Abstract
Low birth weight and anemia are significant public health challenges in developing countries. This study seeks to evaluate the relationship between hemoglobin levels during the first and second trimesters of pregnancy and the occurrence of low birth weight, while accounting for potential confounding factors. This multi-center cross-sectional study was conducted among all pregnant women with COVID-19 and with no history of receiving the COVID-19 vaccine who delivered in public and private hospitals in three counties, Mahabad, Miandoab, Bukan in Iran, using routinely collected maternity and health data on pregnancies. Hemoglobulin levels were measured during the first (6–10th weeks) and second (24–28th weeks) trimesters of pregnancy, and pregnancy outcomes were recorded in the health information system. Hb levels were categorized into four groups as follows: ≥110 g/L, 100–109 g/L, 90–99 g/L, and < 90 g/L for the first trimester and ≥ 105 g/L, 100–104 g/L, 90–99 g/L, and < 90 g/L for the second trimester. Multivariable logistic regression analysis determined the association between hemoglobin levels during pregnancy and low birth weight. P-values < 0.05 were considered statistically significant. A total of 385 mothers with COVID-19 were included. The mean age of COVID-19 pregnant women was 30.01 ± 6.24 years. After multivariable adjustment, Hb levels < 110 g/L in the first trimester had a significant association with low birth weight [OR, 4.13; (95% CI 2.11–8.10)]. Morevoer, Hb levels < 105 g/L in the second trimester was a significant association with low birth weight [OR:3.91; (95% CI:1.98–7.75)]. Maternal anemia during pregnancy, including first and second trimesters, was a significant association with Low birth weight even after adjusting for common confounders. Effective management and monitoring of anemia in pregnant women, particularly in low- and middle-income countries, are crucial for preventing low birth weight.
Keywords: COVID-19, Pregnant women, Low birth weight, Neonatal
Subject terms: Medical research, Epidemiology, Outcomes research
Introduction
Birth weight is an important indicator in evaluating the risk of perinatal mortality and morbidity because it is associated with gestational age1. The Infants with a birth weight of less than 2500 g have a low birth weight (LBW)2. LBW is a fundamental health indicator for evaluating infant status3. It is estimated that 15–20% of all births globally are LBW, totalling over 20 million births annually2. Mortality in LBW infants is higher 40 times than in infants with a weight greater than 2500 g4. LBW impacts the health sector’s economy, increases the risk of death and illness shortly after birth, contributes to non-communicable diseases, and poses a considerable burden on society3,5. Thus, LBW infants may contribute to major public health problems. The etiology of LBW infants is still not fully understood, as it relates to multiple sociodemographic, nutritional, biological, and environmental factors.
Anaemia is a serious global public health problem that particularly affects pregnant women6. Iron deficiency is responsible for 75% of cases of non-physiologic anemia in pregnancy7.
In 2019, the worldwide prevalence of anemia in pregnant women was 36.5%, totalling more than 116 million pregnant women affected by anemia8. The prevalence of anemia and iron deficiency anemia in Iranian pregnant women is estimated to be 17%9, and 13.5%10, respectively. Maternal anemia during pregnancy is one of the important causes of fetal death and an indirect cause of maternal mortality11.
The coronavirus disease 2019 (COVID-19 ) continues to be an alarming global public health crisis12,13. One of the vulnerable groups during the COVID-19 pandemic was pregnant women and infants14. COVID-19 infection during pregnancy is associated with an increased risk of adverse birth outcomes because of the increased number of cesarian15. Significant advancements have been achieved in the COVID-19 vaccination efforts, and it’s essential to maintain this momentum, especially for those who are most vulnerable to the disease16. Pregnant women are strongly encouraged to receive COVID-19 vaccinations due to the associated health benefits for both the mother and the baby17. COVID-19 poses significant risks during pregnancy, affecting both maternal health and fetal outcomes18. The association with altered hemoglobin levels in seropositive individuals underscores the importance of closely monitoring hematological health in pregnant women19. There is a complex interaction between COVID-19 and maternal anemia status19. Moreover, the increased risks of adverse outcomes such as preeclampsia, preterm birth, LBW and stillbirth in pregnant women with COVID-19 necessitate targeted intervention strategies to effectively mitigate these risks18. Some studies have documented that anemia can cause some adverse maternal and birth outcomes20,21, but several studies have shown no association22,23. Also, even though several studies have shown the impact of the COVID-19 pandemic on pregnancy outcomes24,25.
