sets into motion an intricate series of interactions between the host immune system and the invading pathogen. The majority of viral infections stimulate strong cellular immune responses resulting in virus elimination; however, in some cases viruses (e.g., human immunodeficiency virus [HIV] and hepatitis C virus) outpace the immune system, promote a state of immunosuppression, and ultimately persist (28). Viral replication usually stimulates a robust CD8 cytotoxic T-lymphocyte (CTL) response that is responsible for eliminating infected cells and controlling infection. CTLs proliferate and rapidly acquire the ability to produce antiviral molecules, such as gamma interferon (IFN-γ) and perforin (21). Coincident with the development of the CTL response, CD4 T cells proliferate, acquire a Th1 or Th2 phenotype, and produce cytokines that modulate the activation of the immune response and sustain CTL function (2, 5, 12, 22, 24, 26, 44). CD8 T-cell responses to persistent viruses are initially robust, as indicated by their ability to proliferate and secrete antiviral molecules (4, 33, 43, 49). However, associated with the viral transition into persistence, CTLs lose functionality, succumbing either to inactivation and/or physical deletion (10, 11, 27, 49, 52). Previous studies have suggested that CD4 T-cell responses are critical to sustain CD8 T-cell function to prevent persistent infection (2, 3, 6, 26, 33, 43, 44) and to provide signals that promote development of CD8 T-cell memory (19, 39, 41). Accordingly, a disruption of appropriate CD4 T-cell responses could explain the inability to sustain CTL activity and to form CD8 T-cell memory during persistent viral infection (48).
The precise fate of CD4 T-cell responses during chronic infection is controversial. An in-depth analysis of antigen-specific CD4 T-cell responses using transgenic cells showed that these cells were fully functional (i.e., able to produce interleukin-2 [IL-2] and proliferate upon ex vivo stimulation) for at least 60 days during chronic lymphocytic choriomeningitis virus (LCMV) infection (30). Thus, it is unclear why in the presence of a strong CD4 T-cell response, particularly during the early phase of infection, the CTL response was exhausted. One caveat with the aforementioned study (30) was that naïve, antigen-specific CD4 T cells were transferred into mice during the chronic phase of a preexisting persistent viral infection and consequently were exposed to high antigen loads at a time well past the point of physiologic T-cell priming and following the onset of immunosuppression (38). In opposition to these findings, dysfunctional CD4 T-cell responses have been observed during persistent infection with LCMV (7, 10). Analysis of the endogenous CD4 T-cell population by ex vivo cytokine production illustrated a progressive loss in IFN-γ and IL-2, dropping below the level of detection by 200 days postinfection (10). However, in these studies the physical presence of virus-specific cells was not determined; thus, it is unclear whether the dysfunction was due to an overall decrease in the number of virus-specific cells or a progressive loss of functional responsiveness. In a separate study, transgenic, virus-specific CD4 T cells were found to persist for extended periods of time during chronic infection, although the functional capacity of the transgenic cells was not examined (7). Moreover, these studies utilized CD8- or perforin-knockout mice to induce the persistent infection; consequently, it is unclear how CD4 T cells react during a persistent infection in an immunologically competent environment. Presently, there are clear discrepancies concerning CD4 T-cell dynamics during persistent infection and, because augmentation of CD4 T-cell responses can have a profound impact on controlling chronic infection in mice and humans (2, 25), a more thorough understanding of CD4 T-cell dynamics during persistent infection is important both for understanding the molecular basis of viral persistence and for the design of future therapeutic strategies.
Following the initial exposure to antigen, T cells undergo a variety of responses depending on the length of contact with the antigen-presenting cell (APC) (18, 45, 51), the strength of the signal given (32, 40), and the maturation level of the stimulating APC (35). In response to self antigens, T cells proliferate to various degrees and may acquire effector functions; however, they rapidly lose responsiveness in a process termed tolerization or anergy (35). An important factor that determines the fate of T cells during tolerance induction is antigen concentration and the consequent strength of the activating signal (32, 40). In response to high antigen concentrations, CD8 T cells become anergic and persist without effector function (32). Conversely, lower antigenic concentrations induce T-cell exhaustion, in which efficient effector functions initially form but then the cells are deleted. There is also evidence that tolerance is an active mechanism, as blocking of CTLA-4 engagement on the surface of T cells can prevent tolerance (31). The level of proliferation on the other hand does not necessarily appear to be related to the development of T-cell tolerance. Although abortive proliferative responses occur upon activation in the absence of costimulation (16), extensive proliferation can be observed in response to both immunizing and tolerizing antigens (1). Similar T-cell nonresponsiveness, and in some cases deletion, occurs during persistent viral infection (27); however, the relationship of this process to tolerance is unclear. Unlike the response to immunogenic, replicating pathogens that induce high levels of IFN-α/β, inflammatory cytokines, and APC maturation, the antiself response is generated in the absence of inflammatory stimuli. Thus, the environmental cues to which T cells are exposed during the recognition of self versus pathogenic antigens are different, which undoubtedly influences the generation of the responding T-cell populations.
Herein we analyzed CD4 T-cell responses to acute and persistent LCMV infection to determine how the antigenic environment shapes the ensuing functional responses. CD4 T-cell responses to acute and persistent infection were initially similar; however, persistent infection rapidly induced a state of T-cell inactivation. Within 9 days of infection, the vast majority of antigen-specific CD4 T cells were incapable of producing IL-2 or tumor necrosis factor alpha (TNF-α) and ultimately failed to develop memory responses. Interestingly, there was a selection for the few cells that were not functionally inactivated, and these cells persisted during the chronic phase of infection. Mechanistically, the functional inactivation was at the cellular level and was not due to active repression from antigen-presenting cells or from T regulatory cells. Temporally, the CD4 T-cell inactivation coincided with CD8 T-cell exhaustion and suggests a biological mechanism whereby functional disruption of CD4 T cells prevents the appropriate orchestration of the immune response and facilitates viral persistence.
MATERIALS AND METHODS
Mice.
C57BL/6 (H-2b, Thy1.2+) and congenic B6.PL-THY1a/CyJ (Thy1.1+) mice were from the Rodent Breeding Colony at The Scripps Research Institute. The LCMV-GP-specific, CD4+ T-cell receptor (TcR) transgenic (SMARTA) mice were provided by Hans Hengartner and have been described previously (29). All mice were housed under pathogen-free conditions. Mouse handling conformed to the requirements of the National Institutes of Health and The Scripps Research Institute Animal Research Committee.
Virus.
Six- to 8-week-old C57BL/6 mice were infected intravenously (i.v.) with 2 × 106 PFU of LCMV Armstrong clone 53b or LCMV Clone 13 to generate acute or persistent infection, respectively. Stocks were prepared by a single passage on BHK-21 cells, and viral titers were determined by plaque formation on Vero cells. For the rechallenge experiments, mice were infected i.v. with 2 × 106 PFU of Clone 13. The phenotypic and genotypic characterization of both LCMV strains, their passage, and quantitation have been described elsewhere (34).
T-cell isolation, adoptive transfers, and tissue processing.
CD4 T cells were initially purified from the spleens of naïve SMARTA mice by negative selection (StemCell Technologies, Vancouver, B.C., Canada), and 104 purified cells were transferred i.v. into C57BL/6 mice. The mice were then infected 2 days later with LCMV. For all experiments in which intrahepatic lymphocytes were analyzed, the mice were perfused with 25 to 30 ml of 0.9% saline by direct cardiac injection to remove blood from all tissue compartments. Single-cell suspensions from the spleen and liver were then isolated by mechanical dispersion through a 100-μm filter. Red blood cells were lysed in 0.84% ammonium chloride. Intrahepatic lymphocytes were further isolated by centrifugation in 35% Percoll (Amersham Biosciences, Uppsala, Sweden). The absolute number of SMARTA cells in each organ was determined by multiplying the frequency of Thy1.1+ cells (determined flow cytometry) by the total number of cells in each organ.
Intracellular cytokine staining and flow cytometry.
Total splenocytes or intrahepatic lymphocytes were stimulated for 5 to 6 h with 5 μg/ml of the major histocompatibility complex class II-restricted LCMV-GP61-80 peptide (>95% pure; Synpep, Dublin, California) in the presence of 50 U/ml recombinant murine IL-2 (R&D Systems, Minneapolis, Minnesota) and 1 μg/ml brefeldin A (Sigma, St. Louis, MO). The presence of IL-2 in the ex vivo stimulation cultures did not alter cytokine expression or skew the responses toward a Th1 or Th2 phenotype (data not shown). Cells were then stained for surface expression of CD4 (RM4-5), CD8 (53-6.7), and Thy1.1 (OX-7) conjugated to phycoerythrin-Cy7, allophycocyanin-Cy7, and peridinin chlorophyll a protein, respectively (Pharmingen, La Jolla, CA). The cells were then simultaneously fixed and permeabilized with a paraformaldehyde-saponin solution and stained with fluorescein isothiocyanate-, phycoerythrin-, and allophycocyanin-conjugated antibodies to IFN-γ (XMG1.2), TNF-α (MP6-XT22), IL-2 (JES6-5H4), or IL-10 (JES5-16E3) (Pharmingen, La Jolla, CA). Five- and six-color flow cytometric analysis was performed using the Digital LSR II (Becton Dickinson, San Jose, California). Forward versus side scatter profiles were used to define the live population, and gates for cytokine analysis were set based on antibody isotype controls and cells that stained negative for the protein of interest.
