Abstract
Background
Dietary fat malabsorption contributes to poor nutritional status in patients with cystic fibrosis (CF) and exocrine pancreatic insufficiency (EPI). Prescribing gastric acid-reducing agents such as proton-pump inhibitors (PPI) as an adjunct to pancreatic enzyme replacement therapy (PERT) to improve dietary fat absorption has been accepted in clinical practice despite limited evidence.
Aims
This was a pilot randomized, double-blind, placebo-controlled crossover trial of subjects aged 12 and older with CF and EPI assessed on placebo and omeprazole to determine if PPI improved the efficacy of PERT as indicated by measures of dietary fat absorption.
Methods
Fat malabsorption via stool coefficient of fat absorption (CFA) and malabsorption blood test (MBT), gastrointestinal pH (wireless motility capsule [WMC]), and quality of life (QOL) were assessed after 14 days on both placebo or PPI (omeprazole).
Results
Total 19 subjects enrolled, 13 were randomized, and 9 provided paired results on placebo and PPI. The 3 subject results for CFA were as follows: 1 increased, 1 decreased, and 1 was within the reference range in both tests for fat absorption. For 9 MBT subjects, 7 decreased and 2 increased fat absorption. For the 4 WMC studies, no change in transit times, nor in pH profiles were noted. No differences were seen in the domains of the two QOL questionnaires comparing placebo and PPI.
Conclusions
These limited descriptive pilot study results in participants with CF and EPI on PERT evaluated by stool, blood, and QOL tests did not suggest improvement in fat absorption attributable to PPI.
Keywords: Cystic fibrosis, Proton-pump inhibitors, PERT, MBT, CFA, WMC
Introduction
Dietary fat malabsorption contributes to poor nutritional status in patients with cystic fibrosis (CF) and exocrine pancreatic insufficiency (EPI). Impaired pancreatic function has also been associated with increased gastric acidity, which results in gastroesophageal reflux, peptic ulcers, and decreased effectiveness of pancreatic enzyme replacement therapy (PERT) [1]. The use of gastric acid suppression agents including histamine receptor antagonists (H2RA) and proton-pump inhibitors (PPI) is widespread in pediatric patients from preterm infants to young adults to treat many diagnoses including oral feeding intolerance, dyspepsia, gastroesophageal reflux, and functional abdominal pain [2, 3]. In pediatric and adult populations with EPI, adjunctive gastric acid suppression agents have been an established practice intended to improve efficacy of PERT [4]. Specifically in CF, the use of PPI and H2RA with PERT to improve fat absorption has become accepted clinical practice despite limited clinical evidence [5–7]. To date, no study has assessed the effect of gastric acid suppression on duodenal pH or dietary fatty acid absorption in the small intestine in participants with CF and EPI. Clarifying this relationship will provide information on how these common medications may affect nutritional and general health outcomes in this population.
The most recent Cochrane review assessing acid suppression in patients with CF reports that single-center studies showed improvement in abdominal pain and some degree of variably measured fat malabsorption but did not provide data on lung function, quality of life, or survival; therefore, the authors offered no conclusion on whether these agents offered overall benefit in CF [1]. Though ubiquitously used, PPI for any indication is not without risk [8]. PPI use has been associated with an increased risk of asthma and decreased bone density in children and with liver cancer in the general population, though any causal relationship remains in question [9–12]. Acid suppression is also associated with increased risk of serious infection in neonates as well as young children; in the pediatric population with CF whose risk of infection is already elevated from that of the general pediatric population, this risk from acid suppression is almost certainly greater [13]. Due to these risks as well as a lack of clarity about the risk of lifetime PPI use, gastric acid suppression agents should be used only in the presence of potential benefit [14].
The aim of this study was to determine if daily PPI compared to placebo improved the efficacy of PERT as evidenced by changes in dietary fat malabsorption. The primary outcome was the efficacy of the standard dose of omeprazole 20 mg daily to improve dietary fat absorption in subjects with CF and EPI via measurement of the coefficient of fat absorption (CFA) in the stool. Other outcomes sought to determine changes in duodenal pH on and off acid suppression using a wireless motility capsule (WMC) and to characterize changes in plasma fatty acid absorption in the small intestine using the malabsorption blood test (MBT). GI symptoms and other quality of life (QOL) indicators were also assessed.
Methods
This was a single-center pilot randomized, placebo-controlled, double-blinded crossover trial to determine the efficacy of acid suppression with a PPI in improving dietary fat malabsorption and other outcomes in subjects with CF and EPI. The trial was approved by the Children’s Hospital of Philadelphia (CHOP) Institutional Review Board and registered with clinicaltrials.gov (NCT 03551691). All study activities were outpatient and conducted at the CHOP Center for Human Phenomic Science.
