Abstract
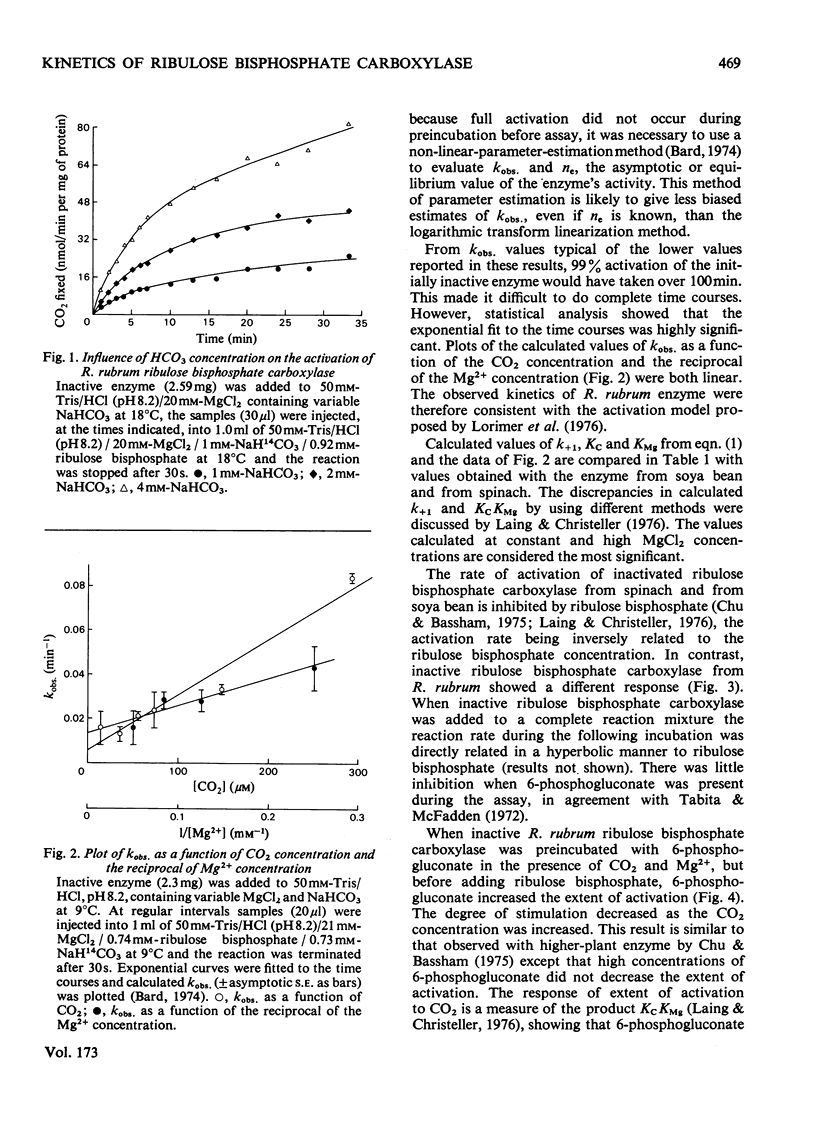
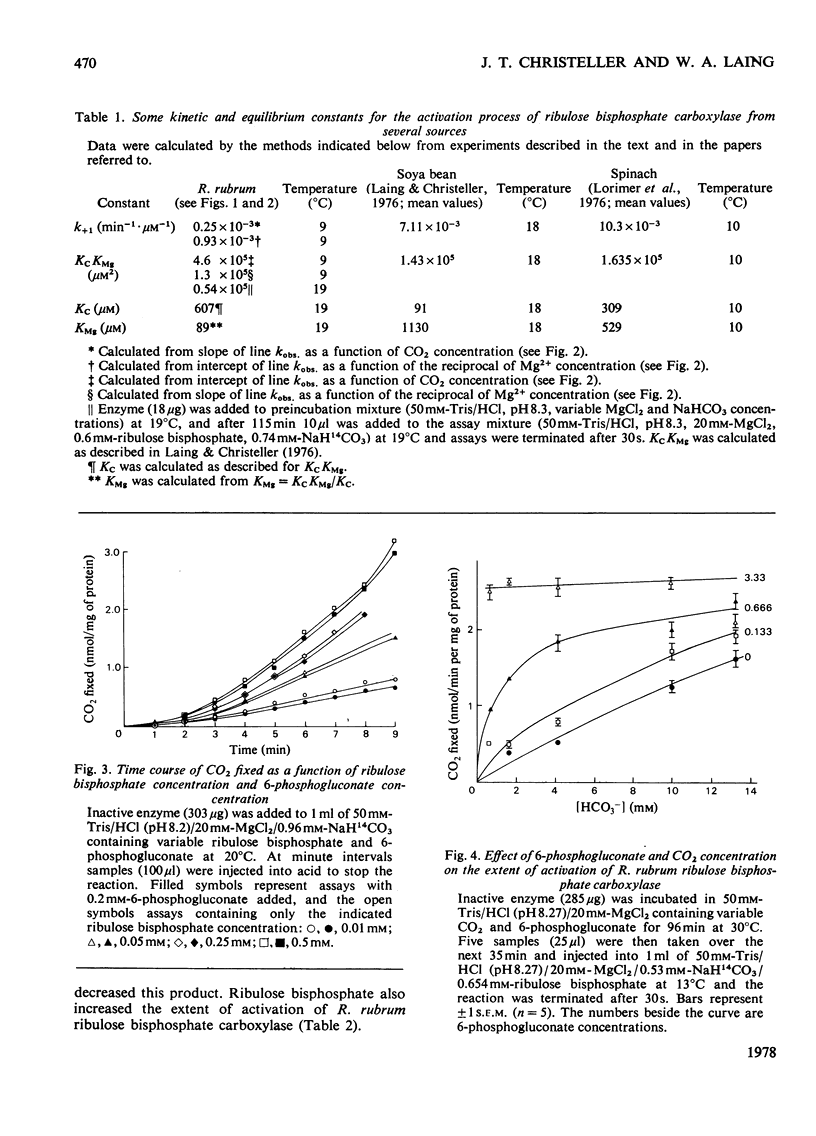
The activation kinetics of purified Rhodospirillum rubrum ribulose bisphosphate carboxylase were analysed. The equilibrium constant for activation by CO(2) was 600 micron and that for activation by Mg2+ was 90 micron, and the second-order activation constant for the reaction of CO(2) with inactive enzyme (k+1) was 0.25 X 10(-3)min-1 . micron-1. The latter value was considerably lower than the k+1 for higher-plant enzyme (7 X 10(-3)-10 X 10(-3)min-1 . micron-1). 6-Phosphogluconate had little effect on the active enzyme, and increased the extent of activation of inactive enzyme. Ribulose bisphosphate also increased the extent of activation and did not inhibit the rate of activation. This effect might have been mediated through a reaction product, 2-phosphoglycolic acid, which also stimulated the extent of activation of the enzyme. The active enzyme had a Km (CO2) of 300 micron-CO2, a Km (ribulose bisphosphate) of 11--18 micron-ribulose bisphosphate and a Vmax. of up to 3 mumol/min per mg of protein. These data are discussed in relation to the proposed model for activation and catalysis of ribulose bisphosphate carboxylase.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bassham J. A., Krause G. H. Free energy changes and metabolic regulation in steady-state photosynthetic carbon reduction. Biochim Biophys Acta. 1969 Oct 21;189(2):207–221. doi: 10.1016/0005-2728(69)90048-6. [DOI] [PubMed] [Google Scholar]
- Bowes G., Ogren W. L., Hageman R. H. Phosphoglycolate production catalyzed by ribulose diphosphate carboxylase. Biochem Biophys Res Commun. 1971 Nov 5;45(3):716–722. doi: 10.1016/0006-291x(71)90475-x. [DOI] [PubMed] [Google Scholar]
- Christeller J. T., Laing W. A. Isotope Discrimination by Ribulose 1,5-Diphosphate Carboxylase: No Effect of Temperature or HCO(3) Concentration. Plant Physiol. 1976 Apr;57(4):580–582. doi: 10.1104/pp.57.4.580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu D. K., Bassham J. A. Activation and inhibition of ribulose 1,5-diphosphate carboxylase by 6-phosphogluconate. Plant Physiol. 1973 Oct;52(4):373–379. doi: 10.1104/pp.52.4.373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu D. K., Bassham J. A. Activation of ribulose 1,5-diphosphate carboxylase by nicotinamide adenine dinucleotide phosphate and other chloroplast metabolites. Plant Physiol. 