Abstract
The tympanic membrane (TM) forms an impenetrable barrier to medical therapies for middle ear (ME) diseases like otitis media. By screening a phage-displayed peptide library, we have previously discovered rare peptides that mediate the active transport of cargo across the intact membrane of animals and humans. Since the M13 filamentous bacteriophage on which the peptides are expressed are large (nearly 1 µm in length), this offers the possibility of noninvasively delivering drugs, large drug packages, or gene therapy to the ME. To evaluate this possibility, EDC chemistry was employed to covalently attach amoxicillin, or neomycin molecules to phage bearing a trans-TM peptide, as a model for large drug packages. Eight hours after application of antibiotic-phage to the TM of infected rats, ME bacterial titers were substantially reduced compared to untreated animals. As a control, antibiotic was linked to wild-type phage, not bearing any peptide, and application to the TM did not affect ME bacteria. The results support the ability of rare peptides to actively deliver pharmacologically relevant amounts of drugs through the intact TM and into the ME. Moreover, since bacteriophage engineered to express peptides are viral vectors, the trans-TM peptides could also transport other viral vectors into the ME.
Keywords: Otitis media, antibiotics, transtympanic drug delivery, active transport, middle ear, otic delivery
Introduction
Middle ear (ME) diseases, such as otitis media (OM), affect up to 90% of children (Casselbrant et al., 1993; Pichichero, 2016), with 10–15% experiencing chronic or recurrent conditions (Teele et al., 1989; Rovers, 2008). According to the American Academy for Pediatrics, over 5 million acute OM (AOM) cases occur annually in US children, resulting in >10 million antibiotic prescriptions, and over 20 million visits for medical care (Kaur et al., 2017). OM is the most common condition treated with antibiotics, and increasing incidence of antibiotic resistance among the organisms responsible for AOM is a cause for concern (Kaur et al., 2017). The insertion of tympanostomy tubes is the most frequently performed ambulatory surgery on children in the United States (Rosenfeld et al., 2022). Cholesteatoma is another aggressive middle ear disease in which epidermal cells invade the ME and exhibit destructive growth that can erode middle and inner ear structures, leading to hearing loss or even complete deafness (Kuo et al., 2015).
OM is primarily treated with systemic antibiotics or, in more persistent cases through surgical interventions such as the installation of pressure equalization tubes (tympanostomy tubes) into a surgical opening in the tympanic membrane (TM) and/or adenoidectomy (Rosa-Olivares et al., 2015). While these treatments are often effective, they come with significant side effects. The widespread use of systemic antibiotics for such a common condition can lead to the development of antibiotic-resistant bacterial strains throughout the body (Fleming-Dutra et al., 2016; Gavrilovici et al., 2022). Due to effects on the gut microbiome they can also produce gastric distress, which can be particularly severe in infants (Johnston et al., 2006; Högenauer et al., 1998). Tympanostomy tubes can lead to TM scarring or tympanosclerosis (Sederberg-Olsen et al., 1989), which can affect hearing and lead to long-term issues. General surgery further carries risks of infection, anesthesia-related complications, and postoperative discomfort. Cholesteatoma must be surgically removed, but complete resection is frequently impossible, leading to a high risk of recurrence requiring additional surgery (Neudert et al., 2014). Drug treatments to inhibit cholesteatoma growth are valuable, but tissue growth inhibitors delivered systemically likely have significant side effects.
Local otic delivery of pharmacotherapy to ME offers a promising alternative to systemic drug administration. This approach allows drugs to directly target the site of infection or inflammation, significantly reducing systemic exposure and minimizing the risk of side effects. By greatly reducing the amounts reaching the gut and other systems, local otic delivery can also limit the development of antibiotic-resistant bacteria in non-targeted sites, which is a critical concern with systemic antibiotics (Hoskison et al., 2013). Moreover, this method can enhance the effectiveness of treatment by achieving higher local drug concentrations, improving therapeutic outcomes for conditions like otitis media, without overloading the body with high systemic doses. However, the TM is an impermeable structure. Surgically breaching the TM is currently required for local drug delivery to the ear. Previous experimental studies have used tissue permeants (Yang et al., 2016) or magnetically driven nanoparticles (Shapiro et al., 2014) to transport drugs across the TM. However, no noninvasive methods have yet been approved for safe and effective use for human application.
Many epithelial barriers have mechanisms for the active transport of macromolecules across their cells (Ménard et al., 2010; Wu et al., 2016). To determine whether this might also be true for the TM, we used the technique of phage display to search for peptides that would be able to cross the membrane. This method, first introduced in 1985 (Smith, 1985), has been used in numerous biological systems to identify peptides and other molecules with specific properties, including drug delivery. The technique employs large combinatorial libraries of bacteriophage, each expressing a random peptide on its surface. The libraries are typically screened through multiple rounds of biopanning for specific structural and/or functional characteristics (Wu et al., 2016). Utilization of phage display can be done without requiring prior knowledge of the structure or necessary interacting ligands. It is based on biological activity outcome to identify a peptide sequence that will interact with the desired biologic target or process. We chose to apply this method on the TM) aiming to identify a cohort of peptide that can target the TM and enter the ME. Repeated sequential screening of the library for trans-TM transport can led to collapse of a naïve library expressing a very large number of peptides to a very small number of clones sequences that can enter the ME.
