Abstract
Aims
There were no previous studies comparing aspirin vs. P2Y12 inhibitor monotherapy following short dual antiplatelet therapy (DAPT) after complex percutaneous coronary intervention (PCI).
Methods and results
We conducted a pre-specified subgroup analysis based on complex PCI in the 1-year results of the STOPDAPT-3 (ShorT and OPtimal Duration of Dual AntiPlatelet Therapy-3) trial, which randomly compared 1-month DAPT followed by aspirin monotherapy (aspirin group) with 1-month prasugrel monotherapy followed by clopidogrel monotherapy (clopidogrel group). The main analysis in the present study was the 30-day landmark analysis. The co-primary endpoints were cardiovascular events (a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, or stroke) and major bleeding (Bleeding Academic Research Consortium 3 or 5). In the 30-day landmark analysis (N = 5833), there were 1415 patients (24.3%) who underwent complex PCI. There was a significant interaction between complex PCI and the effect of the aspirin group relative to the clopidogrel group for cardiovascular events (complex PCI: 3.3% vs. 5.2%, non-complex PCI: 4.3% vs. 3.6%, interaction P = 0.04) and net adverse clinical events (complex PCI: 4.8% vs. 7.2%, non-complex PCI: 5.3% vs. 4.4%, interaction P = 0.02), but not for bleeding events (complex PCI: 2.1% vs. 2.7%, non-complex PCI: 1.7% vs. 1.4%, interaction P = 0.35).
Conclusions
There was a significant interaction between complex PCI and the effect of aspirin monotherapy relative to clopidogrel monotherapy beyond 1 month and up to 1 year for cardiovascular events due to numerically lower risk of aspirin monotherapy in patients with complex PCI, while the effect of aspirin monotherapy relative to clopidogrel monotherapy was not different for bleeding regardless of complex PCI.
Clinical trial registration
ShorT and OPtimal duration of Dual AntiPlatelet Therapy after everolimus-eluting cobalt-chromium stent-3 [STOPDAPT-3]; NCT04609111.
Keywords: Antiplatelet therapy, Coronary stent, Percutaneous coronary intervention, Procedural complexity
Abbreviation list
- ACS
acute coronary syndrome
- CI
confidence interval
- DAPT
dual antiplatelet therapy
- HR
hazard ratio
- PCI
percutaneous coronary intervention
Introduction
Several randomized clinical trials have suggested that the strategy of short duration of dual antiplatelet therapy (DAPT) up to 1–3 months followed by P2Y12 inhibitor monotherapy reduced major bleeding events without increasing cardiovascular events after percutaneous coronary intervention (PCI).1–5 However, the procedural complexity of PCI has been acknowledged as a determinant for cardiovascular events, and short DAPT compared with prolonged DAPT has been reported to increase the risk of cardiovascular events after complex PCI.6 On the other hand, another study showed that P2Y12 inhibitor monotherapy after very short DAPT compared with standard DAPT did not increase cardiovascular events regardless of complex PCI.7,8 As of now, there are no data regarding the optimal antiplatelet monotherapy after very short DAPT in patients who underwent complex PCI. Recently, 1-year results of the STOPDAPT-3 (ShorT and OPtimal Duration of Dual AntiPlatelet Therapy-3) trial reported that aspirin monotherapy compared with clopidogrel monotherapy was associated with comparable cardiovascular and bleeding outcomes beyond 1 month and up to 1 year after PCI.9,10 In the present study, we conducted a pre-specified subgroup analysis stratified by complex and non-complex PCI using the 1-year follow-up data from the STOPDAPT-3 trial.
Methods
Study design and population
The STOPDAPT-3 (NCT04609111) was a physician-initiated, prospective, multicentre, open-label, adjudicator blinded randomized clinical trial, where we compared the group of 1-month prasugrel monotherapy followed by clopidogrel monotherapy with the group of 1-month DAPT followed by aspirin monotherapy in patients planned for PCI in terms of cardiovascular and bleeding endpoints. The details for the study design and the overall results at 30 days and at 1 year were previously reported (Supplemental Appendix A–B).9,10 Briefly, the trial enrolled patients with acute coronary syndrome or those with high bleeding risk by the criteria of the Academic Research Consortium irrespective of acute coronary syndrome who were planned for PCI with cobalt–chromium everolimus-eluting stents (Xience series, ABBOTT vascular). Patients were randomly assigned in a one-to-one fashion to the 1-month aspirin-free prasugrel monotherapy or the 1-month DAPT with aspirin and prasugrel. At 1 month (between 30 and 59 days after the index PCI), patients who had received DAPT were switched to aspirin (81–100 mg/day) monotherapy (aspirin group), and patients who had received prasugrel monotherapy were switched to clopidogrel (75 mg/day) monotherapy (clopidogrel group). Each group of patients were to continue the assigned antiplatelet monotherapy up to 1 year. The ethics committees in all the participating centres approved the study protocol, and informed consent was obtained from all patients.
Application of complex percutaneous coronary intervention criteria
The present study was the pre-specified subgroup analysis stratified by the complex and non-complex PCI. In the present study, patients were divided into two subgroups based on the complex PCI criteria. Giustino et al.6 proposed procedural complexity criteria called ‘complex PCI’, and the criteria were endorsed by the clinical guidelines of the European Society of Cardiology and the Japanese Circulation Society.11,12 Complex PCI was defined as a procedure at the index PCI and the staged PCI with at least one of the following procedural criteria: 3 vessels treated, ≥3 stents implanted, ≥3 lesions treated, bifurcation with 2 stents implanted, total stent length >60 mm, or chronic total occlusion as the target lesion.6 The definition of complex PCI was consistent with that in our previous publications on this issue.8,13
Endpoints
The co-primary cardiovascular endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, or ischaemic stroke, and the co-primary bleeding endpoint was major bleeding defined as the Bleeding Academic Research Consortium (BARC) type 3 or 5.14 The major secondary endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, ischaemic stroke, or major bleeding (BARC 3 or 5), which represented net adverse clinical events for cardiovascular and bleeding events. Myocardial infarction and stent thrombosis were defined by the Academic Research Consortium criteria.15 The definitions of other secondary endpoints are described in the supplemental materials (Supplemental Appendix C–D). The independent clinical event committee adjudicated all the clinical events in a blinded fashion to the assigned group.
Statistical analysis
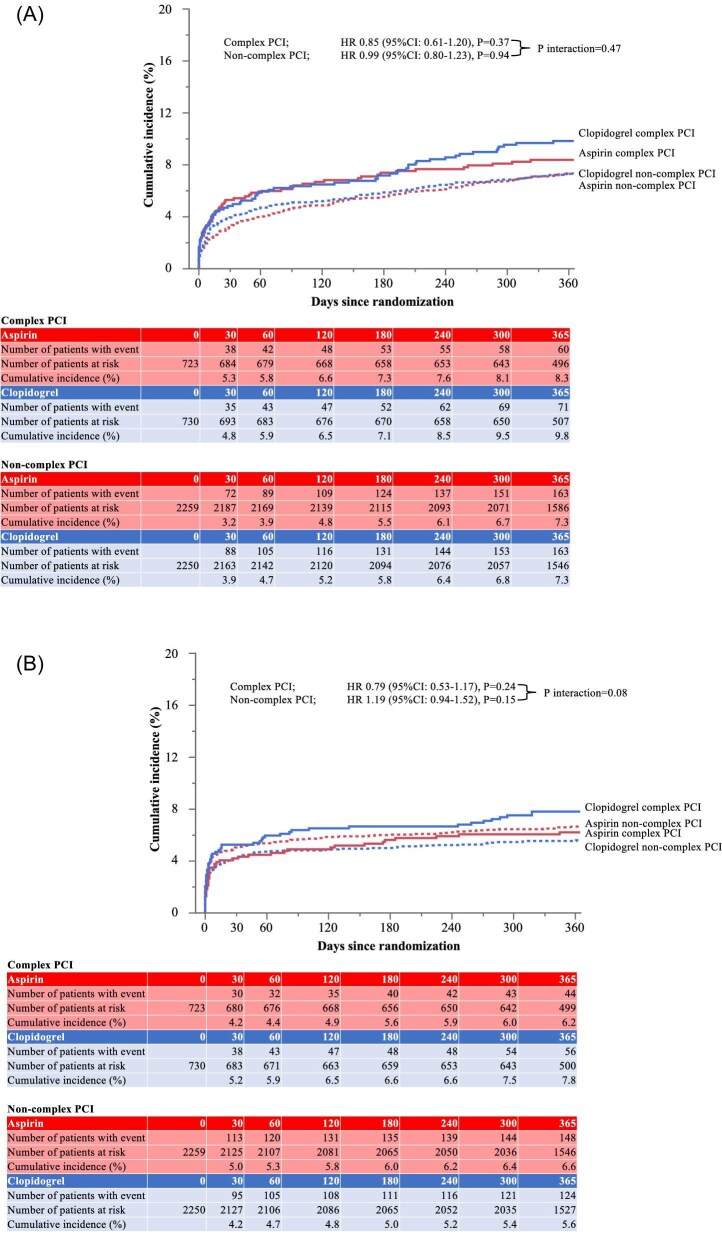
The main analysis in the present study was the 30-day landmark analysis for the comparison between aspirin and clopidogrel monotherapy beyond 30 days and up to 1 year. In the 30-day landmark analysis, we excluded those patients who had the endpoint event of interest before 30 days, and thus the number of patients included in the 30-day landmark analysis was different according to the endpoint event of interest. We also conducted the overall 1-year analysis evaluating throughout 1 year. The overall 1-year analysis set consisted of 5962 patients after excluding 3 patients who did not receive PCI for the absence of suitable coronary lesions, and 37 patients who withdrew consent (Figure 1). The 30-day landmark analysis set consisted of 5833 patients after excluding 129 patients who died by 30 days or lost to follow-up at 30 days (Figure 1).