Cell proliferation and cell cycle analysis.
Surface staining for the ex vivo cell cycle analyses of SMARTA cells was performed as described above using total splenocytes. Stained splenocytes were resuspended in 70% ethanol and put at 4°C overnight. A 1-μg/ml concentration of 4′,6-diamidino-2-phenylindole, dihydrochloride (DAPI; Molecular Probes, Eugene, Oregon) in 0.1% Triton X-100 was then added, and the samples were immediately analyzed on the Digital LSR II. Gates were set such that the fluorescence intensity of the G2/M population was twice that of the G0/G1a peak.
Cell proliferation was measured by carboxyfluorescein diacetate N-succinimidyl ester (CFSE; Molecular Probes) dilution. Naive SMARTA cells were resuspended in phosphate-buffered saline containing 2 μM CFSE and cultured as indicated. The frequency of SMARTA cells that proliferated during culture was determined as described by Gett et al. (13). Briefly, the number of cells in each division peak was divided by 2i (where i equals the number of divisions). The total number of SMARTA cells in each peak was then summed, and this number was divided by the total number of SMARTA cells in the culture. This number was then expressed as the percentage of cells that had proliferated. In vivo proliferation of SMARTA cells was determined by transferring 104 CFSE-labeled SMARTA cells into C57BL/6 mice i.v., followed 2 days later by Armstrong or Clone 13 infection. To ensure that proliferation was a specific response to viral infection and not homeostatic proliferation, CFSE-labeled SMARTA cells were transferred in parallel into mice that did not receive virus.
T-cell culture assays.
To determine the repressive effects of APC, CD4 T cells were isolated from Armstrong- or Clone 13-infected animals that had received SMARTA cells prior to infection by positive selection with CD4 magnetic activated cell sorter beads (Miltenyi, Auburn, California). To isolate APC, splenocytes from Armstrong- or Clone 13-infected animals that had not received SMARTA cells were depleted of T cells with Thy1.2 magnetic activated cell sorter beads (Miltenyi). T cells and APC were mixed together at a 1:4 T cell to APC ratio in the presence of GP61-80 peptide, and intracellular cytokine staining was performed. To evaluate T regulatory function, splenic CD4 T-cell populations were sorted using the FACSVantage (Becton Dickinson). Postsort purities for each population were ≥98%. The sorted populations were then individually mixed with CFSE-labeled naïve SMARTA cells at a 1:1 dilution. T-cell-depleted APC (isolated as described above) from 3-day Armstrong-infected animals or mice persistently infected from birth (carrier mice) were isolated and added to the culture at a 1:1 T cell to APC ratio. No exogenous peptide or cytokines were added to the cultures. The cells were cultured in complete medium (RPMI 1640 supplemented with 10% fetal bovine serum, 1% Na-pyruvate, 10 mM HEPES, 1% nonessential amino acids, and 500 U/ml penicillin and streptomycin) supplemented with 50 μM β-mercaptoethanol for 5 days, and CFSE dilution in the naïve SMARTA cell population was analyzed by flow cytometry. The stimulatory potential of APC during Armstrong and Clone 13 infection was determined by culturing CFSE-labeled naïve SMARTA cells with T-cell-depleted APC at a 1:4 ratio for 5 days. The cells were cultured in complete medium supplemented with 50 μM β-mercaptoethanol. Where indicated, 5 μg/ml of the GP61-80 peptide was added to the cultures.
Statistical analysis.
Student's t tests were performed using the SigmaStat 2.0 software (Systat Software Inc., Richmond, California) to determine statistical significance (P ≤ 0.05). Three to four mice were used in each experimental group, as indicated in the figure legends.
RESULTS
Dynamics of the virus-specific CD4 T-cell response in acute versus persistent infection.
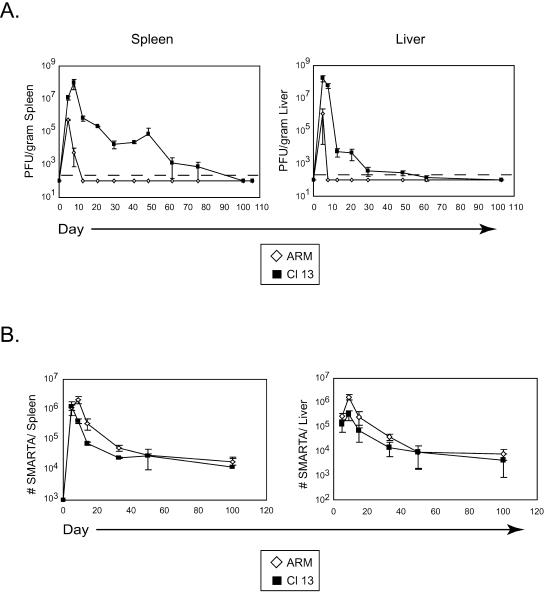
In order to analyze the generation and maintenance of CD4 T-cell responses during acute and persistent infection, we utilized a system in which Thy1.1+, TcR transgenic CD4 T cells (SMARTA cells) specific to the LCMV GP61-80 peptide presented in the context of I-Ab (29) were transferred into Thy1.2+ C57BL/6 mice. The I-Ab GP61-80-restricted response is the dominant CD4 T-cell response to LCMV (17, 46). In an attempt to retain a physiologic setting while maximizing the ability to visualize the response, 104 SMARTA cells were transferred into each mouse prior to infection. Following transfer, C57BL/6 mice were infected with LCMV Armstrong or LCMV Clone 13. The LCMV variant Clone 13, which was derived during the course of a natural infection with Armstrong, induces immunosuppression due to a single amino acid change in the glycoprotein that enables efficient infection of dendritic cells (DC) (4, 34, 37). Although genetically similar (differing by only two nucleotides in the entire genome), Armstrong and Clone 13 are phenotypically distinct viruses that replicate in different cell types and have different clearance rates while sharing identical CD4 and CD8 T-cell epitopes (10). Infection with Armstrong leads to an acute infection that is readily cleared from all organs, including the spleen and liver, by the CTL response within 10 days (Fig. 1A). These organs were selected in this study to represent a lymphoid and nonlymphoid compartment, respectively. During infection with Clone 13, on the other hand, the virus replicates to 2 logs higher in both lymphoid and nonlymphoid tissues and results in a chronic infection that abrogates the CTL response (49) and persists for 60 to 80 days (Fig. 1A), by mechanisms which are still unclear.
FIG. 1.
Kinetics of viral clearance and the LCMV-specific CD4 T-cell response. (A) Spleen and liver tissues were isolated from LCMV Armstrong-infected (ARM) or LCMV Clone 13-infected (Cl 13) animals at the indicated days postinfection, and the titers of infectious virus were determined by plaque assay. Data are expressed as PFU per gram of tissue. The dashed line represents the lower limit of detection, which is 200 PFU/gram of tissue. Each time point represents the average ± standard deviation (SD) of three mice per group. (B) The kinetics of the SMARTA T-cell response. A total of 104 SMARTA cells were transferred into C57BL/6 mice that were subsequently infected with Armstrong (ARM) or Clone 13 (Cl 13). Spleen (left panel) and liver (right panel) cells were isolated at the indicated days postinfection, and the absolute number of SMARTA cells in each organ was determined. Values represent the average ± SD of three mice at each time point and are representative of at least two experiments.
Following infection with Armstrong or Clone 13, SMARTA cells initially expanded with similar kinetics in the spleen and liver (Fig. 1B), and all the SMARTA cells expressed elevated levels of CD44 by day 5 postinfection (data not shown). Moreover, similar levels of CD25 expression were observed early in the response to Armstrong and Clone 13 infection (day 5) (data not shown), further indicating that all the SMARTA cells were activated following . Interestingly, following the initial expansion in the spleen, SMARTA cells in Clone 13-infected mice contracted earlier than during Armstrong infection, as SMARTA cells from Armstrong-infected animals continued to expand through viral clearance and peaked at 9 days postinfection. This early contraction was not observed in the liver (Fig. 1B), wherein the number of SMARTA cells continued to increase through day 9. Nevertheless, the contraction kinetics in the spleen and liver occurred at the same rate during both infections (Fig. 1B), with rates of decline leveling off 30 to 50 days postinfection, temporally consistent with memory development following acute infection (20). Hence, unlike the dominant Db NP396-404-specific CD8 T-cell response, which is deleted during Clone 13 infection (27, 49, 52), the dominant I-Ab GP61-80-restricted CD4 T-cell response is maintained throughout the entire infection. It is interesting that in spite of the premature contraction in the spleen during Clone 13 infection, ultimately similar numbers of virus-specific cells remained. These data suggest that long-term maintenance of virus- specific CD4 T cells is not influenced by antigen clearance kinetics.
