Abstract
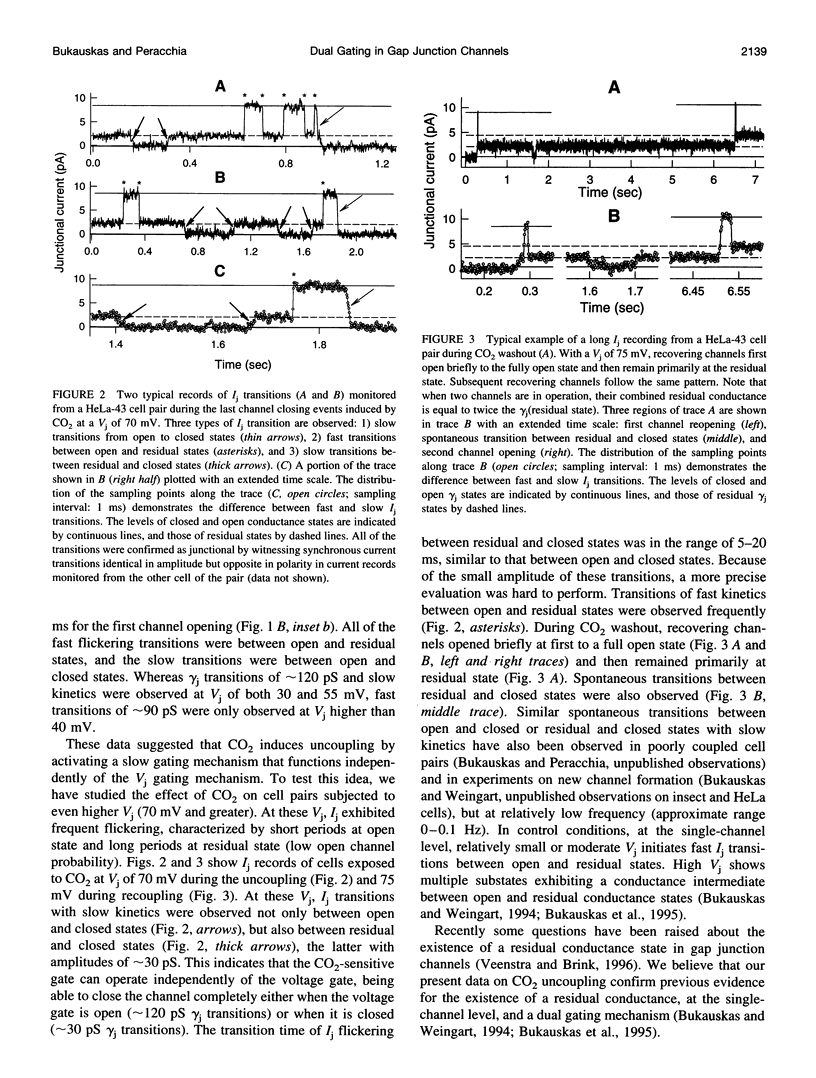
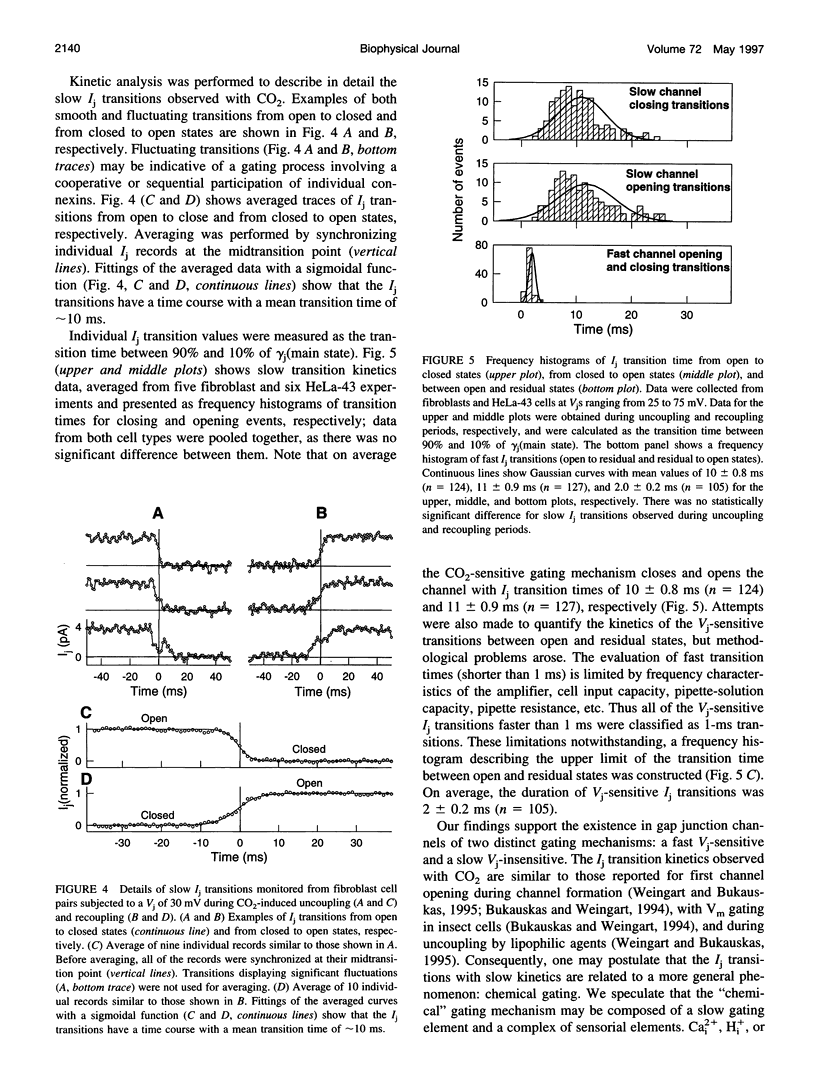
The chemical gating of single-gap junction channels was studied by the dual whole-cell voltage-clamp method in HeLa cells transfected with connexin43 (HeLa43) and in fibroblasts from sciatic nerves. Junctional current (Ij), single-channel conductance, and Ij kinetics were studied in cell pairs during CO2 uncoupling and recoupling at small transjunctional voltages (Vj < 35 mV: Vj gating absent) and at high Vj (Vj > 40 mV: Vj gating strongly activated). In the absence of Vj gating, CO2 exclusively caused Ij slow transitions from open to closed channel states (mean transition time: approximately 10 ms), corresponding to a single-channel conductance of approximately 120 pS. At Vj > 40 mV, Vj gating induced fast Ij flickering between open, gamma j(main state), and residual, gamma j(residual), states (transition time: approximately 2 ms). The ratio gamma j(main state)/gamma j(residual) was approximately 4-5. No obvious correlation between Ij fast flickering and CO2 treatment was noticed. At high Vj, in addition to slow Ij transitions between open and closed states, CO2 induced slow transitions between residual and closed states. During recoupling, each channel reopened by a slow transition (mean transition time: approximately 10 ms) from closed to open state (rarely from closed to residual state). Fast Ij flickering between open and residual states followed. The data are in agreement with the hypothesis that gap junction channels possess two gating mechanisms, and indicate that CO2 induces channel gating exclusively by the slow gating mechanism.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bastide B., Hervé J. C., Cronier L., Délèze J. Rapid onset and calcium independence of the gap junction uncoupling induced by heptanol in cultured heart cells. Pflugers Arch. 1995 Jan;429(3):386–393. doi: 10.1007/BF00374154. [DOI] [PubMed] [Google Scholar]
- Bennett M. V., Barrio L. C., Bargiello T. A., Spray D. C., Hertzberg E., Sáez J. C. Gap junctions: new tools, new answers, new questions. Neuron. 1991 Mar;6(3):305–320. doi: 10.1016/0896-6273(91)90241-q. [DOI] [PubMed] [Google Scholar]
- Bennett M. V., Verselis V. K. Biophysics of gap junctions. Semin Cell Biol. 1992 Feb;3(1):29–47. doi: 10.1016/s1043-4682(10)80006-6. [DOI] [PubMed] [Google Scholar]
- Beyer E. C. Gap junctions. Int Rev Cytol. 1993;137C:1–37. [PubMed] [Google Scholar]
