Abstract
Dipeptidyl peptidase IV (DPP-IV) is a serine protease whose inhibition has been an object of considerable interest in the context of developing novel treatments for type 2 diabetes mellitus. The development of novel DPP-IV inhibitors from natural or synthetic origin has seen a growing scientific interest in recent years, especially during the SARS-CoV-2 pandemic, when DPP-IV inhibitors were found to be of beneficial therapeutic value for COVID-19 patients. The present manuscript aims to summarize the most recent information on the synthesis of different DPP-IV inhibitors, emphasizing the various heterocyclic scaffolds that can be found in them. Special attention is devoted to DPP-IV inhibitors that are currently in clinical trials. Different synthetic approaches for the construction of DPP-IV inhibitors are discussed, as well as the most recent developments in the field.
Keywords: DPP-IV inhibitors, type 2 diabetes, heterocyclic synthesis, drug design, medicinal chemistry
1. Introduction
Dipeptidyl peptidase IV (DPP-IV) can be regarded as a promising therapeutic target in the treatment of type 2 diabetes mellitus [1,2,3,4,5,6]. DPP-IV is involved in the metabolism of incretin hormones (GLP-1 and GIP) in the body, which regulate insulin secretion [7]. Currently, there are twelve drugs that act as DPP-IV inhibitors and have been approved by regulatory agencies. DPP-IV inhibitors attracted even greater scientific interest during the SARS-CoV-2 pandemic, when their ability to mitigate the severity of COVID-19 disease was discovered [8,9,10,11,12]. In recent years, the importance of DPP-IV inhibition has grown beyond the treatment of type 2 diabetes. Recent evidence may indicate that DPP-IV inhibition may be of therapeutic significance in the field of cancer treatment [13,14]. Further research is focused on the potentially favorable cardiovascular effects of DPP-IV inhibition, as well as its effects on immune modulation [15,16,17,18]. A possible connection between DPP-IV inhibition and tackling neurological disorders has also been the subject of research attention [19,20]. However, in 2016, the FDA issued warnings about the increased risk of heart failure when using saxagliptin and alogliptin based on postmarketing studies and clinical trials [21,22,23,24]. The potential uses of DPP-IV inhibitors in treating conditions other than diabetes have been reviewed in the literature [25,26,27,28]. At present, there are twelve DPP-IV inhibitors that have been approved for use in clinical practice by the respective regulatory agencies, and there are many more in various stages of clinical trials [29]. In light of their various therapeutic uses, the development of novel and selective DPP-IV inhibitors remains an ongoing research challenge.
The prevailing classification of DPP-IV inhibitors divides them into two major classes—peptidomimetic and non-peptidomimetic. The peptidomimetic class of inhibitors has two groups—glycine- and β-alanine-based inhibitors. Both classes are designed based on natural DPP-IV substrates and bear a resemblance to them. The non-peptidomimetic series is structurally more diverse compared to the peptidomimetic inhibitor group [29,30]. Among the approved DPP-IV inhibitors, seven belong to the peptidomimetic group, and the remaining five are classified as non-peptidomimetic [29].
The present review aims to provide an overview of the synthetic methodologies that give rise to different heterocyclic DPP-IV inhibitors.
2. Five-Membered Heterocycles
2.1. Five-Membered Heterocycles with One Heteroatom and Their Benzo-Fused Derivatives
2.1.1. Cyanopyrrolidine
Cyanopyrrolidines are among the most prominent and well-researched groups of DPP-IV inhibitors of the peptidomimetic group [31]. These compounds resemble the proline-containing DPP-IV substrate, and several approved drugs contain this fragment as well—Vildagliptin [32], saxagliptin [33], and Denagliptin [34] (Figure 1). The structure–activity relationship and the history of the development of the first inhibitors of this class have been reviewed in the literature [31,35].
Figure 1.
Cyanopyrrolidine-containing DPP-IV inhibitors.
The methods of preparation usually use proline (1) as the starting material, which, upon conversion to amide and the subsequent dehydration, yields the target 2-cyanopyrrolidine (2). This approach was first described by Ashworth et al. [36] (Scheme 1).
Scheme 1.
Reagents and conditions: (i) 2-Nitrobenzenesulfenyl chloride, 2 N NaOH; (ii) N-hydroxysuccinimide, carbodiimide; (iii) conc. NH4OH, dioxane, 94% (over three steps); (iv) imidazole, POCl3, pyridine, 95%; (v) 4 N HCl/dioxane, diethyl ether, 90%.
Trifluoroacetic anhydride has also been widely utilized as a dehydrating agent for the conversion of amide to a nitrile group since the work of Villhauer et al. was published [37]. Further modification of the obtained 2-cyanopyrrolidine, especially its incorporation in peptides, can be achieved by the use of peptide coupling reagents [38]. The solid-phase synthesis of cyanopyrrolidine-containing peptides has also been reported [39]. This process starts with readily available Fmoc-proline being loaded on Rink-amide MBHA resin. The key dehydration reaction for obtaining the 2-cyanopyrrolidine fragment was again performed with trifluoroacetic anhydride [39]. Additional modifications of the pyrrolidine ring, such as the placement of an additional fluorine atom as a way to increase the potency of prospective inhibitors, have also been explored [40]. Fluorinated derivatives were obtained from the methyl ester of Boc-4-hydroxyproline 3 using diethylaminosulfur trifluoride (DAST) as a mild alcohol fluorinating agent [41,42]. The subsequent alkaline ester hydrolysis and coupling reaction with ammonia that were achieved with 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide hydrochloride (EDC) and 1-hydroxybenztriazole (HOBt) produced the fluorinated amide derivative 4 [43] (Scheme 2).
Scheme 2.
Reagents and conditions: (i) DAST, dry CH2Cl2, -78 °C to rt; (ii) 2 equiv. LiOH, AcCN-H2O (3-1), rt; (iii) EDC, HOBt, NH3, DMF-AcCN; (iv) 4 M HCl, ethyl acetate; (v) cyanuric chloride, DMF, quantitative yield; (vi) 2 M HCl, H2O-MeOH, quantitative yield.
The coupling of the obtained pyrrolidine derivatives with amino acids, for example, aspartic and glutamic acid, has given rise to highly potent DPP-IV inhibitors [44,45].
2.1.2. Indole and Isoindole
Inhibitors of DPP-IV do not typically contain indole derivatives. The very first indole-containing DPP-IV inhibitors were documented by Sakashita et al. [46]. The application of NaBH(OAc)3 in a reductive amination process yielded compound 5, which has s 5-methoxyindoline moiety (Scheme 3).
Scheme 3.
Reagents and conditions: (i) NaBH(OAc)3, AcOH, 93%; (ii) H+, 78%.
To develop novel compounds exhibiting enhanced selectivity for DPP-IV in comparison to DPP-9, a series of aryl-substituted indoles 6 and their sulfonamide derivatives were prepared [47]. The incorporation of the aryl group at C-2 was achieved through Suzuki coupling of 7 with aryl boronic acid, followed by the incorporation of an additional amino group introduced using potassium phthalimide in a Hoffman elimination reaction (Scheme 4).
Scheme 4.
Reagents and conditions: (i) MeI, THF, potassium phthalimide, DMF, 150 °C, 5 h, 64%; (ii) THF, CHCl3, pyridinium tribromide, −10 °C, 3 h, 56%; (iii) 2,4-dichlorophenylboronic acid, Pd(PPh3)4, LiCl, Na2CO3, toluene, EtOH, 105 °C, 4 h, 30%; (iv) R1SO2Cl or R1COCl, NaH, DMF, 0 °C, 16 h 31–50%; (v) H2N-NH2, EtOH, rt for 1 h, 84–91%.
According to reports, isoindole derivatives can inhibit both DPP-IV and DPP-8/9 but with greater selectivity for DPP-8/9 [48]. A six-step synthetic route, starting with substituted phthalic anhydride 9 was used to prepare the lead compound 8 (Scheme 5). Key stages from the synthetic procedure include amidation and thermal ring closure to phthalimide derivative 10 and final reduction with borane to isoindole 11. Peptide coupling chemistry was successfully used to incorporate the peptide side chain into the isoindole ring. Further research resulted in the introduction of a more complex peptide side chain, allowing higher selectivity for DPP-8 over DPP-9 [49].
Scheme 5.
Reagents and conditions: (i) NH4OH, THF, thermal dehydration, 95–99%; (ii) borane, TFH; (iii) Et3N, Boc2O, CH2Cl2, 21–33% over two steps; (iv) TFA, CH2Cl2, 95–99%; (v) allo-Ile-Boc, TBTU, Et3N, CH2Cl2, 38–80%; (vi) TFA, CH2Cl2, 95–99%.
2.2. Five-Membered Heterocycles with Two Heteroatom and Their Benzo-Fused Derivatives
2.2.1. Pyrazole
A prominent example of pyrazole in the context of DPP-IV inhibition is the drug Teneligliptin (Figure 2), developed by Yoshida et al. and approved for clinical use in Japan [50].
Figure 2.

Structure of Teneligliptin.
The inclusion of the pyrazole ring system in potential inhibitors significantly affects the anticipated interactions of the inhibitor and the DPP-IV active site, facilitating π-cation interactions with Arg358 and Tyr666, thereby engaging both S1 and S2 pockets of DPP-IV [29,51]. This research conducted by Server et al. on thiosemicarbazone DPP-IV inhibitors featuring a pyrazole ring 12 elucidates this conclusion appropriately (Scheme 6) [52]. The proposed inhibitors were obtained through a two-step synthetic protocol beginning with 4-(1H-pyrazol-1-yl)phenyl isothiocyanate (13), which, when treated with hydrazine hydrate, yielded the corresponding thiosemicarbazide product 14. Condensation with substituted aromatic aldehydes produced the desired thiosemicarbazone functionality [53].
Scheme 6.
Reagents and conditions: (i) NH2NH2·H2O, ethanol, rt, 4 h; (ii) RC6H4CHO, ethanol, reflux, 8 h, 75–90%.
A further instance of functionalizing 1-phenyl-1H-pyrazole derivatives in the development of DPP-IV inhibitors is the application of a one-pot Mannich reaction by Nidhar et al. [54]. The synthesized inhibitors 15 incorporated a β-amino acid side chain, which is crucial for enhancing affinity toward the DPP-IV enzyme (Scheme 7). The authors described 1-phenyl-1H-pyrazole as participating in important hydrophobic interactions with the amino acids Trp629, Ser630, Tyr631, and Tyr547. The synthesized inhibitors exhibited IC50 values in the nanomolar range, indicating their potential for further investigation. The authors also investigated the addition of a chalcone side chain [55].
Scheme 7.
Reagents and conditions: (i) Bi(NO3)2, ethanol, 60–70 °C, 3–5 h, 75–96%.
The authors also investigated the insertion of a persulfonimide side chain to the pyrazole heterocycle using a triazole linker 16, 17 [56]. The addition of the persulfonimide fragment is expected to improve DPP-IV-inhibitory potential by interacting with Arg125, Gly741, and Trp629. Furthermore, the linker chain(1,2,3-triazole) can interact with His740, Val122, and Glu205. The two fragments were linked, and the triazole ring was synthesized using a click-chemistry approach based on a 1,3-dipolar cycloaddition reaction of alkynes and azide-substituted pyrazole 18 (Scheme 8).
Scheme 8.
Reagents and conditions: (i) CuI/DCM and DIPEA and Me6TREN, 90–98%.
Another study described the use of the classical synthetic approach to construct pyrazole derivatives from α,β-unsaturated ketones 19 in reaction with phenylhydrazines, resulting in novel DPP-IV inhibitors featuring an additional furan ring 20 (Scheme 9) [57].
Scheme 9.
Reagents and conditions: (i) Piperonal, 10% NaOH, absolute ethanol, rt 24 h; (ii) 4-substituted phenylhydrazine hydrochloride, AcOH, absolute ethanol, reflux 10 h, 85–87%.
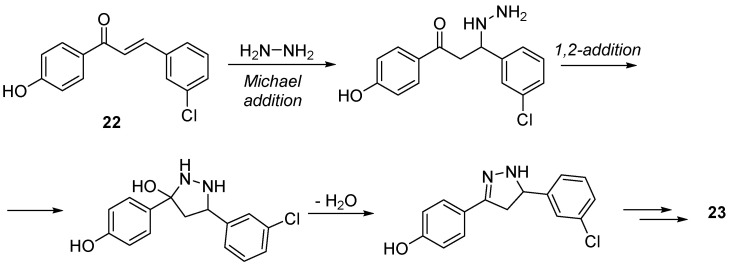
Sharma et al. employed a comparable method for constructing the target heterocycle in their research aimed at identifying novel potent DPP-IV inhibitors (Scheme 10) [58]. Their lead compound 21 was synthesized in a three-step procedure, with the key reaction between the substituted chalcone 22 and hydrazine in an acetic acid medium, resulting in the formation of the heterocycle 23 and achieving N-acylation in a single stage. This approach provides a versatile method for synthesizing pyrazole derivatives through the use of hydrazine and 1,3-difunctional compounds, including 1,3-dicarbonyl compounds, chalcones, β-ketoesters, etc. [59]. The reaction likely occurs through the initial conjugate addition of the hydrazine to the β-position of 22, followed by ring closure via 1,2 addition to the carbonyl group and subsequent elimination of water (Scheme 11) [60]. The final Williamson reaction involving the substituted phenacetyl bromide derivative yielded the final product 21. The obtained compound exhibited an IC50 within the micromolar range.
Scheme 10.
Reagents and conditions: (i) 3-chlorobenzaldehyde, NaOH, MeOH, reflux, 6 h; (ii) NH2NH2·H2O, AcOH, 90 °C, 81%; (iii) 2-bromo-1-(p-tolyl)ethan-1-one, dry acetone, K2CO3, reflux, 8–10 h, 61–89%.
Scheme 11.
Mechanism of the formation of pyrazole-derivative 23.
The pyrazolidine scaffold is present in potential DPP-IV inhibitors, in particular when coupled with isoleucine 24 (Scheme 12) [61]. These inhibitors were derived from previously reported cyano-pyrazolidines, which demonstrated inhibitory activity in the micromolar range [62]. The core heterocycle 25 was synthesized through a reaction between Boc-protected hydrazine 26 and 1,3-dibromopropane in the presence of Et4NBr.
Scheme 12.
Reagents and conditions: (i) 50% NaOH, dibromopropane, Et4NBr, toluene, reflux, 5 h, 87%; (ii) 4 M HCl, dioxane, 100%; (iii) Boc–isoleucine, EDCI, TEA, CH2Cl2, 78%; (iv) RNCO, CH2Cl2, rt, 12 h, 70–80%; (v) 4 M HCl, dioxane–EtOAc, rt, 12 h, 87%.
