Abstract
Aims: Signalling through the Wnt pathway is integrally associated with colon carcinogenesis. Although activating mutations in the genes for adenomatous polyposis coli (APC) and β-catenin are clearly associated with colon cancer, less is understood about the role of the upstream secreted ligands (Wnts) and their receptors (frizzled, Fz) in this process. In other systems, increased Wnt signalling has been shown to alter the expression of components of this pathway. This study was designed to test the hypothesis that colon cancer is characterised by aberrant expression of specific Wnt genes and Fz receptors.
Methods: The expression of Wnt genes was assessed by in situ, antisense RNA hybridisation in paraffin wax embedded samples of normal and malignant human colon tissues with probes specific for the individual Wnt genes. The expression of Fz1 and Fz2 was determined by immunoperoxidase based antibody staining on human tissues.
Results: Changes in the expression of some ligands and receptors were seen in colon cancer. For example, Wnt2 mRNA was detected in colon cancer but was undetectable in normal colonic mucosa. Differential expression of Wnt5a in normal mucosa was also noted, with increased expression at the base of the crypts compared with the luminal villi and slightly increased expression in colon cancer. Wnt7a exhibited minimal expression in both normal and malignant colon tissues, whereas other Wnt ligands including Wnts 1, 4, 5b, 6, 7b, and 10b were expressed equally and strongly in both normal and malignant colon tissues. In defining cellular responses and phenotype, the type and distribution of Fz receptors may be as important as the pattern of Wnt ligand expression. No expression of Fz receptor 1 and 2 was seen in normal colonic mucosa and in well differentiated tumours. However, poorly differentiated tumours exhibited a high degree of Fz receptor expression, especially at the margin of cellular invasion.
Conclusions: These data indicate that the expression of members of the Wnt signal transduction pathway, distinct from APC and β-catenin, is integrally associated with the process of colon carcinogenesis. Wnt2, and possibly Wnt5a, may be involved in the progression from normal mucosa to cancer and the expression of Fz1/2 receptors may be involved in processes associated with tumour invasion. Altered expression of these Wnts and Fz receptors may prove useful as prognostic or diagnostic markers for patients with colon cancer.
Keywords: colon carcinogenesis, signal transduction, in situ hybridisation
Ectopic activation of the Wnt signalling pathway leads to increased cellular growth and division in experimental organisms and mutations in Wnt pathway genes are tightly linked to the genesis of certain cancers in humans.1 Signalling through the Wnt pathway begins with a Wnt ligand, one of a family of at least 16 members in mammals.2 Wnts are secreted growth factors that signal through cell surface Frizzled (Fz) transmembrane receptors to initiate the signalling cascade.3–5 Members of the Dishevelled family are activated upon ligand binding to Fz,6 causing inhibition of glycogen synthase kinase 3β (GSK-3β, Zw3, shaggy). Inhibition of GSK-3β activity prevents phosphorylation of β-catenin (Armadillo, Arm), thus blocking adenomatous polyposis coli (APC) and Axin mediated degradation of β-catenin. Stabilised β-catenin accumulates and binds to members of the lymphoid enhancer factor/T cell factor (LEF/TCF) family of high mobility group box transcription factors in the nucleus,7–10 leading to changes in the transcription of growth regulatory genes. Targets of Wnt/LEF/TCF regulated transcription include the protooncogene myc,11 cyclooxygenase 2,12 matrilysin/MMP-7,13,14 cyclin D1,15,16 and a member of the LEF/TCF family, TCF1.17 Activation of the Wnt pathway also affects the expression of members of another family of signalling molecules—bone morphogenetic protein 6 (BMP-6), a member of the transforming growth factor β (TGF-β) superfamily.18
“Ectopic activation of the Wnt signalling pathway leads to increased cellular growth and division in experimental organisms and mutations in Wnt pathway genes are tightly linked to the genesis of certain cancers in humans”
The role of Wnt signalling in cancer was suggested by the discovery that ectopic expression of mouse Wnt1 (int1) caused the formation of mammary tumours in mice.19 The recognition that the APC tumour suppressor gene functions as a component of the Wnt pathway further implicated Wnt signalling in cancer, particularly colon cancer.20 APC mutations (or mutations in β-catenin or axin) are found in up to 85% of sporadic forms of colon cancer.21,22
Studies in model organisms suggest that the expression of the Wnt genes is controlled by a complex network that uses both positive and negative feedback.23,24 If true in humans, loss of heterozygosity or mutational change of certain members of the Wnt signalling pathway—for example, APC or β-catenin—might be expected to alter the expression of upstream genes, such as those for pathway ligands or receptors. To test this hypothesis, we performed a systematic study of the expression of Wnt ligands and their Fz receptors in normal and malignant human colon tissues. Vider et al reported that low amounts of Wnt2 were expressed in normal colon tissue, but that the gene was overexpressed in tumour tissue samples, and Wnt5a was expressed in both non-tumorous and colonic tumour tissue, although the methodology used did not distinguish between changes in cancer cells themselves and cells derived from surrounding tissues.25 The differential expression of downstream components of the Wnt signalling pathway, specifically members of the LEF/TCF family, has also been suggested. For example, TCF4 is normally expressed in colonic mucosa and cancer,26 TCF1 is overexpressed in colon cancer cell lines,27 and we have recently shown that LEF1 is expressed in colon cancer tissues but not in non-malignant colonic mucosa.28 Given these preliminary data, we have extended our analysis to test the hypothesis that the expression of specific Wnt ligands and Fz receptors would be altered in colon cancer and that these changes may be involved in colon carcinogenesis.
