Abstract
Introduction
This single-centre retrospective analysis of data from three randomised studies and two expanded-access studies compared the effect of interferon (IFN)-alfa, sunitinib, and sorafenib on the occurrence and progression of metastatic bone lesions in patients with renal cell carcinoma (RCC).
Methods
The analysis included 292 patients: 107 received sunitinib 50 mg/day in 6-week cycles (Schedule 4/2), 147 received sorafenib 800 mg/day, and 38 received placebo or IFN-alfa 9 MU t.i.w.
Results
Pre-existing metastatic bone lesions were reported in 82 patients, of which 30 experienced progression. Twenty-three of 210 patients developed new bone lesions. Overall, sunitinib appeared slightly more effective than sorafenib or IFN-alfa at extending mean time to progression of pre-existing bone lesions (P = 0.057). Compared with sorafenib, sunitinib significantly decreased formation (P = 0.034) and prolonged time to occurrence of new bone lesions (P = 0.047).
Conclusion
Further evaluation of the effect of these therapies on bone metastases in RCC is warranted.
Keywords: Renal cell carcinoma, Sunitinib malate, Sorafenib tosylate, IFN-alfa, Metastasis
Introduction
Renal cell carcinoma (RCC), the most common cancer of the kidney, is increasing in incidence worldwide by approximately 2% per year (Ljungberg et al. 2007). It is often asymptomatic, or associated with non-specific symptoms, and approximately 25–30% of patients have metastatic disease at diagnosis (Godley and Taylor 2001; Motzer et al. 1996). The prognosis for patients with metastatic RCC (mRCC) has been extremely poor, with a typical 5-year survival rate of ≤11% (Godley and Taylor 2001; Sachdeva et al. 2008). Although RCC metastases can occur in virtually any organ site, the most frequent sites are the lung, liver, bones, and brain (Janzen et al. 2003; Motzer et al. 1996). Among patients with RCC metastases, about 20–30% have bone involvement (Zekri et al. 2001). Bone metastases are often associated with disseminated disease (Janzen et al. 2003), most frequently affecting the axial skeleton with osteolytic lesions where bone resorption dominates over new bone formation (Coleman 1997). These metastases are associated with significant skeletal morbidity, causing bone pain, fractures, spinal cord compression, and hypercalcaemia of malignancy (Coleman 1997).
mRCC is highly resistant to conventional chemotherapy, radiotherapy, and hormonal therapy (Godley and Taylor 2001; Lilleby and Fossa 2005; Rohrmann et al. 2005). Traditionally, therefore, treatment of bone metastases has generally been considered to be palliative to relieve pain, prevent pathological fractures, and improve physiological function and mobility, and other treatments are necessary (Ljungberg et al. 2007). Systemic treatment options for patients with mRCC include immunotherapy and, more recently, targeted agents, such as the multitargeted receptor tyrosine kinase (RTK) inhibitors sunitinib malate (SUTENT®, Pfizer Inc., New York, NY, USA) and sorafenib tosylate (Nexavar®, Bayer HealthCare Pharmaceuticals Inc., Wayne, NJ, USA and Onyx Pharmaceuticals Inc., Emeryville, CA, USA) (Schoffski et al. 2006).
Cytokine immunotherapy has been in use for several decades; however, evidence suggests that only a small proportion of patients with mRCC, with a good risk profile and clear-cell subtype histology, derive clinical benefit from immunotherapy with interleukin-2 or interferon-alfa (IFN-α; EAU guidelines grade B recommendation) (Ljungberg et al. 2007). Both sunitinib and sorafenib have shown clinical activity in patients with mRCC (Escudier et al. 2007; Motzer et al. 2006, 2007; Ratain et al. 2006). Sunitinib demonstrated significantly superior efficacy compared with IFN-α in treatment-naïve patients with mRCC, and is now considered the reference standard of care in this setting (Motzer et al. 2007). In addition, the results of an ongoing expanded-access programme in patients with mRCC suggest that sunitinib is active in subpopulations not previously studied, including those with non-clear-cell histology, brain metastases, or Eastern Cooperative Oncology Group performance status (ECOG PS) ≥2 (Gore et al. 2009). Sorafenib has been approved for the second-line treatment of mRCC based on data from phase II and III clinical trials (Bukowski et al. 2007; Escudier et al. 2005; Ratain et al. 2006).
However, the efficacy of these agents in the treatment of metastatic bone lesions associated with RCC is unknown. The purpose of the present retrospective analysis was to begin to investigate the effects of sunitinib, sorafenib, or IFN-α on the occurrence and progression of bone metastases in patients with mRCC.
Methods
Study design and patients
This was a retrospective single-centre analysis of data from three randomised phase II and III clinical trials and two expanded-access studies (for sunitinib and sorafenib) in patients with mRCC which evaluated cytokine or RTK treatment (Table 1). Patients were treated between March 2004 and April 2006 at the Oncology Clinic of CSK MON WIM hospital in Warsaw, Poland. The design, rationale, and results of each of these studies have been reported separately (Escudier et al. 2007; Gore et al. 2009; Motzer et al. 2007; Ryan et al. 2007; Szczylik et al. 2007).
