Abstract
Inpatient infection prevention practices vary widely between health care institutions, on a national and international scale. Transmissible infectious diseases pose a risk to vulnerable inpatient populations as well as hospital staff and visitors. Maintaining a high index of suspicion, along with early identification and prompt isolation procedures of patients with these conditions can greatly reduce the likelihood of transmission. We review the presenting features and recommended infection prevention practices for six transmissible infectious diseases known to pose risks to patients, staff and visitors: scabies, tuberculosis, respiratory viruses, varicella zoster virus, Clostridioides difficile, and syphilis.
Keywords: scabies, respiratory viruses, tuberculosis, clostroides difficile, varicella, infection control, syphilis
Background
Hospitals world-wide serve as a catch-all mechanism for patients with a broad range of diseases. Hospital-based providers, in the emergency department and inpatient settings, are faced with rapid identification and triage of transmissible infectious diseases that pose a safety risk to other hospitalized patients and hospital staff. Although the previous three decades have seen a rise in hospital-based infection prevention programs in addition to expanding knowledge about prevention methods such as hand hygiene and appropriate isolation, knowledge amongst providers appears to be variable.1 Outbreaks of nosocomial infections are common world-wide suggesting that there is much progress to be made in improving patient safety in this domain.2–4 We review six commonly presenting conditions associated with high likelihood of nosocomial spread and suggest strategies for effective infection prevention.
Scabies and Crusted (Norwegian) Scabies
Scabies, a condition caused by the mite, Sarcoptes scabiei varietas hominis, is a cutaneous disease characterized by a pruritic rash often affecting immunocompromised, malnourished and immobilized individuals and inadequate self-hygiene.5 The mites reproduce by digging burrows and laying eggs in the stratum corneum of human skin. Cutaneous manifestations include papular, papulo-vesicular or nodular rashes affecting the trunk and extremities.6 Crusted scabies, also known as crusted (Norwegian) scabies, an extreme form of scabies, characterized by uncontrolled mite replication and extensive crusting (hyperkeratosis) of the hands, feet, elbows and neck, can occur in individuals who have chronic diseases, are immunosuppressed, have compromised skin functionality, or have impaired physical capability.7,8 Appearance of scabies in the elderly adult may be atypical; 51% of patients were asymptomatic and 25% lacked evidence of cutaneous burrows.9 Failure to find mites in the skin does not rule out infection.10 The risk of spread, particularly in the hospital, is very high as patients who are not able to mount an immune response are more likely to be impacted. Mites can survive outside the human body for 24-48 hours at room temperature and relatively humid air.5 Infection may be transmitted by a single mite. Missed and delayed diagnosis leads to nosocomial spread with outbreaks lasting three months on average.11,12
An identified outbreak should be brought to the attention of the local health department and patients with an active scabies infestation should be immediately isolated to reduce spread.13 The speed of response and intervention dramatically decreases spread. Topical permethrin and oral ivermectin are used to treat scabies in affected cases thereby also limiting spread.14 Staff and visitors who interact with infected patients should follow strict contact precautions and always wear appropriate protective equipment including gloves, a gown, and shoe covers. The number of people that interact with the patient should be limited to reduce spread. Thorough hand sanitation should be done before and after contact. Similarly to crusted scabies, all persons who interact with patients with non-crusted scabies should avoid direct skin-to-skin contact and wear appropriate PPE including gloves. Thorough hand sanitation should be performed before and after contact.
For both crusted and non-crusted scabies, used linens should be washed and dried at a temperature of at least 50℃ (122℉).15 All persons who have come in contact with the patient, including visitors and family members prior to known infestation, should be identified and treated immediately. Though thorough decontamination measures are not required for non-crusted scabies once patients have been discharged, the process is necessary for crusted scabies.