1974 Oct;54(4):556–559. doi: 10.1104/pp.54.4.556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu D. K., Bassham J. A. Regulation of ribulose 1,5-diphosphate carboxylase by substrates and other metabolites: further evidence for several types of binding sites. Plant Physiol. 1975 Apr;55(4):720–726. doi: 10.1104/pp.55.4.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laing W. A., Christeller J. T. A model for the kinetics of activation and catalysis of ribulose 1,5-bisphosphate carboxylase. Biochem J. 1976 Dec 1;159(3):563–570. doi: 10.1042/bj1590563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laing W. A., Ogren W. L., Hageman R. H. Bicarbonate stabilization of ribulose 1,5-diphosphate carboxylase. Biochemistry. 1975 May 20;14(10):2269–2275. doi: 10.1021/bi00681a035. [DOI] [PubMed] [Google Scholar]
- Lorimer G. H., Badger M. R., Andrews T. J. The activation of ribulose-1,5-bisphosphate carboxylase by carbon dioxide and magnesium ions. Equilibria, kinetics, a suggested mechanism, and physiological implications. Biochemistry. 1976 Feb 10;15(3):529–536. doi: 10.1021/bi00648a012. [DOI] [PubMed] [Google Scholar]
- McFadden B. A. Autotrophic CO2 assimilation and the evolution of ribulose diphosphate carboxylase. Bacteriol Rev. 1973 Sep;37(3):289–319. doi: 10.1128/br.37.3.289-319.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabita F. R., McFadden B. A. D-ribulose 1,5-diphosphate carboxylase from Rhodospirillum rubrum. I. Levels, purification, and effects of metallic ions. J Biol Chem. 1974 Jun 10;249(11):3453–3458. [PubMed] [Google Scholar]
- Tabita F. R., McFadden B. A. D-ribulose 1,5-diphosphate carboxylase from Rhodospirillum rubrum. II. Quaternary structure, composition, catalytic, and immunological properties. J Biol Chem. 1974 Jun 10;249(11):3459–3464. [PubMed] [Google Scholar]
- Tabita F. R., McFadden B. A. Regulation of ribulose-1,5-diphosphate carboxylase by 6-phospho-D-gluconate. Biochem Biophys Res Commun. 1972 Sep 5;48(5):1153–1159. doi: 10.1016/0006-291x(72)90831-5. [DOI] [PubMed] [Google Scholar]
- Weber K., Pringle J. R., Osborn M. Measurement of molecular weights by electrophoresis on SDS-acrylamide gel. Methods Enzymol. 1972;26:3–27. doi: 10.1016/s0076-6879(72)26003-7. [DOI] [PubMed] [Google Scholar]


