Abstract
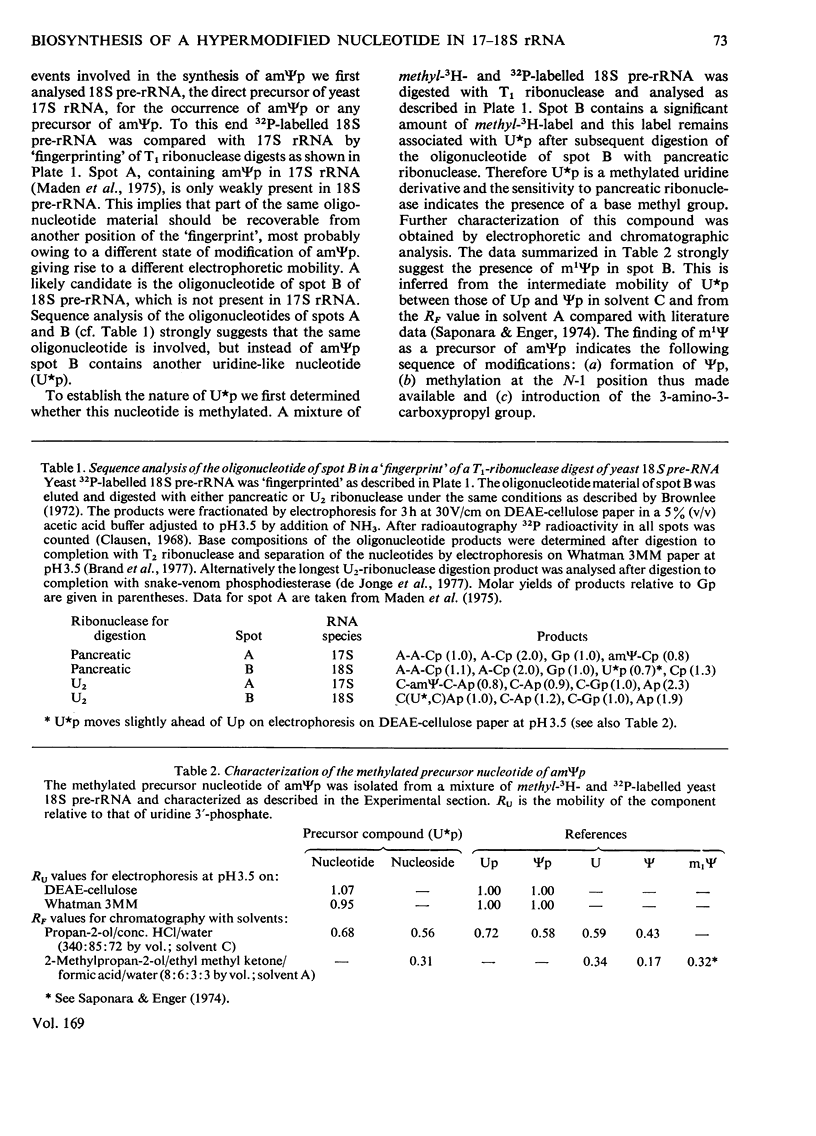
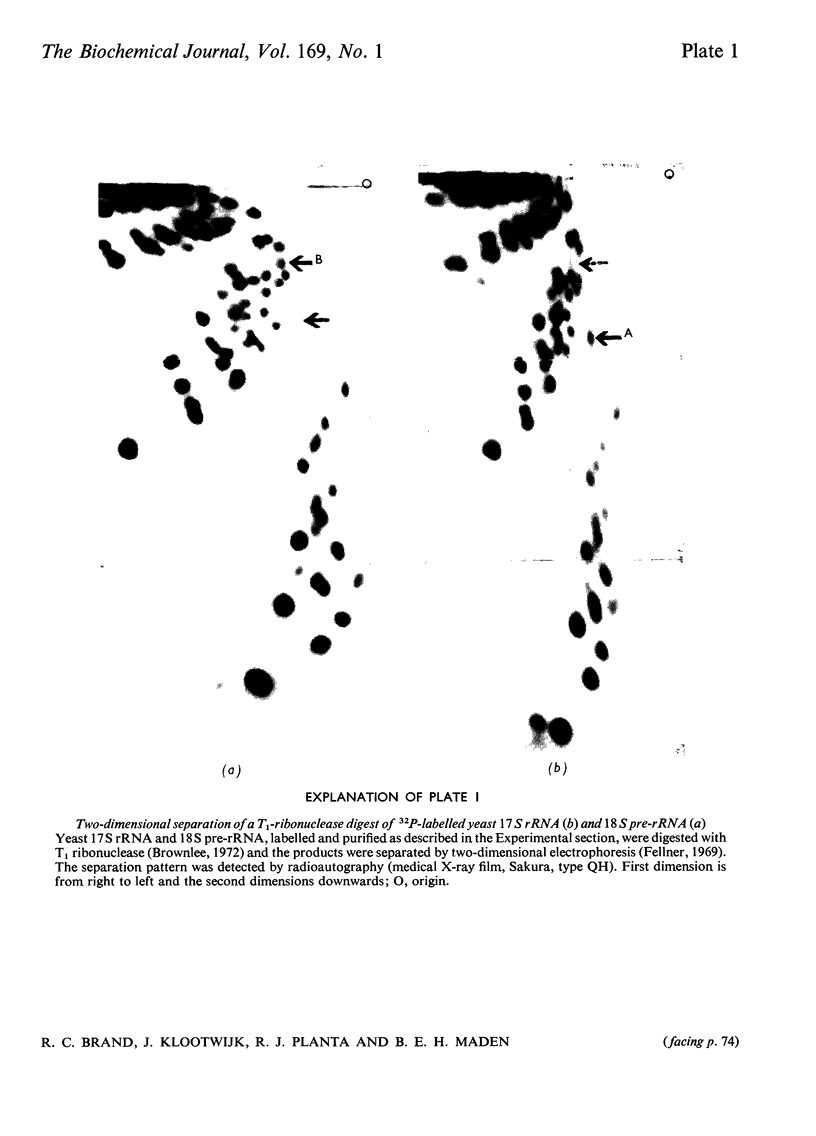
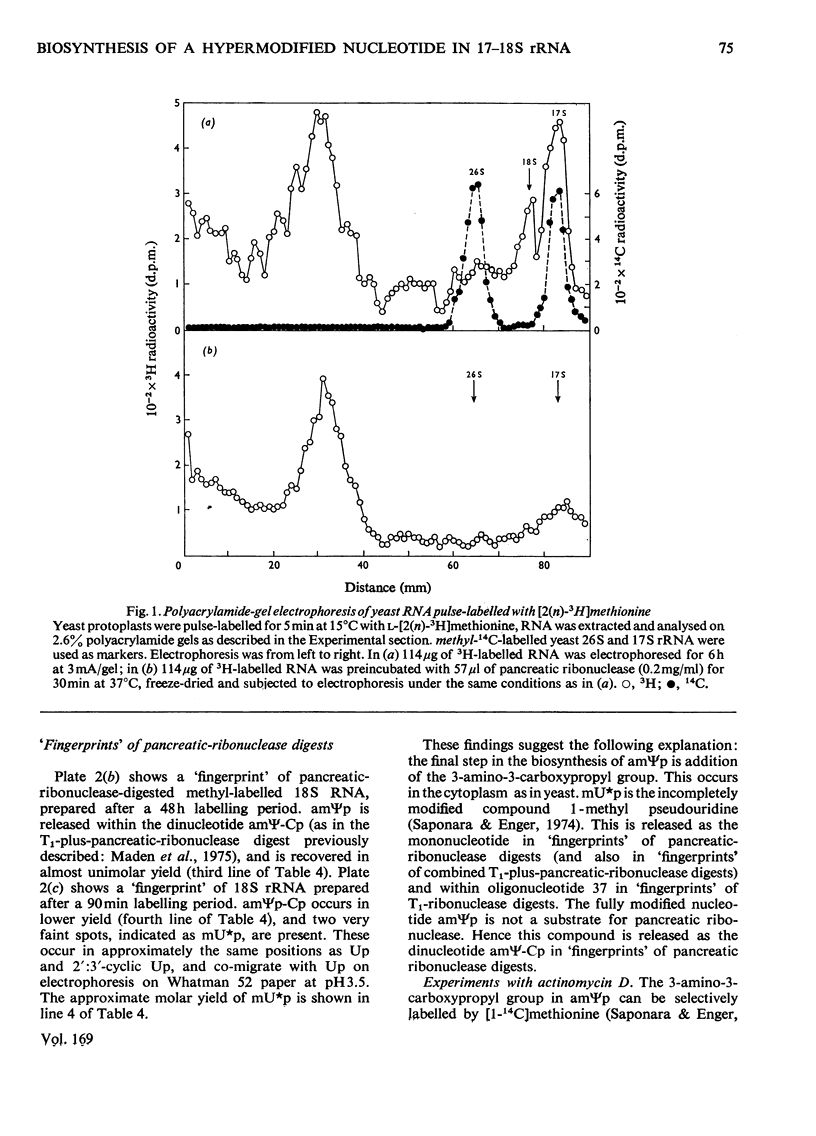
The biosynthesis of a hypermodified nucleotide, similar to or identical with 3-(3-amino-3-carboxypropyl)-1-methylpseudouridine monophosphate, present in Saccharomyces carlsbergensis 17S and HeLa-cell 18S rRNA, was investigated with respect to the sequence of reactions required for synthesis and their timing in ribosome maturation. In both yeast and HeLa cells methylation precedes attachment of the 3-amino-3-carboxypropyl group. In yeast the methylated precursor nucleotide was tentatively characterized as 1-methylpseudouridine. This precursor nucleotide was demonstrated in both 37S and most of the cytoplasmic 18S pre-rRNA (rRNA precursor) molecules. The synthesis of the hypermodified nucleotide is completed just before the final cleavage of 18S pre-rRNA to give 17S rRNA, so that the final addition of the 3-amino-3-carboxypropyl group is a cytoplasmic event. Comparable experiments with HeLa cells indicated that formation of 1-methylpseudouridine occurs at the level of 45S RNA and addition of the 3-amino-3-carboxypropyl group occurs in the cytoplasm on newly synthesized 18S RNA.