In addition, Iran is among low-middle-income countries (LMICs)26 and is the second most sanctioned country worldwide27. Undoubtedly, sanctions can negatively impact lifestyles and health by creating unique economic conditions. Sanctions restricted Iran’s access to COVID-19 vaccines, leading to only 20% of the population being fully vaccinated by December 202128. Sanctions have strained Iran’s healthcare, impacting care for pregnant women. Because of sanctions, women in Iran may experience higher rates of nutritional deficiencies, such as iron deficiency during pregnancy, which can subsequently affect newborn birth weights and the health of the next generation. A current cross-sectional study hypothesizes that low hemoglobin levels and anemia in pregnant women with COVID-19 are associated with LBW babies. Thus, the present study aims to determine the association between anemia and Hb levels in the first and second trimesters of pregnancy with LBW in Iranian women with COVID-19 during a period when Iran lacked access to the COVID-19 vaccine due to sanctions.
Methods
Study design, period, and setting
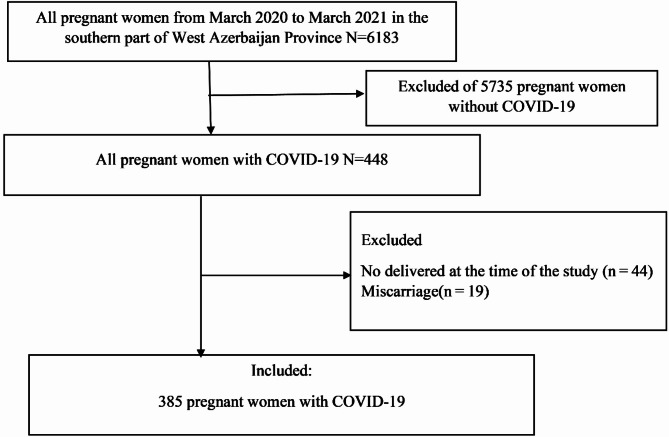
The current multi-center cross-sectional research was performed from March 21, 2020 to March 21, 2021, including all pregnant women with COVID-19 who delivered in public and private hospitals in three counties (Mahabad, Miandoab, Bukan) in the southern part of West Azerbaijan Province in Northwest of Iran. In this multi-center study, infection of COVID-19 was confirmed in all pregnant women with positive RT-PCR test results at any stage of pregnancy, irrespective of clinical signs and symptoms. The inclusion criteria of subjects were as follows: (1) had a positive RT- PCR test for COVID-19 at any stage of pregnancy; (2) had complete medical and health records. 3) had given birth during this study, and 4) did not receive the COVID-19 vaccine. From the time of the first prenatal health care to the time of delivery, all baseline, clinical, laboratory characteristics, maternal and fetal outcomes of the participants were recorded in the National Health System. The study is based on retrospective medical records in the National Health System. Participants were excluded, as they had one of the following conditions: (1) no delivery at the time of the study (n = 44), (2) Miscarriage(n = 19). Finally, a total of 385 pregnant were included in the current study. Figure 1 depicts the flowchart detailing the sample selection process.
Fig. 1.
Participant inclusion flowchart.
Baseline information and confounders/covariates
The following variables were considered as confounders/covariates: Age (years), residency(rural, urban), education (Illiterate, < diploma, diploma, > diploma or more), occupation (housekeeper, employed), parity (nullipara or multipara), history of miscarriage (yes or no), pre-pregnancy BMI (Body Mass Index) (kg/m2), preeclampsia/eclampsia (yes or no), gestational diabetes mellitus(GDM) (yes or no), and preterm birth (PTB) (yes or no) were obtained from the data source by extracting the information recorded in electronic medical records registered in National Health System. Pregnant women with other pregnancy complications, including circulatory diseases, urinary diseases, respiratory diseases, vitro fertilization, thyroid diseases, digestive diseases, coagulation disorders, multiple pregnancies, placenta accrete, placenta previa, antepartum hemorrhage, spectrum, intrauterine infection, scarred uterus, abnormal Amniotic fluid, cervical incompetence, and abnormal placenta were not included in this study.
Outcome variable
The outcome variable in this study was LBW. We followed the International Classification of Diseases, 10th Revision (ICD-10) definition. LBW was defined as a newborn with a birth weight under 2500 g (ICD-10 codes were P05.051 and P07.001)29.
Laboratory assays
Two Hb measurements were taken for routine antenatal care. The first sample was collected at the first (6–10th weeks) trimesters and the second at the second trimesters (24-28th weeks) of gestation. Hb levels were measured with a calibrated laboratory machine (ADVIA 2120 hematology system, SIEMENS health care) immediately after blood sample collection. According to the WHO criteria, anemia in the first trimester of pregnancy is defined as a hemoglobin (Hb) level of < 110 g/L and anemia in the second trimester of pregnancy is defined as a hemoglobin (Hb) level of < 105 g/L30.
Statistical analyses
Categorical and continuous variables were N (%) and mean ± standard deviation (SD), respectively. The chi-square test evaluated the relationship between categorical variables. The hypothesis of normality for all continuous variables was tested using the Kolmogorov-Smirnov test. We employed the independent t-test to compare continuous variables between the two groups. Univariable and multivariable logistic regression analyses were used to investigate the association between low hemoglobin levels and LBW. Variables with p-value < 0.25 in the univariable analysis were entered into a multivariable logistic regression. Assumptions of logistic regression were met. P-values < 0.05 were considered statistically significant. Data analysis was performed using Statistical Package for Social Science (IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp).
