Abstract
Introduction
Functional neurological disorders (FND) are conditions marked by disruptions in brain network function without structural abnormalities. Sleep disturbances, though under-researched, are commonly observed in FND patients and may worsen symptoms and overall health.
Methods
This systematic review had been registered prospectively in PROSPERO with the registration number: CRD42023446306. Search of PubMed, MEDLINE, Embase, and Cochrane databases identified 218 articles. After removing duplicates and applying exclusion criteria, 9 studies were included in the final analysis.
Results
The analysis showed a significant prevalence of sleep disorders among FND patients, with 58% reporting sleep disturbances, similar to other neuropsychiatric conditions. Studies on psychogenic non-epileptic seizures (PNES) indicated poorer subjective sleep quality and higher insomnia rates compared to epilepsy controls. Limited data on specific measures such as wake after sleep onset (WASO) and Epworth Sleepiness Scale (ESS) scores prevented definitive conclusions.
Discussion
This review is the first systematic examination of sleep disorders in FND. The findings reveal a high prevalence of sleep disturbances, especially among PNES patients, correlating with lower quality of life and increased symptom severity. However, the heterogeneity of studies and limited reporting of specific sleep metrics weaken these conclusions. Further research is needed to investigate the direct impact of sleep quality on FND pathogenesis and management.
Conclusion
Sleep disturbances are prevalent in FND patients and can significantly affect their quality of life. Increased awareness and routine evaluation of sleep in FND patients are recommended. Future studies should explore the relationship between sleep deprivation and FND to develop targeted therapeutic interventions.
Supplementary Information
The online version contains supplementary material available at 10.1007/s10072-024-07931-9.
Keywords: Functional neurological disorder, Sleep disorder, Sleep, Non-epileptic attack disorder (NEAD), WASO, ESS
Introduction
Functional neurological disorders (FND) are a spectrum of conditions that manifest primarily through disruptions in brain network function rather than structural changes and one of the most common causes of Neurology outpatient visits or in-patient admissions [1, 2]. Other chronic conditions like chronic pain and fatigue are also associated with FND and might be a core part of the spectrum [3]. These symptoms can be disabling, negatively affecting employment, activities of daily living, mood and quality of life, often to a greater extent than other neurological diseases [4, 5]. Understanding them is of crucial importance for developing a comprehensive therapeutic approach for management of FND which includes physiotherapy, occupational therapy and psychological approaches [6].
The presence of sleep disturbance has been noted in patients with FND, though limited research is currently present on the clinical relevance of sleep disturbance, its extent, broader characteristics, and impact [7]. Sleep disorders are known to be common in chronic diseases and can lead to worsening outcomes such as mood and quality of life [8, 9]. A study by Aasvik et al. (2018), found that the severity of insomnia had a substantial effect on working memory functioning in patients with comorbid Symptoms of Pain, Fatigue, and Mood Disorders [10]. Additionally, a recent study found that poor sleep (objective and subjective) predicted state dissociation and diminished state sense of agency, both of which are important in the pathomechanism of FND [9]. Unfortunately, sleep disorders are not often routinely evaluated for people with FND and the largest international online survey of FND people did not include it [10]. Hence it is important to identify whether sleep is a clinically relevant factor in pathogenesis of FND as therapeutic interventions targeting sleep disturbances may emerge as additional therapeutic avenues. It is important because various treatments such as cognitive behavioural therapy for insomnia (CBT-i), demonstrate effectiveness in enhancing sleep quality, subsequently influencing various outcomes including mood, quality of life, and severity of symptoms, across several neuropsychiatric and chronic disease cohorts [11, 12].
Therefore, the aim of our review is to analyse relevant literature reporting sleep disturbances/disorders in patients diagnosed with FND and if there is any significant correlation that could be addressed clinically in the management of FND.