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brand R. C., Klootwijk J., Van Steenbergen T. J., De Kok A. J., Planta R. J. Secondary methylation of yeast ribosomal precursor RNA. Eur J Biochem. 1977 May 2;75(1):311–318. doi: 10.1111/j.1432-1033.1977.tb11531.x. [DOI] [PubMed] [Google Scholar]
- Brand R. C., Planta R. J. The molecular weights of yeast ribosomal precursor RNAs. Mol Biol Rep. 1975 Dec;2(4):321–325. doi: 10.1007/BF00357019. [DOI] [PubMed] [Google Scholar]
- Clausen T. Measurement of 32P activity in a liquid scintillation counter without the use of scintillator. Anal Biochem. 1968 Jan;22(1):70–73. doi: 10.1016/0003-2697(68)90260-1. [DOI] [PubMed] [Google Scholar]
- De Jonge P., Klootwijk J., Planta R. J. Terminal nucleotide sequences of 17-S ribosomal RNA and its immediate precursor 18-S RNA in yeast. Eur J Biochem. 1977 Jan;72(2):361–369. doi: 10.1111/j.1432-1033.1977.tb11260.x. [DOI] [PubMed] [Google Scholar]
- EAGLE H. Amino acid metabolism in mammalian cell cultures. Science. 1959 Aug 21;130(3373):432–437. doi: 10.1126/science.130.3373.432. [DOI] [PubMed] [Google Scholar]
- Enger M. D., Saponara A. G. Incorporation of 14C from [2-14C]methionine into 18 s but not 28 s RNA of Chinese hamster cells. J Mol Biol. 1968 Apr 14;33(1):319–322. doi: 10.1016/0022-2836(68)90298-2. [DOI] [PubMed] [Google Scholar]
