Abstract
Background
The Clinical Treatment Score post-5 years (CTS5) is a clinicopathological tool designed to estimate late distant recurrence (LDR) in hormone receptor-positive (HR+) breast cancer patients after 5 years of adjuvant endocrine therapy (ET). While intended as a prognostic algorithm, its predictive value for ET extension remains uncertain.
Methods
The score was calculated in 4931 patients from four prospective randomized ABCSG trials (ABCSG-6, -6a, -8, and -16) with 250 LDR events. We assessed its prognostic power, calibration accuracy, and predictive value. Time to LDR was analyzed using Cox regression models.
Results
In our cohorts, the CTS5 provided prognostic information whether used as a continuous or categorical score. In the ABCSG-8 cohort (n = 2054) and the combined ABCSG-6+8 cohort (n = 3308), a higher continuous score was significantly associated with increased LDR risk. The categorical CTS5 showed that high-risk patients had significantly higher LDR rates compared to low- or intermediate-risk patients. The score slightly overestimated LDR risk, regardless of predicted risk. Although no significant predictive value was found on the relative scale, an absolute LDR risk reduction of 23.4 % was found in patients with a high CTS5 of 5 when extended ET was administered additional five than two years. In patients with a CTS5 of 2, no benefit was found when ET was extended to 10 instead of 7 years.
Conclusion
The CTS5 is a valid tool for LDR risk stratification in HR + breast cancer, but should be used cautiously for determining benefits from ET extension, as no significant predictive value was found.
Keywords: CTS5, Adjuvant setting, Endocrine therapy, Predictive value, Late distant recurrence
Highlights
-
•
The CTS5 is an easy-to-use tool predicting late distant recurrence in women with hormone receptor-positive BC after 5 years of endocrine therapy.
-
•
The score slightly tends to overestimate late distant recurrence risk.
-
•
The CTS5 should be used cautiously to evaluate the benefit of extended endocrine therapy, as its predictive value could not be confirmed in our trial.
1. Introduction
The need for a tool to reliably estimate the risk of late distant recurrence (LDR) is especially high in patients with hormone receptor-positive breast cancer (HR + BC). It is known that recurrence can occur over decades, even 30 years or more after primary diagnosis [1]. Larger tumor size and nodal-positivity are associated with an increased risk of LDR [1,2], which can be reduced with extended adjuvant therapy [[3], [4], [5]]. Multigenomic tests, which promise to reliably predict the risk, are expensive, often not easily available, and do not deliver results immediately. The Clinical Treatment Score post-5 years (CTS5) is an easy-to-use and cost-free tool that estimates the LDR risk after 5 years of endocrine therapy (ET) in women with HR + BC [[6], [7], [8], [9], [10], [11], [12]]. The CTS5 calculator offers the advantage of being an online tool, providing immediate results facilitating prompt decision-making in contrast to multigenomic tests. We recently validated the score in the ABCSG-6 and -6a trial, confirming that it is a highly prognostic score, which is well calibrated in low- and intermediate-risk groups but is less accurate in high-risk patients [13]. In the latter case, higher distant recurrences were expected than observed, which was previously also described by Noordhoek et al. in the TEAM and IDEAL trials [6]. Similarly, in a systematic review including 10,425 premenopausal and 30,354 postmenopausal patients, the CTS5 reliably predicted LDR in both cohorts, but overestimated LDR events in high-risk patients [11].
However, regarding the score's predictive value - the potential to identify patients who will benefit from extended ET more than others - controversial results were found [7,8]. In the ABCSG-6a trial, we observed that high-risk patients seemed to benefit more from extending ET than low-risk patients, but the statistical significance for this finding was lacking [13]. Low numbers of high-risk patients as well as of LDR events in our study cohort might be causal for the lack of significance. Therefore, we hereby present a further validation of the CTS5 in a larger patient collective.
2. Methods
The calculation of the CTS5 was performed according to the paper published by Dowsett et al., in 2018 by using the formula:
CTS5 = 0.438 × nodes +0.988 × (0.093 × size + 0.001 × size2 + 0.375 × grade + 0.017 × age).
The separation into three groups was created by using the pre-defined cut-off values [12]. The three risk groups were defined as risk of DR in the years 5–10 in: low (<5 %), intermediate (5 %–10 %), and high risk (>10 %).
At first, the score was calculated in order to evaluate its prognostic (ABCSG-8 cohort) and predictive value (ABCSG-16) in pure randomized controlled trials. To enable an investigation in larger patient cohorts, the score was also tested in combined cohorts with ABCSG-6 and -6a, respectively. Therefore, all four studies will be described shortly below.
In the ABCSG-6 trial, 2020 postmenopausal patients were randomly assigned to five years of tamoxifen with or without concomitant aminoglutethimide for the first two years of ET [14]. After 5 years, 860 recurrence-free patients were prospectively randomized in the ABCSG-6a trial to additional 3 years of anastrozole or no further therapy [15].
The ABCSG-8 trial included 3901 postmenopausal patients with HR + BC in the adjuvant setting. After completion of 2 years of adjuvant tamoxifen, patients were randomly assigned to either 3 years of anastrozole or further 3 years of tamoxifen [16].
The ABCSG-16 trial included 3484 postmenopausal women with HR + BC who had received 5 years of adjuvant ET and were further randomized to either 2 or 5 years of additional anastrozole treatment [17].
The primary endpoint was to evaluate the prognostic performance of the continuous CTS5 score in patients who underwent 5 years of adjuvant ET without therapy extension. This was tested in patients of the ABCSG-8 and the combined ABCSG-6+8 cohorts. Secondary endpoints included the evaluation of the CTS5 risk categories, the calibration performance, and as an exploratory endpoint, the prognostic performance of N- and anatomic stage in ABCSG-8 as well in the combined ABCSG-6+8 cohort.
The predictive performance of the score was tested in the ABCSG-16 trial. Additionally, the calculation of the score in the ABCSG-6a+16 and the ABCSG-6+8 cohorts enabled a comparison of the predictive values in cohorts with and without extended ET.