METHODS AND MATERIALS
Tissue acquisition
Archived, paraffin wax embedded pathological specimens were obtained under an IRB approved protocol (UCI98–20) following oral and written informed consent. Patients were identified through the Chao Family Comprehensive Cancer Center as individuals with recent surgical resection of colonic adenocarcinoma. Samples of normal colonic tissue and of colon cancer tissue were obtained from different cuts of the same surgical specimen for each patient. When possible, histologically normal and malignant tissues were included on a single slide to enable direct comparison of staining intensity. Over 20 patients with colon cancer for whom tissue blocks were available were enrolled on our study.
Cell lines
Human cell lines were obtained from the American Type Culture Collection (Manassas, Virginia, USA). They included: Jurkat, an acute T cell leukemia cell line used as a control for in situ hybridisation, which was maintained in culture in RPMI 1640 medium with 10% fetal bovine serum (FBS); HT29, an adherent colorectal adenocarcinoma cell line that forms well differentiated adenocarcinomas in nude mice and is maintained in culture in DMEM medium with 10% FBS; Colo205, a free floating colorectal adenocarcinoma cell line maintained in RPMI 1640 medium with 10% FBS; CaCo2, an adherent colorectal adenocarcinoma cell line that forms moderately well differentiated adenocarcinomas in nude mice and is maintained in culture in DMEM media with 20% FBS; and PANC1, an adherent epithelioid pancreatic cancer cell line maintained in culture in DMEM media with 10% FBS. For in situ hybridisation and antibody staining controls, cells were pelleted by centrifugation, embedded in paraffin wax, sectioned, and prepared in a similar manner to the tissue samples (described below). To define the cellular localisation of RNA by in situ hybridisation, adherent cells (HT29 and CaCo2) were also grown directly on slides (Nunc Lab-Tek II chamber slide; Nunc, Rochester, New York, USA), fixed, and prepared for staining. Eliminating paraffin wax embedding greatly reduces artifacts caused by dewaxing.
In situ RNA hybridisation
Slides were dewaxed with sequential xylene/alcohol/distilled H2O washes and hybridised with single stranded antisense RNA probes generated from cDNAs using T7 and T3 RNA polymerase promoters in the respective vectors. Probes were labelled with digoxigenin, hydrolysed, and hybridised to tissue sections in a moisture controlled environment at 37°C for 72 hours at conditions optimised for each probe. Positive hybridisation was detected using antidigoxigenin conjugated to alkaline phosphatase to allow visualisation with an alkaline phosphatase substrate. All slides were counterstained lightly for five seconds with a haematoxylin and eosin reagent to reveal the tissue architecture. Probes for Wnts and BMPs generally included non-homologous coding sequences or 3‘ untranslated sequences. Samples from over 20 individuals with colon cancer were analysed for expression, although every sample was not used for each probe. Each probe was tested against a minimum of three samples of normal and malignant tissue from different patients. Most probes were hybridised against between 10 and 15 separate tissue sections. For example, the Wnt2 probe was hybridised against normal colonic mucosa specimens from seven different patients along with hybridisation of the corresponding colon cancer specimens from each of these patients. Control hybridisations (described below) accompanied each of these analyses of human derived tissues, including eight separate hybridisation experiments each against cell lines HT29, Colo205, and CaCo2.
Several controls were used for every experiment. First, each tissue was subjected to hybridisation with either trefoil factor or thymidine kinase (TK) probes to ensure that the tissue was preserved adequately to permit in situ RNA hybridisation. Trefoil factor and TK are differentially expressed in normal colonic mucosa (trefoil factor in goblet cells and TK in the crypts but not the villi). Trefoil factor is frequently overexpressed in colon carcinoma cells and TK is overexpressed in rapidly proliferating tissues. As a negative control, all tissues were also analysed with an identical probe in the sense orientation to ensure that there was no detectable staining and to confirm the absence of non-specific hybridisation. A panel of cell lines provided additional positive and negative controls. The expression of each gene was defined for each cell line by northern blot analysis. Cell lines were then centrifuged, embedded in paraffin wax, and sectioned to mimic the conditions used for the tissue samples. Each probe was tested against a panel of control cell lines to ensure reproducibility and precision when compared with northern blot analyses before use on the tissue sections. The probes were prepared freshly and each batch was subjected to a full battery of positive and negative control testing. This battery of positive and negative controls allowed for extensive quality control as follows: (1) tissue that did not exhibit distinct staining with TK or trefoil factor was discarded to eliminate tissues with extensive RNA degradation, presumably occurring at the time of surgery before fixation; (2) each batch of probe was tested initially against positive and negative control cell lines to ensure adequacy of probe preparation; (3) each analysis of patient tissue included slides stained with antidigoxigenin and alkaline phosphatase reagents to ensure that background staining was minimal; and (4) each analysis of patient tissue included slides for which a sense RNA probe was used to ensure that background staining was minimal.