2.2.2. Isoxazole
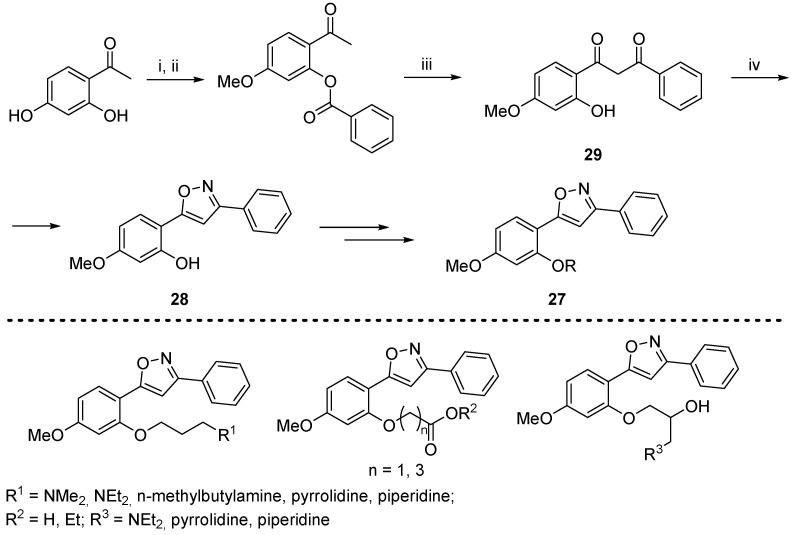
The first documented isoxazole-based DPP-IV inhibitors were developed in work by Kumar et al., which included the synthesis of a series of 3,5-diarylisoxazoles 27 [63]. The key reaction used a classical approach to construct the heterocycle 28 through the interaction of 1,3-dicarbonyl compound 29 and hydroxylamine (Scheme 13). The intermediate 28 was obtained in moderate yield, which is common when unsymmetrical diketones undergo this process [64]. The final target products 27 are synthesized via O-alkylation of the phenolic hydroxyl group.
Scheme 13.
Reagents and conditions: (i) (CH3)2SO4, K2CO3, acetone; (ii) benzoyl chloride, pyridine, 1 h, rt, 82% over two steps; (iii) KOH, pyridine, 8 h, rt, 82%; (iv) NH2OH·HCl, pyridine, 45%.
A recent study by Karandikar et al. examined benzo-fused isoxazoles with an acetamide side chain 30 [65]. The compounds were synthesized through a three-step synthetic strategy, with the key reaction being a Posner ring transformation of 4-hydroxycoumarin 31 to form 1,2-benzisoxazol-3-acetic acid 32 (Scheme 14). The amide side chain was introduced using coupling reagents. The introduction of an additional glycine fragment in the amide side chain, serving as a spacer, improved the DPP-IV-inhibitory activity.
Scheme 14.
Reagents and conditions: (i) Na metal, 70–80 °C, 1 h, 45%; (ii) NH2OH.HCl, NaHCO3, MeOH, 64–66 °C, 15 h, 40%; (iii) NR1R2.TFA/NR1R2, EDCl, HOBt, TEA, DCM, rt, 16 h, 32–71%.
2.2.3. Imidazole
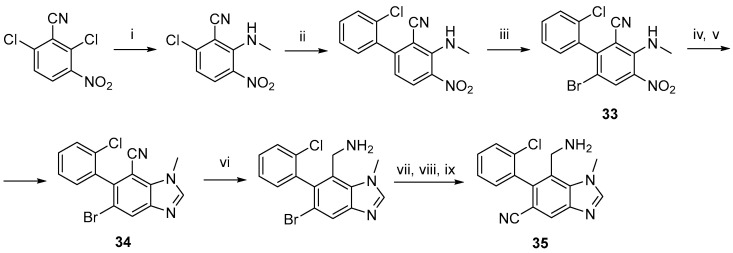
The first documented DPP-IV inhibitors with benzimidazole as a core structure were developed in 2008 by Wallace et al. [66]. The molecular design of the prospective inhibitors was based on available crystallographic data of DPP-IV in conjunction with 2-phenylbenzylamine. The synthetic methodology consists of several steps, with the key ring-forming process being the condensation of substituted o-diamine, formed in situ from 33 with formic acid. Following the cyanide reduction of 34, amine Boc protection, Pd-coupling reaction to introduce the CN group, and final deprotection, the target product 35 was obtained (Scheme 15). The authors described the versatility of the synthetic protocol, enabling the synthesis of molecules with diverse substituents to the heterocycle.
Scheme 15.
Reagents and conditions: (i) CH3NH2, THF, 82%; (ii) o-ClC6H4-B(OH)2, Pd2dba3, DavePhos, K3PO4, DMA, 68 °C, 74%; (iii) Br2, AcOH, 88 °C, 94%; (iv) Fe, AcOH; (v) formic acid, polyphosphoric acid, 73% over two steps; (vi) BH3-THF, reflux then HCl, 33%; (vii) Boc2O; (viii) Zn(CN)2, Pd2dba3, DavePhos, K3PO4, DMA, 92 °C; (ix) TFA, DCM, 70% over three steps.
More recently, Sunil et al. demonstrated that aminosubstituted benzimidazoles 36 could be a novel class of potent DPP-IV inhibitors [67]. The parent 2-amino benzimidazole 37 was prepared by treating o-phenylenediamine with cyanogen bromide (Scheme 16).
Scheme 16.
Reagents and conditions: (i) BrCN, H2O, 4 h, 70%; (ii) chloroacetyl chloride, dry benzene, 0–5 °C, then reflux for 6 h, 85%; (iii) aminoacetic acid, pyridine, EtOH, 6 h, 75%.
Katarina Tomovic et al. recently reported another example of N-substituted benzimidazoles as potential DPP-IV inhibitors [68]. The authors obtained two series of benzimidazole derivatives starting with commercially available substituted 2-mercapto-benzimidazoles (Scheme 17).
Scheme 17.
Reagents and conditions: (i) KMnO4, NaOH, H2O, reflux, 52%; (ii) NH4OH, 150 °C in welded ampoule, 77%; (iii) Tetrabutylammonium bromide, dry K2CO3, 2-chloro-1-phenylethan-1-one, 25 °C, acetonitrile, 82%.
2.3. Five-Membered Heterocycles with Three Heteroatoms and Their Benzo-Fused Derivatives
2.3.1. Triazole
Apart from triazoles fused to larger heterocycles, which are reviewed in Section 4, triazole-based DPP-IV inhibitors can be found in early 2016 in the literature. Gundetti et al. reported the synthesis and DPP-IV-inhibitory activity of 1,2,3-tiazoles 38 designed as analogues of Sitagliptin [69]. The suggested synthetic protocol is based on click chemistry—1,3-dipolar cycloaddition between azide 39 and appropriate alkyne (Scheme 18). Starting with (3R)-N-(tert-butoxycarbonyl)-3-amino-4-(2,4,5-trifluorophenyl)butanoic acid 40, the target compounds are obtained in a total of five synthetic steps and showed nanomolar IC50 values.
Scheme 18.
Reagents and conditions: (i) NaBH4, I2, THF, 88%; (ii) TsCl, TEA, DCM, 76%; (iii) NaN3, DMSO, 80 °C, 85%; (iv) alkynes, Cu(OAc)2, CH3CN, (v) 10% Pd/C-H2, MeOH, 65–85%.
The authors extended the scope of the synthetic methodology to the construction of analogues with bulkier groups 41 attached to the heterocycle [70]. The starting 42 was converted to a terminal alkyne 43 in three steps with the use of the Ohira–Bestmann reagent; subsequent azide cycloaddition yielded the final heterocycle 44 (Scheme 19). The obtained novel inhibitors performed poorly in comparison with the previous series; however, they still showed micromolar range IC50 values. The procedure was further used by the authors to install bulkier substituents to the triazole heterocycle [71].
Scheme 19.
Reagents and conditions: (i) CAN/MeOH, rt, 5 h, 97%; (ii) DIBAL, −78 °C, 3 h, 86%; (iii) Bestmann–Ohira reagent, K2CO3, MeOH, rt, 14 h, 94%; (iv) arylazides, Cu(OAc)2, CH3CN, rt, 3 h, 98–82%; (v) HBr/AcOH, 80 °C, 14 h, 65–85%.
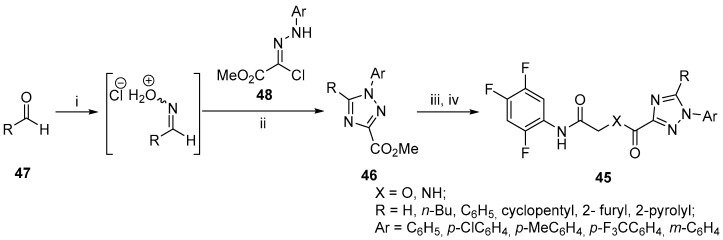
Sitagliptin and Vildagliptin served as important references in the development of novel 1,2,4-triazole derivatives 45 as DPP-IV inhibitors [72]. The suggested inhibitors exhibited IC50 values in the nanomolar range as well as excellent selectivity for DPP-IV over Quiescent cell proline dipeptidase (QPP) and DPP-8/9. The triazole 46 was again constructed via 1,3-dipolar cycloaddition from aldehydes 47 with hydrazonoyl hydrochlorides 48 in the presence of hydroxylamine hydrochloride (Scheme 20) [73]. The process demonstrates a high functional group tolerance and was subsequently adapted by the authors to be able to use nitriles as starting reagents as well [74]. Another variation is the use of Vilsmeier reagent with hydrazonoyl hydrochlorides [75].
Scheme 20.
Reagents and conditions: (i) NH2OH.HCl, toluene; (ii) toluene, reflux, 1–2 h, 28–91%, (iii) DBU, MeOH, r.t., 6 h, >90%; (iv) 2-hydroxy-N-(2,4,5- trifluorophenyl)acetamide or 2-amino-N-(2,4,5-trifluorophenyl)acetamide, Bis(2-oxo-3-oxazolidinyl)phosphinic chloride (BOP-Cl), NEt3, CH2Cl2, 4 h, 71–92%.
The reaction of substituted alkynes with sodium azide as a route to 1,2,3-triazole derivatives 49 containing carboximidamide functionality has also been documented [76]. The compounds demonstrated remarkable DPP-IV-inhibitory activity, exhibiting IC50 values in the nanomolar range. The synthesis consists of three steps, in which the required trichloroacetimidamide 50 is generated in situ and reacted with phenylacetylene to yield the appropriately substituted alkyne 51 (Scheme 21). The final reaction of the obtained 51 with sodium azide occurs at room temperature in the water–methanol medium.
Scheme 21.
Reagents and conditions: (i) acetonitrile, rt, 10 min; (ii) Et3N, CuI, rt, 5 h; (iii) NaN3, methanol-water, rt, 8 h, 60–77% overall yield.
2.3.2. Oxadiazole
A good example of 1,2,4-oxadiazole-based DPP-IV inhibitors can be found in the work of Xu et al. [77]. A series of highly potent and selective DPP-IV inhibitors 52 were prepared by the authors and are recognized as some of the most powerful inhibitors in the literature. The preparation of 52 involves a coupling reaction between aspartic acid derivatives 53 and substituted benzamidoxime, mediated by 1,1-carbonyl diimidazole (CDI) (Scheme 22). The synthetic route consists of five steps, beginning with the Boc-protected methyl ester of aspartic acid 53. The subsequent coupling reaction of 53 introduces a pyrrolidine functionality, followed by enolate alkylation at the α-position in the resulting 54.
Scheme 22.
Reagents and conditions: (i) EDC, HOBt, DIEA, pyrrolidine, DMF; (ii) KHMDS, MeI, −78 °C; (iii) LiOH, THF, H2O; (iv) CDI, 4- methanesulfonylbenzamidoxime, rt, 1 h, then 110 °C, 12 h; (v) TFA/CH2Cl2, 1 h.
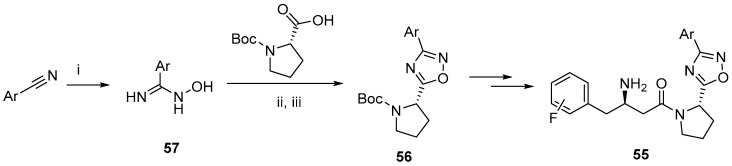
Nordhoff et al. also observed the increased potency of 1,2,4-oxadiazole-based DPP-IV inhibitors that contain a pyrrolidine moiety 55 [78]. The authors investigated the inhibitory activity of 1,3,4-oxadiazole derivatives as DPP-IV inhibitors, but they were outperformed by their 1,2,4- analogues 55. The key ring-forming reaction to obtain 56 was a condensation of amidoxime derivatives 57 with carboxylic acids (Scheme 23).
Scheme 23.
Reagents and conditions: (i) NH2OH, K2CO3, MeOH; (ii) DIC, DCM; (iii) pyridine, reflux.
3. Six-Membered Heterocycles
3.1. Six-Membered Heterocycles with One Heteroatom and Their Benzo-Fused Derivatives
3.1.1. Pyridine, Piperidine, Quinoline, and Isoquinoline
The work of Kaczanowska et al. outlines aminomethyl pyridine derivatives 58 as nanomolar DPP-IV inhibitors [79]. The synthesis of the core heterocycle 59 was based on earlier research by the authors [80]. The process can be described as a base-catalyzed cyclocondensation between unsaturated oxo acid 60 and β-aminocrotonitrile, formed in situ during the course of the reaction (Scheme 24).
Scheme 24.
Reagents and conditions: (i) t-BuOK, AcCN, rt, 16 h, 81%.
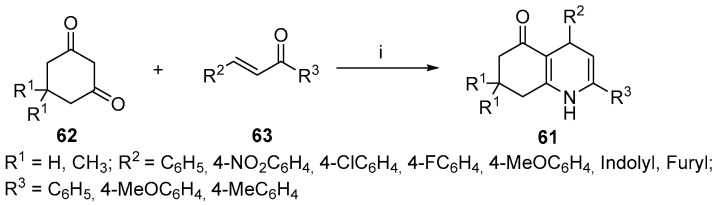
Kumar et al. explored the antihyperglycemic properties of partially saturated quinoline derivatives and evaluated the compounds against several in vitro diabetes models [81]. The authors prepared 2,4-diaryl substituted polyhydroquinolines 61 using a reaction between cyclic 1,3-diketones 62 and chalcones 63 in the presence of ammonium acetate and p-toluenesulfonic acid as a catalyst (Scheme 25). The synthetic protocol was referenced from the literature [82].
Scheme 25.
Reagents and conditions: (i) NH4OAc; 10 mol% p-toluenesulfonic acid, MeOH, reflux, 75–93%.