Ethical consideration
This study was approved by the Research Ethical Review of Urmia University of Medical Sciences (approval number: IR.UMSU.REC.1399.352). The ethics committee of Urmia University of Medical Sciences waived the need for Informed Consent due to the study’s retrospective nature. All methods have been carried out in accordance with the relevant guidelines and regulations.
Results
Demographic characteristics of study participants
A total of 385 pregnant women with COVID-19 were included in this study. Table 1 provides detailed characteristics of the participants. The mean ± SD of the pregnant women’s age was 30.01 ± 6.24 years. The mean ± SD of the pregnant women’s age with normal birth weight infants was 29.84 ± 6.12, and for pregnant women with LBW infants was 30.93 ± 6.81 years. The educational level was under diploma in 209(54.3%) of participants. Most of them lived in urban areas(84.7%) (Table 1).
Table 1.
Demographic characteristics of study participants.
| Variables | Normal birth weight(n = 325) | LBW(n = 60) | Total (n = 385) |
P-value |
|---|---|---|---|---|
| N(%) | N(%) | N(%) | ||
| Age(Mean ± Sd) | 29.84 ± 6.12 | 30.93 ± 6.81 | 30.01 ± 6.24 | 0.214 |
| Residency | ||||
| Rural | 48(14.8) | 11(18.3) | 59(15.3) | 0.480 |
| Urban | 277(85.2) | 49(81.7) | 326(84.7) | |
| Education level | ||||
| Illiterate | 10(3.1) | 3(5) | 13(3.4) | 0.855 |
| < diploma | 176(54.2) | 33(55) | 209(54.3) | |
| Diploma | 64 (19.7) | 12(20) | 76(19.7) | |
| > diploma | 75(23.1) | 12(20) | 87(22.6) | |
| Occupation | ||||
| Housekeeper | 304(93.5) | 57(95) | 361(93.8) | 0.667 |
| Employed | 21(6.5) | 3 (5) | 24(6.2) | |
Data are presented as the mean and standard deviation for continuous variables and as n (%) for categorical variables.
*P < 0.05 was considered as a statistically significant level for the t-test and chi-square test.
Laboratory and clinical characteristics of pregnant women with COVID-19
Laboratory and Clinical records of all pregnant women with COVID-19 who delivered from March 21 2020-March 21 2021 were collected by trimester of pregnancy, This characteristics of these patients are summarized in Table 2. 60(15.6%) cases of LBW were among all deliveries(Table 2).
Table 2.
Laboratory and pregnancy characteristics of pregnant women with COVID-19 (N=385).
| Variables | Normal birth weight(N = 325) | LBW (N = 60) | Total(N = 385) | P-value | |||||
|---|---|---|---|---|---|---|---|---|---|
| First trimester | Second trimester | First trimester | Second trimester | First trimester | Second trimester | First trimester | Second trimester | ||
| Hemoglobin (g/dL), (Mean ± Sd) | 12.37 ± 1.43 | 11.62 ± 1.02 | 11.2 ± 1.81 | 10.82 ± 1.16 | 12.18 ± 1.55 | 11.49 ± 1.08 | < 0.001 | < 0.001 | |
| Hematocrit, (Mean ± Sd) | 38.46 ± 3.07 | 36.03 ± 2.72 | 37.76 ± 2.45 | 35.93 ± 2.42 | 38.35 ± 2.99 | 36.01 ± 2.67 | 0.095 | 0.792 | |
| MCV, (Mean ± Sd) | 84.6 ± 5.27 | 86.92 ± 5.84 | 84.24 ± 6.74 | 85.95 ± 6.32 | 84.54 ± 5.52 | 86.76 ± 5.92 | 0.640 | 0.936 | |
| MCH(Mean Corpuscular Hemoglobin), (Mean ± Sd) | 28.27 ± 2.78 | 28.7 ± 2.4 | 28.07 ± 2.88 | 28.35 ± 2.58 | 28.24 ± 2.79 | 28.64 ± 2.43 | 0.620 | 0.316 | |
| MCHC(Mean Corpuscular Hemoglobin Concentration ), (Mean ± Sd) | 32.83 ± 1.85 | 32.76 ± 1.37 | 32.98 ± 1.63 | 32.63 ± 1.37 | 32.85 ± 1.82 | 32.73 ± 1.36 | 0.560 | 0.497 | |
| Bun(Blood urea nitrogen), (Mean ± Sd) | 14.01 ± 5.07 | - | 15.07 ± 5.5 | - | 14.17 ± 5.29 | - | 0.154 | - | |
