Abstract
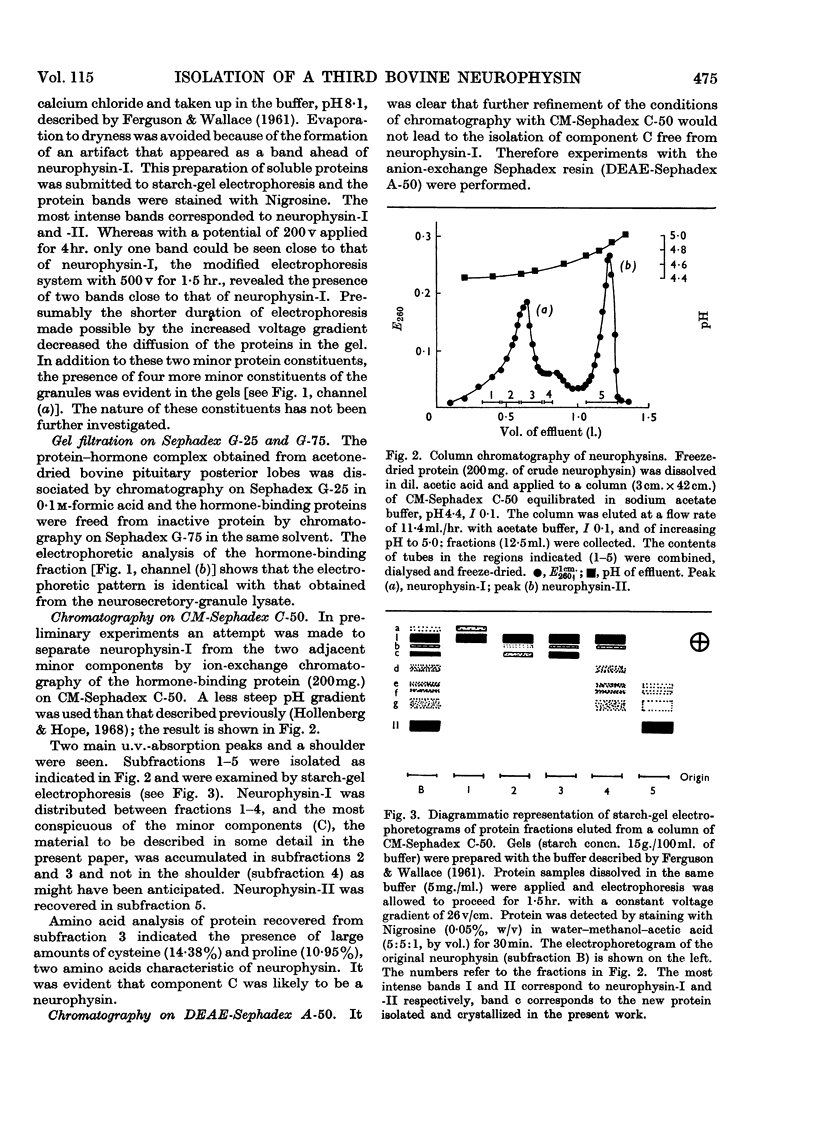
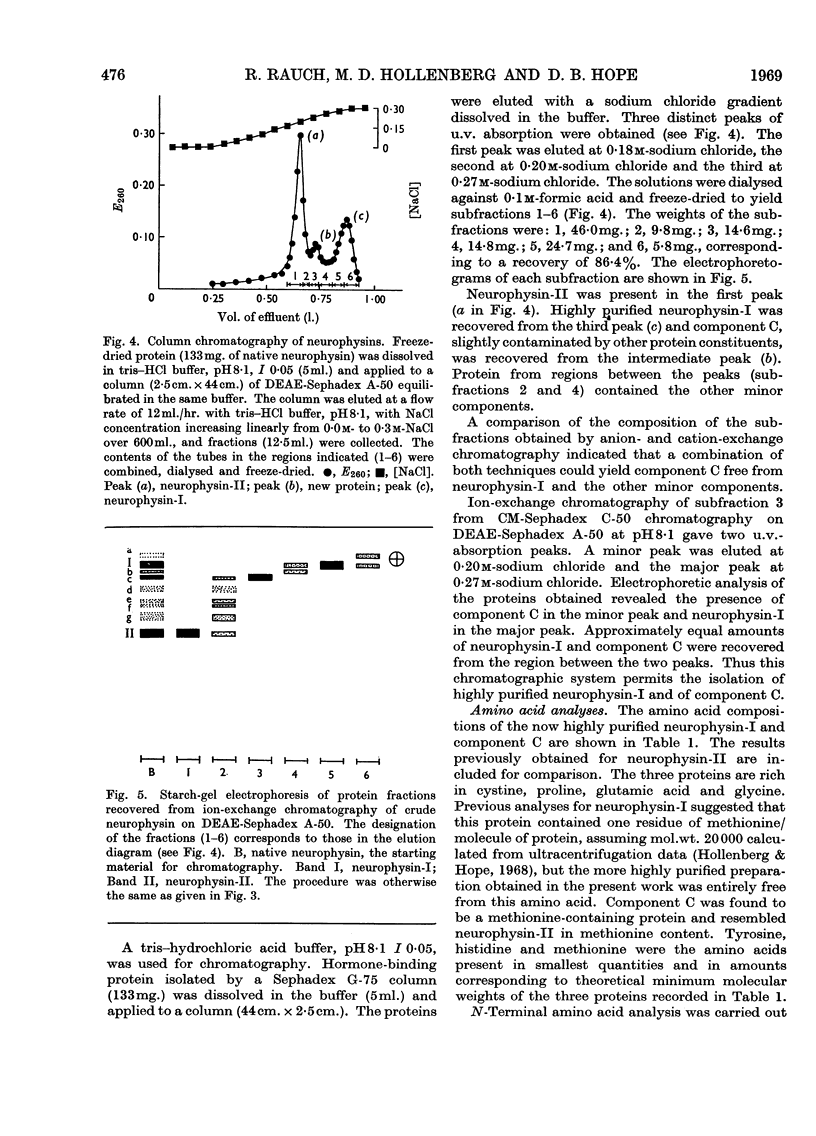
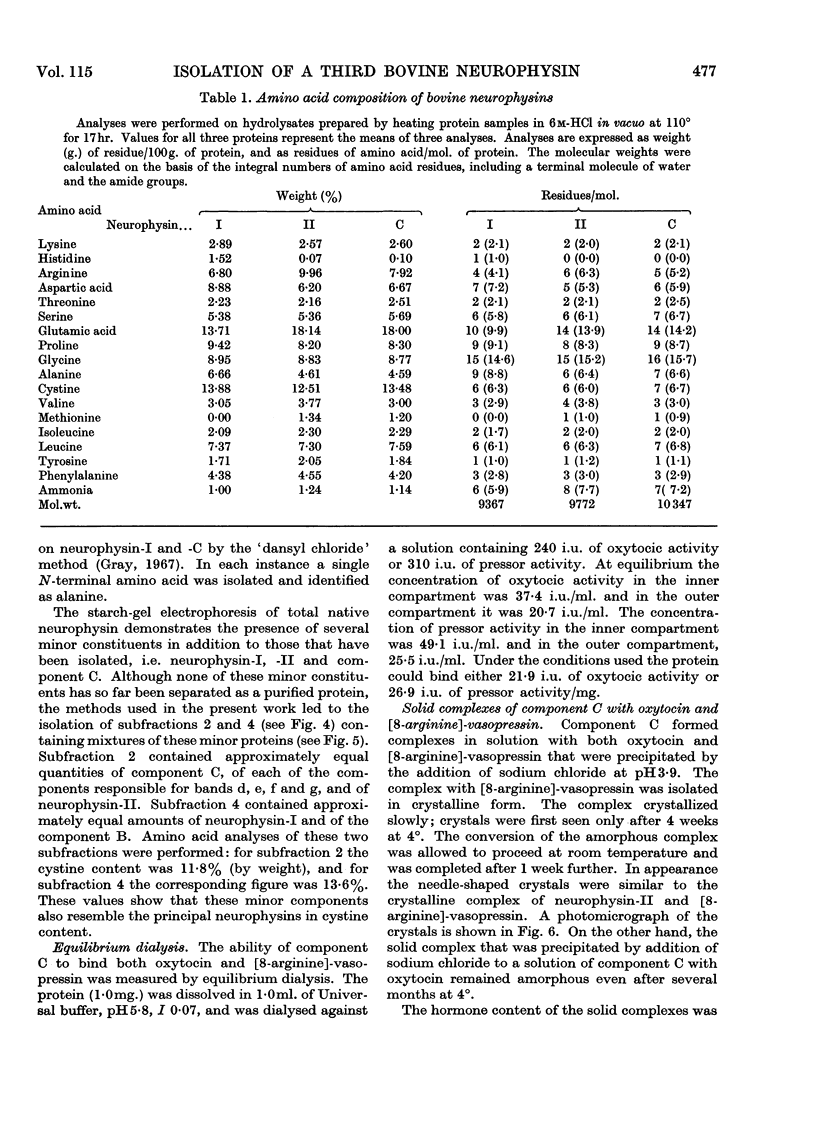
1. A third native hormone-binding protein, neurophysin-C, has been isolated from acetone-desiccated bovine pituitary posterior lobes. 2. This protein was detected in lysates of neurosecretory granules isolated from bovine pituitary posterior lobes. 3. The molecular weight appears to be close to 10000. 4. Neurophysin-C is similar in amino acid composition to neurophysin-I and -II; it contains a single residue of tyrosine and of methionine. The N-terminal amino acid in all three neurophysins is alanine. 5. Neurophysin-C accounts for approximately 15% of the total hormone-binding protein present in the pituitary posterior lobes. 6. The new neurophysin forms complexes with oxytocin as well as with [8-arginine]-vasopressin. The complex with vasopressin has been crystallized. 7. Bioassay of the pressor and oxytocic activities of the protein–hormone complexes shows that neurophysin-C binds one molecule of either vasopressin or oxytocin.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BANGHAM D. R., MUSSETT M. V. Third international standard for posterior pituitary; re-named third international standard for oxytocic, vasopressor and antidiuretic substances in 1956. Bull World Health Organ. 1958;19(2):325–340. [PMC free article] [PubMed] [Google Scholar]
- CHAN W. Y., O'CONNELL M., POMEROY S. R. Effects of the estrous cycle on the sensitivity of rat uterus to oxytocin and desamino-oxytocin. Endocrinology. 1963 Feb;72:279–282. doi: 10.1210/endo-72-2-279. [DOI] [PubMed] [Google Scholar]