Study Subjects
Subjects diagnosed with CF and EPI were eligible for inclusion in the study if they were ≥ 12 years of age at enrollment, had a fecal elastase of < 200 µg/g stool indicating EPI, and in good pulmonary health with FEV1 > 40% predicted. Subjects who were already receiving PPI or H2RA prior to the study were eligible if willing to discontinue the medication 14 days prior to study initiation. Subjects were excluded if they were either pregnant or breastfeeding, had another illness affecting nutritional status, using medication or had another diagnosis affecting dietary fat absorption, or allergy to soy or safflower products. Subject or parent/guardian informed consent and child assent when applicable was obtained prior to all study-related activities. After consent/assent was obtained, a stool sample was collected to confirm EPI via fecal elastase-1 (FE) via quantitative enzyme-linked immunosorbent assay (ARUP Laboratories, Salt Lake City, Utah).
Randomization and Study Procedures
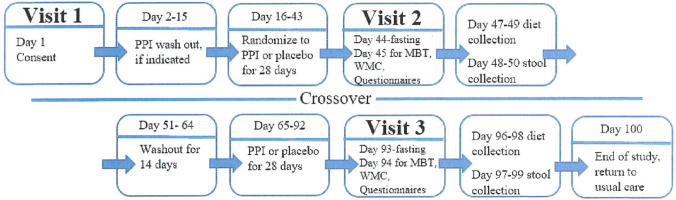
Subjects were randomized by the University of Pennsylvania Investigational Drug Service to receive either omeprazole 20 mg daily or placebo for at least 14 days while maintaining their standard diet and PERT regimen (Fig. 1). All other research personnel were blinded to the randomization throughout the study. After at least 14 days of study medication, subjects underwent the primary (CFA) and other outcome measures: MBT, Patient-Reported Outcomes Measurement Information Systems [PROMIS] questionnaires, Cystic Fibrosis Questionnaire-Revised [CFQ-R], WMC for gastric and duodenal pH assessment (> 18 years of age), and questionnaires assessing adherence and adverse events via recall. Upon completion of study measures, subjects then underwent a 14-day wean and washout period before crossing over to the other treatment arm for 14 days. Study measures were completed at the end of a second treatment period (minimum 14 days, maximum 28 days), and subjects were weaned off the study medication and returned to usual care.
Fig. 1.
Protocol timeline. PPI proton-pump inhibitor, MBT malabsorption blood test, WMC wireless motility capsule
Clinical Data Collection
Demographics and clinical data were collected by study staff. Anthropometrics were obtained in triplicate by the research staff according to standardized techniques, and the mean was used for analysis. Body mass index was calculated (kg/m2) from weight using a digital scale (Seca, Munich, Germany) and height using a stadiometer (Holtain, Crymych, United Kingdom).
Measurement of Fat Malabsorption
Subjects collected stool for 72 h at home for CFA (on and off PPI), and stool was assessed for total fat by nuclear magnetic resonance spectroscopy (Mayo Clinic Laboratories, Rochester, MN). The CFA was derived from the stool collection and 3-day dietary food records. Three-day average fat intake was analyzed by Nutrition Data System 2012 (NCC, University of Minnesota, Minneapolis, MN). CFA was calculated as grams of fat consumed per day minus grams of fat excreted per day, divided by total fat grams consumed.
The MBT was performed at the same two timepoints on PPI and placebo to assess fatty acid absorption in the upper intestine [15–18]. A test meal containing pentadecanoic acid (PA), a 15-carbon free fatty acid, and triheptadecanoin (THA), a triglyceride containing three 17-carbon heptadecanoic fatty acids (HA) requiring hydrolysis by lipase for HA absorption, was administered in the morning after a 12-h fast. Blood samples were taken hourly over a nine-hour period following the test meal. Concentrations of plasma PA and HA were determined (CHOP, Clinical Laboratory). A noncompartmental analysis estimated the area under the curve (AUC) for PA and HA micromolar concentrations to facilitate comparison of relative absorption on PPI and placebo.
Measurement of Gastric and Intestinal pH
In subjects ≥ 18 years of age, gastric and intestinal pH was assessed with the WMC (SmartPill ®, Medtronic Corporation, Minneapolis, MN) at the same two timepoints described above. The WMC possesses sensors to measure pH, pressure, and temperature for approximately 72 h after administration. Anal exit was defined as an acute temperature drop and permanent loss of signal on the receiver. Data were extracted using GIMS software (Gastrointestinal Motility Software [GIMS™] Medtronic Corporation, Minneapolis MN). Available WMC data allowed the calculation of multiple values. The mean 1-min interval pH for stomach was calculated for 30 min prior to gastric emptying time (GET), defined as an abrupt rise in the pH by 3 or more units above the stomach pH. Similarly, small bowel mean 1-min pH increments for 30 min post GET was extracted [19]. The colon mean 1-min pH intervals for 30 min were recorded prior to anal exit of WMC. Corresponding segments also had 5-min pH AUC calculated.
Quality of Life Measurements
The Cystic Fibrosis Questionnaire-Revised (CFQ-R, University of Miami, Miami, FL, adult format) was used to assess QOL after placebo and omeprazole treatment [20, 21]. Subjects ≥ 14 years completed the PROMIS short forms for depression, mobility, pain behavior, social isolation, and GI symptoms including belly pain, constipation, diarrhea, gas and bloating, and reflux [22]. Subjects also completed PROMIS-GI, which included eight symptom scales: reflux, disrupted swallowing, diarrhea, bowel incontinence, nausea and vomiting, constipation, belly pain, and gas, bloating, and flatulence.