| Creatinine, (Mean ± Sd) | 0.76 ± 0.15 | - | 0.8 ± 0.11 | - | 0.77 ± 0.14 | - | 0.086 | - | |
| Pre-pregnancy BMI (kg/m2), (Mean ± Sd) | 27.48 ± 4.85 | - | 28.46 ± 4.27 | - | 27.63 ± 4.76 | - | 0.146 | ||
| Anemia | 39(12) | 37(11.4) | 28(46.7) | 27(45) | - | - | < 0.001 | < 0.001 | |
| Preeclampsia/eclampsia, N(%) | 28 ( 8.6) | - | 17 (28.3) | - | 45(11.7) | - | < 0.001 | - | |
| GDM, N(%) | 35 (10.8) | - | 15(25) | - | 50(13) | - | 0.003 | ||
| Parity, N(%) | |||||||||
| Nullipara | 59 (18.2) | - | 7 (11.7) | - | 66 (17.1) | - | 0.221 | - | |
| Multipara | 266 (81.8) | - | 53 (88.3) | - | 319 (82.9) | - | - | - | |
| History of miscarriage, N(%) | 63 (19.4 ) | - | 21(35) | - | 84 (21.8) | - | 0.007 | - | |
| Blood group, N(%) | |||||||||
| A | 130 (40) | - | 23(38.3) | - | 153 (39.7) | - | 0.947 | - | |
| B | 64 (19.7) | - | 11 (18.3) | - | 75(19.5) | - | - | - | |
| AB | 21 (6.5) | - | 5(8.3) | - | 26(6.8) | - | - | - | |
| O | 110 (33.8) | - | 21(35) | - | 131(34) | - | - | - | |
| Blood Rh, N(%) | |||||||||
| Positive | 298 (91.7) | - | 55(91.7) | - | 353(91.7) | - | 0.995 | ||
| Gestational age at the time of diagnosis of COVID-19, N(%) | |||||||||
| First trimester | 30 (9.2) | - | 6 (10) | - | 63(9.35) | - | 0.543 | - | |
| Second trimester | 144(44.3) | - | 22 (36.7) | - | 166(43.12) | - | - | - | |
| Third trimester | 151 (46.5) | - | 32 (53.3) | - | 183 (47.53) | - | - | - | |
Data are presented as the mean and standard deviation for continuous variables and as n (%) for categorical variables.
*P < 0.05 was considered as a statistically significant level for the t-test and chi-square test.
Association of Hb levels and maternal anemia with LBW in the first trimester
The associations of Hb levels in the first trimester with LBW are shown in Table 3. Model 1 showed that LBW is significantly associated with anemia and Hb levels in the first trimester. After adjusting for age, BMI, parity, history of miscarriages, preeclampsia/eclampsia, GDM, and PTB potential confounders, the results indicated significant associations between Hb levels and LBW. Model 2 showed that the odds of LBW in mothers with Hb level < 110 g/L was 4.13 times greater than in women with Hb level ≥ 110 g/L [OR = 4.13, (95%CI 2.11–8.10)]. Also, the odds of LBW in mothers with Hb level 100–109 g/L and Hb level < 90 g/L were 4.32 and 6.25, respectively times greater than in women with Hb level ≥ 110 g/L [OR = 4.32, (95%CI 1.66–11.21)] and [OR 6.25, (95%CI 2.35–16.67)]( Table 3).
Table 3.
Logistic regression analyses of the association between hb levels (maternal anemia) in first trimester and LBW among Iranian pregnant women with COVID-19, ( N=385).
| Variable | Normal Birth Weight(N = 325) | LBW(N = 60) | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|---|---|
| N (%) | N (%) | OR | 95% CI | P-value | OR | 95% CI | P-value | |
| Anemia(Hb levels (g/L)) | ||||||||
| ≥ 110 | 286(88) | 32(53.4) | Ref | Ref | Ref | Ref | Ref | Ref |
| < 110 | 39(12) | 28(47.6) | 6.42 | 3.49–11.78 | < 0.001 | 4.13 | 2.11–8.10 | < 0.001 |
| Hb levels (g/L) | ||||||||
| ≥ 110 | 286(88) | 32(53.3) | Ref | Ref | Ref | Ref | Ref | Ref |
| 100–109 | 14(4.3) | 13(21.7) | 8.29 | 3.58–19.19 | < 0.001 | 4.32 | 1.66–11.21 | 0.003 |
| 90–99 | 11(3.4) | 5(8.3) | 4.06 | 1.33–12.43 | 0.014 | 1.92 | 0.54–6.85 | 0.31 |
| <90 | 14(4.3) | 10(16.7) | 6.38 | 2.62–15.54 | < 0.001 | 6.25 | 2.35–16.67 | < 0.001 |
Model 1: without adjustment.
Model 2: adjustment for age, BMI, parity and history of miscarriage, preeclampsia/eclampsia, GDM, and PTB.