Tuberculosis
Tuberculosis (TB) is an infectious disease caused by the bacterium Mycobacterium tuberculosis which primarily spreads through airborne particles produced by infected individuals when they speak, cough, sneeze, or sing. Though TB cases have gradually declined in the US since 1993, it remains a significant cause of morbidity and mortality worldwide.16,17 The majority of TB cases in the US are in foreign born individuals from endemic areas of the world. TB is believed to be primarily transmitted indoors via airborne particles and outbreaks have occurred in hospitals and other congregate settings.18 Shared patient rooms, immunocompromised state of hospitalized patients, and delayed isolation may lead to transmission and increased risk of severe disease. The role of the clinician is rapid recognition and isolation of the patient with suspected and/or confirmed pulmonary TB. Factors associated with delayed isolation include older age, comorbid malignancy, admission department other than Infectious Diseases and Pulmonary, and the absence of the diagnosis of active TB on the admission chest radiograph.18 Conditions such as malignancy and autoimmune disease are also associated with delayed diagnosis of TB.19 Clinicians must carefully consider respiratory and constitutional symptoms (cough, weight loss, fatigue) and abnormal chest radiograph findings in patients possessing risk factors for TB including exposure to an infected person, human immunodeficiency virus (HIV) infection, medically underserved populations, immigration from a geographic area with high TB incidence, history of incarceration, and others (Table 1).20
Table 1. Risk factors associated with tuberculosis transmission.
| Immunocompromised state (human immunodeficiency virus, diabetes mellitus, other immunosuppression including biological agents) |
| Injection drug use |
| Alcohol use disorder |
| Housing insecurity |
| Birth in or travel to endemic area |
| Contact with tuberculosis-infected person |
| Personal history of tuberculosis treatment |
| Exposure to high-risk congregate settings (prison, shelter, refugee camp) |
Hospital inpatient interventions are based on a three-level hierarchy of control measures: administrative measures, environmental controls, and use of protective equipment (PPE).21
Developing infection prevention policies, procedures, and guidelines that align with established standards is crucial in preventing TB infections.22
Isolation of patients in airborne infection isolation rooms and management of ventilation and adequate air exchange in the hospital are important in maintaining diligence when considering environmental controls.23
Protective equipment includes N95 masks which should be worn by health workers when entering room of patients with suspected or confirmed TB.
Effective treatment of TB rapidly leads to decreased infectiousness.
Respiratory Viruses
Infection caused by respiratory viruses (RV) can cause upper and lower respiratory infections (URI), with a clinical presentation characterized by myalgias, low-grade fever, nasal congestion, and rhinorrhea.24 URI is benign, is the most common infection worldwide, and is known as the common cold. Over 200 viruses have been found to cause URI. Among them are rhinoviruses, metapneumoviruses, parainfluenza, and strains of adenovirus and coronavirus.25 Influenza and respiratory syncytial virus are RVs that cause illnesses more severe than URI that can be potentially deadly in infants and individuals with comorbidities.26 Hospital acquired influenza when compared to community acquired influenza is associated with a higher rate of ICU admission, need for mechanical ventilation and death.27,28 Furthermore, RVs can be the cause of pandemics, such as the coronavirus SARS-CoV-2, which causes COVID-19 and is more severe than circulating RVs and possibly lethal in healthy individuals. RV transmission is usually seasonal, with higher transmission in cooler months.25 Rhinovirus is the most common pathogen in adults and is the leading cause of community acquired pneumonia in hospitalized patients.29,30
Despite the vast number of respiratory viruses, infection mitigation strategies between them are similar. The cornerstone in preventing RV transmission is appropriate masking and hand hygiene, namely washing hands with soap and water for 20 seconds or using approved alcohol-based hand sanitizer after each encounter with a patient. This has been repeatedly shown to decrease RV transmission both in the community and in hospital settings.26,31 The other hallmark in RV mitigation is source control with surgical grade face masks, which prevent spread of respiratory droplets (those >5µm) from individuals with suspected RV.32 Healthcare workers who interact with suspected or confirmed RV patients should wear surgical masks, don gowns and gloves, and wear eye protection.26 N95 respirators should be used for protection from COVID-19. In periods where RV transmission is high, universal masking by all individuals regardless of symptom status might be recommended by local or CDC guidelines. For patients with confirmed or suspected URI, isolation protocols are often employed. Lastly, a yearly recombinant vaccination and mRNA vaccination exist for influenza and COVID-19, and these have been shown to effectively prevent some infections and markedly reduce the severity of infection with these viruses.26
Varicella Zoster Virus