Statistical Analysis
Descriptive statistics for CFA, duodenal pH, MBT, and other outcomes were calculated. The following was the statistical analysis plan for the study; however, the COVID-19 pandemic limited the number of subjects completing the protocol, and only exploratory statistical evaluations were appropriate due to the small final sample. Means and standard deviations were computed where able and applicable. Paired t tests or Wilcoxon sign rank tests were used to compare results for the total group on and off omeprazole treatment as appropriate. The primary outcome was the change in CFA between the measurements made on and off omeprazole. Exploratory outcomes included concurrent changes in fat absorption as measured by the MBT, changes in intestinal pH as measured by the WMC, and changes in QOL by PROMIS and CFQ-R. For the MBT outcomes, AUC from time zero to nine hours was calculated using the linear trapezoid method. PK parameters were compared for the total group comparing on and off omeprazole using paired t tests or Wilcoxon sign rank tests as appropriate, and between randomization groups at each time point using an unpaired t test or Mann–Whitney U rank sum test as appropriate. Ultimately, non-parametric Mann–Whitney t test or analysis of variance was used to evaluate the differences between placebo and PPI WMC studies using GraphPad Prism 9.4 (San Diego, CA).
Results
A total of 19 subjects enrolled and 13 were randomized and provided test results included in this report. The other six subjects did not return for protocol tests after the randomization visit due to the impact of the COVID-19 pandemic, despite many efforts to accommodate health and scheduling concerns.
Table 1 provides the baseline data on the 13 subjects for genotype, race, age, sex, and anthropometrics and results of all completed CFA and MBT. Three subjects completed the CFA with full results. One subject had increased stool fat loss and one had decreased stool fat loss when placebo and PPI were compared. The remaining subject’s CFA was within the reference range for both placebo and PPI.
Table 1.
Subject characteristics and outcomes at baseline and following active treatment
| ID | Genotype | Age | Race | Sex | Study drug | Baseline* | CFA | MBT | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Weight (kg) | Height (cm) | WAZ | HAZ | BMIZ | % | HA AUC | HA % change | |||||||
| 1 | F508del, F508del | 16 | C | F | Placebo | 61.4 | 148.1 | 0.7 | − 2.2 | 1.5 | 83 | 14.7 | ||
| Active | 60 | 5.9 | − 59.9 | |||||||||||
| 2 | F508del, F508del | 28 | C | M | Placebo | 60.5 | 176.9 | − 1.0 | 0.0 | − 1.5 | 92 | 8.0 | ||
| Active | 96 | 5.5 | − 31.3 | |||||||||||
| 3 | F508del, G542X | 16 | C | M | Placebo | 59.8 | 161.1 | − 0.1 | − 1.6 | 0.8 | 77 | 8.7 | ||
| Active | 84 | 5.6 | − 35.6 | |||||||||||
| 4 | F508del, F508del | 19 | C | F | Placebo | – | 5.1 | |||||||
| Active | 60.1 | 156.4 | 0.3 | − 1.1 | 0.8 | 83 | 4.8 | − 5.9 | ||||||
| 5 | F508del, F508del | 17 | C | M | Placebo | – | 6.6 | |||||||
| Active | 78.2 | 180.8 | 1.1 | 0.8 | 0.8 | 99 | 8.9 | 34.8 | ||||||
| 6 | G1551D, – 7 T/7 T | 18 | C | F | Placebo | – | 11.5 | |||||||
| Active | 50.3 | 154.4 | − 0.8 | − 1.3 | − 0.1 | 89 | 9.3 | − 19.1 | ||||||
| 7 | F508del, F508del | 20 | C | M | Placebo | – | 12.0 | |||||||
| Active | 60.4 | 165.8 | − 1.0 | − 1.5 | − 0.4 | 46 | 7.1 | − 40.8 | ||||||
| 8 | F508del, F508del | 23 | C | M | Placebo | – | – | – | 6.4 | |||||
| Active | – | 11.1 | 73.4 | |||||||||||
| 9 | F508del, F508del | 23 | C | F | Placebo | 59.9 | 148.9 | 0.2 | − 2.2 | 1.1 | 55 | 13.5 | ||
| Active | – | 7.9 | − 41.5 | |||||||||||
| 10 | F508del, F508del | 14 | C | F | Placebo | 40.1 | 150.5 | − 1.2 | − 1.5 | − 0.6 | – | |||
| Active | – | |||||||||||||
| 11 | F508del, F508del | 18 | C | M | Placebo | – | ||||||||
| Active | 74.6 | 179 | 0.6 | 0.4 | 0.4 | – | 7.8 | |||||||
| 12 | F508del, F508del | 19 | C | F | Placebo | – | ||||||||
| Active | – | – | – | 8.1 | ||||||||||
| 13 | S549N, Q98X | 18 | AA | F | Placebo | 54.8 | 155.2 | − 0.2 | − 1.2 | 0.4 | – | |||
| Active | – | |||||||||||||
C Caucasian, AA African–American, M male, F female, WAZ weight-for-age Z-score, HAZ height-for-age Z-score, BMIZ body mass index-for-age Z-score (BMIZ), CFA coefficient of fat absorption, MBT malabsorption blood test
*anthropometric measurements are baseline measures; alignment in the table indicate the study drug that participants were randomized to first
‘–’ in cells indicates measurement/test was not performed (e.g., not able to complete due to the COVID-19 pandemic) or the data were not usable (e.g., incomplete diet record to calculate CFA)
Nine subjects had full results for the MBT. Of these nine, seven (78%) demonstrated decreased plasma HA concentration while taking a PPI, with a mean decrease in HA AUC of 33.4% (range -5.9 to -59.9), indicating decreased duodenal fatty acid absorption. Two subjects had increased HA concentrations (+ 34.8, + 73.4), indicating an increase in fatty acid absorption on PPI relative to placebo.