Table 1.
Overview of studies and treatments received
| Study type | Treatment received | Dosing | Randomisation | Total patients included in current analysis (n) |
|---|---|---|---|---|
| Phase III (Motzer et al. 2007) | Sunitinib | 50 mg/day; schedule 4/2 | 1:1 | 11 |
| IFN-α | 9 MU t.i.w. | 9 | ||
| Expanded-access (Gore et al. 2009) | Sunitinib | 50 mg/day; schedule 4/2 | N/A | 96 |
| Phase II (Szczylik et al. 2007) | Sorafeniba | 400 mg b.i.d | 1:1 | 24 |
| IFN-α | 9 MU t.i.w. | 9 | ||
| Phase III (Escudier et al. 2007) | Sorafenib | 400 mg b.i.d. | 1:1 | 49 |
| Placebo | N/A | 20 | ||
| Expanded-access (Ryan et al. 2007) | Sorafenib | 400 mg b.i.d. | N/A | 74 |
b.i.d. twice daily, N/A not applicable, t.i.w. three times per week
aSorafenib dose could be increased to 1,200 mg daily on first RCC progression
Enrolment criteria used in the three randomised phase II or III clinical trials of sunitinib and sorafenib included age ≥18 years, histologically confirmed mRCC with a clear-cell component, ECOG PS 0 or 1 and adequate organ function. Two of these trials (Motzer et al. 2007; Szczylik et al. 2007) included treatment-naïve patients only, and one (Escudier et al. 2007) included patients with ≥1 prior systemic therapy. Key eligibility criteria for the sunitinib expanded-access study included age ≥18 years, histologically confirmed mRCC, ineligibility for participation in ongoing sunitinib clinical trials, and potential to derive clinical benefit from sunitinib therapy as judged by the investigator; there were no ECOG PS limits and patients with asymptomatic brain lesions were included. The sorafenib expanded-access study enrolled patients aged ≥15 years with histologically confirmed mRCC, ineligible for participation in ongoing sorafenib clinical trials, and with ECOG PS between 0 and 2.
Each study was approved by the institutional review board/ethics committee at the participating centre, and was conducted in accordance with Good Clinical Practice Guidelines. All patients provided written informed consent prior to any study procedures.
Treatment groups analysed
Treatment-naïve patients received IFN-α subcutaneously at 9 MU three times per week, sunitinib administered orally at 50 mg once daily in 6-week cycles of 4 weeks on treatment followed by 2 weeks off treatment (Schedule 4/2), or sorafenib orally at 400 mg twice daily (b.i.d.), with the possibility to increase the daily dose to 1,200 mg on first RCC progression (Table 1). Pre-treated patients received placebo, sunitinib 50 mg/day on Schedule 4/2, or sorafenib 400 mg b.i.d. Dose reductions could be carried out in response to treatment-related adverse events or intolerance. In addition, patients experiencing disease progression while receiving IFN-α or placebo could cross over to the sunitinib or sorafenib treatment groups within the appropriate studies.
Outcomes
The incidences of progression of pre-existing bone lesions and new metastatic bone lesions were measured. The mean time to progression of pre-existing or new metastatic bone lesions was also assessed. Progression of pre-existing lesions was defined as the deterioration of symptoms, requiring intensification of concomitant treatment (including palliative radiotherapy of affected sites) or the appearance of additional bone tumours. For patients without bone involvement at baseline, progression was defined as the occurrence of bone metastatic lesion(s) observed through X-rays, computed tomography (CT), or bone scans. In each study, baseline measurements were collected in patients only if specified by the study protocol. Due to the retrospective nature of this analysis, bone involvement was measured only in those patients where bone involvement was suspected (CT and bone scans) and followed up with X-rays.
Study assessments
Clinical examination and CT were used to assess disease stage at baseline (prior to study treatment). Baseline examinations were also extended to include bone scans verified by X-ray if required by study protocol or if bone involvement was suspected. Follow-up tumour assessments were carried out subsequently using clinical and radiological examinations (X-rays, CT, and bone scans) every 2–4 months.
Statistical analyses
Comparisons between treatment groups were performed using χ2 tests for differences in the incidence of progressive disease verified for statistical significance with the Yates two-sided test at the 5% significance level. Time to tumour lesion progression curves for the treatment groups were generated with the Kaplan–Meier survival analysis method and compared using a log-rank test. Patients treated with second-line sunitinib or sorafenib were excluded from the comparison with the IFN-α/placebo group because of the crossover from IFN-α/placebo to RTK treatment in the second-line studies. All statistical analyses were performed using STATISTICA 6.0 software.
Results
Patient characteristics and treatment
A total of 292 patients was included in the present analysis: 20 patients received placebo, 18 patients received IFN-α, 107 patients received sunitinib (11 first-line and 96 second-line), and 147 patients received sorafenib (67 first-line and 80 second-line; Table 1).