Expression was visualised directly by means of an Olympus B50 microscope system with Nomarski optics and digital capture technology. Photographs of tissue and cell line sections were obtained within 48 hours of completion of the hybridisation to minimise slide artifacts that can appear over time. Probes used for in situ hybridisation included Wnt1, Wnt2, Wnt4, Wnt5a, Wnt5b, Wnt6, Wnt7a, Wnt7b, Wnt10b, BMP2a, and BMP6 obtained from Genome Systems. The genes and the accession and identification numbers from the Image Consortium are listed: Wnt2, AA970688, 1579015; Wnt4, AI139742, 1710594; Wnt5a, W49672, 324901; Wnt5b, A1201930, 1859198; Wnt6, AI127113, 1708122; Wnt7a, AI040985, 1641787; Wnt7b, AA991310, 1608881; Wnt10b, AI144467, 1708905; BMP2a, AA515983, 925036; BMP6, AA573738, 1012778. Wnt11 and BMP4 probes were contaminated with T1 phage and were not available from Genome Systems.
Antibody staining
Paraffin wax embedded samples of normal colon and colon cancers were obtained as described above. Normal colon from 15 different patients, in addition to more than five distinct samples both of well differentiated and distinct samples of poorly differentiated colon cancer were analysed. Polyclonal goat antihuman Fz1+2 antibodies, which react with both human Fz1 and Fz2 cell surface receptors, were obtained from Santa Cruz Biotechnology (Santa Cruz, California, USA). Samples were dewaxed with xylene/ethanol and incubated with primary antibody (2 μg/ml) for 30 minutes at 25°C. This was followed by incubation with biotinylated donkey antigoat secondary antibody (1 μg/ml), and subsequently with horseradish peroxidase and diaminobenzidine (DAB) chromogen. Donkey serum was used as a blocking agent to reduce background staining. Negative controls included slides hybridised with secondary antibody and HRP/DAB chromagen but without primary (anti-Fz) antibody. Additional controls performed with each experiment included staining of paraffin wax embedded cell lines known to express Fz.
RESULTS
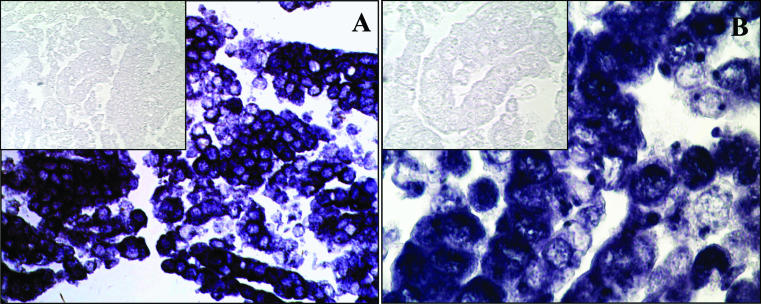
In situ hybridisation was performed on both normal and malignant paraffin wax embedded tissues using digoxigenin labelled antisense RNA probes, along with negative and positive controls, to ensure adequacy of the tissue and probes and absence of non-specific background hybridisation, as outlined in the materials and methods section. Paraffin wax embedded cell lines served as controls for each probe and clearly distinguished positive hybridisation from negative, sense probe, hybridisation (fig 1A ▶). Comparisons of normal tissue with malignant tissue on different regions of the same slide ensured identical treatment of tissues for comparison of expression.
Figure 1.
In situ, anti-sense RNA hybridisation of Wnt5b in HT29 colon cancer cell lines. In situ hybridisation of paraffin wax embedded cells. Dark colouration indicates positive hybridisation. Inserts represent control hybridisation with sense orientation Wnt5b probe in HT29 cells. (A) Original magnification, ×100; (B) original magnification, ×400.
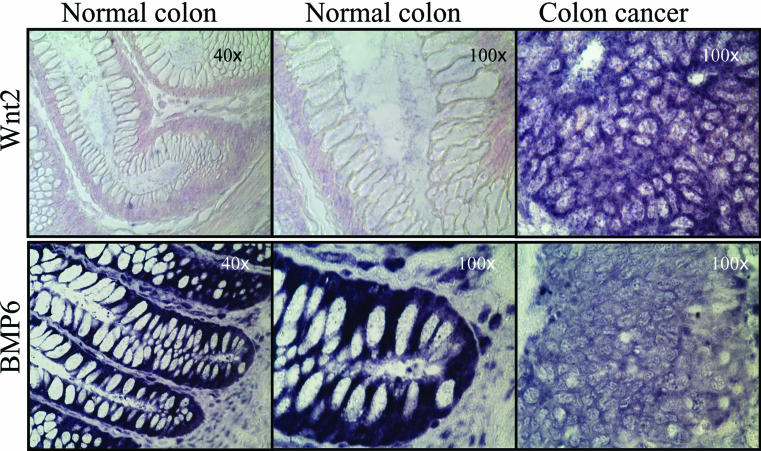
Examination of patient samples showed that most Wnt ligands were expressed in both the normal colonic mucosa and in colon cancer (table 1 ▶). Expression was graded as negative (no cells staining), weakly positive (faint staining), positive (evident staining), and strongly positive. In addition to this quantitative grading, tissue samples in which some cells were positive but others within the same specimen were negative were graded as heterogeneous. If one or more specific tissue location was positive (for example, the base of a crypt), but others were not, this information was also recorded. Abundant expression of Wnt1, Wnt4, Wnt5b, Wnt6, Wnt7b, and Wnt10b was seen in both normal and malignant tissue and in all three colon cancer cell lines tested. Wnt7a message was absent, or at least below the level of detection, in the colon tissues and it was only weakly expressed in HT29 and Colo205. Expression of Wnt7a was not seen in CaCo2 cells. Interestingly, Wnt2 was the only ligand that was specifically activated in colon cancer compared with normal colonic mucosa (table 1 ▶; fig 2 ▶). Specifically, Wnt2 expression was absent in the colonic crypts or villi but was expressed in colon cancer and in the colon cancer cell lines tested. Expression of Wnt5a was noted in the normal colon, with slightly higher signals at the base of the crypt. Wnt5a expression was slightly stronger in cancerous tissues but the proportional increase over normal tissues was less evident than for Wnt2.