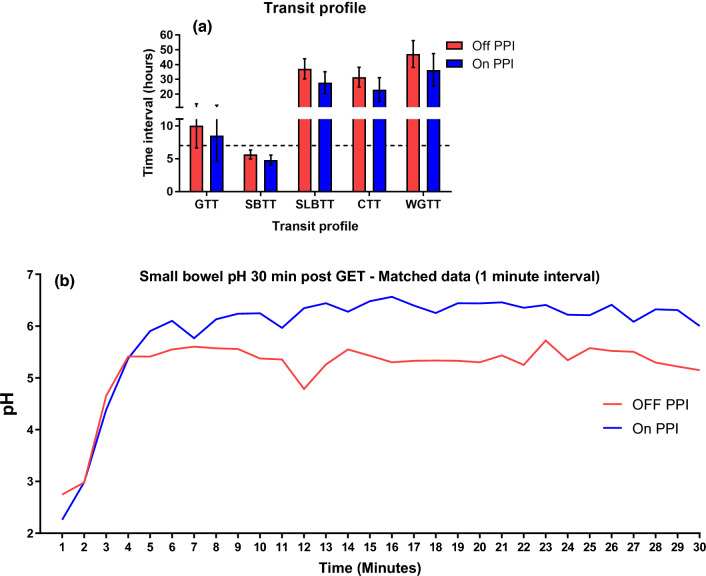
Four subjects completed both WMC evaluations, ultimately demonstrating that gastric transit time and small bowel transit time were not influenced by acid suppression, Fig. 2A. There was a decrease in colonic transit time when subjects were on PPI therapy, but this pattern did not reach statistical significance. No significant changes in gastric, intestinal, or colonic pH 30-min interval profiles were noted, Fig. 2B. There was an expected small decline in intestinal pH when subjects were on placebo compared to the PPI.
Fig. 2.
Measured gastrointestinal transit (a) and post-stomach small intestinal pH (b) profiles on and off treatment. CTT Colonic transit time, GET gastric emptying time, GTT gastric transit time, PPI proton-pump inhibitor, SBTT small bowel transit time, SLBTT small-large bowel transit time, WGTT whole gut transit time
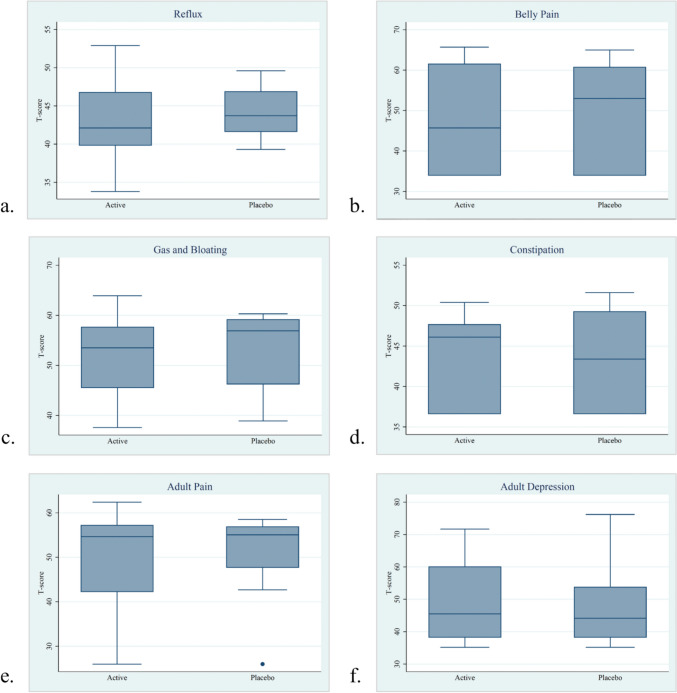
Eleven subjects completed PROMIS questionnaires while on both placebo and PPI (Fig. 3). This included seven adult and four pediatric subjects. In the adult subjects, no significant differences were seen in the QOL metrics nor the GI symptom profiles between treatments. Similarly, the eleven subjects who completed the CFQ-R (Fig. 4) had no significant changes in the domains between treatments. The digest domain trended toward improvement on PPI (p = 0.054).
