Abstract
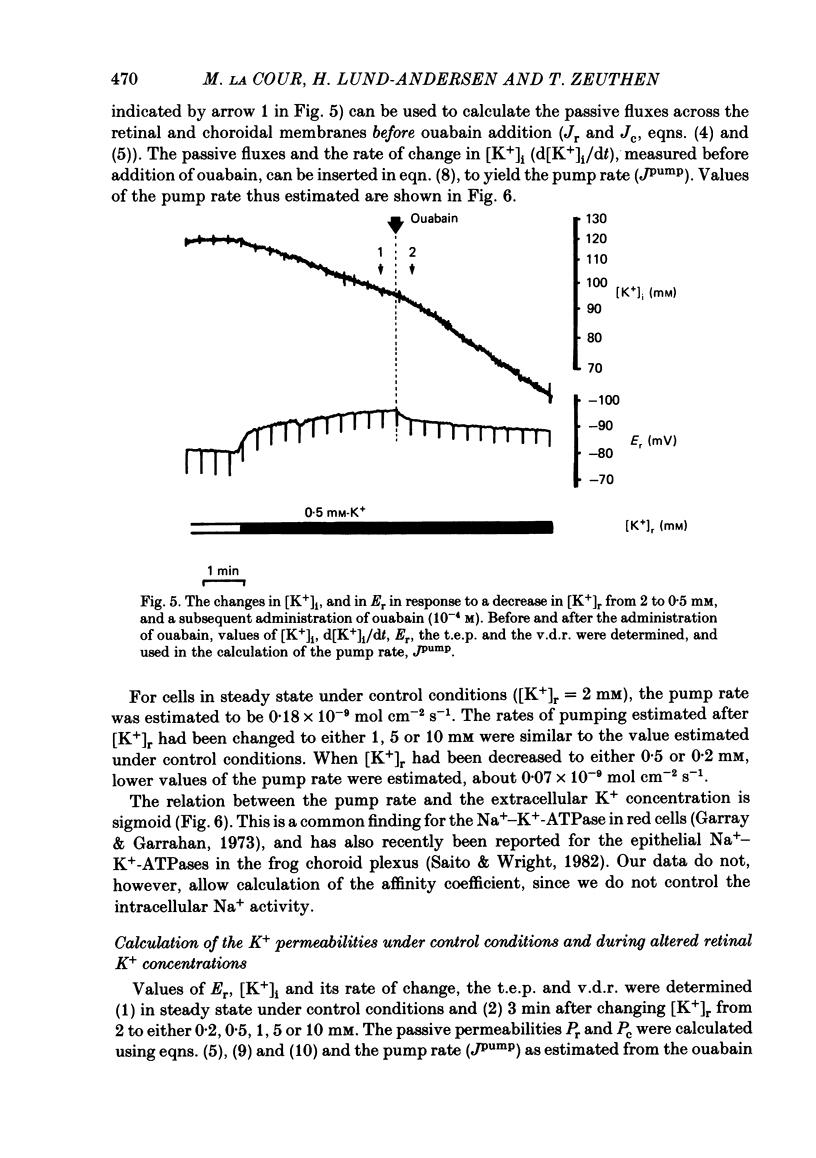
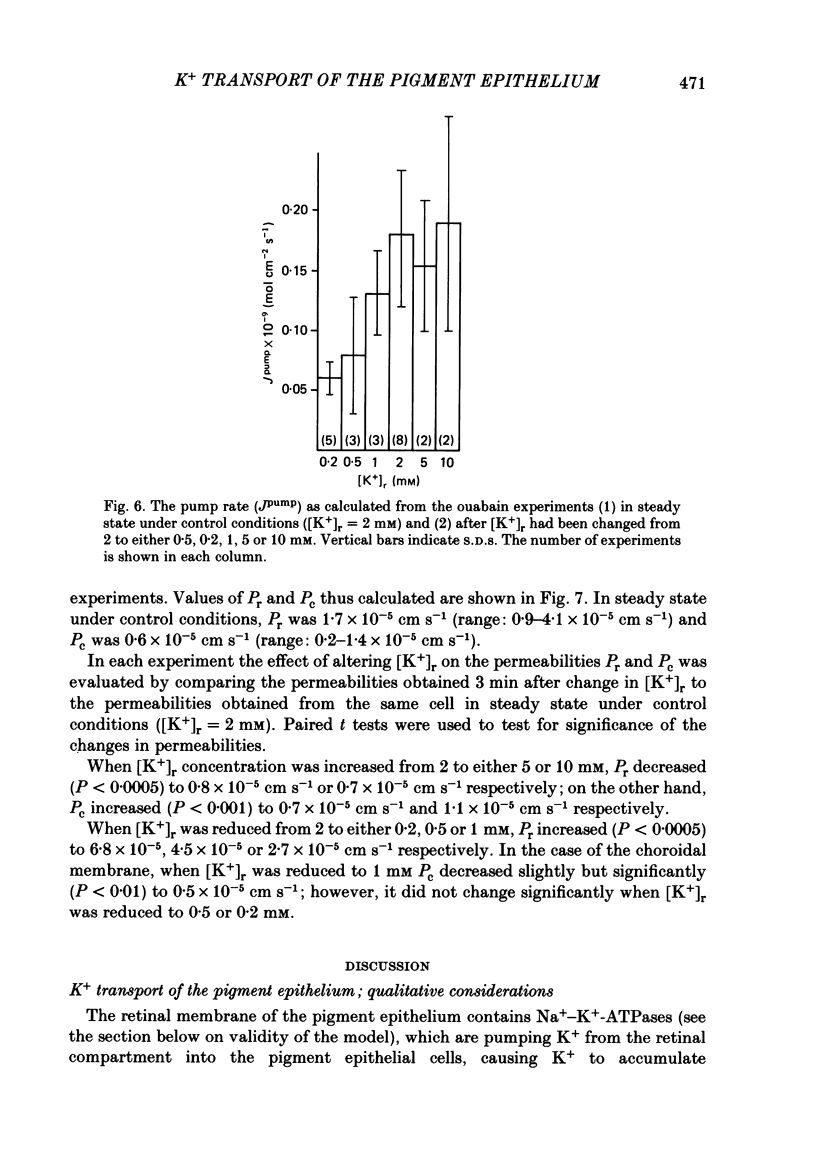
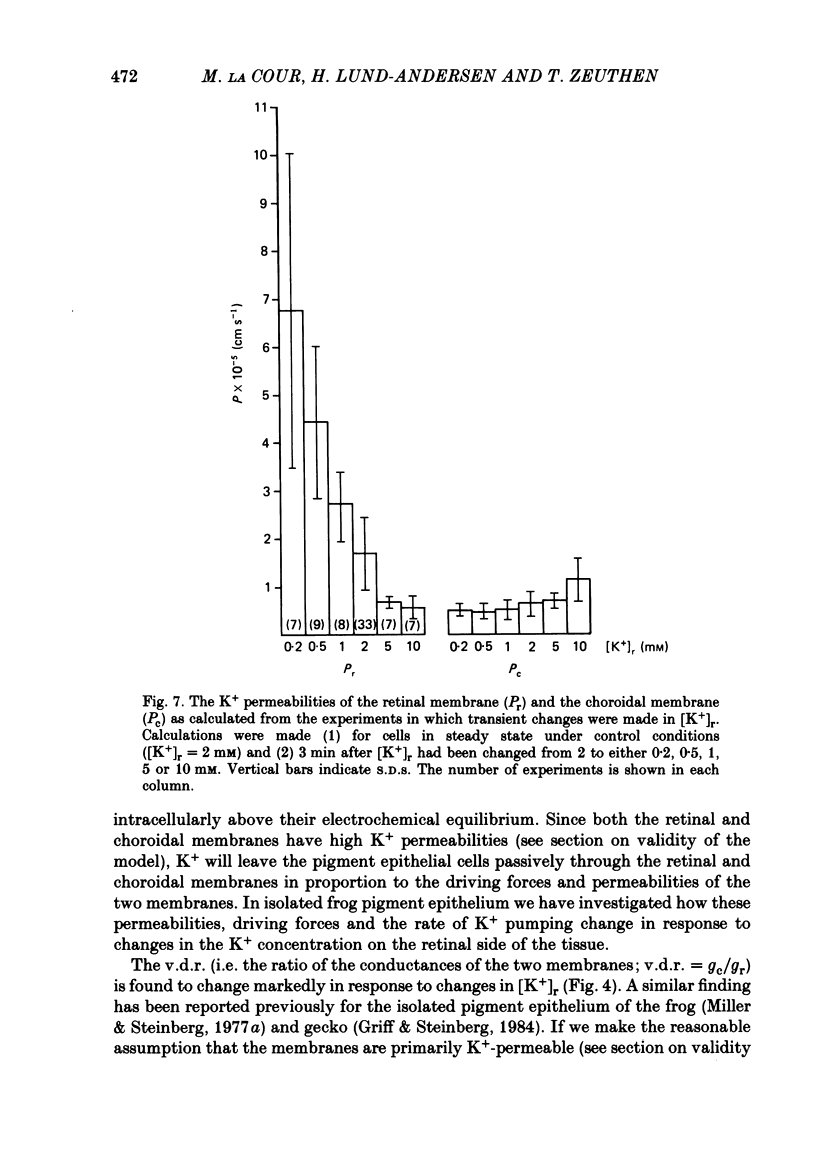
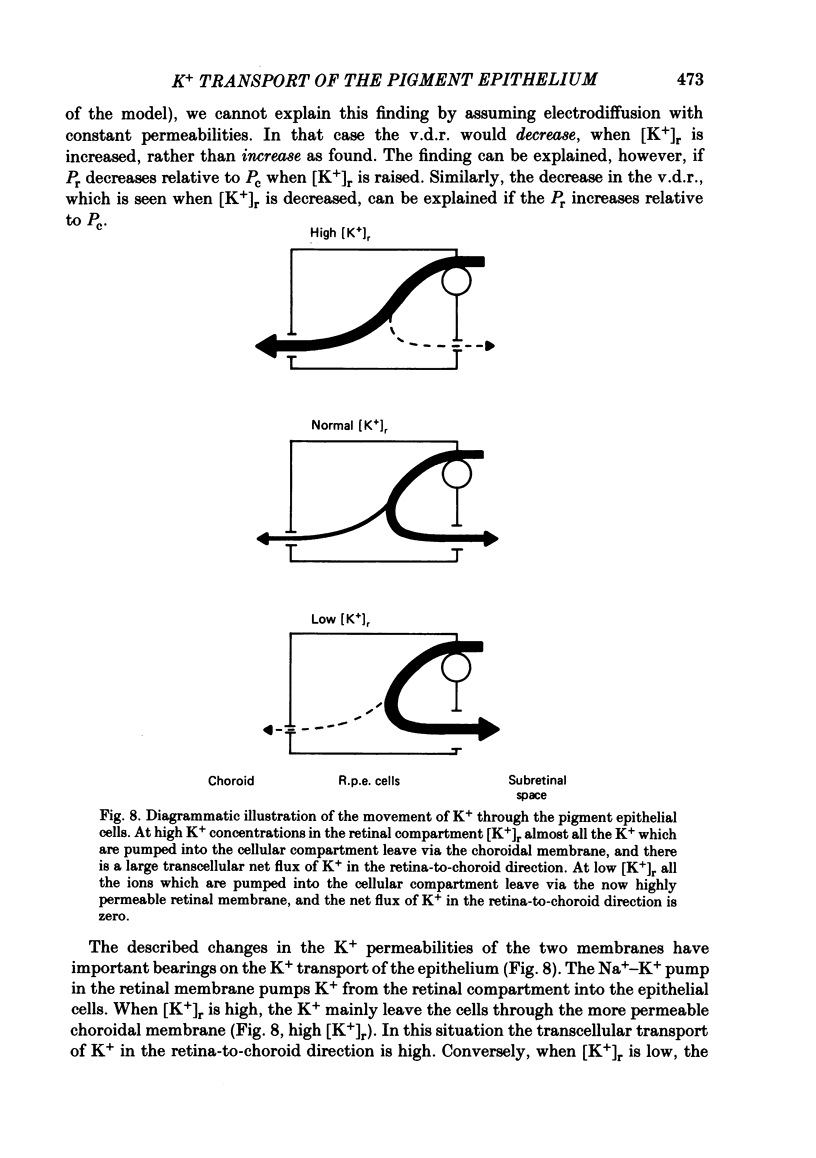
The K+ transport of the isolated retinal pigment epithelium from the bull-frog was studied using micropuncture with double-barrelled ion-selective micro-electrodes. Transient changes of intracellular values of electrical potential and K+ activity were monitored in response to abrupt changes in the K+ concentration on the retinal side of the tissue. The data were interpreted in terms of a simple three-compartment model of the epithelium in which the retinal (or apical) and choroidal (or basal) membranes separate the cellular compartment from the retinal and choroidal compartments. K+ transport across the retinal membrane was described by an active ouabain-sensitive K+ influx in parallel with a passive electrodiffusive K+ efflux. In steady state under control conditions, the active K+ influx (pump rate) averaged 0.18 X 10(-9) mol cm-2 s-1. The electrodiffusive K+ efflux was described by a K+ permeability, which in steady state under control conditions averaged 1.7 X 10(-5) cm s-1. K+ transport across the choroidal membrane was described as purely electrodiffusive. In steady state under control conditions, the K+ permeability of the choroidal membrane averaged 0.6 X 10(-5) cm s-1. When the K+ concentration on the retinal side of the tissue was increased from its control value, the K+ permeability of the retinal membrane decreased and the K+ permeability