Overall, the median age was 57.8 years (range 25.6–78.3), 74% were male and 282 patients (97%) had prior nephrectomy. Pre-existing bone lesions were present in similar proportions across the treatment groups; 8 (21%), 32 (30%), and 42 (29%) patients treated with IFN-α/placebo, sunitinib, and sorafenib, respectively, had pre-existing lesions (Table 2). Of these 82 patients, 35 (43%) had received prior palliative radiotherapy at least 28 days prior to the initiation of therapy with IFN-α/placebo, sunitinib, or sorafenib.
Table 2.
Patient demographics and baseline disease characteristics
| Characteristics | IFN-α/placebo | Sunitinib | Sorafenib | Total | ||
|---|---|---|---|---|---|---|
| (n = 38) | First-line (n = 11) | Second-line (n = 96) | First-line (n = 67) | Second-line (n = 80) | (n = 292) | |
| Median age, years (range) | 59.1 (37.2–74.1) | 55.2 (37.7–70.3) | 56.4 (25.6–75.4) | 60.2 (37.3–75.4) | 56.8 (32.2–78.3) | 57.8 (25.6–78.3) |
| Gender, n (%) | ||||||
| Male | 29 (76) | 10 (91) | 71 (74) | 49 (73) | 57 (71) | 216 (74) |
| Female | 9 (24) | 1 (9) | 25 (26) | 18 (27) | 23 (29) | 77 (26) |
| Nephrectomy, n (%) | 38 (100) | 11 (100) | 92 (96) | 62 (95) | 79 (99) | 282 (97) |
| Pre-existing bone lesions, n (%) | 8 (21) | 4 (36) | 28 (29) | 22 (33) | 20 (25) | 82 (28) |
| Metastatic sites, n (%) | ||||||
| Lung | 29 (76) | 8 (73) | 70 (73) | 56 (84) | 70 (88) | 233 (80) |
| Liver | 7 (18) | 3 (27) | 36 (38) | 20 (30) | 14 (18) | 80 (27) |
| Brain | 0 (0) | 2 (18) | 7 (7) | 3 (5) | 5 (6) | 17 (6) |
| ECOG PS, n (%) | ||||||
| 0 | 24 (63) | 8 (73) | 35 (36) | 22 (33) | 41 (51) | 130 (44) |
| 1 | 14 (37) | 3 (27) | 54 (56) | 40 (60) | 34 (43) | 145 (50) |
| 2 | 0 (0) | 0 (0) | 7 (7) | 5 (7) | 5 (6) | 17 (6) |
| MSKCC risk group, n (%) | ||||||
| Favourable | 3 (8) | 0 (0) | 11 (11) | 8 (12) | 14 (18) | 36 (12) |
| Intermediate | 35 (92) | 11 (100) | 82 (85) | 40 (60) | 60 (75) | 228 (78) |
| Poor | 0 (0) | 0 (0) | 3 (3) | 19 (28) | 6 (8) | 28 (10) |
ECOG PS Eastern Cooperative Oncology Group performance status, MSKCC Memorial Sloan-Kettering Cancer Center
Due to the retrospective nature of the analysis, patients’ baseline characteristics differed with respect to metastases and MSKCC risk groups. While lung metastases were present at baseline in similar proportions in patients, there were variations with respect to liver metastases. Brain lesions were present in similar proportions of patients treated with sunitinib and sorafenib (8 and 5%, respectively) although none was present in patients treated with IFN-α or placebo. More patients receiving first-line sunitinib had brain metastases than those receiving first-line sorafenib (18 vs. 6%, respectively). However, the small number of treatment-naive patients in the sunitinib group did not allow a more effective comparison. Similar numbers of patients in both groups had brain metastases. Similar proportions of treatment-naïve patients in the sorafenib and sunitinib groups had lung and liver metastases. In the second-line setting, patients receiving sunitinib included a significantly larger proportion with liver metastases than those receiving sorafenib (P = 0.006), while the proportion of patients with lung metastases was significantly higher in the sorafenib group than the sunitinib group (P = 0.0027).
Overall, the majority of patients had ECOG PS 0 (44%) or 1 (50%) and were grouped in the MSKCC intermediate-risk category (78%). A larger proportion of the patients treated with IFN-α or placebo (63%) had ECOG PS 0 than those treated with the RTK inhibitors (42%). None of the IFN-α- or placebo-treated patients was considered at poor risk, and 10% of patients treated with the RTK inhibitors were considered at poor risk. A lower proportion of the patients treated with sorafenib were at intermediate risk than those treated with sunitinib (68 and 87%, respectively). All patients receiving sunitinib in the first-line setting were at intermediate risk and none had ECOG PS 2. Conversely, significantly more sorafenib-treated patients were in the MSKCC poor-risk category than in the sunitinib group (17 and 3%, respectively; P = 0.0008). Patient demographics and baseline characteristics are shown in Table 2.