The statistical endpoint is time to LDR, defined as metastatic disease excluding locoregional, ipsilateral or contralateral recurrence after 5 years of ET. Patients, who died without a prior DR, were censored at the time of death. Hence, the estimated risk reflects the “pure” LDR risk (i.e. had patients not died). The 5-year LDR risk starts after completion of 5 years of ET, therefore refers to 10 years after randomization to the respective trial.
2.1. Statistical analysis
The prognostic performance of the score was tested with univariable Cox regression models and Kaplan Meier estimation.
The calibration power was assessed via calibration plot comparing the predicted risk based on CTS5 scores versus the observed risk estimated with Kaplan Meier methodology. Based on the number of events, 4 or 5 equally sized groups of patients with similar CTS5 scores - based on predicted risk - were created to have enough precision of the observed rates. Adding more groups would have resulted in very large confidence intervals. Summary measures of the predicted DR risk within each quartile or quantile group were shown and compared to the estimated DR risk.
The predictive performance of CTS5 was tested by using a Cox model with interaction term. The interaction effect was assessed on the relative (HR scale) and on the absolute scale (difference in LDR risk estimates).
As an exploratory analysis the prognostic performance of the N- and anatomic stage was tested with univariable Cox regression models and Kaplan Meier estimation. A bivariable model was derived to compare the prognostic performance of the CTS5 with the prognostic information that the N- or anatomic stage provided.
3. Results
Out of 3901 patients included in the ABCSG-8 trial, 3784 patients had valid data for CTS5 calculation. Of those, 3287 patients were event-free and still in follow-up at the 5-year follow up. Of those, 1233 patients were enrolled in the ABCSG-16 trial, the other 2054 were therefore included in the primary endpoint-analysis testing the prognostic performance of the continuous CTS5. Furthermore, 784 patients from ABCSG-6 and 470 patients from the control arm of ABCSG-6a, who did not receive extended ET were included in this analysis (Fig. 1).
Fig. 1.
Patient flow of the patients included in this analysis.
3.1. The prognostic performance
Patient characteristics of the ABCSG-8 cohort (n = 2054) are shown in Table 1. A total of 89 LDR events were recorded. An increasing CTS5 was significantly associated with an increased LDR risk (HR 2.81; 95%CI, 2.12–3.73; p < 0.0001). When censoring at year 10, the continuous CTS5 was prognostic (HR 3.07; 95%CI, 2.23–4.21; p < 0.0001) in this study cohort. In the combined study population of ABCSG-6+8 patients (n = 3308; 168 LDR events), the score remained prognostic with a slightly decreased HR of 2.32 (95%CI, 1.94–2.77; p < 0.0001). A sensitivity analysis with study cohort as a stratification factor revealed that the HR effect sizes were marginally different between the cohorts, but a potential differential effect between ABCSG-6 and -8 could not be detected (interaction p-value 0.22).
Table 1.
Patient's characteristics of the main analysis population consisting of 2054 patients from ABCSG-8 without extended ET.
| Low risk N = 1159 |
Intermediate risk N = 661 |
High risk N = 234 |
Total N = 2054 |
|
|---|---|---|---|---|
| Age (years) | ||||
| Mean (SD) | 63.5 (8.2) | 66.9 (7.9) | 67.8 (7.7) | 65.1 (8.2) |
| Median (Q1-Q3) | 63.0 (57.0–70.0) | 67.0 (61.0–74.0) | 68.5 (62.0–75.0) | 65.0 (58.0–72.0) |
| Min - Max | 41 to 80 | 45 to 80 | 49 to 80 | 41 to 80 |
| Anatomic stage | ||||
| IA | 1081 (93.3 %) | 150 (22.7 %) | 0 | 1231 (59.9 %) |
| IIA | 78 (6.7 %) | 463 (70.0 %) | 49 (20.9 %) | 590 (28.7 %) |
| IIB | 0 | 42 (6.4 %) | 123 (52.6 %) | 165 (8.0 %) |
| IIIA | 0 | 6 (0.9 %) | 62 (26.5 %) | 68 (3.3 %) |
| Tumor size (cm) | ||||
| Mean (SD) | 1.2 (0.4) | 2.1 (0.8) | 2.7 (1.2) | 1.7 (0.9) |