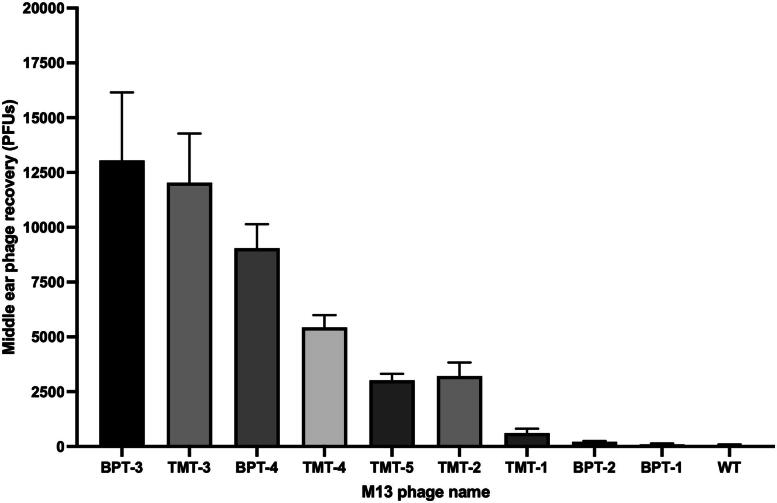
We screened a 12-mer phage library (PhD-12™, New England Biolabs) containing M13 bacteriophage each expressing one of 1010 peptides, as described previously (Kurabi et al., 2016). We tested two screening methods. In the first method, we applied the library to the TM for 2 h, amplified the ME contents in phage host bacteria, and re-applied the resultant library to the TM, for a total of three rounds. For the second method, we employed sequential screening, first for peptides that bound to the TM, then for those that were internalized, and finally for those that penetrated the TM. For each strategy we sequenced the inserts of 30 phage from the final round. The first strategy resulted in library collapse to four peptides (TMT 1–4), with one related peptide (TMT5) found on a repeat screen. The second strategy resulted in sixteen peptide sequences, of which four (BPT 1–4) were recovered more than once in our sequenced sample. In all, the nine peptide phage all crossed the TM more efficiently than wild-type (WT) phage without a peptide, although a range of transport efficiencies was observed (Figure 2). Peptide phage movement across the TM was oxygen and temperature dependent, suggesting active transport. Peptides linked to a DNA template were transported at rates similar to those for peptide phage, indicating that phage is not required for transport (Kurabi et al., 2018). Subsequent structural (Kurabi et al., 2017) and inhibitor studies revealed that transport occurred by the process of transcytosis (Kurabi et al., 2022). Finally, an in vitro assay was used to demonstrate peptide-mediated transport across the intact human TM (Kurabi et al., 2018).
Figure 2.
Phage recovery from the middle ear for nine trans-TM peptide phage, reflecting a range of peptide transport rates (adapted from Kurabi et al., 2016). The initial titers were standardized at 109 PFU/μL. This study utilized the TMT3 synthetic peptide and peptide phage.
These studies identified peptides with the capacity to actively cross the TM carrying large (∼1 µm) phage or DNA cargo. The trans-TM peptides identified are not similar to known cell- or tissue-penetrating peptides. Trans-TM transport offers the possibility of efficiently delivering large-molecular-weight drugs, gene therapy vectors, and other cargo for the treatment of ME disease. The purpose of the present investigation was to determine whether or not peptide-mediated trans-TM transport could carry pharmacologically meaningful quantities of a drug across the intact membrane and influence ME disease.
Materials and methods
Animals
Experiments were performed using 60–90-day-old Sprague Dawley rats (Envigo). All animal studies were performed in accordance with the National Institutes of Health standards for the Care and Use of Animals in Research guidelines, and were approved by the Institutional Animal Use and Care Committee of the VA San Diego Healthcare System (VASDHS). The study is reported in accordance with ARRIVE guidelines.
Infection of the ME
Rats were anesthetized with a Ketamine rodent cocktail (4.0 mg/kg xylazine, 0.75 mg/kg acepromazine, and 40.0 mg/kg ketamine, administered intraperitoneally). The middle ear bulla was accessed ventrally via a midline incision in the neck. A small opening in the bullar bone was created using a 25 g needle, and 5 × 104 CFUs of nontypeable Haemophilus influenzae (NTHi) strain 3655, a nontypeable biotype II clinical isolate derived from an otitis media patient (Kurabi et al., 2016), were injected in 50 μl of saline through a 28 g needle at an angle designed to avoid damage to the tympanic membrane. The NTHi clinical isolate was a gift from Dr. Asa Melhus (Lund University, Sweden). The NTHi strain 3655 was characterized and validated through genome sequencing, PCR amplification using serotype-specific primers, and Gram staining.
At the end of experimental procedures, the rats were euthanized using a three-fold increase in the standard anesthetic cocktail described above, followed by decapitation. The bullae are then carefully dissected to access the middle ear.
Preparation of bacteriophage
Tetracycline resistant E. coli ER2738 carrier bacteria (New England Biolabs) was used to amplify M13 filamentous phage, allowing blue/white screening using IPTG/X-gal LB plates (isopropyl beta-D-thiogalactopyranoside and 5-bromo-4-chloro-3-indolyl-β-D-galactopyranoside) for all the subsequent titration steps. One-liter cultures of E. coli ER2738 bacteria were infected with phage and grown for 5.5 h. The bacteria were removed by centrifugation at 3000 rpms at 4 °C, and phage were then precipitated from the supernatant by the addition of 20% (wt/vol) polyethylene glycol 8000 with 2.5 M NaCl, followed by two rounds of centrifugation at 10,000 rpms at 4 °C as described previously (Kurabi et al., 2016; Kurabi et al., 2018). The final phage pellet was suspended in sterile PBS, and the phage titer was determined by applying various dilutions of the phage preparation together with top agar on LB/IPTG/X-gal agar plates. The final phage preparations were all diluted, using sterile PBS, to a phage concentration of 109 PFU/μL and stored at 4 °C.
