Highlights
-
•
Granulosa Cell Tumor (GCT) represent 2%-5% of ovarian cancers.
-
•
Stereotactic Body Radiation Therapy (SBRT) is considered an effective treatment of oligometastatic gynecologic cancer.
-
•
SBRT for GCT metastasis demonstrates a marked Inhibin B and radiographic response.
-
•
SBRT may be an effective option for local control of GCT recurrence in patients non-amendable to surgical resection.
Keywords: Granulosa cell tumor, Stereotactic body radiation therapy, Inhibin B
Abstract
Stereotactic body radiation therapy (SBRT) is a safe and effective treatment for oligometastatic gynecologic cancers. This report details the case of a 75-year-old woman with recurrent granulosa cell tumor of the left ovary. The patient was diagnosed with a peritoneal recurrence after her inhibin-B level increased to 39.7 pg/mL, and a subsequent computed tomography (CT) scan of the abdomen and pelvis revealed an enlarging left-sided soft tissue mass in the sigmoid mesentery measuring 1.2 × 1.2 cm. She underwent SBRT, delivering 37.5 Gy in 5 fractions to the soft tissue mass. A CT scan post-treatment indicated the mass had decreased to 0.8 cm and that her inhibin B level decreased to < 7 pg/mL. This case highlights the effectiveness of SBRT in the management of oligometastatic ovarian cancer, achieving a marked radiographic and tumor marker response in patients with disease not amenable to surgical resection.
1. Introduction
Stereotactic body radiation therapy (SBRT) has been identified as a safe and effective technique for the local treatment of oligometastatic gynecologic cancer (Sherwani et al., 2023). While there is no universally accepted definition for an oligometastatic state, it commonly is defined by the presence of 1–5 metastases (Hellman and Weichselbaum, 1995).
SBRT is utilized to treat solid tumors in both primary and oligometastatic cancers (Fields et al., 2017, Macchia et al., 2020). Compared to traditional radiotherapy, SBRT employs non-coplanar, volumetric modulated arc therapy (VMAT), which is a radiation delivery method in which the linear accelerator rotates around the patient and facilitates the delivery of radiation from different beam angles and intensities that are not of the same plane. This technique allows for precise tumor targeting and minimizes dose spillage to surrounding healthy tissue. Additionally, daily on-board imaging allows for dose escalation, while maximally sparing adjacent organs at risk (OARs). These considerations are particularly important when treating tumors in the pelvis in which the OARs are mobile. These features make this intervention particularly well-suited for the management of oligometastatic deposits adjacent to sensitive structures such as small bowel (Macchia et al., 2020). Toxicities from SBRT may include diarrhea, rectal and bladder irritation, bowel strictures, urethral injury and nerve damage (Lazzari et al., 2018, Macchia et al., 2023).
Currently, radiation therapy is an acceptable option for palliative management of recurrent, oligometastatic granulosa cell tumor (GCT) (Armstrong et al., 2022). While the benefits of SBRT are recognized in the treatment of oligometastatic ovarian cancer, there is limited data available on the use of SBRT specifically for oligometastatic GCT treatment. GCTs of the ovary represent 2 %-5% of ovarian cancers and originate from sex-cord stromal cells (Koukourakis et al., 2008). The main subtype of GCT is the adult subtype, which represents 95 % of cases. This subtype most commonly presents in perimenopausal and postmenopausal women, with a median age at diagnosis of 508. GCTs are slow growing, with a 20–50 % risk of recurrence for up to 25 years following initial therapy (Koukourakis et al., 2008, Li et al., 2021).
Inhibin B is the primary tumor marker for GCTs. These tumors are often associated with an elevated inhibin B prior to surgery with an immediate decline post resection (Koukourakis et al., 2008). As such, inhibin B is often utilized as a biomarker for tumor recurrence (Koukourakis et al., 2008). In this case report, we present a patient with recurrent GCT who was treated with SBRT and had a marked radiographic and tumor biomarker response.