of the choroidal membrane increased. This caused the epithelium to attain a new steady state in which the cells transported K+ away from the retinal compartment at a high rate. When the K+ concentration on the retinal side of the tissue was decreased from its control value, the K+ permeability of the retinal membrane increased and the pump rate decreased. This caused the epithelial cells to transport K+ from the cellular compartment into the retinal compartment. In effect, the K+ transport of the retinal pigment epithelium depends on the K+ concentration in the retinal compartment in such a way as to keep variations in this concentration at a minimum.
Full text
PDF










Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Dick E., Miller R. F. Light-evoked potassium activity in mudpuppy retina: its relationship to the b-wave of the electroretinogram. Brain Res. 1978 Oct 13;154(2):388–394. doi: 10.1016/0006-8993(78)90711-4. [DOI] [PubMed] [Google Scholar]
- Garay R. P., Garrahan P. J. The interaction of sodium and potassium with the sodium pump in red cells. J Physiol. 1973 Jun;231(2):297–325. doi: 10.1113/jphysiol.1973.sp010234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griff E. R., Steinberg R. H. Changes in apical [K+] produce delayed basal membrane responses of the retinal pigment epithelium in the gecko. J Gen Physiol. 1984 Feb;83(2):193–211. doi: 10.1085/jgp.83.2.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes B. A., Miller S. S., Machen T. E. Effects of cyclic AMP on fluid absorption and ion transport across frog retinal pigment epithelium. Measurements in the open-circuit state. J Gen Physiol. 