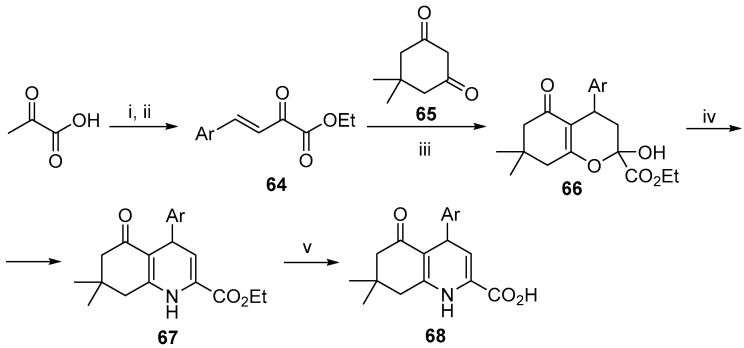
In order to obtain polyhydroquinolines with an ester functionality, the authors employed β,γ-unsaturated α-ketoesters 64. This was achieved through a base-catalyzed reaction between aromatic aldehydes and pyruvic acid, followed by an esterification (Scheme 25) [81]. Subsequent ring-forming reaction with dimedone 65 yielded the chromene derivative 66, which was transformed into the target quinoline derivative 67. The products 67 were transformed into the corresponding carboxylic acids 68 (Scheme 26).
Scheme 26.
Reagents and conditions: (i) KOH, MeOH, rt, 10–15 h; (ii) CH3COCl, EtOH, 70 °C, 6–8 h, 85–93% over two steps; (iii) p-toluenesulfonic acid, MeOH, rt, 78–92%; (iv) NH4OAc, EtOH, reflux, 88–95%; (v) NaOH, MeOH, 80–97%.
Prospective quinoline inhibitors that contain a pyrazole substituent have also been explored [83]. The synthetic pathway starts from commercially available 2-chloroquinoline, which undergoes a reaction with 4-hydroxybenzaldehyde and subsequently with 4-methoxyacetophenone to yield the chalcone derivative 69 (Scheme 27). The cyclocondensation of 69 with hydrazine hydrate or phenylhydrazine gave the final products 70. In another approach, 2-chloroquinoline reacted with 4-hydroxyacetophenone, leading to a product that was condensated with substituted aromatic aldehydes to furnish 71. In a similar matter, the formation of the pyrazole ring was accomplished through the reaction of 71 with hydrazine hydrate or phenylhydrazine, and the target 72 was obtained.
Scheme 27.
Reagents and conditions: (i) 4-hydroxybenzaldehyde, DMF, K2CO3, 100 °C 6 h; (ii) 4-methoxyacetophenone, ethanol, 10% aq. NaOH, stirring for 2 h, 64% over two steps; (iii) hydrazine hydrate, ethanol, reflux 4 h, 80%; (iv) phenylhydrazine ethanol, reflux for 6 h, 75%; (v) 4-hydroxyacetophenone, DMF, K2CO3, reflux for 12 h; (vi) aldehydes (2-chlorobenzaldehyde or 4-methoxybenzaldehyde), ethanol, 10% aq. NaOH, stirring for 2 h, 57–65% over two steps.
3.1.2. Coumarin
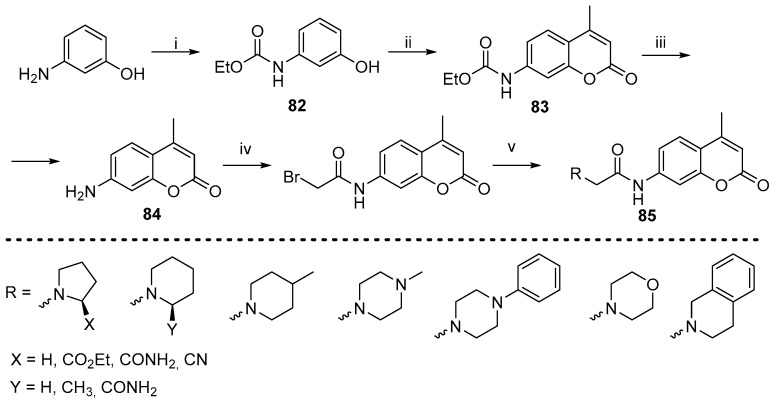
The pioneering research on coumarin derivatives for the inhibition of DPP-IV can be attributed to the work of Soni et al. in 2016 [84]. The authors prepared a series of coumarins featuring an amide side chain 73 and 74 (Scheme 28). The compounds were synthesized using a two-step synthetic route, beginning with the condensation of 2,4-dihydroxybenzaldehyde 75 with acetoacetic or malonic ester to form the core coumarin heterocycle 76 and 77 [85,86]. The final products 73 and 74 were obtained by alkylation of the 7-hydroxyl group in 76 and 77.
Scheme 28.
Reagents and conditions: (i) ethyl acetoacetate, piperidine, 0 °C to rt, overnight; (ii) KSF montmorillonite, 160 °C, 46%; (iii) substituted amine, anhydrous K2CO3, KI, DMF, 70–80 °C, 12–18 h, 43–91%.
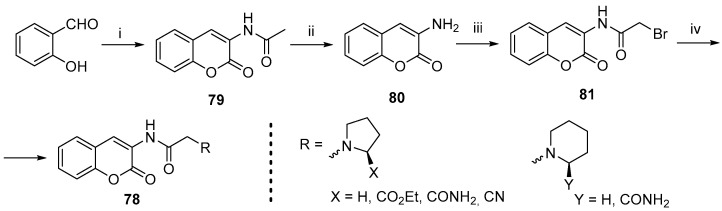
In subsequent research, the authors successfully synthesized a new series of amino-substituted coumarin derivatives [87]. Two novel series of coumarins bearing amine substituent on positions 3- and 7- were obtained. The synthesis of 3-aminocoumarin derivatives 78 was carried out using a two-step approach (Scheme 29). The initial 2-hydroxybenzaldehyde underwent a reaction with N-acetyl glycine in a Perkin reaction, resulting in the formation of the acetamide derivative 79. Acidic deprotection yielded the 3-aminocoumarin as a free base 80 [88]. Acylation with bromoacetyl bromide furnished the substituted 81, which was reacted with various amines to yield the derivatives 78. The 7-aminocoumarin series was synthesized via a Pechmann condensation starting with substituted 3-aminophenol derivative 82 (Scheme 30). The resulting coumarin 83 was converted to its free base 84 and, similarly to the synthesis of the previous series, was alkylated with bromoacetyl bromide and reacted with amine derivatives to yield the final product 85.
Scheme 29.
Reagents and conditions: (i) N-acetyl glycine, Ac2O, NaOAc, reflux, 7 h, 47%; (ii) EtOH, conc. HCl, reflux, 1 h, 61%; (iii) 2-bromoacetyl bromide, TEA, DCM, rt, 2 h, 89%; (iv) substituted amine, Et3N, DMF, rt, 16 h, 49–85%.
Scheme 30.
Reagents and conditions: (i) ethyl chloroformate, EtOAc, rt, 2 h, 48%; (ii) ethyl acetoacetate, 70% EtOH in H2SO4, rt, 16 h, 85%; (iii) AcOH, H2SO4, 106–108 °C, 1 h, 71% (iv) bromo acetyl bromide, Et3N, DCM, rt, 2 h, 93%; (v) substituted amine derivatives, TEA, DMF, rt, 16 h, 47–72%.
The authors employed a similar synthetic procedure to obtain a series of 7-aminocoumarin derivatives featuring a proline and sulfonamide moiety in the side chain [89].
The Pechmann condensation was also used in the recent research by Jasim et al. for the synthesis of coumarins, which incorporated an additional condensated benzene ring (Scheme 31) [90]. The synthetic protocol begins with substituted naphthalene 86, which, upon diazotation and treatment with methanol, is transformed to its methoxy counterpart 87. The Pechmann condensation of 87 with 3-oxoglutaric acid yielded the coumarin derivative 88. The final products 89 were obtained after esterification of the carboxylic group with substituted phenols. In later studies, the authors replaced the methoxy group in 89 with chlorine, therefore enhancing the potency of the obtained compounds [91].
Scheme 31.
Reagents and conditions: (i) NaNO2, MeOH, AcOH, 6 h, rt, 43%; (ii) 3-oxoglutaric acid, conc. H2SO4, sonication, 1.5 h, 30 °C, 88%; or 3-oxoglutaric acid, conc. H2SO4, 20 h, rt, 80%; (iii) substituted phenol derivative, SOCl2, 63–76%.
3.2. Six-Membered Heterocycles with Two Heteroatoms and Their Benzo-Fused Derivatives
3.2.1. Piperazine
Substituted aryl-piperazines containing β-amino acids have been reported as nanomolar DPP-IV inhibitors [92]. The synthetic procedure begins with N-benzyl substituted piperazine 90, which undergoes a coupling reaction with Boc-protected β-aminoacids to furnish 91. After benzyl deprotection, the key intermediate 92 is produced. Subsequent alkylation on the unsubstituted nitrogen in 92 led to the formation of four groups of DPP-IV inhibitors (Scheme 32).
Scheme 32.
Reagents and conditions: (i) (R)-ArCH2CH(NHBoc)CH2CO2H, EDC, HOBt, DIEA, DMF; (ii) H2, Pd(OH)2, MeOH.
Kushwaha et al. synthesized piperazines containing an additional heterocyclic substituent such as pyrrolidine, thiazolidine, piperidine, or morpholine (Scheme 33) [93]. The compounds were obtained from the respective five- or six-membered heterocycle 93, which was treated with chloroacetyl chloride to give the intermediate 94. The final substitution reaction with aryl-piperazine 95 gave the final products 96.
Scheme 33.
Reagents and conditions: (i) chloroacetyl chloride, TEA, DCM, −15 °C to 0 °C, 2 h, 80–93%; (ii) chloroacetic acid, DCC, DCM, 0 °C, 4 h; (iii) TEA, TFH, rt, 6–8 h, 64–87%.
Piperazine can be functionalized with sulfonamide moieties at the two nitrogen atoms, giving rise to sulfonamide-piperazine inhibitors 97 (Scheme 34) [94].
Scheme 34.
Reagents and conditions: (i) NaOH, acetone, reflux, 1.5 h, 1–76%.
3.2.2. Pyridazine
A recent report by Nidhar et al. showed the synthesis of novel pyridazine-acetohydrazide hybrids 98 and their DPP-IV-inhibitory activity in nanomolar concentrations [95]. The heterocycle was formed by a base-catalyzed reaction of 2-cyanoacetohydrazide (99) and benzil (Scheme 35). The obtained core heterocycle 100 was acylated, and the resulting product 101 was further converted to the target 98.
Scheme 35.
Reagents and conditions: (i) benzil, K2CO3, MeOH, reflux, 90–95%; (ii) ethyl 2-chloroacetate, K2CO3, anhydrous DMF, 2 h, good to excellent yield.
3.2.3. Quinazoline and Quinoxaline
The quinazoline heterocycle could be seen in the approved drug Linagliptin (Figure 3), which is a highly potent DPP-IV inhibitor approved by the FDA in 2011 [96].
Figure 3.
Structure of Linagliptin.
Quinazoline derivatives coupled with thiazole have been identified as a novel class of DPP-IV inhibitors that also possess inhibitory potential against DPPH [97]. These molecules were synthesized in five steps, starting with anthranilic acid or its iodo-derivative (Scheme 36). The reaction with benzoyl chloride gave the benzoxazine derivative 102, which reacted with hydrazine monohydrate to form the target quinazoline core 103. Condensation with aldehydes and subsequent treatment with POCl3 resulted in the formation of the intermediate product 104, which, when coupled with thiazole derivatives 105, gave the final products 106.
Scheme 36.
Reagents and conditions: (i) anhydrous pyridine, 2–8 °C, sodium bicarbonate, 1 h, 72–76%; (ii) H2NN2H.H2O, ethanol, 6 h, 68–74%; (iii) aldehydes, ethanol, glacial acetic acid, reflux, 8 h, 66–76%; (iv) anhydrous toluene, DIPEA, POCl3, reflux, 4–6 h, 64–82%; (v) phenol, reflux, 4 h, 70–78%.
The authors also synthesized quinazolines bearing a thiazole ring at C-2 using the same methodology [98]. The obtained compounds showed IC50 values in the nanomolar range as well as good selectivity for DPP-IV over DPP-8/9. A similar synthetic approach could be found in the work of Zayed et al., describing novel quinazolinone derivatives featuring a sulfonamide side chain [99].
Quinoxaline derivatives have also been reported as exhibiting DPP-IV-inhibitory activity and hypoglycemic properties (Scheme 37) [100]. The heterocyclic core 107 was obtained using a base-catalyzed condensation between o-phenylenediamine and oxalic acid, followed by alkylation with methyl iodide. In order to install a sulfonamide functionality, 107 was treated with chlorosulfonic acid to obtain the 6-sulfonyl chloride intermediate 108. After a reaction of 108 with aromatic and heterocyclic amines, the products 109 were obtained. Alternatively, 108 reacted with sulfur-containing drugs such as sulfanilamide, sulfathiazole, sulfapyridine, and sulfadiazine to afford the derivatives 110.
Scheme 37.
Reagents and conditions: (i) oxalic acid, 4 N HCl, reflux, 4 h; (ii) anhydrous K2CO3, DMF, methyl iodide, heating, 6 h, 99%; (iii) ClSO3H, heating, 90 °C, 3 h; (iv) heterocycles or primary amines, EtOH, reflux, 3 h, 72–75%; (v) sulfa drugs, Et3N, EtOH, 6 h, 81–85%.
3.2.4. Pyrimidine
The drug alogliptin (Figure 4) can be viewed as an example of a pyrimidine derivative as a DPP-IV inhibitor [101,102].
Figure 4.

Structure of alogliptin.
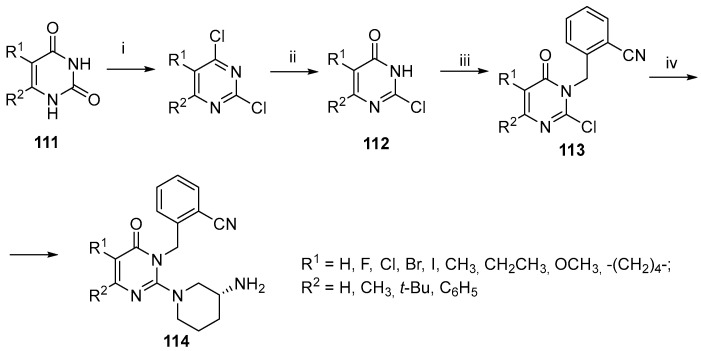
Zhang et al. synthesized pyrimidinone derivatives from pyrimidinediones 111, which underwent chlorination with POCl3, followed by selective alkaline hydrolysis to yield 112 [103]. Alkylation with cyanobenzyl bromide produced a mixture of regioisomers, of which 113 was further used by the authors to synthesize the target inhibitors 114 (Scheme 38).
Scheme 38.
Reagents and conditions: (i) POCl3, dimethylaniline, reflux; (ii) NaOH, 47–76%; (iii) NaH, LiBr, 2-cyanobenzyl bromide, 20–52%; (iv) 3-(R)-aminopiperidine, NaHCO3, 60 °C, 49–74%.