For phage titrations from the middle ear fluid recovered, 1 μL of each sample was serially diluted into a sterile tube with LB media. This was then mixed with 200 μL E. coli ER2738 host strain, then mixed with 3 mL of liquid top agar, vortexed briefly, and poured immediately onto plates containing IPTG/X-gal. Blue plaques of bacteria on the LB/IPTG/X-gal plates were counted and multiplied by the appropriate dilution factors to convert titer results into total plaque-forming units (PFU) per present in the middle ear. Same procedure was applied to titer amplified phage bacteriophage stocks.
Linkage of antibiotic to trans-TM peptide
The trans-TM peptide TMT3 (Kurabi et al., 2016), covalently linked to either amoxicillin or ciprofloxacin, was commercially synthesized (Biosynthesis, Inc., Louisville, TX). Linkage was to the carboxy terminus of the peptide (the end which is linked to phage protein pIII in peptide-phage) allowing the presumed active end of the peptide to remain free. Antibacterial activity of the conjugate was assessed by incubation with NTHi growing in liquid culture and was compared to the unconjugated antibiotic.
Linkage of antibiotic to trans-TM bacteriophage
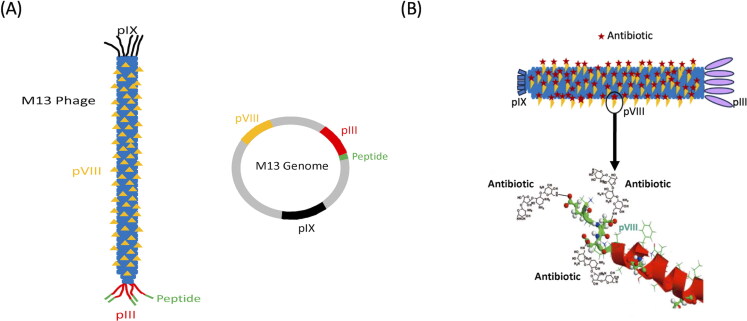
To conjugate antibiotic to bacteriophage expressing a trans-TM peptide we employed 1-Ethyl-3-[3-dimethylaminopropyl] carbodiimide hydrochloride (EDC, ThermoFisher Scientific) as a linking agent. EDC can covalently bind amoxicillin, ciprofloxacin or neomycin to the pVIII bacteriophage protein (Figure 1B). Neomycin was chosen because an EDC procedure for linkage to the phage major coat protein pVIII had previously been developed by Yacoby et al. (Yacoby et al., 2007), and shown to result in the conjugation of several hundred antibiotic molecules per individual phage. The phage pVIII protein, of which 2,700 copies are expressed along the length of the phage capsid (Henry & Pratt, 1969), contains three carboxylic amino acids (Glu2, Asp4 and Asp5) and five free amine groups (Lys8, Lys39, Lys43, Lys44, Lys48) that can be conjugated by application of EDC chemistry. Following their published protocol (Yacoby et al., 2007), two peptide-phage with high trans-TM transport characteristics, TMT3 and BPT3 (Kurabi et al., 2016), were conjugated with neomycin. TMT3 phage was similarly conjugated with amoxicillin or ciprofloxacin.
Figure 1.
(A) Schematic reconstructed representation of M13mp phage vector engineered to express a peptide (in green), whose gene is ligated into the pIII gene (in red). Each bacteriophage particle has five pIII coat protein molecules. In contrast, the cylindrical body of the phage, shown substantially shortened here for display purposes, contains thousands of pVIII coat protein molecules on its surface (in yellow). The helical pIX protein (in black) is at the other cap of the phage. The engineered peptide is displayed at the free N-terminus of pIII protein filaments. (B) Schematic diagram: Linkage of antibiotic to phage using EDC-mediated coupling (image from Yacoby et al., 2007). The red stars represent the antibiotic molecules conjugated with pVIII phage protein via EDC chemistry. For illustration, the carboxyl side chains at the N-terminal helix of pVIII major coat protein are bound to three molecules of neomycin.
Neomycin, amoxicillin or ciprofloxacin were all dissolved in 1.0 mL of conjugation buffer (0.1 M Sodium citrate buffer pH 5, 0.75 M NaCl) at a final concentration of 2.5 mM. For the conjugation, 0.4 mg of EDC reagent was added to the phage (in a 5 mL total volume of conjugation buffer) 1.0 h prior to addition of amoxicillin or neomycin, to allow binding of the appropriate EDC moiety to phage (Terminal COOH, Asp, and Glu). In the case of ciprofloxacin linking, the EDC reagent was added to ciprofloxacin first, to allow binding of the carboxylic acid side group present. This antibiotic mixture was then added to the phage. When the phage and antibiotics were mixed together after the EDC reaction initiation period, an additional 0.3 mg of EDC was added to each mixture tube, and the mixtures incubated at room temperature for 2.0 h in a rotary mixer at room temperature. The targeted drug-carrying phage were separated from the reactants by two dialysis steps (Slide-A-Lyzer Dialysis cassettes from Thermo Scientific) for 16 h against 1,000 volumes of sterile phosphate buffered saline. The antibacterial activity of each antibiotic-phage conjugate was then evaluated by incubation with NTHi in liquid culture, and compared to growth of NTHi alone and with the unbound antibiotic.
The phage concentration of the preparation was determined by measuring the absorption at 269 nm and 320 nm using Nanodrop (ThermoFisher) and then using phage concentration calculator: http://www.abdesignlabs.com/technical-resources/phage-calculator/.
Bacterial growth curves
A stock solution of NTHi (5 mL) was grown, in Brain Heart Infusion (BHI) media enriched with 5% Fildes (BD Diagnostic Systems), overnight at 37 °C with shaking. Next day, a growth curve was started by addition of 100 μL aliquot of NTHi stock culture to 1 mL test tube of fresh BHI media plus 100 μL of antibiotic solution, or antibiotic-peptide, or antibiotic-phage to be tested, and growth was followed by monitoring the OD600 every hour for the first 8 h and then 24 h later. Measurements were carried out in triplicate for each condition, and bacterial growth was analyzed using GraphPad Prism 9.0 software.
