ABSTRACT
Valvular calcification, developing either in the mitral or the aortic valve, is highly prevalent in patients suffering from chronic kidney disease (CKD), in whom their presence correlates with higher cardiovascular and all-cause mortality risk. To date, the exact mechanisms that promote heart valve calcification remain unclear, and none of the treatments tested so far have shown efficacy in preventing valvular fibrocalcific remodelling. It is therefore essential to improve our understanding of the mechanisms involved in the pathological process if we are to find new, effective therapies. The purpose of this review is to (i) summarize our current knowledge of the mechanisms by which CKD and related therapies affect valvular cell activity, (ii) present the latest therapeutic targets identified in preclinical studies, and (iii) discuss the most recent clinical trials evaluating the efficacy of therapies aimed at preventing valvular calcification in CKD.
Keywords: aortic valve calcification, chronic kidney disease, mitral valve calcification
INTRODUCTION
Cardiovascular calcification is a degenerative process characterized by the accumulation of calcium and phosphate salts in the form of hydroxyapatite within the intimal and/or medial layers of the vessel wall, and in the heart valves. Cardiovascular calcification is not only abundant in CKD patients, but has also been shown to have a faster rate of progression [1, 2]. Research has largely focused on the pathophysiology and consequences of vascular calcification; less attention has been paid to heart valve calcification and its impact on the survival of patients with CKD. Epidemiological data report that the aortic valve (AV) and mitral valve (MV) are most commonly affected in CKD patients, resulting in valve stenosis and/or regurgitation [3, 4]. In patients on maintenance haemodialysis (HD), the reported prevalence of AV and MV calcification varies between 25% and 59% [1, 5–7]. Overall, the prevalence of valvular calcification (VC) is eight times higher in patients undergoing HD than in the general population [7]. In HD patients, the presence of VC is correlated with higher cardiovascular and all-cause mortality risk [8–10]. The exact mechanisms that promote heart valve calcification remain unclear, and there is no pharmacotherapy specifically targeting VC to prevent progressive valvular fibrocalcific remodelling. It is therefore essential to improve our understanding of the mechanisms involved in the pathological process if we are to find effective therapies. The purpose of this review is to (i) summarize our current knowledge of the mechanisms by which CKD and related therapies affect valvular cell activity, (ii) present the latest therapeutic targets identified in preclinical studies, and (iii) discuss the most recent clinical trials evaluating the efficacy of therapies aimed at preventing VC in CKD.
VALVULAR CALCIFICATION: MAJOR MECHANISMS AND CONSEQUENCES
Overview of the main cellular and molecular mechanisms
Quiescent valvular interstitial cells (qVICs) are the prominent cell type in healthy valve cusps and leaflets, residing throughout the fibrosa, spongiosa, and atrialis (MV)/ventricularis (AV) layers. VICs are recognized as essential for the homeostasis and remodelling of layer-specific extracellular matrix but also play a key role in pathological leaflet remodelling [11]. In response to injury or altered mechanical forces, qVICs transition towards activated VICs (aVICs), displaying a myofibroblastic phenotype characterized by the expression of α-smooth muscle actin (α-SMA), leading to fibrosis, thickening, and increased valve stiffness. Alternatively, qVICs and aVICs can differentiate into osteoblast-like cells (obVICs). These cells express markers such as runt-related transcription factor 2 (RUNX2), a marker of terminal osteoblastic differentiation, and can secrete a bone-like matrix that can calcify. If not halted, this mineralization process can lead to the formation of bone- and cartilage-like tissues that dramatically decrease the elasticity of valve leaflets. Evidence from histological and in vitro studies suggests that valvular remodelling shares similarities with vascular atherosclerosis. The main cellular and molecular mechanisms leading to VC are summarized in Fig. 1.
Figure 1:
Overview of the main mechanisms leading to valvular remodelling. Among the main mechanisms involved in AV remodelling, shear stress can induce endothelial damage and TGF-β accumulation within the valvular tissue. TGF-β promotes the EndMT of VECs as well as the transformation of qVICs into myofibroblast-activated VICs (aVICs), which can secrete collagen and promote valvular fibrosis and thickening. VECs dysfunction favours the infiltration of oxidized LDL and inflammatory cells, including macrophages, thereby promoting foam cell formation and initiating a process that resembles vascular atherosclerosis. Macrophage secretion of TNF-α and IL-1β promotes the deactivation of aVICs. Subsequent exposure to IL-6 promotes their osteogenic transition towards obVICs capable of promoting the mineralization process. αSMA, α-smooth muscle actin; MФ, macrophage; oxLDL, oxidized LDL; ALP: alkaline phosphatase.
AV calcification
In the AV, infiltration of red blood cells, oxidized lipids, and immune cell (monocytes/lymphocytes) following valvular endothelial injury is the initiating event [12–14]. Subsequent release of growth factors and pro-inflammatory cytokines by infiltrated macrophages and T cells have a profound impact on qVIC physiology. Indeed, secretion of TGF-β (transforming growth factor β) promotes qVIC transition towards pro-fibrotic aVIC. Local dysregulation in the mineral balance promotes the formation of obVICs expressing alkaline phosphatase (ALP), bone morphogenetic protein 2 (BMP2), MSX2, osteopontin (OPN), and RUNX2 [11, 15, 16]. Through a process known as endothelial-to-mesenchymal transition (EndMT), aortic valvular endothelial cells (VECs) can acquire a myofibroblastic phenotype and progressively develop an osteoblast-like phenotype, leading to calcification [17–19]. AV diseases preferentially occur in the aortic side of the valvular leaflets where they are exposed to complex and unstable haemodynamic conditions [20]. Similar to the vasculature, the altered haemodynamics can induce endothelial activation and inflammatory responses in AV leaflets [21], rendering them more prone to calcification and plaque formation [22].
MV calcification
As MV remodelling has been studied less than AV remodelling, it remains unclear to what extent the same mechanistic sequence is involved in the development of MV calcification. The process may be slightly different in the MV compared to the AV due to specific valvular architectures, overall cellular content, and different haemodynamic environments associated with their physical locations. Histologically, mitral annulus specimens show lipid deposition and inflammatory cell infiltration in the vicinity of calcification [23, 24]. Myofibroblastic differentiation of interstitial cells and the presence of lamellar bone have also been reported [25]. Stretching, mechanical stress, inflammation, or turbulent flow can trigger myofibroblastic differentiation of mitral qVICs either directly [26] or through the release of TGF‐β [27]. In vitro, mitral qVICs exposed to osteogenic medium can undergo both myofibroblastic and osteogenic differentiation, and display the ability to calcify over time [28–31]. As with aortic VECs, mitral VECs showed a propensity for osteogenic, as well as chondrogenic differentiation [30].
Consequences for valvular phenotype and function
Process of AV remodelling
The gradual fibro-calcific remodelling of the AV leaflets, called calcific aortic valve disease (CAVD), represents the most prevalent valvular heart disease worldwide [32, 33]. In the early phase of the disease, called AV sclerosis, the valve thickens and calcifies slightly, but these changes do not impede blood flow. Over time, the disease can progress to severe calcification of the valve with impaired leaflet movement and progressive narrowing of AV opening that impedes left ventricular outflow. Without intervention, progressive ventricular hypertrophy ensues ultimately leading to heart failure and death. Patients can experience distressing symptoms such as shortness of breath, angina, and syncope. Calcific aortic stenosis (CAS), the late stage of CAVD, is associated with important cardiovascular morbidity and mortality. Without appropriate and timely treatment the risk of death at 2 years can increase to 50% in the presence of severe CAS [32, 34, 35]. Currently, aortic valve replacement (AVR), either through surgery (SAVR) or transcatheter means (TAVR), remains the mainstay of management for patients with progressive and symptomatic CAS.
Process of MV remodelling
MV calcification results from gradual calcification of the fibrous mitral annulus, predominantly developing along its posterior portion [36]. When the calcification is confined to the annulus, leaflet motion is preserved and ventricular filling is unimpeded. However, calcific degeneration tends to spread further into the leaflets over time, resulting in impaired leaflet mobility and distortion [37]. These anatomical changes can cause both mitral stenosis [37] and mitral regurgitation [38]. While either lesion may be dominant in a given patient, mixed disease is more common. Mitral annular calcification is associated with progressive gradients across the MV [39]. When mitral stenosis or mitral regurgitation becomes severe, symptoms of heart failure, including dyspnoea and exercise intolerance, may occur. Possible extension of calcification into the left ventricle, papillary muscle, chordae tendineae, and left ventricular outflow tract, with occasional continuous calcification of the aorto-mitral curtain extending to the AV have been reported in the most severe cases [40].
VALVULAR CALCIFICATION IN CKD PATIENTS
Prevalence and outcomes
AV calcification
In CKD, the prevalence of CAS gradually increases as eGFR declines [41]. In this population, the pathological remodelling of the AV is more frequent [42], occurs earlier [4], progresses faster [43], and is associated with a worse prognosis [41, 43, 44] than in the general population. In CKD patients, most CAS are of degenerative origin (91%), with bicuspid-related CAS observed in only 7% of patients [45]. To date, the prevalence and progression of CAS in kidney transplant recipients has been poorly studied. Although the exact cellular and molecular mechanisms underlying AV remodelling in CKD patients remain unclear, multiple factors may account for the high prevalence and rapid progression of CAS in the CKD population. These include: the high calcium-phosphate product, accumulation of uremic toxins, low-grade inflammation, and use of anti-vitamin K drugs, together with low levels of calcification inhibitors such as pyrophosphate and fetuin A.
MV calcification
MV calcification is also common in patients with CKD [5, 46, 47]. Interestingly, severe MV calcification predicts CKD [48]. In dialysis patients, the presence of MV calcification correlated significantly with ischaemic heart disease [49]. Mitral annular calcification is associated with increased mortality and significant coronary artery disease in end-stage kidney disease patients. These patients have increased left ventricular cavity size, worse left ventricular systolic function, and higher left ventricular filling pressures compared to patients without MV calcification [47]. MV calcification is highly prevalent in renal transplant patients, with reported rates ranging from 21.5% to 23% [50–52]. Among renal transplant recipients, the burden of pre-transplant MV calcification was reported to be an independent predictor of post-transplant risk of cardiac death and myocardial infarction [52]. Kidney transplantation, which reduces cardiac volume overload, leads to a significant improvement in mitral regurgitation [51]. In 2021, Daragó et al. reported that calcification of the mitral and aortic valves progressed steadily, although not significantly, after kidney transplantation. In this study, the presence of diabetes was significantly associated with the incidence of calcified valves after kidney transplantation.
Clinical and preclinical associations
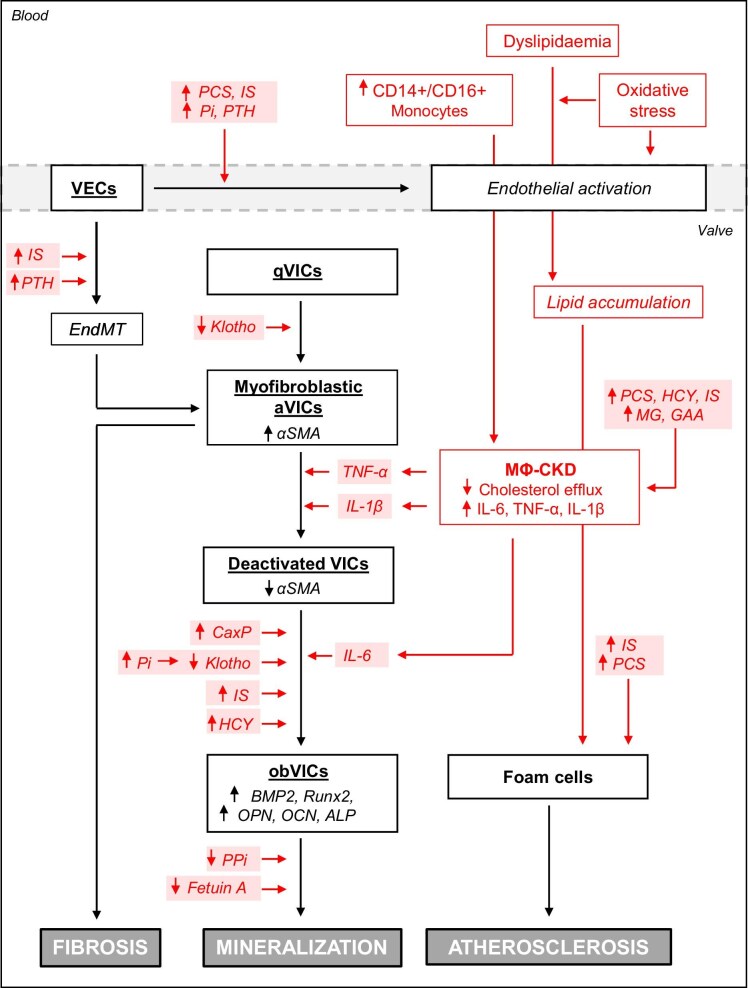
The main factors associated with the risk of developing VC in patients with CKD and the general population are summarized in Table 1. The mechanisms of action are presented in Fig. 2.
Table 1:
Traditional and CKD-specific risk factors for VC. LDL, low density lipoprotein; ApoE, apolipoprotein E; ApoB, apolipoprotein B; ENPP1, ectonucleotide pyrophosphatase/phosphodiesterase 1; VDR, vitamin D receptor; ADMA, asymmetric dimethylarginine; UTs : uremic toxins; CRP: C-reactive protein.
| Main risk factors for VC | |
|---|---|
| Traditional | CKD specific |
|
Non-modifiable risk factors
:
Age Gender (AV: male; MV: female) Bicuspid AV Gene variants for Runx2, ApoE, ApoB, Notch1, ENPP1, VDR Modifiable risk factors: Smoking Comorbidity: Obesity Metabolic syndrome Hypertension Diabetes Familiar hypercholesterolaemia Kidney disease Osteoporosis History of chest irradiation Aortic stenosis (MV) Serum biochemistry: ↑ LDL and lipoprotein a ↑ triglycerides ↓ Vitamin K ↑ Homocystein ↑ Calcium ↑ Phosphate ↓ Magnesium |
Disturbed mineral homeostasis:
↑ Calcium-phosphate product ↑ Serum phosphate ↑ PTH ↑ FGF-23 Loss of calcification inhibitors: ↓ Klotho ↓ Fetuin A ↓ Pyrophosphate ↓ Vitamin K Accumulation of UTs: ↑ Indoxyl-sulfate ↑ Homocystein ↑ ADMA ↑ β2-microglobulin Low-grade inflammation: ↑ CRP ↑ IL-6 |
Figure 2:
Main mechanisms by which CKD-related disorders promote VC. α-SMA, α-smooth muscle actin. GAA, guanidine acetic acid; HCY, homocysteine; IS, indoxyl-sulfate; PCS, paracresyl sulfate; Pi, inorganic phosphate; PPi, pyrophosphate; MG, methylguanidine; ALP: alkaline phosphatase.