Disease progression occurred in 12 patients treated with IFN-α or placebo. These patients did not cross-over to treatment with either RTK inhibitor and were therefore included in the subsequent analysis. The treatment received in each study is summarised in Table 1. At the time of analysis, median follow-up was 11.7 months (range 0.5–41.5). The median treatment duration was 7.3 months (range 0.9–25.5) with IFN-α. The median treatment duration in the first-line setting was 15.1 months (range 3.6–24.4) for sunitinib and 10.6 months (range 0.9–25.5) for sorafenib. In the second-line setting, median treatment duration was 11.5 months (range 0.9–22.0) and 15.5 months (range 0.4–41.5), respectively, for sunitinib and sorafenib. The combined median first- and second-line treatment durations were 11.8 months (range 0.9–24.2) for sunitinib and 7.3 months (range 0.4–41.5) for sorafenib.
Progression of pre-existing metastatic bone lesions
In patients treated with IFN-α in the first-line setting (n = 5), progression of pre-existing metastatic bone lesions occurred in 3 (60.0%) patients. Progression of pre-existing metastatic bone lesions occurred in 1 of 4 patients (25.0%) and 9 of 22 patients (40.9%) receiving first-line sunitinib and sorafenib treatment, respectively (Table 3). The mean time to progression of the pre-existing bone lesions in patients receiving first-line IFN-α, sunitinib, or sorafenib was 12.0, 22.4, and 6.5 months, respectively. The difference between sunitinib and sorafenib with respect to time to progression of pre-existing lesions was not significant (P = 0.96). Comparison between the patients treated with IFN-α and those treated with sunitinib, sorafenib, and the RTK inhibitors combined indicated that the differences were not significant with respect to mean time to progression of pre-existing lesions (P = 0.25, P = 0.97, and P = 0.53, respectively).
Table 3.
Progression of pre-existing metastatic bone lesions in patients with mRCC (n = 82)
| Lesions with progression, n (%) | Mean time to lesion progression (months) | |
|---|---|---|
| First-line treatment | ||
| IFN-α (n = 5) | 3 (60.0) | 12.0 |
| Sunitinib (n = 4) | 1 (25.0) | 22.4 |
| Sorafenib (n = 22) | 9 (40.9) | 6.5 |
| χ2 sunitinib vs. sorafenib | – | 0.36 |
| P (Yates) | – | 0.96 |
| Second-line treatment | ||
| Placebo (n = 3) | 2 (66.6) | 5.6 |
| Sunitinib (n = 28) | 8 (28.6) | 10.1 |
| Sorafenib (n = 20) | 9 (45.0) | 7.0 |
| χ2 sunitinib vs. sorafenib | – | 1.38 |
| P (Yates) | – | 0.38 |
In the second-line setting, 2 of 3 patients (66.6%) receiving placebo; 8 of 28 patients (28.6%) receiving sunitinib, and 9 of 20 patients (45%) treated with sorafenib had progression of pre-existing lesions (Table 3). Compared with first-line treatment, there were smaller differences between the treatment groups with respect to mean time to pre-existing lesion progression; 5.6, 10.1, and 7.0 months in patients treated with placebo, sunitinib, and sorafenib, respectively. The mean time to pre-existing lesion progression did not differ significantly between patients treated with placebo and sunitinib, sorafenib, and the RTK inhibitors combined (P = 0.14, P = 0.53, and P = 0.26, respectively; sunitinib vs. sorafenib, P = 0.38).
New metastatic bone lesions
A total of 210 patients did not have bone lesions at baseline; of these, 65 received first-line and 145 second-line treatment. In patients receiving first-line treatment, new metastatic lesions occurred in 1 of 13 patients (7.7%) treated with IFN-α, 1 of 7 patients (14.3%) treated with sunitinib, and 7 of 45 patients (15.5%) treated with sorafenib (Table 4). The mean time to occurrence of new lesions was longer for sunitinib (18.0 months) than the other agents (6.7 months for IFN-α and 8.3 months for sorafenib); however, these differences did not reach statistical significance (IFN-α vs. sunitinib, sorafenib, and RTK inhibitors combined, P = 0.36, P = 0.83, and P = 0.96, respectively; sunitinib vs. sorafenib, P = 0.63).
Table 4.
Occurrence of metastatic bone lesions in mRCC patients without lesions at baseline (n = 210)
| Lesions with progression, n (%) | Mean time to occurrence of lesion (months) | |
|---|---|---|
| First-line treatment | ||
| IFN-α (n = 13) | 1 (7.7) | 6.7 |
| Sunitinib (n = 7) | 1 (14.3) | 18.0 |
| Sorafenib (n = 45) | 7 (15.5) | 8.3 |
| χ2 sunitinib vs. sorafenib | – | 0.01 |
| P (Yates) | – | 0.63 |
| Second-line treatment | ||
| Placebo (n = 17) | 3 (17.6) | 9.5 |
| Sunitinib (n = 68) | 2 (2.0) | 11.0 |
| Sorafenib (n = 60) | 9 (15.0) | 13.1 |
| χ2 sunitinib vs. sorafenib | – | 5.9 |
| P (Yates) | – | 0.034 |
Among the patients receiving second-line treatment, 3/17 (17.6%) patients receiving placebo developed new metastatic bone lesions (Table 4). In the group of patients receiving sunitinib, a very small number (2 of 68 patients; 2.9%) had new metastatic bone lesions compared with 9 of 60 patients (15.0%) treated with sorafenib. The mean time to occurrence of new bone lesions was 9.5, 11.0, and 13.1 months, respectively, for placebo, sunitinib, and sorafenib, with the difference between sunitinib and sorafenib reaching statistical significance (P = 0.034). The difference between sunitinib and placebo was also significant (P = 0.017). However, when placebo was compared with the sorafenib and the RTK inhibitor-treated groups combined, the differences did not reach statistical significance (P = 0.27 and P = 0.1, respectively).