Table 1.
Expression of Wnts, BMPs, and Fz in human tissues and colon cancer cell lines
| Normal colonic mucosa | Colon cancer | Ht29 | Colo205 | CaCo2 | Panc1 | |
| Wnt 1 | Positive | Positive | Positive | Positive | Positive | |
| Wnt 2 | Negative | Hetero positive | Weakly positive | Weakly positive | Positive | |
| Wnt 4 | Positive | Positive | Positive | Positive | Positive | |
| Wnt 5A | Positive, stronger at base of crypts | Strongly positive | Positive | Positive | Positive | |
| Wnt 5b | Positive | Hetero positive | Positive | Positive | Positive | |
| Wnt6 | Positive | Positive | Positive | Positive | Positive | |
| Wnt 7a | Negative, possible few positive cells at base | Negative | Weakly positive | Weakly positive | Negative | |
| Wnt 7b | Positive | Positive | Positive | Weakly positive | Hetero positive | |
| Wnt 10b | Positive | Positive | Positive | Positive | Positive | |
| BMP 2a | Negative | Negative | Weakly hetero positive | Weakly hetero positive | Weakly hetero positive | Positive |
| BMP6 | Positive | Negative to weakly positive | Positive | Positive | Positive | |
| Fz 1/2 | Negative | Positive, poorly differentiated | Positive | Positive | Positive |
Panc1 cells were used as a positive control for BMP2a only. Hetero indicates heterogeneous staining among cells visualised in the tissue samples. Wnt and BMP mRNA were detected by in situ hybridisation and Fz1/2 protein by immunoperoxidase. See materials and methods section for numbers of samples analysed.
BMP, bone morphogenetic protein; Fz, frizzled.
Figure 2.
Expression, as measured by in situ antisense RNA hybridisation, of Wnt 2 (top row) and bone morphogenetic protein 6 (BMP6; bottom row) in normal colon (left two columns, original magnifications, ×40 and ×100) and colon cancer (right column, original magnification, ×100) from the same individual. The dark purple colouration indicates positive hybridisation. The pink staining is the result of the FAST red dye used to reveal tissue architecture. Hybridisation for a specific probe on normal and cancer tissues was performed concomitantly, under identical conditions, and with freshly prepared probe.
The expression of two bone morphogenetic proteins, BMP2a and BMP6, was also evaluated. BMP6 was expressed in the normal colonic mucosa and in the HT29, Colo205, and CaCo2 cell lines, but expression was greatly reduced in the malignant colon cancer tissues (table 1 ▶; fig 2 ▶). In contrast, minimal expression of BMP2a was detected in normal colon, colon cancer, and the colon cancer cell lines. Panc1, a transformed pancreatic adenocarcinoma cell line, was used as a positive control because it is known to express BMP2a.29 Therefore, at least one member of the TGF-β family of ligands is significantly downregulated in human colon cancer.