Application of antibiotic linked to peptide or phage to the TM in vivo
Immediately after NTHi inoculation of the rat ME, 18 rats were divided into 3 groups, with 6 rats per group. TMT3 peptide linked to amoxicillin (AMX) or to ciprofloxacin (CIP) was applied onto the TM in saline at 103 times the minimal effective dose, as established previously by incubation with NTHi in liquid culture, and allowed to remain on the TM for 4 h. The concentration of AMX-TMT3 peptide was 10 mg/mL, while the concentration of CIP-TMT3 peptide was 1 mg/mL. The ME contents were then removed and NTHi tittered onto chocolate agar plates. The control group had no treatment after NTHi inoculation, where the saline only was applied to TM.
TMT3 phage covalently linked to amoxicillin or neomycin, or BPT3 linked to neomycin, was similarly applied to the TM of infected MEs for 8 h. A total of 64 rats were used in this study, with 8 rats per group. The groups included: (1) TMT3 phage covalently linked to amoxicillin, (2) TMT3 phage covalently linked to neomycin, (3) BPT3 phage covalently linked to neomycin, (4) Wild-type M13 phage conjugated to amoxicillin and (5) neomycin conjugated to wild-type phage, (6) amoxicillin alone, (7) neomycin alone, (8) untreated NTHi-infected control group. NTHi titer of the ME contents were then performed for each of the individual groups respectively. The phage titer was 109 PFU/μL for all constructs applied.
Results
Peptides transport a viral expression vector through the TM
The M13 bacteriophage used to construct the phage library is a bacterial virus that specifically infects E. coli containing a F’ plasmid. The bacteriophage is employed as a viral vector, to induce expression of a modified version of the filamentous phage protein pIII by the bacterial host during phage reproduction. The modified pIII is then assembled within the bacterium onto the surface of the phage progeny. Five copies of the modified pIII are located at one end of the phage particles (Figure 1A). Each pIII has an additional 12 amino acids added to the N-terminal, which is the free end of the pIII filament. As noted above, from 1010 random peptides expressed in the library, we characterized nine that are transported at levels significantly higher than phage without a peptide. The most efficient of these peptides was recovered from the ME at titers 10,000 times higher than those observed with WT phage (Figure 2). Recovery of WT phage from the ME, which averaged around 10 phage particles, seemed most likely to consist of contamination rather than trans-TM transport.
Activity of antibiotics linked to peptide or phage
Linkage to antibiotic risks interfering with bactericidal capacity, due to steric hindrance or blockage of critical molecular sites. Thus, the antibiotic activity of peptide-antibiotic conjugates was assessed in liquid culture by evaluating the growth of nontypeable Haemophilus influenzae (NTHi) bacteria alone and with the addition of different antibiotics and drug constructs over different concentration in an 8 h time period. The minimal bactericidal concentration of amoxicillin linked to the synthetic TMT3 peptide is at ∼100 μg/mL, while that of the free antibiotic was 10 μg/mL. This reduced activity is presumably due to steric hindrance (Figure 3). In contrast, ciprofloxacin linked to the TMT3 peptide showed similar activity (MIC ∼1 μg/ml) compared to ciprofloxacin alone.
Figure 3.
Linkage to TMT3 synthetic peptide does not affect antibiotic activity. Amoxicillin (AMX) and ciprofloxacin (CIP) antibiotics were linked to the TMT3 peptide, and the activity against NTHi in liquid culture was evaluated. Recovery of NTHi after 8 h in liquid culture media with antibiotic alone or antibiotic linked to TMT3 peptide. The MIC is defined as the lowest concentration of test drug at which the % inoculum growth was maintained inhibited.
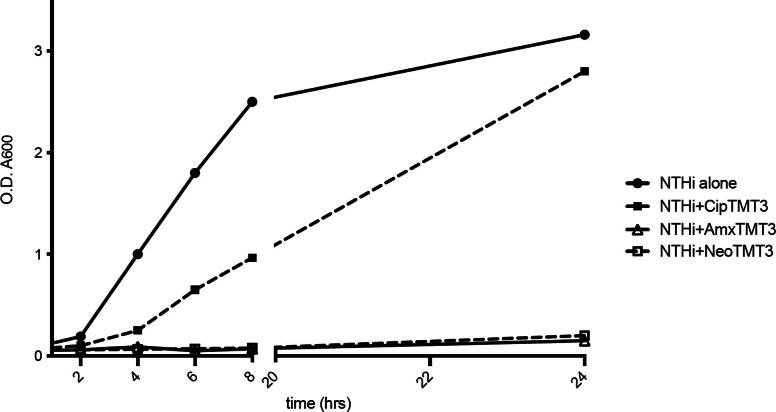
The Antibiotics, neomycin and amoxicillin cross-linked to bacteriophage expressing TMT3 retained bactericidal activity (Figure 4). Looking at the growth curves of NTHi alone (positive control) over time, it was observed that NTHi could not grow with addition of neomycin or amoxicillin conjugated phages applied at 1:10 ratio in vitro culture over 24 h. In contrast, ciprofloxacin is linked to phage and completely loses activity at 8 h. Presumably, active sites necessary for ciprofloxacin activity, such as the fluorine that endows quinolones with functional bactericidal properties and/or the extra carbon atom that dramatically enhances ciprofloxacin activity over that of norfloxacin (Wise et al., 1983), were blocked or substantially altered by the linkage chemistry.
Figure 4.