2. Patient case
The patient is a 75-year-old with recurrent GCT of the ovary. In 2005, she underwent a laparoscopic right salpingo-oophorectomy for an adnexal mass and was diagnosed with Stage II GCT. She underwent surgical staging with a total abdominal hysterectomy and left salpingo-oophorectomy and subsequently received four cycles of adjuvant chemotherapy consisting of bleomycin, etoposide, and cisplatin. In May 2012, she was diagnosed with recurrence and underwent a tumor debulking surgery with optimal cytoreduction followed by intraperitoneal chemotherapy with cisplatin and paclitaxel. She continued maintenance docetaxel until February of 2014. In 2017, she switched to our medical center and was without evidence of disease. She was subsequently placed on maintenance letrozole. In 2019, she was diagnosed with an isolated recurrence of the spleen and underwent an open splenectomy.
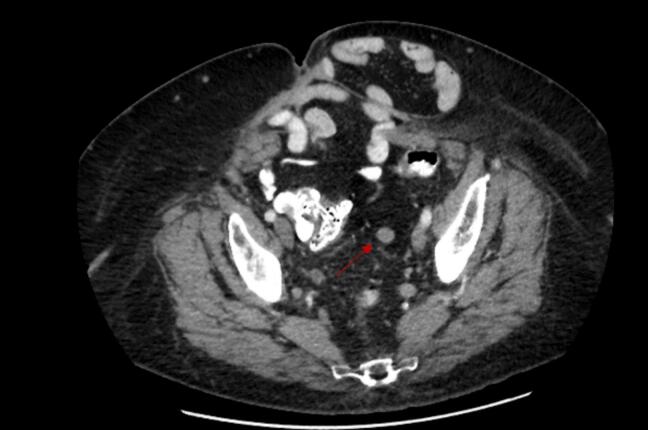
In July 2023, there was an up-trend in her inhibin B level to 17.6 pg/mL, but computed tomography (CT) of her abdomen and pelvis did not show evidence of disease. In February 2024, her inhibin-B more than doubled to 39.7 pg/mL, and subsequent CT abdomen and pelvis showed a peritoneal recurrence – an enlarging left-sided pelvic soft tissue mass in the sigmoid mesentery measuring 1.2 x 1.2 cm (Fig. 1). After discussion at a multi-disciplinary conference, this lesion was presumed to be a recurrence. The lesion was not accessible to percutaneous biopsy, and she had a complex ventral hernia that would significantly increase the complexity of a repeat laparotomy; therefore, the decision was made to proceed with SBRT.
Fig. 1.
CT Image of Peritoneal Implant Prior to SBRT.
During the radiation therapy planning process, the patient underwent CT simulation supine in a custom SBRT Vac-Lok. The planning target volume (PTV) was generated by delineating the gross tumor with a 7 mm margin. The nearest OAR was the small bowel, and a 7 mm expansion was also utilized to generate a small bowel planning organ at risk volume (PRV). The SBRT dosage was chosen based on the acceptable dose and fractionation schedule outlined in the SABR-COMET-10 study (Palma et al., 2019). The patient received 37.5 Gy in 5 fractions delivered every other day to the PTV, which was also in acceptable range for dose tolerance of the small bowel.
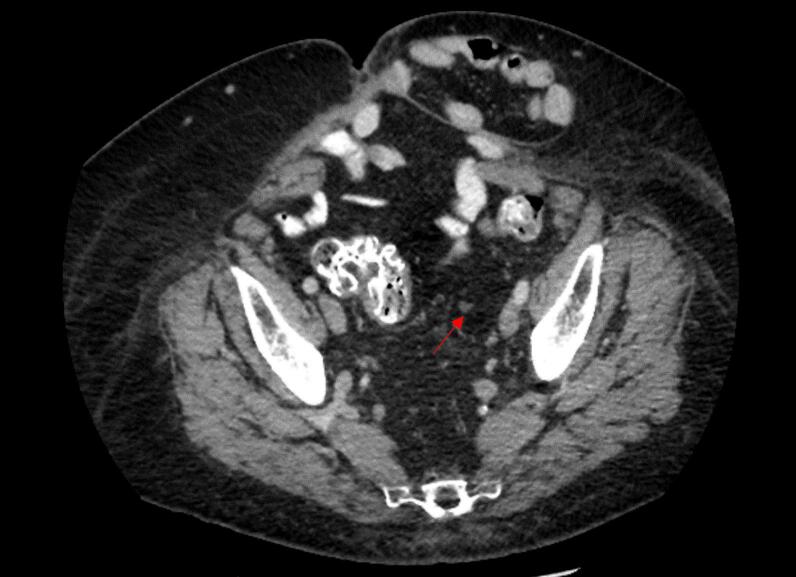
At her 1-month outpatient follow-up, the patient did not have any change in bowel habits and had no dysuria or fatigue. Her inhibin B was < 7 pg/mL at that time. The patient was next seen in the outpatient clinic for her 5-month follow-up. The post-treatment CT scan at that time showed a marked treatment response with the lesion measuring 0.8 cm (Fig. 2). She remained asymptomatic and her inhibin B continued to be < 7 pg/mL. Since her inhibin B remained < 7 pg/mL at that time, surveillance was recommended. At her 8-month follow-up in December of 2024, she continued to feel well overall with no new symptoms. Her inhibin B remained < 7 pg/mL. The follow-up time of 8 months is insufficient to rule out the occurrence of late toxicities following SBRT.