Methods
Search strategy and inclusion criteria
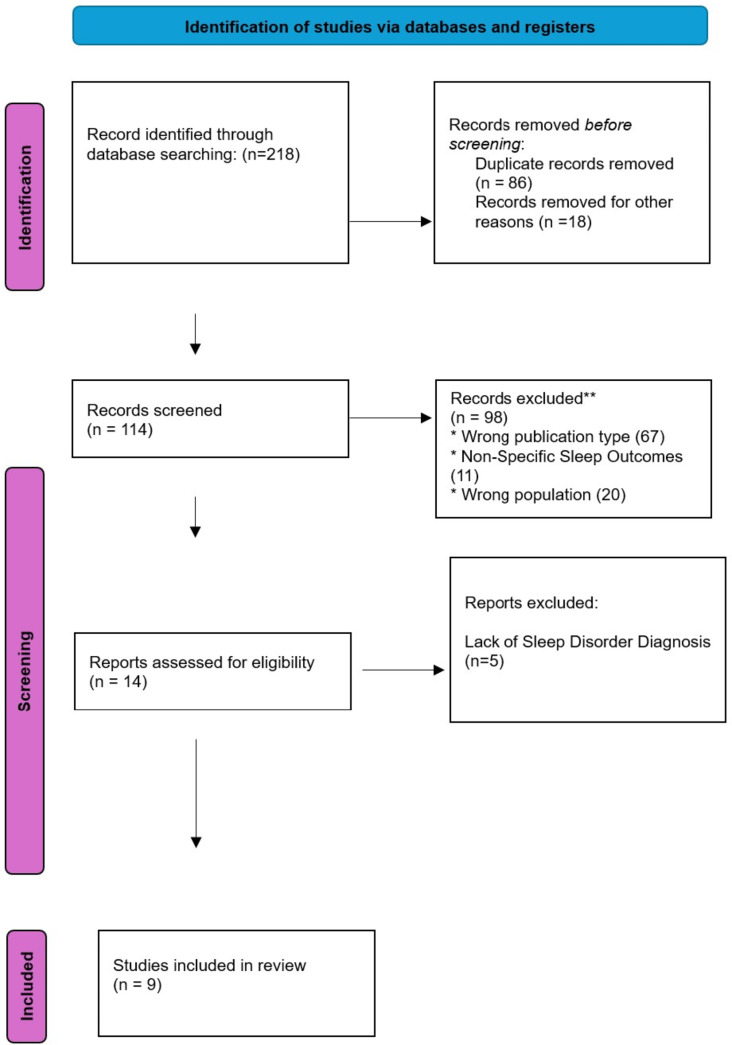
This systematic review was conducted in with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines as demonstrated in Fig. 1. This systematic review had been registered prospectively in the international register PROSPERO under the registration number: CRD42023446306.
Fig. 1.
Preferred reporting items for systematic reviews and meta-analysis (PRISMA)
We searched the PubMed, Embase, Cochrane and Medlinedatabases up to 31st December 2023 for eligible studies. The search strategy was based on the utilisation of MeSH (Medical Subject Headings) term and each keyword search of each database was conducted based on the Boolean operators AND and OR. The searched keywords utilised in the article screening process included the following: “Functional neurological disorder”, “Psychogenic non-epileptic seizures”, “Sleep disorders” “Functional motor disorder” (Supplementary Tables 1–3).
Data extraction
The following data were extracted from each study: The first author’s last name, publication year, sample size, study design, type of FND, and methodology used to assess/monitor sleep. Data were extracted by one reviewer (SK) and checked for accuracy by a second reviewer (AD).
Eligibility criteria
Inclusion criteria
Inclusion criteria were as follows: primary clinical studies involving adult patients who had a diagnosis of FND, systematic reviews looking at similar topic, available full text articles published in English, studies found on the Ovid MEDLINE, Cochrane Register of Clinical Trials, Embase and Pubmed databases.
Exclusion criteria
Non-original articles such as editorials, letters to the editor and conference abstracts were excluded, as well as articles looking at paediatric population and in languages other than English.
Statistical analysis
Data was extracted via a Microsoft word document, then exported to R version 4.0 for analysis and figure generation. We calculated incidence of sleep disorders in patients with FND using the random-effects model which takes into account both within and between study variation (heterogeneity). Secondary analysis was performed comparing Wakefulness after sleep onset (WASO) and Epworth sleepiness scale (ESS) [13] in patients with FND compared to non-FND patients.