Figure 1.
Study flow. PCI, percutaneous coronary intervention.
Categorical variables were presented as number and percentage and were compared using the χ2 test. Continuous variables were expressed as mean ± standard deviation or median with interquartile range and were compared using Student’s t-test or the Wilcoxon rank-sum test depending on their distributions. The cumulative incidences of the endpoints were estimated by the Kaplan–Meier method. The effects of patients with complex PCI relative to those with non-complex PCI, and the effects of the aspirin group relative to the clopidogrel group for the endpoints were expressed as hazard ratios (HRs) with 95% confidence intervals (CIs) by the Cox proportional hazard model. In the present study, we analysed the treatment-by-subgroup interactions in the effects of the aspirin group compared with the clopidogrel group in complex PCI and non-complex PCI subgroups. All reported P values were two-sided. P values <0.05 were considered statistically significant. All analysis was performed with R version 4.2.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Study population
Among 5833 patients in the 30-day landmark analysis, there were 1415 patients (24.3%) who underwent complex PCI (aspirin group: N = 705, and clopidogrel group: N = 710), and 4418 patients (75.7%) who underwent non-complex PCI (aspirin group: N = 2215, and clopidogrel group: N = 2203) (Figure 1). Regarding the criteria for complex PCI, >60 mm total stent length (19.4%) and ≥3 stents implanted (12.1%) were more prevalent than other criteria, whereas bifurcation with 2 stents (1.2%) was less prevalent than other criteria (Table 1).
Table 1.
Prevalence of complex percutaneous coronary intervention
| (A) 30-day landmark analysis set | (B) Overall 1-year analysis set | |||||
|---|---|---|---|---|---|---|
| Overall | Aspirin group |
Clopidogrel group |
Overall | Aspirin group |
Clopidogrel group |
|
| N = 5833 | N = 2920 | N = 2913 | N = 5962 | N = 2982 | N = 2980 | |
| Target of 3 vessels | 263 (4.5) | 135 (4.6) | 128 (4.4) | 264 (4.4) | 135 (4.5) | 129 (4.3) |
| ≥3 stents implanted | 707 (12.1) | 354 (12.1) | 353 (12.1) | 720 (12.1) | 359 (12.0) | 361 (12.1) |
| ≥3 lesions treated | 481 (8.2) | 241 (8.3) | 240 (8.2) | 485 (8.1) | 242 (8.1) | 243 (8.2) |
| Bifurcation with two stents | 72 (1.2) | 32 (1.1) | 40 (1.4) | 78 (1.3) | 34 (1.1) | 44 (1.5) |
| Total stent length >60 mm | 1109 (19.4) | 556 (19.3) | 553 (19.4) | 1141 (19.5) | 471 (19.5) | 570 (19.6) |
| Target of chronic total occlusion | 283 (4.9) | 148 (5.1) | 135 (4.6) | 285 (4.8) | 149 (5.0) | 136 (4.6) |
| Complex PCI | 1415 (24.3) | 705 (24.1) | 710 (24.4) | 1453 (24.3) | 723 (24.2) | 730 (24.5) |
PCI, percutaneous coronary intervention.
Baseline characteristics
Among 5833 patients in the 30-day landmark analysis, patients with complex PCI were older and more often men compared with those without (Table 2). Patients with complex PCI less often presented as acute coronary syndrome, but more often had acute heart failure compared with those without. Patients with complex PCI more often had comorbidities such as history of heart failure, hypertension, hyperlipidaemia, diabetes, left ventricular systolic dysfunction, anaemia, and chronic kidney disease than those without. The prevalence of the high bleeding risk by the criteria of the Academic Research Consortium was higher in patients with complex PCI than in those without. Procedural characteristics were largely different between patients with and without complex PCI. The rate of intracoronary imaging use was >90% regardless of complex PCI. In terms of medications, total heparin dose during the index PCI was slightly higher in patients with complex PCI than in those without. The prescription rates of mineralocorticoid receptor antagonist and β-blockers were higher in patients with complex PCI than in those without. Baseline characteristics in the overall 1-year analysis are shown in Supplementary material online, Table S1.
Table 2.
Baseline characteristics in the 30-day landmark analysis: complex percutaneous coronary intervention vs. non-complex percutaneous coronary intervention
| Complex PCI | Non-complex PCI | ||
|---|---|---|---|
| N = 1415 | N = 4418 | P value | |
| Patient demographics | |||
| Age (year) | 72.2 ± 11.2 | 71.3 ± 11.8 | 0.01 |
| ≥75 | 669 (47.3) | 1981 (44.8) | 0.11 |
| Men | 1126 (79.6) | 3344 (75.7) | 0.003 |
| Body mass index (kg/m2) | 24.0 ± 3.7 | 23.9 ± 3.8 | 0.39 |
| <25 | 926 (65.4) | 2891 (65.4) | 1.00 |
| Clinical presentation | |||
| Acute coronary syndrome | 995 (70.3) | 3358 (76.0) | <0.001 |
| ST-segment elevation myocardial infarction | 570 (40.3) | 1887 (42.7) | |
| Non-ST-segment elevation myocardial infarction | 253 (17.9) | 821 (18.6) | |
| Unstable angina | 172 (12.2) | 650 (14.7) | |
| Non-acute coronary syndrome | 420 (29.7) | 1060 (24.0) | <0.001 |
| Cardiogenic shock | 63 (4.5) | 172 (3.9) | 0.35 |
| Current heart failure | 287 (20.3) | 751 (17.0) | 0.01 |
| History and comorbidities | |||
| Prior PCI | 201 (14.2) | 711 (16.1) | 0.09 |
| Prior coronary artery bypass grafting | 33 (2.3) | 89 (2.0) | 0.47 |
| Prior myocardial infarction | 105 (7.4) | 346 (7.8) | 0.61 |
| Prior stroke | 133 (9.4) | 405 (9.2) | 0.79 |
| Prior heart failure | 359 (25.4) | 942 (21.3) | 0.001 |
| Atrial fibrillation | 123 (8.7) | 419 (9.5) | 0.37 |
| Peripheral artery disease | 90 (6.4) | 240 (5.4) | 0.19 |
| Hypertension | 1157 (81.8) | 3329 (75.4) | <0.001 |
| Hyperlipidaemia | 986 (69.7) | 2951 (66.8) | 0.04 |
| Diabetes | 719 (50.8) | 1906 (43.1) | <0.001 |
| With insulin therapy | 100 (7.1) | 213 (4.8) | 0.001 |
| Current smoker | 335 (23.7) | 1047 (23.7) | 0.99 |
| Left ventricular ejection fraction (%) | 54.1 ± 12.2 | 54.9 ± 11.7 | 0.03 |
| <40 | 153 (11.5) | 371 (9.0) | 0.01 |
| Moderate or severe mitral regurgitation | 36 (2.5) | 107 (2.4) | 0.80 |
| Anaemia | 186 (13.1) | 476 (10.8) | 0.01 |
| Thrombocytopenia | 18 (1.3) | 51 (1.2) | 0.72 |
| Moderate chronic kidney disease | 626 (44.2) | 1748 (39.6) | 0.002 |
| Severe chronic kidney disease | 182 (12.9) | 462 (10.5) | 0.01 |
| Estimated glomerular filtration rate <30 mL/min/1.73 m2 not on dialysis | 88 (6.2) | 210 (4.8) | 0.03 |