Varicella Zoster Virus (VZV) is a highly contagious pathogen that is transmitted via respiratory droplets and is a well-documented source of nosocomial infection. Primary VZV infections result in a diffuse, generally self-limiting, vesicular rash known as “chickenpox”.33 VZV then has the ability to lay dormant and cause endogenous secondary infections decades later, which is known as “shingles”.33 Given the potential for serious complications in neonates and immunocompromised persons, several measures must be put in place to limit the spread of VZV in the hospital setting. The CDC provides a framework for preventing transmission from two perspectives: patient isolation and healthcare personnel prophylaxis.34 With respect to patient isolation, any patient who is admitted to the hospital and is suspected, or confirmed, to have a primary varicella or disseminated VZV infection should be placed on contact and airborne precautions. These precautions should consist of alcohol-based hand hygiene after each patient contact in combination with the donning of a gown, gloves, eye protection, and the use of a certified N95 respirator by personnel in patient care areas. Patients should be kept on these precautions until the lesions have dried and crusted over.35,36 With respect to protection of hospital personnel, the CDC recommends all personnel be adequately immunized to VZV at the time of initial employment; confirmation of sufficient immunization status can be achieved through written documentation show the receipt of the two-dose varicella vaccine series, laboratory evidence of disease-induced immunity, or written documentation of prior varicella diagnosis by a healthcare provider.36
Clostridioides difficile
Clostridioides difficile (formerly known as Clostridium difficile), a spore forming gram-positive anaerobic bacillus, is the leading cause of nosocomial diarrheal illness and is an important focus with respect to institutional infection prevention.37,38 In 2014, C. difficile was the number one organism responsible for health care associated infections.39 Per the current CDC recommendations, any patient with a suspected or confirmed Clostridioides difficile infection (CDI) should be placed on isolation and contact precautions.37 These precautions should consist of, when feasible, placing patients in single-patient rooms, having dedicated patient-care equipment, and ensuring proper contact precautions are adhered to when the patient is transported throughout the facility; all hospital employees who are entering a patient’s room should don the appropriate personal protective equipment, which would consist of a high-quality gowns and gloves at a minimum.37,38 Contact precautions should ideally be in place for at least 48 hours after the resolution of diarrheal symptoms, but can be left in place up to the duration of the hospitalization due to Clostridioides difficile’s prolonged spore shedding time.40 In addition to these measures, adequate hand hygiene, should be employed by all hospital staff members and the patient after each encounter.41 Another essential policy that should be enacted in prevention of CDI is appropriate environmental cleaning procedures; the CDC recommends all CDI patient rooms and equipment undergo a daily cleaning, in addition to a terminal cleaning at the end of the patient’s stay, with a Clostridioides difficile sporicidal agent (EPA List K agents).42 Lastly, healthcare providers should make concerted efforts to practice effective antibiotic stewardship by limiting the use and duration of antibiotics that increase the risk of CDI, such as fluoroquinolones, carbapenems, and 3rd/4th generation cephalosporins.42
Syphilis
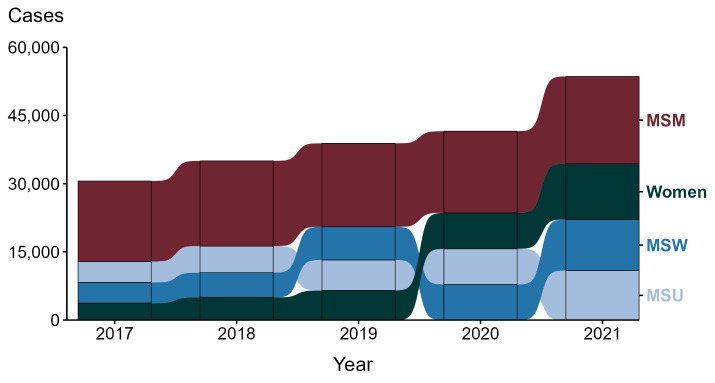
The transmission of syphilis is commonly associated with sexual intercourse but can also occur through other means. In the past, when syphilis was more prevalent, there were reports of occupational infection, where the infectious agent was acquired by a healthcare provider during the provision of a medical service. Prior to the standard practice of using gloves by healthcare providers, there were reports of extragenital syphilitic lesions on the fingers and in the noses of physicians.43–45 A study by Epstein et al. found a report of 51 cases of extragenital chancres in physicians; thirty-five of these lesions occurred on the fingers, six inside the nose, one on an eyelid and one on an arm.43 Routine examinations of patients was the most common means of infection, and not wearing gloves was a recurrent reported reason as to how the transmission occurred. Examination of patients, deliveries, pelvic examinations, needle punctures and tonsillectomies seemed to be the most dangerous procedures.43 Because patients have spirochetemia, syphilis is a blood borne pathogen. Transmission occurs when there is contact with open wet lesions (chancre, mucous patches, condyloma lata, lues maligna).44 Dry and intact lesions of syphilis are non-contagious. Given that occupational infections of syphilis have occurred, it is important to use precautions when treating patients with syphilis in the inpatient setting. The CDC recommends that healthcare providers caring for patients with syphilis infection use standard precautions.35 This includes any or all the following: hand hygiene, the use of gloves, gown, mask, eye protection, or face shield, depending on the anticipated exposure; and safe injection practices. Also, equipment or items in the patient environment likely to have been contaminated with infectious body fluids must be handled in a manner to prevent transmission of infectious agents (e.g., wear gloves for direct contact, contain heavily soiled equipment, properly clean and disinfect or sterilize reusable equipment before use on another patient).35 The number of syphilis cases in the United States has risen over the past several years (Figure 1).