Fig. 2.
CT Image After Completion of SBRT showing a Partial Response.
3. Discussion
SBRT in oligometastatic ovarian cancer is associated with overall favorable response rates. A 2020 systematic review assessed the safety and efficacy of SBRT in gynecological oligometastases and included 16 studies in the analysis with a total of 667 patients. More than half of the patients (57.6 %) had a primary ovarian cancer. The overall response rate with SBRT was 49–97 % and the 2-year local control rate was 71–100 % (Yegya-Raman et al., 2020). The largest contribution of patients for this review came from a 2020 multicenter retrospective study of 261 patients. The main cohort of patients had high-grade serous ovarian cancer. In this study, the complete and partial response rates were 65.2 % and 23.8 % of lesions, respectively, and the overall clinical benefit was 96.4 % (Macchia et al., 2020). There was no germ cell cohort, although there were 16 unspecified malignancies.
The 1999 Wolf et al. retrospective cases series was one of the first cases series to publish on the results of radiotherapy for advanced and recurrent GCTs. There were 14 patients included in this study and 6 patients had a complete clinical response at a median follow-up of 13 years (Wolf et al., 1999). Although most patients received whole-abdomen radiation, it established the use of radiation therapy in GCT treatment. The literature supporting the efficacy of SBRT in patients with oligometastatic GCTs is limited. A 2012 retrospective study of 20 patients included 2 patients with primary GCTs. At a median follow-up of 25 months, local control and overall survival rates were 85 % and 80 % (Chaudhry et al., 2012). Similarly, a 2018 retrospective study with 82 patients with ovarian oligometastases included 7 patients with GCT. Following SBRT therapy, the 2-year local progression-free survival and overall survival rates were 68 % and 71 %, respectively (Lazzari et al., 2018). Most recently, a 2020 retrospective cohort of 35 patients included 1 patient with GCT and found 83 % local control at an average follow-up time of 33.67 months (Kowalchuk et al., 2020).
In the above literature, the response of inhibin B following SBRT was not reported for GCT patients. Additionally, there is insufficient literature on the response of inhibin B after radiotherapy of any form. A 2021 case report presented a patient with pelvic and abdominal GCT recurrence who demonstrated a 98 % decrease in inhibin B following more protracted radiation courses (Singh et al., 2021). While it is can be assumed that inhibin B would respond to SBRT given its established role as a tumor marker for GCT, our case provides the first evidence of inhibin B response post-SBRT.
To our knowledge, this is the first case to demonstrate a rapid inhibin B response to SBRT therapy for GCT and is only the second to detail inhibin B response following any form of radiotherapy. Our patient’s inhibin B decreased from 39.7 pg/mL to < 7 pg/mL following 5 fractions totaling 37.5 Gy of SBRT. In sum, our case demonstrates that SBRT may be an effective option for local control of GCT recurrence, especially for patients not amenable to surgical resection and inhibin B may be a useful marker of treatment response.
Financial support: none.
Informed consent: Written informed consent was obtained from the patient for publication of this case report and accompanying images.