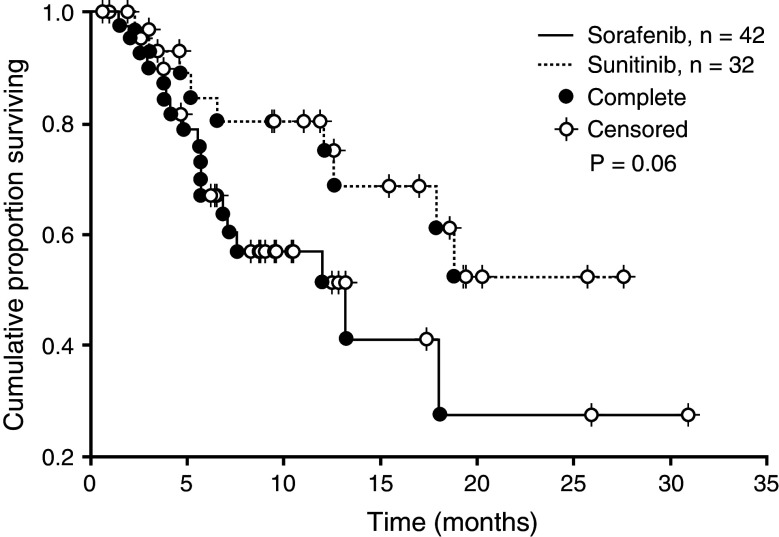
In an overall comparison of sunitinib and sorafenib (including both first- and second-line treatment), sunitinib was associated with a longer mean time to progression of pre-existing metastatic bone lesions, which was close to reaching significance (P = 0.057; Fig. 1). With respect to new metastatic bone lesions (including both first- and second-line treatment), sunitinib was found to significantly prolong mean time to occurrence when compared with sorafenib (P = 0.047; Fig. 2). Due to a logical error, an overall comparison between the RTK inhibitors combined and IFN-α was not possible.
Fig. 1.
Time to progression of pre-existing bone lesions in mRCC patients receiving sunitinib or sorafenib therapy
Fig. 2.
Time to progression as occurrence of new bone lesions in mRCC patients receiving sunitinib or sorafenib therapy
Safety
Adverse-event profiles in each of the study groups have been previously reported (Escudier et al. 2007; Gore et al. 2009; Motzer et al. 2007; Ryan et al. 2007; Szczylik et al. 2007). The tolerability profiles of sunitinib and sorafenib were consistent across the respective randomised and expanded-access studies. In general, the most common non-haematological adverse events observed with both sunitinib and sorafenib included diarrhoea, fatigue, nausea, stomatitis, hand–foot syndrome, and hypertension.
Discussion
This retrospective, single European centre analysis examined the emergence and progression of metastatic bone lesions in five studies of patients with mRCC. Treatment with sunitinib was found both to reduce the incidence of new metastatic bone lesions and significantly to prolong the mean time to occurrence of new lesions compared with sorafenib. In patients receiving first-line treatment, new metastatic lesions occurred in similar numbers of patients treated with sunitinib and sorafenib and, although not statistically significant, sunitinib appeared to prolong the mean time to occurrence of new lesions.
Similar results were observed in patients with pre-existing lesions. Although statistically not significant, both sunitinib and sorafenib prolonged mean time to progression of pre-existing bone metastases; sunitinib slightly more than sorafenib. Among treatment-naïve patients receiving sorafenib, 9 of 22 (40.9%) had progression of the pre-existing lesions, while in sunitinib-treated patients, progression occurred in 1 of 4 (25.0%) patients. The proportion of patients in each treatment group with progression of pre-existing lesions was similar in the pre-treated patients. Additionally, the mean time to lesion progression was longer for sunitinib-treated patients in both the first- and second-line settings than for sorafenib-treated patients.
The presence of bone metastases is a poor prognostic factor for response to therapy and overall survival in patients with mRCC (Negrier et al. 2002). Currently, palliative therapy for bone metastases and related pain relies on treatment with bisphosphonates and radiation therapy. Treatment with radiation therapy or with bisphosphonates, such as zoledronic acid, alone or in combination, may alleviate bone pain caused by metastases and increase patients’ quality of life (Costa and Major 2009; Ljungberg et al. 2007).