Heterogeneity between studies was evaluated using I2 statistics. Publication bias was assessed using Egger’s test and by inspection of funnel plots. Study quality was assessed using the Newcastle-Ottawa scale which ranks the studies on a scale from 0 to 9 based on the selection of the study population, comparability between cases and non-cases and the assessment of the outcome [14].
Results
Study selection
The database search yielded 218 articles (PubMed;110, MEDLINE; 76, Embase; 24 and Cochrane; 8). After the removal of 86 duplicates, 132 articles remained. These were assessed against a predetermined exclusion criteria by title and abstract. 18 were excluded due to inconsistent research objectives. Furthermore, the full text of the remaining 114 articles was attempted to be retrieved. The full text of 114 articles was assessed against the exclusion criteria majority of the papers were of wrong publication type including but not limited to literature reviews, case reports and abstracts; certain studies also reported non-specific sleep outcomes and wrong population. 9 articles were consistent with the inclusion criteria and so were included in this review.
Baseline characteristics
The baseline characteristics of included studies are summarized in Table 1. Most common country of publication was the United Kingdom (4/9). Totally there were 1227 patients out of which 740 reported some form of sleep disorders.
Table 1.
Baseline characteristics and outcomes measured for all the studies included in the final analysis
| First author | Year | Number of patients with reported sleep disorders | Total number of patients diagnosed with FND | Type of FND | Study design |
|---|---|---|---|---|---|
| Nepožitek | 2023 | 33 | 37 | functional motor disorders (FMDs) | Comparative retrospective study |
| Sivathamboo | 2019 | 51 | 93 | psychogenic nonepileptic paroxysms & epileptic seizures | Comparative prospective study |
| Latreille | 2019 | 13 | 17 | psychogenic nonepileptic paroxysms & epileptic seizures | Comparative pilot prospective study |
| Bazil | 2003 | 5 | 8 | psychogenic nonepileptic paroxysms & epileptic seizures | Comparative prospective study |
| Graham | 2017 |
152 + 18 (survey + clinical sample) |
225 + 20 (survey + clinical sample) |
FND | Cohort study |
| Tang | 2016 | 97 | 149 | Globus symptoms | Cohort study |
| Latreille | 2018 | 114 | 149 | psychogenic nonepileptic paroxysms & epileptic seizures | Comparative pilot prospective study |
| Popkirov | 2019 | 11 | 22 | psychogenic nonepileptic paroxysms & epileptic seizures | Comparative prospective study |
| Ducroizet | 2023 | 246 | 527 | FND | Cross-sectional study |
Sleep disorders
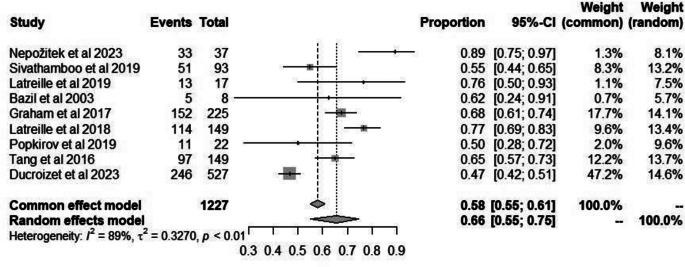
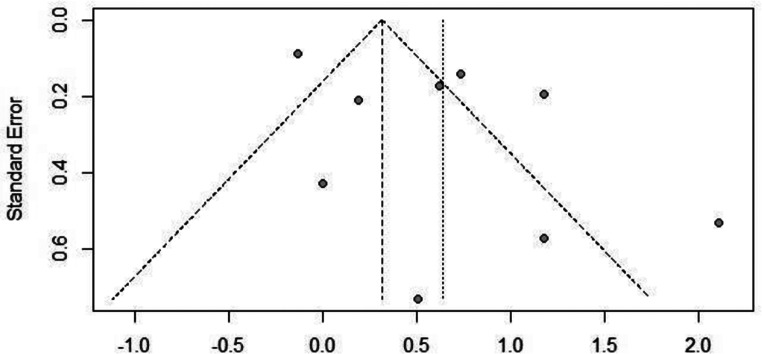
The overall proportion of patients with FND experiencing sleep disorders among all studies was 0.58 (95% CI: 0.55–0.61, I2 = 89%) (Fig. 2). The analysis did find that the presence of sleep disorders among patients with FND was significantly high (p < 0.01). There was no evidence of publication bias with Egger’s test, p = 0.08 (Fig. 3).