Further research on the pyrimidinone derivatives that incorporate an additional 3-aminopiperidine fragment can be seen in the work of Ning Li et al. [104]. The synthesis follows a four-step procedure starting with 6-chlorouracil (115), which undergoes alkylation on both nitrogen atoms to form the precursor 116. After reacting 116 with (R)-3-(Boc-amino) piperidine and subsequent amine deprotection, the final products 117 were obtained (Scheme 39). A similar synthetic approach was employed by Vibhu Jha et al. in their research on novel N-methylated and N-benzylated pyrimidinediones [105]. Modification of the heterocyclic core 117 to incorporate benzoic acid moiety has also been explored [106,107].
Scheme 39.
Reagents and conditions: (i) R1Br, DIPEA, DMF, rt; (ii) R2Br, K2CO3, KI, DMF, rt; (iii) (R)-3-(Boc-Amino)piperidine, K2CO3, DMF, 90 °C, N2 atmosphere; (iv) CH2Cl2, TFA, rt, 65–84%.
Mourad et al. developed novel dihydropyrimidine phthalimide hybrids as analogues of alogliptin and phthalimide-based selective DPP-IV inhibitors [108]. The synthetic protocol starts from appropriately substituted chalcones 118, which reacted with thiourea to form 4,6-diaryl-3,4-dihydropyrimidine-2(1H)-thiones 119. The reaction with α-phthalimido-o-toluyl chloride 120 afforded the target compounds 121 (Scheme 40).
Scheme 40.
Reagents and conditions: (i) NaOH, dil. EtOH, rt; (ii) NaOH, abs. EtOH, 66–76%; (iii) abs. dichloromethane, TEA, rt, 65–75%.
3.2.5. 1,4-Dioxane
Artasensi et al. made a significant advancement in overcoming the limitations of existing multidrug therapies by developing ligands that inhibit both DPP-IV and carbonic anhydrases [109]. The synthetic methodology for their most potent compounds starts with 1,4-benzodioxan-2-carboxylic acid (122), which is converted to the corresponding aldehyde 123 via a three-step sequence (Scheme 41). The β-hydroxyester derivative 124 was obtained via the Reformatsky reaction of 123 with ethyl bromoacetate. Subsequent reduction of the ester group to give primary alcohol, followed by its protection with trityl chloride, afforded 125. The Mitsunobu reaction of 125 gave the azide derivative 126, which, upon deprotection and mesylation, yielded the key intermediate 127. Nucleophilic substitution reaction with 128, followed by sulfonamide deptorection, afforded 129, which, after azide reduction, gave the target compound 130.
Scheme 41.
Reagents and conditions: (i) SOCl2, DCM, reflux; (ii) N,O-dimethylhydroxylamine, DCM, rt, 47% over two steps; (iii) LiAlH4, THF, −20 °C, quantitative yield; (iv) Zn, TBDMSiCl, ethyl bromoacetate, THF, reflux, 89%; (v) LiAlH4, THF, −10 °C, 48%; (vi) TrtCl, TEA, DCM, rt, 96%; (vii) PPh3, diethyl azodicarboxylate (DEAD), diphenylphosphorylazide (DPPA), THF, rt, 32%; (viii) Amberlist 15, DCM/MeOH, reflux, 41%; (ix) MsCl, TEA, DCM, rt, 92%; (x) K2CO3, N,N-dimethylformamide (DMF), 90 °C, 74%; (xi) NH2NH2·H2O, MeOH; (xii) NH2NH2·H2O, PdO, MeOH, reflux, 83%.
3.3. Six-Membered Heterocycles with Three Heteroatoms and Their Benzo-Fused Derivatives
Triazine
1,3,5-Triazines coupled with thiazolidine-2,4-diones have been shown to possess both antibacterial properties and DPP-IV-inhibitory activity [110]. The compounds were prepared using 2,4,6-trichloro-1,3,5-triazine (131), which reacted with amines to form disubstituted amino-1,3,5-triazine 132. The latter reacted with thiazolidine-2,4-dione 133, yielding the final products 134 (Scheme 42).
Scheme 42.
Reagents and conditions: (i) RNH2, NaOH, 40–45 °C, 64–87%; (ii) K2CO3, reflux, 120–135 °C; (iii) conc. H2SO4, H2O, reflux, 80%.
A similar synthetic methodology, starting with 2,4,6-trichloro-1,3,5-triazine 131, has been used to yield 1,3,5-triazine derivatives containing sulfonamide [111] and sulfonamide and morpholine functionalities [112]. Recently, Gupta et al. obtained morpholine-1,3,5-triazine derivatives containing an additional five- or six-membered heterocycle (pyrimidine, pyrazole) [113,114,115].
4. Polycyclic Fused Heterocycles
4.1. Bicyclic 5-5 Systems
A prominent example of fused five-membered heterocycles in the structure of clinically approved DPP-IV inhibitor is the drug Omarigliptin (Figure 5) [116,117].
Figure 5.

Structure of Omarigliptin.
Betancort et al. developed a series of pyrrolo[2,1-b]thiazole derivatives starting with (S)-2-amino-2-phenylacetic acid 135, which was methylated and converted to benzaldimine 136 (Scheme 43) [118]. After alkylating 136 with allyl bromide, and replacing the benzaldimine fragment with a Boc protecting group, the intermediate 137 was obtained. The C=C bond in 137 underwent ozonolysis and reductive work-up, resulting in the cyclic 138. The thiazolidine ring was formed after the condensation of 138 with L-cysteine methyl ester as a mixture of diastereomers 139, and after refluxing in toluene with p-toluene sulfonic acid, the additional pyrrolidine ring in 140 was formed. After deprotecting the amine group and converting the ester to nitrile, the target 141 was formed.
Scheme 43.
Reagents and conditions: (i) SOCl2, MeOH, 0 °C, 2 h, 89%; (ii) PhCHO, TEA, MgSO4, CH2Cl2, rt, 18 h, 97%; (iii) t-BuOK, CH2CHCH2Br, THF, rt, 18 h; (iv) 6 N HCl, EtOAc, 1 h; (v) Boc2O, THF, reflux, 18 h, 87%; (vi) O3, PPh3, CH2Cl2, −78 °C, 36%; (vii) LiOH, THF, 1 h, 95%; (viii) L-Cys-OMe·HCl, NaHCO3, EtOH, 16 h, 51%; (ix) p-TsOH, toluene, reflux, 1 h, 23%; (x) NH3, MeOH, rt, 2 h, 97%; (xi) TFAA, TEA, CH2Cl2, rt, 1 h, 84%; (xii) TFA, CH2Cl2, rt, 1 h, 14% after preparing HPLC; (xiii) HCl (g), CH2Cl2/Et2O, 2 h, 53%.
Zhang et al. developed analogies of Omarigliptin bearing fluorine or CF3-substituted tetrahydropyran rings (Scheme 44) [119]. The synthetic strategy begins with the tetrahydropyran 142, which reacts with morpholine to produce a mixture of enamine derivatives 143a and 143b, which then reacts with Umemoto’s reagent to obtain the trifuoromethylated ketone 144. After reductive amination of 144 with 2-(methylsulfonyl)-2,4,5,6-tetrahydropyrrolo[3,4-c]pyrazole (145), the target 146 was formed. The final Boc deprotection resulted in the lead compound 147. Additionally, t-BuOK treatment of 147 leads to cleavage of the methylsulfonyl group.
Scheme 44.
Reagents and conditions: (i) morpholine, toluene, reflux, Dean–Stark apparatus; (ii) Umemoto’s reagent (S-(trifluoromethyl)dibenzothiophenium trifluoromethanesulfonate), DMAP, DMAc, 57% over two steps; (iii) toluene, reflux, then NaBH(OAc)3, CH3COOH, 1,2-dichloroethane, 72%; (iv) CHCl3, reflux, Dean-Stark apparatus, then NaBH(OAc)3, CH3COOH, 1,2-dichloroethane, 72–75%; (v) TFA, DCM, 77–90%; (vi) t-BuOK, THF, 60%.
4.2. Bicyclic 5-6 Systems
Two prominent examples of DPP-IV inhibitors featuring bicyclic 5-6 heterocycles are Sitagliptin and Anagliptin, respectively (Figure 6).
Figure 6.
Structure of Sitagliptin and alogliptin.
One of the simplest bicyclic 5-6 heterocyclic systems that can be found in DPP-IV inhibitors is the imidazo[1,2-a]pyridine. Qing Lie et al. synthesized imidazo[1,2-a]pyridines and evaluated their DPP-IV-inhibitory activity [120]. The key ring-forming step is a reaction between 2-aminopyridine and phenacetyl bromides 148 (Scheme 45). The resultant 149 is treated with the Vismeier reagent to produce the aldehyde 150, followed by condensation with hydroxylamine hydrochloride to form the oxime 151. After reducing 151 with zinc powder in acetic acid, the resultant amine group was Boc-protected for the purposes of purification, and after final deprotection, the target 152 was obtained.
Scheme 45.
Reagents and conditions: (i) CuBr2, ethyl acetate, CHCl3, reflux, 16 h, 90%; (ii) 2-aminopyridine, NaHCO3, EtOH, rt, 80–84%; (iii) POCl3, DMF, 0–60 °C, 2 h, 65–68%; (iv) NH2OH. HCl, pyridine, EtOH, reflux, 83–90%; (v) Zn, NaOAc, AcOH, rt, 10 h, then Boc2O, NaOH, EtOH, rt, 3 h, then HCl(gas), ethyl acetate/ether, 0 °C, 36–71%.
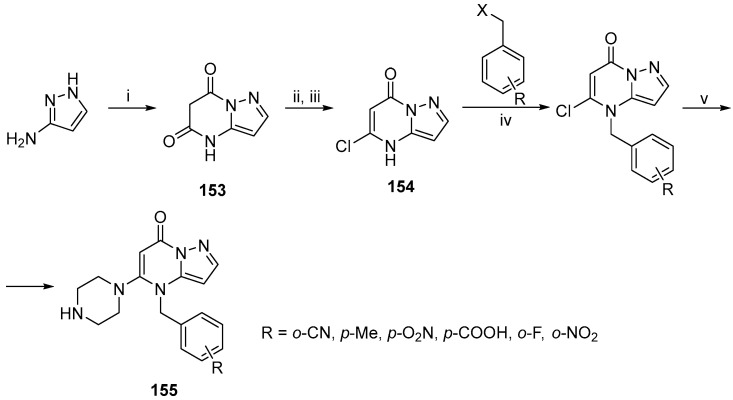
Another heterocyclic system found in compounds that have a variety of interesting biological properties is the pyrazolo[1,5-a]pyrimidine ring system [121,122,123]. Deng et al. synthesized novel pyrazolo[1,5-a]pyrimidin-7(4H)-one derivatives as DPP-4 inhibitors that exhibit high potency and selectivity (Scheme 46) [124]. The synthetic methodology begins with 1H-pyrazol-5-amine, which reacts with diethyl malonate to produce the intermediate 153. Chlorination with POCl3 and selective alkaline hydrolysis furnished the necessary chloro derivative 154. Alkylation of 154 with benzyl halide derivatives and substitution of the chlorine atom gave the prospective inhibitors 155.
Scheme 46.
Reagents and conditions: (i) diethyl malonate, EtONa EtOH, reflux, 71%; (ii) POCl3, N,N-dimethyl-aniline, 100 °C, 80%; (iii) 1 N NaOH, 90 °C; (iv) Cs2CO3 or K2CO3 or DIPEA, DMF, 90 °C, 46–69%; (v) DIPEA, DMF, 90 °C, 20–33%.
Further research by the authors resulted in the development of DPP-IV inhibitors with increased potency compared to 155 by replacing the piperazine fragment in position 5 with (R)-piperidin-3-amine. The synthesis follows the methodology specified in Scheme 46 [125].
Patel et al. developed a series of triazolotriazine derivatives as novel DPP-IV inhibitors based on in silico studies and utilizing Sitagliptin as a reference compound [126]. After bromination of 1,2,4-triazole, the dibromo derivative 156 was obtained, and after N-alkylation with different phenacetyl bromides 157, the substituted derivative 158 was formed. A final ring-closing reaction in which 158 reacted with hydrazine hydrate and subsequently formed the target 159 after nucleophilic substitution (Scheme 47).
Scheme 47.
Reagents and conditions: (i) H2O, KHCO3, Br2 in aqueous solution of KBr, stirring, 30–40 min, 80 °C, 71%; (ii) K2CO3, stirring, 4–6 h, rt, 68–85%; (iii) NH2NH2·H2O 80%, MeOH, reflux, 8–9 h, 62–76%.
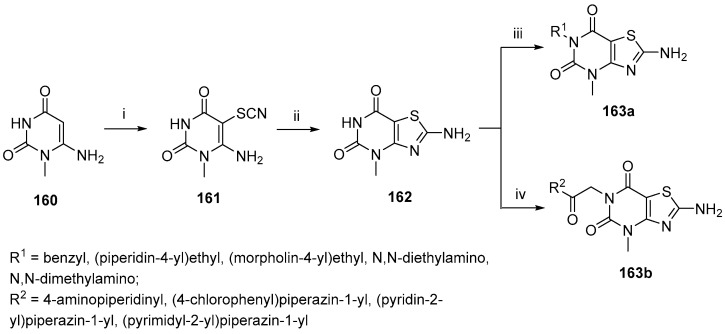
Another heterocyclic fragment that can be found in DPP-IV inhibitors is the thiazolopyrimidine ring system [127]. Sharma et al. reported the synthesis, in silico, and in vitro evaluation of thiazolopyrimidine derivatives as DPP-IV inhibitors, as well as their structure–activity relationship (Scheme 48) [128]. The core heterocyclic structure was formed in two steps, beginning with the 6-amino pyrimidine derivative 160. First, a thiocyanate group was added on C-5, and a cyclization reaction of 161 to give 2-amino-4-methyl-4H-thiazolo [4,5-d]pyrimidine-5,7-dione 162. The target compounds 163a–b were prepared via N-alkylation of 162 with alkyl halides of N-substituted chloracetamides.
Scheme 48.
Reagents and conditions: (i) Br2/KSCN, DMF, 83%; (ii) DMF, 80–120 °C, 89%; (iii) R1Cl, anhydrous K2CO3, DMF, 70 °C, 74–78% (iv) N-substituted chloracetamides, anhydrous K2CO3, DMF, 70–80 °C, 64–68%.
4.3. Bicyclic 6-6 Systems
An important example of a bicyclic system of two fused six-membered heterocycles that has found application in DPP-IV inhibitors is the pyridopyrimidine ring system. It was first described by Betty Lam et al. as a potent and selective DPP-IV inhibitor in 2012 [129]. The synthesis of the compounds started with a Knoevenagel condensation of substituted aromatic aldehydes with malonodinitrile to generate the derivatives 164, which, upon reaction with aminopyrimidine 165, led to the formation of the core heterocycle 166 via a Michael addition reaction (Scheme 49). BH3-mediated reduction of the nitrile group resulted in the formation of 167, while compounds 168 resulted from a three-step synthetic procedure.