In this study, progression of pre-existing lesions remained unaffected by prior radiotherapy (patients with prior radiotherapy vs. patients without prior radiotherapy, P = 0.76; data not shown). Overall, 32/82 (39%) patients treated in the first- and second-line settings experienced progression of pre-existing bone lesions. Thirteen of these 32 patients (41%) had received prior treatment with radiotherapy while 19 (59%) patients had not. Further examination indicated that eight sorafenib-treated patients and four sunitinib-treated patients with progression of pre-existing lesions had received prior radiotherapy. No significant differences were found between the two groups (sunitinib vs. sorafenib; P = 0.59). Bisphosphonates, including pamidronic acid (n = 28), clodronic acid (n = 15), and zoledronic acid (n = 4), were used as concomitant therapy in 51/82 (62%) patients with pre-existing lesions (data not shown). As with prior radiotherapy, concomitant treatment with bisphosphonates did not affect progression of pre-existing lesions when treating patients with the RTK inhibitors; 15 sorafenib-treated patients and eight sunitinib-treated patients with progression of pre-existing lesions received concomitant treatment with bisphosphonates. No differences were observed between the two treatment groups (sunitinib vs. sorafenib; P = 0.14). The impact of prior radiotherapy or concomitant bisphosphonate treatment was not examined in patients with the occurrence of new lesions as these were employed after the patients had reached the primary study endpoint of the disease progression in the skeleton.
The significant morbidity attached to bone lesions underscores the need for effective therapy for these patients. The biological processes regulating bone metastases are still not clearly understood. The high vascularity and rich bone microenvironment may encourage tumour cell growth through increased expression of growth factors and mediators of osteolysis or osteoblastic metastasis (Kominsky et al. 2007; Mundy 2002). Bone lesions in RCC are well vascularised and osteolytic although the exact interactions between the bone microenvironment and RCC cells remain unknown (Kominsky et al. 2007). RCC cells have been shown to overexpress various growth factors, including epidermal growth factor (EGF), which are associated with the development and progression of mRCC and the induction of angiogenic factors such as vascular endothelial growth factor (VEGF) (Weber et al. 2003).
Thus, inhibition of this signalling cascade could inhibit tumour angiogenesis and growth. Blocking EGF receptor activity through the use of PKI 166, a tyrosine kinase inhibitor, resulted in decreased tumour growth and bone lysis in nude mice implanted with human RCC cells obtained from a bone metastasis (Weber et al. 2003). Combining PKI 166 with paclitaxel also resulted in tumour cell and tumour-associated endothelial cell apoptosis and decreased VEGF expression. Similarly, combining a targeted agent such as sunitinib or sorafenib with radiation therapy may result in radio sensitisation or increase sensitivity to the inhibitory effects of the targeted agent (Plastaras et al. 2007; Staehler et al. 2009; Taussky and Soulières 2009). Sunitinib is an oral, multitargeted RTK inhibitor that targets the major subtypes of VEGF receptor (VEGFR-1, -2, and -3) and platelet-derived growth factor receptor (PDGFR-α and -β), stem cell factor receptor (c-KIT), colony stimulating factor receptor type 1 (CSF-1R), FMS-like tyrosine kinase-3 receptor (FLT3), and the glial cell-line-derived neurotrophic factor receptor (RET) (Abrams et al. 2003; Kim et al. 2006; Mendel et al. 2003; O’Farrell et al. 2003). Sorafenib, also an oral, multitargeted kinase inhibitor acts through inhibition of VEGFR-2 and -3, PDGFR-β, FLT3, c-KIT, RET, B-Raf and Raf-1/C-Raf (Beeram et al. 2005; Wilhelm and Chien 2002).
The data used for the present analysis were collected at a single centre, which should preclude variations in consistency of data gathering and analysis. However, this is a retrospective analysis and the results should be considered in that context. The results provide an indication of the potential for targeted therapy in the treatment of bone lesions. In patients without skeletal involvement at the initiation of systemic therapy, progression of mRCC with respect to bone lesions is easily defined through the occurrence of any osteolytic lesions. However, no clear rules define progression in patients with metastatic bone lesions already present at systemic therapy initiation. According to Response Evaluation Criteria in Solid Tumours criteria, metastatic bone tumours represent non-target lesions and only the clear occurrence of additional new metastases can be objectively evaluated as progression. In many cases, such assessments are subjective, rendering comparisons between studies difficult. We, therefore, defined progression as the deterioration of symptoms caused by the bone lesions requiring the intensification of concomitant treatment (including increasing doses of analgesics or opioids and/or palliative radiotherapy of affected site).
In order to evaluate the effect of various treatment options, specifically, IFN-α, sunitinib, sorafenib, or best supportive care (placebo), on the incidence of progression of pre-existing bone metastatic lesions or the formation of new lesions, standard statistical methods have been used in this retrospective analysis. The use of Kaplan–Meier survival analysis verified by log-rank testing allowed assessment of time to lesion progression and the comparison of this parameter between the subgroups. Although in some subgroup analyses, statistical significance was not reached due to the small group sizes, overall, a positive trend towards a longer time to lesion progression was observed in those patients treated with sunitinib.
In summary, these findings from a retrospective analysis add support to the existing data showing that RTK inhibitors are an effective treatment option for mRCC. Further study on the effects of RTK inhibitors, specifically sunitinib, is warranted in bone lesions in mRCC to improve the management and treatment of bone metastases in RCC.