Fig. 2.
Forest plot depicting incidence of reported sleep disorders among patients FND
Fig. 3.
Funnel plot depicting outcome of Egger’s test
Wakefulness after sleep onset (WASO)
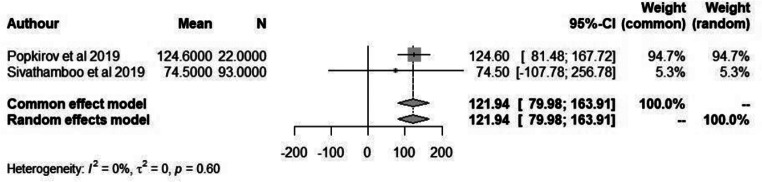
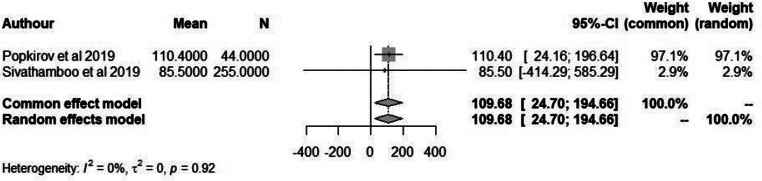
WASO time is an important parameter in measuring quality of sleep and a better reflection of sleep fragmentation. However, only 2 of the included studies reported WASO with an overall mean time of 121.9 min (95% CI: 80.0-163.9) in patients with FND compared to 109.7 (95% CI: 24.7- 194.7) minutes in non-FND patients (Figs. 4 and 5). However, due to the small sample size further studies would need to be conducted in order to determine its significance.
Fig. 4.
Forest plot depicting mean duration of WASO in patients with FND
Fig. 5.
Forest plot depicting mean duration of WASO in non-FND patients
Epworth sleepiness scale (ESS)
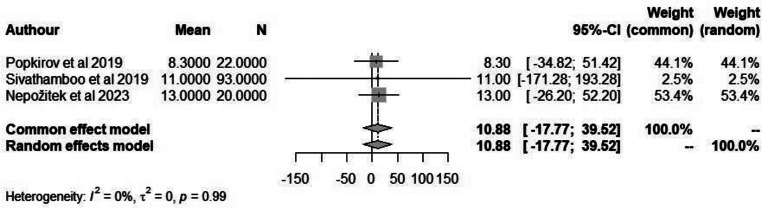
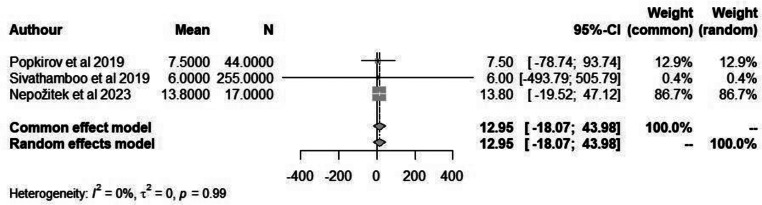
The ESS is a validated tool to assess the extent of subjective daytime sleepiness in eight different everyday life situations. The maximum score is 24, which indicates severe daytime sleepiness. This score suggests that a person is experiencing excessive daytime sleepiness, even in situations where they would typically be expected to stay alert. Despite the extensive use of the ESS in different types of patient populations and even in healthy persons, it has never been specifically validated in patients with FND. When applying a random-effects meta-analysis, we obtained a pooled mean of 10.88 (P = 0.99) in patients with FND compared to 12.95 in non-FND patients (Figs. 6 and 7).
Fig. 6.