Growth curves of NTHi upon treatment with different TMT3 phage-antibiotic conjugated constructs in culture. Neomycin (Neo) and amoxicillin (AMX) molecules crosslinked to bacteriophage retained anti-bacterial activity, but ciprofloxacin (CIP) showed reduced activity. The absorbance level of the bacteria (growth) was measured by monitoring O.D. A600.
Antibiotic linked to a trans-TM peptide failed to reduce ME bacterial load
When either amoxicillin or ciprofloxacin linked to TMT3 peptide 1:1 were applied to the TMs of rats in which the ME had been inoculated with NTHi, no reduction in ME bacterial load was observed after 4 h of incubation (Figure 5A,B). The lack of activity was comparable to that observed when free antibiotic was applied to the TM. This suggested that transport of higher numbers of antibiotic molecules and/or longer dwell times on the TM might be required.
Figure 5.
Antibiotics linked to a trans-TM peptide 1:1 and applied to the TM of an infected ear had no effect on NTHi titers. The initial inoculum concentration of NTHi applied into the middle ear of rats is ∼5x103 CFUs and the drug constructs AMX linked to TMT3 peptide or CIP linked to TMT3 peptide were applied for 4h in vivo on the intact tympanic membrane. The NTHi recovery in the ME were determined from plate counts of formed NTHi colonies in the middle ear fluid (CFUs).
Multiple antibiotic molecules linked to TMT3 or BPT3 phage reduce ME bacterial infection
EDC chemistry covalently links several hundred neomycin antibiotic molecules to M13 phage (Yacoby et al., 2007), and presumably links a similar number of amoxicillin or ciprofloxacin molecules (Figure 1B) utilizing the accessible amine group to link the carboxyl residues of the phage coat proteins. We infected rat MEs with NTHi as described in methods. When amoxicillin or neomycin linked to either TMT3 or BPT3 phage was then applied to their TMs for 8 h, a significant reduction in ME bacterial titers was observed by comparison to infected MEs that were untreated (Figure 6). As expected from the in vitro assessment (Figure 4), ciprofloxacin linked to TMT3 phage had no effect in vivo. Application of experimental control WT phage linked to neomycin also did not reduce ME NTHi titers. To address whether effective bacterial control was limited to TMT3 phage linkage, we also found that neomycin linked to BPT3 produced reduction in ME NTHi that was comparable to that observed with TMT3-NEO.
Figure 6.
Control of middle earbacterial infection by phage-mediated antibiotic delivery across the tympanic membrane (TM). Application of neomycin (Neo) or amoxicillin (AMX) conjugated to a trans-TM phages TMT3 on the intact TM reduced bacterial titers in an infected middle ear compared to without treatment (NTHi alone), or with exposure of the TM to WT phage conjugated to antibiotics. The ciprofloxacin-TMT3 phage (TMT3 + CIP) did not have an effect on NTHi titer since its antibiotic activity was compromised after the EDC crosslinking reaction (see Figure 4).
Discussion
Summary
We previously observed active transport through the intact TM, mediated by rare peptides either expressed on the surface of the M13 bacteriophage or as free peptides linked to a DNA reporter (Kurabi et al., 2016; Kurabi et al., 2018). To test the ability of peptides to transport a drug, individual antibiotic molecules were linked to a peptide. Alternatively, large numbers of antibiotic molecules were linked to phage bearing a peptide as a model drug package. Single antibiotic molecules linked to trans-TM peptide and applied to the TM did not reach the ME at therapeutic concentrations. However, multiple antibiotic molecules attached to trans-TM bacteriophage achieved therapeutic ME levels within hours of application of to the TM.
Delivery of antibiotic to the ME
The results achieved with antibiotic linkage to peptide phage demonstrate that trans-TM peptides can mediate delivery of large numbers of drugs to the ME lumen within hours, quantities sufficient to be pharmacologically effective. Obviously, the use of phage as a delivery substrate is not practical for clinical use. Linkage to bacteriophage can hinder bactericidal activity, as we observed for ciprofloxacin. Alternative drug packaging methods, such as nanoparticles loaded with drugs and labeled with trans-TM peptides, would deliver active antibiotic and be appropriate for clinical use. Bacteriophage can stimulate intracellular innate immune receptors. They have also been shown to enter mammalian cell nuclei and alter gene expression or even nuclear DNA (Bodner et al., 2021). In addition, use of neomycin, used initially because of the published chemistry for linkage (Yacoby et al., 2007), is inappropriate for OM therapy, since this antibiotic may enter the inner ear via the round window membrane, and is ototoxic to inner ear sensory cells. However, the neomycin and bacteriophage linkage experiments provide an important proof of concept for the delivery of clinically relevant drug packages across the intact TM.
Peptides transport a viral expression vector through the TM
The M13 bacteriophage used to construct the phage library is a bacterial virus that infects explicitly E. coli. In the peptide phage library, bacteriophage are employed as viral vectors to induce expression of a modified version of the filamentous phage protein pIII by the bacterial host. The discovery of peptide phage that transit the TM establishes that trans-TM peptides are capable of delivering a viral expression vector, one that is much larger than an adenovirus or adeno-associated virus vector, across the intact TM. Future treatment of genetic middle ear disorders (e.g. Bootpetch et al., 2020; Hirsch et al., 2021; Paff et al., 2021), using gene therapy could be achieved via the attachment of trans-TM peptides to clinically appropriate viral vectors.
Therapeutic implications of direct, active transport across the TM
The isolation of peptides capable of actively transporting large particles (as noted above, M13 phage are approximately 1 µm in length) across the TM provides a potential targeting mechanism for the delivery of therapies into the ME. This could include drug packages, gene therapy vectors or even bactericidal phage. As noted above, the most common current treatment for OM is systemic antibiotics, with a number of problematic side effects. Local application of antimicrobials would significantly reduce exposure to bacteria throughout the body, decreasing the risk of antibiotic-resistant strains. Gastric distress and gut symptoms would also be reduced compared to oral delivery. While some local drug would exit the ME via the Eustachian tube, the amount reaching the oral cavity and gut would be far less than for the oral route, especially given the much higher systemic dose necessary to deliver an effective quantity to the ME.