Acknowledgments
Medical writing support was provided by ACUMED® (Tytherington, UK), with funding from Pfizer Inc.
Conflict of interest statement
The authors have no financial interest to disclose except for Prof. Cezary Szczylik who is a member of advisory boards of pharmaceutical companies: Bayer-Schering, Pfizer and Wyeth.
References
- Abrams TJ, Lee LB, Murray LJ, Pryer NK, Cherrington JM (2003) SU11248 inhibits KIT and platelet-derived growth factor receptor beta in preclinical models of human small cell lung cancer. Mol Cancer Ther 2:471–478 [PubMed] [Google Scholar]
- Beeram M, Patnaik A, Rowinsky EK (2005) Raf: a strategic target for therapeutic development against cancer. J Clin Oncol 23:6771–6790 [DOI] [PubMed] [Google Scholar]
- Bukowski R, Szczylik C, Stadler W, Simantov R, Shan M (2007) Final results of the randomized phase III trial of sorafenib in advanced renal cell carcinoma: survival and biomarker analysis. J Clin Oncol 25(18S):240s (Abstract 5023) [Google Scholar]
- Coleman RE (1997) Skeletal complications of malignancy. Cancer 80:1588–1594 [DOI] [PubMed] [Google Scholar]
- Costa L, Major PP (2009) Effect of bisphosphonates on pain and quality of life in patients with bone metastases. Nat Clin Pract Oncol 6:163–174 [DOI] [PubMed] [Google Scholar]
- Escudier B, Szczylik C, Eisen T, Stadler WM, Schwartz B, Shan M, Bukowski RM (2005) Randomized phase III trial of the Raf kinase and VEGFR inhibitor sorafenib (BAY 43-9006) in patients with advanced renal cell carcinoma (RCC). J Clin Oncol 23(16S):380s (Abstract LBA4510) [Google Scholar]
- Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, Negrier S, Chevreau C, Solska E, Desai AA, Rolland F, Demkow T, Hutson TE, Gore M, Freeman S, Schwartz B, Shan M, Simantov R, Bukowski RM (2007) Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 356:125–134 [DOI] [PubMed] [Google Scholar]
- Godley P, Taylor M (2001) Renal cell carcinoma. Curr Opin Oncol 13:199–203 [DOI] [PubMed] [Google Scholar]
- Gore M, Szczylik C, Porta C, Bracarda S, Hawkins R, Bjarnason G, Oudard S, Lee SH, Haanen J, Castellano D, Vrdoljak E, Schoffski P, Mainwaring P, Nieto A, Yuan J, Bukowski R (2009) Safety and efficacy of sunitinib with subpopulation analysis in an expanded-access trial of metastatic renal cell carcinoma. Lancet Oncol 10:757–763 [DOI] [PubMed] [Google Scholar]
- Janzen NK, Kim HL, Figlin RA, Belldegrun AS (2003) Surveillance after radical or partial nephrectomy for localized renal cell carcinoma and management of recurrent disease. Urol Clin North Am 30:843–852 [DOI] [PubMed] [Google Scholar]
- Kim DW, Jo YS, Jung HS, Chung HK, Song JH, Park KC, Park SH, Hwang JH, Rha SY, Kweon GR, Lee SJ, Jo KW, Shong M (2006) An orally administered multitarget tyrosine kinase inhibitor, SU11248, is a novel potent inhibitor of thyroid oncogenic RET/papillary thyroid cancer kinases. J Clin Endocrinol Metab 91:4070–4076 [DOI] [PubMed] [Google Scholar]
- Kominsky SL, Doucet M, Brady K, Weber KL (2007) TGF-β promotes the establishment of renal cell carcinoma bone metastasis. J Bone Miner Res 21:37–44 [DOI] [PubMed] [Google Scholar]
- Lilleby W, Fossa SD (2005) Chemotherapy in metastatic renal cell cancer. World J Urol 23:175–179 [DOI] [PubMed] [Google Scholar]
- Ljungberg B, Hanbury D, Kuczyk M (2007) Renal cell carcinoma guideline. Eur Urol 51:1502–1510 [DOI] [PubMed] [Google Scholar]
- Mendel DB, Laird AD, Xin X, Louie SG, Christensen JG, Li G, Schreck RE, Abrams TJ, Ngai TJ, Lee LB, Murray LJ, Carver J, Chan E, Moss KG, Haznedar JO, Sukbuntherng J, Blake RA, Sun L, Tang C, Miller T, Shirazian S, McMahon G, Cherrington JM (2003) In vivo antitumor activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: determination of a pharmacokinetic/pharmacodynamic relationship. Clin Cancer Res 9:327–337 [PubMed] [Google Scholar]
- Motzer RJ, Bander NH, Nanus DM (1996) Renal-cell carcinoma. N Engl J Med 335:865–875 [DOI] [PubMed] [Google Scholar]
