Abstract
How to cite this article: Govil D, Pachisia AV. Blue Hopes, Red Flags: The Methylene Blue Dilemma in Sepsis Shock. Indian J Crit Care Med 2025;29(2):95–97.
Keywords: Circulatory failure, Methylene blue, Septic shock, Vasoplegia, Vasopressin, Vasopressors
Introduction
Septic shock is defined as a subset of sepsis in which underlying circulatory and cellular/metabolic abnormalities are profound enough to substantially increase mortality.1 Circulatory failure results in inadequate oxygen delivery, which leads to dysfunction at cellular and metabolic levels. This occurs due to impaired aerobic metabolism, the compensatory shift to accelerated aerobic glycolysis, and delayed hepatic clearance.2 To mitigate the effects of circulatory failure resulting from the loss of vasomotor tone, noradrenaline is the first-line vasoactive agent. Its efficacy stems from predominant α-1 adrenergic receptor activation, which induces vasoconstriction, alongside limited β-1 adrenergic activity, which supports the cardiac output.3 Excessive use of adrenergic agents can lead to adverse effects, primarily due to excessive vasoconstriction and dysrhythmias.4 Because of these adverse effects of catecholamine vasopressors, there is great interest in the use of catecholamine-sparing drugs for distributive shock associated with sepsis. Hence, adding a second agent for the management of septic shock may offer a therapeutic advantage. Based on current evidence, the surviving sepsis campaign guidelines recommend vasopressin as the second-line vasopressor for the management of septic shock.3 Lately, there has been renewed interest in methylene blue as one of the few adjuvant molecules for the management of septic shock because of its catecholamine-sparing effect.
Methylene Blue: Theoretical Insights into Its Action
Methylene blue is an odorless, aromatic, heterocyclic blue solution with the chemical formula C16H18ClN3S and a half-life of 5.25 hours.5 It is metabolized by nicotinamide adenine dinucleotide phosphate (NADP) into leucomethylene blue, which is mostly excreted in urine, giving urine a blue-green color. A small portion of methylene blue is excreted in the urine in its unchanged form. A great deal of caution is advised whenever intravenous methylene blue is considered in patients with glucose-6-phosphate dehydrogenase deficiency, as it will lead to hemolytic anemia in these patients because of the limited ability to metabolize methylene blue.6,7
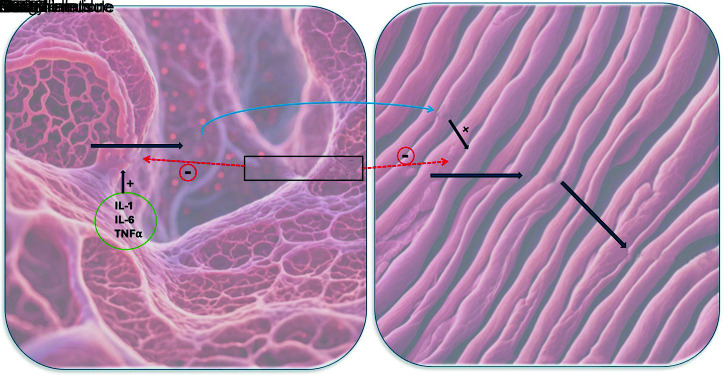
Nitric oxide (NO) is central to the action of methylene blue. Nitric oxide is produced from two forms of nitric oxide synthase (NOS), i.e., Constitutive NOS (cNOS) and inducible NOS (iNOS). cNOS is the constantly active form of enzyme, which is regulated by a negative feedback mechanism. The production of iNOS is induced by endotoxins and cytokines, including interleukin (IL)-1, IL-6, tumor necrosis factor-α, and interferon-γ found in raised levels during sepsis and septic shock, and is not regulated by a negative feedback mechanism. Hence, during septic shock, with a constant increase in inflammatory markers, there is increased production of NO in the endothelium and vascular smooth muscles, culminating in vasodilation. Nitric oxide activates the soluble guanylate cyclase (sGC) enzyme (Fig. 1), which helps in the formation of cyclic guanosine monophosphate (cGMP). Cyclic guanosine monophosphate is the key mediator in activating protein kinase G, promoting calcium reuptake, myosin dephosphorylation, and inducing smooth muscle relaxation.8 Methylene blue binds to the iron heme moiety of sGC, thus inhibiting the action of sGC, and decreasing the cGMP levels, which is responsible for vasodilation.9 In addition, methylene blue is also a direct inhibitor of iNOS.8
Fig. 1.