CaxP product
Elevated CaxP (calcium-phosphate product) product is a strong predictor of VC in HD patients [10, 53, 54]. In vitro, calcium and phosphate, either alone [55–57] or in combination [58, 59], promote VICs osteogenic transition (evidenced by increased BMP2, OPN, RUNX2, SOX9 expression, and ALP activity), and mineralization [55, 60–62]. In rodents with induced renal disease, an increase in phosphate intake is required to induce VC [63–65]. Serum phosphate levels, even within the normal range, are significantly associated with the prevalence of VC both in the general population [66–68] and in patients with reduced renal function [69].
PTH
Elevated levels of PTH (parathyroid hormone) are associated with the presence of AV calcification in patients with preserved renal function [70], with mild to moderate CKD [71] and those receiving maintenance HD [72]. In preclinical studies, elevated PTH (either in vitro or in CKD mice) promotes VEC dysfunction and subsequent EndMT. This favours their transition towards osteoblast-like cells able to express BMP-2, bone sialoprotein, OPN, SOX9, and RUNX2 [73, 74]. The secretome of PTH-treated VEC promotes VICs osteogenic transition, evidenced by increased BMP-2/4 expression, increased secretion of osteocalcin and TGF-β1, and downregulation of collagen I and III [73]. The observation that Notch-1 inhibition in CKD mice prevents PTH-induced EndMT and subsequent AV calcification [74] suggests that targeting PTH or its associated signalling may protect against VC.
FGF-23
High C-terminal FGF-23 (fibroblast growth factor 23) is a determinant of VC in HD patients [75]. In patients with mild to moderate CKD, FGF-23 levels also correlate with the extent of AV calcification [71, 76]. In patients with CAS and preserved renal function, FGF-23 is the most important biomarker associated with the risk of adverse outcomes [77]. Elevated C-terminal FGF-23 levels are associated with increased 1-year mortality in patients undergoing TAVR with an eGFR ≥ 45 ml/min/1.73 m² [78]. However, the mechanisms linking FGF-23 to VC remain unclear.
Klotho
In patients with mild to moderate CKD, AV calcium score negatively correlates with circulating klotho levels [71]. Interestingly, lower klotho expression was observed in human calcified AV tissues [79]. Klotho-deficient mice show calcification at the hinge region of the fibrosa side of the AV [80]. When fed a high-fat diet, these mice show greater collagen levels in the AV than WT mice, indicating that klotho deficiency promotes AV fibrosis [81]. In SAMP1 mice, restoration of circulating klotho levels suppressed inflammation and subsequent myofibroblastic transition, thereby attenuating AV fibrosis [82]. In vitro, expression of klotho decreases in VICs exposed to high-phosphate [79]. Exposure to recombinant klotho markedly reduced phosphate-induced VICs osteogenic transition and mineralization. The observation that recombinant klotho reduces both RUNX2 and ALP basal levels in VICs isolated from calcified valves suggests that klotho may even reverse VIC pro-osteogenic activity [79]. Whether CKD-induced downregulation of klotho in VICs is linked to hyperphosphataemia and contributes to valvular remodelling remains to be investigated.
Loss of calcification inhibitors
AV explants cultured ex vivo secrete pyrophosphate, which effectively protects them against mineralization [83, 84]. Therefore, pyrophosphate deficiency in CKD [85, 86] may have an important impact on CAVD. In peritoneal dialysis (PD) patients, circulating fetuin A levels correlate with the presence of AV leaflet mineralization [87] and predict a worse prognosis [88]. Vitamin K deficiency is highly prevalent in CKD patients, and has been associated with vascular calcification [89–91], vascular stiffness [90, 92], mortality [93], and CV disease. To date, it is unclear whether vitamin K deficiency directly contributes to VC in CKD patients.
Inflammation
Inflammation is a potent inducer of VC. In vitro, macrophage secretion of TNF-α (tumour necrosis factor-α) or IL-1β (interleukin 1β) suppresses myofibroblastic activation of VICs from both aortic and mitral origin, which is necessary for IL-6 (interleukin-6) induced osteogenic transition [31, 95] (Fig. 1). Interestingly, macrophages from rats with CKD show enhanced pro-inflammatory properties when cultured ex vivo compared to those from control rats [96], and AV calcific remodelling in CKD rats is associated with macrophage infiltration and micro-inflammation, as evidenced by elevated NFkB activation [97]. Low-grade inflammation linked to CKD is associated with AV fibrocalcic remodelling and a higher prevalence of cardiovascular events [98–100]. In end-stage kidney disease patients with VC, inflammatory markers such as CRP (C-reactive protein) and IL-6 were shown to predict a worse prognosis [88, 100–102].
Uremic toxins
Uremic toxins (UTs) may play a key role in the onset, progression, and prognosis of VC in CKD. In patients with severe CAS and normal kidney function, serum asymmetric dimethylarginine activity is elevated and positively correlates with CAS severity [103]. Additionally, homocysteine levels are elevated in CAVD compared to those without CAVD [104]. In vitro, exposure to homocysteine promotes VICs osteogenic transition and mineralization. In mice, elevation of plasma homocysteine levels promotes AV fibrocalcic remodelling [105]. In vitro, indoxyl-sulfate promotes osteogenic transition and mineralization of human VICs [58] as well as VECs EndMT [106]. Whether a clinical association exists between circulating levels of asymmetric dimethylarginine, homocysteine, indoxyl-sulfate, and CAS in CKD patients has never been explored. Consistent with previous studies demonstrating a correlation between beta-2 microglobulin and VC [98, 107], Shen et al., recently reported beta-2 microglobulin to be a predictor of VC in HD patients [108, 109]. Given the key role played by UTs in endothelial activation [110, 111], monocyte inflammation [111–116], and foam cell formation [111, 117], it is plausible that UTs may predispose to valvular inflammation/atherogenesis by directly modulating the phenotype of monocytes and macrophages.
Hypomagnesaemia
Clinical studies in patients with advanced CKD have observed an association between low serum magnesium levels, increased vascular calcification, and cardiovascular mortality [118, 119]. Indeed, magnesium has been shown to prevent Pi-induced calcification of VSMCs in vitro through its ability to interfere with the process by which Ca and P crystallize into hydroxyapatite and to induce a signal in the cells that prevents their osteogenic transition [118, 119]. Low serum magnesium levels are recognized as a risk factor for cardiac valve calcification in patients with [120–124] and without renal disease [125]. Despite this association, it appears that the efficacy of magnesium in preventing in vitro the mineralization of aortic or mitral VICs has never been studied. To our knowledge, no interventional studies have been conducted to test the effect of magnesium supplementation on VC in CKD patients with low serum magnesium. Randomized clinical trials evaluating the effects of dietary magnesium supplementation, the use of magnesium-based phosphate binders, or the use of magnesium-enriched haemodialysis/PD solutions may be helpful in evaluating whether magnesium can be used as a therapeutic target to prevent VC in CKD.
Sclerostin
Sclerostin is an inhibitor of Wnt signalling [126] that has been reported to be cardioprotective [127]. In a cohort of 110 patients with CKD stages 3–5, sclerostin was identified as an independent risk factor for VC [128]. However, another study in 80 HD patients failed to demonstrate an association between serum sclerostin and VC [129]. Given that Wnt signalling is a key driver of BMP2-dependent osteogenic signalling in VICs [32], the downregulation of which reduces the osteogenic transition of VICs [130], the possibility that sclerostin elevation may protect against VC cannot be ruled out and needs further investigation.
Monocytes subsets
Three monocyte subsets have been defined in humans as a function of the cell surface expression of the lipopolysaccharide receptor CD14 and the FcIII receptor CD16: the classical (Mon1, CD14++/CD16−); intermediate (Mon 2, CD14++/CD16+); and non-classical (Mon 3, CD14+/CD16++) monocytes [131]. The proportion of pro-inflammatory CD14+/CD16+ monocytes (Mon2/Mon3) is abnormally high in both dialysed and non-dialysed patients with CKD [132] and closely associated with levels of high-sensitivity CRP and IL-6 [133]. In vitro, CD14+/CD16+ cells collected from CKD patients are more atherogenic [134] than Mon1, showing greater capacity to adhere to the endothelium [134], lower cholesterol efflux, avid oxLDL uptake, and potent intracellular production of IL-6, IL-1, and TNF-α [135]. Mon2 subset is increased in patients with CAS without renal impairment [136]. In these patients, high amounts of Mon2 before TAVR is associated with long-term mortality and worse functional outcomes post-TAVR [137]. In non-dialysed CKD patients, Mon2 monocytes are independently associated with cardiovascular events [135, 138]. If a similar association is found in patients with CKD, the measurement of Mon2 levels may be useful in the prediction of patients at risk of developing VC.
Clinical management
AV calcification
In patients with severe CKD, the diagnosis of CAS is challenging because patients remain asymptomatic for long periods and typical symptoms such as dyspnoea or angina may be underestimated due to sedentary lifestyles and confounding factors such as anaemia and volume overload. In HD patients, subtle signs such as hypotension, arrhythmias, and angina should be monitored during the dialysis session to alert the clinician to the presence of haemodynamically significant CAS.
Transthoracic echocardiography is widely used for early CAS detection [139]. It allows accurate diagnosis of the cause of CAS, assessment of haemodynamic severity, measurement of left ventricular size, search for hypertrophy or systolic dysfunction, and can help determine prognosis and optimal timing of valve intervention. Annual monitoring has been suggested for individuals with moderate CAS [34]. However, interpretation of transthoracic echocardiography in patients with CKD is difficult. First, CAS is not the only factor influencing afterload, which may also be influenced by hypertension or arterial stiffness, which are classically high in CKD patients. In addition, systolic dysfunction may decrease the mean pressure gradient, which may lead to an underestimation of the severity of CAS [140]. Therefore, reassessment of CAS severity during the first few months of dialysis is essential. Second, it is important to note that the AV area calculation is based on the continuity equation, which assumes that flows in the outflow tract and AV are equal. Therefore, in patients with septal hypertrophy [141] or severe calcification of the aortic annulus extending into the outflow tract, the AV area calculation may be biased. Finally, the presence of an arteriovenous fistula in HD patients may increase preload, cardiac output, and transvalvular flow [142]. Therefore, measurement of mean transvalvular pressure gradient and AV area during temporary fistula occlusion may theoretically better reflect the severity of CAS. However, because these measurements may underestimate CAS-induced haemodynamic disturbances in the left ventricle, they are not recommended in clinical practice [143].
Other imaging modalities include cardiac computed tomography and cardiac magnetic resonance imaging. Given that VC is strongly correlated with the progression of CAS, the severity of haemodynamic disturbances and the clinical outcome of CAS [144, 145], the 2021 European guidelines recommend the use of cardiac computed tomography to assess the degree of VC in patients with low valve gradient [34]. However, no study has specifically validated this method in the CKD population. Recently, magnetic resonance imaging, which depicts patterns of left ventricular remodelling more accurately than echocardiography, has become the gold standard for non-invasive assessment of the left ventricle [146]. However, given the reluctance to use gadolinium in severe CKD, no study has specifically evaluated this population. In this context, T1 mapping, which can detect myocardial damage including diffuse fibrosis [147] without the use of gadolinium, may be a promising approach in CKD patients.
To date, there are no specific guidelines for the management of CAS in patients with CKD, a population which has been excluded from large randomized trials. In fact, there is no medical treatment and, similar to patients with normal renal function, intervention is recommended in symptomatic patients with severe, high-grade aortic stenosis [mean gradient ≥40 mmHg, peak velocity ≥4.0 m/s, and valve area ≤1.0 cm2 (or ≤0.6 cm2/m)] [34]. Moreover, if AVR is associated with improved outcomes in CKD patients, dialysis remains a preoperative predictor of in-hospital mortality after AVR (whether surgical or transcatheter), which remains twice as high in dialysis versus non-dialysis patients [148–150]. Nevertheless, retrospective studies have demonstrated the benefit of AVR over conservative treatment in CKD [44] and dialysis patients [151]. Although acute kidney injury is a common complication of AVR [152] and is associated with an increased risk of mortality [153, 154], fear of acute kidney injury should not be a deterrent to AVR. In a recent study derived from the PARTNER cohort (n = 5190), Cubeddu et al. provide reassurance by showing that in CKD patients with severe CAS undergoing TAVR, CKD stage is more likely to remain stable or improve than worsen [155]. This post-procedural improvement in renal function, probably related to cardio-renal crosstalk, has also been demonstrated after SAVR [156]. The recent development of contrast-free TAVR may also help reduce the risk of acute kidney injury [157]. To date, severe CAS remains a barrier to access to transplantation or major surgery, and evidence on outcomes with AVR in renal transplant patients remains limited. The increased risk of graft failure with SAVR compared to TAVR reported in retrospective studies may encourage the preference of TAVR in kidney transplant patients with CAS [158]. The benefit of AVR before transplantation needs to be studied.
MV calcification
MV calcification is often asymptomatic until the lesions become severe and lead to degenerative mitral stenosis. It causes dyspnoea, orthopnoea, and cardiac signs associated with right ventricular failure (peripheral oedema, hepatosplenomegaly) [159]. Echocardiography is the gold standard for diagnosis, but the usual parameters have not been validated [34]. Planimetry is less reliable due to diffuse calcium and irregular orifice. The mean transmitral gradient has been shown to have prognostic value [160], which encourages us to perform transthoracic echocardiography after dialysis when blood pressure is controlled in a ‘dry weight’ patient. Medical management of early MV stenosis consists of controlling congestive symptoms with diuretics or ultrafiltration in dialysis patients. Transcatheter and surgical approaches are high-risk procedures and evidence from randomized trials is lacking [161].
PREVENTING VC IN CKD
Over the last 15 years, several therapeutic strategies have been developed and used to prevent the development of VC. The most promising approaches are presented in Table 2.