Association of Hb levels and maternal anemia with LBW in the second trimester
The associations between Hb levels in the second trimester and LBW are presented in Table 4. In Model 1, LBW was found to be significantly associated with anemia and low Hb levels during the second trimester. After adjusting for potential confounders, including maternal age, BMI, parity, history of miscarriages, preeclampsia/eclampsia, gestational diabetes mellitus, and preterm birth, Model 2 confirmed a significant relationship between Hb levels and LBW.
Table 4.
Logistic regression analyses of the association between hb levels (maternal anemia) in second trimester and LBW among Iranian pregnant women with COVID-19, ( N=385).
| Variable | Normal Birth Weight(N = 325) | LBW(N = 60) | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|---|---|
| N (%) | N (%) | OR | 95% CI | P-value | OR | 95% CI | P-value | |
| Anemia(Hb levels (g/L) | ||||||||
| ≥ 105 | 288(88.6) | 33(55) | Ref | Ref | Ref | Ref | Ref | Ref |
| < 105 | 37(11.4) | 27(45) | 6.36 | 3.45–11.75 | < 0.001 | 3.91 | 1.98–7.75 | < 0.001 |
| Hb levels (g/L) | ||||||||
| ≥ 105 | 288(88.6) | 33(55) | Ref | Ref | Ref | Ref | Ref | Ref |
| 100–104 | 17(5.2) | 14(23.3) | 7.18 | 3.25–15.89 | < 0.001 | 4.42 | 1.86–10.52 | < 0.001 |
| 90–99 | 17(5.2) | 10(16.7) | 5.13 | 2.17–12.13 | < 0.001 | 3.01 | 1.15–7.88 | 0.025 |
| < 90 | 3(1) | 3(5) | 8.72 | 1.69-45.00 | 0.010 | 6.27 | 1.07–36.77 | 0.042 |
Model 1: without adjustment.
Model 2: adjustment for age, BMI, parity and history of miscarriage, preeclampsia/eclampsia, GDM, and PTB.
Specifically, the odds of LBW were 3.91 times higher in mothers with Hb levels < 105 g/L than those with Hb levels ≥ 105 g/L [OR = 3.91, 95% CI: 1.98–7.75]. Additionally, the odds of LBW in mothers with Hb levels between 100 and 104 g/L, 90–99 g/L, and < 90 g/L were 4.42, 3.01, and 6.27 times higher, respectively, than in those with Hb levels ≥ 105 g/L [OR = 4.42, 95% CI: 1.86–10.52; OR = 3.01, 95% CI: 1.15–7.88; OR = 6.27, 95% CI: 1.07–36.77] (Table 4).
Odds ratio of LBW in women with anemia in first and second trimesters compare to women without anemia
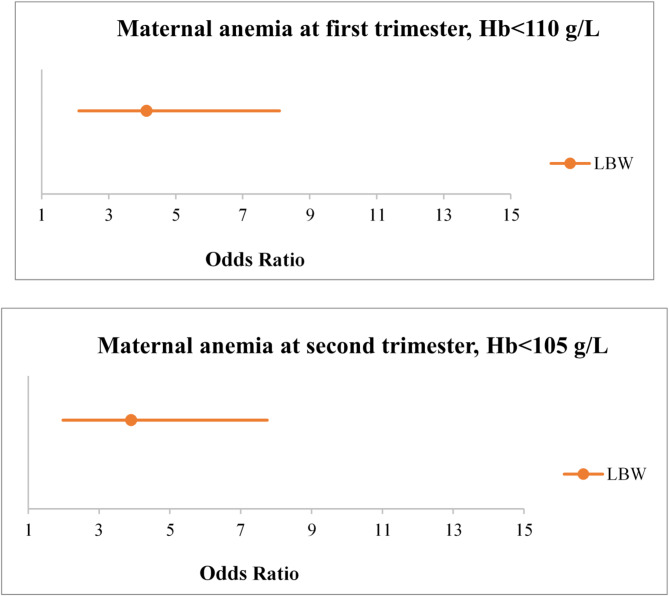
Figure 2 showed that women whose Hb < 110 g/L in the first trimester had increase odds of LBW [OR 4.13; (95% CI, 2.11–8.10)]. The same results were found in women with Hb levels < 105 g/L in the second trimester for LBW [OR, 3.91; (95% CI, 1.98–7.75)].
Fig. 2.
. Odds Ratio of LBW in women with anemia in first and second trimesters compare women without anemia.
Discussion
This study is cross-sectional in its study of the association between maternal anemia during pregnancy and LBW. In this regard, in the first and second trimesters, we observed a significant association between Hb levels and LBW while considering maternal age at birth, pre-pregnancy BMI, parity, history of miscarriage, preeclampsia/eclampsia, GDM, and PTB as confounding factors. In the first trimester, the odds of LBW in pregnant women with Hb level < 110 g/L was higher than in pregnant women with Hb level ≥ 110 g/L. Also, in the second trimester, this association was observed between pregnant women with Hb level < 105 g/L and participants with Hb level ≥ 105 g/L.