Figure 1. Increasing incidence of syphilis in the United States.
Source: Centers for Disease Control. https://www.cdc.gov/std/statistics/2021/overview.htm#Syphilis. Accessed May 26th, 2023. ACRONYMS: MSM = Gay, bisexual, and other men who have sex with men; MSU = Men with unknown sex of sex partners; MSW = Men who have sex with women only.
Conclusion
Infection prevention in the hospital is of paramount importance to patient safety as well as the safety of healthcare workers and visitors. Hospital-based providers must maintain a high level of vigilance for common, highly transmissible conditions and prevent transmission throughout the hospital. Scabies, tuberculosis, respiratory viruses, VZV, CDI, and syphilis are only some of the conditions at risk of nosocomial spread. The task of inpatient infection prevention is shared by providers, nurses, infection prevention teams, environmental services and others who must coordinate efforts in order to improve patient safety and outcomes.
Author Contributions
All authors have reviewed the final manuscript prior to submission. All the authors have contributed significantly to the manuscript, per the International Committee of Medical Journal Editors criteria of authorship.
Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; AND
Drafting the work or revising it critically for important intellectual content; AND
Final approval of the version to be published; AND
Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Disclosures/Conflicts of Interest
The authors declare no conflicts of interest.
Funding Statement
No funding was obtained for this manuscript.
References
- Overconfidence in infection control proficiency. Bushuven Stefan, Juenger Jana, Moeltner Andreas, Dettenkofer Markus. May;2019 American Journal of Infection Control. 47(5):545–550. doi: 10.1016/j.ajic.2018.10.022. https://doi.org/10.1016/j.ajic.2018.10.022 [DOI] [PubMed] [Google Scholar]
- Determining the need for additional training among hospital infection-control workforce – results from a multicentric survey within the multiresistance network of southern Lower Saxony (MRNS), Germany. Barre Felix, Kaba Hani, Dresselhaus Isabella, Mayr Eckart, Voigt Michelle, Schaumann Reiner, Dierks Marie-Luise, Scheithauer Simone. Apr 11;2022 GMS Hyg Infect Control. 17:Doc06. doi: 10.3205/DGKH000409. https://www.egms.de/en/journals/dgkh/2022-17/dgkh000409.shtml [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brady R R W, Rodrigues M A, Harrison R, Rae C, Graham C, Poxton I R, Gibb A P. Scottish Medical Journal. 3. Vol. 57. SAGE Publications; Knowledge of Clostridium difficile infection among UK health-care workers: development of a knowledge assessment tool; pp. 124–130.https://doi.org/10.1258/smj.2012.012015 [DOI] [PubMed] [Google Scholar]
- Knowledge, awareness, and attitude towards infection prevention and management among surgeons: identifying the surgeon champion. Sartelli Massimo, Kluger Yoram, Ansaloni Luca, Coccolini Federico, Baiocchi Gian Luca, Hardcastle Timothy C., Moore Ernest E., May Addison K., Itani Kamal M. F., Fry Donald E., Boermeester Marja A., Guirao Xavier, Napolitano Lena, Sawyer Robert G., Rasa Kemal, Abu-Zidan Fikri M., Adesunkanmi Abdulrashid K., Atanasov Boyko, Augustin Goran, Bala Miklosh, Cainzos Miguel A., Chichom-Mefire Alain, Cortese Francesco, Damaskos Dimitris, Delibegovic Samir, Demetrashvili Zaza, De Simone Belinda, Duane Therese M., Ghnnam Wagih, Gkiokas George, Gomes Carlos A., Hecker Andreas, Karamarkovic Aleksandar, Kenig Jakub, Khokha Vladimir, Kong Victor, Isik Arda, Leppäniemi Ari, Litvin Andrey, Lostoridis Eftychios, Machain Gustavo M., Marwah Sanjay, McFarlane Michael, Mesina Cristian, Negoi Ionut, Olaoye Iyiade, Pintar Tadeja, Pupelis Guntars, Rems Miran, Rubio-Perez Ines, Sakakushev Boris, Segovia-Lohse Helmut, Siribumrungwong Boonying, Talving Peep, Ulrych Jan, Vereczkei András G., Labricciosa Francesco M., Catena Fausto. Aug 17;2018 World Journal of Emergency Surgery. 