Table 2:
Main therapies under investigation to prevent VC. ApoE, apolipoprotein E; HDF, hemodiafiltration; ND, not determined.
| Evidence of efficiency | |||||
|---|---|---|---|---|---|
| Treatment | Mechanism of action | Preclinical | Reference | Clinical | Reference |
| SNF472 | Inhibits crystal formation and growth | ↓ VICs mineralization | [162] | Slowed the progression of AV mineralization in HD patients | [163] |
| ↓ Pig leaflet mineralization | [164] | ||||
| STS | Chelates precipitated calcium to form soluble calcium thiosulfate | In mice: ↓ Vascular calcifciation ↓ TNF-α, IL-1β, IL-6 in serum and aorta |
[165] | Prevented VC in HD patients | [166] |
| Calcimimetics | Decreases serum PTH and CaxP | ↓ PTH in rats, which prevented VECs EndMT, and VC | [74] | Slowed AV calcification in HD patients when given in combination with low-dose vitamin D | [167] |
| Promotes PBMCs-induced decalcification | ↑ CaSR expression in PBMCs from CKD patients, which rescues their capacity to prevent vascular calcification | [168] | |||
| Activation of CaSR in VICs and monocytes | Deleterious effect: ↑ VICs osteogenic transition and calcification |
[56] | |||
| Deleterious effect: ↑ Monocytes/macrophages chemtaxis, infiltration, and inflammation |
[169–171] | ||||
| Phosphate-lowering therapies | Decreases serum phosphorus | In ApoE KO mice with CKD, sevelamer, and lanthanum carbonate reduced valvular atherosclerosis and calcification | [172, 173] | Sevelamer and calcium-based phosphate binder slowed AV and MV calcification in HD patients | [174, 175] |
| Anti-IL-6 | Neutralization of IL-6 | Reduces indoxyl-sulfate-induced VICs mineralization | [58] | Subcutaneous administration of ziltivekimab (CKD stages 5–3) or clazakizumab (HD) reduced hsCRP Clazakizumab also reduced phospholipase A2 and lp(a) |
[176–178] |
| HD | |||||
| HDF, HCO, and MCO dialysis | Improve clearance of uremic retention solute with middle-to-high molecular weight compared to standard high-flux dialysis | ↓ Monocyte inflammation ↓ VSMC calcification (MCO/HCO) |
[179–182] | Reduce systemic inflammation to a greater extent than conventional high-flux dialysis | [180, 183, 184] |
| Acetate-free, citrate-acidified bicarbonate dialysis | Reduces serum calcification propensity | Protects against calcium deposition in rat aortic rings cultured ex vivo | [185] | Improves serum calcification propensity, assessed by T50, in HD patients | [186] |
| Peritoneal dialysis | Greater preservation of residual renal function Improved CKD-MBD More physiological approach to volume removal Reduced valvular shear stress |
ND | |||
| Vitamin K | Carboxylation of matrix-gla protein | ND | Vit K1 improved vitamin K status and retarded thoracic aortic calcification progress but had no effect on VC in HD patients (Vitavask trial) |
[187, 188] | |
SNF472
SNF472 is an intravenous formulation of the hexasodium salt of myoinositol hexaphosphate (phytate; IP6) that selectively and directly inhibits the formation and growth of hydroxyapatite crystals [189, 190]. SNF472 inhibited the calcification of human VICs [162] and pig AV leaflet cultured ex vivo [164]. Consistently, administration of SNF472 significantly attenuated the progression of AV calcification in HD patients [163]. Future studies are needed to assess the potential risks and benefits of this agent, particularly regarding its effect on bone mineral density [191].
Sodium thiosulfate
Several studies suggested that sodium thiosulfate (STS) may effectively prevent cardiovascular calcification linked to CKD by binding calcium ions in the circulation or soft tissues [192–196]. In addition, STS has antioxidant and anti-inflammatory properties that may help prevent tissue damage and remodelling [197]. In mice, targeted delivery of STS effectively reduced TNF-α, IL-6, and IL-1β in both serum and aorta and reduced the progression of vascular calcification [165]. In HD patients, STS therapy was reported to prevent the development of VC [166] as well as the progression of calcification in both the coronary and iliac arteries [198–200]. However, a meta-analysis of 19 cohort studies concluded that intravenous STS was not associated with improvement of skin lesions or survival benefit in CKD patients with calciphylaxis [201]. Additional studies are needed to confirm whether STS can protect against VC.
Calcimimetics
CaxP product and PTH dramatically affect VICs/VECs osteogenic properties. In CKD rats, reduction of PTH level using cinacalcet prevented EndMT and subsequent VC [74]. In HD patients with secondary hyperparathyroidism, Cinacalcet given in combination with fixed low-dose vitamin D sterols slowed AV calcification, although the primary outcome of the study, coronary calcification, was barely significant at 0.07 [167]. These latter data suggest that different mechanisms of calcification may exist between valves and arteries. Consistent with this hypothesis, calcimimetic activation of the calcium-sensing receptor (CaSR) expressed by aortic vascular smooth muscle cells (VSMCs) protects against mineralization [57], whereas their activation of the CaSR expressed by human VICs promotes osteogenic transition and mineralization [56]. Interestingly, peripheral blood mononuclear cells (PBMCs) isolated from CKD patients display lower CaSR expression at their cell surface than PBMCs from healthy donors, which renders them less efficient ex vivo to resorb a pre-established calcification [202]. Pre-incubation of these PBMCs with a calcimimetic significantly rescued their capacity to prevent calcium deposition and to resorb a pre-established calcification [202]. In pro-inflammatory conditions, activation of the monocytes CaSR promotes chemotaxis [203], infiltration [203], and NLRP3-dependent inflammation [169–171]. These adverse off-target effects may partly explain why cinacalcet failed to prevent the risk of death or major adverse cardiovascular events in the CKD population [204].
Phosphate-lowering therapies
As pathophysiological [205] and epidemiological data suggest a role for phosphate in the development [68] and progression [206] of VC, targeting phosphate to slow down VC progression could be a promising avenue. Early studies showed that sevelamer and lanthanum (both non-calcium-based phosphate binders) decreased VC in Apolipoprotein E-deficient mice with CKD [172, 173]. In a randomized clinical trial, Raggi and colleagues treated 186 HD patients with either calcium-based phosphate binders or sevelamer. After 1 year of follow-up they observed that cardiovascular calcification (including AV and MV calcification) slowed in 45% of the patients treated with sevelamer and regressed in 26% compared with only 28% and 10%, respectively, in the calcium group [174]. In a recent cohort study of 1489 adult patients receiving maintenance dialysis, Zhang et al. showed that the use of calcium-based phosphate binders was associated with a higher risk of coronary artery calcification progression compared with the use of non-calcium-based phosphate binders [207], which is consistent with the findings of previous meta-analyses [208]. Some years ago, it was suggested that combined preparations of calcium and magnesium could be effective alternatives to calcium-based agents because the amount of calcium is reduced compared to calcium salt alone and replaced with magnesium, which is a natural antagonist of the mineralization process. In this context, Matias et al. reported that calcium acetate/magnesium carbonate therapy reduced pulse pressure, lowered ventricular mass index, and reduced the progression of AV calcification in HD patients on hemodiafiltration (n = 36) compared with sevelamer (n = 30) or no phosphate binder therapy (n = 72) after 48 months of follow-up [175]. Interestingly, in the randomized LANDMARK study, lanthanum-based treatment for hyperphosphataemia neither reduced the cardiovascular events [209], nor delayed the progression of coronary artery calcification [210] compared with calcium carbonate in haemodialysis patients. In the same vein, in patients with stage 3b or 4 CKD, treatment with lanthanum over 96 weeks did not affect arterial stiffness or aortic calcification compared with placebo [211]. If CKD-MBD (chronic kidney disease-mineral and bone disorders) guidelines recommend lowering elevated phosphate levels towards the normal range in patients with CKD stages 3A to 5D, the optimal target level of serum phosphate remains uncertain. In 2021, the randomized trial EPISODE showed for the first time that strict phosphate control towards the normal range (3.9–4.55 mg/dl) by non-calcium-based phosphate binders delayed the progression of coronary arterial calcification in patients on dialysis [212]. Taken together, these data suggest that lowering phosphate by non-calcium-based phosphate binders may be an interesting strategy to prevent VC. Further studies are needed to evaluate whether phosphate-lowering therapies are effective in slowing VC and to determine the optimal target serum phosphate level to achieve efficiency.
Targeting IL-6
In patients with CAS, plasma levels of inflammatory cytokines such as IL-8, IL-6, TNF, and IL-1β are associated with an increased risk of mortality and hospitalization for heart failure with IL-6 being the best predictor [77]. Genome-wide association study meta-analysis identified the IL-6 locus as significantly associated with CAS [213]. Expression of IL-6 is elevated in CAVD samples and correlates with the remodelling process [214, 215]. Mendelian randomization approach showed that genetically proxied tocilizumab was associated with a reduced risk of CAS [216]. This observation reinforces the idea that targeting IL-6 might be a promising strategy. This seems particularly true in CKD patients, for whom plasma IL-6 independently associated with mortality [217] and with the presence of VC [75]. In preclinical studies, neutralizing IL-6 reduces indoxyl-sulfate-induced human VICs osteogenic transition and calcification [58]. This observation is of particular interest since monoclonal antibodies directed against IL-6, ziltivekimab [176, 177], and clazakizumab [178], are currently in clinical investigation in patients with stage 3–5 CKD and elevated hsCRP [176, 177]. Subcutaneous administration of these anti-IL-6 efficiently reduced median hsCRP levels as well as biomarkers associated with cardiovascular events as compared to placebo group. Studies should soon provide formal evidence of whether or not reducing circulating IL-6 levels leads to reduce major adverse cardiovascular events [177]. If the clinical outcomes are positive, further studies will have to determine whether targeting IL-6 slows VC progression. The fact that clazakizumab reduced secretory phospholipase A2, and lipoprotein a (lp(a)) concentrations relative to placebo is particularly promising. In CKD, AV calcifications and inflammation inversely correlate with bone mineralization [218] in an interdependent bone-vascular axis. Since IL-6 promotes osteoclast formation, the possibility that neutralizing IL-6 may influence the bone mineral density should not be neglected.
Vitamin K supplementation
In the VitaVask trial, vitamin K1 supplementation (5 mg, three times a week for 18 months) failed to slow the progression of calcification in the AV and MV in HD patients, whereas aortic calcification progress was significantly retarded [187]. In the VALKYRIE study, supplementation of HD patients with atrial fibrillation with rivaroxaban (10 mg daily) plus vitamin K2 [menaquinone 7 (MK-7), 2000 µg three times a week] for 18 months improved vitamin K status but did not change the VC score as compared to a supplementation with rivaroxaban or vitamin K antagonists [188]. These data are consistent with those obtained in the AVADEC study, which showed that 2 years of supplementation with MK-7 (720 µg/day) and vitamin D (25 µg/day) did not affect the progression of CAS in elderly men with normal renal function [219]. Interestingly, Kaesler et al. showed that HDL particles isolated from long-term HD patients exhibit an almost absent MK-7 incorporation in vitro compared to those from control subjects [220]. When HDL particles were spiked with MK-7, only those isolated from healthy controls showed significantly reduced uncarboxylated matrix-gla protein levels in VSMCs. This crucial change in MK-7 pharmacology may partly explain why, in contrast to what has been observed in the general population [221], MK-7 in CKD patients, had not shown a clear benefit in preventing VC in VALKYRIE, or cardiovascular calcification in the KURNATOWSKA [222], K4KIDNEYS [223], OIKONOMAKI [224], and RENAKVIT [225] studies.
Lipid-lowering therapies
Despite an association with hypercholesterolaemia, randomized clinical trials failed to demonstrate the efficacy of statins, even in combination with ezetimibe, in delaying CAS progression [226–228]. Whether the off-target effects of statins, which are known to promote cardiovascular calcification [229] and increase circulating lp(a) [230], are responsible for their lack of efficacy remains unclear. Contrary to statins, the absolute reduction in cardiovascular events on proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors is numerically greater in patients with advanced CKD [231]. Whether PCSK9 inhibitors efficiently prevent VC in this population remains to be investigated. If lowering lp(a) appears to be a promising strategy to prevent CAS [232], most CKD patients were excluded from the randomized controlled trials evaluating the efficacy of pelacarsen [a hepatocyte-directed antisense oligonucleotide targeting the lp(a) gene mRNA] in lowering lp(a) levels [233], and ‘significant renal disease’ is an exclusion criterion in ongoing phase 3 trials evaluating the cardiovascular outcomes of novel treatments designed to specifically lower lp(a) [234], suggesting that it will be several years before we know whether lp(a) levels contribute to VC in CKD [235].
Dialysis modalities
Improvement in dialysis could also be considered to be a means to prevent VC. Although it is always difficult to make comparisons between different populations, some data may suggest an advantage of PD over HD in the incidence of CAS. Indeed, studies estimate the prevalence of VC to be ∼32% in patients who have been on PD for at least 31 months [46, 236], while it ranges from 34% to 85% in patients on HD for a similar duration [237–239]. A recent study of 30 PD and 34 HD patients showed a significantly higher prevalence of VC in HD compared to PD patients (70.6% vs. 29.4%), further supporting the benefit of PD [240]. In fact, PD provides a more physiologic approach to volume removal, greater preservation of residual renal function, and is associated with improved CKD-MBD. This approach may therefore have theoretical advantages over HD to reduce the incidence of VC [241]. The absence of fistula may also account for this protective effect [242]. Indeed, the presence of an arteriovenous fistula has important haemodynamic consequences because it creates a high-flow, low-resistance environment that decreases total systemic vascular resistance while increasing venous return to the heart, thereby increasing cardiac output [243]. It is known that temporary compression of the arteriovenous fistula induces a significant decrease in transvalvular flow rate [142]. Besides, revision of the fistula to reduce blood flow decreased Vmax and improved dyspnoea to NYHA class II [244]. In addition, given that changes in shear stress promote valvular endothelial dysfunction [21], as well as monocyte inflammation, adhesion, and lipoprotein uptake through activation of the mechanosensitive ion channel Piezo-1 [245], it is plausible that the arteriovenous fistula directly influences the process of VC.
Besides, it should be noted that patients with no residual renal function or with severe diabetes, who are the most prone to develop VC, are also more likely to be referred to HD rather than PD, which may partly explain this reduced incidence. In this context, it is interesting to note that a recent Chinese multicentre prospective cohort study of 1489 patients aged 18 to 74 years undergoing HD or PD (21.6%) showed no difference in progression of VC after 4 years of follow-up [207]. In light of these conflicting results, further studies seem necessary to reach a firm conclusion regarding the potential benefit of PD over HD on the risk of VC [246].