Persistent infection induces early functional inactivation of CD4 T cells.
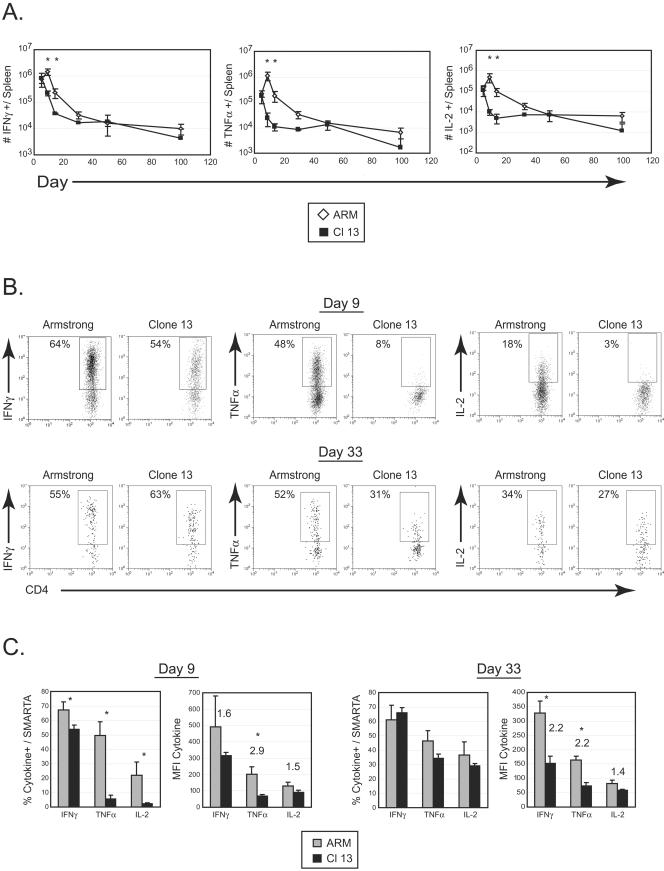
During a developing immune response, CD4 T cells play a crucial role in the production of cytokines that direct immune function, sustain CTL activity when required, and potentiate CD8 T-cell memory generation (36). Consequently, an inability of CD4 T cells to produce cytokines during persistent infection would be expected to have a detrimental impact on the ensuing immune response and help explain the failure to sustain CD8 T-cell responses. By day 5 post-Armstrong infection, the majority of SMARTA cells in the spleen expressed IFN-γ, and the number of IL-2- and TNF-α-producing cells progressively increased through the first 9 days of infection (Fig. 2A). The maximal frequency of splenic IFN-γ- and TNF-α-producing SMARTA cells was observed on day 9 post-Armstrong infection and coincided with the peak of the CD4 T-cell response (Fig. 2B and C). Comparatively, the kinetics of IL-2 production were somewhat delayed, given that the maximum frequency was not reached until 15 to 30 days after Armstrong infection (data not shown), coinciding with the transition into memory (36). In stark contrast, the CD4 T-cell response to a persistent viral infection was dramatically diminished. Although similar frequencies of cytokine-producing SMARTA cells were observed in the spleen during the initial phase of clonal expansion (day 5) (data not shown), by day 9 post-Clone 13 infection there was a statistically significant ninefold decrease in the frequency of both TNF-α- and IL-2-producing cells (Fig. 2C), indicating functional inactivation of the virus-specific CD4 T-cell response. The functional inactivation consequently resulted in a significant decline in the number of cytokine-producing cells capable of stimulating an immune response. This is illustrated by the 47- and 49-fold decrease in the absolute number of TNF-α- and IL-2-secreting SMARTA cells in the spleen, respectively, compared to Armstrong infection. Although some of this loss in cell numbers could be attributed to the early 3-fold contraction of total SMARTA cells observed between days 5 and 9 following Clone 13 infection, it does not account for the entire reduction of TNF-α-producing and particularly IL-2-producing cells, which fell 8- and 11- fold, respectively, in this same time period. On the other hand, the fourfold decrease in the number of IFN-γ-producing cells between days 5 and 9 of Clone 13 infection could be explained by the early contraction. Importantly, the SMARTA cells that were present were unable to produce IL-2 or TNF-α, and a decreased number produced IFN-γ. In addition to the functional inactivation of the majority of SMARTA cells, the few cells that remained responsive produced decreased amounts of cytokine (based on mean fluorescence intensity [MFI]) (Fig. 2C).
FIG.2.
Persistent viral infection induces rapid CD4 T-cell inactivation. (A) Splenocytes from the same experiment shown in Fig. 1B were isolated at the indicated days post-Armstrong (ARM) or post-Clone 13 (Cl 13) infection. The absolute numbers of IFN-γ-producing (left panel), TNF-α-producing (middle panel), and IL-2-producing (right panel) SMARTA cells were assessed by intracellular cytokine staining. Each point represents the average ± standard deviation (SD) for three mice and is representative of at least two experiments. Asterisks denote statistical difference between Armstrong and Clone 13 infection at the indicated time point (P ≤ 0.05). (B) Representative dot plots at 9 (top panels) and 33 (bottom panels) days post-Armstrong or post-Clone 13 infection are shown for the time course illustrated in panel A. These time points were selected to provide an example of the acute (day 9) and chronic (day 33) phase of a persistent viral infection. The production of IFN-γ, TNF-α, and IL-2 was analyzed by intracellular flow cytometry after stimulation with GP61-80 peptide in vitro. Data are from a single Armstrong- or Clone 13-infected mouse. Flow plots are gated on SMARTA cells, and the number indicates the frequency of SMARTA cells that produced each cytokine. (C) The percentage of SMARTA cells producing IFN-γ, TNF-α, and IL-2 in the spleen during the acute phase (day 9) (left panels) and memory or chronic phase (day 33) (right panels) of Armstrong (ARM) or Clone 13 (Cl 13) is shown. The MFI of each cytokine is shown in the right graph of each set. Asterisks denote statistical significance (P ≤ 0.05), and the numbers represent the fold difference in MFI between SMARTA cells from Armstrong and Clone 13 infections. Each point represents the average ± SD for three mice and is representative of at least two experiments.
Functional inactivation was confirmed in the endogenous CD4 T-cell response by staining with major histocompatibility complex class II tetramers loaded with GP61-80 peptide and performing intracellular cytokine analyses (17). Tetramer analyses revealed that endogenous GP61-80-restricted CD4 T cells were not physically deleted after Clone 13 infection; however, very little IL-2 or TNF-α could be detected by intracellular cytokine analyses (data not shown). Similarly, only very low levels of IL-2 or TNF-α production were observed by the subdominant NP309-328-restricted CD4 T-cell response (data not shown). Collectively, these findings demonstrate that there is a global inactivation of the LCMV-specific CD4 T-cell response during the early phases of persistent viral infection and that transferred SMARTA cells accurately reflect the endogenous LCMV-specific CD4 T-cell response.