13(1):37. doi: 10.1186/s13017-018-0198-x. https://doi.org/10.1186/s13017-018-0198-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunderkötter Cord, Wohlrab Johannes, Hamm Henning. Deutsches Ärzteblatt international. 41. Vol. 118. Deutscher Arzte-Verlag GmbH; Scabies: Epidemiology, Diagnosis, and Treatment; pp. 695–704.https://doi.org/10.3238/arztebl.m2021.0296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelman D., Yoshizumi J., Hay R.J., Osti M., Micali G., Norton S., Walton S., Boralevi F., Bernigaud C., Bowen A.C., Chang A.Y., Chosidow O., Estrada-Chavez G., Feldmeier H., Ishii N., Lacarrubba F., Mahé A., Maurer T., Mahdi M.M.A., Murdoch M.E., Pariser D., Nair P.A., Rehmus W., Romani L., Tilakaratne D., Tuicakau M., Walker S.L., Wanat K.A., Whitfeld M.J., Yotsu R.R., Steer A.C., Fuller L.C. British Journal of Dermatology. 5. Vol. 183. Oxford University Press (OUP); The 2020 International Alliance for the Control of Scabies Consensus Criteria for the Diagnosis of Scabies; pp. 808–820.https://doi.org/10.1111/bjd.18943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernigaud C, Fischer K, Chosidow O. Acta Dermato Venereologica. 9. Vol. 100. Medical Journals Sweden AB; The Management of Scabies in the 21st Century: Past, Advances and Potentials; pp. adv00112–234.https://doi.org/10.2340/00015555-3468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crusted Scabies. Palaniappan Vijayasankar, Gopinath Hima, Kaliaperumal Karthikeyan. Mar 3;2021 The American Journal of Tropical Medicine and Hygiene. 104(3):787–788. doi: 10.4269/ajtmh.20-1334. https://doi.org/10.4269/ajtmh.20-1334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scabies outbreaks in ten care homes for elderly people: a prospective study of clinical features, epidemiology, and treatment outcomes. Cassell Jackie A, Middleton Jo, Nalabanda Ananth, Lanza Stefania, Head Michael G, Bostock Jennifer, Hewitt Kirsty, Jones Christopher Iain, Darley Charles, Karir Simran, Walker Stephen L. Aug;2018 The Lancet Infectious Diseases. 18(8):894–902. doi: 10.1016/s1473-3099(18)30347-5. https://doi.org/10.1016/s1473-3099(18)30347-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clinical practices. Scabies. Chosidow Olivier. Apr 20;2006 New England Journal of Medicine. 354(16):1718–1727. doi: 10.1056/nejmcp052784. https://doi.org/10.1056/nejmcp052784 [DOI] [PubMed] [Google Scholar]
- Nosocomial scabies. Vorou R., Remoudaki H.D., Maltezou H.C. Jan;2007 Journal of Hospital Infection. 65(1):9–14. doi: 10.1016/j.jhin.2006.08.012. https://doi.org/10.1016/j.jhin.2006.08.012 [DOI] [PubMed] [Google Scholar]
- Mounsey K. E., Murray H. C., King M., Oprescu F. Epidemiology and Infection. 11. Vol. 144. Cambridge University Press (CUP); Retrospective analysis of institutional scabies outbreaks from 1984 to 2013: lessons learned and moving forward; pp. 2462–2471.https://doi.org/10.1017/s0950268816000443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention CDC - scabies - resources for health professionals - institutional settings - control. [2023-3-24]. https://www.cdc.gov/parasites/scabies/health_professionals/control.html
- Centers for Disease Control and Prevention CDC - scabies - resources for health professionals - institutional settings - single case. [2023-3-24]. https://www.cdc.gov/parasites/scabies/health_professionals/single.html Retrieved March 24, 2023, from.