| Haemodialysis | 94 (6.6) | 252 (5.7) | 0.19 |
| Cancer history | 152 (10.7) | 439 (9.9) | 0.38 |
| ARC-HBR | 815 (57.6) | 2341 (53.0) | 0.002 |
| Procedural characteristics at the index PCI | |||
| Staged PCI | 861 (60.8) | 347 (7.9) | <0.001 |
| Number of procedures | 2.0 (1.0–2.0) | 1.0 (1.0–1.0) | <0.001 |
| Number of target lesions | 2.0 (2.0–3.0) | 1.0 (1.0–1.0) | <0.001 |
| ≥3 | 481 (34.0) | 0 (0.0) | <0.001 |
| Target of two vessels or more | 975 (68.9) | 541 (12.2) | <0.001 |
| Target of three vessels | 258 (18.2) | 0 (0.0) | <0.001 |
| Target of chronic total occlusion | 283 (20.0) | 0 (0.0) | <0.001 |
| Target of bifurcation lesion | 633 (44.7) | 1025 (23.2) | <0.001 |
| Bifurcation with two stents | 72 (5.1) | 0 (0.0) | <0.001 |
| Target lesion location | |||
| Left main coronary artery | 170 (12.0) | 167 (3.8) | <0.001 |
| Left anterior descending coronary artery | 999 (70.6) | 2499 (56.6) | <0.001 |
| Left circumflex coronary artery | 572 (40.4) | 770 (17.4) | <0.001 |
| Right coronary artery | 866 (61.2) | 1408 (31.9) | <0.001 |
| Bypass graft | 5 (0.4) | 12 (0.3) | 0.62 |
| Number of implanted stents | 2.0 (2.0–3.0) | 1.0 (1.0–1.0) | <0.001 |
| ≥3 | 707 (50.0) | 0 (0.0) | <0.001 |
| Minimal stent diameter (mm) | 2.5 (2.25–3.0) | 3.0 (2.5–3.5) | <0.001 |
| Total stent length (mm) | 71.0 (61.0–94.0) | 28.0 (18.0–38.0) | <0.001 |
| >60 | 1109 (79.0) | 0 (0.0) | <0.001 |
| Radial approach only | 958 (67.7) | 3547 (80.3) | <0.001 |
| Use of intracoronary imaging | 1349 (95.3) | 3987 (90.2) | <0.001 |
| Medication at the index PCI | |||
| Loading of prasugrel | 1412 (99.8) | 4394 (99.5) | 0.11 |
| Loading of aspirin | 581 (82.6) | 1800 (81.3) | 0.44 |
| Total heparin dose (IU/kg) during PCI | 129.0 (101.9–156.2) | 118.5 (96.2–147.5) | <0.001 |
| Post-procedural heparin use | 614 (43.4) | 1977 (44.8) | 0.36 |
| Medication at discharge | |||
| Antiplatelet agents | 1405 (99.5) | 4376 (99.5) | 0.93 |
| Aspirin only | 30 (2.1) | 64 (1.5) | |
| Prasugrel only | 674 (47.7) | 2102 (47.8) | |
| DAPT | 685 (48.5) | 2158 (49.1) | |
| Aspirin/prasugrel | 679 (48.1) | 2146 (48.8) | |
| Anticoagulants | 193 (13.7) | 595 (13.5) | 0.90 |
| Warfarin | 30 (2.1) | 123 (2.8) | |
| Direct oral anticoagulants | 163 (11.5) | 472 (10.7) | |
| Renin–angiotensin system inhibitor | 1054 (74.6) | 3281 (74.6) | 0.98 |
| Angiotensin-converting enzyme inhibitor | 452 (32.0) | 1478 (33.6) | |
| Angiotensin-2 receptor blocker | 494 (35.0) | 1540 (35.0) | |
| Angiotensin receptor–neprilysin inhibitor | 114 (8.1) | 277 (6.3) | |
| Mineralocorticoid receptor antagonist | 281 (19.9) | 681 (15.5) | <0.001 |
| β-Blockers | 952 (67.4) | 2792 (63.5) | 0.01 |
| Statins | 1323 (93.7) | 4111 (93.5) | 0.79 |
| High-intensity statin therapy | 726 (51.4) | 2097 (47.7) | 0.02 |
| Proton pump inhibitors | 1239 (87.7) | 3862 (87.8) | 0.93 |
Categorical variables are presented as number and percentage. Continuous variables are presented as mean ± standard deviation or median with interquartile range.
ARC-HBR, Academic Research Consortium for high bleeding risk; DAPT, dual antiplatelet therapy; PCI, percutaneous coronary intervention.
Baseline characteristics were well balanced between the aspirin and clopidogrel groups regardless of complex PCI, except for the higher prescription rate of proton pump inhibitors in the aspirin group than in the clopidogrel group in the 30-day landmark and the overall 1-year analyses (see Supplementary material online, Tables S2 and S3).
As we previously reported, the vast majority of the study patients received the assigned antiplatelet therapy according to the study protocol. Nevertheless, the prescription rate of the assigned antiplatelet therapy was numerically lower and the prescription rate of DAPT beyond 30 days was numerically higher in patients with complex PCI than in those without (see Supplementary material online, Figure S1).
Clinical outcomes: complex percutaneous coronary intervention vs. non-complex percutaneous coronary intervention
In the overall 1-year analysis, the cumulative incidence of the co-primary cardiovascular endpoint at 1 year was higher in patients with complex PCI than in those without (9.1% vs. 7.3%, HR, 1.27; 95% CI, 1.03–1.55; P = 0.02), whereas it was not different between patients with and without complex PCI in the 30-day landmark analysis (4.3% vs. 3.9%, HR, 1.09; 95% CI, 0.81–1.47; P = 0.58) (see Supplementary material online, Tables S4 and S5). The cumulative incidences of the myocardial infarction and coronary revascularization were higher in patients with complex PCI than in those without in the overall 1-year and 30-day landmark analyses (see Supplementary material online, Tables S4 and S5).
In the overall 1-year analysis, the cumulative incidence of the co-primary bleeding endpoint at 1 year was not different between patients with and without complex PCI (7.0% vs. 6.1%, HR, 1.14; 95% CI, 0.91–1.44; P = 0.25), whereas it was higher in patients with complex PCI than in those without in the 30-day landmark analysis (2.4% vs. 1.6%, HR, 1.54; 95% CI 1.01–2.36; P = 0.04) (see Supplementary material online, Tables S4 and S5).
Clinical outcomes in the 30-day landmark analysis: aspirin group vs. clopidogrel group in patients with and without complex percutaneous coronary intervention
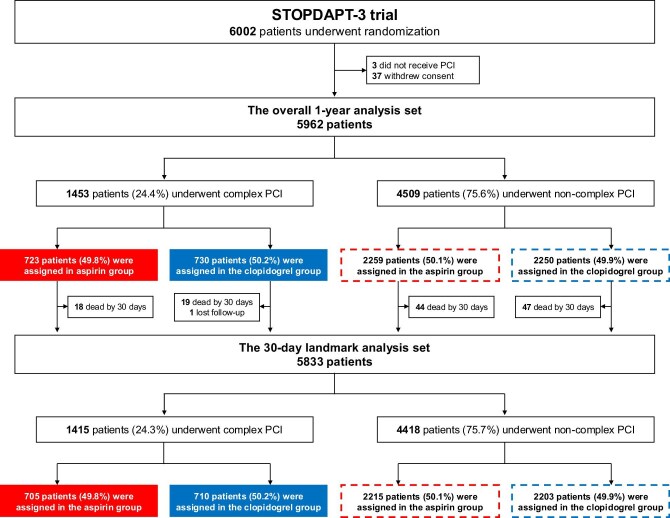
In the 30-day landmark analysis, the cumulative incidence of the co-primary cardiovascular endpoint at 1 year was 3.3% in the aspirin group and 5.2% in the clopidogrel group in patients with complex PCI (HR, 0.52; 95% CI, 0.36–1.05; P = 0.08), while it was 4.3% in the aspirin group and 3.6% in the clopidogrel group in patients without complex PCI (HR, 1.18; 95% CI 0.87–1.60; P = 0.27) (Figure 2A). There was a significant treatment-by subgroup interaction in patients with and without complex PCI for the co-primary cardiovascular endpoint (P for interaction = 0.04). The cumulative incidence of definite stent thrombosis at 1 year was 0.3% in the aspirin group and 0.1% in the clopidogrel group in patients with complex PCI, while it was 0.1% in the aspirin group and 0.1% in the clopidogrel group in patients without complex PCI (Table 3).