Optimizing HD modalities in patients at risk of developing cardiovascular calcification could be an interesting alternative for reducing the prevalence of VC. On-line hemodiafiltration, high cut-off (HCO), and medium cut-off (MCO) dialysers provide an improved clearance of uremic retention solutes with middle-to-high molecular weight compared with conventional high-flux dialysis [247], and could theoretically help to prevent VC. In line with this hypothesis, HCO and MCO dialysers reduce systemic inflammation [180, 183, 184], decrease monocytes expression of inflammatory cytokines [179, 180] and are less prone to induce vascular cell calcification in vitro [181, 182] compared to standard high-flux dialysers. In addition, the composition of dialysis buffers appears to be particularly important. In 2019, Villa-Bellosta et al. exposed rat aortic rings to blood samples collected before and after HD from patients on acetate- (n = 35) or citrate- (n = 25) acidified bicarbonate dialysis. Using this system, they showed that citrate protects against calcium accumulation [185], which suggested that citrate-acidified bicarbonate dialysis may be an alternative approach to reduce ectopic calcification in HD patients. In 2018, Lorenz et al. showed that citrate-acidified acetate-free bicarbonate HD sustainably improved serum calcification propensity as assessed by T50 values in 78 prevalent European HD patients [186]. To date, the impact of dialysis buffers on VC has never been studied.
CONCLUSION
VC is common and, like vascular calcification, is associated with worse outcomes in patients with CKD. Although the genesis of VC involves several mechanisms similar to those that induce vascular calcification, it may also have specific and independent mechanisms. Although the behaviour of VICs has long been compared to that of VSMCs due to their anatomical proximity, it has recently been reported that valve cells exhibit more complex and unique behaviours than vascular cells. Indeed, human VSMCs exposed to an osteogenic environment expressed higher levels of osteogenic markers and exhibited greater matrix remodelling than human VICs from the same patient [248]. These data suggest the existence of cell-mediated differences between vascular and VC processes, which may account for some opposite calcification responses of human VICs compared to VSMCs. Understanding these mechanisms may lead to the development of specific therapeutic strategies to prevent and/or reverse VC. Whether modulation of VC can influence mortality and morbidity in patients with CKD remains to be demonstrated. To date, the paucity of data does not allow us to make robust evidence-based recommendations regarding the best way to monitor and protect heart valves in CKD patients (best monitoring tool, frequency of monitoring, promising preventive, and/or curative treatment). Further research is needed to address these issues.
ACKNOWLEDGEMENTS
The authors are grateful to the Fédération Hospitalo-Universitaire programme ‘CArdiac Research Network on Aortic VAlve and Heart Failure’ (reference: GCS G4 FHU CARNAVAL), the University of Picardie Jules Verne and the Hauts-de-France Regional Council for providing N.I. with a PhD fellowship. The authors would like to thank the French Government for its support through the Investissements d'Avenir programme of the National Research Agency (ANR) (ANR-16-RHUS-0003_STOP-AS).
Contributor Information
Lucie Hénaut, MP3CV Laboratory, UR UPJV 7517, CURS, University of Picardie Jules Verne, Amiens, France.
Alexandre Candellier, MP3CV Laboratory, UR UPJV 7517, CURS, University of Picardie Jules Verne, Amiens, France.
Sharon Huish, Department of Nephrology, Royal Devon University Healthcare NHS Foundation Trust, Exeter, UK; Donal O'Donoghue Renal Research Centre, Northern Care Alliance NHS Foundation Trust, Salford, UK.
Nervana Issa, MP3CV Laboratory, UR UPJV 7517, CURS, University of Picardie Jules Verne, Amiens, France.
Smeeta Sinha, Donal O'Donoghue Renal Research Centre, Northern Care Alliance NHS Foundation Trust, Salford, UK; Manchester Academic Health Sciences Centre, University of Manchester, Manchester, UK.
Ziad A Massy, INSERM Unit 1018, Team 5, CESP, Hôpital Paul Brousse, Paris-Saclay University and Versailles Saint-Quentin-en-Yvelines University (UVSQ), Villejuif, France; Association pour l'Utilisation du Rein Artificiel dans la région parisienne (AURA), Paris, Paris, France; Ambroise Paré University Hospital, APHP, Department of Nephrology Boulogne-Billancourt/Paris, Boulogne-Billancourt/Paris, France.
FUNDING
This paper was published as part of a supplement financially supported by an educational grant from CSL Vifor.
CONFLICT OF INTEREST STATEMENT
Z.A.M. reports grants from the NATIONAL RESEARCH AGENCY during the conduct of the study; grants from Amgen, grants from Sanofi-Genzyme, grants from the French Government, grants from MSD, grants and other from GSK, grants from Lilly, grants from FMC, grants from Baxter, grants from Otsuka, grants and other from AstraZeneca, grants from CSL Vifor, and others from Boehringer, outside the submitted work.
AUTHORS’ CONTRIBUTIONS
L.H., Z.M., and S.S. designed the literature review. L.H. wrote the section on the mechanisms and consequences of VC. L.H., N.I., and S.S. collected information on preclinical and clinical associations. A.C. and Z.M. wrote the section on clinical management of VC. L.H. and A.C. wrote the section on the prevention of VC. L.H. and N.I. designed the tables and figures. All authors critically revised the manuscript.
DATA AVAILABILITY STATEMENT
No new data were generated or analysed in support of this research.
REFERENCES
- 1. Braun J, Oldendorf M, Moshage W et al. Electron beam computed tomography in the evaluation of cardiac calcification in chronic dialysis patients. Am J Kidney Dis 1996;27:394–401. 10.1016/S0272-6386(96)90363-7 [DOI] [PubMed] [Google Scholar]
- 2. Tamashiro M, Iseki K, Sunagawa O et al. Significant association between the progression of coronary artery calcification and dyslipidemia in patients on chronic hemodialysis. Am J Kidney Dis 2001;38:64–9. 10.1053/ajkd.2001.25195 [DOI] [PubMed] [Google Scholar]
- 3. Hoevelmann J, Mahfoud F, Lauder L et al. Valvular heart disease in patients with chronic kidney disease. Herz 2021;46:228–33. 10.1007/s00059-020-05011-0 [DOI] [PubMed] [Google Scholar]
- 4. London GM, Pannier B, Marchais SJ et al. Calcification of the aortic valve in the dialyzed patient. J Am Soc Nephrol 2000;11:778–83. 10.1681/ASN.V114778 [DOI] [PubMed] [Google Scholar]
- 5. Raggi P, Boulay A, Chasan-Taber S et al. Cardiac calcification in adult hemodialysis patients. A link between end-stage renal disease and cardiovascular disease? J Am Coll Cardiol 2002;39:695–701. 10.1016/S0735-1097(01)01781-8 [DOI] [PubMed] [Google Scholar]
- 6. Otto CM, Lind BK, Kitzman DW et al. Association of aortic-valve sclerosis with cardiovascular mortality and morbidity in the elderly. N Engl J Med 1999;341:142–7. 10.1056/NEJM199907153410302 [DOI] [PubMed] [Google Scholar]
- 7. Ureña-Torres P, D'Marco L, Raggi P et al. Valvular heart disease and calcification in CKD: more common than appreciated. Nephrol Dial Transplant 2020;35:2046–53. 10.1093/ndt/gfz133 [DOI] [PubMed] [Google Scholar]
- 8. Wang Z, Jiang A, Wei F et al. Cardiac valve calcification and risk of cardiovascular or all-cause mortality in dialysis patients: a meta-analysis. BMC Cardiovasc Disord 2018;18:12. 10.1186/s12872-018-0747-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Bai J, Zhang X, Zhang A et al. Cardiac valve calcification is associated with mortality in hemodialysis patients: a retrospective cohort study. BMC Nephrol 2022;23:43. 10.1186/s12882-022-02670-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zhang J, Pang Q, Wang S et al. Associated factors of cardiac valve calcification and its prognostic effects among patients with chronic kidney disease: a systematic review and meta-analysis. Front Cardiovasc Med 2023;10:1120634. 10.3389/fcvm.2023.1120634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Liu AC, Joag VR, Gotlieb AI. The emerging role of valve interstitial cell phenotypes in regulating heart valve pathobiology. Am J Pathol 2007;171:1407–18. 10.2353/ajpath.2007.070251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Mohty D, Pibarot P, Després JP et al. Association between plasma LDL particle size, valvular accumulation of oxidized LDL, and inflammation in patients with aortic stenosis. Arterioscler Thromb Vasc Biol 2008;28:187–93. 10.1161/ATVBAHA.107.154989 [DOI] [PubMed] [Google Scholar]
- 13. Morvan M, Arangalage D, Franck G et al. Relationship of iron deposition to calcium deposition in Human aortic valve leaflets. J Am Coll Cardiol 2019;73:1043–54. 10.1016/j.jacc.2018.12.042 [DOI] [PubMed] [Google Scholar]
- 14. Zheng KH, Tsimikas S, Pawade T et al. Lipoprotein(a) and oxidized phospholipids promote valve calcification in patients with aortic stenosis. J Am Coll Cardiol 2019;73:2150–62. 10.1016/j.jacc.2019.01.070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Nagy E, Eriksson P, Yousry M et al. Valvular osteoclasts in calcification and aortic valve stenosis severity. Int J Cardiol 2013;168:2264–71. 10.1016/j.ijcard.2013.01.207 [DOI] [PubMed] [Google Scholar]
- 16. Yang X, Meng X, Su X et al. Bone morphogenic protein 2 induces Runx2 and osteopontin expression in human aortic valve interstitial cells: role of Smad1 and extracellular signal-regulated kinase 1/2. J Thorac Cardiovasc Surg 2009;138:1008–15. 10.1016/j.jtcvs.2009.06.024 [DOI] [PubMed] [Google Scholar]
- 17. Egan KP, Kim JH, Mohler ER et al. Role for circulating osteogenic precursor cells in aortic valvular disease. Arterioscler Thromb Vasc Biol 2011;31:2965–71. 10.1161/ATVBAHA.111.234724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Deb A, Wang SH, Skelding K et al. Bone marrow-derived myofibroblasts are present in adult human heart valves. J Heart Valve Dis 2005;14:674–8. [PubMed] [Google Scholar]
- 19. Liu X, Xu Z. Osteogenesis in calcified aortic valve disease: from histopathological observation towards molecular understanding. Prog Biophys Mol Biol 2016;122:156–61. 10.1016/j.pbiomolbio.2016.02.002 [DOI] [PubMed] [Google Scholar]
- 20. Otto CM, Kuusisto J, Reichenbach DD et al. Characterization of the early lesion of “degenerative” valvular aortic stenosis. Histological and immunohistochemical studies. Circulation 1994;90:844–53. 10.1161/01.CIR.90.2.844 [DOI] [PubMed] [Google Scholar]
- 21. Sucosky P, Balachandran K, Elhammali A et al. Altered shear stress stimulates upregulation of endothelial VCAM-1 and ICAM-1 in a BMP-4- and TGF-beta1-dependent pathway. Arterioscler Thromb Vasc Biol 2009;29:254–60. 10.1161/ATVBAHA.108.176347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Gomel MA, Lee R, Grande-Allen KJ. Comparing the role of mechanical forces in vascular and valvular calcification progression. Front Cardiovasc Med 2018;5:197. 10.3389/fcvm.2018.00197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Arounlangsy P, Sawabe M, Izumiyama N et al. Histopathogenesis of early-stage mitral annular calcification. J Med Dent Sci 2004;51:35–44. [PubMed] [Google Scholar]
- 24. Sell S, Scully RE. Aging changes in the aortic and mitral valves. Histologic and histochemical studies, with observations on the pathogenesis of calcific aortic stenosis and calcification of the mitral annulus. Am J Pathol 1965;46:345–65. [PMC free article] [PubMed] [Google Scholar]
- 25. Mohler ER, Gannon F, Reynolds C et al. Bone formation and inflammation in cardiac valves. Circulation 2001;103:1522–8. 10.1161/01.CIR.103.11.1522 [DOI] [PubMed] [Google Scholar]
- 26. Blomme B, Deroanne C, Hulin A et al. Mechanical strain induces a pro-fibrotic phenotype in human mitral valvular interstitial cells through RhoC/ROCK/MRTF-A and Erk1/2 signaling pathways. J Mol Cell Cardiol 2019;135:149–59. 10.1016/j.yjmcc.2019.08.008 [DOI] [PubMed] [Google Scholar]
- 27. Calafiore AM, Totaro A, Testa N et al. The secret life of the mitral valve. J Card Surg 2021;36:247–59. 10.1111/jocs.15151 [DOI] [PubMed] [Google Scholar]
- 28. Wang X, Deb N, Lacerda CMR. Comparison of serotonin-regulated calcific processes in aortic and mitral valvular interstitial cells. ACS Omega 2021;6:19494–505. 10.1021/acsomega.1c01723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wang X, Ali MS, Lacerda CMR. Osteogenesis inducers promote distinct biological responses in aortic and mitral valve interstitial cells. J Cell Biochem 2019;120:11158–71. 10.1002/jcb.28392 [DOI] [PubMed] [Google Scholar]
- 30. Wylie-Sears J, Aikawa E, Levine RA et al. Mitral valve endothelial cells with osteogenic differentiation potential. Arterioscler Thromb Vasc Biol 2011;31:598–607. 10.1161/ATVBAHA.110.216184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Zhu AS, Mustafa T, Connell JP et al. Tumor necrosis factor alpha and interleukin 1 beta suppress myofibroblast activation via nuclear factor kappa B signaling in 3D-cultured mitral valve interstitial cells. Acta Biomater 2021;127:159–68. 10.1016/j.actbio.2021.03.075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Lindman BR, Clavel MA, Mathieu P et al. Calcific aortic stenosis. Nat Rev Dis Primers 2016;2:16006. 10.1038/nrdp.2016.6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kraler S, Blaser MC, Aikawa E et al. Calcific aortic valve disease: from molecular and cellular mechanisms to medical therapy. Eur Heart J 2022;43:683–97. 10.1093/eurheartj/ehab757 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Vahanian A, Beyersdorf F, Praz F et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease: developed by the Task Force for the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Rev Esp Cardiol (Engl Ed) 2022;75:524. [DOI] [PubMed] [Google Scholar]