- Fellner P. Nucleotide sequences from specific areas of the 16S and 23S ribosomal RNAs of E. coli. Eur J Biochem. 1969 Nov;11(1):12–27. doi: 10.1111/j.1432-1033.1969.tb00733.x. [DOI] [PubMed] [Google Scholar]
- Jeppesen P. G. The nucleotide sequences of some large ribonuclease T 1 products from bacteriophage R17 ribonucleic acid. Biochem J. 1971 Sep;124(2):357–366. doi: 10.1042/bj1240357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan M. S., Maden B. E. Nucleotide sequences within the ribosomal ribonucleic acids of HeLa cells, Xenopus laevis and chick embryo fibroblasts. J Mol Biol. 1976 Feb 25;101(2):235–254. doi: 10.1016/0022-2836(76)90375-2. [DOI] [PubMed] [Google Scholar]
- Klootwijk J., Planta R. J. Analysis of the methylation sites in yeast ribosomal RNA. Eur J Biochem. 1973 Nov 15;39(2):325–333. doi: 10.1111/j.1432-1033.1973.tb03130.x. [DOI] [PubMed] [Google Scholar]
- Klootwijk J., van den Bos R. C., Planta R. J. Secondary methylation of yeast ribosomal RNA. FEBS Lett. 1972 Oct 15;27(1):102–106. doi: 10.1016/0014-5793(72)80419-8. [DOI] [PubMed] [Google Scholar]
- Maden B. E., Forbes J., de Jonge P., Klootwijk J. Presence of a hypermodified nucleotide in HeLa cell 18 S and Saccharomyces carlsbergensis 17 S ribosomal RNAs. FEBS Lett. 1975 Nov 1;59(1):60–63. doi: 10.1016/0014-5793(75)80341-3. [DOI] [PubMed] [Google Scholar]