- Motzer RJ, Rini BI, Bukowski RM, Curti BD, George DJ, Hudes GR, Redman BG, Margolin KA, Merchan JR, Wilding G, Ginsberg MS, Bacik J, Kim ST, Baum CM, Michaelson MD (2006) Sunitinib in patients with metastatic renal cell carcinoma. JAMA 295:2516–2524 [DOI] [PubMed] [Google Scholar]
- Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, Oudard S, Negrier S, Szczylik C, Kim ST, Chen I, Bycott PW, Baum CM, Figlin RA (2007) Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 356:115–124 [DOI] [PubMed] [Google Scholar]
- Mundy GR (2002) Metastasis to bone: causes, consequences and therapeutic opportunities. Nature 2:584–593 [DOI] [PubMed] [Google Scholar]
- Negrier S, Escudier B, Gomez F, Douillard JY, Ravaud A, Chevreau C, Buclon M, Perol D, Lasset C (2002) Prognostic factors of survival and rapid progression in 782 patients with metastatic renal carcinomas treated by cytokines: a report from the Groupe Francais d’Immunotherapie. Ann Oncol 13:1460–1468 [DOI] [PubMed] [Google Scholar]
- O’Farrell AM, Abrams TJ, Yuen HA, Ngai TJ, Louie SG, Yee KW, Wong LM, Hong W, Lee LB, Town A, Smolich BD, Manning WC, Murray LJ, Heinrich MC, Cherrington JM (2003) SU11248 is a novel FLT3 tyrosine kinase inhibitor with potent activity in vitro and in vivo. Blood 101:3597–3605 [DOI] [PubMed] [Google Scholar]
- Plastaras JP, Kim S-H, Liu YY, Dicker DT, Dorsey JF, McDonough J, Cerniglia G, Rajendran RR, Gupta A, Rustgi AK, Diehl JA, Smith CD, Flaherty KT, El-Deiry WS (2007) Cell cycle-dependent and schedule-dependent antitumour effects of sorafenib combined with radiation. Cancer Res 67:9443–9454 [DOI] [PubMed] [Google Scholar]
- Ratain MJ, Eisen T, Stadler WM, Flaherty KT, Kaye SB, Rosner GL, Gore M, Desai AA, Patnaik A, Xiong HQ, Rowinsky E, Abbruzzese JL, Xia C, Simantov R, Schwartz B, O’Dwyer PJ (2006) Phase II placebo-controlled randomized discontinuation trial of sorafenib in patients with metastatic renal cell carcinoma. J Clin Oncol 24:2505–2512 [DOI] [PubMed] [Google Scholar]
- Rohrmann K, Staehler M, Haseke N, Bachmann A, Stief CG, Siebels M (2005) Immunotherapy in metastatic renal cell carcinoma. World J Urol 23:196–201 [DOI] [PubMed] [Google Scholar]
- Ryan CW, Bukowski RM, Figlin R, Knox J, Hutson TE, Dutcher JP, George J, Kirshner J, Humphrey J, Stadler WM, On behalf of the ARCCS Investigators (2007) The Advanced Renal Cell Carcinoma Sorafenib (ARCCS) expanded access trial: Long-term outcomes in first-line patients (pts). J Clin Oncol 25(18S):258s (Abstract 5096) [Google Scholar]
- Sachdeva K, Makhoul I, Javeed M, Curti B (2008) Renal cell carcinoma. eMedicine http://www.emedicine.com/MED/topic2002.htm, 1–23
- Schoffski P, Dumez H, Clement P, Hoeben A, Prenen H, Wolter P, Joniau S, Roskams T, Van PH (2006) Emerging role of tyrosine kinase inhibitors in the treatment of advanced renal cell cancer: a review. Ann Oncol 17:1185–1196 [DOI] [PubMed] [Google Scholar]
- Staehler M, Haseke N, Stadler T, Karl A, Stief CG, Wilkowski R (2009) Effectivity of radiation therapy in combination with multi-kinase inhibition based on sorafenib or sunitinib in progressive metastatic renal cancer. Eur Urol Suppl 8:156 (Abstract 143) [Google Scholar]
- Szczylik C, Demkow T, Staehler M, Rolland F, Negrier S (2007) Randomized phase II trial of first line treatment with sorafenib versus interferon in patients with advanced renal cell carcinoma: final results. J Clin Oncol 25(18S):241s (Abstract 5025) [DOI] [PubMed] [Google Scholar]
- Taussky D, Soulières D (2009) Hypofractionated radiotherapy with concomitant sunitinib—is there a radiosensitizing effect? Can J Urol 16:4599–4600 [PubMed] [Google Scholar]
- Weber KL, Doucet M, Price JE, Baker C, Kim SJ, Fidler IJ (2003) Blockade of epidermal growth factor receptor signaling leads to inhibition of renal cell carcinoma growth in the bone of nude mice. Cancer Res 63:2940–2947 [PubMed] [Google Scholar]
- Wilhelm S, Chien DS (2002) BAY 43-9006: preclinical data. Curr Pharm Des 8:2255–2257 [DOI] [PubMed] [Google Scholar]
- Zekri J, Ahmed N, Coleman RE, Hancock B (2001) The skeletal metastatic complications of renal cell carcinoma. Int J Oncol 19:379–382 [DOI] [PubMed] [Google Scholar]