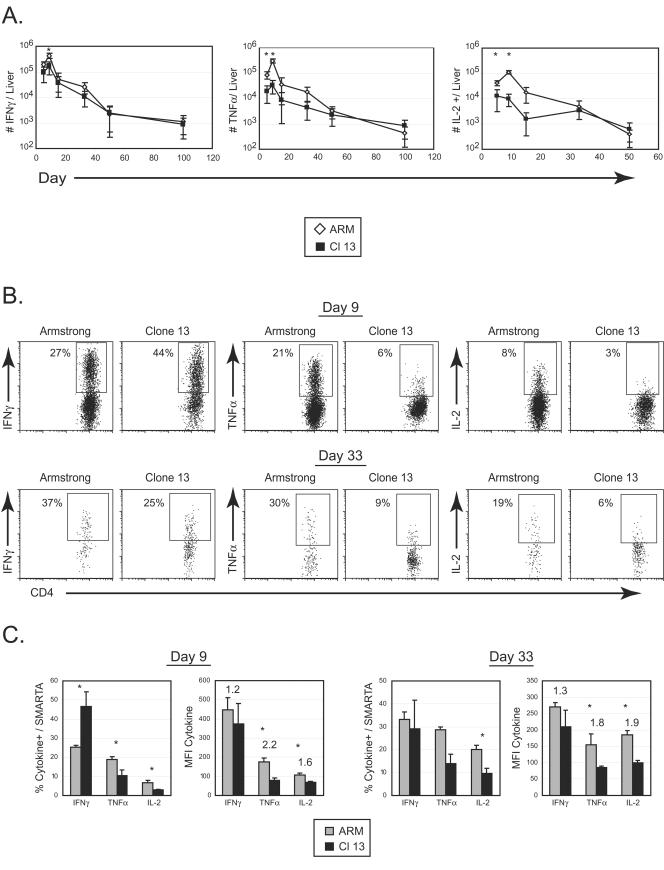
In a nonlymphoid tissue, the number of IFN-γ-, TNF-α-, and IL-2-producing cells also peaked at day 9 and then contracted (Fig. 3); however, the single-cell kinetics differed from the spleen. Following Armstrong infection, the peak frequency of cytokine production was observed on day 5, when approximately 75% and 35% of SMARTA cells produced IFN-γ and TNF-α, respectively, in both infections (data not shown). This is in contrast to the spleen, where the maximum frequency of cytokine production was reached on day 9. Interestingly, the frequency of IFN-γ-producing cells declined in the liver of Armstrong-infected animals by day 9 (temporally corresponding to viral clearance), while the percentage of IFN-γ-producing SMARTA cells during Clone 13 infection remained elevated by comparison (Fig. 3B). Nonetheless, an early 10-fold decrease in the absolute number (Fig. 3A) and a 2-fold decrease in the frequency (Fig. 3B) of TNF-α-producing cells was observed in the liver of Clone 13-infected mice, analogous to splenic results. A lower number and frequency of IL-2-producing SMARTA cells were also observed in livers of Clone 13-infected mice, although it should be noted that the liver did not harbor a large influx of IL-2 producers following either infection (Fig. 3). Collectively, our data demonstrate that CD4 T-cell function is disrupted in both lymphoid and nonlymphoid compartments during the acute phase of persistent infection, precisely at the time when it is required to sustain the immune response.
FIG.3.
T-cell inactivation in a nonlymphoid tissue. (A) Infiltrating liver lymphocytes from the same experiment shown in Fig. 2 were isolated at the indicated days post-Armstrong (ARM) or post-Clone 13 (Cl 13) infection. The absolute number of IFN-γ- (left panel), TNF-α- (middle panel), and IL-2-producing (right panel) SMARTA cells was assessed by intracellular cytokine staining. Each point represents the average ± standard deviation for three mice and is representative of at least two experiments. Asterisks denote statistical significance at the indicated time point (P ≤ 0.05). (B) Representative dot plots at days 9 (top panels) and 33 (bottom panels) post-Armstrong or post-Clone 13 infection are shown for the time course illustrated in panel A. The production of IFN-γ, TNF-α, and IL-2 was analyzed by intracellular flow cytometry. Data are from a single Armstrong- or Clone 13-infected mouse. Flow plots are gated on liver-infiltrating SMARTA cells, and the number in each plot indicates the frequency of SMARTA cells that produced each cytokine. (C) The percentage and MFI of IFN-γ, TNF-α, and IL-2 by liver-infiltrating SMARTA cells were analyzed as described in the legend for Fig. 2C.
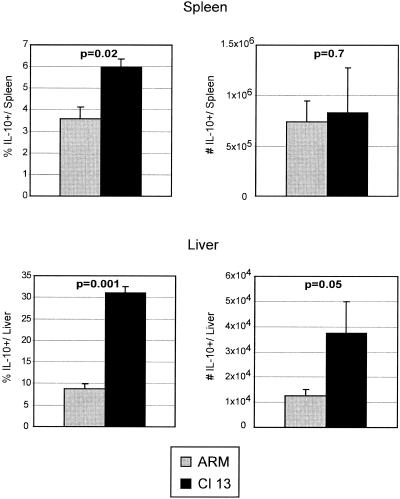
The induction of Th2 cytokine-producing cells during LCMV infection has been reported (50), and inappropriate Th2 responses have been associated with viral persistence. Therefore, it was possible that the observed decrease in IL-2 and TNF-α, both Th1-associated cytokines, coincided with a skewing towards a Th2-type response. To address this possibility, we quantified the frequency of IL-4-, IL-5-, and IL-10-producing SMARTA cells. Following infection with either Armstrong or Clone 13, no IL-4- or IL-5-producing CD4 T cells were observed (data not shown). However, there was a significant increase in the frequency of IL-10-producing cells in both the spleen and, most notably, the liver during the acute phase of persistent infection (Fig. 4). By day 5 post-Clone 13 infection, 30% of all SMARTA cells in the liver produced IL-10 upon stimulation. A significant increase in the absolute number of IL-10-producing cells was also observed in the liver, but not the spleen. All of the IL-10-producing cells coexpressed IFN-γ (data not shown), further suggesting that the SMARTA cells were not skewed toward a Th2 response. As IL-10 is known to suppress T-cell responses, it was interesting that IL-10-producing SMARTA cells were no longer observed by day 9 postinfection (data not shown).
FIG. 4.
Increased IL-10 production during persistent infection. Spleen (top panels) and liver (bottom panels) cells from Armstrong-infected (ARM) or Clone 13-infected (Cl 13) animals were isolated 5 days postinfection. The percentage (left panels) and absolute number (right panels) of IL-10-producing SMARTA cells were analyzed by intracellular flow cytometry. Bar graphs represent the average ± standard deviation from three mice in each group and are representative of three experiments. The corresponding P values for statistically different groups are denoted on the graphs.
As the Clone 13 infection progressed through the chronic phase (days 15 to 50), the absolute number of SMARTA cells in the spleen capable of producing TNF-α and IL-2 closely followed contraction kinetics (Fig. 2A), whereas the frequency of TNF-α- and IL-2-producing SMARTA cells increased (Fig. 2B and C). As the increased frequency was not accompanied by a concomitant increase in the absolute number of cytokine-producing cells, it appears that there is a selective survival of the SMARTA cells that are not inactivated as opposed to a functional resurgence of the remaining cells. To further determine the functionality of these cells, we analyzed their cytokine-producing capacity on a per cell basis. Similar to day 9, during the chronic phase of infection the amount of cytokine produced per cell was decreased in Clone 13 infection compared to Armstrong infection (Fig. 2C). Because the day 9 and day 33 time points were acquired on different days, it was not possible to directly compare the MFI between the time points. However, it should be noted that the decrease in the MFI remained constant at both time points, further supporting a survival advantage for the SMARTA cells that were not completely inactivated. Following the peak of the response, the number of cytokine-producing cells in the liver decreased with a similar kinetics during both types of infection (Fig. 3A). Although not as dramatic as observed in the spleen, the frequency of cytokine-producing cells in the liver also increased during the chronic phase of infection, yet the amount of cytokine produced per cell remained decreased (Fig. 3B), and this was not associated with an increase in the number of cytokine-producing cells (Fig. 3A).
Despite the selection for functionally responsive SMARTA cells over time, the increased frequency of TNF-α and IL-2 producers following Clone 13 infection was short-lived. A significant three- and fourfold decrease in the frequency of TNF-α- and IL-2-producing SMARTA cells, respectively, was observed in the spleen by day 100 postinfection (data not shown). Even the frequency of IFN-γ-producing cells during Clone 13 infection, which had been relatively stable throughout the persistent infection, showed a threefold decrease in the spleen by day 100. Importantly, this functional collapse was not associated with a decrease in the overall number of SMARTA cells (Fig. 1b), which is suggestive of a second round of functional inactivation, rather than physical deletion.
CD4 T cells from persistently infected animals do not respond to rechallenge.
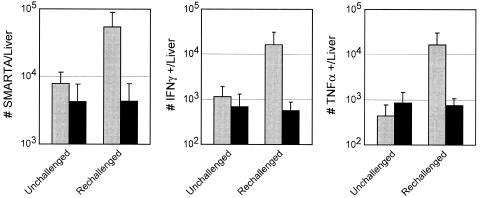
The final collapse in cytokine production following resolution of the persistent infection suggested that the cells might be unable to respond to a reencounter with the same pathogen. To assess the recall responses of CD4 T cells, we rechallenged mice that had been infected with Armstrong or Clone 13 100 days earlier. Although restricted residual viral replication is observed in the brain and kidney at this time (49), it is completely cleared from the other peripheral organs, including the spleen, liver (Fig. 1A), and blood (data not shown). Both groups of mice were rechallenged with Clone 13, and the number/functional capacity of SMARTA cells in the spleen and liver were analyzed. No increase in SMARTA cell numbers or functional capacity was observed in the spleens of Armstrong- or Clone 13-infected animals (data not shown); however, rechallenge induced a sevenfold expansion of SMARTA cells in the liver of animals that had been first infected with Armstrong, whereas no change was observed in animals previously infected with Clone 13 (Fig. 5). Similarly, rechallenge induced a 14- and 36-fold increase in the number of IFN-γ- and TNF-α-producing cells, respectively, in the liver of mice previously infected with Armstrong (Fig. 5), whereas no increase was observed in animals that had been first infected with Clone 13. No increase in IL-2-producing cells in the liver was observed in either case (data not shown). Hence, the prolonged antigen exposure during persistent infection prevents the establishment of CD4 T-cell recall responses within 3 days following reinfection.