Figure 2.
Kaplan–Meier curves in the 30-day landmark analysis. (A) co-primary cardiovascular endpoint: a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, or ischaemic stroke. (B) co-primary bleeding endpoint: BARC 3 or 5 bleeding. BARC, Bleeding Academic Research Consortium; CI, confidence interval; DAPT, dual antiplatelet therapy; HR, hazard ratio; PCI, percutaneous coronary intervention.
Table 3.
Clinical outcomes in the 30-day landmark analysis: aspirin vs. clopidogrel stratified by complex percutaneous coronary intervention /non-complex percutaneous coronary intervention
| Complex PCI | Non-complex PCI | ||||||
|---|---|---|---|---|---|---|---|
| Aspirin group | Clopidogrel group | Aspirin group | Clopidogrel group | ||||
| N = 705 | N = 710 | N = 2215 | N = 2203 | ||||
| Number of patients with event/number of patients (cumulative incidence at 1 year) | Hazard ratio (95% CI) | Number of patients with event/number of patients (cumulative incidence at 1 year) | Hazard ratio (95% CI) | Interaction P | |||
| Co-primary cardiovascular endpoint | |||||||
| A composite of cardiovascular death, myocardial infarction, definite stent thrombosis, or ischaemic stroke | 22/684 (3.3%) | 36/693 (5.2%) | 0.62 (0.36–1.05) | 92/2187 (4.3%) | 77/2163 (3.6%) | 1.18 (0.87–1.60) | 0.04 |
| Co-primary bleeding endpoint | |||||||
| BARC 3 or 5 bleeding | 14/680 (2.1%) | 18/683 (2.7%) | 0.78 (0.39–1.56) | 35/2125 (1.7%) | 30/2127 (1.4%) | 1.17 (0.72–1.90) | 0.35 |
| Major secondary endpoint | |||||||
| A composite of cardiovascular death, myocardial infarction, definite stent thrombosis, ischaemic stroke, or BARC 3 or 5 bleeding | 31/661 (4.8%) | 48/670 (7.2%) | 0.65 (0.42–1.03) | 111/2100 (5.3%) | 91/2098 (4.4%) | 1.22 (0.93–1.61) | 0.02 |
| Secondary endpoints | |||||||
| Death | 24/705 (3.4%) | 27/710 (3.8%) | 0.89 (0.52–1.55) | 91/2215 (4.2%) | 79/2203 (3.6%) | 1.14 (0.85–1.55) | 0.44 |
| Death from cardiovascular causes | 8/705 (1.1%) | 20/710 (2.8%) | 0.40 (0.18–0.91) | 50/2215 (2.3%) | 46/2203 (2.1%) | 1.08 (0.72–1.61) | 0.03 |
| Sudden cardiac death | 2/705 (0.3%) | 7/710 (1.0%) | 0.29 (0.06–1.39) | 21/2215 (1.0%) | 9/2203 (0.4%) | 2.32 (1.06–5.06) | 0.02 |
| Death from non-cardiovascular causes | 16/705 (2.3%) | 7/710 (1.0%) | 2.30 (0.95–5.59) | 41/2215 (1.9%) | 33/2203 (1.5%) | 1.23 (0.78–1.95) | 0.22 |
| Myocardial infarction | 14/693 (2.1%) | 14/700 (2.1%) | 1.01 (0.48–2.13) | 27/2201 (1.3%) | 22/2178 (1.0%) | 1.22 (0.69–2.13) | 0.70 |
| Spontaneous myocardial infarction | 13/693 (1.9%) | 14/700 (2.1%) | 0.94 (0.44–2.00) | 24/2201 (1.1%) | 19/2178 (0.9%) | 1.25 (0.69–2.28) | 0.56 |
| Procedural myocardial infarction | 1/693 (0.1%) | 0/700 (0.0%) | NA | 3/2201 (0.1%) | 3/2178 (0.1%) | 0.99 (0.20–4.90) | NA |
| Myocardial infarction related to the target lesion | 9/693 (1.3%) | 10/700 (1.5%) | 0.91 (0.37–2.24) | 10/2201 (0.5%) | 8/2178 (0.4%) | 1.24 (0.49–3.14) | 0.64 |
| Definite or probable stent thrombosis | 3/701 (0.4%) | 1/706 (0.1%) | 3.01 (0.31–28.95) | 3/2208 (0.1%) | 4/2193 (0.2%) | 0.75 (0.17–3.33) | 0.31 |
| Definite | 2/701 (0.3%) | 1/706 (0.1%) | 2.01 (0.18–22.14) | 2/2208 (0.1%) | 3/2193 (0.1%) | 0.66 (0.11–3.97) | 0.47 |
| Probable | 1/705 (0.1%) | 0/710 (0.0%) | NA | 1/2215 (0.0%) | 1/2203 (0.0%) | 0.99 (0.06–15.90) | NA |
| Stroke | 3/696 (0.4%) | 7/704 (1.0%) | 0.43 (0.11–1.67) | 22/2200 (1.0%) | 22/2186 (1.0%) | 0.99 (0.55–1.79) | 0.27 |
| Ischaemic stroke | 3/696 (0.4%) | 5/704 (0.7%) | 0.61 (0.14–2.54) | 20/2202 (0.9%) | 19/2187 (0.9%) | 1.05 (0.56–1.96) | 0.49 |
| Haemorrhagic stroke | 0/705 (0.0%) | 2/710 (0.3%) | NA | 2/2213 (0.1%) | 3/2202 (0.1%) | 0.66 (0.11–3.96) | NA |
| Any coronary revascularization | 39/696 (5.8%) | 40/705 (5.9%) | 0.99 (0.64–1.55) | 87/2207 (4.1%) | 85/2180 (4.0%) | 1.01 (0.75–1.36) | 0.96 |
| Target lesion revascularization | 19/698 (2.8%) | 21/707 (3.1%) | 0.92 (0.50–1.71) | 26/2208 (1.2%) | 27/2189 (1.3%) | 0.96 (0.56–1.64) | 0.93 |
| Clinically driven | 16/698 (2.4%) | 18/708 (2.6%) | 0.90 (0.46–1.77) | 22/2209 (1.0%) | 20/2190 (1.0%) | 1.09 (0.60–2.00) | 0.68 |
| Non-target lesion revascularization | 22/702 (3.3%) | 27/708 (4.0%) | 0.82 (0.47–1.44) | 68/2213 (3.2%) | 63/2193 (3.0%) | 1.07 (0.76–1.50) | 0.44 |
| Coronary artery bypass grafting | 2/703 (0.3%) | 7/708 (1.0%) | 0.29 (0.06–1.39) | 8/2212 (0.4%) | 9/2200 (0.4%) | 0.88 (0.34–2.29) | 0.23 |
| Bleeding | |||||||
| BARC 2, 3, or 5 bleeding | 20/640 (3.2%) | 25/654 (3.9%) | 0.81 (0.45–1.46) | 66/2039 (3.3%) | 51/2048 (2.5%) | 1.31 (0.91–1.88) | 0.18 |
| BARC 5 | 1/705 (0.1%) | 1/710 (0.1%) | 1.00 (0.06–16.00) | 5/2215 (0.2%) | 5/2203 (0.2%) | 0.99 (0.29–3.43) | 0.99 |