- Centers for Disease Control and Prevention CDC - scabies - resources for health professionals - institutional settings - multiple cases. [2023-3-24]. https://www.cdc.gov/parasites/scabies/health_professionals/multiple.html
- Filardo Thomas D., Feng Pei-Jean, Pratt Robert H., Price Sandy F., Self Julie L. MMWR. Morbidity and Mortality Weekly Report. 12. Vol. 71. Centers for Disease Control MMWR Office; Tuberculosis — United States, 2021; pp. 441–446.https://doi.org/10.15585/mmwr.mm7112a1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanchar Avinash, Swaminathan Soumya. The Indian Journal of Pediatrics. 8. Vol. 86. Springer Science and Business Media LLC; Tuberculosis control: Who perspective and guidelines; pp. 703–706.https://doi.org/10.1007/s12098-019-02989-2 [DOI] [PubMed] [Google Scholar]
- Risk Factors for Delayed Isolation of Patients with Active Pulmonary Tuberculosis in an Acute-care Hospital. Han Jaijun, Nam Bo Da, Park Se Yoon, Park Jebyung, Lee Eunyoung, Lee Eun Jung, Hwang Jung Hwa, Kim Tae Hyong. Mar 19;2019 Scientific Reports. 9(1):4849. doi: 10.1038/s41598-019-41086-4. https://doi.org/10.1038/s41598-019-41086-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Predictive Factors and Clinical Impacts of Delayed Isolation of Tuberculosis during Hospital Admission. Lee Inhan, Kang Soyoung, Chin Bumsik, Joh Joon-Sung, Jeong Ina, Kim Junghyun, Kim Joohae, Lee Ji Yeon. Feb 8;2023 Journal of Clinical Medicine. 12(4):1361. doi: 10.3390/jcm12041361. https://doi.org/10.3390/jcm12041361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosocomial tuberculosis transmission from 2006 to 2018 in Beijing Chest Hospital, China. Xie Zhongyao, Zhou Ning, Chi Yuqing, Huang Guofang, Wang Jingping, Gao Hui, Xie Na, Ma Qianhui, Yang Nan, Duan Zhenlan, Nie Wenjuan, Sun Zhaogang, Chu Naihui. Oct 24;2020 Antimicrobial Resistance & Infection Control. 9(1):165. doi: 10.1186/s13756-020-00831-5. https://doi.org/10.1186/s13756-020-00831-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sosa Lynn E., Njie Gibril J., Lobato Mark N., Bamrah Morris Sapna, Buchta William, Casey Megan L., Goswami Neela D., Gruden MaryAnn, Hurst Bobbi Jo, Khan Amera R., Kuhar David T., Lewinsohn David M., Mathew Trini A., Mazurek Gerald H., Reves Randall, Paulos Lisa, Thanassi Wendy, Will Lorna, Belknap Robert. MMWR. Morbidity and Mortality Weekly Report. 19. Vol. 68. Centers for Disease Control MMWR Office; Tuberculosis screening, testing, and treatment of u.S. Health care personnel: Recommendations from the national tuberculosis controllers association and cdc, 2019; pp. 439–443.https://doi.org/10.15585/mmwr.mm6819a3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. Jensen P.A., Lambert L.A., Iademarco M.F., Ridzon R., CDC 2005MMWR Recomm Rep. 54(RR-17):1–141. [PubMed] [Google Scholar]
- The effectiveness of individual and environmental infection control measures in reducing the transmission of mycobacterium tuberculosis: A systematic review. Fox Greg J, Redwood Lisa, Chang Vicky, Ho Jennifer. 2021Clinical Infectious Diseases. 72(1):15–26. doi: 10.1093/cid/ciaa719. https://doi.org/10.1093/cid/ciaa719 [DOI] [PubMed] [Google Scholar]
- The common cold. Kirkpatrick George L. Dec;1996 Primary Care: Clinics in Office Practice. 23(4):657–675. doi: 10.1016/s0095-4543(05)70355-9. https://doi.org/10.1016/s0095-4543(05)70355-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- The common cold. Heikkinen Terho, Järvinen Asko. Jan;2003 The Lancet. 361(9351):51–59. doi: 10.1016/s0140-6736(03)12162-9. https://doi.org/10.1016/s0140-6736(03)12162-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Healthcare workers. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html