- 35. Tsao CW, Aday AW, Almarzooq ZI et al. Heart disease and stroke statistics-2022 update: a report from the American Heart Association. Circulation 2022;145:e153–e639. [DOI] [PubMed] [Google Scholar]
- 36. Korn D, Desanctis RW, Sell S. Massive calcification of the mitral annulus. A clinicopathological study of fourteen cases. N Engl J Med 1962;267:900–9. 10.1056/NEJM196211012671802 [DOI] [PubMed] [Google Scholar]
- 37. Muddassir SM, Pressman GS. Mitral annular calcification as a cause of mitral valve gradients. Int J Cardiol 2007;123:58–62. 10.1016/j.ijcard.2006.11.142 [DOI] [PubMed] [Google Scholar]
- 38. Fulkerson PK, Beaver BM, Auseon JC et al. Calcification of the mitral annulus: etiology, clinical associations, complications and therapy. Am J Med 1979;66:967–77. 10.1016/0002-9343(79)90452-2 [DOI] [PubMed] [Google Scholar]
- 39. Pressman GS, Agarwal A, Braitman LE et al. Mitral annular calcium causing mitral stenosis. Am J Cardiol 2010;105:389–91. 10.1016/j.amjcard.2009.09.042 [DOI] [PubMed] [Google Scholar]
- 40. Hilmy AM, el-Domaity N, Daabees AY. Effect of ethylenediaminetetraacetic acid (EDTA) on acute mercury poisoning of toad. Comp Biochem Physiol C Comp Pharmacol Toxicol 1986;85:253–4. 10.1016/0742-8413(86)90083-6 [DOI] [PubMed] [Google Scholar]
- 41. Vavilis G, Bäck M, Occhino G et al. Kidney dysfunction and the risk of developing aortic stenosis. J Am Coll Cardiol 2019;73:305–14. 10.1016/j.jacc.2018.10.068 [DOI] [PubMed] [Google Scholar]
- 42. Samad Z, Sivak JA, Phelan M et al. Prevalence and outcomes of left-sided valvular heart disease associated with chronic kidney disease. J Am Heart Assoc 2017;6:e006044. 10.1161/JAHA.117.006044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Candellier A, Bohbot Y, Pasquet A et al. Chronic kidney disease is a key risk factor for aortic stenosis progression. Nephrol Dial Transplant 2023;38:2776–85. 10.1093/ndt/gfad116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Bohbot Y, Candellier A, Diouf M et al. Severe aortic stenosis and chronic kidney disease: outcomes and impact of aortic valve replacement. J Am Heart Assoc 2020;9:e017190. 10.1161/JAHA.120.017190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Kim D, Shim CY, Hong GR et al. Effect of end-stage renal disease on rate of progression of aortic stenosis. Am J Cardiol 2016;117:1972–7. 10.1016/j.amjcard.2016.03.048 [DOI] [PubMed] [Google Scholar]
- 46. Wang AYM, Wang M, Woo J et al. Cardiac valve calcification as an important predictor for all-cause mortality and cardiovascular mortality in long-term peritoneal dialysis patients: a prospective study. J Am Soc Nephrol 2003;14:159–68. 10.1097/01.ASN.0000038685.95946.83 [DOI] [PubMed] [Google Scholar]
- 47. Sharma R, Pellerin D, Gaze DC et al. Mitral annular calcification predicts mortality and coronary artery disease in end stage renal disease. Atherosclerosis 2007;191:348–54. 10.1016/j.atherosclerosis.2006.03.033 [DOI] [PubMed] [Google Scholar]
- 48. Jesri A, Braitman LE, Pressman GS. Severe mitral annular calcification predicts chronic kidney disease. Int J Cardiol 2008;128:193–6. 10.1016/j.ijcard.2007.05.015 [DOI] [PubMed] [Google Scholar]
- 49. Mansur A, Saleem S, Naveed H et al. Mitral annular calcification In stage 5 chronic kidney disease on dialysis therapy. J Ayub Med Coll Abbottabad 2020;32:179–83. [PubMed] [Google Scholar]
- 50. Podestà MA, Cucchiari D, Ciceri P et al. Cardiovascular calcifications in kidney transplant recipients. Nephrol Dial Transplant 2022;37:2063–71. 10.1093/ndt/gfab053 [DOI] [PubMed] [Google Scholar]
- 51. Daragó A, Szabó E, Barkó D et al. Effects of kidney transplantation on valvular heart diseases. Transplant Proc 2021;53:1433–8. 10.1016/j.transproceed.2021.01.043 [DOI] [PubMed] [Google Scholar]
- 52. El Hangouche N, Gomez J, Asfaw A et al. Impact of pretransplant mitral annular calcification on the incidence of cardiac events after renal transplantation. Nephrol Dial Transplant 2020;35:526–33. 10.1093/ndt/gfz063 [DOI] [PubMed] [Google Scholar]
- 53. Ribeiro S, Ramos A, Brandão A et al. Cardiac valve calcification in haemodialysis patients: role of calcium-phosphate metabolism. Nephrol Dial Transplant 1998;13:2037–40. 10.1093/ndt/13.8.2037 [DOI] [PubMed] [Google Scholar]
- 54. Rufino M, García S, Jiménez A et al. Heart valve calcification and calcium x phosphorus product in hemodialysis patients: analysis of optimum values for its prevention. Kidney Int Suppl 2003;63:S115–8. 10.1046/j.1523-1755.63.s85.27.x [DOI] [PubMed] [Google Scholar]
- 55. Kessler JR, Bluemn TS, DeCero SA et al. Exploring molecular profiles of calcification in aortic vascular smooth muscle cells and aortic valvular interstitial cells. J Mol Cell Cardiol 2023;183:1–13. 10.1016/j.yjmcc.2023.08.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Issa H, Hénaut L, Abdallah JB et al. Activation of the calcium-sensing receptor in human valvular interstitial cells promotes calcification. J Mol Cell Cardiol 2019;129:2–12. 10.1016/j.yjmcc.2019.01.021 [DOI] [PubMed] [Google Scholar]
- 57. Hénaut L, Boudot C, Massy ZA et al. Calcimimetics increase CaSR expression and reduce mineralization in vascular smooth muscle cells: mechanisms of action. Cardiovasc Res 2014;101:256–65. 10.1093/cvr/cvt249 [DOI] [PubMed] [Google Scholar]
- 58. Candellier A, Issa N, Grissi M et al. Indoxyl-sulfate activation of the AhR- NF-κb pathway promotes interleukin-6 secretion and the subsequent osteogenic differentiation of human valvular interstitial cells from the aortic valve. J Mol Cell Cardiol 2023;179:18–29. 10.1016/j.yjmcc.2023.03.011 [DOI] [PubMed] [Google Scholar]
- 59. Sanchis P, Ho CY, Liu Y et al. Arterial “inflammaging” drives vascular calcification in children on dialysis. Kidney Int 2019;95:958–72. 10.1016/j.kint.2018.12.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Kruithof BPT, van de Pol V, Los T et al. New calcification model for intact murine aortic valves. J Mol Cell Cardiol 2021;156:95–104. 10.1016/j.yjmcc.2021.03.003 [DOI] [PubMed] [Google Scholar]
- 61. Balogh E, Chowdhury A, Ababneh H et al. Heme-mediated activation of the Nrf2/HO-1 axis attenuates calcification of valve interstitial cells. Biomedicines 2021;9:427. 10.3390/biomedicines9040427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. En Q, Zeping H, Yuetang W et al. Metformin alleviates the calcification of aortic valve interstitial cells through activating the PI3K/AKT pathway in an AMPK dependent way. Mol Med 2021;27:156. 10.1186/s10020-021-00416-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Messaoudi H, Levesque T, Perzo N et al. Subtotal nephrectomy associated with a high-phosphate diet in rats mimics the development of calcified aortic valve disease associated with chronic renal failure. J Clin Med 2023;12:1539. 10.3390/jcm12041539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Wang L, Tang R, Zhang Y et al. A rat model with multivalve calcification induced by subtotal nephrectomy and high-phosphorus diet. Kidney Dis 2020;6:346–54. 10.1159/000506013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Shuvy M, Abedat S, Eliaz R et al. Hyperphosphatemia is required for initiation but not propagation of kidney failure-induced calcific aortic valve disease. Am J Physiol Heart Circ Physiol 2019;317:H695–H704. 10.1152/ajpheart.00765.2018 [DOI] [PubMed] [Google Scholar]
- 66. Linefsky JP, O'Brien KD, Sachs M et al. Serum phosphate is associated with aortic valve calcification in the multi-ethnic Study of Atherosclerosis (MESA). Atherosclerosis 2014;233:331–7. 10.1016/j.atherosclerosis.2013.12.051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Xia C, Lei W, Hu Y et al. Association of serum levels of calcium, phosphate, and vitamin D with risk of developing aortic stenosis: the UK Biobank cohort. Eur J Prev Cardiol 2022;29:1520–8. 10.1093/eurjpc/zwac016 [DOI] [PubMed] [Google Scholar]
- 68. Linefsky JP, O'Brien KD, Katz R et al. Association of serum phosphate levels with aortic valve sclerosis and annular calcification: the cardiovascular health study. J Am Coll Cardiol 2011;58:291–7. 10.1016/j.jacc.2010.11.073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Adeney KL, Siscovick DS, Ix JH et al. Association of serum phosphate with vascular and valvular calcification in moderate CKD. J Am Soc Nephrol 2009;20:381–7. 10.1681/ASN.2008040349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Linhartová K, Veselka J, Sterbáková G et al. Parathyroid hormone and vitamin D levels are independently associated with calcific aortic stenosis. Circ J 2008;72:245–50. 10.1253/circj.72.245 [DOI] [PubMed] [Google Scholar]
- 71. Di Lullo L, Gorini A, Bellasi A et al. Fibroblast growth factor 23 and parathyroid hormone predict extent of aortic valve calcifications in patients with mild to moderate chronic kidney disease. Clin Kidney J 2015;8:732–6. 10.1093/ckj/sfv073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Iwata S, Hyodo E, Yanagi S et al. Parathyroid hormone and systolic blood pressure accelerate the progression of aortic valve stenosis in chronic hemodialysis patients. Int J Cardiol 2013;163:256–9. 10.1016/j.ijcard.2011.06.025 [DOI] [PubMed] [Google Scholar]
- 73. Vadana M, Cecoltan S, Ciortan L et al. Parathyroid hormone induces human valvular endothelial cells dysfunction that impacts the osteogenic phenotype of valvular interstitial cells. Int J Mol Sci 2022;23:3776. 10.3390/ijms23073776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Wang L, Tang R, Zhang Y et al. PTH-induced EndMT via miR-29a-5p/GSAP/Notch1 pathway contributed to valvular calcification in rats with CKD. Cell Prolif 2021;54:e13018. 10.1111/cpr.13018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Kandarini Y, Mahadita GW, Herawati S et al. High C-terminal fibroblast growth factor-23, intact parathyroid hormone, and interleukin-6 as determinants of valvular calcification in regular hemodialysis patients. Int J Gen Med 2022;15:4227–36. 10.2147/IJGM.S359168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Unsal A, Kose Budak S, Koc Y et al. Relationship of fibroblast growth factor 23 with left ventricle mass index and coronary calcification in chronic renal disease. Kidney Blood Press Res 2012;36:55–64. 10.1159/000339026 [DOI] [PubMed] [Google Scholar]
- 77. Vidula MK, Orlenko A, Zhao L et al. Plasma biomarkers associated with adverse outcomes in patients with calcific aortic stenosis. Eur J Heart Fail 2021;23:2021–32. 10.1002/ejhf.2361 [DOI] [PubMed] [Google Scholar]
- 78. Mirna M, Lauten A, Jirak P et al. Serum levels of C-terminal FGF23 (cFGF23) are associated with 1-year-mortality in patients undergoing transcatheter aortic valve replacement (TAVR). Eur J Intern Med 2021;85:98–107. 10.1016/j.ejim.2020.09.022 [DOI] [PubMed] [Google Scholar]
- 79. Li F, Yao Q, Ao L et al. Klotho suppresses high phosphate-induced osteogenic responses in human aortic valve interstitial cells through inhibition of Sox9. J Mol Med 2017;95:739–51. 10.1007/s00109-017-1527-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Gomez-Stallons MV, Wirrig-Schwendeman EE, Hassel KR et al. Bone morphogenetic protein signaling is required for aortic valve calcification. Arterioscler Thromb Vasc Biol 2016;36:1398–405. 10.1161/ATVBAHA.116.307526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Chen J, Lin Y, Sun Z. Deficiency in the anti-aging gene Klotho promotes aortic valve fibrosis through AMPKα-mediated activation of RUNX2. Aging Cell 2016;15:853–60. 10.1111/acel.12494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Chen J, Fan J, Wang S et al. Secreted klotho attenuates inflammation-associated aortic valve fibrosis in senescence-accelerated mice P1. Hypertension 2018;71:877–85. 10.1161/HYPERTENSIONAHA.117.10560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Rathan S, Yoganathan AP, O'Neill CW. The role of inorganic pyrophosphate in aortic valve calcification. J Heart Valve Dis 2014;23:387–94. [PMC free article] [PubMed] [Google Scholar]
- 84. Rattazzi M, Bertacco E, Iop L et al. Extracellular pyrophosphate is reduced in aortic interstitial valve cells acquiring a calcifying profile: implications for aortic valve calcification. Atherosclerosis 2014;237:568–76. 10.1016/j.atherosclerosis.2014.10.027 [DOI] [PubMed] [Google Scholar]
- 85. Lomashvili KA, Khawandi W, O'Neill WC. Reduced plasma pyrophosphate levels in hemodialysis patients. J Am Soc Nephrol 2005;16:2495–500. 10.1681/ASN.2004080694 [DOI] [PubMed] [Google Scholar]
- 86. Laurain A, Rubera I, Duranton C et al. Alkaline phosphatases account for low plasma levels of inorganic pyrophosphate in chronic kidney disease. Front Cell Dev Biol 2020;8:586831. 10.3389/fcell.2020.586831 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Wang AYM, Woo J, Lam CWK et al. Associations of serum fetuin-A with malnutrition, inflammation, atherosclerosis and valvular calcification syndrome and outcome in peritoneal dialysis patients. Nephrol Dial Transplant 2005;20:1676–85. 10.1093/ndt/gfh891 [DOI] [PubMed] [Google Scholar]