| Median (Q1-Q3) | 1.2 (0.9–1.4) | 2.0 (1.6–2.5) | 2.5 (2.0–3.1) | 1.5 (1.1–2.0) |
| Min - Max | 0.2 to 4.0 | 0.6 to 10.0 | 1.2 to 10.0 | 0.2 to 10.0 |
| T-stage | ||||
| pT1a | 4 (0.3 %) | 0 | 0 | 4 (0.2 %) |
| pT1b | 447 (38.6 %) | 18 (2.7 %) | 0 | 465 (22.6 %) |
| pT1c | 691 (59.6 %) | 309 (46.7 %) | 59 (25.2 %) | 1059 (51.6 %) |
| pT2 | 17 (1.5 %) | 327 (49.5 %) | 164 (70.1 %) | 508 (24.7 %) |
| pT3 | 0 | 7 (1.1 %) | 11 (4.7 %) | 18 (0.9 %) |
| Positive nodes (n) | ||||
| Mean (SD) | 0.1 (0.3) | 0.5 (0.9) | 2.6 (1.9) | 0.5 (1.1) |
| Median (Q1-Q3) | 0.0 (0.0–0.0) | 0.0 (0.0–1.0) | 2.0 (1.0–4.0) | 0.0 (0.0–0.0) |
| Min - Max | 0.0 to 3.0 | 0.0 to 9.0 | 0.0 to 9.0 | 0.0 to 9.0 |
| N-stage | ||||
| pN0 | 1098 (94.7 %) | 449 (67.9 %) | 16 (6.8 %) | 1563 (76.1 %) |
| pN1 | 61 (5.3 %) | 206 (31.2 %) | 159 (67.9 %) | 426 (20.7 %) |
| pN2 | 0 | 6 (0.9 %) | 59 (25.2 %) | 65 (3.2 %) |
| Grade | ||||
| G1 | 345 (29.8 %) | 57 (8.6 %) | 9 (3.8 %) | 411 (20.0 %) |
| G2 | 771 (66.5 %) | 563 (85.2 %) | 209 (89.3 %) | 1543 (75.1 %) |
| GX | 43 (3.7 %) | 41 (6.2 %) | 16 (6.8 %) | 100 (4.9 %) |
| CTS5 Score | ||||
| Mean (SD) | 2.6 (0.3) | 3.5 (0.2) | 4.3 (0.4) | 3.1 (0.6) |
| Median (Q1-Q3) | 2.7 (2.4–2.9) | 3.4 (3.3–3.6) | 4.2 (4.0–4.5) | 3.0 (2.6–3.5) |
| Min - Max | 1.8 to 3.1 | 3.1 to 3.9 | 3.9 to 5.2 | 1.8 to 5.2 |
The prognostic performance of the categorical CTS5 was tested in the same data sets. In ABCSG-8, 234 patients were assigned to the high-, 661 to the intermediate- and 1159 to the low-risk group. High-risk patients had a significantly higher LDR risk than low-risk patients (HR 6.13; 95%CI, 3.59–10.49; p < 0.0001) and intermediate-risk patients had a significant higher LDR risk than low-risk patients (HR 2.73; 1.62–4.59; p = 0.0002). The 5- to 10-year LDR-free survival rates were 85.0 % (95%CI, 78.4 %,89.6 %) for high-, 94.2 % (95%CI, 91.4 %,96.0 %) for intermediate- and 97.6 % (95%CI, 96.1 %,98.5 %) for low-risk patients. The KM curves are shown in Fig. 2. 10- to 15-year LDR-free survival rates of 78.0 % (95%CI, 68.1 %,85.1 %) in the high-, 86.9 % (95%CI, 80.2 %,91.4 %) in the intermediate- and 95.1 % (95%CI, 92.0 %,97.0 %) in the low-risk group were observed.
Fig. 2.
5- to 10-year DR-free survival curves according to the CTS5 risk categories in patients from ABCSG-8 (n = 2054).
The score was also of prognostic value when tested in the combined ABCSG-6+8 cohort (n = 3308; 168 LDR events). In those patients, 541 were assigned to the high-, 1101 to the intermediate-, and 1666 to the low-risk group. The score was able to significantly discriminate high- and low-risk patients (HR 4.79; 95%CI, 3.24–7.07; p < 0.0001) as well as intermediate- and low-risk patients (HR 2.32; 95%CI, 1.56–3.45; p < 0.0001). The corresponding KM curves are provided in the Suppl. Fig. 1.
3.2. The calibration accuracy
The calibration accuracy of the CTS5 was tested in patients without extended ET in the ABCSG-8 cohort (n = 2054) and in the combined ABCSG-6+8 cohort (n = 3308).
In the ABCSG-8 cohort, only four rather than five equally sized groups were created due to sample size limitations.
In the first three CTS5-quartiles, the predicted LDR risk was higher than the observed risk (median predicted risk = 2.4 %, 3.7 %, and 5.5 % vs. estimated KM risk = 2.0 %, 2.6 %, and 4.0 %, respectively). In the highest CTS5-quartile, the predicted LDR-risk was lower than the observed risk (median predicted risk = 9.6 %, KM risk = 11.6 %). KM curves are shown in Suppl.Fig. 2.
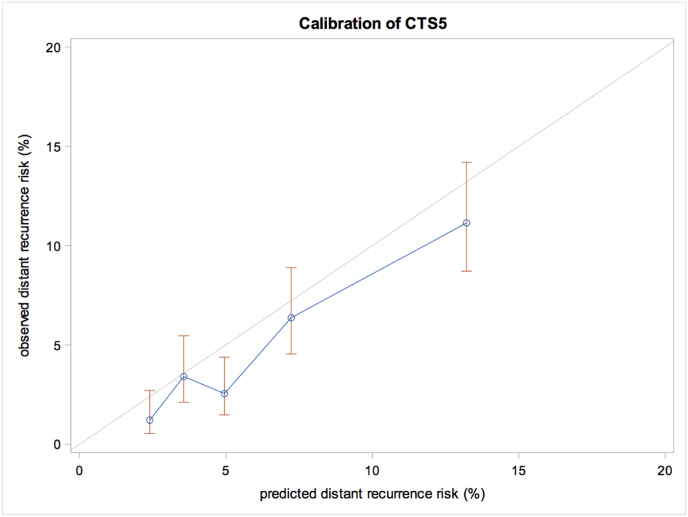
In the combined ABCSG-6+8 cohort with 3308 patients, five CTS5-quantils could be derived. In all five quantiles the predicted risk of LDR was slightly higher than the observed risk. The difference between predicted and observed risk varied from −0.2 % to −2.4 % (Fig. 3).
Fig. 3.
Calibration plot of the CTS5 comparing the observed with the predicted LDR risk when the population of ABCSG-6+8 (n = 3308) was divided into quintiles according to the CTS5 score.