- Influenza in the acute hospital setting [published correction appears in Lancet Infect Dis 2002 Jun;2(6):383] Salgado Cassandra D, Farr Barry M, Hall Keri K, Hayden Frederick G. Mar;2002 The Lancet Infectious Diseases. 2(3):145–155. doi: 10.1016/s1473-3099(02)00221-9. https://doi.org/10.1016/s1473-3099(02)00221-9 [DOI] [PubMed] [Google Scholar]
- Hospital-acquired influenza in the United States, FluSurv-NET, 2011–2012 through 2018–2019. Cummings Charisse N., O’Halloran Alissa C., Azenkot Tali, Reingold Arthur, Alden Nisha B., Meek James I., Anderson Evan J., Ryan Patricia A., Kim Sue, McMahon Melissa, McMullen Chelsea, Spina Nancy L., Bennett Nancy M., Billing Laurie M., Thomas Ann, Schaffner William, Talbot H. Keipp, George Andrea, Reed Carrie, Garg Shikha. 2022Infection Control & Hospital Epidemiology. 43(10):1447–1453. doi: 10.1017/ice.2021.392. https://doi.org/10.1017/ice.2021.392 [DOI] [PubMed] [Google Scholar]
- Shorr Andrew F., Fisher Kristen, Micek Scott T., Kollef Marin H. Chest. 1. Vol. 154. Elsevier BV; The Burden of Viruses in Pneumonia Associated With Acute Respiratory Failure: An Underappreciated Issue; pp. 84–90.https://doi.org/10.1016/j.chest.2017.12.005 [DOI] [PubMed] [Google Scholar]
- Jain Seema, Self Wesley H., Wunderink Richard G., Fakhran Sherene, Balk Robert, Bramley Anna M., Reed Carrie, Grijalva Carlos G., Anderson Evan J., Courtney D. Mark, Chappell James D., Qi Chao, Hart Eric M., Carroll Frank, Trabue Christopher, Donnelly Helen K., Williams Derek J., Zhu Yuwei, Arnold Sandra R., Ampofo Krow, Waterer Grant W., Levine Min, Lindstrom Stephen, Winchell Jonas M., Katz Jacqueline M., Erdman Dean, Schneider Eileen, Hicks Lauri A., McCullers Jonathan A., Pavia Andrew T., Edwards Kathryn M., Finelli Lyn. New England Journal of Medicine. 5. Vol. 373. Massachusetts Medical Society; Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults; pp. 415–427.https://doi.org/10.1056/nejmoa1500245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Physical interventions to interrupt or reduce the spread of respiratory viruses. Jefferson Tom, Del Mar Chris, Dooley Liz, Ferroni Eliana, Al-Ansary Lubna A, Bawazeer Ghada A, van Driel Mieke L, Nair Sreekumaran, Foxlee Ruth, Rivetti Alessandro. Jan 20;2010 Cochrane Database of Systematic Reviews. (1):CD006207. doi: 10.1002/14651858.cd006207.pub3. https://doi.org/10.1002/14651858.cd006207.pub3 [DOI] [PubMed]
- Virology, transmission, and pathogenesis of SARS-CoV-2. Cevik Muge, Kuppalli Krutika, Kindrachuk Jason, Peiris Malik. Oct 23;2020 BMJ. 371:m3862. doi: 10.1136/bmj.m3862. https://doi.org/10.1136/bmj.m3862 [DOI] [PubMed] [Google Scholar]
- Weber David J., Rutala William A., Hamilton Holli. Infection Control & Hospital Epidemiology. 10. Vol. 17. Cambridge University Press (CUP); Prevention and Control of Varicella-Zoster Infections in Healthcare Facilities; pp. 694–705.https://doi.org/10.1017/s0195941700003076 [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention Shingles: Preventing VZV in healthcare settings. [2023-3-24]. https://www.cdc.gov/shingles/hcp/hc-settings.html