| BARC 3 | 13/680 (2.0%) | 17/683 (2.5%) | 0.77 (0.37–1.58) | 30/2125 (1.4%) | 26/2127 (1.3%) | 1.15 (0.68–1.95) | 0.37 |
| BARC 2 | 13/665 (2.0%) | 12/681 (1.8%) | 1.11 (0.50–2.42) | 42/2125 (2.0%) | 28/2123 (1.3%) | 1.51 (0.93–2.43) | 0.51 |
| TIMI major or minor | 11/679 (1.7%) | 18/684 (2.7%) | 0.61 (0.29–1.30) | 30/2127 (1.4%) | 28/2132 (1.3%) | 1.07 (0.64–1.80) | 0.23 |
| TIMI major | 8/692 (1.2%) | 12/696 (1.8%) | 0.67 (0.27–1.64) | 26/2176 (1.2%) | 25/2163 (1.2%) | 1.03 (0.60–1.79) | 0.42 |
| TIMI minor | 3/692 (0.4%) | 7/698 (1.0%) | 0.43 (0.11–1.66) | 9/2166 (0.4%) | 6/2171 (0.3%) | 1.50 (0.54–4.23) | 0.15 |
| GUSTO moderate or severe | 10/690 (1.5%) | 17/692 (2.5%) | 0.59 (0.27–1.28) | 33/2163 (1.6%) | 25/2156 (1.2%) | 1.32 (0.78–2.21) | 0.09 |
| GUSTO severe | 7/695 (1.0%) | 12/702 (1.7%) | 0.59 (0.23–1.49) | 17/2191 (0.8%) | 19/2178 (0.9%) | 0.89 (0.46–1.71) | 0.47 |
| GUSTO moderate | 3/700 (0.4%) | 5/700 (0.7%) | 0.60 (0.14–2.51) | 17/2187 (0.8%) | 10/2179 (0.5%) | 1.69 (0.77–3.70) | 0.21 |
| Intracranial bleeding (BARC 3 or 5) | 3/703 (0.4%) | 5/708 (0.7%) | 0.60 (0.14–2.53) | 8/2211 (0.4%) | 12/2200 (0.6%) | 0.66 (0.27–1.62) | 0.91 |
| Gastrointestinal bleeding (BARC 3 or 5) | 8/700 (1.2%) | 6/708 (0.9%) | 1.35 (0.47–3.90) | 14/2200 (0.7%) | 14/2193 (0.7%) | 1.00 (0.47–2.09) | 0.64 |
| Gastrointestinal bleeding (BARC 2, 3, or 5) | 13/694 (1.9%) | 8/708 (1.1%) | 1.66 (0.69–4.00) | 28/2195 (1.3%) | 27/2186 (1.3%) | 1.03 (0.61–1.75) | 0.36 |
BARC, Bleeding Academic Research Consortium; CI, confidence interval; GUSTO, Global Use of Strategies to Open Occluded Arteries; PCI, percutaneous coronary intervention; TIMI, Thrombolysis in Myocardial Infarction.
The cumulative incidence of the co-primary bleeding endpoint at 1 year was 2.1% in the aspirin group and 2.7% in the clopidogrel group in patients with complex PCI (HR, 0.78; 95% CI, 0.39–1.56; P = 0.48), while it was 1.7% in the aspirin group and 1.4% in the clopidogrel group in patients without complex PCI (HR, 1.17; 95% CI, 0.72–1.90; P = 0.54) (Figure 2B). There was no significant treatment-by subgroup interaction in patients with and without complex PCI for the co-primary bleeding endpoint (P for interaction = 0.35).
Clinical outcomes in the overall 1-year analysis: aspirin group vs. clopidogrel group in patients with and without complex percutaneous coronary intervention
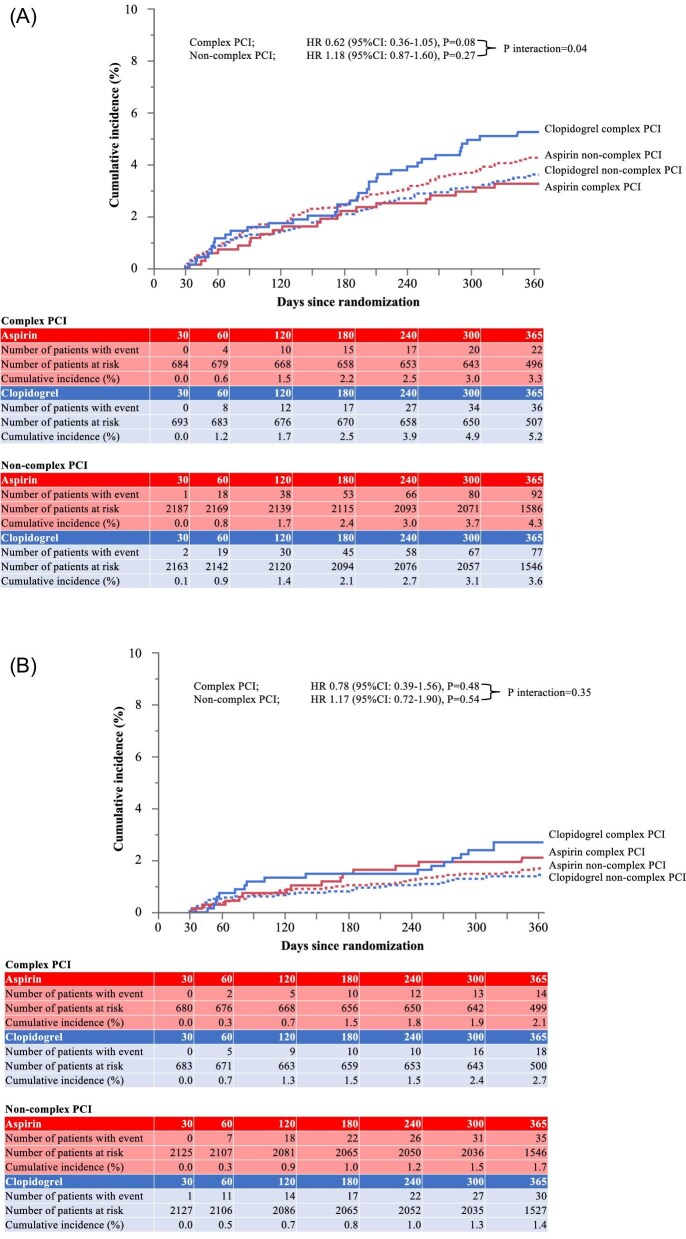
In the overall 1-year analysis, the cumulative incidence of the co-primary cardiovascular endpoint at 1 year was 8.3% in the aspirin group and 9.8% in the clopidogrel group in patients with complex PCI (HR, 0.85; 95% CI 0.61–1.20; P = 0.37), while it was 7.3% in the aspirin group and 7.3% in the clopidogrel group in patients without complex PCI (HR, 0.99; 95% CI 0.80–1.23; P = 0.94) (Figure 3A). There was no significant treatment-by subgroup interaction in patients with and without complex PCI for the co-primary cardiovascular endpoint (P for interaction = 0.47).
Figure 3.
Kaplan–Meier curves in the overall 1-year analysis. (A) co-primary cardiovascular endpoint: a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, or ischaemic stroke. (B) co-primary bleeding endpoint: BARC 3 or 5 bleeding. BARC, Bleeding Academic Research Consortium; CI, confidence interval; DAPT, dual antiplatelet therapy; HR, hazard ratio; PCI, percutaneous coronary intervention.
The cumulative incidence of the co-primary bleeding endpoint at 1 year was 6.2% in the aspirin group and 7.8% in the clopidogrel group in patients with complex PCI (HR, 0.79; 95% CI, 0.53–1.17; P = 0.24), while it was 6.6% in the aspirin group and 5.6% in the clopidogrel group in patients without complex PCI (HR, 1.19; 95% CI 0.94–1.52; P = 0.15) (Figure 3B). There was no significant treatment-by subgroup interaction in patients with and without complex PCI for the co-primary cardiovascular endpoint (P for interaction = 0.08).
Results for the secondary endpoints in the 30-day landmark and the overall 1-year analyses are shown in Table 3 and Supplementary material online, Table S6.
Discussion
The main findings in the present study were as follows: (i) The effect of aspirin monotherapy relative to clopidogrel monotherapy beyond 1 month and up to 1 year was not significant for cardiovascular events regardless of complex PCI, but there was a significant interaction between complex PCI and the effect of aspirin monotherapy relative to clopidogrel monotherapy for cardiovascular events due to numerically lower risk of aspirin monotherapy in patients with complex PCI. (ii) The effect of aspirin monotherapy relative to clopidogrel monotherapy beyond 1 month and up to 1 year was not significant for bleeding events regardless of complex PCI without significant interaction.