Forest plot depicting mean ESS score in patients with FND
Fig. 7.
Forest plot depicting mean ESS score in non-FND patients
Risk of bias
The assessment of bias for retrospective cohort studies, using the Newcastle-Ottowa Scale, is detailed in Fig. 8. The mean score for all studies was 5.5 (out of a maximum total of 9), and no studies were classified to have a high risk of bias.
Fig. 8.
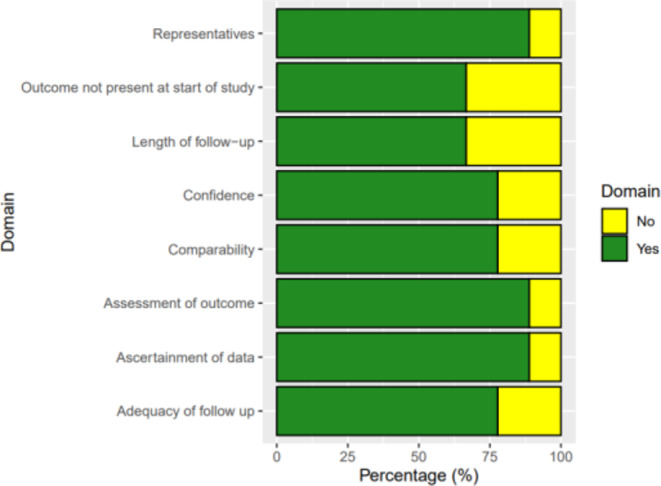
Bar graph depicting Risk of Bias among all included studies
Discussion
Summary of findings
A meta-analysis was conducted on the included studies that reported sleep disturbances/disorders in patients with FND. We found that in patients with FND there were a significant proportion of individuals who experienced some form of sleep disorder. Further analysis was done to establish the possible link between sleep quality and FND by analysis studies that reported WASO and ESS score. However, due to the limited studies reporting this information no significant results were obtained.
Comparison with literature
To our knowledge this is the first systematic review to address the presence of sleep disorders in patients with FND. Although there is a well-established relationship between sleep and psychiatric disorders [15, 16], there are very few data on sleep in FND. Some small studies (n < 20) have reported reduced subjective sleep quality as well as increased rapid-eye-movement sleep duration during polysomnography in patients with Psychogenic Non-epileptic seizures (PNES) [17, 18]. While the results slightly vary between studies; our study found that 58% of patients reported sleep disorders. This is similar to a recent study that found 46.7% of patients with FND experience sleep disturbances [19].
Of the 9 studies included 5 focused on patients with PNES. Latreille et al. (2018) compared sleep-wake patterns in a prospective observational study in PNES and epilepsy patients. Twenty-seven subjects were included in the study, of which 17 had PNES, and 10 were diagnosed epilepsy. Compared to epilepsy controls, the PNES patients showed increased latency of sleep onset (on average about 30 min longer than controls). Otherwise, both groups had a similar sleep architecture. However, the PNES patients subjectively had poorer sleep quality measured by Pittsburgh Sleep Quality Index (10.8 ± 5.1 versus 5.8 ± 2.9; p = 0.01; higher score indicates the worse quality of sleep) and met the clinical criteria for insomnia more often than patients with epilepsy (50% vs. 10%, p = 0.05) [20]. Compared to non-FND patients with FND were more likely to have mild to severe sleep changes, mostly shorter sleep length by up to 1–2 h, and difficulty falling asleep again. These changes in sleep patterns were linked with lower quality of life. These results propose that sleep disturbance is a more pronounced problem in PNES than in epilepsy [20, 21].