1984 Jun;83(6):875–899. doi: 10.1085/jgp.83.6.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karwoski C. J., Proenza L. M. Light-evoked changes in extracellular potassium concentration in munpuppy retina. Brain Res. 1978 Mar 10;142(3):515–530. doi: 10.1016/0006-8993(78)90913-7. [DOI] [PubMed] [Google Scholar]
- Kline R. P., Ripps H., Dowling J. E. Generation of b-wave currents in the skate retina. Proc Natl Acad Sci U S A. 1978 Nov;75(11):5727–5731. doi: 10.1073/pnas.75.11.5727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuura T., Miller W. H., Tomita T. Cone-specific c-wave in the turtle retina. Vision Res. 1978;18(7):767–775. doi: 10.1016/0042-6989(78)90115-3. [DOI] [PubMed] [Google Scholar]
- Miller S. S., Steinberg R. H. Active transport of ions across frog retinal pigment epithelium. Exp Eye Res. 1977 Sep;25(3):235–248. doi: 10.1016/0014-4835(77)90090-2. [DOI] [PubMed] [Google Scholar]
- Miller S. S., Steinberg R. H., Oakley B., 2nd The electrogenic sodium pump of the frog retinal pigment epithelium. J Membr Biol. 1978 Dec 29;44(3-4):259–279. doi: 10.1007/BF01944224. [DOI] [PubMed] [Google Scholar]
- Miller S. S., Steinberg R. H. Passive ionic properties of frog retinal pigment epithelium. J Membr Biol. 1977 Sep 15;36(4):337–372. doi: 10.1007/BF01868158. [DOI] [PubMed] [Google Scholar]
- Miller S. S., Steinberg R. H. Potassium transport across the frog retinal pigment epithelium. J Membr Biol. 1982;67(3):199–209. doi: 10.1007/BF01868661. [DOI] [PubMed] [Google Scholar]