FIG. 5.
Chronic infection prevents the establishment of memory CD4 T-cell responses. A. Mice that had been previously infected with Armstrong (gray bars) or Clone 13 (black bars) were either left alone (left bars in each panel) or were rechallenged (right bars in each panel) with Clone 13. After 3 days, the livers of individual mice were isolated, and the number of SMARTA cells (left panel) was determined. IFN-γ (middle panel) and TNF-α (right panel) production were assessed by intracellular flow cytometry. Values represent the average ± standard deviation of three to four mice in each group.
CD4 T-cell inactivation is not due to active repression by APC or induction of T regulatory activity.
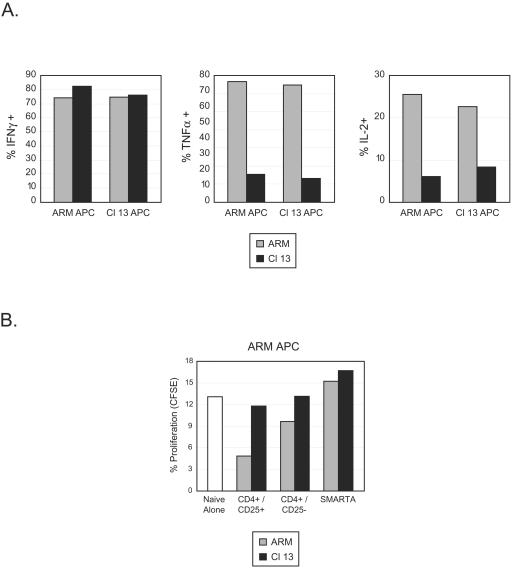
We next sought to determine the mechanisms that induce nonresponsive versus functional CD4 T-cell responses. It has been shown that dendritic cell function is perturbed during persistent LCMV infection (4, 37, 38). Consequently, the DC may express proteins that actively repress T-cell responses in cells that are otherwise functional. To analyze whether functional inactivation resulted from interactions with repressive DC, splenic SMARTA cells were sorted from Armstrong- or Clone 13-infected animals 9 days postinfection and stimulated with T-cell-depleted APC from animals infected in parallel with Armstrong or Clone 13. It was surmised that if the Clone 13 APC were actively suppressive, then SMARTA cells from Armstrong infected animals would appear nonresponsive following an in vitro exposure. However, this was not the case, as Armstrong SMARTA cells produced equivalent amounts of IFNγ, TNF-α, and IL-2 responses regardless of whether Armstrong or Clone 13 APC were used as stimulators (Fig. 6A). Conversely, stimulation of CD4 T cells from Clone 13-infected animals with Armstrong APC did not induce TNF-α or IL-2 production, suggesting that inactivation was not maintained by active repression from APC.
FIG. 6.
CD4 T-cell inactivation is not due to active repression or the generation of T regulatory cells. (A) Splenocytes from Armstrong-infected (ARM) or Clone 13-infected (Cl 13) animals were isolated 9 days postinfection and pooled separately. CD4 T cells from each population were then sorted and incubated with T-cell-depleted APC from Armstrong-infected (left bars in each panel) or Clone 13-infected (right bars in each panel) animals. The cells that served as APC were isolated 9 days postinfection from mice infected in parallel but that did not receive SMARTA cells. GP61-80 peptide was added to each culture, and the production of IFN-γ (left panel), TNF-α (middle panel), and IL-2 (right panel) was quantified by intracellular flow cytometry. Data are from a single experiment and are representative of two to three different experiments each containing pools of three spleens per group. (B) Splenocytes from Armstrong-infected (ARM) or Clone 13-infected (Cl 13) animals were isolated 9 days postinfection and pooled. SMARTA, endogenous CD4+/CD25+, and endogenous CD4+/CD25− populations were sorted by fluorescence-activated cell sorter. CFSE-labeled, naïve SMARTA cells were then mixed with equal numbers of each of these populations. These cell mixtures were then added to T-cell-depleted APC from an animal that had been infected with Armstrong 3 days previously. Naïve SMARTA cell proliferation was assessed after 5 days in culture by flow cytometric analysis of CFSE dilution. Bars represent the frequency of naïve SMARTA cells that proliferated during the culture period. The white bar represents cultures that received CFSE-labeled naïve SMARTA cells and T-cell-depleted APC but none of the sorted CD4 T-cell populations.
Another possibility was that T regulatory (Treg) cells developed early after persistent infection, resulting in suppression of the antiviral response (42). In order to asses this possibility, SMARTA cells as well as endogenous populations of CD4 T cells (CD25+ and CD25−) were isolated at day 9 post-Armstrong or post-Clone 13 infection. These populations were then individually cocultured with CFSE-labeled naïve SMARTA cells and T-cell-depleted APC obtained from Armstrong-infected mice at day 3 postinfection. The APC were isolated at this time to provide an endogenous source of antigen presentation acquired during the priming phase of an active and appropriate immune response. The ability of each CD4 population to suppress naïve T-cell proliferation was then assessed. Culturing naïve SMARTA cells with APC induced proliferation in approximately 13% of the cells (Fig. 6B). Coculture of naïve cells with SMARTA cells from either Armstrong- or Clone 13-infected animals did not substantially decrease proliferation. Similarly, SMARTA cells isolated on day 5 post-Armstrong or post-Clone 13 infection did not affect the naïve cell proliferation (data not shown). Interestingly, the endogenous population of CD4+ CD25+ T cells from Armstrong-infected animals decreased naïve cell proliferation by approximately 60% (Fig. 6B). This was not observed in Clone 13-infected mice, suggesting a regulatory activity of CD4 T cells at the peak of the acute response that is absent during persistent infection. No suppression was observed when the CD4+ CD25− population was used from either type of infection. Likewise, no Treg activity was observed at day 9 post-Clone 13 infection when APC from LCMV carrier mice, persistently infected at birth and presenting high levels of antigen (47), were used as a source of stimulation (data not shown). Taken together, these data indicate that the CD4 T-cell inactivation during persistent infection occurs at the cellular level and is not propagated by regulatory cells.
APC efficiently prime virus-specific CD4 T-cell responses during persistent infection.
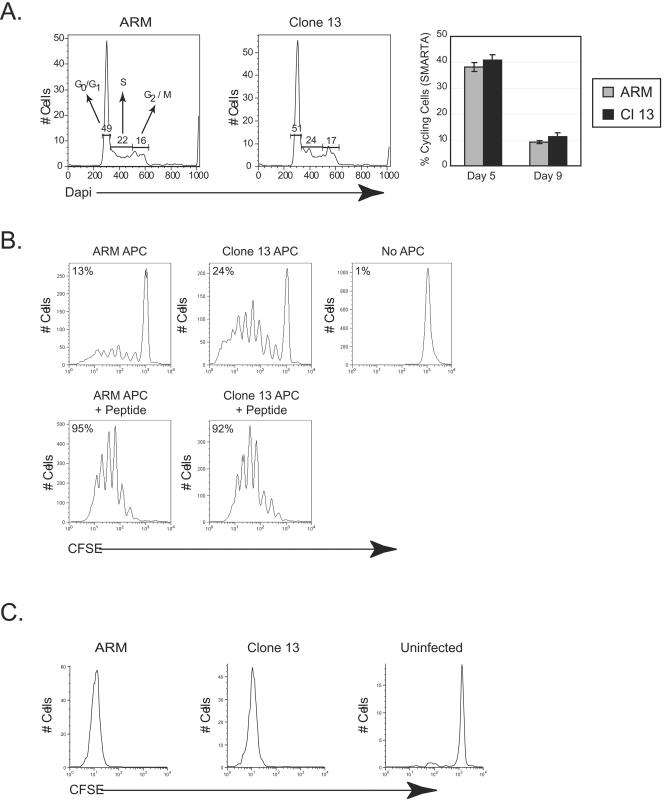
Since CD4 T-cell inactivation did not result from active APC repression, we next determined if the initial priming events set in motion an abortive differentiation pathway. One possible interpretation of the premature contraction observed during Clone 13 infection is that SMARTA cells receive faulty priming signals (similar to the induction of tolerance [23]) and do not expand appropriately. According to this theory, the initial cell expansion would appear similar between Armstrong and Clone 13 (as is observed early postinfection [Fig. 1A]), which would then be followed by a cessation of proliferation and a decline in cell numbers. To determine the efficacy of priming during Clone 13 infection, we conducted detailed analyses of SMARTA cell proliferative responses. Similar frequencies of SMARTA cells were cycling on day 5 post-Armstrong or post-Clone 13 infection (Fig. 5A), indicating that similar numbers of SMARTA cells were induced to proliferate during the initial expansion phase. By day 9 the frequency of proliferating SMARTA cells had decreased to a similar level in response to both infections, illustrating that the frequency of cells in cycle at any given time followed similar kinetics after both infections and that the early contraction during Clone 13 infection was not due to a decrease in cellular proliferation.