Popkirov et al. (2019) examined the occurrence of sleep disorders in a group of 22 PNES patients and 44 epilepsy patients. However, no significant differences were found between the two groups; in patients with epilepsy only three cases of mild or moderate obstructive sleep apnoea–hypopnea syndrome (OSAHS) (7%) and four cases of sleep-disordered breathing (SDB) (9%). Most of the patients were overweight or obese. In 22 patients with dissociative seizures, two (9%) had mild SDB, two (9%) had mild OSAHS and one (5%) had moderate OSAHS [22]. Similar results were obtained by Sivathamboo et al. (2019), Moderate to severe SDB was observed in 26.3% (67/255) of patients with epilepsy and 29.0% (27/93) of patient with PNES. Following adjustment for confounders, pathologic daytime sleepiness predicted moderate to severe SDB in epilepsy (odds ratio [OR] 10.35, 95% confidence interval [CI] 2.09–51.39; p = 0.004). In multivariable analysis, independent predictors for moderate to severe SDB in epilepsy were older age (OR 1.07, 95% CI 1.04–1.10; p < 0.001) and higher body mass index (OR 1.06, 95% CI 1.01–1.11; p = 0.029), and in PNES older age (OR 1.10, 95% CI 1.03–1.16; p = 0.002) [23]. Similar results were also obtained in patients with Functional Motor Disorder (FMD). Nepožitek et al. (2023) found that 23/37 (62%) of patients with FMD experienced sleep disturbances; 35% having restless legs syndrome; 49% obstructive sleep apnoea; and 8% periodic limb movements in sleep; however, the presence of these disorders was not correlated with subjective sleepiness. Patients with FMD with self-reported sleepiness reported higher fatigue (p = 0.002), depression (p = 0.002), and had longer sleep latencies in the MSLT (p < 0.001) compared to the patients with central hypersomnia [24]. Higher Wake After Sleep Onset (WASO) rates are a marker of sleep disruption and the Popkirov et al. paper highlighted this fact. WASO observed in patients with Functional seizures may not be coincidental. Research indicates a potential causal link between sleep disturbances and dissociative experiences [25]. Experimental studies have shown that acute sleep deprivation can increase dissociative symptoms in healthy individuals, as measured by self-report and cognitive tasks [26]. Insomniac patients also tend to score high on the Dissociative Experiences Scale, with these scores correlating with specific EEG findings [27]. The study by Bregman-Hai and Soffer-Dudek (2023) provides further support of this relationship between poor sleep and dissociation. The study found that poor sleep quality and posttraumatic symptoms (PTS) are independent pathways leading to dissociation and disruptions in the sense of agency. Specifically, individuals with low levels of PTS exhibited a stronger relationship between poor sleep (measured by WASO) and dissociative experiences, whereas this relationship was not observed in individuals with high levels of PTS. This suggests that sleep disturbances might contribute to dissociative experiences primarily in those with less severe posttraumatic symptoms via an independent pathway [28].
On the other hand, this opens up the possibility that targeting sleep disruption might be a therapeutic strategy to improve dissociative symptoms. Past research has shown that improving sleep quality in psychiatric patients has been associated with a reduction in dissociative symptoms [29]. However, we need a larger prospective study to explore this possibility in FND population.
Limitations
Our study has several limitations. The presence of sleep disorders in the presence of FND was measured by the proportion of patients experiencing sleep disorders in each study. Due to this the heterogeneity (I2 = 89%) of studies included were high. In order to focus on the effect of sleep disturbances in FND a further analysis was done based on the ESS score and WASO time; however, due to only limited studies reporting this data no significant results were obtained. We also excluded full-text papers not available in English, restricting paper eligibility.
Conclusion
Our study suggests that sleep disorder is common in people FND compared to other medical conditions. However, we need to ask more about sleep during our evaluation of people with FND. Due to limited studies reporting WASO we could not evaluate the direct relationship of sleep deprivation and FND but it remains a tantalizing prospect as a therapeutic target. Hence further research would need to be conducted to determine the significance of our results. Nevertheless, our study suggests that further awareness needs to be raised about sleep disturbances/disorders in patients with FND which could have significant impact on a quality of life of people with FND.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Author contributions
SK: Systematic literature search, drafting and revising the manuscript. A Dutta: Study design, critical revisions to the manuscript, data analysis and interpretation, and assisted in the final approval of the version to be published. Abhijit Das: Conception and design of the study, overall supervision and guidance for the study, data analysis and interpretation, and critically revised the manuscript for important intellectual content.
Funding
None.