- Newman E. A., Frambach D. A., Odette L. L. Control of extracellular potassium levels by retinal glial cell K+ siphoning. Science. 1984 Sep 14;225(4667):1174–1175. doi: 10.1126/science.6474173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman E. A., Odette L. L. Model of electroretinogram b-wave generation: a test of the K+ hypothesis. J Neurophysiol. 1984 Jan;51(1):164–182. doi: 10.1152/jn.1984.51.1.164. [DOI] [PubMed] [Google Scholar]
- Newman E. A. Regional specialization of retinal glial cell membrane. Nature. 1984 May 10;309(5964):155–157. doi: 10.1038/309155a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oakley B., 2nd, Green D. G. Correlation of light-induced changes in retinal extracellular potassium concentration with c-wave of the electroretinogram. J Neurophysiol. 1976 Sep;39(5):1117–1133. doi: 10.1152/jn.1976.39.5.1117. [DOI] [PubMed] [Google Scholar]
- Oakley B., 2nd, Miller S. S., Steinberg R. H. Effect of intracellular potassium upon the electrogenic pump of frog retinal pigment epithelium. J Membr Biol. 1978 Dec 29;44(3-4):281–307. doi: 10.1007/BF01944225. [DOI] [PubMed] [Google Scholar]
- Oakley B., 2nd Potassium and the photoreceptor-dependent pigment epithelial hyperpolarization. J Gen Physiol. 1977 Oct;70(4):405–425. doi: 10.1085/jgp.70.4.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ostwald T. J., Steinberg R. H. Localization of frog retinal pigment epithelium Na+-K+ ATPase. Exp Eye Res. 1980 Sep;31(3):351–360. doi: 10.1016/s0014-4835(80)80043-1. [DOI] [PubMed] [Google Scholar]