To determine if priming by APC obtained during the acute phase of persistent infection induced an abortive proliferative response, naïve SMARTA cells were labeled with CFSE and mixed with APC from animals that were infected with Armstrong or Clone 13 3 days earlier. APC were isolated from animals at day 3 postinfection to ensure that they were matured during the initial phase of each infection and when physiologic priming should occur, but prior to immunosuppression of the DC population observed during Clone 13 infection (4, 37, 38). Importantly, no peptide was added to these cultures, to ensure that only endogenous antigen presentation occurred. Cocultures with Armstrong APC induced 13% of naïve SMARTA cells to proliferate within 5 days, whereas 24% of naïve cells proliferated after culturing with APC from Clone 13-infected animals (Fig. 7B). Moreover, in the Clone 13 APC cultures, 23% of the naïve SMARTA cells that divided underwent four or more divisions, compared to 15% of the naive SMARTA cells stimulated by Armstrong APC. No proliferation occurred in the absence of APC. Importantly, CFSE-labeled naïve SMARTA cells proliferated extensively (>7 cell divisions) in vivo following Armstrong or Clone 13 infection, and no SMARTA cells were observed that had not completely diluted CFSE (Fig. 7C). To determine whether APC from both sources had the same capacity to stimulate naïve cells, coculture experiments were performed using APC coated with the GP61-80 peptide. Stimulation with peptide-labeled APC induced nearly all naïve SMARTA cells to proliferate regardless of the origin of the APC (Fig. 7B). Moreover, greater than 40% of the naïve cells in each case progressed through four or more divisions, illustrating that the inherent stimulatory capacity of APC from each type of infection was similar. Collectively, these studies demonstrate that efficient CD4 T-cell proliferative responses are invoked during the acute phase of persistent infection and that APC during the early phase of both types of infection have a similar potential to stimulate CD4 T-cell responses.
FIG. 7.
Efficient antigen presentation and enhanced stimulatory activity by APC during persistent infection. (A) Splenic SMARTA cells from Armstrong- or Clone 13-infected mice were isolated 5 days postinfection, and the percentages of SMARTA cells (Thy1.1+) in the G0/G1a, S, and G2/M stages of the cell cycle were determined directly ex vivo by intracellular DAPI labeling of DNA. An example of the cell cycle profile on day 5 following Armstrong (left panel) or Clone 13 (middle panel) infection is shown. The percentage of cycling SMARTA cells from day 5 and day 9 post-Armstrong (ARM) and post-Clone 13 (Cl 13) infection is shown (right panel). Values represent the average ± standard deviation of threemice in each group. (B) CFSE-labeled, naïve SMARTA cells were cultured for 5 days either alone (upper right panel) or with T-cell-depleted APC from an animal that had been infected 3 days earlier with Armstrong (upper left panel) or Clone 13 (upper middle panel). In parallel CFSE-labeled naïve SMARTA cells were cultured with T-cell-depleted APC (Armstrong, bottom left panel; Clone 13, bottom right panel) in the presence of exogenous GP61-80 peptide. Histograms are gated on SMARTA cells, and proliferation was assessed by flow cytometry based on the dilution of CFSE. Numbers in the upper left of each panel represent the total percentage of naïve SMARTA cells that had progressed through one or more divisions. These data show a single experiment and are representative of two to three experiments. (C) CFSE-labeled, naïve SMARTA cells were transferred into C57BL/6 mice that were left uninfected (right panel) or that were subsequently infected with Armstrong (left panel) or Clone 13 (middle panel). Splenocytes were isolated 5 days postinfection, and proliferation was analyzed by CFSE dilution. Flow plots are gated on SMARTA cells. Data are from a single mouse and are representative of three animals per group.
DISCUSSION
These studies document that virus-specific CD4 T cells are functionally inactivated early during persistent infection but that a subpopulation of cells that resist inactivation persist through the chronic phase of infection. During the initial expansion phase (through day 5), the CD4 T-cell response to both infections is similar, although the SMARTA cells in Clone 13-infected animals are skewed toward IL-10 production. However, within 9 days following Clone 13 infection, corresponding with the transition into viral persistence, antigen-specific CD4 T cells lose the ability to produce IL-2 and TNF-α. Priming of T cells in the presence of IL-10 has been shown to induce nonresponsiveness in vitro, resulting in the loss of cytokine production and effector function (14). A similar mechanism may be functioning during persistent infection, whereby the CD4 T cells are primed to produce IL-10, which then serves to dampen the resulting response.
It is interesting that an elevated frequency of IL-2- and TNF-α-producing SMARTA cells was noted during the chronic stage of Clone 13 infection. As the increase was not accompanied by a rise in the absolute number of cytokine-producing SMARTA cells, this appears to represent a survival advantage for those cells that can resist the early inactivation rather than a functional restoration of previously inactivated cells. This is further substantiated by the finding that the level of cytokine production by the remaining functionally responsive cells does not change between the acute (day 9) and chronic (day 33) phases of infection, suggesting that these cells are selected for over time. However, it is important to point out that the level of cytokine production per cell during the chronic stage of Clone 13 infection never reaches that observed following Armstrong infection, despite the decline in viral titers. Therefore, it appears that there is an intrinsic defect in the virus-specific CD4 T cells that is separable from the high levels of viral replication. It is conceivable that these remaining cytokine-producing cells represent a population that could be manipulated therapeutically to facilitate viral clearance. In fact, recent studies in humans illustrated that antigen-loaded DC can improve CD4 T-cell responses during chronic HIV infection (25); however, the relative quality of the response, both in terms of effector function on a per cell basis and the fraction of total antigen-specific cells that are activated, remains unknown. Thus, the system utilized herein allows future evaluation of the ability of different therapeutic vaccination strategies to stimulate T-cell responses during chronic infection.
Our results differ from previous observations analyzing virus-specific CD4 T-cell kinetics during persistent infection (30). In an attempt to directly monitor the behavior of virus-specific CD4 T cells during persistent infection, Oxenius et al. transferred naïve SMARTA cells into mice that had a preestablished persistent infection (30). One of the conclusions drawn from their study was that virus-specific CD4 T cells maintained functional responsiveness for nearly 60 days in an environment containing a heavy antigenic burden. If this conclusion is correct, it is unclear why CTL exhaustion would occur in the presence of a fully functional CD4 T-cell response. The major caveat with the study by Oxenius et al. (30) is that naïve SMARTA cells were transferred into mice that were already persistently infected, thus bypassing the normal sequence of events that occur during T-cell priming. Importantly, in our study we transferred naïve SMARTA cells into mice prior to infection and allowed priming to proceed normally. When this occurred the functional responsiveness of CD4 T cells was lost rapidly during persistent infection. A similar loss of cytokine production was observed by Fuller et al. (11); however, in their studies it was not determined whether the loss was due to deletion or inactivation of the virus-specific CD4 T-cell population. This issue was addressed partially in another system (7), where virus-specific CD4 T cells were observed throughout persistent infection. However, the lack of functioning CD8 T cells in these studies made it difficult to deduce how CD4 T cells respond during a physiologic infection in which the CTL response initially participates in viral clearance. Thus, our data agree with the observations of Fuller et al. (10, 11) and extend these studies to show that during the viral transition into persistence the virus-specific CD4 T cells are not deleted but remain in an inactivated state, a state that is phenotypically associated with the failure to control viral infections in humans, such as HIV (15).
Our studies also illustrate the divergent responses of virus-specific CD4 and CD8 T cells to a persistent infection. Unlike the CD8 T-cell compartment, in which one of the dominant epitope-specific responses is deleted during persistent infection with LCMV (49), there does not appear to be deletion of virus-specific CD4 T cells regardless of epitope specificity. This is an important point, because it indicates that virus-specific CD4 T cells are present during the transition into viral persistence, albeit in an inactivated state, which creates the potential to therapeutically restore function. Moreover, in our model system the overall number of virus-specific CD4 T cells present following contraction and during the chronic phase of persistent infection is similar to the number of cells observed during the memory phase of an acutely resolved infection. In contrast, the number of virus-specific CD8 T cells is consistently lower during persistent infection compared to an acutely resolved infection and remains low throughout infection (10, 11, 49). Hence, there appears to be an inherent level of ongoing CD4 T-cell maintenance, regardless of infection kinetics, that is not evident in the CD8 T-cell compartment. Perhaps most exciting is that during the chronic phase of persistent infection, the virus-specific CD4 T-cell population is at least partially responsive, whereas CD8 T-cell responsiveness remains depressed (unpublished observations) (10, 11, 49). Thus, our studies highlight that CD4 and CD8 T-cell responses differently adapt to a persistent viral infection, which suggests that independent mechanisms may be involved in functional inactivation.
