Abstract
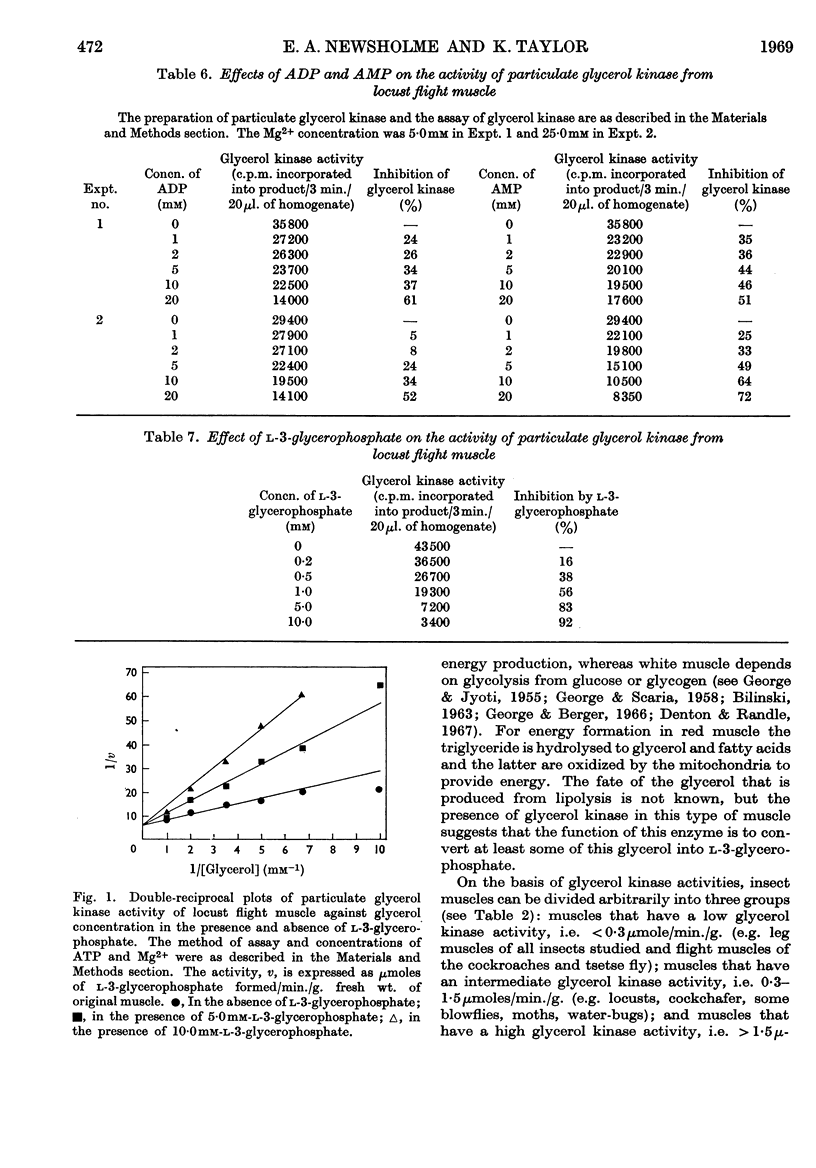
1. Glycerol kinase (EC 2.7.1.30) activity was measured in crude extracts of skeletal muscles by a radiochemical method. The properties of the enzyme from a number of different muscles are very similar to those of the enzyme from rat liver. Glycerol kinase from locust flight muscle was inhibited competitively by l-3-glycerophosphate with a Ki of 4·0×10−4m. 2. The activity of glycerol kinase was measured in a variety of muscles from vertebrates and invertebrates in an attempt to explain the large variation in the activity of this enzyme in different muscles. 3. In vertebrates glycerol kinase activities were generally higher in red muscle than in white muscle; the highest activities (approx. 0·2μmole/min./g. fresh wt.) were found in the red breast muscle of some birds (e.g. pigeon, duck, blue tit) whereas the activities in the white breast muscle of the pheasant and domestic fowl were very low (approx. 0·02μmole/min./g.). 4. On the basis of glycerol kinase activities, muscles from insects can be classified into three groups: muscles that have a low enzyme activity, i.e. <0·3μmole/min./g. (leg muscles of all insects studied and the flight muscles of cockroaches and the tsetse fly); muscles that have an intermediate enzyme activity, i.e. 0·3–1·5μmoles/min./g. (e.g. locusts, cockchafers, moths, water-bugs); and muscles that have a high enzyme activity, i.e. >1·5μmoles/min./g. (e.g. bees, wasps, some blowflies). 5. The function of glycerol kinase in vertebrate and insect muscles that possess a low or intermediate activity is considered to be the removal of glycerol that is produced from lipolysis of triglyceride or diglyceride by the muscle. Therefore in these muscles the activity of glycerol kinase is related to the metabolism of fat, which is used to support sustained muscular activity. A possible regulatory role of glycerol kinase in the initiation of triglyceride or diglyceride lipolysis is discussed. 6. The function of glycerol kinase in the insect muscles that possess a high activity of the enzyme is considered to be related to the high rates of glycolysis that these muscles can perform. The oxidation of extramitochondrial NADH, and therefore the maintenance of glycolysis, is dependent on the functioning of the glycerophosphate cycle; if at any stage of flight (e.g. at the start) the rate of mitochondrial oxidation of l-3-glycerophosphate was less than the activity of the extramitochondrial glycerophosphate dehydrogenase, this compound would accumulate, inhibit the latter enzyme and inhibit glycolysis. It is suggested that such excessive accumulation of l-3-glycerophosphate is prevented by hydrolysis of this compound to glycerol; the latter would have to be removed from the muscle when the accumulation of l-3-glycerophosphate had stopped, and this would explain the presence of glycerol kinase in these muscles and its inhibition by l-3-glycerophosphate.