- 88. Wang AYM, Lam CWK, Wang M et al. Increased circulating inflammatory proteins predict a worse prognosis with valvular calcification in end-stage renal disease: a prospective cohort study. Am J Nephrol 2008;28:647–53. 10.1159/000117817 [DOI] [PubMed] [Google Scholar]
- 89. Schurgers LJ, Barreto DV, Barreto FC et al. The circulating inactive form of matrix gla protein is a surrogate marker for vascular calcification in chronic kidney disease: a preliminary report. Clin J Am Soc Nephrol 2010;5:568–75. 10.2215/CJN.07081009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Thamratnopkoon S, Susantitaphong P, Tumkosit M et al. Correlations of plasma desphosphorylated uncarboxylated matrix gla protein with vascular calcification and vascular stiffness in chronic kidney disease. Nephron 2017;135:167–72. 10.1159/000453368 [DOI] [PubMed] [Google Scholar]
- 91. Delanaye P, Krzesinski JM, Warling X et al. Dephosphorylated-uncarboxylated Matrix Gla protein concentration is predictive of vitamin K status and is correlated with vascular calcification in a cohort of hemodialysis patients. BMC Nephrol 2014;15:145. 10.1186/1471-2369-15-145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Fain ME, Kapuku GK, Paulson WD et al. Inactive matrix gla protein, arterial stiffness, and endothelial function in African American hemodialysis patients. Am J Hypertens 2018;31:735–41. 10.1093/ajh/hpy049 [DOI] [PubMed] [Google Scholar]
- 93. Cheung CL, Sahni S, Cheung BMY et al. Vitamin K intake and mortality in people with chronic kidney disease from NHANES III. Clin Nutr 2015;34:235–40. 10.1016/j.clnu.2014.03.011 [DOI] [PubMed] [Google Scholar]
- 94. Bellinge JW, Dalgaard F, Murray K et al. Vitamin K intake and atherosclerotic cardiovascular disease in the Danish diet cancer and health study. J Am Heart Assoc 2021;10:e020551. 10.1161/JAHA.120.020551 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Grim JC, Aguado BA, Vogt BJ et al. Secreted factors from proinflammatory macrophages promote an osteoblast-like phenotype in valvular interstitial cells. Arterioscler Thromb Vasc Biol 2020;40:e296–e308. 10.1161/ATVBAHA.120.315261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Li C, Ding XY, Xiang DM et al. Enhanced M1 and impaired M2 macrophage polarization and reduced mitochondrial biogenesis via inhibition of AMP kinase in chronic kidney disease. Cell Physiol Biochem 2015;36:358–72. 10.1159/000430106 [DOI] [PubMed] [Google Scholar]
- 97. Shuvy M, Abedat S, Beeri R et al. Uraemic hyperparathyroidism causes a reversible inflammatory process of aortic valve calcification in rats. Cardiovasc Res 2008;79:492–9. 10.1093/cvr/cvn088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Ikee R, Honda K, Ishioka K et al. Differences in associated factors between aortic and mitral valve calcification in hemodialysis. Hypertens Res 2010;33:622–6. 10.1038/hr.2010.44 [DOI] [PubMed] [Google Scholar]
- 99. Schönenberger A, Winkelspecht B, Köhler H et al. High prevalence of aortic valve alterations in haemodialysis patients is associated with signs of chronic inflammation. Nephron Clin Pract 2004;96:c48–55. 10.1159/000076399 [DOI] [PubMed] [Google Scholar]
- 100. Mohamed BA, Yang W, Litt H et al. Valvular calcification, inflammation, and mortality in dialysis patients. J Heart Valve Dis 2013;22:584–90. [PubMed] [Google Scholar]
- 101. Leskinen Y, Paana T, Saha H et al. Valvular calcification and its relationship to atherosclerosis in chronic kidney disease. J Heart Valve Dis 2009;18:429–38. [PubMed] [Google Scholar]
- 102. Takahashi H, Ishii H, Aoyama T et al. Association of cardiac valvular calcifications and C-reactive protein with cardiovascular mortality in incident hemodialysis patients: a Japanese cohort study. Am J Kidney Dis 2013;61:254–61. 10.1053/j.ajkd.2012.09.007 [DOI] [PubMed] [Google Scholar]
- 103. Cagirci G, Cay S, Canga A et al. Association between plasma asymmetrical dimethylarginine activity and severity of aortic valve stenosis. J Cardiovasc Med 2011;12:96–101. 10.2459/JCM.0b013e32833cdcea [DOI] [PubMed] [Google Scholar]
- 104. Wu G, Xian J, Yang X et al. Association between homocysteine levels and calcific aortic valve disease: a systematic review and meta-analysis. Oncotarget 2018;9:8665–74. 10.18632/oncotarget.23938 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Xie W, Shan Y, Wu Z et al. Herpud1 deficiency alleviates homocysteine-induced aortic valve calcification. Cell Biol Toxicol 2023;39:2665–84. 10.1007/s10565-023-09794-w [DOI] [PubMed] [Google Scholar]
- 106. Delgado-Marin M, Sánchez-Esteban S, Cook-Calvete A et al. Indoxyl sulfate-induced valve endothelial cell endothelial-to-mesenchymal transition and calcification in an integrin-linked kinase-dependent manner. Cells 2024;13:481. 10.3390/cells13060481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Ikee R, Honda K, Oka M et al. Association of heart valve calcification with malnutrition-inflammation complex syndrome, beta-microglobulin, and carotid intima media thickness in patients on hemodialysis. Ther Apher Dial 2008;12:464–8. 10.1111/j.1744-9987.2008.00636.x [DOI] [PubMed] [Google Scholar]
- 108. Feng J, Yu L, Li H et al. High serum β2-microglobulin is a significant predictor of mortality in maintenance hemodialysis patients. Semin Dial 2023;36:247–54. 10.1111/sdi.13128 [DOI] [PubMed] [Google Scholar]
- 109. Lu M, Amano S, Miyamoto K et al. Insulin-induced vascular endothelial growth factor expression in retina. Invest Ophthalmol Vis Sci 1999;40:3281–6. [PubMed] [Google Scholar]
- 110. Six I, Maizel J, Barreto FC et al. Effects of phosphate on vascular function under normal conditions and influence of the uraemic state. Cardiovasc Res 2012;96:130–9. 10.1093/cvr/cvs240 [DOI] [PubMed] [Google Scholar]
- 111. Jing YJ, Ni JW, Ding FH et al. p-cresyl sulfate is associated with carotid arteriosclerosis in hemodialysis patients and promotes atherogenesis in apoE-/- mice. Kidney Int 2016;89:439–49. 10.1038/ki.2015.287 [DOI] [PubMed] [Google Scholar]
- 112. Glorieux GL, Dhondt AW, Jacobs P et al. In vitro study of the potential role of guanidines in leukocyte functions related to atherogenesis and infection. Kidney Int 2004;65:2184–92. 10.1111/j.1523-1755.2004.00631.x [DOI] [PubMed] [Google Scholar]
- 113. Schepers E, Barreto DV, Liabeuf S et al. Symmetric dimethylarginine as a proinflammatory agent in chronic kidney disease. Clin J Am Soc Nephrol 2011;6:2374–83. 10.2215/CJN.01720211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Fang P, Zhang D, Cheng Z et al. Hyperhomocysteinemia potentiates hyperglycemia-induced inflammatory monocyte differentiation and atherosclerosis. Diabetes 2014;63:4275–90. 10.2337/db14-0809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Zhang D, Jiang X, Fang P et al. Hyperhomocysteinemia promotes inflammatory monocyte generation and accelerates atherosclerosis in transgenic cystathionine beta-synthase-deficient mice. Circulation 2009;120:1893–902. 10.1161/CIRCULATIONAHA.109.866889 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Zeng X, Dai J, Remick DG et al. Homocysteine mediated expression and secretion of monocyte chemoattractant protein-1 and interleukin-8 in human monocytes. Circ Res 2003;93:311–20. 10.1161/01.RES.0000087642.01082.E4 [DOI] [PubMed] [Google Scholar]
- 117. Matsuo K, Yamamoto S, Wakamatsu T et al. Increased proinflammatory cytokine production and decreased cholesterol efflux due to downregulation of ABCG1 in macrophages exposed to indoxyl sulfate. Toxins 2015;7:3155–66. 10.3390/toxins7083155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Hénaut L, Massy ZA. Magnesium as a calcification inhibitor. Adv Chronic Kidney Dis 2018;25:281–90. 10.1053/j.ackd.2017.12.001 [DOI] [PubMed] [Google Scholar]
- 119. Zaslow SJ, Oliveira-Paula GH, Chen W. Magnesium and vascular calcification in chronic kidney disease: current insights. Int J Mol Sci 2024;25:1155. 10.3390/ijms25021155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Tsai S, Li Y, Wu X. Serum magnesium level and cardiac valve calcification in hemodialysis patients. BMC Cardiovasc Disord 2023;23:610. 10.1186/s12872-023-03662-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Ding Z, Chen W, Zhang C et al. Correlation between serum magnesium level and cardiac valve calcification in patients with chronic kidney disease. Clin Lab 2022;68. 10.7754/Clin.Lab.2021.210533 [DOI] [PubMed] [Google Scholar]
- 122. Silva AP, Viegas CSB, Guilherme P et al. Gla-rich protein, magnesium and phosphate associate with mitral and aortic valves calcification in diabetic patients with moderate CKD. Diagnostics 2022;12:496. 10.3390/diagnostics12020496 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Tzanakis I, Pras A, Kounali D et al. Mitral annular calcifications in haemodialysis patients: a possible protective role of magnesium. Nephrol Dial Transplant 1997;12:2036–7. 10.1093/ndt/12.9.2036 [DOI] [PubMed] [Google Scholar]
- 124. Silva AP, Gundlach K, Büchel J et al. Low magnesium levels and FGF-23 dysregulation predict mitral valve calcification as well as intima media thickness in predialysis diabetic patients. Int J Endocrinol 2015;2015:308190. 10.1155/2015/308190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Hisamatsu T, Miura K, Fujiyoshi A et al. Serum magnesium, phosphorus, and calcium levels and subclinical calcific aortic valve disease: a population-based study. Atherosclerosis 2018;273:145–52. 10.1016/j.atherosclerosis.2018.03.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Kusu N, Laurikkala J, Imanishi M et al. Sclerostin is a novel secreted osteoclast-derived bone morphogenetic protein antagonist with unique ligand specificity. J Biol Chem 2003;278:24113–7. 10.1074/jbc.M301716200 [DOI] [PubMed] [Google Scholar]
- 127. Drechsler C, Evenepoel P, Vervloet MG et al. High levels of circulating sclerostin are associated with better cardiovascular survival in incident dialysis patients: results from the NECOSAD study. Nephrol Dial Transplant 2015;30:288–93. 10.1093/ndt/gfu301 [DOI] [PubMed] [Google Scholar]
- 128. Ji YQ, Guan LN, Yu SX et al. Serum sclerostin as a potential novel biomarker for heart valve calcification in patients with chronic kidney disease. Eur Rev Med Pharmacol Sci 2018;22:8822–9. [DOI] [PubMed] [Google Scholar]
- 129. Khedr A, Hebah H, Gohar S. Relation between sclerostin and valvular calcification in hemodialysis patients. Egypt J Hosp Med 2023;91:3785–9. 10.21608/ejhm.2023.293448 [DOI] [Google Scholar]
- 130. Li SJ, Kao YH, Chung CC et al. HDAC I inhibitor regulates RUNX2 transactivation through canonical and non-canonical Wnt signaling in aortic valvular interstitial cells. Am J Transl Res 2019;11:744–54. [PMC free article] [PubMed] [Google Scholar]
- 131. Ziegler-Heitbrock L, Ancuta P, Crowe S et al. Nomenclature of monocytes and dendritic cells in blood. Blood 2010;116:e74–80. 10.1182/blood-2010-02-258558 [DOI] [PubMed] [Google Scholar]
- 132. Merino A, Portolés J, Selgas R et al. Effect of different dialysis modalities on microinflammatory status and endothelial damage. Clin J Am Soc Nephrol 2010;5:227–34. 10.2215/CJN.03260509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Lee JW, Cho E, Kim MG et al. Proinflammatory CD14(+)CD16(+) monocytes are associated with vascular stiffness in predialysis patients with chronic kidney disease. Kidney Res Clin Pract 2013;32:147–52. 10.1016/j.krcp.2013.08.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Ramírez R, Carracedo J, Merino A et al. CD14+CD16+ monocytes from chronic kidney disease patients exhibit increased adhesion ability to endothelial cells. Contrib Nephrol 2011;171:57–61. 10.1159/000327134 [DOI] [PubMed] [Google Scholar]
- 135. Rogacev KS, Zawada AM, Emrich I et al. Lower apo A-I and lower HDL-C levels are associated with higher intermediate CD14++CD16+ monocyte counts that predict cardiovascular events in chronic kidney disease. Arterioscler Thromb Vasc Biol 2014;34:2120–7. 10.1161/ATVBAHA.114.304172 [DOI] [PubMed] [Google Scholar]
- 136. Hewing B, Au SCD, Ludwig A et al. Severe aortic valve stenosis in adults is associated with increased levels of circulating intermediate monocytes. J Cardiovasc Transl Res 2017;10:27–34. 10.1007/s12265-016-9726-9 [DOI] [PubMed] [Google Scholar]
- 137. Neuser J, Galuppo P, Fraccarollo D et al. Intermediate CD14++CD16+ monocytes decline after transcatheter aortic valve replacement and correlate with functional capacity and left ventricular systolic function. PLoS ONE 2017;12:e0183670. 10.1371/journal.pone.0183670 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Rogacev KS, Seiler S, Zawada AM et al. CD14++CD16+ monocytes and cardiovascular outcome in patients with chronic kidney disease. Eur Heart J 2011;32:84–92. 10.1093/eurheartj/ehq371 [DOI] [PubMed] [Google Scholar]
- 139. Otto CM, Nishimura RA, Bonow RO et al. 2020 ACC/AHA Guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2021;143:e72–e227. [DOI] [PubMed] [Google Scholar]
- 140. Briand M, Dumesnil JG, Kadem L et al. Reduced systemic arterial compliance impacts significantly on left ventricular afterload and function in aortic stenosis: implications for diagnosis and treatment. J Am Coll Cardiol 2005;46:291–8. 10.1016/j.jacc.2004.10.081 [DOI] [PubMed] [Google Scholar]
- 141. Khan AA, Tang GHL, Engstrom K et al. Aortic stenosis with severe asymmetric septal hypertrophy: a novel management strategy to improve TAVR outcomes. JACC Cardiovasc Interv 2019;12:2228–30. 10.1016/j.jcin.2019.06.025 [DOI] [PubMed] [Google Scholar]