The European Society of Cardiology and the Japanese Circulation Society guidelines have suggested that complex PCI is a risk factor of stent-driven recurrent ischaemic events.11,12 Indeed, patients with complex PCI had higher incidence of cardiovascular events compared with those without in the overall 1-year analysis in the present study. Giustino et al.6 reported that prolonged DAPT (12–24 months) compared with short DAPT (3–6 months) reduced major adverse cardiac events and coronary thrombotic events after complex PCI in the individual patient data-level meta-analysis of six randomized clinical trials in which aspirin monotherapy was mainly used after stopping short DAPT. More recently, Gragnano et al.7 showed that P2Y12 inhibitor (ticagrelor, prasugrel, or clopidogrel) monotherapy after very short DAPT (1–3 months) compared with standard DAPT was not associated with an increased risk of cardiovascular events after complex PCI in the individual patient data-level meta-analysis of five randomized clinical trials. In addition, the subgroup analysis of the STOPDAPT-2 Total Cohort (ShorT and OPtimal Duration of Dual AntiPlatelet Therapy-2 Total Cohort) showed that clopidogrel monotherapy after 1-month DAPT compared with standard DAPT was not associated with an increased risk of cardiovascular events regardless of complex PCI.8 When considering the above studies, it might be hypothesized that P2Y12 inhibitor monotherapy might be a preferred antiplatelet monotherapy after stopping short DAPT regimen. Indeed, the HOST-EXAM (HOST-EXtended Antiplatelet Monotherapy) trial demonstrated that clopidogrel monotherapy reduced cardiovascular events compared with aspirin monotherapy in patients with chronic phase after PCI.16 The 5-year follow-up of the STOPDAPT-2 trial also showed that clopidogrel monotherapy was associated with numerically lower risk of cardiovascular events compared with aspirin monotherapy beyond 1 year after PCI.17 Moreover, in an individual patient data-level meta-analysis of randomized clinical trials comparing P2Y12 inhibitor monotherapy with aspirin monotherapy in patients with coronary artery disease, P2Y12 inhibitor monotherapy reduced cardiovascular events compared with aspirin monotherapy.18 However, there were no previous clinical trials comparing aspirin monotherapy to P2Y12 inhibitor monotherapy following short or very short DAPT after complex PCI. In the present study, the incidence of cardiovascular events beyond 1 month and up to 1 year was not different between aspirin monotherapy and clopidogrel monotherapy regardless of complex PCI. There was a significant interaction between complex PCI and the effect of aspirin monotherapy relative to clopidogrel monotherapy for cardiovascular events and the net clinical benefit due to numerically lower risk of aspirin monotherapy in patients with complex PCI. Aspirin monotherapy might be an attractive regimen after stopping short DAPT in patients undergoing complex PCI. However, the positive interaction in the present study should be interpreted with cautious, because the positive interaction was derived from a small subgroup and the results in the entire study population of the 1-year analysis of the STOPDAPT-3 trial were neutral.10 In addition, the result in the 1-year analysis of the STOPDAPT-3 trial was not consistent with the previous studies comparing P2Y12 inhibitor monotherapy with aspirin monotherapy conducted in the chronic phase after PCI.16,17 Further studies would be warranted to evaluate the optimal antiplatelet monotherapy after short DAPT in patients who underwent complex PCI.
The previous studies reported that patients undergoing complex PCI had higher incidence of bleeding events compared with those undergoing non-complex PCI.6–8 The incidence of bleeding events was higher in patients with complex PCI than in those without complex PCI in the 30-day landmark analysis in the present study. In the main analysis of the 1-year results of the STOPDAPT-3, the incidence of major bleeding was not different between aspirin monotherapy and clopidogrel monotherapy, which was consistent with the trials conducted in the chronic phase after PCI such as the 5-year results of the STOPDAPT-2 and an individual patient data-level meta-analysis.10,17,18 In the present study, the incidence of major bleeding was also not different between aspirin monotherapy relative to clopidogrel monotherapy regardless of complex PCI without significant interaction. In the HOST-EXAM trial, clopidogrel monotherapy was associated with lower incidence of major bleeding compared with aspirin monotherapy.16 The higher prescription rate of proton pump inhibitors in the STOPDAPT-3 trial compared with the HOST-EXAM trial might be one of the reasons to cancel out the benefit of clopidogrel over aspirin in reducing bleeding events. To reduce bleeding events after complex PCI, further studies would be needed to evaluate optimal antiplatelet monotherapy and to explore the necessity of prophylactic use of proton pump inhibitors in patients taking P2Y12 inhibitor or aspirin monotherapy.
Limitations
The original 1-year results of the STOPDAPT-3 trial had important limitations.10 Most importantly, the randomization was made only once at the timing of the index PCI and not 1 month. Strictly speaking, this trial was not a randomized comparison between aspirin and clopidogrel used as monotherapy after 1 month. However, the number of study patients was large enough and the number of patients having events within 1 month was not significantly different regardless of complex PCI, and the balance of baseline characteristics in the groups was well maintained in the 30-day landmark analysis.9,10,13 The influence of immortal bias associated with the landmark analysis should be considered. However, it might not be considered large enough to change the results, because the overall 1-year analysis confirmed the consistency of the results. In addition, the changes in the antiplatelet therapy (prasugrel monotherapy to clopidogrel monotherapy in the no-aspirin group and DAPT to aspirin monotherapy in the DAPT group) were mandated not just at 30 days, but between 30 and 59 days after the index PCI. There are other important limitations of this study. First, the present pre-specified subgroup analysis was underpowered, and should be interpreted as exploratory. Second, the prevalence of complex PCI was low. The prevalence of bifurcation with two stents, which was reported as the strongest risk factor for ischaemic events, was much lower than in previous studies.6 Third, the vast majority of the study patients underwent PCI guided by intracoronary imaging devices, which is quite different from the practice in the United States and Europe. The effect of aspirin monotherapy relative to clopidogrel monotherapy might be modified by intracoronary imaging use, especially in patients with complex PCI. Fourth, the prescription rate of DAPT beyond 30 days was numerically higher in patients with complex PCI than in those without.
Conclusion
There was a significant interaction between complex PCI and the effect of aspirin monotherapy relative to clopidogrel monotherapy beyond 1 month and up to 1 year for cardiovascular events due to numerically lower risk of aspirin monotherapy in patients with complex PCI. The effect of aspirin monotherapy relative to clopidogrel monotherapy beyond 1 month and up to 1 year was not different for bleeding events regardless of complex PCI. The positive interaction between complex PCI and the effect of aspirin vs. clopidogrel monotherapy for cardiovascular events should be interpreted with cautious due to the neutral results in the entire study population.
Supplementary Material
Acknowledegments
We appreciate the study investigators for their efforts in enrolling patients and collecting data. We also appreciate the members of Research Institute for Production Development for co-ordinating the study.
Contributor Information
Takenori Domei, Department of Cardiology, Kokura Memorial Hospital, Kitakyushu 802-8555, Japan.
Ko Yamamoto, Department of Cardiology, Kokura Memorial Hospital, Kitakyushu 802-8555, Japan.
Masahiro Natsuaki, Department of Cardiovascular Medicine, Saga University, Saga 849-0937, Japan.
Hirotoshi Watanabe, Department of Cardiology, Hirakata Kohsai Hospital, Hirakata 573-0153, Japan.
Takeshi Morimoto, Department of Clinical Epidemiology, Hyogo College of Medicine, Nishinomiya 663-8501, Japan.
Yuki Obayashi, Department of Cardiovascular Medicine, Kyoto University Graduate School of Medicine, Kyoto 606-8507, Japan.
Ryusuke Nishikawa, Department of Cardiovascular Medicine, Kyoto University Graduate School of Medicine, Kyoto 606-8507, Japan.
Tomoya Kimura, Department of Clinical Epidemiology, Hyogo College of Medicine, Nishinomiya 663-8501, Japan; Department of Cardiology, Japanese Red Cross Wakayama Medical Center, Wakayama 640-8588, Japan.
Kenji Ando, Department of Cardiology, Kokura Memorial Hospital, Kitakyushu 802-8555, Japan.
Satoru Suwa, Department of Cardiology, Juntendo University Shizuoka Hospital, Izunokuni 410-2211, Japan.