Scheme 49.
Reagents and conditions: (i) malonodinitrile, ethanol, 10% aq KOH, 95%; (ii) propanol, 120 °C, 43%; (iii) BH3-THF, THF, 65 °C, 46%.
Fang et al. conducted research on novel tetrahydropyridopyrimidine derivatives that exhibit both GPR119 and DPP-IV-inhibitory activity [130]. The authors synthesized two series of compounds—based either on 4,6-disubstituted tetrahydropyrido[4,3-d]pyrimidine or on 4,7-disubstituted tetrahydropyrido[3,4-d]pyrimidine scaffold. The synthetic methodology for the 4,6-disubstituted tetrahydropyrido[4,3-d]pyrimidine derivatives is outlined in Scheme 50, as the synthesis of both series follows similar steps. The procedure starts with commercially available 1-benzyl-3-carbethoxy-4-piperidone 169, which cyclizes with formamide, followed by chlorination with POCl3 to give 170 [131]. The substitution of chlorine with 2-fluoro-4-cyanoaniline or 2-fluoro-4-methylsulfonyl aniline gives 171. Deprotection of the benzyl group in 171, followed by N-alkylation, afforded the final 172.
Scheme 50.
Reagents and conditions: (i) formamide, NaOEt, EtOH, reflux; (ii) POCl3, reflux; (iii) 4-amino-3-fluorobenzonitrile, NaH, THF, reflux, overnight, 35% or 2-fluoro-4-(methylsulfonyl)aniline, X-Phos, Pd2(dba)3, Cs2CO3, dioxane, reflux, overnight, 83%; (iv) 2-chloroethyl chloroformate, 1,2-dichloroethane, reflux, overnight, then MeOH, reflux, 3 h, 76%; (v) R2Cl, K2CO3, DMF, 70 °C, overnight, 64–94%.
4.4. Tricyclic Systems
An important example of the use of a tricyclic system in DPP-IV inhibitors is the research by Emondson et al. on piperidine-fused benzimidazoles and imidazopyridines, which are described as potent inhibitors in nanomolar concentrations [132]. The synthetic route starts with the cyclic lactam 173, which is protected with 4,4′-dimethoxybenzhydrol and then converted to 174 via ester hydrolysis and Curtius rearrangement (Scheme 51). Exchange of Cbz with the Boc-protecting group and deprotection of lactam nitrogen gave 175, which was used in the key ring-forming reaction with various aromatic halogen-substituted amines 176a–b to furnish the target inhibitors 177a–b.
Scheme 51.
Reagents and conditions: (i) 4,4′-dimethoxybenzhydrol, H2SO4, HOAc, 47%; (ii) LiOH(aq), THF, 80%; (iii) DPPA, TEA, toluene, BnOH, reflux, 65%; (iv) Pd(OH)2, H2, MeOH, Boc2O, 73%; (v) ceric ammonium nitrate, CH3CN, H2O, 0 °C, 75%; (vi) CuI, MeNHCH2CH2NHMe, K2CO3, toluene, reflux; (vii) TFA, CH2Cl2, 33–80% over two steps.
Boehringer et al. demonstrated the promising DPP-IV-inhibitory activity of benzo[a]quinolizidine derivatives with IC50 values in the nanomolar range [133]. The authors employed a four-step synthetic route starting with the 3,4-dihydroisoquinoline derivative 178, which reacted with methyl vinyl ketone to form the key intermediate 179 based on a previously reported procedure [134]. The ketone 179 underwent palladium-catalyzed arylation to give 180 as a single stereoisomer. The final step was the conversion of the carbonyl group to oxime, followed by reduction to give 181, bearing an amino group at C-2 (Scheme 52). Further research by the authors led to the discovery of Carmegliptin, which is a highly potent DPP-IV inhibitor [135]. Other authors have also investigated benzo[a]quinolizidine compounds and their heterocyclic analogues as prospective DPP-IV inhibitors [136,137].
Scheme 52.
Reagents and conditions: (i) water bath, 1 h, then 5% Na2CO3; (ii) Pd(OAc)2, PtBu3, t-BuONa, THF, 21–51%; (iii) NH2OH.HCl, NaOAc, EtOH, 83–99%; (iv) H2, Raney Ni, aq NH3, MeOH, THF, 34–85%.
The work of Wu et al. provides another example of potent tricyclic DPP-IV inhibitors [138]. The authors constructed novel tricyclic guanine derivatives starting with 2,6-dichloropurine 182 using selective hydrolysis to obtain 183. Alkylation gave 184, which was then reacted with methylamine or ethylamine, followed by refluxing with 6 N HCl to give the target heterocycle 185. The obtained 185 was further used for the preparation of the prospective inhibitors 186 (Scheme 53).
Scheme 53.
Reagents and conditions: (i) NaOH, 90%; (ii) BrCH2CN, N,N-Diisopropylethylamine, DMF, 55%; (iii) MeNH2 or EtNH2, 1,4-dioxane, rt; (iv) 6 N HCl, reflux, 65%.
5. Conclusions
This review is focused on heterocyclic scaffolds that have been the subject of research by several different research groups. As can be seen, there is a great structural diversity of heterocycles that could be found in DPP-IV inhibitors. However, the search for novel compounds with improved potency and selectivity is an ongoing challenge in contemporary medicinal chemistry. The development of DPP-IV inhibitors is a multidisciplinary field of research, supported by various in silico drug design approaches, innovative synthetic methodologies, and in vitro methods to assess bioactivity and toxicity.
Acknowledgments
We thank the Medical University—Pleven governing body, as well as the governing body of project No. BG-RRP-2.004-0003, for their continuing support.
Author Contributions
Conceptualization, T.A. and A.P.; software, A.P.; formal analysis, V.P.; investigation, V.P.; data curation, T.A.; writing—original draft preparation, V.P.; writing—review and editing, T.A. and A.P.; visualization, A.P.; supervision, A.P.; project administration, A.P.; funding acquisition, A.P. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding Statement
This study is financed by the European Union-NextGenerationEU through the National Recovery and Resilience Plan of the Republic of Bulgaria, project No. BG-RRP-2.004-0003.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Ahrén B., Simonsson E., Larsson H., Landin-Olsson M., Torgeirsson H., Jansson P.-A., Sandqvist M., Båvenholm P., Efendic S., Eriksson J.W., et al. Inhibition of Dipeptidyl Peptidase IV Improves Metabolic Control Over a 4-Week Study Period in Type 2 Diabetes. Diabetes Care. 2002;25:869–875. doi: 10.2337/diacare.25.5.869. [DOI] [PubMed] [Google Scholar]
- 2.Augustyns K., Veken P.V.d., Senten K., Haemers A. The Therapeutic Potential of Inhibitors of Dipeptidyl Peptidase IV (DPP IV) and Related Proline-Specific Dipeptidyl Aminopeptidases. Curr. Med. Chem. 2005;12:971–998. doi: 10.2174/0929867053507298. [DOI] [PubMed] [Google Scholar]
- 3.Gorrell M.D. Dipeptidyl Peptidase IV and Related Enzymes in Cell Biology and Liver Disorders. Clin. Sci. 2005;108:277–292. doi: 10.1042/CS20040302. [DOI] [PubMed] [Google Scholar]
- 4.Baggio L.L., Drucker D.J. Biology of Incretins: GLP-1 and GIP. Gastroenterology. 2007;132:2131–2157. doi: 10.1053/j.gastro.2007.03.054. [DOI] [PubMed] [Google Scholar]
- 5.Penaforte-Saboia J.G., Couri C.E.B., Albuquerque N.V., Silva V.L.L., Olegario N.B.D.C., Fernandes V.O., Montenegro Junior R.M. Emerging Roles of Dipeptidyl Peptidase-4 Inhibitors in Delaying the Progression of Type 1 Diabetes Mellitus. Diabetes Metab. Syndr. Obes. 2021;14:565–573. doi: 10.2147/DMSO.S294742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Deacon C.F. Dipeptidyl Peptidase 4 Inhibitors in The Treatment of Type 2 Diabetes Mellitus. Nat. Rev. Endocrinol. 2020;16:642–653. doi: 10.1038/s41574-020-0399-8. [DOI] [PubMed] [Google Scholar]
- 7.Kerru N., Singh-Pillay A., Awolade P., Singh P. Current Anti-Diabetic Agents and Their Molecular Targets: A Review. Eur. J. Med. Chem. 2018;152:436–488. doi: 10.1016/j.ejmech.2018.04.061. [DOI] [PubMed] [Google Scholar]
- 8.Nar H., Schnapp G., Hucke O., Hardman T.C., Klein T. Action of Dipeptidyl Peptidase-4 Inhibitors on SARS-CoV-2 Main Protease. ChemMedChem. 2021;16:1425–1426. doi: 10.1002/cmdc.202000921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Solerte S.B., Di Sabatino A., Galli M., Fiorina P. Dipeptidyl Peptidase-4 (DPP4) Inhibition in COVID-19. Acta Diabetol. 2020;57:779–783. doi: 10.1007/s00592-020-01539-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Strollo R., Pozzilli P. DPP4 inhibition: Preventing SARS-CoV-2 infection and/or progression of COVID-19? Diabetes Metab. Res. Rev. 2020;36:e3330. doi: 10.1002/dmrr.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zein A.F.M.Z., Raffaello W.M. Dipeptidyl Peptidase-4 (DPP-IV) Inhibitor was Associated with Mortality Reduction in COVID-19—A Systematic Review and Meta-Analysis. Prim. Care Diabetes. 2022;16:162–167. doi: 10.1016/j.pcd.2021.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nag S., Mandal S., Mukherjee O., Mukherjee S., Kundu R. DPP-4 Inhibitors as a Savior for COVID-19 Patients with Diabetes. Future Virol. 2023;18:321–333. doi: 10.2217/fvl-2022-0112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Busek P., Duke-Cohan J.S., Sedo A. Does DPP-IV Inhibition Offer New Avenues for Therapeutic Intervention in Malignant Disease? Cancers. 2022;14:2072. doi: 10.3390/cancers14092072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sourav D., Subhasis B., Kumar S.K.A., Priyankar P. Critical Role of Dipeptidyl Peptidase IV: A Therapeutic Target for Diabetes and Cancer. Mini-Rev. Med. Chem. 2019;19:88–97. doi: 10.2174/1389557518666180423112154. [DOI] [PubMed] [Google Scholar]
- 15.Kang S.M., Park J.H. Pleiotropic Benefits of DPP-4 Inhibitors Beyond Glycemic Control. Clin. Med. Insights Endocrinol. Diabetes. 2021;14:11795514211051698. doi: 10.1177/11795514211051698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ribeiro-Silva J.C., Marques V.B., dos Santos L. Effects of Dipeptidyl Peptidase 4 Inhibition on The Endothelial Control of The Vascular Tone. Am. J. Physiol. Cell Physiol. 2023;325:C972–C980. doi: 10.1152/ajpcell.00246.2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Akoumianakis I., Antoniades C. Dipeptidyl Peptidase IV Inhibitors as Novel Regulators of Vascular Disease. Vasc. Pharmacol. 2017;96–98:1–4. doi: 10.1016/j.vph.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 18.Reinhold D., Kähne T., Steinbrecher A., Wrenger S., Neubert K., Ansorge S., Brocke S. The Role of Dipeptidyl Peptidase IV (DP IV) Enzymatic Activity in T Cell Activation and Autoimmunity. Biol. Chem. 2002;383:1133–1138. doi: 10.1515/BC.2002.123. [DOI] [PubMed] [Google Scholar]
- 19.Al-Badri G., Leggio G.M., Musumeci G., Marzagalli R., Drago F., Castorina A. Tackling Dipeptidyl Peptidase IV in Neurological Disorders. Neural Regen. Res. 2018;13:26–34. doi: 10.4103/1673-5374.224365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Złotek M., Kurowska A., Herbet M., Piątkowska-Chmiel I. GLP-1 Analogs, SGLT-2, and DPP-4 Inhibitors: A Triad of Hope for Alzheimer’s Disease Therapy. Biomedicines. 2023;11:3035. doi: 10.3390/biomedicines11113035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Layman S.N., Elliott W.V., Neu D.W., Howard T.G., Hamby A. Alogliptin and Heart Failure Outcomes in Patients With Type 2 Diabetes. J. Pharm. Pract. 2024;37:410–414. doi: 10.1177/08971900221135656. [DOI] [PubMed] [Google Scholar]
- 22.Doggrell S.A., Dimmitt S.B. Gliptins-do they increase cardiovascular risk or benefit? Expert Opin. Drug Saf. 2014;13:675–680. doi: 10.1517/14740338.2014.904284. [DOI] [PubMed] [Google Scholar]
- 23.Bowes C.D., Lien L.F., Butler J. Assessment of Heart Failure in Diabetes Cardiovascular Outcomes Trials: Is What We Are Currently Capturing Adequate? Curr. Diabetes Rep. 2019;19:39. doi: 10.1007/s11892-019-1154-1. [DOI] [PubMed] [Google Scholar]