Data availability
All data used in this study is publicly available.
Declarations
Ethical approval
This systematic review was conducted following PRISMA guidelines. As it involves the synthesis of previously published data, no new primary data were collected, and no ethical approval was required. No human participants were directly involved, and all data used were publicly available. The authors declare no conflicts of interest, and the review was conducted with transparency, integrity, and objectivity.
Competing interests
None.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bennett K, Diamond C, Hoeritzauer I, Gardiner P, McWhirter L, Carson A et al (2021) A practical review of functional neurological disorder (FND) for the general physician. Clin Med 21:28. 10.7861/CLINMED.2020-0987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Beharry J, Palmer D, Wu T, Wilson D, Le Heron C, Mason D et al (2021) Functional neurological disorders presenting as emergencies to secondary care. Eur J Neurol 28:1441–1445. 10.1111/ENE.14728 [DOI] [PubMed] [Google Scholar]
- 3.Gilmour GS, Nielsen G, Teodoro T, Yogarajah M, Coebergh JA, Dilley MD et al (2020) Management of functional neurological disorder. J Neurol 267:2164. 10.1007/S00415-020-09772-W [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aarsland D, Batzu L, Halliday GM, Geurtsen GJ, Ballard C, Ray Chaudhuri K et al (2021) Parkinson disease-associated cognitive impairment. Nat Reviews Disease Primers 2021 7:1. 10.1038/s41572-021-00280-3 [DOI] [PubMed] [Google Scholar]
- 5.Dymecka J, Gerymski R, Tataruch R, Bidzan M, Fatigue (2021) Physical disability and self-efficacy as predictors of the Acceptance of Illness and Health-Related Quality of Life in patients with multiple sclerosis. Int J Environ Res Public Health 18. 10.3390/IJERPH182413237 [DOI] [PMC free article] [PubMed]
- 6.Hallett M, Aybek S, Dworetzky BA, McWhirter L, Staab JP, Stone J (2022) Functional neurological disorder: New Phenotypes, Common mechanisms. Lancet Neurol 21:537. 10.1016/S1474-4422(21)00422-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Graham CD, Kyle SD (2017) A preliminary investigation of sleep quality in functional neurological disorders: poor sleep appears common, and is associated with functional impairment. J Neurol Sci 378:163–166. 10.1016/J.JNS.2017.05.021 [DOI] [PubMed] [Google Scholar]
- 8.Duo L, Yu X, Hu R, Duan X, Zhou J, Wang K (2023) Sleep disorders in chronic pain and its neurochemical mechanisms: a narrative review. Front Psychiatry 14. 10.3389/FPSYT.2023.1157790 [DOI] [PMC free article] [PubMed]
- 9.Lanigar S, Bandyopadhyay S (2017) Sleep and Epilepsy: a Complex Interplay. Mo Med 114:453 [PMC free article] [PubMed] [Google Scholar]
- 10.Aasvik J, Stiles TC, Woodhouse A, Borchgrevink P, Inge Landrø N (2018) The Effect of Insomnia on Neuropsychological Functioning in patients with comorbid symptoms of Pain, fatigue, and Mood disorders. Arch Clin Neuropsychol 33:14–23. 10.1093/ARCLIN/ACX040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Finan PH, Buenaver LF, Runko VT, Smith MT (2014) Cognitive-behavioral therapy for Comorbid Insomnia and Chronic Pain. Sleep Med Clin 9:261. 10.1016/J.JSMC.2014.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rossman J (2019) Cognitive-behavioral therapy for Insomnia: an effective and underutilized treatment for Insomnia. Am J Lifestyle Med 13:544. 10.1177/1559827619867677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Johns MW (1991) A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 14:540–545. 10.1093/SLEEP/14.6.540 [DOI] [PubMed] [Google Scholar]