3.3. The predictive performance
The predictive performance was tested for two versus five additional years of ET within the ABCSG-16 cohort (n = 1233; 66 events). The predictive performance of the continuous CTS5 could not be confirmed on the relative scale (interaction p-value 0.47), although there was a numerical trend favoring additional five years of ET in patients with higher CTS5 values. On the absolute scale, this trend was more obvious: LDR rates of patients with a CTS5 of 5 were reduced by 23.4 % when ET was extended for five rather than for two years (5- to 10-year LDR rates in patients with five additional years of ET: 16.2 % (95%CI, 0.0%–30.7 %) versus with two additional years of ET: 39.6 % (95%CI, 9.4%–59.7 %), see Fig. 4). Regarding the categorical CTS5, similar results were found. In detail, a trend for beneficial outcome was observed on the relative scale in the high-risk group when ET was administered for another five years. Although the HR favoring five additional years of ET in high-risk patients was remarkably low (HR 0.57 (95%CI, 0.19, 1.69), the level of significance wasn't reached (interaction p = 0.67). On the absolute scale, a LDR risk reduction in high-risk patients of −11.6 % (95%CI, −25.3 %, 2.1 %) was found when ET was administered for a total of ten years compared to seven years (5-to 10-year LDR rate in patients with five additional years of ET: 6.7 % (95%CI, 2.2 %, 19.4 %) versus two additional years: 18.3 % (95%CI, 9.6 %,33.5 %)). In intermediate-risk patients a difference of −3.5 % (95%CI, −8.7 %, 1.7 %) LDR risk was found between patients with additional five than two years of ET (5-to 10-year LDR in patients with five additional years of ET: 4.4 % (2.1 %,9.0 %) versus with two additional years: 7.9 % (4.6 %,13.2 %)). In low-risk patients the LDR rates were only −0.5 % (95%CI, −2.2 %, 1.1 %) lower when ET was administered additionally five than two years (5- to 10-year LDR in patients with five additional years of ET: 1.1 % (0.4 %,2.8 %) versus with two additional years: 1.6 % (0.7 %,3.5 %)).
Fig. 4.
Five-year LDR risk (starting at randomization to ABCSG-16) according to the continuous CTS5 in patients with either 2 years or 5 years of additional AI therapy. The confidence bands reflect the 95 % pointwise confidence intervals. The semi-transparent bars reflect the numbers of patients in each CTS5 value group.
In order to analyze the CTS5's predictive value in the largest possible cohort, data of 4931 patients with 250 events of the ABCSG-6+8 (non-extended ET) and the ABCSG-6a+16 (extended ET) cohorts were analyzed. Regarding the continuous score, no significant predictive value was found on the relative scale (interaction p-value 0.71). With an HR of 0.72 (95%CI, 0.40, 1.28) in patients with a CTS5 of 2 and an HR of 0.86 (95%CI, 0.50, 1.49) in patients with a CTS5 of 5, a numerical stronger relative benefit of extended ET was found in patients with lower CTS5 values whereas a slightly weaker relative effect was observed in patients with a higher score. On the absolute scale, however, patients with a lower CTS5 score seemed to benefit less from extending ET. The greatest benefit with −2.5 % (95%CI, −10.6, 5.6) in LDR rates was found in patients with a CTS5 of 5 and extended ET (5-to-10-year LDR rate extended ET: 14.8 % (95%CI, 7.8 %,21.2 %) versus non-extended: 17.3 % (95%CI, 12.7 %,21.7 %), see Suppl. Fig. 3). Regarding the categorical score, again, no predictive value was observed on the relative scale (interaction p-value 0.55). On the absolute scale, the 5-to-10-year LDR rate was −1.8 % lower in high-risk patients who received extended ET than in those who did not referring to a marginal benefit of extended ET (5- to 10-year LDR extended ET: 9.8 % (95%CI, 6.2%–15.4 %) versus non-extended ET: 11.6 % (95%CI, 8.9%–15.0 %)). Even in low-risk patients, LDR rates were −1.4 % (95%CI, −2.5, −0.3) lower when ET was extended (5-to-10-years LDR rate extended ET 1.1 % (95%CI, 0.6 %, 2.0 %) versus non-extended: 2.4 % (95%CI, 1.7 %, 3.5 %). In contrast, between intermediate-risk risk patients with or without extended ET, no remarkable difference in 5-to-10-year LDR rates were found (extended: 5.4 % (95%CI, 4.1 %,7.2 %) versus non-extended: 5.1 % (95%CI, 3.4 %,7.5 %)).
3.4. Exploratory analyses
As exploratory endpoints, the prognostic information of the N-stage as well as the anatomic stage was investigated. In a bivariable model, we observed in the ABCSG-8 and in the combined ABCSG-6+8 cohorts that the CTS5 provided prognostic information beyond the N- or anatomic stage. In contrast, if the CTS5 was known, the N- or anatomic stage were not able to provide additional prognostic information. Only in the ABCSG-6 cohort (n = 1254) were the results reversed. In detail, in this cohort the CTS5 could not show independent prognostic information when N- or anatomic stage were known (data is shown in Suppl. Tables 1 and 2 a and b).
The power of N- and anatomic stage to predict the benefit of extended ET was tested in the ABCSG-8, -6+8 and −16 cohorts. In all cohorts, no significant predictive value neither for N- nor for anatomic stage was found on the relative scale. The greatest risk reduction on the absolute scale was detected in the ABCSG-16 cohort: decreased LDR rates were observed in patients with nodal-positive BC (−6.7 %; 95%CI, −13.1 to −0.3) or with IIB/IIIA-stage BC (−11.5 %; 95%CI, −22.8 to −0.2) when five instead of two years of additional ET were administered.
4. Discussion
The CTS5 is a widely used tool estimating the LDR risk in HR + BC patients after completion of five years of ET. It was intended to be a prognostic tool [12], which could be confirmed in several validation studies [[6], [7], [8], [9], [10], [11]]. In a previous analysis, we also verified its prognostic performance, regardless of whether it was used as a continuous or categorical score. It is able to discriminate patients according to their LDR risk accurately [13].
Nevertheless, Noordhoek et al. observed in their validation study - analyzing the IDEAL and TEAM patient cohorts – an overestimation of the recurrence risk in high-risk patients [6]. Shresta et al. confirmed these findings in a recent systematic review reporting a mean 10-year DR risk for high-risk patients of 13.8 %, which significantly differed from the 20.3 % in Dowsett's high-risk group in the original CTS5 cohort [11]. After Noordhoek's validation study, Dowsett replied that including patients with extended ET might be causal for the differences in predicted and observed DR risk rates [18]. In the ABCSG-6 cohort, the calibration accuracy was intentionally tested in 1254 patients without extended ET and we still observed an overestimation of LDR in high-risk patients [13]. A risk difference of the predicted versus the observed risk of −6.0%-points was observed in the highest CTS5-quantile which is comparable to the reported −6.5%-points in the review of Shresta et al. but lower than the −10%-points in Noordhoek's validation. Within the ABCSG-8 and the combined ABCSG-6+8 cohort, we observed that the CTS5 estimated DR risk was practically equivalent to the observed rates, with a modest tendency to overestimate the risk. With −2.4%-points the largest risk difference between predicted versus observed risk was found in the third CTS5-quantil in the ABCSG-6+8 cohort. However, there is evidence that the calibration of the score was more accurate in low-risk than in high-risk cohorts as it was the case in the ABCSG-6 trial including a higher proportion of high-risk patients.