- Saito Y., Wright E. M. Kinetics of the sodium pump in the frog choroid plexus. J Physiol. 1982 Jul;328:229–243. doi: 10.1113/jphysiol.1982.sp014261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimazaki H., Oakley B., 2nd Reaccumulation of [K+]o in the toad retina during maintained illumination. J Gen Physiol. 1984 Sep;84(3):475–504. doi: 10.1085/jgp.84.3.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg R. H., Linsenmeier R. A., Griff E. R. Three light-evoked responses of the retinal pigment epithelium. Vision Res. 1983;23(11):1315–1323. doi: 10.1016/0042-6989(83)90107-4. [DOI] [PubMed] [Google Scholar]
- Steinberg R. H., Oakley B., 2nd, Niemeyer G. Light-evoked changes in [K+]0 in retina of intact cat eye. J Neurophysiol. 1980 Nov;44(5):897–921. doi: 10.1152/jn.1980.44.5.897. [DOI] [PubMed] [Google Scholar]
- Steinberg R. H. Scanning electron microscopy of the bullfrog's retina and pigment epithelium. Z Zellforsch Mikrosk Anat. 1973 Oct 26;143(4):451–463. doi: 10.1007/BF00306765. [DOI] [PubMed] [Google Scholar]
- Zeuthen T. Relations between intracellular ion activities and extracellular osmolarity in Necturus gallbladder epithelium. J Membr Biol. 1982;66(2):109–121. doi: 10.1007/BF01868487. [DOI] [PubMed] [Google Scholar]
- la Cour M. The retinal pigment epithelium controls the potassium activity in the subretinal space. Acta Ophthalmol Suppl. 1985;173:9–10. doi: 10.1111/j.1755-3768.1985.tb06827.x. [DOI] [PubMed] [Google Scholar]