Anemia is a global public health issue, mainly being worst in low and middle-income countries (LMICs)31. People in LMICs often experience nutritional deficiency disorders. In Iran, which is one of the LMICs and is under severe international sanctions, it is more important to pay attention to anemia in pregnant women27. Our results showed a statistically significant association between maternal Hb levels during pregnancy and LBW. In the first trimester, the odds of LBW in anemic pregnant women with COVID-19 was 4.13 times more likely than in nonanemic pregnant women with COVID-19 [OR = 4.13, (95%CI 2.11–8.10)]. Also, in the first trimester, the odds of LBW in mothers with Hb level 100–109 g/L and Hb level < 90 g/L were 4.32 and 6.25, respectively times greater than in women with Hb level ≥ 110 g/L. The results of this study showed in the second trimester, the odds of LBW in mothers with Hb level 100–104 g/L, Hb level 90–99 g/L, and Hb level < 90 g/L were 4.42, 3.01, and 6.27, respectively, times more likely than pregnant women with Hb level ≥ 105 g/L. Kemppinen et al. showed that anemic women and newborns are prone to severe complications like PTB, FGR, and postpartum infections32.In Iranian women, Khezri et al. showed that maternal anemia during pregnancy was related to adverse pregnancy outcomes even after accounting for confounding factors21. A retrospective cohort analysis by Biswas et al. demonstrated that anemia during pregnancy significantly increases the risk of low birth weight (LBW). Similarly, Wahyuni et al. found a strong association between anemia during pregnancy and the incidence of LBW infants33,34. These findings are consistent with the current research. Additionally, a previous study indicated that individuals with COVID-19 who were anemic faced a 70% higher risk of near-term mortality compared to non-anemic individuals35, Moreover, COVID-19 patients with hemoglobin levels above 10 g/dL had a lower risk of mortality compared to those with hemoglobin levels below 10 g/dL35. Anemia reduces oxygen delivery to the fetus by lowering hemoglobin levels, impairing placental function, and disrupting angiogenesis3. This oxygen deficit can restrict fetal growth, leading to adverse pregnancy outcomes such as preterm birth (PTB) or LBW36. Additionally, inadequate nutrient intake during pregnancy, often observed in anemic women, further increases the risk by depriving the fetus of essential nutrients for growth37. Iran is currently grappling with a challenging economic situation due to strict sanctions27, resulting in widespread livelihood and health issues, including insufficient nutrient intake and iron deficiency anemia among pregnant women. Iron deficiency anemia requires careful management because of its health risks for both mother and fetus38. The sanctions have also contributed to reduced weight gain in both pregnant women and newborns, underscoring their adverse effects on maternal and infant nutrition in Iran39.
In Iran, the removal of all international sanctions is crucial to improving overall health, particularly for pregnant women. The sanctions have severely hindered Iranians’ access to essential health services and food security, with high-risk groups, such as pregnant women, being disproportionately affected. To reduce anemia among pregnant women and safeguard the health of future generations, it is essential to improve living conditions, enhance access to health services, ensure food security, and provide free and accessible iron supplements.
Strengths and limitations of the study
The primary strength of this study is its novel focus, being the first, to our knowledge, to examine the association between maternal hemoglobin (Hb) levels during the first and second trimesters of pregnancy and low birth weight (LBW) among infants born to mothers with COVID-19. Additionally, the study benefits from a robust sample size, allowing for a comprehensive analysis of the relationship between maternal Hb levels and LBW in this population. Furthermore, the multi-center design, encompassing data from three counties, enhances the generalizability of the findings.
However, this study has several limitations. First, as a cross-sectional study, causal inferences cannot be drawn from the observed associations. Second, the RT-PCR test for COVID-19 used in the study may have yielded false positives, as its reported sensitivity is 0.68 (95% probability interval [PrI]: 0.63–0.73) and specificity is 0.99 (95% PrI: 0.98–1.00)40. Lastly, the reliance on electronic health records posed limitations due to the lack of detailed information, such as data on certain chronic conditions and comorbidities, which may have influenced the results.
Conclusion
This study highlights the critical importance of managing maternal anemia in pregnant women with COVID-19, as anemia significantly increases the odds of LBW in newborns. To prevent adverse pregnancy outcomes in low- and middle-income countries such as Iran, addressing maternal anemia must be prioritized.
The removal of international sanctions is essential to maintaining and improving the health and nutritional status of pregnant women, reducing iron deficiency anemia, and ensuring the well-being of future generations. Lifting sanctions would improve access to health services, including nutritional supplements, vaccines, and laboratory diagnostics, while enhancing food security for high-risk groups, such as pregnant women. Additionally, ensuring the free and widespread availability of iron supplements for pregnant women is crucial.