While local delivery has advantages, in children it currently requires surgical intervention involving general anesthesia to penetrate the TM while avoiding damage to the delicate structures of the ossicular chain and inner ear. A noninvasive delivery mechanism would significantly enhance the practicality of local delivery of antibiotics for OM.
The simplicity of noninvasive local drug delivery would also be beneficial in developing countries for which limited access to advanced clinical care means that OM is undertreated (Arguedas et al., 2010). According to the WHO, undertreated OM causes an estimated 28,000 deaths/year due to meningitis and is responsible for half of the world’s burden of handicapping hearing loss, making it the world’s leading cause (WHO, 2004, 2020).
It should be noted that widespread antimicrobial resistance has led to a resurgence of the treatment of bacterial infections that have resisted any antibiotic using bacteriophage that are lytic to the infecting organism (Abedon et al., 2011; Lin et al., 2017; Hatfull et al., 2022) (and not linked to any drug). Such lytic bacteriophage therapy has also been effectively applied to resolve longstanding otitis externa due to antibiotic-resistant Pseudomonas infection (Wright et al., 2009). Lytic bacteriophage therapies have particular utility in that, unlike drugs, phage can evolve in step with bacteria (Teklemariam et al., 2023) reducing the development of bacterial resistance to phage (Silva-Valenzuela et al., 2022). The ability to genetically engineer lytic bacteriophages (Hussain et al., 2023) opens additional possibilities, such as inducing trans-TM peptide expression in phage that are lytic for OM organisms, offering a novel, noninvasive therapeutic option.
Bacteriophage have also been shown to infect bacteria in biofilms (Alves et al., 2014), which are widely thought to contribute to OM chronicity (Bakaletz, 2007). Although we did not specifically investigate biofilms in this study, it is important to note that local delivery of antibiotics to the middle ear may not be more effective than systemic administration in addressing biofilms. Biofilms present a significant challenge due to their resistance to conventional antibiotics, regardless of the route of delivery. However, local delivery of anti-biofilm compounds holds promise in directly targeting and disrupting biofilm formation. By applying these agents locally, it may be possible to penetrate the biofilm matrix more effectively, offering a potential advantage over systemic therapies in treating biofilm-associated infections.
Conclusions
We use phage as a model to deliver large numbers of molecules through the TM. We applied EDC chemistry to attach antibiotics to phage bearing a trans-TM peptide and observed reduction in the ME bacterial load. A number of important questions regarding the transport of peptides across the TM remain unanswered. Major issues for potential therapeutic applications are whether peptides have any negative effects on the ME or inner ear. We recently determined that direct exposure of the ME to phages bearing tympanic membrane-penetrating peptides TMT1 through TMT4 did not produce ME inflammation or impact hearing thresholds over the course of 4 days (Kurabi et al., 2018). Detailed and long-term safety studies will help determine the potential clinical potential of active trans-RWM transport. Also, since this study was performed in rats, whether antibiotics can be delivered in effective quantities through the human TM also needs to be evaluated. While both have the same three-layered structure (outer epithelial layer, middle fibrous layer, and inner mucosal layer), the human TM is larger and thicker than in the rat. Although we have shown similar peptide-phage transport across the human TM (Kurabi et al., 2018), drug transport could still be less effective.
Acknowledgements
We would like to acknowledge Mr. Kwang Pak and Mr. Eduardo Chavez for their invaluable technical assistance, and thank Dr. Asa Melhus (Lund University, Sweden) for kindly providing the nontypeable Haemophilus influenzae strain used in this study.
Funding Statement
Supported by a grant from the TRIH initiative of Action on Hearing Loss, by grants [DC012595, DC000129 and DC014801] from the NIH/NIDCD, and grant BX001205 from the Veterans Administration.
Ethical approval
Ethical approval of this study was obtained from the Institutional Animal Care and Use Committee (IACUC) of the San Diego VA Medical Center, protocol number A13-022 titled: Middle Ear Response in Otitis Media. All studies were carried out in accordance with the NIH standards for animal experimentation while adhering to ARRIVE guidelines. Rats are considered a suitable model for experimental OM because their middle ears are anatomically and histologically comparable to those of children and infants (Davidoss et al., 2018). A total of 82 rats were used across all studies. Specifically, 18 rats were sacrificed during the study investigating the application of antibiotics linked to synthetic peptides, while 64 rats were used for the study of EDC-linked antibiotics to phages applied to the tympanic membrane (TM) in vivo. All rats were housed at the VASDHS VMU facility in stable groups with standard environmental enrichment provided to all animals as part of routine care. During experimental procedures, rats were anesthetized with a Ketamine rodent cocktail (4.0 mg/kg xylazine, 0.75 mg/kg Acepromazine, and 40.0 mg/kg ketamine i.p.) for sedation, and analgesics (Buprenorphine 0.1 mg/Kg) during recovery to minimize pain. For euthanasia, a three-fold increase in the standard anesthetic cocktail described above was used followed by decapitation to ensure humane and effective procedures.
Authors contributions
A.K and A.F.R. conceived the study. A.K, and E.S. performed experiments and analyzed data. A.K. wrote the primary manuscript text and prepared the figures. A.F.R and E.S helped refine manuscript drafts. All authors reviewed and approved the manuscript. All authors agree to be accountable for all aspects of the work.
Disclosure statement
From 2008 to 2022, Dr. Allen F. Ryan was an advisor to Otonomy Inc., which developed slow-release drug treatments for the ear. This relationship has been approved by the UCSD Committee on Conflict of Interest, and the company played no part in the research presented here.
