Abstract
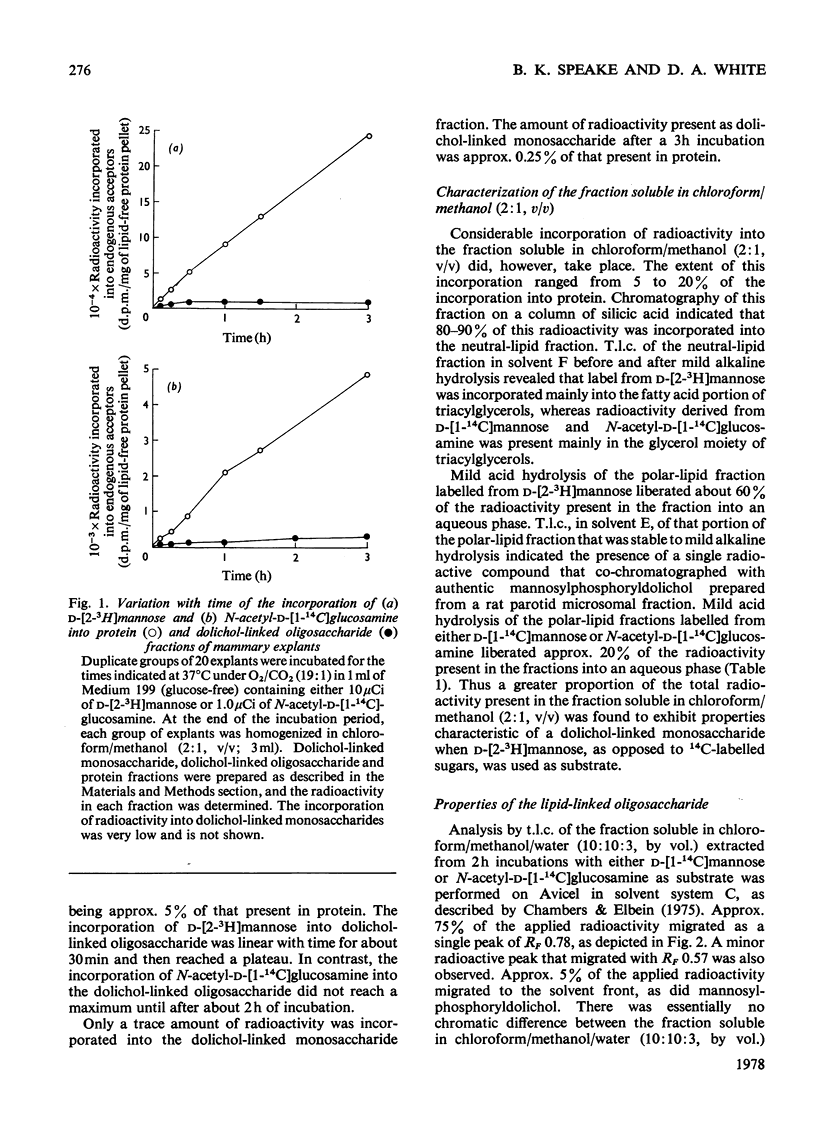
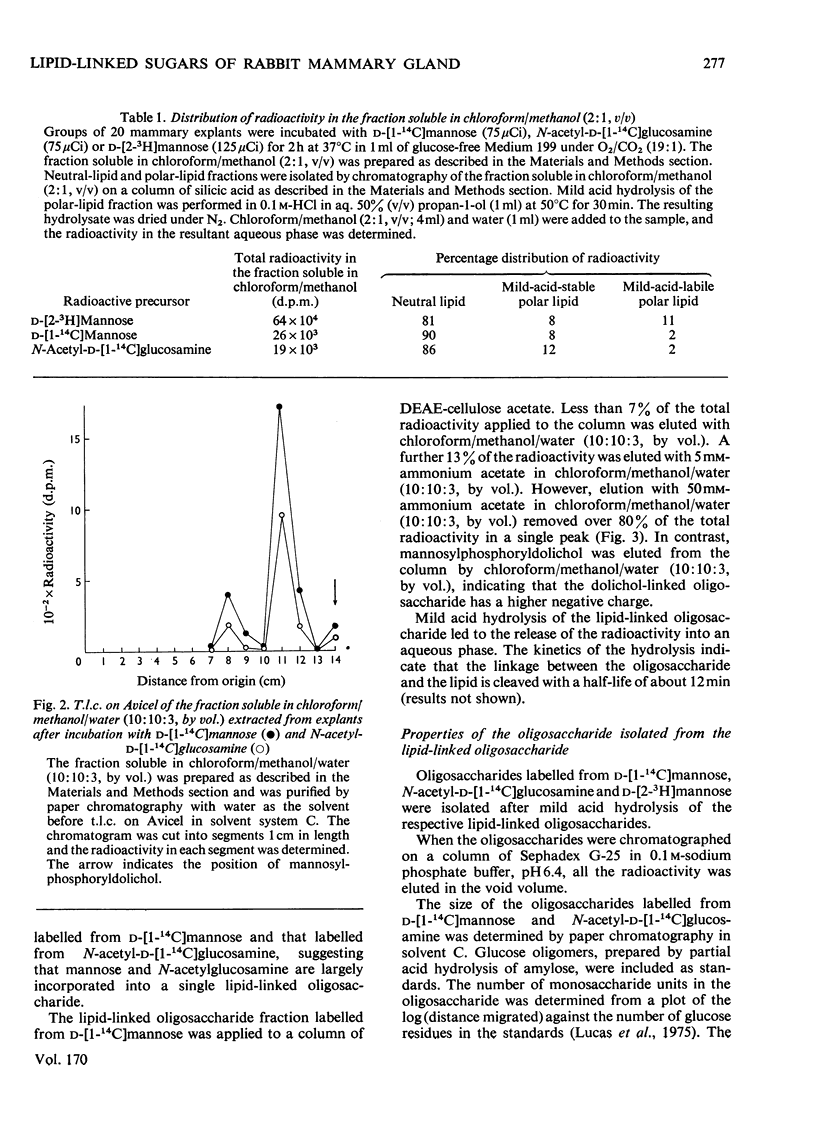
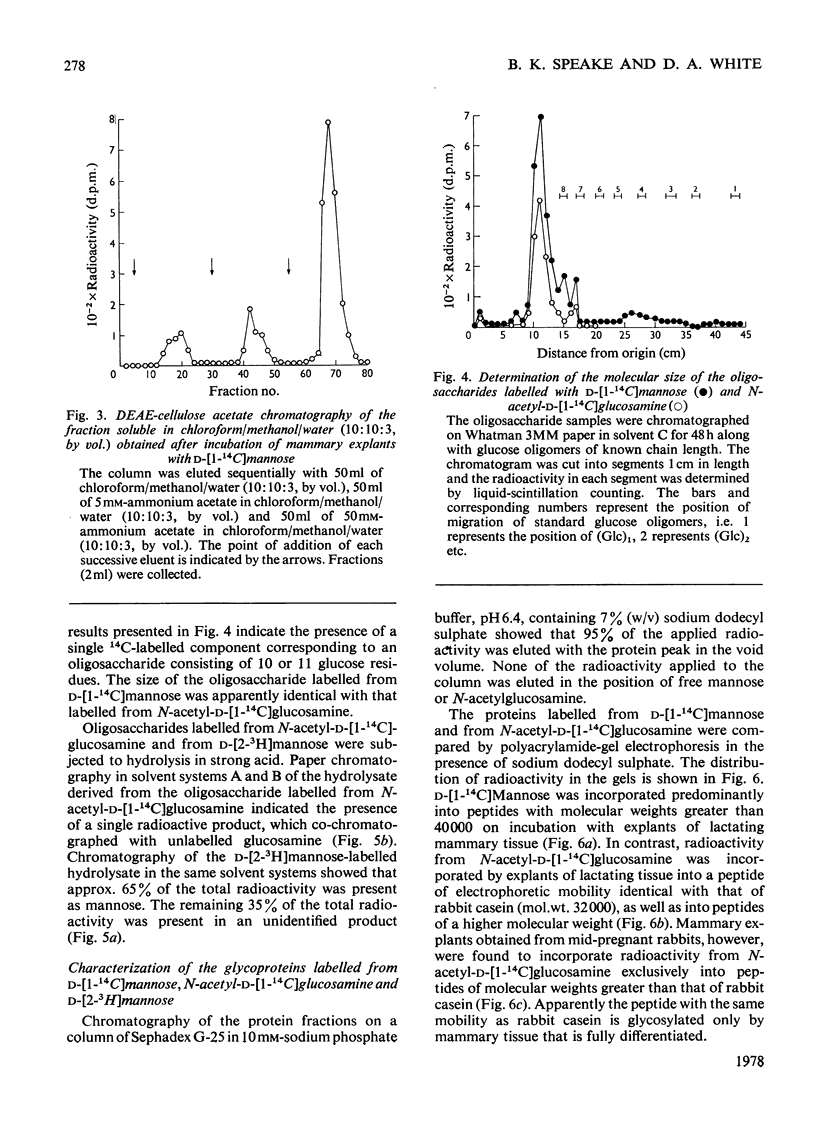
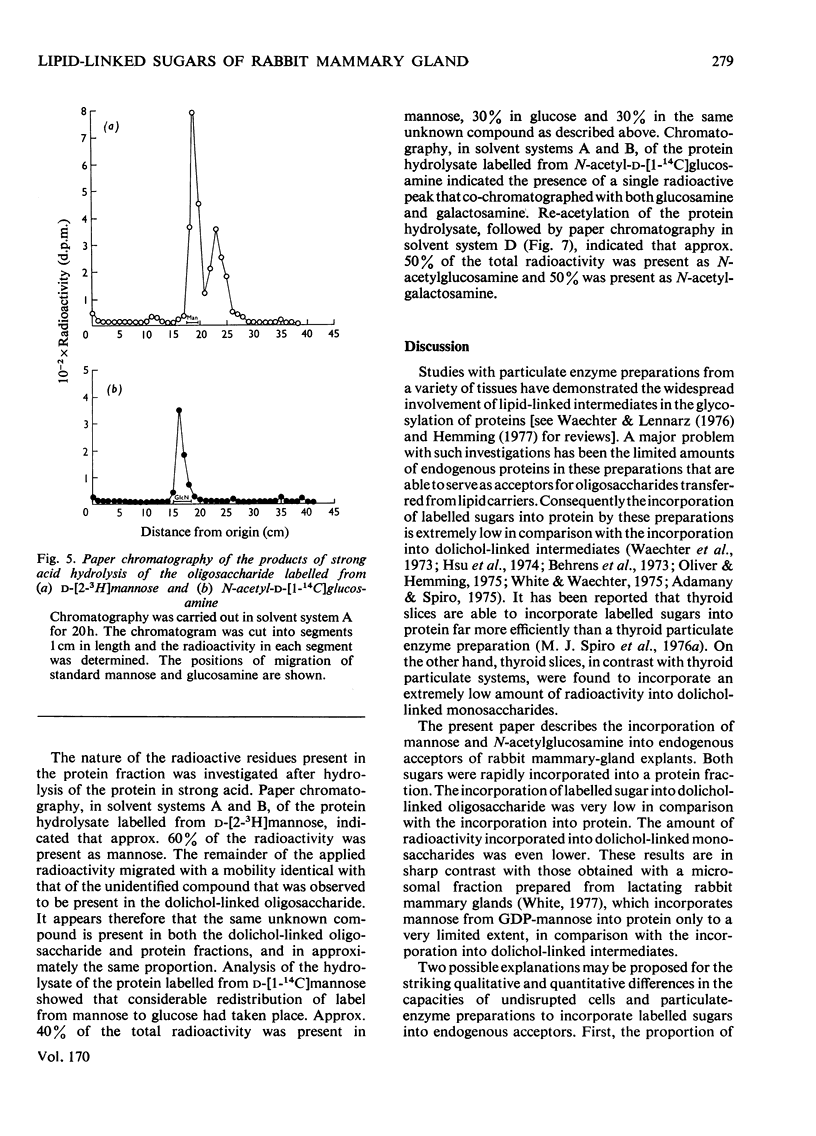
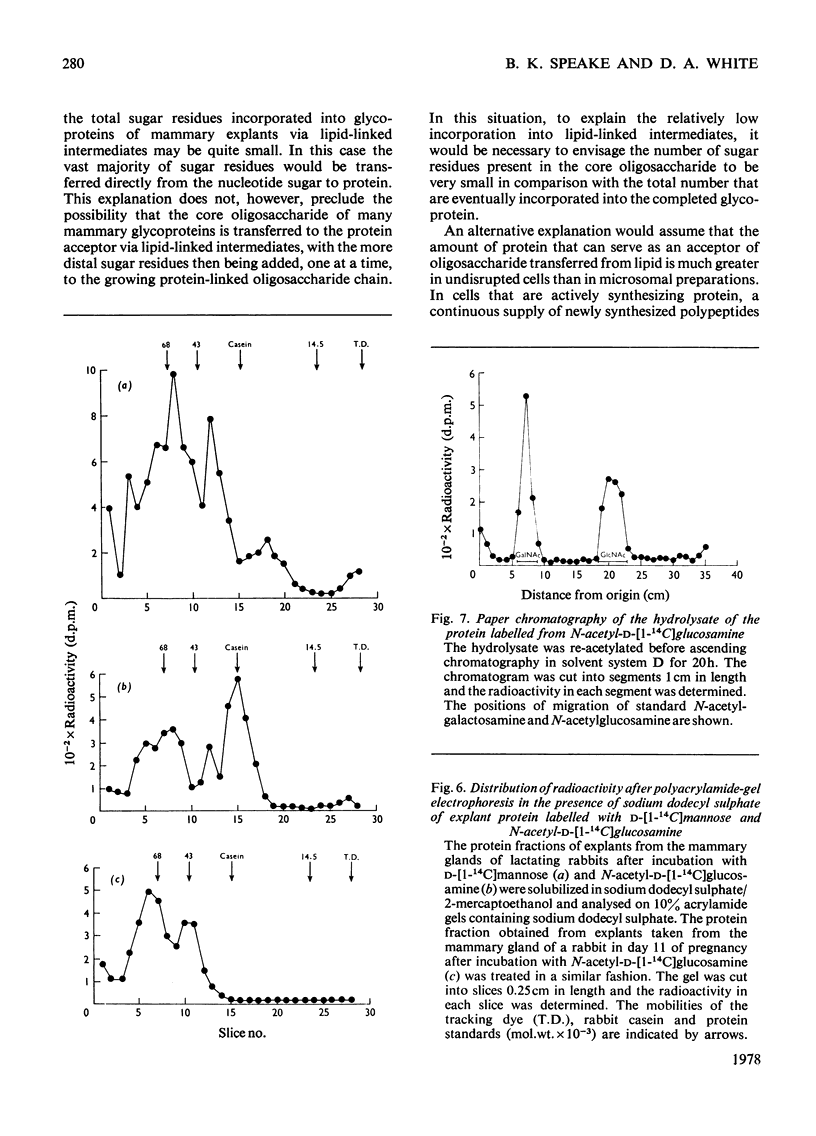
1. The incorporation of d-[1-14C]mannose, d-[2-3H]mannose and N-acetyl-d-[1-14C]-glucosamine into glycoproteins and lipid-linked intermediates of mammary explants obtained from lactating rabbits was studied. The amount of radioactivity incorporated into lipid-linked intermediates was very low compared with the incorporation into protein. Most of the radioactivity incorporated into the chloroform/methanol-soluble fraction was present as neutral lipid. Radioactivity from d-[2-3H]mannose was incorporated mainly into the fatty acid moiety, whereas radioactivity from d-[1-14C]mannose and N-acetyl-d-[1-14C]glucosamine was present in the glycerol moiety of triacylglycerol. 2. The labelled lipid-linked intermediate that was soluble in chloroform/methanol/water (10:10:3, by vol.) was partially characterized and was found to exhibit properties characteristic of an oligosaccharide linked to lipid via a pyrophosphate bridge. It migrated largely as a single zone of radioactivity on t.l.c. and was eluted from a column of DEAE-cellulose acetate as a single peak by 50mm-ammonium acetate. 3. The oligosaccharide moiety was released from the lipid by mild acid hydrolysis. The size of the oligosaccharide was estimated by paper chromatography to be 10 or 11 monosaccharide units. 