- CRESTFIELD A. M., MOORE S., STEIN W. H. The preparation and enzymatic hydrolysis of reduced and S-carboxymethylated proteins. J Biol Chem. 1963 Feb;238:622–627. [PubMed] [Google Scholar]
- DEKANSKI J. The quantitative assay of vasopressin. Br J Pharmacol Chemother. 1952 Dec;7(4):567–572. doi: 10.1111/j.1476-5381.1952.tb00723.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean C. R., Hope D. B., Kazić T. Evidence for the storage of oxytocin with neurophysin-I and of vasopressin with neurophysin-II in separate neurosecretory granules. Br J Pharmacol. 1968 Sep;34(1):192P–193P. [PMC free article] [PubMed] [Google Scholar]
- Dean C. R., Hope D. B. The isolation of neurophysin-I and-II from bovine pituitary neurosecretory granules separated on a large scale from other subcellular organelles. Demonstration of slow equilibration of neurosecretory granules during centrifugation in a sucrose density gradient. Biochem J. 1968 Jan;106(2):565–573. doi: 10.1042/bj1060565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean C. R., Hope D. B. The isolation of purified neurosecretory granules from bovine pituitary posterior lobes. Comparison of granule protein constituents with those of neurophysin. Biochem J. 1967 Sep;104(3):1082–1088. doi: 10.1042/bj1041082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FERGUSON K. A., WALLACE A. L. Starch-gel electrophoresis of anterior pituitary hormones. Nature. 1961 May 13;190:629–630. doi: 10.1038/190629a0. [DOI] [PubMed] [Google Scholar]