NO production and action of methylene blue (IL-1, interleukin-1; IL-6, interleukin-6; TNFα, tumor necrosis factor-alpha; NO nitric oxide, iNOS, inducible nitric oxide synthase; cNOS, constitutive nitric oxide synthase; sGC, soluble guanylyl cyclase; cGMP, cyclic guanosine monophosphate; GTP, guanosine triphosphate)
Intravenously administered methylene blue reaches peak concentrations within 30 minutes, with effects beginning 30–60 minutes after administration. It has a relatively high volume of distribution (255 ± 58 L) and is extensively protein-bound (94%).10
Methylene Blue in Septic Shock
The effects of methylene blue on vasoplegia have been recognized for nearly four decades. However, in recent years, there has been a resurgence of interest in exploring its role in septic shock and assessing its clinical efficacy in this context. More than two decades ago, in 2001, Kirov et al. published a pilot study on the use of methylene blue in human septic shock. The study group (methylene blue) and control group (isotonic saline) both had 10 patients each (as an adjunct to conventional treatment). A methylene blue bolus of 2 mg/kg followed 2 hours later by an infusion was given in a stepwise increasing rate, which was maintained for 1 hour. Methylene blue was able to reduce concurrent adrenergic support without affecting oxygen delivery.11 Subsequently, Memis et al. evaluated the effects of methylene blue on cytokine levels during septic shock in a randomized control trial (RCT). They gave an infusion of methylene blue 0.5 mg/kg/hour in the study group. They concluded that although methylene blue did not have any effect on cytokine levels and mortality, it did significantly increase the mean arterial pressure.12 More recently, Ibarra-Estrada et al., in a single-seater RCT, septic shock patients (based on sepsis-3 criteria, requiring noradrenaline) were assigned to the methylene blue group. This group received 500 mg of methylene blue over 6 hours, once daily, for three doses. The control group received 500 mL of 0.9% saline. The patients in the methylene blue group had a statistically significant early discontinuation of vasopressors, one more day of vasopressor-free days at day 28, along with a shorter intensive care unit (ICU) and hospital length of stay, without affecting the mortality and no serious adverse events.13 Responding to the study by Ibarra-Estrada et al., Dubey et al., in a letter to the editor, have argued that since methylene blue is a hypotonic solution, it should not be diluted in normal saline, as it would lead to drug precipitation. Rather, 5% dextrose or sterile water should be used for methylene blue dilution before giving it intravenously.14 In a case series by Luis-Silva et al. of septic shock patients on noradrenaline (≥0.2 µg/kg/min) and vasopressin (0.04 IU/min), methylene blue was given as a bolus of 3 mg/kg over 20 minutes followed by a maintenance dose of 0.5 mg/kg/min for 48 hours. All patients had a reduction of vasopressor dose and lactate levels with methylene blue, along with a reduction of cytokine levels, suggesting that methylene blue may have a role in decreasing inflammatory activity. A meta-analysis of six RCTs has found low-certainty evidence in support of methylene blue decreasing the duration of vasopressors, hospital length of stay or short-term mortality. The study, however, did not find any increase in adverse events.15 Another meta-analysis, published in the same year, which included 11 studies (including non-randomized trials), found that methylene blue use led to significantly lower mortality in septic shock and cardiac surgery subgroups, along with reduced length of ICU and hospital stay. Methylene blue patients were also found to have higher mean arterial pressure and systemic vascular resistance, with no change in cardiac output.16 The dose of methylene blue required in patients with septic shock is still not well-defined. In a recently published study, Shaker et al. have compared a 1 mg/kg bolus dose to a 4 mg/kg dose. This was followed by a methylene blue infusion at 0.25 mg/kg/hour for 72 hours. Both doses had a significantly lower noradrenaline dose requirement when compared to saline infusion, however, between them, there was no difference. After the use of the logistic regression model, the authors found that the 4 mg/kg group had a reduced risk of mortality, with a hazard ratio of 0.29 (95% CI: 0.09–0.90).17 Interestingly, Kuri et al., in their RCT, have compared methylene blue and vasopressin as second-line agents in septic shock patients requiring noradrenaline ≥0.2 µg/kg/min to maintain a mean arterial pressure of 65 mm Hg. The methylene blue group received an intravenous dose of 1 mg/kg over 30 minutes, followed by a 0.5 mg/kg infusion over 6 hours. The vasopressin group received a vasopressin infusion of 0.04 units/min for 6 hours. The study had 37 patients in each group. They found that there was no difference in the noradrenaline dose between the two groups at zero and six hours. However, there was a significant noradrenaline dose reduction in the vasopressin group between 12 and 24 hours when compared to the methylene blue group (p = 0.042 and 0.009, respectively). They have also found that lactate levels and sequential organ failure assessment (SOFA) scores were significantly lower at 24 hours in the vasopressin group (p = 0.048 and 0.022, respectively).18
Conclusion
Based on the findings of Kuri et al., it can be concluded that, given the current body of evidence, methylene blue cannot be considered a suitable replacement for vasopressin as a second-line agent in the treatment of septic shock.18 At present, its role appears to be limited to that of an adjunctive therapy for patients with refractory shock. Furthermore, the optimal dose and timing of intravenous methylene blue in septic shock remain unclear, as the evidence supporting its use remains limited and inconclusive. Furthermore, further RCTs are essential before methylene blue can be widely accepted in clinical practice for the treatment of septic shock. While existing studies provide some preliminary insights, the current body of evidence is insufficient to establish definitive guidelines regarding its efficacy and safety profile.