4. d-[1-14C]Mannose was incorporated largely into glycopeptides with molecular weights in the range 40000–80000, as determined by polyacrylamide-gel electrophoresis in the presence of sodium dodecyl sulphate. Label from N-acetyl-d-[1-14C]glucosamine was incorporated into a glycopeptide with an electrophoretic mobility identical with that of rabbit casein (mol.wt. 32000) as well as into glycopeptides of higher molecular weight. 5. Approx. 50% of the total radioactivity in the protein labelled from N-acetyl-d-[1-14C]glucosamine was present as galactosamine, a component of the carbohydrate portion of rabbit casein. No labelled galactosamine was present in the lipid-linked oligosaccharide labelled from N-acetyl-d-[1-14C]glucosamine. It thus appears that the lipid-linked oligosaccharide is not involved in the glycosylation of casein.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adamany A. M., Spiro R. G. Glycoprotein biosynthesis: studies on thyroid mannosyltransferases. II. Characterization of a polyisoprenyl mannosyl phosphate and evaluation of its intermediary role in the glycosylation of exogenous acceptors. J Biol Chem. 1975 Apr 25;250(8):2842–2854. [PubMed] [Google Scholar]
- Barman T. E. Purification and properties of bovine milk glyco-alpha-lactalbumin. Biochim Biophys Acta. 1970 Jul 27;214(1):242–244. doi: 10.1016/0005-2795(70)90094-2. [DOI] [PubMed] [Google Scholar]
- Behrens N. H., Carminatti H., Staneloni R. J., Leloir L. F., Cantarella A. I. Formation of lipid-bound oligosaccharides containing mannose. Their role in glycoprotein synthesis. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3390–3394. doi: 10.1073/pnas.70.12.3390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown R. C., Fish W. W., Hudson B. G., Ebner K. E. Isolation and characterization of rat alpha-lactalbumin: a glycoprotein. Biochim Biophys Acta. 1977 Mar 28;491(1):82–92. doi: 10.1016/0005-2795(77)90043-5. [DOI] [PubMed] [Google Scholar]