- Maden B. E., Salim M. The methylated nucleotide sequences in HELA cell ribosomal RNA and its precursors. J Mol Biol. 1974 Sep 5;88(1):133–152. doi: 10.1016/0022-2836(74)90299-x. [DOI] [PubMed] [Google Scholar]
- Penman S., Smith I., Holtzman E. Ribosomal RNA synthesis and processing in a particulate site in the HeLa cell nucleus. Science. 1966 Nov 11;154(3750):786–789. doi: 10.1126/science.154.3750.786. [DOI] [PubMed] [Google Scholar]
- Retèl J., Planta R. J. Ribosomal precursor RNA in Saccharomyces carlsbergensis. Eur J Biochem. 1967 Dec;3(2):248–258. doi: 10.1111/j.1432-1033.1967.tb19524.x. [DOI] [PubMed] [Google Scholar]
- Saponara A. G., Enger M. D. The isolation from ribonucleic acid of substituted uridines containing alpha-aminobutyrate moieties derived from methionine. Biochim Biophys Acta. 1974 Apr 27;349(1):61–77. doi: 10.1016/0005-2787(74)90009-4. [DOI] [PubMed] [Google Scholar]
- Trapman J., Planta R. J. Maturation of ribosomes in yeast. I Kinetic analysis by labelling of high molecular weight rRNA species. Biochim Biophys Acta. 1976 Sep 6;442(3):265–274. doi: 10.1016/0005-2787(76)90301-4. [DOI] [PubMed] [Google Scholar]
- Trapman J., Retèl J., Planta R. J. Ribosomal precursor particles from yeast. Exp Cell Res. 1975 Jan;90(1):95–104. doi: 10.1016/0014-4827(75)90361-4. [DOI] [PubMed] [Google Scholar]