Full text
PDF


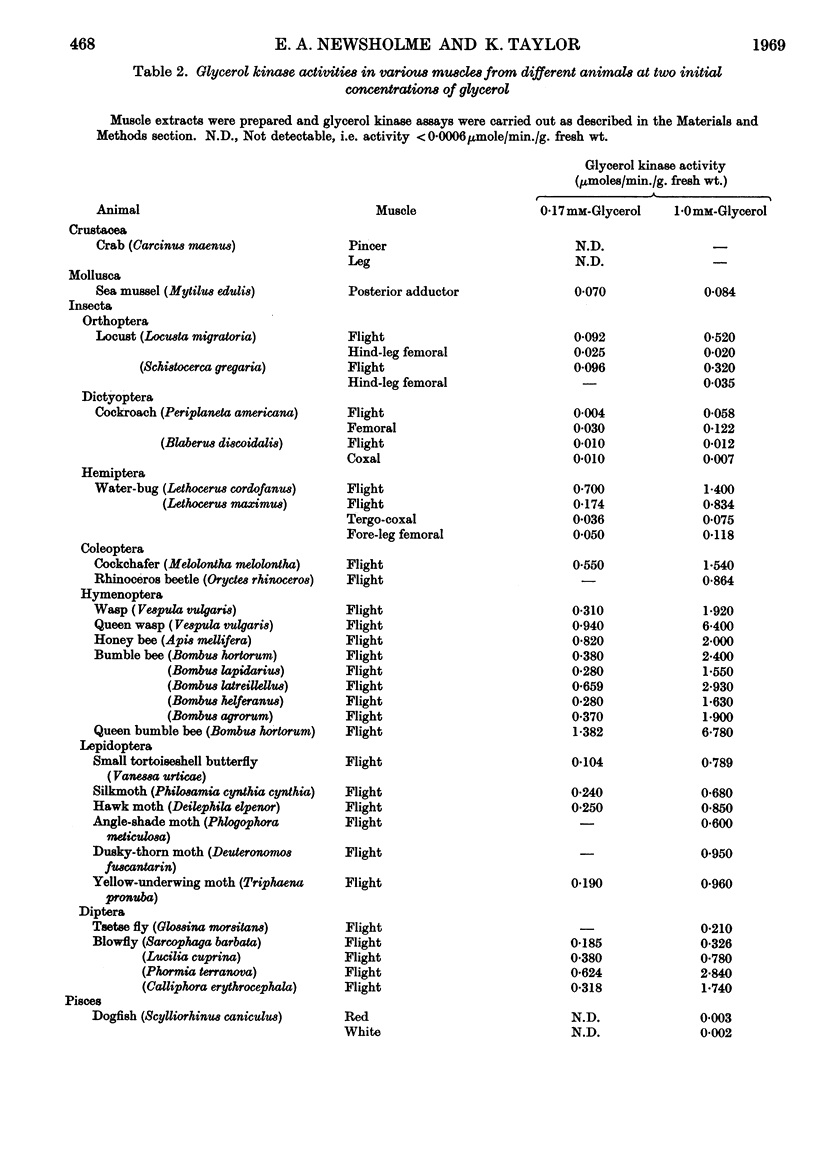





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BILINSKI E. Utilizatimn lipids by fish. I. Fatty acid oxidation by tissue slices from dark and white muscle of rainbow trout (Salmo gairdnerii). Can J Biochem Physiol. 1963 Jan;41:107–112. [PubMed] [Google Scholar]
- BLANCHAER M. C. THE COMPETITIVE INHIBITION OF THE CYTOPLASMIC L-ALPHA-GLYCEROPHOSPHATE DEHYDROGENASE OF SKELETAL MUSCLE BY L-ALPHA-GLYCEROPHOSPHATE. Can J Biochem. 1965 Jan;43:17–24. doi: 10.1139/o65-002. [DOI] [PubMed] [Google Scholar]
- Childress C. C., Sacktor B., Traynor D. R. Function of carnitine in the fatty acid oxidase-deficient insect flight muscle. J Biol Chem. 1967 Feb 25;242(4):754–760. [PubMed] [Google Scholar]
- Denton R. M., Randle P. J. Concentrations of glycerides and phospholipids in rat heart and gastrocnemius muscles. Effects of alloxan-diabetes and perfusion. Biochem J. 1967 Aug;104(2):416–422. doi: 10.1042/bj1040416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newsholme E. A., Robinson J., Taylor K. A radiochemical enzymatic activity assay for glycerol kinase and hexokinase. Biochim Biophys Acta. 1967 Mar 15;132(2):338–346. doi: 10.1016/0005-2744(67)90153-2. [DOI] [PubMed] [Google Scholar]
- Newsholme E. A., Rolleston F. S., Taylor K. Factors affecting the glucose 6-phosphate inhibition of hexokinase from cerebral cortex tissue of the guinea pig. Biochem J. 1968 Jan;106(1):193–201. doi: 10.1042/bj1060193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson J., Newsholme E. A. Some properties of hepatic glycerol kinase and their relation to the control of glycerol utilization. Biochem J. 1969 May;112(4):455–464. doi: 10.1042/bj1120455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SACKTOR B. Cell structure and the metabolism of insect flight muscle. J Biophys Biochem Cytol. 1955 Jan;1(1):29–46. doi: 10.1083/jcb.1.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WIELAND O., SUYTER M. Glycerokinase; Isolierung und Eigenschaften des Enzyms. Biochem Z. 1957;329(4):320–331. [PubMed] [Google Scholar]