- Chambers J., Elbein A. D. Biosynthesis and characterization of lipid-linked sugars and glycoproteins in aorta. J Biol Chem. 1975 Sep 10;250(17):6904–6915. [PubMed] [Google Scholar]
- Heifetz A., Elbein A. D. Biosynthesis of Man-beta-GlcNAc-GlcNAc-pyrophosphoryl-polyprenol by a solubilized enzyme from aorta. Biochem Biophys Res Commun. 1977 Mar 7;75(1):20–28. doi: 10.1016/0006-291x(77)91283-9. [DOI] [PubMed] [Google Scholar]
- Hemming F. W. Dolichol phosphate, a coenzyme in the glycosylation of animal membrane-bound glycoproteins. Biochem Soc Trans. 1977;5(4):1223–1231. doi: 10.1042/bst0051223. [DOI] [PubMed] [Google Scholar]
- Hsu A. F., Baynes J. W., Heath E. C. The role of a dolichol-oligosaccharide as an intermediate in glycoprotein biosynthesis. Proc Natl Acad Sci U S A. 1974 Jun;71(6):2391–2395. doi: 10.1073/pnas.71.6.2391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiely M. L., McKnight G. S., Schimke R. T. Studies on the attachment of carbohydrate to ovalbumin nascent chains in hen oviduct. J Biol Chem. 1976 Sep 25;251(18):5490–5495. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lehle L., Tanner W. Formation of lipid-bound oligosaccharides in yeast. Biochim Biophys Acta. 1975 Aug 13;399(2):364–374. doi: 10.1016/0304-4165(75)90265-2. [DOI] [PubMed] [Google Scholar]
- Lucas J. J., Waechter J., Lennarz W. J. The participation of lipid-linked oligosaccharide in synthesis of membrane glycoproteins. J Biol Chem. 1975 Mar 25;250(6):1992–2002. [PubMed] [Google Scholar]