Given that HR + BC is associated with higher rates of LDR than other BC subtypes [1], ET extension seems a feasible strategy to reduce the risk [15,[19], [20], [21]]. Since there is no evidence that all patients with HR + BC will benefit from ET beyond 5 years, the need for a tool predicting benefits of extended ET is still high [22]. However, the optimal duration of extended ET is not clarified yet but there is evidence that a total duration of 7 or 7.5 years might be beneficial for the oncological outcome [23] while the duration of 10 years showed no superiority in the ABCSG-16([17]) or IDEAL trial [24]. When discussing the predictive power of the score, it has to be kept in mind that a predictive tool is able to anticipate the effect of a specific therapy whereas a prognostic tool predicts patient's outcome relating to characteristics of a disease, irrespective of the administered treatment [25,26].
Noordhoek et al. tested the score's predictive power in over 7000 patients who underwent either 2.5 or 5 years of extended ET. Evidence for the predictive value remained missing. In our previously published analysis, we could underline these findings although we observed an apparent benefit in high-risk patients on the absolute scale when ET was extended (−6.1 %, 95%CI, −14.4–2.3) [13]. Thus, here we investigated the CTS5's predictive power in two larger cohorts. Even in the largest cohort consisting of 4931 patients with 250 events, however, no significant predictive value was observed. Beneficial effects of extending ET were even found in low-risk patients, rendering the score's predictive value doubtful. In the ABCSG-16 cohort, a notable benefit of extending ET was observed in the high-risk group, even though the result did not reach statistical significance. In detail, high-risk patients who underwent ET for 10 years had a 23.4 % lower rate of LDR events than patients of the same risk group with only 7 years of ET. This finding underscores a potential predictive value of the CTS5 in this cohort although, in general, the ABCSG-16 trial couldn't confirm superiority of 10 years of ET over 7 years [17]. Villasco et al. suggested in their trial - including 783 patients of which 180 received extended ET - that the CTS5 was able to predict the benefit of therapy extension while neither N-, anatomic-, disease stage, tumor grade nor Ki67 level did [8]. Considering the small sample size, the results must be interpreted carefully. The score should also be applied with caution in premenopausal women. Lee et al. had shown that its predictive power might be lower in this subgroup [7]. Taken together, these findings may justify the use of the CTS5 for decision-making regarding ET extension in high-risk patients, particularly when multigenomic tests are unavailable or not feasible.
Multigenomic tests such as Oncotype DX([27]), Endopredict [28], Prosigna [29], MammaPrint [30] or Breast Cancer Index (BCI) [31] represent the greatest competitors for clinical assessment tools. Although these tests were intentionally set up as prognostic tools, they are used regularly for prediction. The BCI, for example, uses two genomic assays and estimates not only DR risk but also the benefit of ET extension [32,33]. In the literature, several trials could show superior predictive power of the BCI when compared to the CTS5([34], [35]). Regarding the ability to predict beneficial response of extended endocrine therapy, comparisons of other multigenomic tests with the CTS5 are scarce. Recently, Kitano et al. compared two GEPs with the CTS5, analyzing microarray CEL files of 2454 patients and calculated the CTS5 in 1263 of those cases [36]. By combining two GEPs with the CTS5, they successfully identified intermediate-risk patients with a higher benefit of extended ET. If a precise prediction to whom extended therapy should be applied is requested, a combination of the CTS5 with a GEP might be a valid option.
The strength of this here presented study is the high number of patients from randomized trials. When ABCSG-6+8 patients (non-extended) were combined, data from over 3300 patients was available. Regarding the predictive performance, data of 4931 patients was analyzed.
It has to be emphasized that there were imbalances between patients who were enrolled in ABCSG-16 and those who were not. Younger patients as well as patients with smaller tumors were more likely to be enrolled in ABCSG-16. ABCSG-16 patients were more frequently assigned to the low-risk group. In an exploratory analysis (data not shown) we observed a HR for LDR of 0.65 (95%CI, 0.47–0.90; p = 0.0096) for patients enrolled in ABCSG-16 when compared to those who were not. Additionally, the ABCSG-8 trial didn't include patients with G3 tumors. Since grading is one of the parameters included in the CTS5 formula, patients needed to fulfill additional high-risk criteria to be assigned to the high-risk group. ABCSG-8 patients were in the median older (65 years) than patients of the ATAC (64 years) or BIG 1–98 (61 years) trial, which served as the original CTS5 training and validation cohorts [12]. Additionally, a higher proportion of ABCSG-8 patients had pN0 tumors compared to the ATAC and the BIG 1–98 cohorts. Overall, ABCSG-8 patients were older and had a lower risk profile than those of the original training and validation cohorts. This potential selection bias has to be kept in mind when interpreting the results referring to comparisons of patients with and without extended ET. Nevertheless, the prognostic power of the CTS5 was higher in the ABCSG-8 than in the ABCSG-6 cohort (c-index 0.72 versus 0.68). This might indicate once more a superior applicability of the score in low risk-cohorts.
To conclude, the CTS5 is a reasonably well-calibrated, highly prognostic tool that lacks of predictive power. When addressing the question whether an individual patient will benefit from extended ET, it can guide the decision-making process, nevertheless it has to be kept in mind that confirmatory predictive evidence is lacking.