CRediT authorship contribution statement
Madeline Petty: Writing – review & editing, Writing – original draft. Vasanti Jhaveri: Writing – review & editing, Writing – original draft. Nicholas Taylor: Writing – review & editing, Conceptualization. Benjamin Foster: Writing – review & editing, Conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Armstrong D.K., Alvarez R.D., Backes F.J., et al. NCCN Guidelines® insights: ovarian cancer, Version 3.2022. J Natl Compr Canc Netw. 2022;20(9):972–980. doi: 10.6004/jnccn.2022.0047. [DOI] [PubMed] [Google Scholar]
- Chaudhry H., Oermann E., Suy S., et al. Stereotactic body radiation therapy (SBRT) for single small (<5 cm) extracranial ovarian or uterine cancer metastases: promising local control and normal tissue tolerance with 5 fraction approach. Int. J. Radiat. Oncol. Biol. Phys. 2012;84(3):S448. doi: 10.1016/j.ijrobp.2012.07.1187. [DOI] [Google Scholar]
- Fields E.C., McGuire W.P., Lin L., Temkin S.M. Radiation treatment in women with ovarian cancer: past, present, and future. Front. Oncol.. 2017;7:177. doi: 10.3389/fonc.2017.00177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellman S., Weichselbaum R.R. Oligometastases. J Clin Oncol. 1995;13(1):8–10. doi: 10.1200/JCO.1995.13.1.8. [DOI] [PubMed] [Google Scholar]
- Koukourakis G.V., Kouloulias V.E., Koukourakis M.J., et al. Granulosa cell tumor of the ovary: tumor review. Integr Cancer Ther. 2008;7(3):204–215. doi: 10.1177/1534735408322845. [DOI] [PubMed] [Google Scholar]
- Kowalchuk R.O., Waters M.R., Richardson K.M., et al. Stereotactic body radiation therapy in the treatment of ovarian cancer. Radiat. Oncol. 2020;15(1):108. doi: 10.1186/s13014-020-01564-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazzari R., Ronchi S., Gandini S., et al. Stereotactic body radiation therapy for oligometastatic ovarian cancer: a step toward a drug holiday. Int J Radiat Oncol Biol Phys. 2018;101(3):650–660. doi: 10.1016/j.ijrobp.2018.03.058. [DOI] [PubMed] [Google Scholar]
- Li J., Chu R., Chen Z., et al. Progress in the management of ovarian granulosa cell tumor: a review. Acta Obstet. Gynecol. Scand.. 2021;100(10):1771–1778. doi: 10.1111/aogs.14189. [DOI] [PubMed] [Google Scholar]
- Macchia G., Lazzari R., Colombo N., et al. A large, multicenter, retrospective study on efficacy and safety of stereotactic body radiotherapy (SBRT) in oligometastatic ovarian cancer (MITO RT1 Study): a collaboration of MITO, AIRO GYN, and MaNGO groups. Oncologist. 2020;25(2):e311–e320. doi: 10.1634/theoncologist.2019-0309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macchia G., Pezzulla D., Cilla S., et al. Stereotactic body reirradiation in gynaecological cancer: outcomes and toxicities from a single institution experience. Clin Oncol (r Coll Radiol). 2023;35(10):682–693. doi: 10.1016/j.clon.2023.07.011. [DOI] [PubMed] [Google Scholar]
- Palma D.A., Olson R., Harrow S., et al. Stereotactic ablative radiotherapy for the comprehensive treatment of 4–10 oligometastatic tumors (SABR-COMET-10): study protocol for a randomized phase III trial. BMC Cancer. 2019;19(1):816. doi: 10.1186/s12885-019-5977-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherwani Z., Parikh S., Yegya-Raman N., et al. Stereotactic body radiation therapy in gynecologic oligometastases: an effective but underutilized approach. Cancers (Basel) 2023;15(13):3526. doi: 10.3390/cancers15133526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh S.A., Dahshan B., Krc R.F., McDermott D.M., Jacobson G.M. Radiographic and tumor biomarker response to radiotherapy for recurrent granulosa cell tumor of the ovary. Cureus. 2021;13(2) doi: 10.7759/cureus.13154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf J.K., Mullen J., Eifel P.J., Burke T.W., Levenback C., Gershenson D.M. Radiation treatment of advanced or recurrent granulosa cell tumor of the ovary. Gynecol Oncol. 1999;73(1):35–41. doi: 10.1006/gyno.1998.5287. [DOI] [PubMed] [Google Scholar]
- Yegya-Raman N., Cao C.D., Hathout L., et al. Stereotactic body radiation therapy for oligometastatic gynecologic malignancies: a systematic review. Gynecol. Oncol.. 2020;159(2):573–580. doi: 10.1016/j.ygyno.2020.08.010. [DOI] [PubMed] [Google Scholar]