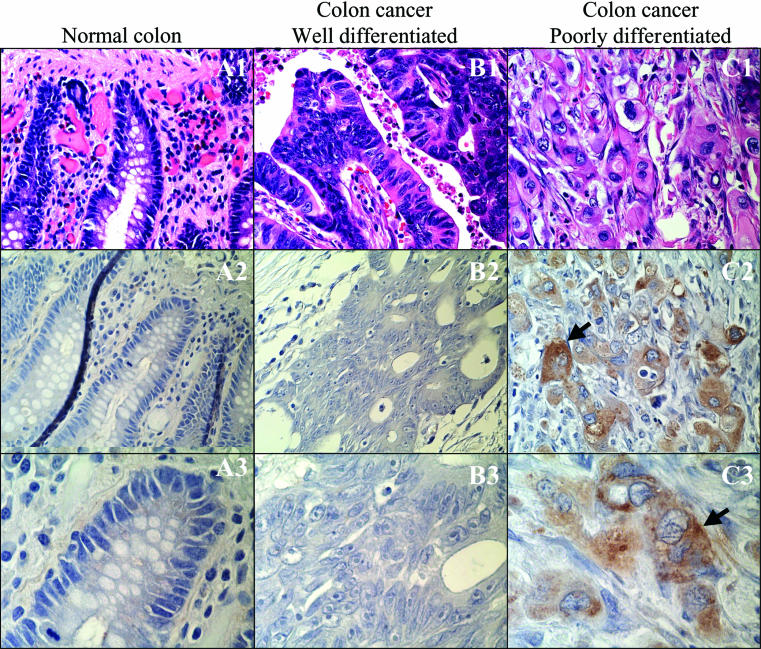
The expression of Fz1 and Fz2 protein was evaluated with a polyclonal antibody that reacts with these two members of the Fz receptor family. Although the expression patterns of different Wnt ligands may direct different cell behaviours, the expression patterns of the Fz receptors are equally important in influencing cell growth. No expression of Fz1/2 could be detected in normal colonic mucosa or in well differentiated colon cancers from six independent patient samples. Repeated attempts were made to detect potentially low amounts of Fz1/2, but no expression was detected. These results are in contrast to the expression of Fz1/2 in samples from three patients with poorly differentiated colon cancer (fig 3 ▶). Not only was Fz1/2 expression readily detectable, but it was greatest in areas where tumour cells were invading adjacent tissues (the “invasion front”). Each of the colon cancer cell lines tested, HT29, Colo205, and CaCo2, exhibited strong expression of Fz1/2 after immunoperoxidase based staining on paraffin wax embedded sections (data not shown).
Figure 3.
Expression, as assessed by antibody based immunoperoxidase methodology, of Fz1/2 receptors in (A1–3) normal colonic mucosa, (B1–3) well differentiated colon carcinoma, and (C1–3) poorly differentiated colon carcinoma. Haematoxylin and eosin staining is depicted in A1, B1, and C1. Experimental conditions are represented in panels A2, B2, and C2 (original magnification, ×40) and A3, B3, and C3 (original magnification, ×100). The brown granular staining in panels C2 and C3 indicates the presence of Fz1/2 receptors.
DISCUSSION
Alterations in the Wnt signalling pathway are involved in colon cancer carcinogenesis in patients with familial polyposis and in the most patients with sporadic colon cancer.30–32 The role of APC mutations in this process, and the resultant accumulation of cytoplasmic and nuclear protoncogenic β-catenin are well described.1,20 Inactivating mutations in APC and activating mutations in β-catenin mimic stimulation of the Wnt pathway, leading to increases in cellular β-catenin and changes in downstream target gene expression. Other possible targets facilitating the initiation of Wnt pathway activation are the Wnt genes and Fz receptor themselves. The data presented here suggest that the regulation of expression of several members of the Wnt ligand family and of specific Fz receptors is key to the process of colon carcinogenesis.
“Surprisingly, we found that the expression of most of the Wnt ligands is fairly ubiquitous in both normal and malignant colon, and that minimal expression differences can be ascertained between the colonic crypts and villi”
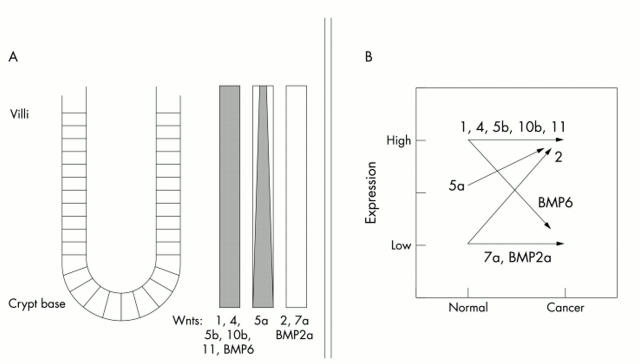
In drosophila and other model organisms, the Wnt signalling pathway regulates the expression of its ligand and receptor genes in an autoactivating loop.33–37 In the colon, where alterations in key Wnt signalling pathway genes are fundamental in malignant transformation, we had initially hypothesised that one or more specific Wnt would exhibit expression that was localised to the base of the adult human colonic crypt, where the population of proliferating stem cells resides, as also postulated by Bienz and Clevers.20 Surprisingly, we found that the expression of most of the Wnt ligands is fairly ubiquitous in both normal and malignant colon, and that minimal expression differences can be ascertained between the colonic crypts and villi. Only Wnt2, and to a lesser degree Wnt5a, appear to be upregulated in the transition between normal and malignant mucosa (fig 4 ▶). Only Wnt5a displays any degree of graded expression between the colonic crypts and villi. The colonic crypts contain proliferating mucosal stem cells give rise to differentiated, non-proliferating mucosal cells that migrate towards the villi.38 In the mouse, TCF4 is required to maintain the proliferative integrity of the undifferentiated crypt stem cell.39 We found no evidence for localised expression of the Wnts tested here. Our current data support an alternative model that any restriction of Wnt signalling in the colon is achieved by restricting other components of the Wnt signal transduction pathway, a notable departure from the strategies used by drosophila, xenopus, and other model systems.
Figure 4.
Diagram representing the expression of various Wnt ligands, BMP2a, and BMP6 in the normal colon and (B) the comparative expression of these molecules in normal and malignant colon.