- Siegel Jane D., Rhinehart Emily, Jackson Marguerite, Chiarello Linda, and the Healthcare Infection Control Practices Advisory Committee 2007 Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings. https://www.cdc.gov/infectioncontrol/guidelines/isolation/index.html [DOI] [PMC free article] [PubMed]
- Immunization of health-care personnel: recommendations of the Advisory Committee on Immunization Practices (ACIP) Advisory Committee on Immunization Practices, Centers for Disease Control and Prevention (CDC) 2011MMWR Recomm Rep. 60:1. https://www.cdc.gov/mmwr/preview/mmwrhtml/rr6007a1.htm [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention CDI Prevention Strategies. [2023-3-24]. https://www.cdc.gov/cdiff/clinicians/cdi-prevention-strategies.html#print
- McDonald L Clifford, Gerding Dale N, Johnson Stuart, Bakken Johan S, Carroll Karen C, Coffin Susan E, Dubberke Erik R, Garey Kevin W, Gould Carolyn V, Kelly Ciaran, Loo Vivian, Shaklee Sammons Julia, Sandora Thomas J, Wilcox Mark H. Clinical Infectious Diseases. 7. Vol. 66. Oxford University Press (OUP); Clinical Practice Guidelines for Clostridium difficile Infection in Adults and Children: 2017 Update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA) pp. e1–e48.https://doi.org/10.1093/cid/cix1085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Multistate point-prevalence survey of health care-associated infections [published correction appears in N Engl J Med. 2022 Jun 16;386(24):2348] Magill Shelley S., Edwards Jonathan R., Bamberg Wendy, Beldavs Zintars G., Dumyati Ghinwa, Kainer Marion A., Lynfield Ruth, Maloney Meghan, McAllister-Hollod Laura, Nadle Joelle, Ray Susan M., Thompson Deborah L., Wilson Lucy E., Fridkin Scott K. Mar 27;2014 New England Journal of Medicine. 370(13):1198–1208. doi: 10.1056/nejmoa1306801. https://doi.org/10.1056/nejmoa1306801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clostridioides difficile skin contamination in patients with C. difficile-associated disease. Bobulsky G. S., Al-Nassir W. N., Riggs M. M., Sethi A. K., Donskey C. J. Feb 1;2008 Clinical Infectious Diseases. 46(3):447–450. doi: 10.1086/525267. https://doi.org/10.1086/525267 [DOI] [PubMed] [Google Scholar]
- Clostridioides difficile colitis: a retrospective study of incidence and severity before and after institution of an alcohol-based hand rub policy. Knight Nicole, Strait Taylor, Anthony Nicholas, Lovell Roger, Norton H. James, Sautter Robert, Scobey Martin. Sep;2010 American Journal of Infection Control. 38(7):523–528. doi: 10.1016/j.ajic.2009.12.008. https://doi.org/10.1016/j.ajic.2009.12.008 [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. Core Elements of Hospital Antibiotic Stewardship Programs. US Department of Health and Human Services, Centers for Disease Control and Prevention; Atlanta, GA: https://www.cdc.gov/antibiotic-use/healthcare/pdfs/hospital-core-elements-H.pdf [Google Scholar]
- Extragenital syphilis in physicians. Epstein E. Aug;1952 Calif Med. 77(2):149–50. [PMC free article] [PubMed] [Google Scholar]
- Nosocomial sexually transmitted diseases. Rein Michael F. Mar;1984 Infection Control. 5(3):117–122. doi: 10.1017/s0195941700059968. https://doi.org/10.1017/s0195941700059968 [DOI] [PubMed] [Google Scholar]
- Occupational infection in health care. The century-old lessons from syphilis. Meyer Gregg S. Nov 8;1993 Archives of Internal Medicine. 153(21):2439–47. doi: 10.1001/archinte.1993.00410210057007. https://doi.org/10.1001/archinte.1993.00410210057007 [DOI] [PubMed] [Google Scholar]