Future research should focus on conducting longitudinal studies with adequate sample sizes to establish a causal relationship between maternal anemia and adverse pregnancy outcomes. These efforts will provide more substantial evidence to guide interventions to improve maternal and infant health outcomes.
Acknowledgements
The authors would like to express their deepest gratitude to all healthcare personnel at Urmia University of Medical Sciences.
Author contributions
R.K.H. and K.E. were the study’s principal investigators and advisors. R.K.H., K.E., and F.R. performed the statistical analysis. S.J. provided critical appraisal and analytical support. All authors contributed to the design and data analysis and assisted in preparing the final version of the manuscript. All authors read and approved the manuscript.
Funding
This research did not receive any specific grants from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Rozhan Khezri, Email: khezri.rojan@yahoo.com.
Kamran Ebrahimi, Email: kamran.ebrahimi4377@gmail.com.
References
- 1.Kramer, M. S. & Victora, C. G. Low Birth Weight and Perinatal Mortality. Nutrition and Health in Developing Countriesp. 57–69 (Springer, 2001).
- 2.World Health Organization (WHO). Global nutrition targets 2025: low birth weight policy brief. Accessed December 30 (2014). Available from: https://www.who.int/publications/i/item/WHO-NMH-NHD-14.5
- 3.Engidaw, M. T., Eyayu, T. & Tiruneh, T. The effect of maternal anaemia on low birth weight among newborns in Northwest Ethiopia. Sci. Rep.12 (1), 15280 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kliegman, R. M., Behrman, R. E., Jenson, H. B. & Stanton, B. M. Nelson Textbook of Pediatrics e-book (Elsevier Health Sciences, 2007).
- 5.Ahmad, M. O., Kalsoom, U., Sughra, U., Hadi, U. & Imran, M. Effect of maternal anaemia on birth weight. J. Ayub Med. Coll. Abbottabad. 23 (1), 77–79 (2011). [PubMed] [Google Scholar]
- 6.Goonewardene, M., Shehata, M. & Hamad, A. Anaemia in pregnancy. Best practice & research Clinical obstetrics & gynaecology. ;26(1):3–24. (2012). [DOI] [PubMed]
- 7.Horowitz, K. M., Ingardia, C. J. & Borgida, A. F. Anemia in pregnancy. Clin. Lab. Med.33 (2), 281–291 (2013). [DOI] [PubMed] [Google Scholar]
- 8.World Health Organization (WHO). THE GLOBAL HEALTH OBSERVATORY/ Anaemia in women and children. (2019). Available from: https://www.who.int/data/gho/data/themes/topics/anaemia_in_women_and_children
- 9.Azami, M., Darvishi, Z., Borji, M. & Sayehmiri, K. The prevalence of anemia among pregnant women in Iran (2005–2016); a systematic review and meta-analysis study. J. School Public. Health Inst. Public. Health Res. ;14(4–5), e38462 (2016).
- 10.Taghizadeh, R., Khadem al-hosseini, M., Janani, L. & Amiri, F. Prevalence of Iron deficiency Anemia among pregnant women referred to Fardis health centers in 2018. Alborz Univ. Med. J.11 (3), 271–279 (2022). [Google Scholar]
- 11.Iqbal, S. & Ekmekcioglu, C. Maternal and neonatal outcomes related to iron supplementation or iron status: a summary of meta-analyses. J. Maternal-Fetal Neonatal Med.32 (9), 1528–1540 (2019). [DOI] [PubMed] [Google Scholar]
- 12.Khezri, R., Rezaei, F. & Motevalli, F. D. Covid-19 pandemic and risk of myopia. Annals Med. Surg. ;79, 1–2 (2022). [DOI] [PMC free article] [PubMed]
- 13.Khezri, R., Nikbakht, H-A., Aljalili, S. & Ghelichi-Ghojogh, M. Is it possible to eradicate the covid-19 or not? New Microbes and New Infections. ;49. (2022). [DOI] [PMC free article] [PubMed]
- 14.Khezri, R., Shojaie, L., Nikbakht, H-A., Jahanian, S. & Ghelichi-Ghojogh, M. Coronavirus Pandemic and Pregnant Mothers (Annals of Medicine and Surgery, 2022). [DOI] [PMC free article] [PubMed]
- 15.Yang, R. et al. Pregnant women with COVID-19 and risk of adverse birth outcomes and maternal-fetal vertical transmission: a population-based cohort study in Wuhan, China. BMC Med.18 (1), 1–7 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.World Health Organization (WHO). Home/Diseases/Coronavirus disease (COVID-19)/COVID-19 vaccines. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines. Cited 25 January 2025.