CRediT authorship contribution statement
Kerstin Wimmer: Writing – original draft, Resources, Project administration, Conceptualization. Dominik Hlauschek: Writing – review & editing, Visualization, Project administration, Methodology, Formal analysis, Data curation, Conceptualization. Marija Balic: Writing – review & editing, Resources. Georg Pfeiler: Writing – review & editing, Resources. Richard Greil: Writing – review & editing, Resources. Christian F. Singer: Writing – review & editing, Resources. Stefan Halper: Resources. Günther Steger: Writing – review & editing, Resources. Christoph Suppan: Writing – review & editing, Resources. Simon Peter Gampenrieder: Writing – review & editing, Resources. Ruth Helfgott: Writing – review & editing, Resources. Daniel Egle: Writing – review & editing, Resources. Martin Filipits: Writing – review & editing, Resources. Raimund Jakesz: Writing – review & editing, Resources. Lidija Sölkner: Writing – review & editing, Methodology, Formal analysis, Data curation, Conceptualization. Christian Fesl: Writing – review & editing, Supervision, Project administration, Methodology, Formal analysis, Data curation, Conceptualization. Michael Gnant: Writing – review & editing, Supervision, Resources, Project administration, Methodology, Conceptualization. Florian Fitzal: Writing – review & editing, Supervision, Resources, Project administration, Methodology, Conceptualization.
Ethics approval and consent to participate
The respective trials ABCSG-06 (NCT00309491), ABCSG-06A (NCT00300508), ABCSG 8 (NCT00291759) and ABCSG 16 (NCT00295620) were conducted by the Austrian Breast Cancer and Colorectal Study Group (ABCSG) and were registered clinical trials. The studies have been approved by the responsible ethics committees for each participating site (e.g. EC Med. Univ. Vienna from 10Apr1990 (ABCSG-6), from 04Dec1995 (ABCSG 6a/1033AU/0001; ABCSG 8 EC Nr. 240/95) and from 13Jan2004 (ABCSG 16 EC Nr. 504/2003). The trials were conducted according to the guidelines of the Declaration of Helsinki and were carried out in accordance with the guidelines for Good Clinical Practice. All patients provided signed informed consent.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of competing interest
All authors have completed and submitted the Declaration of Interest form.
Marija Balic reported having received research funding from Amgen, Celgene, Eli Lilly, Novartis, Roche, Pfizer, Samsung and has an advisory role at Amgen, AstraZeneca, Celgene, Daiichi Sankyo, Eli Lilly, MSD, Novartis, Pierre Fabre, Pfizer, Roche, Samsung and Gilead. Marija Balic also reports to be part of the speakers bureau at Amgen, AstraZeneca, Celgene, Daiichi Sankyo, Eli Lilly, MSD, Novartis, Pierre Fabre, Pfizer, Roche, Seagen and Gilead.
Daniel Egle reports having received honoraria, travel grants and is a member of Advisory Boards at Amgen, AstraZeneca, Daiichi-Sankyo, Gilead, Lilly, MSD, Novartis, Pfizer, Pierre-Fabre, Roche, Sandoz, Seagen and Sirius.
Martin Filipits has received honoraria from Astra Zeneca, Biomedica, Biorad, Boehringer Ingelheim, Eli Lilly, Merck, Novartis, and Pfizer.
Florian Fitzal reports a role as Editor for Oncoplastic Surgery part I and II: Springer and having received travel and scientific support from Comesa (Mentor), Novartis, Roche, Astra Zeneca, Pfizer, Myriad, Nanostring, Bondimed (Polytech, Integra) and Eli Lilly. Florian Fitzal has also an Advisory Role for Pfizer, Astra Zeneca, Lilly and Roche and is founder of the Breast analyzing tool (BAT): breastanalyzing. com.
Simon Peter Gampenrieder reports having received speaking and lecture fees from Novartis Pharma AG, Roche, Bristol Myers Squibb Co, AstraZeneca Pharmaceuticals LP, Merck Sharp & Dohme UK Ltd, Pfizer Inc, Eli Lilly and Company, Seagen Inc; travel reimbursement from Novartis Pharma AG, Roche, Pfizer Inc, Amgen Inc, Shire, Bayer AG, Celgene GmbH, Daiichi Sankyo Inc and funding grants from Roche. Also, Simon Peter Gampenrieder reports to have a consulting and advisory role at Roche, Bristol Myers Squibb Co, AstraZeneca Pharmaceuticals LP, Merck Sharp & Dohme UK Ltd, Eli Lilly and Company and Seagen Inc.
Michael Gnant reports having received personal fees/travel support from Amgen, DaiichiSankyo, AstraZeneca, Eli Lilly, LifeBrain, Nanostring, Novartis, PierreFabre, MSD, also an immediate family member is employed by Sandoz.
Richard Greil reports having received research funding and honoraria from Celgene, Roche, Merck, Takeda, AstraZeneca, Novartis, Amgen, BMS, MSD, Sandoz, Abbvie, Gilead and Daiichi Sankyo. In addition, honoraria were also received from Sanofi.
Richard Greil has also a consulting or Advisory Role at Celgene, Novartis, Roche, BMS, Takeda, Abbvie, Astra Zeneca, Janssen, MSD, Merck, Gilead, Daiichi Sankyo and Sanofi.
Richard Greil was supported for travel, accommodations, expenses by Roche, Amgen, Janssen, Astra Zeneca, Novartis, MSD, Celgene, Gilead, BMS, Abbvie and Daiichi Sankyo.
Georg Pfeiler reports having received grants and honoraria from Pfizer, Roche, Seagen, Daiichi, Lilly, Novartis, AstraZeneca, MSD and UCB.
Christian F. Singer reports that administrative support, statistical analysis, and writing assistance were provided by Medical University of Vienna. Christian F singer reports a relationship with the Medical University of Vienna that includes board membership, consulting or advisory, funding grants, speaking and lecture fees, and travel reimbursement.