The mechanisms underlying functional inactivation of CD4 T cells are of fundamental importance to the eradication of persistent infections that afflict humans. Accordingly, we set out to determine if the functional inactivation was an active suppression maintained by constant extracellular factors present during persistent infection or if it was due to a specific intracellular response that prevented the induction of full effector capacity. It is clear in our model system that APC do not trigger abortive T-cell proliferative responses. This same phenomenon may occur after infection with HIV or hepatitis C virus, as strong T-cell responses can be observed initially which are then followed by a subsequent state of functional decline (33, 43). At the early stages of acute and persistent infections, we observed similar levels of cycling cells in vivo, and all the SMARTA cells proliferated extensively in vivo. Moreover, ex vivo analyses of APC obtained during the priming phase from persistently infected mice revealed their capacity to efficiently induce naïve T-cell proliferation. In fact, they appeared to be better inducers of proliferation than APC from Armstrong-infected animals. Together these data indicate that the initial priming and proliferation events are intact during Clone 13 infection. Moreover, CD4 inactivation cannot be explained by the inability of APC to induce effector responses, as APC from Armstrong- and Clone 13-infected animals stimulated similar levels of cytokine production from CD4 T cells, whereas APC from Armstrong-infected animals did not restore function to inactivated CD4 T cells. These data indicate that APC from persistently infected mice do not actively repress the function of effector CD4 T cells. We also considered the possibility that the dysfunction observed in the CD4 T-cell response was due to the induction of Treg activity. However, neither SMARTA cells nor the endogenous CD25+ population from persistently infected mice substantially suppressed the proliferation of naïve T cells. It was interesting that some Treg activity appeared to develop at the peak of the T-cell response during Armstrong infection, but not Clone 13 infection, yet it is presently unclear whether this is due to a loss of regulatory activity during persistent infection or an alteration in the kinetics of Treg induction. The role and function of Treg cells during chronic infection is currently under investigation. Overall, our data suggest that early CD4 T-cell inactivation occurs at the cellular level following a Clone 13 infection, but it remains to be determined whether this is due to viral persistence, elevated viral replication, or the ability of the virus to target and alter the functionality of APC. Future studies will be important to differentiate the factors that lead to inactivation in order to provide insight into whether T-cell inactivation can be prevented if the infectious process is altered early during the establishment of viral persistence.
Our finding that CD4 T-cell inactivation coincides temporally with the CTL impairment (27, 49, 52) suggests a biological mechanism that may in part explain the failure to sustain a functional CD8 T-cell response. The fact that CD4 T-cell responses during persistent infection are, at the outset, strong suggests that they may initially stimulate some level of CD8 T-cell memory formation, which may explain the low levels of memory CD8 T-cell development observed following the resolution of a chronic infection (48). As CD8 T cells require paracrine IL-2 for sustained expansion (8, 9), the early loss of production by CD4 T cells potentially represents one mechanism whereby CTL responses are exhausted. Although sustained CTL responses may not solely be dependent on CD4 T cells, the early CD4 T-cell inactivation during persistent infection represents yet another level of dysfunction that likely contributes to the failure to control viral replication. Thus, a greater understanding of the early events in T-cell activation during acute and persistent infection as well as elucidation of the molecular mechanisms that regulate the divergent responses may ultimately give rise to interventions that preserve T-cell function and prevent viral persistence.
Acknowledgments
This is publication number 17003-NP from the Division of Virology, Department of Neuropharmacology.
We thank Kurt Edelmann for critical review of the manuscript and P. Truong, A. Tishon, and H. Lewicki for technical assistance.
This work was supported by NIH grants AI09484, DK58541 (M.B.A.O.), NS048866-01 (D.B.M.), and DK55037 (L.T.), a Dana Foundation grant (D.B.M.), and NIH training grant AI07244-22 (D.G.B.).
REFERENCES
- 1.Adler, A. J., C. T. Huang, G. S. Yochum, D. W. Marsh, and D. M. Pardoll. 2000. In vivo CD4+ T cell tolerance induction versus priming is independent of the rate and number of cell divisions. J. Immunol. 164:649-655. [DOI] [PubMed] [Google Scholar]
- 2.Battegay, M., D. Moskophidis, A. Rahemtulla, H. Hengartner, T. W. Mak, and R. M. Zinkernagel. 1994. Enhanced establishment of a virus carrier state in adult CD4+ T-cell-deficient mice. J. Virol. 68:4700-4704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berger, D. P., D. Homann, and M. B. Oldstone. 2000. Defining parameters for successful immunocytotherapy of persistent viral infection. Virology 266:257-263. [DOI] [PubMed] [Google Scholar]
- 4.Borrow, P., C. F. Evans, and M. B. Oldstone. 1995. Virus-induced immunosuppression: immune system-mediated destruction of virus-infected dendritic cells results in generalized immune suppression. J. Virol. 69:1059-1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cardin, R. D., J. W. Brooks, S. R. Sarawar, and P. C. Doherty. 1996. Progressive loss of CD8+ T cell-mediated control of a gamma-herpesvirus in the absence of CD4+ T cells. J. Exp. Med. 184:863-871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chang, K. M., R. Thimme, J. J. Melpolder, D. Oldach, J. Pemberton, J. Moorhead-Loudis, J. G. McHutchison, H. J. Alter, and F. V. Chisari. 2001. Differential CD4+ and CD8+ T-cell responsiveness in hepatitis C virus infection. Hepatology 33:267-276. [DOI] [PubMed] [Google Scholar]
- 7.Ciurea, A., L. Hunziker, P. Klenerman, H. Hengartner, and R. M. Zinkernagel. 2001. Impairment of CD4+ T cell responses during chronic virus infection prevents neutralizing antibody responses against virus escape mutants. J. Exp. Med. 193:297-305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.D'Souza, W. N., and L. Lefrancois. 2003. IL-2 is not required for the initiation of CD8 T cell cycling but sustains expansion. J. Immunol. 171:5727-5735. [DOI] [PubMed] [Google Scholar]
- 9.D'Souza, W. N., K. S. Schluns, D. Masopust, and L. Lefrancois. 2002. Essential role for IL-2 in the regulation of antiviral extralymphoid CD8 T cell responses. J. Immunol. 168:5566-5572. [DOI] [PubMed] [Google Scholar]
- 10.Fuller, M. J., A. Khanolkar, A. E. Tebo, and A. J. Zajac. 2004. Maintenance, loss, and resurgence of T cell responses during acute, protracted, and chronic viral infections. J. Immunol. 172:4204-4214. [DOI] [PubMed] [Google Scholar]
- 11.Fuller, M. J., and A. J. Zajac. 2003. Ablation of CD8 and CD4 T cell responses by high viral loads. J. Immunol. 170:477-486. [DOI] [PubMed] [Google Scholar]
- 12.Gerlach, J. T., H. M. Diepolder, M. C. Jung, N. H. Gruener, W. W. Schraut, R. Zachoval, R. Hoffmann, C. A. Schirren, T. Santantonio, and G. R. Pape. 1999. Recurrence of hepatitis C virus after loss of virus-specific CD4+ T-cell response in acute hepatitis C. Gastroenterology 117:933-941. [DOI] [PubMed] [Google Scholar]
- 13.Gett, A. V., and P. D. Hodgkin. 2000. A cellular calculus for signal integration by T cells. Nat. Immunol. 1:239-244. [DOI] [PubMed] [Google Scholar]
- 14.Groux, H., M. Bigler, J. E. de Vries, and M. G. Roncarolo. 1996. Interleukin-10 induces a long-term antigen-specific anergic state in human CD4+ T cells. J. Exp. Med. 184:19-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Harari, A., F. Vallelian, P. R. Meylan, and G. Pantaleo. 2005. Functional heterogeneity of memory CD4 T cell responses in different conditions of antigen exposure and persistence. J. Immunol. 174:1037-1045. [DOI] [PubMed] [Google Scholar]