In accordance with this alternative model of Wnt signal regulation, Fz expression appears to be highly regulated in the human colon. There was little or no apparent expression in normal colon or well differentiated cancers detected by the Fz1/2 specific antibody used in our study. This raises the possibility that Wnt ligands are signalling through a Fz receptor other than Fz1/2 (there are at least 10 human Fz receptors) or through another type of receptor that has yet to be defined. In striking contrast to the lack of detectable Fz1/2 in normal mucosa and well differentiated colon cancer, abundant Fz expression is seen in poorly differentiated cancers, particularly at the invasion front. Other studies have detected nuclear accumulation of β-catenin, most prominently at the leading edges of colon tumours,40 a localisation pattern that correlates with aggressive tumour invasion.41 Transcriptional activation of Fz gene expression may result from high numbers of nuclear β-catenin–LEF–TCF complexes in these cells. Studies in human and other model systems provide some evidence for similar autocrine feedback loops within the Wnt/Wingless pathway.17,42–45 Alternatively, the expression of cell surface Fz receptors at the invasion front may be the primary cause of the increased nuclear accumulation of β-catenin. Perhaps APC mutations alone are not sufficient to drive nuclear β-catenin accumulation to high levels; other processes ongoing at the invasion front may synergise with a crippled APC dependent degradation pathway to promote the formation of nuclear β-catenin–LEF–TCF complexes. Particularly for the process of tumour invasion, our data suggest that the regulation of Fz receptor expression may be more important than the regulation of Wnt ligand expression, whereas in drosophila the expression of Fz receptors and Wg ligands are equally crucial for normal growth and development.37,46
“Particularly for the process of tumour invasion, our data suggest that the regulation of Frizzled receptor expression may be more important than the regulation of Wnt ligand expression”
BMPs appear to function in opposition to Wnts in drosophila and other model systems.23,24,47–49 The loss of transduction of TGF-β signals mediated by genetic inactivation of the transcription factor Smad4 is thought to be a frequent and important genetic alteration that can promote carcinogenesis in the colon.50,51 Our data suggest that downregulation of BMP6 occurs in colon cancer compared with normal mucosal control samples from the same individual. If the concept of antagonistic activities of the Wnt and BMP pathways holds in the human colon, it is possible that upregulation of the Wnt pathway—for example, increased Wnt2 and Fz expression—might lead to increased repression of the expression of BMP6 or, alternatively, that loss of TGF-β/BMP signalling might lead to increased expression of Wnt pathway genes.
The data presented here indicate that the expression of Wnt and Fz is important in the progression from normal colonic mucosa to a malignant and invasive phenotype. Additional studies are needed to evaluate the role of other Fz receptors and of secreted Fz related proteins, which act to repress Wnt related signalling. Studies designed to elucidate the expression pattern of important Wnt species in early and late adenomatous polyps are under way. More detailed analysis of Wnt gene expression in colonic crypt stem cells may help to define the processes important for stem cell proliferation and differentiation. Such future research in the area of Wnt signalling, in addition to providing a better understanding of the processes involved in colon cancer carcinogenesis, may lead to the development of clinically useful diagnostic and prognostic markers.
Take home messages.
The expression of members of the Wnt signal transduction pathway may be integrally associated with the process of colon carcinogenesis
Wnt2, and possibly Wnt5a, may be involved in the progression from normal mucosa to cancer
The expression of the Fz1/2 receptors may be involved in processes associated with tumour invasion
It appears that the regulation of Wnt signalling in the human colon is achieved by restricting other components of the Wnt signal transduction pathway, unlike the mechanisms of regulation seen in drosophila, xenopus, and other model systems
In the future, the altered expression of these molecules may be useful in the diagnosis and prognosis of colon cancer
Acknowledgments
We are indebted to K Gaudenz for excellent technical assistance and to H Thiesen for critical review of the manuscript. This work was supported by the Program in Cancer Research, Chao Family Comprehensive Cancer Center (RFH, MLW, JLM); a grant from the University of California, Irvine College of Medicine (RFH); and by NIH grants HD36081 and HD36049 (JLM), CA/GM83982 (MLW), and CA82450 (RFH).