Data availability statement
The datasets used and/or analyzed during the current study available from the corresponding author on request. PCR custom primers and NTHi genome sequence are available upon request from corresponding author. Preprint of current manuscript is available at: Peptides Rapidly Transport Antibiotic Across the Intact Tympanic Membrane to Treat a Middle Ear Infection | Research Square: https://assets-eu.researchsquare.com/files/rs-3168421/v1/3cef2e03-51e5-404b-8a62-289d7e237717.pdf?c=1710149937
References
- Abedon ST, Kuhl SJ, Blasdel BG, Kutter EM. (2011). Phage treatment of human infections. Bacteriophage 1:66–85. doi: 10.4161/bact.1.2.15845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alves DR, Gaudion A, Bean JE, et al. (2014). Combined use of bacteriophage K and a novel bacteriophage to reduce Staphylococcus aureus biofilm formation. Appl Environ Microbiol 80:6694–703. doi: 10.1128/AEM.01789-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arguedas A, Kvaerner K, Liese J, et al. (2010). Otitis media across nine countries: disease burden and management. Int J Pediatr Otorhinolaryngol 74:1419–24. doi: 10.1016/j.ijporl.2010.09.022. [DOI] [PubMed] [Google Scholar]
- Bakaletz LO. (2007). Bacterial biofilms in otitis media: evidence and relevance. Pediatr Infect Dis J 26:S17–S9. doi: 10.1097/INF.0b013e318154b273. [DOI] [PubMed] [Google Scholar]
- Bodner K, Melkonian AL, Covert MW. (2021). The enemy of my enemy: new insights regarding bacteriophage-mammalian cell interactions. Trends Microbiol 29:528–41. doi: 10.1016/j.tim.2020.10.014. [DOI] [PubMed] [Google Scholar]
- Bootpetch TC, Hafrén L, Elling CL, et al. (2020). Multi-omic studies on missense PLG variants in families with otitis media. Sci Rep 10:15035. doi: 10.1038/s41598-020-70498-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casselbrant M, Mandel E, Rockette H, Bluestone C. (1993). Incidence of OM and bacteriology of acute OM during the first two years of life. In: Lim D, Bluestone C, Klein J, Nelson J, Ogra P, eds. Recent advances in otitis media. Toronto: Decker Press, 1–3. [Google Scholar]
- Davidoss NH, Varsak YK, Santa Maria PL. (2018). Animal models of acute otitis media-a review with practical implications for laboratory research. Eur Ann Otorhinolaryngol Head Neck Dis 135:183–90. doi: 10.1016/j.anorl.2017.06.013. [DOI] [PubMed] [Google Scholar]
- Fleming-Dutra KE, Hersh AL, Shapiro DJ, Jr, et al. (2016). Prevalence of inappropriate antibiotic prescriptions among us ambulatory care visits, 2010-2011. JAMA 315:1864–73. doi: 10.1001/jama.2016.4151. [DOI] [PubMed] [Google Scholar]
- Gavrilovici C, Spoială E-L, Miron I-C, et al. (2022). Acute otitis media in children-challenges of antibiotic resistance in the post-vaccination era. Microorganisms 10:1598. doi: 10.3390/microorganisms10081598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatfull GF, Dedrick RM, Schooley RT. (2022). Phage therapy for antibiotic-resistant bacterial infections. Annu Rev Med 73:197–211. doi: 10.1146/annurev-med-080219-122208. [DOI] [PubMed] [Google Scholar]
- Henry TJ, Pratt D. (1969). The proteins of bacteriophage M13. Proc Natl Acad Sci U S A 62:800–7. doi: 10.1073/pnas.62.3.800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirsch SD, Elling CL, Bootpetch TC, et al. (2021). The role of CDHR3 in susceptibility to otitis media. J Mol Med 99:1571–83. doi: 10.1007/s00109-021-02118-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Högenauer C, Hammer HF, Krejs GJ, Reisinger EC. (1998). Mechanisms and management of antibiotic-associated diarrhea. Clin Infect Dis 27:702–10. doi: 10.1086/514958. [DOI] [PubMed] [Google Scholar]
- Hoskison E, Daniel M, Al-Zahid S, et al. (2013). Drug delivery to the ear. Ther Deliv 4:115–24. doi: 10.4155/tde.12.130. [DOI] [PubMed] [Google Scholar]
- Hussain W, Yang X, Ullah M, et al. (2023). Genetic engineering of bacteriophages: key concepts, strategies, and applications. Biotechnol Adv 64:108116. doi: 10.1016/j.biotechadv.2023.108116. [DOI] [PubMed] [Google Scholar]
- Johnston BC, Supina AL, Vohra S. (2006). Probiotics for pediatric antibiotic-associated diarrhea: a meta-analysis of randomized placebo-controlled trials. CMAJ 175:377–83. doi: 10.1503/cmaj.051603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaur R, Morris M, Pichichero ME. (2017). Epidemiology of acute otitis media in the postpneumococcal conjugate vaccine era. Pediatrics 140:e20170101. doi: 10.1542/peds.2017-4067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo CL, Liao WH, Shiao AS. (2015). A review of current progress in acquired cholesteatoma management. Eur Arch Otorhinolaryngol 272:3601–9. doi: 10.1007/s00405-014-3291-0. [DOI] [PubMed] [Google Scholar]