- 14.Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25:603–605. 10.1007/S10654-010-9491-Z [DOI] [PubMed] [Google Scholar]
- 15.Krystal AD, PSYCHIATRIC DISORDERS AND SLEEP (2012) Neurol Clin 30:1389. 10.1016/J.NCL.2012.08.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hombali A, Seow E, Yuan Q, Chang SHS, Satghare P, Kumar S et al (2019) Prevalence and correlates of sleep disorder symptoms in psychiatric disorders. Psychiatry Res 279:116–122. 10.1016/J.PSYCHRES.2018.07.009 [DOI] [PubMed] [Google Scholar]
- 17.Bazil CW, Legros B, Kenny E (2003) Sleep structure in patients with psychogenic nonepileptic seizures. Epilepsy Behav 4:395–398. 10.1016/S1525-5050(03)00120-3 [DOI] [PubMed] [Google Scholar]
- 18.Phillips MCL, Costello CA, White EJ, Smit M, Carino J, Strawhorn A et al (2013) Routine polysomnography in an epilepsy monitoring unit. Epilepsy Res 105:401–404. 10.1016/J.EPLEPSYRES.2013.02.015 [DOI] [PubMed] [Google Scholar]
- 19.Ducroizet A, Zimianti I, Golder D, Hearne K, Edwards M, Nielsen G et al (2023) Functional neurological disorder: clinical manifestations and comorbidities; an online survey. J Clin Neurosci 110:116–125. 10.1016/J.JOCN.2023.02.014 [DOI] [PubMed] [Google Scholar]
- 20.Latreille V, Baslet G, Sarkis R, Pavlova M, Dworetzky BA (2018) Sleep in psychogenic nonepileptic seizures: time to raise a red flag. Epilepsy Behav 86:6–8. 10.1016/J.YEBEH.2018.07.001 [DOI] [PubMed] [Google Scholar]
- 21.Vanek J, Prasko J, Ociskova M, Genzor S, Holubova M, Hodny F et al (2021) Sleep disturbances in patients with nonepileptic seizures. Nat Sci Sleep 13:209–218. 10.2147/NSS.S289190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Popkirov S, Stone J, Derry CP (2019) Abnormal sleep in patients with epileptic or dissociative (non-epileptic) seizures: a polysomnography study. Eur J Neurol 26:255–260. 10.1111/ENE.13798 [DOI] [PubMed] [Google Scholar]
- 23.Sivathamboo S, Farrand S, Chen Z, White EJ, Pattichis AA, Hollis C et al (2019) Sleep-disordered breathing among patients admitted for inpatient video-EEG monitoring. Neurology 92:E194–204. 10.1212/WNL.0000000000006776 [DOI] [PubMed] [Google Scholar]
- 24.Nepožitek J, Dostálová S, Věchetová G, Sieger T, Forejtová Z, Nováková L et al (2023) Sleepiness and comorbid sleep disorders in functional motor disorders: a comparative study with central hypersomnia. J Sleep Res e14098. 10.1111/JSR.14098 [DOI] [PubMed]
- 25.van der Kloet D, Merckelbach H, Giesbrecht T, Lynn SJ (2012) Fragmented sleep, fragmented mind: the role of sleep in dissociative symptoms. Perspect Psychol Sci 7(2):159–75 [DOI] [PubMed]
- 26.Giesbrecht T, Smeets T, Leppink J, Jelicic M, Merckelbach H (2007) Acute dissociation after 1 night of sleep loss. J AbnormPsychol 116(3):599–606 [DOI] [PubMed]
- 27.van Heugten-van der Kloet D, Giesbrecht T, Merckelbach H (2015) Sleep loss increases dissociation and affects memory foremotional stimuli. J Behav Ther Exp Psychiatry 47:9–17 [DOI] [PubMed]
- 28.Bregman-Hai N, Soffer-Dudek N (2024) Posttraumatic symptoms and poor sleep are independent pathways to agency disruptions and dissociation: A longitudinal study with objective sleep assessment. J Psychopathol Clin Sci 133(2):192–207 [DOI] [PubMed]
- 29.van der Kloet D, Giesbrecht T, Lynn SJ, Merckelbach H, de Zutter A. Sleep normalization and decrease in dissociativeexperiences: evaluation in an inpatient sample. J Abnorm Psychol 121(1):140–50 [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data used in this study is publicly available.