Fig. 3.
PROMIS and PROMIS-GI selected domains: 3a Reflux, 3b Belly Pain, 3c Gas and Bloating, 3d Constipation, 3e Adult Pain, and 3f Adult Depression. PROMIS measure domains are reported on a T-score, where 50 is the mean of the reference population. Higher numbers indicate more of the domain being measured. PROMIS Patient-reported outcomes measurement information system
Fig. 4.
CFQ-R selected domains: 3a Physical, 3b Emotion, 3c Eating, 3d Treatment Burden, 3e Respiratory, and 3f Digest. The CFQ-R is a disease-specific measure for patients with cystic fibrosis, with higher scores indicating better health-related quality of life in the domain being measured. CFQ-R Cystic fibrosis questionnaire–revised
No significant adverse events were reported in the study population, and subjects did not report any decreased adherence to study drug or PERT via recall questionnaire.
Discussion
Proton-pump inhibitors (PPIs) decrease gastric acid secretion from parietal cells by selectively and irreversibly inhibiting the H+/K+ ATPase. PPIs are pro-drugs that require gastric acid for activation, reacting covalently with cysteine on the luminal surface of the ATPase [23]. Omeprazole, a commonly-used PPI, was patented in 1978 and FDA-approved for adults in 1989 with both prescription and over the counter preparations, in 2002 for children aged 2–16 years old, and in 2008 for children aged 1–2 years old. Label indications include treatment of gastroesophageal reflux disease, erosive esophagitis, eosinophilic esophagitis, gastric or duodenal ulcer, H. pylori eradication, and frequent heartburn [24]. Off-label use in CF intended to enhance fat absorption with PERT is an accepted clinical practice despite limited evidence that PPI improves PERT effectiveness [1].
Patients with CF and EPI have increased duodenal acidity due to impaired or absent pancreatic bicarbonate secretion. Intrinsic pancreatic enzymes and enteric-coated PERT both require the more basic pH typical of the duodenal environment to be activated (intrinsic enzymes) or dissolved and released (PERT) in a timely fashion for digestion of dietary macronutrients. An acidic small bowel milieu will not only impair enzymatic activity but also lead to bile salt precipitation, exacerbating fat malabsorption. Acid suppression has been hypothesized to facilitate fat absorption by raising the duodenal pH to increase lipase bioavailability and thus triglyceride hydrolysis, improving fatty acid absorption, and possibly supporting growth in children and healthy weight for adults. There is no prior evidence that PPI results in any changes in dietary fat absorption in CF or in healthy subjects.
PPIs have been thought to be safe for short- and long-term use [25]. The FDA package insert for omeprazole warns of an increased risk of inflammatory and malabsorptive states, including C. difficile diarrhea, acute interstitial nephritis, atrophic gastritis, systemic lupus erythematosus, hypomagnesemia, vitamin B12 deficiency, iron deficiency, and bone fractures [24].
Recent studies provide evidence for potential PPI risks in adults and children [26]. Lassalle et al. found in a nationwide cohort study a higher rate of severe infections overall in infants through children nine years of age treated with a PPI compared H2RA or antacids. PPI use was associated with increased gastrointestinal, upper respiratory, and central nervous system infections, and the authors recommended judicious use in children [27]. Increased risk of fracture in children exposed to early acid suppression was previously described in 2019 by Malchodi et al. and in 2020 by Wang et al. [10, 28]. In the CF population, pulmonary exacerbation is of particular clinical importance, and McCrory et al. reported higher risk of pulmonary exacerbation in participants with CF taking PPI [29]. Despite these data, acid suppression with PPI has increased over the past two decades, frequently in children. For example, a 2022 report demonstrated that PPIs were being prescribed in Scandinavia more frequently from 2007 to 2020 despite international guidelines recommending otherwise [30].
Given the potential risks of PPI therapy as an adjunct to PERT in patients with CF and EPI, we designed this pilot study to evaluate the potential benefit. CFA results for the three subjects with placebo and PPI exposure were mixed: one subject had increased stool fat loss, one had decreased stool fat loss, and one subject remained within the reference range across both tests. Although PPI use in this study was anticipated to decrease fat malabsorption, prior work has demonstrated that PPI as an adjunct to PERT had no statistically significant effect on CFA [31].
Of the nine subjects who completed the MBT, 78% had unexpectedly decreased plasma HA absorption with omeprazole, indicating decreased intestinal fatty acid absorption. While the intended effect of PPI is to suppress gastric acid secretion and increase the gastric pH and fatty acid absorption, other downstream effects may occur, changing absorption patterns of nutrients including magnesium and vitamin B12 as well as altering enteral bacterial homeostasis [32]. A well-described adverse effect of oral PPI is diarrhea, though the mechanism is not well understood and likely multifactorial. Subjects in this study did not report an increase in diarrheal symptoms. Whether the decrease in HA absorption in these participants was due to the changes in pH, a decrease in cofactors of HA absorption (i.e., bile salts), altered bacterial composition, or an as-yet-unidentified factor remains unclear. Previous studies utilizing the MBT in patients with CF and EPI detected changes in fat absorption with other intervention protocols including dosage and timing of PERT [17, 18].