- Kurabi A, Beasley KA, Chang L, et al. (2017). Peptides actively transported across the tympanic membrane: functional and structural properties. PLoS One 12:e0172158. doi: 10.1371/journal.pone.0172158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurabi A, Pak K, Bernhardt M, et al. (2016). Discovery of a biological mechanism of active transport through the tympanic membrane to the middle ear. Sci Rep 6:22663. doi: 10.1038/srep22663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurabi A, Pak K, Chavez E, et al. (2022). A transcytotic transport mechanism across the tympanic membrane. Sci Rep 12:984. doi: 10.1038/s41598-021-04748-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurabi A, Schaerer D, Noack V, et al. (2018). Active transport of peptides across the intact human tympanic membrane. Sci Rep 8:11815. doi: 10.1038/s41598-018-30031-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin DM, Koskella B, Lin HC. (2017). Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J Gastrointest Pharmacol Ther 8:162–73. doi: 10.4292/wjgpt.v8.i3.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ménard S, Cerf-Bensussan N, Heyman M. (2010). Multiple facets of intestinal permeability and epithelial handling of dietary antigens. Mucosal Immunol 3:247–59. doi: 10.1038/mi.2010.5. [DOI] [PubMed] [Google Scholar]
- Neudert M, Lailach S, Lasurashvili N, et al. (2014). Cholesteatoma recidivism: comparison of three different surgical techniques. Otol Neurotol 35:1801–8. doi: 10.1097/MAO.0000000000000484. [DOI] [PubMed] [Google Scholar]
- Paff T, Omran H, Nielsen KG, Haarman EG. (2021). Current and future treatments in primary ciliary dyskinesia. Int J Mol Sci 22:9834. doi: 10.3390/ijms22189834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pichichero ME. (2016). Ten-year study of acute otitis media in Rochester NY. Pediatr Infect Dis J 35:1027–32. doi: 10.1097/INF.0000000000001216. [DOI] [PubMed] [Google Scholar]
- Rosa-Olivares J, Porro A, Rodriguez-Varela M, et al. (2015). Otitis media: to treat, to refer, to do nothing: a review for the practitioner. Pediatr Rev 36:480–8. doi: 10.1542/pir.36-11-480. [DOI] [PubMed] [Google Scholar]
- Rosenfeld RM, Tunkel DE, Schwartz SR, et al. (2022). Clinical practice guideline: tympanostomy tubes in children (update). Otolaryngol Head Neck Surg 166:S1–S55. doi: 10.1177/01945998211065662. [DOI] [PubMed] [Google Scholar]
- Rovers MM. (2008). The burden of otitis media. Vaccine 26 Suppl 7:G2–4. doi: 10.1016/j.vaccine.2008.11.005. [DOI] [PubMed] [Google Scholar]
- Sederberg-Olsen JF, Sederberg-Olsen AE, Jensen AM. (1989). Late results of treatment with ventilation tubes for secretory otitis media in ENT practice. Acta Otolaryngol 108:448–55. doi: 10.3109/00016488909125552. [DOI] [PubMed] [Google Scholar]
- Shapiro B, Kulkarni S, Nacev A, et al. (2014). Shaping magnetic fields to direct therapy to ears and eyes. Annu Rev Biomed Eng 16:455–81. doi: 10.1146/annurev-bioeng-071813-105206. [DOI] [PubMed] [Google Scholar]
- Silva-Valenzuela CA, Molina-Quiroz RC, Sillankorva S. (2022). Editorial: Phage-bacteria interplay: future therapeutic approaches against antibiotic resistant bacteria. Front Cell Infect Microbiol 12:1124187. doi: 10.3389/fcimb.2022.1124187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith GP. (1985). Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface. Science 228:1315–7. doi: 10.1126/science.4001944. [DOI] [PubMed] [Google Scholar]
- Teele DW, Klein JO, Rosner B. (1989). Epidemiology of otitis media during the first seven years of life in children in greater Boston: a prospective, cohort study. J Infect Dis 160:83–94. doi: 10.1093/infdis/160.1.83. [DOI] [PubMed] [Google Scholar]
- Teklemariam AD, Al-Hindi RR, Qadri I, et al. (2023). The battle between bacteria and bacteriophages: a conundrum to their immune system. Antibiotics 12:381. doi: 10.3390/antibiotics12020381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO (World Health Organization) . (2004). http://www.who.int/pbd/deafness/activities/hearing_care/otitis_media.pdf.
- WHO (World Health Organization) . (2020). https://www.who.int/news-room/fact-sheets/detail/deafness-and-hearing-loss.
- Wise R, Andrews JM, Edwards LJ. (1983). In vitro activity of Bay 09867, a new quinoline derivative, compared with those of other antimicrobial agents. Antimicrob Agents Chemother 23:559–64. doi: 10.1128/AAC.23.4.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright A, Hawkins CH, Anggård EE, Harper DR. (2009). A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; a preliminary report of efficacy. Clin Otolaryngol 34:349–57. doi: 10.1111/j.1749-4486.2009.01973.x. [DOI] [PubMed] [Google Scholar]
- Wu CH, Liu IJ, Lu RM, Wu HC. (2016). Advancement and applications of peptide phage display technology in biomedical science. J Biomed Sci 23:8. doi: 10.1186/s12929-016-0223-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yacoby I, Bar H, Benhar I. (2007). Targeted drug-carrying bacteriophages as antibacterial nanomedicines. Antimicrob Agents Chemother 51:2156–63. doi: 10.1128/AAC.00163-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang R, Sabharwal V, Okonkwo OS, et al. (2016). Treatment of otitis media by transtympanic delivery of antibiotics. Sci Transl Med 8:356ra120. doi: 10.1126/scitranslmed.aaf4363. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study available from the corresponding author on request. PCR custom primers and NTHi genome sequence are available upon request from corresponding author. Preprint of current manuscript is available at: Peptides Rapidly Transport Antibiotic Across the Intact Tympanic Membrane to Treat a Middle Ear Infection | Research Square: https://assets-eu.researchsquare.com/files/rs-3168421/v1/3cef2e03-51e5-404b-8a62-289d7e237717.pdf?c=1710149937