Tsuyoshi Isawa, Department of Cardiology, Sendai Kousei Hospital, Sendai 981-0914, Japan.
Hiroyuki Takenaka, Department of Cardiology, Hirakata Kohsai Hospital, Hirakata 573-0153, Japan.
Tetsuya Ishikawa, Department of Cardiology, Dokkyo Medical University Saitama Medical Center, Koshigaya 343-0845, Japan.
Toshihiro Tamura, Department of Cardiology, Tenri Hospital, Tenri 632-8552, Japan.
Kando Kawahatsu, Department of Cardiology, Teine Keijinkai Hospital, Teine 006-0811, Japan.
Fujio Hayashi, Department of Cardiovascular Center, Japanese Red Cross Osaka Hospital, Osaka 543-8555, Japan.
Mitsuru Abe, Department of Cardiology, Kyoto Medical Center, Kyoto 612-0861, Japan.
Takeshi Serikawa, Department of Cardiology, Fukuoka Wajiro Hospital, Fukuoka 811-0213, Japan.
Hiroyoshi Mori, Showa University Fujigaoka Hospital, Yokohama 227-8501, Japan.
Takayuki Kawamura, Department of Cardiology, Kindai University Faculty of Medicine, Osakasayama 589-8511, Japan.
Arata Hagikura, Division of Cardiology, Tsukazaki Hospital, Himeji 671-1227, Japan.
Naoki Shibata, Ogaki Municipal Hospital, Ogaki 503-8502, Japan.
Koh Ono, Department of Cardiovascular Medicine, Kyoto University Graduate School of Medicine, Kyoto 606-8507, Japan.
Takeshi Kimura, Department of Cardiology, Hirakata Kohsai Hospital, Hirakata 573-0153, Japan.
Funding
Abbott Medical Japan.
Conflicts of interest: K.Y. reports honoraria from Abbott Medical Japan, Daiichi Sankyo, and Boston Scientific. M.N. reports honoraria from Abbott Medical Japan, Daiichi Sankyo, Medtronic, Terumo, Japan Lifeline, Asahi Intecc, Bristol-Myers Squibb, Otsuka, Amgen, Sanofi, Takeda, and Bayer. H.W. reports personal fees from Abbott Medical Japan during the conduct of the study as well as personal fees from Daiichi Sankyo, Kowa, Abiomed, Bayer, Pfizer, Bristol-Myers Squibb, and Otsuka outside the submitted work. T.M. reports lecturer's fees from Abbott, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Japan Lifeline, Pfizer, Tsumura, and UCB; manuscript fee from Pfizer; and advisory board for GlaxoSmithKline, Novartis, and Teijin. S.S. reports personal fees from Abbott Medical Japan and Daiichi Sankyo outside the submitted work. Takeshi Kimura reports grants from Abbott Medical Japan and Boston Scientific and being an advisory board member of Abbott Medical Japan and Terumo Japan. No other disclosures were reported.
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
- 1. Vranckx P, Valgimigli M, Juni P, Hamm C, Steg PG, Heg D, van Es GA, McFadden EP, Onuma Y, van Meijeren C, Chichareon P, Benit E, Möllmann H, Janssens L, Ferrario M, Moschovitis A, Zurakowski A, Dominici M, Van Geuns RJ, Huber K, Slagboom T, Serruys PW, Windecker S; GLOBAL LEADERS investigators . Ticagrelor plus aspirin for 1 month, followed by ticagrelor monotherapy for 23 months vs aspirin plus clopidogrel or ticagrelor for 12 months, followed by aspirin monotherapy for 12 months after implantation of a drug-eluting stent: a multicentre, open-label, randomised superiority trial. Lancet 2018;392:940–949. [DOI] [PubMed] [Google Scholar]
- 2. Watanabe H, Domei T, Morimoto T, Natsuaki M, Shiomi H, Toyota T, Ohya M, Suwa S, Takagi K, Nanasato M, Hata Y, Yagi M, Suematsu N, Yokomatsu T, Takamisawa I, Doi M, Noda T, Okayama H, Seino Y, Tada T, Sakamoto H, Hibi K, Abe M, Kawai K, Nakao K, Ando K, Tanabe K, Ikari Y, Hanaoka KI, Morino Y, Kozuma K, Kadota K, Furukawa Y, Nakagawa Y, Kimura T. Effect of 1-month dual antiplatelet therapy followed by Clopidogrel vs 12-month dual antiplatelet therapy on cardiovascular and bleeding events in patients receiving PCI the STOPDAPT-2 randomized clinical trial. JAMA 2019;321:2414–2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Hahn J-Y, Song YB, Oh J-H, Chun WJ, Park YH, Jang WJ, Im E-S, Jeong J-O, Cho BR, Oh SK, Yun KH, Cho D-K, Lee J-Y, Koh Y-Y, Bae J-W, Choi JW, Lee WS, Yoon HJ, Lee SU, Cho JH, Choi WG, Rha S-W, Lee JM, Park TK, Yang JH, Choi J-H, Choi S-H, Lee SH, Gwon H-C. Effect of P2Y12 inhibitor monotherapy vs dual antiplatelet therapy on cardiovascular events in patients undergoing percutaneous coronary intervention the SMART-CHOICE randomized clinical trial. JAMA 2019;321:2428–2437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mehran R, Baber U, Sharma SK, Cohen DJ, Angiolillo DJ, Briguori C, Cha JY, Collier T, Dangas G, Dudek D, Džavík V, Escaned J, Gil R, Gurbel P, Hamm CW, Henry T, Huber K, Kastrati A, Kaul U, Kornowski R, Krucoff M, Kunadian V, Marx SO, Mehta SR, Moliterno D, Ohman EM, Oldroyd K, Sardella G, Sartori S, Shlofmitz R, Steg PG, Weisz G, Witzenbichler B, Han Y-L, Pocock S, Gibson CM. Ticagrelor with or without aspirin in high-risk patients after PCI. N Engl J Med 2019;381:2032–2042. [DOI] [PubMed] [Google Scholar]
- 5. Kim B-K, Hong S-J, Cho Y-H, Yun KH, Kim YH, Suh Y, Cho JY, Her A-Y, Cho S, Jeon DW, Yoo S-Y, Cho D-K, Hong B-K, Kwon H, Ahn C-M, Shin D-H, Nam C-M, Kim J-S, Ko Y-G, Choi D, Hong M-K, Jang Y. Effect of ticagrelor monotherapy vs ticagrelor with aspirin on major bleeding and cardiovascular events in patients with acute coronary syndrome the TICO randomized clinical trial. JAMA 2020;323:2407–2416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Giustino G, Chieffo A, Palmerini T, Valgimigli M, Feres F, Abizaid A, Costa RA, Hong M-K, Kim B-K, Jang Y, Kim H-S, Park KW, Gilard M, Morice M-C, Sawaya F, Sardella G, Genereux P, Redfors B, Leon MB, Bhatt DL, Stone GW, Colombo A. Efficacy and safety of dual antiplatelet therapy after complex PCI. J Am Coll Cardiol 2016;68:1851–1864. [DOI] [PubMed] [Google Scholar]
- 7. Gragnano F, Mehran R, Branca M, Franzone A, Baber U, Jang Y, Kimura T, Hahn J-Y, Zhao Q, Windecker S, Gibson CM, Kim B-K, Watanabe H, Song YB, Zhu Y, Vranckx P, Mehta S, Hong S-J, Ando K, Gwon H-C, Calabrò P, Serruys PW, Dangas GD, Mcfadden EP, Angiolillo DJ, Heg D, Valgimigli M. P2Y12 inhibitor monotherapy or dual antiplatelet therapy after complex percutaneous coronary interventions. J Am Coll Cardiol 2023;81:537–552. [DOI] [PubMed] [Google Scholar]