In the four subjects who completed both WMC studies on placebo and PPI, there was no indication of significant changes in the transit or pH profiles across different regions of the intestine. Although a small increase in baseline pH profile was observed in the proximal small bowel on PPI, it did not reach statistical significance. Historical use of acid suppression in subjects with CF on PERT with no GERD symptoms was to improve luminal pH to facilitate earlier dissolution of enterically coated enzymes and improve digestion and nutritional assimilation of fat calories [33, 34]. Our results question the need for acid suppression medications in CF care to improve dietary fat malabsorption when direct measurement of the gastric, small bowel, and duodenal pH showed no significant changes on or off PPI.
QOL has rarely been investigated in prior studies of PPI use in subjects with CF, and the PROMIS symptom tool has not to our knowledge been evaluated [35]. No significant differences were seen in the results with placebo and PPI in PROMIS domains of depression, mobility, pain behavior, and social isolation, nor were significant differences seen in the PROMIS-GI symptom profiles including belly pain, constipation, diarrhea, gas and bloating, and reflux. The digest domain approached statistical significance (p = 0.05). Lack of differences may be due to the small number of subjects studied here or due to the short amount of time that participants were treated with PPI. In non-CF adult studies, fewer than 50% of participants were satisfied with their response to PPI treatment when prescribed for symptoms of gastroesophageal reflux disease (regurgitation, heartburn) [35, 36]. Perception of PPI non-response has been associated with psychological distress and lower QOL. In our study, an association with QOL was not seen with PPI non-response; however, the expectation in this study was that PPI may alter fat absorption, not symptoms of reflux. The CFQ-R has been used to report QOL symptoms with PPI. In one adult CF pilot randomized controlled double-blind trial of esomeprazole compared to placebo evaluating the effect of PPI on pulmonary exacerbations, QOL measured by CFQ-R showed no difference between placebo and PPI over 36 weeks [37].
Our study results are limited by the small number of subjects who completed the outcome measurements both on PPI and placebo. The protocol complexity and COVID-19 disruption resulted in formal withdrawal from the study by some subjects, and for other subjects, an inability to complete the protocol within the extended study timeline. The two more novel tests, the MBT and WMC, were well-tolerated and provided meaningful information. They should be considered in the design of future clinical trials required to determine the clinical utility to improve fat absorption with adjunct therapy of PPI with PERT. Subjects were recruited from several CF Centers and there was significant interest in scientific and clinical question posed by this study from the CF teams, subjects, and their care providers, and this may support participation in future PPI and CF research.
Conclusion
Use of PPIs are common in children and adults with CF and EPI with the intent to increase PERT fat absorption efficacy and support healthy weight and fat-soluble vitamin and essential fatty acid status. Our limited descriptive results from participants with CF and EPI on PERT treated with PPI and placebo did not demonstrate improvement in fat absorption attributable to PPI via plasma or stool tests. The unexpected MBT results, arguably more specific than the CFA as a direct measure of fatty acid absorption, suggested that duodenal fat absorption may be impaired rather than enhanced by PPI in this setting. The similarly unexpected WMC pH results suggest that PPI use did not alter gastric, intestinal, or colonic pH. With these preliminary results showing no evidence of efficacy in the basic mechanism to improve outcomes in patients with CF and EPI, coupled with increasing evidence of risks of PPI medications in all clinical populations, further research is needed to justify continued use of these medications in this population.
Acknowledgments
The authors would like to thank the teams at the Cystic Fibrosis Centers at the Children’s Hospital of Philadelphia, Hospital of the University of Pennsylvania, and Geisinger Medical Center for their referral of interested study subjects and support in study efforts.
Author’s contribution
AT, JNB, and VAS are the guarantors and will ensure that questions related to any part of this work are appropriately investigated and resolved. VAS and JNB were responsible for study design; JNB, AT, BDK, and VAS participated in data acquisition. DP and DG were responsible for WMC data extraction and analysis. All authors (AEP, AT, JNB, BDK, VAS, DP, and DG) participated in data interpretation, drafted the manuscript, and were involved in critical manuscript revisions. All authors participated in final manuscript revisions and take responsibility for the integrity of the data and accuracy of the data analysis.
Funding
This work was supported in part by an Investigator Initiated Program award from Chiesi USA, Inc, NIH (UL1RR024134), NIH T32 GM008562-23 (JNB), and the NASPGHAN Foundation/Abbot Nutrition Training Grant (BDK).
Data availability
No datasets were generated or analysed during the current study.