Following authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper: Kerstin Wimmer, Dominik Hlauschek, Günther Steger, Christoph Suppan, Ruth Helfgott, Raimund Jakesz, Lidija Sölkner, Christian Fesl.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.breast.2025.104415.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Pedersen R.N., Esen B.O., Mellemkjaer L., Christiansen P., Ejlertsen B., Lash T.L., et al. The incidence of breast cancer recurrence 10-32 Years after primary diagnosis. J Natl Cancer Inst. 2022;114(3):391–399. doi: 10.1093/jnci/djab202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Negoita S, Ramirez-Pena E. Prevention of Late Recurrence: An Increasingly Important Target for Breast Cancer Research and Control. J Natl Cancer Inst. 2022;114(3):340–341. doi: 10.1093/jnci/djab203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Laenkholm A.V., Jensen M.B., Eriksen J.O., Rasmussen B.B., Knoop A.S., Buckingham W., et al. PAM50 risk of recurrence score predicts 10-year distant recurrence in a comprehensive Danish cohort of postmenopausal women allocated to 5 Years of endocrine therapy for hormone receptor-positive early breast cancer. J Clin Oncol. 2018;36(8):735–740. doi: 10.1200/JCO.2017.74.6586. [DOI] [PubMed] [Google Scholar]
- 4.Burstein H.J., Curigliano G., Thurlimann B., Weber W.P., Poortmans P., Regan M.M., et al. Customizing local and systemic therapies for women with early breast cancer: the St. Gallen International Consensus Guidelines for treatment of early breast cancer 2021. Ann Oncol. 2021;32(10):1216–1235. doi: 10.1016/j.annonc.2021.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thomssen C., Balic M., Harbeck N., Gnant M. St. Gallen/Vienna 2021: a brief summary of the consensus discussion on customizing therapies for women with early breast cancer. Breast Care. 2021;16(2):135–143. doi: 10.1159/000516114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Noordhoek I., Blok E.J., Meershoek-Klein Kranenbarg E., Putter H., Duijm-de Carpentier M., Rutgers E.J.T., et al. Overestimation of late distant recurrences in high-risk patients with ER-positive breast cancer: validity and accuracy of the CTS5 risk score in the TEAM and IDEAL trials. J Clin Oncol. 2020;38(28):3273–3281. doi: 10.1200/JCO.19.02427. [DOI] [PubMed] [Google Scholar]
- 7.Lee J.H., Lee S.K., Chae B.J., Yu J., Lee J.E., Kim S.W., et al. Validation of the clinical treatment score post-five years in breast cancer patients for predicting late distant recurrence: a single-center investigation in Korea. Front Oncol. 2021;11 doi: 10.3389/fonc.2021.691277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Villasco A., Accomasso F., D'Alonzo M., Agnelli F., Sismondi P., Biglia N. Evaluation of the ability of the Clinical Treatment Score at 5 years (CTS5) compared to other risk stratification methods to predict the response to an extended endocrine therapy in breast cancer patients. Breast Cancer. 2021;28(5):1131–1140. doi: 10.1007/s12282-021-01258-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang C., Chen C., Lin Y., Zhou Y., Mao F., Zhu H., et al. Validation of CTS5 model in large-scale breast cancer population and the impact of menopausal and HER2 status on its prognostic value. Sci Rep. 2020;10(1):4660. doi: 10.1038/s41598-020-61648-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dejthevaporn T., Patanayindee P. Clinical treatment score post-5 Years as a tool for risk estimation of late recurrence in Thai patients with estrogen-receptor-positive, early breast cancer: a validation study. Breast Cancer. 2023;17 doi: 10.1177/11782234231186869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shrestha A., Cullinane C., Evoy D., Geraghty J., Rothwell J., Walshe J., et al. Clinical Treatment Score post-5 years as a predictor of late distant recurrence in hormone receptor-positive breast cancer: systematic review and meta-analysis. Br J Surg. 2022;109(5):411–417. doi: 10.1093/bjs/znac008. [DOI] [PubMed] [Google Scholar]
- 12.Dowsett M., Sestak I., Regan M.M., Dodson A., Viale G., Thurlimann B., et al. Integration of clinical variables for the prediction of late distant recurrence in patients with estrogen receptor-positive breast cancer treated with 5 Years of endocrine therapy: CTS5. J Clin Oncol. 2018;36(19):1941–1948. doi: 10.1200/JCO.2017.76.4258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wimmer K., Hlauschek D., Balic M., Pfeiler G., Greil R., Singer C.F., et al. Is the CTS5 a helpful decision-making tool in the extended adjuvant therapy setting? Breast Cancer Res Treat. 2024;205(2):227–239. doi: 10.1007/s10549-023-07186-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schmid M., Jakesz R., Samonigg H., Kubista E., Gnant M., Menzel C., et al. Randomized trial of tamoxifen versus tamoxifen plus aminoglutethimide as adjuvant treatment in postmenopausal breast cancer patients with hormone receptor-positive disease: Austrian breast and colorectal cancer study group trial 6. J Clin Oncol. 2003;21(6):984–990. doi: 10.1200/JCO.2003.01.138. [DOI] [PubMed] [Google Scholar]
- 15.Jakesz R., Greil R., Gnant M., Schmid M., Kwasny W., Kubista E., et al. Extended adjuvant therapy with anastrozole among postmenopausal breast cancer patients: results from the randomized Austrian Breast and Colorectal Cancer Study Group Trial 6a. J Natl Cancer Inst. 2007;99(24):1845–1853. doi: 10.1093/jnci/djm246. [DOI] [PubMed] [Google Scholar]
- 16.Jakesz R., Jonat W., Gnant M., Mittlboeck M., Greil R., Tausch C., et al. Switching of postmenopausal women with endocrine-responsive early breast cancer to anastrozole after 2 years' adjuvant tamoxifen: combined results of ABCSG trial 8 and ARNO 95 trial. Lancet. 2005;366(9484):455–462. doi: 10.1016/S0140-6736(05)67059-6. [DOI] [PubMed] [Google Scholar]