- 24.Vaduganathan M., Singh A., Sharma A., Januzzi J.L., Scirica B.M., Butler J., Zannad F., McGuire D.K., Cannon C.P., Bhatt D.L. Contemporary Trends in Prescription of Dipeptidyl Peptidase-4 Inhibitors in the Context of US Food and Drug Administration Warnings of Heart Failure Risk. Am. J. Cardiol. 2020;125:1577–1581. doi: 10.1016/j.amjcard.2020.01.053. [DOI] [PubMed] [Google Scholar]
- 25.Juillerat-Jeanneret L. Dipeptidyl Peptidase IV and Its Inhibitors: Therapeutics for Type 2 Diabetes and What Else? J. Med. Chem. 2014;57:2197–2212. doi: 10.1021/jm400658e. [DOI] [PubMed] [Google Scholar]
- 26.Kwok A.J., Mashar M., Khavandi K., Sabir I. DPP-IV Inhibitors: Beyond Glycaemic Control? Trends Cardiovas Med. 2014;24:157–164. doi: 10.1016/j.tcm.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 27.Deacon C.F. A Review of Dipeptidyl Peptidase-4 Inhibitors. Hot Topics from Randomized Controlled Trials. Diabetes Obes. Metab. 2018;20:34–46. doi: 10.1111/dom.13135. [DOI] [PubMed] [Google Scholar]
- 28.Lei Y., Hu L., Yang G., Piao L., Jin M., Cheng X. Dipeptidyl Peptidase-IV Inhibition for the Treatment of Cardiovascular Disease-Recent Insights Focusing on Angiogenesis and Neovascularization. Circ. J. 2017;81:770–776. doi: 10.1253/circj.CJ-16-1326. [DOI] [PubMed] [Google Scholar]
- 29.Ambhore J.P., Laddha P.R., Nandedkar A., Ajmire P.V., Chumbhale D.S., Navghare A.B., Kuchake V.G., Chaudhari P.J., Adhao V.S. Medicinal Chemistry of Non-Peptidomimetic Dipeptidyl Peptidase IV (DPP IV) Inhibitors for Treatment of Type-2 Diabetes Mellitus: Insights on Recent Development. J. Mol. Struct. 2023;1284:135249. doi: 10.1016/j.molstruc.2023.135249. [DOI] [Google Scholar]
- 30.Mulakayala N., Reddy Ch U., Iqbal J., Pal M. Synthesis of Dipeptidyl Peptidase-4 Inhibitors: A Brief Overview. Tetrahedron. 2010;66:4919–4938. doi: 10.1016/j.tet.2010.04.088. [DOI] [Google Scholar]
- 31.Jens-Uwe P. 11 Years of Cyanopyrrolidines as DPP-IV Inhibitors. Curr. Top Med. Chem. 2007;7:579–595. doi: 10.2174/156802607780091000. [DOI] [PubMed] [Google Scholar]
- 32.Del Prato S. Dipeptidyl Peptidase 4 Inhibition and Vildagliptin Therapy for Type 2 Diabetes. Int. J. Clin. Pract. 2007;61:38–48. doi: 10.1111/j.1742-1241.2007.01439.x. [DOI] [PubMed] [Google Scholar]
- 33.Augeri D.J., Robl J.A., Betebenner D.A., Magnin D.R., Khanna A., Robertson J.G., Wang A., Simpkins L.M., Taunk P., Huang Q., et al. Discovery and Preclinical Profile of Saxagliptin (BMS-477118): A Highly Potent, Long-Acting, Orally Active Dipeptidyl Peptidase IV Inhibitor for the Treatment of Type 2 Diabetes. J. Med. Chem. 2005;48:5025–5037. doi: 10.1021/jm050261p. [DOI] [PubMed] [Google Scholar]
- 34.Hulin B., Cabral S., Lopaze M.G., Van Volkenburg M.A., Andrews K.M., Parker J.C. New Fluorinated Pyrrolidine and Azetidine Amides as Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. Lett. 2005;15:4770–4773. doi: 10.1016/j.bmcl.2005.07.026. [DOI] [PubMed] [Google Scholar]
- 35.Kumar S., Mittal A., Mittal A. A Review Upon Medicinal Perspective and Designing Rationale of DPP-4 Inhibitors. Bioorg. Med. Chem. 2021;46:116354. doi: 10.1016/j.bmc.2021.116354. [DOI] [PubMed] [Google Scholar]
- 36.Ashworth D.M., Atrash B., Baker G.R., Baxter A.J., Jenkins P.D., Jones D.M., Szelke M. 2-Cyanopyrrolidides as Potent, Stable Inhibitors of Dipeptidyl Peptidase IV. Bioorg. Med. Chem. Lett. 1996;6:1163–1166. doi: 10.1016/0960-894X(96)00190-4. [DOI] [Google Scholar]
- 37.Villhauer E.B., Brinkman J.A., Naderi G.B., Burkey B.F., Dunning B.E., Prasad K., Mangold B.L., Russell M.E., Hughes T.E. 1-[[(3-Hydroxy-1-adamantyl)amino]acetyl]-2-cyano-(S)-pyrrolidine: A Potent, Selective, and Orally Bioavailable Dipeptidyl Peptidase IV Inhibitor with Antihyperglycemic Properties. J. Med. Chem. 2003;46:2774–2789. doi: 10.1021/jm030091l. [DOI] [PubMed] [Google Scholar]
- 38.Ji X., Su M., Wang J., Deng G., Deng S., Li Z., Tang C., Li J., Li J., Zhao L., et al. Design, Synthesis and Biological Evaluation of Hetero-Aromatic Moieties Substituted Pyrrole-2-Carbonitrile Derivatives as Dipeptidyl Peptidase IV Inhibitors. Eur. J. Med. Chem. 2014;75:111–122. doi: 10.1016/j.ejmech.2014.01.021. [DOI] [PubMed] [Google Scholar]
- 39.Jadav P., Bahekar R., Shah S.R., Patel D., Joharapurkar A., Kshirsagar S., Jain M., Shaikh M., Sairam K.V.V.M. Long-Acting Peptidomimetics Based DPP-IV Inhibitors. Bioorg. Med. Chem. Lett. 2012;22:3516–3521. doi: 10.1016/j.bmcl.2012.03.078. [DOI] [PubMed] [Google Scholar]
- 40.Fukushima H., Hiratate A., Takahashi M., Saito-Hori M., Munetomo E., Kitano K., Saito H., Takaoka Y., Yamamoto K. Synthesis and Structure-Activity Relationships of Potent 1-(2-Substituted-Aminoacetyl)-4-Fluoro-2-Cyanopyrrolidine Dipeptidyl Peptidase IV Inhibitors. Chem. Pharm. Bull. 2008;56:1110–1117. doi: 10.1248/cpb.56.1110. [DOI] [PubMed] [Google Scholar]
- 41.Middleton W.J. New Fluorinating Reagents. Dialkylaminosulfur Fluorides. J. Org. Chem. 1975;40:574–578. doi: 10.1021/jo00893a007. [DOI] [Google Scholar]
- 42.Demange L., Ménez A., Dugave C. Practical Synthesis of Boc and Fmoc Protected 4-Fluoro and 4-Difluoroprolines from Trans-4-Hydroxyproline. Tetrahedron Lett. 1998;39:1169–1172. doi: 10.1016/S0040-4039(97)10793-6. [DOI] [Google Scholar]
- 43.Fukushima H., Hiratate A., Takahashi M., Saito M., Munetomo E., Kitano K., Saito H., Takaoka Y., Yamamoto K. Synthesis and Structure–Activity Relationships of Potent 3- or 4-Substituted-2-Cyanopyrrolidine Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. 2004;12:6053–6061. doi: 10.1016/j.bmc.2004.09.010. [DOI] [PubMed] [Google Scholar]
- 44.Tsai T.-Y., Hsu T., Chen C.-T., Cheng J.-H., Chiou M.-C., Huang C.-H., Tseng Y.-J., Yeh T.-K., Huang C.-Y., Yeh K.-C., et al. Rational Design and Synthesis of Potent and Long-Lasting Glutamic Acid-Based Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. Lett. 2009;19:1908–1912. doi: 10.1016/j.bmcl.2009.02.061. [DOI] [PubMed] [Google Scholar]
- 45.Lu I.L., Lee S.-J., Tsu H., Wu S.-Y., Kao K.-H., Chien C.-H., Chang Y.-Y., Chen Y.-S., Cheng J.-H., Chang C.-N., et al. Glutamic Acid Analogues as Potent Dipeptidyl Peptidase IV and 8 Inhibitors. Bioorg. Med. Chem. Lett. 2005;15:3271–3275. doi: 10.1016/j.bmcl.2005.04.051. [DOI] [PubMed] [Google Scholar]
- 46.Sakashita H., Akahoshi F., Yoshida T., Kitajima H., Hayashi Y., Ishii S., Takashina Y., Tsutsumiuchi R., Ono S. Lead Optimization of [(S)-γ-(arylamino)prolyl]thiazolidine Focused on γ-Substituent: Indoline Compounds As Potent DPP-IV Inhibitors. Bioorg. Med. Chem. 2007;15:641–655. doi: 10.1016/j.bmc.2006.10.059. [DOI] [PubMed] [Google Scholar]
- 47.Xiao P.-F., Guo R., Huang S.-Q., Cui H.-J., Ye S., Zhang Z. Discovery of Dipeptidyl Peptidase IV (DPP4) Inhibitors Based on a Novel Indole Scaffold. Chin. Chem. Lett. 2014;25:673–676. doi: 10.1016/j.cclet.2014.03.047. [DOI] [Google Scholar]
- 48.Van Goethem S., Van der Veken P., Dubois V., Soroka A., Lambeir A.-M., Chen X., Haemers A., Scharpé S., De Meester I., Augustyns K. Inhibitors of Dipeptidyl Peptidase 8 and Dipeptidyl Peptidase 9. Part 2: Isoindoline Containing Inhibitors. Bioorg. Med. Chem. Lett. 2008;18:4159–4162. doi: 10.1016/j.bmcl.2008.05.079. [DOI] [PubMed] [Google Scholar]
- 49.Van Goethem S., Matheeussen V., Joossens J., Lambeir A.-M., Chen X., De Meester I., Haemers A., Augustyns K., Van der Veken P. Structure–Activity Relationship Studies on Isoindoline Inhibitors of Dipeptidyl Peptidases 8 and 9 (DPP8, DPP9): Is DPP8-Selectivity an Attainable Goal? J. Med. Chem. 2011;54:5737–5746. doi: 10.1021/jm200383j. [DOI] [PubMed] [Google Scholar]
- 50.Yoshida T., Akahoshi F., Sakashita H., Kitajima H., Nakamura M., Sonda S., Takeuchi M., Tanaka Y., Ueda N., Sekiguchi S., et al. Discovery and Preclinical Profile of Teneligliptin (3-[(2S,4S)-4-[4-(3-methyl-1-phenyl-1H-pyrazol-5-yl)piperazin-1-yl]pyrrolidin-2-ylcarbonyl]thiazolidine): A Highly Potent, Selective, Long-Lasting and Orally Active Dipeptidyl Peptidase IV Inhibitor for the Treatment of Type 2 Diabetes. Bioorg. Med. Chem. 2012;20:5705–5719. doi: 10.1016/j.bmc.2012.08.012. [DOI] [PubMed] [Google Scholar]
- 51.Singhal S., Manikrao Patil V., Verma S., Masand N. Recent Advances and Structure-Activity Relationship Studies Of DPP-4 Inhibitors as Anti-Diabetic Agents. Bioorg. Chem. 2024;146:107277. doi: 10.1016/j.bioorg.2024.107277. [DOI] [PubMed] [Google Scholar]
- 52.Sever B., Soybir H., Görgülü Ş., Cantürk Z., Altıntop M.D. Pyrazole Incorporated New Thiosemicarbazones: Design, Synthesis and Investigation of DPP-4 Inhibitory Effects. Molecules. 2020;25:5003. doi: 10.3390/molecules25215003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Altıntop M.D., Temel H.E., Sever B., Akalın Çiftçi G., Kaplancıklı Z.A. Synthesis and Evaluation of New Benzodioxole- Based Thiosemicarbazone Derivatives as Potential Antitumor Agents. Molecules. 2016;21:1598. doi: 10.3390/molecules21111598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Nidhar M., Kumar V., Mahapatra A., Gupta P., Yadav P., Sonker P., Kumar A., Mishra S., Singh R.K., Tewari A.K. Lead Modification via Computational Studies: Synthesis of Pyrazole-Containing Β-Amino Carbonyls for The Treatment of Type 2 Diabetes. Chem. Biol. Drug Des. 2023;101:638–649. doi: 10.1111/cbdd.14157. [DOI] [PubMed] [Google Scholar]
- 55.Nidhar M., Sonker P., Sharma V.P., Kumar S., Tewari A.K. Design, Synthesis and In-Silico & In Vitro Enzymatic Inhibition Assays of Pyrazole-Chalcone Derivatives as Dual Inhibitors of α-Amylase & DPP-4 Enzyme. Chem. Pap. 2022;76:1707–1720. doi: 10.1007/s11696-021-01985-1. [DOI] [Google Scholar]
- 56.Nidhar M., Khanam S., Sonker P., Gupta P., Mahapatra A., Patil S., Yadav B.K., Singh R.K., Kumar Tewari A. Click Inspired Novel Pyrazole-Triazole-Persulfonimide & Pyrazole-Triazole-Aryl Derivatives; Design, Synthesis, DPP-4 Inhibitor with Potential Anti-Diabetic Agents. Bioorg. Chem. 2022;120:105586. doi: 10.1016/j.bioorg.2021.105586. [DOI] [PubMed] [Google Scholar]
- 57.Temel H.E., Altıntop M.D., Sever B., Özdemir A., Çiftçi G.A. In Vitro Evaluation of 2-Pyrazoline Derivatives as DPP-4 Inhibitors. Turk. J. Biochem. 2023;48:104–109. doi: 10.1515/tjb-2022-0161. [DOI] [Google Scholar]
- 58.Sharma S., Srivastav S., Singh G., Singh S., Malik R., Alam M.M., Shaqiquzamman M., Ali S., Akhter M. In Silico Strategies for Probing Novel DPP-IV Inhibitors as Anti-Diabetic Agents. J. Biomol. Struct. Dyn. 2021;39:2118–2132. doi: 10.1080/07391102.2020.1751714. [DOI] [PubMed] [Google Scholar]
- 59.Elguero J., Silva A.M.S., Tomé A.C. Five-Membered Heterocycles: 1,2-Azoles. Part 1. Pyrazoles. In: Alvarez-Builla J., Vaquero J.J., Barluenga J., editors. Modern Heterocyclic Chemistry. Wiley-VCH Verlag GmbH & Co. KGaA; Weinheim, Germany: 2011. pp. 635–725. [Google Scholar]
- 60.Hoz A.d.l., Alkorta I., Elguero J. The mechanism of the reaction of hydrazines with α,β-unsaturated carbonyl compounds to afford hydrazones and 2-pyrazolines (4,5-dihydro-1H-pyrazoles): Experimental and theoretical results. Tetrahedron. 2021;97:132413. doi: 10.1016/j.tet.2021.132413. [DOI] [Google Scholar]
- 61.Cheon H.G., Kim S.-S., Kim K.-R., Rhee S.-D., Yang S.-D., Ahn J.H., Park S.-D., Lee J.M., Jung W.H., Lee H.S., et al. Inhibition of Dipeptidyl Peptidase IV by Novel Inhibitors with Pyrazolidine Scaffold. Biochem. Pharmacol. 2005;70:22–29. doi: 10.1016/j.bcp.2005.04.004. [DOI] [PubMed] [Google Scholar]