- 8. Yamamoto K, Watanabe H, Morimoto T, Obayashi Y, Natsuaki M, Domei T, Yamaji K, Suwa S, Isawa T, Watanabe H, Yoshida R, Sakamoto H, Akao M, Hata Y, Morishima I, Tokuyama H, Yagi M, Suzuki H, Wakabayashi K, Suematsu N, Inada T, Tamura T, Okayama H, Abe M, Kawai K, Nakao K, Ando K, Tanabe K, Ikari Y, Morino Y, Kadota K, Furukawa Y, Nakagawa Y, Kimura T. Clopidogrel monotherapy after 1-month DAPT in patients with high bleeding risk or complex PCI. JACC Asia 2023;3:31–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Natsuaki M, Watanabe H, Morimoto T, Yamamoto K, Obayashi Y, Nishikawa R, Ando K, Domei T, Suwa S, Ogita M, Isawa T, Takenaka H, Yamamoto T, Ishikawa T, Hisauchi I, Wakabayashi K, Onishi Y, Hibi K, Kawai K, Yoshida R, Suzuki H, Nakazawa G, Kusuyama T, Morishima I, Ono K, Kimura T. An aspirin-free versus dual antiplatelet strategy for coronary stenting: STOPDAPT-3 randomized trial. Circulation 2024;149:585–600. [DOI] [PubMed] [Google Scholar]
- 10. Watanabe H, Natsuaki M, Morimoto T, Yamamoto K, Obayashi Y, Nishikawa R, Kimura T, Ando K, Domei T, Suwa S, Ogita M, Isawa T, Takenaka H, Yamamoto T, Ishikawa T, Hisauchi I, Wakabayashi K, Onishi Y, Hibi K, Kawai K, Yoshida R, Suzuki H, Nakazawa G, Kusuyama T, Morishima I, Ono K, Kimura T. Aspirin versus clopidogrel monotherapy after percutaneous coronary intervention: 1-year follow-up of the STOPDAPT-3 trial. Eur Heart J 2024;45:5042–5054. 10.1093/eurheartj/ehae617 [DOI] [PubMed] [Google Scholar]
- 11. Vrints C, Andreotti F, Koskinas KC, Rossello X, Adamo M, Ainslie J, Banning AP, Budaj A, Buechel RR, Chiariello GA, Chieffo A, Christodorescu RM, Deaton C, Doenst T, Jones HW, Kunadian V, Mehilli J, Milojevic M, Piek JJ, Pugliese F, Rubboli A, Semb AG, Senior R, Ten Berg JM, Van Belle E, Van Craenenbroeck EM, Vidal-Perez R, Winther S, Borger M, Gudmundsdóttir IJ, Knuuti J, Ahrens I, Böhm M, Buccheri S, Capodanno D, Christiansen EH, Collet J-P, Dickstein K, Eek C, Falk V, Henriksen PA, Ibanez B, James S, Kedev S, Køber L, Kyriakou M, Magavern EF, Mcinerny A, Mersha CO, Mihaylova B, Mindham R, Neubeck L, Neumann F-J, Nielsen JC, Paolisso P, Paradies V, Pasquet AA, Piepoli M, Prescott E, Rakisheva A, Rocca B, Ruel M, Sandner S, Saraste A, Szummer K, Vaartjes I, Wijns W, Windecker S, Witkowsky A, Zdrakovic M, Zeppenfeld K, Shuka N, Bouraghda MA, Hayrapetyan HG, Reinstadler SJ, Musayev O, De Pauw M, Kušljugić Z, Gelev V, Skoric B, Karakyriou M, Kovarnik T, Nielsen LH, Abdel-Aziz IS, Ainla T, Porela P, Benamer H, Nadaraia K, Richardt G, Papafaklis MI, Becker D, Gudmundsdóttir IJ, Wolak A, Riccio C, Zholdin BK, Elezi S, Abilova S, Mintale I, Allam B, Badarienė J, Pereira B, Dingli P, Revenco V, Bulatovic N, Benouna EGM, Dedic A, Mitevska I, Angel K, Bryniarski K, Luz AMC, Popescu BA, Bertelli L, Beleslin BD, Hudec M, Fras Z, Freixa-Pamias R, Holm A, Jeger R, Marjeh MYB, Hammami R, Aytekin V, Nesukay EG, Swanson N, Shek AB. 2024 ESC guidelines for the management of chronic coronary syndromes. Eur Heart J 2024;45:3415–3537. 10.1093/eurheartj/ehae177 [DOI] [PubMed] [Google Scholar]
- 12. Nakamura M, Kimura K, Kimura T, Ishihara M, Otsuka F, Kozuma K, Kosuge M, Shinke T, Nakagawa Y, Natsuaki M, Yasuda S, Akasaka T, Kohsaka S, Haze K, Hirayama A. JCS 2020 guideline focused update on antithrombotic therapy in patients with coronary artery disease. Circ J 2020;84:831–865. [DOI] [PubMed] [Google Scholar]
- 13. Yamamoto K, Natsuaki M, Watanabe H, Morimoto T, Obayashi Y, Nishikawa R, Ando K, Suwa S, Isawa T, Takenaka H, Ishikawa T, Tamura T, Kawahatsu K, Hayashi F, Akao M, Serikawa T, Mori H, Kawamura T, Hagikura A, Shibata N, Ono K, Kimura T; STOPDAPT-3 investigators. An aspirin-free strategy for immediate treatment following complex percutaneous coronary intervention. JACC Cardiovasc Interv 2024;17:1119–1130. 10.1016/j.jcin.2024.03.017 [DOI] [PubMed] [Google Scholar]
- 14. Mehran R, Rao SV, Bhatt DL, Gibson CM, Caixeta A, Eikelboom J, Kaul S, Wiviott SD, Menon V, Nikolsky E, Serebruany V, Valgimigli M, Vranckx P, Taggart D, Sabik JF, Cutlip DE, Krucoff MW, Ohman EM, Steg PG, White H. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation 2011;123:2736–2747. [DOI] [PubMed] [Google Scholar]
- 15. Cutlip DE, Windecker S, Mehran R, Boam A, Cohen DJ, Van Es G-A, Gabriel Steg P, Morel M-Angè, Mauri L, Vranckx P, Mcfadden E, Lansky A, Hamon M, Krucoff MW, Serruys PW. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation 2007;115:2344–2351. [DOI] [PubMed] [Google Scholar]
- 16. Koo B-K, Kang J, Park KW, Rhee T-M, Yang H-M, Won K-B, Rha S-W, Bae J-W, Lee NH, Hur S-H, Yoon J, Park T-H, Kim BS, Lim SW, Cho YH, Jeon DW, Kim S-H, Han J-K, Shin E-S, Kim H-S, Koo B-K, Kang J, Park KW, Rhee T-M, Lee H, Yang H-M, Won K-B, Rha S-W, Bae J-W, Lee NH, Hur S-H, Yoon J, Park T-H, Kim BS, Lim SW, Cho YH, Jeon DW, Kim S-H, Han J-K, Shin E-S, Kim H-S, Han K-R, Moon K-W, Oh SK, Kim U, Rhee M-Y, Kim D-I, Kim S-Y, Lee S-Y, Lee SU, Kim S-W, Kim SY, Jeon H-K, Cha KS, Jo S-H, Ryu JK, Suh I-W, Choi H-H, Woo S-I, Chae I-H, Shin W-Y, Kim D-K, Oh JH, Jeong MH, Kim YH. Aspirin versus clopidogrel for chronic maintenance monotherapy after percutaneous coronary intervention (HOST-EXAM): an investigator-initiated, prospective, randomised, open-label, multicentre trial. Lancet 2021;397:2487–2496. [DOI] [PubMed] [Google Scholar]
- 17. Watanabe H, Morimoto T, Natsuaki M, Yamamoto K, Obayashi Y, Nishikawa R, Ando K, Ono K, Kadota K, Suwa S, Morishima I, Yoshida R, Hata Y, Akao M, Yagi M, Suematsu N, Morino Y, Yokomatsu T, Takamisawa I, Noda T, Doi M, Okayama H, Nakamura Y, Hibi K, Sakamoto H, Noguchi T, Kimura T. Clopidogrel vs aspirin monotherapy beyond 1 year after percutaneous coronary intervention. J Am Coll Cardiol 2024;83:17–31. [DOI] [PubMed] [Google Scholar]
- 18. Gragnano F, Cao D, Pirondini L, Franzone A, Kim H-S, Von Scheidt M, Pettersen A-ÅR, Zhao Q, Woodward M, Chiarito M, Mcfadden EP, Park KW, Kastrati A, Seljeflot I, Zhu Y, Windecker S, Kang J, Schunkert H, Arnesen H, Bhatt DL, Steg PG, Calabrò P, Pocock S, Mehran R, Valgimigli M. P2Y12 inhibitor or aspirin monotherapy for secondary prevention of coronary events. J Am Coll Cardiol 2023;82:89–105. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.