Declarations
Conflict of interest
All authors disclose they have no conflicts of interest related to this project and manuscript. The funding sources had no role in study design, analysis, or drafting of the manuscript.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ng SM, Moore HS. Drug therapies for reducing gastric acidity in people with cystic fibrosis. Cochrane Database Syst Rev 2021;4:CD003424. 10.1002/14651858.CD003424.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Omari TI, Haslam RR, Lundborg P et al. Effect of omeprazole on acid gastroesophageal reflux and gastric acidity in preterm infants with pathological acid reflux. J Pediatr Gastroenterol Nutr 2007;44:41–44. 10.1097/01.mpg.0000252190.97545.07. [DOI] [PubMed] [Google Scholar]
- 3.Orel R, Benninga MA, Broekaert IJ et al. Drugs in focus: proton-pump inhibitors. J Pediatr Gastroenterol Nutr 2021;72:645–653. 10.1097/MPG.0000000000003063. [DOI] [PubMed] [Google Scholar]
- 4.DiMagno EP. Gastric acid suppression and treatment of severe exocrine pancreatic insufficiency. Best Pract Res Clin Gastroenterol 2001;15:477–486. 10.1053/bega.2001.0195. [DOI] [PubMed] [Google Scholar]
- 5.Borowitz DS, Grand RJ, Durie PR. Use of pancreatic enzyme supplements for patients with cystic fibrosis in the context of fibrosing colonopathy. J Pediatr 1995;127:681–684. 10.1016/s0022-3476(95)70153-2. [DOI] [PubMed] [Google Scholar]
- 6.Bruno MJ, Rauws EA, Hoek FJ et al. Comparative effects of adjuvant cimetidine and omeprazole during pancreatic enzyme replacement therapy. Dig Dis Sci 1994;39:988–992. 10.1007/BF02087549. [DOI] [PubMed] [Google Scholar]
- 7.Marotta F, O’Keefe SJ, Marks IN et al. Pancreatic enzyme replacement therapy. Importance of gastric acid secretion, H2-antagonists, and enteric coating. Dig Dis Sci 1989;34:456–461. 10.1007/BF01536271. [DOI] [PubMed] [Google Scholar]
- 8.Rosen R, Vandenplas Y, Singendonk M et al. Pediatric gastroesophageal reflux clinical practice guidelines: joint recommendations of the North American society for pediatric gastroenterology, hepatology, and nutrition and the European society for pediatric gastroenterology, hepatology, and nutrition. J Pediatr Gastroenterol Nutr 2018;66:516–554. 10.1097/MPG.0000000000001889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang Y-H, Wintzell V, Ludvigsson JF et al. Association between proton-pump inhibitor use and risk of asthma in children. JAMA Pediatr 2021;175:394–403. 10.1001/jamapediatrics.2020.5710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang Y-H, Wintzell V, Ludvigsson JF et al. Association between proton-pump inhibitor use and risk of fracture in children. JAMA Pediatr 2020;174:543–551. 10.1001/jamapediatrics.2020.0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Freedberg DE, Haynes K, Denburg MR et al. Use of proton-pump inhibitors is associated with fractures in young adults: a population-based study. Osteoporos Int 2015;26:2501–2507. 10.1007/s00198-015-3168-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tran KT, McMenamin ÚC, Hicks B et al. Proton-pump inhibitor and histamine-2 receptor antagonist use and risk of liver cancer in two population-based studies. Aliment Pharmacol Ther 2018;48:55–64. 10.1111/apt.14796. [DOI] [PubMed] [Google Scholar]
- 13.Tan J, Jeffries S, Carr R. A review of histamine-2 receptor antagonist and proton-pump inhibitor therapy for gastroesophageal reflux disease in neonates and infants. Paediatr Drugs 2023;25:557–576. 10.1007/s40272-023-00580-z. [DOI] [PubMed] [Google Scholar]
- 14.Freedberg DE, Kim LS, Yang Y-X. The risks and benefits of long-term use of proton-pump inhibitors: expert review and best practice advice from the American gastroenterological association. Gastroenterology 2017;152:706–715. 10.1053/j.gastro.2017.01.031. [DOI] [PubMed] [Google Scholar]
- 15.Bashaw H, Brownell JN, Schall JI et al. Measures of dietary fat and energy absorption in healthy adults. Pancreas 2020;49:845–854. 10.1097/MPA.0000000000001587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Brownell JN, Schall JI, Stallings VA. Pancreatic function in chronic pancreatitis: a cohort study comparing 3 methods of detecting fat malabsorption and the impact of short-term pancreatic enzyme replacement therapy. Pancreas 2019;48:1068–1078. 10.1097/MPA.0000000000001381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mascarenhas MR, Mondick J, Barrett JS et al. Malabsorption blood test: assessing fat absorption in patients with cystic fibrosis and pancreatic insufficiency. J Clin Pharmacol 2015;55:854–865. 10.1002/jcph.484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Stallings VA, Mondick JT, Schall JI et al. Diagnosing malabsorption with systemic lipid profiling: pharmacokinetics of pentadecanoic acid and triheptadecanoic acid following oral administration in healthy subjects and subjects with cystic fibrosis. Int J Clin Pharmacol Ther 2013;51:263–273. 10.5414/CP201793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gelfond D, Ma C, Semler J et al. Intestinal pH and gastrointestinal transit profiles in cystic fibrosis patients measured by wireless motility capsule. Dig Dis Sci 2013;58:2275–2281. 10.1007/s10620-012-2209-1. [DOI] [PubMed] [Google Scholar]