- Beyer E. C., Kistler J., Paul D. L., Goodenough D. A. Antisera directed against connexin43 peptides react with a 43-kD protein localized to gap junctions in myocardium and other tissues. J Cell Biol. 1989 Feb;108(2):595–605. doi: 10.1083/jcb.108.2.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brockes J. P., Fields K. L., Raff M. C. Studies on cultured rat Schwann cells. I. Establishment of purified populations from cultures of peripheral nerve. Brain Res. 1979 Apr 6;165(1):105–118. doi: 10.1016/0006-8993(79)90048-9. [DOI] [PubMed] [Google Scholar]
- Bukauskas F. F., Elfgang C., Willecke K., Weingart R. Biophysical properties of gap junction channels formed by mouse connexin40 in induced pairs of transfected human HeLa cells. Biophys J. 1995 Jun;68(6):2289–2298. doi: 10.1016/S0006-3495(95)80411-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bukauskas F. F., Weingart R. Voltage-dependent gating of single gap junction channels in an insect cell line. Biophys J. 1994 Aug;67(2):613–625. doi: 10.1016/S0006-3495(94)80521-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burt J. M. Uncoupling of cardiac cells by doxyl stearic acids specificity and mechanism of action. Am J Physiol. 1989 Apr;256(4 Pt 1):C913–C924. doi: 10.1152/ajpcell.1989.256.4.C913. [DOI] [PubMed] [Google Scholar]
- Chow I., Young S. H. Opening of single gap junction channels during formation of electrical coupling between embryonic muscle cells. Dev Biol. 1987 Aug;122(2):332–337. doi: 10.1016/0012-1606(87)90298-3. [DOI] [PubMed] [Google Scholar]
- Crow D. S., Beyer E. C., Paul D. L., Kobe S. S., Lau A. F. Phosphorylation of connexin43 gap junction protein in uninfected and Rous sarcoma virus-transformed mammalian fibroblasts. Mol Cell Biol. 1990 Apr;10(4):1754–1763. doi: 10.1128/mcb.10.4.1754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyoshi H., Boyle M. B., MacKay L. B., Garfield R. E. Voltage-clamp studies of gap junctions between uterine muscle cells during term and preterm labor. Biophys J. 1996 Sep;71(3):1324–1334. doi: 10.1016/S0006-3495(96)79332-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreno A. P., Fishman G. I., Spray D. C. Phosphorylation shifts unitary conductance and modifies voltage dependent kinetics of human connexin43 gap junction channels. Biophys J. 1992 Apr;62(1):51–53. doi: 10.1016/S0006-3495(92)81775-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neyton J., Trautmann A. Single-channel currents of an intercellular junction. 1985 Sep 26-Oct 2Nature. 317(6035):331–335. doi: 10.1038/317331a0. [DOI] [PubMed] [Google Scholar]