- 142. Ennezat PV, Maréchaux S, Pibarot P. From excessive high-flow, high-gradient to paradoxical low-flow, low-gradient aortic valve stenosis: hemodialysis arteriovenous fistula model. Cardiology 2010;116:70–2. 10.1159/000314938 [DOI] [PubMed] [Google Scholar]
- 143. Shroff GR, Bangalore S, Bhave NM et al. Evaluation and management of aortic stenosis in chronic kidney disease: a scientific statement from the American Heart Association. Circulation 2021;143:e1088–e114. 10.1161/CIR.0000000000000979 [DOI] [PubMed] [Google Scholar]
- 144. Pawade T, Clavel MA, Tribouilloy C et al. Computed tomography aortic valve calcium scoring in patients with aortic stenosis. Circ Cardiovasc Imaging 2018;11:e007146. 10.1161/CIRCIMAGING.117.007146 [DOI] [PubMed] [Google Scholar]
- 145. Pawade T, Sheth T, Guzzetti E et al. Why and how to measure aortic valve calcification in patients with aortic stenosis. JACC Cardiovasc Imaging 2019;12:1835–48. 10.1016/j.jcmg.2019.01.045 [DOI] [PubMed] [Google Scholar]
- 146. Treibel TA, Badiani S, Lloyd G et al. Multimodality imaging markers of adverse myocardial remodeling in aortic stenosis. JACC Cardiovasc Imaging 2019;12:1532–48. 10.1016/j.jcmg.2019.02.034 [DOI] [PubMed] [Google Scholar]
- 147. Bing R, Cavalcante JL, Everett RJ et al. Imaging and impact of myocardial fibrosis in aortic stenosis. JACC Cardiovasc Imaging 2019;12:283–96. 10.1016/j.jcmg.2018.11.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Thourani VH, Keeling WB, Sarin EL et al. Impact of preoperative renal dysfunction on long-term survival for patients undergoing aortic valve replacement. Ann Thorac Surg 2011;91:1798–806; discussion 1806-1807. 10.1016/j.athoracsur.2011.02.015 [DOI] [PubMed] [Google Scholar]
- 149. Kumar N, Khera R, Garg N et al. Comparison of outcomes of transcatheter versus surgical aortic valve replacement in patients with chronic kidney disease. Am J Cardiol 2018;121:343–8. 10.1016/j.amjcard.2017.10.029 [DOI] [PubMed] [Google Scholar]
- 150. Conrotto F, Salizzoni S, Andreis A et al. Transcatheter aortic valve implantation in patients with advanced chronic kidney disease. Am J Cardiol 2017;119:1438–42. 10.1016/j.amjcard.2017.01.042 [DOI] [PubMed] [Google Scholar]
- 151. Kawase Y, Taniguchi T, Morimoto T et al. Severe aortic stenosis in dialysis patients. J Am Heart Assoc 2017;6:e004961. 10.1161/JAHA.116.004961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Kumar N, Garg N. Acute kidney injury after aortic valve replacement in a nationally representative cohort in the USA. Nephrol Dial Transplant 2019;34:295–300. 10.1093/ndt/gfy097 [DOI] [PubMed] [Google Scholar]
- 153. Elhmidi Y, Bleiziffer S, Deutsch MA et al. Acute kidney injury after transcatheter aortic valve implantation: incidence, predictors and impact on mortality. Arch Cardiovasc Dis 2014;107:133–9. 10.1016/j.acvd.2014.01.002 [DOI] [PubMed] [Google Scholar]
- 154. Gebauer K, Diller GP, Kaleschke G et al. The risk of acute kidney injury and its impact on 30-day and long-term mortality after transcatheter aortic valve implantation. Int J Nephrol 2012;2012:483748. 10.1155/2012/483748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Cubeddu RJ, Asher CR, Lowry AM et al. Impact of transcatheter aortic valve replacement on severity of chronic kidney disease. J Am Coll Cardiol 2020;76:1410–21. 10.1016/j.jacc.2020.07.048 [DOI] [PubMed] [Google Scholar]
- 156. Najjar M, Yerebakan H, Sorabella RA et al. Reversibility of chronic kidney disease and outcomes following aortic valve replacement. Interact Cardiovasc Thorac Surg 2015;21:499–505. 10.1093/icvts/ivv196 [DOI] [PubMed] [Google Scholar]
- 157. Dakroub A, Chung W, Craft J et al. Zero contrast transcatheter aortic valve replacement in patients with renal dysfunction using a novel, multimodality cardiovascular imaging approach. Kidney Int Rep 2023;8:2168–71. 10.1016/j.ekir.2023.07.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Mentias A, Desai MY, Saad M et al. Transcatheter aortic valve replacement in kidney transplant patients. J Am Coll Cardiol 2020;76:2571–3. 10.1016/j.jacc.2020.09.581 [DOI] [PubMed] [Google Scholar]
- 159. Abramowitz Y, Jilaihawi H, Chakravarty T et al. Mitral annulus calcification. J Am Coll Cardiol 2015;66:1934–41. 10.1016/j.jacc.2015.08.872 [DOI] [PubMed] [Google Scholar]
- 160. Bertrand PB, Churchill TW, Yucel E et al. Prognostic importance of the transmitral pressure gradient in mitral annular calcification with associated mitral valve dysfunction. Eur Heart J 2020;41:4321–8. 10.1093/eurheartj/ehaa819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Sud K, Agarwal S, Parashar A et al. Degenerative mitral stenosis: unmet need for percutaneous interventions. Circulation 2016;133:1594–604. 10.1161/CIRCULATIONAHA.115.020185 [DOI] [PubMed] [Google Scholar]
- 162. Zabirnyk A, Ferrer MD, Bogdanova M et al. SNF472, a novel anti-crystallization agent, inhibits induced calcification in an in vitro model of human aortic valve calcification. Vascul Pharmacol 2019;122–123:106583. 10.1016/j.vph.2019.106583 [DOI] [PubMed] [Google Scholar]
- 163. Raggi P, Bellasi A, Bushinsky D et al. Slowing progression of cardiovascular calcification with SNF472 in patients on hemodialysis: results of a randomized phase 2b study. Circulation 2020;141:728–39. 10.1161/CIRCULATIONAHA.119.044195 [DOI] [PubMed] [Google Scholar]
- 164. Zabirnyk A, Perez MDM, Blasco M et al. A novel ex vivo model of aortic valve calcification. A preliminary report. Front Pharmacol 2020;11:568764. 10.3389/fphar.2020.568764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165. Feng W, Teng Y, Zhong Q et al. Biomimetic grapefruit-derived extracellular vesicles for safe and targeted delivery of sodium thiosulfate against vascular calcification. ACS Nano 2023;17:24773–89. 10.1021/acsnano.3c05261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166. Djuric P, Dimkovic N, Schlieper G et al. Sodium thiosulphate and progression of vascular calcification in end-stage renal disease patients: a double-blind, randomized, placebo-controlled study. Nephrol Dial Transplant 2020;35:162–9. 10.1093/ndt/gfz204 [DOI] [PubMed] [Google Scholar]
- 167. Raggi P, Chertow GM, Torres PU et al. The ADVANCE study: a randomized study to evaluate the effects of cinacalcet plus low-dose vitamin D on vascular calcification in patients on hemodialysis. Nephrol Dial Transplant 2011;26:1327–39. 10.1093/ndt/gfq725 [DOI] [PubMed] [Google Scholar]
- 168. Lanting SM, Way KL, Sabag A et al. Degree of adiposity and obesity severity is associated with cutaneous microvascular dysfunction in type 2 diabetes. Microvasc Res 2021;136:104149. 10.1016/j.mvr.2021.104149 [DOI] [PubMed] [Google Scholar]
- 169. Lee GS, Subramanian N, Kim AI et al. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca2+ and cAMP. Nature 2012;492:123–7. 10.1038/nature11588 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Rossol M, Pierer M, Raulien N et al. Extracellular Ca2+ is a danger signal activating the NLRP3 inflammasome through G protein-coupled calcium sensing receptors. Nat Commun 2012;3:1329. 10.1038/ncomms2339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171. Liu W, Zhang X, Zhao M et al. Activation in M1 but not M2 macrophages contributes to cardiac remodeling after myocardial infarction in rats: a critical role of the calcium sensing receptor/NRLP3 inflammasome. Cell Physiol Biochem 2015;35:2483–500. 10.1159/000374048 [DOI] [PubMed] [Google Scholar]
- 172. Phan O, Ivanovski O, Nguyen-Khoa T et al. Sevelamer prevents uremia-enhanced atherosclerosis progression in apolipoprotein E-deficient mice. Circulation 2005;112:2875–82. 10.1161/CIRCULATIONAHA105.541854 [DOI] [PubMed] [Google Scholar]
- 173. Nikolov IG, Joki N, Nguyen-Khoa T et al. Lanthanum carbonate, like sevelamer-HCl, retards the progression of vascular calcification and atherosclerosis in uremic apolipoprotein E-deficient mice. Nephrol Dial Transplant 2012;27:505–13. 10.1093/ndt/gfr254 [DOI] [PubMed] [Google Scholar]
- 174. Raggi P, Bommer J, Chertow GM. Valvular calcification in hemodialysis patients randomized to calcium-based phosphorus binders or sevelamer. J Heart Valve Dis 2004;13:134–41. [PubMed] [Google Scholar]
- 175. Matias PJ, Jorge C, Azevedo A et al. Calcium acetate/magnesium carbonate and cardiovascular risk factors in chronic hemodialysis patients. Nephron 2016;132:317–26. 10.1159/000444421 [DOI] [PubMed] [Google Scholar]
- 176. Ridker PM, Devalaraja M, Baeres FMM et al. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet 2021;397:2060–9. 10.1016/S0140-6736(21)00520-1 [DOI] [PubMed] [Google Scholar]
- 177. Ridker PM. From RESCUE to ZEUS: will interleukin-6 inhibition with ziltivekimab prove effective for cardiovascular event reduction? Cardiovasc Res 2021;117:e138–e40. 10.1093/cvr/cvab231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178. Chertow GM, Chang AM, Felker GM et al. IL-6 inhibition with clazakizumab in patients receiving maintenance dialysis: a randomized phase 2b trial. Nat Med 2024;30:2328–36. 10.1038/s41591-024-03043-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179. Trojanowicz B, Ulrich C, Fiedler R et al. Modulation of leucocytic angiotensin-converting enzymes expression in patients maintained on high-permeable haemodialysis. Nephrol Dial Transplant 2018;33:34–43. 10.1093/ndt/gfx206 [DOI] [PubMed] [Google Scholar]
- 180. Zickler D, Schindler R, Willy K et al. Medium cut-off (MCO) membranes reduce inflammation in chronic dialysis patients—a randomized controlled clinical trial. PLoS ONE 2017;12:e0169024. 10.1371/journal.pone.0169024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181. Zickler D, Willy K, Girndt M et al. High cut-off dialysis in chronic haemodialysis patients reduces serum procalcific activity. Nephrol Dial Transplant 2016;31:1706–12. 10.1093/ndt/gfw293 [DOI] [PubMed] [Google Scholar]
- 182. Willy K, Hulko M, Storr M et al. In Vitro dialysis of cytokine-rich plasma with high and medium cut-off membranes reduces its procalcific activity. Artif Organs 2017;41:803–9. 10.1111/aor.12884 [DOI] [PubMed] [Google Scholar]
- 183. Girndt M, Fiedler R, Martus P et al. High cut-off dialysis in chronic haemodialysis patients. Eur J Clin Invest 2015;45:1333–40. 10.1111/eci.12559 [DOI] [PubMed] [Google Scholar]
- 184. Fiedler R, Neugebauer F, Ulrich C et al. Randomized controlled pilot study of 2 weeks’ treatment with high cutoff membrane for hemodialysis patients with elevated C-reactive protein. Artif Organs 2012;36:886–93. 10.1111/j.1525-1594.2012.01479.x [DOI] [PubMed] [Google Scholar]
- 185. Villa-Bellosta R, Hernández-Martínez E, Mérida-Herrero E et al. Impact of acetate- or citrate-acidified bicarbonate dialysate on ex vivo aorta wall calcification. Sci Rep 2019;9:11374. 10.1038/s41598-019-47934-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186. Lorenz G, Mayer CC, Bachmann Q et al. Acetate-free, citrate-acidified bicarbonate dialysis improves serum calcification propensity—a preliminary study. Nephrol Dial Transplant 2018;33:2043–51. 10.1093/ndt/gfy134 [DOI] [PubMed] [Google Scholar]
- 187. Saritas T, Reinartz S, Krüger T et al. Vitamin K1 and progression of cardiovascular calcifications in hemodialysis patients: the VitaVasK randomized controlled trial. Clin Kidney J 2022;15:2300–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188. De Vriese AS, Caluwé R, Pyfferoen L et al. Multicenter randomized controlled trial of vitamin K antagonist replacement by Rivaroxaban with or without vitamin K2 in hemodialysis patients with atrial fibrillation: the Valkyrie Study. J Am Soc Nephrol 2020;31:186–96. 10.1681/ASN.2019060579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189. Ferrer MD, Pérez MM, Cànaves MM et al. A novel pharmacodynamic assay to evaluate the effects of crystallization inhibitors on calcium phosphate crystallization in human plasma. Sci Rep 2017;7:6858. 10.1038/s41598-017-07203-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190. Ferrer MD, Ketteler M, Tur F et al. Characterization of SNF472 pharmacokinetics and efficacy in uremic and non-uremic rats models of cardiovascular calcification. PLoS ONE 2018;13:e0197061. 10.1371/journal.pone.0197061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191. Bushinsky DA, Raggi P, Bover J et al. Effects of myo-inositol hexaphosphate (SNF472) on bone mineral density in patients receiving hemodialysis: an analysis of the randomized, placebo-controlled CaLIPSO study. Clin J Am Soc Nephrol 2021;16:736–45. 10.2215/CJN.16931020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192. Cicone JS, Petronis JB, Embert CD et al. Successful treatment of calciphylaxis with intravenous sodium thiosulfate. Am J Kidney Dis 2004;43:1104–8. 10.1053/j.ajkd.2004.03.018 [DOI] [PubMed] [Google Scholar]
- 193. Araya CE, Fennell RS, Neiberger RE et al. Sodium thiosulfate treatment for calcific uremic arteriolopathy in children and young adults. Clin J Am Soc Nephrol 2006;;1:1161–6. 10.2215/CJN.01520506 [DOI] [PubMed] [Google Scholar]
- 194. Ackermann F, Levy A, Daugas E et al. Sodium thiosulfate as first-line treatment for calciphylaxis. Arch Dermatol 2007;143:1336–7; author reply 1338. 10.1001/archderm.143.10.1336 [DOI] [PubMed] [Google Scholar]
- 195. Schlieper G, Brandenburg V, Ketteler M et al. Sodium thiosulfate in the treatment of calcific uremic arteriolopathy. Nat Rev Nephrol 2009;5:539–43. 10.1038/nrneph.2009.99 [DOI] [PubMed] [Google Scholar]