- Frankland B. T., Hollenberg M. D., Hope D. B., Schacter B. A. Dissociation of oxytocin and vasopressin from their carrier protein by chromatography on sephadex G-25. Br J Pharmacol Chemother. 1966 Feb;26(2):502–510. doi: 10.1111/j.1476-5381.1966.tb01931.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hollenberg M. D., Hope D. B. The composition of crystalline complexes of neurophysin-M with [8-arginine]-vasopressin and oxytocin. Biochem J. 1967 Dec;105(3):921–926. doi: 10.1042/bj1050921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hollenberg M. D., Hope D. B. The isolation of the native hormone-binding proteins from bovine pituitary posterior lobes. Crystallization of neurophysin-I and-II as complexes with [8-arginine]-vasopressin. Biochem J. 1968 Jan;106(2):557–564. doi: 10.1042/bj1060557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MUNSICK R. A. Effect of magnesium ion on the response of the rat uterus to neurohypophysial hormones and analogues. Endocrinology. 1960 Mar;6:451–457. doi: 10.1210/endo-66-3-451. [DOI] [PubMed] [Google Scholar]
- Morse D., Horecker B. L. Thin-layer chromatographic separation of DNS-amino acids. Anal Biochem. 1966 Mar;14(3):429–433. doi: 10.1016/0003-2697(66)90285-5. [DOI] [PubMed] [Google Scholar]
- SMITHIES O. Zone electrophoresis in starch gels: group variations in the serum proteins of normal human adults. Biochem J. 1955 Dec;61(4):629–641. doi: 10.1042/bj0610629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WADDELL W. J. A simple ultraviolet spectrophotometric method for the determination of protein. J Lab Clin Med. 1956 Aug;48(2):311–314. [PubMed] [Google Scholar]