- Obaid A. L., Socolar S. J., Rose B. Cell-to-cell channels with two independently regulated gates in series: analysis of junctional conductance modulation by membrane potential, calcium, and pH. J Membr Biol. 1983;73(1):69–89. doi: 10.1007/BF01870342. [DOI] [PubMed] [Google Scholar]
- Spray D. C., Harris A. L., Bennett M. V. Equilibrium properties of a voltage-dependent junctional conductance. J Gen Physiol. 1981 Jan;77(1):77–93. doi: 10.1085/jgp.77.1.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traub O., Eckert R., Lichtenberg-Fraté H., Elfgang C., Bastide B., Scheidtmann K. H., Hülser D. F., Willecke K. Immunochemical and electrophysiological characterization of murine connexin40 and -43 in mouse tissues and transfected human cells. Eur J Cell Biol. 1994 Jun;64(1):101–112. [PubMed] [Google Scholar]
- Trexler E. B., Bennett M. V., Bargiello T. A., Verselis V. K. Voltage gating and permeation in a gap junction hemichannel. Proc Natl Acad Sci U S A. 1996 Jun 11;93(12):5836–5841. doi: 10.1073/pnas.93.12.5836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unwin N. The structure of ion channels in membranes of excitable cells. Neuron. 1989 Dec;3(6):665–676. doi: 10.1016/0896-6273(89)90235-3. [DOI] [PubMed] [Google Scholar]
- Veenstra R. D., DeHaan R. L. Measurement of single channel currents from cardiac gap junctions. Science. 1986 Aug 29;233(4767):972–974. doi: 10.1126/science.2426781. [DOI] [PubMed] [Google Scholar]
- Veenstra R. D. Do connexin channels have a residual conductance state? Biophys J. 1996 Feb;70(2):1082–1084. doi: 10.1016/S0006-3495(96)79654-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veenstra R. D., Wang H. Z., Westphale E. M., Beyer E. C. Multiple connexins confer distinct regulatory and conductance properties of gap junctions in developing heart. Circ Res. 1992 Nov;71(5):1277–1283. doi: 10.1161/01.res.71.5.1277. [DOI] [PubMed] [Google Scholar]
- Verselis V. K., Bennett M. V., Bargiello T. A. A voltage-dependent gap junction in Drosophila melanogaster. Biophys J. 1991 Jan;59(1):114–126. doi: 10.1016/S0006-3495(91)82204-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weingart R., Bukauskas F. F. Gap junction channels of insects exhibit a residual conductance. Pflugers Arch. 1993 Jul;424(2):192–194. doi: 10.1007/BF00374611. [DOI] [PubMed] [Google Scholar]
- Willecke K., Hennemann H., Dahl E., Jungbluth S., Heynkes R. The diversity of connexin genes encoding gap junctional proteins. Eur J Cell Biol. 1991 Oct;56(1):1–7. [PubMed] [Google Scholar]