- 196. Lu Y, Shen L, Zhou L et al. Success of small-dose fractionated sodium thiosulfate in the treatment of calciphylaxis in a peritoneal dialysis patient. BMC Nephrol 2022;23:4. 10.1186/s12882-021-02648-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197. Karageuzyan KG. Oxidative stress in the molecular mechanism of pathogenesis at different diseased states of organism in clinics and experiment. Curr Drug Targets Inflamm Allergy 2005;4:85–98. 10.2174/1568010053622939 [DOI] [PubMed] [Google Scholar]
- 198. Wen W, Portales-Castillo I, Seethapathy R et al. Intravenous sodium thiosulphate for vascular calcification of hemodialysis patients—a systematic review and meta-analysis. Nephrol Dial Transplant 2023;38:733–45. 10.1093/ndt/gfac171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199. Huang C, Duan Z, Xu C et al. Influence of sodium thiosulfate on coronary artery calcification of patients on dialysis: a meta-analysis. Ren Fail 2023;45:2254569. 10.1080/0886022X.2023.2254569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200. Adirekkiat S, Sumethkul V, Ingsathit A et al. Sodium thiosulfate delays the progression of coronary artery calcification in haemodialysis patients. Nephrol Dial Transplant 2010;25:1923–9. 10.1093/ndt/gfp755 [DOI] [PubMed] [Google Scholar]
- 201. Wen W, Portales-Castillo I, Seethapathy R et al. Intravenous sodium thiosulphate for calciphylaxis of chronic kidney disease: a systematic review and meta-analysis. JAMA Netw Open 2023;6:e2310068. 10.1001/jamanetworkopen.2023.10068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202. Mary A, Objois T, Brazier M et al. Decreased monocyte calcium sensing receptor expression in patients with chronic kidney disease is associated with impaired monocyte ability to reduce vascular calcification. Kidney Int 2021;99:1382–91. 10.1016/j.kint.2021.01.026 [DOI] [PubMed] [Google Scholar]
- 203. Olszak IT, Poznansky MC, Evans RH et al. Extracellular calcium elicits a chemokinetic response from monocytes in vitro and in vivo. J Clin Invest 2000;105:1299–305. 10.1172/JCI9799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204. EVOLVE Trial Investigators, Chertow GM, Block GA, Correa-Rotter R et al. Effect of cinacalcet on cardiovascular disease in patients undergoing dialysis. N Engl J Med 2012;367:2482–94. [DOI] [PubMed] [Google Scholar]
- 205. Bäck M, Michel JB. From organic and inorganic phosphates to valvular and vascular calcifications. Cardiovasc Res 2021;117:2016–29. 10.1093/cvr/cvab038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206. Candellier A, Diouf M, Bohbot Y et al. Association between serum phosphate levels and progression of aortic stenosis. JACC: Advances 2024;3:101096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207. Zhang H, Li G, Yu X et al. Progression of vascular calcification and clinical outcomes in patients receiving maintenance dialysis. JAMA Netw Open 2023;6:e2310909. 10.1001/jamanetworkopen.2023.10909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208. Jamal SA, Vandermeer B, Raggi P et al. Effect of calcium-based versus non-calcium-based phosphate binders on mortality in patients with chronic kidney disease: an updated systematic review and meta-analysis. Lancet 2013;382:1268–77. 10.1016/S0140-6736(13)60897-1 [DOI] [PubMed] [Google Scholar]
- 209. Ogata H, Fukagawa M, Hirakata H et al. Effect of treating hyperphosphatemia with lanthanum carbonate vs calcium carbonate on cardiovascular events in patients with chronic kidney disease undergoing hemodialysis: the LANDMARK randomized clinical trial. JAMA 2021;325:1946–54. 10.1001/jama.2021.4807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210. Ogata H, Fukagawa M, Hirakata H et al. LANDMARK Investigators and Committees . Effect of lanthanum carbonate and calcium carbonate on the progression of coronary artery calcification among hemodialysis patients with vascular calcification risk: a randomized controlled trial. Clin Exp Nephrol 2022;26:1223–32. 10.1007/s10157-022-02270-5 [DOI] [PubMed] [Google Scholar]
- 211. Toussaint ND, Pedagogos E, Lioufas NM et al. A randomized trial on the effect of phosphate reduction on vascular end points in CKD (IMPROVE-CKD). J Am Soc Nephrol 2020;31:2653–66. 10.1681/ASN.2020040411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212. Isaka Y, Hamano T, Fujii H et al. Optimal phosphate control related to coronary artery calcification in dialysis patients. J Am Soc Nephrol 2021;32:723–35. 10.1681/ASN.2020050598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213. Thériault S, Dina C, Messika-Zeitoun D et al. Genetic association analyses highlight IL6, ALPL, and NAV1 As 3 new susceptibility genes underlying calcific aortic valve stenosis. Circ Genom Precis Med 2019;12:e002617. 10.1161/CIRCGEN.119.002617 [DOI] [PubMed] [Google Scholar]
- 214. El Husseini D, Boulanger MC, Mahmut A et al. P2Y2 receptor represses IL-6 expression by valve interstitial cells through Akt: implication for calcific aortic valve disease. J Mol Cell Cardiol 2014;72:146–56. 10.1016/j.yjmcc.2014.02.014 [DOI] [PubMed] [Google Scholar]
- 215. Côté N, Mahmut A, Fournier D et al. Angiotensin receptor blockers are associated with reduced fibrosis and interleukin-6 expression in calcific aortic valve disease. Pathobiology 2014;81:15–24. 10.1159/000350896 [DOI] [PubMed] [Google Scholar]
- 216. Ciofani JL, Han D, Nazarzadeh M et al. The effect of immunomodulatory drugs on aortic stenosis: a mendelian randomisation analysis. Sci Rep 2023;13:18810. 10.1038/s41598-023-44387-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217. Barreto DV, Barreto FC, Liabeuf S et al. Plasma interleukin-6 is independently associated with mortality in both hemodialysis and pre-dialysis patients with chronic kidney disease. Kidney Int 2010;77:550–6. 10.1038/ki.2009.503 [DOI] [PubMed] [Google Scholar]
- 218. Vachey C, Candellier A, Toutain S et al. The bone-vascular axis in chronic kidney disease: from pathophysiology to treatment. Curr Osteoporos Rep 2024;22:69–79. 10.1007/s11914-023-00858-8 [DOI] [PubMed] [Google Scholar]
- 219. Diederichsen ACP, Lindholt JS, Möller S et al. Vitamin K2 and D in patients with aortic valve calcification: a randomized double-blinded clinical trial. Circulation 2022;145:1387–97. 10.1161/CIRCULATIONAHA.121.057008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220. Kaesler N, Schreibing F, Speer T et al. Altered vitamin K biodistribution and metabolism in experimental and human chronic kidney disease. Kidney Int 2022;101:338–48. 10.1016/j.kint.2021.10.029 [DOI] [PubMed] [Google Scholar]
- 221. Lees JS, Chapman FA, Witham MD et al. Vitamin K status, supplementation and vascular disease: a systematic review and meta-analysis. Heart 2019;105:938–45. [DOI] [PubMed] [Google Scholar]
- 222. Kurnatowska I, Grzelak P, Masajtis-Zagajewska A et al. Effect of vitamin K2 on progression of atherosclerosis and vascular calcification in nondialyzed patients with chronic kidney disease stages 3-5. Pol Arch Med Wewn 2015;125:631–40. 10.20452/pamw.3041 [DOI] [PubMed] [Google Scholar]
- 223. Witham MD, Lees JS, White M et al. Vitamin K supplementation to improve vascular stiffness in CKD: the K4Kidneys randomized controlled trial. J Am Soc Nephrol 2020;31:2434–45. 10.1681/ASN.2020020225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224. Oikonomaki T, Papasotiriou M, Ntrinias T et al. The effect of vitamin K2 supplementation on vascular calcification in haemodialysis patients: a 1-year follow-up randomized trial. Int Urol Nephrol 2019;51:2037–44. 10.1007/s11255-019-02275-2 [DOI] [PubMed] [Google Scholar]
- 225. Levy-Schousboe K, Frimodt-Møller M, Hansen D et al. Vitamin K supplementation and arterial calcification in dialysis: results of the double-blind, randomized, placebo-controlled RenaKvit trial. Clin Kidney J 2021;14:2114–23. 10.1093/ckj/sfab017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226. Cowell SJ, Newby DE, Prescott RJ et al. A randomized trial of intensive lipid-lowering therapy in calcific aortic stenosis. N Engl J Med 2005;352:2389–97. 10.1056/NEJMoa043876 [DOI] [PubMed] [Google Scholar]
- 227. Rossebø AB, Pedersen TR, Boman K et al. Intensive lipid lowering with simvastatin and ezetimibe in aortic stenosis. N Engl J Med 2008;359:1343–56. 10.1056/NEJMoa0804602 [DOI] [PubMed] [Google Scholar]
- 228. Chan KL, Teo K, Dumesnil JG et al. ASTRONOMER Investigators . Effect of lipid lowering with rosuvastatin on progression of aortic stenosis: results of the Aortic Stenosis Progression Observation: Measuring Effects of Rosuvastatin (ASTRONOMER) Trial. Circulation 2010;121:306–14. 10.1161/CIRCULATIONAHA.109.900027 [DOI] [PubMed] [Google Scholar]
- 229. Xinyu Z, Dongxia M, Yue H et al. Statins accelerate coronary calcification and reduce the risk of cardiovascular events. Cardiol Rev 2023;31:293–8. 10.1097/CRD.0000000000000438 [DOI] [PubMed] [Google Scholar]
- 230. Koutsogianni AD, Liamis G, Liberopoulos E et al. Effects of lipid-modifying and other drugs on lipoprotein(a) levels-potent clinical implications. Pharmaceuticals 2023;16:750. 10.3390/ph16050750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231. Charytan DM, Sabatine MS, Pedersen TR et al. Efficacy and safety of Evolocumab in chronic kidney disease in the FOURIER trial. J Am Coll Cardiol 2019;73:2961–70. 10.1016/j.jacc.2019.03.513 [DOI] [PubMed] [Google Scholar]
- 232. Capoulade R, Chan KL, Yeang C et al. Oxidized phospholipids, lipoprotein(a), and progression of calcific aortic valve stenosis. J Am Coll Cardiol 2015;66:1236–46. 10.1016/j.jacc.2015.07.020 [DOI] [PubMed] [Google Scholar]
- 233. Tsimikas S, Karwatowska-Prokopczuk E, Gouni-Berthold I et al. Lipoprotein(a) reduction in persons with cardiovascular disease. N Engl J Med 2020;382:244–55. 10.1056/NEJMoa1905239 [DOI] [PubMed] [Google Scholar]
- 234. Sosnowska B, Surma S, Banach M. Targeted treatment against lipoprotein (a): the coming breakthrough in lipid lowering therapy. Pharmaceuticals 2022;15:1573. 10.3390/ph15121573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235. Fernandez-Prado R, Perez-Gomez MV, Ortiz A. Pelacarsen for lowering lipoprotein(a): implications for patients with chronic kidney disease. Clin Kidney J 2020;13:753–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236. Wang C, Jiang L, Feng S et al. Risk factor analysis of calcification in aortic and mitral valves in maintenance peritoneal dialysis patients. Kidney Blood Press Res 2013;37:488–95. 10.1159/000355729 [DOI] [PubMed] [Google Scholar]
- 237. Elewa M, Mitra S, Jayanti A. Left-sided valvular heart disease in dialysis recipients: a single-centre observational study. Clin Kidney J 2023;16:1092–101. 10.1093/ckj/sfad020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238. Holden RM, Sanfilippo AS, Hopman WM et al. Warfarin and aortic valve calcification in hemodialysis patients. J Nephrol 2007;20:417–22. [PubMed] [Google Scholar]
- 239. Raggi P, Bellasi A, Gamboa C et al. All-cause mortality in hemodialysis patients with heart valve calcification. Clin J Am Soc Nephrol 2011;6:1990–5. 10.2215/CJN.01140211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240. Rroji M, Seferi S, Cafka M et al. Is residual renal function and better phosphate control in peritoneal dialysis an answer for the lower prevalence of valve calcification compared to hemodialysis patients? Int Urol Nephrol 2014;46:175–82. 10.1007/s11255-013-0438-7 [DOI] [PubMed] [Google Scholar]
- 241. Candellier A, Hénaut L, Morelle J et al. Aortic stenosis in patients with kidney failure: is there an advantage for a PD-first policy? Perit Dial Int 2021;41:158–67. 10.1177/0896860820941371 [DOI] [PubMed] [Google Scholar]
- 242. Ogugua FM, Mathew RO, Ternacle J et al. Impact of arteriovenous fistula on flow states in the evaluation of aortic stenosis among ESKD patients on dialysis. Echocardiography 2024;41:e15728. 10.1111/echo.15728 [DOI] [PubMed] [Google Scholar]
- 243. Basile C, Lomonte C, Vernaglione L et al. The relationship between the flow of arteriovenous fistula and cardiac output in haemodialysis patients. Nephrol Dial Transplant 2008;23:282–7. 10.1093/ndt/gfm549 [DOI] [PubMed] [Google Scholar]
- 244. Kim GY, Lee SH, Kim SH et al. Temporary arteriovenous fistula compression for clinical decision-making in patients on hemodialysis with significant aortic stenosis. J Cardiovasc Imaging 2023;31:118–20. 10.4250/jcvi.2022.0088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245. Baratchi S, Zaldivia MTK, Wallert M et al. Transcatheter aortic valve implantation represents an anti-inflammatory therapy via reduction of shear stress-induced, piezo-1-mediated monocyte activation. Circulation 2020;142:1092–105. 10.1161/CIRCULATIONAHA.120.045536 [DOI] [PubMed] [Google Scholar]
- 246. de la Espriella R, Romero-González G, Núñez J. Valvular heart disease in patients on kidney replacement therapy: “opening Pandora's box”. Clin Kidney J 2023;16:1045–8. 10.1093/ckj/sfad060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247. Rosner MH, Reis T, Husain-Syed F et al. Classification of uremic toxins and their role in kidney failure. Clin J Am Soc Nephrol 2021;16:1918–28. 10.2215/CJN.02660221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248. Ferdous Z, Jo H, Nerem RM. Differences in valvular and vascular cell responses to strain in osteogenic media. Biomaterials 2011;32:2885–93. 10.1016/j.biomaterials.2011.01.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were generated or analysed in support of this research.