- 62.Ahn J.H., Kim H.-M., Jung S.H., Kang S.K., Kim K.R., Rhee S.D., Yang S.-D., Cheon H.G., Kim S.S. Synthesis and DPP-IV Inhibition of Cyano-Pyrazoline Derivatives as Potent Anti-Diabetic Agents. Bioorg. Med. Chem. Lett. 2004;14:4461–4465. doi: 10.1016/j.bmcl.2004.06.046. [DOI] [PubMed] [Google Scholar]
- 63.Kumar A., Maurya R.A., Sharma S., Ahmad P., Singh A.B., Tamrakar A.K., Srivastava A.K. Design and Synthesis of 3,5-Diarylisoxazole Derivatives as Novel Class of Anti-Hyperglycemic and Lipid Lowering Agents. Bioorg. Med. Chem. 2009;17:5285–5292. doi: 10.1016/j.bmc.2009.05.033. [DOI] [PubMed] [Google Scholar]
- 64.Silva A.M.S., Tomé A.C., Pinho e Melo T.M.V.D., Elguero J. Five-Membered Heterocycles: 1,2-Azoles. Part 2. Isoxazoles and Isothiazoles. In: Alvarez-Builla J., Vaquero J.J., Barluenga J., editors. Modern Heterocyclic Chemistry. Wiley-VCH Verlag GmbH & Co. KGaA; Weinheim, Germany: 2011. pp. 727–808. [Google Scholar]
- 65.Karandikar S., Soni R., Soman S.S., Umar S., Suresh B. 1,2-Benzisoxazole-3-Acetamide Derivatives as Dual Agents For DPP-IV Inhibition and Anticancer Activity. Synth. Commun. 2018;48:2877–2887. doi: 10.1080/00397911.2018.1508723. [DOI] [Google Scholar]
- 66.Wallace M.B., Feng J., Zhang Z., Skene R.J., Shi L., Caster C.L., Kassel D.B., Xu R., Gwaltney S.L. Structure-Based Design and Synthesis of Benzimidazole Derivatives as Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. Lett. 2008;18:2362–2367. doi: 10.1016/j.bmcl.2008.02.071. [DOI] [PubMed] [Google Scholar]
- 67.Sreeja S., Arul G.D.A.S., Mathan S. Design, Synthesis and Biological Evaluation of 2-Aminobenzimidazole Derivatives as DPP4 Inhibitors. Curr. Bioact. Compd. 2020;16:696–702. doi: 10.2174/1573407215666190318121902. [DOI] [Google Scholar]
- 68.Tomovic K., Ilic B.S., Smelcerovic Z., Miljkovic M., Yancheva D., Kojic M., Mavrova A.T., Kocic G., Smelcerovic A. Benzimidazole-Based Dual Dipeptidyl Peptidase-4 and Xanthine Oxidase Inhibitors. Chem. Biol. Interact. 2020;315:108873. doi: 10.1016/j.cbi.2019.108873. [DOI] [PubMed] [Google Scholar]
- 69.Gundeti S., Lee J., Park H. Novel 1,2,3-Triazole Analogs of Sitagliptin as DPP4 Inhibitors. B Korean Chem. Soc. 2016;37:1156–1158. doi: 10.1002/bkcs.10828. [DOI] [Google Scholar]
- 70.Vo D.-V., Hong K.H., Lee J., Park H. Synthesis, In Vitro Evaluation, and Computational Simulations Studies of 1,2,3-Triazole Analogues as DPP-4 Inhibitors. Bioorg. Med. Chem. 2021;29:115861. doi: 10.1016/j.bmc.2020.115861. [DOI] [PubMed] [Google Scholar]
- 71.Vo D.-V., Lee J., Park H. 1,2,3-Triazole Analogs with Bulky and Conformationally Rigid Substructures: Synthesis and In Vitro Evaluation as DPP-4 Inhibitors. B Korean. Chem. Soc. 2023;44:425–428. doi: 10.1002/bkcs.12677. [DOI] [Google Scholar]
- 72.Fuh M.-T., Tseng C.-C., Li S.-M., Tsai S.-E., Chuang T.-J., Lu C.-H., Yang Y.-C., Tsai H.J., Wong F.F. Design, Synthesis and Biological Evaluation of Glycolamide, Glycinamide, and β-Amino carbonyl 1,2,4-Triazole Derivatives as DPP-4 Inhibitors. Bioorg. Chem. 2021;114:105049. doi: 10.1016/j.bioorg.2021.105049. [DOI] [PubMed] [Google Scholar]
- 73.Tseng W.-C., Wang L.-Y., Wu T.-S., Wong F.F. ‘One-Flask’ Synthesis to 3,5-Disubstituted 1,2,4-Triazoles from Aldehydes with Hydrazonoyl Hydrochlorides via 1,3-Dipolar Cycloaddition. Tetrahedron. 2011;67:5339–5345. doi: 10.1016/j.tet.2011.05.003. [DOI] [Google Scholar]
- 74.Wang L.-Y., Tsai H.J., Lin H.-Y., Kaneko K., Cheng F.-Y., Shih H.-S., Wong F.F., Huang J.-J. One-Flask Synthesis of 1,3,5-Trisubstituted 1,2,4-Triazoles from Nitriles and Hydrazonoyl Chlorides via 1,3-Dipolar Cycloaddition. RSC Adv. 2014;4:14215–14220. doi: 10.1039/C4RA00113C. [DOI] [Google Scholar]
- 75.Tsai S.-E., Chiang K.-H., Tseng C.-C., Chen N.-W., Chern C.-Y., Wong F.F. Facile One-Pot Synthesis of Methyl 1-Aryl-1H-1,2,4-triazole-3-carboxylates from Nitrilimines with Vilsmeier Reagent. Eur. J. Med. Chem. 2019;2019:1754–1762. doi: 10.1002/ejoc.201801808. [DOI] [Google Scholar]
- 76.Dastjerdi H.F., Naderi N., Nematpour M., Rezaee E., Mahboubi-Rabbani M., Ebrahimi M., Hosseinipoor S., Hosseini O., Tabatabai S.A. Design, Synthesis and Anti-Diabetic Activity of Novel 1,2,3-Triazole-5-Carboximidamide Derivatives as Dipeptidyl Peptidase-4 Inhibitors. J. Mol. Struc. 2020;1221:128745. doi: 10.1016/j.molstruc.2020.128745. [DOI] [Google Scholar]
- 77.Xu J., Wei L., Mathvink R.J., Edmondson S.D., Eiermann G.J., He H., Leone J.F., Leiting B., Lyons K.A., Marsilio F., et al. Discovery of Potent, Selective, and Orally Bioavailable Oxadiazole-Based Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. Lett. 2006;16:5373–5377. doi: 10.1016/j.bmcl.2006.07.061. [DOI] [PubMed] [Google Scholar]
- 78.Nordhoff S., Bulat S., Cerezo-Gálvez S., Hill O., Hoffmann-Enger B., López-Canet M., Rosenbaum C., Rummey C., Thiemann M., Matassa V.G., et al. The Design of Potent and Selective Inhibitors Of DPP-4: Optimization of ADME Properties by Amide Replacements. Bioorg. Med. Chem. Lett. 2009;19:6340–6345. doi: 10.1016/j.bmcl.2009.09.078. [DOI] [PubMed] [Google Scholar]
- 79.Kaczanowska K., Wiesmüller K.-H., Schaffner A.-P. Design, Synthesis, and in Vitro Evaluation of Novel Aminomethyl-pyridines as DPP-4 Inhibitors. Med. Chem. Lett. 2010;1:530–535. doi: 10.1021/ml100200c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kaczanowska K., Eickhoff H., Albert K., Wiesmüller K.-H., Schaffner A.-P. A Simple, Diversity Oriented Synthesis of Highly Substituted Pyridines. J. Heterocycl. Chem. 2011;48:792–798. doi: 10.1002/jhet.614. [DOI] [Google Scholar]
- 81.Kumar A., Sharma S., Tripathi V.D., Maurya R.A., Srivastava S.P., Bhatia G., Tamrakar A.K., Srivastava A.K. Design and Synthesis of 2,4-Disubstituted Polyhydroquinolines as Prospective Antihyperglycemic And Lipid Modulating Agents. Bioorg. Med. Chem. 2010;18:4138–4148. doi: 10.1016/j.bmc.2009.11.061. [DOI] [PubMed] [Google Scholar]
- 82.Sogawa S., Nihro Y., Ueda H., Izumi A., Miki T., Matsumoto H., Satoh T. 3,4-Dihydroxychalcones as Potent 5-Lipoxygenase and Cyclooxygenase Inhibitors. J. Med. Chem. 1993;36:3904–3909. doi: 10.1021/jm00076a019. [DOI] [PubMed] [Google Scholar]
- 83.El Shehry M.F., Ghorab M.M., Abbas S.Y., Fayed E.A., Shedid S.A., Ammar Y.A. Quinoline Derivatives Bearing Pyrazole Moiety: Synthesis and Biological Evaluation as Possible Antibacterial and Antifungal Agents. Eur. J. Med. Chem. 2018;143:1463–1473. doi: 10.1016/j.ejmech.2017.10.046. [DOI] [PubMed] [Google Scholar]
- 84.Soni R., Durgapal S.D., Soman S.S., Georrge J.J. Design, Synthesis and Anti-Diabetic Activity of Chromen-2-One Derivatives. Arab. J. Chem. 2019;12:701–708. doi: 10.1016/j.arabjc.2016.11.011. [DOI] [Google Scholar]
- 85.Shah D.N., Shah N.M. The Fries Isomerization of Acetyl and Benzoyl Esters of Umbelliferones. J. Org. Chem. 1954;19:1681–1685. doi: 10.1021/jo01375a018. [DOI] [Google Scholar]
- 86.Bigi F., Chesini L., Maggi R., Sartori G. Montmorillonite KSF as an Inorganic, Water Stable, and Reusable Catalyst for the Knoevenagel Synthesis of Coumarin-3-carboxylic Acids. J. Org. Chem. 1999;64:1033–1035. doi: 10.1021/jo981794r. [DOI] [PubMed] [Google Scholar]
- 87.Soni R., Soman S.S. Design and Synthesis of Aminocoumarin Derivatives As DPP-IV Inhibitors and Anticancer Agents. Bioorg. Chem. 2018;79:277–284. doi: 10.1016/j.bioorg.2018.05.008. [DOI] [PubMed] [Google Scholar]
- 88.Dakin H.D. The Condensation of Aromatic Aldehydes with Glycine and Acetylglycine. J. Biol. Chem. 1929;82:439–446. doi: 10.1016/S0021-9258(20)78291-8. [DOI] [Google Scholar]
- 89.Durgapal S.D., Soman S.S. Evaluation of Novel Coumarin-Proline Sulfonamide Hybrids as Anticancer and Antidiabetic Agents. Synth. Commun. 2019;49:2869–2883. doi: 10.1080/00397911.2019.1647439. [DOI] [Google Scholar]
- 90.Jasim S.F., Mustafa Y.F. Synthesis and Antidiabetic Assessment of New Coumarin-Disubstituted Benzene Conjugates: An In Silico–In Virto Study. J. Med. Chem. Sci. 2022;5:887–899. doi: 10.26655/jmchemsci.2022.6.3. [DOI] [Google Scholar]
- 91.Waheed S.A., Mustafa Y.F. The In vitro Effects of New Albocarbon-based Coumarins on Blood Glucose-controlling Enzymes. J. Med. Chem. Sci. 2022;5:954–967. doi: 10.26655/jmchemsci.2022.6.9. [DOI] [Google Scholar]
- 92.Brockunier L.L., He J., Colwell L.F., Habulihaz B., He H., Leiting B., Lyons K.A., Marsilio F., Patel R.A., Teffera Y., et al. Substituted Piperazines as Novel Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. Lett. 2004;14:4763–4766. doi: 10.1016/j.bmcl.2004.06.065. [DOI] [PubMed] [Google Scholar]
- 93.Kushwaha R.N., Srivastava R., Mishra A., Rawat A.K., Srivastava A.K., Haq W., Katti S.B. Design, Synthesis, Biological Screening, and Molecular Docking Studies of Piperazine-Derived Constrained Inhibitors of DPP-IV for the Treatment of Type 2 Diabetes. Chem. Biol. Drug Des. 2015;85:439–446. doi: 10.1111/cbdd.12426. [DOI] [PubMed] [Google Scholar]
- 94.Khalaf R.A., Alwarafi E., Sabbah D. Piperazine Sulfonamides as DPP-IV Inhibitors: Synthesis, Induced-Fit Docking and Biological Evaluation. Acta Pharm. 2021;71:631–643. doi: 10.2478/acph-2021-0034. [DOI] [PubMed] [Google Scholar]
- 95.Nidhar M., Kumar V., Mahapatra A., Gupta P., Yadav B.K., Singh R.K., Tewari A.K. Ligand-Based Designing of DPP-4 Inhibitors via Hybridization; Synthesis, Docking, and Biological Evaluation of Pyridazine-Acetohydrazides. Mol. Divers. 2023;27:2729–2740. doi: 10.1007/s11030-022-10577-4. [DOI] [PubMed] [Google Scholar]
- 96.Guo Y.-Y., Zhang J.-Y., Sun J.-F., Gao H. A Comprehensive Review of Small-Molecule Drugs for the Treatment of Type 2 Diabetes Mellitus: Synthetic Approaches and Clinical Applications. Eur. J. Med. Chem. 2024;267:116185. doi: 10.1016/j.ejmech.2024.116185. [DOI] [PubMed] [Google Scholar]
- 97.Ali Z., Akhtar M.J., Haider M.R., Khan A.A., Siddiqui A.A., Yar M.S. Design and Synthesis of Quinazoline-3,4-(4H)-Diamine Endowed with Thiazoline Moiety as New Class For DPP-4 and DPPH Inhibitor. Bioorg. Chem. 2017;71:181–191. doi: 10.1016/j.bioorg.2017.02.004. [DOI] [PubMed] [Google Scholar]
- 98.Ali Z., Akhtar M.J., Siddiqui A.A., Khan A.A., Haider M.R., Yar M.S. Design, Synthesis, and Biological Evaluation of Novel Quinazoline Clubbed Thiazoline Derivatives. Arch. Pharm. 2017;350:1600298. doi: 10.1002/ardp.201600298. [DOI] [PubMed] [Google Scholar]
- 99.Zayed M.F., Hassan M.H. Synthesis and Biological Evaluation Studies of Novel Quinazolinone Derivatives as Antibacterial and Anti-Inflammatory Agents. Saudi Pharm. J. 2014;22:157–162. doi: 10.1016/j.jsps.2013.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Syam Y.M., Anwar M.M., Abd El-Karim S.S., Elseginy S.A., Essa B.M., Sakr T.M. New Quinoxaline Compounds as DPP-4 Inhibitors and Hypoglycemics: Design, Synthesis, Computational and Bio-Distribution Studies. RSC Adv. 2021;11:36989–37010. doi: 10.1039/D1RA06799K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Liu K.K.C., Sakya S.M., O’Donnell C.J., Flick A.C., Ding H.X. Synthetic Approaches to the 2010 New Drugs. Bioorg. Med. Chem. 2012;20:1155–1174. doi: 10.1016/j.bmc.2011.12.049. [DOI] [PubMed] [Google Scholar]
- 102.Feng J., Zhang Z., Wallace M.B., Stafford J.A., Kaldor S.W., Kassel D.B., Navre M., Shi L., Skene R.J., Asakawa T., et al. Discovery of Alogliptin: A Potent, Selective, Bioavailable, and Efficacious Inhibitor of Dipeptidyl Peptidase IV. J. Med. Chem. 2007;50:2297–2300. doi: 10.1021/jm070104l. [DOI] [PubMed] [Google Scholar]