- 17.Gnant M., Fitzal F., Rinnerthaler G., Steger G.G., Greil-Ressler S., Balic M., et al. Duration of adjuvant aromatase-inhibitor therapy in postmenopausal breast cancer. N Engl J Med. 2021;385(5):395–405. doi: 10.1056/NEJMoa2104162. [DOI] [PubMed] [Google Scholar]
- 18.Dowsett M., Sestak I., Cuzick J. Calibration of CTS5 in women with early estrogen receptor-positive breast cancer. J Clin Oncol. 2021;39(4):338–339. doi: 10.1200/JCO.20.02551. [DOI] [PubMed] [Google Scholar]
- 19.Mamounas E.P., Bandos H., Lembersky B.C., Jeong J.H., Geyer C.E., Jr., Rastogi P., et al. Use of letrozole after aromatase inhibitor-based therapy in postmenopausal breast cancer (NRG Oncology/NSABP B-42): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(1):88–99. doi: 10.1016/S1470-2045(18)30621-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Davies C., Pan H., Godwin J., Gray R., Arriagada R., Raina V., et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381(9869):805–816. doi: 10.1016/S0140-6736(12)61963-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Goss P.E., Ingle J.N., Pritchard K.I., Robert N.J., Muss H., Gralow J., et al. Extending aromatase-inhibitor adjuvant therapy to 10 years. N Engl J Med. 2016;375(3):209–219. doi: 10.1056/NEJMoa1604700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Saponaro M., Annunziata L., Turla A., Vigano I., De Laurentiis M., Giuliano M., et al. Extended adjuvant endocrine treatment in luminal breast cancers in the era of genomic tests. Int J Mol Sci. 2022;23(21) doi: 10.3390/ijms232113604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Del Mastro L., Mansutti M., Bisagni G., Ponzone R., Durando A., Amaducci L., et al. Extended therapy with letrozole as adjuvant treatment of postmenopausal patients with early-stage breast cancer: a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22(10):1458–1467. doi: 10.1016/S1470-2045(21)00352-1. [DOI] [PubMed] [Google Scholar]
- 24.Blok E.J., Kroep J.R., Meershoek-Klein Kranenbarg E., Duijm-de Carpentier M., Putter H., van den Bosch J., et al. Optimal duration of extended adjuvant endocrine therapy for early breast cancer; results of the IDEAL trial (BOOG 2006-05) J Natl Cancer Inst. 2018;110(1) doi: 10.1093/jnci/djx134. [DOI] [PubMed] [Google Scholar]
- 25.Oldenhuis C.N., Oosting S.F., Gietema J.A., de Vries E.G. Prognostic versus predictive value of biomarkers in oncology. Eur J Cancer. 2008;44(7):946–953. doi: 10.1016/j.ejca.2008.03.006. [DOI] [PubMed] [Google Scholar]
- 26.Italiano A. Prognostic or predictive? It's time to get back to definitions. J Clin Oncol. 2011;29(35):4718. doi: 10.1200/JCO.2011.38.3729. author reply -9. [DOI] [PubMed] [Google Scholar]
- 27.Paik S., Shak S., Tang G., Kim C., Baker J., Cronin M., et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351(27):2817–2826. doi: 10.1056/NEJMoa041588. [DOI] [PubMed] [Google Scholar]
- 28.Filipits M., Rudas M., Jakesz R., Dubsky P., Fitzal F., Singer C.F., et al. A new molecular predictor of distant recurrence in ER-positive, HER2-negative breast cancer adds independent information to conventional clinical risk factors. Clin Cancer Res. 2011;17(18):6012–6020. doi: 10.1158/1078-0432.CCR-11-0926. [DOI] [PubMed] [Google Scholar]
- 29.Wallden B., Storhoff J., Nielsen T., Dowidar N., Schaper C., Ferree S., et al. Development and verification of the PAM50-based Prosigna breast cancer gene signature assay. BMC Med Genom. 2015;8:54. doi: 10.1186/s12920-015-0129-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cardoso F., van't Veer L.J., Bogaerts J., Slaets L., Viale G., Delaloge S., et al. 70-Gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med. 2016;375(8):717–729. doi: 10.1056/NEJMoa1602253. [DOI] [PubMed] [Google Scholar]
- 31.Jerevall P.L., Ma X.J., Li H., Salunga R., Kesty N.C., Erlander M.G., et al. Prognostic utility of HOXB13:IL17BR and molecular grade index in early-stage breast cancer patients from the Stockholm trial. Br J Cancer. 2011;104(11):1762–1769. doi: 10.1038/bjc.2011.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bartlett J.M.S., Sgroi D.C., Treuner K., Zhang Y., Ahmed I., Piper T., et al. Breast Cancer Index and prediction of benefit from extended endocrine therapy in breast cancer patients treated in the Adjuvant Tamoxifen-To Offer More? (aTTom) trial. Ann Oncol. 2019;30(11):1776–1783. doi: 10.1093/annonc/mdz289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Noordhoek I., Treuner K., Putter H., Zhang Y., Wong J., Meershoek-Klein Kranenbarg E., et al. Breast cancer index predicts extended endocrine benefit to individualize selection of patients with HR(+) early-stage breast cancer for 10 Years of endocrine therapy. Clin Cancer Res. 2021;27(1):311–319. doi: 10.1158/1078-0432.CCR-20-2737. [DOI] [PubMed] [Google Scholar]
- 34.Liefers GJ, Noordhoek I, Putter H, Wong J, Siuliukina N, Zhang Y, et al. Predictive performance of breast cancer index (BCI) and clinical treatment score post-5 years (CTS5) in the IDEAL study. J. Clin. Oncol. 2022;40(16_suppl) 545-545. [Google Scholar]
- 35.Shah S., Shaing C., Khatib J., Lodrigues W., Dreadin-Pulliam J., Anderson B.B., et al. The utility of breast cancer index (BCI) over clinical prognostic tools for predicting the need for extended endocrine therapy: a safety net hospital experience. Clin Breast Cancer. 2022;22(8):823–827. doi: 10.1016/j.clbc.2022.08.003. [DOI] [PubMed] [Google Scholar]
- 36.Kitano S., Tsunashima R., Kato C., Watanabe A., Sota Y., Matsumoto S., et al. Validation of late recurrence prediction by gene expression profiles and clinicopathological factors in estrogen receptor-positive breast cancer. Breast Cancer. 2024;31(5):898–908. doi: 10.1007/s12282-024-01602-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.