- 17.Ciapponi, A. et al. Safety of COVID-19 vaccines during pregnancy: a systematic review and meta-analysis. Vaccine41 (25), 3688–3700 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wei, S. Q., Bilodeau-Bertrand, M., Liu, S. & Auger, N. The impact of COVID-19 on pregnancy outcomes: a systematic review and meta-analysis. Cmaj193 (16), E540–e8 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Surekha, M. V. et al. Impact of COVID-19 during pregnancy on placental pathology, maternal and neonatal outcome - A cross-sectional study on anemic term pregnant women from a tertiary care hospital in Southern India. Front. Endocrinol. (Lausanne). 14, 1092104 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dewey, K. G. & Oaks, B. M. U-shaped curve for risk associated with maternal hemoglobin, iron status, or iron supplementation. Am. J. Clin. Nutr.106 (suppl_6), 1694S–702S (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Khezri, R., Salarilak, S. & Jahanian, S. The association between maternal anemia during pregnancy and preterm birth. Clin. Nutr. ESPEN. 56, 13–17 (2023). [DOI] [PubMed] [Google Scholar]
- 22.Chu, F-C., Shao, S. S. W., Lo, L-M. & Hung, T-H. Association between maternal anemia at admission for delivery and adverse perinatal outcomes. J. Chin. Med. Association. 83 (4), 402–407 (2020). [DOI] [PubMed] [Google Scholar]
- 23.Hämäläinen, H., Hakkarainen, K. & Heinonen, S. Anaemia in the first but not in the second or third trimester is a risk factor for low birth weight. Clin. Nutr.22 (3), 271–275 (2003). [DOI] [PubMed] [Google Scholar]
- 24.Kim, S-Y., Kim, S-Y., Kil, K. & Lee, Y. Impact of COVID-19 mitigation policy in South Korea on the reduction of preterm or low birth weight birth rate: a single center experience. Children8 (5), 332 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Surya, S. S., Jamil, N. A., Cahyanti, D., Rahma, A. & Dewi, T. M. Anemia in Pregnancy and Low Birth Weight Before and During the COVID-19 Pandemic in Kalijambe. (2021).
- 26.World Bank Group. Data: Iran, Islamic Rep. Accessed July 26 (2024). Available from: http://data.worldbank.org/country/iran-islamic-republic
- 27.Number of international sanctions imposed worldwide as of January 12. by target country. [cited 2024 July 26]. (2023). https://www.statista.com/statistics/1294726/number-of-global-sanctions-by-target-country/
- 28.World Health Organization (WHO). Response to COVID-19 in Iran. Cited July 31 (2024). Available from: https://www.who.int/about/accountability/results/who-results-report-2020-mtr/country-story/2021/iran
- 29.Glodean, D. M., Miclea, D. & Popa, A. R. Macrosomia. A systematic review of recent literature. Romanian J. Diabetes Nutr. Metabolic Dis.25 (2), 187–195 (2018). [Google Scholar]
- 30.Organization, W. H. The Global Prevalence of Anaemia in 2011 (World Health Organization, 2015).
- 31.Araujo Costa, E. & de Ayres-Silva, P. Global profile of anemia during pregnancy versus country income overview: 19 years estimative (2000–2019). Ann. Hematol.102 (8), 2025–2031 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kemppinen, L. et al. Gestational iron deficiency anemia is associated with preterm birth, fetal growth restriction, and postpartum infections. J. Perinat. Med.49 (4), 431–438 (2021). [DOI] [PubMed] [Google Scholar]
- 33.Biswas, P., Samsuzzaman, M., Chakraborty, A. & Das, D. K. Maternal anemia and low birth weight in a community development block of purba Bardhaman, West Bengal: a retrospective cohort analysis. J. Maternal and Child Health (2019).
- 34.Wahyuni, S., Putri, A. R. A. & Imbir, S. The Relationship Of Anemia In Pregnancy With The Event Of LBW Babies (Low Birth Weight) at Supiori Hospital. JURNAL KEBIDANAN KESTRA (JKK). ;4(2):108 – 12. (2022).
- 35.Al-Jarallah, M. et al. In‐hospital mortality in SARS‐CoV‐2 stratified by hemoglobin levels: A retrospective study. EJHaem2 (3), 335–339 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Figueiredo, A. C. et al. Maternal anemia and low birth weight: a systematic review and meta-analysis. Nutrients10 (5), 601 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Widiyanto, J. & Lismawati, G. Maternal age and anemia are risk factors of low birthweight of newborn. Enfermeria Clin.29, 94–97 (2019). [Google Scholar]
- 38.Tandon, R., Jain, A. & Malhotra, P. Management of iron deficiency anemia in pregnancy in India. Indian J. Hematol. Blood Transfus.34, 204–215 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gohari, M. et al. Effects of Post-Joint comprehensive plan of action sanctions on the nutritional status and food security of pregnant women and their families (2017–2020). J. Nutrition and Food Security (2022).
- 40.Kostoulas, P., Eusebi, P. & Hartnack, S. Diagnostic accuracy estimates for COVID-19 real-time polymerase chain reaction and lateral flow immunoassay tests with bayesian latent-class models. Am. J. Epidemiol.190 (8), 1689–1695 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.