- 20.Modi AC, Quittner AL. Validation of a disease-specific measure of health-related quality of life for children with cystic fibrosis. J Pediatr Psychol 2003;28:535–545. 10.1093/jpepsy/jsg044. [DOI] [PubMed] [Google Scholar]
- 21.Quittner AL, Buu A, Messer MA et al. Development and validation of the cystic fibrosis questionnaire in the United States: a health-related quality-of-life measure for cystic fibrosis. Chest 2005;128:2347–2354. 10.1378/chest.128.4.2347. [DOI] [PubMed] [Google Scholar]
- 22.Fries JF, Bruce B, Cella D. The promise of PROMIS: using item response theory to improve assessment of patient-reported outcomes. Clin Exp Rheumatol 2005;23:S53–S57. [PubMed] [Google Scholar]
- 23.Shin JM, Kim N. Pharmacokinetics and pharmacodynamics of the proton-pump inhibitors. J Neurogastroenterol Motil 2013;19:25–35. 10.5056/jnm.2013.19.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Anon. Prilosec (omeprazole) [Package Insert] U.S. Food and Drug Administration Web site. 2012
- 25.Gunasekaran TS, Hassall EG. Efficacy and safety of omeprazole for severe gastroesophageal reflux in children. J Pediatr 1993;123:148–154. 10.1016/s0022-3476(05)81561-6. [DOI] [PubMed] [Google Scholar]
- 26.Berry JG, Mansbach JM. Questionable safety of proton-pump inhibitor use in children. JAMA Pediatr 2023;177:1010–1012. 10.1001/jamapediatrics.2023.2906. [DOI] [PubMed] [Google Scholar]
- 27.Lassalle M, Zureik M, Dray-Spira R. Proton-pump inhibitor use and risk of serious infections in young children. JAMA Pediatr 2023;177:1028–1038. 10.1001/jamapediatrics.2023.2900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Malchodi L, Wagner K, Susi A et al. Early acid suppression therapy exposure and fracture in young children. Pediatrics 2019;144:e20182625. 10.1542/peds.2018-2625. [DOI] [PubMed] [Google Scholar]
- 29.McCrory BE, Harper HN, McPhail GL. Use and incidence of adverse effects of proton-pump inhibitors in patients with cystic fibrosis. Pharmacotherapy 2018;38:725–729. 10.1002/phar.2125. [DOI] [PubMed] [Google Scholar]
- 30.Lyamouri M, Mårild K, Nielsen RG et al. Proton-pump inhibitors for infants in three Scandinavian countries increased from 2007 to 2020 despite international recommendations. Acta Paediatr 2022;111:2222–2228. 10.1111/apa.16491. [DOI] [PubMed] [Google Scholar]
- 31.Sander-Struckmeier S, Beckmann K, Janssen-van Solingen G et al. Retrospective analysis to investigate the effect of concomitant use of gastric acid-suppressing drugs on the efficacy and safety of pancrelipase/pancreatin (CREON®) in patients with pancreatic exocrine insufficiency. Pancreas 2013;42:983–989. 10.1097/MPA.0b013e31828784ef. [DOI] [PubMed] [Google Scholar]
- 32.Nehra AK, Alexander JA, Loftus CG et al. Proton-pump inhibitors: review of emerging concerns. Mayo Clin Proc 2018;93:240–246. 10.1016/j.mayocp.2017.10.022. [DOI] [PubMed] [Google Scholar]
- 33.Ferrone M, Raimondo M, Scolapio JS. Pancreatic enzyme pharmacotherapy. Pharmacotherapy 2007;27:910–920. 10.1592/phco.27.6.910. [DOI] [PubMed] [Google Scholar]
- 34.Kraisinger M, Hochhaus G, Stecenko A et al. Clinical pharmacology of pancreatic enzymes in patients with cystic fibrosis and in vitro performance of microencapsulated formulations. J Clin Pharmacol 1994;34:158–166. 10.1002/j.1552-4604.1994.tb03981.x. [DOI] [PubMed] [Google Scholar]
- 35.Yadlapati R, Tye M, Keefer L et al. Psychosocial distress and quality of life impairment are associated with symptom severity in PPI non-responders with normal impedance-pH profiles. Am J Gastroenterol 2018;113:31–38. 10.1038/ajg.2017.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kahrilas PJ, Jonsson A, Denison H et al. Regurgitation is less responsive to acid suppression than heartburn in patients with gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2012;10:612–619. 10.1016/j.cgh.2012.01.022. [DOI] [PubMed] [Google Scholar]
- 37.Canani RB, Cirillo P, Roggero P et al. Therapy with gastric acidity inhibitors increases the risk of acute gastroenteritis and community-acquired pneumonia in children. Pediatrics 2006;117:e817–e820. 10.1542/peds.2005-1655. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.