Orcid
Deepak Govil https://orcid.org/0000-0002-4624-1614
Anant Vikram Pachisia https://orcid.org/0000-0003-1292-1141
Footnotes
Source of support: Nil
Conflict of interest: None
References
- 1.Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;315(8):801–810. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kraut JA, Madias NE. Lactic acidosis. N Engl J Med. 2014;371(24):2309–2319. doi: 10.1056/NEJMra1309483. [DOI] [PubMed] [Google Scholar]
- 3.Guarino M, Perna B, Cesaro AE, Maritati M, Spampinato MD, Contini C, et al. 2023 Update on sepsis and septic shock in adult patients: Management in the emergency department. J Clin Med. 2023;12(9):3188. doi: 10.3390/jcm12093188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dünser MW, Hasibeder WR. Sympathetic overstimulation during critical illness: Adverse effects of adrenergic stress. J Intensive Care Med. 2009;24(5):293–316. doi: 10.1177/0885066609340519. [DOI] [PubMed] [Google Scholar]
- 5.Peter C, Hongwan D, Küpfer A, Lauterburg BH. Pharmacokinetics and organ distribution of intravenous and oral methylene blue. Eur J Clin Pharmacol. 2000;56(3):247–250. doi: 10.1007/s002280000124. [DOI] [PubMed] [Google Scholar]
- 6.Mc Donagh EM, Bautista JM, Youngster I, Altman RB, Klein TE. PharmGKB summary: Methylene blue pathway. Pharmacogenet Genomics. 2013;23(9):498–508. doi: 10.1097/FPC.0b013e32836498f4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Faber P, Ronald A, Millar BW. Methylthioninium chloride: Pharmacology and clinical applications with special emphasis on nitric oxide mediated vasodilatory shock during cardiopulmonary bypass. Anaesthesia. 2005;60(6):575–587. doi: 10.1111/j.1365-2044.2005.04185.x. [DOI] [PubMed] [Google Scholar]
- 8.Hosseinian L, Weiner M, Levin MA, Fischer GW. Methylene blue: Magic bullet for vasoplegia? Anesth Analg. 2016;122(1):194–201. doi: 10.1213/ANE.0000000000001045. [DOI] [PubMed] [Google Scholar]
- 9.Jang DH, Nelson LS, Hoffman RS. Methylene blue for distributive shock: A potential new use of an old antidote. J Med Toxicol. 2013;9(3):242–249. doi: 10.1007/s13181-013-0298-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Puntillo F, Giglio M, Pasqualucci A, Brienza N, Paladini A, Varrassi G. Vasopressor-sparing action of methylene blue in severe sepsis and shock: A narrative review. Adv Ther. 2020;37(9):3692–3706. doi: 10.1007/s12325-020-01422-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kirov MY, Evgenov OV, Evgenov NV, Egorina EM, Sovershaev MA, Sveinbjørnsson B, et al. Infusion of methylene blue in human septic shock: A pilot, randomized, controlled study. Crit Care Med. 2001;29(10):1860–1867. doi: 10.1097/00003246-200110000-00002. [DOI] [PubMed] [Google Scholar]
- 12.Memis D, Karamanlioglu B, Yuksel M, Gemlik I, Pamukcu Z. The influence of methylene blue infusion on cytokine levels during severe sepsis. Anaesth Intensive Care. 2002;30(6):755–762. doi: 10.1177/0310057X0203000606. [DOI] [PubMed] [Google Scholar]
- 13.Ibarra-Estrada M, Kattan E, Aguilera-González P, Sandoval-Plascencia L, Rico-Jauregui U, Gómez-Partida CA, et al. Early adjunctive methylene blue in patients with septic shock: A randomized controlled trial. Crit Care. 2023;27(1):110. doi: 10.1186/s13054-023-04397-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dubey D, Ray P, Imran A. Methylene blue as adjunctive therapy in septic shock: Correct drug diluent derives optimal efficacy. Crit Care. 2023;27(1):337. doi: 10.1186/s13054-023-04615-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fernando SM, Tran A, Soliman K, Flynn B, Oommen T, Wenzhe L, et al. Methylene blue in septic shock: A systematic review and meta-analysis. Crit Care Explor. 2024;6(7):e1110. doi: 10.1097/CCE.0000000000001110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pruna A, Bonaccorso A, Belletti A, Turi S, Di Prima AL, D'amico F, et al. Methylene blue reduces mortality in critically ill and perioperative patients: A meta-analysis of randomized trials. J Cardiothorac Vasc Anesth. 2024;38(1):268–274. doi: 10.1053/j.jvca.2023.09.037. [DOI] [PubMed] [Google Scholar]
- 17.Shaker EH, Soliman AM, Bedewy AAE, Elrawas MM. Comparative study between high and low dose methylene blue infusion in septic cancer patients: A randomized, blinded, controlled study. BMC Anesthesiol. 2025;25(1):15. doi: 10.1186/s12871-024-02792-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kuri HR, Sharma A, Meshram T, Kothari N, Goyal S, Paliwal B, et al. Effects of early use of methylene blue and vasopressin on noradrenaline dose in septic shock: A randomized controlled trial. Indian J Crit Care Med. 2025;29(2):108–112. doi: 10.5005/jp-journals-10071-24905. [DOI] [PMC free article] [PubMed] [Google Scholar]