Abbreviations
APC, adenomatous polyposis coli
BMP, bone morphogenetic protein
DAB, diaminobenzidine
FBS, fetal bovine serum
Fz, Frizzled
GSK-3β, glycogen synthase kinase 3β
LEF, lymphoid enhancer factor
TCF, T cell factor
TGF-β, transforming growth factor β
TK, thymidine kinase
REFERENCES
- 1.Polakis P. The oncogenic activation of β-catenin. Current Opin Genet Dev 1999;9:15–21. [DOI] [PubMed] [Google Scholar]
- 2.Cadigan KM, Nusse R. Wnt signaling: a common theme in animal development. Genes Dev 1997;11:3286–305. [DOI] [PubMed] [Google Scholar]
- 3.Bhanot P, Brink M, Samos CH, et al. A new member of the Frizzled family from drosophila functions as a Wingless receptor. Nature 1996;282:225–30. [DOI] [PubMed] [Google Scholar]
- 4.Yang-Snyder J, Miller JR, Brown JD, et al. A frizzled homolog functions in a vertebrate Wnt signaling pathway. Current Biol 1996;6:1302–6. [DOI] [PubMed] [Google Scholar]
- 5.He X, Saint-Jeannet J-P, Wang Y, et al. A member of the Frizzled protein family mediating axis induction by Wnt5a. Science 1997;275:1652–4. [DOI] [PubMed] [Google Scholar]
- 6.Klingensmith J, Nusse R, Perrimon N. The drosophila segment polarity gene dishevelled encodes a novel protein required for response to the wingless signal. Genes Dev 1994;8:118–30. [DOI] [PubMed] [Google Scholar]
- 7.Behrens J, von Kries JP, Kuhl M, et al. Functional interaction of β-catenin with the transcription factor LEF1. Nature 1996;382:638–42. [DOI] [PubMed] [Google Scholar]
- 8.Van de Wetering M, Cavallo R, Dooijes D, et al. Armadillo coactivates transcription driven by the product of the drosophila segment polarity gene dTCF. Cell 1997;88:789–99. [DOI] [PubMed] [Google Scholar]
- 9.Riese J, Yu X, Munnerlyn A, et al. Lef1, a nuclear factor coordinating signaling inputs from wingless and decapentaplegic. Cell 1997;88:777–87. [DOI] [PubMed] [Google Scholar]
- 10.Brannon M, Gomperts M, Sumoy L, et al. A β-catenin-XTCF3 complex binds to the siamois promoter to regulate dorsal axis specification in xenopus. Genes Dev 1997;11:2359–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.He TC, Sparks AB, Rago C, et al. Identification of c-MYC as a target of the APC pathway. Science 1998;281:1509–12. [DOI] [PubMed] [Google Scholar]
- 12.Howe LR, Subbaramaiah K, Chung WJ, et al. Transcriptional activation of cyclooxygenase-2 in Wnt-1-transformed mouse mammary epithelial cells. Cancer Res 1999;59:1572–7. [PubMed] [Google Scholar]
- 13.Crawford HC, Fingleton BM, Rudolph-Owen LA, et al. The metalloproteinase matrilysin is a target of beta-catenin transactivation in intestinal tumors. Oncogene 1999;18:2883–91. [DOI] [PubMed] [Google Scholar]
- 14.Brabletz T, Jung A, Dag S, et al. Beta-catenin regulates the expression of the matrix metalloproteinase-7 in human colorectal cancer. Am J Pathol 1999;155:1033–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Testu O, McCormick F. Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature 1999;398:422–6. [DOI] [PubMed] [Google Scholar]
- 16.Stutman M, Zhurinsky J, Simcha I, et al. The cyclin D1 gene is a target of the beta-catenin/LEF1 pathway. Proc Natl Acad Sci U S A 1999;96:5522–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Roose J, Huls G, van Beest M, et al. Synergy between tumor suppressor APC and the β-catenin-Tcf4 target Tcf1. Science 1999;285:1923–6. [DOI] [PubMed] [Google Scholar]
- 18.Raida M, Sarbia M, Clement JH, et al. Expression, regulation and clinical significance of bone morphogenetic protein 6 in esophageal squamous cell carcinoma. Int J Cancer 1999;83:38–44. [DOI] [PubMed] [Google Scholar]
- 19.Nusse R, Varmus HE. Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell 1982;31:99–109. [DOI] [PubMed] [Google Scholar]
- 20.Bienz M, Clevers H. Linking colorectal cancer to Wnt signaling. Cell 2000;103:311–20. [DOI] [PubMed] [Google Scholar]
- 21.Miyoshi Y, Nagase H, Ando H, et al. Somatic mutations of the APC gene in colorectal tumors: mutation cluster region in the APC gene. Hum Mol Genet 1992;1:229–33. [DOI] [PubMed] [Google Scholar]
- 22.Potter JD. Colorectal cancer: molecules and populations. J Natl Cancer Inst 1999;91:916–32. [DOI] [PubMed] [Google Scholar]
- 23.Brook WJ, Cohen SM. Antagonistic interactions between wingless and decapentaplegic responsible for dorsal–ventral pattern in the drosophila leg. Science 1996;273:1373–7. [DOI] [PubMed] [Google Scholar]
- 24.Theisen H, Haerry TE, O’Conner MD, et al. Developmental territories created by mutual antoagonism between Wingless and Decapentaplegic. Development 1996;122:3939–48. [DOI] [PubMed] [Google Scholar]
- 25.Vider B-Z, Zimber A, Chastre E, et al. Evidence for the involvement of the Wnt2 gene in human colorectal cancer. Oncogene 1996;12:153–8. [PubMed] [Google Scholar]