- MORGAN J. F., MORTON H. J., PARKER R. C. Nutrition of animal cells in tissue culture; initial studies on a synthetic medium. Proc Soc Exp Biol Med. 1950 Jan;73(1):1–8. doi: 10.3181/00379727-73-17557. [DOI] [PubMed] [Google Scholar]
- Oliver G. J., Hemming F. W. The transfer of mannose to dolichol diphosphate oligosaccharides in pig liver endoplasmic reticulum. Biochem J. 1975 Nov;152(2):191–199. doi: 10.1042/bj1520191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parodi A. J., Behrens N. H., Leloir L. F., Carminatti H. The role of polyprenol-bound saccharides as intermediates in glycoprotein synthesis in liver. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3268–3272. doi: 10.1073/pnas.69.11.3268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parodi A. J., Staneloni R., Cantarella A. I., Leloir L. F., Behrens N. H., Carminatti H., Levy J. A. Further studies on a glycolipid formed from dolichyl-D-glucosyl monophosphate. Carbohydr Res. 1973 Feb;26(2):393–400. doi: 10.1016/s0008-6215(00)84527-9. [DOI] [PubMed] [Google Scholar]
- Robbins P. W., Krag S. S., Liu T. Effects of UDP-glucose addition on the synthesis of mannosyl lipid-linked oligosaccharides by cell-free fibroblast preparations. J Biol Chem. 1977 Mar 10;252(5):1780–1785. [PubMed] [Google Scholar]
- Speake B. K., Dils R., Mayer R. J. Regulation of enzyme turnover during tissue differentiation. Interactions of insulin, prolactin and cortisol in controlling the turnover of fatty acid synthetase in rabbit mammary gland in organ culture. Biochem