- 16.Harding, F. A., J. G. McArthur, J. A. Gross, D. H. Raulet, and J. P. Allison. 1992. CD28-mediated signalling co-stimulates murine T cells and prevents induction of anergy in T-cell clones. Nature 356:607-609. [DOI] [PubMed] [Google Scholar]
- 17.Homann, D., L. Teyton, and M. B. Oldstone. 2001. Differential regulation of antiviral T-cell immunity results in stable CD8+ but declining CD4+ T-cell memory. Nat. Med. 7:913-919. [DOI] [PubMed] [Google Scholar]
- 18.Iezzi, G., K. Karjalainen, and A. Lanzavecchia. 1998. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 8:89-95. [DOI] [PubMed] [Google Scholar]
- 19.Janssen, E. M., E. E. Lemmens, T. Wolfe, U. Christen, M. G. von Herrath, and S. P. Schoenberger. 2003. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 421:852-856. [DOI] [PubMed] [Google Scholar]
- 20.Kaech, S. M., S. Hemby, E. Kersh, and R. Ahmed. 2002. Molecular and functional profiling of memory CD8 T cell differentiation. Cell 111:837-851. [DOI] [PubMed] [Google Scholar]
- 21.Kagi, D., B. Ledermann, K. Burki, R. M. Zinkernagel, and H. Hengartner. 1996. Molecular mechanisms of lymphocyte-mediated cytotoxicity and their role in immunological protection and pathogenesis in vivo. Annu. Rev. Immunol. 14:207-232. [DOI] [PubMed] [Google Scholar]
- 22.Kalams, S. A., S. P. Buchbinder, E. S. Rosenberg, J. M. Billingsley, D. S. Colbert, N. G. Jones, A. K. Shea, A. K. Trocha, and B. D. Walker. 1999. Association between virus-specific cytotoxic T-lymphocyte and helper responses in human immunodeficiency virus type 1 infection. J. Virol. 73:6715-6720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lenschow, D. J., T. L. Walunas, and J. A. Bluestone. 1996. CD28/B7 system of T cell costimulation. Annu. Rev. Immunol. 14:233-258. [DOI] [PubMed] [Google Scholar]
- 24.Lichterfeld, M., D. E. Kaufmann, X. G. Yu, S. K. Mui, M. M. Addo, M. N. Johnston, D. Cohen, G. K. Robbins, E. Pae, G. Alter, A. Wurcel, D. Stone, E. S. Rosenberg, B. D. Walker, and M. Altfeld. 2004. Loss of HIV-1-specific CD8+ T cell proliferation after acute HIV-1 infection and restoration by vaccine-induced HIV-1-specific CD4+ T cells. J. Exp. Med. 200:701-712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lu, W., L. C. Arraes, W. T. Ferreira, and J. M. Andrieu. 2004. Therapeutic dendritic-cell vaccine for chronic HIV-1 infection. Nat. Med. 10:1359-1365. [DOI] [PubMed] [Google Scholar]
- 26.Matloubian, M., R. J. Concepcion, and R. Ahmed. 1994. CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. J. Virol. 68:8056-8063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Moskophidis, D., F. Lechner, H. Pircher, and R. M. Zinkernagel. 1993. Virus persistence in acutely infected immunocompetent mice by exhaustion of antiviral cytotoxic effector T cells. Nature 362:758-761. [DOI] [PubMed] [Google Scholar]
- 28.Oldstone, M. B. 1989. Viral persistence. Cell 56:517-520. [DOI] [PubMed] [Google Scholar]
- 29.Oxenius, A., M. F. Bachmann, R. M. Zinkernagel, and H. Hengartner. 1998. Virus-specific MHC-class II-restricted TCR-transgenic mice: effects on humoral and cellular immune responses after viral infection. Eur. J. Immunol. 28:390-400. [DOI] [PubMed] [Google Scholar]
- 30.Oxenius, A., R. M. Zinkernagel, and H. Hengartner. 1998. Comparison of activation versus induction of unresponsiveness of virus-specific CD4+ and CD8+ T cells upon acute versus persistent viral infection. Immunity 9:449-457. [DOI] [PubMed] [Google Scholar]
- 31.Perez, V. L., L. Van Parijs, A. Biuckians, X. X. Zheng, T. B. Strom, and A. K. Abbas. 1997. Induction of peripheral T cell tolerance in vivo requires CTLA-4 engagement. Immunity 6:411-417. [DOI] [PubMed] [Google Scholar]
- 32.Rocha, B., A. Grandien, and A. A. Freitas. 1995. Anergy and exhaustion are independent mechanisms of peripheral T cell tolerance. J. Exp. Med. 181:993-1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rosenberg, E. S., J. M. Billingsley, A. M. Caliendo, S. L. Boswell, P. E. Sax, S. A. Kalams, and B. D. Walker. 1997. Vigorous HIV-1-specific CD4+ T cell responses associated with control of viremia. Science 278:1447-1450. [DOI] [PubMed] [Google Scholar]
- 34.Salvato, M., P. Borrow, E. Shimomaye, and M. B. Oldstone. 1991. Molecular basis of viral persistence: a single amino acid change in the glycoprotein of lymphocytic choriomeningitis virus is associated with suppression of the antiviral cytotoxic T-lymphocyte response and establishment of persistence. J. Virol. 65:1863-1869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schwartz, R. H. 1996. Models of T cell anergy: is there a common molecular mechanism? J. Exp. Med. 184:1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Seder, R. A., and R. Ahmed. 2003. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nat. Immunol. 4:835-842. [DOI] [PubMed] [Google Scholar]
- 37.Sevilla, N., S. Kunz, A. Holz, H. Lewicki, D. Homann, H. Yamada, K. P. Campbell, J. C. de La Torre, and M. B. Oldstone. 2000. Immunosuppression and resultant viral persistence by specific viral targeting of dendritic cells. J. Exp. Med. 192:1249-1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sevilla, N., D. B. McGavern, C. Teng, S. Kunz, and M. B. Oldstone. 2004. Viral targeting of hematopoietic progenitors and inhibition of DC maturation as a dual strategy for immune subversion. J. Clin. Investig. 113:737-745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Shedlock, D. J., and H. Shen. 2003. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300:337-339. [DOI] [PubMed] [Google Scholar]
- 40.Singh, N. J., and R. H. Schwartz. 2003. The strength of persistent antigenic stimulation modulates adaptive tolerance in peripheral CD4+ T cells. J. Exp. Med. 198:1107-1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sun, J. C., and M. J. Bevan. 2003. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science 300:339-342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Suvas, S., U. Kumaraguru, C. D. Pack, S. Lee, and B. T. Rouse. 2003. CD4+ CD25+ T cells regulate virus-specific primary and memory CD8+ T cell responses. J. Exp. Med. 198:889-901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Thimme, R., D. Oldach, K. M. Chang, C. Steiger, S. C. Ray, and F. V. Chisari. 2001. Determinants of viral clearance and persistence during acute hepatitis C virus infection. J. Exp. Med. 194:1395-1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tishon, A., H. Lewicki, G. Rall, M. Von Herrath, and M. B. Oldstone. 1995. An essential role for type 1 interferon-gamma in terminating persistent viral infection. Virology 212:244-250. [DOI] [PubMed] [Google Scholar]
- 45.van Stipdonk, M. J., G. Hardenberg, M. S. Bijker, E. E. Lemmens, N. M. Droin, D. R. Green, and S. P. Schoenberger. 2003. Dynamic programming of CD8+ T lymphocyte responses. Nat. Immunol. 4:361-365. [DOI] [PubMed] [Google Scholar]
- 46.Varga, S. M., and R. M. Welsh. 1998. Detection of a high frequency of virus-specific CD4+ T cells during acute infection with lymphocytic choriomeningitis virus. J. Immunol. 161:3215-3218. [PubMed] [Google Scholar]
- 47.Volkert, M., K. Bro-Jorgensen, and O. Marker. 1975. Persistent LCM virus infection in the mouse: immunity and tolerance. Bull. W. H. O. 52:471-478. [PMC free article] [PubMed] [Google Scholar]
- 48.Wherry, E. J., D. L. Barber, S. M. Kaech, J. N. Blattman, and R. Ahmed. 2004. Antigen-independent memory CD8 T cells do not develop during chronic viral infection. Proc. Natl. Acad. Sci. USA 101:16004-16009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wherry, E. J., J. N. Blattman, K. Murali-Krishna, R. van der Most, and R. Ahmed. 2003. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J. Virol. 77:4911-4927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Whitmire, J. K., M. S. Asano, K. Murali-Krishna, M. Suresh, and R. Ahmed. 1998. Long-term CD4 Th1 and Th2 memory following acute lymphocytic choriomeningitis virus infection. J. Virol. 72:8281-8288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Williams, M. A., and M. J. Bevan. 2004. Shortening the infectious period does not alter expansion of CD8 T cells but diminishes their capacity to differentiate into memory cells. J. Immunol. 173:6694-6702. [DOI] [PubMed] [Google Scholar]
- 52.Zajac, A. J., J. N. Blattman, K. Murali-Krishna, D. J. Sourdive, M. Suresh, J. D. Altman, and R. Ahmed. 1998. Viral immune evasion due to persistence of activated T cells without effector function. J. Exp. Med. 188:2205-2213. [DOI] [PMC free article] [PubMed] [Google Scholar]