- 26.Korinek V, Barker N, Morin P, et al. Constitutive transcriptional activation by a β-catenin–TCF complex in APC−/− colon carcinoma. Science 1997;275:1784–7. [DOI] [PubMed] [Google Scholar]
- 27.Meyer K, Hieronymus T, Castrop J, et al. Ectopic activation of lymphoid high mobility group-box transcription factor TCF-1 and overexpression in colorectal cancer cells. Int J Cancer 1997;72:625–30. [DOI] [PubMed] [Google Scholar]
- 28.Hovanes K, Munguia JE, Truong T, et al. Beta-catenin-sensitive isoforms of lymphoid enhancer factor-1 are selectively expressed in colon cancer. Nat Genet 2001;58:53–7. [DOI] [PubMed] [Google Scholar]
- 29.Kleff J, Maruyama H, Ishiwata T, et al. Bone morphogenetic protein 2 exerts diverse effects on cell growth in vitro and is expressed in human pancreatic cancer in vivo. Gastroenterology 1999;116:1202–16. [DOI] [PubMed] [Google Scholar]
- 30.Groden J, Thliveris A, Samowitz W, et al. Identification and characterization of the familial adenomatous polyposis coli gene. Cell 1991;66:589–600. [DOI] [PubMed] [Google Scholar]
- 31.Kinzler KW, Nilbert MC, Su LK, et al. Identification of FAP locus genes from chromosome 5q21. Science 1991;253:661–5. [DOI] [PubMed] [Google Scholar]
- 32.Nagase H, Nakamura Y. Mutations of the APC (adenomatous polyposis coli) gene. Hum Mutat 1993;2:425–34. [DOI] [PubMed] [Google Scholar]
- 33.Marsh JL, Theisen H. Regeneration in insects. Semin Cell Dev Biol 1999;10:365–75. [DOI] [PubMed] [Google Scholar]
- 34.Dierick H, Bejsovec A. Cellular mechanisms of wingless/Wnt signal transduction. Current Top Dev Biol 1999;43:153–90. [DOI] [PubMed] [Google Scholar]
- 35.Baker NE. Localization of transcripts from the wingless gene in whole drosophila embryos. Development 1998;103:289–98. [DOI] [PubMed] [Google Scholar]
- 36.Baker NE. Transcription of the segment polarity gene wingless in the imaginal discs of drosophila, and the phenotype of a pupal-lethal wg mutation. Development 1998;102:489–97. [DOI] [PubMed] [Google Scholar]
- 37.Noordermeer J, Johnston P, Tijsewijk F, et al. The consequences of ubiquitous expression of the wingless gene in the drosophila embryo. Development 1992;116:711–19. [DOI] [PubMed] [Google Scholar]
- 38.Wong WM, Wright NA. Cell proliferation in gastrointestinal mucosa. J Clin Pathol 1999;52:321–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Korinek V, Barker N, Moerer P, et al. Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat Genet 1998;19:379–83. [DOI] [PubMed] [Google Scholar]
- 40.Brabletz T, Jung A, Hermann K, et al. Nuclear overexpression of the oncoprotein β-catenin in colorectal cancer is localized predominantly at the invasion front. Pathol Res Pract 1998;194:701–4. [DOI] [PubMed] [Google Scholar]
- 41.Miyazawa K, Iwaya K, Kuroda M, et al. Nuclear accumulation of beta-catenin in intestinal type gastric carcinoma: correlation with early tumor invasion. Vichows Arch 2000;437:508–13. [DOI] [PubMed] [Google Scholar]
- 42.Lescher B, Haenig B, Kospert A. sFRP-2 is a target of the Wnt-4 signaling pathway in developing metanephric kidney. Dev Dyn 1998;213:440–51. [DOI] [PubMed] [Google Scholar]
- 43.Cadigan KM, Fish MP, Rulifson EJ, et al. Wingless repression of drosophila frizzled 2 expression shapes wingless morphogen gradient in the wing. Cell 1998;93:767–77. [DOI] [PubMed] [Google Scholar]
- 44.Hooper JE. Distinct pathways for autocrine and paracrine Wingless signaling in drosophila embryos. Nature 1994;372:461–4. [DOI] [PubMed] [Google Scholar]
- 45.Yoffe KB, Manoukian AS, Wilder EL, et al. Evidence for engrailed-independent wingless autoregulation in drosophila. Dev Biol 1995;170:636–50. [DOI] [PubMed] [Google Scholar]
- 46.Muller H, Samanta R, Wieschaus E. Wingless signaling in the drosophila embryo: zygotic requirements and the role of the frizzled genes. Development 1999;126:577–86. [DOI] [PubMed] [Google Scholar]
- 47.Baker JC, Beddington RS, Harland RM. Wnt signaling in xenopus embryos inhibits BMP4 expression and activates neural development. Genes Dev 1999;13:3149–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Heslip TR, Theisen H, Walker H, et al. Shaggy and dishevelled exert opposite effects on Wingless and Decapentaplegic expression and on positional identity in imaginal discs. Development 1997;124:1069–78. [DOI] [PubMed] [Google Scholar]
- 49.Hirsinger E, Duprez D, Jouve C, et al. Noggin acts downstream of Wnt and Sonic Hedgehog to antagonize BMP4 in avian somite patterning. Development 1997;124:4605–14. [DOI] [PubMed] [Google Scholar]
- 50.Moskaluk CA, Kern SE. Cancer gets mad: DPC4 and other TCFβ pathway genes in human cancer. Biochem Biophys Acta 1996;1288:M31–3. [DOI] [PubMed] [Google Scholar]
- 51.Riggens GJ, Thiagalingam S, Rozenblum E, et al. Mad-related genes in the human. Nat Genet 1996;13:347–9. [DOI] [PubMed] [Google Scholar]