J. 1976 Feb 15;154(2):359–370. doi: 10.1042/bj1540359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speake B. K., Dils R., Mayer R. J. Regulation of enzyme turnover during tissue differention. Studies on the effects of hormones on the turnover of fatty acid synthetase in rabbit mammary gland in organ culture. Biochem J. 1975 May;148(2):309–320. doi: 10.1042/bj1480309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiro M. J., Spiro R. G., Bhoyroo V. D. Lipid-saccharide intermediates in glycoprotein biosynthesis. I. Formation of an oligosaccharide-lipid by thyroid slices and evaluation of its role in protein glycosylation. J Biol Chem. 1976 Oct 25;251(20):6400–6408. [PubMed] [Google Scholar]
- Spiro M. J., Spiro R. G., Bhoyroo V. D. Lipid-saccharide intermediates in glycoprotein biosynthesis. III. Comparison of oligosaccharide-lipids formed by slices from several tissues. J Biol Chem. 1976 Oct 25;251(20):6420–6425. [PubMed] [Google Scholar]
- Spiro R. G., Spiro M. J., Bhoyroo V. D. Lipid-saccharide intermediates in glycoprotein biosynthesis. II. Studies on the structure of an oligosaccharide-lipid from thyroid. J Biol Chem. 1976 Oct 25;251(20):6409–6419. [PubMed] [Google Scholar]
- Strong C. R., Dils R. Fatty acid biosynthesis in rabbit mammary gland during pregnancy and early lactation. Biochem J. 1972 Aug;128(5):1303–1309. doi: 10.1042/bj1281303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Struck D. K., Lennarz W. J. Evidence for the participation of saccharide-lipids in the synthesis of the oligosaccharide chain of ovalbumin. J Biol Chem. 1977 Feb 10;252(3):1007–1013. [PubMed] [Google Scholar]