- 103.Zhang Z., Wallace M.B., Feng J., Stafford J.A., Skene R.J., Shi L., Lee B., Aertgeerts K., Jennings A., Xu R., et al. Design and Synthesis of Pyrimidinone and Pyrimidinedione Inhibitors of Dipeptidyl Peptidase IV. J. Med. Chem. 2011;54:510–524. doi: 10.1021/jm101016w. [DOI] [PubMed] [Google Scholar]
- 104.Li N., Wang L.-J., Jiang B., Guo S.-J., Li X.-Q., Chen X.-C., Luo J., Li C., Wang Y., Shi D.-Y. Design, Synthesis and Biological Evaluation of Novel Pyrimidinedione Derivatives as DPP-4 Inhibitors. Bioorg. Med. Chem. Lett. 2018;28:2131–2135. doi: 10.1016/j.bmcl.2018.05.022. [DOI] [PubMed] [Google Scholar]
- 105.Jha V., Bhadoriya K.S. Synthesis, Pharmacological Evaluation and Molecular Docking Studies of Pyrimidinedione Based DPP-4 Inhibitors as Antidiabetic Agents. J. Mol. Struct. 2018;1158:96–105. doi: 10.1016/j.molstruc.2018.01.014. [DOI] [Google Scholar]
- 106.Huang J., Deng X., Zhou S., Wang N., Qin Y., Meng L., Li G., Xiong Y., Fan Y., Guo L., et al. Identification of Novel Uracil Derivatives Incorporating Benzoic Acid Moieties as Highly Potent Dipeptidyl Peptidase-IV Inhibitors. Bioorg. Med. Chem. 2019;27:644–654. doi: 10.1016/j.bmc.2019.01.001. [DOI] [PubMed] [Google Scholar]
- 107.Li Q., Deng X., Jiang N., Meng L., Xing J., Jiang W., Xu Y. Identification and Structure–Activity Relationship Exploration of Uracil-Based Benzoic Acid and Ester Derivatives as Novel Dipeptidyl Peptidase-4 Inhibitors for The Treatment of Type 2 Diabetes Mellitus. Eur. J. Med. Chem. 2021;225:113765. doi: 10.1016/j.ejmech.2021.113765. [DOI] [PubMed] [Google Scholar]
- 108.Mourad A.A.E., Khodir A.E., Saber S., Mourad M.A.E. Novel Potent and Selective DPP-4 Inhibitors: Design, Synthesis and Molecular Docking Study of Dihydropyrimidine Phthalimide Hybrids. Pharmaceuticals. 2021;14:144. doi: 10.3390/ph14020144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Artasensi A., Angeli A., Lammi C., Bollati C., Gervasoni S., Baron G., Matucci R., Supuran C.T., Vistoli G., Fumagalli L. Discovery of a Potent and Highly Selective Dipeptidyl Peptidase IV and Carbonic Anhydrase Inhibitor as “Antidiabesity” Agents Based on Repurposing and Morphing of WB-4101. J. Med. Chem. 2022;65:13946–13966. doi: 10.1021/acs.jmedchem.2c01192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Srivastava J.K., Dubey P., Singh S., Bhat H.R., Kumawat M.K., Singh U.P. Discovery of Novel 1,3,5-Triazine-Thiazolidine-2,4-Diones as Dipeptidyl Peptidase-4 Inhibitors with Antibacterial Activity Targeting the S1 Pocket for the Treatment of Type 2 Diabetes. RSC Adv. 2015;5:14095–14102. doi: 10.1039/C4RA16903D. [DOI] [Google Scholar]
- 111.Gao H.-D., Liu P., Yang Y., Gao F. Sulfonamide-1,3,5-Triazine–Thiazoles: Discovery of A Novel Class of Antidiabetic Agents via Inhibition Of DPP-4. RSC Adv. 2016;6:83438–83447. doi: 10.1039/C6RA15948F. [DOI] [Google Scholar]
- 112.Wang Y., Tang X., Yi L. Design and Discovery of Novel 1,3,5-Triazines as Dipeptidyl Peptidase-4 Inhibitor against Diabetes. Pharmacology. 2019;103:273–281. doi: 10.1159/000494060. [DOI] [PubMed] [Google Scholar]
- 113.Gupta A., Bhat H.R., Singh U.P. Discovery of Novel Hybrids of Morpholino-1,3,5-Triazine-Pyrimidine as An Anti-Diabetic Agent in High-Fat, Low-Dose Streptozotocin-Induced Diabetes in Wistar Rats: An In-Vitro, In-Silico and In-Vivo Study. J. Mol. Struct. 2023;1294:136478. doi: 10.1016/j.molstruc.2023.136478. [DOI] [Google Scholar]
- 114.Gupta A., Bhat H.R., Singh U.P. Discovery of Imeglimin-Inspired Novel 1,3,5-Triazine Derivatives as Antidiabetic Agents in Streptozotocin-Induced Diabetes in Wistar Rats Via Inhibition of DPP-4. RSC Med. Chem. 2023;14:1512–1536. doi: 10.1039/D3MD00085K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Gupta A., Bhat H.R., Singh U.P. Discovery of Novel 1,3,5-triazine Derivatives As an Antidiabetic Agent in Wistar Rats via Inhibition of DPP-4. Future Med. Chem. 2023;15:829–852. doi: 10.4155/fmc-2022-0312. [DOI] [PubMed] [Google Scholar]
- 116.Biftu T., Sinha-Roy R., Chen P., Qian X., Feng D., Kuethe J.T., Scapin G., Gao Y.D., Yan Y., Krueger D., et al. Omarigliptin (MK-3102): A Novel Long-Acting DPP-4 Inhibitor for Once-Weekly Treatment of Type 2 Diabetes. J. Med. Chem. 2014;57:3205–3212. doi: 10.1021/jm401992e. [DOI] [PubMed] [Google Scholar]
- 117.Biftu T., Qian X., Chen P., Feng D., Scapin G., Gao Y.-D., Cox J., Sinha Roy R., Eiermann G., He H., et al. Novel Tetrahydropyran Analogs as Dipeptidyl Peptidase IV Inhibitors: Profile of Clinical Candidate (2R,3S,5R)-2-(2,5-difluorophenyl)-5-[2-(methylsulfonyl)-2,6-dihydropyrrolo[3,4-c]pyrazol-5(4H)-yl]tetrahydro-2H-pyran-3-amine (23) Bioorg. Med. Chem. Lett. 2013;23:5361–5366. doi: 10.1016/j.bmcl.2013.07.061. [DOI] [PubMed] [Google Scholar]
- 118.Betancort J.M., Winn D.T., Liu R., Xu Q., Liu J., Liao W., Chen S.-H., Carney D., Hanway D., Schmeits J., et al. Bicyclic Cyanothiazolidines as Novel Dipeptidyl Peptidase 4 Inhibitors. Bioorg. Med. Chem. Lett. 2009;19:4437–4440. doi: 10.1016/j.bmcl.2009.05.048. [DOI] [PubMed] [Google Scholar]
- 119.Zhang C., Ye F., Wang J., He P., Lei M., Huang L., Huang A., Tang P., Lin H., Liao Y., et al. Design, Synthesis, and Evaluation of a Series of Novel Super Long-Acting DPP-4 Inhibitors for the Treatment of Type 2 Diabetes. J. Med. Chem. 2020;63:7108–7126. doi: 10.1021/acs.jmedchem.0c00374. [DOI] [PubMed] [Google Scholar]
- 120.Li Q., Zhou M., Han L., Cao Q., Wang X., Zhao L., Zhou J., Zhang H. Design, Synthesis and Biological Evaluation of Imidazo [1,2-a]pyridine Derivatives as Novel DPP-4 Inhibitors. Chem. Biol. Drug Des. 2015;86:849–856. doi: 10.1111/cbdd.12560. [DOI] [PubMed] [Google Scholar]
- 121.Sheng Y., Sun B., Guo W.-T., Liu X., Wang Y.-C., Xie X., Xiao X.-L., Li N., Dong D.-L. (4-[6-(4-isopropoxyphenyl)pyrazolo [1,5-a]pyrimidin-3-yl] quinoline) Is a Novel Inhibitor of Autophagy. Br. J. Pharmacol. 2014;171:4970–4980. doi: 10.1111/bph.12821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Sheng Y., Sun B., Xie X., Li N., Dong D. DMH1 (4-[6-(4-isopropoxyphenyl)pyrazolo [1,5-a]pyrimidin-3-yl]quinoline) Inhibits Chemotherapeutic Drug-Induced Autophagy. Acta Pharm. Sin. B. 2015;5:330–336. doi: 10.1016/j.apsb.2014.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Guerrini G., Ciciani G., Daniele S., Di Cesare Mannelli L., Ghelardini C., Martini C., Selleri S. Synthesis and Pharmacological Evaluation of Pyrazolo [1,5-a]pyrimidin-7(4H)-one Derivatives As Potential GABAA-R Ligands. Bioorg. Med. Chem. 2017;25:1901–1906. doi: 10.1016/j.bmc.2017.02.013. [DOI] [PubMed] [Google Scholar]
- 124.Deng X., Shen J., Zhu H., Xiao J., Sun R., Xie F., Lam C., Wang J., Qiao Y., Tavallaie M.S., et al. Surrogating And Redirection of Pyrazolo [1,5-a]pyrimidin-7(4H)-one Core, a Novel Class of Potent and Selective DPP-4 Inhibitors. Bioorg. Med. Chem. 2018;26:903–912. doi: 10.1016/j.bmc.2018.01.006. [DOI] [PubMed] [Google Scholar]
- 125.Shen J., Deng X., Sun R., Tavallaie M.S., Wang J., Cai Q., Lam C., Lei S., Fu L., Jiang F. Structural Optimization of Pyrazolo [1,5-a]pyrimidine Derivatives as Potent and Highly Selective DPP-4 Inhibitors. Eur. J. Med. Chem. 2020;208:112850. doi: 10.1016/j.ejmech.2020.112850. [DOI] [PubMed] [Google Scholar]
- 126.Patel B.D., Bhadada S.V., Ghate M.D. Design, Synthesis and Anti-Diabetic Activity of Triazolotriazine Derivatives as Dipeptidyl Peptidase-4 (DPP-4) Inhibitors. Bioorg. Chem. 2017;72:345–358. doi: 10.1016/j.bioorg.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 127.Rohilla A., Gupta T., Pathak A., Akhtar M.J., Haider M.R., Haider K., Shahar Yar M. Emergence of Promising Novel DPP-4 Inhibitory Heterocycles as Anti-Diabetic Agents: A Review. Arch. Pharm. 2018;351:1800127. doi: 10.1002/ardp.201800127. [DOI] [PubMed] [Google Scholar]
- 128.Sharma M., Gupta M., Singh D., Kumar M., Kaur P. Synthesis, Evaluation and Molecular Docking of Thiazolopyrimidine Derivatives as Dipeptidyl Peptidase IV Inhibitors. Chem. Biol. Drug Des. 2012;80:918–928. doi: 10.1111/cbdd.12041. [DOI] [PubMed] [Google Scholar]
- 129.Lam B., Zhang Z., Stafford J.A., Skene R.J., Shi L., Gwaltney S.L. Structure-Based Design of Pyridopyrimidinediones as Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. Lett. 2012;22:6628–6631. doi: 10.1016/j.bmcl.2012.08.110. [DOI] [PubMed] [Google Scholar]
- 130.Fang Y., Zhang S., Wu W., Liu Y., Yang J., Li Y., Li M., Dong H., Jin Y., Liu R., et al. Design and Synthesis of Tetrahydropyridopyrimidine Derivatives as Dual GPR119 and DPP-4 Modulators. Bioorg. Chem. 2020;94:103390. doi: 10.1016/j.bioorg.2019.103390. [DOI] [PubMed] [Google Scholar]
- 131.Gozalbes R., Simon L., Froloff N., Sartori E., Monteils C., Baudelle R. Development and Experimental Validation of a Docking Strategy for the Generation of Kinase-Targeted Libraries. J. Med. Chem. 2008;51:3124–3132. doi: 10.1021/jm701367r. [DOI] [PubMed] [Google Scholar]
- 132.Edmondson S.D., Mastracchio A., Cox J.M., Eiermann G.J., He H., Lyons K.A., Patel R.A., Patel S.B., Petrov A., Scapin G., et al. Aminopiperidine-Fused Imidazoles as Dipeptidyl Peptidase-IV Inhibitors. Bioorg. Med. Chem. Lett. 2009;19:4097–4101. doi: 10.1016/j.bmcl.2009.06.011. [DOI] [PubMed] [Google Scholar]
- 133.Boehringer M., Fischer H., Hennig M., Hunziker D., Huwyler J., Kuhn B., Loeffler B.M., Luebbers T., Mattei P., Narquizian R., et al. Aryl- and Heteroaryl-Substituted Aminobenzo[a]quinolizines as Dipeptidyl Peptidase IV Inhibitors. Bioorg. Med. Chem. Lett. 2010;20:1106–1108. doi: 10.1016/j.bmcl.2009.12.025. [DOI] [PubMed] [Google Scholar]
- 134.Brossi A., Lindlar H., Walter M., Schnider O. Syntheseversuche in der Emetin-Reihe. 1. Mitteilung. 2-Oxo-hydrobenzo[a]chinolizine. Helv. Chim. Acta. 1958;41:119–139. doi: 10.1002/hlca.660410117. [DOI] [Google Scholar]
- 135.Mattei P., Boehringer M., Di Giorgio P., Fischer H., Hennig M., Huwyler J., Koçer B., Kuhn B., Loeffler B.M., MacDonald A., et al. Discovery of Carmegliptin: A Potent and Long-Acting Dipeptidyl Peptidase IV Inhibitor for The Treatment of Type 2 Diabetes. Bioorg. Med. Chem. Lett. 2010;20:1109–1113. doi: 10.1016/j.bmcl.2009.12.024. [DOI] [PubMed] [Google Scholar]
- 136.Pashev A., Burdzhiev N., Stanoeva E. Novel Angularly Substituted [1,4]thiazino [3,4-a]isoquinoline Carboxylic Acids Prepared By Cyclic Imine-Cyclic Anhydride Reaction. J. Heterocycl. Chem. 2023;60:513–518. doi: 10.1002/jhet.4601. [DOI] [Google Scholar]
- 137.Pashev A., Petrov V., Pesheva A., Petrova L., Ilieva K., Stavreva G., Atanasova M., Cheshmedzhieva D., Altankov G., Aleksandrova T. Angular-Substituted [1,4]Thiazino [3,4-a]Isoquinolines: Biological Evaluation and In Silico Studies on DPP-IV Inhibition. Int. J. Mol. Sci. 2024;25:11753. doi: 10.3390/ijms252111753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Wu W.-L., Hao J., Domalski M., Burnett D.A., Pissarnitski D., Zhao Z., Stamford A., Scapin G., Gao Y.-D., Soriano A., et al. Discovery of Novel Tricyclic Heterocycles as Potent and Selective DPP-4 Inhibitors for the Treatment of Type 2 Diabetes. Med. Chem. Lett. 2016;7:498–501. doi: 10.1021/acsmedchemlett.6b00027. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.