- Struck D. K., Lennarz W. J. Utilization of exogenous GDP-mannose for the synthesis of mannose-containing lipids and glycoproteins by oviduct cells. J Biol Chem. 1976 Apr 25;251(8):2511–2519. [PubMed] [Google Scholar]
- TREVELYAN W. E., PROCTER D. P., HARRISON J. S. Detection of sugars on paper chromatograms. Nature. 1950 Sep 9;166(4219):444–445. doi: 10.1038/166444b0. [DOI] [PubMed] [Google Scholar]
- Waechter C. J., Lennarz W. J. The role of polyprenol-linked sugars in glycoprotein synthesis. Annu Rev Biochem. 1976;45:95–112. doi: 10.1146/annurev.bi.45.070176.000523. [DOI] [PubMed] [Google Scholar]
- Waechter C. J., Lucas J. J., Lennarz W. J. Evidence for xylosyl lipids as intermediates in xylosyl transfers in hen oviduct membranes. Biochem Biophys Res Commun. 1974 Jan 23;56(2):343–350. doi: 10.1016/0006-291x(74)90848-1. [DOI] [PubMed] [Google Scholar]
- Waechter C. J., Lucas J. J., Lennarz W. J. Membrane glycoproteins. I. Enzymatic synthesis of mannosyl phosphoryl polyisoprenol and its role as a mannosyl donor in glycoprotein synthesis. J Biol Chem. 1973 Nov 10;248(21):7570–7579. [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- White D. A. The biosynthesis of mannose-containing lipids as intermediates in the glycosylation of proteins of rabbit mammary gland [proceedings]. Biochem Soc Trans. 1977;5(4):1022–1023. doi: 10.1042/bst0051022. [DOI] [PubMed] [Google Scholar]
- White D. A., Waechter C. J. A mannosyl-carrier lipid of bovine adrenal meddulla and rat parotid. Biochem J. 1975 Mar;146(3):645–651. doi: 10.1042/bj1460645. [DOI] [PMC free article] [PubMed] [Google Scholar]


